Published online Jun 27, 2023. doi: 10.4240/wjgs.v15.i6.1056
Peer-review started: January 16, 2023
First decision: January 31, 2023
Revised: February 3, 2023
Accepted: April 17, 2023
Article in press: April 17, 2023
Published online: June 27, 2023
Processing time: 149 Days and 20.7 Hours
Gastrointestinal complications are common in patients undergoing various forms of cancer treatments, including chemotherapy, radiation therapy, and molecular-targeted therapies. Surgical complications of oncologic therapies can occur in the upper gastrointestinal tract, small bowel, colon, and rectum. The mechanisms of action of these therapies are different. Chemotherapy includes cytotoxic drugs, which block the activity of cancer cells by targeting intracellular DNA, RNA, or proteins. Gastrointestinal symptoms are very common during chemotherapy, due to a direct effect on the intestinal mucosa resulting in edema, inflammation, ulceration, and stricture. Serious adverse events have been described as complications of molecular targeted therapies, including bowel perforation, bleeding, and pneumatosis intestinalis, which may require surgical evaluation. Radiotherapy is a local anti-cancer therapy, which uses ionizing radiation to cause inhibition of cell division and ultimately lead to cell death. Complications related to radio
Core Tip: Gastrointestinal complications are common in patients undergoing various forms of cancer treatments, including chemotherapy, radiation therapy, and molecular-targeted therapies. Surgical complications of oncologic therapies can occur in the upper gastrointestinal tract, small bowel, colon, and rectum. Treatment of the different gastrointestinal complications should be tailored to the individual patient and based on the underlying pathophysiology of the complication.
- Citation: Fico V, Altieri G, Di Grezia M, Bianchi V, Chiarello MM, Pepe G, Tropeano G, Brisinda G. Surgical complications of oncological treatments: A narrative review. World J Gastrointest Surg 2023; 15(6): 1056-1067
- URL: https://www.wjgnet.com/1948-9366/full/v15/i6/1056.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v15.i6.1056
Oncological treatments have greatly improved in the past few decades, thanks to the introduction of new therapies, such as immunologic agents or molecular targeted therapies, used alone or in combination with traditional chemotherapy and radiotherapy. The mechanisms of action of the various cancer therapies are different. Chemotherapy includes cytotoxic drugs, which block the activity of cancer cells by targeting intracellular DNA, RNA, or proteins[1,2].
Gastrointestinal symptoms are very common during chemotherapy, due to a direct effect on the intestinal mucosa resulting in edema, inflammation, ulceration, and stricture[3].
The development of molecular targeted therapies was due to the advances in oncological molecular biology. They include monoclonal antibody to vascular endothelial growth factor (VEGF) and epidermal growth factor receptor (EGFR), and tyrosine kinase inhibitors[1]. These drugs modify biological characteristics of tumor cells and have a key role to selectively block some mechanisms related to cell growth, proliferation, and invasion[2]. Serious adverse events have been described as complications of molecular targeted therapies, including bowel perforation, bleeding, and pneumatosis intestinalis (PI), which may require surgical evaluation[2,4-10].
To date, immunotherapy represents the standard of care for different types of cancer. Several agents, such as cytotoxic T-lymphocyte-associated protein-4 (CTLA-4) and inhibitors of programmed cell death protein-1 (PD-1) and its ligand (PD-L1), inhibit tumor growth through the stimulation of the body’s immune system against cancer. Immune-related adverse events mostly affect the gastrointestinal system, with heterogeneous symptoms that evolve into bowel ischemia or perforation, rarely[11,12].
Radiotherapy is a local anti-cancer therapy, which uses ionizing radiation to cause inhibition of cell division and ultimately lead to cell death[1]. Complications related to radiotherapy can be both acute and chronic. Acute symptoms occur within 2 mo and usually resolve in 3 mo[13,14]. Chronic symptoms, instead, occur months to years after radiotherapy. A high radiation dose, wide radiation area, long-term radiotherapy, and concurrent chemotherapy, are the factors related to an increased risk of toxicity[15]. The incidence of severe intestinal injury after abdominopelvic radiotherapy is about 4%-8%, and the main potentially surgical complications are perforation, strictures, abscesses, fistulas, and bleeding[16].
Ablative therapies, such as radiofrequency, laser, microwave, cryoablation, and chemical ablation with acetic acid or ethanol, can cause thermal or chemical injuries to the nearby structures[2].
As cancer treatments improve and new drugs are introduced, complications associated with oncologic therapies also increase. Many of these complications are life-threatening and have a high morbidity. As such, they require a prompt diagnosis. Therefore, it is crucial for surgeons to know the different complications and the therapies that can cause them, in order to ensure an immediate surgical treatment, if needed. In addition, knowing the stage and the prognosis of the disease is fundamental, and a multidisciplinary approach is necessary in order to personalize the surgical treatment. The purpose of this narrative review is to describe the complications related to different oncologic therapies that may require surgical interventions.
Neutropenic enterocolitis or typhlitis is typically diagnosed in patients with severe neutropenia related to oncologic treatment. This is a clinical syndrome characterized by abdominal pain, especially in the right lower quadrant, and fever. A systematic review by Gorschlüter et al[17] showed an incidence of 5.3% of neutropenic enterocolitis in patients treated for hematologic cancers or treated with high dose chemotherapy for solid tumors. Moreover, 7.0% of individuals undergoing myelosuppressive chemotherapy courses for hematologic malignancies will develop Clostridium difficile–associated diarrhea, of whom 8.2% will develop severe enterocolitis, compared with the 2.8% incidence in general inpatient cohorts[18,19].
On computed tomography (CT), the cecum is most frequently affected by circumferential wall thickening with involvement of pericolonic fat. The most severe form of neutropenic enterocolitis can be characterized by bowel necrosis and perforation. Therefore, a right colectomy should be performed to prevent complications, if there is no improvement in clinical condition within 2-3 d of conservative treatment[20,21].
Radiotherapy can also cause enterocolitis, and the sigmoid colon and rectum are the most affected segments in patients treated for pelvic cancers. Acute enterocolitis, due to edema, inflammation, and atrophy related to mucosal stem cell damage, manifests with abdominal pain, nausea, and diarrhea, and it is usually self-limiting in 2-6 wk with symptomatic treatments[1,15,22].
The pathophysiological mechanism that determines the development of chronic enterocolitis is based on the gradual increase in fibrosis of the intestinal wall, due to collagen deposition[22]. Radiotherapy-related vascular injury causing ischemia is another significant factor.
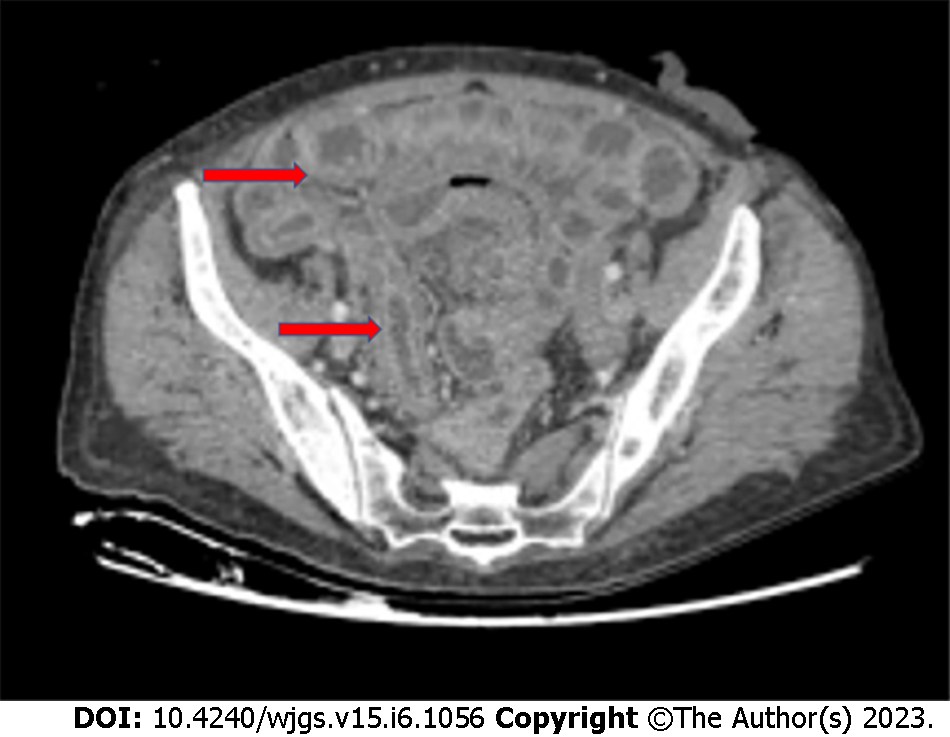
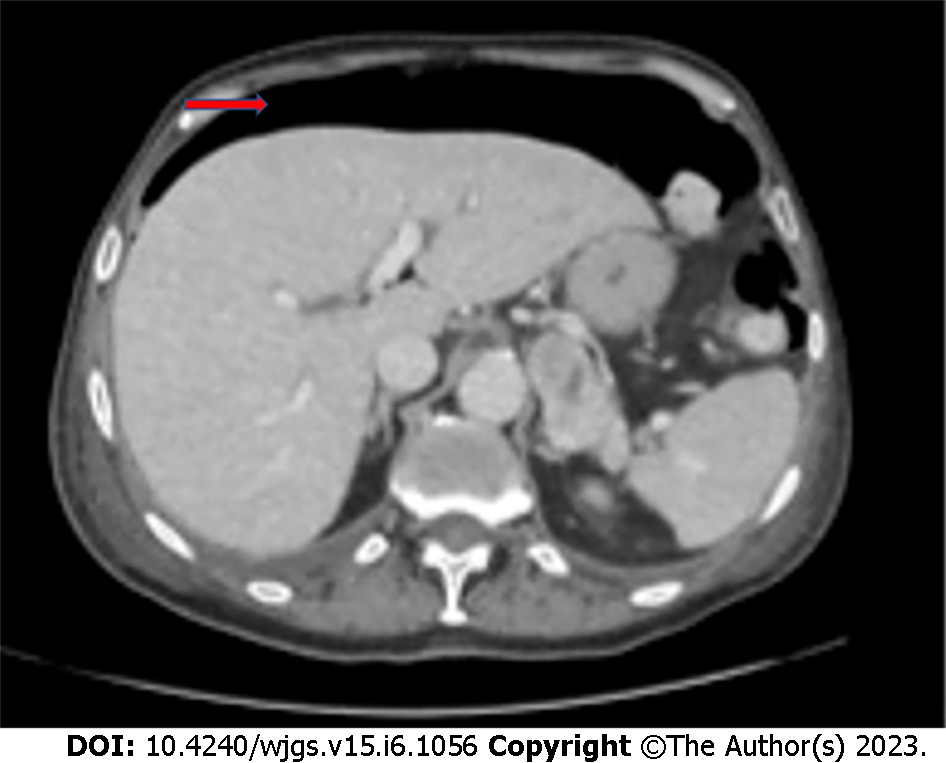
Chronic radiation enteritis affects 5% of patients treated with a dose of 45 Gy, reaching 50% in those treated with 65 Gy[23,24], and the terminal ileum is more commonly affected (Figure 1). Chronic radiation colitis occurs in 1%-5% of patients[25] and symptoms usually develop 6-12 mo after treatment. Bleeding, fistulas, abscesses, and stricture causing intestinal obstruction are the clinical manifestations of radiotherapy-related enterocolitis that may involve the surgeon.
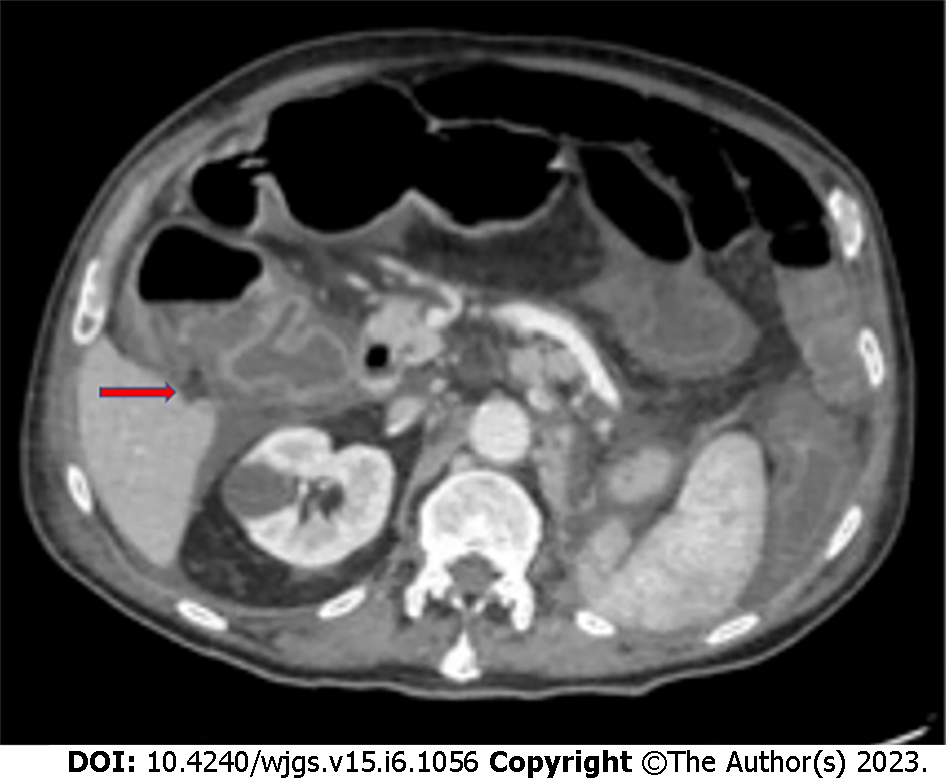
Patients undergoing chemotherapy may develop Clostridium difficile colitis (Figure 2), especially when treated with cyclophosphamide, methotrexate, fluorouracil, and doxorubicin[26]. Indications for surgery are the same as for antibiotic-related pseudomembranous colitis (i.e., perforation, fulminant toxic megacolon, and organ failure).
The most common gastrointestinal complications in case of treatment with checkpoint inhibitors are diarrhea and colitis, mainly in patients treated with anti-CTLA-4 (ipilimumab)[27]. Enterocolitis associated with immunotherapy has an incidence of 2.0%[28], which increases to 40.0% in patients on ipilimumab[29], and usually develops after 6-7 wk of treatment. Bowel perforation and death occur respectively in 1.0% and 0.8% of patients[27-31].
To sum up, surgery is required in all the enterocolitis cases consequent to oncological treatment if there is evidence of persistent bleeding, ischemia, perforation, or clinical worsening despite conservative treatment.
PI is a rare clinical condition characterized by the presence of air in the thickness of the intestinal wall. It is difficult to estimate the incidence of PI, as it is very often asymptomatic. However, its overall incidence, based on autopsy findings, is 0.03%[32].
PI can be idiopathic (about 15% of cases), when a cause cannot be identified, or secondary (about 85% of cases)[33]. In these cases, PI is associated with gastrointestinal or pulmonary diseases, mechanical ventilation, endoscopic procedures, infections, and drugs.
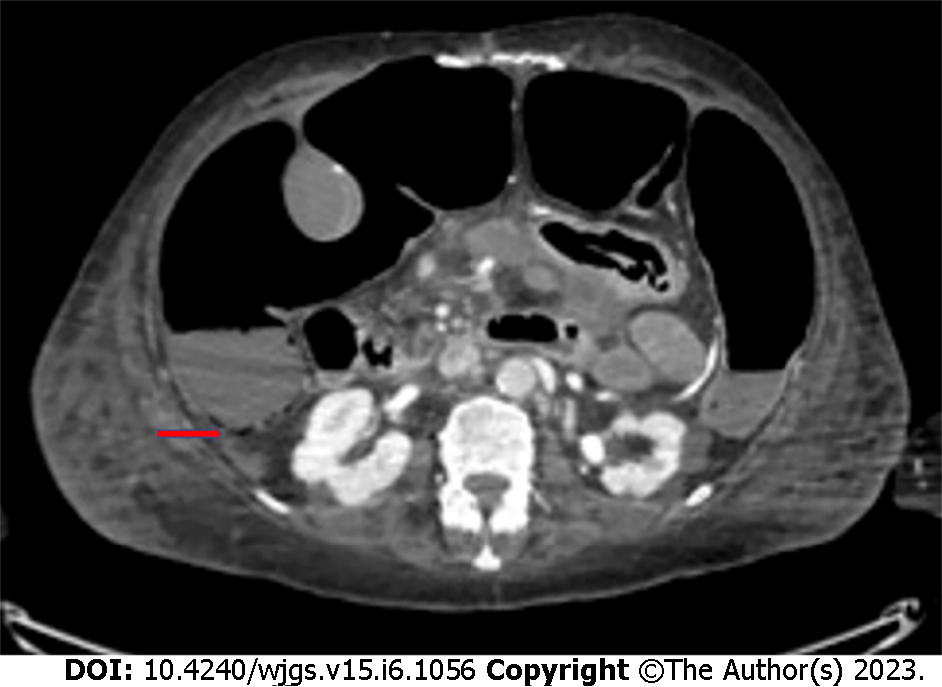
PI (Figure 3) can also occur as a complication of oncological medical therapies, including cytotoxic agents (cyclophosphamide, cytarabine, vincristine, doxorubicina, etoposide, docetaxel, irinotecan, and cisplatin) and molecular targeted agents (tyrosine kinase inhibitors such as imatinib, sunitinib, lenvatinib, and erlotinib; anti-VEGF monoclonal antibodies such as bevacizumab or anti-EGFR monoclonal antibodies such as cetuximab)[34,35].
The pathophysiological mechanisms underlying intestinal pneumatosis are not yet completely understood. As regards chemotherapeutic drugs, the most probable pathogenetic mechanism is the cytotoxic or ischemic damage caused by these drugs to the mucous layer of the intestinal wall. This damage would lead to the entry of gas, which is physiologically contained in the intestinal lumen, into the intestinal wall[36].
Chemotherapy-induced PI is also due to the myelosuppressive effects of drugs, which induce bone marrow aplasia and inhibit the regeneration process of damaged tissue[6]. Targeted therapies, on the other hand, are specific drugs that act as anti-VEGF/VEGFR, anti-EGFR, anti-PDGFR, and c-KIT inhibitors. These can determine a decrease in capillary density causing ischemia (anti-VEGF/VEGFR), a decrease in the efficiency in repairing intestinal damage (anti-EGFR, anti-PDGRF, and c-KIT inhibitors), and a reduction in intestinal motility (c-KIT inhibitors) by acting on Cajal cells[22,37-39].
According to a recent paper by Gazzaniga et al[6], PI mainly occurs in stage IV cancer patients (69.4% vs 11.1% of patients treated with a neoadjuvant therapy and 2.8% in adjuvant setting), and with the use of targeted therapies. PI is asymptomatic in most cases, and it is very often an occasional finding on CT performed in oncologic patients to monitor response to chemotherapy. No therapy is required in asymptomatic patients with PI. If present, symptoms can be extremely variable and may be indicative of bowel ischemia. The presence of hepatic and portomesenteric venous gas on a CT scan, associated with abdominal pain and alterations of blood tests and vital parameters, can be indicative of an ischemic pathology[40]. Hence, they require a prompt surgical exploration.
In fact, several studies in the literature demonstrated that the presence of gas in the portal vein is correlated to a transmural bowel ischemia in more than 90% of patients, and it is linked to a poor prognosis[41,42].
Therefore, it is very important to discriminate the cases in which surgery is necessary, to perform an immediate laparoscopy or laparotomy to avoid the progression of necrosis.
Intestinal obstructions represent an extremely common clinical condition in cancer patients, and they are caused by the tumor mass in most cases. Nevertheless, they can also be an effect of oncological therapies. Radiotherapy can induce a process of fibro-apoptosis which reduces the elasticity of the wall of the hollow viscera until it determines a stenosis. Small bowel strictures consequent to radiation therapy are a rare complication, caused by wall thickening and edema, which develop in 6-12 mo and occur especially in the terminal ileum, owing to its fixed position[22,43]. Intestinal obstructions caused by chronic radiation enteritis should be initially treated conservatively by fluid infusion, nasogastric tube placement, and possible use of laxatives[4,5].
Surgical treatment is indicated if there is no clinical response to medical therapy. One-third of patients with chronic radiation enteritis require surgery, approximately. Surgery is associated with a high morbidity rate and a high risk of reoperation. It is fundamental to resect the entire bowel involved in the stricture to prevent recurrence of obstruction, and to reduce complication and mortality rates[24]. Radiation therapy can also cause strictures of the esophagus and rectum[44,45]. In these cases, endoscopy is the treatment of choice with endoscopic dilatation and placement of self-expanding stents. Intestinal strictures caused by cytotoxic drugs, such as 5-fluorouracil and monoclonal antibodies (i.e., nivolumab) are extremely rare, but described in the literature[46,47].
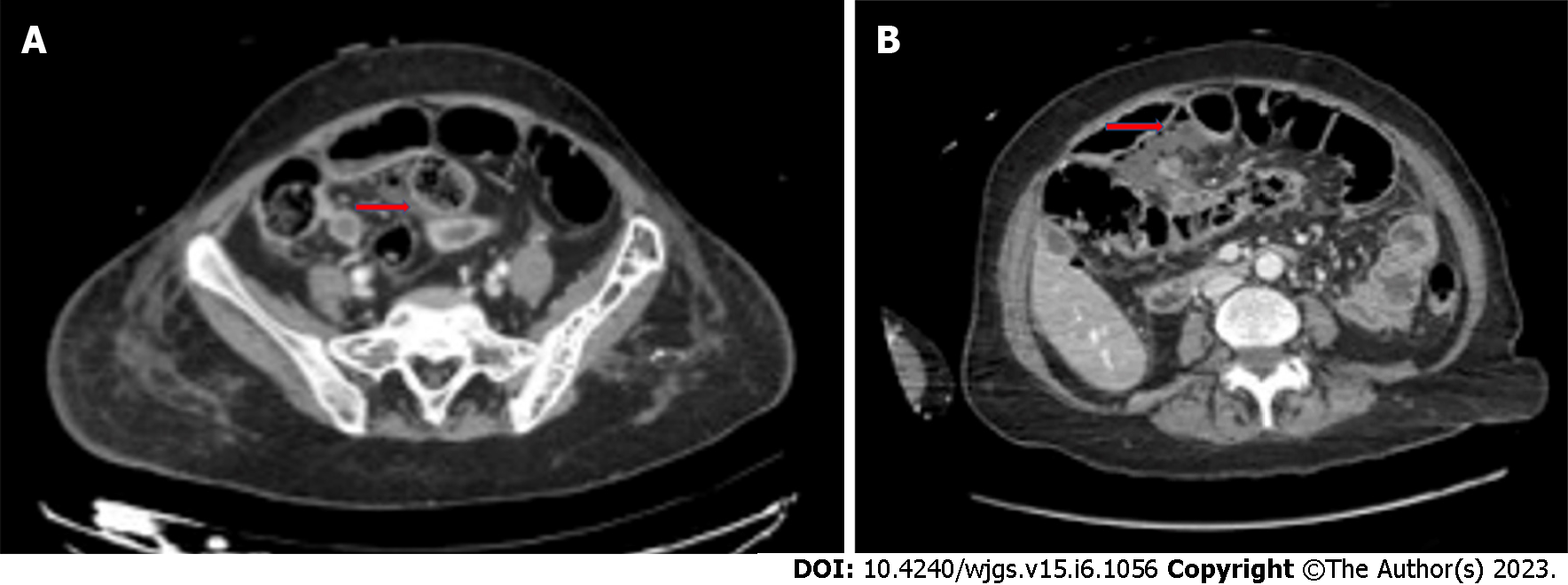
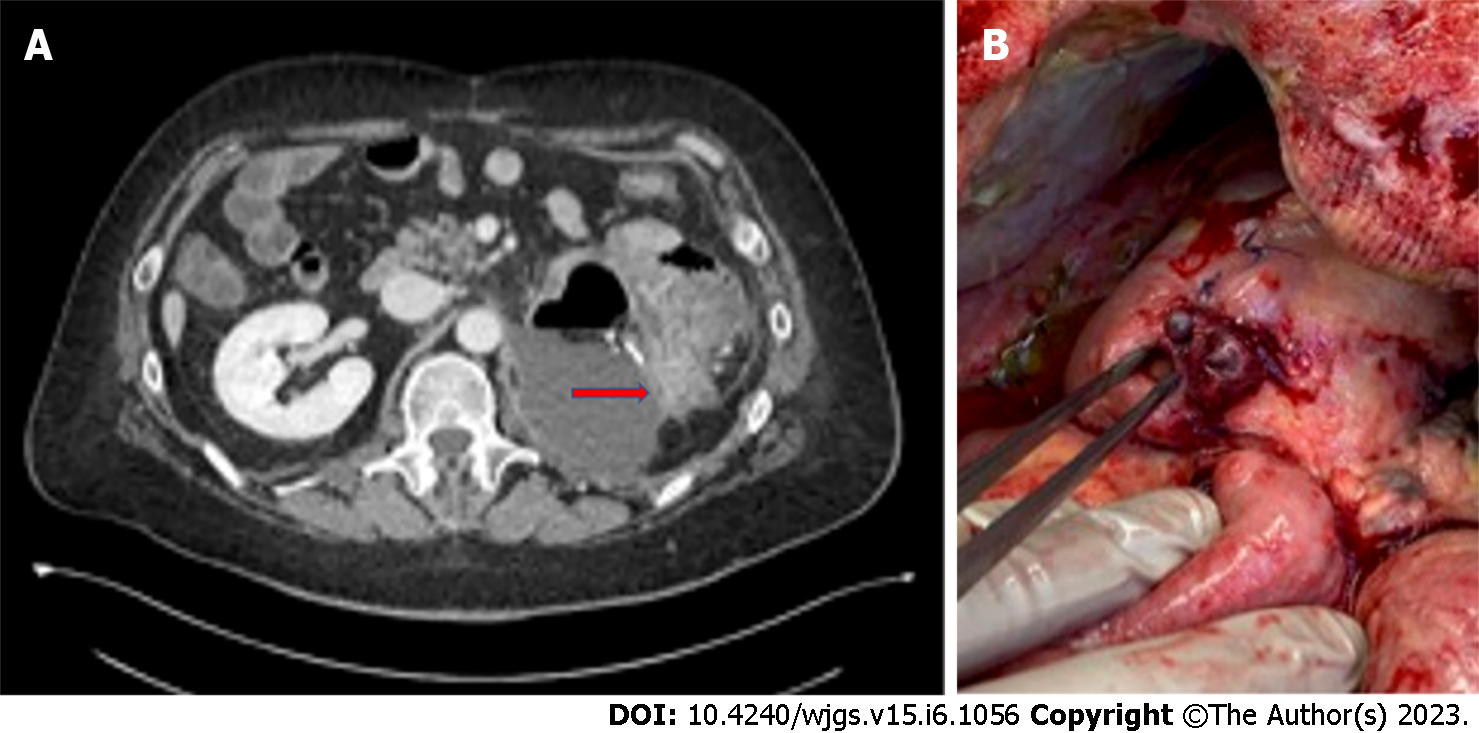
Bowel perforation is a rare but serious complication of cancer treatments. It has been reported in association with chemotherapy, molecular targeted therapies, immunotherapy, ablative techniques for solid tumors, and radiation therapy. Several mechanisms may be responsible for gastrointestinal perforation from oncologic treatments. Anticancer drugs induce vascular damage by thrombosis and thromboembolism, and when intestinal vessels are involved, bowel ischemia with perforation may occur[12]. Perforation of the gastrointestinal tract can also occur after prolonged obstruction[48] or due to treatment responses with tumor lysis, as in cases of lymphomas or gastrointestinal stromal tumors[20]. Finally, bowel perforation can be a result of other complications of oncologic therapies, like pneumatosis or enterocolitis. Management of perforation with no generalized peritonitis may be based on placement of image-guided percutaneous drainage in case of fluid collections. If there is a free perforation, instead, urgent laparotomy is needed, primarily to limit septic complications, which are characterized by a very high mortality rate in patients with neutropenia[20]. Gastrointestinal perforation has been reported in the literature with several chemotherapy agents, including fluorouracil, taxols, cisplatin, interleukin-2, and mytomicin[49-52]. Among the molecular targeted therapies, bevacizumab is most commonly associated with gastrointestinal perforation (Figure 4), with an incidence of 0.9%[53], and a correlation with late anastomotic leakage[54]. Risk factors for bevacizumab-related perforation are specific tumors (colorectal, prostate, and gynecological cancers), combination with other treatments, such as oxaliplatin and taxanes, presence of a primary tumor in situ, and recent history of endoscopy or abdominal radiotherapy[53,55-57].
Bowel perforation occurs in 80% of patients during the first 6 mo after bevacizumab administration[58], and the most common sites of perforation are the colon, small intestine and stomach[9]. The pathophysiological mechanisms underlying bowel perforation from molecular targeted therapy are different: The antiangiogenic action, which reduces capillary density of the mucosa layer and compromises intestinal wall integrity; the tumor lysis, in response to treatment; the increased risk of thromboembolic events in mesenteric vessels; and the regression of normal blood vessels[8,59].
Several studies in the literature also show an association between gastrointestinal perforation and antiangiogenic tyrosine kinase inhibitors, like erlotinib, regorafenib, sunitinib, and sorafenib[10,60-64] (Figure 5).
The incidence of tyrosine kinase inhibitors-related bowel perforation is still unknown, since there are mainly case reports in the literature. Intestinal perforation after immunotherapy is a rare event (Figure 6). A case report by Patel et al[31] described a jejunal perforation after treatment with ipilimumab and nivolumab for metastatic melanoma, related to tumor regression. Another paper by Romano et al[65] reported a small bowel perforation in a patient treated with nivolumab for metastatic lung cancer.Radiofrequency or micro-wave ablation can cause injuries to nearby organs. Bowel perforation with formation of abscesses and fistulas or free peritonitis, can be due to ablative therapies on liver cancer or, to a greater extent, on solid renal tumors, for direct thermal or chemical injuries[2,3]. In the literature, 4%-8% of patients treated with abdominopelvic radiation therapy can develop serious complications such as fistulas, perforation, or abscesses[16,66].
A recent paper by Zhan et al[67] showed that both long course and short course radiotherapy as neoadjuvant treatment for locally advanced rectal cancer increased the risk of anastomotic leakage, without a rise in postoperative mortality.
Risk factors for bowel perforation following radiotherapy are radiation dose, size of irradiation field, and the combination with other cancer treatments[14]. Several studies describe cases of intestinal perforation following the use of radiotherapy together with antiangiogenetic agents, like dabrafenib and trametinib for pelvic bone melanoma metastases[68], sorafenib in renal cancer patients[69], and gefitinib in a patient with lung cancer receiving lumbar irradiation[70]. The precise pathophysiology of radiotherapy-related bowel perforation is still unclear, but stem-cell and microvascular damage seems to have a pivotal role in gastrointestinal injuries affecting these patients.
Bleeding events in cancer patients can be caused by the disease itself or by medical treatments and require a surgical intervention, rarely. However, the surgeon may be involved in the multidisciplinary management of the patient or in case of failure of conservative treatments. Oncological therapies can affect the risk of hemorrhage both through alteration of the number or function of platelets and effect on the coagulation process. Some chemotherapeutic agents and anti-angiogenic targeted therapies are associated with increased bleeding tendency. For example, gastrointestinal bleeding has been described in patients receiving bevacizumab or in patients with gastrointestinal stromal tumors receiving imatinib or sunitinib[71,72].
Patients may present with different severity symptoms: Visible bleeding such as hematemesis, melaena, and hematuria, or occult bleeding for intraperitoneal or retroperitoneal hemorrhages. Treatment includes initial management by fluid infusion and blood transfusion. Endoscopy is a minimally invasive method to control the bleeding in the gastrointestinal tract, lungs, and bladder[73].
Angiography and interventional radiologic embolization of blood vessels represent additional minimally invasive bleeding control techniques. Nevertheless, these techniques present some technical issues: Accessibility of target blood vessels, subsequent ischemia of important non-target organs, and the availability of appropriate expertise[4,5]. Surgical treatment is reserved for patients with hemodynamic instability or in case of failure of other bleeding control techniques.
Rectal bleeding has been reported to occur in up to 53% of patients who received pelvic radiotherapy, but only 6% of these cases require interventions. The dose of radiotherapy is closely related to the risk of bleeding. The onset of rectal bleeding is described in the literature from 3 mo to 12 mo after radiotherapy.
Medical treatments for rectal bleeding after radiation therapy include sucralfate enemas, long term treatment with metronidazole, vitamin A, and hyperbaric oxygen therapy[74]. Endoscopic thermal therapies are frequently used in rectal bleeding and among these, argon plasma coagulation is the treatment of choice[75]. Radiologic embolization and surgery are required very rarely.
Granulocyte growth factor (G-CSF), also known as colony stimulating factor 3 (CSF 3), is a glycoprotein that stimulates the bone marrow to produce granulocytes and stem cells and release them into the bloodstream. This drug is widely used to treat neutropenia, a frequent side effect of many chemotherapy drugs[76]. It is also used to increase the content of hematopoietic stem cells before a bone marrow donation. Although G-CSF is generally well tolerated, a rare side effect of this drug is splenic rupture[77]. The mechanism underlying splenic damage is likely related to massive extramedullary hematopoiesis resulting in splenomegaly, splenic congestion, and nontraumatic rupture of the viscera. The patients generally present abdominal pain, mostly reported in the left hypochondrium, tenderness, anemia on blood tests and, in the most severe cases, hemodynamic instability. If a splenic rupture is suspected, a CT scan of the abdomen is required. Embolization of the splenic vessels is a valid option for stable patients and in hospital centers with the availability of interventional radiology. On the other hand, splenic rupture represents a surgical emergency for patients with hemodynamic instability. Splenic damage is also reported in the literature in patients treated with imatinib or idarubicin[78,79].
Non-occlusive mesenteric ischemia (NOMI) is another rare but serious complication of oncological treatments. A recent paper by Nagano et al[80] describes three cases of NOMI in patients undergoing chemotherapy for head and neck cancers. Prompt diagnosis and emergency surgical treatment are needed to reduce mortality rate and improve prognosis of patients with NOMI and bowel necrosis. Acute cholecystitis has been described in patients undergoing oncological therapies with antiangiogenetic targeted agents, including sunitinib, sorafenib, and bevacizumab[81]. Furthermore, a case of acute cholecystitis in a patient with metastatic renal cell carcinoma during therapy with everolimus, an inhibitor of the mammalian target of rapamycin, is reported in the literature[82]. Alithiasic cholecystitis has been described in patients with hematological diseases treated with vincristine, cyclophosphamide, or cytosine-arabinoside[83]. The proposed pathogenetic mechanism for the onset of acute cholecystitis during oncological therapies is related to the presence of microvascular ischemia or to an altered lipid metabolism, with consequent formation of gallstones. Symptoms and ultrasonographic findings, which include gallbladder distension, edema, hyperemia, pericholecystic fluid, and stranding, are analogous to those found in acute cholecystitis due to another etiology. In patients with acute cholecystitis, it is necessary to suspend cancer therapy temporarily or permanently. Some patients were treated exclusively with antibiotic therapy until symptoms resolved, and others underwent urgent chole
The development of specific cancer treatments has improved long-term survival in cancer patients. As a result, the risk of developing a second cancer after a primary oncologic treatment also increases, especially in long-survivor cancer patients.
The risk of a second tumor after radiotherapy is reported to be 0.1% to 1.0%[1] and radiation-induced cancers can be sarcomas, lymphomas, mesotheliomas, and carcinomas. The time to develop a post-radiation sarcoma is estimated to be 4-17 years[86]. Tamoxifen, a selective estrogen receptor modulator, is a chemotherapeutic agent used for the treatment of breast cancer. It is well known that tamoxifen is associated with an increased risk of endometrial cancer (two-to-three times higher than that in normal population)[87] and uterine sarcoma in postmenopausal patients. The onset of both tumors is related to the dose and time of therapy[88], and for this reason it is more frequent in long-survivor breast cancer patients for this reason.
Oncologic therapies have greatly improved over the past few years. As a result, complications related to cancer treatments have also increased. Gastrointestinal complications that most frequently require surgery are bowel perforations and obstructions (if conservative treatment fails). However, even for rarer complications, such as splenic rupture or diaphragmatic injury, emergency surgical treatment is necessary. Hence, it is essential for surgeons to be aware of new cancer therapies and their side effects, in order to act promptly if surgery is needed. It is also essential to keep in mind that the treatment of different gastrointestinal complications should be tailored to the individual patient and based on the underlying pathophysiology of the complication.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Surgery
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): 0
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Amagai T, Japan; Oley MH, Indonesia S-Editor: Chen YL L-Editor: Wang TQ P-Editor: Yu HG
| 1. | Ganeshan DM, Salem U, Viswanathan C, Balachandran A, Garg N, Silverman P, Bhosale P. Complications of oncologic therapy in the abdomen and pelvis: a review. Abdom Imaging. 2013;38:1-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 14] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 2. | Birch JC, Khatri G, Watumull LM, Arriaga YE, Leyendecker JR. Unintended Consequences of Systemic and Ablative Oncologic Therapy in the Abdomen and Pelvis. Radiographics. 2018;38:1158-1179. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Andreyev HJ, Davidson SE, Gillespie C, Allum WH, Swarbrick E; British Society of Gastroenterology; Association of Colo-Proctology of Great Britain and Ireland; Association of Upper Gastrointestinal Surgeons; Faculty of Clinical Oncology Section of the Royal College of Radiologists. Practice guidance on the management of acute and chronic gastrointestinal problems arising as a result of treatment for cancer. Gut. 2012;61:179-192. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 177] [Cited by in RCA: 213] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 4. | Bosscher MR, van Leeuwen BL, Hoekstra HJ. Surgical emergencies in oncology. Cancer Treat Rev. 2014;40:1028-1036. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 5. | Bosscher MR, van Leeuwen BL, Hoekstra HJ. Mortality in emergency surgical oncology. Ann Surg Oncol. 2015;22:1577-1584. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 6. | Gazzaniga G, Villa F, Tosi F, Pizzutilo EG, Colla S, D'Onghia S, Di Sanza G, Fornasier G, Gringeri M, Lucatelli MV, Mosini G, Pani A, Siena S, Scaglione F, Sartore-Bianchi A. Pneumatosis Intestinalis Induced by Anticancer Treatment: A Systematic Review. Cancers (Basel). 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 7. | Shinagare AB, Howard SA, Krajewski KM, Zukotynski KA, Jagannathan JP, Ramaiya NH. Pneumatosis intestinalis and bowel perforation associated with molecular targeted therapy: an emerging problem and the role of radiologists in its management. AJR Am J Roentgenol. 2012;199:1259-1265. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 76] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 8. | Sliesoraitis S, Tawfik B. Bevacizumab-induced bowel perforation. J Am Osteopath Assoc. 2011;111:437-441. [PubMed] |
| 9. | Yoshimoto T, Yoshikawa K, Higashijima J, Miyatani T, Tokunaga T, Nishi M, Takasu C, Kashihara H, Takehara Y, Shimada M. Bevacizumab-associated intestinal perforation and perioperative complications in patients receiving bevacizumab. Ann Gastroenterol Surg. 2020;4:151-155. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 10. | Ouchi R, Okada K, Usui K, Kurata N, Suzuki S, Nagao M, Watanabe Y, Koyama K. Intestinal Perforation in a Patient with Colon Cancer during Treatment with Regorafenib: A Case Report and Review of the Literature. Tohoku J Exp Med. 2021;254:207-211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 11. | Rajha E, Chaftari P, Kamal M, Maamari J, Chaftari C, Yeung SJ. Gastrointestinal adverse events associated with immune checkpoint inhibitor therapy. Gastroenterol Rep (Oxf). 2020;8:25-30. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 12. | Reginelli A, Sangiovanni A, Vacca G, Belfiore MP, Pignatiello M, Viscardi G, Clemente A, Urraro F, Cappabianca S. Chemotherapy-induced bowel ischemia: diagnostic imaging overview. Abdom Radiol (NY). 2022;47:1556-1564. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Yeoh EK, Horowitz M. Radiation enteritis. Surg Gynecol Obstet. 1987;165:373-379. [PubMed] |
| 14. | Zheng Y, Gao W, Spratt DE, Sun Y, Xing L. Management of gastrointestinal perforation related to radiation. Int J Clin Oncol. 2020;25:1010-1015. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 15. | O'Reilly M, Mellotte G, Ryan B, O'Connor A. Gastrointestinal side effects of cancer treatments. Ther Adv Chronic Dis. 2020;11:2040622320970354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 48] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 16. | Andreyev J. Gastrointestinal complications of pelvic radiotherapy: are they of any importance? Gut. 2005;54:1051-1054. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 157] [Article Influence: 7.9] [Reference Citation Analysis (1)] |
| 17. | Gorschlüter M, Mey U, Strehl J, Ziske C, Schepke M, Schmidt-Wolf IG, Sauerbruch T, Glasmacher A. Neutropenic enterocolitis in adults: systematic analysis of evidence quality. Eur J Haematol. 2005;75:1-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 182] [Cited by in RCA: 168] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 18. | Gorschlüter M, Glasmacher A, Hahn C, Schakowski F, Ziske C, Molitor E, Marklein G, Sauerbruch T, Schmidt-Wolf IG. Clostridium difficile infection in patients with neutropenia. Clin Infect Dis. 2001;33:786-791. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 114] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 19. | Loo VG, Bourgault AM, Poirier L, Lamothe F, Michaud S, Turgeon N, Toye B, Beaudoin A, Frost EH, Gilca R, Brassard P, Dendukuri N, Béliveau C, Oughton M, Brukner I, Dascal A. Host and pathogen factors for Clostridium difficile infection and colonization. N Engl J Med. 2011;365:1693-1703. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 590] [Cited by in RCA: 615] [Article Influence: 43.9] [Reference Citation Analysis (0)] |
| 20. | Sussman JJ. Surgical emergencies in the cancer patient. In: Norton JA, editor Surgery: Basic science and clinical evidence. New York: Springer-Verlag, 2007: 2117. [DOI] [Full Text] |
| 21. | Morgan C, Tillett T, Braybrooke J, Ajithkumar T. Management of uncommon chemotherapy-induced emergencies. Lancet Oncol. 2011;12:806-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 27] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 22. | Viswanathan C, Bhosale P, Ganeshan DM, Truong MT, Silverman P, Balachandran A. Imaging of complications of oncological therapy in the gastrointestinal system. Cancer Imaging. 2012;12:163-172. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 23. | Wobbes T, Verschueren RC, Lubbers EJ, Jansen W, Paping RH. Surgical aspects of radiation enteritis of the small bowel. Dis Colon Rectum. 1984;27:89-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 31] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Perrakis N, Athanassiou E, Vamvakopoulou D, Kyriazi M, Kappos H, Vamvakopoulos NC, Nomikos I. Practical approaches to effective management of intestinal radiation injury: benefit of resectional surgery. World J Gastroenterol. 2011;17:4013-4016. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 11] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 25. | Donner CS. Pathophysiology and therapy of chronic radiation-induced injury to the colon. Dig Dis. 1998;16:253-261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 45] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 26. | Anand A, Glatt AE. Clostridium difficile infection associated with antineoplastic chemotherapy: a review. Clin Infect Dis. 1993;17:109-113. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 205] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 27. | Lomax AJ, McNeil C. Acute management of autoimmune toxicity in cancer patients on immunotherapy: Common toxicities and the approach for the emergency physician. Emerg Med Australas. 2017;29:245-251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 28. | De Velasco G, Je Y, Bossé D, Awad MM, Ott PA, Moreira RB, Schutz F, Bellmunt J, Sonpavde GP, Hodi FS, Choueiri TK. Comprehensive Meta-analysis of Key Immune-Related Adverse Events from CTLA-4 and PD-1/PD-L1 Inhibitors in Cancer Patients. Cancer Immunol Res. 2017;5:312-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 310] [Cited by in RCA: 357] [Article Influence: 44.6] [Reference Citation Analysis (0)] |
| 29. | Kumar V, Chaudhary N, Garg M, Floudas CS, Soni P, Chandra AB. Current Diagnosis and Management of Immune Related Adverse Events (irAEs) Induced by Immune Checkpoint Inhibitor Therapy. Front Pharmacol. 2017;8:49. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 296] [Cited by in RCA: 430] [Article Influence: 53.8] [Reference Citation Analysis (0)] |
| 30. | Hodi FS, O'Day SJ, McDermott DF, Weber RW, Sosman JA, Haanen JB, Gonzalez R, Robert C, Schadendorf D, Hassel JC, Akerley W, van den Eertwegh AJ, Lutzky J, Lorigan P, Vaubel JM, Linette GP, Hogg D, Ottensmeier CH, Lebbé C, Peschel C, Quirt I, Clark JI, Wolchok JD, Weber JS, Tian J, Yellin MJ, Nichol GM, Hoos A, Urba WJ. Improved survival with ipilimumab in patients with metastatic melanoma. N Engl J Med. 2010;363:711-723. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10799] [Cited by in RCA: 11768] [Article Influence: 784.5] [Reference Citation Analysis (0)] |
| 31. | Patel KR, Lee LY, Tripathy A, McKean D. Case of small bowel perforation secondary to nivolumab and ipilimumab related tumour regression. BMJ Case Rep. 2020;13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 32. | Kang G. Benign pneumatosis intestinalis: Dilemma for primary care clinicians. Can Fam Physician. 2017;63:766-768. [PubMed] |
| 33. | Heng Y, Schuffler MD, Haggitt RC, Rohrmann CA. Pneumatosis intestinalis: a review. Am J Gastroenterol. 1995;90:1747-1758. [PubMed] |
| 34. | Mimatsu K, Oida T, Kawasaki A, Kano H, Kuboi Y, Aramaki O, Amano S. Pneumatosis cystoides intestinalis after fluorouracil chemotherapy for rectal cancer. World J Gastroenterol. 2008;14:3273-3275. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 13] [Cited by in RCA: 16] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 35. | Chaudhry NS, Bi WL, Gupta S, Keraliya A, Shimizu N, Chiocca EA. Pneumatosis Intestinalis After Molecular-Targeted Therapy. World Neurosurg. 2019;125:312-315. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 36. | Pieterse AS, Leong AS, Rowland R. The mucosal changes and pathogenesis of pneumatosis cystoides intestinalis. Hum Pathol. 1985;16:683-688. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 74] [Cited by in RCA: 66] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 37. | Vijayakanthan N, Dhamanaskar K, Stewart L, Connolly J, Leber B, Walker I, Trus M. A review of pneumatosis intestinalis in the setting of systemic cancer treatments, including tyrosine kinase inhibitors. Can Assoc Radiol J. 2012;63:312-317. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 38. | Lee YS, Han JJ, Kim SY, Maeng CH. Pneumatosis cystoides intestinalis associated with sunitinib and a literature review. BMC Cancer. 2017;17:732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 39. | Lee JY, Han HS, Lim SN, Shim YK, Choi YH, Lee OJ, Lee KH, Kim ST. Pneumatosis intestinalis and portal venous gas secondary to Gefitinib therapy for lung adenocarcinoma. BMC Cancer. 2012;12:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 40. | Knechtle SJ, Davidoff AM, Rice RP. Pneumatosis intestinalis. Surgical management and clinical outcome. Ann Surg. 1990;212:160-165. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 161] [Cited by in RCA: 154] [Article Influence: 4.4] [Reference Citation Analysis (1)] |
| 41. | Lassandro F, Scaglione M, Rossi G, Grassi R, Romano L. Portomesenteric vein gas: diagnostic and prognostic value. Emerg Radiol. 2002;9:96-99. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 19] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 42. | Wiesner W, Mortelé KJ, Glickman JN, Ji H, Ros PR. Pneumatosis intestinalis and portomesenteric venous gas in intestinal ischemia: correlation of CT findings with severity of ischemia and clinical outcome. AJR Am J Roentgenol. 2001;177:1319-1323. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 243] [Cited by in RCA: 208] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 43. | Theis VS, Sripadam R, Ramani V, Lal S. Chronic radiation enteritis. Clin Oncol (R Coll Radiol). 2010;22:70-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 157] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 44. | Murro D, Jakate S. Radiation esophagitis. Arch Pathol Lab Med. 2015;139:827-830. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 57] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 45. | Tagkalidis PP, Tjandra JJ. Chronic radiation proctitis. ANZ J Surg. 2001;71:230-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 73] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 46. | Kelvin FM, Gramm HF, Gluck WL, Lokich JJ. Radiologic manifestations of small-bowel toxicity due to floxuridine therapy. AJR Am J Roentgenol. 1986;146:39-43. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 18] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 47. | Tso DK, Avery LL, Lev MH, Kamalian S. Nivolumab-induced small bowel obstruction and perforation: a rare but life-threatening side effect of immunotherapy. Emerg Radiol. 2020;27:107-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 48. | Katabathina VS, Restrepo CS, Betancourt Cuellar SL, Riascos RF, Menias CO. Imaging of oncologic emergencies: what every radiologist should know. Radiographics. 2013;33:1533-1553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 49. | Heimann DM, Schwartzentruber DJ. Gastrointestinal perforations associated with interleukin-2 administration. J Immunother. 2004;27:254-258. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 50. | Cronin CG, O'Connor M, Lohan DG, Keane M, Roche C, Bruzzi JF, Murphy JM. Imaging of the gastrointestinal complications of systemic chemotherapy. Clin Radiol. 2009;64:724-733. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 51. | Liaw CC, Huang JS, Wang HM, Wang CH. Spontaneous gastroduodenal perforation in patients with cancer receiving chemotherapy and steroids. Report of four cases combining 5-fluorouracil infusion and cisplatin with antiemetics dexamethasone. Cancer. 1993;72:1382-1385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 52. | Rose PG, Piver MS. Intestinal perforation secondary to paclitaxel. Gynecol Oncol. 1995;57:270-272. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 32] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 53. | Hapani S, Chu D, Wu S. Risk of gastrointestinal perforation in patients with cancer treated with bevacizumab: a meta-analysis. Lancet Oncol. 2009;10:559-568. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 320] [Cited by in RCA: 307] [Article Influence: 19.2] [Reference Citation Analysis (0)] |
| 54. | Garant A, Des Groseilliers S, Martin L, Ferland É, Vuong T. Late anastomotic dehiscence during bevacizumab therapy for patients with colorectal cancer. Clin Oncol (R Coll Radiol). 2011;23:497-498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 55. | Kabbinavar FF, Flynn PJ, Kozloff M, Ashby MA, Sing A, Barr CE, Grothey A. Gastrointestinal perforation associated with bevacizumab use in metastatic colorectal cancer: results from a large treatment observational cohort study. Eur J Cancer. 2012;48:1126-1132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 39] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 56. | Badgwell BD, Camp ER, Feig B, Wolff RA, Eng C, Ellis LM, Cormier JN. Management of bevacizumab-associated bowel perforation: a case series and review of the literature. Ann Oncol. 2008;19:577-582. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 121] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 57. | Bonifazi M, Rossi M, Moja L, Scigliano VD, Franchi M, La Vecchia C, Zocchetti C, Negri E. Bevacizumab in clinical practice: prescribing appropriateness relative to national indications and safety. Oncologist. 2012;17:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 58. | Hurwitz H, Fehrenbacher L, Novotny W, Cartwright T, Hainsworth J, Heim W, Berlin J, Baron A, Griffing S, Holmgren E, Ferrara N, Fyfe G, Rogers B, Ross R, Kabbinavar F. Bevacizumab plus irinotecan, fluorouracil, and leucovorin for metastatic colorectal cancer. N Engl J Med. 2004;350:2335-2342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7832] [Cited by in RCA: 7727] [Article Influence: 368.0] [Reference Citation Analysis (1)] |
| 59. | Rutkowski P, Ruka W. Emergency surgery in the era of molecular treatment of solid tumours. Lancet Oncol. 2009;10:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 27] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 60. | Cheon YH, Kim MJ, Kang MG, Kim HJ, Lee SS, Kim CY, Jeon DH, Kim YE, Lee GW. Bowel perforation after erlotinib treatment in a patient with non-small cell lung cancer. Yonsei Med J. 2011;52:695-698. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 61. | Walraven M, Witteveen PO, Lolkema MP, van Hillegersberg R, Voest EE, Verheul HM. Antiangiogenic tyrosine kinase inhibition related gastrointestinal perforations: a case report and literature review. Angiogenesis. 2011;14:135-141. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 54] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 62. | Okamoto I, Miyazaki M, Morinaga R, Kaneda H, Ueda S, Hasegawa Y, Satoh T, Kawada A, Fukuoka M, Fukino K, Tanigawa T, Nakagawa K. Phase I clinical and pharmacokinetic study of sorafenib in combination with carboplatin and paclitaxel in patients with advanced non-small cell lung cancer. Invest New Drugs. 2010;28:844-853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 37] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 63. | Eng FC, Easson AM, Szentgyorgyi E, Knox JJ. Sorafenib and surgical complications: a case report of adverse reaction to sorafenib during treatment for renal cell carcinoma. Eur J Surg Oncol. 2009;35:219-221. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 64. | Frieling T, Heise J, Wassilew SW. Multiple colon ulcerations, perforation and death during treatment of malignant melanoma with sorafenib. Dtsch Med Wochenschr. 2009;134:e1-e2, 1464. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 65. | Romano S, Lassandro F, Scaglione M, Romano L, Rotondo A, Grassi R. Ischemia and infarction of the small bowel and colon: spectrum of imaging findings. Abdom Imaging. 2006;31:277-292. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 66. | Andreyev HJ, Vlavianos P, Blake P, Dearnaley D, Norman AR, Tait D. Gastrointestinal symptoms after pelvic radiotherapy: role for the gastroenterologist? Int J Radiat Oncol Biol Phys. 2005;62:1464-1471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 102] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 67. | Zhan TC, Zhang DK, Gu J, Li M. Surgical complications after different therapeutic approaches for locally advanced rectal cancer. World J Gastrointest Oncol. 2019;11:393-403. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 68. | Anker CJ, Grossmann KF, Atkins MB, Suneja G, Tarhini AA, Kirkwood JM. Avoiding Severe Toxicity From Combined BRAF Inhibitor and Radiation Treatment: Consensus Guidelines from the Eastern Cooperative Oncology Group (ECOG). Int J Radiat Oncol Biol Phys. 2016;95:632-646. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 104] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 69. | Inoue T, Kinoshita H, Komai Y, Kawabata T, Kawa G, Uemura Y, Matsuda T. Two cases of gastrointestinal perforation after radiotherapy in patients receiving tyrosine kinase inhibitor for advanced renal cell carcinoma. World J Surg Oncol. 2012;10:167. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 31] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 70. | Muraoka T, Tsukuda K, Toyooka S, Kagawa S, Naomoto Y, Takemoto M, Katsui K, Kanazawa S, Maki Y, Masuda H, Harada M, Asano H, Naito M, Miyoshi S. Ileal perforation induced by acute radiation injury under gefitinib treatment. Int J Clin Oncol. 2011;16:774-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 71. | Demetri GD, von Mehren M, Blanke CD, Van den Abbeele AD, Eisenberg B, Roberts PJ, Heinrich MC, Tuveson DA, Singer S, Janicek M, Fletcher JA, Silverman SG, Silberman SL, Capdeville R, Kiese B, Peng B, Dimitrijevic S, Druker BJ, Corless C, Fletcher CD, Joensuu H. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347:472-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3203] [Cited by in RCA: 3107] [Article Influence: 135.1] [Reference Citation Analysis (0)] |
| 72. | Liu Y, Zhang HL, Zhang Y, Mei JZ, Lin HW, Guo YW, Li RJ, Meng XR, Liu GJ, Li M, Xiao P, Bai H. Digestive tract hemorrhage due to complications with gastrointestinal stromal tumor treated with sunitinib: A case report. Oncol Lett. 2013;5:699-701. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 73. | Pereira J, Phan T. Management of bleeding in patients with advanced cancer. Oncologist. 2004;9:561-570. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 180] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 74. | Cavcić J, Turcić J, Martinac P, Jelincić Z, Zupancić B, Panijan-Pezerović R, Unusić J. Metronidazole in the treatment of chronic radiation proctitis: clinical trial. Croat Med J. 2000;41:314-318. [PubMed] |
| 75. |
Andreyev HJ.
Argon plasma coagulation in chronic radiation proctitis: Postgate |
| 76. | Aapro MS, Cameron DA, Pettengell R, Bohlius J, Crawford J, Ellis M, Kearney N, Lyman GH, Tjan-Heijnen VC, Walewski J, Weber DC, Zielinski C; European Organisation for Research and Treatment of Cancer (EORTC) Granulocyte Colony-Stimulating Factor (G-CSF) Guidelines Working Party. EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphomas and solid tumours. Eur J Cancer. 2006;42:2433-2453. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 415] [Cited by in RCA: 369] [Article Influence: 19.4] [Reference Citation Analysis (0)] |
| 77. | Masood N, Shaikh AJ, Memon WA, Idress R. Splenic rupture, secondary to G-CSF use for chemotherapy induced neutropenia: a case report and review of literature. Cases J. 2008;1:418. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 78. | Elliott MA, Mesa RA, Tefferi A. Adverse events after imatinib mesylate therapy. N Engl J Med. 2002;346:712-713. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 79. | Hernández R, del Cañizo MC, López C, González MI, Vázquez ML, Caballero MD, San Miguel JF. Pathologic rupture of the spleen during induction with ATRA in a patient with acute promyelocytic leukemia. Med Oncol. 2000;17:337-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 80. | Nagano H, Fujiwara Y, Matsuzaki H, Umakoshi M, Ohori J, Kurono Y. Three cases of non-occlusive mesenteric ischemia that developed after head and neck cancer therapy. Auris Nasus Larynx. 2021;48:1193-1198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 81. | Tirumani SH, Krajewski KM, Shinagare AB, Jagannathan JP, Ramaiya NH. Gallbladder complications associated with molecular targeted therapies: clinical and imaging features. Clin Imaging. 2014;38:50-55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 82. | Cetin B, Coskun U, Yildiz R, Buyukberber S, Baykara M, Benekli M. Acute cholecystitis in a patient with metastatic renal cell carcinoma treated with everolimus. J Oncol Pharm Pract. 2011;17:274-278. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 83. | Büyükaşik Y, Koşar A, Demiroğlu H, Altinok G, Ozcebe OI, Dündar S. Acalculous acute cholecystitis in leukemia. J Clin Gastroenterol. 1998;27:146-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 84. | Kogut MJ, Bastawrous S, Padia S, Bhargava P. Hepatobiliary oncologic emergencies: imaging appearances and therapeutic options. Curr Probl Diagn Radiol. 2013;42:113-126. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 85. | Kim KR, Thomas S. Complications of image-guided thermal ablation of liver and kidney neoplasms. Semin Intervent Radiol. 2014;31:138-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 86. | Sheppard DG, Libshitz HI. Post-radiation sarcomas: a review of the clinical and imaging features in 63 cases. Clin Radiol. 2001;56:22-29. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 118] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 87. | Committee Opinion No. 601: Tamoxifen and uterine cancer. Obstet Gynecol. 2014;123:1394-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 88. | Fong K, Causer P, Atri M, Lytwyn A, Kung R. Transvaginal US and hysterosonography in postmenopausal women with breast cancer receiving tamoxifen: correlation with hysteroscopy and pathologic study. Radiographics. 2003;23:137-50; discussion 151. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 21] [Article Influence: 1.0] [Reference Citation Analysis (0)] |