Published online Aug 27, 2025. doi: 10.4240/wjgs.v17.i8.108312
Revised: May 29, 2025
Accepted: July 11, 2025
Published online: August 27, 2025
Processing time: 125 Days and 0.8 Hours
Pancreatic duct stones (PDS) pose substantial therapeutic challenges owing to their tendency to induce recurrent pain and discomfort via mechanical irritation and ductal obstruction, resulting in a marked decline in patients’ quality of life. The pathogenesis of pancreatic duct calculi remains incompletely understood due to its multifactorial and intricate nature. Advances in minimally invasive endoscopic interventions have broadened the available treatment modalities for PDS. This review summarizes current perspectives on the mechanisms driving stone formation alongside evolving therapeutic strategies.
Core Tip: Pancreatic duct stones represent a complication associated with chronic pancreatitis. This review highlights recent progress in understanding the pathophy
- Citation: Wu S, Zheng Y, Mou JW, Zhang YJ, Zhu KX. Mechanism and treatment of pancreatic duct stones. World J Gastrointest Surg 2025; 17(8): 108312
- URL: https://www.wjgnet.com/1948-9366/full/v17/i8/108312.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v17.i8.108312
Pancreatic duct stones (PDS) represent a prevalent complication of chronic pancreatitis (CP)[1]. CP arises from diverse etiologies that induce structural remodeling of the pancreas and disrupt both exocrine and endocrine functions, ultimately promoting the intraductal deposition of calcium carbonate or proteins, which aggregate into stones or protein plugs. Obstruction caused by PDS elevates intraductal pressure, elicits abdominal pain, and compromises pancreatic function. Clinically, patients often exhibit persistent epigastric pain, steatorrhea, malnutrition, and a spectrum of complications associated with pancreatic insufficience[2]. PDS are classified as either true calculi - located within the pancreatic duct - or pseudocalculi, referring to calcifications within the pancreatic parenchyma. Management strategies encompass pharmacological interventions, minimally invasive endoscopic procedures, and surgical approaches. The advent and refinement of endoscopic techniques, owing to their reduced invasiveness, have positioned them as the preferred initial treatment modality for many patient[3]. A comprehensive understanding of the mechanisms driving stone development is essential for refining both prophylactic and interventional strategies. This review consolidates existing evidence on PDS pathogenesis and recent clinical management innovations.
Multiple risk factors have been implicated in the development of PDS, including CP, chronic alcohol consumption, hypercalcemia, malnutrition, and parasitic infections of the biliary or pancreatic ducts. The underlying mechanisms of stone formation are multifactorial and remain incompletely defined. Current clinical and experimental findings provide several key insights.
Pancreatic stone protein (PSP), synthesized and secreted by pancreatic acinar cells, was first isolated from human PDS in 1979[4]. PSP is detectable in both normal and pathological pancreatic secretions and is encoded by the regenerating gene family. The human regenerating gene, a single-copy gene comprising six exons and five introns with a total length of 3.0 kb, is located on chromosome IIp1 bands 2-3. In vitro studies by Multigner et al[5] suggested a strong association between PSP levels in pancreatic fluid and stone formation. These findings indicate that while PSP is a physiological component of pancreatic secretions, its concentration is significantly reduced in individuals who develop PDS. This observation supports the hypothesis that PSP may serve as an endogenous inhibitor of calcium carbonate crystallization, primarily by impeding crystal nucleation and subsequent stone growth. Persistent high protein levels in pancreatic fluid, induced by diverse pathogenic stimuli, promotes intraductal protein aggregate formation. These aggregates trigger epithelial hypertrophy and squamous metaplasia, diminishing PSP synthesis and its inhibitory capacity. The resulting environment favors calcium carbonate precipitation and the organization of protein aggregates into stones. Contradictory evidence challenges the proposed inhibitory role of PSP[6]. One study identified PSP-S1 - a cleavage product of PSP generated by trypsin - is prevalent in pancreatic fluid and constitutes the primary component of the organic matrix in PDS[4]. PSP-S1 is insoluble at physiological pH, predisposing it to aggregation and subsequent stone formation. This suggests that, under certain conditions, PSP or its derivatives may actually facilitate lithogenesis rather than prevent it. Divergent experimental protocols likely account for the contradictory interpretations. In the latter experiment[4], physiological levels of NaCl and phosphate ions within pancreatic fluid markedly suppressed crystal nucleation. Such discrepancies highlight the unresolved nature of PSP’s role in PDS development and highlight the necessity for rigorously designed investigations to clarify its mechanistic contribution.
Cystic fibrosis transmembrane transduction regulator (CFTR) (also known as cystic fibrosis transmembrane conductance regulator protein), a cyclic adenosine monophosphate-regulated chloride channel, is selectively localized to the apical membrane of pancreatic ductal epithelial cells and plays a pivotal role in regulating the secretion of pancreatic fluid and bicarbonate[7]. In the context of CP, the trafficking and membrane localization of CFTR are frequently disrupted, re
Lactoferrin (LF), a member of the transferrin family and an iron-binding glycoprotein comprising approximately 700 amino acids, is found in human milk and various other secretions. LF was first isolated from bovine milk by Sorensen in 1939, while iron-binding proteins were not identified in human milk until 1961 by Blanc in France. Subsequent research by Hayakawa et al[13] demonstrated that LF concentrations were markedly elevated in patients with chronic calcific pancreatitis compared to both healthy controls and individuals with non-calcific CP or suspected pancreatitis. Immunohistochemical analyses revealed strong LF staining in the cytoplasm of squamous epithelial cells within pancreatic ducts containing LF plugs. Additionally, LF accumulation was markedly elevated in glandular cells and protein plugs of dilated ducts in comparison to normal pancreatic tissue[14]. These observations suggest that LF may contribute to the nucleation and early aggregation of proteinaceous material within the pancreatic ductal system. Further studies have proposed that LF interacts with eosinophilic proteins—potentially synthesized by LF itself - to form insoluble protein complexes that deposit within the ducts and act as precursors to PDS formation[15]. However, whether LF found in PDS originates from intrinsic pancreatic secretions or is transported via systemic circulation remains unresolved, necessitating further investigation to clarify its role in lithogenesis.
Osteopontin (OPN), an acidic phosphoprotein initially identified by Oldberg in 1986 during research on bone-specific cell adhesion proteins, constitutes a major component of the bone matrix. Nakamura et al[16] reported OPN expression in pancreatic acinar cells, ductal epithelium, and infiltrating macrophages in patients diagnosed with CP. Notably, OPN was present in PDS but undetectable in normal pancreatic tissue. Detection of OPN mRNA within stone specimens further substantiates its local synthesis. Quantitative analyses revealed markedly elevated OPN levels in patients with PDS compared to those with CP absent of calculi. Given OPN’s established involvement in urinary stone formation, it is postulated to contribute to calcium salt deposition in the pancreas as well[17], suggesting a role in the development of PDS. Nonetheless, the underlying molecular pathways through which OPN mediates stone formation remain unclear.
The calcium-sensing receptor (CaSR), a G-protein-coupled receptor essential for maintaining calcium balance, was first cloned from bovine parathyroid tissue by Brown et al[18] in 1993. It is widely expressed in tissues involved in calcium regulation, including pancreatic ductal cells. CaSR regulates responses to elevated extracellular calcium by enhancing ductal fluid secretion, thereby lowering the calcium concentration in pancreatic fluid and preventing stone formation under hypercalcemic conditions[19]. Mutations in the CaSR gene, such as the p.A986S homozygous variant, have been significantly associated with CP[20]. These genetic variants disrupt calcium regulation both intracellularly and extracellularly, leading to elevated calcium ion levels in pancreatic secretions and promoting the deposition of insoluble calcium salts, which ultimately drives PDS development.
In addition to the mechanisms previously described, ethyl alcohol, pancreatic duct obstruction, and recurrent episodes of CP contribute significantly to the pathogenesis of PDS. Sarles et al[21] demonstrated that ethanol stimulates pancreatic acinar cell secretion, promoting the formation of proteinaceous emboli containing calcium carbonate, which progressively develop into PDS. Experimental models of PDS have been successfully established through intentional pancreatic duct ligation in animals, confirming that impaired pancreatic fluid outflow can precipitate stone formation[22]. In recurrent CP, sustained inflammation and parenchymal necrosis induce lipolysis, enabling the liberated fatty acids to chelate calcium ions, forming insoluble calcium salts that precipitate within the ductal system.
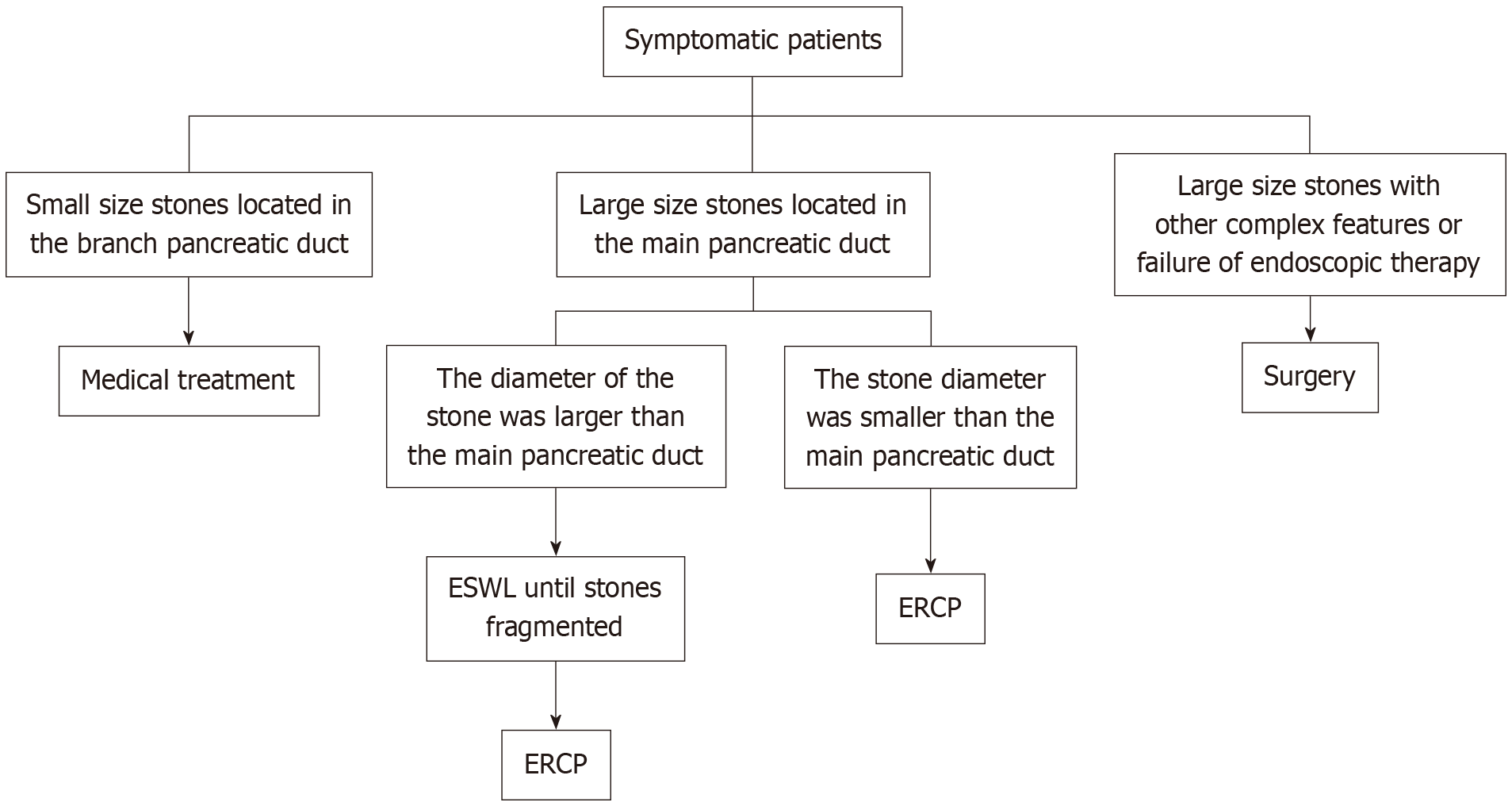
Management of PDS is primarily guided by stone burden and anatomical localization. Therapeutic approaches include pharmacological treatment, endoscopic intervention, and surgical resection. Conservative endoscopic methods have restricted applicability, limited by their narrow clinical indications. In contrast, minimally invasive endoscopic techniques have emerged as the preferred initial strategy due to reduced procedural trauma, the capacity to delay or avert surgical intervention, and their predictive value for operative outcomes. Numerous studies indicate that removing obstructive calculi from the main pancreatic duct significantly mitigates symptoms in most patients with CP[23]. In cases of painful obstructive CP, surgery provides more durable analgesia[24] and remains the definitive option when endoscopic measures prove inadequate. Our suggested general approach for managing PDS patients is summarized in Figure 1.
Oral pancreatic enzyme replacement, analgesics, and strict adherence to a low-fat diet represent the core therapeutic strategies for CP management, regardless of PDS status. These measures do not alter stone burden but primarily function by suppressing cholecystokinin-mediated exocrine stimulation and reducing intraductal pressure, thereby mitigating pain associated with ductal hypertension[25]. No pharmacologic agent currently offers both a favorable safety profile and proven efficacy for PDS dissolution. Although trimethadione, an older antiepileptic, has demonstrated some capacity to dissolve PDS[26]. its clinical applicability is constrained by severe adverse effects, including hematotoxicity, hepatorenal impairment, and neuropsychological complications during prolonged use. The lack of a non-invasive pharmacologic solution highlights the imperative for further therapeutic exploration.
Minimally invasive endoscopic therapy remains the preferred treatment modality for PDS, primarily targeting stone extraction to relieve ductal obstruction and associated pain. While most evidence suggests that endoscopic interventions do not enhance pancreatic function, magnetic resonance cholangiopancreatography imaging has indicated potential improvement in exocrine function post-treatment[27]. Endoscopic techniques include lithotripsy, per-oral pancreatoscopy (POP), and extracorporeal shock wave lithotripsy (ESWL).
Endoscopic treatment of PDS: Endoscopic techniques encompass endoscopic retrograde cholangiopancreatography (ERCP) paired with endoscopic sphincterotomy (EST), along with lithotripsy using balloon, basket, or stent placement. The European Society of Gastrointestinal Endoscopy recommends ERCP with EST as a first-line intervention for symptomatic obstructive CP involving the main pancreatic duct in the head or body of the pancreas[28]. ERCP facilitates pain relief by performing EST, dilating strictures, extracting PDS via balloon or basket, and reducing intraductal pressure through pancreatic duct stenting, thereby restoring ductal patency and mitigating symptoms.
The combination of EST with transpancreatic duct balloon dilatation, stone extraction, and basket lithotripsy represents a safe and effective approach for removing impacted stones from the main pancreatic duct, offering advantages such as minimal invasiveness, procedural efficiency, and improved safety profile[29]. These techniques facilitate the clearance of PDS, allowing evacuation of stones, sludge, and debris from the pancreatic ductal system into the duodenal lumen. Basket mechanical lithotripsy, which involves fragmenting stones using a retrieval basket, enables the removal of otherwise difficult-to-extract calculi[30]. However, this method is limited by its ineffectiveness in ducts with a diameter of less than 6 mm. For embedded stones, mechanical fragmentation with a basket enhances accessibility for extraction. Despite its utility, basket lithotripsy carries inherent risks, including basket entrapment, wire fracture, and pancreatic duct injury, particularly when manipulating stones larger than the distal ductal diameter[31]. In contrast, stone extraction balloons offer a safer alternative during ERCP. Unlike baskets, balloons can be deflated, thereby minimizing the risk of entrapment within the duct, and are associated with a lower complication rate[32]. However, their mechanical li
In cases of pancreatic duct stenosis, stent placement is essential to achieve ductal dilatation and facilitate stone extraction. Benign strictures typically arise from periductal inflammation and fibrosis[35]. and in patients with CP, these strictures often consist of dense fibrotic tissue. As a result, balloon dilatation alone rarely yields durable therapeutic efficacy. Standard management involves sequential stenting, employing stents of increasing caliber over a 6-12 months period to progressively expand the duct and resolve the stricture. This strategy has been associated with effective pain mitigation, with immediate relief reported in 65%-95% of cases and sustained benefit observed in 32%-68%[36], su
POP combined with electrohydraulic lithotripsy or laser lithotripsy: First introduced by Kawai et al[40] in 1974, POP enables direct endoscopic access to the main pancreatic duct, permitting both targeted biopsy and therapeutic manipulation. In the management of CP complicated by PDS, the integration of POP with either electrohydraulic lithotripsy (EHL) or laser lithotripsy (LL) has increasingly been adopted as a therapeutic strategy[41]. EHL delivers high-amplitude hydraulic shockwaves via dual electrodes to fragment intraductal calculi, while LL utilizes concentrated high-energy laser pulses for stone disintegration. European Society of Gastrointestinal Endoscopy guidelines recommend POP-guided lithotripsy as an alternative when ERCP combined with ESWL is contraindicated or fails to achieve ductal clearance[28]. Comparative studies suggest that LL outperforms EHL in both technical and clinical metrics, likely due to its superior capability in disintegrating high-density stones[42]. A meta-analysis reported a pooled technical success rate of 76.8% for POP-guided lithotripsy, accompanied by a 14.9% complication rate. The most frequently encountered adverse events included abdominal pain (4.7%), post-procedural acute pancreatitis (7%), bleeding (3.4%), and ductal perforation (4.3%)[43]. Although effective, the application of POP-guided lithotripsy remains constrained by the requirement for specialized instrumentation and advanced endoscopic proficiency, both essential for reducing procedural risk and optimizing outcomes.
ESWL: ESWL, developed in the 1980s, employs externally generated focused shock waves to fragment intraductal stones. First applied in gastroenterology by Sauerbruch et al[44] in 1987. Since its adoption, ESWL has become a major therapeutic approach for managing PDS, primarily by decreasing stone burden to promote spontaneous passage or improve the efficiency of subsequent ERCP. Indications include large or multiple stones, especially in the presence of pancreatic duct stenosis, with repeated sessions often required for optimal outcomes[28]. In selected patients, ESWL is recognized as either an adjunct or a first-line intervention[23,28]. For radiopaque stones > 5 mm located in the pancreatic head or body, clinical guidelines recommend ESWL followed by ERCP to achieve definitive clearance[28]. Recent evidence reported a 50% complete clearance rate post-ESWL[45], while long-term follow-up indicates that combining ESWL with endoscopic drainage provides sustained pain relief and can prevent surgical intervention in nearly two-thirds of cases[46,47]. However, concerns have emerged regarding combined therapy. A comparative study evaluating ESWL alone (n = 26) vs ESWL with ERCP (n = 29) revealed that the addition of ERCP was associated with a higher incidence of pancreatic pain and increased overall cost[48]. Consequently, ESWL monotherapy remains a safe and effective strategy in appropriately selected cases. Reported complications include skin contusion, bleeding, infection, papillary impaction, and the formation of Steinstrasse - accumulated fragmented stones within the duct[49]. Post-ESWL pancreatitis has been observed in 6.3%-12.5% of patients undergoing the procedure alone for calcific CP[50,51]. In cases of PDS with coexisting ductal stenosis, insufficient drainage following ESWL may result in obstructive stone accumulation requiring ERCP for resolution. Furthermore, stones exceeding 10 mm often necessitate multiple treatment sessions, highlighting the importance of experienced clinical operators.
Surgical management remains the definitive approach for PDS, aiming to achieve complete stone clearance, mitigate pain, preserve pancreatic function, and optimize long-term outcomes and postoperative quality of life. Comparative studies by Díte et al[52] and Cahen et al[53] demonstrated substantially higher pain relief rates following surgery (75%-86%) compared to endoscopic treatment (32%-61%). Indications for surgery include: (1) Extensive or multiple PDS and complex ductal strictures; (2) Inflammatory mass in the pancreatic head or disease confined to the pancreatic tail; and (3) Clinical suspicion of malignancy not definitively excluded by nonoperative evaluation.
The two principal surgical strategies involve drainage of pancreatic secretions and resection of affected pancreatic segments. Drainage procedures offer particular benefit for patients presenting with PDS and significant main duct dilatation, due to their procedural simplicity, reduced parenchymal loss, and superior preservation of endocrine and exocrine function. This technique is appropriate for cases with stones confined to the main pancreatic duct and ac
Primary surgical interventions for pancreatic resection include pancreaticoduodenectomy (Whipple’s procedure), duodenum-preserving pancreatic head resection (DPPHR), limited resection combined with pancreatic ductal longitudinal jejunostomy (Frey’s procedure), and total pancreatectomy with autologous islet cell transplantation (TPAIT). The Frey procedure is most appropriate for individuals presenting with pancreatic head calculi and distal ductal dilatation, whereas DPPHR and Whipple’s are generally indicated when malignancy coexists with pancreatic head stones. These approaches may also be considered in the context of obstructive complications. By targeting diseased pancreatic seg
In summary, the pathogenesis of PDS remains incompletely understood. Current hypotheses suggest that PSP impedes calcium carbonate crystal nucleation, thereby limiting ductal stone formation. While LF and OPN are consistently identified within PDS, their mechanistic contributions remain to be clearly defined. Aberrant localization of CFTR in pancreatic ductal epithelial cells has also been implicated in lithogenesis. Elucidation of the molecular functions of PSP, LF, and OPN through gene-editing technologies and validated animal models is essential for delineating their roles in stone formation and identifying potential therapeutic targets. Gene therapy offers a promising approach to restoring proper CFTR localization, thereby reducing the propensity for stone development. Additionally, the discovery of novel litholytic agents may help overcome current limitations in pharmacologic management. The CaSR, a key modulator of calcium balance, has been implicated in PDS pathogenesis through genetic mutations, although its precise molecular role remains unresolved. Clarifying these mechanisms necessitates rigorously designed, multicenter studies with substantial cohort sizes. Technological advancements in minimally invasive endoscopic therapy have broadened the interventional spectrum for PDS, incorporating modalities such as ESWL, ERCP with EST, and adjunctive tools including retrieval baskets, extraction balloons, and pancreatic duct stents. Integration of multiple techniques represents a forward-looking approach, offering potential reductions in residual stone rates and enhanced therapeutic outcomes. In particular, POP combined with EHL or LL has demonstrated efficacy in managing calculi of diverse sizes and anatomical distributions. Each approach entails specific advantages and constraints, highlighting the importance of further prospective, ran
| 1. | Farnbacher MJ, Schoen C, Rabenstein T, Benninger J, Hahn EG, Schneider HT. Pancreatic duct stones in chronic pancreatitis: criteria for treatment intensity and success. Gastrointest Endosc. 2002;56:501-506. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 23] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 2. | Beyer G, Habtezion A, Werner J, Lerch MM, Mayerle J. Chronic pancreatitis. Lancet. 2020;396:499-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 162] [Cited by in RCA: 310] [Article Influence: 62.0] [Reference Citation Analysis (1)] |
| 3. | Singh VK, Yadav D, Garg PK. Diagnosis and Management of Chronic Pancreatitis: A Review. JAMA. 2019;322:2422-2434. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 301] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 4. | De Caro A, Lohse J, Sarles H. Characterization of a protein isolated from pancreatic calculi of men suffering from chronic calcifying pancreatitis. Biochem Biophys Res Commun. 1979;87:1176-1182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 113] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 5. | Multigner L, De Caro A, Lombardo D, Campese D, Sarles H. Pancreatic stone protein, a phosphoprotein which inhibits calcium carbonate precipitation from human pancreatic juice. Biochem Biophys Res Commun. 1983;110:69-74. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 99] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 6. | De Reggi M, Gharib B, Patard L, Stoven V. Lithostathine, the presumed pancreatic stone inhibitor, does not interact specifically with calcium carbonate crystals. J Biol Chem. 1998;273:4967-4971. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 7. | Choi JY, Muallem D, Kiselyov K, Lee MG, Thomas PJ, Muallem S. Aberrant CFTR-dependent HCO3- transport in mutations associated with cystic fibrosis. Nature. 2001;410:94-97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 298] [Cited by in RCA: 309] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 8. | Ko SB, Azuma S, Yoshikawa T, Yamamoto A, Kyokane K, Ko MS, Ishiguro H. Molecular mechanisms of pancreatic stone formation in chronic pancreatitis. Front Physiol. 2012;3:415. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 9. | Ko SB, Mizuno N, Yatabe Y, Yoshikawa T, Ishiguro H, Yamamoto A, Azuma S, Naruse S, Yamao K, Muallem S, Goto H. Corticosteroids correct aberrant CFTR localization in the duct and regenerate acinar cells in autoimmune pancreatitis. Gastroenterology. 2010;138:1988-1996. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 75] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 10. | Gelrud A, Sheth S, Banerjee S, Weed D, Shea J, Chuttani R, Howell DA, Telford JJ, Carr-Locke DL, Regan MM, Ellis L, Durie PR, Freedman SD. Analysis of cystic fibrosis gener product (CFTR) function in patients with pancreas divisum and recurrent acute pancreatitis. Am J Gastroenterol. 2004;99:1557-1562. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 65] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 11. | Tabary O, Zahm JM, Hinnrasky J, Couetil JP, Cornillet P, Guenounou M, Gaillard D, Puchelle E, Jacquot J. Selective up-regulation of chemokine IL-8 expression in cystic fibrosis bronchial gland cells in vivo and in vitro. Am J Pathol. 1998;153:921-930. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 134] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 12. | Bertin C, Pelletier AL, Vullierme MP, Bienvenu T, Rebours V, Hentic O, Maire F, Hammel P, Vilgrain V, Ruszniewski P, Lévy P. Pancreas divisum is not a cause of pancreatitis by itself but acts as a partner of genetic mutations. Am J Gastroenterol. 2012;107:311-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 142] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 13. | Hayakawa T, Naruse S, Kitagawa M, Nakae Y, Harada H, Ochi K, Kuno N, Kurimoto K, Hayakawa S. Pancreatic stone protein and lactoferrin in human pancreatic juice in chronic pancreatitis. Pancreas. 1995;10:137-142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 14] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 14. | Nagai H, Ohtsubo K. Pancreatic lithiasis in the aged. Its clinicopathology and pathogenesis. Gastroenterology. 1984;86:331-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 41] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Jin CX, Hayakawa T, Kitagawa M, Ishiguro H. Lactoferrin in chronic pancreatitis. JOP. 2009;10:237-241. [PubMed] |
| 16. | Nakamura M, Oka M, Iizuka N, Kawauchi S, Gondo T, Ueno T, Tangoku A. Osteopontin expression in chronic pancreatitis. Pancreas. 2002;25:182-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 14] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 17. | Langdon A, Grohe B. The osteopontin-controlled switching of calcium oxalate monohydrate morphologies in artificial urine provides insights into the formation of papillary kidney stones. Colloids Surf B Biointerfaces. 2016;146:296-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 18. | Brown EM, Gamba G, Riccardi D, Lombardi M, Butters R, Kifor O, Sun A, Hediger MA, Lytton J, Hebert SC. Cloning and characterization of an extracellular Ca(2+)-sensing receptor from bovine parathyroid. Nature. 1993;366:575-580. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1993] [Cited by in RCA: 1765] [Article Influence: 55.2] [Reference Citation Analysis (0)] |
| 19. | Rácz GZ, Kittel A, Riccardi D, Case RM, Elliott AC, Varga G. Extracellular calcium sensing receptor in human pancreatic cells. Gut. 2002;51:705-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 54] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 20. | Masson E, Chen JM, Férec C. Overrepresentation of Rare CASR Coding Variants in a Sample of Young French Patients With Idiopathic Chronic Pancreatitis. Pancreas. 2015;44:996-998. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Sarles H. Chronic calcifying pancreatitis. Scand J Gastroenterol. 1985;20:651-659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 10] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 22. | Tanaka T, Miura Y, Matsugu Y, Ichiba Y, Ito H, Dohi K. Pancreatic duct obstruction is an aggravating factor in the canine model of chronic alcoholic pancreatitis. Gastroenterology. 1998;115:1248-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 24] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 23. | Dumonceau JM, Devière J, Le Moine O, Delhaye M, Vandermeeren A, Baize M, Van Gansbeke D, Cremer M. Endoscopic pancreatic drainage in chronic pancreatitis associated with ductal stones: long-term results. Gastrointest Endosc. 1996;43:547-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 154] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 24. | Chinnakotla S, Radosevich DM, Dunn TB, Bellin MD, Freeman ML, Schwarzenberg SJ, Balamurugan AN, Wilhelm J, Bland B, Vickers SM, Beilman GJ, Sutherland DE, Pruett TL. Long-term outcomes of total pancreatectomy and islet auto transplantation for hereditary/genetic pancreatitis. J Am Coll Surg. 2014;218:530-543. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 114] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 25. | Isaksson G, Ihse I. Pain reduction by an oral pancreatic enzyme preparation in chronic pancreatitis. Dig Dis Sci. 1983;28:97-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 121] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 26. | Noda A, Okuyama M, Murayama H, Takeuchi K, Yokota T, Kobayashi T, Takayama T. Dissolution of pancreatic stones by oral trimethadione in patients with chronic calcific pancreatitis. J Gastroenterol Hepatol. 1994;9:478-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 27. | Bali MA, Sztantics A, Metens T, Arvanitakis M, Delhaye M, Devière J, Matos C. Quantification of pancreatic exocrine function with secretin-enhanced magnetic resonance cholangiopancreatography: normal values and short-term effects of pancreatic duct drainage procedures in chronic pancreatitis. Initial results. Eur Radiol. 2005;15:2110-2121. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 58] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 28. | Dumonceau JM, Delhaye M, Tringali A, Arvanitakis M, Sanchez-Yague A, Vaysse T, Aithal GP, Anderloni A, Bruno M, Cantú P, Devière J, Domínguez-Muñoz JE, Lekkerkerker S, Poley JW, Ramchandani M, Reddy N, van Hooft JE. Endoscopic treatment of chronic pancreatitis: European Society of Gastrointestinal Endoscopy (ESGE) Guideline - Updated August 2018. Endoscopy. 2019;51:179-193. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 168] [Cited by in RCA: 248] [Article Influence: 41.3] [Reference Citation Analysis (0)] |
| 29. | Maydeo A, Bhandari S, Bapat M. Endoscopic balloon sphincteroplasty for extraction of large radiolucent pancreatic duct stones (with videos). Gastrointest Endosc. 2009;70:798-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 30. | Thomas M, Howell DA, Carr-Locke D, Mel Wilcox C, Chak A, Raijman I, Watkins JL, Schmalz MJ, Geenen JE, Catalano MF. Mechanical lithotripsy of pancreatic and biliary stones: complications and available treatment options collected from expert centers. Am J Gastroenterol. 2007;102:1896-1902. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 74] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 31. | Nabi Z, Lakhtakia S. Endoscopic management of chronic pancreatitis. Dig Endosc. 2021;33:1059-1072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | ASGE Technology Committee; Adler DG, Conway JD, Farraye FA, Kantsevoy SV, Kaul V, Kethu SR, Kwon RS, Mamula P, Pedrosa MC, Rodriguez SA, Tierney WM. Biliary and pancreatic stone extraction devices. Gastrointest Endosc. 2009;70:603-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 28] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 33. | Tadenuma H, Ishihara T, Yamaguchi T, Tsuchiya S, Kobayashi A, Nakamura K, Sakurada R, Saisho H. Long-term results of extracorporeal shockwave lithotripsy and endoscopic therapy for pancreatic stones. Clin Gastroenterol Hepatol. 2005;3:1128-1135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 52] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 34. | Sharzehi K. Management of Pancreatic Duct Stones. Curr Gastroenterol Rep. 2019;21:63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 35. | Choi EK, Lehman GA. Update on endoscopic management of main pancreatic duct stones in chronic calcific pancreatitis. Korean J Intern Med. 2012;27:20-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 36. | Adler JM, Gardner TB. Endoscopic Therapies for Chronic Pancreatitis. Dig Dis Sci. 2017;62:1729-1737. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 37. | Wang DQ, Portincasa P, Liu M, Tso P. Overcoming Ductal Block: Emergency ERCP and Sphincterotomy Plus Common Bile Duct Stenting Improves Therapeutic Outcomes in Severe Gallstone Pancreatitis. Dig Dis Sci. 2022;67:11-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 38. | Adler DG, Lichtenstein D, Baron TH, Davila R, Egan JV, Gan SL, Qureshi WA, Rajan E, Shen B, Zuckerman MJ, Lee KK, VanGuilder T, Fanelli RD. The role of endoscopy in patients with chronic pancreatitis. Gastrointest Endosc. 2006;63:933-937. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 87] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 39. | Wang P, Li ZS, Liu F, Ren X, Lu NH, Fan ZN, Huang Q, Zhang X, He LP, Sun WS, Zhao Q, Shi RH, Tian ZB, Li YQ, Li W, Zhi FC. Risk factors for ERCP-related complications: a prospective multicenter study. Am J Gastroenterol. 2009;104:31-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 331] [Article Influence: 20.7] [Reference Citation Analysis (1)] |
| 40. | Kawai K, Akasaka Y, Murakami K, Tada M, Koli Y. Endoscopic sphincterotomy of the ampulla of Vater. Gastrointest Endosc. 1974;20:148-151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 514] [Cited by in RCA: 454] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 41. | Han S, Miley A, Akshintala V, Freeman ML, Kahaleh M, Othman M, Patel S, Papachristou GI, Raijman I, Sankey N, Sayana H, Singh V, Tarnasky P, Trikudanathan G, Shah RJ. Per-oral pancreatoscopy-guided lithotripsy vs. extracorporeal shock wave lithotripsy for treating refractory main pancreatic duct stones in chronic pancreatitis: Protocol for an open-label multi-center randomized clinical trial. Pancreatology. 2022;22:1120-1125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 14] [Reference Citation Analysis (0)] |
| 42. | Brewer Gutierrez OI, Raijman I, Shah RJ, Elmunzer BJ, Webster GJM, Pleskow D, Sherman S, Sturgess RP, Sejpal DV, Ko C, Maurano A, Adler DG, Mullady DK, Strand DS, DiMaio CJ, Piraka C, Sharahia R, Dbouk MH, Han S, Spiceland CM, Bekkali NLH, Gabr M, Bick B, Dwyer LK, Han D, Buxbaum J, Zulli C, Cosgrove N, Wang AY, Carr-Locke D, Kerdsirichairat T, Aridi HD, Moran R, Shah S, Yang J, Sanaei O, Parsa N, Kumbhari V, Singh VK, Khashab MA. Safety and efficacy of digital single-operator pancreatoscopy for obstructing pancreatic ductal stones. Endosc Int Open. 2019;7:E896-E903. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 38] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 43. | Saghir SM, Mashiana HS, Mohan BP, Dhindsa BS, Dhaliwal A, Chandan S, Bhogal N, Bhat I, Singh S, Adler DG. Efficacy of pancreatoscopy for pancreatic duct stones: A systematic review and meta-analysis. World J Gastroenterol. 2020;26:5207-5219. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 44. | Sauerbruch T, Holl J, Sackmann M, Werner R, Wotzka R, Paumgartner G. Disintegration of a pancreatic duct stone with extracorporeal shock waves in a patient with chronic pancreatitis. Endoscopy. 1987;19:207-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 71] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 45. | Guda NM, Partington S, Freeman ML. Extracorporeal shock wave lithotripsy in the management of chronic calcific pancreatitis: a meta-analysis. JOP. 2005;6:6-12. [PubMed] |
| 46. | Kozarek RA, Brandabur JJ, Ball TJ, Gluck M, Patterson DJ, Attia F, France R, Traverso LW, Koslowski P, Gibbons RP. Clinical outcomes in patients who undergo extracorporeal shock wave lithotripsy for chronic calcific pancreatitis. Gastrointest Endosc. 2002;56:496-500. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 47. | Delhaye M, Arvanitakis M, Verset G, Cremer M, Devière J. Long-term clinical outcome after endoscopic pancreatic ductal drainage for patients with painful chronic pancreatitis. Clin Gastroenterol Hepatol. 2004;2:1096-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 134] [Cited by in RCA: 129] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 48. | Dumonceau JM, Costamagna G, Tringali A, Vahedi K, Delhaye M, Hittelet A, Spera G, Giostra E, Mutignani M, De Maertelaer V, Devière J. Treatment for painful calcified chronic pancreatitis: extracorporeal shock wave lithotripsy versus endoscopic treatment: a randomised controlled trial. Gut. 2007;56:545-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 197] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 49. | Yi JH, Li ZS, Hu LH. Adverse events of pancreatic extracorporeal shock wave lithotripsy: a literature review. BMC Gastroenterol. 2023;23:360. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 50. | Inui K, Tazuma S, Yamaguchi T, Ohara H, Tsuji T, Miyagawa H, Igarashi Y, Nakamura Y, Atomi Y. Treatment of pancreatic stones with extracorporeal shock wave lithotripsy: results of a multicenter survey. Pancreas. 2005;30:26-30. [PubMed] |
| 51. | Ohara H, Hoshino M, Hayakawa T, Kamiya Y, Miyaji M, Takeuchi T, Okayama Y, Gotoh K. Single application extracorporeal shock wave lithotripsy is the first choice for patients with pancreatic duct stones. Am J Gastroenterol. 1996;91:1388-1394. [PubMed] |
| 52. | Díte P, Ruzicka M, Zboril V, Novotný I. A prospective, randomized trial comparing endoscopic and surgical therapy for chronic pancreatitis. Endoscopy. 2003;35:553-558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 384] [Cited by in RCA: 323] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 53. | Cahen DL, Gouma DJ, Nio Y, Rauws EA, Boermeester MA, Busch OR, Stoker J, Laméris JS, Dijkgraaf MG, Huibregtse K, Bruno MJ. Endoscopic versus surgical drainage of the pancreatic duct in chronic pancreatitis. N Engl J Med. 2007;356:676-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 579] [Cited by in RCA: 507] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 54. | Khaled YS, Ammori MB, Ammori BJ. Laparoscopic lateral pancreaticojejunostomy for chronic pancreatitis: a case report and review of the literature. Surg Laparosc Endosc Percutan Tech. 2011;21:e36-e40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 15] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 55. | Ratnayake CBB, Kamarajah SK, Loveday BPT, Nayar M, Oppong K, White S, French JJ, Windsor JA, Pandanaboyana S. A Network Meta-analysis of Surgery for Chronic Pancreatitis: Impact on Pain and Quality of Life. J Gastrointest Surg. 2020;24:2865-2873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 1.4] [Reference Citation Analysis (0)] |