Published online Jan 27, 2020. doi: 10.4240/wjgs.v12.i1.28
Peer-review started: August 3, 2019
First decision: September 10, 2019
Revised: September 24, 2019
Accepted: November 26, 2019
Article in press: November 26, 2019
Published online: January 27, 2020
Processing time: 145 Days and 15.4 Hours
Gastrointestinal neurofibromas are commonly found in patients diagnosed with neurofibromatosis type 1. However, isolated gastrointestinal neurofibromas are a rare entity and only fourteen cases of isolated colorectal neurofibromas have been documented in literature. Isolated gastrointestinal neurofibromas have not been associated with Lynch syndrome (LS). Patients with LS are at an increased risk of colorectal cancer, and are recommended to undergo screening colonoscopy.
A 33-year-old healthy female with a family history of LS was found to have unresectable polyp in the ascending colon on screening colonoscopy suspicious for malignancy. The patient was asymptomatic and had no stigmata of neurofibromatosis. A staging workup for colorectal cancer revealed no evidence of metastatic disease. A discussion with the patient resulted in the decision to undergo a segmental resection with ongoing surveillance. The patient underwent a laparoscopic right hemicolectomy. Histopathology was consistent with a gastrointestinal neurofibroma. Post-operatively, the patient recovered well. She will not require further treatment with regards to her colonic neurofibroma, but will continue to follow-up for ongoing surveillance of her LS.
We present the first case of an isolated colonic neurofibroma in a patient with LS. This case explores considerations for the management of isolated gastrointestinal neurofibromas given the lack of guidelines in literature.
Core tip: Gastrointestinal neurofibromas are rarely found in isolation in the absence of systemic neurofibromatosis. We present herein, the first case of an isolated colonic neurofibroma in a patient with Lynch syndrome. There are currently no guidelines for the management of isolated gastrointestinal neurofibromas due to the lack of studies. We recommend considering establishing a diagnosis with endoscopic mucosal biopsy or ultrasound guided biopsy, reserving resection for patients with symptomatic disease or alternative indications, and continuing to follow patients for the surveillance of neurofibromatosis type 1 to reduce associated morbidity and mortality.
- Citation: Sun WY, Pandey A, Lee M, Wasilenko S, Karmali S. Isolated colonic neurofibroma in the setting of Lynch syndrome: A case report and review of literature. World J Gastrointest Surg 2020; 12(1): 28-33
- URL: https://www.wjgnet.com/1948-9366/full/v12/i1/28.htm
- DOI: https://dx.doi.org/10.4240/wjgs.v12.i1.28
Lynch syndrome (LS), or hereditary nonpolyposis colorectal cancer (HNPCC), is an autosomal dominant disorder caused by a germline mutation in one of the DNA mismatch repair genes (MLH1, MSH2, MSH6, or PMS2) that predisposes patients to various malignancies, of which colorectal cancer (CRC) is the most common[1]. Given the increased risk, patients diagnosed with LS are recommended to undergo cancer screening, including screening colonoscopy for CRC every two years beginning at the age of 20 to 25[2]. LS has also been associated with an increased risk of endometrial, gastric, ovarian, hepatobiliary, urinary tract, small bowel, and other cancers[1]. Additionally, the association of LS with neurofibromatosis has been reported in literature[3].
Neurofibromas are benign nerve sheath tumours originating from the peripheral nervous system containing an amalgamation of Schwann cells and fibroblasts[4]. While rare, malignant transformation into neurofibrosarcoma, or malignant peripheral nerve sheath tumour (MPNST) can occur[5]. Neurofibromas are typically associated with neurofibromatosis Type 1 (NF1), also known as von Recklinghausen disease. NF1 is an autosomal dominant disorder due to a mutation in chromosome 17[4]. Classic features of NF1 include café-au-lait macules, Lisch nodules, and neurofibromas of the skin; however, the cardiovascular system, eyes, bones, and gastrointestinal system can also be affected[6].
In patients with NF1 or multiple endocrine neoplasia type 2B (MEN 2B), gastrointestinal neurofibromas is relatively common, affecting approximately 25% of patients[7]. Most gastrointestinal neurofibromas occur in the stomach and small intestine[8]. These manifestations are typically asymptomatic, but symptoms may include abdominal pain, gastrointestinal bleeding, obstruction, and palpable masses[6]. However, isolated gastrointestinal neurofibromas in patients without a history of NF1 or MEN 2B are extremely rare[7]. In this report, we will present a rare case of isolated colonic neurofibroma in a patient with a history of LS.
A 33-year-old female with LS diagnosed with positive MSH6 mutation presented for routine screening gastroscopy and colonoscopy.
The patient was asymptomatic with no gastrointestinal issues or stigmata of neurofibromatosis.
The patient had a family history significant for LS and colorectal cancer in multiple family members. The patient was also diagnosed with LS with positive MSH6 mutation. She was otherwise healthy. There was no family history of NF1.
Physical examination revealed a benign abdomen. Cutaneous examinations revealed no evidence of café-au-lait spots or neurofibromatosis.
Complete blood count, comprehensive metabolic panel, liver panel, and coagulation studies were all within normal limits. The patient also had a normal carcinoembryonic antigen (CEA) level of 1.3 µg/L (normal range < 5.0 µg/L).
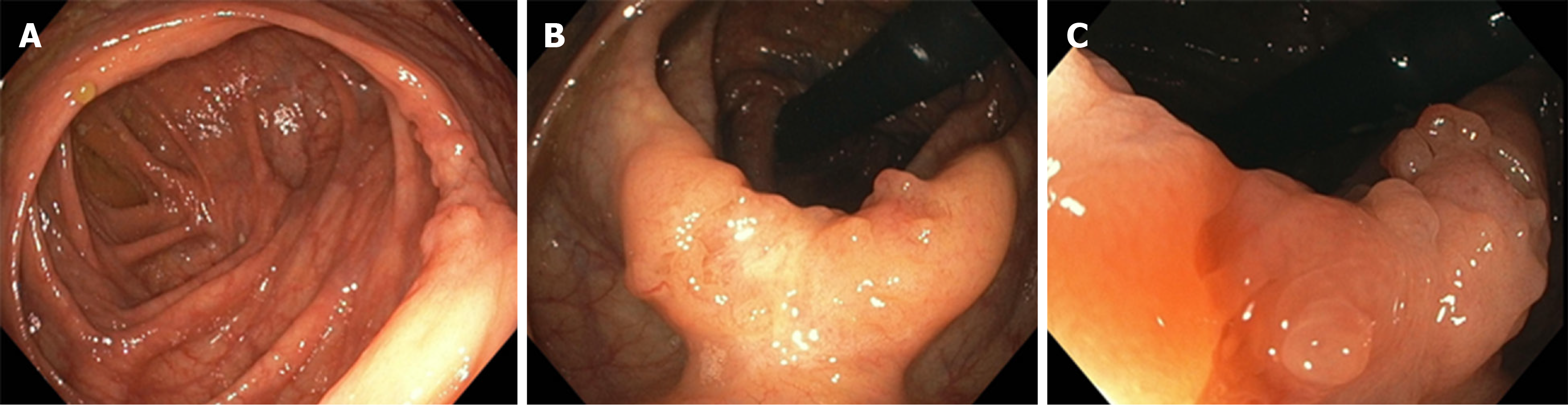
Initial screening gastroscopy was unremarkable. Random biopsies revealed no diagnostic abnormality. Initial screening colonoscopy demonstrated 2 to 2.5 cm of abnormal appearing mucosa in the ascending colon with central tethering. An attempt to lift the area of concern with methylene blue was unsuccessful. The area was tattooed and biopsied, which was non-diagnostic. The patient underwent a repeat colonoscopy after two months. The suspicious area was again appreciated appearing as sessile, serrated, polypoid tissue measuring approximately 2.5 cm wrapped around a haustral fold in the ascending colon (Figure 1). Biopsies were obtained again, but the polyp was unresectable. The biopsies revealed sessile serrated polyp, negative for dysplasia. A staging workup was completed, which included a negative computerized tomography (CT) scan of the chest, abdomen and pelvis.
Following two non-diagnostic biopsies, a referral to general surgery was made for the management of the unresectable polyp in the setting of LS. A discussion with the patient regarding the management included the recommendation of a subtotal colectomy versus a segmental resection, for which the patient elected for the latter. The patient underwent a laparoscopic right hemicolectomy.
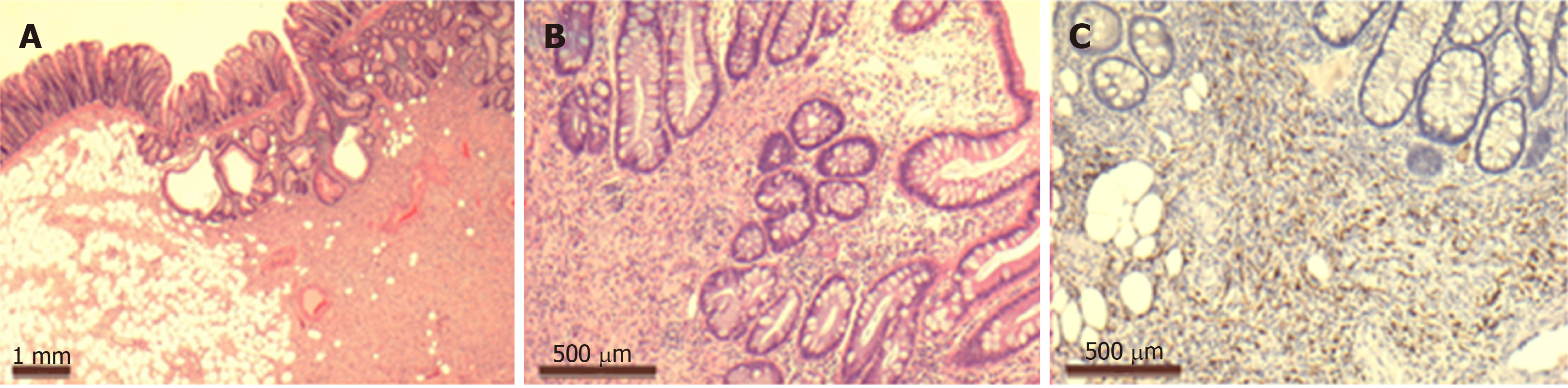
Gross pathological examination revealed a 2.2 cm × 1.8 cm × 0.4 cm ill-defined polypoid lesion adjacent to the cecum. Microscopic examination demonstrated an area of reactive serrated mucosa overlying an ill-defined submucosal mass extending into superficial muscularis propria and overlying mucosa. The mass consisted of short small monomorphic spindle cells within a collagenous background (Figure 2A). The spindle cells were intermixed with scattered mast cells (Figure 2B). Immunohistochemistry showed the spindle cells were positive for S100 (Figure 2C). Fifteen lymph nodes were identified and negative for malignancy. Overall, the histomorphology and phenotype by ancillary immunohistochemistry were consistent with a gastrointestinal neurofibroma.
The patient tolerated the procedure well and was discharged on postoperative day three. She was seen one month later in follow-up and had recovered well. She will not require any further treatment with regards to her isolated colonic neurofibroma, but will continue to follow-up for ongoing surveillance of her LS.
In this report, we presented the first case of isolated colonic neurofibroma in the setting of LS. Screening colonoscopy is routinely performed in patients with LS to diagnose colorectal cancers at an earlier stage. Commonly, clinicians and patients with non-diagnostic biopsies of suspicious colonic masses are faced with a dilemma: To resect or not resect. Given the patient in this study was at an increased risk of CRC due to LS, aggressive management of the mass with resection is appropriate. However, while submucosal aetiologies are more rare, it is also important to consider them in patients with non-diagnostic biopsies of colonic masses that may appear to have endoscopic features of submucosal origin. When conventional endoscopic mucosal biopsies fail to diagnose masses, the use of endoscopic ultrasound (EUS) guided biopsies may be considered to aid in the diagnosis[9].
Given the clinical context in this study, the patient was presented with the option of segmental resection versus subtotal colectomy. The patient expressed a desire for future pregnancy, and therefore, we elected for segmental resection to reduce the risk of infertility. However, total colectomy with ileoanal anastomosis would be the preferred primary treatment for patients with colon cancer or colon neoplasia unresectable by endoscopy[1]. The patient will require ongoing surveillance for CRC in the remainder of her colon. Furthermore, the patient would have required a completion proctocolectomy if her pathology confirmed a CRC.
Surprisingly, the final pathology revealed a gastrointestinal neurofibroma. Furthermore, the patient did not exhibit any features of NF1, thus, classifying this case as an isolated colonic neurofibroma. There have only been fourteen cases of isolated colorectal neurofibromas reported in the English literature (Table 1). The average age of presentation was 51 years. Nine patients (64%) were female, and four (29%) were asymptomatic.
| Ref. | Year ofpublication | Age (yr) | Sex | Presentation | Location of neurofibroma |
| Keith et al[15] | 1937 | 50 | F | Pain | Rectum |
| Woolf[16] | 1938 | 70 | M | Asymptomatic | Rectum |
| Butler et al[17] | 1959 | 45 | F | Pain, bleeding, tenesmus | Rectum |
| Geboes et al[18] | 1978 | NA | NA | NA | Rectum |
| Abramson et al[19] | 1997 | 53 | M | Bleeding | Transverse colon |
| Bononi et al[13] | 2000 | 68 | F | Tenesmus, bleeding | Sigmoid colon |
| Panteris et al[20] | 2005 | 65 | F | Bloody diarrhoea | Descending colon |
| Carter et al[7] | 2008 | 52 | F | Non-bloody diarrhoea, pain | Diffuse colonic involvement |
| Hindy et al[8] | 2012 | 59 | M | Asymptomatic | Transverse colon |
| Chelimilla et al[14] | 2013 | 70 | F | Asymptomatic | Ascending colon |
| Bilal et al[12] | 2016 | 52 | M | Pain | Proximal descending colon |
| Ahn et al[4] | 2016 | 26 | F | Asymptomatic | Sigmoid colon |
| Adioui et al[11] | 2018 | 29 | F | Pain, abdominal mass | Sigmoid colon |
| Miao et al[21] | 2018 | 24 | F | Pain, mass in stool | Ileocecal valve |
While LS has not been associated with neurofibromas, MSH6 mutation has been associated with NF1 phenotype in literature[10]. Given that gastrointestinal neurofibromas are rarely found in isolation, some have recommended referring patients with isolated gastrointestinal neurofibromas for the workup and surveillance of NF1[4,8,11-14]. We also agree with the need to screen patients with isolated gastrointestinal neurofibromas for NF1 given the associated morbidity and mortality with these conditions.
We present the first case of an isolated colonic neurofibroma in a patient with LS. Given the risk of colorectal cancer, the patient had a non-diagnostic polyp resected. There are currently no guidelines for the management of isolated gastrointestinal neurofibromas due to the lack of studies. We recommend considering establishing a diagnosis with endoscopic mucosal biopsy or ultrasound guided biopsy, reserving resection for patients with symptomatic disease or alternative indications, and continuing to follow patients for the surveillance of NF1 to reduce associated morbidity and mortality.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Canada
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Hashimoto N, Seow-Choen F S-Editor: Gong ZM L-Editor: A E-Editor: Ma YJ
| 1. | Giardiello FM, Allen JI, Axilbund JE, Boland CR, Burke CA, Burt RW, Church JM, Dominitz JA, Johnson DA, Kaltenbach T, Levin TR, Lieberman DA, Robertson DJ, Syngal S, Rex DK; US Multi-Society Task Force on Colorectal Cancer. Guidelines on genetic evaluation and management of Lynch syndrome: a consensus statement by the US Multi-Society Task Force on colorectal cancer. Gastroenterology. 2014;147:502-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 332] [Cited by in RCA: 361] [Article Influence: 32.8] [Reference Citation Analysis (0)] |
| 2. | Syngal S, Brand RE, Church JM, Giardiello FM, Hampel HL, Burt RW; American College of Gastroenterology. ACG clinical guideline: Genetic testing and management of hereditary gastrointestinal cancer syndromes. Am J Gastroenterol. 2015;110:223-262; quiz 263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 957] [Cited by in RCA: 1086] [Article Influence: 108.6] [Reference Citation Analysis (0)] |
| 3. | Bandipalliam P. Syndrome of early onset colon cancers, hematologic malignancies & features of neurofibromatosis in HNPCC families with homozygous mismatch repair gene mutations. Fam Cancer. 2005;4:323-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 67] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 4. | Ahn S, Chung CS, Kim KM. Neurofibroma of the Colon: A Diagnostic Mimicker of Gastrointestinal Stromal Tumor. Case Rep Gastroenterol. 2016;10:674-678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 5. | Dunn GP, Spiliopoulos K, Plotkin SR, Hornicek FJ, Harmon DC, Delaney TF, Williams Z. Role of resection of malignant peripheral nerve sheath tumors in patients with neurofibromatosis type 1. J Neurosurg. 2013;118:142-148. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 6. | Donk W, Poyck P, Westenend P, Lesterhuis W, Hesp F. Recurrent abdominal complaints caused by a cecal neurofibroma: a case report. World J Gastroenterol. 2011;17:3953-3956. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 7] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 7. | Carter JE, Laurini JA. Isolated intestinal neurofibromatous proliferations in the absence of associated systemic syndromes. World J Gastroenterol. 2008;14:6569-6571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 25] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 8. | Hindy P, Parvin R, Hanna K, Andrawes S, Gress F, Goodman A. An isolated neurofibromal polyp of the colon. Case Rep Gastroenterol. 2012;6:58-62. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 9. | Vander Noot MR, Eloubeidi MA, Chen VK, Eltoum I, Jhala D, Jhala N, Syed S, Chhieng DC. Diagnosis of gastrointestinal tract lesions by endoscopic ultrasound-guided fine-needle aspiration biopsy. Cancer. 2004;102:157-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 140] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 10. | Pemov A, Sung H, Hyland PL, Sloan JL, Ruppert SL, Baldwin AM, Boland JF, Bass SE, Lee HJ, Jones KM, Zhang X; NISC Comparative Sequencing Program, Mullikin JC, Widemann BC, Wilson AF, Stewart DR. Genetic modifiers of neurofibromatosis type 1-associated café-au-lait macule count identified using multi-platform analysis. PLoS Genet. 2014;10:e1004575. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 11. | Adioui T, Tamzaourte M, Nejjari F, Chakkor A, Elkaoui H, Boudhas A, Oukabli M, Rouibaa F, Aourarh A, Zentar A. Isolated Neurofibroma of the Sigmoid Colon: a Case Report and Review of the Literature. J Gastrointest Cancer. 2018;49:195-199. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 12. | Bilal M, Bilimoria F, Clarke K. An isolated colonic neurofibroma. Ann Gastroenterol. 2016;29:381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 13. | Bononi M, De Cesare A, Stella MC, Fiori E, Galati G, Atella F, Angelini M, Cimitan A, Lemos A, Cangemi V. Isolated intestinal neurofibromatosis of colon. Single case report and review of the literature. Dig Liver Dis. 2000;32:737-742. [PubMed] |
| 14. | Chelimilla H, Chandrala CK, Niazi M, Kumbum K. Incidental finding of isolated colonic neurofibroma. Case Rep Gastroenterol. 2013;7:369-375. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Keith A, Albert R. A case of neurofibroma of the rectal wall. Trans Am Proctol Soc. 1937;38:68. |
| 16. | Woolf MS. Neurofibroma of the Rectum. Cal West Med. 1938;49:463-464. [PubMed] |
| 17. | Butler DB, Hanna E. Neurogenic tumor of the rectum. Dis Colon Rectum. 1959;2:291-293. [PubMed] |
| 18. | Geboes K, De Wolf-Peeters C, Rutgeerts P, Vantrappen G, Desmet V. Submucosal tumors of the colon: experience with twenty-five cases. Dis Colon Rectum. 1978;21:420-425. [PubMed] |
| 19. | Abramson LP, Orkin BA, Schwartz AM. Isolated colonic neurofibroma manifested by massive lower gastrointestinal bleeding and intussusception. South Med J. 1997;90:952-954. [PubMed] |
| 20. | Panteris V, Vassilakaki T, Vaitsis N, Elemenoglou I, Mylonakou I, Karamanolis DG. Solitary colonic neurofibroma in a patient with transient segmental colitis: case report. World J Gastroenterol. 2005;11:5573-5576. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 24] [Cited by in RCA: 25] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 21. | Miao Y, Wang JJ, Chen ZM, Zhu JL, Wang MB, Cai SQ. Neurofibroma discharged from the anus with stool: A case report and review of literature. World J Clin Cases. 2018;6:455-458. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 4] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |