Published online Apr 15, 2023. doi: 10.4239/wjd.v14.i4.396
Peer-review started: December 25, 2022
First decision: January 9, 2023
Revised: January 15, 2023
Accepted: March 20, 2023
Article in press: March 20, 2023
Published online: April 15, 2023
Processing time: 108 Days and 0.5 Hours
The global prevalence of obesity is increasing rapidly with an exponential rise in incidence of type 2 diabetes mellitus in recent years. ‘Diabesity’, the term coined to show the strong interlink between obesity and diabetes, is the direct cons-equence of the obesity pandemic, and poses significant challenges in the management of the disease. Without addressing the clinical and mechanistic complications of obesity such as metabolic-associated fatty liver disease and obstructive sleep apnoea, a rational management algorithm for diabesity cannot be developed. Several classes of anti-diabetic medications including insulins, sulphonylureas, thiazolidinediones and meglitinides are associated with the risk of weight gain and may potentially worsen diabesity. Therefore, appropriate selection of antidiabetic drug regimen is crucial in the medical management of diabesity. The role of non-pharmacological measures such as dietary adjustments, exercise interventions and bariatric procedures should also be emphasised. Unfortunately, the importance of appropriate and optimal management of diabesity is often overlooked by medical professionals when achieving adequate glycemic control which results in inappropriate management of the disease and its complications. This review provides a narrative clinical update on the evidence behind the management of diabesity.
Core Tip: The worldwide prevalence of diabesity is increasing exponentially because of the global obesity pandemic in the past few decades. This is also expected to increase the prevalence of other associated chronic disorders such as hypertension, dyslipidemia, metabolic-associated fatty liver disease (MAFLD), obstructive sleep apnoea (OSA), and cardiovascular disease. Rational management of diabesity involves prompt control of diabetes while also optimally addressing other comorbidities such as obesity, hypertension, dyslipidemia, MAFLD, OSA and cardiovascular risk. Appropriate selection of the antidiabetic therapeutic agents and early planning of metabolic surgery when indicated are crucial to scientifically approach patients with diabesity. This evidence-based review addresses the key issues regarding management of diabesity.
- Citation: Michaelidou M, Pappachan JM, Jeeyavudeen MS. Management of diabesity: Current concepts. World J Diabetes 2023; 14(4): 396-411
- URL: https://www.wjgnet.com/1948-9358/full/v14/i4/396.htm
- DOI: https://dx.doi.org/10.4239/wjd.v14.i4.396
Obesity has become a global pandemic over the past three decades because of the increase in prevalence of adverse lifestyle behaviours such as over-consumption of processed food and physical inactivity in the latter half of twentieth century. Consequently, a tsunami of more than fifty obesity-related problems has emerged which threatens human health and wellbeing on a global level. 800 million people around the world at present live with obesity, with an estimated health expenditure of one trillion United States dollars budgeted for managing obesity-related conditions in the year 2025[1]. There is also an expected increase in childhood obesity to 250 million by the year 2030. Unless there is a massive global action plan from health agencies and administrators to curtail the obesity pandemic, this is likely to continue to significantly impact individuals and healthcare systems.
The worst metabolic consequence of obesity is type 2 diabetes mellitus (T2DM), which substantially increases cardiovascular disease-related morbidity and mortality. Other obesity associated com-orbidities such as dyslipidemia, hypertension, metabolic-associated fatty liver disease (MAFLD), obstructive sleep apnoea (OSA), heart failure, ischaemic heart disease, cancers and osteoarthritis further increase the morbidity, mortality, and health-related quality of life, especially when these patients additionally have T2DM. The term “diabesity” was coined by Sims et al[2] to describe the very strong pathophysiological link between diabetes and excess body weight in 1970s. Visceral adiposity, which leads to insulin resistance, is the putative mechanism in the development of diabesity. Therefore, an ideal treatment strategy for the disease should also include optimal management of obesity[3-5]. The real-world approach to the management of diabetes often ignores these crucial issues by simply focusing on glycaemic control. This manuscript provides an up-to-date overview of diabesity, in terms of pathophysiology and management. Particularly, we have reviewed and summarised newly published data from clinical trials that have shown the significant impact of glucagon-like peptide-1 (GLP-1) and sodium glucose cotransporter-2 (SGLT-2) inhibitors on diabesity. Therefore, we elaborate the conceptual, pathobiological, mechanistic, and therapeutic aspects of diabesity in this comprehensive review.
The link between obesity and diabetes has been described in numerous studies to date. The key pathophysiological process is thought to be the development of insulin resistance, which, in turn, is associated with significant cardiovascular and metabolic morbidities, such as the development of T2DM[6]. Insulin resistance is described as inefficient glucose metabolism despite adequate insulin secretion[7]. In the presence of insulin resistance, a variety of insulin-sensitive tissues including the liver, skeletal muscle, as well as adipose tissue exhibit altered biochemical and cellular pathways[8].
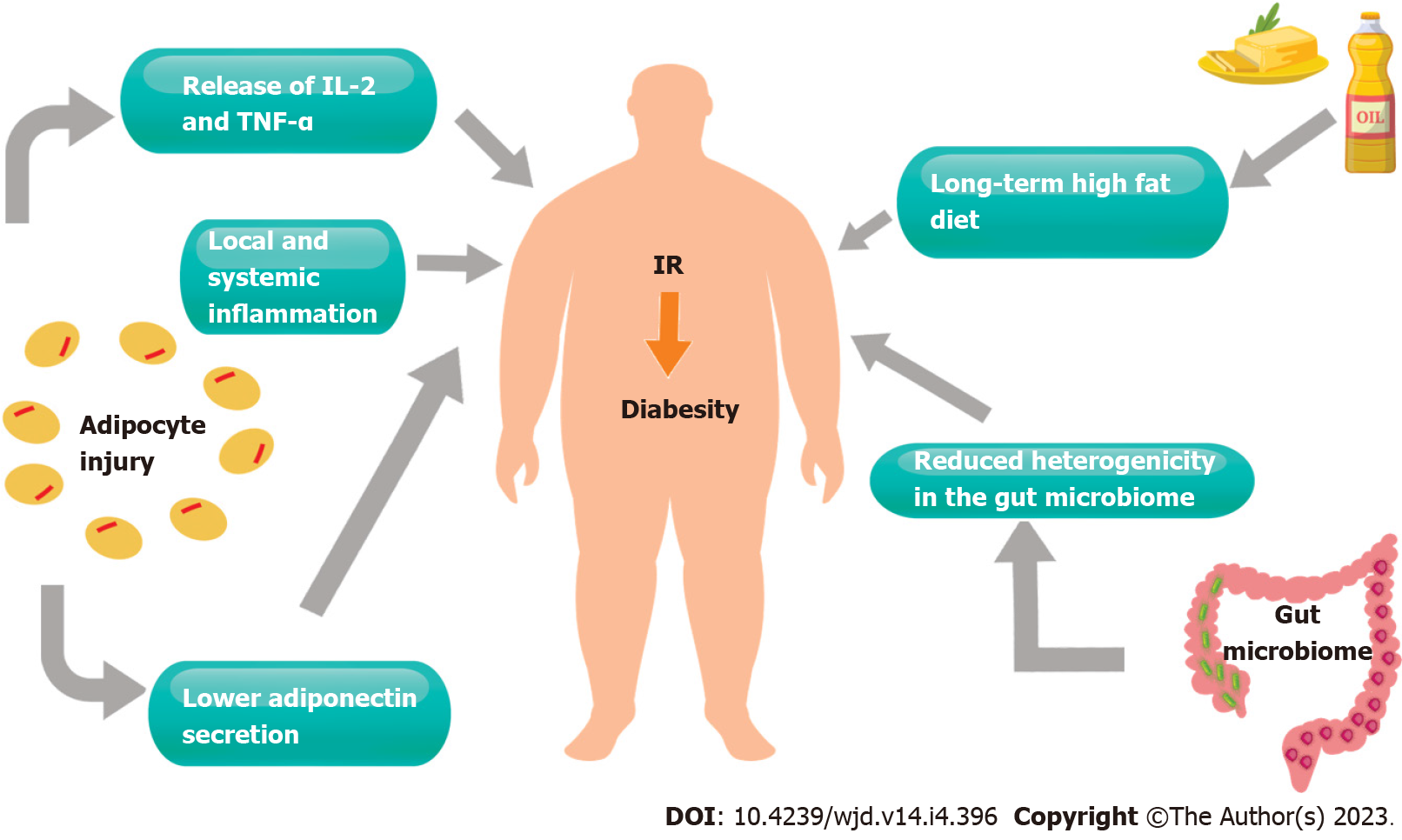
There is a plethora of proposed pathophysiological processes for the development of diabetes in obesity. Perhaps the most described pathophysiological theory is that of chronic inflammation, with adipocytes shown to secrete a variety of pro-inflammatory substances including interleukin-6, tumor necrosis factor alpha and inflammatory cytokines, which, in turn, result in local and systemic inflammatory processes[9]. The resulting oxidative stress in adipocytes has been associated with lower adiponectin secretion, a protein that has been linked to improved glucose regulation and reduced incidence of metabolic syndrome[10].
Another proposed mechanism is that of excess circulating lipid substrates, such as fatty acids, due to excessive consumption through a high fat diet, which also results in chronic inflammation and oxidative stress[11,12]. A long-term high fat diet may result in lipid deposition in tissues that would not normally be targeted by insulin. Triglyceride accumulation in the liver and skeletal muscle has also been shown to promote insulin resistance through impaired insulin signaling[13]. More recent literature describes the potential association between mitochondrial dysfunction, particularly in skeletal muscle, and diabetes. Mitochondrial dysfunction may be a consequence of pro-inflammatory stress and oxidation, affecting the organelle function leading to insulin resistance, at least in skeletal muscle tissues[14]. Further research is required to characterise the interplay between energy metabolism and the development of diabetes at mitochondrial level.
Recent publications highlight the impact of gut bacteria on obesity and insulin resistance. The gut microbiome has been shown to have a significant impact on nutrient metabolism. Individuals with reduced heterogeneity in their gut microbiome have been found to be at a greater risk of obesity and insulin resistance[15]. Linking to excess fat consumption, research suggests that excess lipid intake may result in bacterial production of short chain fatty acids, which in turn affect energy balance and metabolism[16,17] (Figure 1).
Metabolic dysfunction, and the inflammatory processes in obesity and T2DM, have been closely associated with non-alcoholic fatty liver disease. This has prompted the use of the new term, MAFLD, given the significant link between fatty liver disease and insulin resistance[18,19]. A variety of pathophysiological processes, as described in the previous section, result in excess adipose tissue accumulation in the liver. As a result, hepatic cellular damage, oxidative stress, and insulin resistance develop in such cases. A study investigating the presence of fatty liver disease vs simple hepatic steatosis in patients with T2DM or metabolic syndrome, revealed that almost all diabetic patients showed evidence of steatohepatitis on liver biopsy, without necessarily showing derangement in their liver function (98.6% of patients with T2DM vs 58.5% of patients with metabolic syndrome, P < 0.0001). This suggests that MAFLD may be one of the early end-organ complications of T2DM and metabolic syndrome, with early occult onset and progression without any clinical signs[20]. Hence early identification and management may have a significant impact on mortality and morbidity. MAFLD has also been directly associated with increased cardiovascular morbidity and both micro- and macrovascular complications in diabetic patients, further solidifying the need for early identification and management[21].
OSA involves partial or complete obstruction of the airway during sleep, which results in transient hypoxaemia, sleep restriction and reduction in intrathoracic pressures[22]. These have been linked to the activation of the hypothalamic pituitary adrenal axis resulting in excess stress hormone release and increased cardiovascular morbidity. OSA and hypoxia have also been linked to alterations in adipokine levels as well as oxidative stress. All, in turn, lead to the accumulation of excess adipose tissue and the development insulin resistance[23]. The reverse has also been reported, i.e., OSA could be aggravated by T2DM and obesity[23,24]. One could argue that a patient with both conditions would be at risk of significant morbidity, with diabetes that would be challenging to control without optimising their sleep apnoea. Sleep apnoea and T2DM have both been associated with increased cardiovascular morbidity and mortality, hence a combination of the two may prove more detrimental[25]. Evidence suggests that the prolonged use of continuous positive airway pressure ventilation (CPAP), other than improving sleep quality and reducing hypoxaemia, may also improve glucose tolerance and insulin sensitivity in obese individuals[26-28]. A study of 40 T2DM patients treated with CPAP for 3 mo, assessed the effect of CPAP therapy on insulin resistance using hyperinsulinaemic euglycaemic clamp. Results revealed significant improvement in insulin sensitivity, as early as 2 d after commencing treatment with CPAP (5.75 ± 4.20 vs 6.79 ± 4.91 micromol/kg/min at 2 d; P = 0.003). The improvement persisted after 3 mo of CPAP therapy[28]. Hence, early identification and treatment of OSA in diabesity patients with CPAP may help to reduce cardiovascular complications and improve glycaemic control.
It has been proposed that diabesity significantly contributes to endothelial damage and heart failure, which are both exacerbated by dyslipidaemia. Through the mechanism of altered mitochondrial energy expenditure as previously described, diabesity may have a direct effect on mitochondrial function in cardiac muscle. This may result in inefficient energy production and expenditure, causing cardiac stress[29]. Additionally, it has been reported that patients with diabetes exhibit cardiac oxidative stress and poor utilisation of energy substrates, in turn causing dysfunction in cardiac muscle contraction[29,30]. Increased consumption of fats, which results in increased circulatory fatty acids, may also contribute to cardiac steatosis which will also impair cardiac function. Studies using cardiac magnetic resonance (MR) spectroscopy have demonstrated cardiac steatosis, both in patients with diabetes, as well as in those with obesity[31]. Hence diabesity may have a direct link to structural alterations and stress of cardiac muscle, leading to heart failure. This is further supported by the Framingham study, which also demonstrated that an elevated glycated haemoglobin (HbA1c) level was associated with more severe heart failure[32].
The association between diabetes, obesity and macrovascular disease has been well established through the years[33,34]. Burke et al[34] demonstrated that obese individuals without metabolic complications had a significantly higher coronary calcium score when compared to non-obese patients (17% increase in risk of higher coronary artery calcium scores). This suggests that obesity alone, even without the presence of diabetes, would result in increased coronary artery disease risk. Kronmal et al[35] investigated the association between coronary artery calcium progression and a multitude of risk factors which included T2DM in 5756 individuals. Results demonstrated that T2DM, particularly of longer duration, was associated with progression of coronary artery calcium. This suggests that patients with diabetes are at significantly increased risk of myocardial events[35]. Therefore, in the diabesity epidemic, it would be of paramount importance that the patient’s cardiovascular risk is stratified and addressed early on, in order to prevent significant cardiovascular events, perhaps with coronary artery calcium assessment. Recent advances in cardiac imaging modalities, including cardiac MR and computed tomography coronary angiography, may allow for early identification and management of coronary artery disease in diabesity patients as shown in recent trials[29,36].
It has been suggested that the development of diabesity-related kidney disease may be mediated through insulin resistance. Insulin is essential for normal glomerular function and podocyte biology, and dysfunctional insulin signaling has been shown to dysregulate vascular endothelial growth factor A signaling pathway and affects glucose transport[37]. Studies demonstrated podocyte damage, glomerulosclerosis, and albuminuria occur in patients with insulin resistance[38]. In more recent years, studies have also suggested that proximal tubule cells have a role in nutrient regulation in the kidney. Excessive amounts of glucose, fatty acids, and amino acids present in the proximal tubule due to obesity or diabetes, will cause dysregulation in the relevant pathways normally protecting kidney, and consequently result in tubular injury, fibrosis, and inflammation[37-39]. Therefore, medications such as SGLT-2 inhibitors may help to prevent the presence of excessive nutrients in proximal tubule cells, due to induction of glycosuria, and therefore help to minimise tubular damage through this mechanism[40].
As mentioned earlier, T2DM and obesity are inter-linked and share common pathophysiological mechanisms of disease origin, especially adverse lifestyle behaviours. Hence, the first step in the management of diabesity, prior to medical intervention would be to attempt weight loss via lifestyle modifications such as changes in diet and exercise. Multiple studies have shown that weight loss can induce remission of T2DM. It has been suggested that weight loss of 15 kg can induce remission of T2DM in around 70% of individuals[41,42]. Achieving weight loss through dietary restriction can be difficult and challenging, and ultimately, it aims to reverse the mechanisms induced by nutrient overconsumption. Therefore, setting realistic stepwise targets may aid patients in achieving sustained weight loss. Smaller degrees of weight loss such as 5 kg have been shown to decrease fasting blood glucose and improve the insulin sensing ability of adipose, skeletal muscle and liver tissues. Stepwise incremental weight loss has been shown to result in a gradual improvement in HbA1c[43].
Larger randomised controlled trials have been conducted in this field, such as the look-AHEAD trial, which demonstrated that remission of diabetes correlated with the degree of weight loss and was inversely correlated with the duration of T2DM[44]. A similar primary care study in the United Kingdom, DiRECT, randomised patients in a control group (standard diabetes care) or a structured weight management program. The weight management program group showed greater weight loss [10 kg vs 1 kg, adjusted difference -8.8 kg, 95% confidence interval (CI): -10.3 to -7.3; P < 0.0001], greater incidence of diabetes remission (46% vs 4%, odds ratio = 19.7, 95%CI: 7.8 to 49.8; P < 0.0001), with the degree of weight loss correlating to the improvement in diabetes management[45]. However, large scale trials mainly involved short-term high intensity weight loss programmes. We could argue that a long-term, permanent change in diet would be desired to achieve sustainable benefits in our patient cohort. Ajala et al[40] has reviewed the beneficial effects of possible dietary modifications in overweight patients with diabetes. Their study demonstrated that a lower carbohydrate, lower glycaemic index, Mediterranean style diet and high protein diet led to significant improvement in glycaemic control [HbA1c change of -0.12% (P = 0.04), -0.14% (P = 0.008), -0.47% (P < 0.00001), and -0.28% (P < 0.00001), respectively][46]. Mediterranean and high protein diets have also been shown to reduce cardiovascular risk in diabetes patients and should be considered[46,47]. More recent research (Ozsoy et al[48], 2022) investigated the possibility of modulating gut microbiota through diet, hence altering neurotransmitter pathways of satiety and insulin sensitivity. It was suggested that perhaps the Mediterranean diet would be successful in achieving a varied balance in the gut microbiome.
Regular physical activity, regardless of the associated weight loss, has been shown to improve HbA1c and insulin sensitivity. Exercise comes with a variety of benefits including reduction in visceral adipose tissue, improvement in lipids and blood pressure and hence a reduction in the cardiometabolic risk[49,50]. Effects seem to be proportional to energy expenditure, i.e., total calories burnt, rather than the type of exercise itself[50,51]. Therefore, exercise programmes should be personalised and tailored to patient preference and co-morbidities. Despite the improvement in biochemical parameters, exercise alone does not seem to lead to the remission of diabetes[49-52], therefore a combined diet and exercise programme is recommended in order to achieve the best possible outcome. Results from the Malmö study further support this, where patients on a combined diet and exercise modification programme demonstrated long-term sustainable benefits, with half the T2DM patients in remission at 5 years. Improvement in glycaemic control was proportional to the degree of weight loss (r = 0.19, P < 0.02) and also to change in fitness levels (r = 0.22, P < 0.02)[53].
Metformin is one of the first-line medications for the management of T2DM and is known for its good safety profile and low cost. Metformin works by inhibiting hepatic gluconeogenesis, improving insulin sensitivity in skeletal muscle and reduction of appetite, although its mechanism of action is not yet fully understood[54,55]. It is one of the antidiabetic medications recommended globally as first-line therapy for T2DM[56]. Previous studies have demonstrated significant weight loss benefits, ranging between 0.6 to 2.9 kg[5]. Wu et al[57] has proposed that, in addition to the known mechanisms of action of metformin, this medication may also positively affect the composition of the gut microbiome and may potentially lead to improved glucose metabolism in the gut, resulting in weight loss. Day et al[58] has suggested that metformin also increases the expression of growth differentiating factor 15 (GDF-15), which results in appetite suppression and weight loss. However, more in vivo studies are required to further investigate this association. Multiple health benefits have been demonstrated with metformin use, which include reversibility of hepatic steatosis and a lipid lowering effect. It has been suggested that these are indirect effects of weight loss associated with metformin, rather than a direct effect on hepatic lipid metabolism and storage[59]. A recent review by Lv et al[60] looking into the clinical benefits of metformin, demonstrated significant benefits in a variety of health issues including chronic kidney disease, cardiovascular disease and several cancers by preventing growth and metastasis of tumor cells and the effect on the gut microbiome[60-62].
Insulin therapy is recommended in patients with significantly elevated HbA1c, where the aim is rapid and effective optimisation of glycaemic control. This may be the case in patients with significant diabetic complications or patients requiring urgent surgical procedures[5]. However, insulin therapy has been associated with significant weight gain, especially when compared to other hypoglycaemic agents. Weight gain ranging between 1.56-5.75 kg has been reported with insulin use, which may make clinicians reluctant to prescribe it in overweight/obese diabetic patients[63]. It has been previously demonstrated that the dose as well as the type of insulin may influence the degree of weight gain in diabetic patients. Greater doses of insulin have been associated with greater weight gain[64]. Basal insulin has been shown to have a lesser effect on weight than pre-mixed insulin[65]. Freemantle et al[66] suggested that the use of glargine insulin resulted in significantly less weight change when compared to pre-mixed insulin. Results when using glargine were comparable with detemir, degludec, and neutral protamine Hagedorn insulin. Therefore, careful consideration, with joint decision making between patients and specialists, is recommended when prescribing insulin in patients with diabesity, to minimise the risk of weight gain while achieving the benefits of rapid optimisation of glycaemic control.
Sulfonylureas are generally not recommended in patients with obesity, due to significant risk of hypoglycaemia, weight gain, reduced efficacy, and the need for careful dose titration in patients with renal disease[67]. Hypoglycaemia risk has been reported to vary between sulfonylurea agents, with gliclazide having the lowest reported incidence among this class of medications[68]. Usually prescribed in combination with other agents, sulfonylureas have been reported to result in weight gain of approximately 2.01-2.3 kg[63,69]. Therefore, sulfonylureas appear to pose significant risk of hypoglycaemia, as well as weight gain, and poorer outcomes in patients with diabesity. Furthermore, recent large randomised controlled trials did not show any significant change in cardiovascular risk, in patients treated with sulfonylureas vs other oral antihyperglycaemic agents such as pioglitazone or linagliptin[70-72].
Thiazolidinediones are the only oral antihyperglycaemic agents that specifically target and improve the phenomenon of insulin resistance. Through the activation of the peroxisome proliferator-activated receptor gamma system, they increase glucose utilisation, decrease hepatic steatosis, and decrease circulating free fatty acids[73]. These oral agents, despite having a low risk of hypoglycaemia, and relatively favourable efficacy in HbA1c reduction, have been also associated with significant weight gain between 2.3-4.25 kg[63]. Weight gain in patients on this class of medication is not entirely attributed to the deposition of adipose tissue, as patients may experience fluid retention as a side effect of thiazolidinedione use, which also leads to apparent weight gain[74]. A recent review (Lebovitz et al[73]) summarised the different trials looking into the effects of pioglitazone and rosiglitazone on cardiovascular risk. It has been suggested that both medications have been superior to sulfonylureas, with pioglitazone showing a more favourable efficacy and safety profile.
Hesitation to prescribe these agents has also risen from previously reported risk of bladder malignancy, osteoporosis, and heart failure. More recent literature (Lewis et al[75]) suggests that these risks may not be as grave as previously thought. Recent retrospective follow-up studies of patients diagnosed with bladder cancer, did not find a significant association between pioglitazone use and the development of bladder malignancy[75,76]. However, the literature relating to thiazolidinedione use and fracture risk confirms increased risk of fractures in female patients, regardless of menopausal status. The TOSCA-IT study did not find a significantly increased risk of fractures when comparing patients on pioglitazone vs sulfonylurea therapy[70]. Although it has been proven that thiazolidinediones increase the incidence of heart failure, there is no causal link to progression, severity, or death from heart failure in such patients[73]. Therefore, overall data may suggest superiority of this class of medication over sulfonylureas in patients diabesity especially when they have co-existing MAFLD.
Dipeptidyl peptidase-4 (DPP-4) agents are considered as “weight neutral” agents, with minimal effect on overall body weight[63]. This class of medications works by inhibiting the breakdown of GLP-1 and glucose dependent insulinotropic peptide (GIP) in the gut, maximising the duration of action of these incretin hormones[5]. They have been reported to positively impact HbA1c control, with about an overall 0.75% reduction when prescribed alongside other antihyperglycaemics[77]. This class of medications is not generally used as monotherapy, rather than in combination with other oral antihyperglycaemic agents. The addition of DPP-4 inhibitors to metformin therapy, showed favourable weight outcomes when compared to the addition of sulfonylureas or thiazolidinedione in recent trials[78]. However, caution needs to be exercised when prescribing this class of medications to patients with cardiovascular disease, as the use of DPP-4 inhibitors has been shown to increase the incidence of hospitalisation from heart failure. Saxagliptin, vidagliptin and alogliptin have all been associated with an increased incidence of hospitalisation for heart failure in diabetic patients[79]. It is thought that clinical deterioration is evident shortly after treatment is initiated, and this requires careful titration and clinical monitoring.
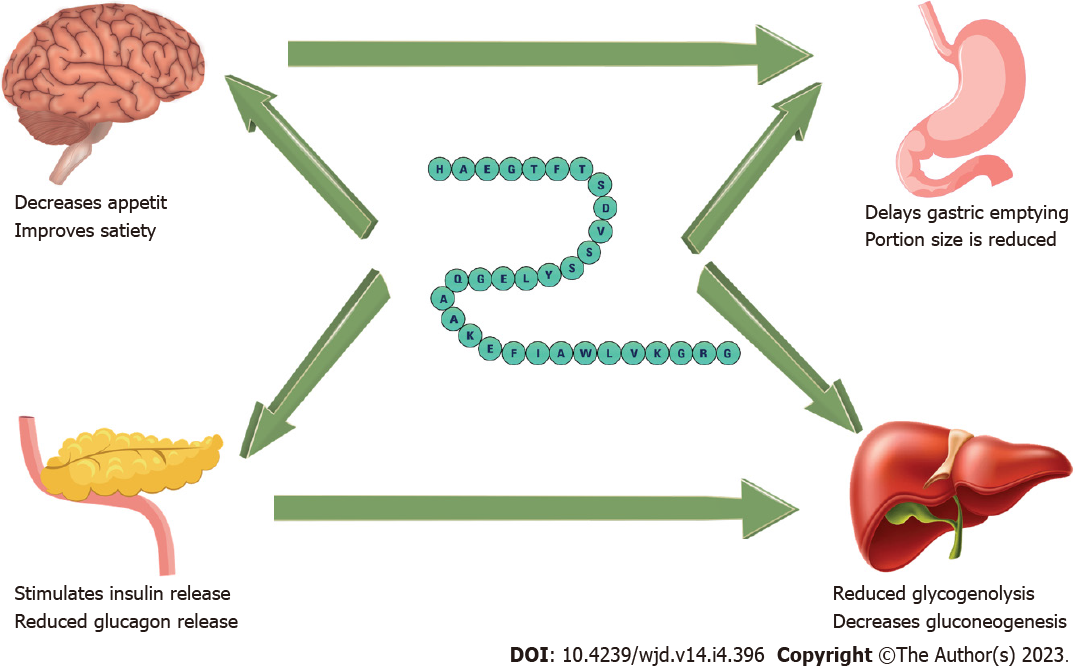
GLP-1 receptor agonists work by the stimulation of insulin secretion and suppression of glucagon production. Furthermore, they modulate receptors in the gut to delay gastric emptying, as well as neurons involved in appetite regulation. These mechanisms lead to reduced appetite and improved feeling of satiety, hence contributing to weight loss[63]. Current preparations include subcutaneous injections and oral tablets. Injections vary in frequency, from daily (exenatide, lixisenatide, and liraglutide) to once weekly (exenatide-extended release, dulaglutide, albiglutide, and semaglutide). A recently approved oral form of semaglutide is also licensed for use, which has shown comparable effects to the subcutaneous semaglutide preparation[80].
There have been multiple research trials with significant data, surrounding the use of GLP-1 inhibitors in diabetes, as well as obesity alone, in the recent years, as summarised in Table 1. Data has shown an association with significant weight loss, ranging between 1.14-6.9 kg, and an improvement in HbA1c between 1%-2%[63]. High dose liraglutide has been approved for use in the management of obesity. Semaglutide, has also recently been shown to be beneficial in the management obese non-diabetic patients. A recent study (Wilding et al[81]) showed that the use of 2.4 mg semaglutide alongside lifestyle intervention, resulted in sustainable weight loss after 68 wk (-14.9% for semaglutide vs -2.4% for placebo). However, a significant percentage of patients in the semaglutide group had to discontinue therapy due to side effects (4.5% for semaglutide vs 0.8% for placebo). Research has also demonstrated that the improvement in glycaemic control achieved by GLP-1 agonists was comparable to that of insulin[82]. Therefore, GLP-1 agonists should be the first-line injectable therapy for consideration in diabesity patients, with remarkable benefits on body weight while also resulting in improved glycaemic control.
| Drug molecule | Mean weight reduction in kg (95%CI) | Mean % HbA1c reduction (95%CI) | Follow-up period (yr) |
| Exenatide (weekly) | -1.27 (-1.4 to -1.13) | -0.53 (-0.57 to -0.50) | 3.2 |
| Liraglutide | -2.3 (-2.5 to -2.0) | -0.40 (-0.45 to -0.34) | 3.8 |
| Lixisenatide | -0.7 (-0.9 to -0.5) | -0.27 (-0.31 to -0.22) | 2.1 |
| Albiglutide | -0.83 (-1.06 to -0.60) | -0.52 (-0.58 to -0.45) | 1.5 |
| Dulaglutide | -1.46 (-1.67 to -1.25) | -0.61 (-0.65 to -0.58) | 5.4 |
| Semaglutide | -2.87 (-3.47 to –2.28) for 0.5 mg | -0.66 (-0.80 to – 0.52) for 0.5 mg | 2.0 |
| -4.5 (-4.94 to -3.75) for 1 mg | -1.05 (-1.19 to -0.91) for 1 mg |
Additionally, GLP-1 agonists have been shown to have significant systemic benefits, including reduction in cardiovascular risk in high-risk patients[83], and reduction in urinary albumin excretion. The 2019 REWIND trial investigated the effect of dulaglutide on renal outcomes, and reported improved morbidity, particularly a reduction in the incidence of new macroalbuminuria, and slower renal function decline[84]. Another trial conducted by Mann et al[85], comparing liraglutide with placebo, showed reduced rates of development of renal disease (hazard ratio = 0.78; 95%CI: 0.67 to 0.92; P = 0.003), particularly macroalbuminuria (hazard ratio = 0.74; 95%CI: 0.60 to 0.91; P = 0.004).
Figure 2 shows the mechanisms of actions of GLP-1 agonists in improving diabesity. A concern with the use of GLP-1 agonists is the high rates of discontinuation by patients, often due to pronounced gastrointestinal side effects of nausea, diarrhoea or vomiting which are commonly reported. Mostly, these are self-limiting presentations that resolve with time[80]. Furthermore, the use of GLP-1 agonists was previously thought to be associated with pancreatitis, and the development of certain types of cancer, such as pancreatic and thyroid cancer[86]. However, a meta-analysis by Abd El Aziz et al[87] argues that there is no significant risk of developing pancreatitis or malignancy secondary to GLP-1 agonist therapy. It is also thought that GLP-1 agonists are expected to cause a modest rise in lipase and amylase, which occasionally led to the misdiagnosis of pancreatitis in the previously reported data.
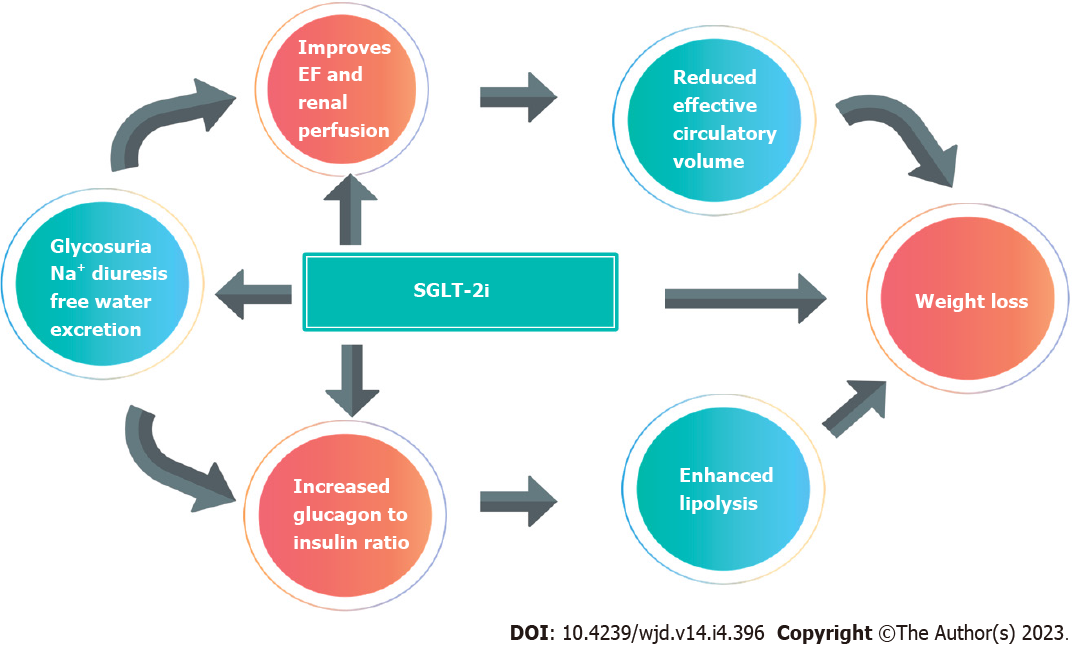
Use of SGLT-2 inhibitors have been an important area of research in the last few years in many clinical trials, with great developments and changes in diabetes, cardiovascular and renal guidelines recently (Table 2). Their main mechanism of action is the reduction of glucose reabsorption in the renal tubules, resulting in increased glucose excretion in the urine[88]. They have been associated with a reduction in HbA1c of about 0.69% and a weight loss between 0.9-2.5 kg[63]. Weight loss is thought to occur due to caloric deficit from impaired glucose reabsorption from the kidney. Studies have shown improvement in waist circumference, central obesity, and visceral adiposity, all of which are features of diabesity as well as metabolic syndrome[89].
| Ref. | Drug molecule | Mean weight reduction in kg (SE/95%CI) | Mean % haemoglobin reduction (95%CI) | Follow-up |
| Neal et al[104], 2017 (CANVAS) | Canagliflozin | -1.60 (- 1.70 to -1.51) | -0.58 (-0.61 to -0.56) | 3.5 yr |
| Wiviott et al[105], 2019 (DECLARE) | Dapagliflozin | -1.80 (-2.00 to -1.70) | -0.42 (-0.45 to -0.40) | 4.2 yr |
| Häring et al[106], 2014 (EMPA-REG) | Empagliflozin | -1.63 (-2.11 to -1.5) for 10 mg | -0.57 (-0.70 to -0.43) for 10 mg | 24 wk |
| -2.01 (-2.49 to -1.53) for 25 mg | -0.64 (-0.77 to -0.50) for 25 mg | |||
| Rosenstock et al[107], 2018 (VERTIS-MET) | Ertugliflozin | -3.00 (-3.30 to -2.70) for 5 mg | -0.70 (-0.90 to -0.50) for 5 mg | 24 wk |
| -2.90 (-3.20 to -2.60) for 15 mg | -0.90 (-1.00 to -0.70) for 15 mg | |||
| Dagogo-Jack et al[108], 2018 (VERTIS-SITA) | Ertugliflozin | -2.0 (-2.7 to -1.4) for 5 mg | -0.70 (-0.90 to -0.50) for 5 mg | 26 wk |
| -1.7 (-2.4 to -1.1) for 10 mg | -0.80 (-0.90 to -0.60) for 10 mg |
There has been extensive research into the role of SGLT-2 inhibitors in improving diabetes, cardiovascular and renal morbidity. A recent meta-analysis by Giugliano et al[90] revealed a reduction in hospitalisation or death from heart failure by 23%, with significantly reduced risk in every trial included. Results were comparable in patients with and without diabetes, indicating that SGLT-2 inhibitors have significant cardiovascular benefits in both groups of patients. Therefore, significant cardiovascular risk may be observed in diabesity patients.
Another meta-analysis of 8 trials, demonstrated the benefits on renal mortality and morbidity through the use of SGLT-2 inhibitors, with an overall reduced risk by 35%[91]. Data was strongest for canagliflozin, with significantly positive results in every trial[90,91]. The EMPEROR-R trial further demonstrated that empagliflozin improved renal outcomes in patients with reduced ejection fraction or other types of heart failure[92].
The main risks associated with the use of SGLT-2 inhibitors include increased incidence of urinary tract infections and genital thrush, due to glycosuria, as well as euglycaemic ketoacidosis. However, significant benefits have been reported in obesity, renal and cardiovascular health, making this class of medication one of the most important in the management of diabetes and obesity, whether they are in co-existence or not. Patient counselling will help to identify and manage side effects promptly and efficiently (Figure 3).
Amylin is a pancreatic hormone secreted in response to the presence of nutrients in the gut. It modulates calcitonin and amylin receptors, and thereby regulates energy utilisation as well as body weight. This is achieved through the enhancement of satiety and delay in gastric emptying. Treatment with amylin analogues is not currently licensed in England but licensed and used abroad. It involves mealtime injections, similar to insulin[93]. Pramlintide, a type of amylin analogue has been associated with a reduction in HbA1c and also a weight loss of 1.5-2.5 kg[63]. Several studies have shown a significant reduction in body weight, with or without the presence of diabetes. However, most of these trials are pre-clinical and further research is needed to further evaluate the beneficial effects and safety profile of amylin analogues in day-to-day clinical practice[93].
In recent years, diabetic medications have been used more and more in the management of obesity. As discussed above, metformin, GLP-1 analogues and SGLT-2 inhibitors show significant weight loss effects. Amylin analogues, although not globally licensed, also demonstrate a beneficial weight loss effect. The recent literature mentions the discovery of GLP/GIP/glucagon receptor poly-agonists, with affinity to more than one receptor[94]. While most medications in this category are still in the experimental phase, a GLP1/GIP poly-agonist, Tirzepatide, demonstrated to have significant effects on weight loss in a recent phase 3 trial. This is a medication administered via weekly subcutaneous injection. This medication has recently been licensed for use in obese patients with T2DM[95].
Current developments in anti-obesity drugs seem to target leptin, ghrelin, GDF-15 pathways. Leptin sensitisers show promising results in patients with genetic obesity, although there is no significant weight reduction in obese patients without genetic cause, such as most diabesity patients[94]. Combination therapy with amylin analogues (pramlintide) has shown greater effects in weight reduction in obese rats (12.7%, P < 0.01)[96]. Hence the role of leptin sensitisers in managing diabesity is currently very limited and requires further investigation. Melanocortin 4 receptors (MC4R), stimulated by leptin to regulate energy expenditure, are other potential targets of novel anti-obesity medications. MC4R agonists, particularly setmelanotide, were licensed in the treatment of genetic obesity, but again, their effect on polygenic obesity is not clear at this stage[94]. Treatments involving ghrelin modulation, a hormone that stimulates appetite, adipose tissue deposition and insulin secretion, are still under trials with no significant benefits demonstrated so far[94]. GDF-15 has been linked to metabolic disease, cardiovascular disease, and cancer. As mentioned earlier, metformin modulation of GDF-15 levels is a proposed mechanism of weight loss[58]. Exogenous administration of GDF15 has been shown to reduce body weight, however evidence comes from animal studies, therefore further research is required in this area[94].
Endoscopic bariatric surgery is a rapidly developing intervention that has revolutionised bariatric surgery. Endoscopic methods target the stomach, and through a variety of techniques aim to restrict gastric capacity, emptying or food absorption[97,98]. Intragastric balloon treatment (IGB) is the most widely used device since its introduction in 1982. IGB is generally recommended for patients with a body mass index (BMI) between 35-40 kg/m2, or as an interim measure, with an aim for more definitive bariatric surgery, in patients with BMI > 40 kg/m2. Studies have shown a BMI reduction of 5.7 kg/m2 and improvement in diabetes and cardiovascular parameters with IGB[99]. Further techniques have been developed more recently, such as endoscopic sleeve surgery, where the capacity of the stomach is reduced. In this procedure, sutures are applied along the gastric wall, with the help of endoscopic equipment. Studies showed a 18% total body weight loss at 24 mo, with relatively low adverse effect profile[100].
Bariatric surgery continues to be the most effective mode of definitive treatment for marked weight loss and diabesity. A recent review by Roth et al[101] reviewed the current literature around weight loss and the side effect profile depending on the type of metabolic surgery. Duodenal switch was associated with the most weight loss, of 70%-80% at 2 years, and resolution of obesity and diabetes related co-morbidities. Roux-en-Y bypass and sleeve gastrectomy showed similar reduction in BMI, ranging between 60%-70%, with studies failing to demonstrate superiority of one over the other. Lower rates of diabetes remission were observed in insulin dependent diabetics following bariatric surgery compared to patients on oral agents, with rates of 16% vs 56% at 12 years. In terms of cardiovascular disease, metabolic surgery was shown to improve 5-year survival from coronary artery disease, in diabetic patients who are insulin treated[101]. Robotic assisted surgery is becoming increasingly more prominent, with data so far showing similar outcomes in robotic and laparoscopic bariatric procedures[102].
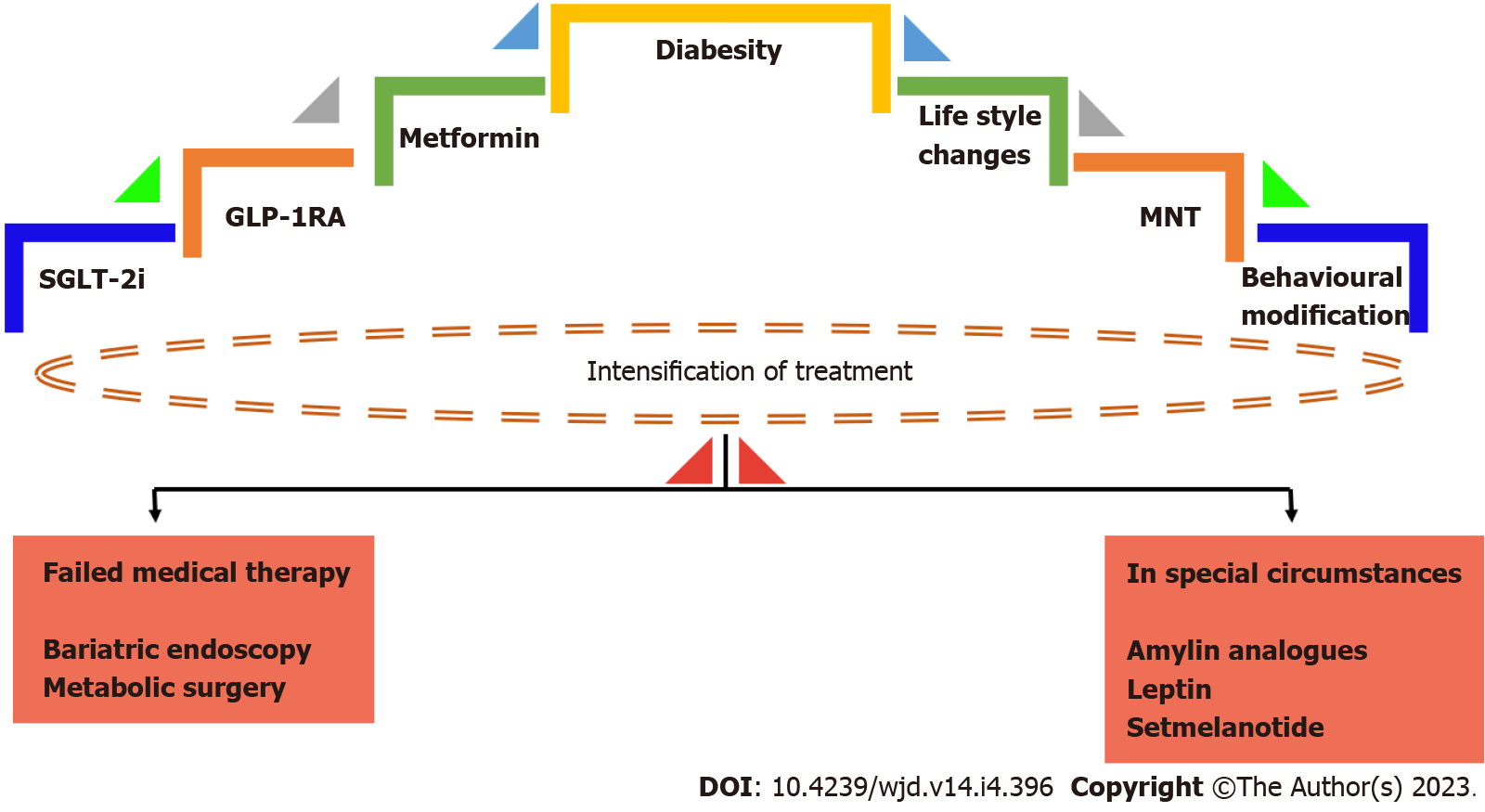
The last few years have seen significant developments in the management of obesity and diabetes. Numerous trials revealed the benefits of SGLT-2 inhibitors and GLP-1 agonists in both achieving good glycaemic control as well as weight loss as summarised earlier. Ongoing research and developments in anti-obesity pharmacopeia show promising results, with anti-obesity medication targeting various hormones involved in the pathophysiological process of appetite, satiety, and adipose tissue deposition. This area of research is still in its infancy, with most trials still in the early stages. Studies in human subjects are yet to be developed. Modulation of leptin and ghrelin shows promising results in animal studies and shall be a future topic of research. The new concept of the gut microbiome and its effects on energy expenditure and metabolism is another area where further research is required, in order to further understand its contribution to obesity. With rapidly evolving technology, it is expected that less invasive endoscopic and robotic bariatric techniques will soon be developed for bariatric surgery. An algorithm for management of diabesity is shown in Figure 4.
With the obesity epidemic, diabetes, and the evident link between the two, the diabesity, is a widely recognised emerging public health issue. Understanding the pathophysiological mechanisms involved in the interplay between diabetes and obesity is paramount, both for primary as well as secondary care physicians, in order to guide the clinical management of such patients. Our paper offers a summary of the current concepts in the pathophysiology and management of diabesity. The emerging theories around the impact of the gut microbiome on diabetes and obesity are still in their infancy but show promising results so far. The potential role of metformin in modulating the gut microbiome is an area that requires further investigation. Future research and better understanding of the involvement of gut bacteria in energy expenditure and the development of diabesity may guide the development of modulatory treatments and reduce risk in affected individuals.
Diabesity is associated with a host of complications, including MAFLD. Our review of literature suggests that the majority of patients with T2DM exhibit hepatic steatosis in the initial stages of their diabetes, without derangement of liver function. Occult liver disease may be difficult to diagnose unless liver biopsy is carried out, and understandably this would not be indicated as a baseline investigation in all obese diabetic patients. Clinicians should be mindful of the likelihood of MAFLD in obese diabetics, and offer management to improve weight and glycaemic control, which will in turn aid to reduce the long-term consequences of hepatic steatosis. Early identification and management of OSA is also important, as intervention in the form of CPAP has been shown to improve insulin resistance. Therefore, early diagnosis and management of OSA may aid glycaemic control, whereas missing this diagnosis may make glycaemic management more challenging, leading to further complications. Additionally, monitoring for cardiovascular disease, potentially with coronary artery calcium score, as well as early cardiology referral where indicated, would be important in order to address and minimize cardiovascular mortality and morbidity.
Lifestyle modification remains paramount, but it is important to set realistic achievable targets which are agreed between the patient and the multi-disciplinary team. From our review, we could infer that exercise alone is not expected to result in significant improvement in diabetic control, unless accompanied by weight loss. On reviewing the recent literature, the common theme appears to be that a Mediterranean diet may be beneficial in achieving weight loss and better glycaemic control in diabesity patients. The Mediterranean diet has been shown to have a greater impact on overall HbA1c when compared to low carbohydrate, high protein, and low fat dietary modifications. When combined with exercise, dietary modification appears to be more effective, with a significant chance of inducing T2DM remission if the patient is motivated and adheres to the interventions offered.
Rapid advances in pharmacotherapy have revealed the dual action of medications such as GLP-1 agonists and SGLT-2 inhibitors in improving diabetes and achieving weight loss. The most recent relevant trials as summarised in our review show promising results, with improvement in both weight and HbA1c with these agents. GLP-1 agonists and SGLT-2 inhibitors are expected to play a pivotal role in the management of diabesity in the coming years, with an increasing number of patients being prescribed these medications.
Newly proposed mechanisms of action of metformin to improve weight and glycaemic control have been published, and these include a potential effect on the gut microbiome, and an association with increased GDF15 levels. Further research is required to confirm these modulatory effects, but metformin remains one of the first line medications for the treatment of diabesity. Rapid advances in technology show a promising future for bariatric surgery, which can be very effective in achieving weight loss and reversing diabetes. Therefore, an early referral to weight management services for patients that fulfil the relevant criteria, and timely intervention could lead to early resolution of the disease.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Endocrinology and metabolism
Country/Territory of origin: United Kingdom
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Emran TB, Bangladesh; Millman JF, Japan; Peng XC, China S-Editor: Wang JJ L-Editor: A P-Editor: Wang JJ
| 1. | Obesity fact sheet. World Obesity Day 2022. [cited 3 October 2022]. Available from: https://www.worldobesityday.org/assets/downloads/Obesity_factsheet_one_pager.pdf. |
| 2. | Sims EA, Danforth E Jr, Horton ES, Bray GA, Glennon JA, Salans LB. Endocrine and metabolic effects of experimental obesity in man. Recent Prog Horm Res. 1973;29:457-496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 88] [Article Influence: 1.7] [Reference Citation Analysis (2)] |
| 3. | Pappachan JM, Viswanath AK. Medical Management of Diabesity: Do We Have Realistic Targets? Curr Diab Rep. 2017;17:4. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 4. | El Khoury L, Chouillard E, Chahine E, Saikaly E, Debs T, Kassir R. Metabolic Surgery and Diabesity: a Systematic Review. Obes Surg. 2018;28:2069-2077. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Pappachan JM, Fernandez CJ, Chacko EC. Diabesity and antidiabetic drugs. Mol Aspects Med. 2019;66:3-12. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 6. | Barazzoni R, Gortan Cappellari G, Ragni M, Nisoli E. Insulin resistance in obesity: an overview of fundamental alterations. Eat Weight Disord. 2018;23:149-157. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 229] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 7. | Luzi L, Petrides AS, De Fronzo RA. Different sensitivity of glucose and amino acid metabolism to insulin in NIDDM. Diabetes. 1993;42:1868-1877. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 36] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 8. | Wilcox G. Insulin and insulin resistance. Clin Biochem Rev. 2005;26:19-39. [PubMed] |
| 9. | Kern PA, Saghizadeh M, Ong JM, Bosch RJ, Deem R, Simsolo RB. The expression of tumor necrosis factor in human adipose tissue. Regulation by obesity, weight loss, and relationship to lipoprotein lipase. J Clin Invest. 1995;95:2111-2119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 862] [Cited by in RCA: 851] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 10. | Furukawa S, Fujita T, Shimabukuro M, Iwaki M, Yamada Y, Nakajima Y, Nakayama O, Makishima M, Matsuda M, Shimomura I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J Clin Invest. 2004;114:1752-1761. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3370] [Cited by in RCA: 3834] [Article Influence: 191.7] [Reference Citation Analysis (0)] |
| 11. | Boden G, Chen X. Effects of fat on glucose uptake and utilization in patients with non-insulin-dependent diabetes. J Clin Invest. 1995;96:1261-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 230] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 12. | Boden G. Obesity, insulin resistance and free fatty acids. Curr Opin Endocrinol Diabetes Obes. 2011;18:139-143. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 627] [Cited by in RCA: 608] [Article Influence: 43.4] [Reference Citation Analysis (0)] |
| 13. | Ritter O, Jelenik T, Roden M. Lipid-mediated muscle insulin resistance: different fat, different pathways? J Mol Med (Berl). 2015;93:831-843. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 53] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 14. | Barazzoni R. Skeletal muscle mitochondrial protein metabolism and function in ageing and type 2 diabetes. Curr Opin Clin Nutr Metab Care. 2004;7:97-102. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 29] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 15. | Le Chatelier E, Nielsen T, Qin J, Prifti E, Hildebrand F, Falony G, Almeida M, Arumugam M, Batto JM, Kennedy S, Leonard P, Li J, Burgdorf K, Grarup N, Jørgensen T, Brandslund I, Nielsen HB, Juncker AS, Bertalan M, Levenez F, Pons N, Rasmussen S, Sunagawa S, Tap J, Tims S, Zoetendal EG, Brunak S, Clément K, Doré J, Kleerebezem M, Kristiansen K, Renault P, Sicheritz-Ponten T, de Vos WM, Zucker JD, Raes J, Hansen T; MetaHIT consortium, Bork P, Wang J, Ehrlich SD, Pedersen O. Richness of human gut microbiome correlates with metabolic markers. Nature. 2013;500:541-546. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2727] [Cited by in RCA: 3205] [Article Influence: 267.1] [Reference Citation Analysis (2)] |
| 16. | Geurts L, Neyrinck AM, Delzenne NM, Knauf C, Cani PD. Gut microbiota controls adipose tissue expansion, gut barrier and glucose metabolism: novel insights into molecular targets and interventions using prebiotics. Benef Microbes. 2014;5:3-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 205] [Article Influence: 18.6] [Reference Citation Analysis (1)] |
| 17. | Kimura I, Ozawa K, Inoue D, Imamura T, Kimura K, Maeda T, Terasawa K, Kashihara D, Hirano K, Tani T, Takahashi T, Miyauchi S, Shioi G, Inoue H, Tsujimoto G. The gut microbiota suppresses insulin-mediated fat accumulation via the short-chain fatty acid receptor GPR43. Nat Commun. 2013;4:1829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 833] [Cited by in RCA: 1081] [Article Influence: 90.1] [Reference Citation Analysis (0)] |
| 18. | Sakurai Y, Kubota N, Yamauchi T, Kadowaki T. Role of Insulin Resistance in MAFLD. Int J Mol Sci. 2021;22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 235] [Article Influence: 58.8] [Reference Citation Analysis (0)] |
| 19. | Eslam M, Sanyal AJ, George J; International Consensus Panel. MAFLD: A Consensus-Driven Proposed Nomenclature for Metabolic Associated Fatty Liver Disease. Gastroenterology. 2020;158:1999-2014.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2367] [Cited by in RCA: 2200] [Article Influence: 440.0] [Reference Citation Analysis (1)] |
| 20. | Masarone M, Rosato V, Aglitti A, Bucci T, Caruso R, Salvatore T, Sasso FC, Tripodi MF, Persico M. Liver biopsy in type 2 diabetes mellitus: Steatohepatitis represents the sole feature of liver damage. PLoS One. 2017;12:e0178473. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 76] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 21. | Targher G, Lonardo A, Byrne CD. Nonalcoholic fatty liver disease and chronic vascular complications of diabetes mellitus. Nat Rev Endocrinol. 2018;14:99-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 297] [Article Influence: 42.4] [Reference Citation Analysis (0)] |
| 22. | Muraki I, Wada H, Tanigawa T. Sleep apnea and type 2 diabetes. J Diabetes Investig. 2018;9:991-997. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 86] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 23. | Zhang SX, Khalyfa A, Wang Y, Carreras A, Hakim F, Neel BA, Brady MJ, Qiao Z, Hirotsu C, Gozal D. Sleep fragmentation promotes NADPH oxidase 2-mediated adipose tissue inflammation leading to insulin resistance in mice. Int J Obes (Lond). 2014;38:619-624. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 90] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 24. | Resnick HE, Redline S, Shahar E, Gilpin A, Newman A, Walter R, Ewy GA, Howard BV, Punjabi NM; Sleep Heart Health Study. Diabetes and sleep disturbances: findings from the Sleep Heart Health Study. Diabetes Care. 2003;26:702-709. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 334] [Cited by in RCA: 329] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 25. | Strand LB, Carnethon M, Biggs ML, Djoussé L, Kaplan RC, Siscovick DS, Robbins JA, Redline S, Patel SR, Janszky I, Mukamal KJ. Sleep Disturbances and Glucose Metabolism in Older Adults: The Cardiovascular Health Study. Diabetes Care. 2015;38:2050-2058. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 26. | Garcia JM, Sharafkhaneh H, Hirshkowitz M, Elkhatib R, Sharafkhaneh A. Weight and metabolic effects of CPAP in obstructive sleep apnea patients with obesity. Respir Res. 2011;12:80. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 55] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 27. | Trenell MI, Ward JA, Yee BJ, Phillips CL, Kemp GJ, Grunstein RR, Thompson CH. Influence of constant positive airway pressure therapy on lipid storage, muscle metabolism and insulin action in obese patients with severe obstructive sleep apnoea syndrome. Diabetes Obes Metab. 2007;9:679-687. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 64] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 28. | Harsch IA, Schahin SP, Radespiel-Tröger M, Weintz O, Jahreiss H, Fuchs FS, Wiest GH, Hahn EG, Lohmann T, Konturek PC, Ficker JH. Continuous positive airway pressure treatment rapidly improves insulin sensitivity in patients with obstructive sleep apnea syndrome. Am J Respir Crit Care Med. 2004;169:156-162. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 356] [Cited by in RCA: 325] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 29. | Ng ACT, Delgado V, Borlaug BA, Bax JJ. Diabesity: the combined burden of obesity and diabetes on heart disease and the role of imaging. Nat Rev Cardiol. 2021;18:291-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 180] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 30. | Montaigne D, Marechal X, Coisne A, Debry N, Modine T, Fayad G, Potelle C, El Arid JM, Mouton S, Sebti Y, Duez H, Preau S, Remy-Jouet I, Zerimech F, Koussa M, Richard V, Neviere R, Edme JL, Lefebvre P, Staels B. Myocardial contractile dysfunction is associated with impaired mitochondrial function and dynamics in type 2 diabetic but not in obese patients. Circulation. 2014;130:554-564. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 240] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 31. | McGavock JM, Lingvay I, Zib I, Tillery T, Salas N, Unger R, Levine BD, Raskin P, Victor RG, Szczepaniak LS. Cardiac steatosis in diabetes mellitus: a 1H-magnetic resonance spectroscopy study. Circulation. 2007;116:1170-1175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 440] [Cited by in RCA: 482] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 32. | Kannel WB, Hjortland M, Castelli WP. Role of diabetes in congestive heart failure: the Framingham study. Am J Cardiol. 1974;34:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1406] [Cited by in RCA: 1456] [Article Influence: 28.5] [Reference Citation Analysis (0)] |
| 33. | Echouffo-Tcheugui JB, Short MI, Xanthakis V, Field P, Sponholtz TR, Larson MG, Vasan RS. Natural History of Obesity Subphenotypes: Dynamic Changes Over Two Decades and Prognosis in the Framingham Heart Study. J Clin Endocrinol Metab. 2019;104:738-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 55] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 34. | Burke GL, Bertoni AG, Shea S, Tracy R, Watson KE, Blumenthal RS, Chung H, Carnethon MR. The impact of obesity on cardiovascular disease risk factors and subclinical vascular disease: the Multi-Ethnic Study of Atherosclerosis. Arch Intern Med. 2008;168:928-935. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 199] [Cited by in RCA: 185] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 35. | Kronmal RA, McClelland RL, Detrano R, Shea S, Lima JA, Cushman M, Bild DE, Burke GL. Risk factors for the progression of coronary artery calcification in asymptomatic subjects: results from the Multi-Ethnic Study of Atherosclerosis (MESA). Circulation. 2007;115:2722-2730. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 375] [Cited by in RCA: 429] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 36. | SCOT-HEART Investigators, Newby DE, Adamson PD, Berry C, Boon NA, Dweck MR, Flather M, Forbes J, Hunter A, Lewis S, MacLean S, Mills NL, Norrie J, Roditi G, Shah ASV, Timmis AD, van Beek EJR, Williams MC. Coronary CT Angiography and 5-Year Risk of Myocardial Infarction. N Engl J Med. 2018;379:924-933. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 686] [Cited by in RCA: 957] [Article Influence: 136.7] [Reference Citation Analysis (0)] |
| 37. | Hinden L, Kogot-Levin A, Tam J, Leibowitz G. Pathogenesis of diabesity-induced kidney disease: role of kidney nutrient sensing. FEBS J. 2022;289:901-921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Lavin DP, White MF, Brazil DP. IRS proteins and diabetic complications. Diabetologia. 2016;59:2280-2291. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 71] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 39. | Cai T, Ke Q, Fang Y, Wen P, Chen H, Yuan Q, Luo J, Zhang Y, Sun Q, Lv Y, Zen K, Jiang L, Zhou Y, Yang J. Sodium-glucose cotransporter 2 inhibition suppresses HIF-1α-mediated metabolic switch from lipid oxidation to glycolysis in kidney tubule cells of diabetic mice. Cell Death Dis. 2020;11:390. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 89] [Cited by in RCA: 123] [Article Influence: 24.6] [Reference Citation Analysis (0)] |
| 40. | Vallon V, Rose M, Gerasimova M, Satriano J, Platt KA, Koepsell H, Cunard R, Sharma K, Thomson SC, Rieg T. Knockout of Na-glucose transporter SGLT2 attenuates hyperglycemia and glomerular hyperfiltration but not kidney growth or injury in diabetes mellitus. Am J Physiol Renal Physiol. 2013;304:F156-F167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 254] [Cited by in RCA: 309] [Article Influence: 23.8] [Reference Citation Analysis (0)] |
| 41. | Hughes TA, Gwynne JT, Switzer BR, Herbst C, White G. Effects of caloric restriction and weight loss on glycemic control, insulin release and resistance, and atherosclerotic risk in obese patients with type II diabetes mellitus. Am J Med. 1984;77:7-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 105] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 42. | Steven S, Taylor R. Restoring normoglycaemia by use of a very low calorie diet in long- and short-duration Type 2 diabetes. Diabet Med. 2015;32:1149-1155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 77] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 43. | Wing RR, Koeske R, Epstein LH, Nowalk MP, Gooding W, Becker D. Long-term effects of modest weight loss in type II diabetic patients. Arch Intern Med. 1987;147:1749-1753. [PubMed] |
| 44. | Gregg EW, Chen H, Wagenknecht LE, Clark JM, Delahanty LM, Bantle J, Pownall HJ, Johnson KC, Safford MM, Kitabchi AE, Pi-Sunyer FX, Wing RR, Bertoni AG; Look AHEAD Research Group. Association of an intensive lifestyle intervention with remission of type 2 diabetes. JAMA. 2012;308:2489-2496. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 500] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 45. | Lean ME, Leslie WS, Barnes AC, Brosnahan N, Thom G, McCombie L, Peters C, Zhyzhneuskaya S, Al-Mrabeh A, Hollingsworth KG, Rodrigues AM, Rehackova L, Adamson AJ, Sniehotta FF, Mathers JC, Ross HM, McIlvenna Y, Stefanetti R, Trenell M, Welsh P, Kean S, Ford I, McConnachie A, Sattar N, Taylor R. Primary care-led weight management for remission of type 2 diabetes (DiRECT): an open-label, cluster-randomised trial. Lancet. 2018;391:541-551. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1267] [Cited by in RCA: 1229] [Article Influence: 175.6] [Reference Citation Analysis (0)] |
| 46. | Ajala O, English P, Pinkney J. Systematic review and meta-analysis of different dietary approaches to the management of type 2 diabetes. Am J Clin Nutr. 2013;97:505-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 492] [Cited by in RCA: 470] [Article Influence: 39.2] [Reference Citation Analysis (0)] |
| 47. | Magkos F, Hjorth MF, Astrup A. Diet and exercise in the prevention and treatment of type 2 diabetes mellitus. Nat Rev Endocrinol. 2020;16:545-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 258] [Article Influence: 51.6] [Reference Citation Analysis (0)] |
| 48. | Ozsoy S, Sultanoglu N, Sanlidag T. The role of Mediterranean diet and gut microbiota in type-2 diabetes mellitus associated with obesity (diabesity). J Prev Med Hyg. 2022;63:E87-E92. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 49. | Boulé NG, Haddad E, Kenny GP, Wells GA, Sigal RJ. Effects of exercise on glycemic control and body mass in type 2 diabetes mellitus: a meta-analysis of controlled clinical trials. JAMA. 2001;286:1218-1227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1201] [Cited by in RCA: 1147] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 50. | Di Loreto C, Fanelli C, Lucidi P, Murdolo G, De Cicco A, Parlanti N, Ranchelli A, Fatone C, Taglioni C, Santeusanio F, De Feo P. Make your diabetic patients walk: long-term impact of different amounts of physical activity on type 2 diabetes. Diabetes Care. 2005;28:1295-1302. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 122] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 51. | Balducci S, Zanuso S, Cardelli P, Salvi L, Bazuro A, Pugliese L, Maccora C, Iacobini C, Conti FG, Nicolucci A, Pugliese G; Italian Diabetes Exercise Study (IDES) Investigators. Effect of high- versus low-intensity supervised aerobic and resistance training on modifiable cardiovascular risk factors in type 2 diabetes; the Italian Diabetes and Exercise Study (IDES). PLoS One. 2012;7:e49297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 69] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 52. | Balducci S, Sacchetti M, Haxhi J, Orlando G, D’Errico V, Fallucca S, Menini S, Pugliese G. Physical exercise as therapy for type 2 diabetes mellitus. Diabetes Metab Res Rev. 2014;30 Suppl 1:13-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 130] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 53. | Eriksson KF, Lindgärde F. Prevention of type 2 (non-insulin-dependent) diabetes mellitus by diet and physical exercise. The 6-year Malmö feasibility study. Diabetologia. 1991;34:891-898. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 684] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 54. | LaMoia TE, Shulman GI. Cellular and Molecular Mechanisms of Metformin Action. Endocr Rev. 2021;42:77-96. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 512] [Cited by in RCA: 433] [Article Influence: 108.3] [Reference Citation Analysis (0)] |
| 55. | Hostalek U, Gwilt M, Hildemann S. Therapeutic Use of Metformin in Prediabetes and Diabetes Prevention. Drugs. 2015;75:1071-1094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 186] [Cited by in RCA: 182] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 56. | Lazzaroni E, Ben Nasr M, Loretelli C, Pastore I, Plebani L, Lunati ME, Vallone L, Bolla AM, Rossi A, Montefusco L, Ippolito E, Berra C, D’Addio F, Zuccotti GV, Fiorina P. Anti-diabetic drugs and weight loss in patients with type 2 diabetes. Pharmacol Res. 2021;171:105782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 57. | Wu H, Esteve E, Tremaroli V, Khan MT, Caesar R, Mannerås-Holm L, Ståhlman M, Olsson LM, Serino M, Planas-Fèlix M, Xifra G, Mercader JM, Torrents D, Burcelin R, Ricart W, Perkins R, Fernàndez-Real JM, Bäckhed F. Metformin alters the gut microbiome of individuals with treatment-naive type 2 diabetes, contributing to the therapeutic effects of the drug. Nat Med. 2017;23:850-858. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 824] [Cited by in RCA: 1135] [Article Influence: 141.9] [Reference Citation Analysis (0)] |
| 58. | Day EA, Ford RJ, Smith BK, Mohammadi-Shemirani P, Morrow MR, Gutgesell RM, Lu R, Raphenya AR, Kabiri M, McArthur AG, McInnes N, Hess S, Paré G, Gerstein HC, Steinberg GR. Metformin-induced increases in GDF15 are important for suppressing appetite and promoting weight loss. Nat Metab. 2019;1:1202-1208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 203] [Article Influence: 33.8] [Reference Citation Analysis (0)] |
| 59. | Petrie JR, Chaturvedi N, Ford I, Brouwers MCGJ, Greenlaw N, Tillin T, Hramiak I, Hughes AD, Jenkins AJ, Klein BEK, Klein R, Ooi TC, Rossing P, Stehouwer CDA, Sattar N, Colhoun HM; REMOVAL Study Group. Cardiovascular and metabolic effects of metformin in patients with type 1 diabetes (REMOVAL): a double-blind, randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2017;5:597-609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 250] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 60. | Lv Z, Guo Y. Metformin and Its Benefits for Various Diseases. Front Endocrinol (Lausanne). 2020;11:191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 310] [Article Influence: 62.0] [Reference Citation Analysis (0)] |
| 61. | Bell S, Farran B, McGurnaghan S, McCrimmon RJ, Leese GP, Petrie JR, McKeigue P, Sattar N, Wild S, McKnight J, Lindsay R, Colhoun HM, Looker H. Risk of acute kidney injury and survival in patients treated with Metformin: an observational cohort study. BMC Nephrol. 2017;18:163. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 62] [Cited by in RCA: 61] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 62. | Mohan M, Al-Talabany S, McKinnie A, Mordi IR, Singh JSS, Gandy SJ, Baig F, Hussain MS, Bhalraam U, Khan F, Choy AM, Matthew S, Houston JG, Struthers AD, George J, Lang CC. A randomized controlled trial of metformin on left ventricular hypertrophy in patients with coronary artery disease without diabetes: the MET-REMODEL trial. Eur Heart J. 2019;40:3409-3417. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 109] [Article Influence: 21.8] [Reference Citation Analysis (0)] |
| 63. | Apovian CM, Okemah J, O'Neil PM. Body Weight Considerations in the Management of Type 2 Diabetes. Adv Ther. 2019;36:44-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 64] [Cited by in RCA: 147] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 64. | Lasserson DS, Glasziou P, Perera R, Holman RR, Farmer AJ. Optimal insulin regimens in type 2 diabetes mellitus: systematic review and meta-analyses. Diabetologia. 2009;52:1990-2000. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 81] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 65. | Valentine V, Goldman J, Shubrook JH. Rationale for, Initiation and Titration of the Basal Insulin/GLP-1RA Fixed-Ratio Combination Products, IDegLira and IGlarLixi, for the Management of Type 2 Diabetes. Diabetes Ther. 2017;8:739-752. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 66. | Freemantle N, Chou E, Frois C, Zhuo D, Lehmacher W, Vlajnic A, Wang H, Chung HW, Zhang Q, Wu E, Gerrits C. Safety and efficacy of insulin glargine 300 u/mL compared with other basal insulin therapies in patients with type 2 diabetes mellitus: a network meta-analysis. BMJ Open. 2016;6:e009421. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 47] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 67. | Schwartz S, Herman M. Revisiting weight reduction and management in the diabetic patient: Novel therapies provide new strategies. Postgrad Med. 2015;127:480-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 68. | Leiter LA. Latest Evidence on Sulfonylureas: What's New? Diabetes Ther. 2020;11:15-22. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 69. | McIntosh B, Cameron C, Singh SR, Yu C, Ahuja T, Welton NJ, Dahl M. Second-line therapy in patients with type 2 diabetes inadequately controlled with metformin monotherapy: a systematic review and mixed-treatment comparison meta-analysis. Open Med. 2011;5:e35-e48. [PubMed] |
| 70. | Vaccaro O, Masulli M, Nicolucci A, Bonora E, Del Prato S, Maggioni AP, Rivellese AA, Squatrito S, Giorda CB, Sesti G, Mocarelli P, Lucisano G, Sacco M, Signorini S, Cappellini F, Perriello G, Babini AC, Lapolla A, Gregori G, Giordano C, Corsi L, Buzzetti R, Clemente G, Di Cianni G, Iannarelli R, Cordera R, La Macchia O, Zamboni C, Scaranna C, Boemi M, Iovine C, Lauro D, Leotta S, Dall'Aglio E, Cannarsa E, Tonutti L, Pugliese G, Bossi AC, Anichini R, Dotta F, Di Benedetto A, Citro G, Antenucci D, Ricci L, Giorgino F, Santini C, Gnasso A, De Cosmo S, Zavaroni D, Vedovato M, Consoli A, Calabrese M, di Bartolo P, Fornengo P, Riccardi G; Thiazolidinediones Or Sulfonylureas Cardiovascular Accidents Intervention Trial (TOSCA. IT) study group; Italian Diabetes Society. Effects on the incidence of cardiovascular events of the addition of pioglitazone versus sulfonylureas in patients with type 2 diabetes inadequately controlled with metformin (TOSCA.IT): a randomised, multicentre trial. Lancet Diabetes Endocrinol. 2017;5:887-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 186] [Cited by in RCA: 193] [Article Influence: 24.1] [Reference Citation Analysis (0)] |
| 71. | ADVANCE Collaborative Group; Patel A, MacMahon S, Chalmers J, Neal B, Billot L, Woodward M, Marre M, Cooper M, Glasziou P, Grobbee D, Hamet P, Harrap S, Heller S, Liu L, Mancia G, Mogensen CE, Pan C, Poulter N, Rodgers A, Williams B, Bompoint S, de Galan BE, Joshi R, Travert F. Intensive blood glucose control and vascular outcomes in patients with type 2 diabetes. N Engl J Med. 2008;358:2560-2572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4759] [Cited by in RCA: 4888] [Article Influence: 287.5] [Reference Citation Analysis (0)] |
| 72. | Nath B, Suvarna VR. CARdiovascular Outcome study of LINAgliptin versus glimepiride in patients with T2D trial. Perspect Clin Res. 2020;11:44-46. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 73. | Lebovitz HE. Thiazolidinediones: the Forgotten Diabetes Medications. Curr Diab Rep. 2019;19:151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 223] [Article Influence: 37.2] [Reference Citation Analysis (0)] |
| 74. | Fonseca V. Effect of thiazolidinediones on body weight in patients with diabetes mellitus. Am J Med. 2003;115 Suppl 8A:42S-48S. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 223] [Cited by in RCA: 211] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 75. | Lewis JD, Habel LA, Quesenberry CP, Strom BL, Peng T, Hedderson MM, Ehrlich SF, Mamtani R, Bilker W, Vaughn DJ, Nessel L, Van Den Eeden SK, Ferrara A. Pioglitazone Use and Risk of Bladder Cancer and Other Common Cancers in Persons With Diabetes. JAMA. 2015;314:265-277. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 227] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 76. | Tuccori M, Filion KB, Yin H, Yu OH, Platt RW, Azoulay L. Pioglitazone use and risk of bladder cancer: population based cohort study. BMJ. 2016;352:i1541. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 151] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 77. | Sherifali D, Nerenberg K, Pullenayegum E, Cheng JE, Gerstein HC. The effect of oral antidiabetic agents on A1C levels: a systematic review and meta-analysis. Diabetes Care. 2010;33:1859-1864. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 259] [Cited by in RCA: 243] [Article Influence: 16.2] [Reference Citation Analysis (0)] |
| 78. | McIntosh B, Cameron C, Singh SR, Yu C, Dolovich L, Houlden R. Choice of therapy in patients with type 2 diabetes inadequately controlled with metformin and a sulphonylurea: a systematic review and mixed-treatment comparison meta-analysis. Open Med. 2012;6:e62-e74. [PubMed] |
| 79. | Packer M. Do DPP-4 Inhibitors Cause Heart Failure Events by Promoting Adrenergically Mediated Cardiotoxicity? Circ Res. 2018;122:928-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 64] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 80. | Nauck MA, Quast DR, Wefers J, Meier JJ. GLP-1 receptor agonists in the treatment of type 2 diabetes - state-of-the-art. Mol Metab. 2021;46:101102. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 399] [Cited by in RCA: 850] [Article Influence: 212.5] [Reference Citation Analysis (0)] |
| 81. | Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, McGowan BM, Rosenstock J, Tran MTD, Wadden TA, Wharton S, Yokote K, Zeuthen N, Kushner RF; STEP 1 Study Group. Once-Weekly Semaglutide in Adults with Overweight or Obesity. N Engl J Med. 2021;384:989-1002. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 699] [Cited by in RCA: 2177] [Article Influence: 544.3] [Reference Citation Analysis (0)] |
| 82. | Abd El Aziz MS, Kahle M, Meier JJ, Nauck MA. A meta-analysis comparing clinical effects of short- or long-acting GLP-1 receptor agonists versus insulin treatment from head-to-head studies in type 2 diabetic patients. Diabetes Obes Metab. 2017;19:216-227. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 95] [Cited by in RCA: 116] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 83. | Kristensen SL, Rørth R, Jhund PS, Docherty KF, Sattar N, Preiss D, Køber L, Petrie MC, McMurray JJV. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776-785. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 729] [Cited by in RCA: 1058] [Article Influence: 176.3] [Reference Citation Analysis (0)] |
| 84. | Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, Probstfield J, Botros FT, Riddle MC, Rydén L, Xavier D, Atisso CM, Dyal L, Hall S, Rao-Melacini P, Wong G, Avezum A, Basile J, Chung N, Conget I, Cushman WC, Franek E, Hancu N, Hanefeld M, Holt S, Jansky P, Keltai M, Lanas F, Leiter LA, Lopez-Jaramillo P, Cardona Munoz EG, Pirags V, Pogosova N, Raubenheimer PJ, Shaw JE, Sheu WH, Temelkova-Kurktschiev T; REWIND Investigators. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet. 2019;394:131-138. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 425] [Cited by in RCA: 399] [Article Influence: 66.5] [Reference Citation Analysis (0)] |
| 85. | Mann JFE, Ørsted DD, Brown-Frandsen K, Marso SP, Poulter NR, Rasmussen S, Tornøe K, Zinman B, Buse JB; LEADER Steering Committee and Investigators. Liraglutide and Renal Outcomes in Type 2 Diabetes. N Engl J Med. 2017;377:839-848. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 740] [Cited by in RCA: 866] [Article Influence: 108.3] [Reference Citation Analysis (0)] |
| 86. | Nauck MA, Friedrich N. Do GLP-1-based therapies increase cancer risk? Diabetes Care. 2013;36:S245-S252. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 103] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 87. | Abd El Aziz M, Cahyadi O, Meier JJ, Schmidt WE, Nauck MA. Incretin-based glucose-lowering medications and the risk of acute pancreatitis and malignancies: a meta-analysis based on cardiovascular outcomes trials. Diabetes Obes Metab. 2020;22:699-704. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 101] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 88. | Pfister M, Whaley JM, Zhang L, List JF. Inhibition of SGLT2: a novel strategy for treatment of type 2 diabetes mellitus. Clin Pharmacol Ther. 2011;89:621-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 89. | Li J, Gong Y, Li C, Lu Y, Liu Y, Shao Y. Long-term efficacy and safety of sodium-glucose cotransporter-2 inhibitors as add-on to metformin treatment in the management of type 2 diabetes mellitus: A meta-analysis. Medicine (Baltimore). 2017;96:e7201. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 36] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 90. | Giugliano D, Longo M, Scappaticcio L, Bellastella G, Maiorino MI, Esposito K. SGLT-2 inhibitors and cardiorenal outcomes in patients with or without type 2 diabetes: a meta-analysis of 11 CVOTs. Cardiovasc Diabetol. 2021;20:236. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 91. | Giugliano D, Longo M, Caruso P, Maiorino MI, Bellastella G, Esposito K. Sodium-glucose co-transporter-2 inhibitors for the prevention of cardiorenal outcomes in type 2 diabetes: An updated meta-analysis. Diabetes Obes Metab. 2021;23:1672-1676. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 30] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 92. | Packer M, Butler J, Zannad F, Pocock SJ, Filippatos G, Ferreira JP, Brueckmann M, Jamal W, Zeller C, Wanner C, Anker SD; EMPEROR Study Group. Empagliflozin and Major Renal Outcomes in Heart Failure. N Engl J Med. 2021;385:1531-1533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 82] [Article Influence: 20.5] [Reference Citation Analysis (0)] |
| 93. | Mathiesen DS, Bagger JI, Knop FK. Long-acting amylin analogues for the management of obesity. Curr Opin Endocrinol Diabetes Obes. 2022;29:183-190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 22] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 94. | Müller TD, Blüher M, Tschöp MH, DiMarchi RD. Anti-obesity drug discovery: advances and challenges. Nat Rev Drug Discov. 2022;21:201-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 475] [Cited by in RCA: 549] [Article Influence: 183.0] [Reference Citation Analysis (0)] |
| 95. | Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, Kiyosue A, Zhang S, Liu B, Bunck MC, Stefanski A; SURMOUNT-1 Investigators. Tirzepatide Once Weekly for the Treatment of Obesity. N Engl J Med. 2022;387:205-216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 232] [Cited by in RCA: 1508] [Article Influence: 502.7] [Reference Citation Analysis (0)] |
| 96. | Roth JD, Roland BL, Cole RL, Trevaskis JL, Weyer C, Koda JE, Anderson CM, Parkes DG, Baron AD. Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc Natl Acad Sci U S A. 2008;105:7257-7262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 332] [Article Influence: 19.5] [Reference Citation Analysis (0)] |
| 97. | Rothstein RI. Bariatric and Metabolic Endoscopy. Gastrointest Endosc Clin N Am. 2017;27:xv-xvi. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 98. | Li SH, Wang YJ, Zhang ST. Development of Bariatric and Metabolic Endoscopy. Chin Med J (Engl). 2018;131:88-94. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 99. | Imaz I, Martínez-Cervell C, García-Alvarez EE, Sendra-Gutiérrez JM, González-Enríquez J. Safety and effectiveness of the intragastric balloon for obesity. A meta-analysis. Obes Surg. 2008;18:841-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 271] [Article Influence: 15.9] [Reference Citation Analysis (0)] |
| 100. | Lopez-Nava G, Sharaiha RZ, Vargas EJ, Bazerbachi F, Manoel GN, Bautista-Castaño I, Acosta A, Topazian MD, Mundi MS, Kumta N, Kahaleh M, Herr AM, Shukla A, Aronne L, Gostout CJ, Abu Dayyeh BK. Endoscopic Sleeve Gastroplasty for Obesity: a Multicenter Study of 248 Patients with 24 Months Follow-Up. Obes Surg. 2017;27:2649-2655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 148] [Cited by in RCA: 151] [Article Influence: 21.6] [Reference Citation Analysis (0)] |
| 101. | Roth AE, Thornley CJ, Blackstone RP. Outcomes in Bariatric and Metabolic Surgery: an Updated 5-Year Review. Curr Obes Rep. 2020;9:380-389. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 102. | Ecker BL, Maduka R, Ramdon A, Dempsey DT, Dumon KR, Williams NN. Resident education in robotic-assisted vertical sleeve gastrectomy: outcomes and cost-analysis of 411 consecutive cases. Surg Obes Relat Dis. 2016;12:313-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 103. | Qin J, Song L. Glucagon-like peptide-1 (GLP-1) receptor agonists and cardiovascular events in patients with type 2 diabetes mellitus: a meta-analysis of double-blind, randomized, placebo-controlled clinical trials. BMC Endocr Disord. 2022;22:125. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 104. | Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, Shaw W, Law G, Desai M, Matthews DR; CANVAS Program Collaborative Group. Canagliflozin and Cardiovascular and Renal Events in Type 2 Diabetes. N Engl J Med. 2017;377:644-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4498] [Cited by in RCA: 5388] [Article Influence: 673.5] [Reference Citation Analysis (0)] |
| 105. | Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, Silverman MG, Zelniker TA, Kuder JF, Murphy SA, Bhatt DL, Leiter LA, McGuire DK, Wilding JPH, Ruff CT, Gause-Nilsson IAM, Fredriksson M, Johansson PA, Langkilde AM, Sabatine MS; DECLARE–TIMI 58 Investigators. Dapagliflozin and Cardiovascular Outcomes in Type 2 Diabetes. N Engl J Med. 2019;380:347-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4652] [Cited by in RCA: 4242] [Article Influence: 707.0] [Reference Citation Analysis (0)] |
| 106. | Häring HU, Merker L, Seewaldt-Becker E, Weimer M, Meinicke T, Broedl UC, Woerle HJ; EMPA-REG MET Trial Investigators. Empagliflozin as add-on to metformin in patients with type 2 diabetes: a 24-week, randomized, double-blind, placebo-controlled trial. Diabetes Care. 2014;37:1650-1659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 265] [Cited by in RCA: 295] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 107. | Rosenstock J, Frias J, Páll D, Charbonnel B, Pascu R, Saur D, Darekar A, Huyck S, Shi H, Lauring B, Terra SG. Effect of ertugliflozin on glucose control, body weight, blood pressure and bone density in type 2 diabetes mellitus inadequately controlled on metformin monotherapy (VERTIS MET). Diabetes Obes Metab. 2018;20:520-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 139] [Article Influence: 19.9] [Reference Citation Analysis (0)] |
| 108. | Dagogo-Jack S, Liu J, Eldor R, Amorin G, Johnson J, Hille D, Liao Y, Huyck S, Golm G, Terra SG, Mancuso JP, Engel SS, Lauring B. Efficacy and safety of the addition of ertugliflozin in patients with type 2 diabetes mellitus inadequately controlled with metformin and sitagliptin: The VERTIS SITA2 placebo-controlled randomized study. Diabetes Obes Metab. 2018;20:530-540. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 121] [Article Influence: 17.3] [Reference Citation Analysis (0)] |