Published online Jul 27, 2012. doi: 10.4240/wjgs.v4.i7.171
Revised: June 22, 2012
Accepted: June 29, 2012
Published online: July 27, 2012
AIM: To employ, in such conditions, a biological graft such as bovine pericardium that offers resistance to infection.
METHODS: In our surgical department, from January 2006 to June 2010, 48 patients underwent abdominal wall reconstruction using acellular bovine pericardium; of these 34 patients had a contaminated wound due to diffuse peritonitis (complicated diverticulitis, bowel perforation, intestinal infarction, strangled hernia, etc.) and 14 patients had hernia relapse on infected synthetic mesh.
RESULTS: In our series, one patient died of multi-organ failure 3 d after surgery. After placement of the pericardium mesh four cases of hernia relapse occurred.
CONCLUSION: Recurrence rate is similar to that of prosthetic mesh repair and the application of acellular bovine pericardium (Tutomesh®, Tutogen Medical Gmbh Germany) is moreover a safe and feasible option that can be employed to manage complicated abdominal wall defects where prosthetic mesh is unsuitable.
- Citation: D’Ambra L, Berti S, Feleppa C, Magistrelli P, Bonfante P, Falco E. Use of bovine pericardium graft for abdominal wall reconstruction in contaminated fields. World J Gastrointest Surg 2012; 4(7): 171-176
- URL: https://www.wjgnet.com/1948-9366/full/v4/i7/171.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v4.i7.171
Incisional hernia is one of the most common complications after laparotomy occuring in 10% to 20% of cases[1]. The incidence of this post-surgical ventral defect is largely correlated to wound-healing complications such as infections, rather than the type of wound incision, or the technique and materials used for abdominal wall closure[2].
Incisional hernia repair can be performed by simple primary closure, staged or delayed reconstruction, surgical flaps or by prosthesis repair made by laparoscopic or open methods. The use of prosthetic mesh significantly decreases the hernia recurrence rate, but it cannot be employed in particular conditions such as: septic contaminated or potentially contaminated surgical fields, pre-irradiated tissue, and in hernia recurrence on infected mesh[3].
The ideal material to use in such conditions is able to: resist infection after its implantation; become completely remodelled into host tissue with mechanical and biological proprieties identical to those of the missing tissue; became rapidly revascularized and to maintain its original physical characteristics. Bioprosthetic meshes have properties that may come close to ideal material. Recent studies have shown the use of acellular dermal allograft and porcine small intestine submucosa as valid solutions for complicated ventral hernia repair[4]. Bovine pericardium is largely employed in paediatric and heart surgery and has replaced dura madre patches as the standard biomaterial for the closure of anterior abdominal wall defects in omphaloceles and gastroschisis[5,6].
Our study aimed to show the application of acellular bovine pericardium patch (Tutomesh®, Tutogen Medical Gmbh Germany) in ventral hernia repair as a safe, feasible option with a minimal complication rate.
In our surgical department, between January 2006 and June 2010, 48 patients with ventral incisional hernia underwent abdominal wall reconstruction using acellular bovine pericardium. Of these, 34 patients were operated as an emergency, having a contaminated wound due to peritonitis as a result of circumstances such as complicated diverticulitis, bowel perforation, intestinal infarction, strangled hernia, etc. The other 14 patients had an hernia relapse on infected synthetic mesh. There were 29 female and 19 male patients, with a median age of 64 years (range 38 to 84 years), and the abdominal wall defects were always localized along the abdominal midline with surface area range of 20 to 600 cm2.
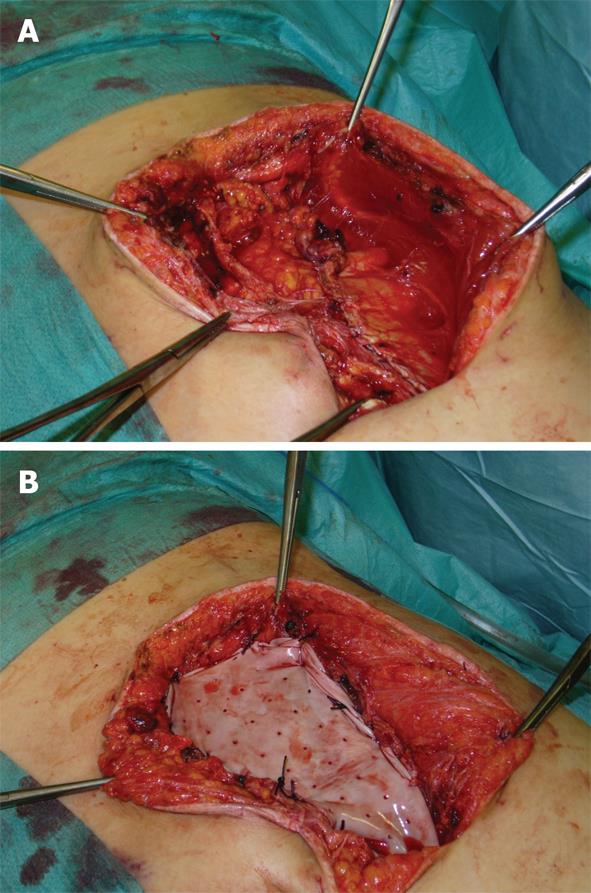
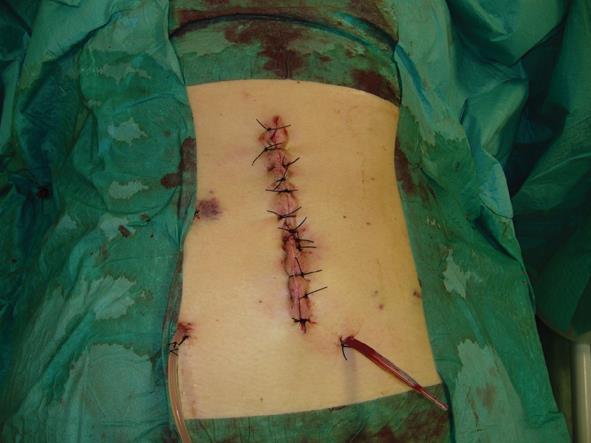
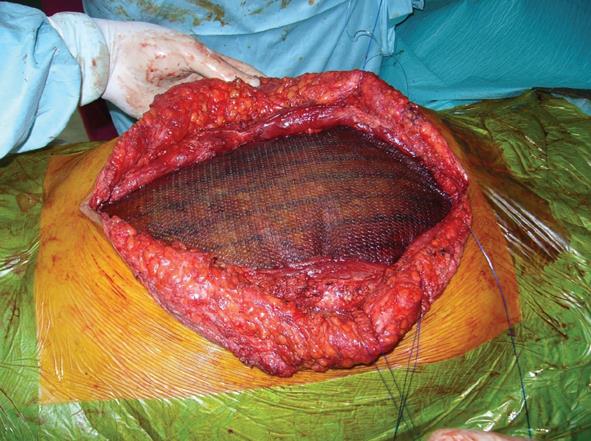
All thirty-four patients admitted with local or diffuse peritonitis underwent a complete cleaning of the peritoneal cavity using hot hydro saline solution with complete evacuation of purulent collections and optimal bowel debridement with removal of contaminated fibrin sites on peritoneal and visceral surfaces. After the surgical management of the principal acute pathology, a comprehensive preparation of the edge of the abdominal wall defect was carried out. The ideal scenario is to close the peritoneum and the posterior rectus sheath and then place the mesh over it, using the sublay technique, avoiding contact with the visceral loop. This is not always achievable, and in our series we have used the sublay technique in twenty-six cases, with retromuscolar mesh placement, fixing the graft with a non-absorbable interrupted suture according to the Rives procedure (Table 1). The anterior rectus sheath, subcutaneous and cutaneous layer were then close with an interrupted non-absorbable suture (Figures 1 and 2). A subcutaneous over-mesh drain tube, model Pean-Rose, was previously positioned. In the other eight cases we corrected the wall abdominal defect using a bridging inlay technique with a interrupted non-absorbable suture to the edge of abdominal wall defect (Figure 3).
| Diagnosis | Technique | n (%) |
| Incisional hernia on contaminated wounds | Sublay | 26 (54) |
| Inlay | 8 (17) | |
| Removal of infected mesh | Sublay | 14 (29) |
In the fourteen patients affected by hernia relapse on infected synthetic mesh we completely removed the prosthetic material and then positioned a bovine pericardium graft using the sublay technique,fixing it with an interrupted non-absorbable suture and then closed the anterior rectus sheath, subcutaneous and cutaneous layer. Hernia relapse was found in the abdominal mid-line in 12 cases, and in the supra pubic area in 2 cases, always due to infection of the synthetic mesh employed to repair potential contaminated defect.
We observed all the patients with a mean follow-up of 22 mo, ranging from 1 to 48 mo, as shown in Table 2.
| Complications | n (%) |
| Death | 1 (2) |
| Wound dehiscence | 2 (4) |
| Seroma | 3 (6) |
| Relapse | 4 (8) |
One patient died on the third day after surgery because of multi organ failure. Four cases of hernia relapse occurred after placement of the pericardium mesh. The first case occurred after 4 mo in an obese, diabetic patient who underwent abdominal reconstruction with a bovine pericardium graft during an Hartmann colonic resection for pelvic peritonitis. The peritonitis was caused by a colorectal anastomotic leak which occurred 10 d after a laparoscopic low anterior resection for rectal cancer. The second case of relapse occurred in a 43-year-old patient, 6 mo after the application of a pericardium allograft during acute peritonitis induced by a venous intestinal infarction due to a thrombosis of superior mesenteric vein. This patient underwent four consecutive interventions, starting with a laparostomy. The third case occurred in a patient affected by chronic renal failure, diabetes and Alzheimer disease 10 d after an operation for pelvic abscess due to sigmoid diverticular perforation.
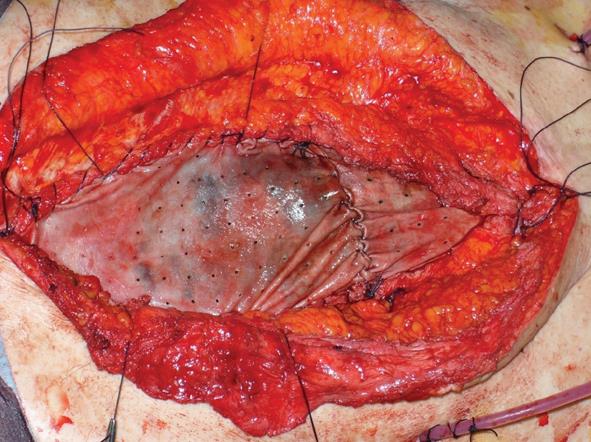
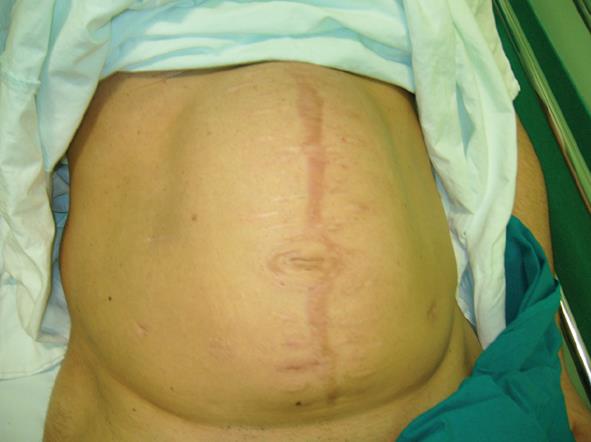
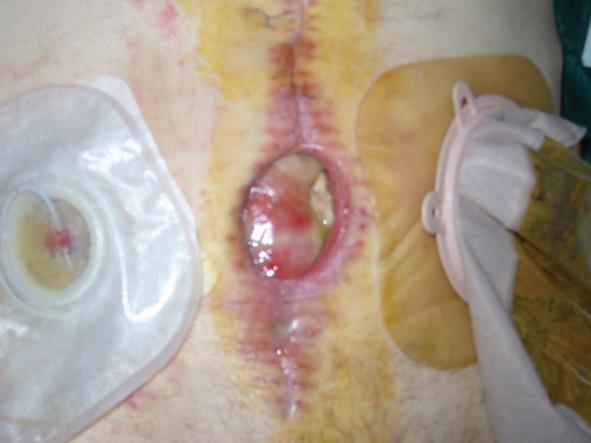
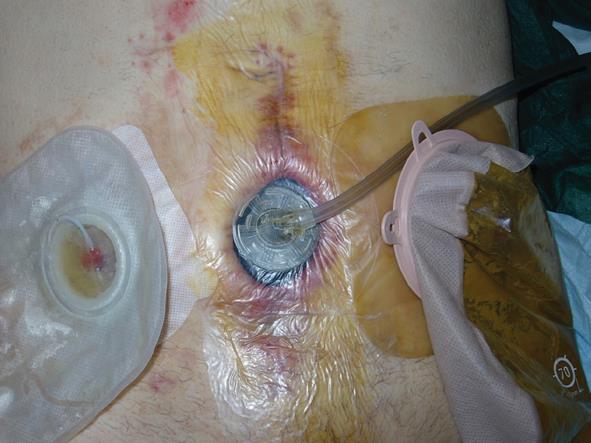
The fourth case of relapse was in a patient who underwent rectal resection with partial cistectomy and distal ureterectomy due to an advanced rectal cancer. Eight days post-operative this patient was affected by severe purulent pelviperitonitis caused by a complete deihscence of the cisto-ileal patch utilized to establish the continuity of urinary tract. He then underwent an ureterocutaneous anastomosis and Hartmann’s colostomy. During the latter operation the abdominal wall was reconstructed by applying a bovine pericardium graft. However, on the twelfth post-operative day this was completely detached from the fascial edge. In the first two cases mentioned, relapse occurred at 4 and 6 mo, respectively, after initial surgery and the abdominal wall defect was then repaired using synthetic mesh placed under muscular layer according to the Rives technique (Figures 4 and 5). In the other two cases, we employed vacuum-assisted closure (VAC) therapy to obtain the complete closure of the abdominal wound. The VAC device was maintained on a continuous suction mode with a negative pressure of -75 mmHg to 125 mmHg and the VAC sponge was changed every 3 d (Figures 6 and 7). At the sixth month follow-up the latter two patients demonstrated a complete restitutio ad integrum of their wound with a residual and partial diastasis of the rectus abdominis.
Ventral incisional hernia continues to be a surgical challenge. The incidence of incisional hernia after abdominal surgery ranges from 10% to 20%, with 74% of cases occurring within 3 years of laparotomy[1]. There are general and local factors that contribute to the establishment of a ventral incisional hernia. General factors include obesity, diabetes, advanced age, corticosteroid therapy, radio and/or chemotherapy, dyspnoea, coughing, vomiting, postoperative ileus, work, constipation, pulmonary failure, vascular surgery, multiple interventions, enterostomies and collagen diseases. Local factors include wound infection, laparotomy over than 10 cm, surgical technique used (continuous vs interrupted suture), suture material, intolerance to material and tissue elasticity. Since the introduction of prosthetic mesh in 1958, the repair of incisional hernia has been facilitated because mesh allows a tension free repair of the abdominal wall defect and has a much lower recurrence rate than primary repair[2,7]. However, mesh repair is associated with a complication rate of 5% to 17%, including seroma formation, visceral erosion, enterocutaneous fistula creation, adhesion formation, bowel obstruction, skin erosion and wound infection[8]. Furthermore, it is well known that implantation of mesh material into contaminated or potentially contaminated abdominal wall defects or in patients with high risk of post operative infection, results in a higher risk of mesh complications including infection. Over the years, several surgical procedures have been developed to manage these abdominal wall defects without using mesh, primarily using reconstructive plastic surgery with autologous flap repair or the component separation technique. Even in these cases, postoperative complications and hernia relapse are high, ranging respectively from 20% to 43% and 8% to 32%, respectively[9-11].
For these reasons, in cases of contaminated or potentially contaminated abdominal wall defect, abdominal wall defect in pre-irradiated tissues, giant ventral hernia with loss of domain or recurrent hernia on infected mesh, the use a mesh material able to resist infection after its implantation is strongly recommended. Such material can be completely remodelled into host tissue with mechanical and biological proprieties identical to those of the missing tissue, and is able to become rapidly revascularized and maintain its original physical characteristics. Biologic meshes have proprieties that may come close to ideal. At the moment various type of biologic prosthesis are commercially available that differ in dimension, cost, and scientific-clinical support. Surgisis® (Biodesign™; CBI West Lafayette, IN, USA) is an acellular collagen matrix from porcine small intestinal submucosa; Alloderm® (Lifecell Corporation, Brachburg, NJ, USA) is an acellular human dermal collagen matrix; Permacol® (Tissue Science Laboratories plc, Covington, GA, USA) is an acellular porcine dermal collagen matrix.
No long-term studies are not yet available, and most of the current evidence supporting the use biologic mesh in this setting is based on case reports and small case series. The literature does not yet include any clinical trials comparing the advantages and disadvantages of the various meshes.
A homologous “dura mater” biomaterial was first utilized in 1950 for neonatal abdominal wall defect repair but was abandoned in 1990. Pursuant to the appearance of spongiform encephalopathy (Creutzfeld-Jacobs S), dura mater patches were withdrawn, although no cases of infection were ever found.
The biomaterial used in this study was acellular bovine pericardium (Tutomesh®, Tutogen Medical Gmbh Germany). This material has actually replaced dura mater in cardiac and neonatal surgery for treatment of gastroschisis and omphalocele. It is a cadaveric pure punched collagen membrane, a very soft and flexible heterologous graft of bovine pericardium carved and processed aseptically, and is completely antigen-less (bacteria, virus, prion, spore). The graft consists of collagenous connective tissue with three-dimensional intertwined fibers, giving it multidirectional mechanical strength, and can be implanted regardless of the direction of the graft. Collagenous connective tissue with multidirectional fibers retains the mechanical strength and elasticity of the native tissue, while providing the basic structure to support replacement by new endogenous tissue. It is indicated for use in general, gynecological, cardiac and plastic surgery, such as in repair of pericardial structures, soft tissue deficiencies, rectal and vaginal prolapse, and hernias, gastric banding, muscle flap reinforcement, and reconstruction of the pelvic floor[12].
Bovine pericardium is indicated for abdominal wall defect repair because it is easy to sutureor glue and does not require overlap, resists infection and does not migrate. It acts as a valid substrate for tissutal regeneration, causes minimal tissutal irritation with minor creation of seroma and does not cause peritoneal adhesions, demonstrating at the same time an excellent mechanical stability of 16 to 42 N/cm (maximum abdominal wall tension measured).
In our series we placed pericardium graft in a retromuscolar sublay manner except in eight cases, in which the mesh was fixed, according to the inlay technique, at the edges of the fascial defect. Four cases of hernia relapse occurred after inlay technique placement. For this reason we suggest the securing of the mesh, using a tension-free procedure, behind the abdominal rectus muscle over the posterior rectus sheath to chive closure of the peritoneum. This retromuscolar mesh implantation assures better outcomes in terms of relapse rate and of diastasis recti abdominis. When the ideal placement of the biological graft is not possible, it is recommended that the contaminated wall defect is managed in a bridging manner, placing the mesh according to the inlay technique and providing a temporary restoration of abdominal wall integrity. In alater operation, implantation of a composite synthetic mesh will be able to re-establish the abdominal wall integrity.
The data available in the literature on biological graft implants for ventral incisional hernia repair are scarce and frequently in the form of case series. Post et al[13] in their series of 13 incisional hernias treated with sublay bovine pericardium graft, found no occurrence of relapse, pain and infection. Lindermann et al[14] reporting their experiences from employing bovine pericardium mesh in 37 incisional and 2 parastomal hernias, described 4 cases of recurrence in the group treated with the inlay technique.
From analysis of the literature data it appears clear that the correction of contaminated abdominal wall defects with a biological mesh using the technique inlay significantly increases the recurrence rate in comparison to corrections performed using the sublay technique. For this reason, the use of biological mesh in a bridging manner (inlay) provides a solution which is temporary but absolutely not definitive, requiring a future mesh implantation to definitively correct the defect[4]. On the other hand, when the biological mesh is fixed retromuscularly employing a sublay technique the recurrence rate seems comparable to other synthetic prosthesis[9,15].
In conclusion, the acellular bovine pericardium graft seems to be a valid tool to use to meet the complex surgical challenge of repairing contaminated or potentially contaminated wall abdominal defect or the replacement an infected synthetic prosthesis. This biological mesh is relatively inexpensive compared with other available biomaterials used in abdominal wall reconstruction[16], and when fixed using the sublay technique provides a recurrence rate similar to that achieved with conventional synthetic prostheses used in less complicated areas of surgery.
Complex abdominal wall defects are difficult to manage because the reconstruction of the wall integrity can not be achieved using synthetic mesh because of the high infection rate. In recent years biological materials have obtained consensus for their employment in contaminated fields due to their optimal tissutal tolerance and their unique ability to provide a safety scaffold in the repair of complicated wall defects.
Homologous “dura mater” biomaterial was first utilized in clinical practice in 1950 for neonatal abdominal wall defect repair but was abandoned in 1990. Acellular bovine pericardium has actually replaced dura mater in cardiac and neonatal surgery for treatment of gastroschisis and omphalocele. It is a cadaveric pure punched collagen membrane, a very soft and flexible heterologous graft of bovine pericardium carved and processed aseptically, and is completely antigen-less (bacteria, virus, prion, spore). The graft consists of collagenous connective tissue with three-dimensional intertwined fibers. Collagenous connective tissue with multidirectional fibers retains the mechanical strength and elasticity of the native tissue, while providing the basic structure to support replacement by new endogenous tissue. It is indicated for use in general, gynecological, cardiac and plastic surgery, such as repair of pericardial structures, soft tissue deficiencies, rectal and vaginal prolapse, hernias, gastric banding, muscle flap reinforcement, and reconstruction of the pelvic floor.
Acellular bovine pericardium allows management of complex defects which cannot be otherwise resolved.
Bovine pericardium is indicated for abdominal wall defect repair because it is easy to suture or glue and does not require overlap, is infection resistant and does not migrate. It acts as valid substrate for tissutal regeneration, causes minimal tissutal irritation with only minor creation of seroma and it does not cause peritoneal adhesions, demonstrating at the same time an excellent mechanical stability.
This is a good study in which authors analyze the effective safety and utility of acellular bovine pericardium in managing of complicated abdominal wall defect.
Peer reviewer: Stefano Scabini, MD, Department of Emato-Oncologic, Aou San Martino, Largo R Benzi 10, Genoa 16132, Italy
S- Editor Wang JL L- Editor Hughes D E- Editor Zheng XM
| 1. | Burger JW, Luijendijk RW, Hop WC, Halm JA, Verdaasdonk EG, Jeekel J. Long-term follow-up of a randomized controlled trial of suture versus mesh repair of incisional hernia. Ann Surg. 2004;240:578-583; discussion 583-585. [PubMed] |
| 2. | Luijendijk RW, Hop WC, van den Tol MP, de Lange DC, Braaksma MM, IJzermans JN, Boelhouwer RU, de Vries BC, Salu MK, Wereldsma JC. A comparison of suture repair with mesh repair for incisional hernia. N Engl J Med. 2000;343:392-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1317] [Cited by in RCA: 1201] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 3. | Leber GE, Garb JL, Alexander AI, Reed WP. Long-term complications associated with prosthetic repair of incisional hernias. Arch Surg. 1998;133:378-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 652] [Cited by in RCA: 588] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 4. | Jin J, Rosen MJ, Blatnik J, McGee MF, Williams CP, Marks J, Ponsky J. Use of acellular dermal matrix for complicated ventral hernia repair: does technique affect outcomes. J Am Coll Surg. 2007;205:654-660. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 183] [Cited by in RCA: 170] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 5. | Morell VO, Wearden PA. Experience with bovine pericardium for the reconstruction of the aortic arch in patients undergoing a Norwood procedure. Ann Thorac Surg. 2007;84:1312-1315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 6. | Hutson JM, Azmy AF. Preserved dura and pericardium for closure of large abdominal wall and diaphragmatic defects in children. Ann R Coll Surg Engl. 1985;67:107-108. [PubMed] |
| 7. | USHER FC. Hernia repair with Marlex mesh. An analysis of 541 cases. Arch Surg. 1962;84:325-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 104] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 8. | Kim H, Bruen K, Vargo D. Acellular dermal matrix in the management of high-risk abdominal wall defects. Am J Surg. 2006;192:705-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 139] [Cited by in RCA: 139] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 9. | Mathes SJ, Steinwald PM, Foster RD, Hoffman WY, Anthony JP. Complex abdominal wall reconstruction: a comparison of flap and mesh closure. Ann Surg. 2000;232:586-596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 214] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 10. | de Vries Reilingh TS, van Goor H, Rosman C, Bemelmans MH, de Jong D, van Nieuwenhoven EJ, van Engeland MI, Bleichrodt RP. "Components separation technique" for the repair of large abdominal wall hernias. J Am Coll Surg. 2003;196:32-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 218] [Cited by in RCA: 176] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 11. | Lowe JB, Lowe JB, Baty JD, Garza JR. Risks associated with "components separation" for closure of complex abdominal wall defects. Plast Reconstr Surg. 2003;111:1276-1283; quiz 1284-1285; discussion 1286-1288. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 90] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 12. | Cavallaro A, Lo Menzo E, Di Vita M, Zanghì A, Cavallaro V, Veroux PF, Cappellani A. Use of biological meshes for abdominal wall reconstruction in highly contaminated fields. World J Gastroenterol. 2010;16:1928-1933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 79] [Cited by in RCA: 65] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Post C, Koziol D. Bovine pericardium for the repair of large abdominal wall hernias. 81 Annual Meeting of the Association of Bavarian Surgeons;. 2004;Jul 21-23; Munich, Germany. |
| 14. | Lindermann M, Urbach V, Paolucci V. Incisional hernia repair with a bioimplant derived from bovine pericardium. 81 Annual Meeting of the Association of Bavarian Surgeons;. 2004;Jul 21-23; Munich, Germany. |
| 15. | Gupta A, Zahriya K, Mullens PL, Salmassi S, Keshishian A. Ventral herniorrhaphy: experience with two different biosynthetic mesh materials, Surgisis and Alloderm. Hernia. 2006;10:419-425. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 116] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 16. | Limpert JN, Desai AR, Kumpf AL, Fallucco MA, Aridge DL. Repair of abdominal wall defects with bovine pericardium. Am J Surg. 2009;198:e60-e65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |