Published online Nov 21, 2019. doi: 10.3748/wjg.v25.i43.6404
Peer-review started: September 2, 2019
First decision: September 19, 2019
Revised: October 15, 2019
Accepted: October 30, 2019
Article in press: October 30, 2019
Published online: November 21, 2019
Processing time: 80 Days and 6.6 Hours
Developing mitochondrial regulators/nutrients from natural products to remedy mitochondrial dysfunction represent attractive strategies for therapy of non-alcoholic fatty liver disease (NAFLD). Polygonatum kingianum (PK) has been traditionally used in China as a medicinal and nutritional ingredient for centuries and can alleviate high-fat diet (HFD)-induced NAFLD by promoting mitochondrial functions. To date, the underlying molecular mechanism of PK for treating mitochondrial dysfunctions and thus alleviating NAFLD remains unclear.
To identify the molecular mechanism behind the mitochondrial regulatory action of PK against HFD-induced NAFLD in rats.
NAFLD model was induced in rats with HFD. The rats were intragastrically administered PK (4 g/kg per day) for 14 wk. Metabolites in hepatic mitochondrial samples were profiled through ultra-high performance liquid chromatography/mass spectrometry followed by multivariate statistical analysis to find the potential biomarkers and metabolic pathways.
PK significantly restored the metabolites’ levels in the mitochondrial samples. Ten potential biomarkers were identified in the analyzed samples. These biomarkers are involved in riboflavin metabolism.
PK can alleviate HFD-induced NAFLD by regulating the riboflavin metabolism and further improving the mitochondrial functions. Thus, PK is a promising mitochondrial regulator/nutrient for alleviating NAFLD-associated diseases.
Core tip: We identified the molecular mechanism behind the mitochondrial regulatory action of Polygonatum kingianum against high-fat diet (HFD)-induced non-alcoholic fatty liver disease (NAFLD) in rats using an integrated mitochondrial metabolomic method. The results indicated that Polygonatum kingianum can alleviate HFD-induced NAFLD by regulating riboflavin metabolism, increasing flavin mononucleotide content and further improving mitochondrial functions. Polygonatum kingianum as a promising mitochondrial regulator/nutrient can alleviate NAFLD-associated diseases.
- Citation: Yang XX, Wei JD, Mu JK, Liu X, Li FJ, Li YQ, Gu W, Li JP, Yu J. Mitochondrial metabolomic profiling for elucidating the alleviating potential of Polygonatum kingianum against high-fat diet-induced nonalcoholic fatty liver disease. World J Gastroenterol 2019; 25(43): 6404-6415
- URL: https://www.wjgnet.com/1007-9327/full/v25/i43/6404.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i43.6404
Non-alcoholic fatty liver disease (NAFLD), the most prevalent chronic liver disease, is characterized by the accumulation of lipids in the liver, and it can progress to inflammatory non-alcoholic steatohepatitis, fibrosis, cirrhosis, and hepatocellular carcinoma and eventually culminate in liver failure[1,2]. Apart from dieting and regular exercising, no other treatment is recommended for NAFLD[3].
Although the underlying mechanism of NAFLD has not yet been clarified, mitochondrial dysfunction might be involved in the pathogenesis and development of NAFLD[1,4]. Mitochondria play a crucial role in energy production, apoptotic cell death, oxidative stress, calcium homeostasis, and lipid metabolism[5]. Recent scientific reports suggest that natural products, such as Shexiang Baoxin Pill, Cyclocarya paliurus, and epigallocatechin gallate can treat NAFLD by regulating the mitochondrial function[6]. Thus, mitochondrial regulators/nutrients can be developed from natural products that which can be utilized for regulating NAFLD-associated mitochondrial dysfunction.
Polygonatum kingianum (PK) has been traditionally used in China as a medicinal and nutritional ingredient for centuries. Many varieties of compounds have been isolated from PK, such as polysaccharides, steroidal saponins, triterpenoid saponins, homoisoflavanones, flavonoids, alkaloids, lignins, and lectins. Among these compounds, the main active compounds are polysaccharides, steroidal saponins, triterpenoid saponins, and homoisoflavanones[7]. PK possesses various pharma-cological activities, such as immuno-stimulatory, anti-aging, blood glucose, and lipid regulatory properties[8,9]. Our previous studies have reported that PK can alleviate high-fat diet (HFD)-induced NAFLD by significantly promoting mitochondrial functions. Therefore, the herb can be utilized for treating mitochondrial dysfunction and alleviate NAFLD as a mitochondrial regulator/nutrient[6]. Additionally, PK can alleviate HFD-induced dyslipidemia through remedying a large number of endogenous metabolites in serum, urine, and liver samples[10]. However, the underlying molecular mechanism of PK for treating mitochondrial dysfunctions and thus alleviating NAFLD remains unclear.
Metabolomics can comprehensively profile and characterize the intermediates and end products of cellular metabolism in body fluids, tissues, cells, etc. It is a novel approach for evaluating the efficacies and the mechanism of action of natural drugs[11]. However, metabolic profiling of whole cell (body fluid or tissue) is probably unsuitable for monitoring mitochondrial alterations after drug treatment (though mitochondria contribute a small fraction to cellular contents). Thus, mitochondrial metabolomics can be applied for studying mitochondria-related diseases, as this technique can identify potential functional alterations in multiple metabolic pathways and signaling networks at the subcellular level[12].
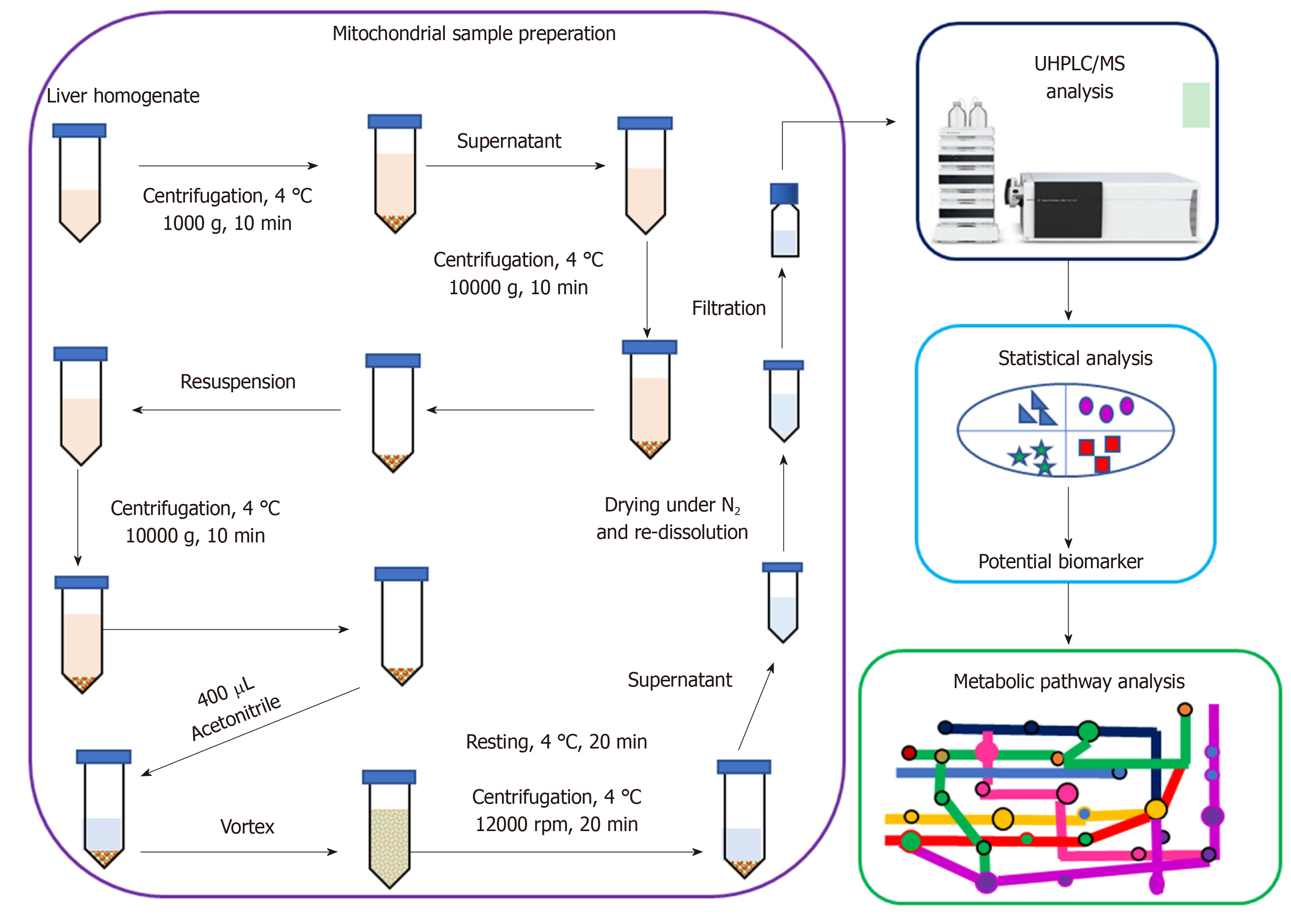
The present study was aimed at elucidating the underlying molecular mechanism of PK for regulating mitochondrial dysfunction and thus alleviating HFD-induced NAFLD in rats. An integrated mitochondrial metabolomic method, based on ultra-high performance liquid chromatography/mass spectrometry (UHPLC/MS), was applied for analyzing mitochondrial samples from rat liver (Figure 1). The results indicate that PK can alleviate HFD-induced NAFLD by regulating many endogenous mitochondrial metabolites in hepatic mitochondrial samples. Thus, PK may be applied as a promising mitochondrial regulator/nutrient for alleviating NAFLD-associated diseases.
HPLC-grade acetonitrile and formic acid were purchased from Merck (Darmstadt, Germany). Basic rodent diet was purchased from Suzhou Shuangshi Experimental Animal Feed Technology Co., Ltd. (Suzhou, China). Simvastatin was provided by Hangzhou Merck East Pharmaceutical Co., Ltd. (Hangzhou, China). High-purity deionized-water was prepared with a Milli-Q System (Millipore, Bedford, MA, United States). Cholesterol, refined lard, and eggs were supplied by Beijing Boao Extension Co., Ltd. (Beijing, China), Sichuan Green Island Co., Ltd. (Chengdu, China), and Wal-Mart Supermarket (Kunming, China), respectively. All other reagents were of analytical grade. The rhizome of PK was obtained from Wenshan Shengnong Trueborn Medicinal Materials Cultivation Cooperation Society (Wenshan, China) on 7 April 2017. Samples were authenticated by Professor Jie Yu, and a voucher specimen of PK (No. 8426) was deposited in College of Pharmaceutical Science, Yunnan University of Chinese Medicine (Kunming, China).
PK was extracted following a previously described method[6]. Briefly, the dried rhizome of PK was permeated in a five-fold volume of Shaoxing Rice Wine (Beijing Ershang Wangzhihe Food Co., Ltd., Beijing, China) and then steamed in a steam sterilizer (LDZX-50 KBS, Shanghai Shenan Medical Instrument Factory, Shanghai, China) for 2.5 h at 120 °C. The steamed samples were dried at 60 °C. Then, the obtained materials were pulverized, immersed in seven-fold volumes of water for 30 min and decocted for 60 min. The extracted liquids were filtered and collected. The filter residues were decocted with seven-fold volumes of water for another 60 min, and the extracted solution was leached. The filtrates were mixed and concentrated by an rotatory evaporator (R-210; Büchi Labortechnik AG, Flawil, Switzerland) under reduced pressure at 50 °C. The concentrates were then lyophilized by an freeze dryer (FD5-3; SIM International Group Co. Ltd., Newark, DE, United States). The powder was preserved in a desiccator until use.
The animal experiments were performed following the Guide for the Care and Use of Laboratory Animals as published by the US National Institutes of Health and approved by the Institutional Ethical Committee on Animal Care and Experimentations of Yunnan University of Chinese Medicine (R-0620160026) (Kunming, China). Special care was taken to minimize the animals’ suffering.
Male Sprague-Dawley rats (200 ± 50 g) were supplied by Dashuo Biotech Co., Ltd. (Chengdu, China). The animals were bred under a controlled environment (60 ± 10% humidity; 22 ± 1 °C temperature; and a 12 h/12 h light/dark cycle) with ad libitum access to commercial laboratory food and tap water. Rats were randomized into four groups (n = 5 rats per group), including normal control (normal saline), model (normal saline), simvastatin (1.8 mg/kg per day), and PK groups (4 g/kg). They were intragastrically administered with the tested samples (or normal saline) once a day for 14 wk. Simvastatin and PK were separately dissolved in normal saline. NAFLD was triggered by feeding with HFD (comprised of 1% cholesterol, 10% refined lard, 10% eggs, and 79% basic feed) for 14 wk in all groups except the normal group. After the last administration, rats were fasted for 12 h and anesthetized by chloral hydrate. The liver was collected and stored at -80 °C until use.
Firstly, liver mitochondria were isolated following a previously described method[5]. Briefly, rat liver (0.1 g) was quickly placed into ice-cold isolation buffer (210 nmol/L mannitol, 70 nmol/L sucrose, 10 nmol/L Tris base, 1 nmol/L EDTA, and 0.5 nmol/L EGTA, pH 7.4) to remove blood, minced into 1 mm3, and then homogenized with isolation buffer with a Dounce glass homogenizer (Kimble/Kontes, Vineland, NJ, United States). After centrifuging at 1000 × g for 10 min, the supernatant was collected and centrifuged at 10000 × g for 10 min. The pellet was resuspended in isolation buffer and centrifuged at 10000 × g for 10 min to isolate mitochondria. Next, the isolated mitochondria were resuspended in 400 μL of acetonitrile. After vortex-mixing and allowing to stand for 20 min at 4 °C, the samples were centrifuged at 12000 rpm for 15 min at 4 °C. The collected supernatants were dried under a nitrogen stream. The residues were re-dissolved in acetonitrile (100 μL) for mitochondrial metabolomics analysis.
UHPLC/MS analyses were performed using a UHPLC (Dionex Ultimate 3000 system) coupled with a hybrid quadrupole-orbitrap mass spectrometer (Thermo Scientific Q-Exactive TM) with a heated-electrospray ionization probe (Thermo Fisher Scientific, San Jose, CA, United States). The UHPLC system comprised of a quaternary pump, a temperature controlled autosampler, a column box, and a photodiode array detector.
The UHPLC conditions were as follows: (1) Chromatographic column: Thermo C18 column (100 mm × 2.1 mm I.D., 1.9 μm); (2) Column temperature: 30 °C; (3) Mobile phase: Acetonitrile (A) and 0.1% formic acid (B) with a gradient program (0-3 min, 5% A; 3-5 min, 5% A-23% A; 5-10 min, 23% A-43% A; 10-13 min, 43% A-64% A; 13-16 min, 64% A-85% A; 16-18 min, 85% A-100% A; 18-20 min, 100% A-100% A); (4) flow rate: 0.2 mL/min; and (5) Sample injection volume: 4 μL.
Mass spectrometry conditions were as follows: (1) Mode: Positive and negative ion; (2) Heat block and curved desolvation line temperature: 250 °C; nebulizing nitrogen gas flow: 1.5 L/min; interface voltage: (+) 3.5 kV, (-) ﹣2.8 kV; (3) Dynamic exclusion time: 10 s; (4) Mass range: MS, m/z 100-1000; MS2 and MS3, m/z 50-1000; and (5) Workstation: Xcalibur 3.0.63 for liquid chromatography coupled with data processing, molecular prediction, and precise molecular weight calculations.
All the UHPLC/MS files were exported in Xcalibur Raw File (.raw) format and transformed to *.cdf format by Xcalibur 2.0 software (Thermo Fisher Scientific, Waltham, MA, United States). Then, the transformed file was transferred to XCMS online to extract all data (grouping and comparison). Afterward, multivariate data analysis, including principal components analysis (PCA) and orthogonal partial least squares discriminant analysis (OPLS-DA), was undertaken through SIMCA-P 14.1 software (Umetrics, Umeå, Sweden). Next, potential biomarkers were chosen in conformity with the parameters of variable importance in the projection (VIP > 1.0) from OPLS-DA. The metabolites were identified using the METLIN database and compared with previously reported data. Additionally, biochemical reactions involving the confirmed metabolites were searched by the Kyoto Encyclopedia of Genes and Genomes in MetaboAnalyst 4.0 online.
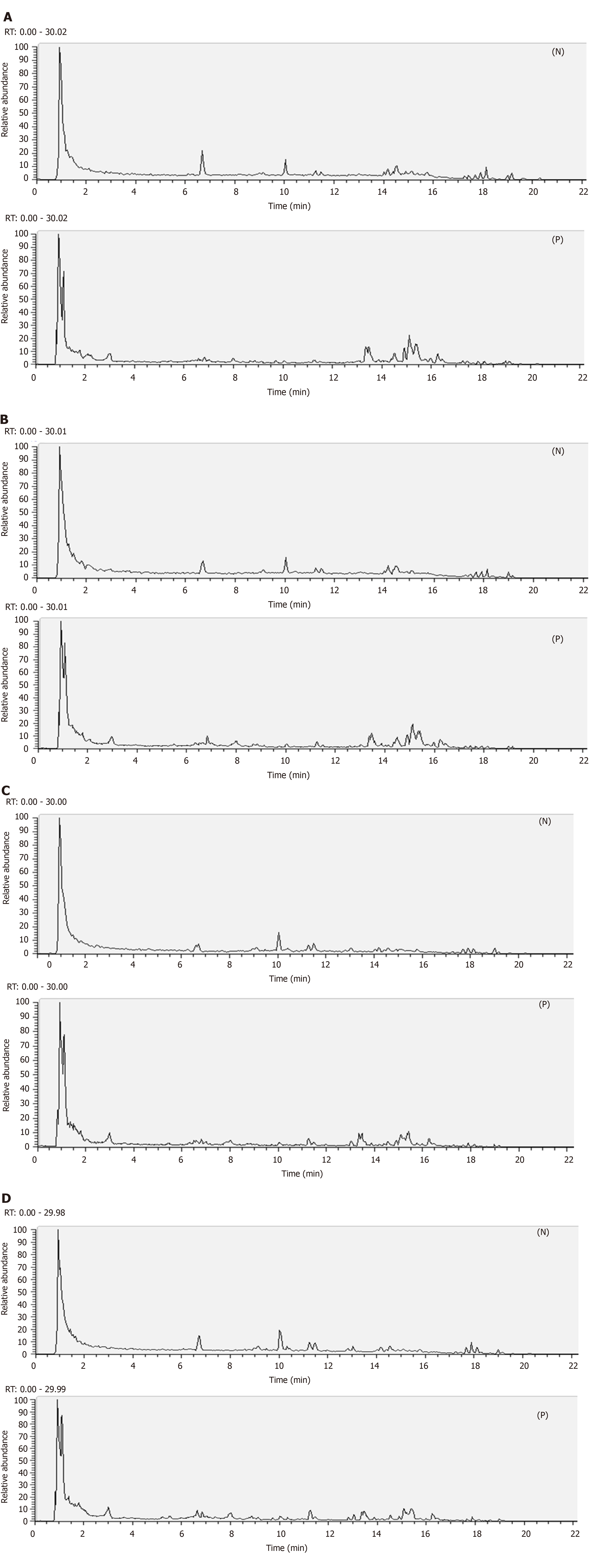
Isolation conditions of the mitochondrial samples on the UHPLC column were optimized regarding peak number and peak shape. Figure 2 displays the representative total ion current profiles of the mitochondrial samples in the four groups at positive and negative ion modes. Many metabolites of the mitochondrial samples were profiled by UHPLC/MS. Furthermore, remarkable differences were noticed in peak number and intensity between the four groups, indicating different metabolomic states in different groups. Thus, the endogenous metabolites in the liver mitochondria were significantly changed after the 14 wk of HFD-feeding and administration of PK and simvastatin. In addition, the significant differences between PK and simvastatin groups indicated the different regulatory mechanisms of the two drugs to the metabolites in the mitochondrial samples.
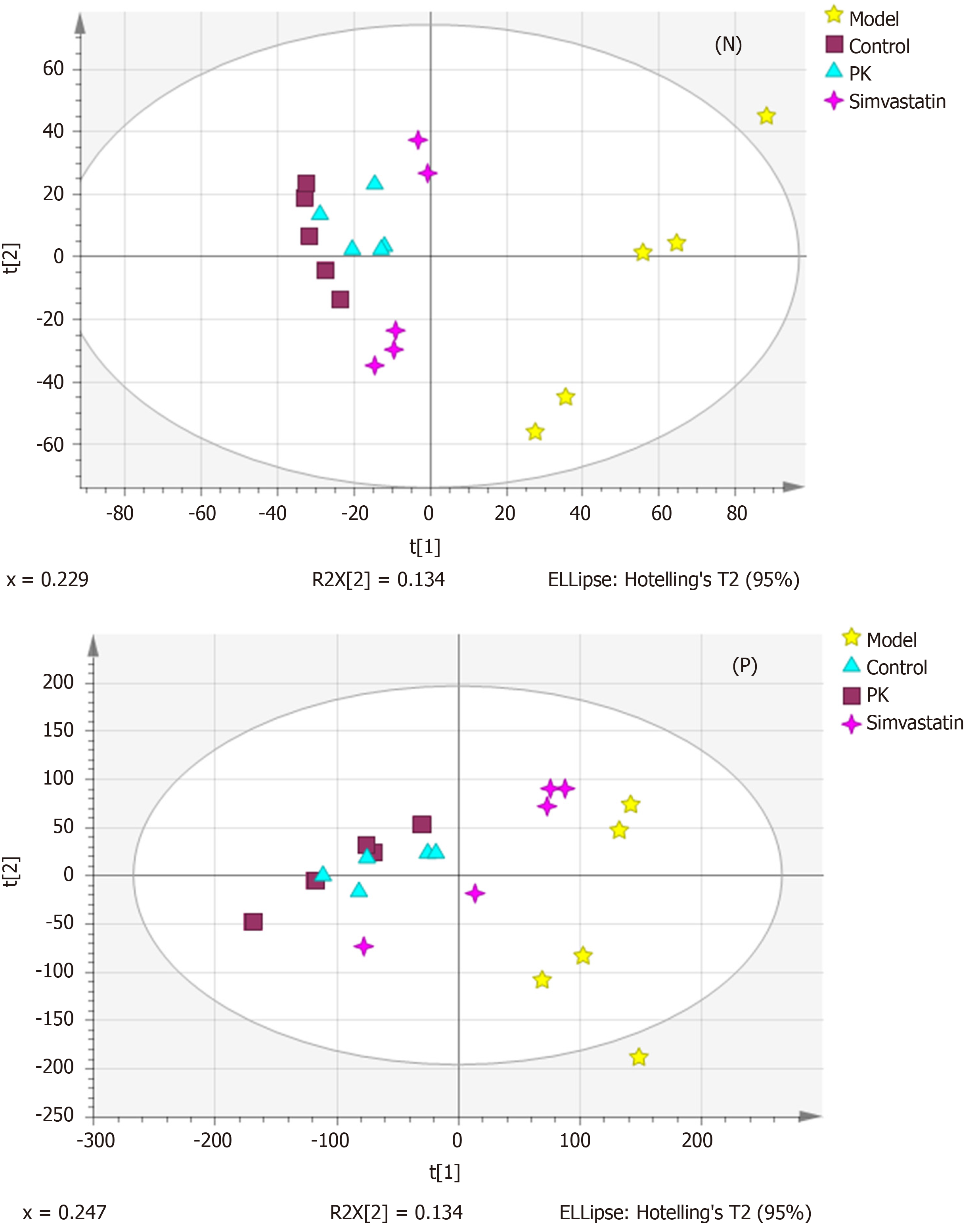
PCA and OPLS-DA are commonly applied for multivariate data analysis due to their ability to analyze highly multivariate, collinear, and possibly incomplete data. Figure 3 shows the PCA score plots of mitochondrial samples in positive and negative ion modes. In the negative ion mode, the normal and model groups were significantly separated, indicating significant differences in the metabolic state between them. The sample sets of PK group showed a tendency to close the normal group and were nearer to the control group than the simvastatin group. In the positive ion mode, the four groups were almost separated, although significant differences were noticed among the sample sets within the same group. Moreover, the normal and model groups were significantly separated, while the PK group approached to the normal group and were nearer to the normal group than the simvastatin group. This phenomenon suggested that PK treatment significantly prevented HFD-induced pathological changes in a better manner than simvastatin.
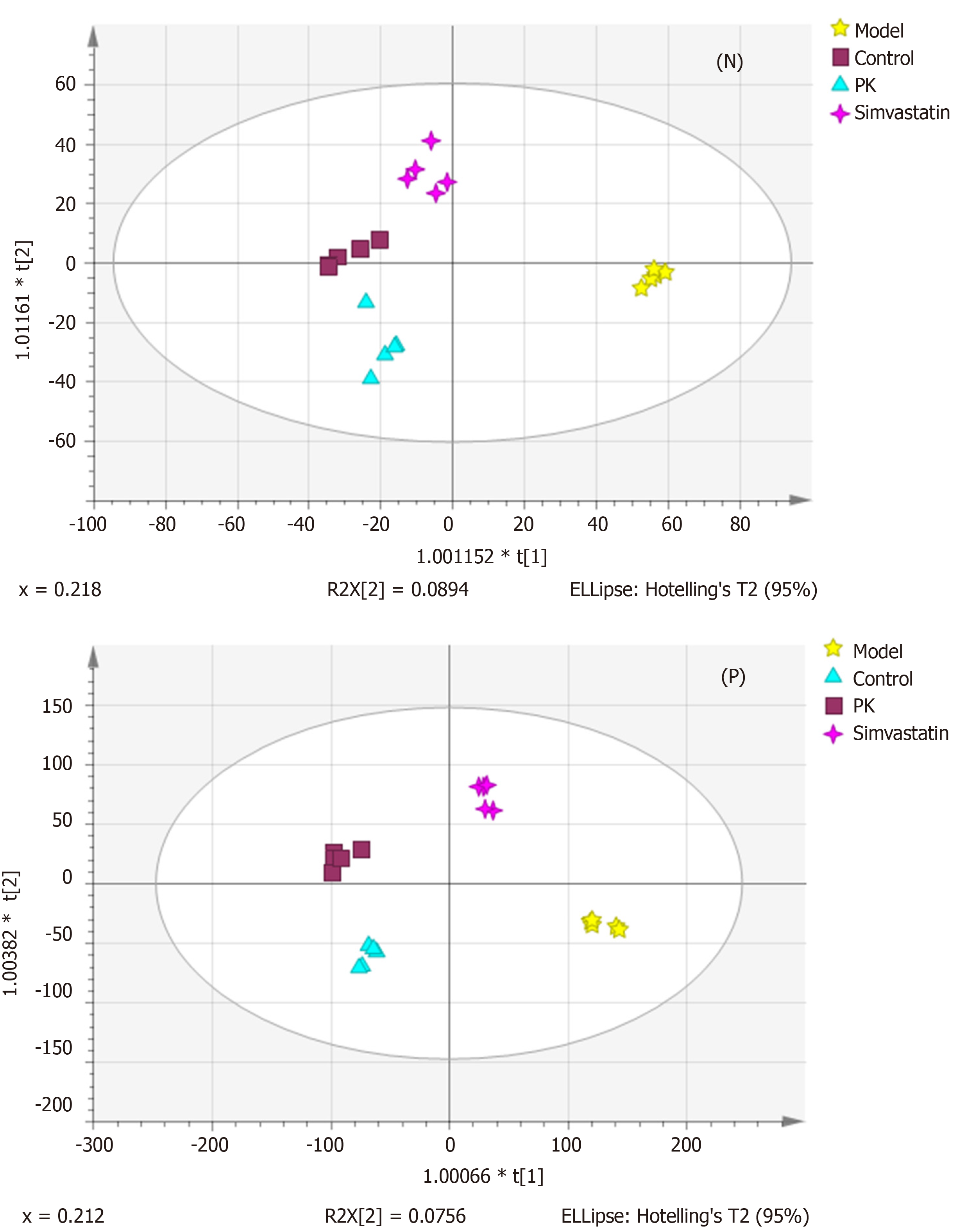
OPLS-DA was performed to validate further the sample isolation process in the four groups, maximize separation between the groups, and find biomarkers in them (Figure 4). The four groups were also remarkably isolated in positive and negative ion modes. The PK and simvastatin groups were both near to the control group, which further demonstrated that PK strongly impeded HFD-induced pathological changes.
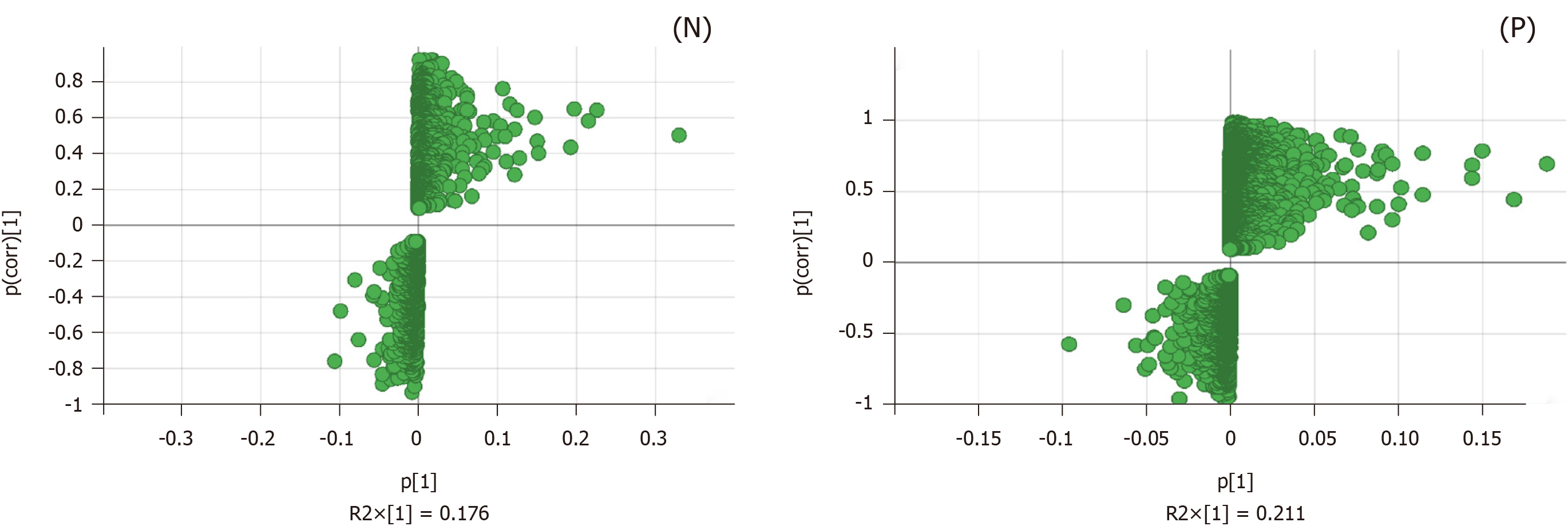
We evaluated the constituent changes after treatment; an S-Plot loading diagram was developed on the basis of OPLS-DA. Each point in Figure 5 represents a variable, i.e. the biomarker responsible for the dissimilarity between the model and PK groups. The importance of each variable to the classification was assessed with the VIP value, and the ingredients with VIP > 1.0 were chosen as potential biomarkers.
Ten molecules were identified from the mitochondrial samples, including eight in the negative ion mode and two in the positive ion mode. Chemically, these compounds were organic acids, amino acids, nucleosides, and organic salts (Table 1).
| Retention time, min | Molecular weight, Da | VIP | Potential biomarker | Formula | Change trend |
| Model-PKRP | |||||
| ESI- | |||||
| 15.0667 | 338.208 | 1.57637 | PGJ2 | C20H30O4 | Up |
| 5.52016 | 203.082 | 1.39807 | Ascorbate-2-sulfate | C6H8O9S | Down |
| 2.09975 | 267.074 | 1.36283 | Allopurinol-1-ribonucleoside | C10H12N4O5 | Down |
| 1.78773 | 298.069 | 1.33035 | D-4'-Phosphopantothenate | C9H18NO8P | Up |
| 15.384 | 293.18 | 1.24297 | Sodium tetradecyl sulfate | C14H30O4S | Up |
| 20.3338 | 383.189 | 1.19012 | Bortezomib | C19H25BN4O4 | Up |
| 3.96113 | 254.08 | 1.14379 | Pantothenic acid | C9H17NO5 | Up |
| 6.51405 | 455.097 | 1.11557 | Flavin mononucleotide | C17H21N4O9P | Up |
| ESI+ | |||||
| 7.0308 | 301.208 | 1.53312 | Pinolenic acid | C18H30O2 | Up |
| 1.09652 | 266.124 | 1.44566 | N6-Methyl-2'-deoxyadenosine | C11H15N5O3 | Up |
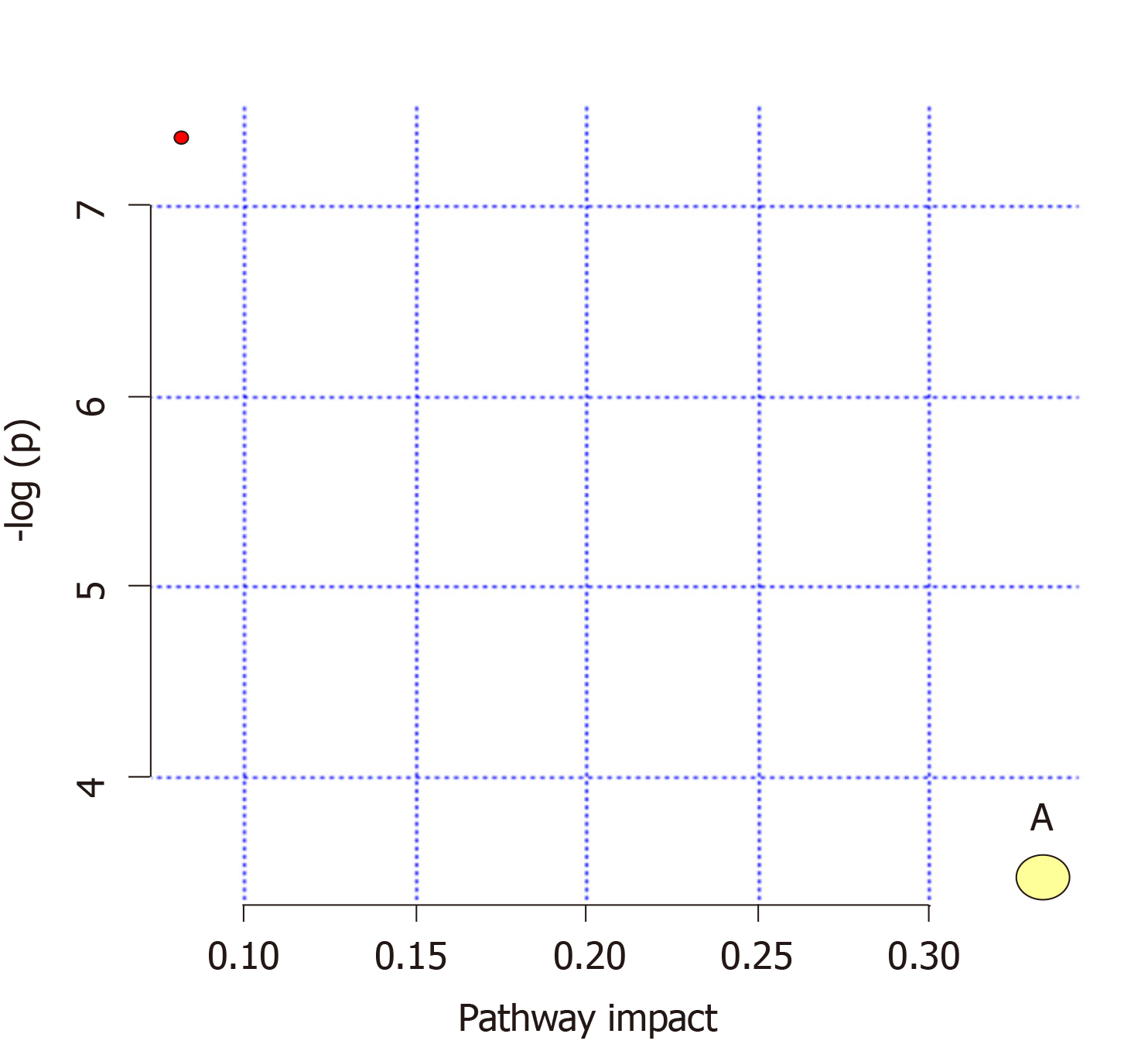
Pathways (influence values greater than 0.1) were chosen as the metabolic pathways[13]. Pathway impact plots were established to visualize the impact of the altered metabolic pathways (Figure 6). Based on the results, the main intervened pathways in mitochondrial samples involved riboflavin metabolism (Table 2).
| No | Pathway | Match status | P-value | Impact | Details |
| 1 | Riboflavin metabolism | 1/11 | 0.031049 | 0.33333 | KEGG |
NAFLD, a chronic liver disease, is associated with excessive lipid accumulation in the liver[14]. Mitochondrial dysfunction is the underlying mechanism of NAFLD. In our previous investigation, PK was established as a potential mitochondrial regulator. PK can treat mitochondrial dysfunctions and alleviate HFD-induced NAFLD. In the present study, a mitochondrial metabolomic method, based on UHPLC/MS analysis of hepatic mitochondrial samples, was applied for investigating the underlying mechanism of PK for regulating mitochondrial function and treating HFD-induced NAFLD in rats. PK and simvastatin showed the different regulatory mechanisms to the metabolites in the mitochondrial samples. Moreover, PK significantly prevented HFD-induced pathological changes in a better manner than simvastatin, which may be attributed to the multifarious constituents and pharmacological mechanism of PK.
In the present study, 10 potential biomarkers involved in riboflavin metabolism were identified from the mitochondrial samples. Flavin mononucleotide (FMN), also known as riboflavin-5-phosphate, is involved in riboflavin metabolism. FMN, an auxiliary group of flavoproteins, is involved in the electron transport mechanism of the biological oxidation processes (e.g., respiration). This biomolecule is synthesized by riboflavin kinase from riboflavin (vitamin B2). Intracellular free radicals are produced at mitochondria, which can oxidize the mitochondrial enzyme complexes and induce mitochondrial respiratory dysfunction. FMN is the active auxiliary group of the flavoproteins in mitochondrial complex I. Studies have shown that FMN can increase the activity of mitochondrial complex I and promote oxygen consumption rate during mitochondrial respiration[15]. Additionally, FMN can be hydrolyzed to riboflavin and phosphoric acid by phosphatase enzyme. Riboflavin, an auxiliary group of flavin dehydrogenases, serves as a hydrogen donor in bio-oxidation. Lack of riboflavin would impair electron transport in the respiratory chain and result in insufficient ATP production. Thus, bio-oxidation and metabolic statuses are affected in the body. Hepatic proteomic analysis identified that riboflavin deficiency might cause a significant decrease in the expression of key proteins that are involved in the beta-oxidation of fatty acid, electron transport of respiratory chain and tricarboxylic acid cycle, and fat accumulation[16]. In this study, FMN content was significantly reduced after HFD-feeding, which caused mitochondrial respiratory dysfunction. However, the FMN content was remarkably increased after the treatment of PK extract, suggesting an increase in the activity of mitochondrial complex I and oxygen consumption during mitochondrial respiration and ATP production. Higher FMN content resisted fat accumulation in the NAFLD rats. The results indicated that PK extract improved mitochondrial function by accelerating riboflavin metabolism and enhancing FMN content in hepatic mitochondria and thus alleviated NAFLD.
In conclusion, PK extract significantly restored different endogenous metabolites (including riboflavin) in the hepatic mitochondrial samples from HFD-induced NAFLD rats. The results suggest that PK can alleviate HFD-induced NAFLD by regulating riboflavin metabolism, increasing FMN content, and further improving mitochondrial functions. Thus, PK as a promising mitochondrial regulator/nutrient can alleviate NAFLD-associated diseases.
Non-alcoholic fatty liver disease (NAFLD) is the most prevalent chronic liver disease. Mitochondrial dysfunction is the mechanism of NAFLD. Developing mitochondrial regulators/nutrients from natural products to remedy mitochondrial dysfunction represents an attractive strategy for NAFLD therapy. Polygonatum kingianum (PK) has been traditionally used in China as a medicinal and nutritional ingredient for centuries and can alleviate high-fat diet (HFD)-induced NAFLD by significantly promoting mitochondrial functions.
To date, the underlying molecular mechanism of PK for treating mitochondrial dysfunctions and thus alleviating NAFLD remains unclear.
We aimed to identify the molecular mechanism behind the mitochondrial regulatory action of PK against HFD-induced NAFLD in rats.
NAFLD model was induced in rats with HFD. The rats were intragastrically administered PK (4 g/kg per day) for 14 wk. Metabolites in hepatic mitochondrial samples were profiled through ultra-high performance liquid chromatography/mass spectrometry followed by multivariate statistical analysis to find the potential biomarkers and metabolic pathways.
PK significantly restored the metabolites’ levels in the mitochondrial samples. Ten potential biomarkers were identified in the analyzed samples. These biomarkers are involved in riboflavin metabolism.
PK can alleviate HFD-induced NAFLD by regulating riboflavin metabolism and further improving the mitochondrial functions.
PK is a promising mitochondrial regulator/nutrient for alleviating NAFLD-associated diseases.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Adams JD, Shimizu Y S-Editor: Tang JZ L-Editor: Filipodia E-Editor: Ma YJ
| 1. | Le J, Jia W, Sun Y. Sennoside A protects mitochondrial structure and function to improve high-fat diet-induced hepatic steatosis by targeting VDAC1. Biochem Biophys Res Commun. 2018;500:484-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 2. | Im AR, Kim YH, Kim YH, Yang WK, Kim SH, Song KH. Dolichos lablab Protects Against Nonalcoholic Fatty Liver Disease in Mice Fed High-Fat Diets. J Med Food. 2017;20:1222-1232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 3. | Lindquist C, Bjørndal B, Rossmann CR, Svardal A, Hallström S, Berge RK. A fatty acid analogue targeting mitochondria exerts a plasma triacylglycerol lowering effect in rats with impaired carnitine biosynthesis. PLos One. 2018;13:e0194978. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Simões ICM, Fontes A, Pinton P, Zischka H, Wieckowski MR. Mitochondria in non-alcoholic fatty liver disease. Int J Biochem Cell Biol. 2018;95:93-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 124] [Cited by in RCA: 183] [Article Influence: 22.9] [Reference Citation Analysis (0)] |
| 5. | Pathania D, Millard M, Neamati N. Opportunities in discovery and delivery of anticancer drugs targeting mitochondria and cancer cell metabolism. Adv Drug Deliv Rev. 2009;61:1250-1275. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 212] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 6. | Yang XX, Wang X, Shi TT, Dong JC, Li FJ, Zeng LX, Yang M, Gu W, Li JP, Yu J. Mitochondrial dysfunction in high-fat diet-induced nonalcoholic fatty liver disease: The alleviating effect and its mechanism of Polygonatum kingianum. Biomed Pharmacother. 2019;117:109083. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 37] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 7. | Zhao P, Zhao C, Li X, Gao Q, Huang L, Xiao P, Gao W. The genus Polygonatum: A review of ethnopharmacology, phytochemistry and pharmacology. J Ethnopharmacol. 2018;214:274-291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 142] [Cited by in RCA: 206] [Article Influence: 29.4] [Reference Citation Analysis (0)] |
| 8. | Yan H, Lu J, Wang Y, Gu W, Yang X, Yu J. Intake of total saponins and polysaccharides from Polygonatum kingianum affects the gut microbiota in diabetic rats. Phytomedicine. 2017;26:45-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 157] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 9. | Lu JM, Wang YF, Yan HL, Lin P, Gu W, Yu J. Antidiabetic effect of total saponins from Polygonatum kingianum in streptozotocin-induced daibetic rats. J Ethnopharmacol. 2016;179:291-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 49] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 10. | Yang XX, Wei JD, Mu JK, Liu X, Dong JC, Zeng LX, Gu W, Li JP, Yu J. Integrated metabolomic profiling for analysis of antilipidemic effects of Polygonatum kingianum extract on dyslipidemia in rats. World J Gastroenterol. 2018;24:5505-5524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 11. | Tao Y, Chen X, Li W, Cai B, Di L, Shi L, Hu L. Global and untargeted metabolomics evidence of the protective effect of different extracts of Dipsacus asper Wall. ex C.B. Clarke on estrogen deficiency after ovariectomia in rats. J Ethnopharmacol. 2017;199:20-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 12. | Liu X, Xu G. Recent advances in using mass spectrometry for mitochondrial metabolomics and lipidomics - A review. Anal Chim Acta. 2018;1037:3-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Xie HH, Xie T, Xu JY, Shen CS, Lai ZJ, Xu NS, Wang SC, Shan JJ. Metabolomics study of aconitine and benzoylaconine induced reproductive toxicity in BeWo cell. Fenxi Huaxue. 2015;43:1808–1813. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 14. | Priore P, Cavallo A, Gnoni A, Damiano F, Gnoni GV, Siculella L. Modulation of hepatic lipid metabolism by olive oil and its phenols in nonalcoholic fatty liver disease. IUBMB Life. 2015;67:9-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 15. | Holt PJ, Efremov RG, Nakamaru-Ogiso E, Sazanov LA. Reversible FMN dissociation from Escherichia coli respiratory complex I. Biochim Biophys Acta. 2016;1857:1777-1785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 38] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 16. | Tang J, Hegeman MA, Hu J, Xie M, Shi W, Jiang Y, de Boer V, Guo Y, Hou S, Keijer J. Severe riboflavin deficiency induces alterations in the hepatic proteome of starter Pekin ducks. Br J Nutr. 2017;118:641-650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |