Published online Apr 27, 2024. doi: 10.4240/wjgs.v16.i4.1195
Peer-review started: December 25, 2023
First decision: January 11, 2024
Revised: January 24, 2024
Accepted: March 19, 2024
Article in press: March 19, 2024
Published online: April 27, 2024
Processing time: 118 Days and 21.7 Hours
Percutaneous transhepatic stent placement has become a common strategy for the postoperative treatment of portal vein (PV)/superior mesenteric veins (SMV) ste
Herein, we reported a case of intestinal edema and SMV stenosis 2 d after pan
Stent placement is effective and feasible for treating acute PV/SMV stenosis after pancreatic surgery even within postoperative 3 d.
Core Tip: Portal vein (PV)/superior mesenteric veins (SMV) stenosis/occlusion is an uncommon but severe complication after pancreatic surgery. Stent placement for acute PV/SMV stenosis within 3 d postoperatively was rarely reported. We reported a case showing that percutaneous transhepatic stent placement was an effective and feasible treatment for acute SMV stenosis on postoperative day 2 after the Whipple procedure and could relieve patients’ symptoms. Complications after SMV stent placement, such as bleeding, infection, pancreatic fistula and bile leakage should be fully noticed and carefully managed, especially when considering their interactive effects. Anticoagulation was initiated for preventing stent thrombosis, but it increased risk of bleeding.
- Citation: Lin C, Wang ZY, Dong LB, Wang ZW, Li ZH, Wang WB. Percutaneous transhepatic stenting for acute superior mesenteric vein stenosis after pancreaticoduodenectomy with portal vein reconstruction: A case report. World J Gastrointest Surg 2024; 16(4): 1195-1202
- URL: https://www.wjgnet.com/1948-9366/full/v16/i4/1195.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v16.i4.1195
Portal vein (PV) or superior mesenteric vein (SMV) stenosis can occur as a postoperative complication and has been widely reported after liver transplantation surgery[1,2]. PV/SMV stenosis can also be a complication of pancreatic sur
Two years after transverse colon cancer surgery, four months since finding pancreatic head mass.
A 65-year-old woman underwent PD with PV and SMV resection and reconstruction using an autologous great saphe
The patient had a history of hypertension, diabetes mellitus, and stroke.
The patient had no positive findings in personal and family history.
No jaundice in the patient’s skin and sclera. Courvoisier’s sign (-), bowel sounds 3 times per minute, and rectal exami
No significant laboratory examinations were found for the patient.
The imaging examinations were shown in history of present illness section.
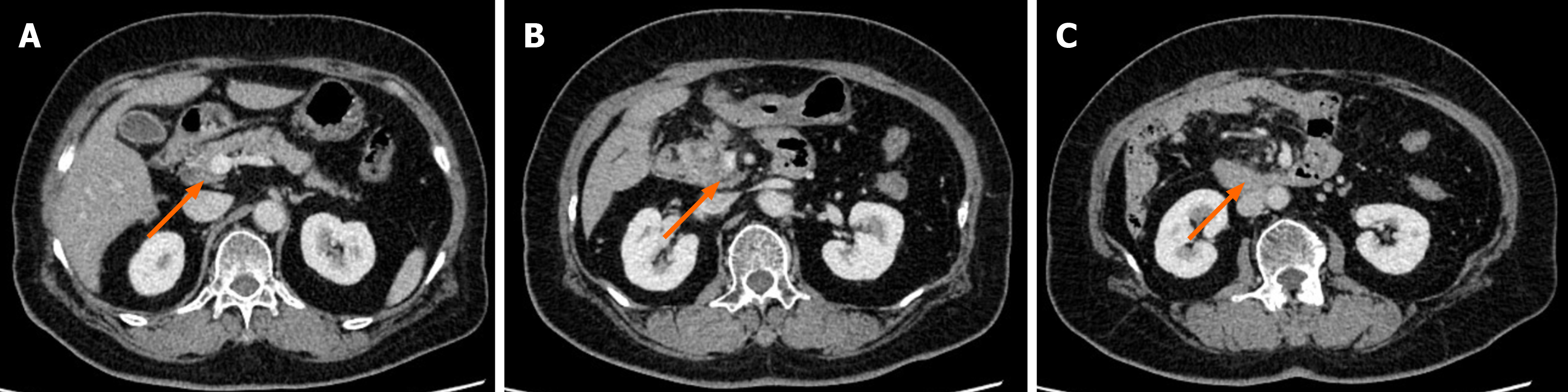
During PD surgery, the mass was confirmed to violate the SMV; therefore, 3 cm of the SMV was resected, and 5 cm of the autologous great saphenous vein was used to reconstruct the SMV. The beginning of the jejunum (length: 20 cm) was also resected after the first jejunal vein was disrupted. On POD 1, the drainage volume of the operative gravidity drain placed close to the biliary-enteric anastomosis was 2800 mL, and the patient’s bowel sounds were absent. Ultrasonography re
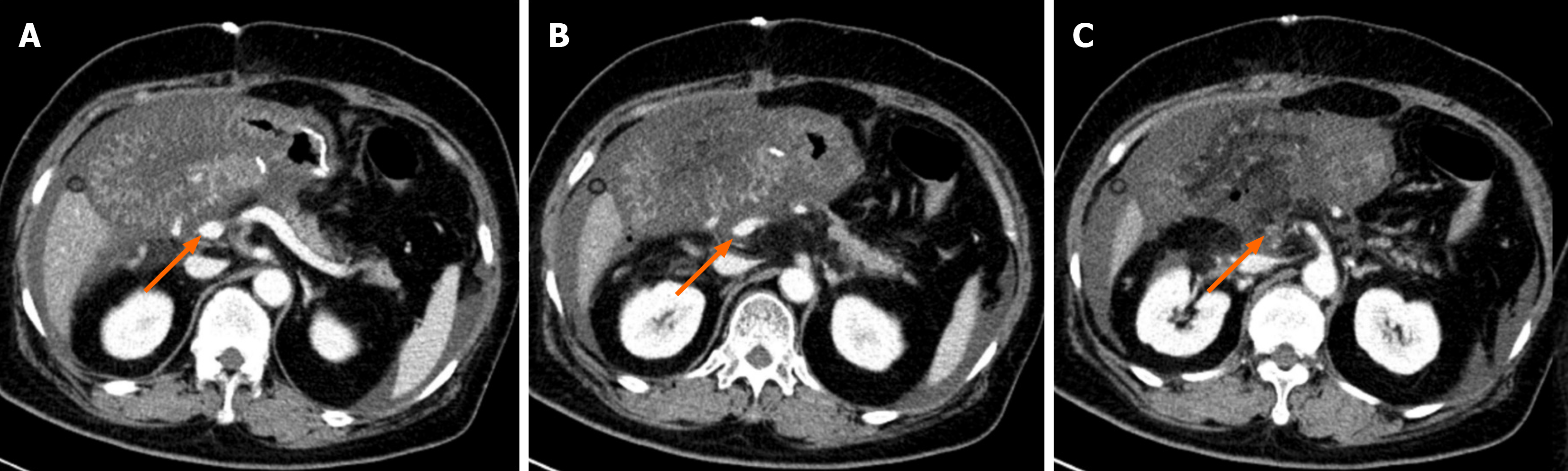
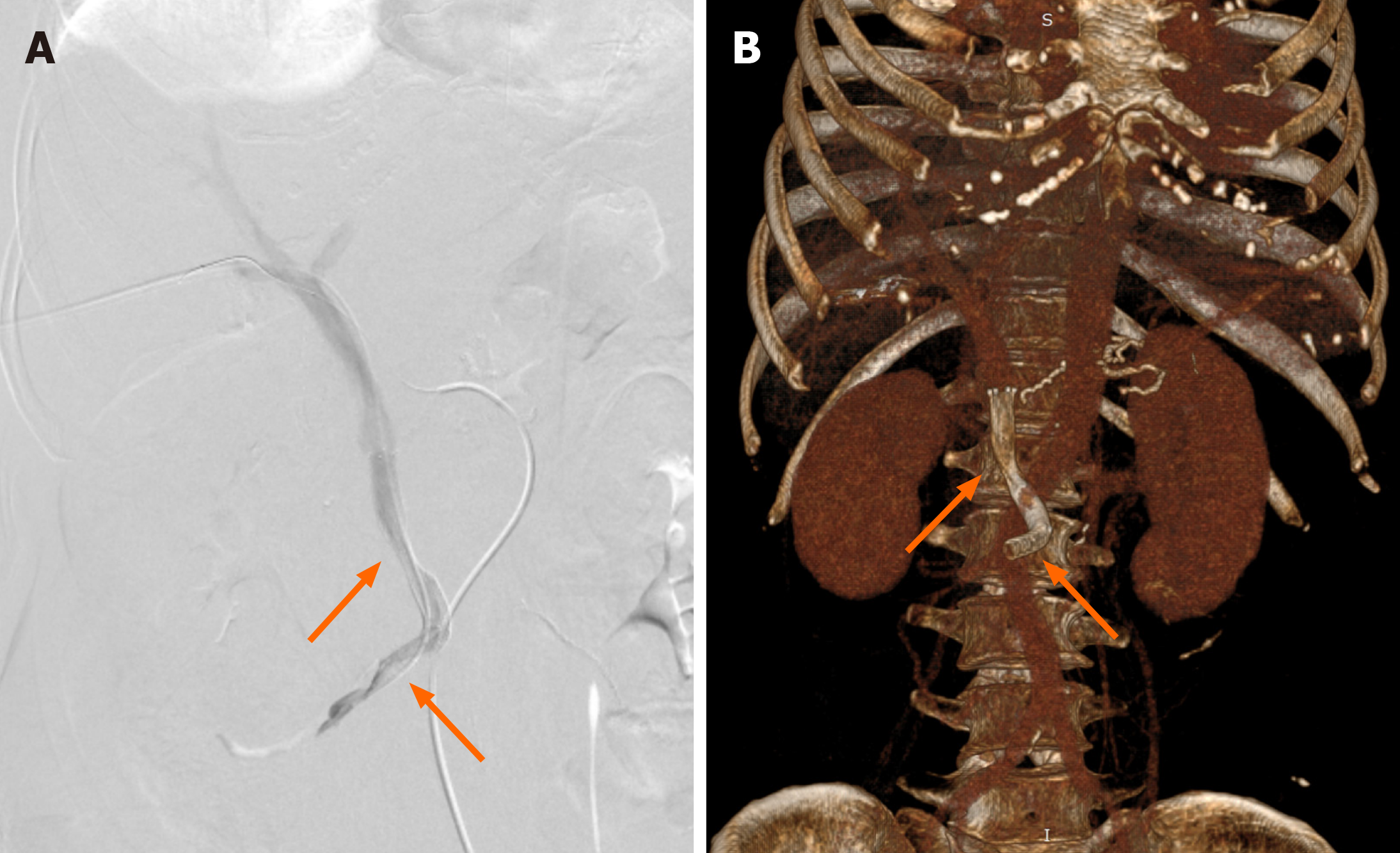
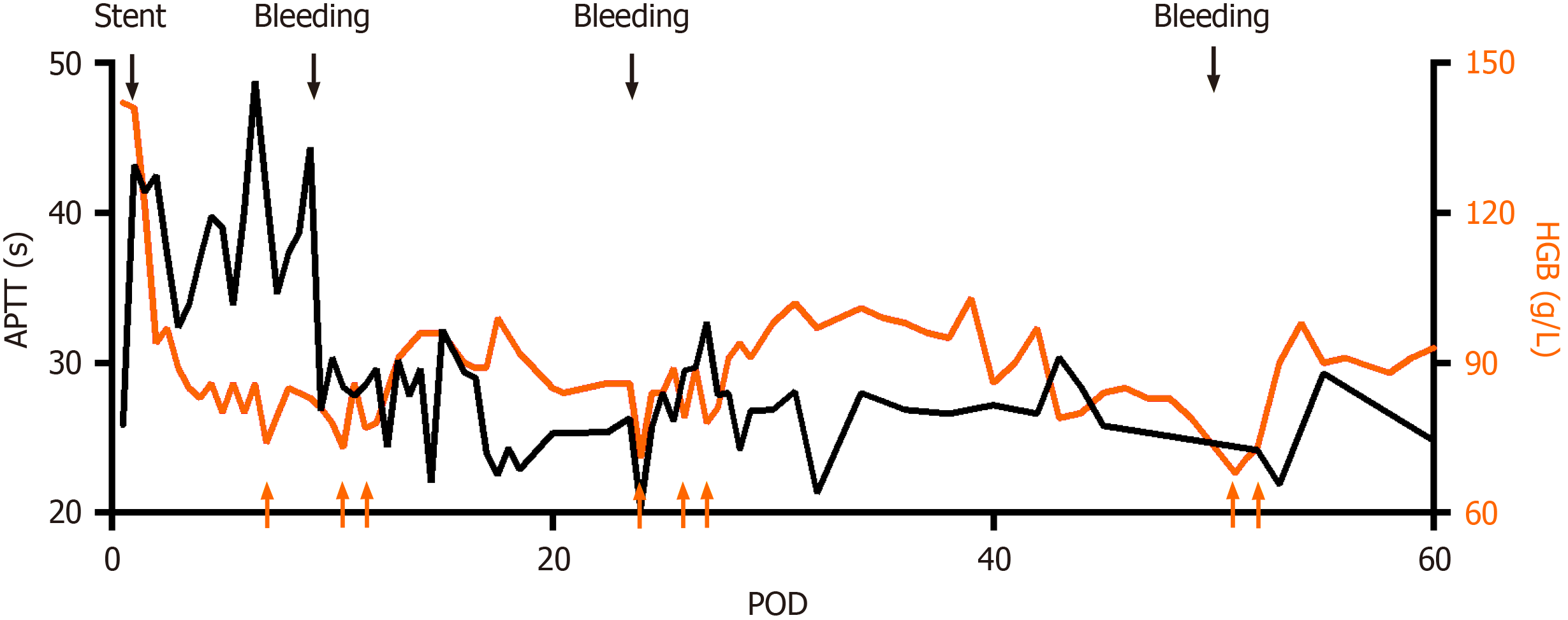
Percutaneous transhepatic stent placement was performed to reestablish blood flow in the PV (Figure 3). Heparin was administered after stent placement to prevent thrombosis with an activated partial thromboplastin time (APTT) goal of 35-40 s, which was 1.5 times of the patient’s baseline level before surgery[12] (Figure 4). On POD 7, thrombosis formation was found in right peroneal vein; therefore, we tested the activity of antithrombin to evaluate heparin’s anticoagulant activity. The activity of AT-III was 67%, suggesting an increased risk of developing abnormal thrombosis and impaired sensitivity to heparin, since heparin functions through antithrombin. Fresh frozen plasma was transfused to rescue sensi
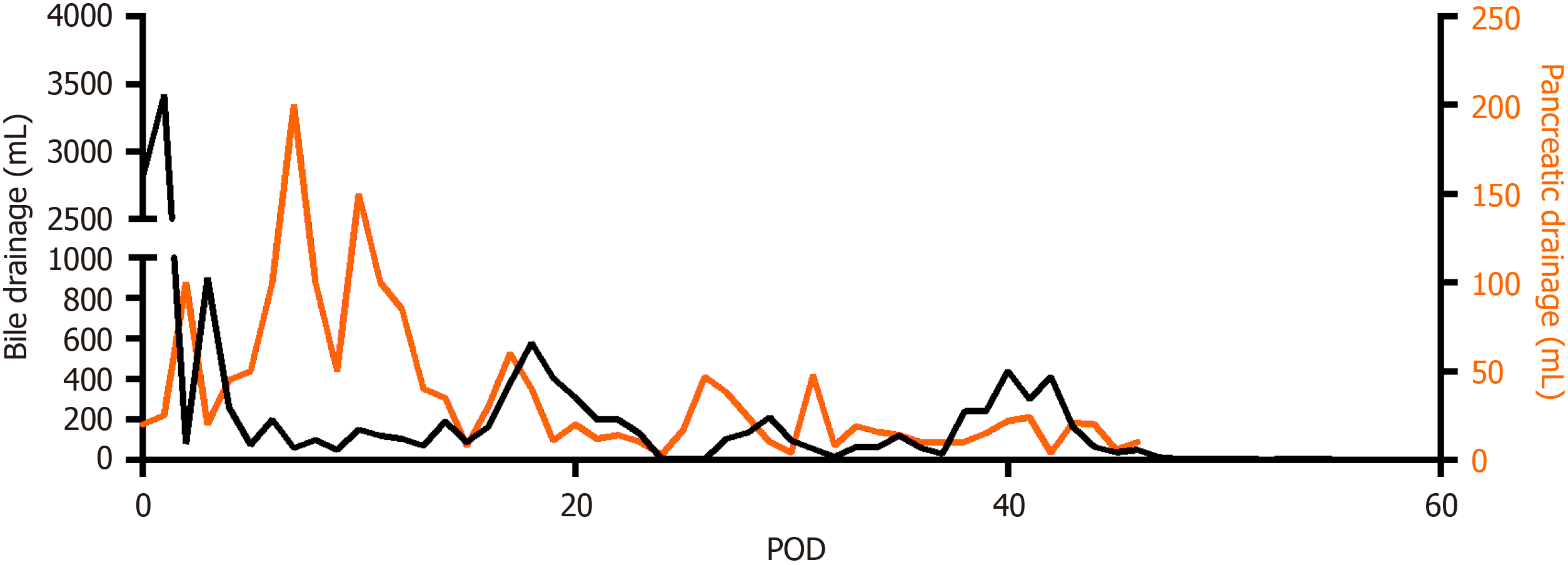
Pancreatic fistulas and bile leakage were also found after stent placement, which was inferred to be a result of the resolution of intestinal edema after re-establishment of PV blood flow. A pancreatic fistula was diagnosed by a daily drainage volume of 100-200 mL nearby the pancreaticojejunal anastomosis (Figure 5), and the drainage fluid was cloudy as rice water with rising amylase. Bile leakage was diagnosed using Atrovirens drainage fluid with a high level of direct bilirubin. Proton pump inhibitors and enzyme inhibitors were administered, and the drainage tubes were carefully ad
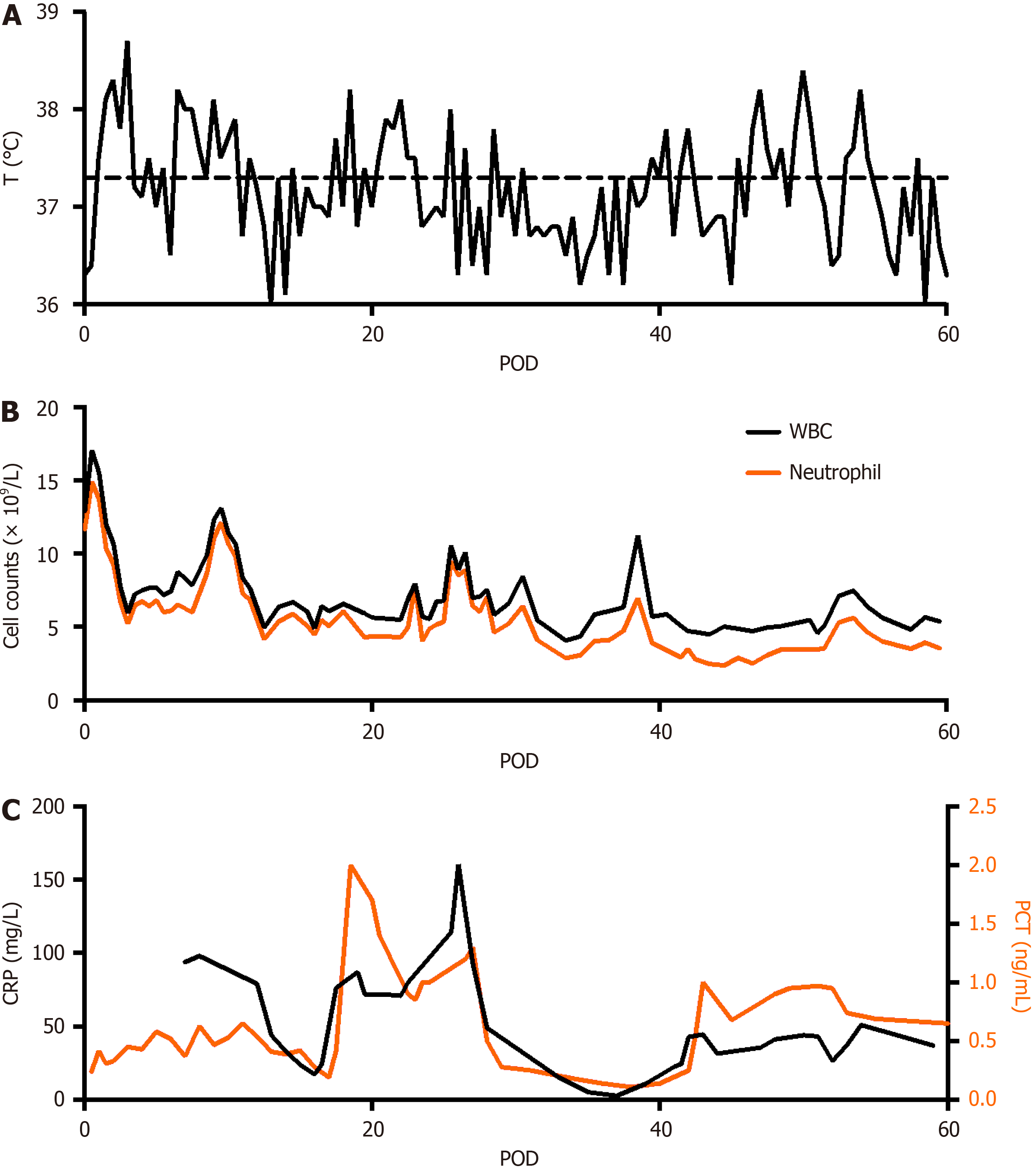
The patient experienced several episodes of infection and fever (Figure 6). During the first week after surgery, septic shock occurred, which probably originated from pancreatic fistula, bile leakage, damage to intestinal barrier function, and translocation of gut bacteria. Retrograde infection from the drainage tubes also aggravated the infection. Intermittent fever was found in the whole POM 1, with peak temperature 37.8-38.5 °C. Blood and sputum cultures were negative and drainage cultures were positive for Pseudomonas aeruginosa, Enterococcus faecium, and Acinetobacter baumannii. Antibiotic use was adjusted several times according to drug susceptibility test results. The patient was finally discharged from the hospital 2 months after surgery. The patient did not present any abdominal symptoms related to PV/SMV stenosis after surgery for 8 months.
Pancreatectomy combined with PV resection and reconstruction was used to treat advanced pancreatic cancers that invaded the portal and SMVs[13,14]. However, there were no standardized guidelines on the choice of reconstruction mode, including primary repair, autologous grafting, or synthetic grafting[3,15]. Primary repair is technically easy but might result in high tension for long segmental resections. Autologous grafting requires a suitable length and caliber, and frequently used materials include the great saphenous vein, femoral vein, external iliac vein, umbilical vein patch, pa
Acute PV stenosis following PD was a rare but severe complication. There were few reports on its management, and the reported treatments included early systematic anticoagulation, surgical repair, and percutaneous transhepatic PV stent placement[20-23]. Early systematic anticoagulation was the first-line treatment for PV thrombosis; however, the failure rate could be as high as 62%[22,23], especially in patients with complications such as ascites. Surgery is typically not performed because severe adhesions at the surgical site would make surgical repair difficult[22,23]. Percutaneous transhepatic PV stent placement is preferred for PV stenosis after hepatic transplantation surgery[24,25]; however, its use after pancreatic surgery was rare, especially within 3 PODs. For this patient, we chose to place a stent to re-establish blood flow instead of performing anticoagulation because the occlusion blocked anticoagulation drugs. The reported patency rate was 66.7%-80%[7,8,11,14] for PV stenting after pancreatic surgery. However, these patency rates could not be directly adopted in our case, since the majority of reported cases concerned surgeries conducted > 30 d. Moreover, in those studies, only a small proportion of patients received PV reconstruction during surgery. Therefore, our case was more vulnerable to thrombosis formation. However, due to our successful management, the patient did not present any abdominal symptoms related to PV/SMV stenosis after surgery for 10 months, and CT scan revealed patent stents.
In this patient, two stents were placed, one coated and one uncoated. The uncoated stent was intended for the preservation of blood supply of SMV branches, protecting the blood supply for both key anastomoses. After the stent place
This case showed a successful example of using percutaneous transhepatic stent placement for treating acute SMV stenosis after Whipple surgery. The effectiveness of stenting has been validated by PV angiography. Although severe complications occurred after this procedure, such as bleeding, infection, pancreatic fistula and bile leakage, our treatment was successful in relieving the patient’s symptoms. This is the first report on stent placement, management of anticoagulation usage, and complications after stenting for acute PV/SMV stenosis after pancreatic surgery with PV reconstruction within postoperative 3 d.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Batta A, India S-Editor: Wang JJ L-Editor: A P-Editor: Xu ZH
| 1. | Kyaw L, Lai NM, Iyer SG, Loh DSKL, Loh SEK, Mali VP. Percutaneous transhepatic interventional therapy of portal vein stenosis in paediatric liver transplantation: A systematic review of efficacy and safety. Pediatr Transplant. 2022;26:e14187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 2. | Sharshar M, Yagi S, Iida T, Yao S, Miyachi Y, Macshut M, Iwamura S, Hirata M, Ito T, Hata K, Taura K, Okajima H, Kaido T, Uemoto S. Liver transplantation in patients with portal vein thrombosis: A strategic road map throughout management. Surgery. 2020;168:1160-1168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Dua MM, Tran TB, Klausner J, Hwa KJ, Poultsides GA, Norton JA, Visser BC. Pancreatectomy with vein reconstruction: technique matters. HPB (Oxford). 2015;17:824-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 4. | Kang MJ, Jang JY, Chang YR, Jung W, Kim SW. Portal vein patency after pancreatoduodenectomy for periampullary cancer. Br J Surg. 2015;102:77-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 5. | Fujii T, Nakao A, Yamada S, Suenaga M, Hattori M, Takami H, Inokawa Y, Kanda M, Sugimoto H, Nomoto S, Murotani K, Kodera Y. Vein resections >3 cm during pancreatectomy are associated with poor 1-year patency rates. Surgery. 2015;157:708-715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 6. | Snyder RA, Prakash LR, Nogueras-Gonzalez GM, Kim MP, Aloia TA, Vauthey JN, Lee JE, Fleming JB, Katz MHG, Tzeng CD. Vein resection during pancreaticoduodenectomy for pancreatic adenocarcinoma: Patency rates and outcomes associated with thrombosis. J Surg Oncol. 2018;117:1648-1654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Kim KR, Ko GY, Sung KB, Yoon HK. Percutaneous transhepatic stent placement in the management of portal venous stenosis after curative surgery for pancreatic and biliary neoplasms. AJR Am J Roentgenol. 2011;196:W446-W450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 8. | Mugu VK, Thompson SM, Fleming CJ, Yohanathan L, Truty MJ, Kendrick ML, Andrews JC. Evaluation of Technical Success, Efficacy, and Safety of Portomesenteric Venous Intervention following Nontransplant Hepatobiliary or Pancreatic Surgery. J Vasc Interv Radiol. 2020;31:416-424.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 9. | Beyer LP, Wohlgemuth WA, Uller W, Pregler B, Goessmann H, Niessen C, Haimerl M, Stroszczynski C, Müller-Wille R. Percutaneous treatment of symptomatic superior mesenteric vein stenosis using self-expanding nitinol stents. Eur J Radiol. 2015;84:1964-1969. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 10. | Ferral H, Alonzo MJ, Datri J, Hogg ME, Marsh R, Talamonti MS. Endovascular management of portal vein obstruction in hepatobiliary cancer patients. J Surg Oncol. 2022;125:392-398. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 11. | Cao G, Ko GY, Sung KB, Yoon HK, Gwon DI, Kim JH. Treatment of postoperative main portal vein and superior mesenteric vein thrombosis with balloon angioplasty and/or stent placement. Acta Radiol. 2013;54:526-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 12. | Ouaïssi M, Sielezneff I, Pirro N, Bon Mardion R, Chaix JB, Merad A, Berdah S, Moutardier V, Cresti S, Emungania O, Anderson L, Christian B, Bernard S. Therapeutic anticoagulant does not modify thromboses rate vein after venous reconstruction following pancreaticoduodenectomy. Gastroenterol Res Pract. 2008;2008:896320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 13. | Ohgi K, Sugiura T, Yamamoto Y, Okamura Y, Ito T, Ashida R, Aramaki T, Uesaka K. Benign Portal Vein Stenosis After Pancreaticoduodenectomy. World J Surg. 2019;43:2623-2630. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 23] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 14. | Khan A, Kleive D, Aandahl EM, Fosby B, Line PD, Dorenberg E, Guvåg S, Labori KJ. Portal vein stent placement after hepatobiliary and pancreatic surgery. Langenbecks Arch Surg. 2020;405:657-664. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (1)] |
| 15. | Gao W, Dai X, Dai C, Jiang K, Wu J, Li Q, Guo F, Chen J, Wei J, Lu Z, Tu M, Miao Y. Comparison of patency rates and clinical impact of different reconstruction methods following portal/superior mesenteric vein resection during pancreatectomy. Pancreatology. 2016;16:1113-1123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 16. | Shao Y, Yan S, Zhang QY, Shen Y, Zhang M, Wang WL, Zheng SS. Autologous falciform ligament graft as A substitute for mesentericoportal vein reconstruction in pancreaticoduodenectomy. Int J Surg. 2018;53:159-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 17. | Shao Y, Feng J, Jiang Y, Hu Z, Wu J, Zhang M, Shen Y, Zheng S. Feasibility of mesentericoportal vein reconstruction by autologous falciform ligament during pancreaticoduodenectomy-cohort study. BMC Surg. 2021;21:4. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 18. | De Pauw V, Pezzullo M, Bali MA, El Moussaoui I, Racu ML, D'haene N, Bouchart C, Closset J, Van Laethem JL, Navez J. Peritoneal patch in vascular reconstruction during pancreaticoduodenectomy for pancreatic cancer: a single Centre experience. Acta Chir Belg. 2023;123:257-265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 19. | Dokmak S, Aussilhou B, Sauvanet A, Nagarajan G, Farges O, Belghiti J. Parietal Peritoneum as an Autologous Substitute for Venous Reconstruction in Hepatopancreatobiliary Surgery. Ann Surg. 2015;262:366-371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 78] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 20. | Woodrum DA, Bjarnason H, Andrews JC. Portal vein venoplasty and stent placement in the nontransplant population. J Vasc Interv Radiol. 2009;20:593-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 42] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 21. | You Y, Heo JS, Han IW, Shin SH, Shin SW, Park KB, Cho SK, Hyun D. Long term clinical outcomes of portal vein stenting for symptomatic portal vein stenosis after pancreaticoduodenectomy. Medicine (Baltimore). 2021;100:e27264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 22. | Cho CW, Park YJ, Kim YW, Choi SH, Heo JS, Choi DW, Kim DI. Follow-up results of acute portal and splenic vein thrombosis with or without anticoagulation therapy after hepatobiliary and pancreatic surgery. Ann Surg Treat Res. 2015;88:208-214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 23. | Belnap L, Zaveri H, Cottam D, Surve A, Rodgers GM, Drury C. What is the optimal treatment of superior mesenteric vein/portal vein thrombosis after bariatric surgery? Surg Obes Relat Dis. 2017;13:533-537. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 24. | Zhou ZQ, Lee JH, Song KB, Hwang JW, Kim SC, Lee YJ, Park KM. Clinical usefulness of portal venous stent in hepatobiliary pancreatic cancers. ANZ J Surg. 2014;84:346-352. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Hiyoshi M, Fujii Y, Kondo K, Imamura N, Nagano M, Ohuchida J. Stent Placement for Portal Vein Stenosis After Pancreaticoduodenectomy. World J Surg. 2015;39:2315-2322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |