Published online Sep 26, 2013. doi: 10.5662/wjm.v3.i3.27
Revised: July 16, 2013
Accepted: August 16, 2013
Published online: September 26, 2013
Processing time: 96 Days and 18.4 Hours
AIM: To develop a method for studying myocardial area at risk (AAR) in ischemic heart disease in correlation with cardiac magnetic resonance imaging (cMRI).
METHODS: Nine rabbits were anesthetized, intubated and subjected to occlusion and reperfusion of the left circumflex coronary artery (LCx) to induce myocardial infarction (MI). ECG-triggered cMRI with delayed enhancement was performed at 3.0 T. After euthanasia, the heart was excised with the LCx re-ligated. Bifunctional staining was performed by perfusing the aorta with a homemade red-iodized-oil (RIO) dye. The heart was then agar-embedded for ex vivo magnetic resonance imaging and sliced into 3 mm-sections. The AAR was defined by RIO-staining and digital radiography (DR). The perfusion density rate (PDR) was derived from DR for the AAR and normal myocardium. The MI was measured by in vivo delayed enhancement (iDE) and ex vivo delayed enhancement (eDE) cMRI. The AAR and MI were compared to validate the bifunctional straining for cardiac imaging research. Linear regression with Bland-Altman agreement, one way-ANOVA with Bonferroni’s multiple comparison, and paired t tests were applied for statistics.
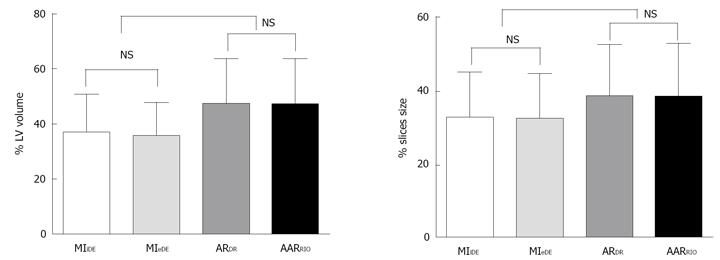
RESULTS: All rabbits tolerated well the surgical procedure and subsequent cMRI sessions. The open-chest occlusion and close-chest reperfusion of the LCx, double suture method and bifunctional staining were successfully applied in all animals. The percentage MI volumes globally (n = 6) and by slice (n = 25) were 36.59% ± 13.68% and 32.88% ± 12.38% on iDE, and 35.41% ± 12.25% and 32.40% ± 12.34% on eDE. There were no significant differences for MI determination with excellent linear regression correspondence (rglobal = 0.89; rslice = 0.9) between iDE and eDE. The percentage AAR volumes globally (n = 6) and by slice (n = 25) were 44.82% ± 15.18% and 40.04% ± 13.64% with RIO-staining, and 44.74% ± 15.98% and 40.48% ± 13.26% by DR showing high correlation in linear regression analysis (rglobal = 0.99; rslice = 1.0). The mean differences of the two AAR measurements on Bland-Altman were almost zero, indicating RIO-staining and DR were essentially equivalent or inter-replaceable. The AAR was significantly larger than MI both globally and slice-by-slice (P < 0.01). After correction with the background and the blank heart without bifunctional staining (n = 3), the PDR for the AAR and normal myocardium was 32% ± 15% and 35.5% ± 35%, respectively, which is significantly different (P < 0.001), suggesting that blood perfusion to the AAR probably by collateral circulation was only less than 10% of that in the normal myocardium.
CONCLUSION: The myocardial area at risk in ischemic heart disease could be accurately determined postmortem by this novel bifunctional staining, which may substantially contribute to translational cardiac imaging research.
Core tip: For developing therapeutic procedures/drugs aimed at modulating infarct size after coronary artery disease, it is important not only to measure myocardial infarction but also to know the extent of area at risk (AAR). However, determination of the AAR both in vivo and ex vivo can be challenging, and controversial. In this experiment we sought to develop a reliable method to accurately localize the culprit coronary occlusion for postmortem verification after in vivo cardiac magnetic resonance imaging and to establish a new bifunctional staining as a standard reference for ex vivo area at risk identification, which may substantially contribute to translational cardiac imaging research.
-
Citation: Feng Y, Ma ZL, Chen F, Yu J, Cona MM, Xie Y, Li Y, Ni Y. Bifunctional staining for
ex vivo determination of area at risk in rabbits with reperfused myocardial infarction. World J Methodol 2013; 3(3): 27-38 - URL: https://www.wjgnet.com/2222-0682/full/v3/i3/27.htm
- DOI: https://dx.doi.org/10.5662/wjm.v3.i3.27
Acute occlusion of a coronary artery initiates an expanding process of myocardial infarction (MI) and this process ends in complete necrosis of the myocardial tissue within the perfusion bed if without a prompt therapeutic intervention. To develop therapeutic procedures and/or drugs aimed at modulating infarct size, it is very important not only to measure the MI size but also to know the extent of perfusion defect zone after coronary occlusion, which is also known as the area at risk (AAR) of myocardium[1]. Since the AAR includes both infracted/nonviable and viable/salvageable myocardium, detection of the AAR may provide an accurate index in clinical practice and preclinical research for diagnostic, therapeutic and prognostic assessment of ischemic heart disease[2,3]. To define the AAR by both in vivo and ex vivo imaging can be challenging. Currently, the most widely used methods for in vivo AAR identification are contrast enhanced computed tomography[4] or cardiac magnetic resonance imaging (cMRI)[5], radionuclide perfusion imaging[6], and contrasted echocardiography[7]. However, determination of the AAR proves difficult and controversial in clinical cardiology[8]. The T2-weighted cMRI may highlight myocardial edema and therefore often overestimate infarct size, and sestamibi single photon emission computed tomography is limited by its low spatial resolution and radioactive nature. Compared to the AAR, MI can be evaluated in vivo by delayed enhancement (DE) cMRI, an established technique that is widely accepted in clinic to reveal acute and chronic MI in patients[9]. This technique has been validated in animal models with reperfused MI using extracellular contrast agents[10,11].
Experimentally, to validate in vivo imaging observations, a variety of ex vivo methods have been developed and attempted to directly visualize and quantify the AAR and MI in animal experiments, because exact postmortem detection of myocardial AAR and MI is deemed necessary as the gold reference. A few decades ago, some investigators tried to assess the AAR through postmortem intracoronary injections of colored industrial dyes[12] or barium gels[13], which stained the normal myocardium but left the AAR unstained. However, highly viscous injectants and undetermined pressure could lead to inaccurate AAR delineation. Nowadays, widespread techniques used for determination of coronary perfusion or AAR are blue dyes (typically Evans blue) and radiolabeled or colored microspheres, with which the heart is perfused at the end of experiments after re-occlusion of the coronary artery[1,14,15]. Triphenyltetrazolium chloride (TTC) histochemical staining is usually combined with a blue dye for simultaneous visualization of normal myocardium, AAR and MI[16]. These methods have their limitations. For instance, the most commonly used water-soluble blue dye Evans-blue may precipitate out of solution and affect the subsequent TTC staining[17] . These dyes are also prone to smearing during slicing, resulting in an ambiguous border definition[17]. The radiolabeled microspheres need the complex procedures and operators have to be exposed to radiation, in addition to the difficulty in some institutions due to unavailable nuclear facilities. The colored microspheres require tissue lyses for counting without morphometry. Therefore, such analyses still remain technically challenging.
To precisely recognize the location of previously occluded coronary branch is crucial for subsequent ex vivo calculation of AAR size. Few study detailed the technical information for accurate coronary re-ligation[18].
Given the suboptimal features of above-mentioned dyes and microspheres for visualizing the AAR in preclinical study, we sought to: (1) develop a reliable method to accurately localize the previous coronary occlusion site for postmortem re-ligation; (2) establish a new bifunctional staining as standard reference for ex vivo AAR evaluation; (3) validate the method by quantitative measurement and morphometry; and (4) compare the AAR defined by the new staining to the cMRI findings at a 3.0 T clinic magnet with histological verifications.
The above goals could have been realized only when has a prior-established methodology, i.e., an occlusion/reperfusion based rabbit model of acute MI and a technical platform for pre- and postmortem imaging co-localization now become available[19,20]. The rabbit as a mediate size experimental animal can be easily handled, with favorable cost-effectiveness and compatibility to clinically available MRI facilities with the entire heart section includable in a standard histological glass slide. The resulted outcomes are deemed pertinent to human patients[19,21].
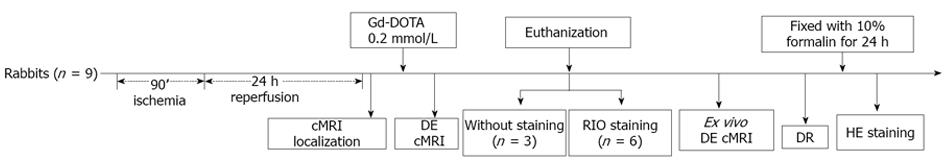
This experimental study was carried out according to a protocol (Figure 1) that was approved by the Animal Care and Use Committee of KU Leuven, Belgium.
The custom-made bifunctional dye of red iodized oil (RIO) was formulated by diluting 20 mg oil-red-O dye (Sigma-Aldrich) in 100 mL of any brand of commercially available iodized oil such as Lipiodol ultra fluid (Guerbet, France), which is also known as ethiodized oil consisting of about 30%-40% iodine and poppyseed oil originally used as a radio-opaque contrast agent to outline structures in radiological investigations or in combination for transarterial chemoembolization of malignant tumors[22,23].
A total of 9 male New Zealand white rabbits (Animal House, KU Leuven, Belgium), weighing 3.2-4.0 kg, were included in this study. Animals were housed under standard laboratory conditions.
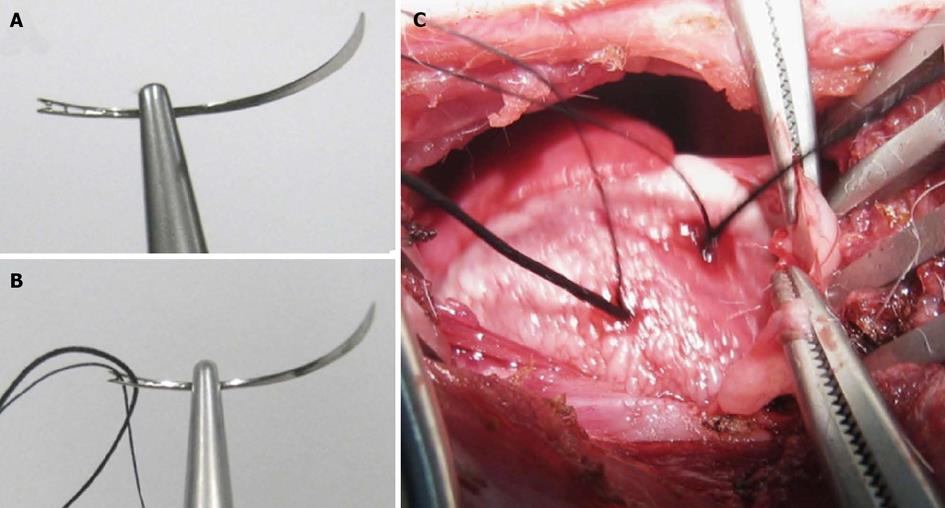
The model of acute reperfused MI was prepared by slight modification on a previously introduced method[19]. After sedation, endotracheal intubation and mechanical ventilation, the rabbit received intravenous (IV) injection of pentobarbital (Nembutal, Sanofi Sante Animale, Brussels, Belgium) at 40 mg/kg per hour to maintain anesthesia during the open-chest operation to dissect the paricardium and expose the left circumflex coronary branch (LCx). For making a removable coronary ligation in vivo and precise re-ligation ex vivo, we used a 1/2 circle shape triangular needle (Sutura, Inc. Fountain Valley, CA, United States) with spring eyes at the end (Figure 2A). Two silk sutures could be easily placed through the separate eyes (Figure 2B): the thicker one (2-0) was used for the LCx ligation that could be removed for reperfusion and the thinner one (5-0) was spared for later ex vivo LCx re-occlusion in order to perform postmortem bifunctional staining (Figure 2C). The acute reperfused MI was induced by tying the thicker suture with a detachable single knot for 90 min, and LCx reperfusion was achieved by pulling the exteriorized end of suture in the closed-chest condition as detailed previously[19]. The ECG (Accusync® 71, Milford, Connecticut, Unites States) was monitored to confirm the formation of MI and to detect the occurrence of arrhythmia, which was treated by IV dose of Xylocaine (AstraZeneca SA, Brussels, Belgium) at 2-4 mg/kg if needed. Buprenorfine hydrochloride (Temgesic; Schering-Plough, Brussels, Belgium) of 0.216 mg was intramuscularly injected to relieve pain. All rabbits were allowed to recover and kept alive until cMRI scanning 24 h after LCx reperfusion.
The rabbit was gas-anesthetized through a mask with 2% isoflurane in the mixture of 20% oxygen and 80% room air and placed supinely in a plastic holder. Using a commercial 8-channel phased array knee coil, the rabbit was subjected to cMRI at a 3.0 T clinical MRI scanner (Trio, Siemens, Erlangen, Germany) with a maximum gradient capability of 45 mT/m. The ECG and respiration were triggered and gated by a Small Animal Monitoring System (SA Instruments, Inc. Stony Brook, New York, United States). Two ECG surface electrodes were attached to the shaved thorax skin with apparently apical pulse and the left leg. The respiration control sensor was attached on the middle of the rabbit abdomen. All data were acquired during free breathing of the animal. After determining the cardiac axes with localizers, the DE cMRI using T1-weighted 3-dimensional segmented k-space inversion recovery turbo fast low angle shot sequence was performed 20 min after intravenous administration of 0.2 mmol/kg Gd-DOTA (Dotarem, Guerbet, France). Short-axis DE cMRI was acquired to depict the presence, location, and extent of the MI. The imaging parameters were: TR: 396 ms; TE: 1.54 ms; TI: 360 ms; FOV: 240 × 180 mm²; FA: 15°; and in-plane resolution: 1.1 × 0.8 mm². The ex vivo higher resolution DE-cMRI was also obtained immediately after animal sacrifice using the same MRI unit with a wrist coil as previously described[19]. The DE cMRI using an extracellular nonspecific contrast agent has been applied in both animal MI models and clinical patients as a gold standard technique for identification and quantification of the MI[10,11]. Therefore, the final MI size was also defined by in vivo and ex vivo DE-cMRI in this study.
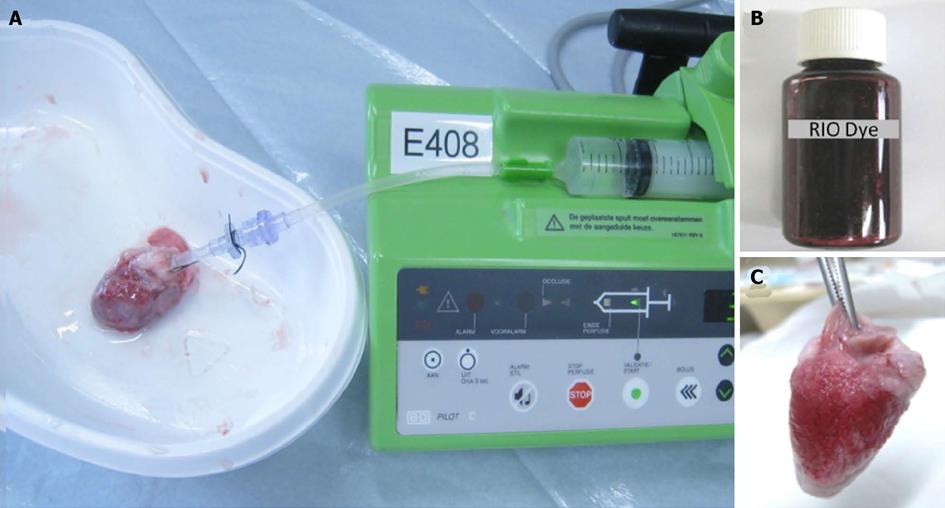
Bifunctional staining of the RIO dye refers to: (1) visualization, location and delineation of the AAR by using its red staining function for further morphometric quantification; and (2) microangiographic examination of the AAR by using the digital radiography (DR) function to compare the perfusion density rate (PDR) between the AAR and normal myocardium. This staining served as a postmortem index for regional myocardial blood flow. Briefly, after the last in vivo cMRI, the rabbit was IV injected with Heparin at 300 IU/kg to prevent coagulation. Under deep anesthesia and thoracotomy, the heart was excised with a section of the aortic arch isolated and pericardial tissue trimmed. As shown in Figure 3A, a plastic catheter filled with 0.9% saline was inserted into the aorta and anchored with its tip 1 cm above the aortic valves. Care was taken to avoid air bubbles entering the catheter that was connected to a 10 mL syringe fixed on the injection bump (BD PILOT, Becton Dickinson Infusion System, France). Under a perfusion pressure of 100 mmHg, 10 mL saline was infused until the eluate from the coronary sinus became clear. The heart was rinsed to wash out remaining blood. Then, the 5-0 suture spared at the coronary occlusion site during open-chest operation was tightened for causing re-occlusion, and 4 mL of RIO dye (Figure 3B) was infused through the aortic catheter for 15 min under the same pressure by using the same pump as mentioned above. The infusion was stopped when the normal myocardium was completely stained red by the RIO dye (Figure 3C). Upon this perfusion staining technique with the LCx re-ligated, the normal myocardium stained brick red and the AAR remained uncolored. Afterwards, the heart was imbedded in agar solution for ex vivo cMRI, followed by serial sectioning into 6-8 slices of 3.0 mm thickness to match with the corresponding short axis cMRI images. Such stained heart sections were exposed by DR with a mammographic unit (Embrace; Agfa-Ge-vaert, Mortsel, Belgium) at 25 KV and 18 mA, and photographed by a digital camera before and after 24 h of fixation with 10% formalin. The fixed heart sections were imbedded into paraffin and processed for histology with 5 micron thick hematoxylin-eosin (HE) stained slices for microscopic inspection.
The MI size on cMRI was determined in consensus by three authors with the built-in software of Siemens workstation (SyngoMR A30). To facilitate cardiac morphometric comparisons with different imaging methods, the dimension was calculated as a value relative to the entire left ventricular myocardium. The MI size (%) on DE cMRI was derived semi-automatically by computer to count all enhanced pixels with the signal intensity higher than two standard deviations above the mean of image intensities in the remote NM, which were divided by all ventricular pixels on the same short-axis image. Digital photographs of RIO-staining sections and DR images were transferred to a personal computer for planimetric analysis by using Image J 1.38x software (Research Services Branch, NIH, Bethesda, MD, United States). The AAR defined as non-stained area by RIO-dye and non-opaque area by DR was contoured with consensus on the computer screen. Due to partial volume effect and fatty tissue interference on DE cMRI, the apical and basal slices were excluded and the remaining slices (n = 25) obtained from 6 rabbits were used for the comparisons of the corresponding bifunctional staining sections.
The measurement of perfusion density rate (PDR%) measurement was performed on DR images of the heart sections using the same Image J 1.38x software. The blank myocardial density refers to the DR density of the heart tissues from control group (n = 3) without RIO dye staining. The AAR of low density and the normal myocardium of high density were delineated in all sections (n = 25) with an operator-defined region of interest. The mean gray value (GV) of all sections were automatically generated and normalized by the background GV. PDR% refers to the increased perfusion density relative to background, which was calculated by the following formula: PDR% = (GVregion of interest-GVblank myocardial density)/GVbackground× 100.
Numerical data were expressed as mean ± SD. Comparisons among multiple factors were performed using one way ANOVA, followed by the Bonferroni’s test to identify any differences between each two techniques. All tests were two tailed. The correlations were tested by Spearman’s method. The linear regression and the means of Bland-Altman analysis were used for correlation and agreement, respectively. Correlations were described by the correlation coefficient with 95%CIs. A difference was considered statistically significant if the P value was less than 0.05. The paired student t test was used for comparison of the PDR between the AAR and MI. The statistical analyses were performed with Analyse-it® 2.14 for Excel and GraphPad Prisma® 5.0.
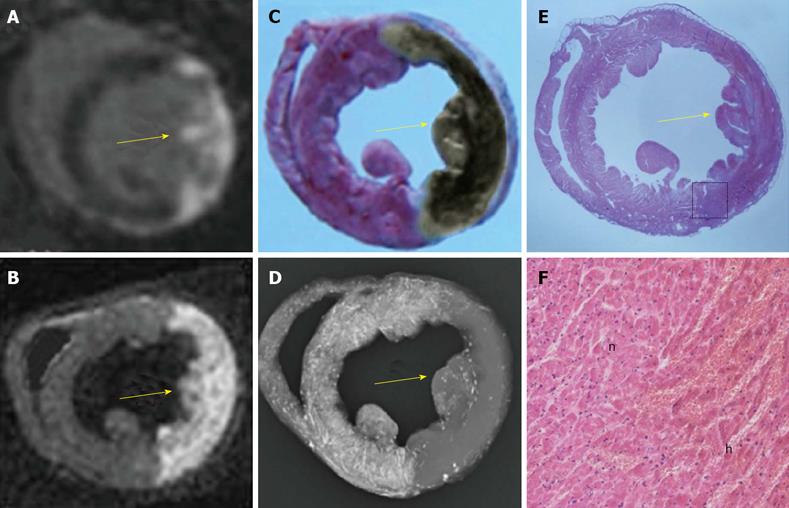
All animals survived the anesthesia, surgical procedures for inducing acute reperfused MI and subsequent cMRI. As one of the critical steps, the double suture method was successfully applied in all animals, which ensured later bifunctional staining. The normal myocardium from all rabbits was well perfused by the RIO dye. Figure 4 demonstrated a case with a large reperfused MI by different techniques. The hyperenhanced transmural MI appeared similarly on both iDE and eDE (Figure 4A, B), which was somewhat smaller than the AAR as an unstained region by RIO (Figure 4C) and non-opacified region on DR (Figure 4D). With this bifunctional staining, the large haemorrhagic MI appeared brown greyish (Figure 4C) after overnight Formalin fixation. The myocardial necrosis with haemorrhage was finally confirmed by macro- and microscopic views of HE stained slice (Figure 4E, F).
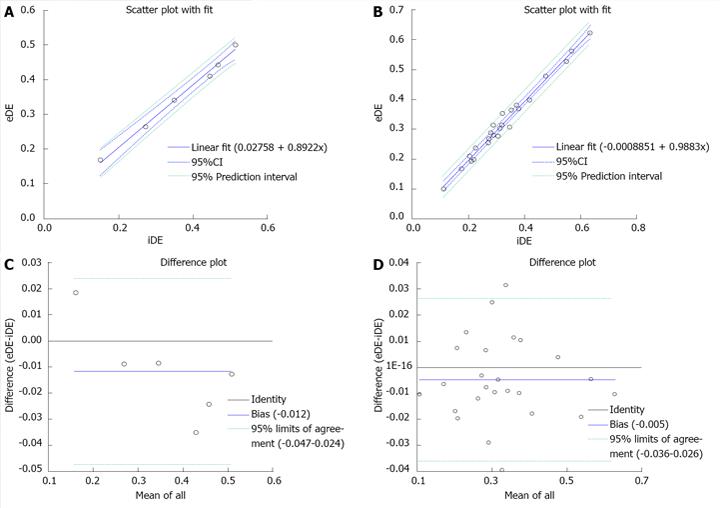
The location and extent of the MI were clearly shown as the hyperenhanced region on DE cMRI in all cases. The percentage MI sizes globally (n = 6) and slice-by-slice (n = 25) were 36.59% and 35.41% on in vivo DEcMRI (iDE), and 32.88% and 32.40% on ex vivo DEcMRI (eDE). There was no significant difference between iDE and eDE. Linear regression analyses showed an excellent correlation both globally and slice-by-slice between MI volumes on iDE and eDE (r = 0.89, 0.98) (Figure 5A, B). By Bland-Altman analysis, there was a 1.2% bias with 95%CIs of 2.4% to 4.7% in global heart (Figure 5C) and 0.5% bias with 95%CIs of 2.6% to 3.6% by slice (Figure 5D), indicating high agreement of the two measurement methods.
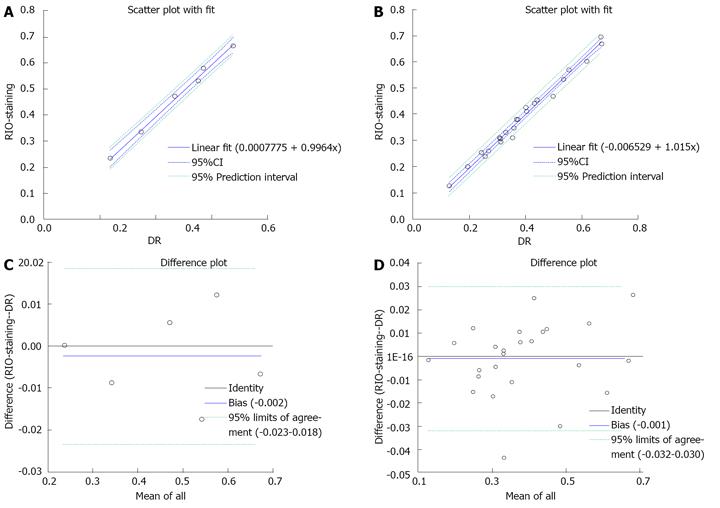
The RIO dye was successfully formulated (Figure 3B). It showed homogenous distribution into the accessible cardiac tissues. After staining, the normal myocardium appeared scarlet red (Figure 3C) and rose red (Figure 4C) before and after formalin fixation and as more opacified region on DR images (Figure 4D), whereas the AAR showed as non-RIO-stained region on heart slices (Figure 4C) and the less opacified region on DR image (Figure 4D). By bifunctional staining, the percentage AARglobal was 44.8% ± 15.18% and 44.74% ± 15.98%, and percentage AARslice was 40.04% ± 13.64% and 40.48% ± 13.26% by RIO-staining and DR, respectively. There was no significant difference between RIO-staining and DR (P = 0.9). Linear regression analyses showed an excellent correlation both globally and slice-by-slice between AAR volumes defined by RIO-staining and DR (r = 0.99, 1.0) (Figure 6A, B). The limits of Bland-Altman agreement were 3.2%-3.0% (Figure 6C) by slice and 2.3%-1.8% (Figure 6D) in global measurements with RIO-staining and DR respectively. The mean difference was almost zero, indicating a very high agreement between RIO-staining and DR techniques.
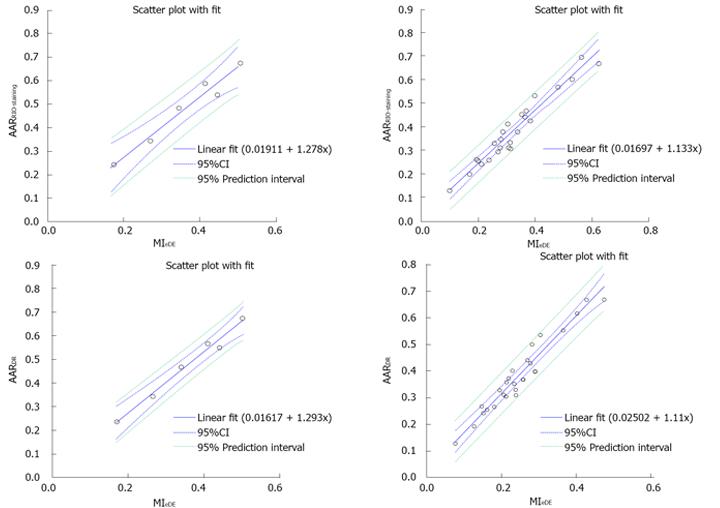
The MI quantified from both iDE and eDE cMRI were significantly smaller than the AARRIO-staining and AARDR with bifunctional staining (P < 0.01; Figure 7), confirming that the final MI was smaller than ischemic region. However, the MI on eDE-cMRI correlated well with AARRIO-staining and AARDR (Figure 8), suggesting that the bifunctional staining with RIO dye could successfully delineate and characterize the AAR.
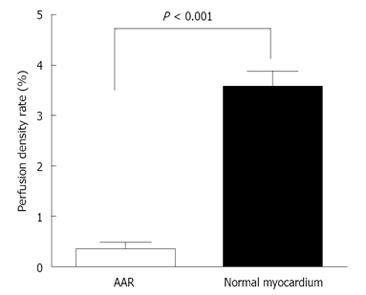
After aortic infusion with the RIO dye while the LCx was re-ligated, the PDR of the normal myocardium and AAR became 354.89% ± 35.18% and 32.28% ± 15.06% relative to the equipment background. The PDR was significantly lower in the AAR, i.e., only about 9.10% ± 3.93% of the PDR in the normal myocardium (P < 0.001) (Figure 9), suggesting insufficient collateral coronary blood supply during the 90 min occlusion, leading to the rabbit model of reperfused MI.
We studied myocardial AAR in the rabbit heart after 90 min of LCx occlusion followed by reperfusion. The AAR refers to the myocardium without blood supply during coronary artery occlusion. The final MI was detected by in vivo and ex vivo DE cMRI. The main outcomes of the present study are: (1) the newly developed method of bifunctional staining enabled reliable high-quality images; (2) both in vivo and ex vivo DE cMRI allowed visualization and quantification of the MI; (3) though measured with different methods, the dimension of the AAR was significantly larger than that of the MI, suggesting the potential of this method to help discriminate the well perfused normal myocardium from the non-perfused nonviable and/or less perfused by viable probably salvageable myocardium; and (4) the PDR was 10 times lower in the AAR compared to the normal myocardium, suggesting that the bifunctional RIO dye may facilitate not only morphometry but also perfusion quantification.
The animal models of reperfused MI have been extensively applied for cardiovascular research both in large and small animals. How to accurately re-ligate coronary artery branch to replicate the exact occlusion site prior to reperfusion appears crucial. The majority of the authors used the snare-loop method for coronary artery occlusion-reperfusion on the open-chest MI model with later postmortem vessel re-ligation[24]. Other studies described re-occlusion of coronary branches with a balloon catheter under the close-chest conditions[25]. However, there is a lack of detailed information about how to imitate postmortem the exact in vivo artery occlusion, which is though a prerequisite for defining the AAR or ischemic area. We have previously introduced an effective method for in vivo LCx occlusion and reperfusion[19]. In this study we modified that method by using the double sutures to precisely co-localize LCx obstruction both in vivo and ex vivo. Within the same puncture, one suture was for in vivo coronary occlusion and reperfusion, and the other suture was left spared in vivo but tightened with a knot to re-occlude the coronary branch ex vivo. This double suture method was successfully validated in all animals, which is a critical step to ensure later bifunctional staining to obtain the desired results as presented.
In the cardiac imaging research on the ischemic heart disease using animal hearts, a most crucial issue is adequate staining of the AAR and discrimination between risk and non-risk zones, because a correctly defined borderline between the ischemic and necrotic zones will have a direct impact on MI diagnosis and therapeutic interventions. In this article we described a new bifunctional staining method to define the AAR, which, to our best knowledge, has not been reported in the literature. Function 1 is to visualize the normal myocardium in red color but the AAR uncolored utilizing the oil-red-o component of the custom-made RIO dye, which facilitates morphometric delineation, quantification and comparison; whereas function 2 is to enable visualization and volumetric density measurement of the non-opacified normal myocardium and the opacified AAR by utilizing the iodized oil component of the RIO dye for making DR to extrapolate, quantify and evaluate the difference in hemodynamic features such as coronary vessel density, interstitial distribution space and/or regional myocardial blood flow/volume between the normal and ischemic myocardium.
There have been some methods reported in the literature for postmortem delineation of the AAR. Coronary infusion of hydrophilic Evans-blue solution is the most common technique to stain the remote myocardium while the insulted coronary branch is re-ligated[25-27]. Evans-blue is a water-soluble intravital dye with strong albumin binding capacity and has been used in hemodynamic studies for decades[28,29]. More recently Evans-blue has been found with high necrosis avidity and used to clearly mark the well-reperfused necrotic tissues[30-32]. Therefore, there is a risk of underestimation of the AAR due to contamination of the inwards diffused Evans-blue to the ischemic necrosis[15]. Some other groups tried to use TTC for demarcating the AAR, however TTC perfusion induced severe tissue contraction, which may affect the geometry of heart and might prevent subsequent successful dye perfusion[17]. Other studies used fluorescein dyes under dark background or radio- and color-labeled microspheres to define or quantify the AAR[33-35]. However, all these methods are only unifunctional and/or lacking required accuracy. The custom-made bifunctional RIO dye as introduced in this study appears a much better alternative to Evans blue. The first component of RIO dye is oil-red-O, which is a stable lipophilic pigment and can be easily diluted in any lipid media such as the iodized oil as the second component of this dye. The distribution of the RIO dye in the cardiac tissue after intracoronary infusion seems only pressure dependent rather than being diffusible afterwards driven by its concentration gradient due to the fact that a hydrophobic dye exists in the hydrophilic environment of myocardium, resulting in a more accurate delineation of the borders of the AAR without infiltration of the dye into the adjacent ischemic region.
Suspension of barium sulphate has also been used for postmortem coronary angiography[13,36]. However, the heavy particles tended to precipitate in its water media, leading to suboptimal perfusion to vascular bed with smaller vessels. As shown in this study, the second component of iodized oil in the RIO dye solved the problem well in DR and its quantification. Thus, the golden combination of the two dye components facilitated successfully the bifunctional staining and enabled comprehensive AAR study.
We have gained the following experiences: (1) heparinizing the animal prior to sacrifice and rinsing the coronary bed with normal saline beforehand proved beneficial for the next bifunctional staining of the AAR by RIO dye perfusion; (2) controlling adequate pressure and observing epicardial red coloration during dye infusion were crucial for the success of procedure; and (3) fixing the heart sections with formalin overnight helped color differentiation between the normal myocardium (rose red) and AAR (gray or brown grayish depending on the degree of hemorrhage) and later planimetric quantification due to a clear borderline.
Myocardial blood volume (capillary bed) and myocardial blood flow can be determined by microsphere methods in animals after excision of the heart[33,35]. Such methods, however, require cutting the myocardium into discrete samples, which may be a combination of normal tissue, border zone, hyperemic tissue, and severely ischemic tissue. Thus, the processing of selected tissue samples can bias the results as well as blur the exact relation to overall myocardial morphology. Although the degree of resolution for flow measurement could possibly be improved by cutting myocardium into smaller samples, this becomes tedious and impractical for studying flow throughout the heart. Besides, it is more difficult to get topographic information.
Using the PDR derived from microangiography on DR to indirectly evaluate vascular blood perfusion to viable and necrotic tissues has been reported[37]. In our experiment, The PDR was significantly lower in AAR, which was about 9% of the normal myocardium. This finding is close to the results of Connelly’s group on myocardium blood flow measured by radioactive microspheres on the similar animal setting[38]. The other group showed that in the reperfusion MI dog model, myocardial samples with only mild (about 20%-30%) reductions in myocardial blood flow compared to non-ischemic regions[39], which appears higher than our results probably due to the richer collaterals of coronary artery in dogs than in rabbit[40]. The application of this new staining method on our rabbit model was successful and can be translated to other species.
Accurate quantitative measurement of MI size is an important aspect of cardiovascular research. The in vivo cMRI for accurate evaluation of MI has been described previously in rabbit models[19]. In this study we have performed the high resolution ex vivo cMRI with extensive postmortem evaluations on rabbit hearts, which were supported by histological staining and consistent to in vivo cMRI. The bifunctional staining could be incorporated into the study using clinically available MRI facilities to acquire outcomes that are more pertinent to human patients.
First, the AAR was demarcated by intracoronarily infusing the heart with the RIO dye after local re-occlusion. Such a procedure was time-consuming with possible technical failure to obtain homogeneous staining of all accessible normal myocardium. Secondly, the postmortem dye distribution may differ from in vivo blood perfusion of the normal myocardium when the related artery is occluded, because normal coronary flow is influenced by the intraventricular pressure so the flow in postmortem infusion may not exactly simulate the in vivo flow pattern. Furthermore, in this experiment by using RIO dye, we could achieve accurate delineation of the AAR postmortem, but we could hardly discriminate the MI from any ischemic but still viable tissues within the AAR. Considering these drawbacks, we have to further optimize this experimental setting for determination of both AAR and MI under both in vivo and ex vivo conditions.
In conclusion, this article introduced a novel bifunctional staining method to accurately visualize and quantify AAR postmortem, which was successfully applied in rabbit models with ischemia-reperfusion MI. The key advantages include a newly developed bifunctional RIO dye, a new ex vivo staining method with greater color intensity in the remote normal myocardium and improved contrast for border delineation to obtain both morphometric and functional information for reliable AAR measurement. This methodology is deemed of high research values and to substantially contribute to the translational research in cardiovascular sciences.
The corresponding author Yicheng Ni is currently a Bayer Lecture Chair holder.
For developing therapeutic procedures/drugs aimed at modulating infarct size after coronary artery disease, it is important not only to measure myocardial infarction but also to know the extent of area at risk (AAR). However, determination of the AAR both in vivo and ex vivo can be challenging, and controversial.
In this experiment the authors sought to develop a reliable method to accurately localize the culprit coronary occlusion for postmortem verification after in vivo cardiac magnetic resonance imaging and to establish a new bifunctional staining as a standard reference for ex vivo area at risk identification, which may substantially contribute to translational cardiac imaging research.
The myocardial area at risk in ischemic heart disease could be accurately determined postmortem by this novel bifunctional staining, which may substantially contribute to translational cardiac imaging research.
This is a conscientious and carefully written preclinical paper.
P- Reviewer Chang ST S- Editor Song XX L- Editor A E- Editor Lu YJ
| 1. | Aletras AH, Tilak GS, Natanzon A, Hsu LY, Gonzalez FM, Hoyt RF, Arai AE. Retrospective determination of the area at risk for reperfused acute myocardial infarction with T2-weighted cardiac magnetic resonance imaging: histopathological and displacement encoding with stimulated echoes (DENSE) functional validations. Circulation. 2006;113:1865-1870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 433] [Cited by in RCA: 419] [Article Influence: 22.1] [Reference Citation Analysis (0)] |
| 2. | Berry C, Kellman P, Mancini C, Chen MY, Bandettini WP, Lowrey T, Hsu LY, Aletras AH, Arai AE. Magnetic resonance imaging delineates the ischemic area at risk and myocardial salvage in patients with acute myocardial infarction. Circ Cardiovasc Imaging. 2010;3:527-535. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 110] [Cited by in RCA: 100] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 3. | Darsee JR, Kloner RA, Braunwald E. Demonstration of lateral and epicardial border zone salvage by flurbiprofen using an in vivo method for assessing myocardium at risk. Circulation. 1981;63:29-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Mewton N, Rapacchi S, Augeul L, Ferrera R, Loufouat J, Boussel L, Micolich A, Rioufol G, Revel D, Ovize M. Determination of the myocardial area at risk with pre- versus post-reperfusion imaging techniques in the pig model. Basic Res Cardiol. 2011;106:1247-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 5. | Friedrich MG, Abdel-Aty H, Taylor A, Schulz-Menger J, Messroghli D, Dietz R. The salvaged area at risk in reperfused acute myocardial infarction as visualized by cardiovascular magnetic resonance. J Am Coll Cardiol. 2008;51:1581-1587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 351] [Article Influence: 20.6] [Reference Citation Analysis (0)] |
| 6. | Ceriani L, Verna E, Giovanella L, Bianchi L, Roncari G, Tarolo GL. Assessment of myocardial area at risk by technetium-99m sestamibi during coronary artery occlusion: comparison between three tomographic methods of quantification. Eur J Nucl Med. 1996;23:31-39. [PubMed] |
| 7. | Villanueva FS, Glasheen WP, Sklenar J, Kaul S. Characterization of spatial patterns of flow within the reperfused myocardium by myocardial contrast echocardiography. Implications in determining extent of myocardial salvage. Circulation. 1993;88:2596-2606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 103] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 8. | Arai AE, Leung S, Kellman P. Controversies in cardiovascular MR imaging: reasons why imaging myocardial T2 has clinical and pathophysiologic value in acute myocardial infarction. Radiology. 2012;265:23-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 40] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 9. | Bondarenko O, Beek AM, Hofman MB, Kühl HP, Twisk JW, van Dockum WG, Visser CA, van Rossum AC. Standardizing the definition of hyperenhancement in the quantitative assessment of infarct size and myocardial viability using delayed contrast-enhanced CMR. J Cardiovasc Magn Reson. 2005;7:481-485. [PubMed] |
| 10. | Oshinski JN, Yang Z, Jones JR, Mata JF, French BA. Imaging time after Gd-DTPA injection is critical in using delayed enhancement to determine infarct size accurately with magnetic resonance imaging. Circulation. 2001;104:2838-2842. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 164] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 11. | Kim RJ, Fieno DS, Parrish TB, Harris K, Chen EL, Simonetti O, Bundy J, Finn JP, Klocke FJ, Judd RM. Relationship of MRI delayed contrast enhancement to irreversible injury, infarct age, and contractile function. Circulation. 1999;100:1992-2002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1774] [Cited by in RCA: 1744] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 12. | Lowe JE, Reimer KA, Jennings RB. Experimental infarct size as a function of the amount of myocardium at risk. Am J Pathol. 1978;90:363-379. [PubMed] |
| 13. | Ohzono K, Koyanagi S, Urabe Y, Harasawa Y, Tomoike H, Nakamura M. Transmural distribution of myocardial infarction: difference between the right and left ventricles in a canine model. Circ Res. 1986;59:63-73. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 14. | Hale SL, Alker KJ, Kloner RA. Evaluation of nonradioactive, colored microspheres for measurement of regional myocardial blood flow in dogs. Circulation. 1988;78:428-434. [PubMed] |
| 15. | Redel A, Jazbutyte V, Smul TM, Lange M, Eckle T, Eltzschig H, Roewer N, Kehl F. Impact of ischemia and reperfusion times on myocardial infarct size in mice in vivo. Exp Biol Med (Maywood). 2008;233:84-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 43] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 16. | Khalil PN, Siebeck M, Huss R, Pollhammer M, Khalil MN, Neuhof C, Fritz H. Histochemical assessment of early myocardial infarction using 2,3,5-triphenyltetrazolium chloride in blood-perfused porcine hearts. J Pharmacol Toxicol Methods. 2006;54:307-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 17. | Bohl S, Medway DJ, Schulz-Menger J, Schneider JE, Neubauer S, Lygate CA. Refined approach for quantification of in vivo ischemia-reperfusion injury in the mouse heart. Am J Physiol Heart Circ Physiol. 2009;297:H2054-H2058. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Rodrigues AC, Hataishi R, Ichinose F, Bloch KD, Derumeaux G, Picard MH, Scherrer-Crosbie M. Relationship of systolic dysfunction to area at risk and infarction size after ischemia-reperfusion in mice. J Am Soc Echocardiogr. 2004;17:948-953. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 19. | Feng Y, Xie Y, Wang H, Chen F, Ye Y, Jin L, Marchal G, Ni Y. A modified rabbit model of reperfused myocardial infarction for cardiac MR imaging research. Int J Cardiovasc Imaging. 2009;25:289-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 22] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Feng Y, Chen F, Xie Y, Wang H, Cona MM, Yu J, Li J, Bogaert J, Janssens S, Oyen R. Lipomatous metaplasia identified in rabbits with reperfused myocardial infarction by 3.0 T magnetic resonance imaging and histopathology. BMC Med Imaging. 2013;13:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 11] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 21. | van der Laarse A, van der Wall EE. Rabbit models: ideal for imaging purposes? Int J Cardiovasc Imaging. 2009;25:299-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 22. | Eurvilaichit C. Lipiodol enhanced CT scanning of malignant hepatic tumors. J Med Assoc Thai. 2000;83:398-406. [PubMed] |
| 23. | Kurokohchi K, Deguchi A, Masaki T, Himoto T, Yoneyama H, Kobayashi M, Maeta T, Kiuchi T, Kohi F, Miyoshi H. Successful treatment of hypovascular advanced hepatocellular carcinoma with lipiodol-targetting intervention radiology. World J Gastroenterol. 2007;13:4398-4400. [PubMed] |
| 24. | Higuchi T, Taki J, Nakajima K, Kinuya S, Namura M, Tonami N. Time course of discordant BMIPP and thallium uptake after ischemia and reperfusion in a rat model. J Nucl Med. 2005;46:172-175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 25. | Dębiński M, Buszman PP, Milewski K, Wojakowski W, Jackiewicz W, Pająk J, Szurlej D, Fryc-Stanek J, Wiernek S, Jelonek M. Intracoronary adiponectin at reperfusion reduces infarct size in a porcine myocardial infarction model. Int J Mol Med. 2011;27:775-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 26. | Michael LH, Entman ML, Hartley CJ, Youker KA, Zhu J, Hall SR, Hawkins HK, Berens K, Ballantyne CM. Myocardial ischemia and reperfusion: a murine model. Am J Physiol. 1995;269:H2147-H2154. [PubMed] |
| 27. | Poulsen RH, Bøtker HE, Rehling M. Postreperfusion myocardial technetium-99m-sestamibi defect corresponds to area at risk: experimental results from an ischemia-reperfusion porcine model. Nucl Med Biol. 2011;38:819-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 28. | Heltne JK, Farstad M, Lund T, Koller ME, Matre K, Rynning SE, Husby P. Determination of plasma volume in anaesthetized piglets using the carbon monoxide (CO) method. Lab Anim. 2002;36:344-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 25] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 29. | Fry DL. Certain histological and chemical responses of the vascular interface to acutely induced mechanical stress in the aorta of the dog. Circ Res. 1969;24:93-108. [PubMed] |
| 30. | Ni Y. Metalloporphyrins and functional analogues as MRI contrast agents. Curr Med Imaging Rev. 2008;4:96-112. [RCA] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 58] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 31. | Li J, Cona MM, Chen F, Feng Y, Zhou L, Yu J, Nuyts J, de Witte P, Zhang J, Himmelreich U. Exploring theranostic potentials of radioiodinated hypericin in rodent necrosis models. Theranostics. 2012;2:1010-1019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 32. | Wang H, Miranda Cona M, Chen F, Li J, Yu J, Feng Y, Peeters R, De Keyzer F, Marchal G, Ni Y. Comparison between nonspecific and necrosis-avid gadolinium contrast agents in vascular disrupting agent-induced necrosis of rodent tumors at 3.0T. Invest Radiol. 2011;46:531-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 33. | Malik AB, Kaplan JE, Saba TM. Reference sample method for cardiac output and regional blood flow determinations in the rat. J Appl Physiol. 1976;40:472-475. [PubMed] |
| 34. | Deveci D, Egginton S. Development of the fluorescent microsphere technique for quantifying regional blood flow in small mammals. Exp Physiol. 1999;84:615-630. [PubMed] |
| 35. | Chien GL, Anselone CG, Davis RF, Van Winkle DM. Fluorescent vs. radioactive microsphere measurement of regional myocardial blood flow. Cardiovasc Res. 1995;30:405-412. [PubMed] [DOI] [Full Text] |
| 36. | Jugdutt BI, Hutchins GM, Bulkley BH, Pitt B, Becker LC. Effect of indomethacin on collateral blood flow and infarct size in the conscious dog. Circulation. 1979;59:734-743. [PubMed] |
| 37. | Wang H, Sun X, Chen F, De Keyzer F, Yu J, Landuyt W, Vandecaveye V, Peeters R, Bosmans H, Hermans R. Treatment of rodent liver tumor with combretastatin a4 phosphate: noninvasive therapeutic evaluation using multiparametric magnetic resonance imaging in correlation with microangiography and histology. Invest Radiol. 2009;44:44-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 38. | Connelly CM, Leppo JA, Weitzman PW, Vogel WM, Apstein CS. Effect of coronary occlusion and reperfusion on myocardial blood flow during infarct healing. Am J Physiol. 1989;257:H365-H374. [PubMed] |
| 39. | Chu A, Cobb FR. Reperfusion alters the relation between blood flow and the remaining myocardial infarction. Circulation. 1989;79:884-889. [PubMed] |