Published online Sep 28, 2014. doi: 10.3748/wjg.v20.i36.13139
Revised: March 29, 2014
Accepted: April 30, 2014
Published online: September 28, 2014
Processing time: 270 Days and 15.8 Hours
AIM: To identify pathologic features associated with this “ulcerative colitis (UC)-like” subgroup of Crohn’s disease (CD).
METHODS: Seventeen subjects diagnosed as having UC who underwent proctocolectomy (RPC) from 2003-2007 and subsequently developed CD of the ileal pouch were identified. UC was diagnosed based on pre-operative clinical, endoscopic, and pathologic studies. Eighteen patients who underwent RPC for UC within the same time period without subsequently developing CD were randomly selected and used as controls. Pathology reports and histological slides were reviewed for a wide range of gross and microscopic pathological features, as well as extent of disease. The demographics, gross description and histopathology of the resection specimens were reviewed and compared between the two groups.
RESULTS: Patients with “UC-like” CD were on average 13 years younger than those with “true” UC (P < 0.01). More severe disease in the proximal involved region and active ileitis with/without architectural distortion were observed in 6 of 17 (35%) and 7 of 17 (41%) “UC-like” CD cases, respectively, but in none of the “true” UC cases (P < 0.05). Active appendicitis occurred in 8 of 16 (50%) “UC-like” CD cases but in only two (11%) “true” UC cases (P < 0.05). Conspicuous lamina propria neutrophils were more specific for “UC-like” CD (76% vs 22%, P < 0.05). In addition, prominent lymphoid aggregates tended to be more common in “UC-like” CD (P = 0.07). The “true” UC group contained a greater number of cases with severe activity (78% vs 47%). Therefore, the features more commonly seen in “UC-like” CD were not due to a more severe disease process. Crohn’s granulomas and transmural inflammation in non-ulcerated areas were absent in both groups.
CONCLUSION: More severe disease in the proximal involved region, terminal ileum involvement, active appendicitis, and prominent lamina propria neutrophils may be morphological factors associated with “UC-like” CD.
Core tip: Despite well-established clinical and pathological criteria, a fraction of Crohn’s disease (CD) cases are diagnosed as ulcerative colitis (UC). Given the significant difference in standard surgical management for UC and Crohn’s colitis, it is critical to identify as many factors as possible which may help distinguish the two entities. In this study we have identified several pathologic features which may assist in identifying a subgroup of inflammatory bowel disease cases known as “UC-like” CD. More severe disease in the proximal involved region, terminal ileum involvement, active appendicitis, and prominent lamina propria neutrophils may be morphological factors associated with “UC-like” CD.
- Citation: James SD, Wise PE, Zuluaga-Toro T, Schwartz DA, Washington MK, Shi C. Identification of pathologic features associated with “ulcerative colitis-like” Crohn’s disease. World J Gastroenterol 2014; 20(36): 13139-13145
- URL: https://www.wjgnet.com/1007-9327/full/v20/i36/13139.htm
- DOI: https://dx.doi.org/10.3748/wjg.v20.i36.13139
Crohn’s disease (CD) and ulcerative colitis (UC) are the two major types of idiopathic inflammatory bowel disease (IBD). The exact pathogenesis of these disorders is not well understood. The most credited models suggest that the intestinal flora triggers and drives an aberrant intestinal immune response with subsequent inflammation in a genetically susceptible host[1]. CD is characterized by skip lesions, transmural inflammation, granulomas, fistulae and frequently, terminal ileum involvement; whereas, UC is featured by continuous colorectal disease, often more prominent distally, with predominant mucosal involvement.
Approximately 20% of CD cases only involve the colon (Crohn’s colitis)[2,3]. Thereby, differentiation between UC and Crohn’s colitis can be very difficult, even in total colectomy specimens. Some cases of Crohn’s colitis may show only mucosal involvement without traditional features of Crohn’s disease, such as transmural lymphoid aggregates, deep or fissuring ulcers, sinus tracts, or fistulas. This subgroup of patients has been referred to as “superficial Crohn’s colitis” or “ulcerative colitis-like Crohn’s colitis’ in previous studies”[4-8]. Up to 10% of IBD cases are characterized as indeterminate colitis. In these cases, clinical and pathological data are unsuccessful in confidently distinguishing the two entities.
Approximately 25% of patients with UC undergo a colectomy due to failure of medical therapy, unacceptable side effects of chronic therapy, and occurrence of acute complications (i.e., fulminant colitis, severe bleeding, toxic megacolon, perforation) or development of malignancy[9]. Standard surgical management of UC for those patients who do not require a permanent ileostomy is a total proctocolectomy. This is often as a 2- or 3-stage procedure, followed by ileal pouch-anal anastomosis (IPAA), also known as a restorative proctocolectomy (RPC)[10,11]. This procedure involves the creation of a pouch of two or more loops of small intestine that are sutured or stapled together to create a reservoir for stool to “replace” the removed rectum. This reservoir is then attached to the anus to reestablish intestinal continuity. IPAA is generally contraindicated in CD. The creation of a pouch frequently causes reactivation of Crohn’s with high risk of pouchitis and fistula development[12-15]. Significant morbidity has been reported with pouch failure rates that range between 30%-90%[16-19]. Therefore, it is important to distinguish Crohn’s colitis from UC.
In the present study, we set out to examine the gross and histologic features that are more frequently associated with CD, using colectomy specimens from patients who were diagnosed with UC on initial clinical and pathological evaluation. Until now, there are no studies which examine those pathologic features that may be useful in identifying this “UC-like” Crohn’s disease subgroup. The aim of this study was to identify pathologic features in resection specimens associated with the “UC-like” subgroup of CD.
There were approximately two-hundred patients who underwent RPC for UC between January 1, 2003-December 31, 2007 at Vanderbilt University Medical Center. UC was diagnosed based on pre-operative clinical, endoscopic, and pathologic studies. The study group consisted of seventeen subjects diagnosed with UC who underwent RPC during 2003-2007 and subsequently developed CD of the ileal pouch based on pouch and pre-pouch ileum biopsies and/or development of peri-anal fistulizing disease. This group was defined as the “UC-like” CD group. Eighteen patients who underwent restorative proctocolectomy for UC within the same time period, without subsequently developing CD, were randomly selected and used as controls. This group was designated as the “true” UC group. Patient’s with indeterminate colitis were excluded. This study was approved by the Vanderbilt Institutional Review Board.
Patient demographics and clinical data were collected from the electronic medical record. Pathology reports and histological slides were reviewed for a wide range of gross and microscopic pathological features, as well as extent of disease. The gross features were evaluated by review of patients’ surgical pathology reports for the presence or absence of the following: skip lesions, cobblestone mucosa, pseudopolyps, terminal ileum involvement, ulceration, and the distribution and severity of the disease. A gross description of the ileum was not provided for one of the “UC-like” CD and three of the “true” UC resection specimens.
Microscopic evaluation was performed on routinely processed formalin-fixed resection specimens by two of the pathologists (James SD and Shi C). The presence or absence of ileitis, active appendicitis, appendiceal fibrous obliteration, granulomas, Paneth cell metaplasia, pyloric gland metaplasia, prominent lamina propria neutrophils, and prominent lymphoid aggregates were assessed. Prominent lymphoid aggregates were defined as equal to or more than 10 lymphoid aggregates per section. The disease activity was classified as (1) mild (cryptitis and occasional crypt abscess, without ulcers); (2) moderate (cryptitis, prominent crypt abscess, and occasional ulceration); and (3) severe (cryptitis, prominent crypt abscess and prominent ulceration). Prominence of architectural distortion, basal plasmacytosis, inflammatory infiltrates in the muscularis mucosae, submucosal fibrosis, and thickening of the muscularis mucosa were also assessed.
Student t test was used to compare the patients’ age. Fisher’s exact test was used to compare other demographics and pathologic findings between the two groups. A P value of < 0.05 was considered statistically significant.
The “UC-like” CD group included 9 males and 8 females. The “true” UC group was composed of 10 males and 8 females. The ”UC-like” CD group was on average 13 years younger than the “true” UC group at the time of total colectomy (31.1 ± 10.3 vs 44.1 ± 16.2, P < 0.01). The mean time from total colectomy to diagnosis of CD was 27.8 ± 5.8 mo (range: 4-83 mo). The median follow up time was 74 and 57 mo for “UC-like” CD group and true “UC” group, respectively.
Gross terminal ileum involvement was identified in 2 of 16 (13%) “UC-like” CD cases, but in none of the “true” UC cases (Table 1). However, statistically there was no difference between the two groups. Discontinuous involvement of the colon was observed in 2/17 (12%) and 1/18 (6%) of the “UC-like” CD cases and the “true” UC cases, respectively (P > 0.05). Cobblestoning, pseudopolyps, and gross ulceration were not different among the two groups.
| Gross features | “UC-like” CD | “True” UC | P value |
| Terminal ileum involvement | 2/16 (13) | 0/15 (0) | 0.48 |
| Discontinuous involvement | 2/17 (12) | 1/18 (6) | 0.60 |
| Pan-colitis | 7/17 (41) | 8/18 (44) | 1.00 |
| Cobblestone mucosa | 6/17 (35) | 9/18 (50) | 0.50 |
| Pseudopolyps | 8/17 (47) | 11/18 (61) | 0.32 |
| Ulceration | 7/17 (41) | 8/18 (44) | 1.00 |
| Degree of severity | |||
| Distal > proximal | 7/17 (41) | 11/18 (61) | 0.32 |
| Evenly distributed | 4/17 (24) | 7/18 (39) | 0.47 |
| Proximal > distal | 6/17 (35) | 0/18 (0) | < 0.01b |
The severity of the disease was also assessed grossly and compared among different regions of the involved colon. In the “UC”-like CD cases, there were 3 distribution patterns of the severity: distal more severe than proximal in 7 (41%), evenly distributed in 4 (24%), and proximal more severe than distal in 6 (35%) of the 17 cases; whereas in the 18 “true” UC cases, 11 (61%) displayed more severe disease in the distal region, 7 (39%) showed no difference in disease severity among the regions, and none (0%) demonstrated more severe disease in the proximal area. More severe disease in the proximal region than in the distal region was more commonly seen in the “UC-like” CD (P < 0.01).
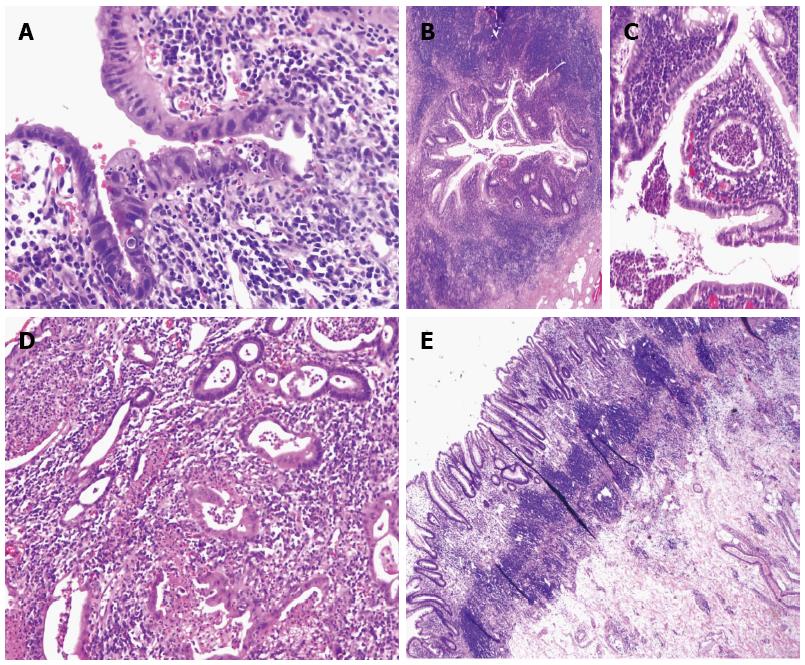
Active ileitis (Figure 1A) was observed in 7 of 17 (41%) “UC-like” CD cases, but in none of the “true” UC cases (P < 0.05). Among 7 cases with active inflammation in the terminal ileum, 4 displayed crypt distortion and villous blunting with cryptitis, crypt abscess, and scattered to prominent lamina propria neutrophils; however, pyloric gland metaplasia was not identified.
Active appendicitis (Figure 1B and C) occurred in 8 of 16 (50%) “UC-like” CD cases but in only two of 18 (11%) “true” UC cases (P < 0.05) (Figure 1). Among the 8 “UC-like” CD cases with active appendicitis, 3 had only distal disease, and 7 displayed mildly to moderately active colitis, whereas all of the “true” UC disease cases with active appendicitis had severely active pan-colitis. Unlike acute appendicitis seen in general populations, the active inflammation was more prominent in the proximal appendix in both UC and CD cases with active appendicitis, with some of them sparing the tip of the appendix. Fibrous obliteration of the appendix was present in 1 of 16 (6%) “UC-like” CD cases, compared to 5 of 18 (28%) “true” UC cases (P > 0.05).
Prominent lamina propria neutrophils were more specific for “UC-like” CD (76% vs 22%, P < 0.05) (Figure 1D). In addition, prominent lymphoid aggregates (Figure 1E) tended to be more common in “UC-like” CD with 8 of 17 (47%) showing a marked increase, compared to 3 of 18 (17%, P = 0.07) in the “true” UC group. Severely active colitis was observed in 8 of 17 (47%) “UC-like” CD cases and 14 of 18 (78%) “true” UC cases (P < 0.05). Therefore, the association of active ileitis, active appendicitis, prominent lamina propria neutrophils and prominent lymphoid aggregates with “UC-like” CD is less likely due to the activity of the disease present in these cases.
None of the cases in both groups showed transmural inflammation in the areas without ulceration. Granulomas directly associated with crypt disruption were identified in one of the “UC-like” CD cases; however, no CD-type granulomas were present in any of these cases. Deep ulcers were observed in the both groups, especially in the cases with severe activity; however, no fissures (deep and narrow ulcers) were identified. An aphthous ulcer was present in one of the “UC-like” CD cases.
Severe architectural distortion was seen in 8 of 17 (47%) “UC-like” CD cases and 12 of 18 (67%) “true” UC cases (P = 0.49). Prominent basal plasmacytosis, inflammatory infiltrates in the muscularis mucosae, submucosal fibrosis, thickening of the muscularis mucosae, and Paneth cell metaplasia were all seen in similar percentages among the two groups (Table 2).
| Microscopic features | “UC-like” CD | “True”UC | P value |
| Active ileitis | 7/17 (41) | 0/18 (0) | < 0.05a |
| Appendiceal activity | 8/16 (50) | 2/18 (11) | < 0.05a |
| Appendiceal fibrous obliteration | 1/16 (6) | 5/18 (28) | 0.18 |
| Severe activity | 8/17 (47) | 14/18 (78) | < 0.05a |
| Prominent lamina propria neutrophils | 13/17 (76) | 4/18 (22) | < 0.05a |
| Prominent lymphoid aggregates | 8/17 (47) | 3/18 (17) | 0.07 |
| Prominent chronicity/architectural distortion | 8/17 (47) | 12/18 (67) | 0.49 |
| Prominent basal plasmacytosis | 4/17 (24) | 6/18 (33) | 0.71 |
| Prominent muscularis mucosa inflammation | 8/17 (47) | 9/18 (50) | 1.00 |
| Prominent submucosal fibrosis | 3/17 (18) | 3/18 (17) | 1.00 |
| Prominent thickening of muscularis mucosa | 4/17 (24) | 5/18 (28) | 1.00 |
| Paneth cell metaplasia | 8/17 (47) | 6/18 (33) | 1.00 |
Distinguishing Crohn’s colitis from UC histologically can be extremely difficult, if not impossible in some cases. In early stages of IBD it is common to have overlapping clinical and pathologic features which make a precise diagnosis challenging and oftentimes subjective[20,21]. Despite well-established clinical and pathological criteria, a fraction of CD cases are diagnosed as UC. This subgroup has been identified in previous studies; however, none solely focus on distinguishing pathologic features. Given the significant difference in standard surgical management for UC and Crohn’s colitis, it is critical to identify as many factors as possible which may help distinguish the two entities. A study evaluating intestinal complications of IBD noted that all studies of IPAA for presumed UC contained 2%-7% of patients in whom the correct diagnosis proved to actually be CD[22]. In another study, which examined the long-term results of patients with CD treated with IPAA, 21 of the 37 patients had a proctocolectomy specimen that was initially diagnosed as UC. After review of the proctocolectomy specimens, four of the patients had their diagnosis changed to CD[13]. IPAA is widely accepted as the procedure of choice for UC patients. Conversely, this procedure is generally contraindicated in patients with CD because of negative outcomes associated with post-procedural complications. Our goal was to identify pathologic characteristics which may help distinguish the “UC-like” CD subgroup from “true” UC.
In this study we found that the “UC-like” CD group was on average 13 years younger than the “true” UC group at the time of total colectomy. A large study evaluating preoperative factors predictive of subsequent diagnosis of CD after IPAA also reported younger age in the “UC-like” CD patients[23]. In addition, perianal fistula, anal fissure, and colorectal stricture were found to be significant predictive factors. This was not observed in our study, which may due a couple of factors. Our colorectal surgeons generally are conservative in practice and will not proceed with an IPAA if these features are identified preoperatively and there remains clinical or pathologic suspicion for CD. With that said, patients who may fall into the category of indeterminate colitis are managed medically and re-evaluated until further characterization can be made. In our study, patients who underwent an IPAA had a working diagnosis of UC. Granulomas were seen in one “UC-like” CD case, which were not felt to be associated with CD but with crypt disruption. Also, a “UC-like” CD case contained an aphthous ulcer present in the colon. Some cases of UC may possess this finding. These findings in isolation would not have changed the management of these patients. However, seeing they may be associated with “UC-like” CD, stronger consideration should be taken if identified prior to surgery.
Overall, gross pathologic features were mostly non-contributory in identifying “UC-like” CD. Two cases of “UC-like” CD did show gross terminal ileum involvement, compared to none of the “true” UC cases. Though there was no statistically significant difference, this may potentially serve as an informative feature to note during pre- or post-operative examination. A cobblestone appearance of the mucosa is generally regarded as a feature of CD, but was noted more often in the “true” UC group. Skip lesions are also most associated with CD; however, they were seen rarely and in close frequency as that observed in the “true” UC group in our study. These findings highlight the strong degree of overlap in both forms of IBD, making the two entities difficult to distinguish on gross examination alone. Interestingly, when we looked into the distribution of disease severity, we found that approximately one third of the “UC-like” CD cases had more severe disease in the proximal region than in the distally involved region, which was not present in the “true” UC group. Therefore, assessment of the distribution of the disease severity, even in patients treated medically for IBD prior to surgery, may be useful in identifying “UC-like” CD.
Microscopic examination resulted in the identification of several significant features associated with “UC-like” CD, including active ileitis (41% of “UC-like” CD cases, but in none of the “true” UC cases). When evaluating the terminal ileum, distinguishing backwash ileitis, characterized by active ileitis with/without architectural distortion in UC patients with severe pan-colitis, from Crohn’s disease can be challenging. Both clinical and pathological features must be taken into consideration. Backwash ileitis has been reported in up 30% percent of UC cases[23,24]. The absence of ileitis in the “true” UC group may be due to small sample size. It may also represent the declining prevalence in UC due to medical management in UC cases with pancolitis and terminal ileum involvement prior to surgery. Two of the seventeen “UC-like” CD cases with active ileitis had active chronic colitis that was limited to the distal colon, which was strongly suggestive of Crohn’s colitis.
Only one of the two cases with gross terminal ileum involvement contained activity microscopically. One possibility is that the changes observed grossly were that of chronic injury instead of an active inflammatory process. Also of consideration is the possibility of sampling error. Lastly, the gross and microscopic discrepancy could further illustrate the low diagnostic yield of gross features in these cases.
Active appendicitis occurred in 50% of “UC-like” CD cases, but in only 11% of “true” UC cases. The prevalence of appendiceal involvement in IBD is unknown. The data surrounding its occurrence in CD are few and inconsistent with ranges between 0%-54%[25-27]. Similarly, appendiceal involvement in ulcerative colitis has been reported with a wide range of variation (12%-87%)[28-30]. Despite the variable and inconsistent data, active appendicitis was found to be a useful feature in identifying “UC-like” CD in the present study.
Prominent lamina propria neutrophils were more specific for “UC-like” CD. The hallmark of active IBD is cryptitis and/or crypt abscess formation with pronounced neutrophilic infiltration into the lamina prominent. Although its mere presence does not help discern between CD and UC, our findings suggest that the magnitude and degree of activity may be informative in distinguishing these two entities.
Lastly, increased and prominent lymphoid aggregates tended to be more common in “UC-like” CD (47% vs 18%). Transmural distribution of lymphoid aggregates is considered a histological characteristic of Crohn’s disease[31], which may explain the observed increase in our “UC-like” CD group. However, lymphoid aggregates were found no deeper than the submucosa. It has also been reported that a marked increase in aggregate diameter is particularly seen in Crohn’s disease[32]. Taken together, examination of the number, distribution, and size of lymphoid aggregates in IBD are informative diagnostic measures[33].
In conclusion, we have identified several pathologic features that may assist in identifying a subgroup of IBD cases known as “UC-like” CD. In isolation, the diagnostic predictive value of these features is limited due to the large degree of overlap in both the gross and histologic findings. However, gross assessment of disease distribution and directed microscopic examination with documentation of terminal ileum involvement, active appendicitis, lamina propria neutrophils, and increased and prominent lymphoid aggregates may be suggestive of Crohn’s colitis. Use of these results may permit creation of a scoring system to validate and better prospectively predict the likelihood of developing CD after RPC for UC and thus impact clinical decision-making. Further studies are warranted.
Despite well-established clinical and pathological criteria, a fraction of Crohn’s disease (CD) cases are diagnosed as ulcerative colitis (UC). Given the significant difference in standard surgical management for UC and Crohn’s colitis, it is critical to identify as many factors as possible that may help distinguish the two entities.
A subgroup of inflammatory bowel disease cases have been identified in the literature referred to as “superficial Crohn’s colitis” or “ulcerative colitis-like Crohn’s colitis” in which traditional features of CD are not seen. Many of these studies have focused on clinical predictive factors and surgical outcomes. In this study, the authors identify pathologic features that may contribute to the identification of this “UC-like” CD.
Previous studies have reported that perianal fistula, anal fissure, and colorectal stricture are useful predictive factors in identifying cases of “UC-like” CD. Conversely, in this study gross pathologic features were non-contributory with a great degree of overlap between the study and control group. However, microscopic features such as terminal ileum involvement, active appendicitis, lamina propria neutrophils, and increased and prominent lymphoid aggregates were found to be suggestive of CD.
By identifying several prospective pathologic predictive factors to better determine the likelihood of developing CD after underwent proctocolectomy (RPC) for suspected UC cases, improved clinical and surgical decision-making can be made, ultimately decreasing the prevalence of adverse outcomes seen in these circumstances.
“UC-like” CD refers to patients who were diagnosed with UC based on pre-operative clinical, endoscopic, and pathologic studies, underwent RPC and subsequently developed CD of the ileal pouch based on pouch and pre-pouch ileum biopsies and/or development of peri-anal fistulizing disease.
This is a single centre, retrospective case control study with patients who had undergone RPC for “true UC” compared with patients who were subsequently diagnosed with CD after surgery. Histopathological features were compared between 17 “UC-like” CD patients and 18 “true UC” patients to identify factors predictive of CD.
P- Reviewer: Coffey JC S- Editor: Qi Y L- Editor: A E- Editor: Ma S
| 1. | Abraham C, Cho JH. Inflammatory bowel disease. N Engl J Med. 2009;361:2066-2078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1967] [Cited by in RCA: 2197] [Article Influence: 137.3] [Reference Citation Analysis (6)] |
| 2. | Lapidus A, Bernell O, Hellers G, Löfberg R. Clinical course of colorectal Crohn’s disease: a 35-year follow-up study of 507 patients. Gastroenterology. 1998;114:1151-1160. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 151] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 3. | Lapidus A, Bernell O, Hellers G, Persson PG, Löfberg R. Incidence of Crohn’s disease in Stockholm County 1955-1989. Gut. 1997;41:480-486. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 108] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Kleer CG, Appelman HD. Surgical pathology of Crohn’s disease. Surg Clin North Am. 2001;81:13-30, vii. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 5. | Odze R. Diagnostic problems and advances in inflammatory bowel disease. Mod Pathol. 2003;16:347-358. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 109] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Harpaz N, Friedman S, George J. Superficial Crohn’s colitis: pathological and clinical features including long-term follow up. Mod Pathol. 2001;14:86A. |
| 7. | McQuillan AC, Appelman HD. Superficial Crohn’s disease: a study of 10 patients. Surg Pathol. 1989;2:231-239. |
| 8. | Soucy G, Wang HH, Farraye FA, Schmidt JF, Farris AB, Lauwers GY, Cerda SR, Dendrinos KG, Odze RD. Clinical and pathological analysis of colonic Crohn’s disease, including a subgroup with ulcerative colitis-like features. Mod Pathol. 2012;25:295-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Cima RR. Timing and indications for colectomy in chronic ulcerative colitis: Surgical consideration. Dig Dis. 2010;28:501-507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 10. | Parks AG, Nicholls RJ. Proctocolectomy without ileostomy for ulcerative colitis. Br Med J. 1978;2:85-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 968] [Cited by in RCA: 913] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 11. | Taylor BA, Dozois RR. The J ileal pouch-anal anastomosis. World J Surg. 1987;11:727-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 30] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 12. | Foley EF, Schoetz DJ, Roberts PL, Marcello PW, Murray JJ, Coller JA, Veidenheimer MC. Rediversion after ileal pouch-anal anastomosis. Causes of failures and predictors of subsequent pouch salvage. Dis Colon Rectum. 1995;38:793-798. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 81] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 13. | Sagar PM, Dozois RR, Wolff BG. Long-term results of ileal pouch-anal anastomosis in patients with Crohn‘s disease. Dis Colon Rectum. 1996;39:893-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 142] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 14. | Murrell ZA, Melmed GY, Ippoliti A, Vasiliauskas EA, Dubinsky M, Targan SR, Fleshner PR. A prospective evaluation of the long-term outcome of ileal pouch-anal anastomosis in patients with inflammatory bowel disease-unclassified and indeterminate colitis. Dis Colon Rectum. 2009;52:872-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 35] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 15. | Le Q, Melmed G, Dubinsky M, McGovern D, Vasiliauskas EA, Murrell Z, Ippoliti A, Shih D, Kaur M, Targan S. Surgical outcome of ileal pouch-anal anastomosis when used intentionally for well-defined Crohn’s disease. Inflamm Bowel Dis. 2013;19:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 47] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 16. | Braveman JM, Schoetz DJ, Marcello PW, Roberts PL, Coller JA, Murray JJ, Rusin LC. The fate of the ileal pouch in patients developing Crohn’s disease. Dis Colon Rectum. 2004;47:1613-1619. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 101] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 17. | Brown CJ, Maclean AR, Cohen Z, Macrae HM, O’Connor BI, McLeod RS. Crohn’s disease and indeterminate colitis and the ileal pouch-anal anastomosis: outcomes and patterns of failure. Dis Colon Rectum. 2005;48:1542-1549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 108] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 18. | Hartley JE, Fazio VW, Remzi FH, Lavery IC, Church JM, Strong SA, Hull TL, Senagore AJ, Delaney CP. Analysis of the outcome of ileal pouch-anal anastomosis in patients with Crohn’s disease. Dis Colon Rectum. 2004;47:1808-1815. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 86] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 19. | Regimbeau JM, Panis Y, Pocard M, Bouhnik Y, Lavergne-Slove A, Rufat P, Matuchansky C, Valleur P. Long-term results of ileal pouch-anal anastomosis for colorectal Crohn’s disease. Dis Colon Rectum. 2001;44:769-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 109] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 20. | Price AB. Overlap in the spectrum of non-specific inflammatory bowel disease--’colitis indeterminate’. J Clin Pathol. 1978;31:567-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 221] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 21. | Wells AD, McMillan I, Price AB, Ritchie JK, Nicholls RJ. Natural history of indeterminate colitis. Br J Surg. 1991;78:179-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 73] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 22. | Petras RE, Oakley JR. Intestinal complications of inflammatory bowel disease: pathologic aspects. Semin Colon Rectal Surg. 1992;3:160-72. |
| 23. | Melton GB, Kiran RP, Fazio VW, He J, Shen B, Goldblum JR, Achkar JP, Lavery IC, Remzi FH. Do preoperative factors predict subsequent diagnosis of Crohn’s disease after ileal pouch-anal anastomosis for ulcerative or indeterminate colitis? Colorectal Dis. 2010;12:1026-1032. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 32] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 24. | Haskell H, Andrews CW, Reddy SI, Dendrinos K, Farraye FA, Stucchi AF, Becker JM, Odze RD. Pathologic features and clinical significance of “backwash” ileitis in ulcerative colitis. Am J Surg Pathol. 2005;29:1472-1481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 124] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 25. | Goldstein N, Dulai M. Contemporary morphologic definition of backwash ileitis in ulcerative colitis and features that distinguish it from Crohn disease. Am J Clin Pathol. 2006;126:365-376. [PubMed] |
| 26. | Ripollés T, Martínez MJ, Morote V, Errando J. Appendiceal involvement in Crohn’s disease: gray-scale sonography and color Doppler flow features. AJR Am J Roentgenol. 2006;186:1071-1078. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 27. | Kahn E, Markowitz J, Daum F. The appendix in inflammatory bowel disease in children. Mod Pathol. 1992;5:380-383. [PubMed] |
| 28. | Stangl PC, Herbst F, Birner P, Oberhuber G. Crohn’s disease of the appendix. Virchows Arch. 2002;440:397-403. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 29. | Groisman GM, George J, Harpaz N. Ulcerative appendicitis in universal and nonuniversal ulcerative colitis. Mod Pathol. 1994;7:322-325. [PubMed] |
| 30. | Matsumoto T, Nakamura S, Shimizu M, Iida M. Significance of appendiceal involvement in patients with ulcerative colitis. Gastrointest Endosc. 2002;55:180-185. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 67] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 31. | Scott IS, Sheaff M, Coumbe A, Feakins RM, Rampton DS. Appendiceal inflammation in ulcerative colitis. Histopathology. 1998;33:168-173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 52] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 32. | Tanaka M, Riddell RH. The pathological diagnosis and differential diagnosis of Crohn’s disease. Hepatogastroenterology. 1990;37:18-31. [PubMed] |
| 33. | Nascimbeni R, Di Fabio F, Di Betta E, Mariani P, Fisogni S, Villanacci V. Morphology of colorectal lymphoid aggregates in cancer, diverticular and inflammatory bowel diseases. Mod Pathol. 2005;18:681-685. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 1.3] [Reference Citation Analysis (0)] |