Published online Feb 25, 2018. doi: 10.5495/wjcid.v8.i1.1
Peer-review started: September 16, 2017
First decision: October 23, 2017
Revised: November 9, 2017
Accepted: November 27, 2017
Article in press: November 27, 2017
Published online: February 25, 2018
Processing time: 97 Days and 23.8 Hours
Mucormycosis is a rare fungal infection which is largely diagnosed in immune-compromised patients. The infection can cause pulmonary, rhinocerebral, skin and soft tissue, central nervous system and gastrointestinal system. The gastrointestinal involvement is the rarest presentation, especially the stomach. We present a 38 years old HIV positive female seen in Steve Biko Academic Hospital with 2 wk history of cough, shortness of breath and lethargy. She was clinically found to be pale, pyrexial, dyspnoea and had severe oral thrush. Blood tests revealed hemoglobin of 6 g/dL CD4 count of 63 cells/uL. Chest X-ray showed multi-lobe pneumonia and gastroscopy confirmed esophageal candidiasis and nodular gastritis. She continued to deteriorate despite antibiotics, antifungal and antituberculous treatment. She developed upper gastrointestinal bleed on day 34, and gastroscopy showed a plaque like lesion in the stomach, histology of which confirmed mucormycosis. She improved on Amphotericin B but subsequently deteriorated and demised a few days later. These cases illustrate the occurrence of a rare fungal infection of the stomach and the poor mortality when diagnosis is delayed. Mucormycosis can co-exist with candidiasis and clinicians should have a high index of suspicion especially in patients not responding to appropriate treatment.
Core tip: Gastric mucormycosis is very rare; co-infection with candida albicans is even more rare. Candidiasis masked the presence of mucormycosis in this patient and was suspected because of poor response to diflucan. Treatment with amphotericin B was delayed but once instituted, the response was good. Awareness of this co-existence of mucormycosis and candida infection in immune-compromised patient should be improved.
- Citation: Kgomo MK, Elnagar AA, Mashoshoe K, Thomas P, Van Hougenhouck-Tulleken WG. Gastric mucormycosis: A case report. World J Clin Infect Dis 2018; 8(1): 1-3
- URL: https://www.wjgnet.com/2220-3176/full/v8/i1/1.htm
- DOI: https://dx.doi.org/10.5495/wjcid.v8.i1.1
Mucormycosis is a rare infection which is largely diagnosed in immune-compromised patients[1], the infection can cause pulmonary, rhinocerebral, skin and soft tissue, central nervous system, gastrointestinal and disseminated disease, with gastrointestinal involvement being the rarest presentation[1].
All portions of the gastrointestinal system can be affected by infection with mucormycosis[1]. In patients found to have gastrointestinal invasive mucormycosis, “the common site of infection is the stomach, ileum and colon”[1,2]. Several predisposing conditions for gastric mucormycosis have been identified, they include malnutrition, persistent ingestion of non-nutritional substances (pica), gastric ulcers, severe systemic illness, age extremes and systemic immunosuppression[3]. The infection is seen in association with underlying conditions, such as diabetes mellitus especially associated with ketoacidosis, hematological malignancy, stem cell or solid organ transplants, severe burns or trauma, treatment with deferoxamine or iron overload states, steroids treatment, immunodeficiency states [e.g., acquired immune deficiency syndrome (AIDS)], injection drug use and malnutrition[3]. Outcome and mortality of zygomycosis varies with the underlying condition and site of infection, it is however very high in general[4]. Invasive mucormycosis is reported to be very rare in immune-compromised patients with primary gastrointestinal disease being the least common[5]. It has previously been diagnosed by imprint cytology[6]. Delaying amphotericin B therapy is associated with poor outcome[7].
We present a 38 years old female seen in our hospital with a 2 wk history of productive cough dyspnoea and lethargy. She had no background history of any medical conditions and according to her, she has been well prior to the presentation. On clinical examination she had oral thrush, more than 1cm cervical and axillary lymphadenopathy, and pallor with an ejection systolic murmur. Her blood pressure was 92/57, temperature 38.7, pulse of 136, saturation at room air of 89%, with respiratory rate of 30 bpm with a normal chest on auscultation.
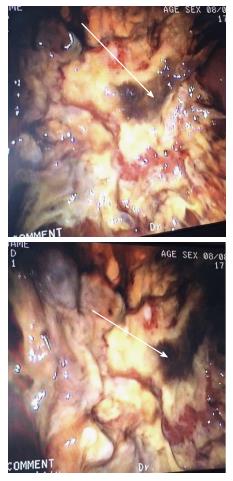
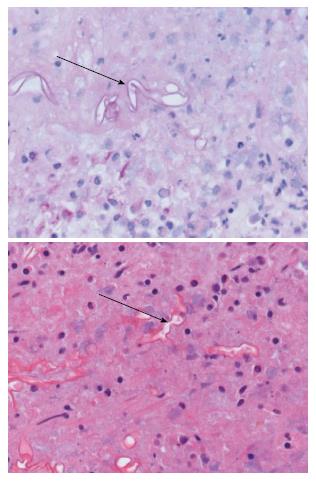
On admission her hemoglobin was 6.0 g/dL with low MCV and MCHC. Her ferritin was 2988 μg/L with a low transferrin saturation of 5%. Her HIV test came back positive, with absolute CD4 count of 63 cells/μL and HIV viral load 405139 copies/mL. She was assessed as having multi-lobar pneumonia, HIV complicated by septic shock and anemia. Gastroscopy done as part of investigation for anemia confirmed esophageal candidiasis and chronic active follicular gastritis. She was started on fluconazole and tuberculosis treatment with rifafour after proven disseminated tuberculosis in sputum and bone marrow aspirate. Despite anti-tuberculosis treatment, anti-fungal cover and broad spectrum antibiotic, patient continued to deteriorate, with worsening sepsis complicated by acute kidney injury requiring hemodialysis. Thirty four days after the initial gastroscopy she developed upper gastrointestinal bleeding, evidenced by coffee ground vomiting and melena stools. Repeat gastroscopy showed a plaque-like mass lesion involving the entire stomach, as shown in Figure 1 below. Biopsy of the mass was performed and sent for histology. Histology of the mass showed masses of fungal organisms admixed with inflammatory debris, the fungal organisms were broad hyphae without septae or acute angle branching. The morphological appearance was in keeping with that of mucormycosis as shown in Figure 2 below.
She was started on amphotericin B after which she improved, inotropic and hemodialysis support was stopped. She however developed a new septic episode while on amphotericin B, complicated by refractory septic shock and demised a few days later.
Mucormycosis is a rare infection which is largely diagnosed in immune-compromised patients[1]. With HIV pandemic in various parts of the world, it is important that health care workers are aware of this infection so that therapy can be instituted early to help reduce the high mortality associated with delay in instituting therapy. The mortality depends on the underlying condition and site of infection. Our patient had many other complications which contributed to her demise.
We present a 38 years old female seen in our hospital with a 2 wk history of productive cough dyspnoea and lethargy.
On clinical examination she had oral thrush, more than 1 cm cervical and axillary lymphadenopathy, pallor with an ejection systolic murmur, hypotension, tachycardia and respiratory failure.
Differential diagnosis of human immunodeficiency virus infection with candidiasis, TB pneumonia and gastrointestinal bleed was made.
She was found to have severe anemia, acquired immune deficiency syndrome with pneumonia, esophageal candidiasis and gastric mucormycosis and gastric bleed.
CXR showed multi-lobar pneumonia and gastroscopy showed gastric plaques as shown in Figure 1.
Gastric biopsy showed fungal hyphae consistent with mucormycosis as shown in Figure 2.
She was started on anti-tuberculosis treatment, fluconazole broad spectrum antibiotics followed a few days later by amphotericin B.
She failed to respond to fluconazole and only responded to amphotericin B.
Mucormycosis is an invasive fungal infection seen only in patients with reduced immune system.
Mucormycosis infect the stomach and can co-exist with candida.
Manuscript source: Unsolicited manuscript
Specialty type: Infectious diseases
Country of origin: South Africa
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Krishnan T, McQuillan GM, Sundar S S- Editor: Ji FF L- Editor: A E- Editor: Lu YJ
| 1. | Shiva Prasad BN, Shenoy A, Nataraj KS. Primary gastrointestinal mucormycosis in an immunocompetent person. J Postgrad Med. 2008;54:211-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 2. | Spellberg B. Gastrointestinal mucormycosis: an evolving disease. Gastroenterol Hepatol (N Y). 2012;8:140-142. [PubMed] |
| 3. | Kontoyiannis DP, Lewis RE. Invasive zygomycosis: update on pathogenesis, clinical manifestations, and management. Infect Dis Clin North Am. 2006;20:581-607, vi. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 185] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 4. | Roden MM, Zaoutis TE, Buchanan WL, Knudsen TA, Sarkisova TA, Schaufele RL, Sein M, Sein T, Chiou CC, Chu JH. Epidemiology and outcome of zygomycosis: a review of 929 reported cases. Clin Infect Dis. 2005;41:634-653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1793] [Cited by in RCA: 1969] [Article Influence: 98.5] [Reference Citation Analysis (1)] |
| 5. | Dioverti MV, Cawcutt KA, Abidi M, Sohail MR, Walker RC, Osmon DR. Gastrointestinal mucormycosis in immunocompromised hosts. Mycoses. 2015;58:714-718. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 54] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 6. | Tathe SP, Dani AA, Chawhan SM, Meshram SA, Randale AA, Raut WK. Gastric mucormycosis: Diagnosis by imprint cytology. Diagn Cytopathol. 2016;44:820-822. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 7. | Chamilos G, Lewis RE, Kontoyiannis DP. Delaying amphotericin B-based frontline therapy significantly increases mortality among patients with hematologic malignancy who have zygomycosis. Clin Infect Dis. 2008;47:503-509. [PubMed] |