INTRODUCTION
The role of insulin in regulating glucose disposal in peripheral tissues, such as skeletal muscle and adipose tissue is well established. In the 50s, when it was discovered that insulin stimulated glucose uptake and disposal into the muscles, this effect was thought to be the most important way by which insulin regulated glycaemia in vivo. When the glucose/fatty acid regulatory cycle was discovered in the 60s a new insight of the way that insulin regulates glucose metabolism was introduced[1]. However, apart from its direct action on cells, insulin is also a vasoactive hormone, and it is now recognized that its vascular and metabolic actions are closely linked. Baron et al[2-4] originally introduced the concept that insulin might control its own access and that of other substrates - like glucose, lipids and several signalling molecules- to peripheral tissues, by increasing blood flow, and that this effect is compromised in states of insulin resistance.
In states of metabolic dysregulation, as in diabetes and obesity, there is deterioration in the cellular effects of insulin in peripheral tissues, which leads to a reduced ability of the latter to stimulate glucose uptake from the skeletal muscle and adipose tissue, as well as to inhibit lipolysis in fat depots[5]. Apart from the defects at the cellular level, the metabolic derangement could also be a result of the inability of insulin to cause vasodilatation and delivery of substrates to peripheral tissues especially in the postprandial period. This could contribute to the progression to type 2 diabetes as well as to the development of atherosclerosis, which is often evident even before overt hyperglycaemia develops[6].
In this review we summarize the current understanding of insulin action on peripheral blood flow and its implications on metabolic impairment both under fasting and postprandial conditions in type 2 diabetes.
INSULIN AND THE VASCULATURE IN NORMAL METABOLIC PHYSIOLOGY
Skeletal muscle
In skeletal muscle, insulin promotes the rate of glucose transport and the activities of hexokinase and 6-phosphofructokinase and subsequently the rate of glycolysis. In terms of protein metabolism, insulin increases synthesis and decreases degradation of proteins, in favour of an anabolic process[1]. Insulin also enhances vasodilatation and capillary recruitment, consequently increasing the flow of nutrients in peripheral tissues and especially in skeletal muscle[7]. It acts through traditional insulin receptors on the vascular endothelium to stimulate production of nitric oxide and induce vasodilatation[8]. The endothelial insulin response is mediated through a PI3-kinase pathway, which after several intermediate steps ends up activating endothelial nitric oxide synthase (eNOS)[6].
Blood flow is highly important for the metabolic function of skeletal muscle and under normal conditions increases after meal ingestion and during exercise and a correlation between the rate of insulin stimulated glucose uptake and the extent of vasodilatation seems to exist[9].
Insulin stimulates skeletal muscle glucose disposal and total muscle blood flow in a time- and dose-dependent fashion. In vivo, it enhances nitric oxide synthase-dependent vascular actions, in order to increase total skeletal muscle blood flow and to recruit muscle capillaries (by relaxing resistance and terminal arterioles, respectively). It is speculated that enhancing blood flow in this way on resistance vessels may induce the delivery of glucose and insulin to peripheral tissues and thus contribute to overall glucose disposal.
Capillary blood volume increases when precapillary arterioles dilate, thus increasing the flow to previously unperfused or underperfused areas, and total blood flow to skeletal muscle increases when larger resistance vessels relax[10].
Insulin increases tissue perfusion by augmenting microvasculature and, at normal concentrations, the rise in total muscle blood flow follows 60-90 min later[11,12].
Both haemodynamic effects of insulin, muscle blood flow increase and capillary recruitment seem to be independent of each other. Capillary recruitment occurs earlier in vivo, and at lower doses of insulin[13].
Insulin resistance may correlate to endothelial dysfunction in many ways, including dysregulation of sub-cellular signalling pathways that influence both insulin action and nitric oxide production[14,15].
Adipose tissue
Subcutaneous adipose tissue represents about 85% of whole body fat stores in subjects with various degrees of adiposity. Its main metabolic role is the storage of triglycerides which derive from energy overflow, and the release of stored lipids when other tissues are in need. Adipose tissue metabolism is under distinct control: usually, when a person consumes a meal, within the first hour postprandially, fat catabolism converts to fat storage, while the opposite happens in the case of physical activity. Adipose tissue interacts with the circulation by providing or drawing triglycerides and non-esterified fatty acids depending on metabolic needs. There are two kinds of triglyceride-rich lipoproteins: (1) chylomicrons, the largest particles, that carry the fat from absorbed nutrients within the intestine; and (2) very-low-density lipoproteins, that carry “endogenous” triglycerides and are released by the liver. Chylomicron- triglycerides are preferably stored within adipose tissue, and the fatty acid composition of adipose tissue (i.e., the kind of fatty acids that form its triglycerides) usually represents the composition of a person’s dietary fat intake, suggesting that adipose tissue triglycerides derive mainly from the ingested fat through diet. However, a proportion of plasma triglycerides are endogenously produced from non-lipid substrates (de novo lipogenesis) in adipose tissue[16-22].
In terms of metabolic regulation, adipose tissue can be divided into central (abdominal) and peripheral (lower body) depots. An unfavourable metabolic profile has been related to central fat accumulation (visceral and subcutaneous, each with distinct metabolic, endocrine and paracrine characteristics and blood flow rates)[23,24].
Adipose tissue regulates its metabolism, at least in part, by increasing its blood flow rate mainly in the early postprandial period[16]. Capillary perfusion is essential for that function. In the case of increased energy demands, as in physical activity, blood flow increases to facilitate the delivery of lipolytic products to peripheral tissues. Furthermore, after meal consumption, it helps the delivery of ingested substrates to fat depots for storage[16]. Adipose tissue blood flow responses are subject to adrenergic stimulation or inhibition. Adrenaline administration stimulates postprandial increases whereas beta-blockers inhibit the latter. Genetic studies in subcutaneous adipose tissue biopsies have identified expression of the type A receptor of A natriuretic peptide and of the synthase of nitric oxide, and have found an association of those with post-challenge blood flow responses[16,25-28].
Lipid kinetics and subcutaneous adipose tissue blood flow alterations are closely linked. More specifically, blood flow rises in response to an increased demand for lipolytic products as energy, or that for cleavage of free fatty acids from the circulation. In euglycaemic subjects with normal weight blood flow peaks within the first hour after a glucose load or a mixed meal. This facilitates the postprandial delivery of energy substrates and insulin to the fat depot, leading to adipose tissue lipoprotein lipase stimulation which stores circulating triglycerides and the suppression of hormone-sensitive lipase, which results in the inhibition of endogenous lipolysis[16,29-31].
On the other hand, visceral adiposity exerts even more unfavourable metabolic actions. Increased visceral fat has been associated with atherogenic dyslipidaemia and the development of atherosclerosis, even in non-diabetic individuals[32]. Although increased abdominal fat is in general positively associated with markers of inflammation and atherosclerosis, visceral fat is more strongly correlated with C-reactive protein, monocyte chemoattractant protein-1, interleukin-6 and isoprostanes independently of total adiposity, indicating a major role in systemic inflammation[33]. Furthermore, visceral fat has been more strongly related to hypertension both in men and women, and provides information towards the latter above BMI and waist circumference. However, subcutaneous adipose tissue is also contributing to vascular dysfunction, possibly through the actions of leptin apart from the presence of insulin resistance[34]. Both adipose tissue beds’ size has been found correlated to adipose tissue blood flow, independently of BMI, leptin or adiponectin concentrations[35].
INSULIN AND BLOOD FLOW IN TYPE 2 DIABETES
Skeletal muscle
Insulin provokes microvascular recruitment in skeletal muscle[10]. Impaired muscle blood flow as a facet of insulin resistance in subjects with either dysglycaemia or diabetes is well recognized in the literature. In the early 90s Steinberg et al[36] have shown that obese insulin resistant subjects present with an endothelial dysfunction and during a euglycaemic hyperinsulinaemic clamp they fail to increase endothelium-dependent vasodilation. In these trials, catheterizations of the femoral artery was used to measure the response to an intra-arterial vasodilator stimulus, comparing control to euglycemic-hyperinsulinemic clamp conditions[36].
Thereafter, it was suggested that since insulin exerts its vasodilatory effects through endothelial nitric oxide release, in vivo stopping nitric oxide production could inhibit insulin’s vasoactive actions in skeletal muscle and consequently reduce glucose uptake[37]. Moreover in obese insulin resistant subjects, insulin resistance in skeletal muscle was promoted by the increased endogenous endothelin action[38].
A rat model of insulin resistance has shown that endothelial-dependent vasodilation is blunted, in part due to an unresponsive nitric oxide synthase to insulin, leading to decreased nitric oxide levels in the endothelial cells[39,40].
In type 2 diabetes and other insulin-resistant states, impaired suppression of adipose tissue lipolysis and postprandial hyperglycemia favour non-esterified fatty acid utilization and oxidation and increase glucose uptake from insulin independent tissues (like liver). Dyslipidaemia, usually related to lack of insulin sensitivity, enhances atherosclerosis and triggers inflammation in endothelial cells[41].
In insulin-resistant patients basal blood flow is generally not altered[42-44]. Laakso et al[45], demonstrated that insulin cannot effectively increase muscle blood flow in type 2 diabetic patients, using the combined euglycemic clamp and leg balance techniques during different insulin infusions. They also concluded that impaired insulin-dependent rise in skeletal muscle blood flow can be attributed to the diabetic milieu and not to obesity, in a study of obese diabetic patients[45].
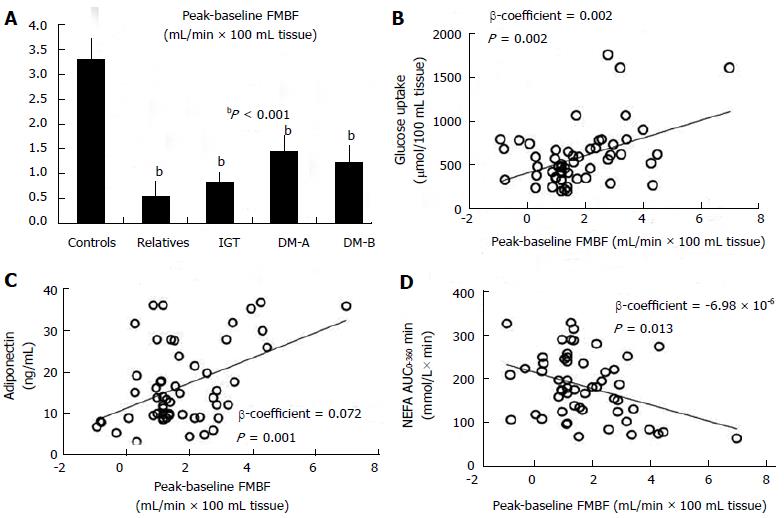
Lambadiari et al[46] studied simultaneously lean subjects with insulin sensitivity varying from normoglycaemic insulin-resistant first-degree relatives of diabetic subjects to prediabetic and diabetic patients with either isolated postprandial hyperglycaemia or overt diabetes[46]. They demonstrated that using a physiological mixed meal as a stimulus, the postprandial augmentation in forearm muscle blood flow is blunted throughout all stages of metabolic impairment compared to controls; this occurs even before overt hyperglycaemia develops. The latter affected glucose disposal in muscle, which was also unresponsive after meal delivery and was also positively correlated to the post-load muscle blood flow differences. Lipid substrates affected blood flow peak as well. Triglyceride levels had a negative impact on blood flow responsiveness in the fed as well as in the fasting period. Post-challenge non-esterified fatty acids levels exhibited a negative effect on blood flow responsiveness, suggesting a possible mechanism for the decrease in muscle glucose clearance after the meal. A lower serum adiponectin level was also seen in the diabetic and the prediabetic insulin-resistant subjects, with the latter being positively related to the decreased postload blood flow rise[46] (Figure 1).
Figure 1 Forearm muscle blood flow peak-baseline values in all groups (A), and associations between peak-baseline forearm muscle blood flow and forearm muscle glucose uptake (B), plasma adiponectin (C) and postprandial non-esterified fatty acids (D), in subjects at all stages of type 2 diabetes.
A: bP overall < 0.001; B: Forearm muscle glucose uptake = 427.9 + 101.4 peak-baseline FMBF, P = 0.001; C: Adiponectin = 12.17 + 3.05 peak-baseline FMBF, P < 0.001; D: Postprandial NEFA (AUC0-360) = 209.5 - 18.52 peak-baseline FMBF, P = 0.005. FMBF: Forearm muscle blood flow; NEFAs: Non-esterified fatty acids. IGT: Impaired glucose tolerance; DM: Diabetes mellitus. Adapted from Lambadiari et al[46].
In subjects with morbid obesity postprandial muscle blood flow was also blunted in a study by the same group and this contributed to the decrease in muscle glucose uptake postprandially[47]. The same was observed by the same group in another insulin resistant state, such as hypothyroidism, in which a decreased postprandial blood flow response was coupled with an impairment in muscle glucose uptake[48]. In a study by Magalhães et al[49] administration of metformin to non-obese type 2 diabetic patients increased post-load forearm muscle blood flow and lowered free fatty acids, thus improving glucose oxidation and insulin sensitivity in the muscle bed.
However, there is not universal agreement with the above mentioned results, since numerous studies have failed to reveal a defect in insulin-mediated blood flow in type 2 diabetic patients[42,50]. There is a certain discrepancy since the literature either confirms or not a substantial[45,51], or an unimportant correlation between insulin kinetics, muscle blood flow and glucose disposal[52,53].
Some of these discrepancies may at least partially explained by the different studies populations and by the experimental protocol used. The commonly used clamp technique is not physiological, because these exceptionally high insulin concentrations are not normally present for long after meal consumption. Hence, one could question the physiological significance of such an increase in blood flow rates. A normal stimulus, such as a mixed meal, can provide evidence of a real life metabolic state[46,54]. However, not only the type of meal but the method for the detection of blood flow is important in this evaluation.
Adipose tissue
In lean insulin-sensitive subjects, abdominal adipose tissue blood flow increases by two- to four-times in response to feeding. The same seems to be true for blood flow in lower body fat depots (thigh) and forearm tissues. Physiologically, adipose tissue blood flow peaks within half to one hour after nutrient ingestion. This rise coincides with plasma insulin peak and the inhibition of lipolysis[23].
By studying obese or diabetic individuals in the 90s, Jansson et al[55,56] detected impairment in adipose tissue blood flow response as a facet of insulin resistance coupled with hypertension and elevated lipolysis products.
Since then, numerous studies have shown that in states of decreased insulin sensitivity, as in “diabesity’’, the postprandial increase in adipose tissue blood flow is reduced[57-60]. Karpe et al[61] showed that the postprandial blood flow rise is associated with insulin sensitivity independently of weight. Moreover, they showed that hyperinsulinaemia affects adipose tissue blood flow indirectly by stimulation of sympathetic activity[61].
Previous reports in healthy subjects by the same research group have demonstrated that nitric oxide determines the actual rate of adipose tissue blood flow, whereas postprandial augmentation of it is mainly under adrenergic regulation in vivo, and that blood flow regulation and lipolysis are co-regulated[25].
Dimitriadis et al[62] showed an altered fasting and postprandial adipose tissue blood flow in all stages of metabolic regulation, from the prediabetic state to clinical diabetes, even in lean first-degree relatives of diabetic patients. This study, using a mixed meal as a stimulus, showed significant association of postprandial adipose tissue blood flow with insulin sensitivity. Basal and post-challenge triglycerides were negatively correlated to the responsiveness of adipose tissue blood flow; the same was true for postprandial non-esterified fatty acids but not for fasting values[62] (Figure 2).
Figure 2 Plasma glucose, and adipose tissue blood flow in healthy subjects (control), first degree relatives of subjects with type 2 diabetes (relatives), subjects with impaired glucose tolerance, subjects with type 2 diabetes with postprandial hyperglycemia and normal fasting plasma glucose levels (diabetes mellitus group A) and subjects with type 2 diabetes with both postprandial and fasting hyperglycemia (diabetes mellitus group B).
“P” stands for overall comparison (repeated measures ANOVA) between control and patient groups. IGT: Impaired glucose tolerance. Adapted from Dimitriadis et al[62].
Fatty acid overflow (mainly palmitic acid), a well recognized factor to interfere with insulin sensitivity, causes both cellular and vascular insulin dysfunction[63]. The increased rate of lipolysis in diabetes may result in increased lipid oxidation and a decreased glucose oxidation rate[61,64].
Impairment in blood flow response of adipose tissue has been found in other insulin resistance states. Mitrou et al[47] study in morbidly obese subjects, shows a drop in postprandial adipose tissue blood flow response and in glucose disposal per 100 mL fat tissue. However, glucose fractional extraction from subcutaneous fat depot was unaltered and glucose uptake per total fat mass was increased. Thus, it seems that although an expanded adipose tissue causes insulin resistance, total fat mass provides a buffer for glucose overflow and compensates for insulin resistance.
Diabetic subjects fail to increase adipose tissue blood flow during prolonged exercise of moderate intensity, in combination to the inability to regulate non-esterified fatty acid mobilization and adipose tissue glucose clearance[65]. Exercise augments adipose tissue lipolysis in diabetic patients, but due to an impaired blood flow response, a high proportion of free fatty acids that come from lipolysis cannot be released into the circulation. Visceral glucose release is lower than whole-body glucose utilisation during exercise and post-exercise recovery[66].
The cause of the impairment in postprandial adipose tissue blood flow reactivity in insulin resistance is still obscure. One potential explanation is the downregulation of the adrenergic receptor during chronic sympathetic stimulation in a milieu of long-standing hyperinsulinaemia. Sympathetic nervous system overactivity induces oxidative stress. Increased levels of circulating free oxygen radicals consumes nitric oxide, and inhibits physiological insulin-dependent vasodilatation[23]. Interestingly, the transcription of eNOS and natriuretic peptide receptor-A, which are expressed in adipose tissue and interfere with vasoactive actions, was associated with adipose tissue blood flow responsiveness to feeding. This finding suggests that part of blood flow regulation is at a transcriptional level and it is independent of adiposity[28].
At the bottom line, adipose tissue is an important buffer against the postprandial spill-over of nonesterified fatty acids in the circulation, thus protecting other peripheral tissues. This buffering effect is dysregulated in states of an over-expanded inflammatory, hypoxic adipose tissue, where the postprandial blood flow response is minimized, potentially leading to atherogenic dyslipidaemia[67].
CONCLUSION
Resistance in the haemodynamic actions of insulin is essential for the development of type 2 diabetes and insulin resistant states as well as their complications, namely cardiovascular disease, the development of which often precedes overt hyperglycaemia and which is the primary cause of mortality within the diabetic population.
Insulin normally stimulates microvascular perfusion (capillary recruitment) of skeletal muscle and subcutaneous adipose tissue and thus increases blood flow mainly after meal ingestion or physical exercise. This effect is impaired in insulin resistance and type 2 diabetes early during metabolic dysregulation development and reflects early-onset vascular dysfunction. Failure of insulin to increase muscle blood flow results in the inability to regulate its own delivery and that of other substrates and hormones and consequently to a decrease in glucose disposal. In fat depots blood flow is closely related to triglyceride clearance and non-esterified fatty acid kinetics. Therefore, we may speculate that dysregulation of post-challenge blood flow responsiveness in skeletal muscle and adipose tissue may together underlie some of the detrimental aspects of insulin resistance.