Published online Feb 15, 2025. doi: 10.4239/wjd.v16.i2.97287
Revised: October 13, 2024
Accepted: November 22, 2024
Published online: February 15, 2025
Processing time: 216 Days and 18.8 Hours
Macrophages are central to the orchestration of immune responses, inflammatory processes, and the pathogenesis of diabetic complications. The dynamic pola
To investigate the inhibitory effect of dapagliflozin on macrophage M1 pola
We established a murine model of type 2 diabetes mellitus and harvested pe
Dapagliflozin attenuated M1 macrophage polarization and mitigated apoptosis in the abdominal macrophages of diabetic mice, evidenced by the downregulation of proapoptotic genes (Caspase 3), inflammatory cytokines [in
Dapagliflozin attenuates the polarization of macrophages toward the M1 phenotype, thereby mitigating inflammation and promoting macrophage apoptosis. These effects are likely mediated through the inhibition of the PI3K/AKT signaling pathway.
Core Tip: Sodium-glucose cotransporter-2 inhibitors, a new type of hypoglycemic drug, are widely used in clinical practice. However, their specific protective mechanism in diabetic kidneys has not been fully elucidated. We induced macrophage polarization and apoptosis in a mouse model of type 2 diabetes mellitus in vitro and treated it with dapagliflozin. Ultimately, our study revealed that dapagliflozin attenuates macrophage M1 polarization and apoptosis, reduces inflammatory responses, thereby protecting the kidney, and is associated with the inhibition of the phosphoinositide 3-kinase/protein kinase B signaling pathway.
- Citation: Xiong SX, Huang LJ, Liu HS, Zhang XX, Li M, Cui YB, Shao C, Hu XL. Dapagliflozin exerts anti-apoptotic effects by mitigating macrophage polarization via modulation of the phosphoinositide 3-kinase/protein kinase B signaling pathway. World J Diabetes 2025; 16(2): 97287
- URL: https://www.wjgnet.com/1948-9358/full/v16/i2/97287.htm
- DOI: https://dx.doi.org/10.4239/wjd.v16.i2.97287
Sodium-glucose cotransporter (SGLT)-2 inhibitors such as dapagliflozin and empagliflozin represent novel therapeutic options against type 2 diabetes mellitus (T2DM). By curbing renal glucose reabsorption and fostering urinary glucose excretion, SGLT-2 inhibitors effectively lower circulating plasma glucose levels[1]. The clinical benefits of these drugs stem from their remarkable cardiovascular benefits, which transcend diabetic status[2]. The mechanism by which SGLT-2 inhibitors specifically ameliorate diabetic nephropathy is not fully understood.
Could SGLT-2 inhibitors orchestrate macrophage polarization? Recent reports suggest the modulatory effects of SGLT-2 inhibitors on macrophage phenotypic transitions[3]. Macrophages are pivotal constituents within the innate immune system, orchestrating host defense, inflammation, and tissue equilibrium[4]. Macrophages possess remarkable plasticity to recalibrate their phenotypes and functionalities in response to dynamic microenvironmental cues-a phenomenon termed polarization. When broadly delineated into classically activated (M1-type) and alternatively activated (M2-type) macrophages, these two groups wield opposing cytokine repertoires[5]: M1-type macrophages propagate inflammation via proinflammatory cytokines, and M2-type macrophages mitigate inflammatory cascades through inhibitory cytokines[5]. The heterogeneous landscape of pro- and anti-inflammatory macrophages enables the elucidation of the mechanisms of chronic inflammation. Importantly, macrophage polarization has emerged as a linchpin in the pathogenesis of insulin resistance, the onset and progression of which are heralded by the shift in the M1/M2 ratio[6]. Targeting M1-type macrophages, arbiters of the proinflammatory milieu, offers a promising avenue for combating insulin resistance[7]. In addition, diabetic nephropathy (DN) is a serious microvascular complication. There is increasing evidence that the levels of proinflammatory factors increase with the development of DN[8]. The differentiation of M2-type macrophages reduces systemic and local renal inflammation in DN[9]. Consequently, macrophage polarization is highly important in the ongoing battle against DN. Recently, the renoprotective effect of SGLT-2 inhibitors on DN was shown to be achieved by attenuating renal oxidative stress and inflammatory responses[10]. Therefore, we hypothesized that the renoprotective effect of SGLT-2 inhibitors on DN is mediated by the modulation of macrophage polarization.
Notably, the nexus between macrophage apoptosis and T2DM further highlights this intricate interplay[11]. Recent investigations underscore the pivotal roles of lipid peroxidation and the inflammatory milieu in instigating macrophage apoptosis[12]. Concurrently, macrophage polarization, a key component of persistent systemic inflammation, has emerged as a putative mediator of macrophage apoptosis. Furthermore, divergent outcomes of apoptosis across different cell types have been observed with SGLT-2 inhibitors[13,14], but their potential impact on macrophage apoptosis remains uncharted.
Thus, we postulate that SGLT-2 inhibitors might inhibit macrophage M1 polarization, thereby ameliorating macro
The human monocyte cell line (THP-1) was procured from the Chinese Academy of Sciences in Shanghai, China. THP-1 cells were nurtured in RMPI-1640 medium enriched with 10% fetal bovine serum.
Specific pathogen free-grade C57BL/6J mice (male, 6 weeks old, sourced from Hangzhou Ziyuan Experimental Animal Science and Technology Co., Ltd.) were meticulously chosen for experimental endeavors, with a regimen of four mice per cage. Following a week-long acclimatization period, eight mice were arbitrarily allocated to a regimen rich in high-sugar and high-fat sustenance, whereas the control cohort received a basal diet. T2DM was provoked through the weekly intraperitoneal administration of streptozotocin (60 mg/kg) overnight after four weeks of dietary intervention. Non
A total of 4-5 mL of RMPI-1640 medium was instilled into the peritoneal cavity of each mouse, and after lavage, the medium was collected. The resulting cell suspension was subjected to two rounds of centrifugation followed by resus
THP-1 cells were pretreated with various concentrations of dapagliflozin (12.5, 25, 50, 100, 200, or 400 μmol/L) for 12 or 24 hours. To determine the optimal concentration of dapagliflozin, cell viability was assessed in a cell counting kit 8 assay. The cell supernatants were subsequently harvested, combined with kit reagents, and incubated at ambient temperature according to the manufacturer’s specifications (Biyuntian Institute of Biotechnology). The resulting absorbance at 450 nm was then quantified with an enzymatic marker.
The cell populations were harvested, subjected to dual washing, and subsequently resuspended at a concentration of 1 × 106 cells/mL. Next, the cells (0.5 × 106) were incubated with 10 μL of Annexin V and 5 μL of propidium iodide (PI) at room temperature and shielded from light for 15 minutes. Flow cytometric analysis was then conducted to scrutinize the fluorescence intensity of Annexin V/PI-stained cells, and apoptosis quantification was carried out using Becton Dickinson fluorescence-activated cell sorting software.
After incubation, the cells were collected into centrifuge tubes and centrifuged at 1000 rpm/minute for 5 minutes. The supernatant was carefully removed, and the cells were subsequently rinsed with phosphate buffered saline (PBS). A fresh aliquot of 100 μL of PBS was added to resuspend the cells. Next, 10 μL of fluorescein isothiocyanate-labeled cluster of differentiation (CD) 86 antibody was added, and the mixture was incubated at 4°C in the dark for 20 minutes. After washing with PBS, the cells were resuspended, and the expression of CD86, a surface molecule on the macrophage membrane, was assessed by flow cytometry.
The cytokine concentrations within the supernatant of each macrophage group were assessed using interleukin (IL)-6, tumor necrosis factor (TNF)-α, and IL-1β assay kits, following the prescribed methodology provided by the manufacturer.
Total cellular RNA was extracted from the cellular milieu using TRIzol reagent, followed by reverse transcription into complementary DNA using a reverse transcription kit (ABclonal Biotechnology, Wuhan, Hubei province, China). Subsequently, quantitative real-time polymerase chain reaction (qRT-PCR) was performed with a qRT-PCR kit from the same source. Amplification proceeded through 40 cycles at 95°C for 30 seconds, followed by denaturation at 95°C for 5 seconds and annealing/extension at 60°C for 30 seconds. The quantification of mRNA levels was then standardized relative to the expression of GAPDH mRNA, employing the 2-ΔΔCt method. The primer sequences are listed in Supplementary Table 1.
Protein expression was assessed by protein blotting, employing GAPDH as an internal reference standard. Equivalent amounts (20-40 μg) of protein samples were loaded and subsequently incubated overnight at 4°C with primary antibodies (dilution factor of 1:1000) targeting phosphoinositide 3-kinase (PI3K), protein kinase B (AKT), phosphorylated protein kinase B (P-AKT), and GAPDH. The membranes were subsequently agitated with goat anti-rabbit antibodies (dilution factor of 1:10000) for 2 hours at ambient temperature. The protein bands were then visualized using enhanced chemiluminescence reagents and quantified in ImageJ software.
The data collected are presented as the mean ± SD. Statistical analysis was tested by one-way analysis of variance followed by Student-Newman-Keuls-q test (statistical product and service solutions 26.0, Chicago, IL, United States). A value of P < 0.05 was regarded as statistically significant.
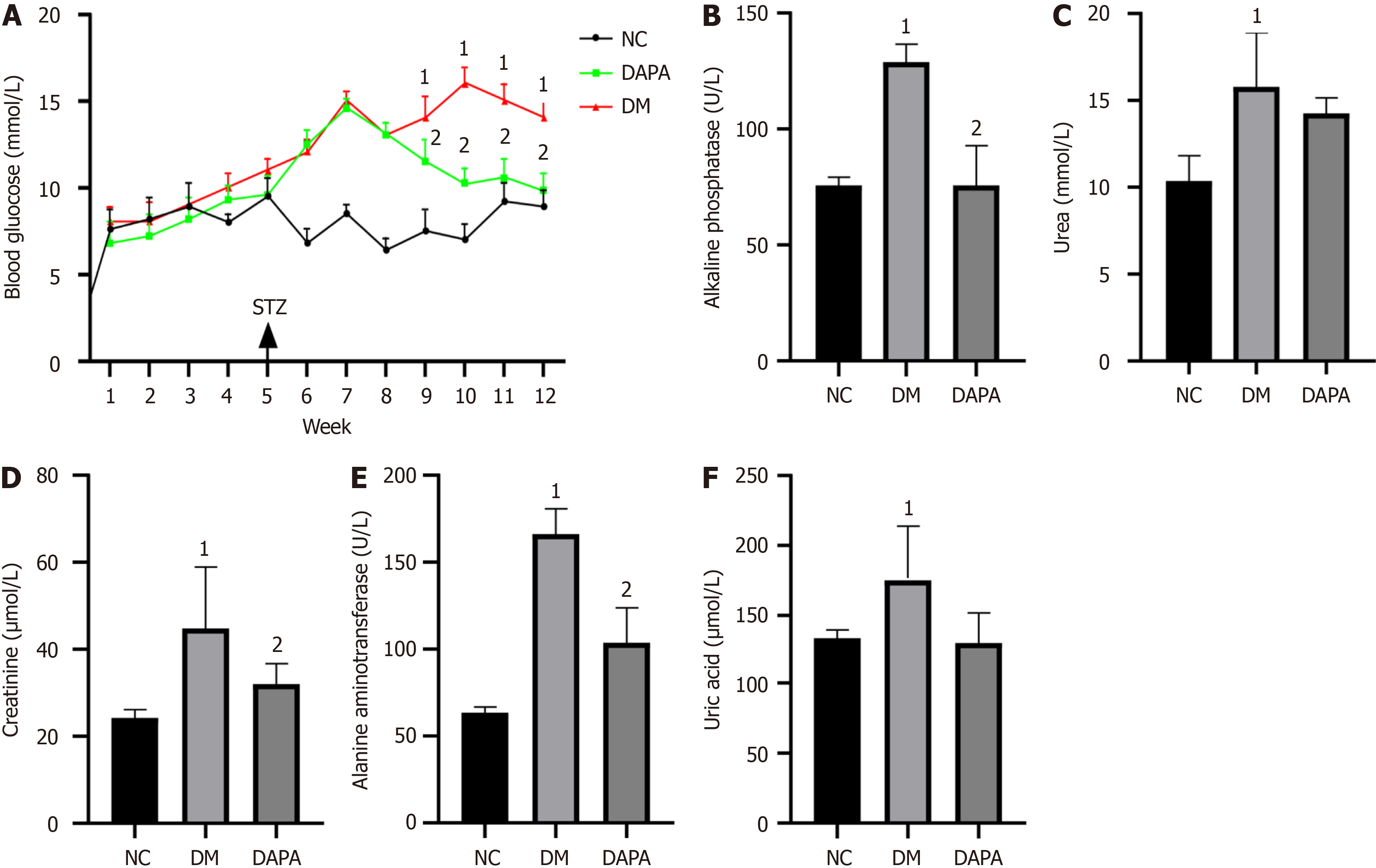
In contrast to their normal counterparts, mice with T2DM presented markedly elevated blood glucose levels, alongside statistically distinct alterations in biochemical parameters such as alanine aminotransferase, alkaline phosphatase, creatinine, uric acid, and urea (P value < 0.05). Following a regimen of dapagliflozin administration for a duration of 2 months, a noteworthy reduction in blood glucose levels was observed relative to those in untreated T2DM-afflicted mice, accompanied by substantial declines in the serum levels of alanine aminotransferase, alkaline phosphatase, and creatinine (P value < 0.05, refer to Figure 1).
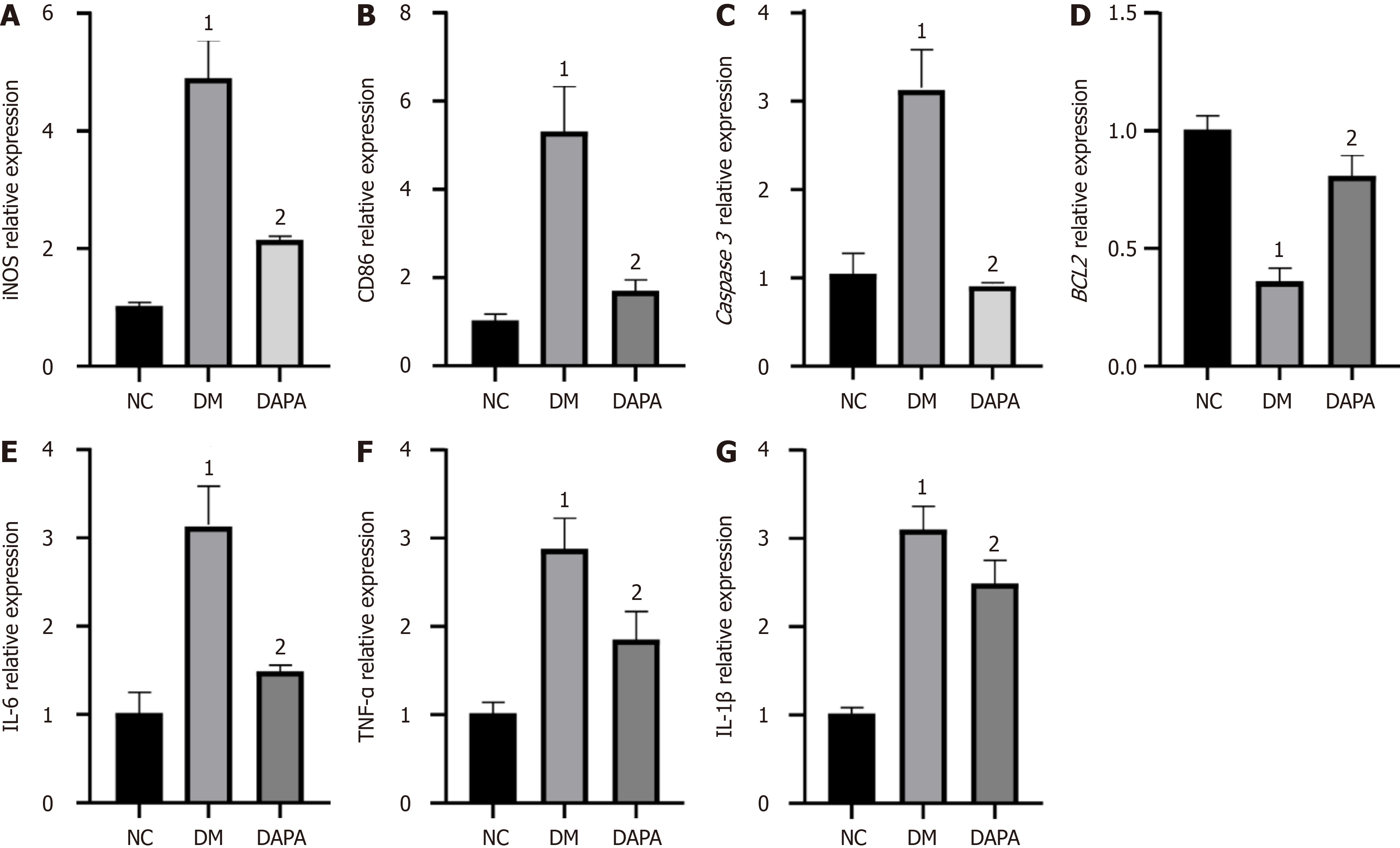
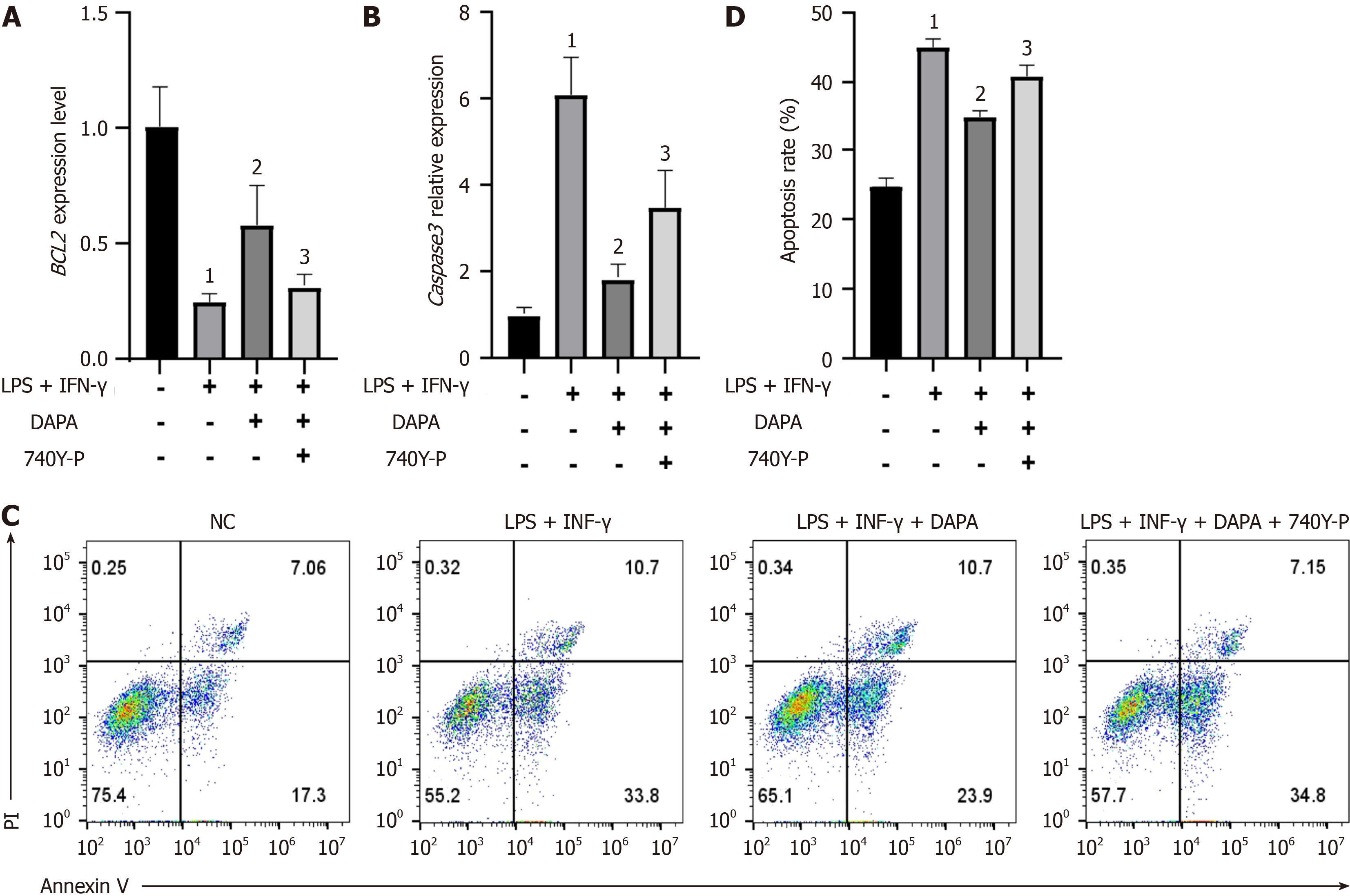
We also revealed notable upregulations of the expression levels of proapoptotic-related factor (Caspase 3), M1-type macrophage surface marker [inducible nitric oxide synthase (iNOS), CD86], and proinflammatory cytokine (IL-6, IL-1β, TNF-α) genes within peritoneal macrophages derived from T2DM-afflicted mice when juxtaposed with those from their normal counterparts. Conversely, the expression levels of antiapoptotic factors (BCL2) significantly decreased (P value < 0.05). Notably, subsequent administration of dapagliflozin inhibited apoptosis, M1 polarization, and inflammatory cytokine expression in the peritoneal macrophages of T2DM-afflicted mice (P value < 0.05, refer to Figure 2).
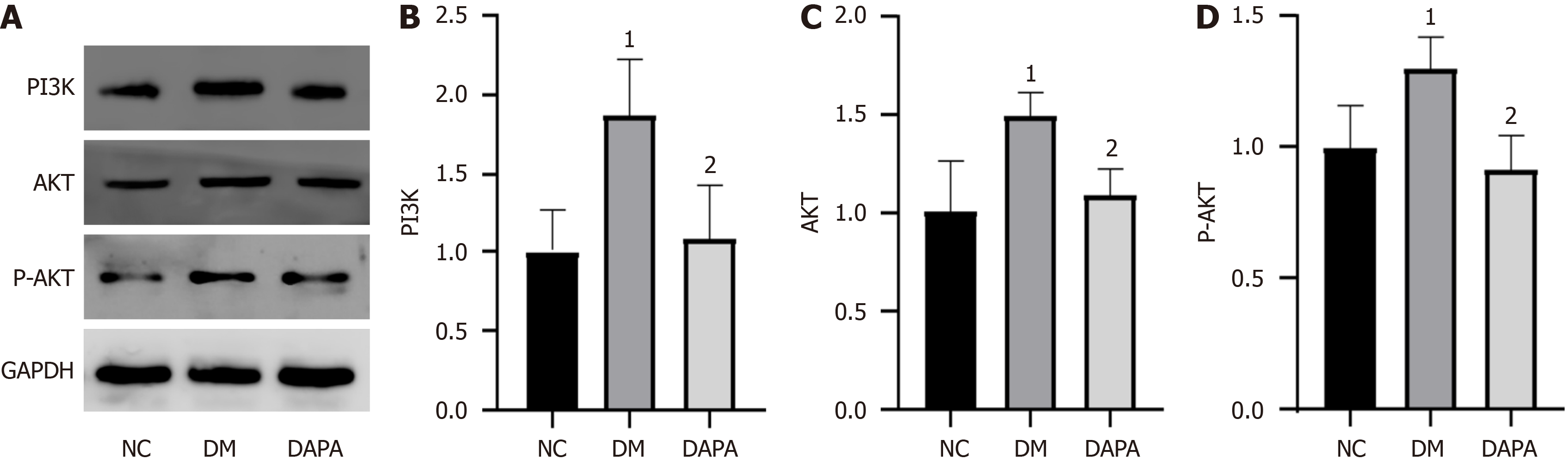
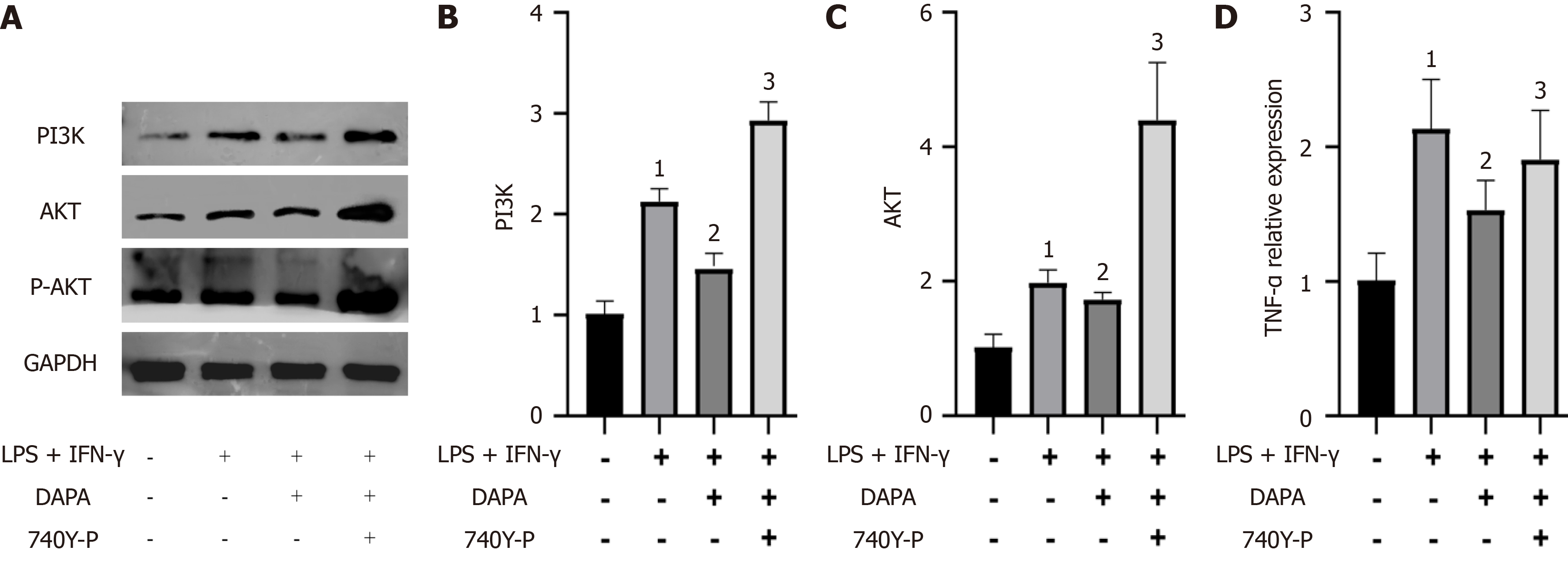
We delved deeper into the potential mechanism underlying the action of dapagliflozin within our experimental paradigm. Our findings revealed substantial inhibition of the protein expression levels of PI3K, AKT, and P-AKT in mouse peritoneal macrophages after in vivo administration of dapagliflozin. In essence, dapagliflozin inhibited the activation of the PI3K/AKT signaling pathway (P value < 0.05, refer to Figure 3).
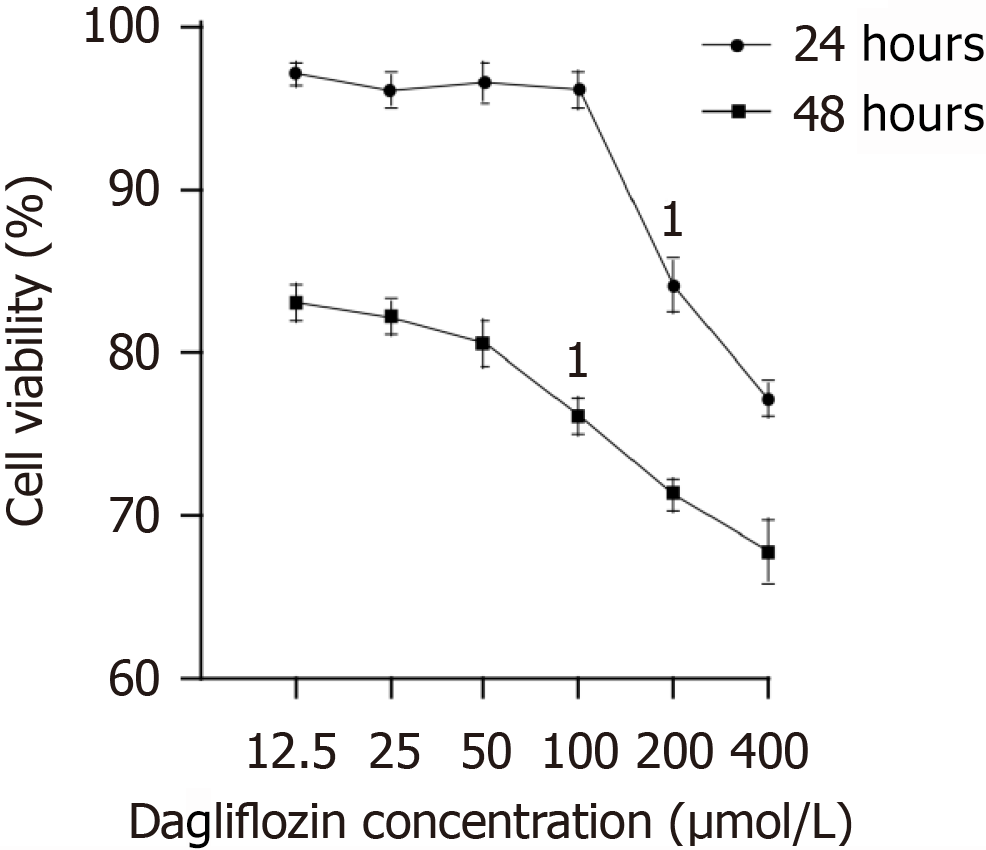
As shown in Figure 4, the viability of macrophages subjected to dapagliflozin treatment at concentrations of 25, 50, and 100 μM for 24 hours exhibited no discernible effect. However, at concentrations of 200 and 400 μM for 24 hours, as well as at all concentrations (25, 50, 100, 200, and 400 μM) for 48 hours, macrophage viability significantly decreased. Guided by these findings, we determined that dapagliflozin at 100 μM for 24 hours was the optimal condition for subsequent experiments (refer to Figure 4).
In contrast to the normal control cohort, THP-1 cells subjected to lipopolysaccharides (LPS) + interferon-γ (IFN-γ) induction exhibited a noteworthy increase in Caspase 3 mRNA expression alongside a reduction in BCL2 mRNA expression. Conversely, in the LPS + IFN-γ + dapagliflozin treatment group, Caspase 3 mRNA expression significantly decreased, whereas BCL2 mRNA expression notably increased compared with the levels in the LPS + IFN-γ group. Following treatment with 740Y-P (a PI3K/AKT signaling pathway agonist), Caspase 3 mRNA expression was once again greater in the 740Y-P-treated group than in the drug-treated group, with a concurrent downregulation of BCL2 mRNA expression; all disparities observed were statistically significant (refer to Figure 5). Apoptosis ratio analysis by flow cytometry further corroborated these findings, confirming the ability of dapagliflozin to prevent THP-1 cell apoptosis, possibly through PI3K/AKT pathway inhibition.
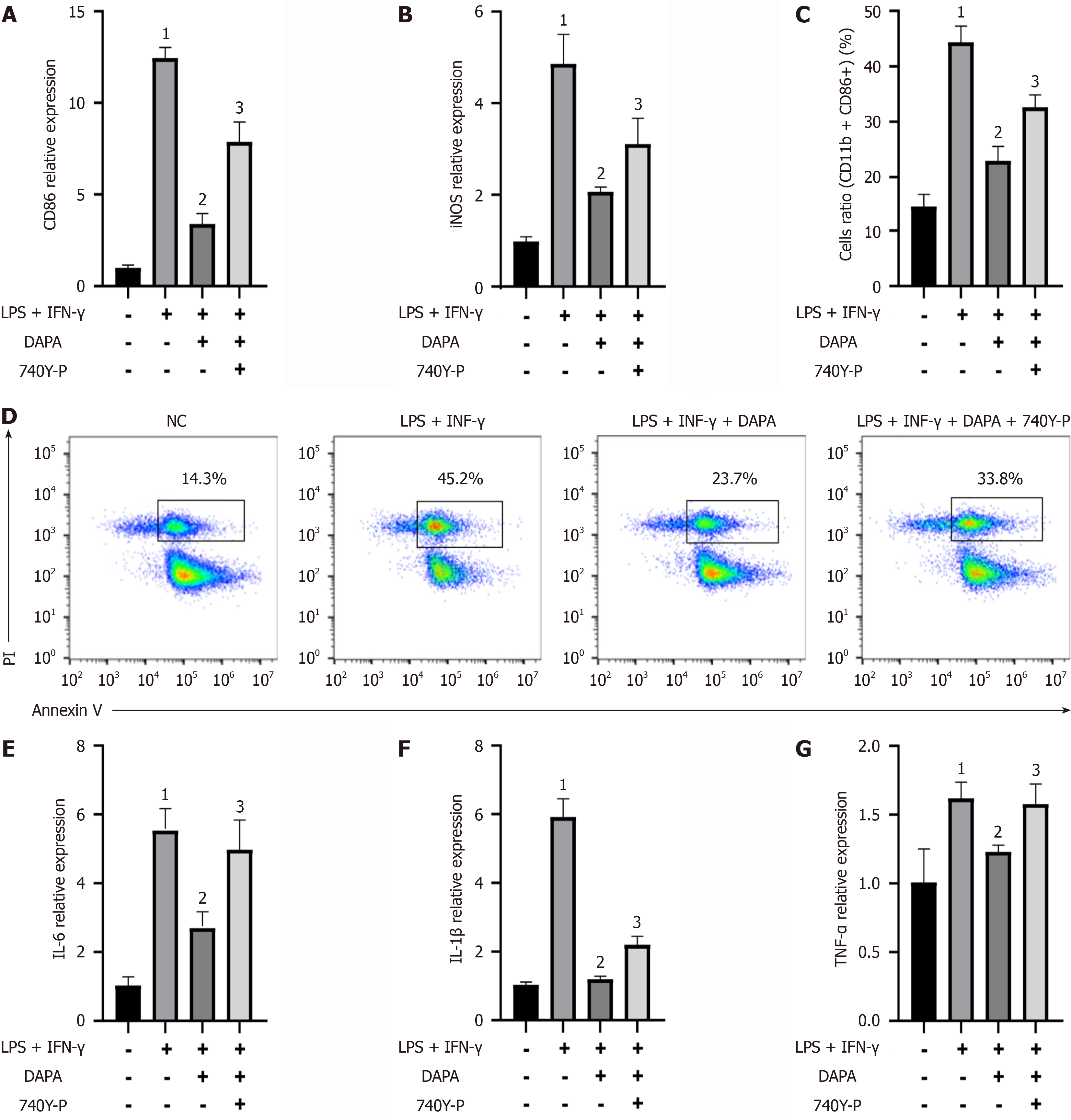
We observed marked increases in the gene expression levels of M1 macrophage surface markers (CD86 and iNOS) and inflammatory factors (IL-6, IL-1β, and TNF-α) in THP-1 cells stimulated with LPS + IFN-γ compared with those in the normal control group. Compared with those in the THP-1 cells alone, the gene expression levels of M1 macrophage surface markers and inflammatory factors in the LPS + IFN-γ group were greater. Conversely, in the IFN-γ + dapagliflozin group, significant reductions in the expression levels of M1 macrophage surface markers and inflammatory factors were observed. However, upon the addition of 740Y-P, the expression levels of these markers and factors rebounded, surpassing those of the LPS + IFN-γ + dapagliflozin group. Similar trends were observed by flow cytometry analysis, in which CD86 expression levels were elevated in the LPS + IFN-γ group compared with those in the normal control group and decreased in the LPS + IFN-γ + dapagliflozin group compared with those in the LPS + IFN-γ group (Figure 6). Furthermore, by enzyme linked immunosorbent assay, we detected alterations in the concentrations of secreted inflammatory factors in each group. Compared with normal control cells, THP-1 cells stimulated with LPS + IFN-γ presented increased secretion levels of IL-6, TNF-α, and IL-1β. These levels were significantly attenuated following dapagliflozin treatment. However, the administration of 740Y-P reversed the effects of dapagliflozin, resulting in a resurgence of inflammatory factor secretion. These findings underscore the potential of dapagliflozin in mitigating the inflammatory response mediated by LPS + IFN-γ, possibly through the inhibition of the PI3K/AKT pathway (refer to Table 1).
| Groups | IL-6 content (pg/mL) | TNF-α content (pg/mL) | IL-1β content (pg/mL) |
| Normal control group | 37.61 ± 17.33 | 64.93 ± 16.13 | 27.96 ± 1.93 |
| LPS + IFN-γ | 196.87 ± 5.041 | 105.01 ± 7.721 | 89.19 ± 3.351 |
| LPS + IFN-γ + dapagliflozin | 62.80 ± 22.122 | 71.08 ± 11.932 | 39.57 ± 3.362 |
| LPS + IFN-γ + dapagliflozin + 740Y-P | 158.54 ± 14.303 | 105.65 ± 7.853 | 53.81 ± 2.143 |
Finally, we investigated the impact of dapagliflozin on the expression levels of members of the PI3K/AKT pathway in THP-1 cells. Our findings revealed significant increases in the expression levels of PI3K, AKT, and P-AKT in THP-1 cells stimulated with LPS + IFN-γ (P value < 0.05). However, dapagliflozin administration notably attenuated the activation of the PI3K/AKT pathway induced by LPS + IFN-γ (P value < 0.05), with the PI3K/AKT agonist 740Y-P effectively counteracting the inhibitory effects of dapagliflozin (P value < 0.05, refer to Figure 7). These results strongly imply that dapagliflozin inhibits the activation of the PI3K/AKT pathway.
Approximately 537 million individuals worldwide are affected by diabetes, with the numbers increasing rapidly, particularly among the younger populace, a trend that is cause for concern[15]. Diabetes and its associated complications pose a grave threat to the well-being and security of all humanity. Reports indicate that nearly 4.2 million people worldwide will succumb to diabetes and its complications in 2019, accounting for 11.3% of all deaths worldwide[16]. Within the patho
The kidney plays a pivotal role in maintaining glucose homeostasis, with SGLT-2 inhibitors emerging as novel therapeutics for T2DM, exemplified by dapagliflozin and empagliflozin[20]. SGLT-2, a low-affinity, high-capacity transporter protein, is exclusively expressed in the proximal tubules of the kidneys and is responsible for reabsorbing 90% of glucose from urine[21]. SGLT-2 inhibitors, by impeding the function of SGLT-2, facilitate the excretion of a substantial quantity of glucose in the urine, thereby effectively lowering blood glucose levels[22]. The glucose-lowering effect of SGLT-2 inhibitors is independent of insulin, thus minimizing the risk of hypoglycemia. Recent studies have demonstrated the renoprotective effects of SGLT-2 inhibitors in patients with T2DM, extending their renoprotective advantages to the general population[23]. This renoprotective mechanism may be attributed to the ability of SGLT-2 inhibitors to curtail glucose and sodium reabsorption, thereby increasing the concentration of sodium ions traversing the macula densa[24]. Consequently, the body constricts small glomerular arteries, attenuating renal blood flow and intraglomerular pressure and thus ameliorating the state of glomerular hyperfiltration. Moreover, in vivo experiments in animal models have revealed the potential of SGLT-2 inhibitors (dapagliflozin and empagliflozin) to alleviate diabetes-induced renal and hepatic inflammation[25,26]. Our study echoes these findings, demonstrating the efficacy of da
Macrophages play important roles in the development of DN and are responsible for the interstitial proliferation of renal tissue and irreversible pathological changes in the glomeruli. In experimental models of DM, macrophage infiltration occurs in the early stages of DM and is strongly associated with renal injury. Furthermore, therapeutic strategies aimed at reducing monocyte infiltration may attenuate the occurrence of kidney injury[27]. However, in our study, dapagliflozin was found to reduce macrophage apoptosis. To address this paradoxical result, we further investigated whether it was related to macrophage polarization.
Notably, recent discoveries have revealed the capacity of SGLT-2 inhibitors to modulate macrophage polarization toward the M2 phenotype[3,28,29]. Consistent with these findings, our study revealed that dapagliflozin attenuates macrophage polarization toward the M1 phenotype, thereby mitigating the inflammatory response. The exploration of heterogeneous profiles of pro- and anti-inflammatory macrophages has been pivotal in recent years. Macrophage polarization orchestrates alterations in the functional phenotype of macrophages in response to the microenvironmental milieu[30]. Distinct macrophage phenotypes exhibit divergent modes of activation and immune functions. M1-type macrophages, through the secretion of proinflammatory cytokines such as IL-6, IL-1β, TNF-α, IL-12, and IL-23, promote the exacerbation and perpetuation of inflammatory responses, with the aim of eradicating bacterial, fungal, and viral infections[31]. Conversely, M2-type macrophages foster inflammatory responses by facilitating the expression of arginase-1, mannose receptor (CD206), and other proinflammatory cytokines, increasing the expression of the anti-inflammatory factor IL-10, and increasing the production of the chemokines CCL17 and CCL22, thereby promoting tissue repair, angiogenesis, and metabolism[32]. Under conditions of normal homeostasis within the internal milieu of an organism, M2-type macrophages dominate, exerting immunosuppressive effects to sustain a balanced immune microenvironment[33]. However, during the onset of T2DM, metabolic syndrome, obesity, and related metabolic disorders, adipocytes within the body undergo proliferation, hypertrophy, or even necrosis, disrupting the equilibrium of the normal immune microenvironment[33]. Consequently, macrophages within adipose tissue undergo polarization toward the M1 phenotype, leading to a relative reduction in the proportion of M2-type macrophages. These imbalances in the original ratio and functions of M1- and M2-type macrophages become exacerbated with the progression of T2DM, perpetuating a state of severe imbalance. Chronic, low-grade inflammation infiltrates adipose tissue, exacerbating insulin resistance and precipitating a spectrum of systemic complications[34]. Macrophages located at sites of renal injury in DN are predominantly of the M1 type. M1 macrophages can exacerbate diabetic renal injury, which is associated with the absence of cyclooxygenase-2[35]. Moreover, inhibition of M1 macrophage activation and promotion of M2 macrophage tran
In our study, we found that dapagliflozin modulated macrophage polarization and attenuated macrophage apoptosis. Increased release of TNF-α during macrophage polarization to the M1 phenotype promotes macrophage apoptosis[36]. Based on our results, it is reasonable to assume that dapagliflozin ameliorates the persistent inflammatory state in DN by decreasing M1 macrophage polarization and reducing M2-type macrophage apoptosis.
The PI3K/AKT signaling pathway orchestrates a myriad of cellular processes, including cell survival, proliferation, growth, metabolism, and angiogenesis, and functions as a linchpin in the regulation of the cell cycle and glucose metabolism[37]. Central to the insulin pathway, the PI3K/AKT signaling axis governs hepatic glycogen synthesis, gluconeogenesis, and lipid synthesis[38]. Nonetheless, few studies have investigated the role of the PI3K/AKT signaling pathway in macrophages.
In our current investigation, we revealed that dapagliflozin can reverse macrophage apoptosis and M1 polarization induced by LPS + IFN-γ through its ability to modulate the macrophage phenotype. Intriguingly, treatment with 740Y-P reversed the effects of dapagliflozin, increasing macrophage apoptosis and promoting the M1 phenotype. Moreover, we meticulously scrutinized the expression profiles of proteins affiliated with the PI3K/AKT signaling pathway in each treatment cohort, revealing that dapagliflozin quelled the activation of this pivotal pathway. These findings suggest that the decrease in M1 polarization and the antiapoptotic efficacy of dapagliflozin are intricately associated with the PI3K/AKT signaling pathway.
In summary, our study elucidates the mechanism by which dapagliflozin impedes cellular M1 polarization, thus mitigating macrophage apoptosis. This delineation underscores the pivotal role of macrophage polarization in the amelioration of macrophage apoptosis by dapagliflozin. Such insights not only deepen our comprehension of the drug’s glucose-lowering mechanism but also provide a novel trajectory for investigating macrophage polarization in the realm of future DM therapeutics. Moreover, our findings shed light on the prospect of dapagliflozin regulating macrophage polarization via the PI3K/AKT signaling pathway, offering a distinctive strategic angle in the management of DM and its chronic complications.
Nevertheless, it is imperative to acknowledge the limitations of our study, which has primarily served as a preliminary exploration into the potential of dapagliflozin to modulate macrophage polarization through the inhibition of the PI3K/AKT signaling pathway. We will continue to explore the downstream target genes of the PI3K/AKT signaling pathway. Hence, while our findings furnish foundational experimental data, they pave the way for future endeavors aimed at further probing this intriguing avenue.
Dapagliflozin curtails the polarization of macrophages toward the M1 phenotype, mitigating inflammation and enhan
We acknowledge the Anhui Province Key Laboratory of Basic and Translational Research of Inflammation-related Diseases for providing a platform for the authors’ work.
| 1. | Lee SG, Lee SJ, Lee JJ, Kim JS, Lee OH, Kim CK, Kim D, Lee YH, Oh J, Park S, Jeon OH, Hong SJ, Ahn CM, Kim BK, Ko YG, Choi D, Hong MK, Jang Y. Anti-Inflammatory Effect for Atherosclerosis Progression by Sodium-Glucose Cotransporter 2 (SGLT-2) Inhibitor in a Normoglycemic Rabbit Model. Korean Circ J. 2020;50:443-457. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 51] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 2. | Verma A, Patel AB, Waikar SS. SGLT2 Inhibitor: Not a Traditional Diuretic for Heart Failure. Cell Metab. 2020;32:13-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 3. | Yan P, Song X, Tran J, Zhou R, Cao X, Zhao G, Yuan H. Dapagliflozin Alleviates Coxsackievirus B3-induced Acute Viral Myocarditis by Regulating the Macrophage Polarization Through Stat3-related Pathways. Inflammation. 2022;45:2078-2090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 4. | Sica A, Mantovani A. Macrophage plasticity and polarization: in vivo veritas. J Clin Invest. 2012;122:787-795. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3696] [Cited by in RCA: 4740] [Article Influence: 364.6] [Reference Citation Analysis (1)] |
| 5. | Wang Y, Shi R, Zhai R, Yang S, Peng T, Zheng F, Shen Y, Li M, Li L. Matrix stiffness regulates macrophage polarization in atherosclerosis. Pharmacol Res. 2022;179:106236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 40] [Article Influence: 13.3] [Reference Citation Analysis (0)] |
| 6. | Yao J, Wu D, Qiu Y. Adipose tissue macrophage in obesity-associated metabolic diseases. Front Immunol. 2022;13:977485. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 68] [Reference Citation Analysis (0)] |
| 7. | Lee JH, Lee SH, Lee EH, Cho JY, Song DK, Lee YJ, Kwon TK, Oh BC, Cho KW, Osborne TF, Jeon TI, Im SS. SCAP deficiency facilitates obesity and insulin resistance through shifting adipose tissue macrophage polarization. J Adv Res. 2023;45:1-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 8. | Liu J, Zhang Y, Sheng H, Liang C, Liu H, Moran Guerrero JA, Lu Z, Mao W, Dai Z, Liu X, Zhang L. Hyperoside Suppresses Renal Inflammation by Regulating Macrophage Polarization in Mice With Type 2 Diabetes Mellitus. Front Immunol. 2021;12:733808. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 9. | Zhang Y, Le X, Zheng S, Zhang K, He J, Liu M, Tu C, Rao W, Du H, Ouyang Y, Li C, Wu D. MicroRNA-146a-5p-modified human umbilical cord mesenchymal stem cells enhance protection against diabetic nephropathy in rats through facilitating M2 macrophage polarization. Stem Cell Res Ther. 2022;13:171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 78] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 10. | Lin B, Koibuchi N, Hasegawa Y, Sueta D, Toyama K, Uekawa K, Ma M, Nakagawa T, Kusaka H, Kim-Mitsuyama S. Glycemic control with empagliflozin, a novel selective SGLT2 inhibitor, ameliorates cardiovascular injury and cognitive dysfunction in obese and type 2 diabetic mice. Cardiovasc Diabetol. 2014;13:148. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 220] [Cited by in RCA: 313] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 11. | Li Q, Park K, Xia Y, Matsumoto M, Qi W, Fu J, Yokomizo H, Khamaisi M, Wang X, Rask-Madsen C, King GL. Regulation of Macrophage Apoptosis and Atherosclerosis by Lipid-Induced PKCδ Isoform Activation. Circ Res. 2017;121:1153-1167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Su LJ, Zhang JH, Gomez H, Murugan R, Hong X, Xu D, Jiang F, Peng ZY. Reactive Oxygen Species-Induced Lipid Peroxidation in Apoptosis, Autophagy, and Ferroptosis. Oxid Med Cell Longev. 2019;2019:5080843. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1314] [Cited by in RCA: 1206] [Article Influence: 201.0] [Reference Citation Analysis (0)] |
| 13. | Abdel-Rafei MK, Thabet NM, Rashed LA, Moustafa EM. Canagliflozin, a SGLT-2 inhibitor, relieves ER stress, modulates autophagy and induces apoptosis in irradiated HepG2 cells: Signal transduction between PI3K/AKT/GSK-3β/mTOR and Wnt/β-catenin pathways; in vitro. J Cancer Res Ther. 2021;17:1404-1418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 14. | Nasiri-Ansari N, Nikolopoulou C, Papoutsi K, Kyrou I, Mantzoros CS, Kyriakopoulos G, Chatzigeorgiou A, Kalotychou V, Randeva MS, Chatha K, Kontzoglou K, Kaltsas G, Papavassiliou AG, Randeva HS, Kassi E. Empagliflozin Attenuates Non-Alcoholic Fatty Liver Disease (NAFLD) in High Fat Diet Fed ApoE((-/-)) Mice by Activating Autophagy and Reducing ER Stress and Apoptosis. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 207] [Article Influence: 51.8] [Reference Citation Analysis (0)] |
| 15. | Ahmad E, Lim S, Lamptey R, Webb DR, Davies MJ. Type 2 diabetes. Lancet. 2022;400:1803-1820. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 517] [Article Influence: 172.3] [Reference Citation Analysis (0)] |
| 16. | Saeedi P, Salpea P, Karuranga S, Petersohn I, Malanda B, Gregg EW, Unwin N, Wild SH, Williams R. Mortality attributable to diabetes in 20-79 years old adults, 2019 estimates: Results from the International Diabetes Federation Diabetes Atlas, 9(th) edition. Diabetes Res Clin Pract. 2020;162:108086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 262] [Cited by in RCA: 395] [Article Influence: 79.0] [Reference Citation Analysis (1)] |
| 17. | Martín-Saladich Q, Simó R, Aguadé-Bruix S, Simó-Servat O, Aparicio-Gómez C, Hernández C, Ramirez-Serra C, Pizzi MN, Roque A, González Ballester MA, Herance JR. Insights into Insulin Resistance and Calcification in the Myocardium in Type 2 Diabetes: A Coronary Artery Analysis. Int J Mol Sci. 2023;24. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 18. | Yunna C, Mengru H, Lei W, Weidong C. Macrophage M1/M2 polarization. Eur J Pharmacol. 2020;877:173090. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 275] [Cited by in RCA: 1436] [Article Influence: 287.2] [Reference Citation Analysis (0)] |
| 19. | Vasamsetti SB, Coppin E, Zhang X, Florentin J, Koul S, Götberg M, Clugston AS, Thoma F, Sembrat J, Bullock GC, Kostka D, St Croix CM, Chattopadhyay A, Rojas M, Mulukutla SR, Dutta P. Apoptosis of hematopoietic progenitor-derived adipose tissue-resident macrophages contributes to insulin resistance after myocardial infarction. Sci Transl Med. 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 19] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 20. | Bays H. From victim to ally: the kidney as an emerging target for the treatment of diabetes mellitus. Curr Med Res Opin. 2009;25:671-681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 65] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 21. | Vaduganathan M, Docherty KF, Claggett BL, Jhund PS, de Boer RA, Hernandez AF, Inzucchi SE, Kosiborod MN, Lam CSP, Martinez F, Shah SJ, Desai AS, McMurray JJV, Solomon SD. SGLT-2 inhibitors in patients with heart failure: a comprehensive meta-analysis of five randomised controlled trials. Lancet. 2022;400:757-767. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 514] [Article Influence: 171.3] [Reference Citation Analysis (0)] |
| 22. | Roden M, Merker L, Christiansen AV, Roux F, Salsali A, Kim G, Stella P, Woerle HJ, Broedl UC; EMPA-REG EXTEND™ MONO investigators. Safety, tolerability and effects on cardiometabolic risk factors of empagliflozin monotherapy in drug-naïve patients with type 2 diabetes: a double-blind extension of a Phase III randomized controlled trial. Cardiovasc Diabetol. 2015;14:154. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 96] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 23. | Kang A, Jardine MJ. SGLT2 inhibitors may offer benefit beyond diabetes. Nat Rev Nephrol. 2021;17:83-84. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 32] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 24. | Sugiyama S, Jinnouchi H, Kurinami N, Hieshima K, Yoshida A, Jinnouchi K, Nishimura H, Suzuki T, Miyamoto F, Kajiwara K, Jinnouchi T. The SGLT2 Inhibitor Dapagliflozin Significantly Improves the Peripheral Microvascular Endothelial Function in Patients with Uncontrolled Type 2 Diabetes Mellitus. Intern Med. 2018;57:2147-2156. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 25. | Tahara A, Kurosaki E, Yokono M, Yamajuku D, Kihara R, Hayashizaki Y, Takasu T, Imamura M, Li Q, Tomiyama H, Kobayashi Y, Noda A, Sasamata M, Shibasaki M. Effects of SGLT2 selective inhibitor ipragliflozin on hyperglycemia, hyperlipidemia, hepatic steatosis, oxidative stress, inflammation, and obesity in type 2 diabetic mice. Eur J Pharmacol. 2013;715:246-255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 249] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 26. | Terami N, Ogawa D, Tachibana H, Hatanaka T, Wada J, Nakatsuka A, Eguchi J, Horiguchi CS, Nishii N, Yamada H, Takei K, Makino H. Long-term treatment with the sodium glucose cotransporter 2 inhibitor, dapagliflozin, ameliorates glucose homeostasis and diabetic nephropathy in db/db mice. PLoS One. 2014;9:e100777. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 203] [Cited by in RCA: 263] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 27. | Calle P, Hotter G. Macrophage Phenotype and Fibrosis in Diabetic Nephropathy. Int J Mol Sci. 2020;21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 176] [Article Influence: 35.2] [Reference Citation Analysis (0)] |
| 28. | Lee TM, Chang NC, Lin SZ. Dapagliflozin, a selective SGLT2 Inhibitor, attenuated cardiac fibrosis by regulating the macrophage polarization via STAT3 signaling in infarcted rat hearts. Free Radic Biol Med. 2017;104:298-310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 378] [Article Influence: 47.3] [Reference Citation Analysis (0)] |
| 29. | Abdollahi E, Keyhanfar F, Delbandi AA, Falak R, Hajimiresmaiel SJ, Shafiei M. Dapagliflozin exerts anti-inflammatory effects via inhibition of LPS-induced TLR-4 overexpression and NF-κB activation in human endothelial cells and differentiated macrophages. Eur J Pharmacol. 2022;918:174715. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 84] [Article Influence: 28.0] [Reference Citation Analysis (0)] |
| 30. | Eshghjoo S, Kim DM, Jayaraman A, Sun Y, Alaniz RC. Macrophage Polarization in Atherosclerosis. Genes (Basel). 2022;13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 69] [Article Influence: 23.0] [Reference Citation Analysis (0)] |
| 31. | Chistiakov DA, Bobryshev YV, Nikiforov NG, Elizova NV, Sobenin IA, Orekhov AN. Macrophage phenotypic plasticity in atherosclerosis: The associated features and the peculiarities of the expression of inflammatory genes. Int J Cardiol. 2015;184:436-445. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 146] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 32. | Sanjurjo L, Aran G, Téllez É, Amézaga N, Armengol C, López D, Prats C, Sarrias MR. CD5L Promotes M2 Macrophage Polarization through Autophagy-Mediated Upregulation of ID3. Front Immunol. 2018;9:480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 70] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 33. | Sun C, Sun L, Ma H, Peng J, Zhen Y, Duan K, Liu G, Ding W, Zhao Y. The phenotype and functional alterations of macrophages in mice with hyperglycemia for long term. J Cell Physiol. 2012;227:1670-1679. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 77] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 34. | Fadini GP, Cappellari R, Mazzucato M, Agostini C, Vigili de Kreutzenberg S, Avogaro A. Monocyte-macrophage polarization balance in pre-diabetic individuals. Acta Diabetol. 2013;50:977-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 49] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 35. | Wang X, Yao B, Wang Y, Fan X, Wang S, Niu A, Yang H, Fogo A, Zhang MZ, Harris RC. Macrophage Cyclooxygenase-2 Protects Against Development of Diabetic Nephropathy. Diabetes. 2017;66:494-504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 71] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 36. | Stratos I, Behrendt AK, Anselm C, Gonzalez A, Mittlmeier T, Vollmar B. Inhibition of TNF-α Restores Muscle Force, Inhibits Inflammation, and Reduces Apoptosis of Traumatized Skeletal Muscles. Cells. 2022;11. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 37. | Chen S, Peng J, Sherchan P, Ma Y, Xiang S, Yan F, Zhao H, Jiang Y, Wang N, Zhang JH, Zhang H. TREM2 activation attenuates neuroinflammation and neuronal apoptosis via PI3K/Akt pathway after intracerebral hemorrhage in mice. J Neuroinflammation. 2020;17:168. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 150] [Cited by in RCA: 230] [Article Influence: 46.0] [Reference Citation Analysis (0)] |
| 38. | Xiao H, Sun X, Lin Z, Yang Y, Zhang M, Xu Z, Liu P, Liu Z, Huang H. Gentiopicroside targets PAQR3 to activate the PI3K/AKT signaling pathway and ameliorate disordered glucose and lipid metabolism. Acta Pharm Sin B. 2022;12:2887-2904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 92] [Article Influence: 30.7] [Reference Citation Analysis (0)] |