Published online Feb 24, 2024. doi: 10.5306/wjco.v15.i2.317
Peer-review started: October 9, 2023
First decision: November 23, 2023
Revised: December 6, 2023
Accepted: January 8, 2024
Article in press: January 8, 2024
Published online: February 24, 2024
Processing time: 133 Days and 17.4 Hours
Limonin is one of the most abundant active ingredients of Tetradium ruticarpum. It exerts antitumor effects on several kinds of cancer cells. However, whether limonin exerts antitumor effects on colorectal cancer (CRC) cells and cancer stem-like cells (CSCs), a subpopulation responsible for a poor prognosis, is unclear.
To evaluate the effects of limonin on CSCs derived from CRC cells.
CSCs were collected by culturing CRC cells in serum-free medium. The cytotoxicity of limonin against CSCs and parental cells (PCs) was determined by cholecystokinin octapeptide-8 assay. The effects of limonin on stemness were detected by measuring stemness hallmarks and sphere formation ability.
As expected, limonin exerted inhibitory effects on CRC cell behaviors, including cell proliferation, migration, invasion, colony formation and tumor formation in soft agar. A relatively low concentration of limonin decreased the expression stemness hallmarks, including Nanog and β-catenin, the proportion of aldehyde dehydrogenase 1-positive CSCs, and the sphere formation rate, indicating that limonin inhibits stemness without presenting cytotoxicity. Additionally, limonin treatment inhibited invasion and tumor formation in soft agar and in nude mice. Moreover, limonin treatment significantly inhibited the phosphorylation of STAT3 at Y705 but not S727 and did not affect total STAT3 expression. Inhibition of Nanog and β-catenin expression and sphere formation by limonin was obviously reversed by pretreatment with 2 μmol/L colievlin.
Taken together, these results indicate that limonin is a promising compound that targets CSCs and could be used to combat CRC recurrence and metastasis.
Core Tip: Colorectal cancer (CRC) is recognized worldwide as one of the most common malignancies and frequently leads to cancer-related death and the subpopulation termed cancer stem-like cell contributes critically to recurrence and metastasis. However, therapeutic molecule or strategy targeting to this subpopulation is still largely unknown. Here, we introduce Limonin, is one of the most abundant active ingredients of Tetradium ruticarpum, as a promising molecule targeting to cancer stem-like cells for CRC therapy.
- Citation: Zhang WF, Ruan CW, Wu JB, Wu GL, Wang XG, Chen HJ. Limonin inhibits the stemness of cancer stem-like cells derived from colorectal carcinoma cells potentially via blocking STAT3 signaling. World J Clin Oncol 2024; 15(2): 317-328
- URL: https://www.wjgnet.com/2218-4333/full/v15/i2/317.htm
- DOI: https://dx.doi.org/10.5306/wjco.v15.i2.317
Colorectal cancer (CRC) is recognized worldwide as one of the most common malignancies and frequently leads to cancer-related death. Currently, numerous treatment strategies for aggressive CRC have been developed; however, promising and novel strategies still need to be developed[1,2]. Accumulating evidence demonstrates that the presence of a small subpopulation of cancer cell-derived parental cells (PCs) termed cancer stem-like cells (CSCs) contributes to tumor recurrence and metastasis. The presence of CSCs is considered the main cause of therapy resistance in several kinds of cancers, including CRC[3]. CSCs are characterized by diverse cancer-initiating properties, including self-renewal capacity and metastatic potential[4,5]. Several putative markers, such as Nanog[6] and β-catenin[7], were reported to be highly expressed in CSCs derived from CRC and correlated with high-risk cases in CRC patients. Targeting CSCs to overcome their promoting effects on chemoresistance or radioresistance has been a recent research focus.
Limonin, a triterpenoid compound, is one of the most abundant molecules in Tetradium ruticarpum[8]. It has several bioactivities, including anti-schistosomal, anti-malarial, antiviral, anti-inflammatory, antioxidant and cholesterol-lowering activities[9-13]. Limonin was also found to exert antitumor effects. Chen et al[8] reported that limonin inhibits angiogenesis and metastasis of human breast cancer. It has also been reported that limonin increases the radiosensitivity of nasopharyngeal carcinoma[14]. Importantly, limonin was reported to be involved in the regulation of the stemness of CSCs. A previous report showed that limonin inhibits the protein level of P-glycoprotein in human colon and leukemia cell lines, which display a high proportion of CSCs[15]. In nasopharyngeal carcinoma cells, limonin inhibits stemness and malignant behaviors by inhibiting cell cycle distribution[14]. However, the roles of limonin in CSCs in CRC cell progression have never been disclosed.
STAT3 is a transcription factor that exerts multiple important biological roles in the processes of normal and transformed cells[16]. Moreover, STAT3 is considered to exert an essential role in maintaining hallmark genes that are critical for the stem cell phenotype, including aldehyde dehydrogenase (ALDH)[17]. STAT3 activation was also reported to be critical in regulating the maintenance of the stemness of CSCs.
In hepatocellular cancer, STAT3 activation promotes stemness by inducing epithelial mesenchymal transition progression[18]. In HER2-negative breast cancer, STAT3 activation was also observed to be correlated with CSC properties[19]. Hence, in this study, we explored potential of utilizing limonin to inhibit CSCs among CRC cells by regulating STAT3 activation.
The human CRC cell lines, HCT116 and SW480, were kept in liquid nitrogen and maintained in DMEM with addition of 10% fetal bovine serum (FBS), 100 μ/mL of penicillin, 100 μg/mL of streptomycin. Cell culture was kept in 5% CO2 incubator at 37 ℃ and medium was replaced every 2-3 d. For enriching CSCs, cells were cultured in DMEM/Ham Nutrient Mixture F-12 (F-12) (at ratio of 1:1) supplemented with epidermal growth factor (EGF, 20 ng/mL), basic Fibroblast Growth Factor basic (bFGF, 10 ng/mL), and 2% B27 for 14-21 d.
The 5 × 103 cells were seeded in each well on a 96-well plate and allowed to attach overnight. To detect the cell viability, 10 μL of cholecystokinin octapeptide-8 was added into each well for 2 h extra incubation, followed by an absorbance measurement at 450 nm using a Multiskan spectrum microplate reader (Thermo Electron Corporation, Waltham, MA, United States). All assays were performed in triplicate and independently repeated three times.
To perform limonin (Sigma-Aldrich, Cat. No.: L9647, St. Louis, MO, United States) treatment, 10-60 μmol/L of limonin was supplemented and after 24-h incubation, cells were employed for following assays.
The 1 × 106 cells were collected and washed using ice-cold PBS for three times. Cells were suspended in 300 μL of ice-cold PBS and fixed with 700 μL of ice-cold 99.5% ethanol overnight. After three careful washed using ice-cold PBS, cells were stained with 50 μg/mL propidium iodide (PI, Sigma–Aldrich, St. Louis, MO, United States) and analyzed by Navios flow cytometers (Beckman Coulter, Brea, CA, United States).
The 24-well transwell chemotaxis chamber technique (Millipore, Billerica, MA, United States) was employed to detect migration capacity. DMEM/F12 medium with addition of 10% FBS was placed in the low chamber room. CSC spheres were trypsinized and single cell suspension were collected. Then, 1 × 104 cells were suspended in Serum-free medium with addition of 2% B 27, 10 ng/mL EGF, and 20 ng/mL bFGF and seeded into the upper chamber (pore size, 8 μm). The chamber was placed in incubator for 24 h incubation at 37 °C. 24 h later, the chamber was obtained and the cells on the upper surface of chamber were wiped away using a cotton swab. Cells were fixed using 4% paraformaldehyde for 15 min at 37 °C and then stained with 0.05% crystal violet for 30 min at room temperature followed by 3 washes with ice-cold PBS. The number of cells that have migrated to the lower surface of the chamber were counted in 5 randomly selected fields using a X71 (U-RFL-T) fluorescence microscope (Olympus, Melville, NY). To detect the invasion capacity, same amount of cells were seeded into chamber pre-coated using Matrigel for 48-h incubation. Same procedure was performed to detect the cells on the lower membrane of chambers. Each assay was performed in triplicate wells.
The 5 × 103 cells were suspended in serum-free medium with addition of 2% B 27, 10 ng/mL EGF and 20 ng/mL bFGF. Cells were dissolved in 0.3% low-melting agarose (Sigma–Aldrich, St. Louis, MO, United States) and were allowed to grow for 14 days and colonies were identified as having > 50 μm in diameter.
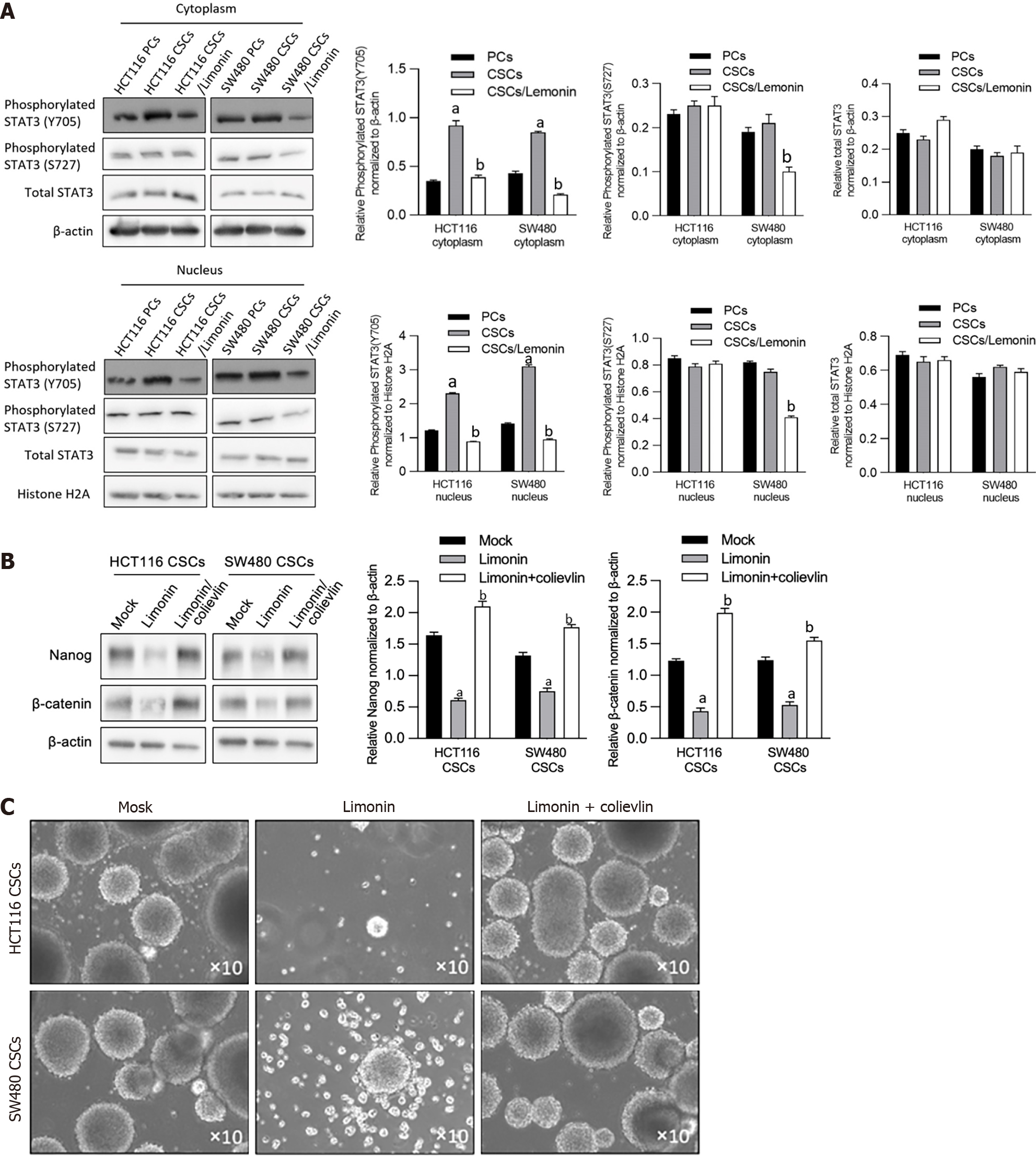
To obtain total protein, 100 μL of lysis buffer containing 1% NP-40, 100 mmol/L Tris-HCL pH 7.5, 150 mmol/L NaCL, 5 mmol/L EDTA was added into 1 × 106 cells and maintained on ice for 30 min. Then, sample was centrifuged at 12000 g, 4 ℃, for 10 min. Supernatant containing total protein was collected and measured using Pierce BCA protein assay kit (Thermo Scientific, Waltham, MA, United States). 20 μg of the total protein was fractionated in an 8% SDS-PAGE gel and transferred to a Nitrocellulose membrane, which were then blocked using PBS containing 0.25% Tween 20 (PBS-T), 5% skim milk for 1 h at room temperature. Then, membrane was incubated with primary antibodies described as followed for 1 h at room temperature: Anti-Nanog antibody (cat. No.: ab109250), anti-β-catenin antibody (cat. No.: ab32572), anti-β-actin antibody (cat. No.: ab8226), anti-STAT3 antibody (cat. No.: ab68153), anti-phosphorylated STAT3 (Y705) antibody (cat. No.: ab278669), anti-phosphorylated STAT3 (S727) antibody (cat. No.: ab32143), anti-Histone H2A antibody (cat. No.: 177308). All antibodies were diluted in 1:1000 using PBS-T. After three washes with PBS-T, membranes were incubated with the corresponding secondary antibodies goat anti-rabbit IgG antibody diluted in 1:5000 (cat. No.: ab7090) for 2 h at room temperature. Obtained membranes were detected using SuperSignal Wester Femto Maximum Sensitivity Substrate (Thermo Scientific, Waltham, MA, United States). Blots were imaged and analyzed with Quantity One (Bio-Rad Laboratories, Hercules, CA, United States).
To flow cytometric analysis of ALDH1, cells were re-suspended and incubated with ALDH1-FITC antibody (Millipore, Billerica, MA, United States), according to the manufacturer’s instructions for 30 min on ice, washed with PBS three times, and fixed with 4% paraformaldehyde for later analysis using BD FACS Canto II, BD Biosciences, San Jose, CA, United States.
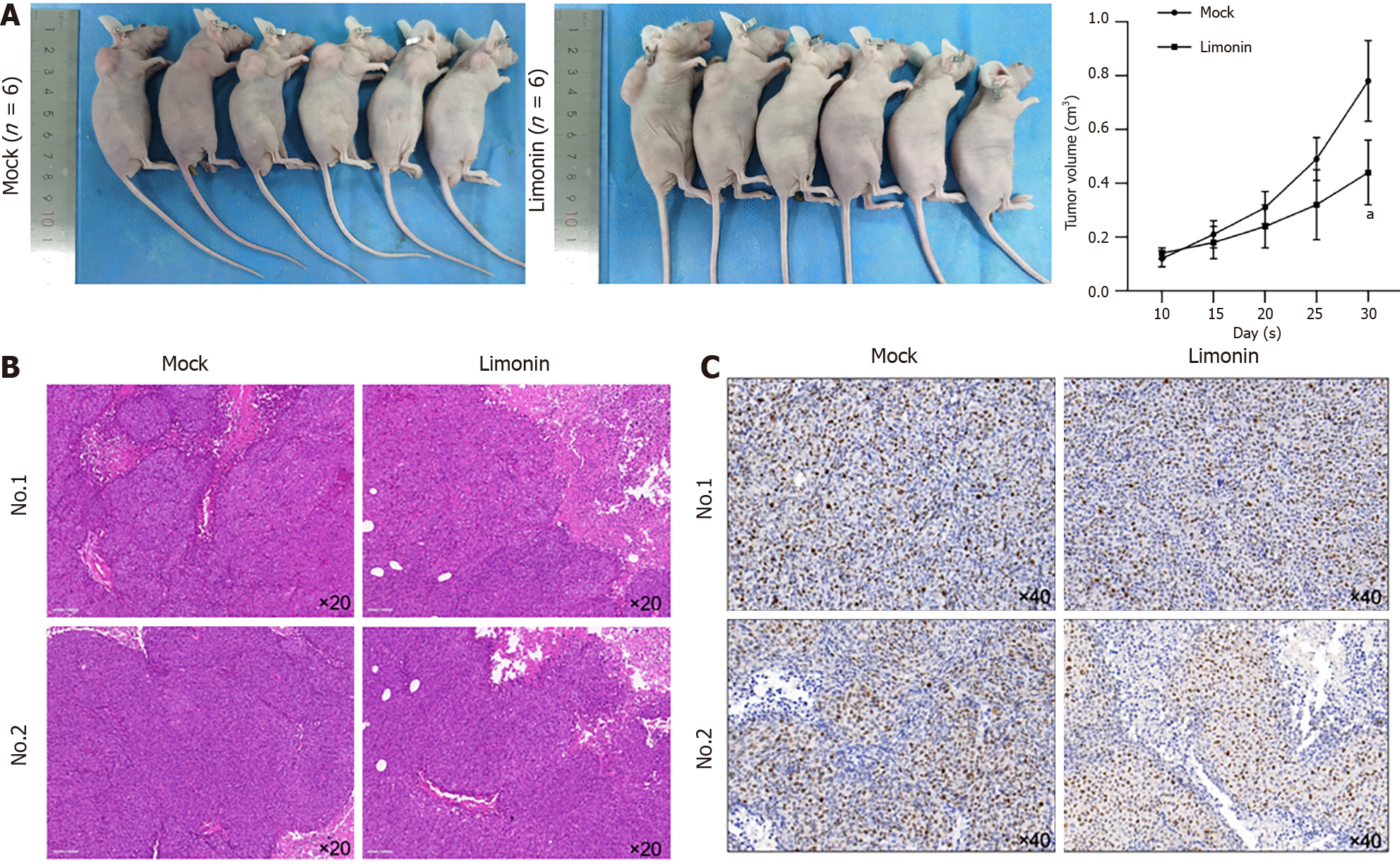
All the animal experiments were conducted according to the Animal ethics committee in Affiliated Hospital of Nanjing University of Traditional Chinese Medicine (No. 2019 DW-13-02). 12 Four-week-old female BALB/c nude mice were purchased from Nanjing JunKe Laboratory Animal Technology Co. Ltd, (Nanjing, China). All animals were raised in the SPF animal facilities (temperature 21–25 °C; humidity 50%–60%; well-ventilated cages) kept on 12 h/12 h light–dark cycle. Food and water were available ad libitum. Mock group of CRC model was established by injecting 2 × 105 CSCs derived from HCT116 cells, into the skin of each nude mouse (6 nude mice). Treated group was established by injecting 2 × 105 CSCs pretreated with limonin for 48 h. We observed the growth of the tumors, and the tumor size was measured at day 10, 15, 20, 25 and 30. One month after the injection, the subcutaneous tumors of nude mice were excised.
After all rats were sacrificed, tumor samples were fixed in 10% formalin for 24 h and then were dehydrated by washing in ascending grades of ethanol. Samples were then sectioned and embedded in paraffin wax for hematoxylin and eosin (H&E) staining. The stained sections were observed for morphological changes. Images were taken under 200 × magnification at five random fields of view under a X71 (U-RFL-T) fluorescence microscope (Olympus, Melville, NY).
A standard two-tailed unpaired student t test was used to calculate differences between samples. One-way analysis of variance was used to determine statistically significant differences from the mean in the xenograft study.
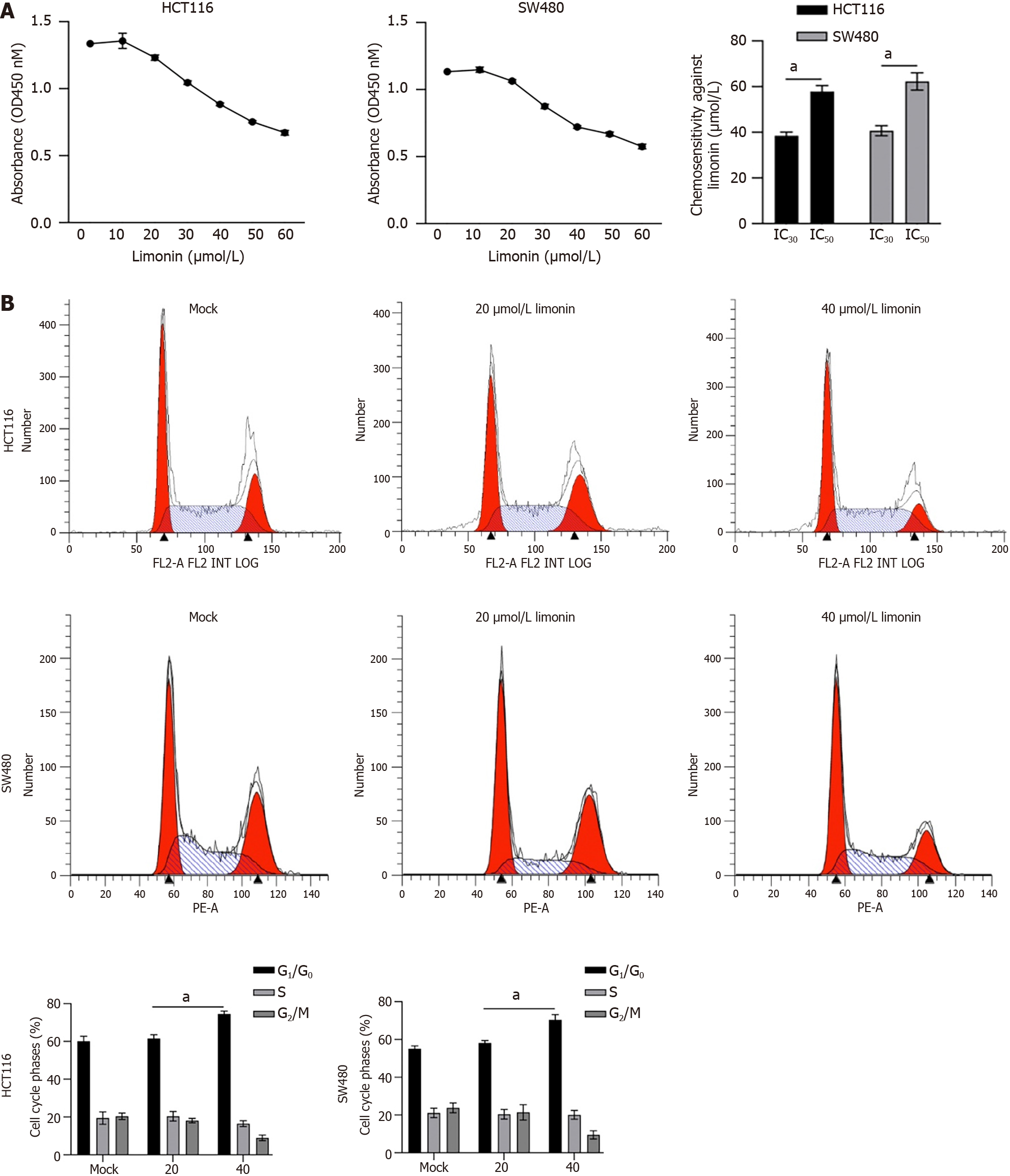
It has been reported that limonin exert broad and effective anticancer activity, including liver cancer[20], intestinal cancer[21], and breast cancer[22]. To evaluate whether limonin exerts cytotoxicity against CRC cells, 10-60 μmol/L of limonin was cultured with HCT116 or SW480 cells for 24 h. As it is shown in Figure 1A, HCT116 showed 30% Inhibitory Concentration (IC30) value of 38.9 ± 1.5 μmol/L and half inhibitory concentration (IC50) value of 58.4 ± 2.1 μmol/L; SW480 showed IC30 value of 42.3 ± 2.1 μmol/L and IC50 value of 63.2 ± 1.7 μmol/L. Then, 20 or 40 μmol/L of limonin was employed for 24-h incubation and HCT116 or SW480 cell cycle distribution were analyzed by performing PI staining followed by flow cytometry assay. Expectedly, 40 μmol/L of limonin, but not 20 μmol/L of limonin treatment, increased proportion of G1/G0 phase (Figure 1B).
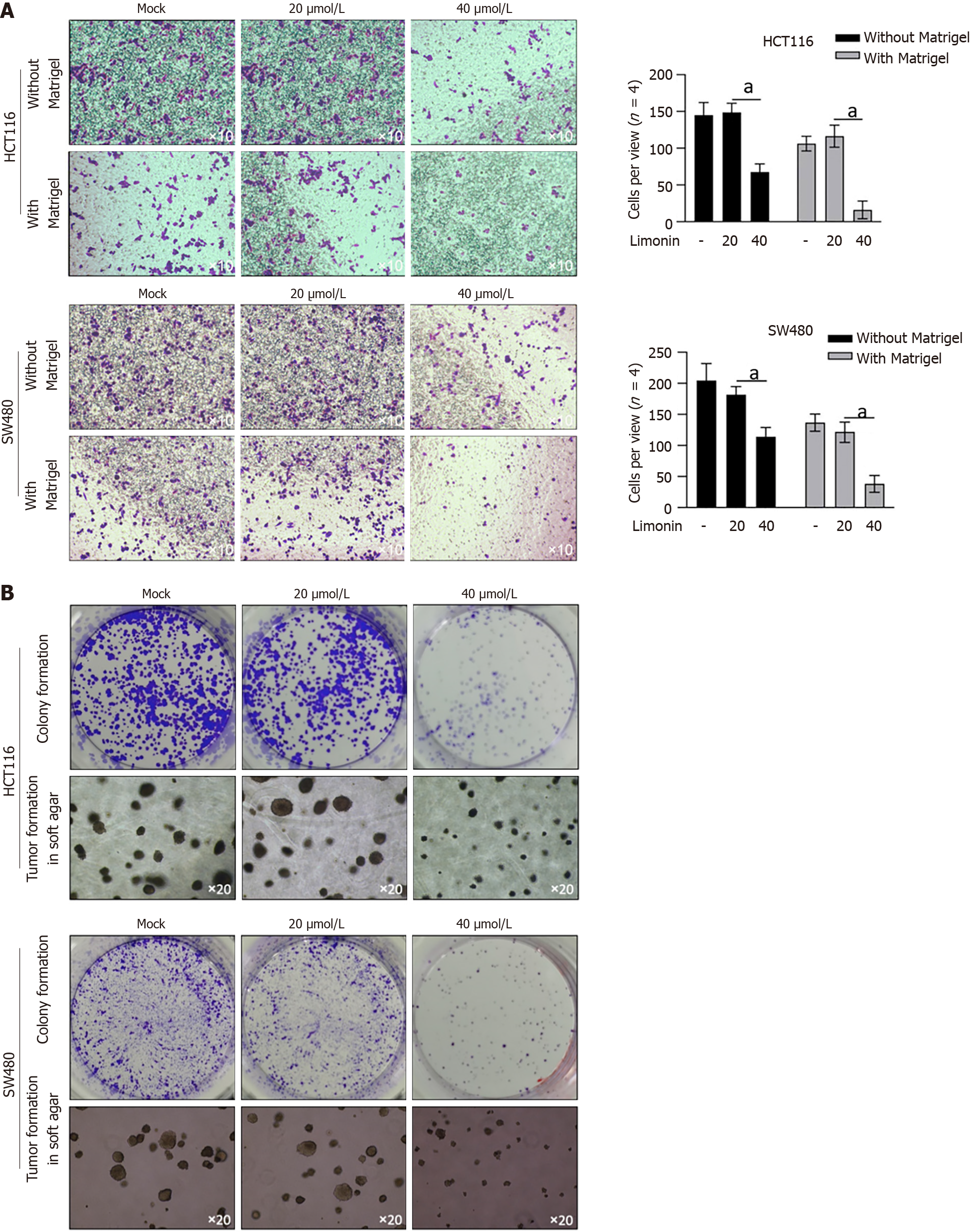
To further confirm its anti-tumor effects on other malignant behaviors, we accessed its effects on migration and invasion. As presented in Figure 2A, 40 μmol/L of limonin treatment significantly decreased transferred cells to lower membrane in transwell without or with Matrigel, demonstrating that limonin decreased cell migration and invasion in colorectal cells. Moreover, after 10-14 d incubation with limonin, formed colonies and tumors > 50 μm in diameter decreased obviously (Figure 2B).
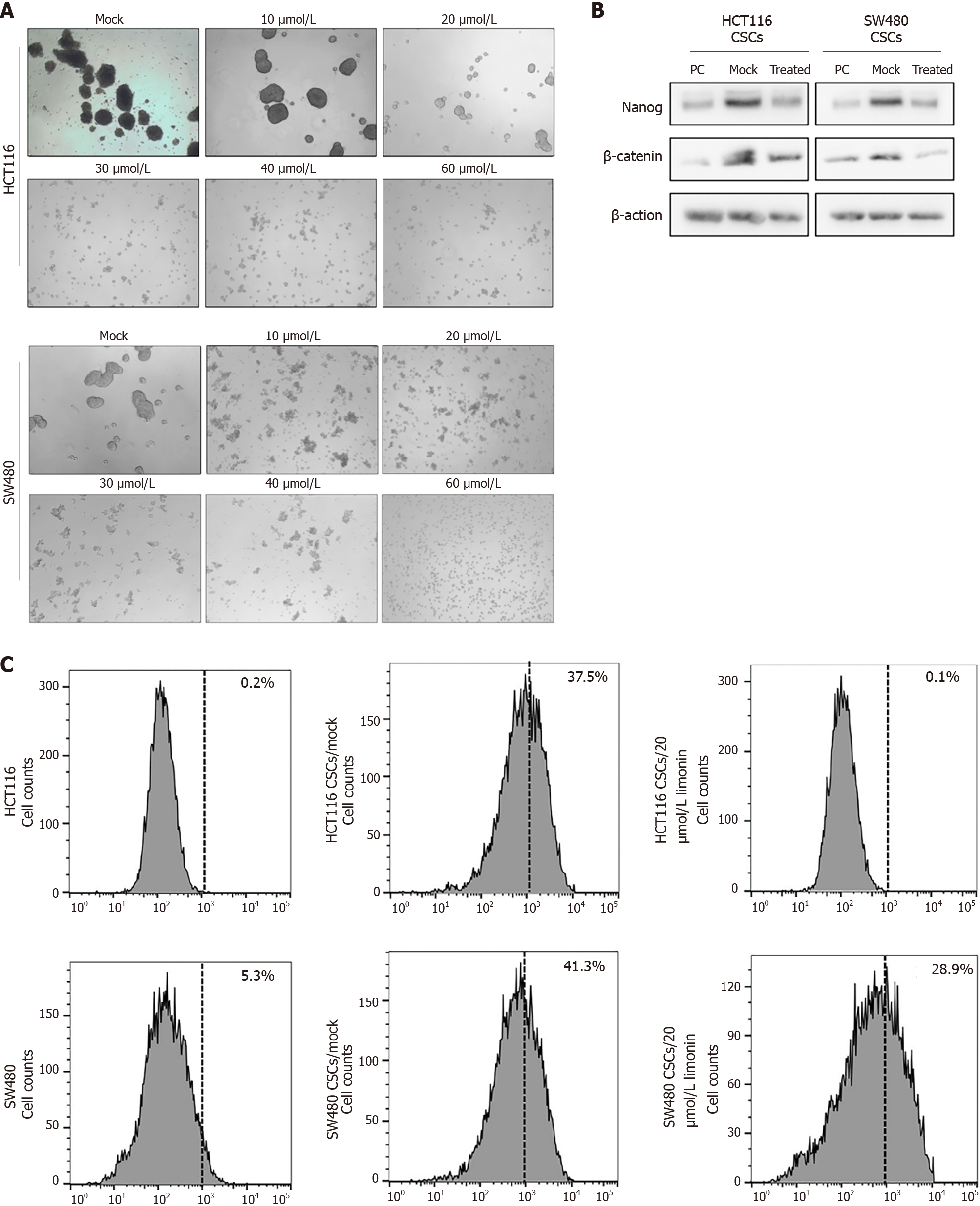
To evaluate the effects of limonin on stemness of CSCs enriched from CRC cells, including HCT116 and SW480, we added limonin ranged from 10-60 μmol/L for 96-h incubation. It is observed that limonin dissociated spheroids obviously (Figure 3A). Notably, 20 μmol/L of limonin completely dissociated spheroids, which presented no detectable cytotoxicity to PCs of HCT116 and SW480 (Figure 1A). By detecting stemness hallmarkers, including Nanog and β-catenin, compared to PCs, CSCs derived from HCT116 or SW480 presented obvious higher levels of Nanog and β-catenin, which was obviously reversed by addition of 20 μmol/L of limonin (Figure 3B). Moreover, we then also detected proportion of ALDH1 in CSCs after limonin treatment. As illustrated in Figure 3C, CSCs in both HCT116 and SW480 presented higher proportion of ALDH1 positive population, and expectedly, addition of limonin decreased ALDH1-positive subpopulation.
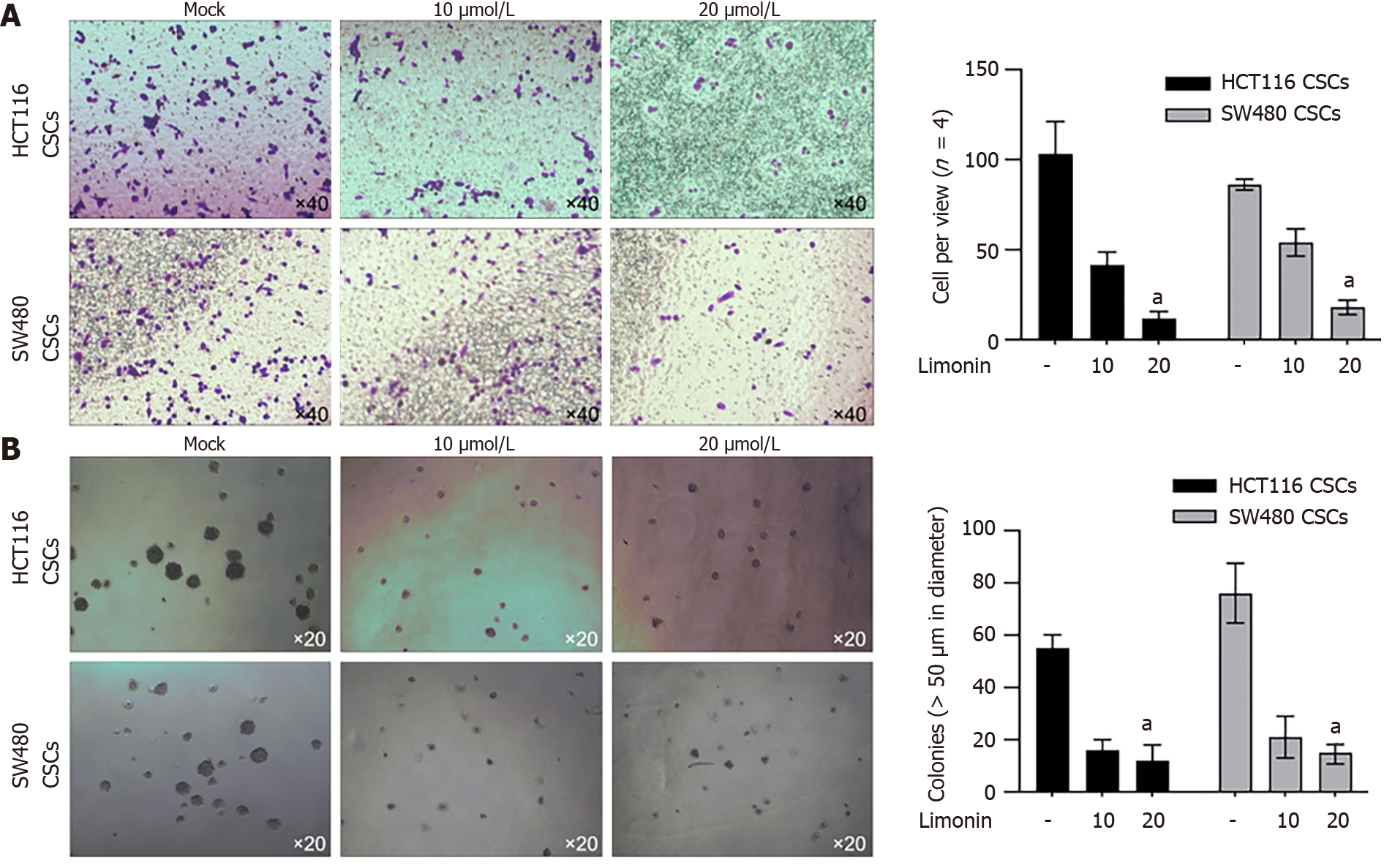
To further confirm, whether the anti-stemness effects of limonin also affects malignancies in CSCs derived from HCT116 and SW480, we treated CSCs with 20 μmol/L of limonin for 24 h. Moreover, 10 and 20 μmol/L of limonin significantly decreased invasion (Figure 4A) and tumor formation in soft agar (Figure 4B), indicating its effects on malignant behaviors.
We then transplanted CSCs pre-treated with limonin for 48 h into nude mice for extra 30-d maintenance. As presented in Figure 5A, limonin pre-treatment significantly decreased tumor formation. By detecting H&E staining, limonin pre-treatment slightly affects pathological structure (Figure 5B), and obviously decreased positive proportion of Ki67, indicating that inhibition of stemness decreased tumor formation in nude mice (Figure 5C). Taken together, these results indicate that, limonin might inhibited stemness and subsequently decreased malignant behaviors, including invasion and tumor formation.
We then explored the potential mechanism contributing to limonin-resulted roles on the stemness and malignancies of CSCs derived from CRC cells. By considering that STAT3 signaling is mostly engaged in CSC progression[23], we evaluated the expression and location of phosphorylated STAT3 (p-STAT3) in CSCs and PCs derived from HCT116 or SW480, and found that the expression of p-STAT3 (Y705) in the cytoplasmic and nuclei fractions was significantly increased in CSCs compared to PCs subpopulation, which is reversed by limonin treatment (Figure 6A). Notably, the expression of p-STAT3 (S727) and total STAT3 protein levels were unaffected. To further confirm whether affected STAT3 signaling is responsible for stemness, we employed a STAT3 activator, colievlin (2 μmol/L), for CSC pretreatment. As presented in Figure 6B and C, inhibition of sphere formation induced by limonin was obviously reversed by pre-treatment of 2 μmol/L of colievlin. These results indicate that, limonin inhibits stemness, at least partially, via inhibiting STAT3 signaling.
Recently, natural products have received increasing attention as novel therapeutics for a wide range of cancers because they have a wide range of anticancer effects and, unlike existing chemoagents, have few side effects. Limonin was reported to have anticancer effects in various studies[8,14,15]. We selected limonin as a novel therapeutic candidate, which may target the CSCs of CRC cells, as these compounds have not yet been studied to prevent recurrence or metastasis induced by the existence of CSCs.
Our results indicated that limonin exerts antitumor effects on malignant behaviors in CRC cells and inhibits the stemness of CSCs derived from CRC cells. Notably, a relatively low concentration of limonin, which fails to exert cytotoxicity, inhibits the stemness of CSCs, demonstrating that limonin regulates CSCs and PCs in different manners. The presence of limonin promotes the dissociation of CSC spheres and decreases the protein levels of Nanog and β-catenin and the percentage of the ALDH1-positive cells without obviously affecting cell viability and cell cycle distribution. Cell proliferation is a critical tumor-promoting process in CRC cells and is considered a leading cause of poor prognosis. Our results indicate that limonin could specifically target the stemness of CSCs without affecting other malignant behaviors and may be considered a specific stemness inhibitor in CRC.
Accumulating evidence suggests tumor-initiating effects of STAT3 activation in several kinds of cancers, including breast cancer[24], lung cancer[25], and CRC[26]. Zhang et al[27] reported that activated STAT3 increased the number of breast cancer stem cells, which was reversed by microRNA-7. In lung cancer, activation of interleukin-6/JAK/STAT3 signaling could enhance lung cancer initiation by promoting the CSC subpopulation[28]. Additionally, in CRC, STAT3 activation was also reported to be tightly correlated with tumor initiation and progression by regulating the abundance of CSCs[26]. These results demonstrated that STAT3 is critical in regulating the stemness of CSCs. Consistently, in our results, limonin treatment significantly decreased STAT3 activation by inhibiting its phosphorylation, which is correlated with the decreased stemness of CSCs. The addition of 2.5 μmol/L colivelin, a STAT3 activator, significantly reversed the inhibitory role of limonin on the stemness of CSCs derived from CRC cells. This indicates that limonin may, at least partially, inhibit the stemness of CSCs by inhibiting STAT3 activation. In this study, we failed to evaluate the effect of high concentrations of limonin on CSCs, which should be investigated in future studies.
STAT3 phosphorylation is closely related to tumor growth, invasion and migration in CRC[29]. Limonin treatment obviously suppressed metastasis and tumor growth mediated by CSCs, which is consistent with previous findings. However, we failed to detect the effect of limonin on cell proliferation after 24 h of treatment. We hypothesized that in CSCs, relatively short-term treatment inhibits STAT3 activation and leads to loss of stemness instead of cell proliferation capacity. In further investigations, it is worth revealing the exact effects of long-term and short-term limonin treatment.
In conclusion, we demonstrated that limonin acts as an antitumor agent by targeting both CSCs and PCs among CRC cells. Furthermore, limonin inhibits STAT3 activation and inhibits the stemness of CSCs derived from CRC cells. Taken together, these findings highlight that limonin could be used as a promising therapeutic agent to CSCs in CRC.
Colorectal cancer (CRC) is one of the most common types of cancer globally. In the last few decades, research efforts have been directed towards understanding the cellular and genetic mechanisms underlying the development and progression of CRC. This has led to the identification and study of a specific subset of cells within colorectal tumors, known as cancer stem cells or cancer stem-like cells (CSCs). The concept of CSCs originated in the mid-20th century but was not widely accepted until the late 1990s. The idea behind CSCs is that a small proportion of cancer cells within a tumor have stem cell-like properties, including the ability to self-renew and differentiate, which contribute to tumor initiation, progression, metastasis, and recurrence. They are also often associated with resistance to traditional cancer therapies.
Several studies have suggested that limonin, a natural compound found in citrus fruits, can exert anti-tumor effects. These effects appear to be due to multiple mechanisms, including the induction of apoptosis (programmed cell death), the inhibition of cancer cell proliferation, and the suppression of metastasis. Based on this, we then investigated whether it exerts suppressing effects on CSCs.
In this research, we evaluated the effects of limonin, a natural compound which is one of the most abundant active ingredients of Tetradium ruticarpum on stemness of CSCs derived from colorectal carcinoma cells. We also investigated the potential mechanism of limonin on stemness of CSCs.
We performed cellular experiment to verify the suppressing effects of limonin on CSCs and performed animal experiment to verify its effects in vivo.
Limonin was found to have suppressive effects on CRC cell activities, which included cell growth, movement, invasion, colony growth and tumor development in soft agar. Even at relatively low concentrations, limonin reduced the expression of stemness markers such as Nanog and β-catenin, decreased the percentage of aldehyde dehydrogenase 1-positive CSCs, and lowered the rate of sphere formation, showing that limonin hinders stemness without cytotoxicity. Furthermore, the treatment of limonin reduced invasion and tumor development in soft agar and in nude mice. Also, limonin treatment notably inhibited the phosphorylation of STAT3 at Y705 but not S727 and had no effect on total STAT3 expression. The limonin-induced reduction in Nanog and β-catenin expression and sphere formation was noticeably offset by a pretreatment with 2 μmol/L colievlin.
In summary, the findings suggest that limonin holds potential as a compound targeting CSCs, and might be utilized for tackling the recurrence and metastasis of CRC.
Can limonin to be used as a chemotherpeutic sensitizor?
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Oncology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Aktas S, Turkey; Haddadi S, Algeria S-Editor: Fan JR L-Editor: A P-Editor: Yu HG
| 1. | Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18694] [Cited by in RCA: 21370] [Article Influence: 2137.0] [Reference Citation Analysis (3)] |
| 2. | Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383:1490-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1965] [Cited by in RCA: 2294] [Article Influence: 208.5] [Reference Citation Analysis (1)] |
| 3. | Park JH, Kim YH, Shim S, Kim A, Jang H, Lee SJ, Park S, Seo S, Jang WI, Lee SB, Kim MJ. Radiation-Activated PI3K/AKT Pathway Promotes the Induction of Cancer Stem-Like Cells via the Upregulation of SOX2 in Colorectal Cancer. Cells. 2021;10. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 4. | Olivares-Urbano MA, Griñán-Lisón C, Marchal JA, Núñez MI. CSC Radioresistance: A Therapeutic Challenge to Improve Radiotherapy Effectiveness in Cancer. Cells. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 148] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 5. | Singh SK, Hawkins C, Clarke ID, Squire JA, Bayani J, Hide T, Henkelman RM, Cusimano MD, Dirks PB. Identification of human brain tumour initiating cells. Nature. 2004;432:396-401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5422] [Cited by in RCA: 5562] [Article Influence: 264.9] [Reference Citation Analysis (0)] |
| 6. | Song KH, Choi CH, Lee HJ, Oh SJ, Woo SR, Hong SO, Noh KH, Cho H, Chung EJ, Kim JH, Chung JY, Hewitt SM, Baek S, Lee KM, Yee C, Son M, Mao CP, Wu TC, Kim TW. HDAC1 Upregulation by NANOG Promotes Multidrug Resistance and a Stem-like Phenotype in Immune Edited Tumor Cells. Cancer Res. 2017;77:5039-5053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 70] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 7. | Escudero-Paniagua B, Bartolomé RA, Rodríguez S, De Los Ríos V, Pintado L, Jaén M, Lafarga M, Fernández-Aceñero MJ, Casal JI. PAUF/ZG16B promotes colorectal cancer progression through alterations of the mitotic functions and the Wnt/β-catenin pathway. Carcinogenesis. 2020;41:203-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 8. | Chen J, Liu BX, Shen Q, Li N, Ling J, Xiao M, Jiao HY, Li T. Limonin inhibits angiogenesis and metastasis of human breast cancer cells by suppressing the VEGFR2/IGFR1-mediated STAT3 signaling pathway. Transl Cancer Res. 2020;9:6820-6832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Reference Citation Analysis (0)] |
| 9. | Barreca D, Gattuso G, Bellocco E, Calderaro A, Trombetta D, Smeriglio A, Laganà G, Daglia M, Meneghini S, Nabavi SM. Flavanones: Citrus phytochemical with health-promoting properties. Biofactors. 2017;43:495-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 233] [Article Influence: 29.1] [Reference Citation Analysis (0)] |
| 10. | Eraky MA, El-Kholy AA, Rashed GA, Hammam OA, Moharam AF, Abou-Ouf EA, Aly NS, Kishik SM, Abdallah KF, Hamdan DI. Dose-response relationship in Schistosoma mansoni juvenile and adult stages following limonin treatment in experimentally infected mice. Parasitol Res. 2016;115:4045-4054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | Gualdani R, Cavalluzzi MM, Lentini G, Habtemariam S. The Chemistry and Pharmacology of Citrus Limonoids. Molecules. 2016;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 106] [Cited by in RCA: 111] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 12. | Hu M, Peng S, He Y, Qin M, Cong X, Xing Y, Liu M, Yi Z. Lycorine is a novel inhibitor of the growth and metastasis of hormone-refractory prostate cancer. Oncotarget. 2015;6:15348-15361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 65] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 13. | Tundis R, Loizzo MR, Menichini F. An overview on chemical aspects and potential health benefits of limonoids and their derivatives. Crit Rev Food Sci Nutr. 2014;54:225-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 93] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 14. | Gao L, Sang JZ, Cao H. Limonin enhances the radiosensitivity of nasopharyngeal carcinoma cells via attenuating Stat3-induced cell stemness. Biomed Pharmacother. 2019;118:109366. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 15. | El-Readi MZ, Hamdan D, Farrag N, El-Shazly A, Wink M. Inhibition of P-glycoprotein activity by limonin and other secondary metabolites from Citrus species in human colon and leukaemia cell lines. Eur J Pharmacol. 2010;626:139-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 102] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 16. | Galoczova M, Coates P, Vojtesek B. STAT3, stem cells, cancer stem cells and p63. Cell Mol Biol Lett. 2018;23:12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 198] [Article Influence: 28.3] [Reference Citation Analysis (0)] |
| 17. | Pattabiraman DR, Weinberg RA. Tackling the cancer stem cells - what challenges do they pose? Nat Rev Drug Discov. 2014;13:497-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 660] [Cited by in RCA: 794] [Article Influence: 72.2] [Reference Citation Analysis (0)] |
| 18. | Gujral TS, Chan M, Peshkin L, Sorger PK, Kirschner MW, MacBeath G. A noncanonical Frizzled2 pathway regulates epithelial-mesenchymal transition and metastasis. Cell. 2014;159:844-856. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 212] [Cited by in RCA: 281] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 19. | Zhou J, Wulfkuhle J, Zhang H, Gu P, Yang Y, Deng J, Margolick JB, Liotta LA, Petricoin E 3rd, Zhang Y. Activation of the PTEN/mTOR/STAT3 pathway in breast cancer stem-like cells is required for viability and maintenance. Proc Natl Acad Sci U S A. 2007;104:16158-16163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 512] [Cited by in RCA: 559] [Article Influence: 31.1] [Reference Citation Analysis (0)] |
| 20. | Rahman A, Siddiqui SA, Jakhar R, Kang SC. Growth inhibition of various human cancer cell lines by imperatorin and limonin from poncirus trifoliata rafin. Seeds. Anticancer Agents Med Chem. 2015;15:236-241. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 21. | Shimizu S, Miyamoto S, Fujii G, Nakanishi R, Onuma W, Ozaki Y, Fujimoto K, Yano T, Mutoh M. Suppression of intestinal carcinogenesis in Apc-mutant mice by limonin. J Clin Biochem Nutr. 2015;57:39-43. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 22. | Fan S, Zhang C, Luo T, Wang J, Tang Y, Chen Z, Yu L. Limonin: A Review of Its Pharmacology, Toxicity, and Pharmacokinetics. Molecules. 2019;24. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 82] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 23. | Li Y, Rogoff HA, Keates S, Gao Y, Murikipudi S, Mikule K, Leggett D, Li W, Pardee AB, Li CJ. Suppression of cancer relapse and metastasis by inhibiting cancer stemness. Proc Natl Acad Sci U S A. 2015;112:1839-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 300] [Cited by in RCA: 379] [Article Influence: 37.9] [Reference Citation Analysis (0)] |
| 24. | Banerjee K, Resat H. Constitutive activation of STAT3 in breast cancer cells: A review. Int J Cancer. 2016;138:2570-2578. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 355] [Cited by in RCA: 468] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 25. | Lánczky A, Nagy Á, Bottai G, Munkácsy G, Szabó A, Santarpia L, Győrffy B. miRpower: a web-tool to validate survival-associated miRNAs utilizing expression data from 2178 breast cancer patients. Breast Cancer Res Treat. 2016;160:439-446. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 590] [Article Influence: 65.6] [Reference Citation Analysis (0)] |
| 26. | Pignon JC, Grisanzio C, Geng Y, Song J, Shivdasani RA, Signoretti S. p63-expressing cells are the stem cells of developing prostate, bladder, and colorectal epithelia. Proc Natl Acad Sci U S A. 2013;110:8105-8110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 177] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 27. | Zhang H, Cai K, Wang J, Wang X, Cheng K, Shi F, Jiang L, Zhang Y, Dou J. MiR-7, inhibited indirectly by lincRNA HOTAIR, directly inhibits SETDB1 and reverses the EMT of breast cancer stem cells by downregulating the STAT3 pathway. Stem Cells. 2014;32:2858-2868. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 284] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 28. | Hsu HS, Lin JH, Hsu TW, Su K, Wang CW, Yang KY, Chiou SH, Hung SC. Mesenchymal stem cells enhance lung cancer initiation through activation of IL-6/JAK2/STAT3 pathway. Lung Cancer. 2012;75:167-177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 71] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 29. | Han C, Sun B, Zhao X, Zhang Y, Gu Q, Liu F, Zhao N, Wu L. Phosphorylation of STAT3 Promotes Vasculogenic Mimicry by Inducing Epithelial-to-Mesenchymal Transition in Colorectal Cancer. Technol Cancer Res Treat. 2017;16:1209-1219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |