Published online Mar 1, 2004. doi: 10.3748/wjg.v10.i5.755
Revised: August 11, 2003
Accepted: August 18, 2003
Published online: March 1, 2004
AIM: To investigate the inhibitory effect of dietary and medicinal formula Ganfujian granule on diethylnitrosamine (DEN)-induced hepatocarcinoma in rats.
METHODS: Male SD rats had free access to water containing 0.1 g/L DEN for 16 weeks, during which the rats fed with standard diet or administration of Ganfujian granule (30.4 g/Kg in diet). At weeks 4, 8, 12 and 16 of hepatocarcinogenesis 5 rats of each group were sacrificed, and at week 20 another 30 rats were sacrificed from each group. The end point for survival observation was at week 28.Immunochemistry methods were used to examine the effect of Ganfujian granule on the process of hepatocarcinogenesis including proliferation of hepatocytes and cell cycle modulation.
RESULTS: Ganfujian granule could reduce and delay the incidence of hepatocarcinoma in rats and prolong the survival of animals. In addition, Ganfujian granule had a marked inhibitory effect on high expression of cyclin dependent kinase (CDK4) during the whole process of hepatocarcinogenesis and cyclin D1 at week 16 and the number of proliferating cell nuclear antigen (PCNA) positive cells in different stages of hepatocarcinogenesis.
CONCLUSION: Ganfujian granule can reduce and delay the incidence of hepatocarcinoma in rats by exerting direct or indirect effects on cell cycle and inhibiting uncontrolled proliferation of hepatocytes.
- Citation: Qian Y, Ling CQ. Preventive effect of Ganfujian granule on experimental hepatocarcinoma in rats. World J Gastroenterol 2004; 10(5): 755-757
- URL: https://www.wjgnet.com/1007-9327/full/v10/i5/755.htm
- DOI: https://dx.doi.org/10.3748/wjg.v10.i5.755
Liver cancer is one of the common malignancies in the world, especially in Asia and Africa. Despite many advances in the treatment of this disease in recent decades, its long-term therapeutic outcome remains poor and prognosis is devastating. Prevention seems to be the best strategy in lowering the present prevalence of the disease. Much work has been done in the prevention of liver cancer, including monitoring of high risk populations (residents in areas of high incidence, cirrhosis of various causes and patients with chronic hepatitis)[1,2], extensive immunization to prevent HBV infection[3], and use of interferon to alleviate HCV infection, both providing hope of lowering or delaying the complication of hepatic cirrhosis or progression to liver cancer in chronic hepatitis patients[4]. More and more efforts have been made in search of natural materials and foods as a means of chemical prevention of liver cancer[5]. Characterized by low toxicity and effectiveness, traditional Chinese herbs have aroused more interest in the prevention of tumors[6]. Ganfujian granule is an oral granule consisting of dietary and medicinal Chinese herbs for preventing liver cancer, developed by our laboratory based on the basic traditional Chinese medicine (TCM) theories of “invigorating spleen and soothing liver” as the key principle in combination with our clinical experience in preventing and treating liver cancer. Our previous experiments have demonstrated that Ganfujian granule can effectively prevent the formation of liver preneoplastic lesions[7]. In the present study we attempted to observe the effect of Ganfujian granule on DEN induced liver cancer in rats and to explore the potential preventive mechanism from the perspective of modulation of proliferation of hepatocytes.
Male SD rats (160-180 g in body mass) were obtained from the Animal Center of the Second Military Medical University of Shanghai. DEN and 2-bromo-3’-deoxyuridine (BrdU) marker were from Sigma, CDK4 sheep monoclonal antibody and cyclin D1 monoclonal antibody were from Santa Cruz, PCNA and BrdU test kit from DAKO.
Animal model The rats had free access to food and water throughout the study. The basal diet was a standard diet. The Ganfujian granule-containing diets were prepared by mixing 3.04% Ganfujian granule with the standard diet (the Ganfujian granule amount for the rats was converted from the kg body mass daily for adults). After a 5 d acclimatization period, 165 rats were assigned into group I (carcinogen-exposed control) and group II (Ganfujian granule treatment). Fresh sterile water was used to prepare DEN solution of 100 µg/mL concentration, to which the rats had free access. The rats in group I were fed with basal diet and those in group II with Ganfujian granule containing diet. At wk 16 DEN water was discontinued and the food for the animals in group II was changed into basal diet. Five rats from each group were sacrificed at wk 4, 8, 12 and 16 of DEN hepatocarcinogenesis. Livers of the rats were resected, cut into 3 mm slices and fixed. Another 30 rats from each group were sacrificed at wk 20 to observe incidence rate of liver cancer. The remaining animals were raised continuously to observe survival. The end point of observation was at wk 28.
Immunohistochemical staining The liver tissues were fixed in 40 g/L buffered formaldehyde and processed for embedding in paraffin, cut into 4 µm thick slices, deparaffinized with xylene and washed with alcohol, and then processed for immunohistochemistry by using a streptavidin-biotin-peroxidase complex methods. Cyclin D1 rat monoclonal antibody, CDK4 rabbit polyclonal antibody and PCNA rat monoclonal antibody were used as primary antibodies. As controls, known positive tissue sections were used, and for negative controls exposure to the primary antibody was omitted.
To determine labeling index (LI), 5 rats from each group at wk 20 received intraperitoneal injections of 2-bromo-3’-deoxyuridine( BrdU ) 35 mg/kg body mass 1 h before death, BrdU incorporation into nuclei was determined immunohistochemically with a ‘BrdU test kit’.
Staining of cyclin D1 and CDK4 foci in liver tissue section was analyzed and quantified using an image analyzer (HPIAS-1000 highly clear color pathological graph analysis system). With the gray scale unified, five different fields were selected for each section to conduct automatic measurement by selecting the mean gray scale as the index of measurement, ×400. The system uses 256 as the maximal gray of white and 0 as the minimal gray of black. The smaller the mean gray scale, the higher the positive reaction. The brownish yellow particles and dark blue particles covering the nuclei represented the positive signal of PCNA and BrdU respectively. The number of positive liver cells was counted. Ten un-overlapped high power fields (×40) of the same transmigration were used for counting the positive and negative cells (100 cells).
Data relative to tumor incidence were analyzed by Fisher’s exact test. Survival functions were analyzed by the log-rank analysis. The differences between groups were analyzed by Student’s t test, and the two-tailed statistical significance was determined. The statistical calculations were carried out by SPSS and NOSA software packages.
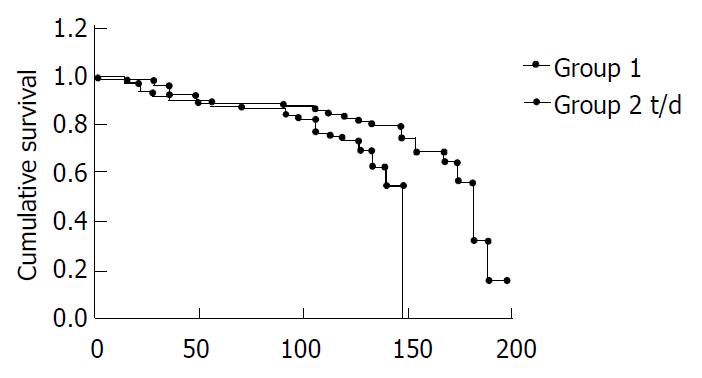
The liver surface of rats in the group I grew coarse gradually between 4-8 wk of DEN hepatocarcinogenesis, on which small granules of local focus were seen. Light microscopy showed that liver cells became degenerative and locally necrotic. Cirrhosis was evidently seen in 40% at wk 12, and at wk 16, cirrhosis was seen in all the rats (100%). More and larger nodules were seen on the liver surface, the liver became smaller, and deranged pseudolobuli of different sizes were seen histologically. Early cancer change was seen in 20% rats. At wk 20, all the 30 rats (100%) developed hepatocarcinoma. Liver impairment of group II was milder than that of group I at various stages of hepatocarcinogenesis. No cirrhosis was seen at wk 12, 60% developed cirrhosis at wk 16, but no cancerous change was observed, and at wk 20, 24 of the 30 rats (80%) developed liver cancer. Fisher’s exact test analysis showed that the incidence rate of liver cancer between the two groups was significant (P < 0.05). Using 28 wk as the end point of observation, the longest survival of rats in group I was 20 wk, and 28 wk in group II. Log-rank test showed that survival of rats in group II was longer than rats in group I, and risk of death in group I was lower than that in group II (P < 0.05, Figure 1).
The number of PCNA positive hepatocytes increased gradually with progression of hepatocarcinogenes is at the early stage, and showed a tendency of sharp increase after 12 wk. Two-tailed t test showed that the number of PCNA positive hepatocytes of group II was lower than that of group I, and the difference between two groups was evident between 12 and 20 wk (P < 0.05), (Table 1). The number of BrdU positively labeled hepatocytes was also smaller in group II at wk 20 of hepatocarcinogenesis (28.80 ± 0.65 vs 45.71 ± 1.90, P < 0.05).
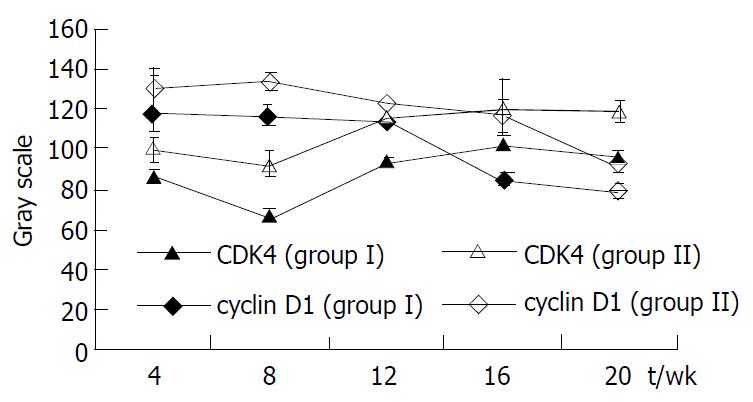
The gray scale of positively stained cyclin D1 of hepatocytes in rats treated with DEN decreased continuously, that is, positive expression of cyclin D1 showed an increasing tendency, reaching the peak at wk 16-20 of hepatocarcinogenesis. Positive expression of CDK4 reached the peak at wk 8, decreased gradually afterwards, and rose again gradually at wk 20. Linear regression correlation analysis showed that there was no significant correlation between two modulators (P > 0.05). Positive expression of CDK4 in group II was significantly lower than that in group I during different stages of hepatocarcinogenesis (P < 0.05) and the expression of cyclin D1 was significantly lower at wk 12 and 20 (P < 0.05), (Figure 2).
Carcinogenesis is a multi-stage process characterized by continuous change in specific heredity and phenotype. The whole process of carcinogenesis is accompanied by subsequent activation of a group of protooncogenes and inactivation of cancer suppressor genes, leading to continuous accumulation of uncontrolled proliferation of cells[8]. Therefore, the liver backgrounds such as nodular cirrhosis and viral hepatitis, and abnormal expression of related genes and proteins play crucial roles in the process of carcinogenesis. In our model of using DEN as the chemical carcinogen, the rat liver underwent different stages of inflammatory change, cirrhosis, carcinogenesis and progression. The incidence rate of liver cancer was 100%. Almost all these liver cancers developed on the basis of cirrhosis. This process might imitate the pathogenesis of human liver cancer to some extent[9,10]. Therefore, our model provides necessary evidence for dynamic observation of the whole process of hepatocarcinogenetic evolution from cancer initiation to malignant transformation based on the change of cell proliferation modulations.
Ganfujian granule is composed of dietary and medicinal Chinese herbs including Chinese yam (Rhizoma Dioscoreae), hawthorn fruit (Fructus Crataegi) and Chinese date (Fructus Ziziphi Jujubae). It is atoxic for long-term use. As a basic formula, it has achieved good therapeutic effects in clinical treatment of liver diseases. In the present study, we observed its effect on DEN induced liver cancer in rats and found that in group I 100% of these rats presented with nodular cirrhosis at wk 16, of which 20% were accompanied by early cancer change at wk 20, all rats developed liver cancer. The longest survival of rats was 20 wk. In group II, cirrhosis was found in 60% rats at wk 16, liver cancer was found in 80% at wk 20. The longest survival of rats was 28 wk. Statistical results showed that Ganfujian granule reduced and delayed the occurrence of liver cancer in rats and prolonged survival of these animals.
Abnormal proliferation of cells is the main feature of carcinogenesis, and therefore exploration of drugs that can affect malignant proliferation of liver cells is of primary importance in chemical prevention of liver cancer. PCNA, a polypeptide chain derived and purified from the cell nucleus, is directly involved in DNA replication. It has been found that the content and positive expression of PCNA were a common index for proliferation of hepatocytes at late G1 stage and early S stage[11]. As BrdU may seep into cells during DNA synthesis, it can be used to reflect the number of S stage cells. Both PCNA and BrdU are good indexes reflecting proliferation of cells. The positive expression of two proteins was mainly found in the pre-cancerous proliferation focus and cancerous liver tissue during DEN induced hepatocarcinogenesis[9]. Their high expression suggested that the ability of cell proliferation became stronger, and this was closely related to malignant cell proliferation and carcinogenesis[12,13]. In the early stage of our experimental model, PCNA positive hepatocytes increased gradually and this increasing tendency became more evident between 12 and 20 wk (the period during which cirrhosis-liver cancer formed). The inhibitory effect of Ganfujian granule on PCNA positive cells was most prominent during this period. In addition, Ganfujian granule also reduced the number of BrdU positively labeled cells at wk 20, suggesting that Ganfujian granule had the action to suppress malignant proliferation of hepatocytes in experimental liver cancer.
The events of cell cycle are normally controlled by the clock within the cell, which is reflected by periodic activation of cyclin D/CDK complex. Irregular regulation may lead to abnormal cleavage of cells and is closely related to malignant transformation of cells[12-15]. In our experiment, we found that positive expression of cyclin D1 tended to increase gradually during DEN induced hepatocarcinogenesis, reaching the peak at the end stage of carcinogenesis. On the other hand, positive expression of CDK4 was active during the early stage of carcinogenesis, became weaker during the middle stage, and rose gradually in the late stage. In group II, positive expression of CDK4 at various stages of carcinogenesis and that of cyclin D1 at wk 16 were significantly lower than those in group I. These findings indicate that DEN induced hepatocarcinogenesis was accompanied by continuous accumulation of cell cycle positive modulators such as cyclin D and CDK, leading to disturbance of cell cycle and uncontrolled proliferation. Ganfujian granule can suppress over-expression of these modulators, regulate the process of cell cycle and capture over-proliferation of hepatocytes that escape the G1-S check-point so as to suppress uncontrolled proliferation and hepatocarcinogenesis.
Based on the results from our experiment, it is concluded that the dietary and medicinal formula Ganfujian granule is able to reduce and delay the occurrence of liver cancer by affecting the abnormal expression of multiple proteins related to cell proliferation cycle at different stages of hepatocarcinogenesis. As a liver cancer chemical preventive agent, clinical application of Gangfujian granule is promising.
Edited by Wang XL Proofread by Zhu LH
| 1. | Lemoine A, Azoulay D, Jezequel-Cuer M, Debuire B. [Hepatocellular carcinoma]. Pathol Biol (Paris). 1999;47:903-910. [PubMed] |
| 2. | Wang QH, liu XF. Study on monitoring of high risk populations of liver cancer. Guowai Yixue Zhongliu Fence. 2002;29:449-451. |
| 3. | Chang MH, Chen DS. Prospects for hepatitis B virus eradication and control of hepatocellular carcinoma. Baillieres Best Pract Res Clin Gastroenterol. 1999;13:511-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 4. | Baffis V, Shrier I, Sherker AH, Szilagyi A. Use of interferon for prevention of hepatocellular carcinoma in cirrhotic patients with hepatitis B or hepatitis C virus infection. Ann Intern Med. 1999;131:696-701. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 59] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Young KJ, Lee PN. Intervention studies on cancer. Eur J Cancer Prev. 1999;8:91-103. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 6. | Li ZQ. Traditional Chinese medicine for primary liver cancer. World J Gastroenterol. 1998;4:360-364. [PubMed] |
| 7. | Qian Y, Ling CQ, Yu CQ, Pan RP, Zhang YN, Wang YZ. Ob-structive effect of three traditional Chinese prescriptions on liver preneoplastic lesion in rats. Disi Junyi Daxue Xuebao. 1999;20:916-918. |
| 8. | Wei W, Gong JP, Qiu FZ. Relationship between cell proliferation and apoptosis during the normal and abnormal liver tissue proliferation. Zhonghua Shiyan Waikexue Zazhi. 2001;18:156-158. |
| 9. | Zhang XL, Shi JQ, Bian XW. Quantitative study on morphologic features and proliferative activity during DEN induced hepatoc-arcinogenesis in rats. Disan Junyi Daxue Xuebao. 2001;23:304-307. |
| 10. | Ling CQ, Qian Y, Zhao JA, Jin Y. Expression of c-myc IGF-II gene and CycinD1 protein in experimental hepatocarcinoma. Shijie Huaren Xiaohua Zazhi. 2001;9:1452-1453. |
| 11. | Lu S, Zhan AH, Huang XX, Ren YJ. Expression and significance of proliferating cell nuclear antigen in resistance of lithium car-bonate to Aflatoxin B1 induced hepatocarcinogenesis in rats. Aibian Jibian Tubian. 2002;14:84-86. |
| 12. | Kui Y, Lin C, Wu W. Relationship between several important modulators of cell cycle G1-S check point and tumor. Guowai Yixue Fenzi Shengwuxue Fence. 1998;20:10-13. |
| 13. | Liu LX, Jiang HC, Zhu AL, Wang XQ, Zhu J. Cell cycle and growth regulators gene expression in liver cancer tissues and adjacent normal tissues. Zhonghua Shiyan Waikexue Zazhi. 2001;18:123-126. |
| 14. | Yuan JH, Zhang RP, Zhang RG, Guo LX, Wang XW, Luo D, Xie Y, Xie H. Growth-inhibiting effects of taxol on human liver cancer in vitro and in nude mice. World J Gastroenterol. 2000;6:210-215. [PubMed] |
| 15. | Yang LJ, Si XH. Expression and significance of cyclin D1 in hu-man hepatocellular carcinoma. Shiyong Zhongliuxue Zazhi. 2000;15:124-126. |