Published online Jan 26, 2021. doi: 10.4330/wjc.v13.i1.28
Peer-review started: August 30, 2020
First decision: October 23, 2020
Revised: November 7, 2020
Accepted: December 22, 2020
Article in press: December 22, 2020
Published online: January 26, 2021
Processing time: 137 Days and 10.6 Hours
Doxorubicin and other anthracycline derivatives inhibit topoisomerase II and is an important class of cytotoxic chemotherapy in cancer treatment. The use of anthracycline is limited by dose-dependent cardiotoxicity, which may manifest initially as asymptomatic cardiac dysfunction with subsequent progression to congestive heart failure. Despite baseline assessment and periodic monitoring of cardiac function for patients receiving anthracycline agents, there are unmet needs in prediction and prevention of anthracycline-induced cardiotoxicity (AIC).
A 35-year-old African American female was found to have a 9-cm high-grade osteosarcoma of right femur and normal baseline cardiac function with left ventricular ejection fraction of approximately 60%-70% determined by transthoracic and dobutamine stress echocardiogram. She underwent perioperative doxorubicin and cisplatin chemotherapy with 3 cycles before surgery and 3 cycles after surgery, and received a total of 450 mg/m2 doxorubicin at the end of her treatment course. She was evaluated regularly during chemotherapy without any cardiac or respiratory symptoms. Approximately two months after her last chemotherapy, the patient presented to the emergency department with dyspnea for one week and was intubated for acute hypoxic respiratory failure. Echocardiogram showed an ejection fraction of 5%-10% with severe biventricular failure. Despite attempts to optimize cardiac function, the patient’s hemodynamic status continued to decline, and resuscitation was not successful on the seventh day of hospitalization. The autopsy showed no evidence of osteosarcoma, and the likely cause of death was cardiac failure with the evidence of pulmonary congestion, liver congestion, and multiple body cavity effusions.
We present a case of 35-year-old African American female developing cardiogenic shock shortly after receiving a cumulative dose of 450 mg/m2 doxorubicin over 9 mo. Cardiac monitoring and management of patients receiving anthracycline chemotherapy have been an area of intense research since introduction of these agents in clinical practice. We have reviewed literature and recent advances in the prediction and prevention of AIC. Although risk factors currently identified can help stratify patients who need closer monitoring, there are limitations to our current understanding and further research is needed in this field.
Core Tip: Anthracyclines may exert a direct toxic effect on cardiac myocytes, precipitating symptomatic heart failure. The case presented demonstrates an example of acute heart failure in a well-compensated young adult who did not at first glance warrant greater than routine cardiac surveillance during doxorubicin treatment course. Utilization of cardioprotective agents and cardiac strain markers such as troponin and brain natriuretic peptide may help to prevent and identify cardiac dysfunction in asymptomatic patients. Prevention of anthracycline-induced cardiotoxicity and cardiovascular toxicities of other anti-cancer therapy requires multidisciplinary approaches such as modification of cardiovascular risk factors, active management of comorbidities, and pharmacologic therapy in selected patients.
- Citation: Chong EG, Lee EH, Sail R, Denham L, Nagaraj G, Hsueh CT. Anthracycline-induced cardiotoxicity: A case report and review of literature. World J Cardiol 2021; 13(1): 28-37
- URL: https://www.wjgnet.com/1949-8462/full/v13/i1/28.htm
- DOI: https://dx.doi.org/10.4330/wjc.v13.i1.28
Anthracycline is a class of commonly used agents for the treatment of solid and hematologic cancers. Cardiotoxicity is a well-documented side effect of anthracycline, likely due to free radical damage and DNA strand breakage in cardiomyocytes[1]. Anthracycline-induced cardiotoxicity (AIC) accounts for greater than 30% of cardiotoxicity from cancer-related therapy[2]. When symptoms and signs of cardiotoxicity such as congestive heart failure are identified early, discontinuation of anthracycline, initiating appropriate medical management followed by frequent monitoring of cardiac function can help to alleviate further decline of cardiac function. When a patient is asymptomatic, it is difficult to predict adverse outcomes or plan appropriate frequency of cardiac monitoring.
Here we present a case of an unusual course of AIC in a 35-year-old African American female who received a cumulative dose of 450 mg/m2 doxorubicin over 9 mo. We also discuss measures and methods that have been reported for early identification and prevention of deterioration of cardiac function.
A 35-year-old African American female without significant past medical history presented to the orthopedic clinic for evaluation of a right knee mass measuring about 5 cm × 9 cm × 6 cm at distal thigh that had been growing in size over the past six months.
A magnetic resonance imaging (MRI) of the knee showed a 5.6 cm × 9.1 cm × 6.0 cm tumor of the distal right femur, and biopsy revealed high-grade osteoblastic osteosarcoma. Further imaging studies including (18)F-fluorodeoxyglucose positron emission tomography/computed tomography (CT) did not indicate distant metastatic disease, and she was referred to medical oncology clinic for pre-operative chemotherapy. At the time of her pre-treatment evaluation, the patient had no chronic medical issues and was not taking any medications. There was no cancer or cardiovascular disease in her family. She had normal complete blood count and comprehensive metabolic panel results. CT scan of chest showed no evidence of pulmonary metastasis, and baseline transthoracic and dobutamine stress echocardiogram showed normal cardiac function with normal right ventricular systolic function and left ventricular ejection fraction (LVEF) 60%-70%.
The patient underwent three cycles of cisplatin and doxorubicin before undergoing resection of the tumor and a total right knee replacement. She had a follow-up echocardiogram after 2 cycles of chemotherapy which again showed normal cardiac function with LVEF 70%. Additionally, she had serial blood tests done for troponin and brain natriuretic peptide (BNP) during hospitalization for neutropenic infection with respiratory symptoms after cycle 2 chemotherapy, and dehydration with hypokalemia after cycle 3 chemotherapy; all the troponin levels were within normal limit except transient increase in BNP with subsequent normalization was noted after cycle 3 chemotherapy. Surgical pathology showed 5% tumor necrosis, and restaging CT and bone scan after surgery showed no evidence of metastatic disease. The patient subsequently completed three more cycles of cisplatin and doxorubicin with good tolerance. While on chemotherapy treatment, she experienced fatigue, nausea, alopecia, and neutropenic infection despite using granulocyte colony stimulating factor, prophylactic antibiotics, and other supportive care measures. She was evaluated every 3-4 wk with history and physical examination, as well as routine laboratory tests.
Approximately two months after her last chemotherapy, she was seen in the medical oncology clinic with an unremarkable history and physical examination. One week later, she presented to the emergency department with complaints of sudden onset of dyspnea, palpitations, and left-sided chest pain. CT angiography of chest showed no pulmonary embolism, but found new right lower lobe indeterminate nodules, measuring up to 7 mm, a small focus of airspace disease in the peripheral right lower lobe, and mild interstitial pulmonary edema. Echocardiogram showed LVEF 5%-10% with severe decrease in right ventricular systolic function. Her serial serum troponin levels were significantly and persistently elevated.
No significant past medical history.
No cancer or cardiovascular disease in her family.
A 5 cm × 9 cm × 6 cm tumor was noted at the distal right thigh.
Normal complete blood count and comprehensive metabolic panel results.
An MRI of the knee showed a 5.6 cm × 9.1 cm × 6.0 cm tumor of the distal right femur, and biopsy revealed high-grade osteoblastic osteosarcoma. Further imaging studies including (18)F-fluorodeoxyglucose positron emission tomography/CT did not indicate distant metastatic disease, and she was referred to medical oncology clinic for pre-operative chemotherapy. At the time of her pre-treatment evaluation, the patient had no chronic medical issues and was not taking any medications. CT scan of chest showed no evidence of pulmonary metastasis, and baseline transthoracic and dobutamine stress echocardiogram showed normal cardiac function with normal right ventricular systolic function and LVEF 60%-70%.
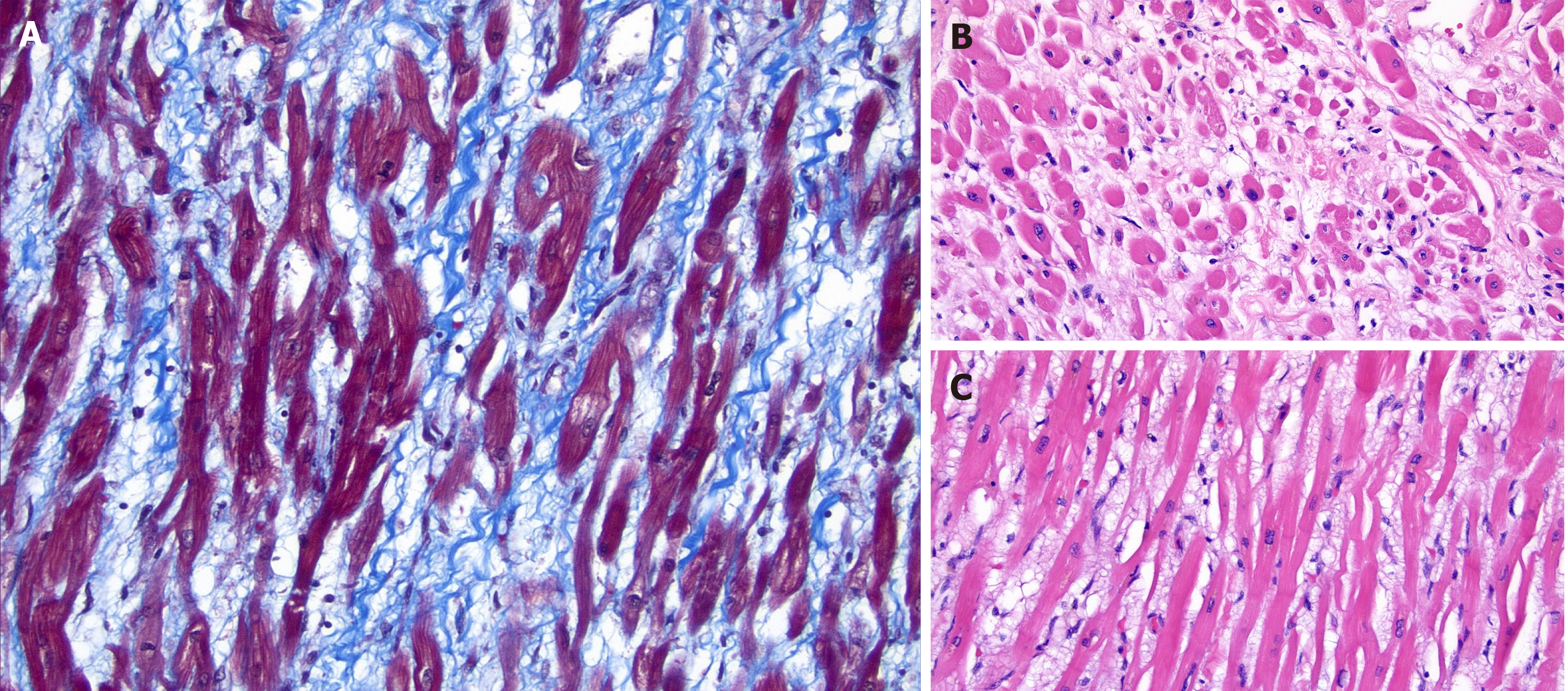
A subsequent autopsy showed no evidence of cancer, and microscopic evaluation of the myocardium was significant for prominent interstitial edema, minimally thinned myocytes, and very focal, minimal interstitial fibrosis (Figure 1).
She was intubated for acute hypoxic respiratory failure due to cardiogenic shock and started on inotropic and vasopressor support. She subsequently developed cardiorenal syndrome. Due to history of malignancy, she was not a candidate for cardiac transplant. An Impella ventricular assist device was placed emergently through the right femoral artery to increase cardiac output on the seventh day of hospitalization.
After placement of ventricular assist device, she became hemodynamically unstable and unresponsive to fluid resuscitation. A hematoma was noted at right femoral access site with drop in hemoglobin from 8.0 g/dL to 6.3 g/dL. Emergent transfusions were ordered, but the patient decompensated and was found to be pulseless. Advanced cardiac life support was initiated, and bedside echocardiogram showed a pericardial effusion with concern for a cardiac tamponade, so an emergent pericardiocentesis was performed at bedside. Unfortunately, restoration of spontaneous circulation was unable to be obtained and the patient expired.
While the rapid onset of the patient’s symptoms presented a broad differential diagnosis for her acute heart failure, the lack of evidence for obvious infection, intracardiac malignancy, prior history of coronary artery disease, or cardiac risk factors prompted suspicion for a diagnosis of anthracycline induced cardiotoxicity. This patient had an initial ejection fraction of 70%, which was reduced to 10% in nine months. The echocardiogram did not show apical ballooning, which would be typical for stress or Takutsubo cardiomyopathy. Therefore, patient’s cause of death is likely from AIC which may manifest either as early side effect with transient cardiac arrhythmia occurring immediately after first dose, or late side effect with cardiomyopathy occurring 2-3 mo or years after therapy. Most AIC develops within one year after completion of treatment[3]. Early AIC does not lead to subsequent development of cardiomyopathy, and therefore is not considered as an indication to stop anthracycline treatment once cardiac arrhythmia is controlled. The estimated risk of impaired cardiac function is about 1% to 2% at a cumulative dose of 300 mg/m2 of doxorubicin, and this risk increases significantly to more than 5% when the cumulative dose of doxorubicin exceeds 450 mg/m2[4].
Retrospective studies and meta-analyses have identified risk factors associated with AIC including: Cumulative dose, administration schedule, duration of infusion, age (> 65 years or < 18 years), female gender, African-American race, history of chest irradiation, use of other cardiac toxic agents such as trastuzumab, existing cardiovascular disease such as coronary artery disease, hypertension and metabolic syndrome[5-7]. Increased risks for AIC have also been associated with genetic polymorphisms in ATP-binding cassette transporter, TOP2 regulator gene RARG, uridine diphosphate glucuronosyltransferase 1A6 glucuronidation gene UGT1A6, carbonyl reductase genes that reduce anthracyclines to cardiotoxic alcohol metabolites, the myofilament splice variant gene CELF4, the hyaluronic acid gene HAS3, and rs28714259 from chromosome 15[8-10]. Early identification of patients at risk of AIC is key to preventing cardiotoxicity[11]. We have outlined strategies to mitigate selected modifiable risk factors associated with AIC in Table 1.
| Risk factor | Risk-reduction strategy |
| Hypertension | Lifestyle modification and pharmacologic control |
| Diabetes | Lifestyle modification and pharmacologic control |
| Dyslipidemia | Lifestyle modification and pharmacologic control |
| Smoking | Smoking cessation |
| Coronary artery disease | Lifestyle modification and pharmacologic control |
| Obesity | Lifestyle modification and weight management |
Dexrazoxane (ICRF-187) was approved by the United States Food and Drug Administration in 1995 as a cardioprotectant for reducing the incidence and severity of cardiomyopathy associated with doxorubicin administration in women with metastatic breast cancer who have received a cumulative doxorubicin dose of 300 mg/m2 and who will continue to receive doxorubicin therapy[12]. In randomized studies, 3% of patients treated with dexrazoxane developed CHF compared with 22% of patients who did not receive dexrazoxane. Its mechanisms of action include reduction of cardiotoxic reactive oxygen species, iron chelation, and catalytic inhibitor of topoisomerase II[13]. It should be noted that there have been some concerns in utilizing this agent due to its side effect of secondary malignancy and decreased antitumor effect of anthracycline[14]. Alterations in infusion schedule and the use of pegylated formulation have also shown to reduce the risk of AIC[15]. A study in pediatric population suggested that infusions greater than 6 h reduce the risk of clinical heart failure when compared to those that received IV pushes[16].
Cardiac monitoring and management of patients receiving anthracyclines have been an area of active research since introduction of these agents in clinical practice and are summarized in Table 2[17-21]. In 2017, the American Society of Clinical Oncology (ASCO) published clinical practice guidelines for the prevention and monitoring of cardiac dysfunction in adult cancer survivors[21]. The ASCO guidelines recommend comprehensive assessment in patients with cancer that includes a history and physical examination, screening for cardiovascular disease risk factors, and an echocardiogram before initiation of anthracycline and other cardiotoxic therapies. Echocardiogram may be performed during cardiotoxic cancer treatment and between 6 mo to 12 mo after completion of therapy in asymptomatic patients considered to be at increased risk for cardiac dysfunction. Patients found to have asymptomatic cardiac dysfunction during routine surveillance should be referred to a cardiologist for further assessment and management.
| Method | Description |
| Biomarkers | Serial measurement of troponin and brain natriuretic peptide |
| Imaging studies | Regular monitoring of left ventricular function with echocardiogram or cardiac MRI |
| Pharmacologic agents | Concurrent use of dexrazoxane (for patients receiving doxorubicin ≥ 250 mg/m2 or epirubicin ≥ 600 mg/m2) |
| Administration strategies | Limiting total dose of anthracycline therapy |
| Continuous infusion | |
| Divided doses | |
| Liposomal formulation |
It is well known that monitoring LVEF alone during anthracycline treatment is insufficient to detect subclinical changes or predict early declines in cardiac function[22]. Studies have shown that persistent elevation of cardiac biomarkers such as troponin and BNP during anthracycline treatment is a harbinger of subsequent LVEF reduction[23,24]. Guidelines from ASCO, European Society for Medical Oncology (ESMO) and Italian Society of Cardiology support periodic measurements of troponin and BNP during anthracycline therapy in patients with risk factors for AIC[18,20,21]. Cardiology consultation and initiation of heart failure therapy with neurohormonal inhibitor such as angiotensin converting enzyme inhibitor (ACE-I) are recommended when cardiac biomarkers are persistently elevated. In addition, these guidelines recommend dexrazoxane treatment, continuous infusion and switching to liposomal formulation in patients planning to receive high-dose anthracyclines (doxorubicin ≥ 250 mg/m2 or epirubicin ≥ 600 mg/m2).
Neurohormonal blocking agents such as beta-blockers (BB), ACE-I, angiotensin receptor blockers (ARB), and aldosterone antagonists have been investigated as prevention and treatment of AIC. In preclinical studies, these agents have shown to suppress neurohormonal activation, reduce left ventricular remodeling, and prevent or delay the onset of cardiac symptoms[25]. However, clinical studies have shown mixed results in using neurohormonal inhibitors at the time of chemotherapy to prevent AIC.
The Carvedilol Effect in Preventing Chemotherapy Induced Cardiotoxicity (CECCY; ClinicalTrials.gov Identifier: NCT01724450) phase III trial was the largest BB prevention study which randomized carvedilol vs placebo as 1:1 in 192 women with HER2-negative breast cancer receiving doxorubicin 240 mg/m2 (60 mg/m2 every 3 wk) with cyclophosphamide followed by paclitaxel[26]. Patients were given incremental dosing of carvedilol or placebo during chemotherapy. The carvedilol group did not meet the primary endpoint which was prevention of early systolic dysfunction within 6 mo of starting doxorubicin. LVEF reduction of at least 10% was noted in 14.5% of the carvedilol group vs 13.5% of the placebo group (P = 1.0). Nevertheless, the carvedilol group had lower troponin I levels over 24 wk (P = 0.003) and a lower incidence of LV diastolic dysfunction (P = 0.04). A meta-analysis of 633 patients from 8 randomized trials (including CECCY) comparing carvedilol vs placebo to prevent AIC showed significantly reduced rates of low LVEF favoring the carvedilol group (3.2% vs 5.8%; OR: 0.42; 95%CI: 0.18-0.99; P = 0.05)[27]. In addition, there were significantly smaller reductions in LVEF in carvedilol-treated patients compared to placebo group.
Prophylactic use of ACE-I to prevent AIC has also been under active investigation. Janbabai et al[28] randomized 69 cancer patients receiving enalapril or placebo during doxorubicin treatment of cumulative dose of 365 mg/m2. The primary end point was change in LVEF measured by echocardiogram from baseline to 6 mo after randomization. There was no change in mean LVEF at 6 mo from baseline in the enalapril treated group, yet there was significant drop in mean LVEF at 6 mo from baseline in control group (46.31% ± 7.04% vs 59.61% ± 5.7%; P < 0.001). This study also showed that serum troponin I and creatinine kinase-MB levels were significantly higher 1 mo after the initiation of chemotherapy in the control group than in the enalapril group, suggesting a cardioprotective effect from enalapril. Cardinale et al[29] randomized 114 cancer patients with elevated troponin levels after starting high-dose chemotherapy with either concurrent or prior anthracycline exposure to enalapril vs placebo. The primary endpoint was an absolute decrease of more than 10% in LVEF with a decline below 50%, which was prevented in enalapril treatment group when compared with placebo (0% vs 43%; P < 0.001).
The phase III International Cardio Oncology Society-ONE trial randomized 273 cancer patients taking anthracycline treatment (40% of them receiving median cumulative dose of 240 mg/m2 doxorubicin) to receive enalapril from initiation of chemotherapy (the prevention group) or during chemotherapy with increased serum troponin level (the troponin-triggered group) (ClinicalTrials.gov Identifier: NCT01968200)[30]. The primary outcome was the incidence of troponin elevation above the threshold at any time during the trial for up to 1 year, and troponin level was measured before and after each anthracycline treatment. There was no difference in the incidence of troponin increase (23% in the prevention vs 26% in the troponin-triggered group; P = 0.50). Two patients in the prevention and one patient in the troponin-triggered group developed LV dysfunction. This study indicates in patients receiving low cumulative dose of anthracycline, a troponin-triggered strategy to use ACE-I for prevention of AIC seems to be a more convenient and sensible option.
In the OVERCOME phase III trial (ClinicalTrials.gov Identifier: NCT01110824), Bosch et al[31] randomized 90 patients with malignant hematologic disorders and normal LVEF in 1:1 ratio to receive either enalapril and carvedilol or nothing during anthracycline therapy. The primary efficacy endpoint was the absolute change from baseline in LVEF, measured by echocardiogram and cardiac MRI at 6 mo after randomization. LVEF did not change in the intervention arm with a median cumulative anthracycline dose of 290 mg/m2 but significantly decreased in control group with a median cumulative dose of anthracycline of 240 mg/m2, leading to a -3.1% absolute difference by echocardiography (P = 0.035). However, the interventional group did not show any statistically significant reduction in the incidence of heart failure or decreased LVEF > 10% (9.5% vs 19%, respectively, P = 0.22). The clinical relevance of combined treatment strategy with ACE-I and BB is currently explored in a phase III SAFE trial (ClinicalTrials.gov Identifier: NCT02236806) which uses 2 × 2 factorial design to randomize 480 breast cancer patients[32].
In the PRADA phase II trial (ClinicalTrials.gov Identifier: NCT01434134), Gulati et al[33] randomized 120 women with breast cancer receiving adjuvant chemotherapy with epirubicin-based regimen to one of four groups: Candesartan plus placebo, metoprolol plus placebo, candesartan plus metoprolol, and placebo plus placebo. The primary endpoint was change in LVEF measured by MRI after completion of adjuvant treatment. No effect of metoprolol on the overall decline in LVEF was observed. Candesartan group had a smaller mean decline in LVEF than the placebo group (0.8% vs 2.6%, P = 0.03), indicating prophylactic use of ARB prevents AIC. There are several ongoing large-scale phase II/III randomized clinical trials exploring novel pharmacological intervention in preventing AIC (Table 3). These approaches include hydroxymethylglutaryl-CoA reductase inhibitor (statins) and LCZ696 (neprilysin inhibitor plus angiotensin receptor blocker).
| Trial No./phase/name | Patient (n) and selection | Randomization schema | Primary endpoint |
| NCT02236806/Phase III/Cardiotoxicity prevention in breast cancer patients treated with anthracyclines and/or trastuzumab (SAFE) | 480 non-metastatic breast cancer receiving anthracycline-based regimens with or without trastuzumab | 2 × 2 factorial design. Arm 1: Bisoprolol plus ramipril; Arm 2: Bisoprolol plus placebo; Arm 3: Ramipril plus placebo; and Arm 4: Placebo | Maximum change in left ventricular ejection fraction at months 6, 9, 12 and 24, compared to baseline |
| NCT03265574/Phase III/Can we prevent chemotherapy-related heart damage in patients with breast cancer? (PROACT) | 170 breast cancer receiving epirubicin-based adjuvant chemotherapy | Open-label comparing enalapril versus standard care | Cardiac troponin T release during epirubicin treatment |
| NCT03760588/Phase II/Prevention of cardiac dysfunction during breast cancer therapy (PRADAII) | 300 breast cancer receiving anthracycline-based adjuvant chemotherapy | Double-blinded comparing LCZ696 (neprilysin inhibitor plus angiotensin receptor blocker) versus placebo | Left ventricular ejection fraction at 18 mo |
| ISRCTN24439460/Phase II/Can heart muscle injury related to chemotherapy be prevented? (cardiac CARE) | 168 breast cancer or non-Hodgkin’s lymphoma with elevated troponin I level during anthracycline-based chemotherapy | Open-label comparing carvedilol plus candesartan versus standard care | Left ventricular ejection fraction at 6 mo |
| NCT02943590/Phase II/Statins to prevent the cardiotoxicity from anthracyclines (STOP-CA) | 300 newly diagnosed non-Hodgkin’s lymphoma receiving doxorubicin-based regimens | Double-blinded comparing atorvastatin versus placebo | Left ventricular ejection fraction at 12 mo |
| NCT01988571/Phase II/Preventing anthracycline cardiovascular toxicity with statins (PREVENT) | 279 breast cancer receiving anthracycline-based adjuvant chemotherapy | Double-blinded comparing atorvastatin versus placebo | Left ventricular ejection fraction at 24 mo |
Anthracyclines have the potential to exert a direct toxic effect on cardiac myocytes, precipitating symptomatic heart failure. The case presented above demonstrates an example of acute heart failure in a well-compensated young adult who did not, at first glance, warrant greater than routine cardiac surveillance during her treatment course. Most cardiotoxicity after anthracycline-containing therapy occurs within the first year and is associated with the anthracycline dose and LVEF at the end of treatment. Early detection and prompt therapy of cardiotoxicity appear crucial for substantial recovery of cardiac function. When symptoms of heart failure are identified early, discontinuation of anthracycline followed by frequent monitoring of cardiac function can help to alleviate further decline. Yet, when a patient is asymptomatic, it is difficult to predict how often cardiac testing should be done.
While published ASCO recommendations regarding baseline and subsequent cardiac imaging are readily available, there are no clear current consensus for criteria by which to stratify asymptomatic patients who do not fall into the high-risk categories. In patients with cardiovascular risk factors, an increased frequency of cardiac imaging should be considered. As evidence mounts regarding the role of ethnicity or genetic polymorphisms in prognosticating the relative risk of chemotherapy side effects, these risk factors could potentially factor in towards an overall risk assessment and for cardiac monitoring frequency.
Furthermore, while there is evidence that cardioprotective agents such as ACE-I, ARB and BB may help to reduce the severity of LVEF depression following anthracycline exposure, it is not clear whether the observed modest reduction in risk translates to actual reduction in symptomatic heart failure and death. More studies may be needed to explore the utility of cardiac strain markers such as troponin and BNP in identifying early-onset cardiac dysfunction in asymptomatic patients.
Prevention of AIC and cardiovascular toxicities of other anti-cancer therapy requires multidisciplinary approaches such as modification of cardiovascular risk factors, active management of co-morbidities, and pharmacologic therapy in selected patients[34]. The emergence of cardio-oncology program provides opportunities for comprehensive management and in-depth investigation, and is supported by the 2019 American Heart Association Scientific Statement on Cardio-Oncology Rehabilitation to manage cardiovascular outcomes in cancer patients and survivors[35].
Manuscript source: Unsolicited manuscript
Specialty type: Cardiac and cardiovascular systems
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Feng MJ S-Editor: Huang P L-Editor: A P-Editor: Li JH
| 1. | Marinello J, Delcuratolo M, Capranico G. Anthracyclines as Topoisomerase II Poisons: From Early Studies to New Perspectives. Int J Mol Sci. 2018;19:3480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 100] [Cited by in RCA: 164] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 2. | Bovelli D, Plataniotis G, Roila F; ESMO Guidelines Working Group. Cardiotoxicity of chemotherapeutic agents and radiotherapy-related heart disease: ESMO Clinical Practice Guidelines. Ann Oncol. 2010;21 Suppl 5:v277-v282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 247] [Cited by in RCA: 282] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 3. | Cardinale D, Colombo A, Bacchiani G, Tedeschi I, Meroni CA, Veglia F, Civelli M, Lamantia G, Colombo N, Curigliano G, Fiorentini C, Cipolla CM. Early detection of anthracycline cardiotoxicity and improvement with heart failure therapy. Circulation. 2015;131:1981-1988. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 838] [Cited by in RCA: 1189] [Article Influence: 118.9] [Reference Citation Analysis (0)] |
| 4. | McGowan JV, Chung R, Maulik A, Piotrowska I, Walker JM, Yellon DM. Anthracycline Chemotherapy and Cardiotoxicity. Cardiovasc Drugs Ther. 2017;31:63-75. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 625] [Cited by in RCA: 654] [Article Influence: 81.8] [Reference Citation Analysis (0)] |
| 5. | Lotrionte M, Biondi-Zoccai G, Abbate A, Lanzetta G, D'Ascenzo F, Malavasi V, Peruzzi M, Frati G, Palazzoni G. Review and meta-analysis of incidence and clinical predictors of anthracycline cardiotoxicity. Am J Cardiol. 2013;112:1980-1984. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 274] [Cited by in RCA: 261] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 6. | Armstrong GT, Oeffinger KC, Chen Y, Kawashima T, Yasui Y, Leisenring W, Stovall M, Chow EJ, Sklar CA, Mulrooney DA, Mertens AC, Border W, Durand JB, Robison LL, Meacham LR. Modifiable risk factors and major cardiac events among adult survivors of childhood cancer. J Clin Oncol. 2013;31:3673-3680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 405] [Cited by in RCA: 540] [Article Influence: 45.0] [Reference Citation Analysis (0)] |
| 7. | Agunbiade TA, Zaghlol RY, Barac A. Heart Failure in Relation to Anthracyclines and Other Chemotherapies. Methodist Debakey Cardiovasc J. 2019;15:243-249. [PubMed] |
| 8. | Visscher H, Ross CJ, Rassekh SR, Barhdadi A, Dubé MP, Al-Saloos H, Sandor GS, Caron HN, van Dalen EC, Kremer LC, van der Pal HJ, Brown AM, Rogers PC, Phillips MS, Rieder MJ, Carleton BC, Hayden MR; Canadian Pharmacogenomics Network for Drug Safety Consortium. Pharmacogenomic prediction of anthracycline-induced cardiotoxicity in children. J Clin Oncol. 2012;30:1422-1428. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 282] [Cited by in RCA: 301] [Article Influence: 21.5] [Reference Citation Analysis (0)] |
| 9. | Schneider BP, Shen F, Gardner L, Radovich M, Li L, Miller KD, Jiang G, Lai D, O'Neill A, Sparano JA, Davidson NE, Cameron D, Gradus-Pizlo I, Mastouri RA, Suter TM, Foroud T, Sledge GW Jr. Genome-Wide Association Study for Anthracycline-Induced Congestive Heart Failure. Clin Cancer Res. 2017;23:43-51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 68] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 10. | Wang X, Sun CL, Quiñones-Lombraña A, Singh P, Landier W, Hageman L, Mather M, Rotter JI, Taylor KD, Chen YD, Armenian SH, Winick N, Ginsberg JP, Neglia JP, Oeffinger KC, Castellino SM, Dreyer ZE, Hudson MM, Robison LL, Blanco JG, Bhatia S. CELF4 Variant and Anthracycline-Related Cardiomyopathy: A Children's Oncology Group Genome-Wide Association Study. J Clin Oncol. 2016;34:863-870. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 105] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 11. | Armenian S, Bhatia S. Predicting and Preventing Anthracycline-Related Cardiotoxicity. Am Soc Clin Oncol Educ Book. 2018;38:3-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 102] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 12. | Swain SM, Whaley FS, Gerber MC, Weisberg S, York M, Spicer D, Jones SE, Wadler S, Desai A, Vogel C, Speyer J, Mittelman A, Reddy S, Pendergrass K, Velez-Garcia E, Ewer MS, Bianchine JR, Gams RA. Cardioprotection with dexrazoxane for doxorubicin-containing therapy in advanced breast cancer. J Clin Oncol. 1997;15:1318-1332. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 441] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 13. | Langer SW. Dexrazoxane for the treatment of chemotherapy-related side effects. Cancer Manag Res. 2014;6:357-363. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 77] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 14. | Lenneman CG, Sawyer DB. Cardio-Oncology: An Update on Cardiotoxicity of Cancer-Related Treatment. Circ Res. 2016;118:1008-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 317] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 15. | van Dalen EC, Michiels EM, Caron HN, Kremer LC. Different anthracycline derivates for reducing cardiotoxicity in cancer patients. Cochrane Database Syst Rev. 2010;CD005006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 66] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 16. | Ewer MS, Jaffe N, Ried H, Zietz HA, Benjamin RS. Doxorubicin cardiotoxicity in children: comparison of a consecutive divided daily dose administration schedule with single dose (rapid) infusion administration. Med Pediatr Oncol. 1998;31:512-515. [PubMed] [DOI] [Full Text] |
| 17. | American Academy of Pediatrics Section on Hematology/Oncology Children's Oncology Group. Long-term follow-up care for pediatric cancer survivors. Pediatrics. 2009;123:906-915. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 146] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 18. | Curigliano G, Cardinale D, Suter T, Plataniotis G, de Azambuja E, Sandri MT, Criscitiello C, Goldhirsch A, Cipolla C, Roila F; ESMO Guidelines Working Group. Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann Oncol. 2012;23 Suppl 7:vii155-vii166. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 523] [Cited by in RCA: 559] [Article Influence: 46.6] [Reference Citation Analysis (0)] |
| 19. | Plana JC, Galderisi M, Barac A, Ewer MS, Ky B, Scherrer-Crosbie M, Ganame J, Sebag IA, Agler DA, Badano LP, Banchs J, Cardinale D, Carver J, Cerqueira M, DeCara JM, Edvardsen T, Flamm SD, Force T, Griffin BP, Jerusalem G, Liu JE, Magalhães A, Marwick T, Sanchez LY, Sicari R, Villarraga HR, Lancellotti P. Expert consensus for multimodality imaging evaluation of adult patients during and after cancer therapy: a report from the American Society of Echocardiography and the European Association of Cardiovascular Imaging. J Am Soc Echocardiogr. 2014;27:911-939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 827] [Cited by in RCA: 942] [Article Influence: 85.6] [Reference Citation Analysis (0)] |
| 20. | Spallarossa P, Maurea N, Cadeddu C, Madonna R, Mele D, Monte I, Novo G, Pagliaro P, Pepe A, Tocchetti CG, Zito C, Mercuro G. A recommended practical approach to the management of anthracycline-based chemotherapy cardiotoxicity: an opinion paper of the working group on drug cardiotoxicity and cardioprotection, Italian Society of Cardiology. J Cardiovasc Med (Hagerstown). 2016;17 Suppl 1:S84-S92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 21. | Armenian SH, Lacchetti C, Barac A, Carver J, Constine LS, Denduluri N, Dent S, Douglas PS, Durand JB, Ewer M, Fabian C, Hudson M, Jessup M, Jones LW, Ky B, Mayer EL, Moslehi J, Oeffinger K, Ray K, Ruddy K, Lenihan D. Prevention and Monitoring of Cardiac Dysfunction in Survivors of Adult Cancers: American Society of Clinical Oncology Clinical Practice Guideline. J Clin Oncol. 2017;35:893-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 619] [Cited by in RCA: 652] [Article Influence: 81.5] [Reference Citation Analysis (0)] |
| 22. | Jensen BV, Skovsgaard T, Nielsen SL. Functional monitoring of anthracycline cardiotoxicity: a prospective, blinded, long-term observational study of outcome in 120 patients. Ann Oncol. 2002;13:699-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 253] [Cited by in RCA: 243] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 23. | Dolci A, Dominici R, Cardinale D, Sandri MT, Panteghini M. Biochemical markers for prediction of chemotherapy-induced cardiotoxicity: systematic review of the literature and recommendations for use. Am J Clin Pathol. 2008;130:688-695. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 131] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 24. | Romano S, Fratini S, Ricevuto E, Procaccini V, Stifano G, Mancini M, Di Mauro M, Ficorella C, Penco M. Serial measurements of NT-proBNP are predictive of not-high-dose anthracycline cardiotoxicity in breast cancer patients. Br J Cancer. 2011;105:1663-1668. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 104] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 25. | Bansal N, Adams MJ, Ganatra S, Colan SD, Aggarwal S, Steiner R, Amdani S, Lipshultz ER, Lipshultz SE. Strategies to prevent anthracycline-induced cardiotoxicity in cancer survivors. Cardiooncology. 2019;5:18. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 95] [Article Influence: 15.8] [Reference Citation Analysis (0)] |
| 26. | Avila MS, Ayub-Ferreira SM, de Barros Wanderley MR Jr, das Dores Cruz F, Gonçalves Brandão SM, Rigaud VOC, Higuchi-Dos-Santos MH, Hajjar LA, Kalil Filho R, Hoff PM, Sahade M, Ferrari MSM, de Paula Costa RL, Mano MS, Bittencourt Viana Cruz CB, Abduch MC, Lofrano Alves MS, Guimaraes GV, Issa VS, Bittencourt MS, Bocchi EA. Carvedilol for Prevention of Chemotherapy-Related Cardiotoxicity: The CECCY Trial. J Am Coll Cardiol. 2018;71:2281-2290. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 350] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 27. | Kheiri B, Abdalla A, Osman M, Haykal T, Chahine A, Ahmed S, Osman K, Hassan M, Bachuwa G, Bhatt DL. Meta-Analysis of Carvedilol for the Prevention of Anthracycline-Induced Cardiotoxicity. Am J Cardiol. 2018;122:1959-1964. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 28. | Janbabai G, Nabati M, Faghihinia M, Azizi S, Borhani S, Yazdani J. Effect of Enalapril on Preventing Anthracycline-Induced Cardiomyopathy. Cardiovasc Toxicol. 2017;17:130-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 29. | Cardinale D, Colombo A, Sandri MT, Lamantia G, Colombo N, Civelli M, Martinelli G, Veglia F, Fiorentini C, Cipolla CM. Prevention of high-dose chemotherapy-induced cardiotoxicity in high-risk patients by angiotensin-converting enzyme inhibition. Circulation. 2006;114:2474-2481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 685] [Cited by in RCA: 700] [Article Influence: 36.8] [Reference Citation Analysis (0)] |
| 30. | Cardinale D, Ciceri F, Latini R, Franzosi MG, Sandri MT, Civelli M, Cucchi G, Menatti E, Mangiavacchi M, Cavina R, Barbieri E, Gori S, Colombo A, Curigliano G, Salvatici M, Rizzo A, Ghisoni F, Bianchi A, Falci C, Aquilina M, Rocca A, Monopoli A, Milandri C, Rossetti G, Bregni M, Sicuro M, Malossi A, Nassiacos D, Verusio C, Giordano M, Staszewsky L, Barlera S, Nicolis EB, Magnoli M, Masson S, Cipolla CM; ICOS-ONE Study Investigators. Anthracycline-induced cardiotoxicity: A multicenter randomised trial comparing two strategies for guiding prevention with enalapril: The International CardioOncology Society-one trial. Eur J Cancer. 2018;94:126-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 163] [Article Influence: 23.3] [Reference Citation Analysis (0)] |
| 31. | Bosch X, Rovira M, Sitges M, Domènech A, Ortiz-Pérez JT, de Caralt TM, Morales-Ruiz M, Perea RJ, Monzó M, Esteve J. Enalapril and carvedilol for preventing chemotherapy-induced left ventricular systolic dysfunction in patients with malignant hemopathies: the OVERCOME trial (preventiOn of left Ventricular dysfunction with Enalapril and caRvedilol in patients submitted to intensive ChemOtherapy for the treatment of Malignant hEmopathies). J Am Coll Cardiol. 2013;61:2355-2362. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 413] [Cited by in RCA: 467] [Article Influence: 38.9] [Reference Citation Analysis (0)] |
| 32. | Meattini I, Curigliano G, Terziani F, Becherini C, Airoldi M, Allegrini G, Amoroso D, Barni S, Bengala C, Guarneri V, Marchetti P, Martella F, Piovano P, Vannini A, Desideri I, Tarquini R, Galanti G, Barletta G, Livi L. SAFE trial: an ongoing randomized clinical study to assess the role of cardiotoxicity prevention in breast cancer patients treated with anthracyclines with or without trastuzumab. Med Oncol. 2017;34:75. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 33. | Gulati G, Heck SL, Ree AH, Hoffmann P, Schulz-Menger J, Fagerland MW, Gravdehaug B, von Knobelsdorff-Brenkenhoff F, Bratland Å, Storås TH, Hagve TA, Røsjø H, Steine K, Geisler J, Omland T. Prevention of cardiac dysfunction during adjuvant breast cancer therapy (PRADA): a 2 × 2 factorial, randomized, placebo-controlled, double-blind clinical trial of candesartan and metoprolol. Eur Heart J. 2016;37:1671-1680. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 396] [Cited by in RCA: 492] [Article Influence: 54.7] [Reference Citation Analysis (0)] |
| 34. | Fradley MG, Brown AC, Shields B, Viganego F, Damrongwatanasuk R, Patel AA, Hartlage G, Roper N, Jaunese J, Roy L, Ismail-Khan R. Developing a Comprehensive Cardio-Oncology Program at a Cancer Institute: The Moffitt Cancer Center Experience. Oncol Rev. 2017;11:340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 35. | Gilchrist SC, Barac A, Ades PA, Alfano CM, Franklin BA, Jones LW, La Gerche A, Ligibel JA, Lopez G, Madan K, Oeffinger KC, Salamone J, Scott JM, Squires RW, Thomas RJ, Treat-Jacobson DJ, Wright JS; American Heart Association Exercise; Cardiac Rehabilitation; and Secondary Prevention Committee of the Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; and Council on Peripheral Vascular Disease. Cardio-Oncology Rehabilitation to Manage Cardiovascular Outcomes in Cancer Patients and Survivors: A Scientific Statement From the American Heart Association. Circulation. 2019;139:e997-e1012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 149] [Cited by in RCA: 301] [Article Influence: 60.2] [Reference Citation Analysis (0)] |