Published online Mar 15, 2024. doi: 10.4239/wjd.v15.i3.475
Peer-review started: December 15, 2023
First decision: December 23, 2023
Revised: January 5, 2024
Accepted: February 18, 2024
Article in press: February 18, 2024
Published online: March 15, 2024
Processing time: 90 Days and 19.5 Hours
Dietary fiber (DF) intake may have a protective effect against type 2 diabetes (T2D); however, its relationship with diabetic kidney disease (DKD) remains unclear.
To investigate the potential association between DF intake and the prevalence of DKD in individuals diagnosed with T2D.
This cross-sectional study used data from the National Health and Nutrition Examination Survey collected between 2005 and 2018. DF intake was assessed through 24-h dietary recall interviews, and DKD diagnosis in individuals with T2D was based on predefined criteria, including albuminuria, impaired glomerular filtration rate, or a combination of both. Logistic regression analysis was used to assess the association between DF intake and DKD, and comprehensive subgroup and sensitivity analyses were performed.
Among the 6032 participants, 38.4% had DKD. With lower DF intake-T1 (≤ 6.4 g/1000 kcal/day)-as a reference, the adjusted odds ratio for DF and DKD for levels T2 (6.5-10.0 g/1000 kcal/day) and T3 (≥ 10.1 g/1000 kcal/day) were 0.97 (95%CI: 0.84-1.12, P = 0.674) and 0.79 (95%CI: 0.68-0.92, P = 0.002), respectively. The subgroup analysis yielded consistent results across various demographic and health-related subgroups, with no statistically significant interactions (all P > 0.05).
In United States adults with T2D, increased DF intake may be related to reduced DKD incidence. Further research is required to confirm these findings.
Core Tip: This cross-sectional analysis of National Health and Nutrition Examination Survey data, spanning 2005 to 2018, explored the potential correlation between dietary fiber (DF) intake and the prevalence of diabetic kidney disease (DKD) in individuals diagnosed with type 2 diabetes (T2D). The study, which involved 6032 participants, reveals that higher DF intake, particularly in the tertile with ≥ 10.1 g/1000 kcal/day, is associated with a statistically significant reduction in DKD incidence. These findings suggest a potential protective effect of increased DF intake against DKD in adults with T2D in the United States. Further investigation is warranted to corroborate these observations.
- Citation: Jia XH, Wang SY, Sun AQ. Dietary fiber intake and its association with diabetic kidney disease in American adults with diabetes: A cross-sectional study. World J Diabetes 2024; 15(3): 475-487
- URL: https://www.wjgnet.com/1948-9358/full/v15/i3/475.htm
- DOI: https://dx.doi.org/10.4239/wjd.v15.i3.475
Diabetes is a collective term for metabolic disorders characterized by hyperglycemia stemming from deficiencies in insulin secretion, insulin action, or both[1]. With the continuous expansion of the global economy and improved life expectancy, the prevalence of diabetes has increased. According to data from the International Diabetes Federation Diabetes Atlas, an estimated 783.2 million individuals aged 20-79 years worldwide will be living with diabetes by 2045[2]. Diabetic kidney disease (DKD) is a complication associated with prolonged diabetes that affects approximately 40% of individuals with type 2 diabetes (T2D) and is one of the most prevalent diabetic complications[3]. DKD markedly increases the risk of cardiovascular events and progression to end-stage renal disease[4,5], culminating in the need for dialysis or renal transplantation. Existing therapeutic approaches offer only symptomatic relief and cannot impede the progression of DKD into chronic kidney disease (CKD)[6]. Therefore, delaying or ameliorating the onset and progression of DKD is crucial.
Dietary fiber (DF) is a composite material composed of indigestible carbohydrates and lignin and resists degradation in the upper gastrointestinal tract. Their primary sources are whole grains, fruits, vegetables, and legumes[7]. Previous studies have highlighted the multifaceted advantages of DF, including its ability to modulate blood glucose[8,9] and lipid levels[9], enhance insulin sensitivity[10], ameliorate inflammatory responses[9], reduce the onset of diabetes[11-13], and lower the prevalence of cardiovascular diseases[14,15] and CKD[16]. However, large-scale studies assessing the association between DF intake and the incidence of DKD in diabetic populations are lacking. Therefore, the current study aimed to investigate whether a higher DF intake reduces the occurrence of DKD in individuals with T2D.
The National Health and Nutrition Examination Survey (NHANES) was designed to assess the health and nutritional status of both adults and children in the United States. This program employed a comprehensive approach that combined structured interviews and physical examinations to amass extensive datasets. NHANES operates under the National Center for Health Statistics (NCHS), in partnership with the United States Department of Agriculture, which is responsible for the compilation and dissemination of dietary and nutritional data. Prior to participating in this survey, all NHANES participants provided written informed consent, and the study was approved by the NCHS Institutional Review Board[17]. The survey included intricate measurements, standardized interviews, and laboratory assessments to gather information related to demographics, dietary habits, physical parameters, laboratory diagnostics, and health-related parameters.
This cross-sectional investigation draws from data acquired from NHANES surveys conducted between 2005 and 2018. For more comprehensive details, individuals can access the NHANES resources via www.cdc.gov/nchs/nhanes. The current analysis included participants aged 20 years and older who had completed both structured questionnaire surveys and rigorous laboratory examinations. Pregnant women and individuals with missing critical variables were excluded from the analysis.
All NHANES participants were eligible to undergo two 24-h dietary recall interviews. The first dietary recall interview took place at the Mobile Examination Center, and the second interview occurred via telephone three to ten days later. To ensure the integrity and availability of raw data, data from the first 24-h dietary recall were utilized. Recognizing that total calorie intake may influence dietary data, the data for DF and protein intake used in this study were adjusted for total calorie intake.
The diagnosis of T2D was based on the criteria established by the American Diabetes Association[18] and self-reported questionnaires. Participants were classified as having T2D if they met any of the following conditions: (1) A documented previous diagnosis by a medical practitioner; (2) current utilization of insulin or oral hypoglycemic medications; (3) fasting plasma glucose (FPG) ≥ 7.0 mmol/L; (4) glycosylated hemoglobin (HbA1c) ≥ 6.5%; and or (5) plasma glucose ≥ 11.1 mmol/L 2 h post oral glucose tolerance test. The diagnosis of DKD in patients with T2D was established when the albumin-to-creatinine ratio (ACR) exceeded 30 mg/g and/or the estimated glomerular filtration rate (eGFR) fell below 60 mL/min/1.73 m2[19]. ACR was computed by assessing the urine albumin-to-urine creatinine ratio, whereas eGFR was determined using the CKD Epidemiology Collaboration algorithm[20].
The current study examined variables that could influence the association between DF intake and DKD. These variables included age, sex, race/ethnicity, educational level, marital status, family income, body mass index (BMI), smoking status, daily calorie and protein intake, dietary supplement usage, history of coronary heart disease (CHD), hypertension, family history of diabetes, age at initial diabetes diagnosis, insulin and glucose-lowering drug utilization, blood pressure, and various laboratory tests including HbA1c, fasting plasma insulin (FINS), FPG, total cholesterol (TC), alanine aminotransferase (ALT), aspartate aminotransferase, serum uric acid (SUA), blood urea nitrogen, serum creatinine (sCr), HbA1c, urine albumin, and urine creatinine. Race/ethnicity was categorized into non-Hispanic white, non-Hispanic black, Mexican American, or other ethnic groups. Educational level was stratified by the number of years of completed education as follows: < 9 years, 9-12 years, and > 12 years. Marital status was classified as being married, living with a partner, or living alone, based on questionnaire responses. Family income was determined using the family poverty income ratio and categorized as low (≤ 1.3), medium (1.3–3.5), and high (> 3.5) income levels[17]. Smoking status was divided into three categories: Never smoked, former smoker, and current smoker. Never-smokers were individuals who had smoked < 100 cigarettes in their lifetime. Former smokers were defined as those who had smoked more than 100 cigarettes in their lifetime but were not currently smoking. Current smokers were defined as individuals who had smoked at least 100 cigarettes in their lifetime and were currently smoking. The use of dietary supplements was defined as the use of dietary supplements or medications in the past month. The diagnosis of CHD was ascertained through a questionnaire item inquiring about prior diagnosis of CHD. The diagnosis of hypertension included systolic blood pressure ≥ 140 mmHg and/or diastolic blood pressure ≥ 90 mmHg, self-reported diagnosis with hypertension, or being informed about hypertension on two or more occasions. A family history of diabetes was determined through questions concerning close relatives with diabetes. The duration of diabetes was calculated by subtracting the age at initial diagnosis with diabetes from the age t time of the interview. Those diagnosed within the last year were categorized as having diabetes for half a year. The use of insulin- and glucose-lowering drugs was determined based on specific questionnaire responses. BMI was calculated using height and weight measurements. The homeostasis model assessment of insulin resistance (HOMA-IR), employed to evaluate insulin resistance, was calculated using the formula HOMA-IR = FINS (μU/mL) × FPG (mmol/L)/22.5[21]. Further details regarding the measurement techniques used for these variables are available at www.cdc.gov/nchs/nhanes/.
Data analyses were conducted using R Statistical Software (http://www.R-project.org, The R Foundation) and the Free Statistics analysis platform (Beijing, China). Continuous variables are presented as means ± SD or medians (interquartile range), while categorical variables are reported as frequencies or percentages. To compare baseline characteristics between groups based on DF intake, independent t-tests were used for continuous variables, and chi-square tests were used for categorical variables. Univariate logistic regression analysis was performed to explore the relationship between the potential factors and DKD. Subsequently, a multivariate logistic regression analysis was performed to ascertain the association between DF intake and DKD. Model 1 was adjusted for age, sex, race/ethnicity, educational level, marital status, and family income. Model 2 incorporated all the covariates from Model 1 and included additional adjustments for smoking status, CHD, hypertension, and daily calorie and protein consumption. Model 3 extended the adjustments to encompass the duration of diabetes; insulin usage; and HbA1c, HOMA-IR, TC, ALT, SUA, and HbA1c levels. To explore the potential nonlinear relationship between DF intake and DKD, restricted cubic splines (RCS) were applied while controlling for all covariates in the full model (Model 3). Subgroup analyses were conducted by stratifying the data into five subgroups based on age (20-60 years vs ≥ 60 years), sex (male vs female), BMI (< 25 kg/m2vs ≥ 25 kg/m2), HbA1c level (< 7.0% vs ≥ 7.0%), and eGFR level (< 90 mL/min/1.73 m2vs ≥ 90 mL/min/1.73 m2). Additionally, this study explored the interactions between DF intake and these factors. To ensure the robustness of the findings, the relationship between DF intake and DKD was re-examined after excluding extreme energy intake values (< 500 kcal/day or > 5000 kcal/day). To address missing data, multiple imputation techniques were employed to fill existing gaps and facilitate a comprehensive analysis.
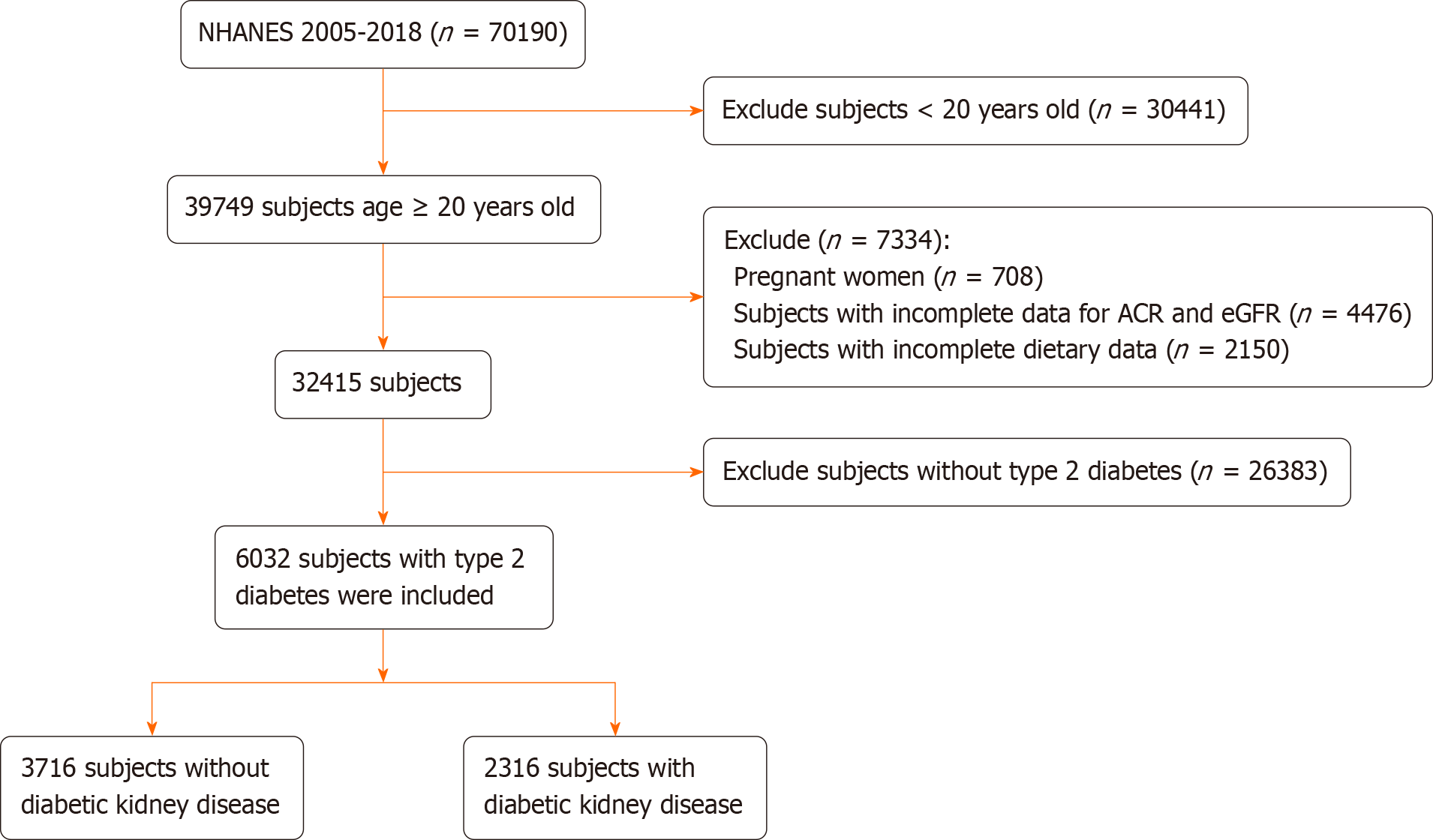
This study included 70190 participants from NHANES 2005-2018, of whom 39749 were aged 20 years or older. However, the study excluded pregnant women (n = 708), individuals with missing ACR and eGFR data (n = 4476), those lacking dietary data (n = 2150), and those who did not meet the diagnostic criteria for T2D (n = 26383). This resulted in a final sample size of 6032 participants, of which 2316 were diagnosed with DKD. The inclusion and exclusion criteria are shown in Figure 1.
Table 1 presents the baseline characteristics of all the participants categorized into tertiles based on their DF intake. In total, 2316 (38.4%) patients had DKD. The average age of the study participants was 61.1 years, and 2892 (47.9%) were female. Individuals who consumed more DF tended to be older, female, Mexican American, married, living with a partner, or a never smoker, or have a lower educational level, lower BMI, lower daily calorie consumption, and higher protein consumption. Moreover, this group was more likely to use dietary supplements and glucose-lowering drugs and had a longer duration of T2D. Notably, they displayed lower levels of FINS, HOMA-IR, TC, SUA, sCr, HbA1c, and diastolic blood pressure.
| Variables | Total | Dietary fiber intake, g/1000 kcal/d | P value | ||
| T1 (≤ 6.4) | T2 (6.5-10.0) | T3 (≥ 10.1) | |||
| N | 6032 | 1978 | 2000 | 2054 | |
| Age (yr), mean (SD) | 61.1 (13.5) | 58.3 (14.2) | 61.8 (13.3) | 63.0 (12.6) | < 0.001 |
| Sex, n (%) | < 0.001 | ||||
| Male | 3140 (52.1) | 1146 (57.9) | 1011 (50.5) | 983 (47.9) | |
| Female | 2892 (47.9) | 832 (42.1) | 989 (49.5) | 1071 (52.1) | |
| Race/ethnicity, n (%) | < 0.001 | ||||
| Non-Hispanic white | 2210 (36.6) | 799 (40.4) | 789 (39.5) | 622 (30.3) | |
| Non-Hispanic black | 1481 (24.6) | 626 (31.6) | 465 (23.2) | 390 (19.0) | |
| Mexican American | 1126 (18.7) | 237 (12.0) | 355 (17.8) | 534 (26.0) | |
| Others | 1215 (20.1) | 316 (16.0) | 391 (19.6) | 508 (24.7) | |
| Educational level (yr), n (%) | < 0.001 | ||||
| < 9 | 1067 (17.7) | 265 (13.4) | 288 (14.4) | 514 (25.0) | |
| 9-12 | 2406 (39.9) | 863 (43.6) | 801 (40.1) | 742 (36.1) | |
| > 12 | 2559 (42.4) | 850 (43.0) | 911 (45.6) | 798 (38.9) | |
| Marital status, n (%) | 0.013 | ||||
| Married or living with a partner | 3649 (60.5) | 1164 (58.8) | 1190 (59.5) | 1295 (63.0) | |
| Living alone | 2383 (39.5) | 814 (41.2) | 810 (40.5) | 759 (37.0) | |
| Family income, n (%) | 0.340 | ||||
| Low | 2151 (35.7) | 706 (35.7) | 682 (34.1) | 763 (37.1) | |
| Medium | 2453 (40.7) | 795 (40.2) | 836 (41.8) | 822 (40.0) | |
| High | 1428 (23.7) | 477 (24.1) | 482 (24.1) | 469 (22.8) | |
| BMI (kg/m2), mean (SD) | 32.3 (7.4) | 33.1 (7.9) | 32.5 (7.3) | 31.5 (6.8) | < 0.001 |
| Smoking status, n (%) | < 0.001 | ||||
| Never | 3029 (50.2) | 893 (45.1) | 952 (47.6) | 1184 (57.6) | |
| Former | 2032 (33.7) | 601 (30.4) | 749 (37.5) | 682 (33.2) | |
| Current | 971 (16.1) | 484 (24.5) | 299 (14.9) | 188 (9.2) | |
| CHD, n (%) | 607 (10.1) | 192 (9.7) | 206 (10.3) | 209 (10.2) | 0.807 |
| Hypertension, n (%) | 3942 (65.4) | 1281 (64.8) | 1316 (65.8) | 1345 (65.5) | 0.780 |
| Calorie consumption (1000 kcal/d), mean (SD) | 1.9 (0.9) | 2.1 (1.0) | 1.9 (0.9) | 1.7 (0.7) | < 0.001 |
| Protein consumption (g/1000 kcal/d), mean (SD) | 41.2 (13.3) | 40.4 (14.7) | 40.9 (12.6) | 42.2 (12.5) | < 0.001 |
| Dietary fiber (g/1000 kcal/d), median (IQR) | 8.2 (5.7, 11.4) | 4.7 (3.7, 5.7) | 8.1 (7.3, 9.0) | 13.1 (11.3, 16.1) | < 0.001 |
| Dietary supplements taken, n (%) | 3300 (54.7) | 981 (49.6) | 1105 (55.2) | 1214 (59.1) | < 0.001 |
| Family history of diabetes, n (%) | 3840 (63.7) | 1276 (64.5) | 1265 (63.2) | 1299 (63.2) | 0.632 |
| Duration of diabetes (yr), median (IQR) | 8.0 (3.0, 15.0) | 8.0 (3.0, 15.0) | 9.0 (3.0, 15.0) | 9.0 (4.0, 16.0) | < 0.001 |
| Insulin use, n (%) | 1160 (19.2) | 384 (19.4) | 378 (18.9) | 398 (19.4) | 0.900 |
| Glucose-lowering drugs use, n (%) | 4092 (67.8) | 1237 (62.5) | 1393 (69.7) | 1462 (71.2) | < 0.001 |
| HbA1c (%), mean (SD) | 7.2 (1.7) | 7.3 (1.9) | 7.2 (1.7) | 7.2 (1.7) | 0.404 |
| FPG (mmol/L), mean (SD) | 8.6 (3.4) | 8.7 (3.6) | 8.6 (3.2) | 8.5 (3.3) | 0.088 |
| FINS (μU/mL), median (IQR) | 14.1 (8.5, 23.9) | 15.3 (8.9, 26.1) | 14.3 (8.5, 23.9) | 13.3 (8.2, 21.9) | < 0.001 |
| HOMA-IR, median (IQR) | 5.1 (2.8, 8.9) | 5.4 (3.0, 10.1) | 5.1 (2.9, 8.9) | 4.8 (2.7, 8.1) | < 0.001 |
| TC (mmol/L), mean (SD) | 4.8 (1.2) | 4.9 (1.3) | 4.8 (1.2) | 4.8 (1.2) | < 0.001 |
| ALT (U/L), median (IQR) | 22.0 (16.0, 30.0) | 22.0 (16.0, 31.0) | 21.0 (16.0, 29.0) | 22.0 (17.0, 29.0) | 0.191 |
| AST (U/L), median (IQR) | 23.0 (19.0, 28.0) | 23.0 (19.0, 28.0) | 23.0 (19.0, 28.0) | 23.0 (19.0, 28.0) | 0.231 |
| SUA (µmol/L), mean (SD) | 344.4 (94.4) | 352.5 (99.4) | 343.4 (92.2) | 337.6 (90.9) | < 0.001 |
| BUN (mmol/L), median (IQR) | 5.0 (3.9, 6.8) | 5.0 (3.9, 6.4) | 5.4 (3.9, 6.8) | 5.4 (3.9, 6.8) | < 0.001 |
| sCr (µmol/L), median (IQR) | 79.6 (63.6, 97.2) | 80.4 (67.2, 99.0) | 79.6 (64.5, 97.2) | 76.0 (61.9, 95.5) | < 0.001 |
| ACR (mg/g), median (IQR) | 12.9 (6.8, 40.6) | 12.5 (6.7, 43.5) | 13.1 (6.8, 43.8) | 12.9 (6.8, 35.9) | 0.487 |
| eGFR (ml/min/1.73 m2), mean (SD) | 83.3 (24.3) | 83.9 (25.0) | 82.4 (24.1) | 83.7 (23.9) | 0.103 |
| Hemoglobin (g/dL), mean (SD) | 13.9 (1.6) | 14.0 (1.7) | 13.8 (1.6) | 13.7 (1.6) | < 0.001 |
| Systolic blood pressure (mmHg), mean (SD) | 132.0 (20.1) | 131.4 (19.8) | 132.0 (20.1) | 132.7 (20.4) | 0.124 |
| Diastolic blood pressure (mmHg), mean (SD) | 69.3 (13.5) | 70.9 (13.5) | 69.3 (13.5) | 68.0 (13.2) | < 0.001 |
| DKD, n (%) | 2316 (38.4) | 765 (38.7) | 803 (40.2) | 748 (36.4) | 0.048 |
Univariate analysis revealed that age; race/ethnicity; educational level; marital status; family income; smoking status; CHD; hypertension; daily calorie and DF consumption; duration of diabetes; insulin usage; and levels of HbA1c, FPG, HOMA-IR, TC, ALT, SUA, and HbA1c were associated with DKD (Table 2).
| Variables | OR (95%CI) | P value |
| Age (yr) | 1.04 (1.04-1.05) | < 0.001 |
| Sex, n (%) | ||
| Male | 1 (reference) | |
| Female | 0.93 (0.84-1.03) | 0.178 |
| Race/ethnicity, n (%) | ||
| Non-Hispanic white | 1 (reference) | |
| Non-Hispanic black | 1.11 (0.97-1.27) | 0.115 |
| Mexican American | 0.80 (0.69-0.93) | 0.003 |
| Others | 0.72 (0.62-0.83) | < 0.001 |
| Educational level (yr), n (%) | ||
| < 9 | 1 (reference) | |
| 9-12 | 0.97 (0.84-1.12) | 0.651 |
| > 12 | 0.73 (0.63-0.85) | < 0.001 |
| Marital status, n (%) | ||
| Married or living with a partner | 1 (reference) | |
| Living alone | 1.45 (1.31-1.61) | < 0.001 |
| Family income, n (%) | ||
| Low | 1 (reference) | |
| Medium | 0.96 (0.85-1.08) | 0.466 |
| High | 0.62 (0.54-0.72) | < 0.001 |
| BMI (kg/m2) | 1.00 (0.99-1.01) | 0.605 |
| Smoking status, n (%) | ||
| Never | 1 (reference) | |
| Former | 1.34 (1.20-1.51) | < 0.001 |
| Current | 1.08 (0.93-1.25) | 0.328 |
| CHD, n (%) | ||
| No | 1 (reference) | |
| Yes | 2.16 (1.82-2.56) | < 0.001 |
| Hypertension, n (%) | ||
| No | 1 (reference) | |
| Yes | 2.24 (2.00-2.52) | < 0.001 |
| Calorie consumption (1000 kcal/d) | 0.83 (0.78-0.89) | < 0.001 |
| Protein consumption (g/1000 kcal/d) | 1.00 (1.00-1.00) | 0.965 |
| Dietary fiber consumption (g/1000 kcal/d) | 0.98 (0.97-0.99) | < 0.001 |
| Dietary supplements taken, n (%) | ||
| No | 1 (reference) | |
| Yes | 1.06 (0.95-1.17) | 0.313 |
| Family history of diabetes, n (%) | ||
| No | 1 (reference) | |
| Yes | 1.04 (0.93-1.16) | 0.487 |
| Duration of diabetes (yr) | 1.04 (1.03-1.04) | < 0.001 |
| Insulin use, n (%) | ||
| No | 1 (reference) | |
| Yes | 2.32 (2.03-2.64) | < 0.001 |
| Glucose-lowering drugs use, n (%) | ||
| No | 1 (reference) | |
| Yes | 1.03 (0.92-1.15) | 0.576 |
| HbA1c (%) | 1.15 (1.11-1.18) | < 0.001 |
| FPG (mmol/L) | 1.06 (1.04-1.07) | < 0.001 |
| FINS (μU/mL) | 1.00 (1.00-1.00) | 0.132 |
| HOMA-IR | 1.01 (1.00-1.01) | < 0.001 |
| TC (mmol/L) | 0.95 (0.91-0.99) | 0.012 |
| ALT (U/L) | 1.00 (0.99-1.00) | 0.019 |
| AST (U/L) | 1.00 (1.00-1.00) | 0.816 |
| SUA (µmol/L) | 1.00 (1.00-1.01) | < 0.001 |
| Hemoglobin (g/dL) | 0.81 (0.79-0.84) | < 0.001 |
To ascertain the independent association between DF intake and DKD, multivariate analyses were performed with adjustments in the three models. As indicated in Table 3, DF intake and DKD were inversely related, with an adjusted OR value of 0.97 (95%CI: 0.96-0.98, P < 0.001). Compared with individuals with a lower DF intake belonging to Tertile 1 (T1, ≤ 6.4 g/1000 kcal/day), the unadjusted OR values for DF intake and DKD in Tertile 2 (T2, 6.5-10.0 g/1000 kcal/day) and Tertile 3 (T3, ≥ 10.1 g/1000 kcal/day) were 0.97 (95%CI: 0.84-1.12, P = 0.674) and 0.79 (95%CI: 0.68-0.92, P = 0.002), respectively. Accordingly, the relationship between DF intake and DKD exhibited an inverse linear pattern (nonlinear, P = 0.814) in RCS analysis (Supplementary Figure 1).
| Variable | Total, n | Events, n (%) | Model 1 | Model 2 | Model 3 | |||
| OR (95%CI) | P value | OR (95%CI) | P Value | OR (95%CI) | P value | |||
| DF intake (g/1000 kcal/d) | 6032 | 2316 (38.4) | 0.97 (0.96-0.98) | < 0.001 | 0.97 (0.96-0.98) | < 0.001 | 0.97 (0.96-0.98) | < 0.001 |
| Tertile groups (g/1000 kcal/d) | ||||||||
| T1 (≤ 6.4) | 1978 | 765 (38.7) | 1 (reference) | 1 (reference) | 1 (reference) | |||
| T2 (6.5-10.0) | 2000 | 803 (40.2) | 0.96 (0.84-1.10) | 0.544 | 0.97 (0.84-1.11) | 0.642 | 0.97 (0.84-1.12) | 0.674 |
| T3 (≥ 10.1) | 2054 | 748 (36.4) | 0.78 (0.68-0.90) | 0.001 | 0.79 (0.68-0.91) | 0.001 | 0.79 (0.68-0.92) | 0.002 |
| P for trend | < 0.001 | 0.001 | 0.002 | |||||
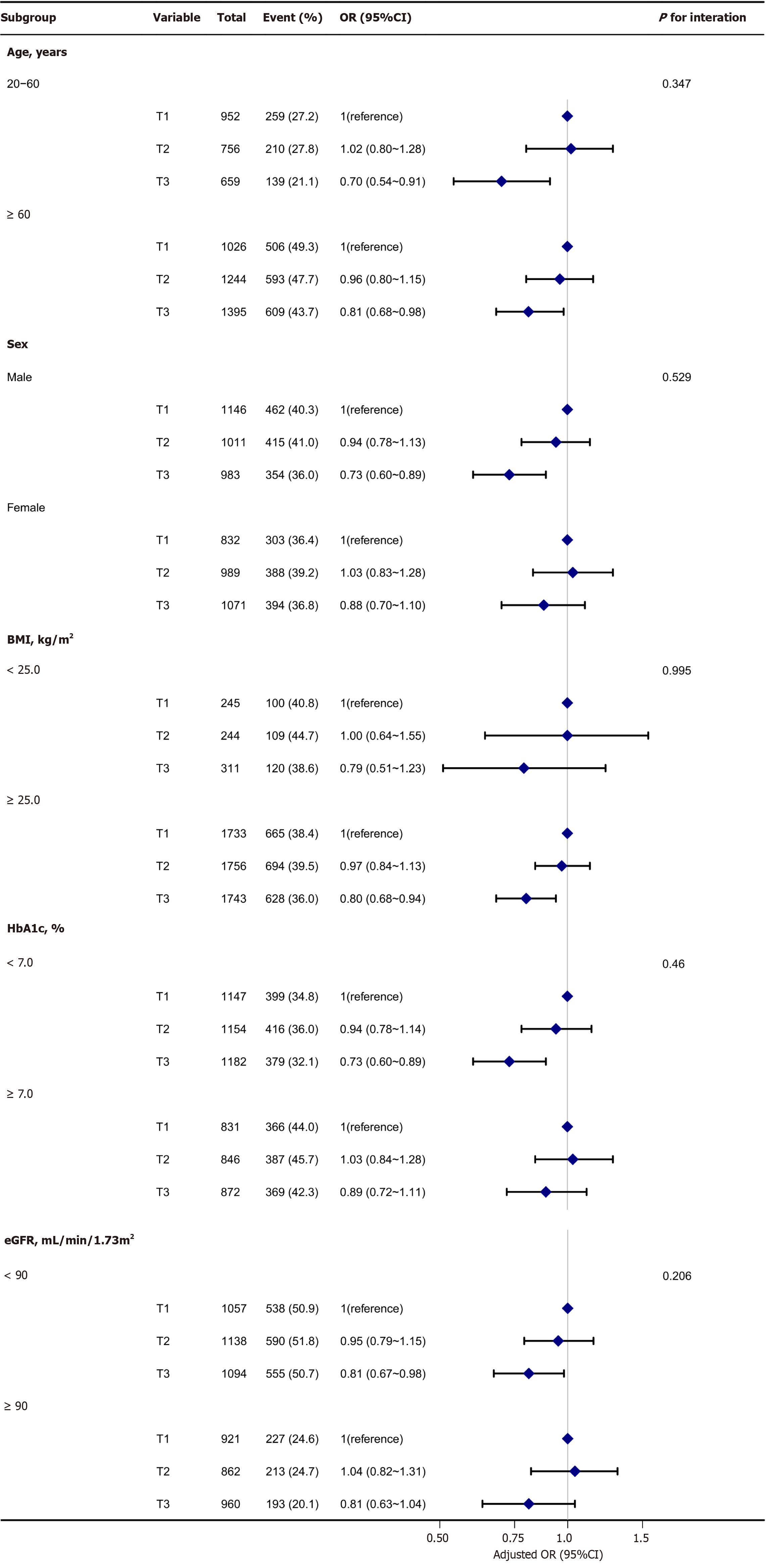
The association between DF intake and DKD was consistent across multiple subgroups. As illustrated in Figure 2, no significant interaction was detected in any subgroup after stratification by age, sex, BMI, HbA1c level, or eGFR (P > 0.05).
After excluding individuals with extreme energy intake, 5910 individuals remained, and the association between DF intake and DKD was stable. Compared with individuals with a lower DF intake, T1 (≤ 6.4 g/1000 kcal/day), the unadjusted OR values for DF intake and DKD in T2 (6.5-10.0 g/1000 kcal/day), and T3 (≥ 10.1 g/1000 kcal/day) were 1.00 (95%CI: 0.86-1.16, P = 0.996), and 0.82 (95%CI: 0.70-0.96, P = 0.011), respectively (Supplementary Table 1).
This cross-sectional study of adults with T2D in the United States revealed an inverse correlation between DF intake and the occurrence of DKD. Higher intake of DF may be associated with a reduced incidence of DKD. Subgroup and sensitivity analyses further reinforced the stability of these findings.
Numerous studies have highlighted the potential benefits of increasing DF intake in reducing the incidence of diabetes. A seven-year prospective study discovered that individuals with lower total DF intake were more susceptible to developing diabetes[11]. Additionally, an analysis of multiple prospective cohort studies supported the association between increased daily intake of cereal fiber and decreased risk of T2D[12]. The potential protective effects of DF intake against diabetes may be linked to its ability to enhance blood sugar control, regulate lipid levels, manage body weight, and modulate inflammatory responses[9]. These effects were also observed in the present study. The high DF intake group exhibited lower FINS, HOMA-IR, TC, and BMI.
Research indicates that the protective effects of cereal fibers against diabetes may be attributed to their modulation of the gut microbiota through various mechanisms, including the enhancement of glucose tolerance, reduction of inflammation, and alteration of immune responses via energy metabolism pathways such as colonic fermentation and production of short-chain fatty acids (SCFAs)[22]. DF serves as the primary energy source for most gut microbiota and undergoes fermentation in the intestine, resulting in the production of active intestinal metabolites called SCFAs such as butyrate, propionate, and acetate[23]. Studies have revealed a deficiency in SCFAs, particularly butyrate, in the gut of individuals with T2D[24]. This insufficiency is predominantly ascribed to alterations in the composition of the gut microbiota, resulting in diminished production of SCFAs[25]. In a randomized controlled trial, Zhao et al[24] identified a distinct cohort of SCFA producers that contributed to the amelioration of T2D through augmented SCFA production. Notably, an increase in DF intake can modulate the generation of SCFA metabolites[24]. Inflammation and fibrosis play pivotal roles in the pathophysiology of DKD. Significantly, discernible differences exist in the abundance and composition of the gut microbiota between individuals with DKD and healthy controls[26]. Research suggests that the gut microbiota may affect DKD development by modulating endocrine functions and the composition of microbial metabolic byproducts within the gut[27]. Notably, SCFAs derived from the gut microbiota can regulate inflammation, oxidative stress, fibrosis, and energy metabolism, thus offering preventive and therapeutic potential in DKD[6]. Experimental evidence indicates a notable decrease in DKD incidence in diabetes-induced animals fed a high-fiber diet, emphasizing the potential renoprotective role of DF. This protective effect is attributed to its capacity to reshape the gut microbiota ecology, mitigate dysbiosis, and stimulate the proliferation of SCFA-producing bacteria. Consequently, SCFA concentrations increase, exerting a protective effect against DKD through the GPR43 and GPR109A pathways[28,29]. Simultaneously, in vitro experiments have suggested that SCFAs modulate inflammatory responses in renal tubular cells and podocytes under hyperglycemic conditions[29]. Butyrate, a SCFA, may alleviate renal inflammation and fibrosis in mice through various pathways[30,31], thereby mitigating DKD. Furthermore, research indicates that SCFAs enhance autophagic capability in renal tubular cells of diabetic mice through the histone deacetylase/unc-51 Like autophagy-activating kinase 1 axis, thus alleviating renal fibrosis[32]. The correlation between the gastrointestinal system and the kidneys is called the “gut-kidney axis”[33]. These studies underscore the significance of gut microbiota in DKD, highlighting the interplay between DF, gut microbiota, SCFAs, and renal health.
To the best of our knowledge, this is the first study to investigate the correlation between DF intake and DKD in a large cohort of patients with T2D. However, the current study had certain limitations. First, akin to most cross-sectional investigations, it can only delineate an association between DF consumption and DKD but cannot establish causation. Second, the data on DF intake relied on 24-hour dietary recall information from the NHANES, which may be susceptible to recall bias. Third, distinct sources of DF may have different effects. Unfortunately, specific data about the sources of DF consumption in NHANES were not obtained. Future studies should consider including these factors. Finally, the current investigation focused on the adult population with diabetes in the United States. To address this limitation, we aim to validate current findings in future studies that include a more diverse population.
In an adult population with T2D in the United States, elevated DF intake may be associated with a decreased incidence of DKD. However, further research is required to confirm this hypothesis.
This study focused on the relationship between dietary fiber (DF) intake and diabetic kidney disease (DKD) in individuals with type 2 diabetes (T2D). The prevalence of T2D is increasing globally. The protective effect of DF against T2D is acknowledged, but its specific impact on DKD remains unclear. DKD poses a substantial health burden, underscoring the importance of investigating modifiable factors such as DF intake for potential preventive strategies.
The main topics driving this research included understanding the potential protective role of DF against DKD, a complication frequently associated with T2D. Key problems to be addressed include the lack of conclusive evidence on the DF-DKD relationship and the need for targeted interventions to mitigate DKD risk in T2D patients. Solving these problems is crucial for future research on diabetes management and the prevention of kidney disease.
The primary objective of this study was to investigate the association between DF intake and the prevalence of DKD in T2D individuals. Realizing these objectives contributes to filling gaps in the current knowledge regarding the role of DF in DKD prevention, providing insights for future research to refine dietary recommendations for individuals with T2D.
This study employed a cross-sectional design utilizing National Health and Nutrition Examination Survey data collected between 2005 and 2018. DF intake was assessed through 24-h dietary recall interviews, and DKD diagnosis was based on predefined criteria, including albuminuria and impaired glomerular filtration rate. This study employed various statistical methods including multiple regression models, restricted cubic splines, stratified analysis with interactions, and sensitivity analysis.
Of the 6032 participants, 38.4% presented with DKD. The study reveals a significant association between higher DF intake and reduced odds of DKD, particularly in the highest intake tier (T3: ≥ 10.1 g/1000 kcal/day).
This study proposes that an increased DF intake is associated with a reduced incidence of DKD in adults with T2D. These findings contribute to the field by suggesting potentially modifiable factors for DKD prevention of T2D in individuals.
Future research should delve deeper into the mechanisms underlying this observed association and explore the feasibility of dietary interventions to prevent or manage DKD in T2D patients. Additionally, longitudinal studies are warranted to establish causality and inform evidence-based dietary guidelines for individuals with T2D at risk of DKD.
The authors express gratitude to the Clinical Scientists' team for their invaluable contributions and extend appreciation to the Free Statistics Analysis Platform for their expertise in statistical analysis. Their involvement has significantly elevated the quality and outcomes of this study.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Endocrinology and metabolism
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Alsaidan A, Saudi Arabia; Horowitz M, Australia S-Editor: Qu XL L-Editor: A P-Editor: Zhao YQ
| 1. | Du L, Li Q, Yi H, Kuang T, Tang Y, Fan G. Gut microbiota-derived metabolites as key actors in type 2 diabetes mellitus. Biomed Pharmacother. 2022;149:112839. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 67] [Article Influence: 22.3] [Reference Citation Analysis (0)] |
| 2. | Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, Stein C, Basit A, Chan JCN, Mbanya JC, Pavkov ME, Ramachandaran A, Wild SH, James S, Herman WH, Zhang P, Bommer C, Kuo S, Boyko EJ, Magliano DJ. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract. 2022;183:109119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3033] [Cited by in RCA: 4820] [Article Influence: 1606.7] [Reference Citation Analysis (36)] |
| 3. | Alicic RZ, Rooney MT, Tuttle KR. Diabetic Kidney Disease: Challenges, Progress, and Possibilities. Clin J Am Soc Nephrol. 2017;12:2032-2045. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1257] [Cited by in RCA: 1797] [Article Influence: 224.6] [Reference Citation Analysis (0)] |
| 4. | Ruiz-Ortega M, Rodrigues-Diez RR, Lavoz C, Rayego-Mateos S. Special Issue "Diabetic Nephropathy: Diagnosis, Prevention and Treatment". J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 83] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 5. | Iatcu CO, Steen A, Covasa M. Gut Microbiota and Complications of Type-2 Diabetes. Nutrients. 2021;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 208] [Article Influence: 52.0] [Reference Citation Analysis (0)] |
| 6. | Tao P, Ji J, Wang Q, Cui M, Cao M, Xu Y. The role and mechanism of gut microbiota-derived short-chain fatty in the prevention and treatment of diabetic kidney disease. Front Immunol. 2022;13:1080456. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 7. | Weickert MO, Pfeiffer AFH. Impact of Dietary Fiber Consumption on Insulin Resistance and the Prevention of Type 2 Diabetes. J Nutr. 2018;148:7-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 309] [Article Influence: 44.1] [Reference Citation Analysis (0)] |
| 8. | Xie Y, Gou L, Peng M, Zheng J, Chen L. Effects of soluble fiber supplementation on glycemic control in adults with type 2 diabetes mellitus: A systematic review and meta-analysis of randomized controlled trials. Clin Nutr. 2021;40:1800-1810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 9. | Reynolds AN, Akerman AP, Mann J. Dietary fibre and whole grains in diabetes management: Systematic review and meta-analyses. PLoS Med. 2020;17:e1003053. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 256] [Cited by in RCA: 293] [Article Influence: 58.6] [Reference Citation Analysis (0)] |
| 10. | Weickert MO, Roden M, Isken F, Hoffmann D, Nowotny P, Osterhoff M, Blaut M, Alpert C, Gögebakan O, Bumke-Vogt C, Mueller F, Machann J, Barber TM, Petzke KJ, Hierholzer J, Hornemann S, Kruse M, Illner AK, Kohl A, Loeffelholz CV, Arafat AM, Möhlig M, Pfeiffer AF. Effects of supplemented isoenergetic diets differing in cereal fiber and protein content on insulin sensitivity in overweight humans. Am J Clin Nutr. 2011;94:459-471. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 121] [Cited by in RCA: 134] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 11. | Wannamethee SG, Whincup PH, Thomas MC, Sattar N. Associations between dietary fiber and inflammation, hepatic function, and risk of type 2 diabetes in older men: potential mechanisms for the benefits of fiber on diabetes risk. Diabetes Care. 2009;32:1823-1825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 105] [Cited by in RCA: 94] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 12. | Yao B, Fang H, Xu W, Yan Y, Xu H, Liu Y, Mo M, Zhang H, Zhao Y. Dietary fiber intake and risk of type 2 diabetes: a dose-response analysis of prospective studies. Eur J Epidemiol. 2014;29:79-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 167] [Cited by in RCA: 180] [Article Influence: 16.4] [Reference Citation Analysis (0)] |
| 13. | Jin F, Zhang J, Shu L, Han W. Association of dietary fiber intake with newly-diagnosed type 2 diabetes mellitus in middle-aged Chinese population. Nutr J. 2021;20:81. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 14. | Partula V, Deschasaux M, Druesne-Pecollo N, Latino-Martel P, Desmetz E, Chazelas E, Kesse-Guyot E, Julia C, Fezeu LK, Galan P, Hercberg S, Mondot S, Lantz O, Quintana-Murci L, Albert ML, Duffy D; Milieu Intérieur Consortium, Srour B, Touvier M. Associations between consumption of dietary fibers and the risk of cardiovascular diseases, cancers, type 2 diabetes, and mortality in the prospective NutriNet-Santé cohort. Am J Clin Nutr. 2020;112:195-207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 81] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 15. | Kwon YJ, Lee HS, Park G, Kim HM, Lee JW. Association of Dietary Fiber Intake with All-Cause Mortality and Cardiovascular Disease Mortality: A 10-Year Prospective Cohort Study. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 13] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 16. | Mirmiran P, Yuzbashian E, Asghari G, Sarverzadeh S, Azizi F. Dietary fibre intake in relation to the risk of incident chronic kidney disease. Br J Nutr. 2018;119:479-485. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 17. | Liu H, Wang L, Chen C, Dong Z, Yu S. Association between Dietary Niacin Intake and Migraine among American Adults: National Health and Nutrition Examination Survey. Nutrients. 2022;14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 93] [Reference Citation Analysis (0)] |
| 18. | American Diabetes Association. Standards of medical care in diabetes--2010. Diabetes Care. 2010;33 Suppl 1:S11-S61. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2231] [Cited by in RCA: 2287] [Article Influence: 152.5] [Reference Citation Analysis (1)] |
| 19. | de Boer IH, Rue TC, Hall YN, Heagerty PJ, Weiss NS, Himmelfarb J. Temporal trends in the prevalence of diabetic kidney disease in the United States. JAMA. 2011;305:2532-2539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 639] [Cited by in RCA: 758] [Article Influence: 54.1] [Reference Citation Analysis (0)] |
| 20. | Inker LA, Eneanya ND, Coresh J, Tighiouart H, Wang D, Sang Y, Crews DC, Doria A, Estrella MM, Froissart M, Grams ME, Greene T, Grubb A, Gudnason V, Gutiérrez OM, Kalil R, Karger AB, Mauer M, Navis G, Nelson RG, Poggio ED, Rodby R, Rossing P, Rule AD, Selvin E, Seegmiller JC, Shlipak MG, Torres VE, Yang W, Ballew SH, Couture SJ, Powe NR, Levey AS; Chronic Kidney Disease Epidemiology Collaboration. New Creatinine- and Cystatin C-Based Equations to Estimate GFR without Race. N Engl J Med. 2021;385:1737-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1478] [Cited by in RCA: 2355] [Article Influence: 588.8] [Reference Citation Analysis (0)] |
| 21. | Matthews DR, Hosker JP, Rudenski AS, Naylor BA, Treacher DF, Turner RC. Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia. 1985;28:412-419. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22373] [Cited by in RCA: 24522] [Article Influence: 613.1] [Reference Citation Analysis (1)] |
| 22. | Davison KM, Temple NJ. Cereal fiber, fruit fiber, and type 2 diabetes: Explaining the paradox. J Diabetes Complications. 2018;32:240-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 57] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 23. | Gowd V, Xie L, Zheng X, Chen W. Dietary fibers as emerging nutritional factors against diabetes: focus on the involvement of gut microbiota. Crit Rev Biotechnol. 2019;39:524-540. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 41] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 24. | Zhao L, Zhang F, Ding X, Wu G, Lam YY, Wang X, Fu H, Xue X, Lu C, Ma J, Yu L, Xu C, Ren Z, Xu Y, Xu S, Shen H, Zhu X, Shi Y, Shen Q, Dong W, Liu R, Ling Y, Zeng Y, Zhang Q, Wang J, Wang L, Wu Y, Zeng B, Wei H, Zhang M, Peng Y, Zhang C. Gut bacteria selectively promoted by dietary fibers alleviate type 2 diabetes. Science. 2018;359:1151-1156. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1058] [Cited by in RCA: 1544] [Article Influence: 220.6] [Reference Citation Analysis (68)] |
| 25. | Snelson M, de Pasquale C, Ekinci EI, Coughlan MT. Gut microbiome, prebiotics, intestinal permeability and diabetes complications. Best Pract Res Clin Endocrinol Metab. 2021;35:101507. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 26. | Cheng X, Zhou T, He Y, Xie Y, Xu Y, Huang W. The role and mechanism of butyrate in the prevention and treatment of diabetic kidney disease. Front Microbiol. 2022;13:961536. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 27. | Lin JR, Wang ZT, Sun JJ, Yang YY, Li XX, Wang XR, Shi Y, Zhu YY, Wang RT, Wang MN, Xie FY, Wei P, Liao ZH. Gut microbiota and diabetic kidney diseases: Pathogenesis and therapeutic perspectives. World J Diabetes. 2022;13:308-318. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 7] [Cited by in RCA: 27] [Article Influence: 9.0] [Reference Citation Analysis (1)] |
| 28. | Li YJ, Ma J, Loh YW, Chadban SJ, Wu H. Short-chain fatty acids directly exert anti-inflammatory responses in podocytes and tubular epithelial cells exposed to high glucose. Front Cell Dev Biol. 2023;11:1182570. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 29. | Li YJ, Chen X, Kwan TK, Loh YW, Singer J, Liu Y, Ma J, Tan J, Macia L, Mackay CR, Chadban SJ, Wu H. Dietary Fiber Protects against Diabetic Nephropathy through Short-Chain Fatty Acid-Mediated Activation of G Protein-Coupled Receptors GPR43 and GPR109A. J Am Soc Nephrol. 2020;31:1267-1281. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 237] [Article Influence: 47.4] [Reference Citation Analysis (0)] |
| 30. | Zhou T, Xu H, Cheng X, He Y, Ren Q, Li D, Xie Y, Gao C, Zhang Y, Sun X, Xu Y, Huang W. Sodium Butyrate Attenuates Diabetic Kidney Disease Partially via Histone Butyrylation Modification. Mediators Inflamm. 2022;2022:7643322. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 31. | Du Y, Yang YT, Tang G, Jia JS, Zhu N, Yuan WJ. Butyrate alleviates diabetic kidney disease by mediating the miR-7a-5p/P311/TGF-β1 pathway. FASEB J. 2020;34:10462-10475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 32. | Ma X, Wang Q. Short-Chain Fatty Acids Attenuate Renal Fibrosis and Enhance Autophagy of Renal Tubular Cells in Diabetic Mice Through the HDAC2/ULK1 Axis. Endocrinol Metab (Seoul). 2022;37:432-443. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 19] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 33. | Evenepoel P, Poesen R, Meijers B. The gut-kidney axis. Pediatr Nephrol. 2017;32:2005-2014. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 209] [Article Influence: 26.1] [Reference Citation Analysis (0)] |