Published online Sep 27, 2023. doi: 10.4240/wjgs.v15.i9.2052
Peer-review started: June 14, 2023
First decision: July 3, 2023
Revised: July 11, 2023
Accepted: July 26, 2023
Article in press: July 26, 2023
Published online: September 27, 2023
Processing time: 100 Days and 4.8 Hours
Ultrasound is a vital tool for the diagnosis and management of colorectal cancer (CRC). Contrast-enhanced ultrasound (CEUS) is a non-invasive, safe, and cost-effective method for evaluating tumour blood vessels, that play a crucial role in tumour growth and progression.
To explore CEUS’s role in the quantitative evaluation of CRC blood vessels and their correlation with angiogenesis markers and prognosis.
This study prospectively enrolled 100 patients with CRC confirmed by histo
Quantitative CEUS parameters (PI, TTP, and AUC) showed significant correlations with VEGF expression (P < 0.001) and MVD (P < 0.001), indicating a strong link between tumour blood vessels and angiogenesis. Increased PI, reduced TTP, and expanded AUC values were significantly related to higher tumour stage (P < 0.001), lymph node metastasis (P < 0.001), and distant metastasis (P < 0.001). Furthermore, these parameters were recognized as independent predictors of overall survival and disease-free survival in multivariate analysis (P < 0.001).
CEUS has a high potential in guiding treatment planning and predicting patient outcomes. However, more com
Core Tip: This study investigates the role of contrast-enhanced ultrasound (CEUS) in evaluating colorectal cancer (CRC) blood vessels and their correlation with angiogenesis markers and prognosis. The study enrolled 100 patients with CRC, and quantitative CEUS parameters showed significant correlations with angiogenesis markers and clinicopathological features. The study concludes that CEUS has high potential in guiding treatment planning and predicting patient outcomes but requires further validation through multicentre studies.
- Citation: Li MH, Li WW, He L, Li JF, Zhang SY. Quantitative evaluation of colorectal tumour vasculature using contrast-enhanced ultrasound: Correlation with angiogenesis and prognostic significance. World J Gastrointest Surg 2023; 15(9): 2052-2062
- URL: https://www.wjgnet.com/1948-9366/full/v15/i9/2052.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i9.2052
Colorectal cancer (CRC) is the third most prevalent cancer and the second leading cause of cancer-related fatalities globally, with approximately 1.8 million new cases and nearly 900000 deaths in 2021[1]. The high mortality rate is attributed to late diagnosis, insufficient staging, and ineffective treatment approaches. Therefore, early detection and precise staging are crucial for optimizing treatment plans and improving patient outcomes[2]. In recent decades, advancements in medical imaging techniques have significantly improved diagnostic precision in CRC[3]. Among these imaging methods, contrast-enhanced ultrasound (CEUS) has gained increased interest for its potential in evaluating tumour blood flow and vascularization.
Tumour vasculature plays a critical role in its growth, progression, and metastasis. Rapidly expanding tumours require a substantial blood supply to ensure sufficient delivery of nutrients and oxygen, while simultaneously eliminating waste products[4]. This demand for blood supply is met through angiogenesis, a process of formation of new blood vessels from pre-existing ones. Angiogenesis is regulated by a fine equilibrium between pro-angiogenic and anti-angiogenic factors, with vascular endothelial growth factor (VEGF) being the strongest promoter of angiogenesis[5]. Tumour blood vessel density, also known as microvessel density (MVD), serves as a surrogate marker for angiogenesis and has been associated with tumour aggressiveness and unfavourable prognosis in various cancers, including CRC[6].
CEUS is a relatively novel imaging technique that employs intravenously administered microbubble contrast agents to improve ultrasound images[7]. The echogenicity of microbubbles enhances visualization of blood flow within the vasculature. By utilizing the non-linear behaviour of microbubbles, CEUS can produce real-time, high-resolution images of tissue perfusion and vascular structure with a high signal-to-noise ratio[8,9]. Compared with other imaging techniques, such as ultrasound, computed tomography (CT), magnetic resonance imaging (MRI), etc., CEUS has several advantages for CRC imaging. First, CEUS can provide high-resolution images of tumour vasculature without being affected by bone or gas interference[10]. Second, CEUS can offer real-time dynamic information on tissue perfusion and blood flow velocity with a high temporal resolution[11]. Third, CEUS can provide quantitative parameters for tumour vascularization analysis using time-intensity curve (TIC) analysis[12]. Fourth, CEUS is a non-invasive, safe, and cost-effective method that does not expose patients to ionizing radiation or nephrotoxic contrast agents[13].
Several studies have examined the potential of CEUS in assessing tumour vasculature in CRC[14-17]. Most of these studies have only focused on qualitative or semi-quantitative analyses, such as visual grading of enhancement patterns or evaluating TIC parameters without thoroughly investigating their correlation with angiogenesis markers and prognostic factors. Additionally, a majority of these studies have been limited by small sample sizes and a lack of standardized CEUS examination and analysis techniques.
Considering these limitations, the present study aimed to explore the role of CEUS in quantitatively evaluating colorectal tumour vasculature and its correlation with angiogenesis markers (VEGF and MVD) and prognostic factors. We hypothesized that quantitative CEUS parameters would significantly correlate with angiogenesis markers, and elevated CEUS-derived parameter values would be associated with aggressive tumour features and poor prognosis. To investigate these hypotheses, we conducted a prospective study with a relatively large cohort of patients with histologically confirmed CRC who underwent preoperative CEUS examinations using standardized techniques. Moreover, we used a comprehensive approach to data analysis, including assessment of TIC-derived parameters and their correlation with angiogenesis markers, clinicopathological characteristics, and survival outcomes.
This study aimed to provide valuable insights into the potential of CEUS as a non-invasive tool for evaluating tumour vasculature in CRC and establish its clinical utility in guiding treatment decisions and predicting patient outcomes. The study’s findings may contribute to the growing evidence supporting the use of CEUS in CRC management and pave the way for future large-scale, multicentre trials to validate and expand upon these findings.
This study prospectively enrolled 100 patients with histologically confirmed CRC from January 2020 to December 2022. Inclusion criteria were as follows: (1) Age ≥ 18 years; (2) Pathologically confirmed primary colorectal adenocarcinoma; (3) No history of chemotherapy or radiotherapy; and (4) Ability to provide informed consent. Exclusion criteria were as follows: (1) Contraindications to ultrasound contrast agents, such as severe allergic reactions; (2) Pregnancy or lactation; and (3) Severe comorbidities affecting survival outcomes, such as end-stage renal disease, chronic heart failure, or uncontrolled diabetes. This study was approved by the Xinjiang Medical University Affiliated Cancer Hospital institutional review board and adhered to the standards of the Declaration of Helsinki.
All patients underwent CEUS examinations within two weeks before surgery using a high-end ultrasound system (LOGIQ E9, GE Healthcare, Milwaukee, WI, United States) with a 1-6 MHz convex array transducer (C1-6, GE Heal
A 2.4 mL dose of SonoVue (Bracco, Milan, Italy), a microbubble contrast agent containing sulphur hexafluoride gas encapsulated in a phospholipid shell, was administered intravenously as a single bolus injection, followed by a 10 mL saline flush. CEUS examination began immediately after injection and continued for 5 min. Mechanical index was set at a low level (≤ 0.1) to minimize microbubble destruction. Imaging parameters, including gain, time gain compensation, and focal zone, were adjusted to optimize image quality and maintain consistent contrast-enhanced image appearance. Experienced sonographers, blinded to patients’ clinical information, performed all CEUS examinations.
CEUS images were stored in DICOM format and analysed offline using VueBox software (Bracco, Milan, Italy). Regions of interests (ROIs) were manually drawn to encompass the entire enhancing tumour area on CEUS images. The software generated TICs for the selected ROIs, displaying the change in intensity over time. Three quantitative CEUS parameters were derived from the TIC analysis: Peak intensity (PI), time to peak (TTP), and area under the curve (AUC). PI repre
During surgery, cancerous tissue samples were collected, preserved in 40 g/L neutral buffered formaldehyde, and embedded in paraffin before being sliced into 4 μm-thick sections. These sections were deparaffinised, rehydrated, and subjected to antigen retrieval using a citrate buffer (pH = 6.0) and microwave heat treatment. Subsequently, they were treated with 3% hydrogen peroxide for 10 min to neutralize endogenous peroxidase activity and incubated overnight at 4 °C with primary antibodies targeting VEGF (1:200, rabbit polyclonal, Abcam, Cambridge, United Kingdom) and CD34 (1:100, mouse monoclonal, Dako, Glostrup, Denmark). After rinsing with phosphate-buffered saline, the sections were treated with secondary antibodies conjugated to horseradish peroxidase for 1 h at room temperature and counterstained with haematoxylin. Immunoreactivity was detected using a 3,3’-diaminobenzidine substrate. The percentage of VEGF-positive tumor cells and CD34-positive microvessels were counted using ImageJ software (National Institutes of Health, Bethesda, MD, United States). Five random fields of view per section were selected and captured at 400 × magnification. The number of positive cells or microvessels and the total number of cells or microvessels were counted manually. The percentage was calculated as the ratio of positive cells or microvessels to total cells or microvessels multiplied by 100.
VEGF expression was evaluated with a semi-quantitative measurement method, which accounted for both the percentage of tumour cells expressing VEGF and the intensity of staining. The percentage of VEGF-positive tumour cells were scored as follows: 0 (0%), 1 (1%-25%), 2 (26%-50%), 3 (51%-75%), and 4 (76%-100%). The intensity of staining was rated using the following categories: 0 (absent), 1 (faint), 2 (moderate), and 3 (vivid). The percentage and intensity scores were multiplied to calculate the final VEGF expression score, ranging from 0-12. High VEGF expression was defined as a score of 6 or higher.
MVD was assessed by enumerating CD34-positive microvessels in the highly vascularized areas of the tumour, termed as ‘hotspots’. These hotspots were located using a low-magnification (100 ×) lens, followed by counting of the microvessels under high magnification (400 ×). Each endothelial cell or cluster, clearly distinguishable from adjacent microvessels, tumour cells, and connective tissues, qualified as a countable microvessel, regardless of lumen presence. MVD was expressed as the mean quantity of microvessels within each high-power field.
The patients’ clinicopathological characteristics, CEUS parameters, and immunohistochemical findings are summarized using descriptive statistics. The relationships between CEUS parameters, angiogenesis markers, and clinicopathological factors were examined using either the Spearman’s rank correlation coefficient or Pearson’s correlation coefficient, depending on the situation. The Mann-Whitney U test or Kruskal-Wallis test was employed, as necessary, to evaluate differences in CEUS parameters concerning VEGF expression and MVD.
The Kaplan-Meier method was used to analyse survival, and the log-rank test was used to evaluate the differences in overall survival (OS) and disease-free survival (DFS) based on CEUS parameters, VEGF expression, and MVD. To identify independent prognostic factors for OS and DFS, a multivariate analysis was conducted using the Cox proportional hazards model. All statistical analyses were performed using the SPSS software (version 26.0, IBM Corp., NY, United States), and a two-sided P value < 0.05 was considered to be statistically significant. The statistical method of this study was reviewed by Li Y from the Baoding Maternal and Child Health Hospital.
The study enrolled 100 patients, including 57 men and 43 women, with a median age of 63 years (range: 38-84 years). Patients’ clinicopathological traits are shown in Table 1. A majority of the tumours were located in the rectum (n = 52), while others were found in the sigmoid colon (n = 26), ascending colon (n = 12), and descending colon (n = 10). Based on the TNM staging system, 21 patients had stage I tumours, 29 had stage II tumours, 35 had stage III tumours, and 15 had stage IV tumours. Lymph node metastasis was observed in 50 patients, whereas 15 patients displayed distant metastasis.
| Characteristic | Number of patients | Percentage |
| Total patients | 100 | 100% |
| Men | 57 | 57% |
| Women | 43 | 43% |
| Tumour location | ||
| Rectum | 52 | 52% |
| Sigmoid colon | 26 | 26% |
| Ascending colon | 12 | 12% |
| Descending colon | 10 | 10% |
| Tumour stage | ||
| Stage I | 21 | 21% |
| Stage II | 29 | 29% |
| Stage III | 35 | 35% |
| Stage IV | 15 | 15% |
| Metastasis | ||
| Lymph node metastasis | 50 | 50% |
| Distant metastasis | 15 | 15% |
The average values of PI, TTP and AUC were 23.6 ± 7.8 dB, 16.5 ± 5.6 s and 1032.6 ± 361.3 dB × s, respectively. High VEGF expression was detected in 56 patients, and the median MVD was 52.5 (range: 12-125). Table 2 shows the correlations between CEUS parameters and angiogenesis markers. Significant positive correlations were observed between PI and both VEGF expression (r = 0.73, P < 0.001) and MVD (r = 0.75, P < 0.001). TTP demonstrated significant negative correlations with both VEGF expression (r = -0.68, P < 0.001) and MVD (r = -0.72, P < 0.001). Additionally, AUC demonstrated significant positive correlations with both VEGF expression (r = 0.71, P < 0.001) and MVD (r = 0.74, P < 0.001).
| Parameters | Pearson correlation coefficient (r) | P value |
| PI and VEGF | 0.73 | < 0.001 |
| PI and MVD | 0.75 | < 0.001 |
| TTP and VEGF | -0.68 | < 0.001 |
| TTP and MVD | -0.72 | < 0.001 |
| AUC and VEGF | 0.71 | < 0.001 |
| AUC and MVD | 0.74 | < 0.001 |
Table 3 shows the correlations between CEUS parameters, angiogenesis indicators, and clinicopathological characteristics. As expected, high VEGF expression and MVD were related to advanced tumour stage (P < 0.001), lymph node metastasis (P < 0.001), and distant metastasis (P < 0.001). Additionally, high PI, short TTP, and large AUC values were associated with advanced tumour stage (P < 0.001), lymph node metastasis (P < 0.001), and distant metastasis (P < 0.001). No sig
| Tumour stage | Lymph node metastasis | Distant metastasis | Age (yr) | Sex | |
| PI | P < 0.001 | P < 0.001 | P < 0.001 | NS | NS |
| TTP | P < 0.001 | P < 0.001 | P < 0.001 | NS | NS |
| AUC | P < 0.001 | P < 0.001 | P < 0.001 | NS | NS |
| VEGF expression | P < 0.001 | P < 0.001 | P < 0.001 | NS | NS |
| MVD | P < 0.001 | P < 0.001 | P < 0.001 | NS | NS |
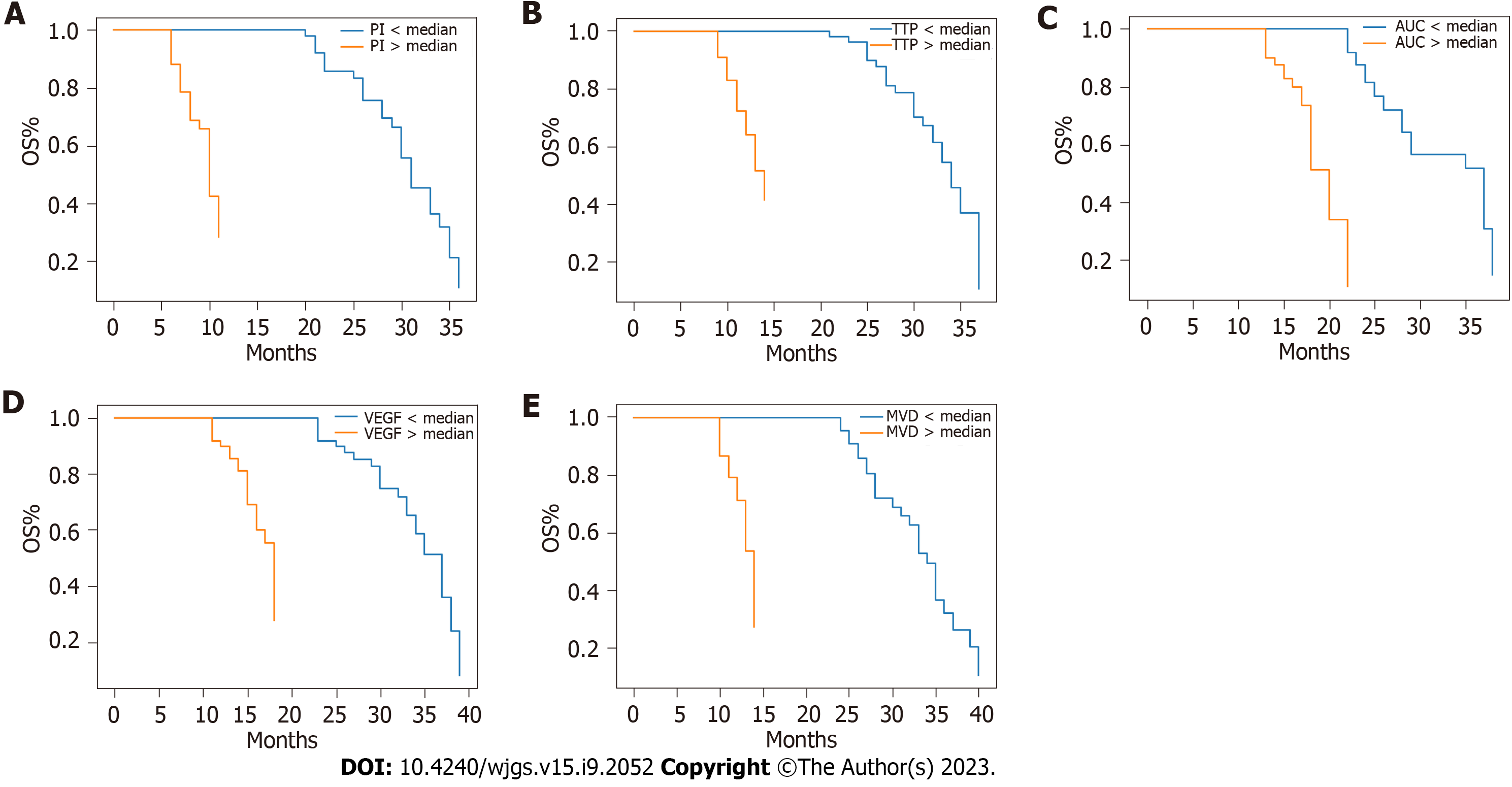
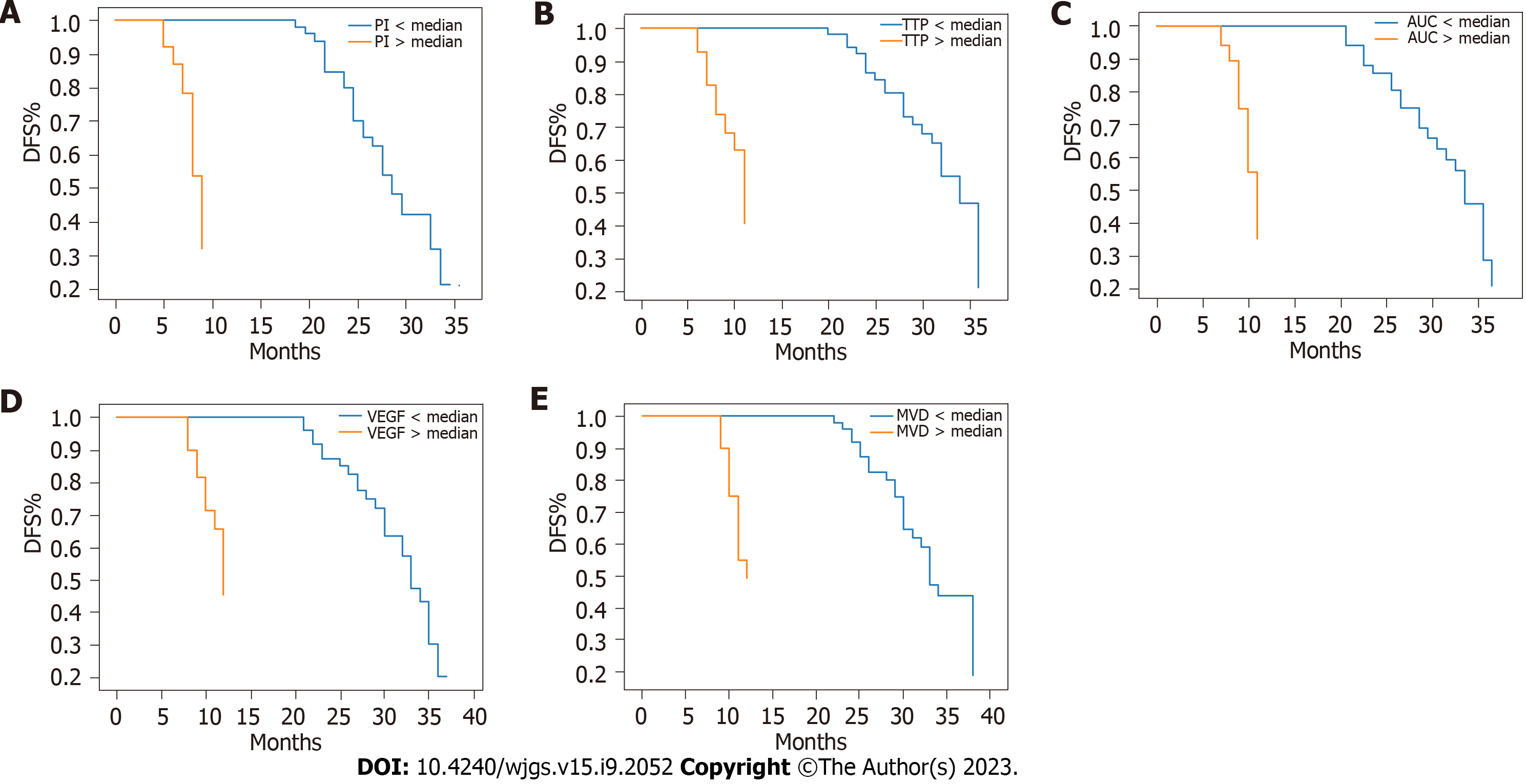
The average follow-up period was 26 mo (range: 3-36 mo). During this period, 28 patients experienced tumour recurrence and 22 patients died. The OS and DFS rates at 3 years were 78% and 72%, respectively. Figures 1 and 2 show the Kaplan-Meier survival curves for OS and DFS based on CEUS parameters, VEGF expression, and MVD, respectively. Poor OS (P < 0.001) and DFS (P < 0.001) were significantly associated with high PI, short TTP, and large AUC values. Additionally, poor OS (P < 0.001) and DFS (P < 0.001) were significantly associated with high VEGF expression and MVD. The multivariate analysis revealed that OS was independently influenced by prognostic factors, including PI [hazard ratio (HR) = 2.55, 95% confidence interval (CI): 1.36-4.78, P = 0.003], TTP (HR = 2.34, 95%CI: 1.24-4.41, P = 0.008), AUC (HR = 2.62, 95%CI: 1.38-4.96, P = 0.003), VEGF expression (HR = 2.47, 95%CI: 1.31-4.65, P = 0.005), and MVD (HR = 2.81, 95%CI: 1.49-5.30, P = 0.001). Similarly, DFS was also independently affected by prognostic factors, including PI (HR = 2.38, 95%CI: 1.28-4.42, P = 0.006), TTP (HR = 2.26, 95%CI: 1.20-4.26, P = 0.011), AUC (HR = 2.54, 95%CI: 1.34-4.81, P = 0.004), VEGF expression (HR = 2.31, 95%CI: 1.24-4.32, P = 0.008), and MVD (HR = 2.67, 95%CI: 1.42-5.03, P = 0.002). Table 4 summarizes the results of the multivariate analysis.
| Prognostic factor | Hazard ratio (OS) | 95%CI (OS) | P value (OS) | Hazard ratio (DFS) | 95%CI (DFS) | P value (DFS) |
| VEGF expression | 2.47 | 1.31-4.65 | 0.005 | 2.31 | 1.24-4.32 | 0.008 |
| MVD | 2.81 | 1.49-5.30 | 0.001 | 2.67 | 1.42-5.03 | 0.002 |
| PI | 2.55 | 1.36-4.78 | 0.003 | 2.38 | 1.28-4.42 | 0.006 |
| TTP | 2.34 | 1.24-4.41 | 0.008 | 2.26 | 1.20-4.26 | 0.011 |
| AUC | 2.62 | 1.38-4.96 | 0.003 | 2.54 | 1.34-4. 81 | 0.004 |
Our study demonstrated significant correlations between quantitative CEUS parameters (PI, TTP, and AUC) and angiogenesis markers (VEGF expression and MVD) in CRC. High PI, short TTP, and large AUC values were significantly associated with aggressive tumour features and unfavourable prognosis, independent of other clinicopathological factors. These results suggest that CEUS may be a useful non-invasive imaging modality for assessing tumour vasculature in CRC and could potentially help in guiding treatment planning and predicting patient outcomes.
The significant correlations between CEUS parameters and angiogenesis markers observed in our study align with the findings of previous literature[18-20]. The positive correlations between PI and VEGF expression and MVD can be explained by the generally greater density of blood vessels in tumours with high VEGF expression and MVD, which leads to increased blood flow and higher PI values on CEUS. Similarly, the negative correlations between TTP and VEGF expression and MVD can be attributed to the faster blood flow in tumours with high VEGF expression and MVD, resulting in a shorter time required to reach PI on CEUS.
The associations between CEUS parameters and clinicopathological characteristics observed in our study are also consistent with the findings of previous research. High PI, short TTP, and large AUC values have been found to correlate with advanced stage, lymph node metastasis, and distant metastasis in various cancers, including CRC[18,21,22]. These associations can be explained by the fact that aggressive tumours typically exhibit higher angiogenesis, leading to increased blood flow and more pronounced contrast enhancement on CEUS. Our survival analysis revealed that high PI, short TTP, and large AUC values were significantly associated with poor OS and DFS, independent of other clinicopathological factors. These results suggest that CEUS parameters could serve as potential prognostic biomarkers in CRC. Furthermore, the significant associations between high VEGF expression, MVD, and poor survival outcomes observed in our study are consistent with those in previous studies, further emphasizing the importance of tumour vasculature in CRC progression and prognosis[23,24].
CEUS provides multiple benefits compared with other imaging techniques, such as CT and MRI, in evaluating tumour vasculature. It is a real-time, non-invasive, and radiation-free imaging modality that provides high spatial and temporal resolution, allowing for a detailed assessment of tumour blood flow and microvasculature[25,26]. Moreover, microbubble contrast agents used in CEUS are purely intravascular, which enables a more accurate evaluation of blood vessel density and perfusion characteristics compared to the contrast agents used in CT and MRI, which have a tendency to extravasate into the interstitial space[27]. Furthermore, CEUS is generally less expensive than CT and MRI, making it a more cost-effective option for patients and healthcare systems[28].
In addition to assessing tumour vasculature, CEUS has also been investigated for other clinical applications in CRC, including detecting and characterizing primary tumours, lymph node staging, and assessing treatment response[29,30]. It has shown to improve the accuracy of primary tumour detection in CRC compared to that of conventional ultrasound, particularly in early-stage tumours, where the sensitivity and specificity of CEUS are reported to be high[31]. Further
CEUS has demonstrated potential utility in identifying metastatic lymph nodes in CRC patients[33]. Several studies have reported that evaluation of lymph node vascularization using CEUS can help differentiate metastatic from non-metastatic lymph nodes, with higher accuracy than conventional ultrasound or CT[34-36]. However, further research is warranted to establish standardized criteria for assessing lymph node involvement using CEUS and to compare its performance with that of other imaging modalities, such as MRI or positron emission tomography.
Monitoring treatment response is another potential clinical application for CEUS in CRC. In recent years, neoadjuvant chemoradiotherapy has become a standard treatment approach for locally advanced rectal cancer, with the goal of downstaging the tumour and improving the likelihood of complete surgical resection[37]. Monitoring the response to neoadjuvant therapy is crucial for determining the optimal timing of surgery and predicting patient outcomes. CEUS has been investigated as a non-invasive method for monitoring changes in tumour vasculature during neoadjuvant treat
Although our study yielded encouraging results, it had certain limitations. First, a relatively small sample size could have affected the statistical power of our analysis. Future research with larger sample size is essential to validate and broaden the implications of our findings. Second, the retrospective design of our study may have led to selection bias; therefore, prospective studies are required to confirm the prognostic significance of CEUS parameters in CRC. Third, our study primarily focused on the correlations between CEUS parameters and angiogenesis markers; however, the underlying biological mechanisms remain to be elucidated. Therefore, further investigations, such as lab-based and animal studies, are required to explore the molecular pathways linking CEUS parameters to angiogenesis and tumour progression in CRC.
Our study demonstrated that quantitative CEUS parameters were significantly associated with angiogenesis markers and prognostic factors in CRC. These findings suggest that CEUS could be a valuable non-invasive tool for assessing tumour vasculature in CRC and may have potential clinical utility in guiding treatment decisions and predicting patient outcomes. Future studies should focus on validating and expanding upon our findings in large cohorts and exploring the underlying biological mechanisms linking CEUS parameters to angiogenesis and tumour progression in CRC. Moreover, the potential applications of CEUS in other aspects of CRC management, such as primary tumour detection, lymph node staging, and treatment response assessment, should be further investigated to fully understand the clinical potential of this imaging modality.
The background of this research study highlights the importance of ultrasound in the diagnosis and management of colorectal cancer (CRC). Contrast-enhanced ultrasound (CEUS) is a non-invasive, safe, and cost-effective method that allows for the evaluation of tumor blood vessels. Tumor angiogenesis, which involves the formation of new blood vessels, plays a critical role in tumor growth and progression.
The aim of this study was to investigate the role of CEUS in quantitatively assessing CRC blood vessels and their cor
Explore the role of CEUS in quantitative assessment of blood vessels in CRC and its correlation with angiogenic markers and prognosis. This study aims to prospectively recruit 100 CRC patients confirmed by histopathology and conduct preoperative CEUS examination.
This study enrolled 100 patients with confirmed CRC through histopathology. Preoperative CEUS examinations were performed on all patients. From the CEUS images, quantitative parameters including peak intensity (PI), time to peak (TTP), and area under the curve (AUC) were derived using time-intensity curve analysis. During surgery, tumor tissue samples were obtained and examined immunohistochemically for the expression of angiogenesis markers, such as vascular endothelial growth factor (VEGF) and microvessel density (MVD). The researchers used appropriate statistical tests to evaluate the correlations between CEUS parameters, angiogenesis markers, and clinicopathological features.
The study findings revealed significant associations between the quantitative CEUS parameters (PI, TTP, and AUC) and VEGF expression, MVD, tumor stage, lymph node metastasis, distant metastasis, overall survival, and disease-free survival. The study concluded that CEUS holds potential for guiding treatment planning and predicting patient outcomes in CRC management but emphasized the need for more comprehensive multicenter studies to validate its clinical utility.
Demonstrated that CEUS has a significant role in the quantitative evaluation of CRC blood vessels. The quantitative CEUS parameters (PI, TTP, and AUC) showed strong correlations with angiogenesis markers, specifically VEGF expre
The study demonstrates the potential of CEUS in guiding treatment decisions and predicting patient outcomes in CRC, further comprehensive studies involving multiple centers are necessary to validate its clinical utility.
Thank you to all those who have contributed to this manuscript.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Garcia K, Spain; Johnson CH, United States S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
| 1. | Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J Clin. 2021;71:209-249. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75126] [Cited by in RCA: 64637] [Article Influence: 16159.3] [Reference Citation Analysis (176)] |
| 2. | Benson AB, Venook AP, Al-Hawary MM, Cederquist L, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Engstrom PF, Grem JL, Grothey A, Hochster HS, Hoffe S, Hunt S, Kamel A, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Mulcahy MF, Murphy JD, Nurkin S, Saltz L, Sharma S, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Wuthrick E, Gregory KM, Gurski L, Freedman-Cass DA. Rectal Cancer, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:874-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 683] [Article Influence: 113.8] [Reference Citation Analysis (0)] |
| 3. | Goiffon RJ, O'Shea A, Harisinghani MG. Advances in radiological staging of colorectal cancer. Clin Radiol. 2021;76:879-888. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 4. | Carmeliet P, Jain RK. Angiogenesis in cancer and other diseases. Nature. 2000;407:249-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6437] [Cited by in RCA: 6489] [Article Influence: 259.6] [Reference Citation Analysis (0)] |
| 5. | Ferrara N, Gerber HP, LeCouter J. The biology of VEGF and its receptors. Nat Med. 2003;9:669-676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6747] [Cited by in RCA: 6952] [Article Influence: 316.0] [Reference Citation Analysis (0)] |
| 6. | Weidner N. Current pathologic methods for measuring intratumoral microvessel density within breast carcinoma and other solid tumors. Breast Cancer Res Treat. 1995;36:169-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 517] [Cited by in RCA: 565] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 7. | Blomley MJ, Cooke JC, Unger EC, Monaghan MJ, Cosgrove DO. Microbubble contrast agents: a new era in ultrasound. BMJ. 2001;322:1222-1225. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 321] [Cited by in RCA: 250] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 8. | Dietrich CF, Averkiou M, Nielsen MB, Barr RG, Burns PN, Calliada F, Cantisani V, Choi B, Chammas MC, Clevert DA, Claudon M, Correas JM, Cui XW, Cosgrove D, D'Onofrio M, Dong Y, Eisenbrey J, Fontanilla T, Gilja OH, Ignee A, Jenssen C, Kono Y, Kudo M, Lassau N, Lyshchik A, Franca Meloni M, Moriyasu F, Nolsøe C, Piscaglia F, Radzina M, Saftoiu A, Sidhu PS, Sporea I, Schreiber-Dietrich D, Sirlin CB, Stanczak M, Weskott HP, Wilson SR, Willmann JK, Kim TK, Jang HJ, Vezeridis A, Westerway S. How to perform Contrast-Enhanced Ultrasound (CEUS). Ultrasound Int Open. 2018;4:E2-E15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 246] [Cited by in RCA: 222] [Article Influence: 31.7] [Reference Citation Analysis (33)] |
| 9. | Dietrich CF, Ignee A, Frey H. Contrast-enhanced endoscopic ultrasound with low mechanical index: a new technique. Z Gastroenterol. 2005;43:1219-1223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 73] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 10. | Claudon M, Dietrich CF, Choi BI, Cosgrove DO, Kudo M, Nolsøe CP, Piscaglia F, Wilson SR, Barr RG, Chammas MC, Chaubal NG, Chen MH, Clevert DA, Correas JM, Ding H, Forsberg F, Fowlkes JB, Gibson RN, Goldberg BB, Lassau N, Leen EL, Mattrey RF, Moriyasu F, Solbiati L, Weskott HP, Xu HX. Guidelines and good clinical practice recommendations for contrast enhanced ultrasound (CEUS) in the liver--update 2012: a WFUMB-EFSUMB initiative in cooperation with representatives of AFSUMB, AIUM, ASUM, FLAUS and ICUS. Ultraschall Med. 2013;34:11-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 224] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 11. | Quaia E. The real capabilities of contrast-enhanced ultrasound in the characterization of solid focal liver lesions. Eur Radiol. 2011;21:457-462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 27] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 12. | Wei K, Le E, Bin JP, Coggins M, Thorpe J, Kaul S. Quantification of renal blood flow with contrast-enhanced ultrasound. J Am Coll Cardiol. 2001;37:1135-1140. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 170] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 13. | Senior R, Becher H, Monaghan M, Agati L, Zamorano J, Vanoverschelde JL, Nihoyannopoulos P. Contrast echocardiography: evidence-based recommendations by European Association of Echocardiography. Eur J Echocardiogr. 2009;10:194-212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 236] [Cited by in RCA: 246] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 14. | Wang S, Yang W, Zhang H, Xu Q, Yan K. The Role of Contrast-Enhanced Ultrasound in Selection Indication and Improveing Diagnosis for Transthoracic Biopsy in Peripheral Pulmonary and Mediastinal Lesions. Biomed Res Int. 2015;2015:231782. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 15. | Cartana ET, Gheonea DI, Cherciu IF, Streaţa I, Uscatu CD, Nicoli ER, Ioana M, Pirici D, Georgescu CV, Alexandru DO, Şurlin V, Gruionu G, Săftoiu A. Assessing tumor angiogenesis in colorectal cancer by quantitative contrast-enhanced endoscopic ultrasound and molecular and immunohistochemical analysis. Endosc Ultrasound. 2018;7:175-183. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 16. | Lassau N, Chebil M, Chami L, Bidault S, Girard E, Roche A. Dynamic contrast-enhanced ultrasonography (DCE-US): a new tool for the early evaluation of antiangiogenic treatment. Target Oncol. 2010;5:53-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 75] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 17. | Lassau N, Bonastre J, Kind M, Vilgrain V, Lacroix J, Cuinet M, Taieb S, Aziza R, Sarran A, Labbe-Devilliers C, Gallix B, Lucidarme O, Ptak Y, Rocher L, Caquot LM, Chagnon S, Marion D, Luciani A, Feutray S, Uzan-Augui J, Coiffier B, Benastou B, Koscielny S. Validation of dynamic contrast-enhanced ultrasound in predicting outcomes of antiangiogenic therapy for solid tumors: the French multicenter support for innovative and expensive techniques study. Invest Radiol. 2014;49:794-800. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 94] [Cited by in RCA: 101] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 18. | Lassau N, Koscielny S, Chami L, Chebil M, Benatsou B, Roche A, Ducreux M, Malka D, Boige V. Advanced hepatocellular carcinoma: early evaluation of response to bevacizumab therapy at dynamic contrast-enhanced US with quantification--preliminary results. Radiology. 2011;258:291-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 150] [Cited by in RCA: 158] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 19. | Wang Y, Li L, Wang YX, Cui NY, Zou SM, Zhou CW, Jiang YX. Time-intensity curve parameters in rectal cancer measured using endorectal ultrasonography with sterile coupling gels filling the rectum: correlations with tumor angiogenesis and clinicopathological features. Biomed Res Int. 2014;2014:587806. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 14] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Lassau N, Lamuraglia M, Chami L, Leclère J, Bonvalot S, Terrier P, Roche A, Le Cesne A. Gastrointestinal stromal tumors treated with imatinib: monitoring response with contrast-enhanced sonography. AJR Am J Roentgenol. 2006;187:1267-1273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 135] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 21. | Albrecht T, Blomley M, Bolondi L, Claudon M, Correas JM, Cosgrove D, Greiner L, Jäger K, Jong ND, Leen E, Lencioni R, Lindsell D, Martegani A, Solbiati L, Thorelius L, Tranquart F, Weskott HP, Whittingham T; EFSUMB Study Group. Guidelines for the use of contrast agents in ultrasound. January 2004. Ultraschall Med. 2004;25:249-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 314] [Cited by in RCA: 252] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 22. | Lamuraglia M, Escudier B, Chami L, Schwartz B, Leclère J, Roche A, Lassau N. To predict progression-free survival and overall survival in metastatic renal cancer treated with sorafenib: pilot study using dynamic contrast-enhanced Doppler ultrasound. Eur J Cancer. 2006;42:2472-2479. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 120] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 23. | Des Guetz G, Uzzan B, Nicolas P, Cucherat M, Morere JF, Benamouzig R, Breau JL, Perret GY. Microvessel density and VEGF expression are prognostic factors in colorectal cancer. Meta-analysis of the literature. Br J Cancer. 2006;94:1823-1832. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 255] [Cited by in RCA: 284] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 24. | Gao J, Knutsen A, Arbman G, Carstensen J, Frånlund B, Sun XF. Clinical and biological significance of angiogenesis and lymphangiogenesis in colorectal cancer. Dig Liver Dis. 2009;41:116-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 25. | Piscaglia F, Nolsøe C, Dietrich CF, Cosgrove DO, Gilja OH, Bachmann Nielsen M, Albrecht T, Barozzi L, Bertolotto M, Catalano O, Claudon M, Clevert DA, Correas JM, D'Onofrio M, Drudi FM, Eyding J, Giovannini M, Hocke M, Ignee A, Jung EM, Klauser AS, Lassau N, Leen E, Mathis G, Saftoiu A, Seidel G, Sidhu PS, ter Haar G, Timmerman D, Weskott HP. The EFSUMB Guidelines and Recommendations on the Clinical Practice of Contrast Enhanced Ultrasound (CEUS): update 2011 on non-hepatic applications. Ultraschall Med. 2012;33:33-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 721] [Cited by in RCA: 680] [Article Influence: 52.3] [Reference Citation Analysis (0)] |
| 26. | Correas JM, Bridal L, Lesavre A, Méjean A, Claudon M, Hélénon O. Ultrasound contrast agents: properties, principles of action, tolerance, and artifacts. Eur Radiol. 2001;11:1316-1328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 273] [Article Influence: 11.9] [Reference Citation Analysis (0)] |
| 27. | Wilson SR, Burns PN. Microbubble-enhanced US in body imaging: what role? Radiology. 2010;257:24-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 314] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 28. | Strobel D, Seitz K, Blank W, Schuler A, Dietrich C, von Herbay A, Friedrich-Rust M, Kunze G, Becker D, Will U, Kratzer W, Albert FW, Pachmann C, Dirks K, Strunk H, Greis C, Bernatik T. Contrast-enhanced ultrasound for the characterization of focal liver lesions--diagnostic accuracy in clinical practice (DEGUM multicenter trial). Ultraschall Med. 2008;29:499-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 176] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 29. | Piscaglia F, Bolondi L; Italian Society for Ultrasound in Medicine and Biology (SIUMB) Study Group on Ultrasound Contrast Agents. The safety of Sonovue in abdominal applications: retrospective analysis of 23188 investigations. Ultrasound Med Biol. 2006;32:1369-1375. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 596] [Cited by in RCA: 533] [Article Influence: 28.1] [Reference Citation Analysis (0)] |
| 30. | Puli SR, Reddy JB, Bechtold ML, Choudhary A, Antillon MR, Brugge WR. Accuracy of endoscopic ultrasound to diagnose nodal invasion by rectal cancers: a meta-analysis and systematic review. Ann Surg Oncol. 2009;16:1255-1265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 91] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 31. | Greis C. Technology overview: SonoVue (Bracco, Milan). Eur Radiol. 2004;14 Suppl 8:P11-P15. [PubMed] |
| 32. | Rappeport ED, Loft A, Berthelsen AK, von der Recke P, Larsen PN, Mogensen AM, Wettergren A, Rasmussen A, Hillingsoe J, Kirkegaard P, Thomsen C. Contrast-enhanced FDG-PET/CT vs. SPIO-enhanced MRI vs. FDG-PET vs. CT in patients with liver metastases from colorectal cancer: a prospective study with intraoperative confirmation. Acta Radiol. 2007;48:369-378. [PubMed] [DOI] [Full Text] |
| 33. | Quaia E, Calliada F, Bertolotto M, Rossi S, Garioni L, Rosa L, Pozzi-Mucelli R. Characterization of focal liver lesions with contrast-specific US modes and a sulfur hexafluoride-filled microbubble contrast agent: diagnostic performance and confidence. Radiology. 2004;232:420-430. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 355] [Cited by in RCA: 338] [Article Influence: 16.1] [Reference Citation Analysis (0)] |
| 34. | Sidhu PS, Cantisani V, Dietrich CF, Gilja OH, Saftoiu A, Bartels E, Bertolotto M, Calliada F, Clevert DA, Cosgrove D, Deganello A, D'Onofrio M, Drudi FM, Freeman S, Harvey C, Jenssen C, Jung EM, Klauser AS, Lassau N, Meloni MF, Leen E, Nicolau C, Nolsoe C, Piscaglia F, Prada F, Prosch H, Radzina M, Savelli L, Weskott HP, Wijkstra H. The EFSUMB Guidelines and Recommendations for the Clinical Practice of Contrast-Enhanced Ultrasound (CEUS) in Non-Hepatic Applications: Update 2017 (Long Version). Ultraschall Med. 2018;39:e2-e44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 653] [Cited by in RCA: 589] [Article Influence: 84.1] [Reference Citation Analysis (1)] |
| 35. | Rubaltelli L, Khadivi Y, Tregnaghi A, Stramare R, Ferro F, Borsato S, Fiocco U, Adami F, Rossi CR. Evaluation of lymph node perfusion using continuous mode harmonic ultrasonography with a second-generation contrast agent. J Ultrasound Med. 2004;23:829-836. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 80] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 36. | Liu LN, Xu HX, Lu MD, Xie XY, Wang WP, Hu B, Yan K, Ding H, Tang SS, Qian LX, Luo BM, Wen YL. Contrast-enhanced ultrasound in the diagnosis of gallbladder diseases: a multi-center experience. PLoS One. 2012;7:e48371. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 37. | Sauer R, Liersch T, Merkel S, Fietkau R, Hohenberger W, Hess C, Becker H, Raab HR, Villanueva MT, Witzigmann H, Wittekind C, Beissbarth T, Rödel C. Preoperative versus postoperative chemoradiotherapy for locally advanced rectal cancer: results of the German CAO/ARO/AIO-94 randomized phase III trial after a median follow-up of 11 years. J Clin Oncol. 2012;30:1926-1933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1251] [Cited by in RCA: 1489] [Article Influence: 114.5] [Reference Citation Analysis (0)] |
| 38. | Giesel FL, Choyke PL, Mehndiratta A, Zechmann CM, von Tengg-Kobligk H, Kayser K, Bischoff H, Hintze C, Delorme S, Weber MA, Essig M, Kauczor HU, Knopp MV. Pharmacokinetic analysis of malignant pleural mesothelioma-initial results of tumor microcirculation and its correlation to microvessel density (CD-34). Acad Radiol. 2008;15:563-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |