Published online Dec 28, 2020. doi: 10.4329/wjr.v12.i12.272
Peer-review started: October 28, 2020
First decision: November 20, 2020
Revised: December 7, 2020
Accepted: December 16, 2020
Article in press: December 16, 2020
Published online: December 28, 2020
Processing time: 60 Days and 23.3 Hours
With each day the number coronavirus disease 2019 (COVID-19) cases continue to rise rapidly and our imaging knowledge of this disease is expeditiously evolving. The role of chest computed tomography (CT) in the screening or diagnosis of COVID-19 remains the subject of much debate. Despite several months having passed since identifying the disease, and numerous studies related to it, controversy and concern still exists regarding the widespread use of chest CT in the evaluation and management of COVID-19 suspect patients. Several institutes and organizations around the world have released guidelines, recommendations and statements against the use of CT for diagnosing or screening COVID-19 infection and advocating its use only for those cases with a strong clinical suspicion of complication or an alternate diagnosis. However, these guidelines and recommendations are in disagreement with majority of the widely available literature, which strongly favour CT as a pivotal tool in the early diagnosis, management and even follow-up of COVID-19 infection. This article besides comprehensively reviewing the current status quo on COVID-19 disease in general, also writes upon the current consensus statements/recommendations on the use of diagnostic imaging in COVID-19 as well as highlighting the precautions and various disinfection procedures being employed world-wide at the workplace to prevent the spread of infection.
Core Tip: As per the recommendations of various national and international organizations, chest imaging [chest radiograph/computed tomography (CT)] is not indicated as a screening tool in asymptomatic coronavirus disease 2019 (COVID-19) patients or in those with mild respiratory features unless the patients are at risk for disease progression. However, these suggestions remain a subject of debate as many studies have shown that especially chest CT can identify lung findings in patients with negative reverse transcriptase polymerase chain reaction or even before the test becomes positive. An added advantage with chest CT is that, based on the lung findings, CT can classify patients into mild, moderate or severe disease category, as patients with more extensive pulmonary disease may benefit from follow-up CTs and potential management options, taking into consideration that the disease has the potential to cause fibrosis. Several chest CT findings typical for COVID-19 have been reported, with the most common one being multifocal ground glass opacities or consolidation usually in a bilateral, peripheral, subpleural, lower lobe and posterior distribution.
- Citation: Mathew RP, Jose M, Jayaram V, Joy P, George D, Joseph M, Sleeba T, Toms A. Current status quo on COVID-19 including chest imaging. World J Radiol 2020; 12(12): 272-288
- URL: https://www.wjgnet.com/1949-8470/full/v12/i12/272.htm
- DOI: https://dx.doi.org/10.4329/wjr.v12.i12.272
In December 2019, an outbreak of pneumonia of unknown cause occurred in Wuhan, the capital of Hubei province, a city with a large population of approximately 11 million people in central China. The outbreak was initially attributed to the Wuhan Huanan (South China) Seafood Wholesale Market, that sells a variety of seafood and exotic meat. Within a month the disease spread throughout the country. By means of deep sequencing analysis, the pathogen was identified as a novel enveloped RNA beta-corona virus, initially named as 2019 novel corona virus (2019-nCoV)[1,2]. By January 30, 2020, the World Health Organization (WHO) declared the virus outbreak a global public health emergency and on February 11, 2020, the WHO announced a new name for the pandemic disease caused by this pathogen: Corona Virus Disease 2019 or coronavirus disease 2019 (COVID-19). In addition, the Coronavirus Study Group of the International Committee on Taxonomy of Viruses renamed 2019-nCoV, as severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2)[3]. COVID-19 was declared a pandemic on March 11, 2020.
Hence, we conducted a review of current literature, focusing on the epidemiology, patient demographics, clinical symptoms of COVID-19 while emphasizing on the current status of chest imaging in the diagnosis, management and follow up of these patients.
Prior to December 2019, there were only 6 coronavirus species that produced human infection: Two belonging to the Alphacoronavirus genus [human coronavirus 229E (HCoV-229E) and HCoV-NL63] and four belonging to the Betacoronairus genus [HCoV-OC43, HCoV-HKU1, Middle East respiratory syndrome (MERS-CoV) and SARS-CoV]. As of December 2019, there are now 7 species, with the newest member being the SARS-CoV-2[4]. The SARS-CoV-2 shares close to 80% sequence identity with SARS-CoV and about 50% with MERS-CoV. SARS-CoV-2 is currently thought to originate from a bat host, as studies have shown that the SARS-CoV-2 is closely related to the coronaviruses seen in Chinese horseshoe bats, which are the natural reservoirs of SARS-CoV[5-7]. Additionally, an intermediary animal host in close contact with human could also have played a role in the disease transmission, i.e., the virus jumped from an intermediary host (e.g., SARS-CoV is believed to have been transmitted from bats to humans via Himalayan civets, Chinese ferret badgers and raccoon dogs sold at the wet markets of Guangdong)[7].
Human-to-human transmission of SARS-CoV-2 has been confirmed by droplets, contact and fomite. As of now, no definite evidence exists for intrauterine vertical transmission[8]. As per current literature, COVID-19 has a median incubation period of 5 d, with 97.5% of the infected patients developing symptoms within 11-12 d[9]. The case fatality rate for COVID-19 is influenced by age, varying from 0.3 deaths per 1000 cases in patients aged 5-17 years, to approximately 305 deaths per 1000 cases among patients aged ≥ 85 in the United States. The cases fatality rate among patients hospitalized in the intensive care unit (ICU) is up to 40%[10]. The basic reproduction number or R0 (pronounced “R naught”) represents the transmissibility of a virus, and indicates the average number of new or secondary infections caused by an infected person in a totally susceptible population. It is used to measure the transmission potential of a communicable disease. For disease with an R0 < 1, the disease transmission is likely to die out, while for those with R0 > 1, the number of infections is likely to increase exponentially. The estimated basic reproduction number or R0 (pronounced “R naught”) for COVID-19 is around 2-3[11]. Currently, the global number of confirmed cases stands close to 42 million with over 1 million confirmed deaths worldwide[12].
SARS-CoV-2 affects males more than females, as the latter tend to have reduced susceptibility to viral infections, which could be attributed to the X chromosome and sex hormones, which play an important role in innate and adaptive immunity[13].
Several studies have shown that, at the time of admission, more than 80% of symptomatic patients tend to clinically present with fever and/or cough[14], with or without additional symptoms such as - shortness of breath (30%), muscle ache, confusion, headache, sore throat, rhinorrhoea, anosmia, chest pain, diarrhoea, nausea and vomiting, sputum production and hemoptysis; while > 90% will have more than one sign or symptom[14]. Also, > 50% will have underlying co-morbidities such as hypertension, cardiovascular disease, cerebrovascular disease and diabetes[14], and studies have shown that SARS-CoV-2 will more likely infect older aged males with co-morbidities leading to respiratory failure from severe alveolar damage[14]. Additionally, patients may have normal or low white blood cell counts, lymphopenia or thrombocytopenia and increased C-reactive protein levels[8,14]. In short, any individual having a fever with upper respiratory tract symptoms with lymphopenia or leukopenia, especially with a close contact or travel history should be suspected to have SARS-CoV-2 infection. Elderly patients with underlying risk factors are more prone to COVID-19 infection and have poorer clinical outcome. Some of these risk factors include-age > 65 years, cardiovascular disease, diabetes, chronic lung disease, hypertension and immunocompromised individuals[14,15].
Approximately 20% of the patients with COVID-19 and 41% who are hospitalized progress to acute respiratory distress syndrome[16]. Other reported complications in COVID-19 patients include myocarditis, cardiomyopathy, ventricular arrhythmias, hemodynamic instability[17,18], acute cerebrovascular disease and encephalitis[19,20]. Vascular thromboembolic events may be seen in 10%-25% of the hospitalized patients[21,22], which further increases to 31%-59% in ICU patients.
There are two broad categories of SARS–CoV-2 tests: Those that detect the virus itself and those that detect the host’s response to the virus. The most widely used and accepted test for detecting SARS-CoV-2, is by identifying the viral RNA through nucleic acid amplification, using real time reverse transcription polymerase chain reaction (RT-PCR) assay. Samples are taken by swabs from the nasopharynx and/or oropharynx, with the latter considered less sensitive than the former. For patients with pneumonia, in addition to the above-mentioned samples, lower respiratory tract secretions (e.g., sputum and bronchoalveolar lavage fluid) are also collected and tested. The detection rates in each sample type will vary from patient to patient and can change over the course of the patient’s illness. A negative RT-PCR does not rule SARS-CoV-2 infection[23]. The second available test is serology, i.e., identifying IgM, IgA, IgG or total antibodies (typically in blood). However, factors such as host immunity and time can influence the development of antibodies, and studies have shown that patients with SARS-CoV-2 seroconvert between days 7 and 11 following exposure to the virus. Hence, because of this delay, serology is not useful in the setting of acute illness[23].
RT-PCR remains the standard reference for the diagnosis of COVID-19. However, one needs to keep in mind that the test is limited by sample collection, patient viral load, transportation and variation in kit performance from different manufacturers. RT-PCR kit availability is also an issue in some centres. In addition, although the RT-PCR tests have a high specificity, studies have shown that the sensitivity is only around 30%-70% at initial presentation[24,25]. Therefore, a negative RT-PCR does not rule out COVID-19 disease, necessitating the need for repeated tests. Additionally, such patients pose a significant risk of unchecked transmission of infection to their community given the high contagious nature of the virus[23-25].
Other samples that can be collected include-fibrobronchoscope brush biopsy, stool and urine. Wang et al[26] evaluated 1070 specimens collected from 205 patients with COVID-19 to investigate the biodistribution of SARS-CoV-2. The found that bronchoalveolar lavage fluid specimens had the highest positive rates (93%), followed by sputum (72%), nasal swabs (63%), fibrobronchoscope brush biopsy (46%), pharyngeal swabs (32%), faeces (29%) and blood (1%). None of the urine samples tested positive.
The WHO has provided statements on terminologies that may be used as guidance when adopting various measures including infection control for COVID-19[27], and these include: (1) Suspect case of COVID-19: (a) Any patient with an acute respiratory illness (fever with clinical signs or symptoms of respiratory disease e.g., cough, shortness of breath, etc.) along with a history of travel or residence in a location with reported community spread of COVID-19 in the last 2 wk prior to onset of symptoms; (b) Any patient with an acute respiratory illness and who has been in contact with a confirmed or probable COVID-19 case in the last 2 wk prior to onset of symptoms; or (c) Any patient with severe acute respiratory illness requiring hospitalization and in the absence of an alternative diagnosis explaining the clinical presentation; (2) Probable case of COVID-19 where the laboratory test result of a suspected patient is inconclusive or a suspect patient for whom testing could not be performed for various reasons; and (3) Confirmed case of COVID-19 refers to any patient with a positive RT-PCR test irrespective of the signs and symptoms is a confirmed case.
The diameter of the coronavirus particle is extremely small, about 60-140 nm; and as a result, the virus can easily reach the terminal lung structures such as alveolar septum, alveolar wall, and the interlobular septum, causing lymphocytic infiltration and interstitial edema[28]. Therefore, imaging plays an important role in detecting changes in the lungs.
Chest radiographs: Despite their high specificity, chest radiographs (CXRs) are less sensitive than computed tomography (CT) and RT-PCR for detecting COVID-19 infection related opacities[8,29]. Choi et al[29] evaluated 20 pairs of CT and CXRs done on the same day from 17 patients diagnosed with COVID-19, with one of their objectives being, to assess the visibility of COVID-19 lesions on CXRs. They found that CXRs had a sensitivity and specificity of 25% and 90%, respectively. In another study involving 64 RT-PCR confirmed COVID-19 patients, base line CXRs had a sensitivity of 69% compared to 91% for initial RT-PCR[30]. As a result, experts suggest that CXRs should not be recommended as the first line imaging modality for evaluating COVID-19 disease[8,29-31].
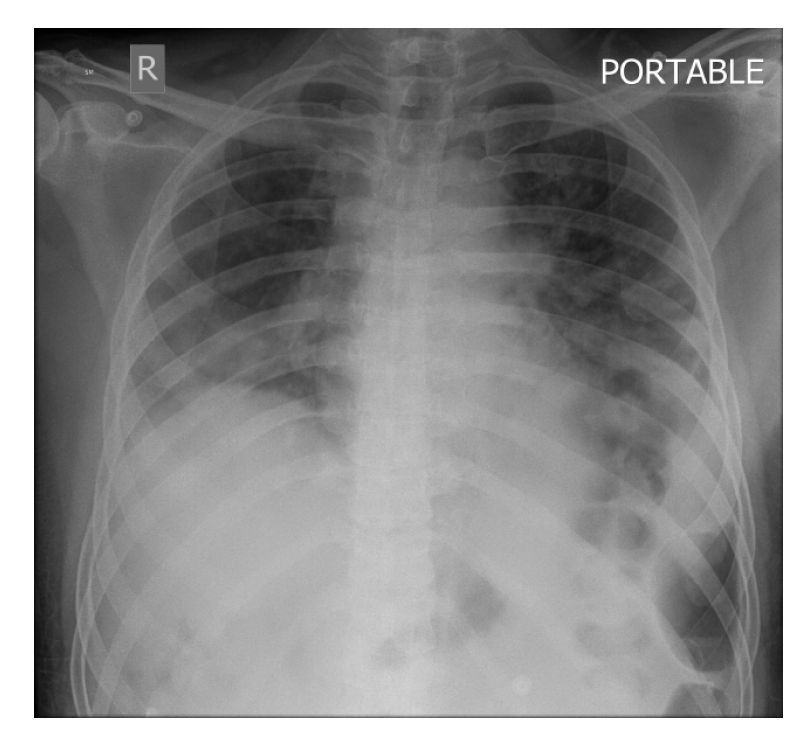
Yoon et al[31] compared CXRs of 9 patients with COVID-19 pneumonia with chest CT. They found that a considerable proportion of COVID-19 patients can present with “normal” appearing CXRs. In fact, a suspicious area of ground glass opacity and focal atelectasis on CXRs of different 2 patients, turned out to be prominent breast tissue on CT. They concluded CXRs when positive may show patchy or diffuse asymmetric airspace infiltrates/opacities with peripheral predilection[8,31,32]. In severe cases, multiple alveolar consolidation may be seen in both lungs (Figure 1). Progression to critical disease can manifest as a “white lung” with a small amount of pleural effusion[32].
CT: Chest CT is now considered as the first-line imaging modality in highly suspected cases of COVID-19 pneumonia. CT has demonstrated its ability to identify COVID-19 patients with negative RT-PCR. In addition, it is also useful for monitoring patients during treatment[28,33]. CT has a reported sensitivity and specificity ranging from 60%-98% and 25%-53% respectively for COVID-19, while the positive and negative predictive values are estimated at 92% and 42% respectively. As a result of its low negative predictive value CT may not be of value as a screening tool for COVID-19, especially in the early stages of the disease[34].
In one of the largest studies till date, involving 1014 COVID-19 patients, Ai et al[35] investigated the diagnostic value and consistency of chest CT in comparison to RT-PCR in patients with COVID-19 pneumonia. They found that CT had a sensitivity of 75% in patients with negative RT-PCR tests, while RT-PCR had a positivity rate of only 59%. They also identified that by analysing serial RT-PCR assays and chest CTs, the mean interval between the initial negative to positive RT-PCR results was 5.1 ± 1.5 d, with about 60%-93% of the patients have an initial positive chest CT consistent with COVID-19 disease, much before the initial positive RT-PCR test results. Additionally, they also found that chest CT played a crucial role in follow up, as 42% of the cases showed improvement on follow up CT, much before the RT-PCR results turned negative[35]. However, one must keep in mind that COVID-19 can present with atypical patterns in immunosuppressed patients as well as those with underlying lung pathologies such as fibrosis, diffuse emphysema, etc. Ironically, close to 50% of patients with COVID-19 can have normal chest CT examinations 2 d after onset of symptoms, and conversely, patients with normal chest CT may show sudden severe pneumonic changes within 2 d of start of symptoms[36].
Studies have shown that initial chest imaging with CT can show abnormality in 85%-97% of patients with COVID-19 pneumonia, with close to 75%-83% of the patients having bilateral lung disease during the acute phase, manifesting as subpleural and peripheral based areas of ground glass opacities (GGO) and consolidation mostly involving the lower lobes[32,34,36]. Elderly patients with progressive consolidation on serial chest CTs suggest poor prognosis. Besides its role in the acute phase of the disease, chest CT is also recommended for follow-up of patients recovering from COVID-19 pneumonia[37]. As per the 6th version of the diagnosis and treatment program guidelines published by the National Health Commission of China, one of the diagnostic criteria for COVID-19 is based on imaging features, as interpreted by radiologists. In fact, in a study conducted by Li et al[38], involving 51 patients with confirmed COVID-19 pneumonia, the missed diagnosis rate for CT was < 4%. A meta-analysis conducted by Adams et al[39] showed that 10.6% of symptomatic RT-PCR confirmed COVID-19 patients had normal chest CT, showing that the real sensitivity for chest CT could be much lower than that reported by the various initial studies, further reinforcing the fact that a normal CT study does not rule out a COVID-19 infection.
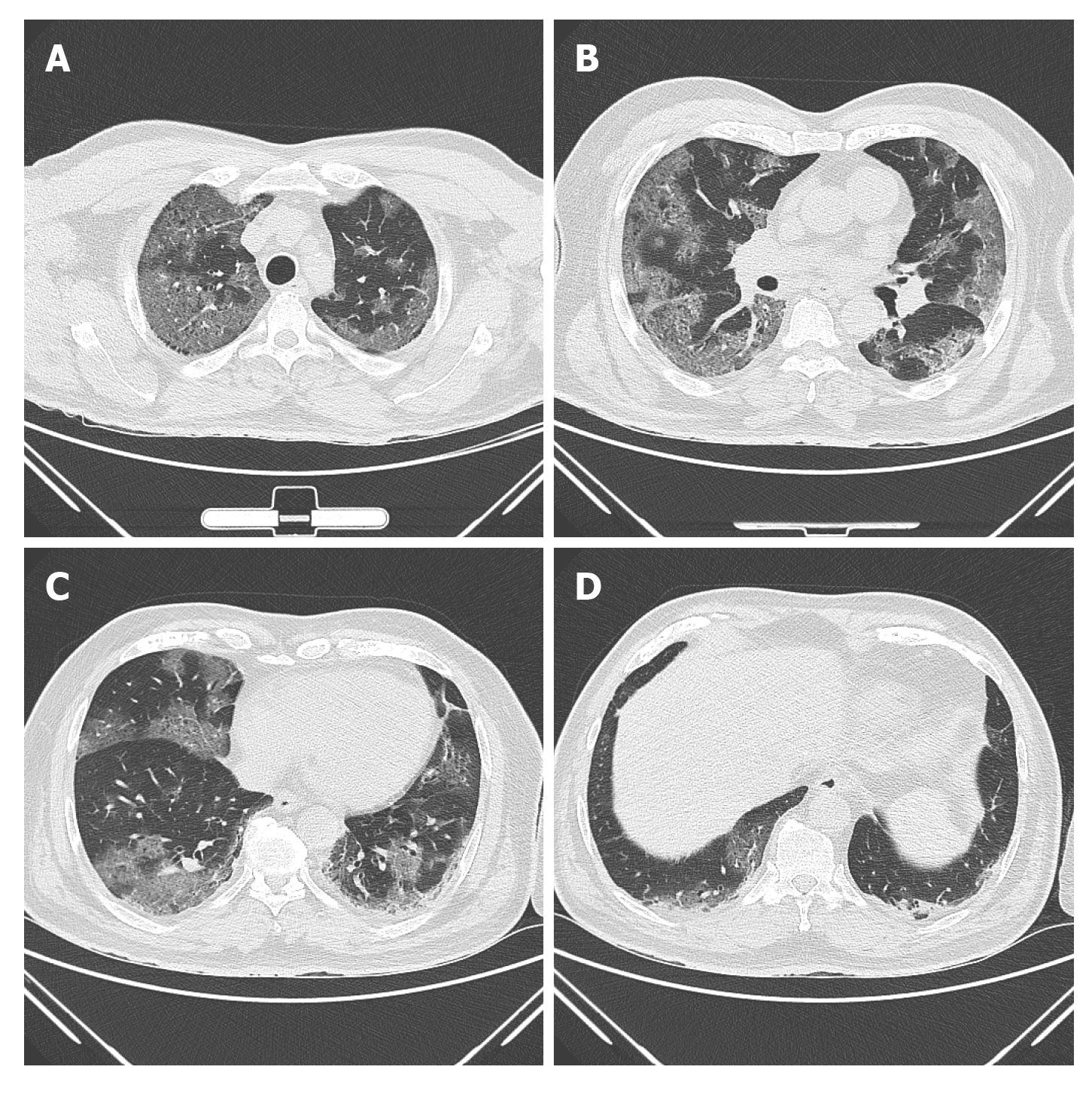
GGO and consolidations are the two main lesions seen in COVID-19 patients, and these can be single or multiple, and unilateral or bilateral. Although most of these lesions are predominantly subpleural and peripheral based (Figure 2A-D), they can also occur along the bronchovascular bundles. Additional, but uncommon findings that have been observed on chest CT include-consolidation with vascular enlargement/thickening, interlobular septal thickening or reticulations appearing as crazy-paving pattern, traction bronchiectasis, positive air bronchogram sign, air-trapping and “reversed halo” sign[33,37,38,40], and discrete nodules with or without a CT halo sign[35,38,40]. CT features of disease progression are increasing density of consolidation, extension of disease to upper lobes and increasing number of GGOs. The identification of fibrosis and resolution of GGOs or consolidation on follow up CT indicates improvement[41]. Features such as cavitation[41], bronchial wall thickening, mucoid impaction, pneumothorax, lymphadenopathy and pleural effusion are very rare and considered as atypical findings[13,25,34,40-42].
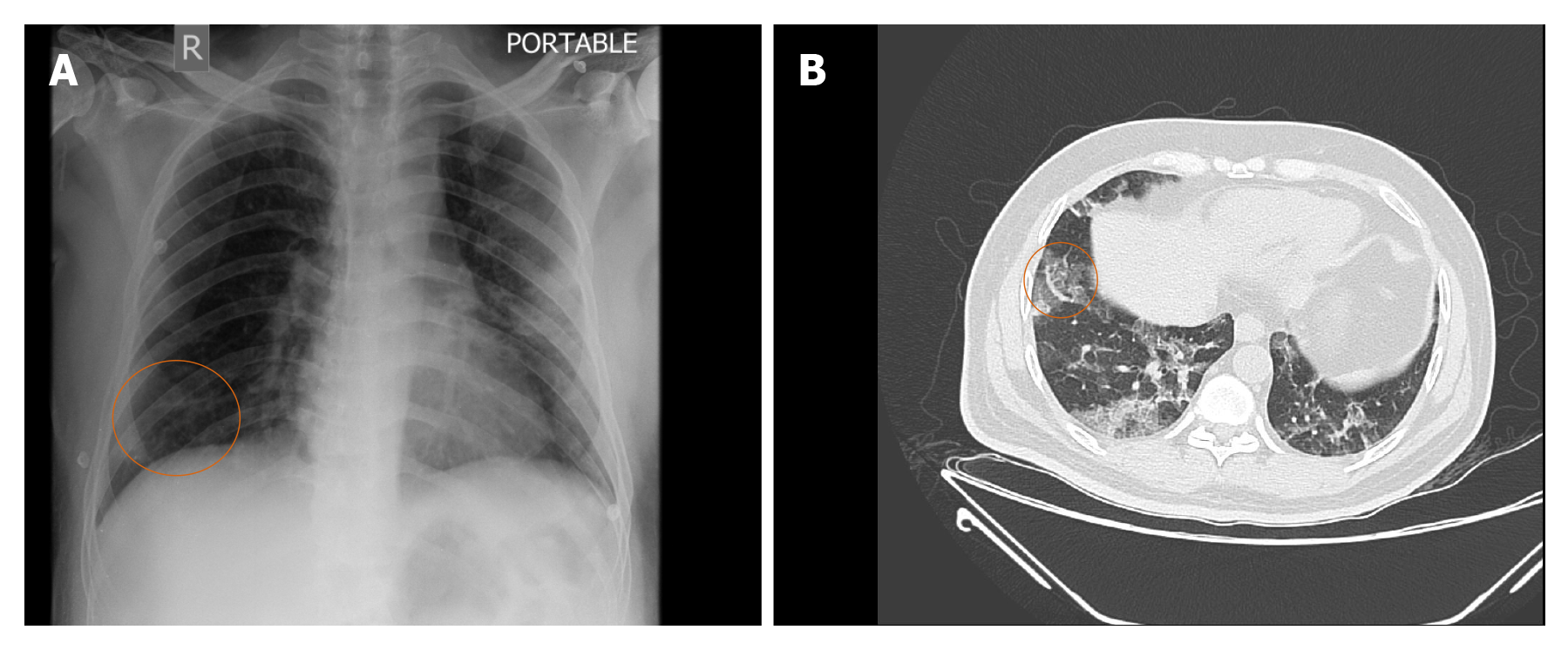
Pulmonary vascular manifestations on CT: Vascular pathology plays an important role in the pathophysiology of COVID-19 as indicated by the frequent presence of medium to small vessel enlargement and regional mosaic perfusion patterns on the CT of COVID-19 patients. Medium to small vessel dilatation is not just confined to the areas of the affected lung and most commonly involves the subpleural vessels[43]. Lang et al[43] retrospectively evaluated the CT pulmonary angiography (CTPA) of 48 patients with confirmed COVID-19 for pulmonary vascular abnormalities. Of the 48 patients enrolled in their study, a subset of 25 patients underwent dual energy CT (DECT). On DECT a mosaic perfusion pattern was observed in 96% of the cases, while regional hyperaemia with overlapping areas of pulmonary opacity or immediately peripheral to the opacities were seen in 52% of the cases. Opacities associated with corresponding oligemia were seen in 96% and hyperaemic halo in 36% of the patients. On CTPA they found that 15% of the patients had pulmonary emboli. Dilated vessels were seen in 85% of the cases with 78% having vessel enlargement within the lung opacity and 55% having vessel enlargement outside of the lung opacities. A plausible explanation attributed to this finding by authors was an inflammatory mediated vasodilatory response to COVID-19 infection leading to an intrapulmonary shunting towards the affected areas of reduced gas exchange and ventilation perfusion mismatch[43], although other experts[44] felt it was due to an underlying pulmonary thrombotic angiopathy. The presence of this dilated, tortuous, branching, non-tapering, peripheral vessels on CT has been termed by some as the vascular “tree-in-bud” pattern (Figure 3A and B). The recognition of vascular tree in bud pattern in COVID-19 is unique, as generally the lung parenchymal tree-in-bud pattern that we commonly refer to on CT is usually associated with peripheral airway disease secondary to an underlying infectious etiology, unlike the vascular pattern that has only been documented in the context of pulmonary tumor thrombotic microangiopathy. Patel et al[44] in their study showed that the presence of vascular tree-in-bud pattern is strongly associated with a longer duration of hospitalization (≥ 10 d) and ventilation, and hence may prove to be a useful prognostic biomarker for poor outcomes in patients with COVID-19.
Current role of chest CT in COVID-19: Dangis et al[45] recently investigated whether low dose (submillisievert) chest CT could rapidly, accurately and reproducibly stratify patients with COVID-19. They found that when compared to RT-PCR, low dose chest CT had excellent sensitivity (86.7%), specificity (93.6%), positive predictive value (91.1%), negative predictive value (90.3%) and accuracy (90.2%) in diagnosing COVID-19 patients in the emergency department (ED). These values improved when the low dose CT was performed in patients having symptoms of more than 2 d duration. They noted that in patients with positive chest CT findings, the likelihood of COVID-19 disease increased from 43.2% (pre-test probability) to approximately 91% (post-test probability), while in those with no lung findings on CT, the likelihood of disease reduced to 9.6% for all patients and to 3.7% in patients with symptoms for > 48 h. When compared to RT-PCR, the median time for CT image acquisistion to report was 25 min (interquartile range: 13-49 min)[45]. They concluded from their study that in addition to rapidly and accurately assessing COVID-19 patients in the ED, chest CT has the added advantage of offering alternate diagnosis in the subset of patients presenting to the ED during the current pandemic[45].
Only limited literature exists on the potential role of contrast enhanced CT (CECT) in COVID-19 patients. Based on recent reports of COVID-19 infection being associated with coagulopathy and thrombotic complications[46], Grillet et al[47] routinely performed CECT in COVID-19 patients who presented to their institution with severe symptoms, and retrospectively investigated the lung parenchyma of 100 such cases and found that pulmonary embolism (PE) was quite frequent (23%) in this subset of patients. A plausible explanation being that COVID-19 patients with severe symptoms tend to be males and often end up on mechanical ventilation, both of which are recognized risk factors for developing PE[48]. Similarly, Léonard-Lorant et al[48], evaluated 106 COVID-19 patients with CTPA and found that 30% of the patients had acute PE on CTPA. They also noted that COVID-19 positive patients with PE had higher D-dimer levels than those without PE, and were also more likely to require ICU management[48]. Although, both of these studies were limited by their preliminary nature, retrospective pattern, as well as the small sample size, their studies highlighted the possible role of CECT in COVID-19 patients presenting with severe symptoms.
However, despite several studies as mentioned above proving the potential application of CT in the evaluation and management of COVID-19 infection, confusions and concerns still exist. As the number of cases keep increasing on a daily basis our knowledge of the imaging features of COVID-19 are expeditiously evolving. Kim et al[49], recently conducted a meta-analysis to evaluate the diagnostic performance of chest CT and RT-PCR for COIVD-19. They found the pooled sensitivity for chest CT was 94% and that for RT-PCR was 89%, while the pooled specificity for CT was only 37%. As a result of the low specificity for chest CT, a wide discrepancy was identified in the PPV between chest CT and RT-PCR in COVID-19 low prevalence countries. They noted that in countries with a prevalence of less than 10%, the PPV of RT-PCR was more than ten times than that of chest CT. Their findings confirmed that the use of chest CT in COVID-19 low prevalence regions can lead to a large number of false positive results, which can ultimately lead to unwarranted additional imaging, unnecessary radiation exposure, escalation of medical costs, increased workload on medical personnel, difficulties with disinfection procedures and not to mention patient anxiety. The majority of the studies demonstrating excellent diagnostic performance of chest CT for COVID-19 were from China, where the pooled prevalence was 39%. They also noted that factors such as distribution of disease severity, proportion of patients with co-morbidities and the number of asymptomatic patients could significantly affect the sensitivity of chest CT. They concluded from their investigation that chest CT for the primary screening or diagnosis of COVID-19 would not be useful in low prevalence regions due to the high rate of false positives[49]. Similar concerns were also raised by Hope et al[50]. They argued that since CT findings of COVID-19 infection were not specific, similar results would also be seen in other viral pneumonia epidemics such as influenza, and also pointed out that CT would have a low PPV unless the prevalence of COVID-19 was high. They strongly felt that CT added no diagnostic value and could in turn give false reassurances if the images were normal. More importantly, they were totally opposed to categorizing CT as a pivotal tool for COVID-19 diagnosis and felt that it was a distraction and even dangerous.
Recently, several consensus statements and recommendations have been released by various international organizations on the role of imaging (CXR/chest CT) in suspected COVID-19 patients, and these have been elaborated in Table 1[51-54].
| Fleischner Society Consensus Statement |
| Mild respiratory diseases are those with no evidence of pulmonary dysfunction or damage (e.g., no or minimal dyspnea, no hypoxemia, etc.) |
| Moderate-severe respiratory disease: Evidence of significant pulmonary dysfunction or damage (e.g., moderate-severe dyspnea, hypoxemia, etc.) |
| Main Recommendations: |
| Asymptomatic COVID-19 patients: Imaging is not routinely indicated for screening |
| Mild respiratory features of COVID-19: Imaging is not indicated unless they at risk for disease progression, i.e., the patient has underlying risk factors (refer text) |
| Moderate-severe respiratory features of COVID-19 regardless of COVID-19 test result: Imaging is indicated |
| Patients with COVID-19 and with evidence of worsening respiratory symptoms/signs: Imaging is indicated |
| Healthcare facility with limited resources including limited access to CT: CXR may be done in this category of patients. However, CT may be warranted in those cases where there is evidence of respiratory worsening |
| Additional recommendations:No role for daily CXRs in stable intubated patients with COVID-19 |
| CT is indicated in those patients that have recovered from COVID-19 but show functional impairment and/or hypoxemia |
| Patients found to have incidental finding on CT suggestive of COVID-19 should undergo a COVID-19 testing |
| American College of Radiology (ACR) Recommendations |
| Viral testing (RT-PCR) remains the only accepted method for diagnosing COVID-19 disease. Confirmation with viral testing is required even if the CXR or CT findings are suggestive of the disease |
| CXR or CT is not currently recommended to diagnose COVID-19 |
| The findings on chest imaging in COVID-19 patients are not specific and can overlap with other diseases such as influence, H1N1, SARS and MERS; thereby limiting the specificity of chest CT |
| British Society of Thoracic Imaging (BSTI) Guidelines |
| No role for chest CT in the diagnosis of COVID-19 unless, the patient is seriously ill or if PCR testing is unavailable |
| The current position is that there is no recommended use of CT, beyond “routine clinical care” |
| Imaging (CXR or CT) may be useful in guiding patient management decisions, complications or when looking for alternate diagnosis |
| Canadian Association of Thoracic Radiology/Canadian Association of Radiologists Consensus Statement |
| RT-PCR assay remains the gold standard for diagnosis |
| Chest radiograph: Should not be used to exclude COVID-19 infection. CXR is often normal early in the disease, and even when present the imaging features are non-specific for COVID-19. CXRs are most useful when an alternate diagnosis is suspected e.g., pneumothorax, pulmonary edema, large pleural effusions, lung mass or lung collapse. The study needs to be limited to those in whom the findings are expected to change the management in order to reduce the risk of transmission of the virus to healthcare workers |
| Chest CT: Similar to CXR, CT should only be performed if the results are expected to influence patient management. CT should not be used to routinely screen patients for possible COVID-19. Although not limited to, potential indications for CT chest in COVID-19 include detection of intrathoracic complications (e.g., pulmonary embolism), immunosuppressed or high-risk patients with suspected respiratory infection and a negative chest radiograph, initial negative RT-PCR result and normal CXR but high clinical suspicion for COVID-19 infection or clinical deterioration. In patients requiring urgent intervention or procedure, it is impractical to wait 24 h for a swab result to decide on the level of PPE required. In such situations CT chest may be used as additional tool to guide the use of PPE. Although a negative CT does not completely rule out COVID-19 infection, it can help stratify the patients into low or high-risk category |
The Radiological Society of North America (RSNA) has provided reporting guidelines for chest CT findings suggestive of COVID-19 pneumonia with an aim of helping the interpreting radiologist to recognize COVID-19 findings, reduce reporting variability, decrease unambiguity in the chest interpretations and to improve the communication with the patient’s referring clinician. Four categories for standardized COVID-19 reporting have been proposed by the RSNA and these have been explained in detail in Table 2[55].
| COVID-19 pneumonia imaging classification | CT findings | Rationale |
| Typical appearance | Peripheral, bilateral, ground-glass opacities with or without consolidation or a “crazy-paving” pattern | CT imaging features most specifically associated with COVID-19 |
| Multifocal ground-glass opacities of rounded morphology with or without consolidation or visible intralobular lines (“crazy-paving” pattern) | ||
| Reverse halo sign or other findings of organizing pneumonia | ||
| Indeterminate appearance | Absence of typical features + the presence of the following features: Multifocal, diffuse, perihilar, or unilateral ground-glass opacity with or without consolidation lacking a specific distribution and that are nonrounded or non-peripheral | Non-specific CT imaging features of COVID-19 pneumonia |
| Few small ground-glass opacities, with a nonrounded and non-peripheral distribution | ||
| Atypical appearance | Absence of typical or indeterminate features + the presence of the following features: Isolated lobar or segmental consolidation without ground-glass opacities; discrete small nodules (centrilobular, “tree-in-bud” appearance); lung cavitation; smooth interlobular septal thickening with pleural effusion | Rarely associated with or not reported features of COVID-19 |
| Negative for pneumonia | No features of pneumonia on chest CT | No features of pneumonia |
For prognostic purposes some have proposed the use of a chest CT severity score as a standardized evaluation of the degree of lung involvement in COVID-19. However, such prediction models are at a high risk for bias and poorly reported, making their performance sub-optimal. Hence, it is not advised to use any prediction models in current clinical practice[55].
Despite the suggestions by most national and international organizations to limit the use of the chest CT in COVID-19 patients to those with severe disease and to patients in whom an alternate diagnosis is suspected, the recommendations remain controversial. Several studies have shown that chest CT is useful for the follow-up of COVID-19 patients to assess disease recovery or progression. Fu et al[56] evaluated the follow-up chest CTs of 317 COVID-19 patients who had initial negative chest CT at the time of active disease (i.e., RT-PCR positive). They found that 34.5% of the patients developed new pulmonary lesions mainly in the form of spherical/patchy GGO, mostly in the left lower lobe, and among these patients, 60% developed new or aggravated symptoms with reduced lymphocyte count. They concluded from their study, that new pulmonary lesion may develop in COVID-19 patients during the period of treatment, and hence chest CT is necessary for monitoring the disease especially in those patients with worsening symptoms or laboratory indicators. Using follow-up chest CT, Zhong et al[57] evaluated the dissipation and outcome of pulmonary lesions identified on the initial chest CT examinations of 52 patients who recovered from moderate and severe COVID-19 disease (33 moderate and 19 severe cases). The time interval to the first follow-up CT was 29 d to 62 d after onset of symptoms and 11 d to 34 d following discharge. Follow-up CTs of 14 patients (42.4%) with moderate disease and 2 patients (10.5%) with severe disease returned to normal, while the remaining 36 patients (69.2%) had residual pulmonary lesions which included-inhomogeneous low-density GGO (91.67%); fibrous bands (52.78%) presenting as strips of variable thickness and length, occasionally adhering to the pleura and pulling it; consolidation (13.89%) presenting as small or dissipated hyperdense opacities when compared to prior imaging; interlobular and/or intralobular septal thickening (8.3%); subpleural reticulations/bands (13.9%) appearing as linear shadows 1 cm below and parallel to the pleura and traction bronchiectasis (11%) presenting as localized columnar extension of the bronchi to the periphery of the lungs. Guan et al[58] evaluated the follow-up chest CTs of 54 patients with COVID-19 (10 recovered patients and 42 patients with progressed disease), the mean interval period between the initial and follow-up CT being 7.82 ± 3.74 d. Two of the 54 cases with COVID-19 showed no pneumonia on follow-up CT. Of the remaining 52 cases, the most common follow-up finding was GGO (96.1%, 50/52), followed by crazy-paving pattern (88.5%, 46/52), consolidation (78.8%, 41/52), irregular lines (71.1%, 37/52), and air bronchogram sign (69.2%, 36/52). Three cases developed pleural effusion. The follow-up CTs of patients belonging to the recovery group, showed reduced crazy-paving pattern, consolidation and air bronchogram sign, while irregular lines were slightly increased. Of the findings, the reduction in the air bronchogram sign was statistically significant. Patients belonging to the progression group showed an increase in all the above findings, with the increase in consolidation being statistically significant. Their study proved that recognition of CT manifestations (irrespective of the patient’s symptoms) can help stage the disease severity thereby aiding in the clinical diagnosis and management. Tabatabaei et al[59], evaluated 52 cases of COVID-19 pneumonia with at least two chest CTs and mean 3-mo interval between the initial and follow-up CT, to assess the rate of complete resolution, and determine the individuals at risk for residual abnormalities. They found that the most common lung findings in patients with residual disease was GGO (54.5%), followed by mixed ground glass with subpleural parenchymal bands (31.8%) and pure parenchymal bands (13.7%). They found that the extent of lung involvement on initial CT can predict the outcome of pulmonary findings in a mid-term follow-up, and hence, patients with more extensive pulmonary disease may benefit from follow-up CTs and potential management options, taking into consideration that the disease has the potential to cause fibrosis.
Fluorodeoxyglucose positron emission tomography/CT: Only limited literature exists describing the chest findings on fluorodeoxyglucose positron emission tomography/CT (FDG-PET/CT) in suspected and confirmed COVID-19 patients. Currently, this imaging modality is not recommended for the diagnosis or management of COVID-19 disease, and some authorities feel it increases the risk of spread of disease due to the long duration required for the imaging acquisition[60]. Lung lesions of COVID-19 patients on FDG-PET/CT tend to be FDG avid[61,62] and can be sometimes be confused with lung malignancy[62]. However, a few experts believe the FDG-PET/CT may have role in COVID-19 disease, as the diagnostic tool has a proven sensitivity to detect and monitor inflammatory diseases (e.g., viral pneumonia), monitor the disease progression as well as treatment outcomes[63]. COVID-19 pneumonia lesions demonstrate high FDG uptake, and the high uptake may indicate lesions requiring a longer time to heal, which can be positively correlated with erythrocyte sedimentation rate values[61,63]. Additionally, asymptomatic patients with COVID-19 pneumonia can present as incidental lesions on nuclear medicine studies being done for oncologic indications in regions with a high COVID-19 prevalence[64].
Artificial intelligence (AI) experts believe that by using machine learning algorithms, based on the available large-scale information of COVID-19 patients, data can be integrated and analysed to better understand the pattern of disease spread, improve diagnostic speed and accuracy, develop new and effective treatment and to potentially identify the most susceptible individuals based on their genetic and physiological make up. Some of the areas where AI have been successfully used in COVID-19 disease include: (1) Taxonomic classification of COVID-19 genomes; (2) CRISPR-based COVID-19 detection assay; (3) Survival prediction of patients with severe COVID-19 disease; and (4) Identifying potential drugs against COVID-19[65]. More recently Li et al[66] proposed a three-dimensional (3D) deep learning model, referred to as COVID-19 detection neural network (COVNet), to diagnose COVID-19 disease using chest CT. Community acquired pneumonia and other non-pneumonia were included in their study to test the robustness of the model. They found that their model achieved high sensitivity and high specificity of 90% and 96% respectively in detecting COVID-19. Therefore, AI algorithms may also prove useful in providing new insights for COVID-19 differential diagnosis. Additionally, AI can help in risk prioritization, reduce turnaround time, and ease the burden of the radiologists and enhance rapid triaging.
Based only on imaging, it is difficult to differentiate COVID-19 from pneumonias caused by other pathogens such as: Influenza A or B, cytomegalovirus, adenovirus, respiratory syncytial virus, SARS-CoV, MERS-CoV, other viral, bacterial, mycoplasma and chlamydial pneumonias. However, some features on chest CT may help differentiate COVID-19 from these infections, and these have been highlighted in Table 3[67,68].
| COVID-19 | Other viral pneumonias | Bacterial/mycoplasma and chlamydial pneumonias |
| Early stage: Peripheral pure GGO | Interstitial inflammation | Bronchial pneumonia, lobar pneumonia |
| Progressive stage: Peripheral multiple GGOs, consolidation, crazy-paving pattern, peripheral vascular “tree-in-bud” pattern | Hyperdense reticular patterns or multiple high-density fibrous streaks | Bronchial wall thickening |
| Advanced stage: Extensive exudative lesions, lung “whiteout”, fibrosis | Localized pulmonary edema and/or atelectasis | Centrilobular nodules |
| Multiple consolidations | ||
| Air bronchogram | ||
| Tree-in-bud sign | ||
| Pleural effusion |
It is now widely accepted that imaging plays a crucial role in the diagnosis, management and follow up of COVID-19 patients. However, the radiologists and the radiology trainees are not immune to the SARS-CoV-2. Some of the workplace safety and precautionary steps that can be adopted and various disinfection procedures that should be followed following examination of a confirmed or suspected COVID-19 patient have been highlighted in Table 4[32,36,69-74].
| Precautions to be adopted by the radiologists, radiology staff, and trainees at the workplace and the various disinfection procedures that should be followed |
| As a standard practice with any major public health emergencies from infectious diseases, any patient or personnel entering the health care facility should be screened by infrared temperature detector which measures the individual’s head temperature and displayed on the device |
| Mandatory wearing of surgical masks by all patients and staff prior to entering the hospital and at all times within the hospital premises |
| Isolating of symptomatic workers and social distancing to the risk of transmission from asymptomatic careers |
| Separating workstations by at least 6 feet |
| Reducing one-on-one consultations and conferences |
| Rotating the radiologists and staffs within the department in to 2-3 teams, off and on service together to limit the chance of spread of infection within small teams e.g., instituting a 1-week-on, 1-week-off work schedule so an adequate reserve capacity is maintained in case one group has to be quarantined |
| While continuing to provide emergency and oncology services, non-urgent outpatient visits, imaging studies and interventional procedures should be rescheduled or postponed after discussing with the patient’s referring physician/surgeon. These steps not only allow to free limited resources for the COVID-19 patients, but also allows to maintain safe distance among patients in the waiting, thereby, reducing the risk of transmission of the disease |
| Enabling teleradiology services, so that radiologists can interpret images remotely from home |
| Access to PPE especially to those directly dealing with confirmed and suspected COVID-19 cases (including nurses, trainees, technicians and radiologists). It is imperative to practise sterile donning and doffing of PPE in advance, and training should be provided including for safe disposal. A PPE equipment comprises of-medical protective clothing (non-sterile waterproof gown and sterile surgical gown), surgical cap, N95 mask or Ffp2/3 mask, double sterile gloves, face shields and goggles or adhering mask which also provides eye protection |
| Wide implementation of supportive protection: By using disposable medical caps, medical protective face masks (antifog type or N95 respirators, disposable latex gloves (double layered) and strictly implementing good hand hygiene with alcohol-based rub containing at least 60%-95% alcohol or by washing hands with soap and water for at least 20 s |
| Mandatory wearing of surgical masks by all patients prior to undergoing imaging |
| When transporting the patient for any imaging or to IR section, identify the lowest traffic/risk path, including avoiding areas with critically ill patients, if possible |
| Maintain spatial distance of at least 1 meter whenever possible between patient and staff during transfer |
| If possible, divide the radiology department into 4 zones: (1) Contaminated zone: Connected to the fever clinic, fever access way, the CT and DR examination rooms; (2) Semi-contaminated: Includes the fever-CT and fever DR control rooms and other patient examination access areas; (3) Buffer zone: Access areas for medical personnel ad a dressing area for technologists; and (4) Clean zones: Includes administrative office and the diagnostic room (rooms containing the work stations and storage rooms for medical supplies). Patients are strictly forbidden from entering the clean zones |
| No observers, students or unauthorized personnel should be allowed to enter the radiology department |
| Warning signs should be hung on the doors to the entrance of his hospital as well as the radiology department reminding the mandatory use of face masks as well as maintaining a safe distance |
| In hospital with more than one CT and one DR systems, one CT scanner and one DR system closest to the emergency department can be exclusively designated for confirmed and suspected COVID-19 patients |
| Disposable sheets should be used for every CT examination, which should be changed after each study |
| For ultrasound studies or USG guided procedures: Use single-use sterile gel, and the USG machine and transducer should have an extra-long cord and covered in a double bag |
| Equipment and environment sterilization procedures: (1) Object surface sterilization: Object surface is cleaned with 1000 mg/L chlorine containing disinfectant, wiped twice with 75% ethanol for non-corrosion resistance, once every 4 h; (2) Equipment sterilization: Cleaned with 2000 mg/L chlorine containing disinfectant. The DR detector and CT gantry in the affected area should be disinfected after each examination, using solutions suggested by the manufacturer/ vendor or wiped with 75% alcohol solution; (3) Equipment located in the buffer zone: Are wiped with 500-1000 mg/L chlorinated disinfectant or alcohol-containing disposable disinfectant wipes twice a day; (4) Sterilization of the air: All central air conditioners should be turned off to prevent air contamination with each other. In the contaminated area: The door is opened for ventilation, each time more than 30 min, once every 4 h. The air sterilizer is continuously sterilized or the ultraviolet ray is continuously used in the unmanned state for 1 h, four times a day. During disinfection the inner shielding should be closed. Other ambient air is sprayed with 1000 mg/L chlorinated disinfectant and ventilated twice a day; (5) Floor disinfection: The floors are wiped with 1000 mg/L chlorinated disinfectant; and (6) Disinfection of the bed: Both the bed and floors are wiped with 2000 mg/L chlorine-containing disinfectant. Visible contamination: Disposable absorbents should be used initially, to completely remove the pollutants, following which a cloth soaked with 2000 mg/L chlorine-containing disinfectant should be used for 30 min before wiping |
| Additional guidelines for IR staff & procedures: (1) If possible, perform IR procedures at the patient’s bedside to minimize transfer, at the discretion of the interventional radiologist; (2) Identify a procedure room with adequate ventilation/air-exchange (ideally a negative pressure room), with a minimum of 6 air changes per hour; (3) Minimize the number of healthcare professionals/support staff involved in the care of the COVID-19 patients whenever possible; (4) Ensure proper disinfection of re-usable eye protection (e.g., leaded glasses) and lead aprons, and for proper cleaning of the room and equipment; (5) Place appropriate signs indicating the presence of a COVID-19 patient (e.g., “COVID-19 patient: DO NOT ENTER”) on the room entrance; and (6) For AGP, such as gastrostomy/gastrojejunostomy/jejunostomy feeding tube insertion, chest tube insertion, lung/mediastinal biopsy, bronchial artery embolization, etc.; the staff and physicians should wear the following PPE: Gowns, gloves, N95 or equivalent respirator and eye protection (goggles or face shield) |
To date no effective drug or vaccine has been discovered for treating COVID-19. However, there are many ongoing clinical trials evaluating potential treatments, and many efforts are underway to develop vaccines[55,74]. The management of these patients is symptomatic, with oxygen therapy being the first step for addressing respiratory impairment. In most patients with complicated disease, an intensive care would be required[75].
The clinical features, course and outcome of COVID-19 are variable and continuously evolving. Nearly all national and international organizations have recommended to not use CT for diagnosing COVID-19, despite several studies having shown that CT can identify lung findings in patients with negative RT-PCR or even before the test becomes positive. Despite the controversy, it is essential for the radiologist to correlate the imaging findings with clinical history and RT-PCR test results. CT has also proven to be a valuable tool in monitoring disease recovery or progression. With the number of cases rising rapidly with each day, it is essential to maintain robust infection control strategies such as-maintaining social distancing, strict hand hygiene, limiting the number of active staff only to the required minimum, regular disinfection of fomites and making PPE readily available for the staff dealing with confirmed COVID-19 cases, to name a few.
Manuscript source: Invited manuscript
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: India
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Zhu JB S-Editor: Huang P L-Editor: A P-Editor: Wu YXJ
| 1. | Li Q, Guan X, Wu P, Wang X, Zhou L, Tong Y, Ren R, Leung KSM, Lau EHY, Wong JY, Xing X, Xiang N, Wu Y, Li C, Chen Q, Li D, Liu T, Zhao J, Liu M, Tu W, Chen C, Jin L, Yang R, Wang Q, Zhou S, Wang R, Liu H, Luo Y, Liu Y, Shao G, Li H, Tao Z, Yang Y, Deng Z, Liu B, Ma Z, Zhang Y, Shi G, Lam TTY, Wu JT, Gao GF, Cowling BJ, Yang B, Leung GM, Feng Z. Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus-Infected Pneumonia. N Engl J Med. 2020;382:1199-1207. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11224] [Cited by in RCA: 9308] [Article Influence: 1861.6] [Reference Citation Analysis (0)] |
| 2. | Zhu N, Zhang D, Wang W, Li X, Yang B, Song J, Zhao X, Huang B, Shi W, Lu R, Niu P, Zhan F, Ma X, Wang D, Xu W, Wu G, Gao GF, Tan W; China Novel Coronavirus Investigating and Research Team. A Novel Coronavirus from Patients with Pneumonia in China, 2019. N Engl J Med. 2020;382:727-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18987] [Cited by in RCA: 17604] [Article Influence: 3520.8] [Reference Citation Analysis (0)] |
| 3. | Lai CC, Liu YH, Wang CY, Wang YH, Hsueh SC, Yen MY, Ko WC, Hsueh PR. Asymptomatic carrier state, acute respiratory disease, and pneumonia due to severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2): Facts and myths. J Microbiol Immunol Infect. 2020;53:404-412. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 610] [Cited by in RCA: 554] [Article Influence: 110.8] [Reference Citation Analysis (0)] |
| 4. | Song Z, Xu Y, Bao L, Zhang L, Yu P, Qu Y, Zhu H, Zhao W, Han Y, Qin C. From SARS to MERS, Thrusting Coronaviruses into the Spotlight. Viruses. 2019;11:59. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 688] [Cited by in RCA: 705] [Article Influence: 117.5] [Reference Citation Analysis (0)] |
| 5. | Lu R, Zhao X, Li J, Niu P, Yang B, Wu H, Wang W, Song H, Huang B, Zhu N, Bi Y, Ma X, Zhan F, Wang L, Hu T, Zhou H, Hu Z, Zhou W, Zhao L, Chen J, Meng Y, Wang J, Lin Y, Yuan J, Xie Z, Ma J, Liu WJ, Wang D, Xu W, Holmes EC, Gao GF, Wu G, Chen W, Shi W, Tan W. Genomic characterisation and epidemiology of 2019 novel coronavirus: implications for virus origins and receptor binding. Lancet. 2020;395:565-574. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8473] [Cited by in RCA: 7585] [Article Influence: 1517.0] [Reference Citation Analysis (0)] |
| 6. | Ge XY, Li JL, Yang XL, Chmura AA, Zhu G, Epstein JH, Mazet JK, Hu B, Zhang W, Peng C, Zhang YJ, Luo CM, Tan B, Wang N, Zhu Y, Crameri G, Zhang SY, Wang LF, Daszak P, Shi ZL. Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor. Nature. 2013;503:535-538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1165] [Cited by in RCA: 1306] [Article Influence: 108.8] [Reference Citation Analysis (0)] |
| 7. | Guan Y, Zheng BJ, He YQ, Liu XL, Zhuang ZX, Cheung CL, Luo SW, Li PH, Zhang LJ, Guan YJ, Butt KM, Wong KL, Chan KW, Lim W, Shortridge KF, Yuen KY, Peiris JS, Poon LL. Isolation and characterization of viruses related to the SARS coronavirus from animals in southern China. Science. 2003;302:276-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1674] [Cited by in RCA: 1596] [Article Influence: 72.5] [Reference Citation Analysis (0)] |
| 8. | Zu ZY, Jiang MD, Xu PP, Chen W, Ni QQ, Lu GM, Zhang LJ. Coronavirus Disease 2019 (COVID-19): A Perspective from China. Radiology. 2020;296:E15-E25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1106] [Cited by in RCA: 948] [Article Influence: 189.6] [Reference Citation Analysis (1)] |
| 9. | Lauer SA, Grantz KH, Bi Q, Jones FK, Zheng Q, Meredith HR, Azman AS, Reich NG, Lessler J. The Incubation Period of Coronavirus Disease 2019 (COVID-19) From Publicly Reported Confirmed Cases: Estimation and Application. Ann Intern Med. 2020;172:577-582. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4286] [Cited by in RCA: 3360] [Article Influence: 672.0] [Reference Citation Analysis (1)] |
| 10. | Wiersinga WJ, Rhodes A, Cheng AC, Peacock SJ, Prescott HC. Pathophysiology, Transmission, Diagnosis, and Treatment of Coronavirus Disease 2019 (COVID-19): A Review. JAMA. 2020;324:782-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2691] [Cited by in RCA: 3167] [Article Influence: 633.4] [Reference Citation Analysis (0)] |
| 11. | Liu Y, Gayle AA, Wilder-Smith A, Rocklöv J. The reproductive number of COVID-19 is higher compared to SARS coronavirus. J Travel Med. 2020;27:taaa021. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2041] [Cited by in RCA: 1578] [Article Influence: 315.6] [Reference Citation Analysis (0)] |
| 12. | Johns Hopkins University School of Medicine. Coronavirus Resource Center. [accessed October 25, 2020] Available from: https://coronavirus.jhu.edu/. |
| 13. | Jaillon S, Berthenet K, Garlanda C. Sexual Dimorphism in Innate Immunity. Clin Rev Allergy Immunol. 2019;56:308-321. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 380] [Article Influence: 63.3] [Reference Citation Analysis (0)] |
| 14. | Chen N, Zhou M, Dong X, Qu J, Gong F, Han Y, Qiu Y, Wang J, Liu Y, Wei Y, Xia J, Yu T, Zhang X, Zhang L. Epidemiological and clinical characteristics of 99 cases of 2019 novel coronavirus pneumonia in Wuhan, China: a descriptive study. Lancet. 2020;395:507-513. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14869] [Cited by in RCA: 12963] [Article Influence: 2592.6] [Reference Citation Analysis (1)] |
| 15. | Wu Z, McGoogan JM. Characteristics of and Important Lessons From the Coronavirus Disease 2019 (COVID-19) Outbreak in China: Summary of a Report of 72 314 Cases From the Chinese Center for Disease Control and Prevention. JAMA. 2020;323:1239-1242. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11409] [Cited by in RCA: 11488] [Article Influence: 2297.6] [Reference Citation Analysis (0)] |
| 16. | Wang D, Hu B, Hu C, Zhu F, Liu X, Zhang J, Wang B, Xiang H, Cheng Z, Xiong Y, Zhao Y, Li Y, Wang X, Peng Z. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. JAMA. 2020;323:1061-1069. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14113] [Cited by in RCA: 14751] [Article Influence: 2950.2] [Reference Citation Analysis (0)] |
| 17. | Hendren NS, Drazner MH, Bozkurt B, Cooper LT Jr. Description and Proposed Management of the Acute COVID-19 Cardiovascular Syndrome. Circulation. 2020;141:1903-1914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 395] [Cited by in RCA: 405] [Article Influence: 81.0] [Reference Citation Analysis (0)] |
| 18. | Long B, Brady WJ, Koyfman A, Gottlieb M. Cardiovascular complications in COVID-19. Am J Emerg Med. 2020;38:1504-1507. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 668] [Cited by in RCA: 648] [Article Influence: 129.6] [Reference Citation Analysis (0)] |
| 19. | Helms J, Kremer S, Merdji H, Clere-Jehl R, Schenck M, Kummerlen C, Collange O, Boulay C, Fafi-Kremer S, Ohana M, Anheim M, Meziani F. Neurologic Features in Severe SARS-CoV-2 Infection. N Engl J Med. 2020;382:2268-2270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1741] [Cited by in RCA: 1892] [Article Influence: 378.4] [Reference Citation Analysis (0)] |
| 20. | Mao L, Jin H, Wang M, Hu Y, Chen S, He Q, Chang J, Hong C, Zhou Y, Wang D, Miao X, Li Y, Hu B. Neurologic Manifestations of Hospitalized Patients With Coronavirus Disease 2019 in Wuhan, China. JAMA Neurol. 2020;77:683-690. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4761] [Cited by in RCA: 4692] [Article Influence: 938.4] [Reference Citation Analysis (0)] |
| 21. | Levi M, Thachil J, Iba T, Levy JH. Coagulation abnormalities and thrombosis in patients with COVID-19. Lancet Haematol. 2020;7:e438-e440. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 878] [Cited by in RCA: 1058] [Article Influence: 211.6] [Reference Citation Analysis (0)] |
| 22. | Middeldorp S, Coppens M, van Haaps TF, Foppen M, Vlaar AP, Müller MCA, Bouman CCS, Beenen LFM, Kootte RS, Heijmans J, Smits LP, Bonta PI, van Es N. Incidence of venous thromboembolism in hospitalized patients with COVID-19. J Thromb Haemost. 2020;18:1995-2002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1017] [Cited by in RCA: 1091] [Article Influence: 218.2] [Reference Citation Analysis (0)] |
| 23. | Patel R, Babady E, Theel ES, Storch GA, Pinsky BA, St George K, Smith TC, Bertuzzi S. Report from the American Society for Microbiology COVID-19 International Summit, 23 March 2020: Value of Diagnostic Testing for SARS-CoV-2/COVID-19. mBio. 2020;11:e00722-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 217] [Cited by in RCA: 241] [Article Influence: 48.2] [Reference Citation Analysis (0)] |
| 24. | Yang Y, Yang MH, Shen CG, Wang FX, Yuan J, Li JX, Zhang MX, Wang ZQ, Xing L, Wei JL, Peng L, Wong G, Zheng HX, Wu WB, Liao MF, Feng K, Li JM, Yang QT, Zhao JJ, Zhang Z, Liu L, Liu YX. Evaluating the accuracy of different respiratory specimens in the laboratory diagnosis and monitoring the viral shedding of 2019-nCoV infections. medRxiv. 2020;. [RCA] [DOI] [Full Text] [Cited by in Crossref: 313] [Cited by in RCA: 224] [Article Influence: 44.8] [Reference Citation Analysis (0)] |
| 25. | Kanne JP, Little BP, Chung JH, Elicker BM, Ketai LH. Essentials for Radiologists on COVID-19: An Update-Radiology Scientific Expert Panel. Radiology. 2020;296:E113-E114. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 367] [Cited by in RCA: 421] [Article Influence: 84.2] [Reference Citation Analysis (0)] |
| 26. | Wang W, Xu Y, Gao R, Lu R, Han K, Wu G, Tan W. Detection of SARS-CoV-2 in Different Types of Clinical Specimens. JAMA. 2020;323:1843-1844. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1894] [Cited by in RCA: 2658] [Article Influence: 531.6] [Reference Citation Analysis (0)] |
| 27. | World Health Organization. Laboratory testing for 2019 novel coronavirus (2019-nCoV) in suspected human cases [accessed September 6, 2020]. Available from: https://www.who.int/publications-detail/Laboratorytesting-for-2019-novel-coronavirus-in-suspected-humancases-20200117. |
| 28. | Kim EA, Lee KS, Primack SL, Yoon HK, Byun HS, Kim TS, Suh GY, Kwon OJ, Han J. Viral pneumonias in adults: radiologic and pathologic findings. Radiographics. 2002;22:S137-S149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 238] [Cited by in RCA: 215] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 29. | Choi H, Qi XL, Yoon SH, Park SJ, Lee KH, Kim JY, Lee YK, Ko H, Kim KH, Park CM, Kim YH, Le JQ, Hong JH, Kim H, Hwang EJ, Yoo SJ, Nam JG, Lee CH, Goo JM. Extension of coronavirus disease 2019 (COVID-19) on chest CT and implications for chest radiograph interpretation. Radiol Cardiothorac Imag. 2020;2:e204001. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 30. | Wong HYF, Lam HYS, Fong AH, Leung ST, Chin TW, Lo CSY, Lui MM, Lee JCY, Chiu KW, Chung TW, Lee EYP, Wan EYF, Hung IFN, Lam TPW, Kuo MD, Ng MY. Frequency and Distribution of Chest Radiographic Findings in Patients Positive for COVID-19. Radiology. 2020;296:E72-E78. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 832] [Cited by in RCA: 825] [Article Influence: 165.0] [Reference Citation Analysis (1)] |
| 31. | Yoon SH, Lee KH, Kim JY, Lee YK, Ko H, Kim KH, Park CM, Kim YH. Chest Radiographic and CT Findings of the 2019 Novel Coronavirus Disease (COVID-19): Analysis of Nine Patients Treated in Korea. Korean J Radiol. 2020;21:494-500. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 431] [Cited by in RCA: 374] [Article Influence: 74.8] [Reference Citation Analysis (0)] |
| 32. | Lei Y, Zhang HW, Yu J, Patlas MN. COVID-19 Infection: Early Lessons. Can Assoc Radiol J. 2020;71:251-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 33. | Zhao W, Zhong Z, Xie X, Yu Q, Liu J. Relation Between Chest CT Findings and Clinical Conditions of Coronavirus Disease (COVID-19) Pneumonia: A Multicenter Study. AJR Am J Roentgenol. 2020;214:1072-1077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 665] [Cited by in RCA: 695] [Article Influence: 139.0] [Reference Citation Analysis (0)] |
| 34. | Simpson S, Kay FU, Abbara S, Bhalla S, Chung JH, Chung M, Henry TS, Kanne JP, Seth Kligerman, Ko JP, Litt H. Radiological Society of North America Expert Consensus Statement on Reporting Chest CT Findings Related to COVID-19. Endorsed by the Society of Thoracic Radiology, the American College of Radiology, and RSNA. Radiol Cardiothorac Imag. 2020;. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 382] [Cited by in RCA: 375] [Article Influence: 75.0] [Reference Citation Analysis (0)] |
| 35. | Ai T, Yang Z, Hou H, Zhan C, Chen C, Lv W, Tao Q, Sun Z, Xia L. Correlation of Chest CT and RT-PCR Testing for Coronavirus Disease 2019 (COVID-19) in China: A Report of 1014 Cases. Radiology. 2020;296:E32-E40. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3614] [Cited by in RCA: 3283] [Article Influence: 656.6] [Reference Citation Analysis (0)] |
| 36. | Sverzellati N, Milone F, Balbi M. How imaging should properly be used in COVID-19 outbreak: an Italian experience. Diagn Interv Radiol. 2020;26:204-206. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 37. | Hosseiny M, Kooraki S, Gholamrezanezhad A, Reddy S, Myers L. Radiology Perspective of Coronavirus Disease 2019 (COVID-19): Lessons From Severe Acute Respiratory Syndrome and Middle East Respiratory Syndrome. AJR Am J Roentgenol. 2020;214:1078-1082. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 257] [Cited by in RCA: 265] [Article Influence: 53.0] [Reference Citation Analysis (0)] |
| 38. | Li Y, Xia L. Coronavirus Disease 2019 (COVID-19): Role of Chest CT in Diagnosis and Management. AJR Am J Roentgenol. 2020;214:1280-1286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 662] [Cited by in RCA: 652] [Article Influence: 130.4] [Reference Citation Analysis (0)] |
| 39. | Adams HJA, Kwee TC, Yakar D, Hope MD, Kwee RM. Systematic Review and Meta-Analysis on the Value of Chest CT in the Diagnosis of Coronavirus Disease (COVID-19): Sol Scientiae, Illustra Nos. AJR Am J Roentgenol. 2020;215:1342-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 40. | Bai HX, Hsieh B, Xiong Z, Halsey K, Choi JW, Tran TML, Pan I, Shi LB, Wang DC, Mei J, Jiang XL, Zeng QH, Egglin TK, Hu PF, Agarwal S, Xie FF, Li S, Healey T, Atalay MK, Liao WH. Performance of Radiologists in Differentiating COVID-19 from Non-COVID-19 Viral Pneumonia at Chest CT. Radiology. 2020;296:E46-E54. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 726] [Cited by in RCA: 726] [Article Influence: 145.2] [Reference Citation Analysis (0)] |
| 41. | Kong W, Agarwal PP. Chest Imaging Appearance of COVID-19 Infection. Radiol Cardiothorac Imag. . [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 224] [Cited by in RCA: 178] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 42. | Song F, Shi N, Shan F, Zhang Z, Shen J, Lu H, Ling Y, Jiang Y, Shi Y. Emerging 2019 Novel Coronavirus (2019-nCoV) Pneumonia. Radiology. 2020;297:E346. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 43. | Lang M, Som A, Carey D, Reid N, Mendoza DP, Flores EJ, Li MD, Shepard JAO, Little BP. Pulmonary Vascular Manifestations of COVID-19 Pneumonia. Radiol Cardiothorac Imag. 2020;2:3. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 99] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 44. | Patel BV, Arachchillage DJ, Ridge CA, Bianchi P, Doyle JF, Garfield B, Ledot S, Morgan C, Passariello M, Price S, Singh S, Thakuria L, Trenfield S, Trimlett R, Weaver C, Wort SJ, Xu T, Padley SPG, Devaraj A, Desai SR. Pulmonary Angiopathy in Severe COVID-19: Physiologic, Imaging, and Hematologic Observations. Am J Respir Crit Care Med. 2020;202:690-699. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 225] [Cited by in RCA: 226] [Article Influence: 45.2] [Reference Citation Analysis (0)] |
| 45. | Dangis A, Gieraerts C, Bruecker YD, Janssen L, Valgaeren H, Obbels D, Gillis M, Ranst MV, Frans J, Demeyere A, Symons R. Accuracy and reproducibility of low-dose submillisievert chest CT for the diagnosis of COVID-19. Radiol Cardiothorac Imag. 2020;. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 79] [Cited by in RCA: 80] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 46. | Lushina N, Kuo JS, Shaikh HA. Pulmonary, Cerebral, and Renal Thromboembolic Disease in a Patient with COVID-19. Radiology. 2020;296:E181-E183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 47. | Grillet F, Behr J, Calame P, Aubry S, Delabrousse E. Acute Pulmonary Embolism Associated with COVID-19 Pneumonia Detected with Pulmonary CT Angiography. Radiology. 2020;296:E186-E188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 401] [Cited by in RCA: 387] [Article Influence: 77.4] [Reference Citation Analysis (0)] |
| 48. | Léonard-Lorant I, Delabranche X, Séverac F, Helms J, Pauzet C, Collange O, Schneider F, Labani A, Bilbault P, Molière S, Leyendecker P, Roy C, Ohana M. Acute Pulmonary Embolism in Patients with COVID-19 at CT Angiography and Relationship to d-Dimer Levels. Radiology. 2020;296:E189-E191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 385] [Cited by in RCA: 443] [Article Influence: 88.6] [Reference Citation Analysis (0)] |
| 49. | Kim H, Hong H, Yoon SH. Diagnostic Performance of CT and Reverse Transcriptase Polymerase Chain Reaction for Coronavirus Disease 2019: A Meta-Analysis. Radiology. 2020;296:E145-E155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 354] [Cited by in RCA: 365] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 50. | Hope MD, Raptis CA, Shah A, Hammer MM, Henry TS; six signatories. A role for CT in COVID-19? Lancet. 2020;395:1189-1190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 135] [Cited by in RCA: 135] [Article Influence: 27.0] [Reference Citation Analysis (0)] |
| 51. | Rubin GD, Ryerson CJ, Haramati LB, Sverzellati N, Kanne JP, Raoof S, Schluger NW, Volpi A, Yim JJ, Martin IBK, Anderson DJ, Kong C, Altes T, Bush A, Desai SR, Goldin J, Goo JM, Humbert M, Inoue Y, Kauczor HU, Luo F, Mazzone PJ, Prokop M, Remy-Jardin M, Richeldi L, Schaefer-Prokop CM, Tomiyama N, Wells AU, Leung AN. The Role of Chest Imaging in Patient Management During the COVID-19 Pandemic: A Multinational Consensus Statement From the Fleischner Society. Chest. 2020;158:106-116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 447] [Cited by in RCA: 486] [Article Influence: 97.2] [Reference Citation Analysis (0)] |
| 52. | American College of Radiology. ACR Recommendations for the use of Chest Radiography and Computed Tomography (CT) for Suspected COVID-19 Infection. 2020 Available from: https://www.acr.org/Advocacy-and-Economics/ACR-Position-Statements/Recommendations-for-Chest-Radiography-and-CT-for-Suspected-COVID19-Infection. |
| 53. | Thoracic Imaging in COVID-19 Infection. Guidance for the Reporting Radiologist British Society of Thoracic Imaging. Version 2, 2020. Available from: https://www.bsti.org.uk/media/resources/files/BSTI_COVID19_Radiology_Guidance_version_2_16.03.20.pdf. |
| 54. | Dennie C, Hague C, Lim RS, Manos D, Memauri BF, Nguyen ET, Taylor J. Canadian Society of Thoracic Radiology/Canadian Association of Radiologists Consensus Statement Regarding Chest Imaging in Suspected and Confirmed COVID-19. Can Assoc Radiol J. 2020;71:470-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 55. | Kwee TC, Kwee RM. Chest CT in COVID-19: What the Radiologist Needs to Know. Radiographics. 2020;40:1848-1865. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 263] [Cited by in RCA: 250] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 56. | Fu B, Hu L, Lv F, Huang J, Li W, Ouyang Y, Chu Z. Follow-Up CT Results of COVID-19 Patients with Initial Negative Chest CT. Infect Drug Resist. 2020;13:2681-2687. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 57. | Zhong L, Zhang S, Wang J, Zhao X, Wang K, Ding W, Xing Z, Shen J. Analysis of Chest CT Results of Coronavirus Disease 2019 (COVID-19) Patients at First Follow-Up. Can Respir J. 2020;2020:5328267. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 58. | Guan CS, Wei LG, Xie RM, Lv ZB, Yan S, Zhang ZX, Chen BD. CT findings of COVID-19 in follow-up: comparison between progression and recovery. Diagn Interv Radiol. 2020;26:301-307. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 59. | Tabatabaei SMH, Rajebi H, Moghaddas F, Ghasemiadl M, Talari H. Chest CT in COVID-19 pneumonia: what are the findings in mid-term follow-up? Emerg Radiol. 2020;27:711-719. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 60. | Joob B, Wiwanitkit V. 18F-FDG PET/CT and COVID-19. Eur J Nucl Med Mol Imaging. 2020;47:1348. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 61. | Qin C, Liu F, Yen TC, Lan X. 18F-FDG PET/CT findings of COVID-19: a series of four highly suspected cases. Eur J Nucl Med Mol Imaging. 2020;47:1281-1286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 178] [Cited by in RCA: 205] [Article Influence: 41.0] [Reference Citation Analysis (0)] |
| 62. | Zou S, Zhu X. FDG PET/CT of COVID-19. Radiology. 2020;296:E118. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 79] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 63. | Deng Y, Lei L, Chen Y, Zhang W. The potential added value of FDG PET/CT for COVID-19 pneumonia. Eur J Nucl Med Mol Imaging. 2020;47:1634-1635. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 64. | Albano D, Bertagna F, Bertoli M, Bosio G, Lucchini S, Motta F, Panarotto MB, Peli A, Camoni L, Bengel FM, Giubbini R. Incidental Findings Suggestive of COVID-19 in Asymptomatic Patients Undergoing Nuclear Medicine Procedures in a High-Prevalence Region. J Nucl Med. 2020;61:632-636. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 131] [Article Influence: 26.2] [Reference Citation Analysis (0)] |
| 65. | Alimadadi A, Aryal S, Manandhar I, Munroe PB, Joe B, Cheng X. Artificial intelligence and machine learning to fight COVID-19. Physiol Genomics. 2020;52:200-202. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 225] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 66. | Li L, Qin L, Xu Z, Yin Y, Wang X, Kong B, Bai J, Lu Y, Fang Z, Song Q, Cao K, Liu D, Wang G, Xu Q, Fang X, Zhang S, Xia J, Xia J. Using Artificial Intelligence to Detect COVID-19 and Community-acquired Pneumonia Based on Pulmonary CT: Evaluation of the Diagnostic Accuracy. Radiology. 2020;296:E65-E71. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1260] [Cited by in RCA: 939] [Article Influence: 187.8] [Reference Citation Analysis (3)] |
| 67. | Dai WC, Zhang HW, Yu J, Xu HJ, Chen H, Luo SP, Zhang H, Liang LH, Wu XL, Lei Y, Lin F. CT Imaging and Differential Diagnosis of COVID-19. Can Assoc Radiol J. 2020;71:195-200. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 170] [Cited by in RCA: 182] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 68. | Wang H, Wei R, Rao G, Zhu J, Song B. Characteristic CT findings distinguishing 2019 novel coronavirus disease (COVID-19) from influenza pneumonia. Eur Radiol. 2020;30:4910-4917. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 69] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 69. | Zhang HW, Yu J, Xu HJ, Lei Y, Pu ZH, Dai WC, Lin F, Wang YL, Wu XL, Liu LH, Li M, Mo YQ, Zhang H, Luo SP, Chen H, Lyu GW, Zhou ZG, Liu WM, Liu XL, Song HY, Chen FZ, Zeng L, Zhong H, Guo TT, Hu YQ, Yang XX, Liu PN, Li DF. Corona Virus International Public Health Emergencies: Implications for Radiology Management. Acad Radiol. 2020;27:463-467. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 70. | Alvin MD, George E, Deng F, Warhadpande S, Lee SI. The Impact of COVID-19 on Radiology Trainees. Radiology. 2020;296:246-248. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 97] [Cited by in RCA: 119] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 71. | Huang Z, Zhao S, Li Z, Chen W, Zhao L, Deng L, Song B. The Battle Against Coronavirus Disease 2019 (COVID-19): Emergency Management and Infection Control in a Radiology Department. J Am Coll Radiol. 2020;17:710-716. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 104] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 72. | Chandy PE, Nasir MU, Srinivasan S, Klass D, Nicolaou S, B Babu S. Interventional radiology and COVID-19: evidence-based measures to limit transmission. Diagn Interv Radiol. 2020;26:236-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 73. | 73 The Canadian Association for Interventional Radiology (CAIR) and Canadian Association of Radiologists (CAR) Guidelines for Interventional Radiology Procedures for the Patients with Suspected or Confirmed COVID-19 [accessed October 6, 2020]. Available from: https://car.ca/covid-19/. |
| 74. | An P, Ye Y, Chen M, Chen Y, Fan W, Wang Y. Management strategy of novel coronavirus (COVID-19) pneumonia in the radiology department: a Chinese experience. Diagn Interv Radiol. 2020;26:200-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 5.2] [Reference Citation Analysis (0)] |