Published online Jul 16, 2013. doi: 10.4253/wjge.v5.i7.308
Revised: May 29, 2013
Accepted: June 18, 2013
Published online: July 16, 2013
Processing time: 142 Days and 6.2 Hours
Chronic radiation proctitis is a complication that occurs in patients who receive radiation therapy for pelvic malignancies. The common presentation is with rectal bleeding, but also rectal pain, diarrhea, tenesmus and even passage of mucus can occur. The optimal treatment of bleeding due to radiation proctitis remains unclear. Among various therapeutic options, medical management is generally ineffective and surgical intervention has a high incidence of morbidity. Promising advances have been made in endoscopic therapy, including argon plasma coagulation (APC), formalin application as well as new techniques such as radiofrequency ablation and cryoablation. APC is a safe, highly effective and long-lasting therapy in patients with rectal bleeding associated with radiation proctitis. It has been shown that several sessions of APC reduce the rate of bleeding and therefore the blood transfusion requirements. Moreover, the effect of treatment is long lasting. However, best results are achieved in patients with mild to moderate radiation proctitis, leaving space for alternative treatments for patients with more severe disease. In patients with severe or refractory radiation proctitis intra rectal formalin application is an appropriate treatment option. Radiofrequency ablation and cryoablation have shown efficacy as alternative methods in a limited number of patients with refractory chronic radiation proctitis.
Core tip: Chronic radiation proctitis presents with rectal bleeding, pain, diarrhea, tenesmus and passage of mucus. Among other therapeutic options, endoscopic therapy with argon plasma coagulation (APC) is a safe and highly effective in patients with rectal bleeding associated with radiation proctitis. Although best results are achieved in patients with mild to moderate lesions, APC therapy reduces the rate of bleeding and blood transfusion requirements and its effect last for long. In patients with severe or refractory radiation proctitis intra rectal formalin application,radiofrequency ablation and cryoablation have shown efficacy in a limited number of patients.
- Citation: Karamanolis G, Psatha P, Triantafyllou K. Endoscopic treatments for chronic radiation proctitis. World J Gastrointest Endosc 2013; 5(7): 308-312
- URL: https://www.wjgnet.com/1948-5190/full/v5/i7/308.htm
- DOI: https://dx.doi.org/10.4253/wjge.v5.i7.308
The rectum is often injured during pelvic radiation due to its fixed position and its anatomical proximity to the radiated target organ such as prostate and cervix. Radiation proctitis, usually mild, is a complication that occurs in up to 15% of patients who receive radiation therapy for pelvic malignancies. Radiation damage may occur in acute or chronic form. Acute complications are seen during or up to 6 wk after radiotherapy, whereas late radiation injury usually occurs in the first 2-3 years after treatment[1-3]. A change in the treatment practices has recently occurred toward escalating radiation doses with improved local control. Conformal radiotherapy of pelvic tumors focuses on reducing irradiation of organs at risk such as rectum[4,5]. Although the incidence of complications has been reduced using this new technology, rectal wall damage continues to be an important side effect of pelvic radiotherapy[6-9].
The common presentation of radiation proctitis is with rectal bleeding, but also rectal pain, diarrhea, tenesmus and even passage of mucus can occur. In approximately 35% of patients the symptoms are mild and settle spontaneously over several months without any treatment. However, rectal bleeding due to chronic radiation proctitis may lead to anemia and necessitate repeated blood transfusions. Medical treatment with salicylates, sucralfate or corticosteroids enemas is usually not beneficial[10-14]. Thus, alternative treatments including endoscopic ones have been used. Among endoscopic treatments, argon plasma coagulation (APC), a nontouch thermo ablative therapy, is increasingly recommended as first line treatment for patients with radiation proctitis.
For evaluation of endoscopic severity of radiation proctitis, a scoring system with measurement of three independent factors (telangiectasia distribution, surface area involved and the presence of fresh blood) was proposed[15] (Table 1). A cumulative score was calculated and three categories of endoscopic severity of radiation proctitis were derived: grade A (mild, 2 points), grade B (moderate, 3 points), and grade C (severe 4/5 points).
| Distribution of telangiectasias | Surface area covered by telangiectasias | Presence of fresh blood |
| Distal rectum (within 10 cm from anal verge): 1 point | Less than 50%: 1 point | No fresh blood: 0 points |
| Entire rectum +/- sigmoid (more than 10 cm from anal verge: 2 points | More than 50%: 2 points | Fresh blood : 1 point |
Recently, we prospectively investigated in a large number of patients the effectiveness of APC in treating patients with various endoscopic grading of radiation proctitis (mild, moderate, and severe) using a modified scoring system with measurement of two independent factors for evaluation of endoscopic severity: telangiectasia distribution and surface area involved[16]. For APC application, an ERBE APC 300 (ERBE Elektromedizin, Tubingen, Germany) argon delivery unit and a 2.3 mm diameter front-firing APC probe inserted through the working channel of the flexible sigmoidoscopy were used. The argon flow rate and the electrical power were set at 2.0 L/min and 40 W, respectively.
Our results showed that APC was successful in all patients with mild and in almost all patients with moderate radiation proctitis. In contrary, in the presence of severe mucosal damage APC failed in 50% of patients. Patients with mild proctitis required 1-2 sessions of APC, while patients with moderately to severe form required a statistically significantly higher number of APC sessions. Our results were in accordance with the existing literature; APC is the preferred method in patients with rectal bleeding associated with mild to moderate radiation proctitis, while in cases of severe and diffuse involvement of the rectum multiple treatments sessions are required and success is less certain[17-26]. We also presented long-term follow up of patients successfully treated with APC and showed that during a follow-up of a mean of 17.9 mo (range 6-33 mo) about 90% of these patients remain in clinical remission.
Till now there is no consensus for the optimal APC settings (power and gas flow rate) for successful and safe coagulation. In the literature the power setting for APC ranged from 25-80 W and for the argon flow rate ranged from 0.6-2 L/min[21]. In our study low-power settings (argon flow rate and electrical power were set at 2.0 L/min and 40 W, respectively) were used. Although these settings were among the lowest reported in the literature seemed adequate for successful coagulation and also carried low rate of complications.
The optimal number of treatment sessions is still unknown. APC is traditionally not applied in 1 treatment session, particularly in patients with severe disease, because of the concern regarding strictures formation. For therapeutic success, the median number of sessions per patient was ranged from 1 to 3.7[27]. Similarly to previous reports, multiple sessions of APC were performed in our patients with a maximum of eight sessions in a patient with severe radiation proctitis.
In severe cases of radiation proctitis and in cases resistant to other treatment modalities intra-rectal formalin is a useful strategy[28]. Formalin is a mixture of methanol and formaldehyde which covalently binds to proteins, and causes cell necrosis. It acts as a haemostatic agent causing chemical cauterization to control bleeding from telengiectatic mucosal and submucosal vessels. Most used 4% dilute formalin applied to the rectum mucosa either by direct application of formalin-soaked gauze or by ‘instilling’ the solution in single or multiple aliquots down the operating channel of a colonoscope. Various volumes of formalin and different mucosal contact time were reported. Mean number of treatment ranged from 1.1-3.4 per patient. Cessation of bleeding occurred in most studies in the range of 60%-100%[28-38]. Median follow ups to a period of two years have shown only a minimal relapse among responders. Reported side effects include anal stenosis, fissures, fecal incontinence and ulceration of mucosa.
The theoretical benefits of radiofrequency ablation are extrapolated are studies treating gastric antral vascular ectasia and Barrett’s esophagus. Effective control of lower gastrointestinal bleeding in patients with refractory chronic radiation proctitis using radiofrequency ablation (RFA) with the Halo90 system has been recently reported[39-41]. RFA was performed in an outpatient practice using a single use Halo90 electrode catheter that was fit on the distal end of a standard flexible sigmoidoscope. An energy density of 12 J/cm2 at a power density of 40 W/cm2 was chosen based on previous studies performed, which showed no transmural injury at these settings. In all cases, the procedure was well tolerated and hemostasis was achieved after 1 or 2 RFA sessions. Re-epithelialization of squamous mucosa was observed over areas of prior hemorrhage. Patients were symptom free on follow-up up to 19 mo after treatment.
Cryoablation involves noncontact application of liquid nitrogen or carbon dioxide gas to tissue for superficial ablation[42]. Cryospray ablation has been used to treat esophageal high-grade dysplasia and early cancer. In two recent studies endoscopic cryoablation was performed in 20 patients with hemorrhagic radiation proctitis[43,44]. Endoscopic severity and subjective clinical scores improved in all patients. Cryoablation was performed with a catheter placed through the endoscope under direct endoscopic visualization to approximately 0.5 to 1.0 cm from the tip of the endoscope. The spray was applied for 5 s and the treatment area was then allowed to thaw no less than 45 s before initiating subsequent cryospray applications. Required sessions ranged from one to four and endoscopic score significantly improved, as well as, rectal pain and rectal bleeding. Although patients tolerated the procedure well, one patient experienced a cecal perforation[43] after therapy probably due to over insufflation during the procedure.
Hyperbaric oxygen therapy (HBOT) is the use of 100% oxygen at pressures greater than atmospheric pressure. The patient breathes 100% oxygen intermittently, while the pressure of the treatment chamber is increased to greater than 1 atmosphere absolute. HBOT promotes angiogenesis and hyperoxygenation to the irradiated tissues. Increasing the oxygen content to the surrounding tissues markedly increases the overall oxygen gradient between these tissues and the central hypoxic area. The increased oxygen gradient is the essential catalytic factor for angiogenesis[45].
Unfortunately, the research into the use of HBOT in radiation proctitis is heterogeneous in terms of duration of treatment, number of treatments and pressures of HBOT used. Warren et al[46] reported a response rate of 64%, with complete symptomatic resolution in 57%, in 14 cases of radiation proctitis treated with varying doses of HBOT. Girnius et al[47] reported nine patients with refractory haemorrhagic proctitis who had failed previous therapy; all patients had some response to HBOT and seven had complete resolution of their rectal bleeding. Jones et al[48] also found that 8 out of 10 patients with refractory radiation proctitis responded to HBOT. Dall’Era et al[49] found that a total of 48% of 27 patients with treatment-resistant radiation proctitis had complete resolution of bleeding and 28% of them had significantly fewer bleeding episodes. Similarly, a recent study reported that HBOT significantly improved the healing responses in patients with refractory radiation proctitis, generating an absolute risk reduction of 32% (number needed to treat of 3)[50]. Although HBOT appears to be of value for refractory radiation proctitis, the quality of current data is poor with marked variability between studies. Moreover, the cost of HBO is high enough, and it is not widely applicable.
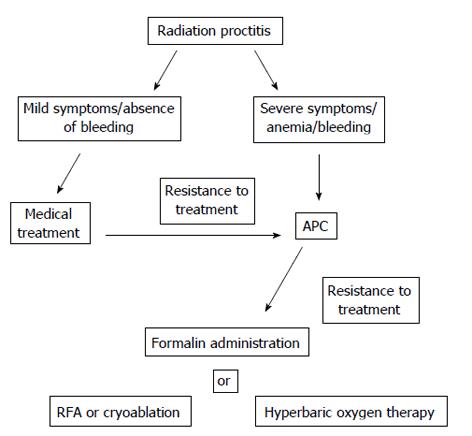
Based on currently data, APC is the favored treatment for bleeding from chronic radiation proctitis. APC is a safe, highly effective and long-lasting therapy in patients with rectal bleeding associated with endoscopic mild radiation proctitis. In severe radiation proctitis multiple APC applications are usually required and success is likely to be more limited. In these patients other treatment options such as intra rectal formalin application should be considered. New therapeutic endoscopic modalities, including radiofrequency ablation and cryoablation, showed effective control of lower gastrointestinal bleeding in patients with refractory chronic radiation proctitis. As the number of patients treated with these new modalities was limited, further studies are needed to identify their safety and efficacy. Figure 1 summarized the treatment modalities available for patients with radiation proctitis.
P- Reviewer Supiot S S- Editor Wen LL L- Editor A E- Editor Zhang DN
| 1. | Deitel M, Vasic V. Major intestinal complications of radiotherapy. Am J Gastroenterol. 1979;72:65-70. |
| 2. | Pedersen D, Bentzen SM, Overgaard J. Early and late radiotherapeutic morbidity in 442 consecutive patients with locally advanced carcinoma of the uterine cervix. Int J Radiat Oncol Biol Phys. 1994;29:941-952. |
| 3. | Schultheiss TE, Lee WR, Hunt MA, Hanlon AL, Peter RS, Hanks GE. Late GI and GU complications in the treatment of prostate cancer. Int J Radiat Oncol Biol Phys. 1997;37:3-11. |
| 4. | Kutcher GJ, Burman C. Calculation of complication probability factors for non-uniform normal tissue irradiation: the effective volume method. Int J Radiat Oncol Biol Phys. 1989;16:1623-1630. |
| 5. | Lebesque JV, Bruce AM, Kroes AP, Touw A, Shouman RT, van Herk M. Variation in volumes, dose-volume histograms, and estimated normal tissue complication probabilities of rectum and bladder during conformal radiotherapy of T3 prostate cancer. Int J Radiat Oncol Biol Phys. 1995;33:1109-1119. |
| 6. | Storey MR, Pollack A, Zagars G, Smith L, Antolak J, Rosen I. Complications from radiotherapy dose escalation in prostate cancer: preliminary results of a randomized trial. Int J Radiat Oncol Biol Phys. 2000;48:635-642. |
| 7. | Lee WR, Hanks GE, Hanlon AL, Schultheiss TE, Hunt MA. Lateral rectal shielding reduces late rectal morbidity following high dose three-dimensional conformal radiation therapy for clinically localized prostate cancer: further evidence for a significant dose effect. Int J Radiat Oncol Biol Phys. 1996;35:251-257. |
| 8. | Dearnaley DP, Khoo VS, Norman AR, Meyer L, Nahum A, Tait D, Yarnold J, Horwich A. Comparison of radiation side-effects of conformal and conventional radiotherapy in prostate cancer: a randomised trial. Lancet. 1999;353:267-272. |
| 9. | O’Brien PC, Hamilton CS, Denham JW, Gourlay R, Franklin CI. Spontaneous improvement in late rectal mucosal changes after radiotherapy for prostate cancer. Int J Radiat Oncol Biol Phys. 2004;58:75-80. |
| 10. | Kochhar R, Patel F, Dhar A, Sharma SC, Ayyagari S, Aggarwal R, Goenka MK, Gupta BD, Mehta SK. Radiation-induced proctosigmoiditis. Prospective, randomized, double-blind controlled trial of oral sulfasalazine plus rectal steroids versus rectal sucralfate. Dig Dis Sci. 1991;36:103-107. |
| 11. | Kochhar R, Sriram PV, Sharma SC, Goel RC, Patel F. Natural history of late radiation proctosigmoiditis treated with topical sucralfate suspension. Dig Dis Sci. 1999;44:973-978. |
| 12. | Ladas SD, Raptis SA. Sucralfate enemas in the treatment of chronic postradiation proctitis. Am J Gastroenterol. 1989;84:1587-1589. |
| 13. | Talley NA, Chen F, King D, Jones M, Talley NJ. Short-chain fatty acids in the treatment of radiation proctitis: a randomized, double-blind, placebo-controlled, cross-over pilot trial. Dis Colon Rectum. 1997;40:1046-1050. |
| 14. | Pinto A, Fidalgo P, Cravo M, Midões J, Chaves P, Rosa J, dos Anjos Brito M, Leitão CN. Short chain fatty acids are effective in short-term treatment of chronic radiation proctitis: randomized, double-blind, controlled trial. Dis Colon Rectum. 1999;42:788-795; discussion 795-796. |
| 15. | Zinicola R, Rutter MD, Falasco G, Brooker JC, Cennamo V, Contini S, Saunders BP. Haemorrhagic radiation proctitis: endoscopic severity may be useful to guide therapy. Int J Colorectal Dis. 2003;18:439-444. |
| 16. | Karamanolis G, Triantafyllou K, Tsiamoulos Z, Polymeros D, Kalli T, Misailidis N, Ladas SD. Argon plasma coagulation has a long-lasting therapeutic effect in patients with chronic radiation proctitis. Endoscopy. 2009;41:529-531. |
| 17. | Sebastian S, O’Connor H, O’Morain C, Buckley M. Argon plasma coagulation as first-line treatment for chronic radiation proctopathy. J Gastroenterol Hepatol. 2004;19:1169-1173. |
| 18. | Silva RA, Correia AJ, Dias LM, Viana HL, Viana RL. Argon plasma coagulation therapy for hemorrhagic radiation proctosigmoiditis. Gastrointest Endosc. 1999;50:221-224. |
| 19. | Dees J, Meijssen MA, Kuipers EJ. Argon plasma coagulation for radiation proctitis. Scand J Gastroenterol Suppl. 2006;175-178. |
| 20. | Villavicencio RT, Rex DK, Rahmani E. Efficacy and complications of argon plasma coagulation for hematochezia related to radiation proctopathy. Gastrointest Endosc. 2002;55:70-74. |
| 21. | Lam MC, Parliament M, Wong CK. Argon plasma coagulation for the treatment of hemorrhagic radiation colitis. Case Rep Gastroenterol. 2012;6:446-451. |
| 22. | Kwan V, Bourke MJ, Williams SJ, Gillespie PE, Murray MA, Kaffes AJ, Henriquez MS, Chan RO. Argon plasma coagulation in the management of symptomatic gastrointestinal vascular lesions: experience in 100 consecutive patients with long-term follow-up. Am J Gastroenterol. 2006;101:58-63. |
| 23. | Swan MP, Moore GT, Sievert W, Devonshire DA. Efficacy and safety of single-session argon plasma coagulation in the management of chronic radiation proctitis. Gastrointest Endosc. 2010;72:150-154. |
| 24. | D’Costa SM, Yao H, Bilimoria SL. Transcription and temporal cascade in Chilo iridescent virus infected cells. Arch Virol. 2001;146:2165-2178. |
| 25. | Rotondano G, Bianco MA, Marmo R, Piscopo R, Cipolletta L. Long-term outcome of argon plasma coagulation therapy for bleeding caused by chronic radiation proctopathy. Dig Liver Dis. 2003;35:806-810. |
| 26. | Taïeb S, Rolachon A, Cenni JC, Nancey S, Bonvoisin S, Descos L, Fournet J, Gérard JP, Flourié B. Effective use of argon plasma coagulation in the treatment of severe radiation proctitis. Dis Colon Rectum. 2001;44:1766-1771. |
| 27. | Wilson SA, Rex DK. Endoscopic treatment of chronic radiation proctopathy. Curr Opin Gastroenterol. 2006;22:536-540. |
| 28. | Leiper K, Morris AI. Treatment of radiation proctitis. Clin Oncol (R Coll Radiol). 2007;19:724-729. |
| 29. | Rubinstein E, Ibsen T, Rasmussen RB, Reimer E, Sørensen BL. Formalin treatment of radiation-induced hemorrhagic proctitis. Am J Gastroenterol. 1986;81:44-45. |
| 30. | Seow-Choen F, Goh HS, Eu KW, Ho YH, Tay SK. A simple and effective treatment for hemorrhagic radiation proctitis using formalin. Dis Colon Rectum. 1993;36:135-138. |
| 31. | Saclarides TJ, King DG, Franklin JL, Doolas A. Formalin instillation for refractory radiation-induced hemorrhagic proctitis. Report of 16 patients. Dis Colon Rectum. 1996;39:196-199. |
| 32. | Ismail MA, Qureshi MA. Formalin dab for haemorrhagic radiation proctitis. Ann R Coll Surg Engl. 2002;84:263-264. |
| 33. | Chautems RC, Delgadillo X, Rubbia-Brandt L, Deleaval JP, Marti MC, Roche B. Formaldehyde application for haemorrhagic radiation-induced proctitis: a clinical and histological study. Colorectal Dis. 2003;5:24-28. |
| 34. | Roche B, Chautems R, Marti MC. Application of formaldehyde for treatment of hemorrhagic radiation-induced proctitis. World J Surg. 1996;20:1092-1094; discussion 1092-1094;. |
| 35. | Parikh S, Hughes C, Salvati EP, Eisenstat T, Oliver G, Chinn B, Notaro J. Treatment of hemorrhagic radiation proctitis with 4 percent formalin. Dis Colon Rectum. 2003;46:596-600. |
| 36. | de Parades V, Etienney I, Bauer P, Bourguignon J, Meary N, Mory B, Sultan S, Taouk M, Thomas C, Atienza P. Formalin application in the treatment of chronic radiation-induced hemorrhagic proctitis--an effective but not risk-free procedure: a prospective study of 33 patients. Dis Colon Rectum. 2005;48:1535-1541. |
| 37. | Pironi D, Panarese A, Vendettuoli M, Pontone S, Candioli S, Manigrasso A, De Cristofaro F, Filippini A. Chronic radiation-induced proctitis: the 4 % formalin application as non-surgical treatment. Int J Colorectal Dis. 2013;28:261-266. |
| 38. | Nelamangala Ramakrishnaiah VP, Javali TD, Dharanipragada K, Reddy KS, Krishnamachari S. Formalin dab, the effective way of treating haemorrhagic radiation proctitis: a randomized trial from a tertiary care hospital in South India. Colorectal Dis. 2012;14:876-882. |
| 39. | Nikfarjam M, Faulx A, Laughinghouse M, Marks JM. Feasibility of radiofrequency ablation for the treatment of chronic radiation proctitis. Surg Innov. 2010;17:92-94. |
| 40. | Zhou C, Adler DC, Becker L, Chen Y, Tsai TH, Figueiredo M, Schmitt JM, Fujimoto JG, Mashimo H. Effective treatment of chronic radiation proctitis using radiofrequency ablation. Therap Adv Gastroenterol. 2009;2:149-156. |
| 41. | Eddi R, Depasquale JR. Radiofrequency ablation for the treatment of radiation proctitis: a case report and review of literature. Therap Adv Gastroenterol. 2013;6:69-76. |
| 42. | Ritch CR, Katz AE. Update on cryotherapy for localized prostate cancer. Curr Urol Rep. 2009;10:206-211. |
| 43. | Hou JK, Abudayyeh S, Shaib Y. Treatment of chronic radiation proctitis with cryoablation. Gastrointest Endosc. 2011;73:383-389. |
| 44. | Moawad FJ, Maydonovitch CL, Horwhat JD. Efficacy of cryospray ablation for the treatment of chronic radiation proctitis in a pilot study. Dig Endosc. 2013;25:174-179. |
| 45. | Bennett MH, Feldmeier J, Hampson N, Smee R, Milross C. Hyperbaric oxygen therapy for late radiation tissue injury. Cochrane Database Syst Rev. 2012;5:CD005005. |
| 46. | Warren DC, Feehan P, Slade JB, Cianci PE. Chronic radiation proctitis treated with hyperbaric oxygen. Undersea Hyperb Med. 1997;24:181-184. |
| 47. | Girnius S, Cersonsky N, Gesell L, Cico S, Barrett W. Treatment of refractory radiation-induced hemorrhagic proctitis with hyperbaric oxygen therapy. Am J Clin Oncol. 2006;29:588-592. |
| 48. | Jones K, Evans AW, Bristow RG, Levin W. Treatment of radiation proctitis with hyperbaric oxygen. Radiother Oncol. 2006;78:91-94. |
| 49. | Dall’Era MA, Hampson NB, Hsi RA, Madsen B, Corman JM. Hyperbaric oxygen therapy for radiation induced proctopathy in men treated for prostate cancer. J Urol. 2006;176:87-90. |
| 50. | Clarke RE, Tenorio LM, Hussey JR, Toklu AS, Cone DL, Hinojosa JG, Desai SP, Dominguez Parra L, Rodrigues SD, Long RJ. Hyperbaric oxygen treatment of chronic refractory radiation proctitis: a randomized and controlled double-blind crossover trial with long-term follow-up. Int J Radiat Oncol Biol Phys. 2008;72:134-143. |