INTRODUCTION
To prevent chronic diabetic complications, it is necessary to try to achieve normoglycemia as much as possible. Previously, glycemic control used to be evaluated by plasma glucose or urinary glucose. However, these indicators fluctuate continuously due to factors such as dietary intake, and it was difficult to evaluate glycemic control correctly by taking measurements at a particular time. Therefore, hemoglobin A1c (HbA1c), which reflects mean plasma glucose during the past 1 to 2 mo, was introduced as a glycemic control indicator[1], and is now widely used in clinical practice for diabetes mellitus. HbA1c can be used to evaluate glycemic control status; if poor glycemic control is observed, it is possible to make additions, changes, etc. to the treatment of diabetes mellitus[2].
Large-scale researches such as the Diabetes Control and Complications Trial revealed that HbA1c is related to the development and progression of diabetic microangiopathy[3]. That is, the development and progression of diabetic microangiopathy can be prevented by maintaining excellent glycemic control using HbA1c as an indicator. Recently, it also became possible to use HbA1c for the diagnosis of diabetes mellitus[4].
However, the following problems of HbA1c were pointed out: (1) abnormal HbA1c values may be observed because of variantl hemoglobin, hemolytic anemia, etc.; (2) HbA1c does not correctly reflect short-term glycemic control status; and (3) HbA1c does not correctly reflect postprandial plasma glucose/fluctuation of plasma glucose. Accordingly, new glycemic control indicators such as fructosamine, 1,5-anhydroglucitol (1,5-AG), and glycated albumin (GA) were introduced. Although these indicators compensate the disadvantages of HbA1c, they have their own disadvantages. For example, 1,5-AG is affected by the threshold of urinary glucose excretion in the kidney, and fructosamine and GA are affected by albumin metabolism[5].
Because fetal hemoglobin (HbF) is the main hemoglobin in neonates, HbA1c cannot be used as a glycemic control indicator in neonates. Therefore, glycemic control in neonatal diabetes mellitus (NDM) was traditionally performed using blood glucose measured by self-monitoring of blood glucose as an indicator, without using a glycemic control indicator. We demonstrated that GA, which is not affected by HbF, reflects glycemic control in NDM and can be used as a glycemic control indicator in NDM[6]. We also obtained various other findings about GA and HbA1c in neonates/infants or NDM. In this review, we outlined the most recent findings concerning glycemic control indicators in neonates or NDM.
NEONATAL DIABETES MELLITUS
NDM is a type of diabetes mellitus caused by single-gene abnormality which develops acutely in insulin dependent state; NDM accounts for the majority of cases of diabetes mellitus which develops within 6 mo after birth[7]. The frequency of NDM according to this definition is 1 in 89000 births, showing that NDM is a rare disease[8]. So far, more than 20 causative genes of NDM have been discovered; genetic mutations of some kind have been identified in not less than 70% of patients[8,9]. NDM is similar to type 1 diabetes mellitus in terms of the form of development (diabetes mellitus develops acutely); however, type 1 diabetes mellitus very rarely develops within 6 mo after birth, judging from studies on the frequency of human leukocyte antigen risk alleles and the presence of pancreatic autoantibodies[10,11]. Based on the clinical course, NDM is classified into two major categories: transient NDM (TNDM) and permanent NDM (PNDM)[12]. TNDM is a condition in which insulin secretion is restored spontaneously and normoglycemia is achieved without treatment; PNDM is a condition in which remission is not achieved and life-long treatment is required. The frequency of TNDM is about 60%, and that of PNDM is about 40%.
Although patients with TNDM require insulin therapy at the time of onset because of marked hyperglycemia, they can be weaned from insulin therapy at an average of 3 mo after the start of treatment[13-16]. This is called the remission period. However, in about half of patients, diabetes mellitus relapses from childhood to adolescence[14,17]. In 70% of patients with TNDM, the cause is overexpression of an imprinted gene PLAGL1 which is located in the chromosome 6q24 region and is expressed from paternal allele (6q24-TNDM)[14,15,18,19]. In 25% of patients with TNDM, mutations of KCNJ11 and ABCC8 genes which encode the ATP-sensitive potassium channel (KATP channel) essential for glucose-stimulated insulin secretion have been identified (KATP-TNDM)[14,20,21]. 6q24-TNDM has the following characteristics: (1) it often develops within 1 wk after birth; (2) it is often diagnosed asymptomatically on routine blood collection; and (3) it is rarely accompanied by ketoacidosis[15,19]. On the other hand, the time of diagnosis of KATP-TNDM is 1 to 4 mo after birth, which is later than that of 6q24-TNDM[14].
The main causes of PNDM are KATP channel abnormality [KCNJ11 gene (31%); ABCC8 gene (10%)] and insulin gene mutations (12%); the median age at the time of diagnosis is 8 wk after birth and 10 wk after birth, respectively[9]. In contrast to TNDM, PNDM shows symptoms such as dehydration, poor sucking, and poor weight gain at the time of onset and is often accompanied by ketoacidosis[15,19]. A large proportion of other causative genes are expressed by autosomal recessive inheritance and account for about 10% of PNDM. In about 35% of patients with NDM, causative genes have not been identified[7].
Insulin therapy is required at the time of onset of NDM regardless of disease type in order to improve metabolic abnormality and weight increase[22]. It has been reported that because neonates have a small body and then receive a small dose of insulin, excellent glycemic control is achieved by an insulin pump which is capable of fine regulation[23-25]. As a treatment after withdrawal from the acute phase, a switch to high-dose administration of sulfonylurea (SU) drugs is an effective causal therapy for KATP channel abnormality; in not less than 90% of patients, a dramatic improvement of glycemic control is observed immediately without hypoglycemia and is maintained for a long period[26-28]. Therefore, when NDM is diagnosed, it is important to determine by gene analysis whether or not KATP channel abnormality is present. Early diagnosis makes it possible to switch to SU drugs during infancy, resulting in an extremely high quality-of-life[29-32].
1,5-ANHYDROGLUCITOL IN NEONATES
1,5-AG is a polyol with a structure in which hydroxyl at the 1st position of glucose is reduced; 1,5-AG is contained in a wide variety of food, but is hardly metabolized in the body[33]. Therefore, after being absorbed from the intestine, 1,5-AG contained in food is widely distributed in various organs to form an internal pool. The amount of 1,5-AG supplied from daily food intake is smaller than the internal pool, and so there is no change in serum 1,5-AG concentration before and after meal. Excessive intake of 1,5-AG is excreted in urine.
Usually, about 180 g of glucose is excreted daily from glomeruli; about 100% of the excreted glucose is reabsorbed by sodium glucose cotransporter 2 (SGLT2), which is located in proximal renal tubules and are specific to glucose[34], and SGTL1, which is located downstream of SGLT2. After the onset of diabetes mellitus, excretion of glucose will increase; when the increased excretion of glucose exceeds the reabsorption capacity of SGLT2 and SGLT1, reabsorption of glucose via 1,5-AG/mannose/fructose cotransporter (SGLT4), which is located downstream of SGLT2 and SGLT1, will start. Because glucose is usually not present, 99.9% of 1,5-AG is reabsorbed by SGLT4; however, this reabsorption mechanism is common to glucose; therefore, if inflow of glucose into tubules increases, reabsorption of 1,5-AG will be inhibited[35-37]. Therefore, in a hyperglycemic condition, excretion of 1,5-AG into urine will increase and serum 1,5-AG will decrease. Thus, serum 1,5-AG is a glycemic control indicator which reflects the degree of urinary glucose excretion.
Because serum 1,5-AG increases and decreases by excretion of urinary glucose, serum 1,5-AG reflects short-term changes in glycemic control more subtly than HbA1c. When glycemic control has worsened rapidly, serum 1,5-AG will decrease rapidly because the increased excretion of a large amount of glucose will inhibit reabsorption of 1,5-AG via SGLT4. In patients with marked hyperglycemia and a high excretion of urinary glucose, serum 1,5-AG will not increase in a short period even if glycemic control has improved rapidly because the internal pool of 1,5-AG has decreased.
Serum 1,5-AG is also affected by the threshold for urinary glucose excretion, and therefore shows a low level in renal glycosuria in which the threshold decreases. In addition, serum 1,5-AG shows an abnormally low level in conditions such as chronic renal failure in which reabsorption of 1,5-AG decreases[38-40], pregnancy[41], oxyhyperglycemia in which urinary glucose is observed transiently[42], patients receiving long-term hyperalimentation[43], and liver cirrhosis[44,45]. One of the causes of an abnormally high level of 1,5-AG is oral administration of a kind of Chinese medicines such as Ninjin-yoei-to and Kami-kihi-to which contain large amounts of 1,5-AG[46].
It is known that serum 1,5-AG during the neonatal period shows an apparently low level[47]. This is considered to be due to a small intake of 1,5-AG during the neonatal period. We reported that serum 1,5-AG is significantly lower in subjects with a habit of consuming dairy products than in subjects without such a habit[48]. The fact that breast milk or formula which contains galactose is the main source of nutrition during the neonatal period may be related to a low level of serum 1,5-AG in neonates.
FRUCTOSAMINE IN NEONATES
Protein undergoes glycation reaction in accordance with plasma glucose concentration, and ketoamine, an early Maillard reaction product, is produced via aldimine. Because the side chain binding of ketoamine takes a fructose structure, ketoamine is generically named fructosamine. Fructosamine is measured using the property that fructose-lysine (fructosamine), in which glucose is bound to the lysine residues of protein, has reducing ability under alkaline conditions. A large proportion of measurements are made by the chemical method; measurements are made by colorimetric determination by producing reduction color reaction using nitroblue tetrazolium (NBT) as a chromogen. Because 60% to 70% of serum protein is albumin, the main component of fructosamine is glycated albumin, but fructosamine contains glycated lipoprotein and glycated globulin as well. Fructosamine is not affected by anemia or variant hemoglobin. In addition, because the turnover of albumin, which accounts for the most part of serum protein, is faster than that of hemoglobin, it is possible to evaluate short-term glycemic control by measuring fructosamine[49]. A low fructosamine level is observed in hyperthyroidism[50,51] and nephrotic syndrome[52] in which protein (albumin) metabolism is accelerated; a high fructosamine level is observed in hypothyroidism[50,51] in which protein (albumin) metabolism is prolonged.
HbA1c and GA are glycation products of hemoglobin and albumin (single proteins), respectively, whereas fructosamine is the generic name of all glycated proteins and lacks specificity. Because albumin accounts for 60% to 70% of serum protein, fructosamine has similar properties to GA; however, there is a problem that because other glycated proteins are measured as well, a high fructosamine level is observed in myeloma[53]. Because HbA1c and GA are expressed as the ratio of hemoglobin and the ratio of albumin, respectively, they are not affected by dilution of serum; on the other hand, because fructosamine is expressed as reducing ability per 1 mL of serum, it is affected by serum protein concentration, and an apparently low level of fructosamine is observed in dilutional anemia. The level of fructosamine in young children is lower than that in adults[54], which is also partly due to low serum protein concentration. Because fructosamine is measured by colorimetric determination based on reduction color reaction, fructosamine is affected by bilirubin with reducing ability, etc. It is considered that the effects of ascorbic acid and vitamin E are mild; however, if a large amount of ascorbic acid or vitamin E is consumed, measurement of fructosamine may be affected.
GLYCEMIC CONTROL INDICATORS OF CORD BLOOD
The composition of hemoglobin in healthy adults is as follows: adult hemoglobin (HbA): 97%; HbA2: 2.5%; HbF: 0.5%[55]. On the other hand, HbF accounts for 80% to 90%, and HbA accounts for only 10% to 20% immediately after birth. After then, HbF decreases logarithmically and is replaced by HbA; by 6 mo after birth, the largest proportion of Hb is HbA; however, it is not until 1 year after birth when the proportion of HbF decreases to less than 1% (level of HbF in adults)[56,57]. Therefore, it is difficult to use the cation exchange high-performance liquid chromatography (HPLC) method, the immunological (latex immunoturbidimetry; LA) method, and the enzyme method which specifically measure HbA1c as glycemic control indicators in NDM.
We measured glycohemoglobin (GHb) in cord blood by various methods[58]. GHb measured by the HPLC method was less than the detection limit when Arkray’s HA-8180 was used and was as low as 1.8% ± 0.2% when Tosoh’s G8 was used. GHb measured by the LA method was less than the detection limit; HbA1c measured by the enzyme method was 1.1% ± 0.3%. Because these methods for measuring GHb measure HbA1c specifically and do not measure glycated HbF, the result is less than sensitivity or a very low level, and it was confirmed that these methods cannot be used as glycemic control indicators in NDM.
It is considered that measurement of GHb by the affinity method using boronic acid may be used as a glycemic control indicator during the neonatal period as well because it measures all glycated hemoglobins[59,60]. It has been reported that GHb in cord blood is higher in patients whose mother has diabetes mellitus than in patients whose mother does not have diabetes mellitus[61-63]. Our investigation revealed that GHb was 3.9% ± 0.2%, which was slightly lower than the reference value for adults (4.6% to 6.2%)[58]. Plasma glucose in cord blood was normal (94 ± 27 mg/dL); therefore, it is considered that the low GHb levels were due to shortened life span of red blood cells[64].
GA in cord blood was 9.4% ± 1.1%, which was slightly lower than the reference value for adults (11.6% to 16.2%)[58]. We demonstrated that low GA levels are observed in neonates because albumin metabolism in neonates is accelerated[65,66]. Low GA levels in cord blood are considered to be due to accelerated metabolism of albumin.
The level of 1,5-AG in cord blood measured in pregnant women including those with diabetes mellitus was similar to that in maternal blood at the time of delivery[67]. This finding was considered to be due to the fact that 1,5-AG in maternal blood was distributed in the fetus via the placenta.
The above results show that both GHb measured by the affinity method and GA were slightly lower than the reference value for adults, but could be used as glycemic control indicators in NDM. On the other hand, HbA1c measured by the HPLC method, the LA method, or the enzyme method and 1,5-AG cannot be used as glycemic control indicators.
GLYCEMIC CONTROL INDICATORS IN NDM: HBA1C AND GA
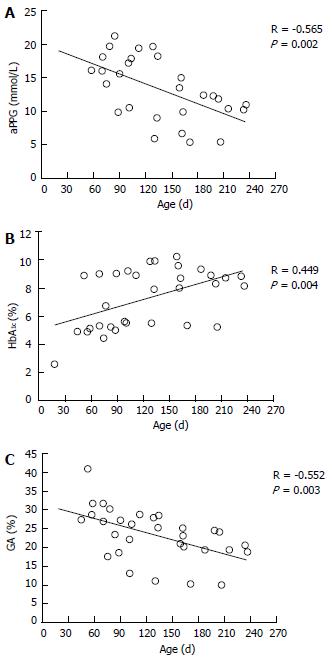
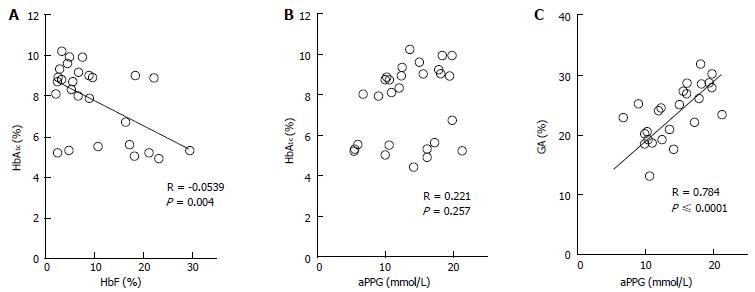
The etiologic diagnosis and treatment of NDM have been making rapid progress; however, there have been few studies on glycemic control indicators useful for evaluating the diagnosis of NDM and effects of therapy. Therefore, we hypothesized that GA is a useful glycemic control indicator in NDM[58] and conducted an investigation[6]. We found that GA, as a glycemic control indicator in NDM, has various advantages: (1) GA is not affected by HbF; (2) unlike fructosamine, GA is not affected by serum protein (albumin) because it is expressed as a ratio to albumin; (3) unlike fructosamine, GA is not affected by other proteins and has a high specificity because it reflects glycation products of a single protein (albumin); (4) GA reflects plasma glucose during a shorter period than HbA1c; and (5) HbA1c reflects mean plasma glucose, whereas GA reflects fluctuation of plasma glucose (postprandial hyperglycemia) in addition to mean plasma glucose[68-70]. HbA1c (%) is expressed as HbA1c/total Hb; therefore, if HbF is high, a relatively low HbA1c level will be observed. At the time of onset of NDM (mostly 1 to 2 mo after birth), a large amount of HbF remains in blood; therefore, a lower HbA1c level is observed in relation to plasma glucose level. In addition, it is estimated that during infancy, during which HbA increases, if plasma glucose level is constant, HbA1c will increase. In fact, in an investigation of five patients with NDM (age at the time of diagnosis: 38 ± 20 d), plasma glucose was markedly high [29.7 ± 13.1 mmol/L (535 ± 236 mg/dL)], whereas HbA1c measured by the HPLC method was within the normal range (5.4% ± 2.6%)[6]. As the course of treatment progressed, plasma glucose tended to decrease (Figure 1A), whereas HbA1c tended to increase (Figure 1B). A significant negative correlation was observed between HbA1c and HbF (Figure 2A), whereas no significant correlation was observed between HbA1c and plasma glucose level (Figure 2B). On the other hand, GA at the time of diagnosis was abnormally high (33.3% ± 6.9%)[6]. In contrast to HbA1c, GA decreased as treatment progressed (Figure 1C) and showed a strong positive correlation with plasma glucose level (Figure 2C). Thus, it was found that GA, but not HbA1c, is an appropriate glycemic control indicator in NDM.
Figure 1 Time course of average preprandial plasma glucose for 1 mo (A), HbA1c (B), and glycated albumin (C) according to treatment in 5 patients with neonatal diabetes mellitus (modified from Ref[6], with permission from Copyright Clearance Center Inc.
).
Figure 2 Correlations between HbA1c and HbF (A) and between HbA1c and average preprandial plasma glucose for 1 mo (B) and correlation between glycated albumin and average preprandial plasma glucose in 5 patients with neonatal diabetes mellitus (C) (modified from Ref[6], with permission from Copyright Clearance Center Inc.
).
From what age can HbA1c be used as a glycemic control indicator? Alternatively, if the effect of HbF is excluded or if a different principle of measurement is employed, might HbA1c be an appropriate indicator? And when using GA as a glycemic control indicator in NDM, what should be taken into account? In the following chapters, we will discuss these issues in relation to the current status and challenges in infants and NDM.
HBA1C IN NEONATES AND NDM
As mentioned above, when HbA1c is expressed as HbA1c/total Hb, it cannot be used as a glycemic control indicator in NDM. There are two ways to eliminate the effect of HbF. One way is to determine the HbA1c level corrected by HbF (HbF corrected HbA1c) by the formula: HbA1c/(total Hb-HbF), resulting in the correction of an apparently low HbA level. The other way is to determine GHb relative to all hemoglobins including HbF and to use this as a glycemic control indicator. For the latter, it is possible to measure all GHb by the affinity method[71].
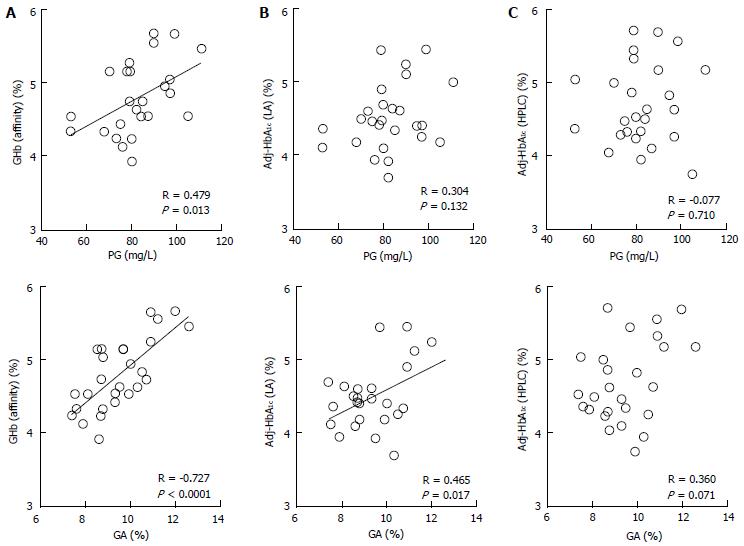
We measured HbA1c by the HPLC method and the LA method in 26 healthy infants (0 to 8 mo old), calculated HbA1c values corrected by HbF [Adj-HbA1c (HPLC) and Adj-HbA1c (LA), respectively], measured GHb by the affinity method [GHb (Affinity)], and evaluated correlations between these values and plasma glucose and between these values and GA[72]. As a result, only GHb (Affinity) had a significant correlation with both plasma glucose and GA (Figure 3A). Adj-HbA1c (LA) was correlated only with GA (Figure 3B); Adj-HbA1c (HPLC) was not correlated with either plasma glucose or GA (Figure 3C). These results suggest that GHb (Affinity) may be used as a glycemic control indicator in NDM. In this research, however, GHb (Affinity) within one month was lower than the reference range of HbA1c during 8 to 12 mo (4.8% to 6.0%)[73], and a large proportion of GHb values from 1 to 5 mo were lower than the reference range. The following three factors are thought to contribute together to this finding. The first factor is the effect of a low plasma glucose level during infancy, especially within one month after birth[65,74]. The second factor is the short half-life of red blood cells (about 90 d) during infancy[64]. The third factor is the glycation rate of HbF which is considered to be lower than that of HbA. In this regard, Little et al[75] reported that GHb measured by the affinity method is low when a sample which contains not less than 15% of HbF is used. In the LA method, HbA1c is measured using antibodies which specifically recognize peptides including glycated valine of hemoglobin β-chain N-terminal[76]. Theoretically, when interpreting Adj-HbA1c (LA) levels, it is necessary to consider a low plasma glucose level and shortened half-life of red blood cells of the infant; however, it is considered that Adj-HbA1c (LA) may be used as a glycemic control indicator; in fact, a correlation between Adj-HbA1c (LA) and GA was observed. However, the LA method is too complicated to be used in clinical settings because it is necessary to measure HbF using the HPLC method. In addition, our investigation revealed that Adj-HbA1c (HPLC) is not an appropriate indicator for the evaluation of HbA1c in infants. In the HPLC analysis, HbF and HbA1c migrate to adjacent locations. When a high HbF level is observed, separation of HbF and HbA1c becomes insufficient and so HbA1c cannot be measured correctly, which is considered to be one of the causes of the above-mentioned phenomenon. On the other hand, Little et al[75] and Rohlfing et al[77] reported on HbF-corrected HbA1c as follows: if HbF is not more than 30%, HbA1c measured by the HPLC method using Tosoh’s G7 and G8 can be used as a glycemic control indicator. However, they did not use samples which contained 30% or more of HbF, and they did not state whether or not Hb in the samples used was derived from infants; therefore, these facts may be the reason for the difference from our data obtained from samples of infants.
Figure 3 Correlations between glycated hemoglobin measure by various methods and plasma glucose or glycated albumin.
Correlations between GHb measured by the affinity method [GHb (affinity)] (A), HbF-adjusted HbA1c measured by the immunological method [Adj-HbA1c (LA)] (B), and HbF-adjusted HbA1c measured by the HPLC method [Adj-HbA1c (HPLC)] (C), and PG or GA in 26 healthy infants were shown (modified from Ref[72], with permission from Copyright Clearance Center Inc.). GA: Glycated albumin; PG: Plasma glucose; GHb: Glycated hemoglobin; HbF: Fetal hemoglobin.
So far, there have been no studies on the age at which HbA1c can be used for patients with NDM, and so research is needed to clarify the relationship between mean plasma glucose and HbA1c and between CGM and HbA1c. Regarding the reference value of HbA1c in healthy infants, there is only a report by Jansen et al[73] who investigated 100 healthy infants of 8 to 12 mo old. In that report, the reference value of HbA1c for infants was 4.8% to 6.0%, which was similar to the reference value of HbA1c for adults (4.6% to 6.2%). From our results, HbA1c levels in most infants of 6 mo of age or older were also within the reference range shown by Jansen et al[73] HbF decreases to less than 5% by 6 mo after birth[56,57]; therefore, it is considered possible to use HbA1c as a glycemic control indicator in patients with NDM of 6 mo of age or older.
GA IN NEONATES AND NDM
GA is a useful glycemic control indicator under conditions in which hemoglobin metabolism is affected. On the other hand, abnormal albumin metabolism affects GA. It has been reported under various conditions that GA shows a low level when albumin metabolism is accelerated and shows a high level when albumin metabolism is suppressed[78].
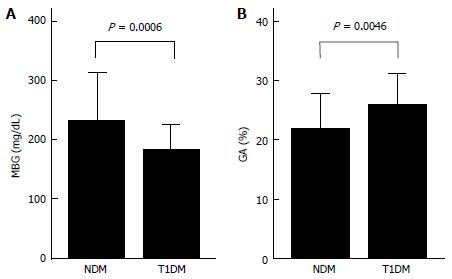
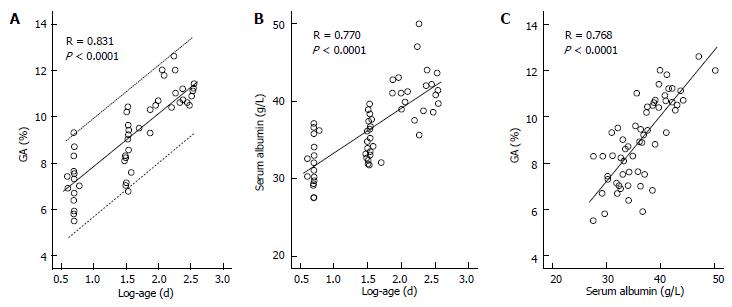
While GA is a useful glycemic control indicator in patients with NDM, it is necessary to keep in mind the following characteristics of GA during infancy: (1) it shows a lower level in relation to plasma glucose; and (2) it shows a positive correlation with logarithmically transformed age[65,66]. For GA in healthy infants, before the currently widely used enzyme method was developed[79], it had already been reported that GA measured by the HPLC method was lower than the reference value for adults[54]. It is known that protein metabolism is accelerated during infancy[80,81]. In addition, it has been reported that albumin synthesis is accelerated as well[82]. Therefore, acceleration of albumin metabolism may contribute to a low GA level during infancy. We compared the relationship between GA and plasma glucose level in patients with NDM and in patients with juvenile type 1 diabetes mellitus (T1DM), and found that patients with NDM had higher plasma glucose levels but lower GA levels than patients with T1DM (Figure 4); thus, we obtained a result which supports the phenomenon of accelerated metabolism of albumin during infancy[65]. In addition, we investigated in healthy infants the relationship between change in GA according to age and plasma glucose and between change in GA according to age and serum albumin. As a result, a strong positive correlation was observed between GA and logarithmically transformed age in days (Figure 5A), and multivariate analysis revealed that age and serum albumin affect GA levels more significantly than plasma glucose[66]. Because GA is expressed as a percentage relative to serum albumin, it is not affected by serum albumin, which is an advantage of GA over fructosamine[54]. However, an increase in serum albumin associated with aging is observed during infancy (Figure 5B) and there is a positive correlation between GA and serum albumin during this period (Figure 5C)[66]. Accordingly, we determined the reference value of GA in infants according to age in mo from the regression equation of GA and age, and proposed that a comparison between GA level and the reference value[65].
Figure 4 Comparisons of mean blood glucose for 1 mo (A) and GA (B) in 6 patients with neonatal diabetes mellitus and in 18 patients with type 1 diabetes mellitus (modified from Reference[65], with permission from Copyright Clearance Center Inc.
).
Figure 5 Correlations between glycated albumin and age and between serum albumin and age and correlation between glycated albumin and serum albumin in healthy infants.
A: Correlation between glycated albumin (GA) and log-age. The dotted line shows the 95%CI; B: Correlation between serum albumin and log-age; C: Correlation between GA and serum albumin (modified from Ref[66], with permission from Copyright Clearance Center Inc.).
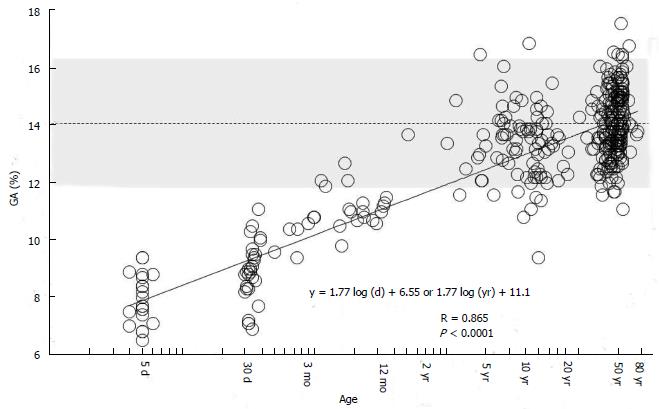
On the other hand, we found that regardless of age, GA can be evaluated based on the reference value for adults without using the reference value for infants by determining age adjusted GA (Aa-GA)[83]. We investigated GA in 376 subjects without diabetes mellitus of a wide range of age (neonates, children, and adults), and found that GA can be expressed as a primary regression equation of logarithmically transformed age (Figure 6). Based on this equation, the following formula for calculating Aa-GA was derived: Aa-GA = GA × 14.0 /[1.77 × log-age (d) + 6.55] or Aa-GA = GA × 14.0 /[1.77 × log-age (yr) + 11.1]. As mentioned above, GA in NDM shows an apparently low level; therefore, if GA in NDM is compared with the reference value for adults, the glycemic control status may be underestimated. By calculating Aa-GA and comparing it with the reference value for adults, it is possible to accurately evaluate the glycemic control status in NDM. The advantages of evaluating Aa-GA by the reference value for adults instead of evaluating GA by the reference value for infants according to age in month are as follows: (1) it is not necessary to consider the reference value according to age in month; and (2) regardless of age, it is possible to make comparisons of longitudinal changes in glycemic control status.
Figure 6 Correlation between glycated albumin and age in days (logarithmic transformation) in 376 healthy subjects (age: 4 d to 78 years).
The dotted line indicates the mean reference value for adults (14%), and the shading indicates the range of reference values for adults (11.7% to 16.2%) (modified from Ref[83], with permission from Royal Society of Medicine).
It is known that because the half-life of GA is shorter than that of HbA1c, GA reflects short-term plasma glucose correctly[84,85]. This characteristic also indicates the usefulness of GA as a glycemic control indicator in NDM. Because a large proportion of NDM develops within one month, the duration of the hyperglycemic status is short. This form of development is similar to that of fulminant type 1 diabetes mellitus[86]. In fulminant type 1 diabetes mellitus, pancreatic beta cells are destroyed in a very short period, and ketoacidosis develops shortly after the onset of diabetic symptoms. Therefore, at the time of onset, HbA1c is normal or only slightly high, but GA is already obviously high[87]. We reported that GA at the time of onset of NDM was abnormally high (33.6 ± 6.9%) in all patients[6], and an abnormally high GA level in NDM may be useful for differential diagnosis from transient hyperglycemia. In addition, when evaluating remission of patients with TNDM and when evaluating the effect of SU drugs administered to patients with PNDM, it will be possible to promptly evaluate an improvement of such glycemic control by using GA[5].
CONCLUSION
The usefulness of GA as a glycemic control indicator in NDM was demonstrated. However, it was found that GA is affected by albumin metabolism and shows an apparently low level. Therefore, it is necessary to compare GA with the reference value according to age or to calculate age-adjusted GA (Aa-GA). On the other hand, HbA1c measured by the HPLC method, the LA method, or the enzyme method does not correctly reflect the glycemic control status because it is affected by a high HbF level. GHb measured by the affinity method reflects the glycemic control status in NDM; however, this method is currently hardly used and cannot easily measure GHb routinely. In addition, it is unknown whether the kinetics of glycation reaction of HbF are similar to those of HbA. Taking into account such circumstances, it is desirable to select GA as a glycemic control indicator for patients with NDM and to evaluate the glycemic control status using Aa-GA.