Published online Jul 16, 2013. doi: 10.4253/wjge.v5.i7.332
Revised: March 14, 2013
Accepted: April 9, 2013
Published online: July 16, 2013
Processing time: 280 Days and 14.1 Hours
AIM: To investigate the use of fully covered metal stents in benign biliary strictures (BBS) and bile leaks.
METHODS: We studied 17 patients, at Harbor-UCLA Medical center (Los Angeles), with BBS (n = 12) and bile leaks (n = 5) from July 2007 to February 2012 that had received placement of fully covered self-expanding metal stents (FCSEMs). Fourteen patients had endoscopic placement of VIABIL® (Conmed, Utica, New York, United States) stents and three had Wallflex® (Boston Scientific, Mass) stents. FCSEMS were 8 mm or 10 mm in diameter and 4 cm to 10 cm in length. Patients were followed at regular intervals to evaluate for symptoms and liver function tests. FCSEMS were removed after 4 or more weeks. Resolution of BBS and leak was documented cholangiographically following stent removal. Stent patency can be defined as adequate bile and contrast flow from the stent and into the ampulla during endoscopic retrograde cholangiopancreatography (ERCP) without clinical signs and/or symptoms of biliary obstruction. Criterion for bile leak resolution at ERCP is defined as absence of contrast extravasation from the common bile duct, cystic duct remanent, or gall bladder fossa. Rate of complications such as migration, and in-stent occlusion were recorded. Failure of endoscopic therapy was defined as persistent biliary stenosis or continuous biliary leakage after 12 mo of stent placement.
RESULTS: All 17 patients underwent successful FCSEMS placement and removal. Etiologies of BBS included: cholecystectomies (n = 8), cholelithiasis (n = 2), hepatic artery compression (n = 1), pancreatitis (n = 2), and Whipple procedure (n = 1). All bile leaks occurred following cholecystectomy. The anatomic location of BBS varied: distal common bile duct (n = 7), common hepatic duct (n = 1), hepaticojejunal anastomosis (n = 2), right intrahepatic duct (n = 1), and choledochoduodenal anastomatic junction (n = 1). All bile leaks were found to be at the cystic duct. Twelve of 17 patients had failed prior stent placement or exchange. Resolution of the biliary strictures and bile leaks was achieved in 16 of 17 patients (94%). The overall median stent time was 63 d (range 27-251 d). The median stent time for the BBS group and bile leak group was 62 ± 58 d (range 27-199 d) and 92 ± 81 d (range 48-251 d), respectively. All 17 patients underwent successful FCSEMS removal. Long term follow-up was obtained for a median of 575 d (range 28-1435 d). Complications occurred in 5 of 17 patients (29%) and included: migration (n = 2), stent clogging (n = 1), cholangitis (n = 1), and sepsis with hepatic abscess (n = 1).
CONCLUSION: Placement of fully covered self-expanding metal stents may be used in the management of benign biliary strictures and bile leaks with a low rate of complications.
Core tip: We studied 17 patients with Benign Biliary Strictures (BBS) (n = 12) and bile leaks (n = 5) from July 2007 to February 2012 that had fully covered self-expanding metal stents (FCSEMs) placed. Twelve of 17 patients had failed prior stent placement or exchange. After a median stent time of 63 d, we found 16 of 17 patients (94%) had complete resolution of biliary strictures and bile leaks. We reported complications in 5 of 17 patients (29%) which included: migration (n = 2), stent clogging (n = 1), cholangitis (n = 1), and sepsis with hepatic abscess (n = 1).
- Citation: Lalezari D, Singh I, Reicher S, Eysselein VE. Evaluation of fully covered self-expanding metal stents in benign biliary strictures and bile leaks. World J Gastrointest Endosc 2013; 5(7): 332-339
- URL: https://www.wjgnet.com/1948-5190/full/v5/i7/332.htm
- DOI: https://dx.doi.org/10.4253/wjge.v5.i7.332
Benign biliary diseases, including benign biliary strictures (BBS) and bile duct leaks, are common and management can be potentially challenging. BBS may occur as a result of chronic pancreatitis, postoperative anastomotic strictures following cholecystectomy and liver transplantation, choledocholithiasis, sclerosing cholangitis and other cholangiopathies[1-3]. Complications of biliary strictures include cholangitis, secondary biliary cirrhosis and end stage liver disease (ESLD). Because of the severity of these complications urgent decompression of strictures is required.
Bile leaks occur after abdominal surgery classically following cholecystectomy, traumatic injury[4], liver transplantation or hepatic resection[5,6]. The most common location of bile leaks is the cystic duct stump followed by the duct of Luschka[6]. Many of these patients require external biliary drainage or develop an internal biliary leak resulting in a biloma, peritonitis or abscess formation[6]. The first-line intervention in treatment of bile leaks involves placement of transpapillary biliary plastic stents and/or sphincterotomy[7,8]. Stent placement has been effective in the closure of 70%-100% of postcholecystectomy bile leaks[8].
Complex leaks are defined as those that are refractory to endoscopic intervention with biliary sphincterotomy or plastic stent placement, bile leaks following orthotopic liver transplantation or complicated cholecystectomy with large leaks. Previous studies have shown single large diameter or multiple stents across the site of the leak are superior in complex leaks[6,8]. Bile leaks can further be classified by endoscopic retrograde cholangiography (ERC) into low grade (leak identified only after intrahepatic opacification) or high grade (leak observed before intrahepatic opacification)[8]. In a study by Sandha et al[8] of 204 patients with bile leaks, transpapillary biliary stenting was found to be more effective than sphincterotomy alone in patients with high grade leaks.
Management of BBS includes decompression by endoscopic retrograde cholangiopancreatography (ERCP) with sphincterotomy and stent placement, and ultimately choledochojejunostomy. Recent studies have shown that therapeutic ERC with stent placement in BBS is a potential, although not equivalent, alternative to surgery. Compared with surgery, stent placement has lower rates of stricture recurrence, lower cost, and lower overall morbidity and mortality[9]. The standard of care for repairing bile leaks involves placement of transpapillary biliary stents with or without sphincterotomy.
Currently, plastic stents are the only Food and Drug Administration (FDA) approved therapy used in benign biliary conditions to treat biliary strictures and bile leaks. Plastic stent patency, usually 3-4 mo in malignant strictures, has been limited secondary to occlusion due to deposition of a bacterial biofilm within the stent lumen[10,11]. Self-expanding metal stents (SEMS) provide prolonged stent patency of up to 9 mo and can be deployed from a small diameter delivery system that can expand to a large diameter (10 mm) permitting improved biliary drainage. Initially, SEMS were bare metal (uncovered) meaning they had no coating material covering the metal stent. A major challenge with uncovered SEMS was in-stent epithelial hyperplasia accounting for the difficult removability of the stents and poor long term patency[2]. Partially covered self-expanding metal stents (PCSEMS) have had some success in BBS and bile leaks but were limited by the susceptibility to in-stent hyperplasia, migration, and difficultly in extraction due to mucosal hyperplasia at the uncovered proximal ends[1,2]. Advances in the development of endoprosthesis led to Fully Covered SEMS (FCSEMS) which are coated circumferentially with a material that prevents stent occlusion and imbedding due to bacterial colonization, tissue hyperplasia, and tumor ingrowth thereby increasing the duration of stent patency and permitting easier stent retrieval. Moreover, FCSEMS use in BBS and bile leaks may result in fewer endoscopic sessions and not require placement of multiple plastic stents[12]. Previous studies have shown successful outcomes of FCSEMS used in malignant biliary strictures however the data for use in benign biliary disease remains limited and conflicting (Table 1). Our studies primary aim was to evaluate the efficacy, patency and rate of complications with placement of FCSEMS for BBS and bile leaks.
| Ref. | No. of patients/No. stents | BBS or bile leaks | Etiology | Stent type | Time to removal | Results (success rate ) | Complications |
| Benign biliary strictures | |||||||
| Deviere et al[18] | 20/20 | BBS | CP | Wallstent FCSEMS | NA1 | 90% | Epithelial hyperplasia (2) |
| Cantù et al[19] | 14/14 | BBS | CP | pCSEMS | 21 mo (median) | 37.5% at 30-mo fu | Cholestasis (7), cholangitis (5), duodenal migration (2), cholecystitis (1) |
| Kuo et al[20] | 3/4 | BBS | OLT | FCSEMS | 32 d (median) | 100% | Septicemia (1), misplacement (1) |
| Kahaleh et al[2] | 79/79 | BBS | CP, OLT, BC, INF, surgical | pCSEMS | 4 mo (median) | 90% ITT 75% | Migrations (11) |
| Cahen et al[21] | 6/6 | BBS | CP | FCSEMS | 3-6 mo (median) | 66% | Migration (2), recurrent stricture (1) |
| Mahajan[3] | 44/44 | BBS | CP, gallstone related, OLT, AP, PSC | FCSEMS | 3.3 mo (median)s | 83% 77% ITT (3 patients died) | Post ERCP pancreatitis (1), mucousal ulceration and bleeding (1) |
| Garcia-Cano[22] | 20/20 | BBS | Biliary fistula, perforation of papilla, to remove uncovered stents, benign strictures, CBD stones | Wallstent FCSEMS | 132 d (median) | 70% | Pancreatitis (3) |
| Sauer et al[23] | 19/19 | BBS and bile leaks | Liver transplant | Wallflex | 11.7 wk (mean) | 79% | Proximal migration (1), distal migration (5), occlusion (1), de novo stricture (2) |
| Bile leaks | |||||||
| Wang et al[24] | 13 | Complex bile leaks | Chole and OLT | Viabil FCSEMS | 103 d | 85% ITT | Mucousal ulcerations (4), de novo choledocholithiasis/ luminal debris (10), strictures (2) |
| Sandha et al[8] | 97 | High grade leaks | Chole | FCSEMS | 42 d | 100% | Post ERCP pancreatitis (2), duodenal perforation (1) |
| Kahaleh et al[1] | 16/16 | Bile leaks | Chole, OLT | Wallstent FCSEMS | 3 mo | 93% | Stent migration (2) |
| Sauer et al[23] | 19/19 | BBS and bile leaks | Liver transplant | Wallflex | 11.7 wk (mean) | 79% | Proximal migration (1), distal migration (5), occlusion (1), de novo stricture (2) |
From July 2007 and February 2012, seventeen patients diagnosed at Harbor-UCLA Medical center with BBS and bile leaks who had undergone endoscopic placement of a FCSEMS were included in this study. Twelve patients were females and five patients were males; the mean age was 50.5 ± 16.5 years (range 27-77 years). Patient records were reviewed retrospectively. The most common presenting symptom among patients was abdominal pain. All strictures had brushings and biopsies to rule out malignancy. Fourteen patients had endoscopic placement of VIABIL® (Conmed, Utica, New York, United States) stents and three had Wallflex® (Boston Scientific, Mass) stents. An 8 or 10 mm diameter FCSEM Viabil® (Conmed, Utica, New York, United States) or Wallflex® (Boston Scientific, Mass) was deployed over a guidewire under endoscopic and fluoroscopic visualization across the biliary stricture or bile leak. The length of the stents varied ranging from 4 to 10 cm. When the gallbladder was present FCSEMS were placed below the cystic duct to avoid cholecystitis. All patients had undergone a biliary sphincterotomy prior to placement of FCSEMS.
Stent patency can be defined as adequate bile and contrast flow from the stent and into the ampulla during ERCP without clinical signs and/or symptoms of biliary obstruction (e.g., RUQ pain/tenderness, elevated alkaline phosphatase ± bilirubin, ect.). Stent placement was confirmed fluoroscopically and endoscopically. Repeat cholangiogram after stent deployment revealed the absence of a leak. Criterion for bile leak resolution at ERCP is defined as absence of contrast extravasation from the CBD, cystic duct remanent, or gall bladder fossa.
Stents were removed after at least a month, only after liver function test (LFT) normalization and resolution of symptoms, using rat-tooth forceps or a snare. After stent removal, a cholangiogram was performed to document resolution of BBS and sealed leaks. In time of follow-up, LFTs were reviewed after stent extraction and routinely during follow up course. Stent duration was expressed as median ± SD. All patients were contacted by a physician after stent removal, as a follow-up to evaluate for biliary pain and jaundice. Any patients who developed signs of biliary obstruction underwent follow-up ERCP to re-evaluate for stricture re-occurrence. Failure of endoscopic therapy was defined as persistent biliary stenosis or continuous biliary leakage after 12 mo of stent placement. Patients who failed stent therapy were referred for surgical intervention.
From July 2007 to February 2012, seventeen patients with BBS (12 patients) or bile leaks (5 patients) underwent successful placement of FCSEMS. Etiologies of BBS included: cholecystectomies (n = 8), cholelithiasis (n = 2), hepatic artery compression (n = 1), pancreatitis (n = 2), and Whipple procedure (n = 1). All bile leaks occurred following cholecystectomy. Etiologies of BBS and bile leaks are shown in Table 2. The anatomic location of BBS varied: distal common bile duct (n = 7), common hepatic duct (n = 1), hepaticojejunal anastomosis (n = 2), right intrahepatic duct (n = 1), and choledochoduodenal anastomatic junction (n = 1). All bile leaks were found to be at the cystic duct. One patient was had a previous ERCP with intraductal ultrasonography (IDUS) showing possible hepatic artery compression leading to the common hepatic duct stricture. A repeat ERCP with IDUS for stent placement demonstrated a normal hepatic artery. Twelve of 17 patients had failed prior stent placement or exchange with plastic and/or metal stents (seven having multiple stents). Because several of our subjects were referred to us who had stents placed at other facilities we were unable to determine the length of previous stenting. In patients with hepaticojejunal anastomotic strictures stents were placed percutaneously.
| Case No. | Age, yr | Sex | Etiology | Location | FCSEMS (mm), type | Complications | Duration of stenting (d) | Follow-up after removal (d) | Results |
| Benign biliary strictures | |||||||||
| 1 | 47 | M | Biliary anastomosis/Whipple | Choledocho-duodenal anastomosis | 10 mm × 0 mm Viabil | None | 89 | 1435 | Patent |
| 2 | 77 | F | Cholecystectomy | Distal CBD | 10 mm × 10 cm wallstent | None | 92 | 1210 | Patent |
| 3 | 36 | F | Cholelithiasis | Distal CBD | 10 mm × 6 cm Viabil | None | 37 | 1138 | Patent |
| 4 | 51 | F | Cholelithiasis | Distal CBD | 10 mm × 6 cm Viabil | None | 161 | 1131 | Patent |
| 5 | 73 | M | Cholecystectomy | Right intrahepatic duct | 10 mm × 10 cm Viabil | Solid debris in lumen | 160 | 1112 | Patent |
| 6 | 76 | F | Cholecystectomy | H-J anastomosis | 8 mm × 6 cm Viabil | None | 29 | 55 | Patent |
| 7 | 27 | F | Cholecystectomy | H-J anastomosis | 10 mm × 4 cm Viabil | None | 42 | 176 | Patent |
| 8 | 47 | F | Compression by hepatic artery | CHD | 80 mm × 8 cm wallstent | None | 35 | 302 | Patent |
| 9 | 36 | F | Cholecystectomy | CBD | 10 mm × 80 mm Viabil | Migration | 27 | 463 | Not patent |
| 10 | 45 | F | Chronic pancreatitis/ cholecystectomy | Distal CBD | 10 mm × 10 cm Viabil | None | 63 | 36 | Patent |
| 11 | 42 | F | Pancretitis and pancreatic head necrosis | Distal CBD | 10 mm × 6 cm wallstent | Recurrent cholangitis (n = 2), migration | 199 | 122 | Patent |
| 12 | 35 | M | Cholecystectomy | Distal CBD | 10 mm × 8 cm Viabil | Abd pain following day of stent placement; repeat ERCP showed residual bile duct stones | 57 | 41 | Patent |
| Bile leaks | |||||||||
| 13 | 75 | F | Cholecystectomy | Bile leak (high grade); complex | 10 mm × 80 mm Viabil | None | 99 | 1364 | Sealed |
| 14 | 49 | F | Cholecystectomy | Bile Leak (high grade) | 10 mm × 10 cm Viabil | None | 92 | 1294 | Sealed |
| 15 | 30 | F | Cholecystectomy | Bile leak (low grade); complex | 10 mm × 80 mm Viabil | Occluded stent after lost to follow-up (eight and a half months) | 251 | 1007 | Sealed |
| 16 | 64 | M | Cholecystectomy | Bile leak (high grade) | 8 mm × 8 cm Viabil | None | 62 | 575 | Sealed |
| 17 | 50 | M | Cholecystectomy | Bile leak (low grade) | 10 mm × 80 mm Viabil | Hepatic abscess and Sepsis | 48 | 28 | Sealed |
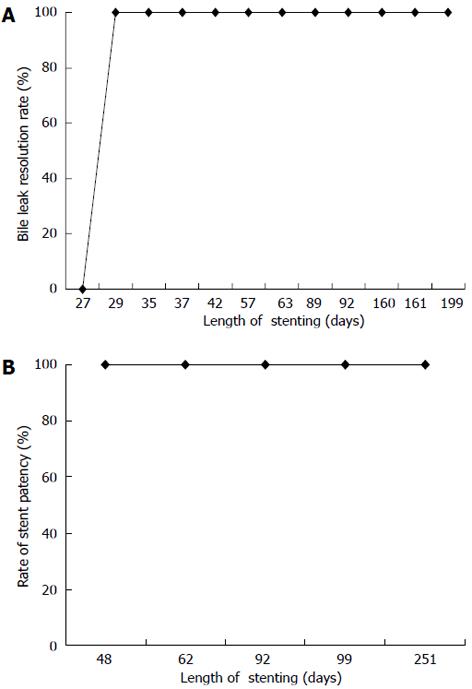
The median stent time was for all subjects was 63 d (mean 90.7 ± 65 d; range 27-199 d). The median stent time for the BBS group and bile leak group was 62 ± 58 d (range 27-199 d) and 92 ± 81 d (range 48-251 d), respectively. All 17 patients underwent successful FCSEMS removal. Resolution of biliary strictures and bile leaks was achieved in 16 of 17 patients (94%) (Figure 1). One patient with a bile leak did not have her stent removed until eight and a half months after placement due to loss of follow-up. An ERCP showed a sealed bile leak but an occluded stent which was extracted. One patient who failed stent therapy for BBS was referred to surgery. Long term follow-up, which included labs and symptom assessment, was obtained for a median of 575 d (range 28-1435 d). Follow-up was longer than one year in 10 cases (58%). No patients reported recurrent biliary pain or jaundice on follow-up. All patients liver function tests normalized. Figure 2 demonstrate stent placement for BBS and bile leaks.
Complications occurred in 5 of 17 patients (29%). Two patients were found to have proximal stent migration. The first was a Viabil metal stent seen to have migrated proximally. The time elapsed from stent placement to migration of FCSEMS was 27 d. Using rat-toothed forcepts the migrated stent was grasped at the distal end and removed. Prior to removal of the stent because the stent had migrated inwards, dilation of the sphincterotomy site was performed after which the distal end of the stent could be seen. The CBD stricture had resolved at the time of extraction. The second migration involved a Wallstent that migrated proximally. The time elapsed from the stent placement to migration was 66 d. A second Wallstent was placed into a migrated Wallstent. Both Wallstents were removed after 133 d.
Two additional patients had stent occlusion caused by intraluminal debris and bile duct stones. The time elapsed from stent placement to occlusion caused by intraluminal debris and bile duct stones were 160 and 251 d, respectively.
One patient developed recurrent cholangitis due to stent occlusion. One patient was found to have a hepatic abscess of the right liver lobe after stent placement for a bile leak. He later developed sepsis and was transferred to the intensive care unit (ICU). Overall, two patients had de novo choledocholithiasis and/or lumen debris that required multiple balloon sweeps and irrigation of the bile duct.
Placement of plastic stents with or without sphincterotomy is the most popular and accepted therapy for treatment of BBS and bile leaks in most centers. Short-term patency rates, limited stent diameter and requirement of multiple endoscopic sessions with stenting have led to the development of SEMS[13-15].
The overall success rate in our study for BBS and bile leaks was 94% (16/17). In the subset of patients with only BBS the success rate was 92% (11/12) (Figure 1A). Our results compare favorably to past results including a case series by Mahajan et al[3] of 44 patients with BBS who were treated with FCSEMS (Viabil, Conmed) and demonstrated a success rate of 83% (34/41) after median stent time of 3.3 mo (Table 1).
In our study, FCSEMS were removed only when all criteria described earlier in methods were met. All stents were able to be removed without any difficulty with median duration of 89 d (range 29-428 d). Although there is no consensus on the optimal duration of biliary stenting some advocate for no longer than 6 mo[2]. Several randomized trials have shown that FCSEMS remained patent for up to a median of 9 mo[16,17]. Our stricture recurrence rate was 0% after a median follow-up of 575 d. Dumonceau et al[5] reported a stricture recurrence was 19% on 36 patients who underwent plastic stent placement for BBS during a mean follow-up period of 44 mo.
Patients presenting with bile leaks had placement of Viabil FCSEMS. Viabil stents (Conmed, Utica, NY) are entirely covered with polytetrafluoroethylene and fluorinated ethylene propylene (ePTFE/FEP) liner that acts as a barrier to tissue ingrowth permitting long stent duration and easier extraction. Moreover, the anchoring fins placed on each tail end aid in prevent stent migration. Although our results are limited by the small sample size we were able to achieve 100% success rate in resolution of biliary leaks with temporary placement of FCSEMS for a mean of 110 ± 81 d (range, 48-251 d) (Figure 1B). The overall success rate for the subset of patients with bile leaks was 100%. Our results again compare favorably to past studies (Table 1). Three of 5 patients had high grade leaks and two of 5 patients had complex bile leaks refractory to previous stenting (Table 2). One patient had a biloma secondary to bile leak in the cystic duct and required percutaneous drain. She was lost to follow-up after eight and a half months and returned with abdominal pain. An ERCP revealed an occluded stent and intrahepatic ductal dilation. The stent was removed and a follow-up ERCP revealed a sealed bile leak.
Twelve patients had failed prior plastic stent placement and were referred to Harbor UCLA for refractory strictures (seven having had ≥ 2 stents). These subjects had fully covered metal stents placed for recurrent strictures. Because several of our subjects had stents placed at outside hospitals we are unable to determine the length of stenting, diameter or type of stent placed for all of these twelve patients. We did not encounter technical difficulties of FCSEMS placement in patients who had failed prior plastic stent therapy. There was no significant difference in total stenting time for patients that had failed prior plastic stenting and new onset strictures or bile leaks.
Upon stent removal, three patients were found to have biliary sludge and/or luminal debris requiring multiple balloon sweeps and irrigation. The incidence of sludge/luminal debris was noted to be proportional to the duration of stenting. One patient was found to have a hepatic abscess of the right liver lobe on computed tomography scan after stent placement for a bile leak proximal to the cystic duct. The patient was transferred to the ICU for sepsis. It is unclear whether the abscess developed as a result of ERCP or secondary to an underlying infection. One patient had developed cholangitis due to an occluded stent. This patient was treated successfully with placement of another stent within the original stent, multiple balloon sweeps to remove sludge, and a course of antibiotics. This compares favorably to rates of cholangitis observed with plastic stent placement for BBS of 18%[5]. The patient with stenting of a BBS localized to the right hepatic duct had an additional anastomosis done by surgery between the other hepatic duct and the duodenum. It is important to mention that unilateral placement of FCSEMS beyond the hepatic hilum harbors the risk of occluding the contralateral hepatic duct and side branches of the right or left hepatic ducts (depending on Bismuth stage of stricture). All complications in our study were treated conservatively.
Overall, our study showed that temporary placement of FCSEMS successfully treated BBS and bile leaks with excellent long-term patency rates and relatively few complications. FCSEMS may provide an effective method in management of BBS and bile leaks while allowing easy endoscopic removability. FCSEMS can be easily removed after insertion and remain in place for several months although there is insufficient data as to what the optimal duration of placement is. The high cost of FCSEMS may be offset by a reduction in ERCP sessions and recurrent stenting for recurrent strictures[14,15].
Management of bile leaks and benign biliary strictures (BBS) involves placement of plastic and uncovered metal stents which have been associated with limited long term stent patency secondary to stent lumen occlusion and epithelial hyperplasia, respectively.
Recent advances in development of endoprosthesis have led to the development of fully covered self-expanding metal stents (FCSEMs) which are coated circumferentially with a material that prevents stent occlusion due to bacterial colonization, tissue hyperplasia, and tumor in growth thereby increasing the duration of stent patency in BBS and bile leaks.
In this retrospective review of patients with BBS and Bile leaks treated with FCSEMS, stricture and bile leak resolution was achieved in 16 of 17 patients after a median follow-up time of 575 d after stent extraction.
This study demonstrated that temporary placement of FCSEMS successfully treated BBS and bile leaks with excellent long-term patency rates and relatively few complications.
The manuscript entitled “An evaluation of fully covered self-expanding metal stents in benign biliary strictures and complex bile leaks” provided valuable data about the safety and efficacy of FCSEMS for endoscopic treatment of benign biliary strictures and bile leaks.
P- Reviewer Pozsar J S- Editor Song XX L- Editor A E- Editor Zhang DN
| 1. | Kahaleh M, Sundaram V, Condron SL, De La Rue SA, Hall JD, Tokar J, Friel CM, Foley EF, Adams RB, Yeaton P. Temporary placement of covered self-expandable metallic stents in patients with biliary leak: midterm evaluation of a pilot study. Gastrointest Endosc. 2007;66:52-59. |
| 2. | Kahaleh M, Behm B, Clarke BW, Brock A, Shami VM, De La Rue SA, Sundaram V, Tokar J, Adams RB, Yeaton P. Temporary placement of covered self-expandable metal stents in benign biliary strictures: a new paradigm (with video). Gastrointest Endosc. 2008;67:446-454. |
| 3. | Mahajan A, Ho H, Sauer B, Phillips MS, Shami VM, Ellen K, Rehan M, Schmitt TM, Kahaleh M. Temporary placement of fully covered self-expandable metal stents in benign biliary strictures: midterm evaluation (with video). Gastrointest Endosc. 2009;70:303-309. |
| 4. | Bridges A, Wilcox CM, Varadarajulu S. Endoscopic management of traumatic bile leaks. Gastrointest Endosc. 2007;65:1081-1085. |
| 5. | Dumonceau JM, Devière J, Delhaye M, Baize M, Cremer M. Plastic and metal stents for postoperative benign bile duct strictures: the best and the worst. Gastrointest Endosc. 1998;47:8-17. |
| 6. | Katsinelos P, Kountouras J, Paroutoglou G, Chatzimavroudis G, Germanidis G, Zavos C, Pilpilidis I, Paikos D, Papaziogas B. A comparative study of 10-Fr vs. 7-Fr straight plastic stents in the treatment of postcholecystectomy bile leak. Surg Endosc. 2008;22:101-106. |
| 7. | Kaffes AJ, Hourigan L, De Luca N, Byth K, Williams SJ, Bourke MJ. Impact of endoscopic intervention in 100 patients with suspected postcholecystectomy bile leak. Gastrointest Endosc. 2005;61:269-275. |
| 8. | Sandha GS, Bourke MJ, Haber GB, Kortan PP. Endoscopic therapy for bile leak based on a new classification: results in 207 patients. Gastrointest Endosc. 2004;60:567-574. |
| 9. | Davids PH, Tanka AK, Rauws EA, van Gulik TM, van Leeuwen DJ, de Wit LT, Verbeek PC, Huibregtse K, van der Heyde MN, Tytgat GN. Benign biliary strictures. Surgery or endoscopy. Ann Surg. 1993;217:237-243. |
| 10. | Levy MJ, Baron TH, Gostout CJ, Petersen BT, Farnell MB. Palliation of malignant extrahepatic biliary obstruction with plastic versus expandable metal stents: An evidence-based approach. Clin Gastroenterol Hepatol. 2004;2:273-285. |
| 11. | Siegel JH, Pullano W, Kodsi B, Cooperman A, Ramsey W. Optimal palliation of malignant bile duct obstruction: experience with endoscopic 12 French prostheses. Endoscopy. 1988;20:137-141. |
| 12. | Pausawasadi N, Soontornmanokul T, Rerknimitr R. Role of fully covered self-expandable metal stent for treatment of benign biliary strictures and bile leaks. Korean J Radiol. 2012;13 Suppl 1:S67-S73. |
| 13. | García-Cano J. Use of fully covered self-expanding metal stents in benign biliary diseases. World J Gastrointest Endosc. 2012;4:142-147. |
| 14. | Behm B, Brock A, Clarke BW, Adams RB, Northup PG, Yeaton P, Kahaleh M. Cost analysis of temporarily placed covered self expandable metallic stents versus plastic stents in biliary strictures related to chronic pancreatitis (abstract). Gastrointest Endosc. 2007;65:AB211. |
| 15. | Draganov P, Hoffman B, Marsh W, Cotton P, Cunningham J. Long-term outcome in patients with benign biliary strictures treated endoscopically with multiple stents. Gastrointest Endosc. 2002;55:680-686. |
| 16. | Soderlund C, Linder S. Covered metal versus plastic stents for malignant common bile duct stenosis: a prospective, randomized, controlled trial. Gastrointest Endosc. 2006;63:986-995. |
| 17. | Davids PH, Groen AK, Rauws EA, Tytgat GN, Huibregtse K. Randomised trial of self-expanding metal stents versus polyethylene stents for distal malignant biliary obstruction. Lancet. 1992;340:1488-1492. |
| 18. | Deviere J, Cremer M, Baize M, Love J, Sugai B, Vandermeeren A. Management of common bile duct stricture caused by chronic pancreatitis with metal mesh self expandable stents. Gut. 1994;35:122-126. |
| 19. | Cantù P, Hookey LC, Morales A, Le Moine O, Devière J. The treatment of patients with symptomatic common bile duct stenosis secondary to chronic pancreatitis using partially covered metal stents: a pilot study. Endoscopy. 2005;37:735-739. |
| 20. | Kuo MD, Lopresti DC, Gover DD, Hall LD, Ferrara SL. Intentional retrieval of viabil stent-grafts from the biliary system. J Vasc Interv Radiol. 2006;17:389-397. |
| 21. | Cahen DL, Rauws EA, Gouma DJ, Fockens P, Bruno MJ. Removable fully covered self-expandable metal stents in the treatment of common bile duct strictures due to chronic pancreatitis: a case series. Endoscopy. 2008;40:697-700. |
| 22. | García-Cano J, Taberna-Arana L, Jimeno-Ayllón C, Martínez-Fernández R, Serrano-Sánchez L, Reyes-Guevara AK, Viñuelas-Chicano M, Gómez-Ruiz CJ, Morillas-Ariño MJ, Pérez-García JI. Use of fully covered self-expanding metal stents for the management of benign biliary conditions. Rev Esp Enferm Dig. 2010;102:526-532. |