Peer-review started: May 27, 2020
First decision: June 5, 2020
Revised: August 19, 2020
Accepted: September 14, 2020
Article in press: September 14, 2020
Published online: November 29, 2020
Processing time: 181 Days and 17.1 Hours
Contrast-induced nephropathy (CIN) is a reversible form of acute kidney injury that occurs within 48-72 h of exposure to intravascular contrast material. CIN is the third leading cause of hospital-acquired acute kidney injury and accounts for 12% of such cases. Risk factors for CIN development can be divided into patient- and procedure-related. The former includes pre-existing chronic renal insufficiency and diabetes mellitus. The latter includes high contrast volume and repeated exposure over 72 h. The incidence of CIN is relatively low (up to 5%) in patients with intact renal function. However, in patients with known chronic renal insufficiency, the incidence can reach up to 27%.
To examine the association between renal enhancement pattern on non-contrast enhanced computed tomographic (CT) images obtained immediately following hepatic artery embolization with development of CIN.
Retrospective review of all patients who underwent hepatic artery embolization between 01/2010 and 01/2011 (n = 162) was performed. Patients without intraprocedural CT imaging (n = 51), combined embolization/ablation (n = 6) and those with chronic kidney disease (n = 21) were excluded. The study group comprised of 84 patients with 106 procedures. CIN was defined as 25% increase above baseline serum creatinine or absolute increase ≥ 0.5 mg/dL within 72 h post-embolization. Post-embolization CT was reviewed for renal enhancement patterns and presence of renal artery calcifications. The association between non-contrast CT findings and CIN development was examined by Fisher’s Exact Test.
CIN occurred in 11/106 (10.3%) procedures (Group A, n = 10). The renal enhancement pattern in patients who did not experience CIN (Group B, n = 74 with 95/106 procedures) was late excretory in 93/95 (98%) and early excretory (EE) in 2/95 (2%). However, in Group A, there was a significantly higher rate of EE pattern (6/11, 55%) compared to late excretory pattern (5/11) (P < 0.001). A significantly higher percentage of patients that developed CIN had renal artery calcifications (6/11 vs 20/95, 55% vs 21%, P = 0.02).
A hyperdense renal parenchyma relative to surrounding skeletal muscle (EE pattern) and presence of renal artery calcifications on immediate post-HAE non-contrast CT images in patients with low risk for CIN are independently associated with CIN development.
Core Tip: Contrast-induced nephropathy (CIN) is a reversible form of acute kidney injury that occurs within 48-72 h of exposure to intravascular contrast material. CIN is the third leading cause of hospital-acquired acute kidney injury and accounts for 12% of such cases. Identification of early indicators of CIN development is important in taking timely management strategies. There is no known mechanism for early identification and timely initiation of preventive measures in patients otherwise considered low risk for development of CIN after transarterial hepatic artery embolization. This work helps clinicians identify patients at risk of CIN at the time of hepatic artery embolization.
- Citation: Soliman MM, Sarkar D, Glezerman I, Maybody M. Findings on intraprocedural non-contrast computed tomographic imaging following hepatic artery embolization are associated with development of contrast-induced nephropathy . World J Nephrol 2020; 9(2): 33-42
- URL: https://www.wjgnet.com/2220-6124/full/v9/i2/33.htm
- DOI: https://dx.doi.org/10.5527/wjn.v9.i2.33
Contrast-induced nephropathy (CIN) is a reversible form of acute kidney injury (AKI) that occurs within 48-72 h of exposure to intravascular contrast material[1]. The most widely accepted definition of CIN is a 25% increase in serum creatinine (sCr) concentration from baseline sCr, or an absolute increase of at least 0.5 mg/dL which appears within 48-72 h after contrast media exposure, is maintained for 3-5 d and returns to baseline over 7-10 d, after excluding other causes[2,3].
CIN is the third leading cause of hospital-acquired AKI and accounts for 12% of such cases[4]. Risk factors for CIN development can be divided into patient- and procedure-related. The former includes pre-existing chronic renal insufficiency and diabetes mellitus. The latter includes high contrast volume and repeated exposure over 72 h[4-7]. The incidence of CIN is relatively low (up to 5%) in patients with intact renal function[8]. However, in patients with known chronic renal insufficiency, the incidence can reach up to 27%[8]. CIN more commonly develops following intraarterial contrast exposure (e.g., diagnostic arteriograms, coronary interventions, transarterial therapies) than with peripheral intravenous administration (e.g., computed tomography)[9,10]. Although the acute decline of renal function in CIN is reversible within 1-2 wk, a higher 1-mo and 1-year mortality rates have been reported in those patients, particularly following arterial angiography[11,12].
Identification of early indicators of CIN development is important in taking timely management strategies. The elevation in sCr occurs 2-3 d following contrast exposure, and is a poorly-timed indicator[13,14]. Persistent renal enhancement after intravenous or intraarterial contrast administration on non-contrast enhanced computed tomographic (CT) images obtained within 7 d post-contrast exposure has been associated with development of CIN[15]. There is no known mechanism for early identification and timely initiation of preventive measures in patients otherwise considered low risk for development of CIN after transarterial hepatic artery embolization[16-18].
When available, a non-contrast CT or cone beam CT imaging of upper abdomen is routinely performed at the end of hepatic artery embolization (HAE) to assess contrast retention pattern in embolized tumors as a surrogate for treatment response[19-21]. This volume of imaging also covers the kidneys. The aim of this study is to identify early predictors for CIN by examining the appearance of kidneys on non-contrast intraprocedural CT images.
The local Institutional Review Board approved this retrospective review (Protocol 16-402) of all patients who underwent HAE between January 2010 and January 2011. Informed consent from individual patients for the retrospective review was waived.
For the hepatic artery embolization procedure for treatment of primary or secondary hepatic malignancies, all patients were chosen based on multidisciplinary consensus and were seen in a dedicated outpatient interventional radiology clinic, where history, laboratory tests and imaging were reviewed. Eligibility for transarterial HAE was based on clinical status and laboratory test results (Child-Pugh A or B, Total serum bilirubin < 2 mg/dL, Eastern Coorpative Oncology Group performance status < 3). The type of embolization (bland vs chemoembolization) was based on multidisciplinary consensus. Informed consent was obtained for HAE from all patients prior to the procedure.
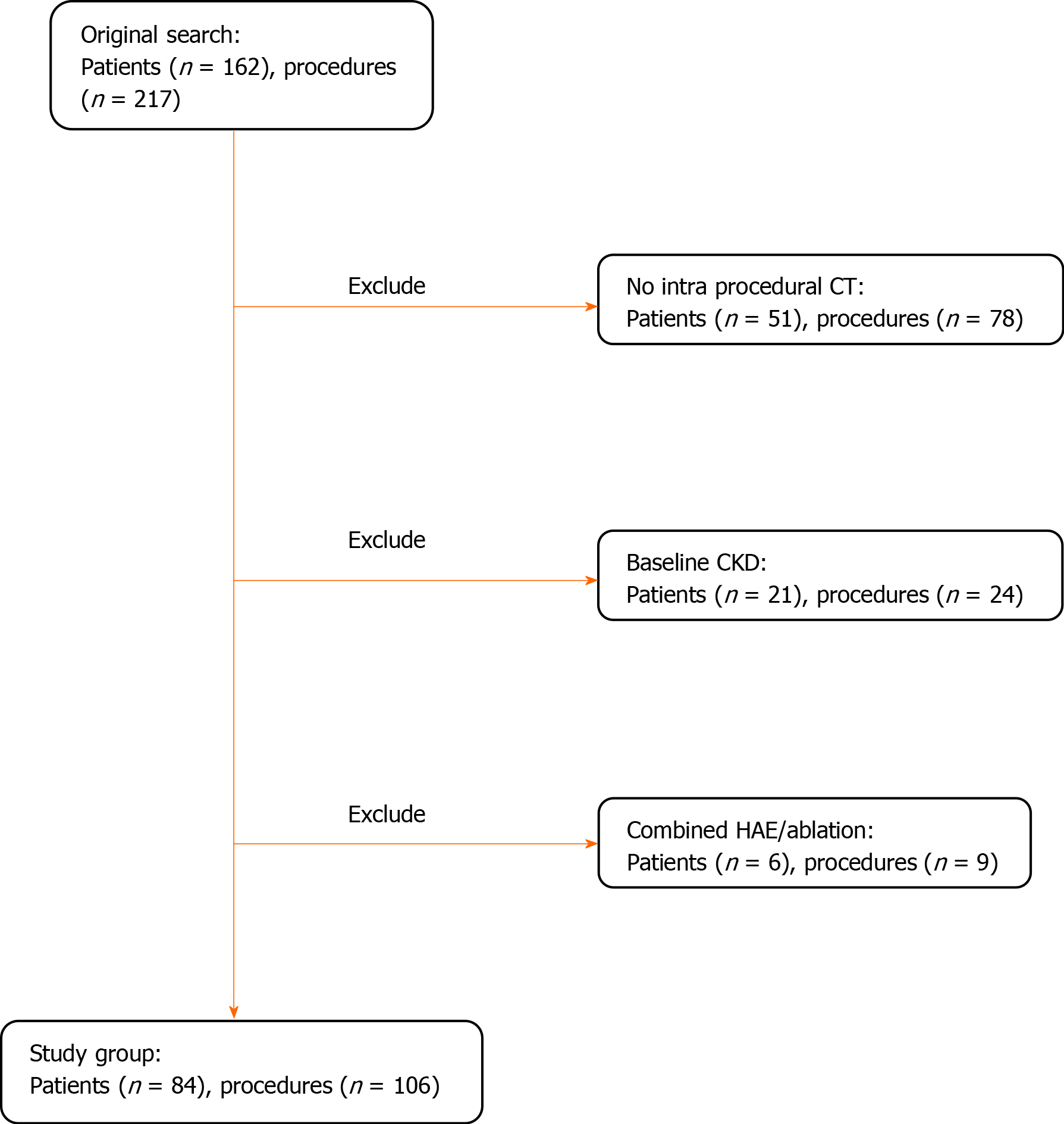
All patients who underwent HAE for treatment of primary or secondary hepatic malignancies within the defined time period were included in the study (n = 162). A total of 217 procedures were performed. Based on the procedure suite, post-embolization non-contrast CT images were available in 139/217 (64%) procedures. Patients with known chronic kidney disease (CKD) stage 3 or higher [GFR < 60 mL/min/1.72 m2 using the CKD-EPI formula] were excluded (n = 21) since they are typically admitted for IV fluid hydration prior to the procedure and are at high risk for CIN. Patients who underwent percutaneous ablation in combination to embolization were excluded due to delay in post embolization non-contrast CT acquisition (n = 6). A total of 84 patients (106 procedures) were included in the final analysis (Figure 1).
Patient electronic records were reviewed for age, gender, ethnicity, type of liver malignancy, presence and etiology of cirrhosis, presence of medical comorbidities related to chronic kidney disease (diabetes mellitus, hypertension, coronary artery disease, prior history of contrast allergies, and number of embolization procedures performed. Laboratory test results were reviewed for sCr prior to embolization, and post-embolization peak sCr in the first 72 h following embolization. Procedural details including type of embolization (bland vs chemoembolization), total contrast volume administered, and total procedure duration (defined by total sedation or anesthesia time) were recorded. CIN was defined as 25% increase in sCr above baseline or an absolute increase of ≥ 0.5 mg/dL in sCr within 72 h of embolization. Patients were divided into two groups based on the change in post-embolization sCr: Group A developed CIN and Group B did not develop CIN. Immediate post-embolization non-contrast CT images was reviewed for renal enhancement pattern and presence of renal artery calcifications. All data was compiled in a Health Insurance Portability and Accountability Act-compliant database.
Patient demographics are recorded in Table 1. A total of 84 patients (53 males, 31 females; mean age 63 years, range: 29-87 years) with 106 procedures were included in the final analysis.
| Clinical characteristics | Value |
| Sex | |
| Male | 53 |
| Female | 31 |
| Age (Yr, average, range) | 63 (29-87) |
| Ethnicity | |
| Caucasian | 62 |
| African American | 8 |
| Hispanic | 4 |
| Asian | 10 |
| Medical comorbidities | |
| Diabetes mellitus | 17 |
| CAD/HTN | 29 |
| Cirrhotic? | |
| Yes | 38 |
| No | 46 |
| Etiology of cirrhosis | |
| HCV | 16 |
| HBV | 6 |
| EtOH | 4 |
| NASH | 3 |
| Hemochromatosis | 1 |
| Unknown | 8 |
| Type of liver malignancy | |
| Primary | |
| HCC | 40 |
| Other | 5 |
| Secondary | |
| NET | 20 |
| GIST | 4 |
| Other | 15 |
| Number of treatments (n = 106) | |
| 1 | 66 |
| 2 | 14 |
| 3 | 4 |
Pre-procedural imaging was reviewed prior to the procedure. General endotracheal anesthesia (n = 85) or conscious sedation (n = 21) were used. Patients were prepped and draped in a standard sterile fashion. Following ultrasound (US)-guided common femoral artery access and utilizing Seldinger technique, a 5 French guide catheter was advanced through a 6 French vascular sheath into the origin of the celiac axis or superior mesenteric artery. Angiograms were obtained using Iohexol (Omnipaque 300; GE Healthcare, Marlborough, MA, United States). A 2.4-2.8 French microcatheter was advanced over a micro guidewire co-axially through the guide catheter for superselective catherization of target branches based on angiographic findings and predetermined area of treatment. Treatment was done with either Embospheres (Merit Medical, South Jordan, UT, United States), Embozene (Boston Scientific, Burlington, MA, United States) and Poly-Vinyl Alcohol particles under continuous angiographic monitoring until complete arterial stasis was achieved. Hemostasis was obtained with a femoral closure device. All patients underwent bland HAE with an average procedural duration of 111.8 min, and average total administered contrast volume of 174.3 mL (average, median, range; Group A: 155, 152.5, 50-320 mL. Group B: 161, 150, 58-350 mL). Post-embolization non-contrast CT was obtained immediately after achieving hemostasis (10-20 min after embolization completion) in all study patients. Patients were transferred to the post-anesthesia care unit and were subsequently admitted for symptomatic management of post-embolization syndrome.
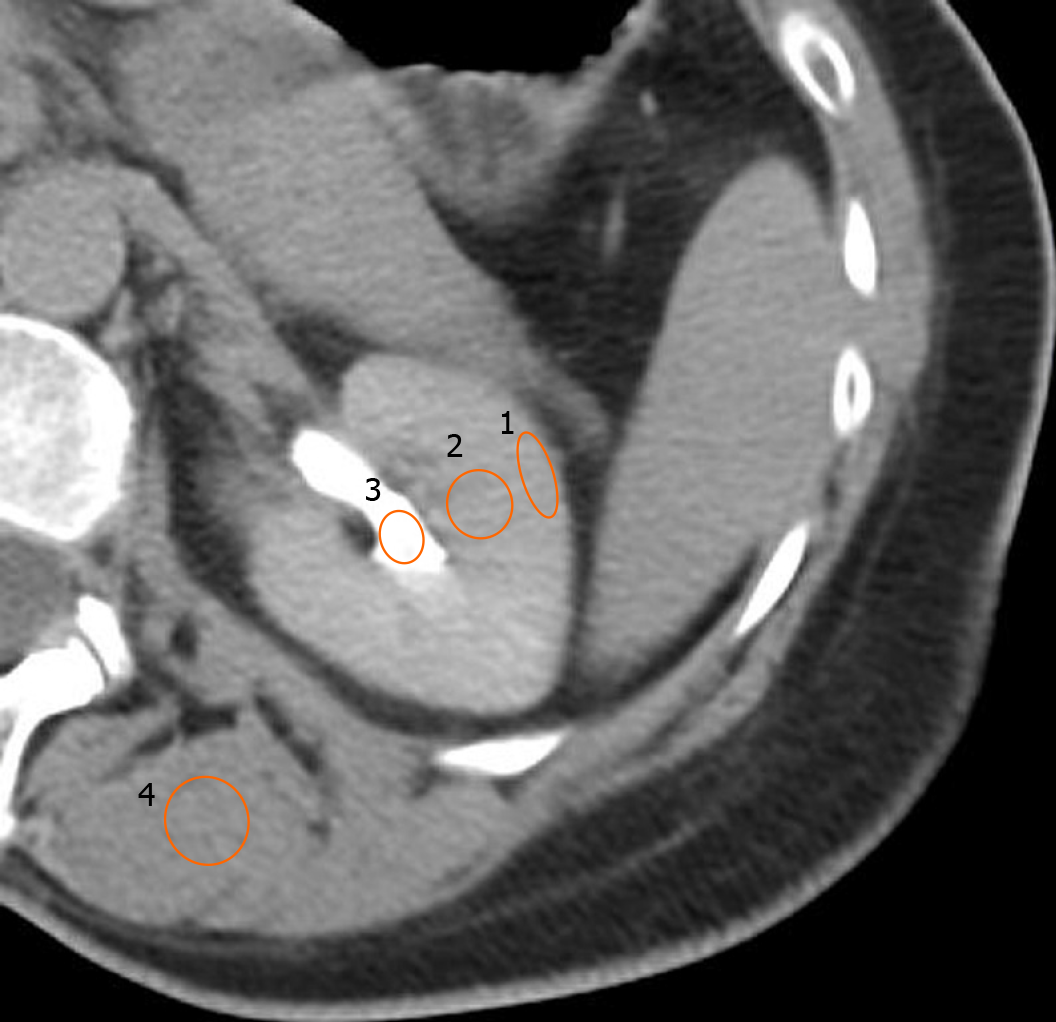
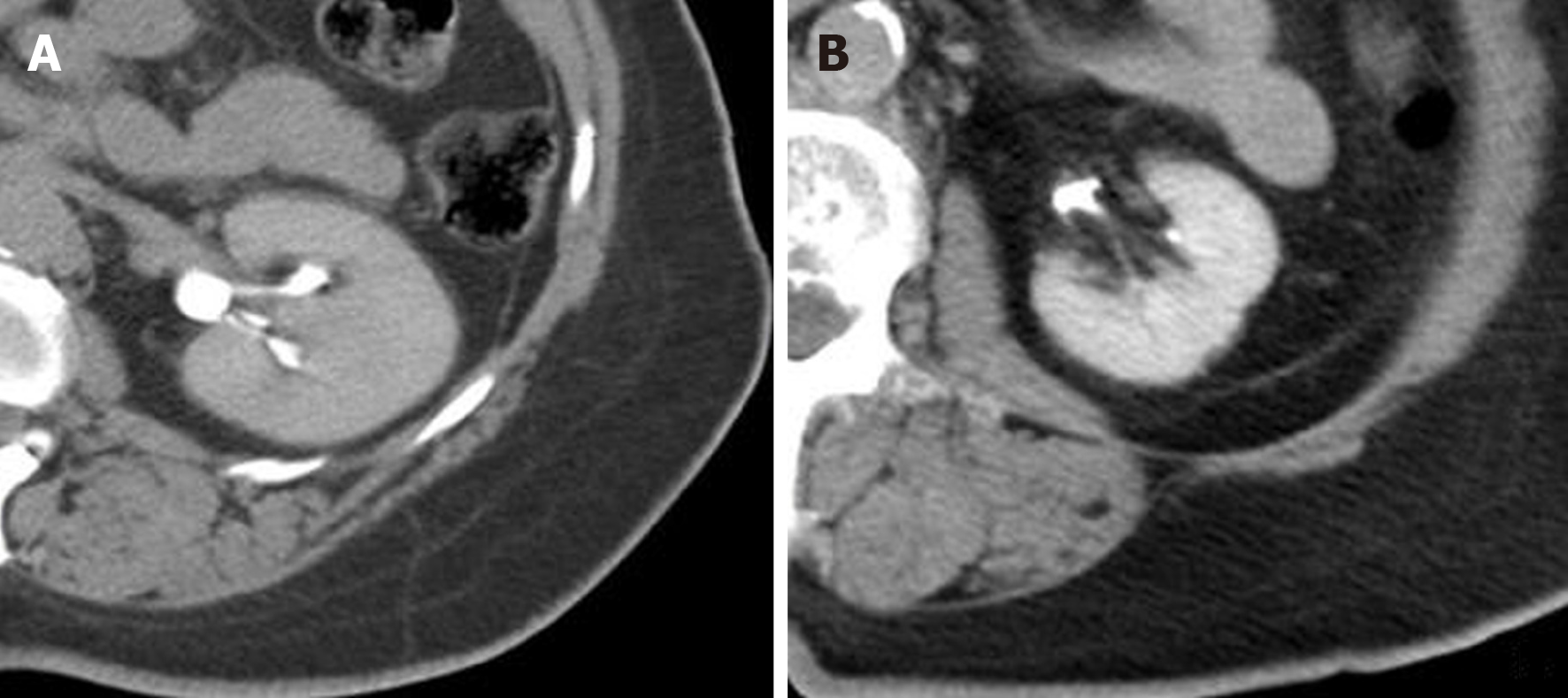
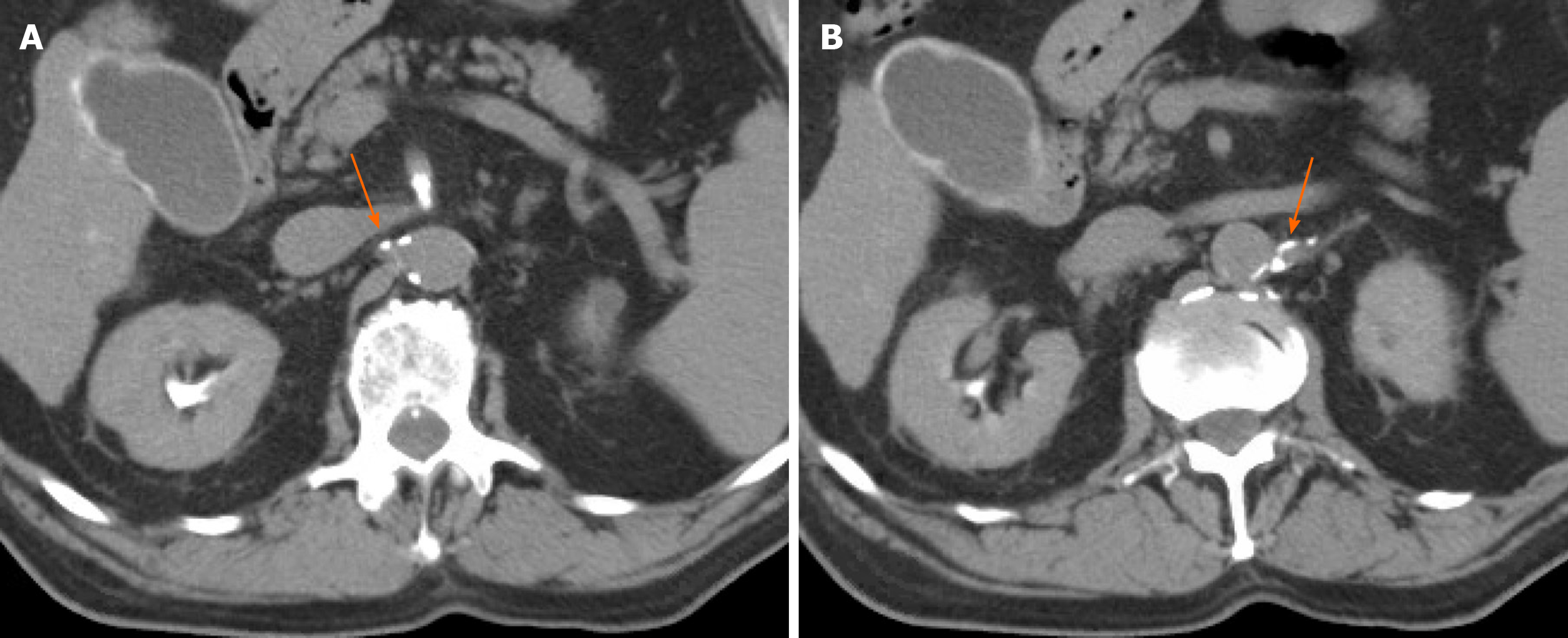
The assessment of renal enhancement pattern and presence of renal artery calcifications was performed by two independent readers. In the case of disagreement, a third reader was involved to assess the CT findings. Regions-of-interest were drawn on the renal cortex, medulla and pelvis to accurately determine the phase or pattern of renal enhancement and categorize them based on predetermined thresholds as either: Corticomedullary, nephrographic, early excretory (EE) or late excretory (LE) (Figures 2 and 3)[22,23]. Corticomedullary pattern occurs 25-80 s after contrast injection and is defined by intense enhancement of the renal cortex relative to the renal medulla, with a difference in Hounsfield units (HU) approaching 100 HU. Nephrographic pattern occurs 85-120 s after contrast injection and is defined by homogenous enhancement of the renal parenchyma (cortex and medulla) with HU range of 120-170. The excretory patterns occur 3-5 min after contrast injection. EE pattern is defined by the opacification of the renal calyces/pelvis, with hyperdense renal parenchyma relative to surrounding skeletal muscle. LE pattern is defined by a renal parenchyma iso-dense to skeletal muscle, with contrast fully opacifying the renal pelvis and ureter. Due to timing of post HAE intraprocedural CT imaging (10-20 min after completion of embolization), the corticomedullary and nephrographic patterns were not seen in our patient cohort. Renal enhancement pattern was divided into only 2 later patterns of EE or LE. Any discernible plaque along the renal arteries with more than 130 Hounsfield units were considered calcifications (Figure 4).
Descriptive statistics (mean, standard deviation) were used to describe the patient demographics and procedural details. Continuous data are reported as the mean value ± SD. A Mann-Whitney U Test was performed to examine difference in baseline serum creatinine, and total contrast volume and procedure duration between the two patient groups. A Fisher’s Exact Test was utilized to compare the prevalence of diabetes mellitus amongst the two patient groups. The association between renal enhancement pattern, presence of renal artery calcifications and the development of CIN (Group A vs B) were analyzed by Chi-Square test of Independence. P value 0.05 was considered to indicate statistical significance. All analyses were performed with SPSS statistical software version 12.0 (SPSS, Inc., Chicago, IL, United States).
There was no significant difference between the baseline sCr (P = 0.46), total contrast volume administered (P = 0.61), total procedure duration (P = 0.25), or prevalence of diabetes mellitus (P = 0.38) amongst the two groups.
The renal enhancement patterns are recorded in Table 2. In patients who did not experience CIN (Group B, n = 74 with 95 procedures) the renal enhancement pattern was late excretory in 93/95 procedures (98%) and early excretory in 2/95 procedures (2%). CIN occurred following 11/106 (11.2%) procedures in 10 distinct patients (Group A). In this group, there was a significantly higher rate of early excretory renal enhancement (6/11 procedures, 55%) compared to late excretory pattern (5/11 procedures, 45%) (P < 0.001). Renal artery calcifications were detected in 26 post-embolization non-contrast CT scans. A significantly higher percentage of patients that developed CIN had detectable renal artery calcifications (6/11 vs 20/95, 55% vs 21% respectively, P = 0.01).
| Association between non-contrast CT findings and CIN (n = 106) | |||
| Renal enhancement pattern | Group A (n = 11) (%) | Group B (n = 95) (%) | P value |
| Early excretory | 6 (55%) | 2 (2%) | < 0.001 |
| Late excretory | 5 (45%) | 93 (98%) | |
| Renal artery calcifications | Group A | Group B | |
| Yes | 6 (55%) | 20 (21%) | 0.01 |
| No | 5 (45%) | 75 (79%) | |
Patients in Group A were hospitalized for a median of 4 d post-embolization (IQR: 3-10 d). Peak sCr was reached in a median of 2 d post-embolization (IQR: 1-2.5 d). Those who stayed in the hospital for 2 d or longer (n = 9) demonstrated complete normalization of sCr prior to discharge. No patient required hemodialysis or hemofiltration. One patient required a nephrology consultation due to oliguria despite Foley catheter placement.
The findings of this study suggest an association between the renal enhancement pattern on the immediate post-HAE non-contrast enhanced CT imaging and the development of CIN in low-risk patients. It also suggests an association between renal artery calcifications and development of CIN.
The cornerstone of CIN management is prevention. Risk stratification, IV fluid administration and avoidance of nephrotoxic medications are all essential in prevention[24]. Randomized clinical trials have consistently shown the efficacy of IV fluid hydration in the prevention of CIN in high-risk patient populations[25-28]. All investigated fluid regimens included a pre-procedural component and a post-procedural component (e.g., IV 0.9% normal saline 12 h pre-HAE and 12- h post-HAE). The consensus is that both components contribute to volume expansion and prevention of CIN development[24]. No head-to-head study was performed investigating the efficacy of pre-procedural hydration compared to post-procedural hydration.
High-risk patients are followed by nephrologists in the outpatient setting, are typically admitted the day before an interventional procedure for IV fluid administration and are followed by nephrologists during their admission and after discharge. Conversely, low-risk patients typically arrive on the day of the procedure and no special renal protective precautions are taken.
Currently there is no mechanism for early identification and timely initiation of preventive measures for patients who do not have risk factors for development of CIN after transarterial hepatic artery embolization[16-18]. In this study, we propose that risk stratification could be initiated within the interventional radiology procedure room. The proposed method is simple with no need for any calculations or image processing. If in a patient low risk for development of CIN, the renal parenchyma is hyperdense compared to adjacent skeletal muscle at the end of hepatic artery embolization, preventive measures should be initiated immediately. In this group of low-risk patients with no history of CKD, CIN or contrast-allergies, an early excretory pattern on the post-embolization CT imaging was associated with the development of CIN. In addition, the presence of renal artery calcifications was found to be independently associated with CIN development. This is in agreement with prior studies showing the association of renal artery calcification with renal function[29,30]. Renal artery calcification can be identified prior to embolization intervention by reviewing CT scan of abdomen. These patients should be treated like those at risk for CIN with pre and post procedural hydration. For those patients stratified based on immediate post procedural CT imaging renal enhancement patterns, a nephrology consultation should be initiated, along with rapid initiation of IV fluid hydration to prevent CIN development. These patients should also be marked for preventive measures for their future angiographic interventions.
This study has several limitations. Inherent to its retrospective design and small sample size, extensive multivariable logistic regression and analysis of potential confounders are limited. The method used to determine renal enhancement patterns is not free of subjectivity despite utilization of multiple readers. Furthermore, the exact timing of non-contrast CT acquisition was variable and could be a potential confounder.
Larger, prospective studies should be conducted with standardized post-HAE CT acquisition timing to investigate this association.
Hyperdense renal parenchyma relative to surrounding skeletal muscle (Early excretory renal enhancement pattern) on immediate post HAE non-contrast CT images and also the presence of renal artery calcification are associated with the development of CIN.
Hyperdense renal parenchyma relative to surrounding skeletal muscle (Early excretory renal enhancement pattern) on immediate post HAE non-contrast CT images and also the presence of renal artery calcification are associated with the development of CIN.
Contrast-induced nephropathy (CIN) is a reversible form of acute kidney injury that occurs within 48-72 h of exposure to intravascular contrast material. A higher 1-mo and 1-year mortality rates have been reported in these patients, particularly following arterial angiography.
The cornerstone of CIN management is prevention.
To help with early identification and timely initiation of preventive measures in patients otherwise considered low risk for development of CIN after transarterial hepatic artery embolization.
Retrospective review of all patients who underwent hepatic artery embolization between 2010 and 2011 (n = 162) was performed. After removing exclusions, the study group comprised of 84 patients with 106 procedures. CIN was defined as 25% increase above baseline serum creatinine or absolute increase ≥ 0.5 mg/dL within 72 h post-embolization. Post-embolization computed tomographic (CT) was reviewed for renal enhancement patterns and presence of renal artery calcifications. The association between non-contrast CT findings and CIN development was examined by Fisher’s Exact Test.
CIN occurred in 11/106 (10.3%) procedures (Group A, n = 10). The renal enhancement pattern in patients who did not experience CIN (Group B, n = 74 with 95/106 procedures) was late excretory in 93/95 (98%) and early excretory (EE) in 2/95 (2%). However, in Group A, there was a significantly higher rate of EE pattern (6/11, 55%) compared to late excretory pattern (5/11) (P < 0.001). A significantly higher percentage of patients that developed CIN had renal artery calcifications (6/11 vs 20/95, 55% vs 21%, P = 0.02).
A hyperdense renal parenchyma relative to surrounding skeletal muscle (EE pattern) and presence of renal artery calcifications on immediate post-HAE non-contrast CT images in patients with low risk for CIN are independently associated with CIN development.
Prospective studies are required to further assess the findings of this study.
Manuscript source: Unsolicited manuscript
Specialty type: Urology and nephrology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: El-Serafy AS S-Editor: Zhang L L-Editor: A P-Editor: Wang LL
| 1. | Parfrey PS, Griffiths SM, Barrett BJ, Paul MD, Genge M, Withers J, Farid N, McManamon PJ. Contrast material-induced renal failure in patients with diabetes mellitus, renal insufficiency, or both. A prospective controlled study. N Engl J Med. 1989;320:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 726] [Cited by in RCA: 661] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 2. | Goldenberg I, Matetzky S. Nephropathy induced by contrast media: pathogenesis, risk factors and preventive strategies. CMAJ. 2005;172:1461-1471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 226] [Cited by in RCA: 240] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 3. | Gleeson TG, Bulugahapitiya S. Contrast-induced nephropathy. AJR Am J Roentgenol. 2004;183:1673-1689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 286] [Cited by in RCA: 278] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 4. | Hou SH, Bushinsky DA, Wish JB, Cohen JJ, Harrington JT. Hospital-acquired renal insufficiency: a prospective study. Am J Med. 1983;74:243-248. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 869] [Cited by in RCA: 777] [Article Influence: 18.5] [Reference Citation Analysis (0)] |
| 5. | Mehran R, Nikolsky E. Contrast-induced nephropathy: definition, epidemiology, and patients at risk. Kidney Int Suppl. 2006;S11-S15. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 494] [Cited by in RCA: 574] [Article Influence: 30.2] [Reference Citation Analysis (0)] |
| 6. | McCullough PA, Adam A, Becker CR, Davidson C, Lameire N, Stacul F, Tumlin J; CIN Consensus Working Panel. Risk prediction of contrast-induced nephropathy. Am J Cardiol. 2006;98:27K-36K. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 285] [Cited by in RCA: 285] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 7. | Mehran R, Aymong ED, Nikolsky E, Lasic Z, Iakovou I, Fahy M, Mintz GS, Lansky AJ, Moses JW, Stone GW, Leon MB, Dangas G. A simple risk score for prediction of contrast-induced nephropathy after percutaneous coronary intervention: development and initial validation. J Am Coll Cardiol. 2004;44:1393-1399. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 246] [Cited by in RCA: 690] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 8. | Morcos SK, Thomsen HS, Webb JA. Contrast-media-induced nephrotoxicity: a consensus report. Contrast Media Safety Committee, European Society of Urogenital Radiology (ESUR). Eur Radiol. 1999;9:1602-1613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 363] [Cited by in RCA: 359] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 9. | Kooiman J, Pasha SM, Zondag W, Sijpkens YW, van der Molen AJ, Huisman MV, Dekkers OM. Meta-analysis: serum creatinine changes following contrast enhanced CT imaging. Eur J Radiol. 2012;81:2554-2561. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 91] [Cited by in RCA: 101] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 10. | Karlsberg RP, Dohad SY, Sheng R; Iodixanol Peripheral CTA Study Investigator Panel. Contrast-induced acute kidney injury (CI-AKI) following intra-arterial administration of iodinated contrast media. J Nephrol. 2010;23:658-666. [PubMed] |
| 11. | Coca SG, Peixoto AJ, Garg AX, Krumholz HM, Parikh CR. The prognostic importance of a small acute decrement in kidney function in hospitalized patients: a systematic review and meta-analysis. Am J Kidney Dis. 2007;50:712-720. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 159] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 12. | Lindsay J, Canos DA, Apple S, Pinnow E, Aggrey GK, Pichard AD. Causes of acute renal dysfunction after percutaneous coronary intervention and comparison of late mortality rates with postprocedure rise of creatine kinase-MB vs rise of serum creatinine. Am J Cardiol. 2004;94:786-789. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 13. | Tublin ME, Murphy ME, Tessler FN. Current concepts in contrast media-induced nephropathy. AJR Am J Roentgenol. 1998;171:933-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 56] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 14. | Mohammed NM, Mahfouz A, Achkar K, Rafie IM, Hajar R. Contrast-induced Nephropathy. Heart Views. 2013;14:106-116. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 127] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 15. | Chou SH, Wang ZJ, Kuo J, Cabarrus M, Fu Y, Aslam R, Yee J, Zimmet JM, Shunk K, Elicker B, Yeh BM. Persistent renal enhancement after intra-arterial vs intravenous iodixanol administration. Eur J Radiol. 2011;80:378-386. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 16. | Huo TI, Wu JC, Lee PC, Chang FY, Lee SD. Incidence and risk factors for acute renal failure in patients with hepatocellular carcinoma undergoing transarterial chemoembolization: a prospective study. Liver Int. 2004;24:210-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 54] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 17. | Cho HS, Seo JW, Kang Y, Bae EJ, Kim HJ, Chang SH, Park DJ. Incidence and risk factors for radiocontrast-induced nephropathy in patients with hepatocellular carcinoma undergoing transcatheter arterial chemoembolization. Clin Exp Nephrol. 2011;15:714-719. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 18. | Hayakawa K, Tanikake M, Kirishima T, Yoshinami N, Shintani H, Yamamoto E, Morimoto T. The incidence of contrast-induced nephropathy (CIN) following transarterial chemoembolisation (TACE) in patients with hepatocellular carcinoma (HCC). Eur Radiol. 2014;24:1105-1111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 19. | Wang X, Erinjeri JP, Jia X, Gonen M, Brown KT, Sofocleous CT, Getrajdman GI, Brody LA, Thornton RH, Maybody M, Covey AM, Siegelbaum RH, Alago W, Solomon SB. Pattern of retained contrast on immediate postprocedure computed tomography (CT) after particle embolization of liver tumors predicts subsequent treatment response. Cardiovasc Intervent Radiol. 2013;36:1030-1038. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 20. | Herber S, Biesterfeld S, Franz U, Schneider J, Thies J, Schuchmann M, Düber C, Pitton MB, Otto G. Correlation of multislice CT and histomorphology in HCC following TACE: predictors of outcome. Cardiovasc Intervent Radiol. 2008;31:768-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | Hu J, Maybody M, Cao G, Wang X, Chen H, Zhu X, Yang R, Wang X. Lipiodol retention pattern assessed by cone beam computed tomography during conventional transarterial chemoembolization of hepatocellular carcinoma: accuracy and correlation with response. Cancer Imaging. 2016;16:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 22. | Yuh BI, Cohan RH. Different phases of renal enhancement: role in detecting and characterizing renal masses during helical CT. AJR Am J Roentgenol. 1999;173:747-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 92] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 23. | Sheth S, Fishman EK. Multi-detector row CT of the kidneys and urinary tract: techniques and applications in the diagnosis of benign diseases. Radiographics. 2004;24:e20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 30] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 24. | Levey AS, Stevens LA, Schmid CH, Zhang YL, Castro AF 3rd, Feldman HI, Kusek JW, Eggers P, Van Lente F, Greene T, Coresh J; CKD-EPI (Chronic Kidney Disease Epidemiology Collaboration). A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150:604-612. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20319] [Cited by in RCA: 20160] [Article Influence: 1260.0] [Reference Citation Analysis (0)] |
| 25. | Gupta RK, Bang TJ. Prevention of Contrast-Induced Nephropathy (CIN) in Interventional Radiology Practice. Semin Intervent Radiol. 2010;27:348-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (1)] |
| 26. | Briguori C, Visconti G, Focaccio A, Airoldi F, Valgimigli M, Sangiorgi GM, Golia B, Ricciardelli B, Condorelli G; REMEDIAL II Investigators. Renal Insufficiency After Contrast Media Administration Trial II (REMEDIAL II): RenalGuard System in high-risk patients for contrast-induced acute kidney injury. Circulation. 2011;124:1260-1269. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 172] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 27. | Solomon R, Werner C, Mann D, D'Elia J, Silva P. Effects of saline, mannitol, and furosemide on acute decreases in renal function induced by radiocontrast agents. N Engl J Med. 1994;331:1416-1420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 909] [Cited by in RCA: 763] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 28. | Mueller C, Buerkle G, Buettner HJ, Petersen J, Perruchoud AP, Eriksson U, Marsch S, Roskamm H. Prevention of contrast media-associated nephropathy: randomized comparison of 2 hydration regimens in 1620 patients undergoing coronary angioplasty. Arch Intern Med. 2002;162:329-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 582] [Cited by in RCA: 504] [Article Influence: 21.9] [Reference Citation Analysis (0)] |
| 29. | Vashishtha D, McClelland RL, Ix JH, Rifkin DE, Jenny N, Allison M. Relation Between Calcified Atherosclerosis in the Renal Arteries and Kidney Function (from the Multi-Ethnic Study of Atherosclerosis). Am J Cardiol. 2017;120:1434-1439. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Roseman DA, Hwang SJ, Manders ES, O'Donnell CJ, Upadhyay A, Hoffmann U, Fox CS. Renal artery calcium, cardiovascular risk factors, and indexes of renal function. Am J Cardiol. 2014;113:156-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |