Published online May 24, 2021. doi: 10.5306/wjco.v12.i5.335
Peer-review started: February 2, 2021
First decision: March 8, 2021
Revised: March 19, 2021
Accepted: April 25, 2021
Article in press: April 25, 2021
Published online: May 24, 2021
Processing time: 109 Days and 7.7 Hours
Hepatic Langerhans cell histiocytosis (LCH) is characterized by proliferation and accumulation of Langerhans cells in the liver, causing liver dysfunction or forming a mass lesion. The liver can be involved in isolation, or be affected along with other organs. A common clinical hepatic presentation is cholestasis with pruritis, fatigue and direct hyperbilirubinemia. In late stages, there may be hypoalbuminemia. Liver biopsy may be required for the diagnosis of hepatic LCH. Histologic finding may be diverse, including lobular Langerhans cell infiltrate with mixed inflammatory background, primary biliary cholangitis-like pattern, sclerosing cholangitis-like pattern, and even cirrhosis at later stages. Because of its non-specific injury patterns with broad differential diagnosis, establishing a diagnosis of hepatic LCH can be challenging. Hepatic LCH can easily be missed unless this diagnosis is considered at the time of biopsy interpretation. A definitive diagnosis relies on positive staining with CD1a and S100 antigen. Liver involvement is a high risk feature in LCH. The overall prognosis of hepatic LCH is poor. Treating at an early stage may improve the outcome. Systemic chemotherapy is the mainstay of treatment and liver transplantation may be offered. New molecular markers involved in pathogenesis of LCH are being explored with a potential for targeted therapy. However, further studies are needed to improve outcome.
Core Tip: Hepatic Langerhans cell histiocytosis (LCH) is characterized by proliferation and accumulation of Langerhans cells in the liver, causing liver dysfunction or forming a mass lesion. There are diverse injury patterns on liver biopsy, including lobular Langerhans cell infiltrate with mixed inflammatory background, primary biliary cholangitis-like pattern, sclerosing cholangitis-like pattern, and even cirrhosis at later stage. The overall prognosis of hepatic LCH is poor. New molecular markers involved in pathogenesis of LCH are being explored with a potential for targeted therapy.
- Citation: Fu Z, Li H, Arslan ME, Ells PF, Lee H. Hepatic Langerhans cell histiocytosis: A review. World J Clin Oncol 2021; 12(5): 335-341
- URL: https://www.wjgnet.com/2218-4333/full/v12/i5/335.htm
- DOI: https://dx.doi.org/10.5306/wjco.v12.i5.335
Langerhans cell histiocytosis (LCH) is characterized by proliferation and accumulation of dendritic antigen-presenting histiocytes, Langerhans cells, in tissue. Langerhans cells express CD1a, S-100 and langerin proteins, and show Birbeck granules on ultrastructural examination[1]. LCH is a rare disease with an annual incidence of about 5 cases per 1 million population. The disease is more common in Caucasian population of northern European descent, with a male predominance. It can affect any age group but most cases occur in children[1,2]. The etiology is unknown. However, there is a strong association between pulmonary LCH and smoking[3,4].
Some authors have considered that LCH is an abnormal reactive process to an inciting event, such as viral infection, given the fact that multiple cytokines are involved in the process and LCH has been reported to regress spontaneously when it’s an isolated lesion[5,6]. However, the revised 4th edition World Health Organization[1] classified LCH as a clonal neoplastic process. Recently, Murakami et al[7] proposed that both BRAF mutation and Interleukin-1 Loop amplification play important roles in the pathogenesis of LCH[7]. More recently, other groups found that BRAFV600E mutation or alternative activating MAPK pathway gene mutations are almost universally identified in LCH[8,9]. These studies provide a potential for molecular alteration-based targeted therapy for LCH.
LCH demonstrates a variable clinical picture and course. It can involve a single organ system (SS-LCH) or multiple organ systems (MS-LCH). While any organ can be affected, bone is the most frequent site occurring in 80% of cases of LCH; it can be unifocal or multifocal. A third of LCH cases involve the skin and a quarter involve the pituitary gland. Other organs, such as liver, spleen, lungs, lymph nodes, gas
When LCH involves a single organ, the course may be indolent and have a favorable survival. Su et al[11] reported 100% 5-year survival in pediatric LCH patients when the disease involved only bone[11]. In a long-term (over 17 years) follow-up study of pediatric LCH, SS-LCH showed a 100% regression rate and low relapse vs 73% regression in MS-LCH[12].
LCH involving the skin, bone, lymph nodes or pituitary gland is considered “low-risk” because of its good response to treatment. Involvement of the lungs, hematopoietic system and spleen, and liver is considered “high risk” with an unfavorable outcome[1,6,12,13].
In this review, we focus on hepatic LCH and provide a brief overview of its clinical presentation, laboratory/imaging findings and current treatment. Its pathologic findings and differential diagnosis are also reviewed.
The diagnosis of hepatic LCH in a patient with known LCH requires one or more of the followings: (1) hepatomegaly, defined as a liver edge greater than 3 cm below the costal margin at the mid clavicular line (confirmed by ultrasound); (2) liver dysfunction defined either by abnormal serum biochemical tests including bilirubin greater than 3 times the upper limit of normal, protein less than 55 g/L, albumin less than 3 g/dL, transaminases [alanine aminotransferase (ALT)/ aspartate aminotransferase (AST)] greater than 3 times normal, or by clinical entities including an intrahepatic nodular mass or ascites or edema, not as a result of other causes; or (3) histopathological findings of active disease[14,15].
The liver may be affected in isolation[16-19], or involved along with other organ systems such as lymph nodes, skin and lungs.
In pediatric LCH, the frequency of liver involvement is variable but may be high (15%-60%). It carries a poor prognosis. In adult LCH, liver involvement is relatively uncommon with an incidence of 16%-27%. However, it is probably under recognized. One study of multisystemic LCH in adults reported liver involvement in 87%[14,18,20].
There are 2 stages of hepatic LCH: An early stage with liver parenchymal infiltration by Langerhans cells and a late stage with biliary sclerosis[18,20]. Clinical manifestations differ based on the stage.
A common presentation is jaundice with direct hyperbilirubinemia and hypoalbuminemia. Patients may present with fatigue and pruritus[21]. Clinically, hepatic LCH may mimic other conditions such as chronic destructive cholangitis, metabolic disease, hepatitis, neoplasia obstructing biliary tract and inherited deficient conjugation of bilirubin. It can also present as Reye syndrome, chronic inflammatory bowel disease, or hemochromatosis[14]. In late stages, patients may present with severe sclerosing cholangitis and liver failure. A subset of patients progresses to develop cirrhosis, which may lead to portal hypertension and secondary hypersplenism[22,23].
Laboratory tests show abnormal serum liver tests with mild to moderately elevated ALT and AST. Cholestatic biochemical profiles are seen with increased total bilirubin, γ glutamyl transferases and anaplastic lymphoma kinase phosphatase. The albumin level is often low. The prothrombin time may be prolonged due to decreased clotting factor. Depending on liver function, clotting factors may be depleted. Complete blood count is usually normal, although the platelet count may be low in patients with portal hypertension and splenomegaly. Abdominal contrast-enhanced computed tomography may demonstrate hepatomegaly with solitary or multiple hypodense hepatic nodules, which can be confluent[18]. At a late stage, magnetic resonance imaging may show biliary tree abnormalities[20].
Liver biopsy is the cornerstone of the diagnosis of LCH and disease staging. Liver biopsy can show various injury patterns. In an early stage, lobular Langerhans cell infiltrate mixed with lymphocytes can be seen. The Langerhans cells may form focal aggregates or be multinucleated[18]. In addition to lymphocytes, mature eosinophils, neutrophils and plasma cells can also be noted. A definitive diagnosis can be rendered based on positive immunohistochemical staining with CD1a and S100 antigen of the Langerhans cells.
At a later stage, Langerhans cells may infiltrate the bile ducts and cause sclerosing cholangitis. In some cases, histologic features of sclerosing cholangitis may be seen in a biopsy without identifiable Langerhans cells[17,22,24]. This may be due to selective involvement of the major bile ducts by Langerhans cells; large ducts are unlikely to be sampled in the usual needle biopsy[25]. Chronic non-suppurative destructive cholangitis injury pattern has been also reported[26].
Notably, hepatic LCH may mimic primary biliary cholangitis (PBC). Rush et al[19] reported hepatic LCH presenting as a small noncaseating granuloma in the portal tract with rare multinucleated epithelioid giant cells within the portal inflammatory infiltrate. The patient underwent a liver transplantation for the presumed diagnosis of advanced anti-mitochondrial antibody (AMA)-negative PBC. However, the disease recurred in the allograft. The diagnosis of hepatic LCH was finally rendered three years after transplantation in the allograft biopsy. Four years later (7 years after the transplantation), the patient lost the liver graft. LCH diagnosis was confirmed in the explanted allograft liver[19].
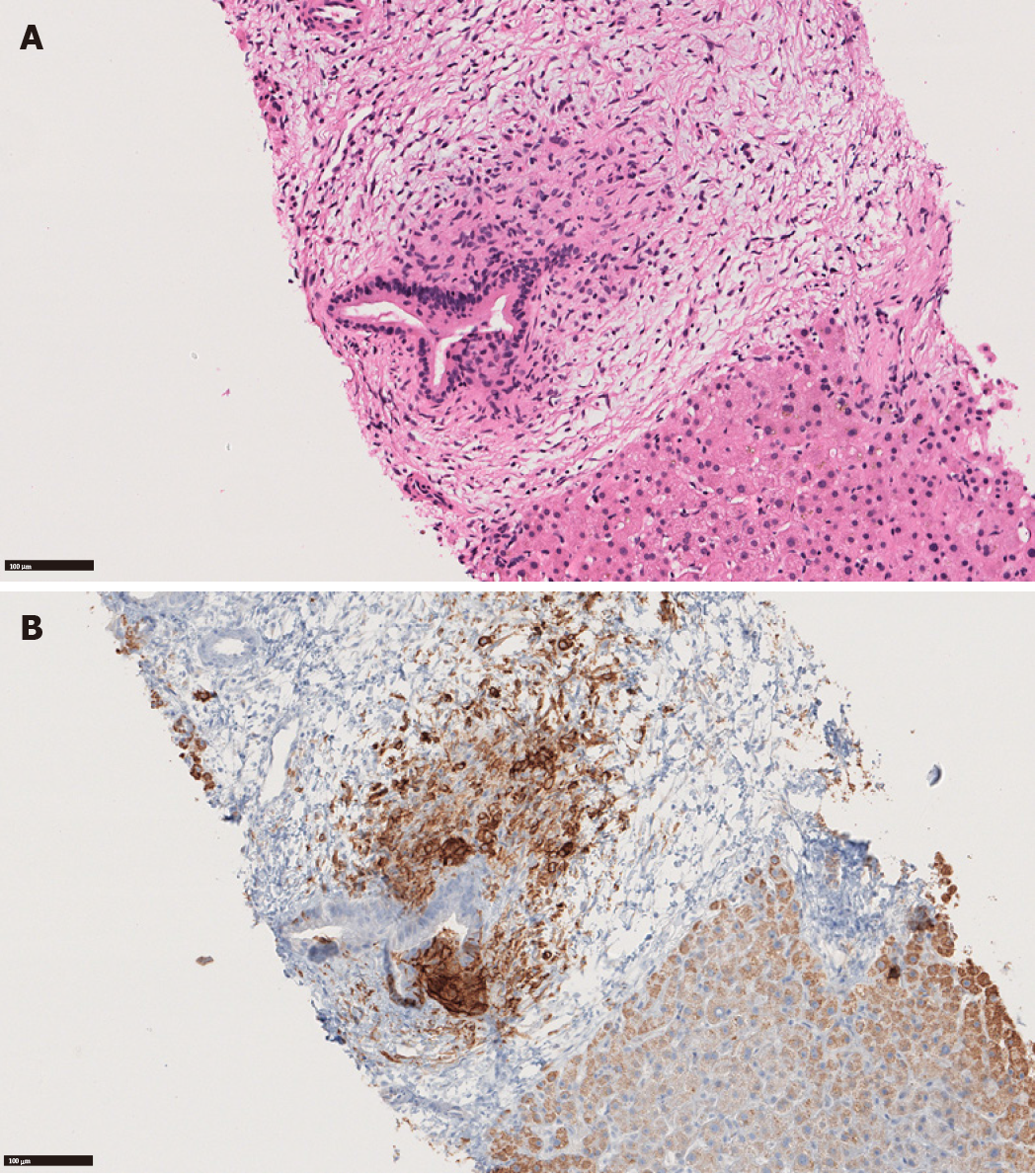
Similarly, our group reported a case of hepatic LCH that histologically and pathologically mimicked PBC. A 65-year-old man presented with intermittent pruritus with cholestatic biochemistry profile for years. The liver biopsy showed portal histiocytic aggregate (non-necrotizing granuloma) encasing a damaged bile duct (Figure 1). Tests for autoantibodies including AMA were negative, therefore AMA-negative PBC was suspected. One month later, multiple skin lesions developed and a diagnosis of LCH was rendered on a skin biopsy. In light of the positive skin biopsy, the previous liver biopsy was re-examined. The histiocytes surrounding the duct were positive for CD1a and S100, confirming the diagnosis of hepatic LCH, retrospectively[21].
These two cases highlight the difficulty of rendering a diagnosis of hepatic LCH without appropriate clinical context.
In summary, hepatic LCH may present as a non-specific inflammatory process with varying injury pattern. Therefore, the diagnosis of hepatic LCH can easily be missed when this diagnosis is not considered. In addition, its patchy nature and limited sampling may further hinder the diagnosis of hepatic LCH[21]. In late stage MS-LCH, even though the clinical picture may be strongly indicative of hepatic LCH, it may be difficult to render a definitive diagnosis on the initial biopsy and a repeat biopsy may be necessary[6].
There are a variety of differential diagnoses for hepatic LCH due to its nonspecific presentations and morphologic features.
The morphological and clinical findings of hepatic LCH may resemble primary sclerosing cholangitis (PSC). A classic “beaded” appearance of extrahepatic biliary tree by endoscopic retrograde cholangiopancreatography and periductal onion-skin type fibrosis with negative stains for S100 and CD1a may be helpful in the diagnosis of PSC.
Granulomatous reaction with ductular proliferation raises a differential diagnosis of PBC. The presence of a dense portal lymphoplasmacytic infiltrate, mild degree of interface activity, and negativity for S100 and CD1a in conjunction with positivity for AMA can be helpful for PBC diagnosis. However, as demonstrated above, it can be very difficult to differentiate hepatic LCH from AMA negative PBC in a small liver biopsy, especially in the absence of appropriate clinical context.
In hepatic LCH, the presence of granulomatous inflammation also raises a differential diagnosis of infection. Grocott (or Gomori) methenamine silver and acid-fast bacteria stains may help to rule out infectious etiology, although they have low sensitivity[27-30]. Tissue culture may be helpful in these scenarios. Other differential diagnosis includes sarcoidosis, foreign-body type giant cell reactions, drug-induced liver injury, and non-Hodgkin’s and Hodgkin’s lymphoma.
Additionally, myeloproliferative disorders and myeloid leukemias can express CD1a and/or S100 protein, mimicking LCH. However, these are distinguished by sinusoidal infiltrative pattern of the neoplastic cells[25].
Depending on clinical course and degree of organ dysfunction, variable treatment options are offered for LCH. Usually, patients with single site SS-LCH are observed only or offered monotherapy, such as oral 6-mercaptopurine and methotrexate, indomethacin, bisphosphonates, and hydroxyurea. Patients with MS-LCH usually benefit from systemic therapy, such as the vinblastine-prednisone combination [6,15,20].
Recently, molecular markers such as BRAFV600E and MAP2K1 have been identified in LCH pathogenesis. These markers may serve as molecular targets for precision therapy in the future. For instance, vemurafenib, a BRAF inhibitor, had prolonged efficacy in patients with BRAF V600-mutant LCH[31]. In addition, MEK inhibitors showed near-universal responses in patients with histiocytoses, including LCH, regardless of tumor genotype[32]. However, prospective clinical trials would be required to determine optimal duration of therapy and to explore a potential for combination with other targeted or cytotoxic therapies.
The optimal treatment for hepatic LCH remains to be determined. Currently, systemic chemotherapy is the mainstay of treatment. Yi et al[33] reported that earlier systemic chemotherapy has led to a relatively better outcome in hepatic LCH. However, the therapy was not effective once cirrhosis developed[33]. Therefore, early diagnosis and treatment of hepatic LCH would be crucial. Unfortunately, a subset of hepatic LCH patients develops severe sclerosing cholangitis that progresses to cirrhosis. In this case, liver transplantation may be the treatment of choice and should be considered early in the disease course[34]. Tang et al[35] reported a case of hepatic LCH that was successfully treated by liver transplantation with subsequent tacrolimus and mycophenolate mofetil as immunosuppressants[35].
Further collaborative studies regarding the biology, clinical presentation and outcome of hepatic LCH would be required to explore and refine variable treatment options.
In LCH, liver involvement has a significant bearing on survival. The overall prognosis of hepatic LCH is poor with a fatality rate of 30%-50% (versus < 10% without liver involvement) and median survival of 9 years[6,18,20]. The 3-year survival rate of LCH with liver involvement is 51.8%, compared with that of 96.7% without liver involvement[6]. In Abdallah et al[20]’s study, 30% of patients died due to sclerosing cholangitis complicated by secondary cirrhosis[20]. Therefore, it is very important to identify liver involvement at an early reversible stage. Early detection and treatment may improve the outcome[6,18].
While hepatic LCH is relatively uncommon, it portends a poor prognosis. Early detection and treatment of hepatic LCH may allow a better prognosis. Liver biopsy plays an important role in the diagnosis and management of hepatic LCH. Unfortunately, the histomorphology is non-specific and needs to be differentiated from other granulomatous processes, such as PBC or infection. A definitive diagnosis requires confirmatory immunohistochemical staining with CD1a and S100. Currently, systemic chemotherapy is the mainstay of treatment for hepatic LCH. Further studies are required to explore other treatment modalities including molecularly targeted therapy.
Manuscript source: Invited manuscript
Specialty type: Oncology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Soldera J S-Editor: Fan JR L-Editor: A P-Editor: Wu YXJ
| 1. | Swerdlow SH, Campo E, Harris NL, Jaffe ES, Pileri SA, Stein H, Thiele J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues (Revised 4th Edition). Lyon: IARC, 2017: 470-472. |
| 2. | Pileri SA, Grogan TM, Harris NL, Banks P, Campo E, Chan JK, Favera RD, Delsol G, De Wolf-Peeters C, Falini B, Gascoyne RD, Gaulard P, Gatter KC, Isaacson PG, Jaffe ES, Kluin P, Knowles DM, Mason DY, Mori S, Müller-Hermelink HK, Piris MA, Ralfkiaer E, Stein H, Su IJ, Warnke RA, Weiss LM. Tumours of histiocytes and accessory dendritic cells: an immunohistochemical approach to classification from the International Lymphoma Study Group based on 61 cases. Histopathology. 2002;41:1-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 491] [Cited by in RCA: 450] [Article Influence: 19.6] [Reference Citation Analysis (0)] |
| 3. | Vassallo R, Ryu JH, Schroeder DR, Decker PA, Limper AH. Clinical outcomes of pulmonary Langerhans'-cell histiocytosis in adults. N Engl J Med. 2002;346:484-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 295] [Cited by in RCA: 240] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 4. | Bernstrand C, Cederlund K, Sandstedt B, Ahström L, Lundell M, Dahlquist G, Henter JI. Pulmonary abnormalities at long-term follow-up of patients with Langerhans cell histiocytosis. Med Pediatr Oncol. 2001;36:459-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 35] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Tillotson CV, Anjum F, Patel BC. Langerhans Cell Histiocytosis. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2021. [PubMed] |
| 6. | Henter JI, Tondini C, Pritchard J. Histiocyte disorders. Crit Rev Oncol Hematol. 2004;50:157-174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 116] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 7. | Murakami I, Matsushita M, Iwasaki T, Kuwamoto S, Kato M, Nagata K, Horie Y, Hayashi K, Imamura T, Morimoto A, Imashuku S, Gogusev J, Jaubert F, Takata K, Oka T, Yoshino T. Interleukin-1 loop model for pathogenesis of Langerhans cell histiocytosis. Cell Commun Signal. 2015;13:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 8. | Chakraborty R, Burke TM, Hampton OA, Zinn DJ, Lim KP, Abhyankar H, Scull B, Kumar V, Kakkar N, Wheeler DA, Roy A, Poulikakos PI, Merad M, McClain KL, Parsons DW, Allen CE. Alternative genetic mechanisms of BRAF activation in Langerhans cell histiocytosis. Blood. 2016;128:2533-2537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 123] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 9. | Héritier S, Hélias-Rodzewicz Z, Chakraborty R, Sengal AG, Bellanné-Chantelot C, Thomas C, Moreau A, Fraitag S, Allen CE, Donadieu J, Emile JF. New somatic BRAF splicing mutation in Langerhans cell histiocytosis. Mol Cancer. 2017;16:115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 10. | Narayanasamy Rajavelu T, Abimannane A, Chinnaiah Govindhareddy DK, Kayal S, Kar R. Langerhans' Cell Histiocytosis Masquerading as Caroli's Disease. J Pediatr Hematol Oncol. 2020;42:e620-e622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 11. | Su M, Gao YJ, Pan C, Chen J, Tang JY. Outcome of children with Langerhans cell histiocytosis and single-system involvement: A retrospective study at a single center in Shanghai, China. Pediatr Hematol Oncol. 2018;35:385-392. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 12. | İnce D, Demirağ B, Özek G, Erbay A, Ortaç R, Oymak Y, Kamer S, Yaman Y, Kundak S, Vergin C. Pediatric langerhans cell histiocytosis: single center experience over a 17-year period. Turk J Pediatr. 2016;58:349-355. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Gadner H, Grois N, Arico M, Broadbent V, Ceci A, Jakobson A, Komp D, Michaelis J, Nicholson S, Pötschger U, Pritchard J, Ladisch S; Histiocyte Society. A randomized trial of treatment for multisystem Langerhans' cell histiocytosis. J Pediatr. 2001;138:728-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 343] [Cited by in RCA: 296] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 14. | Haupt R, Minkov M, Astigarraga I, Schäfer E, Nanduri V, Jubran R, Egeler RM, Janka G, Micic D, Rodriguez-Galindo C, Van Gool S, Visser J, Weitzman S, Donadieu J; Euro Histio Network. Langerhans cell histiocytosis (LCH): guidelines for diagnosis, clinical work-up, and treatment for patients till the age of 18 years. Pediatr Blood Cancer. 2013;60:175-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 394] [Cited by in RCA: 419] [Article Influence: 34.9] [Reference Citation Analysis (0)] |
| 15. | Rodriguez-Galindo C, Allen CE. Langerhans cell histiocytosis. Blood. 2020;135:1319-1331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 204] [Article Influence: 40.8] [Reference Citation Analysis (0)] |
| 16. | Finn LS, Jaffe R. Langerhans' cell granuloma confined to the bile duct. Pediatr Pathol Lab Med. 1997;17:461-468. [PubMed] |
| 17. | Kaplan KJ, Goodman ZD, Ishak KG. Liver involvement in Langerhans' cell histiocytosis: a study of nine cases. Mod Pathol. 1999;12:370-378. [PubMed] |
| 18. | Araujo B, Costa F, Lopes J, Castro R. Adult langerhans cell histiocytosis with hepatic and pulmonary involvement. Case Rep Radiol. 2015;2015:536328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 19. | Rush N, Saxena R, Lin J. Langerhans Cell Histiocytosis With Solitary Hepatic Involvement in an Adult. AJSP Rev Rep. 2020;25:40-44. [DOI] [Full Text] |
| 20. | Abdallah M, Généreau T, Donadieu J, Emile JF, Chazouillères O, Gaujoux-Viala C, Cabane J. Langerhans' cell histiocytosis of the liver in adults. Clin Res Hepatol Gastroenterol. 2011;35:475-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 21. | Li H, Ells P, Arslan ME, Robstad KA, Lee H. Hepatic Langerhans Cell Histiocytosis (LCH) Presenting as a Harbinger of Multisystem LCH. Cureus. 2020;12:e8591. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 22. | Hatemi I, Baysal B, Senturk H, Behzatoglu K, Bozkurt ER, Ozbay G. Adult Langerhans cell histiocytosis and sclerosing cholangitis: a case report and review of the literature. Hepatol Int. 2010;4:653-658. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 14] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 23. | Pirovino M, Jeanneret C, Läng RH, Luisier J, Bianchi L, Spichtin H. Liver cirrhosis in histiocytosis X. Liver. 1988;8:293-298. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 24. | Liu DG, Zhang YX, Li F. Multisystem Langerhans cell histiocytosis with liver dysfunction as the first presentation: A case report. Oncol Lett. 2012;3:391-394. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 25. | Jaffe R. Liver involvement in the histiocytic disorders of childhood. Pediatr Dev Pathol. 2004;7:214-225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 42] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 26. | Haas S, Theuerkauf I, Kühnen A, Wickesberg A, Fischer HP. [Langerhans' cell histiocytosis of the liver. Differential diagnosis of a rare chronic destructive sclerosing cholangitis]. Pathologe. 2003;24:119-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 27. | Zink AR, Nerlich AG. Molecular strain identification of the Mycobacterium tuberculosis complex in archival tissue samples. J Clin Pathol. 2004;57:1185-1192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 28. | Fukunaga H, Murakami T, Gondo T, Sugi K, Ishihara T. Sensitivity of acid-fast staining for Mycobacterium tuberculosis in formalin-fixed tissue. Am J Respir Crit Care Med. 2002;166:994-997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 61] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 29. | Reza Kermanshahi T, Rhatigan R. Comparison between PAS and GMS stains for the diagnosis of onychomycosis. J Cutan Pathol. 2010;37:1041-1044. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 30. | Azar MM, Slotkin R, Abi-Raad R, Liu Y, Grant MH, Malinis MF. Gomori Methenamine Silver Stain on Bronchoalveolar Lavage Fluid Is Poorly Sensitive for Diagnosis of Pneumocystis jiroveci Pneumonia in HIV-Negative Immunocompromised Patients and May Lead to Missed or Delayed Diagnoses. Arch Pathol Lab Med. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 31. | Diamond EL, Subbiah V, Lockhart AC, Blay JY, Puzanov I, Chau I, Raje NS, Wolf J, Erinjeri JP, Torrisi J, Lacouture M, Elez E, Martínez-Valle F, Durham B, Arcila ME, Ulaner G, Abdel-Wahab O, Pitcher B, Makrutzki M, Riehl T, Baselga J, Hyman DM. Vemurafenib for BRAF V600-Mutant Erdheim-Chester Disease and Langerhans Cell Histiocytosis: Analysis of Data From the Histology-Independent, Phase 2, Open-label VE-BASKET Study. JAMA Oncol. 2018;4:384-388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 286] [Article Influence: 47.7] [Reference Citation Analysis (0)] |
| 32. | Diamond EL, Durham BH, Ulaner GA, Drill E, Buthorn J, Ki M, Bitner L, Cho H, Young RJ, Francis JH, Rampal R, Lacouture M, Brody LA, Ozkaya N, Dogan A, Rosen N, Iasonos A, Abdel-Wahab O, Hyman DM. Efficacy of MEK inhibition in patients with histiocytic neoplasms. Nature. 2019;567:521-524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 230] [Cited by in RCA: 261] [Article Influence: 43.5] [Reference Citation Analysis (0)] |
| 33. | Yi X, Han T, Zai H, Long X, Wang X, Li W. Liver involvement of Langerhans' cell histiocytosis in children. Int J Clin Exp Med. 2015;8:7098-7106. [PubMed] |
| 34. | Braier J, Ciocca M, Latella A, de Davila MG, Drajer M, Imventarza O. Cholestasis, sclerosing cholangitis, and liver transplantation in Langerhans cell Histiocytosis. Med Pediatr Oncol. 2002;38:178-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 72] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 35. | Tang Y, Zhang Z, Chen M, Ju W, Wang D, Ji F, Ren Q, Guo Z, He X. Severe sclerosing cholangitis after Langerhans cell histiocytosis treated by liver transplantation: An adult case report. Medicine (Baltimore). 2017;96:e5994. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |