Published online May 21, 2016. doi: 10.3748/wjg.v22.i19.4757
Peer-review started: January 26, 2016
First decision: March 21, 2016
Revised: March 29, 2016
Accepted: April 7, 2016
Article in press: April 7, 2016
Published online: May 21, 2016
Processing time: 112 Days and 20.2 Hours
AIM: To explore Chinese physicians’ perceptions towards fecal microbiota transplantation (FMT) and to provide information and an assessment of FMT development in China.
METHODS: A self-administered questionnaire was developed according to the FMT practice guidelines and was distributed to physicians in hospitals via Internet Research Electronic Data Capture (REDcap) software and electronic mails to assess their attitudes toward and knowledge of FMT. The questionnaire included a brief introduction of FMT that was followed by 20 questions. The participants were required to respond voluntarily, under the condition of anonymity and without compensation. Except for the fill-in-the-blank questions, all of the other questions were required in the REDcap data collection systems, and the emailed questionnaires were completed based on eligibility.
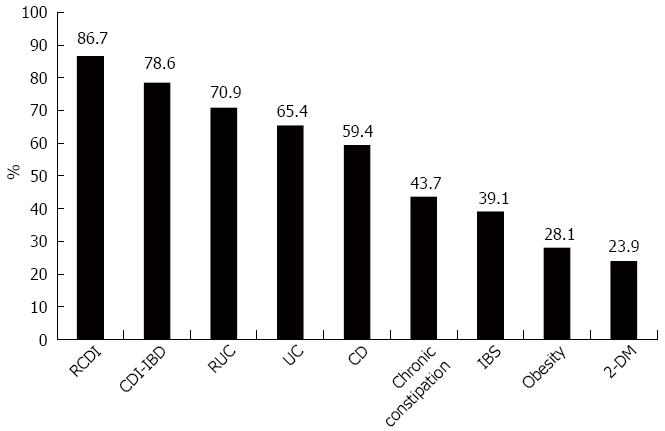
RESULTS: Up to December 9, 2014, 844 eligible questionnaires were received out of the 980 distributed questionnaires, with a response rate of 86.1%. Among the participants, 87.3% were from tertiary hospitals, and there were 647 (76.7%) gastroenterologists and 197 (23.3%) physicians in other departments (non-gastroenterologists). Gastroenterologists’ awareness of FMT prior to the survey was much higher than non-gastroenterologists’ (54.3 vs 16.5%, P < 0.001); however, acceptance of FMT was not statistically different (92.4 vs 87.1%, P = 0.1603). Major concerns of FMT included the following: acceptability to patients (79.2%), absence of guidelines (56.9%), and administration and ethics (46.5%). On the basis of understanding, the FMT indications preferred by physicians were recurrent Clostridium difficile infection (86.7%), inflammatory bowel disease combined with Clostridium difficile infection (78.6%), refractory ulcerative colitis (70.9%), ulcerative colitis (65.4%), Crohn’s disease (59.4%), chronic constipation (43.7%), irritable bowel syndrome (39.1%), obesity (28.1%) and type 2 diabetes (23.9%). For donor selection, the majority of physicians preferred individuals with a similar gut flora environment to the recipients. 76.6% of physicians chose lower gastrointestinal tract as the administration approach. 69.2% of physicians considered FMT a safe treatment.
CONCLUSION: Chinese physicians have awareness and a high acceptance of FMT, especially gastroenterologists, which provides the grounds and conditions for the development of this novel treatment in China. Physicians’ greatest concerns were patient acceptability and absence of guidelines.
Core tip: Perceptions and attitudes toward fecal microbiota transplantation (FMT) by physicians and patients play an important role in determining its acceptability. We investigated Chinese physicians’ acceptance levels of FMT, their concerns about FMT, and their perspectives of FMT techniques. The few data about the perceptions of physicians toward FMT are all from Western countries; this is the first study of physicians’ perceptions of FMT in an Asian country. Additionally, our study was representative with a large respondent number (844) and a large coverage area of China (22 out of 34 provinces); thus it can provide preliminary information for the development of FMT in China.
- Citation: Ren RR, Sun G, Yang YS, Peng LH, Wang SF, Shi XH, Zhao JQ, Ban YL, Pan F, Wang XH, Lu W, Ren JL, Song Y, Wang JB, Lu QM, Bai WY, Wu XP, Wang ZK, Zhang XM, Chen Y. Chinese physicians’ perceptions of fecal microbiota transplantation. World J Gastroenterol 2016; 22(19): 4757-4765
- URL: https://www.wjgnet.com/1007-9327/full/v22/i19/4757.htm
- DOI: https://dx.doi.org/10.3748/wjg.v22.i19.4757
Fecal microbiota transplantation (FMT) refers to the instillation of fecal suspension from a healthy person into the gastrointestinal (GI) tract of a patient to cure a certain disease by restoring the construction of intestinal flora. FMT is by no means a new concept. Fecal medicine was recorded 3000 years ago in the “Collection of 52 Prescriptions”[1,2], which was described as the oldest traditional prescription book in China. Later, during the Eastern Han dynasty in the 2nd century A.D. in China, Zhang Zhongjing described the use of a human fecal suspension by mouth to treat food poisoning in “Jin Gui Yao Lüe” (Synopsis of Golden Chamber)[3]. To our knowledge, this was the first literary record of using human fecal liquid to treat diseases. Then, Ge Hong, Sun Simiao, Li Shizhen, etc., described a series of prescriptions using fecal suspensions or dry feces to treat abdominal diseases in their famed traditional Chinese medicine books[4-6]. The first description of FMT in Western countries was in 1958, when four patients with pseudomembranous colitis were cured using fecal enemas[7]. However, FMT did not gain public attention until recently and only after several studies reported that fecal suspension had astounding efficacy for recurrent Clostridium difficile infection (RCDI)[8,9]. Since then, FMT, an ancient medicine, has become a hot topic and interest has surged in recent years. Currently, more than 40 reports are available about treating RCDI with FMT, with similarly high reported efficacy. FMT was recommended by the American CDI guidelines in 2013 if there was a third recurrence after a pulsed vancomycin regimen[10]. As FMT may restore the dysbiosis of gut microbiota, it is also proposed in treating other GI diseases and non-GI diseases, which have been considered to be linked to the composition of gut microbiome, with associations described between intestinal flora, immune system, and active metabolites[11], such as in inflammatory bowel diseases (IBD), chronic constipation, type 2 diabetes mellitus, metabolic syndrome, and symptoms of Parkinson’s disease[12-15]. However, using fecal suspension to treat diseases other than CDI is still speculative, even for IBD.
The perceptions and attitudes toward FMT held by physicians and patients play an important role in determining its acceptability. A few reports discuss patients’ attitudes towards the acceptance of FMT[16,17]. Despite the unappealing nature of stool, 46% of patients with ulcerative colitis were willing to accept FMT as a treatment, and if it was recommended by their physicians, up to 94% of patients with recurrent CDI are ready to accept FMT[16]. One study reported that 97% of patients with RCDI who had undergone FMT once were willing to accept the treatment again, and an equal number of patients (53%) chose FMT as the treatment of first choice[17]. Nevertheless, minimal data exist regarding physicians’ perception of this technique[18,19]. The acceptance of FMT in Asian countries remains unknown. Therefore, this survey was designed to evaluate Chinese physicians’ perceptions, and especially their acceptance of FMT. We will compare the different views about FMT technology, to provide information and an assessment of the future development of FMT.
The study was conducted from June 2014 to September 2014. A self-administered questionnaire was developed according to the practice guidelines and other literature on FMT[9,20] and was distributed to physicians via Internet Research Electronic Data Capture (REDcap) software[21] and emails. The participants were a convenience sample of physicians working in hospitals and practicing gastroenterology; other specialists, such as those physicians working in endocrinology, pediatrics, general surgery, and neurosurgery, were also included in the study. These physicians were recruited through gastroenterology associations and their subspecialty groups in different provinces.
The questionnaire included a brief introduction of FMT, followed by 20 questions, which were comprised of three sections: demographic information of the interviewees, their attitudes toward FMT, and FMT technique-associated questions (see Supplementary material). The participants were required to respond voluntarily and under the condition of anonymity and without compensation. Except for the fill-in-the-blank questions, all other questions were required in the REDcap system. The email questionnaires were completed according to eligibility.
Study data were collected and managed using REDCap tools hosted at the General Hospital of the Chinese PLA. REDCap was used to manage study data and perform the descriptive analysis. The data were also analyzed using Microsoft Excel and JMP 10.0.0 software. Continuous data are presented as the mean ± SD and analyzed by the ANOVA test. Categorical data are presented as percentages and were analyzed by the χ2 test. Univariate analysis and multivariate logistic regression analysis were employed to identify the impact of various factors on physicians’ preferences for FMT. Odds ratios (ORs) and 95% confidence intervals were calculated and a P-value less than 0.05 was considered statistically significant.
Up until December 9, 2014, 844 eligible questionnaires were received out of the 980 distributed questionnaires, with a response rate of 86.1%. Respondents were selected from six different regions of China, and the study included respondents from most areas of China (22 out of 34 provinces). There were 449 (53.2%) females and 395 (46.8%) males with an average age of 36.1 ± 9.2 years (age range: 19-81 years). The majority of respondents were gastroenterologists (76.7%, 647/844), and most of them were associated with tertiary hospitals (87.3%, 737/844). More than half of the physicians were qualified postgraduates or above, and almost half of the physicians held senior professional titles and had worked in gastroenterology for more than 6 years (Table 1).
| Characteristic | n = 844 |
| Age, mean ± SD (range) | 36.1 ± 9.2 (19-81) |
| Gender, male | 395 (46.8) |
| Region | |
| North West | 211 (32.7) |
| North | 152 (23.6) |
| East | 100 (15.5) |
| North East | 83 (12.9) |
| South West | 68 (10.5) |
| South Central | 31 (4.8) |
| Missing data | 199 |
| Education | |
| College degree | 295 (35.0) |
| Postgraduate degree | 341 (40.4) |
| Doctoral degree | 188 (22.3) |
| Post-doctoral degree | 19 (2.3) |
| Professional title | |
| Resident physician | 291 (34.6) |
| Attending physician | 210 (24.9) |
| Associated chief physician | 198 (23.5) |
| Chief physician | 143 (17.0) |
| Level of hospital | |
| Community hospital | 15 (1.8) |
| Secondary hospital | 88 (10.4) |
| Tertiary hospital | 737 (87.3) |
| Profession | |
| Gastroenterologist | 647 (76.7) |
| General surgeons | 49 (5.8) |
| Endocrinologist | 28 (3.3) |
| Others | 120 (14.2) |
| Working time in gastroenterology (yr) | |
| < 2 | 295 (35.3) |
| 3-5 | 111 (13.3) |
| 6-10 | 106 (12.7) |
| 10-20 | 188 (22.5) |
| > 20 | 135 (16.2) |
Among the physicians, 607 (71.9%) had heard of FMT prior to the survey, but only 45.6% (385/844) had an awareness or understanding of FMT (i.e., “had knowledge of FMT principles and technology”). The primary advertising approach included conferences (60.3%, mainly domestic conferences), professional journals (54.8%) and communication with colleagues (42.1%). Gastroenterologists’ prior awareness of FMT was much higher than non-gastroenterologists’ (54.3 vs 16.5%, P < 0.001), they were more interested in FMT training (92.4 vs 81.4%, P < 0.001), and they showed a more positive attitude to the feasibility (74.5 vs 59.3%, P < 0.001) and potential (71.5 vs 53.9%, P < 0.001) of FMT. However, the acceptance of FMT was similarly high among gastroenterologists and non-gastroenterologists (92.4 vs 87.1%, P = 0.1603).
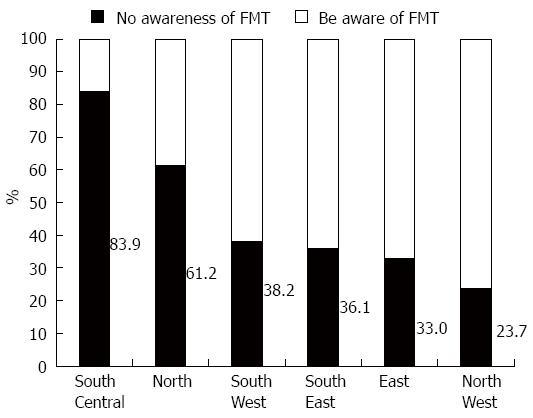
In the univariate analysis, significant factors (P < 0.05) that influenced physicians’ awareness of FMT included age, educational background, professional designation, level of hospital, region, department and working experience in gastroenterology. The multivariate logistic regression analysis confirmed that physicians with a higher education (OR = 1.958, 95%CI: 1.402-2.733, P < 0.001) and a higher professional title (OR = 1.676, 95%CI: 1.133-2.480, P = 0.010) were more likely to understand FMT, and gastroenterologists were more likely to comprehend FMT than physicians in other departments (OR = 4.182, 95%CI: 1.895-9.229, P < 0.001). Physicians in different regions had significantly different understandings of FMT (P < 0.001) (Figure 1 and Table 2).
| Variable | P-vaule | OR | 95%CI |
| Age | 0.160 | 1.391 | 0.878-2.203 |
| Region | < 0.0011 | ||
| Region (North) | < 0.0011 | 0.288 | 0.163-0.508 |
| Region (North East) | 0.0111 | 0.385 | 0.185-0.800 |
| Region (South Central) | 0.058 | 3.005 | 0.963-9.376 |
| Region (East) | 0.089 | 0.555 | 0.282-1.093 |
| Region (South West) | 0.051 | 0.467 | 0.217-1.003 |
| Educational background | < 0.0011 | 1.958 | 1.402-2.733 |
| Professional title | 0.0101 | 1.676 | 1.133-2.480 |
| Level of hospital | 0.069 | 1.759 | 0.958-3.228 |
| Department | 0.0011 | ||
| Department (gastroenterology) | < 0.0011 | 4.182 | 1.895-9.229 |
| Department (general surgery) | 0.104 | 2.429 | 0.834-7.073 |
| Department (endocrinology) | 0.903 | 0.919 | 0.235-3.584 |
| Working time on gastroenterology | 0.476 | 1.090 | 0.860-1.383 |
The acceptance rate of the 385 physicians who had knowledge of FMT was 91.9%. Of these physicians, 59.5% (229/385) were willing to choose FMT ahead of other treatments, and 80.8% (126/156) of physicians who declined FMT as the first treatment selected FMT as an alternative treatment.
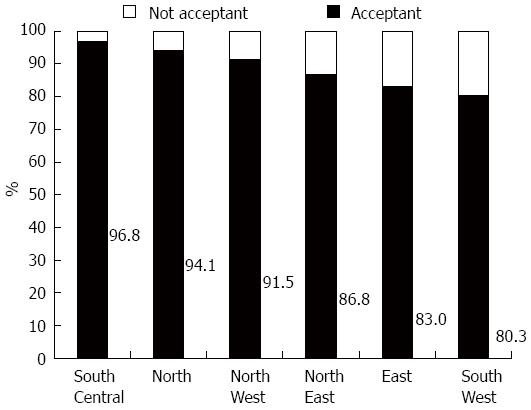
A univariate analysis revealed that only geographic region can significantly influence physicians’ acceptance (P < 0.05). Factoring the significant variables in a univariate analysis and those affecting the acceptance of FMT, such as age, educational background, professional title, hospital level, department, working time in gastroenterology and understandings of FMT into the multivariate logistic regression analysis, it was unexpectedly discovered that understandings of FMT, hospital level and region were all statistically significant (Table 3). Physicians with a greater comprehension of FMT were more likely to accept FMT (OR = 3.265, 95%CI: 1.555-6.855, P = 0.002). The higher the level of hospital physicians worked at, the less likely they were to accept FMT (OR = 0.359, 95%CI: 0.134-0.961, P = 0.041). The lowest acceptance of FMT (80.3%) was observed among physicians working in Southwest China, followed by those in the East (83.0%). Acceptance rate of physicians was above 85% in all other regions (P = 0.007) (Figure 2).
| Variable | P-vaule | OR | 95%CI |
| Age | 0.672 | 1.155 | 0.593-2.250 |
| Region | 0.0071 | ||
| Region (North) | 0.838 | 1.101 | 0.437-2.773 |
| Region (North East) | 0.095 | 0.412 | 0.146-1.167 |
| Region (South Central) | 0.748 | 1.419 | 0.168-11.975 |
| Region (East) | 0.096 | 0.456 | 0.180-1.151 |
| Region (South West) | 0.0061 | 0.264 | 0.102-0.683 |
| Educational background | 0.945 | 1.016 | 0.657-1.570 |
| Professional title | 0.757 | 0.913 | 0.513-1.624 |
| Level of hospital | 0.0411 | 0.359 | 0.134-0.961 |
| Department | 0.910 | ||
| Department (gastroenterology) | 0.510 | 1.291 | 0.604-2.760 |
| Department (general surgery) | 0.778 | 1.177 | 0.379-3.657 |
| Department (endocrinology) | 0.598 | 1.463 | 0.356-6.020 |
| Working time on gastroenterology | 0.683 | 0.933 | 0.670-1.299 |
| Understanding of FMT | 0.0021 | 3.265 | 1.555-6.855 |
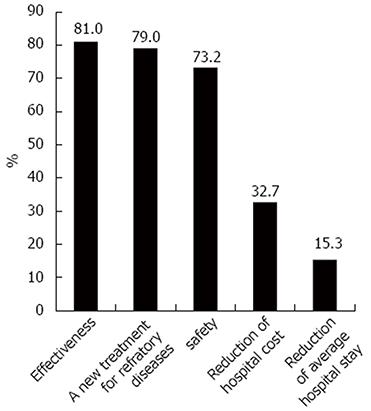
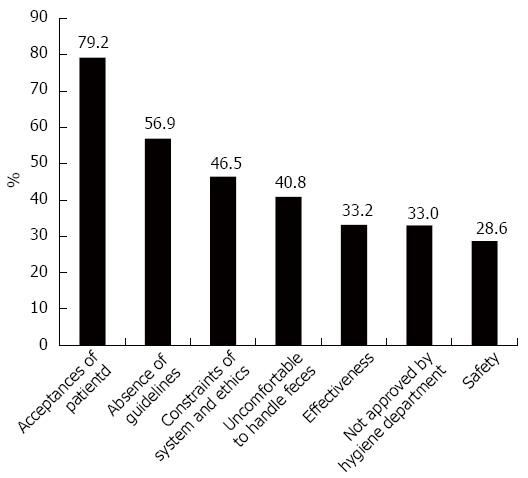
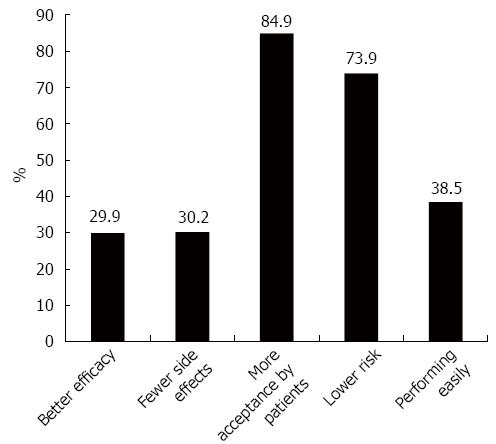
The three most frequent reasons for choosing FMT were as follows: efficacy (81.0%), a new treatment option for refractory diseases (79.0%) and safety (73.2%) (Figure 3). Primary barriers for the clinical application of FMT included patients’ acceptance (79.2%), absence of guidelines (56.9%) and systemic and ethical constraints (46.5%) (Figure 4).
Although we provided a brief description of FMT in the questionnaire, there were some questions about the details of FMT procedures. Therefore, it might not have been reasonable to ask physicians who had no awareness of FMT to analyze FMT technology. To disclose the physicians’ true perceptions of FMT procedures, we excluded physicians who had no knowledge of FMT in the following analysis.
Indications: The majority of physicians (86.7%) selected recurrent RCDI, followed by other diseases such as inflammatory bowel disease with CDI, refractory ulcerative colitis, ulcerative colitis, and Crohn’s disease (Figure 5).
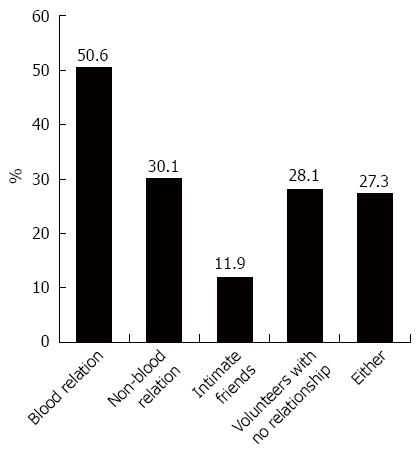
Donor selection: Most participants preferred someone who had a similar microbiota environment to the recipient, including blood relatives (50.6%), non-blood relatives (30.1%) and intimate friends (11.9%) (Figure 6). Only 28.1% of participants selected volunteers with no relationship, and 27.3% held the view that either of the above was an option contingent on the health of the donor; 29.7% of physicians were more inclined to prefer children donors, 35.4% selected adults, and 34.9% preferred both.
Selection of the administration route: Overall, 76.6% of the respondents preferred the lower GI tract as the route of administration, with the primary reasons being that patients would more likely accept this route (84.9%) and that it had lower risk (73.9%) (Figure 7). Only 13.9% of the physicians selected the upper GI tract, and others (7.1%) thought that both approaches were acceptable. With regard to the site for performing FMT, nearly half of the physicians (44.9%) preferred the Endoscopy Center, and only 21.3% preferred wards.
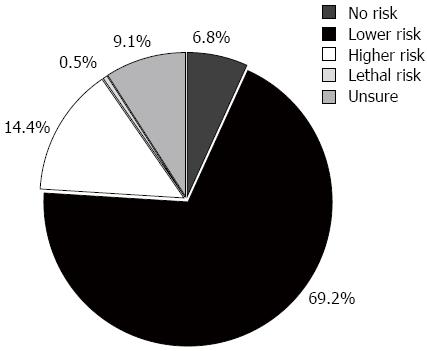
Risk of FMT: Most participants (69.2%) held the opinion that FMT has a low risk with transient abdominal symptoms such as diarrhea, and 14.4% of physicians thought that FMT had a high and even lethal risk (Figure 8). The vast majority of these respondents thought that disease history (93.5%), stool and blood examinations (92.7% and 90.9%) were all necessary considerations before qualifying as a donor.
The evolution of FMT has been rapid and certain. Physicians’ and patients’ awareness and perceptions of FMT are critical factors in determining FMT popularity. Our study was the first of its kind to investigate physicians’ perceptions of FMT in an Asian country. Although there were only 844 physicians in our survey, which is a small proportion of the entire Chinese physician population (more than 200 millions), this survey covered most areas of China (22 out of 34 provinces) and was representative to some extent. This investigation will, undoubtedly provide information of FMT development in China and hopefully in other Asian countries.
Our investigation found high levels of FMT perception, as the vast majority of physicians had heard of FMT prior to this survey and nearly half of understood it well. Among these physicians, gastroenterologists had a better awareness and a more favorable attitude toward the development of this novel method than non-gastroenterologists, which was expected. All the physicians had a very high level of acceptance of FMT and a high interest in FMT training. In our study, geographical region was an important factor affecting physician perceptions of FMT. The significant geographical differences may be related to the differences in the economy, the frequency of information communication, and the uneven distribution of medical resources. Northwest China is less developed than other areas, and it has fewer medical resources and a slower spread of new knowledge and technology.
Chinese physicians’ responses regarding the acceptance of FMT were somewhat astonishing. The high acceptance rate may be related to knowledge of Chinese traditional medicine in which FMT had originated. In this study, for the first time, the attitude of physicians toward FMT as an acceptable treatment was directly assessed. The results revealed that although human beings have a natural aversion to fecal material, the overwhelming majority of physicians were willing to accept FMT as a treatment method. A multivariate analysis revealed that increased awareness of FMT among physicians will enhance the likelihood of its acceptance. Conversely, the technique was less likely to be accepted by physicians working in higher level hospitals. It is possible that the higher level hospitals were more rigorous and cautious in the administration and implementation of new technologies.
Physicians accepted FMT as a treatment modality mainly on account of its effectiveness and safety, and they considered it an optional therapy for refractory diseases. This result was consistent with clinical studies, which reported that FMT was effective and safe in some diseases that were refractory to standard therapy or had shown frequent recurrence. Currently, there are few data about physicians’ attitudes about FMT. In one investigation, 65% (83/135) of physicians had neither offered nor referred a patient for FMT, with the most common reasons being lack of appropriate clinical indication (33%), patients’ acceptance or otherwise (24%) and institutional or logistical barriers (23%)[18]. In our investigation, the primary concern of Chinese physicians was the patients’ acceptance, followed by the absence of guidelines and system and ethical constraints, similar to physicians overseas. This result suggests that the standardization and extension of FMT are imperative.
In addition to recurrent CDI, physicians showed interest in the use of FMT for many other diseases. Several studies have confirmed the astounding efficacy of FMT in the treatment of RCDI. Studies on IBD, IBS, and chronic constipation treatment with FMT followed suit. Further, FMT has a potential therapeutic value in non-GI diseases associated with gut flora, such as obesity, metabolic syndrome and chronic fatigue syndrome, which is based on preliminary case reports or animal experiments[22]. The results of our survey on the selection of potential FMT indications were consistent with these studies, although additional rigorous studies are needed to determine the efficacy of FMT for these diseases.
Until now, there is no evidence that stool material from related donors was better than that from unrelated donors. One argument for the use of related donors is that they are presumed to have shared gut flora exposures; however, they are also more likely to test for infectious disease markers than unrelated volunteer donors[23]. A long-term multicenter follow-up study showed that CDI cure rates were not influenced by the donor-recipient relationship[24], which provided grounds for the commercialization of frozen fecal microbiota and the development of FMT. Nevertheless, donors with different genders, ages, diets or lifestyles may have varying effects on the efficacy of FMT, which should be confirmed by further studies.
FMT is often delivered via the lower GI route, including via colonoscopy and retention enema, and/or via the upper GI route, such as by gastroduodenoscopy, a nasoenteric tube and oral pills. To date, the optimal approach is still unclear, and approximately 75% of cases with RCDI are administered via the lower GI tract, and 25% via the upper GI tract[25]. A systematic review reported that FMT administered by colonoscopy had a higher cure rate (91%) than other routes for RCDI[26]. However, a recent RCT demonstrated a remarkable cure rate using the nasoenteric tube compared to colonoscopy[27]. Our results revealed that the vast majority of physicians (76.6%) preferred the lower GI tract with the primary argument that it may be easily accepted by patients psychologically. Another reason for the selection of the lower GI tract was that it may theoretically have a lower risk with easier colonization in situ, compared with the upper routes through which the small intestinal bacterial may overgrow and whether the stool suspension can reach the entire colon is unknown.
In terms of risk, although the majority of physicians in our survey considered FMT safe, an overwhelming majority of physicians suggested rigorous screening of donors to lessen the risk, including collection of a detailed disease history, and stool and blood examinations. Transient abdominal discomfort such as bloating, diarrhea and abdominal cramps have been observed after FMT and often disappeared within two days after treatment[24,25]. However, limited long-term safety data exist. Reports of concurrent infections after FMT treatment exist. Elizabeth et al[28] described a patient with refractory ulcerative colitis who acquired cytomegalovirus infection after FMT, which revealed a potential risk of FMT, although it was not confirmed whether the virus was directly from the donor. Cases involving norovirus[29], S.typhi, and Blastocystishominis infections have been reported. In our research center, despite rigorous screening, a patient developed an infection with two opportunistic pathogens, Proteusmirabilis and Candidaalbicans following FMT[30]. We still have limited knowledge of the impact of FMT on the intestinal flora and subsequent secondary infections after it. Therefore, the clinical utility of FMT must follow a strict and standardized protocol. It is recommended that patients undergo FMT in a hospital instead of at home. A standard protocol to screen donors is imperative.
In summary, this study is the largest survey of physicians’ perceptions of FMT and it is the first time that physicians’ perception of the indications, donors, and other technology associated with FMT have been evaluated in an Asian country. The keen interest, high acceptance and good understanding of FMT provide the grounds and conditions for the development of this novel treatment in China. The need to establish a standard procedure and protocol cannot be overstated.
The authors thank the physicians in the Department of Gastroenterology of PLA General Hospital for their help in reviewing the survey as well as all of the physicians who voluntarily participated in the survey.
While there has been growing interest in fecal microbiota transplantation (FMT), it is still in early phases worldwide. Physicians’ and patients’ perceptions and attitudes toward FMT play an important role in determining its acceptability. This article explores Chinese physicians’ perceptions towards FMT to provide information and an assessment of FMT development in China.
There are a few reports discussing patients’ attitudes towards the acceptance of FMT. Nevertheless, few studies exist regarding physicians’ perceptions of this technique; all of these studies were conducted in Western countries. The acceptance of FMT in Asian countries remains unknown.
This is the first study to acquire physicians’ perceptions of FMT in an Asian country. This study was representative with a large respondent number (844 eligible questionnaires were collected) and a vast coverage area of China (22 out of 34 provinces); thus, it can provide preliminary information for the FMT development in China. Additionally, the authors reviewed the literature and traced the history of human fecal medicine back 3000 years to the “Collection of 52 Prescriptions”, and they found that the first use of human fecal suspension by mouth occurred 2nd century.
The keen interest and high acceptance of FMT provide the grounds and conditions for the development of this novel treatment in China. Nevertheless, guidelines and strict protocols are necessary to implement this technique.
FMT refers to the instillation of fecal suspension from a healthy person into the gastrointestinal tract of a patient to cure a certain disease by restoring the construction of the intestinal flora.
The strongest point of this manuscript is being the first of its kind in China and other Asian countries. The idea is original and interesting, exploring the knowledge and attitudes regarding fecal microbiota transplantation (a very hot topic in gastroenterology nowadays) among Chinese physicians. The results give some ideas regarding how FMT might impact on clinical practice in the foreseeable future and provide important findings.
P- Reviewer: Ponte A, Serban DE S- Editor: Yu J L- Editor: Wang TQ E- Editor: Zhang DN
| 1. | Yang Y. [Gut microbiota research: highlights and commentary]. Zhonghua Nei Ke Zazhi. 2015;54:396-398. [PubMed] |
| 2. | Zhou DS, He QH. Paraphrase of “Wu Shi Er Fang”. Shanxi: Shanxi Science and Technology Press 2013; 175-176. |
| 3. | Zhang ZJ. Jin Gui Yao Lüe. Beijing: People’s Medical Publishing House 2005; . |
| 4. | Ge H. Zhou Hou Bei Ji Fang. Tianjin: Tianjin Science and Technology Press 2000; . |
| 5. | Sun SM. Qian Jin Yi Fang. Shanxi: Shanxi Science and Technology Press 2010; . |
| 6. | Li SZ. Ben Cao Gang Mu. Beijing: People’s Medical Publishing House 2004; . |
| 7. | Eiseman B, Silen W, Bascom GS, Kauvar AJ. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery. 1958;44:854-859. [PubMed] |
| 8. | Schwan A, Sjölin S, Trottestam U, Aronsson B. Relapsing clostridium difficile enterocolitis cured by rectal infusion of homologous faeces. Lancet. 1983;2:845. [PubMed] |
| 9. | Bakken JS, Borody T, Brandt LJ, Brill JV, Demarco DC, Franzos MA, Kelly C, Khoruts A, Louie T, Martinelli LP. Treating Clostridium difficile infection with fecal microbiota transplantation. Clin Gastroenterol Hepatol. 2011;9:1044-1049. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 650] [Cited by in RCA: 683] [Article Influence: 48.8] [Reference Citation Analysis (0)] |
| 10. | Surawicz CM, Brandt LJ, Binion DG, Ananthakrishnan AN, Curry SR, Gilligan PH, McFarland LV, Mellow M, Zuckerbraun BS. Guidelines for diagnosis, treatment, and prevention of Clostridium difficile infections. Am J Gastroenterol. 2013;108:478-498; quiz 499. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1179] [Cited by in RCA: 1186] [Article Influence: 98.8] [Reference Citation Analysis (0)] |
| 11. | Cho I, Blaser MJ. The human microbiome: at the interface of health and disease. Nat Rev Genet. 2012;13:260-270. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2636] [Cited by in RCA: 2253] [Article Influence: 173.3] [Reference Citation Analysis (0)] |
| 12. | Borody TJ, Warren EF, Leis S, Surace R, Ashman O. Treatment of ulcerative colitis using fecal bacteriotherapy. J Clin Gastroenterol. 2003;37:42-47. [PubMed] |
| 13. | Pinn DM, Aroniadis OC, Brandt LJ. Is fecal microbiota transplantation the answer for irritable bowel syndrome? A single-center experience. Am J Gastroenterol. 2014;109:1831-1832. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 70] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 14. | Vrieze A, Van Nood E, Holleman F, Salojärvi J, Kootte RS, Bartelsman JF, Dallinga-Thie GM, Ackermans MT, Serlie MJ, Oozeer R. Transfer of intestinal microbiota from lean donors increases insulin sensitivity in individuals with metabolic syndrome. Gastroenterology. 2012;143:913-916.e7. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1881] [Cited by in RCA: 2011] [Article Influence: 154.7] [Reference Citation Analysis (0)] |
| 15. | Wang ZK, Yang YS, Chen Y, Yuan J, Sun G, Peng LH. Intestinal microbiota pathogenesis and fecal microbiota transplantation for inflammatory bowel disease. World J Gastroenterol. 2014;20:14805-14820. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 76] [Cited by in RCA: 90] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 16. | Kahn SA, Vachon A, Rodriquez D, Goeppinger SR, Surma B, Marks J, Rubin DT. Patient perceptions of fecal microbiota transplantation for ulcerative colitis. Inflamm Bowel Dis. 2013;19:1506-1513. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 17. | Zipursky JS, Sidorsky TI, Freedman CA, Sidorsky MN, Kirkland KB. Patient attitudes toward the use of fecal microbiota transplantation in the treatment of recurrent Clostridium difficile infection. Clin Infect Dis. 2012;55:1652-1658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 126] [Article Influence: 9.7] [Reference Citation Analysis (0)] |
| 18. | Zipursky JS, Sidorsky TI, Freedman CA, Sidorsky MN, Kirkland KB. Physician attitudes toward the use of fecal microbiota transplantation for the treatment of recurrent Clostridium difficile infection. Can J Gastroenterol Hepatol. 2014;28:319-324. [PubMed] |
| 19. | Sofi AA, Georgescu C, Sodeman T, Nawras A. Physician outlook toward fecal microbiota transplantation in the treatment of Clostridium difficile infection. Am J Gastroenterol. 2013;108:1661-1662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 20. | Agito MD, Atreja A, Rizk MK. Fecal microbiota transplantation for recurrent C difficile infection: ready for prime time? Cleve Clin J Med. 2013;80:101-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 21. | Harris PA, Taylor R, Thielke R, Payne J, Gonzalez N, Conde JG. Research electronic data capture (REDCap)--a metadata-driven methodology and workflow process for providing translational research informatics support. J Biomed Inform. 2009;42:377-381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38562] [Cited by in RCA: 36728] [Article Influence: 2295.5] [Reference Citation Analysis (0)] |
| 22. | Smits LP, Bouter KE, de Vos WM, Borody TJ, Nieuwdorp M. Therapeutic potential of fecal microbiota transplantation. Gastroenterology. 2013;145:946-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 421] [Cited by in RCA: 472] [Article Influence: 39.3] [Reference Citation Analysis (1)] |
| 23. | Starkey JM, MacPherson JL, Bolgiano DC, Simon ER, Zuck TF, Sayers MH. Markers for transfusion-transmitted disease in different groups of blood donors. JAMA. 1989;262:3452-3454. [PubMed] |
| 24. | Brandt LJ, Aroniadis OC, Mellow M, Kanatzar A, Kelly C, Park T, Stollman N, Rohlke F, Surawicz C. Long-term follow-up of colonoscopic fecal microbiota transplant for recurrent Clostridium difficile infection. Am J Gastroenterol. 2012;107:1079-1087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 499] [Article Influence: 38.4] [Reference Citation Analysis (0)] |
| 25. | Brandt LJ, Aroniadis OC. An overview of fecal microbiota transplantation: techniques, indications, and outcomes. Gastrointest Endosc. 2013;78:240-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 187] [Article Influence: 15.6] [Reference Citation Analysis (0)] |
| 26. | Gough E, Shaikh H, Manges AR. Systematic review of intestinal microbiota transplantation (fecal bacteriotherapy) for recurrent Clostridium difficile infection. Clin Infect Dis. 2011;53:994-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 808] [Cited by in RCA: 720] [Article Influence: 51.4] [Reference Citation Analysis (0)] |
| 27. | van Nood E, Vrieze A, Nieuwdorp M, Fuentes S, Zoetendal EG, de Vos WM, Visser CE, Kuijper EJ, Bartelsman JF, Tijssen JG. Duodenal infusion of donor feces for recurrent Clostridium difficile. N Engl J Med. 2013;368:407-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2582] [Cited by in RCA: 2677] [Article Influence: 223.1] [Reference Citation Analysis (0)] |
| 28. | Hohmann EL, Ananthakrishnan AN, Deshpande V. Case Records of the Massachusetts General Hospital. Case 25-2014. A 37-year-old man with ulcerative colitis and bloody diarrhea. N Engl J Med. 2014;371:668-675. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 60] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 29. | Schwartz M, Gluck M, Koon S. Norovirus gastroenteritis after fecal microbiota transplantation for treatment of Clostridium difficile infection despite asymptomatic donors and lack of sick contacts. Am J Gastroenterol. 2013;108:1367. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 115] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 30. | Ren R, Sun G, Yang Y, Peng L, Zhang X, Wang S, Dou Y, Zhang X, Wang Z, Bo X. [A pilot study of treating ulcerative colitis with fecal microbiota transplantation]. Zhonghua Nei Ke Zazhi. 2015;54:411-415. [PubMed] |