Published online Nov 19, 2023. doi: 10.5498/wjp.v13.i11.967
Peer-review started: July 25, 2023
First decision: August 24, 2023
Revised: September 6, 2023
Accepted: September 27, 2023
Article in press: September 27, 2023
Published online: November 19, 2023
Processing time: 114 Days and 21.3 Hours
Cerebrotendinous xanthomatosis (CTX) is a rare autosomal recessive lipid-storage disorder caused by mutations in CYP27A1. Psychiatric manifestations in CTX are rare and nonspecific, and they often lead to considerable diagnostic and treatment delay.
A 33-year-old female patient admitted to the psychiatric ward for presentation of delusions, hallucinations, and behavioral disturbance is reported. The patient presented with cholestasis, cataract, Achilles tendon xanthoma, and cerebellar signs in adulthood and with intellectual disability and learning difficulties in childhood. After the characteristic CTX findings on imaging were obtained, a pathological examination of the Achilles tendon xanthoma was refined. Re-placement therapy was then initiated after the diagnosis was clarified by genetic analysis. During hospitalization in the psychiatric ward, the nonspecific psychiatric manifestations of the patient posed difficulty in diagnosis. After the patient’s history of CTX was identified, the patient was diagnosed with organic schizophrenia-like disorder, and psychotic symptoms were controlled by replacement therapy combined with antipsychotic medication.
Psychiatrists should be aware of CTX, its psychiatric manifestations, and clinical features and avoid misdiagnosis of CTX for timely intervention.
Core Tip: Cerebrotendinous xanthomatosis (CTX) is a rare autosomal recessive lipid-storage disorder, characterized by diverse systemic and neuropsychiatric manifestations. Psychiatric manifestations in CTX are typically nonspecific, and past cases have reported a scarcity of psychotic symptoms, particularly delusions and hallucinations. In this report, we present a case of CTX exhibiting a hallucinatory-paranoid syndrome and describe the diagnostic and therapeutic processes associated with CTX. The long-term treatment effect in CTX may depend on age at start of treatment and psychiatrists should be cognizant of CTX to facilitate timely intervention.
- Citation: Ling CX, Gao SZ, Li RD, Gao SQ, Zhou Y, Xu XJ. Cerebrotendinous xanthomatosis presenting with schizophrenia-like disorder: A case report. World J Psychiatry 2023; 13(11): 967-972
- URL: https://www.wjgnet.com/2220-3206/full/v13/i11/967.htm
- DOI: https://dx.doi.org/10.5498/wjp.v13.i11.967
Cerebrotendinous xanthomatosis (CTX) is a rare autosomal recessive inherited metabolic lipid-storage disorder related to bile acid biosynthesis pathways. It is caused by biallelic pathogenic variants in CYP27A1 (2q35), which codes sterol 27-hydroxylase, a mitochondrial enzyme of the cytochrome P450 oxidase system. Reduction in the activity of this enzyme leads to an increased formation and storage of abnormal lipid content in several tissues, especially tendons, lenses, and the peripheral and central nervous systems. Typically, the affected individuals develop tendon xanthomas, cataracts, dementia, pyramidal paresis, cerebellar ataxia, and peripheral neuropathy. As epidemiologic data are limited and CTX may be substantially underdiagnosed, the prevalence of the disease is underestimated. No consensus exists on the prevalence of CTX, with an estimated rate of < 3:100000–5:100000 worldwide[1]. A recent worldwide genetic epidemiological study estimated the incidence of CTX to be 1:36042-1:468624 and highest among South Asians and East Asians, followed by North Americans, Europeans, and Africans[2]. The psychiatric manifestations of CTX are variable, with the most common manifestations being intellectual disability, behavioral disorders, and mood disorders. Few patients have delusions, hallucinations, paranoid ideas, or catatonia, similar to the features of schizophrenia. Thus, making a timely diagnosis is difficult for clinicians. Early diagnosis and treatment initiation in patients with CTX is of great importance because significant reversal of the disease progression could be achieved. Consequently, an enhanced understanding of this rare disease is necessary.
A 33-year-old woman was admitted to the psychiatric acute ward with the following symptoms: delusions of persecution, auditory hallucination, impulsive behavior, and emotional instability.
For 6 mo, the patient showed hostility towards her colleagues, firmly believing that they wanted to harm her. She could hear abusive voices of colleagues when she was alone. The false belief and voice interfered with the social relations and behavior of the patient, leading to outrunning behavior. In addition, the patient exhibited impulsive acts and temperamental behavior, such as tearing people's clothes, smashing things, standing outside naked when she became excited, and running around at midnight. She also experienced severe depression for several days, with crying and expressing a desire to commit suicide by euthanasia.
According to her mother, the patient has been weak and obtuse since childhood and was unable to pass primary school exams. She required supervision and care from her family in her personal life. Despite being able to work, she could not perform her job well and did not get along with her colleagues, leading to her dismissal from several companies. At age 18, she underwent cataract surgery, and at age 26, she underwent cholecystectomy for cholestasis.
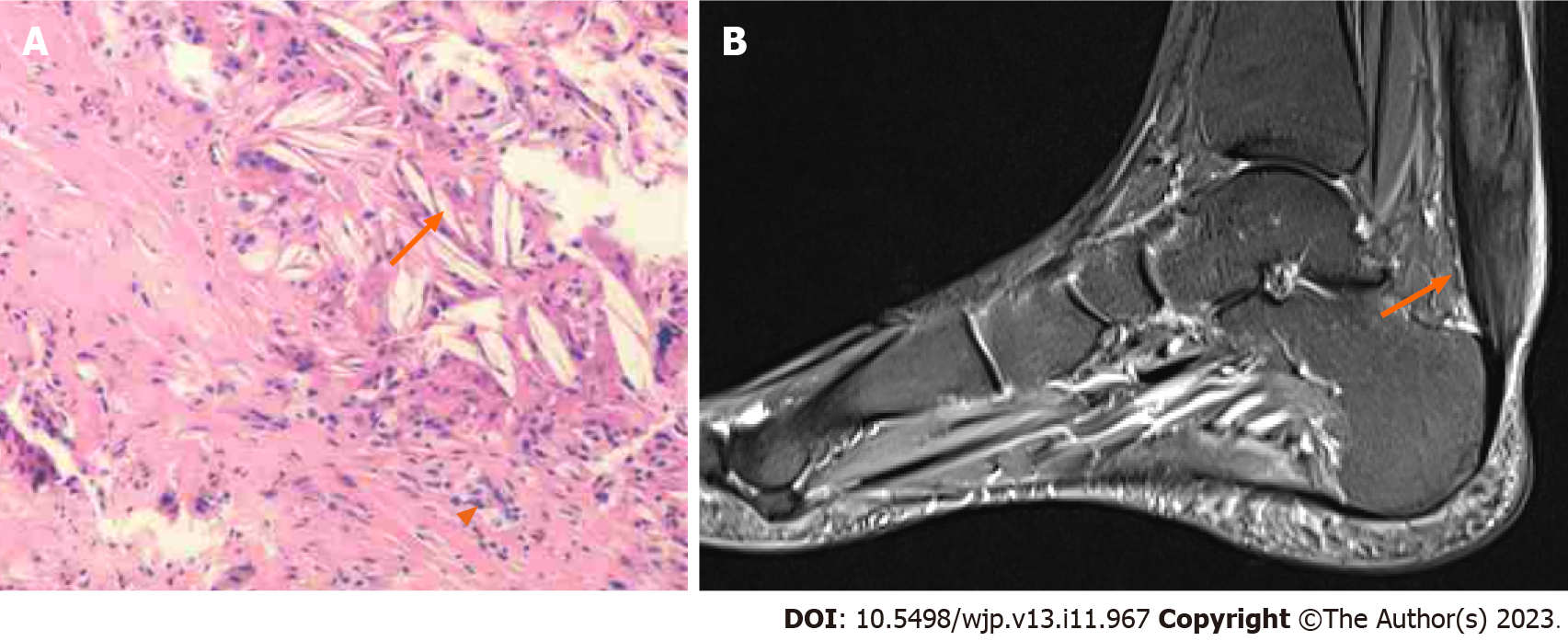
A review of the patient’s medical records revealed that she presented with swelling in her left Achilles tendon, pain, and skewed walking to the right side 5 years ago. Three years ago, she was admitted to the neurology department, where biochemical tests revealed an elevated total biliary acid level of 11.2 µmol/L (normal value ≤ 10.0) and decreased levels of chenodeoxycholic acid and ursodeoxycholic acid. Subsequently, a biopsy of the left Achilles tendon was performed, revealing numerous lipid crystals and a few foamy macrophages (Figure 1A). The patient underwent genetic analysis, which identified two mutations in the CYP27A1 gene associated with CTX: c.1263 + 3G>C and c.379C>T. On the basis of these findings, the patient was ultimately diagnosed with CTX and prescribed chenodeoxycholic acid (750 mg/d) and rosuvastatin (10 mg/d).
There was no family history of psychiatric diseases.
During hospitalization in the psychiatric ward, the patient’s brief psychiatric rating sale (BPRS) score was 40, her Wechsler intelligence scale score was 65, and the total biliary acid returned to normal levels. Nervous system examination showed normal muscle tone in the extremities, grade 5 muscle strength, instability in the heel-knee-tibia test of the right lower extremity, and a positive Romberg sign. In the mental status examination, the patient exhibited the following: verbal auditory hallucination; delusions of persecution; negative perceptions; irritability; childish emotions; poor general knowledge, understanding, judgment, and calculation; impulsivity; and lack of insight.
After nearly 3 years of adequate chenodeoxycholic acid treatment, the total biliary acid level returned to normal.
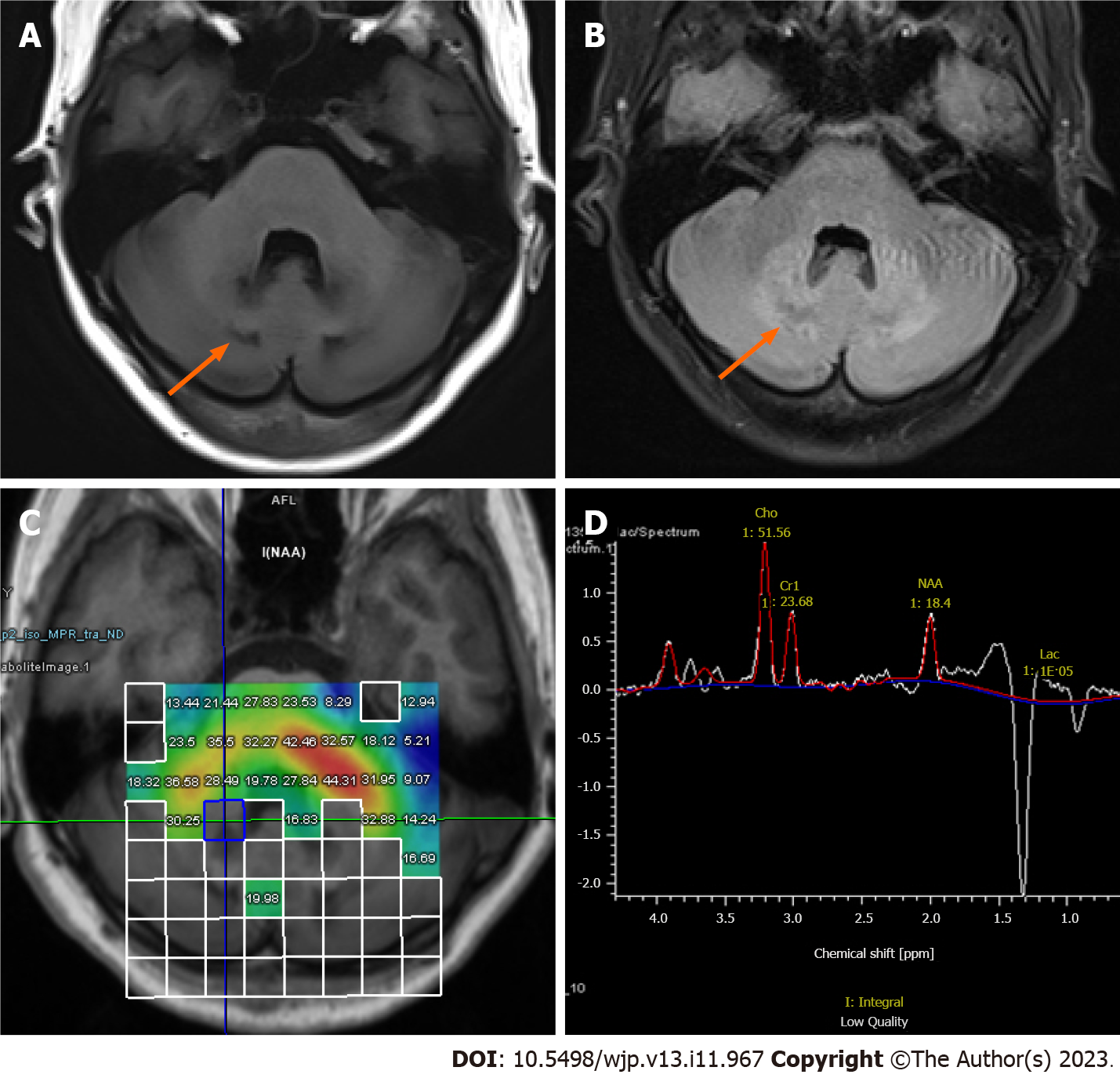
Brain magnetic resonance imaging (MRI) revealed symmetrical patchy abnormal signals in the dentate nuclei and deep medulla of the bilateral cerebellar hemispheres. These signals exhibited a slight decrease in signal intensity on T1-weighted images (T1WI) (Figure 2A) and a slight increase in signal intensity on fluid attenuated inversion recovery (FLAIR) (Figure 2B). Left-ankle-joint MRI showed that the inhomogeneous area had a considerable local thickening of the left Achilles tendon. The area displayed clear boundaries measuring approximately 6.6 cm × 1.2 cm and exhibited a slightly elevated signal intensity on T1WI and a similar slightly elevated signal intensity on the proton density weighted image, accompanied by linear hypointensity within (Figure 1B). Brain magnetic resonance spectroscopy (MRS) showed decreases in N-acetylaspartate (NAA) intensities and increases in lactate and lipid signals (Figure 2C and D).
In accordance with International Classification of Diseases-10 criteria, the patient received the following diagnoses: Organic schizophrenia-like disorder and CTX.
Concurrent with chenodeoxycholic acid treatment for the primary condition, we administered olanzapine to manage the patient's psychotic symptoms, and sodium valproate to stabilize mood while augmenting the antipsychotic medication.
After treatment, the psychotic symptoms were remarkably relieved, her emotions were relatively stable, and impulsive or noisy behavior did not manifest itself. The reassessed BPRS score was 21. During the 3-mo outpatient follow-up, the patient took medication on time and occasionally experienced emotional fluctuations but was able to listen to persuasion and did not exhibit any more impulsive behavior or running.
The clinical presentation of CTX is characterized by diverse systemic and neuropsychiatric manifestations and combinations of symptoms that vary from patient to patient. Of these, psychiatric disturbances are considered a strong diagnostic indicator of CTX, but the symptoms are so variable, as in this case, that making a timely diagnosis is difficult for clinicians. CTX is estimated to have a 15–20-year diagnostic delay[3,4].
The lack of specificity of the early clinical symptoms of CTX is the main reason why the disease is difficult to diagnose in a timely manner, and clinicians must often consider the clinical features of multiple systems, biochemical tests, and radiological examination. Among them, the dentate nucleic signal change in MRI is considered a strong diagnostic indicator[3]. The distinctive neuroradiological findings of CTX show symmetrical abnormalities in the bilateral cerebellar dentate nuclei region with a low signal intensity on T1WI and a high signal intensity on T2 weighted image and FLAIR. The brain MRS shows decreases in NAA intensities and increases in lactate signals[5]. The gold standard for diagnosing CTX is the analysis of the CYP27A1 gene. In the present case, two mutations were confirmed in the CYP27A1 gene by genetic sequencing, with c.379C > G being previously reported as pathogenic and considered one of the most frequent mutations in the Chinese population[6]. However, the relationship between the unreported mutation at the c.1263 + 3G > C locus and the CTX disease phenotype must be further explored.
The psychiatric manifestations of CTX among the Chinese population are infrequently discussed, but foreign literature suggests that intellectual disability, attention deficit hyperactivity disorder, and autism spectrum disorder are often present in childhood or adolescence, with mood and impulsive behavioral symptoms becoming more prominent in adulthood[7]. In the present case, the patient exhibited psychiatric manifestations consistent with those in previous studies. A notable detail that the patient also exhibited hallucinatory-paranoid syndrome, which was scant in past cases and is the first description in Asian population[8]. Psychotic manifestations in CTX are rare, especially in younger patients, and they are often misattributed to psychiatric disease. Most of the psychiatric manifestations of CTX appear after xanthomas and neurological symptoms[7], allowing psychiatrists to identify this organic disorder more easily. Chenodeoxycholic acid is currently the first-line treatment for CTX, and its combination with antipsychotic medication may be beneficial for most patients if psychotic symptoms are present[8]. In the present case, the bile acid level returned to normal after nearly 3 years of adequate chenodeoxycholic acid treatment. The psychiatric symptoms remarkably improved after the combination of the antipsychotic olanzapine and the mood stabilizer sodium valproate during hospitalization. Moreover, the condition was stable at the 3-mo follow-up, indicating that such drugs have good efficacy on psychiatric symptoms associated with CTX.
The long-term treatment effect in CTX may depend on the age at start of treatment. At a point in time demarcation, two different therapeutic effects were observed. A study showed that patients with CTX who started chenodeoxycholic acid treatment before the age of 24 have a better prognosis[9], whereas a follow-up revealed minimal progression in MRI imaging compared with baseline[10].
In psychiatric clinical practice, patients with intellectual disability come to the clinic because of concomitant psychiatric behavioral symptoms, and psychiatric specialists need to pay attention to the differential diagnosis, especially when patients are found to have Achilles tendon xanthomas, juvenile cataracts, childhood-onset chronic diarrhea, and cholestasis. Thus, further improvement of biochemical tests, such as serum cholestanol, radiological examination of the Achilles tendons and brain, and genetic analysis, is needed to avoid a missed or misdiagnosis of CTX for timely intervention.
We would like to thank the patient for her understanding and willingness to participate in this study.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Psychiatry
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kar SK, India; Tom MR, United States S-Editor: Lin C L-Editor: Filipodia P-Editor: Lin C
| 1. | Lorincz MT, Rainier S, Thomas D, Fink JK. Cerebrotendinous xanthomatosis: possible higher prevalence than previously recognized. Arch Neurol. 2005;62:1459-1463. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 90] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 2. | Appadurai V, DeBarber A, Chiang PW, Patel SB, Steiner RD, Tyler C, Bonnen PE. Apparent underdiagnosis of Cerebrotendinous Xanthomatosis revealed by analysis of ~60,000 human exomes. Mol Genet Metab. 2015;116:298-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 73] [Cited by in RCA: 82] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 3. | Mignarri A, Gallus GN, Dotti MT, Federico A. A suspicion index for early diagnosis and treatment of cerebrotendinous xanthomatosis. J Inherit Metab Dis. 2014;37:421-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 115] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 4. | Pilo-de-la-Fuente B, Jimenez-Escrig A, Lorenzo JR, Pardo J, Arias M, Ares-Luque A, Duarte J, Muñiz-Pérez S, Sobrido MJ. Cerebrotendinous xanthomatosis in Spain: clinical, prognostic, and genetic survey. Eur J Neurol. 2011;18:1203-1211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 102] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 5. | Koyama S, Sekijima Y, Ogura M, Hori M, Matsuki K, Miida T, Harada-Shiba M. Cerebrotendinous Xanthomatosis: Molecular Pathogenesis, Clinical Spectrum, Diagnosis, and Disease-Modifying Treatments. J Atheroscler Thromb. 2021;28:905-925. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 6. | Tao QQ, Zhang Y, Lin HX, Dong HL, Ni W, Wu ZY. Clinical and genetic characteristics of Chinese patients with cerebrotendinous xanthomatosis. Orphanet J Rare Dis. 2019;14:282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 7. | Fraidakis MJ. Psychiatric manifestations in cerebrotendinous xanthomatosis. Transl Psychiatry. 2013;3:e302. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 52] [Cited by in RCA: 62] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 8. | Berginer VM, Foster NL, Sadowsky M, Townsend JA 3rd, Siegel GJ, Salen G. Psychiatric disorders in patients with cerebrotendinous xanthomatosis. Am J Psychiatry. 1988;145:354-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Stelten BML, Huidekoper HH, van de Warrenburg BPC, Brilstra EH, Hollak CEM, Haak HR, Kluijtmans LAJ, Wevers RA, Verrips A. Long-term treatment effect in cerebrotendinous xanthomatosis depends on age at treatment start. Neurology. 2019;92:e83-e95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 67] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 10. | Stelten BML, Lycklama À Nijeholt GJ, Hendriks E, Kluijtmans LAJ, Wevers RA, Verrips A. Long-term MRI Findings in Patients With Cerebrotendinous Xanthomatosis Treated With Chenodeoxycholic Acid. Neurology. 2022;99:559-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |