Published online Dec 16, 2010. doi: 10.4253/wjge.v2.i12.404
Revised: October 26, 2010
Accepted: November 2, 2010
Published online: December 16, 2010
In this report, a patient was exposed to an herbal remedy for hypercholesterolemia. She became acutely jaundiced while taking the remedy and presented for medical care. Endoscopic ultrasound was utilized, and found a distal common bile duct mass. Endoscopic retrograde cholangiopancreatography guided bile duct biopsies revealed that the mass was cholangiocarcinoma (CCA). This case highlights a unique association between autoimmune hepatitis and CCA. It also highlights that EUS can be safely used in patients with cirrhosis to spare invasive evaluation such as exploratory laporotomy for diagnosis and staging of cholangiocarcinoma.
- Citation: Rial NS, Henderson JT, Bhattacharyya AK, Nadir A, Cunningham JT. Use of endoscopic ultrasound for diagnosis of cholangiocarcinoma in auto-immune hepatitis. World J Gastrointest Endosc 2010; 2(12): 404-407
- URL: https://www.wjgnet.com/1948-5190/full/v2/i12/404.htm
- DOI: https://dx.doi.org/10.4253/wjge.v2.i12.404
Cholangiocarcinoma (CCA) is an adenocarcinoma arising from the epithelial tissue of the intra-hepatic (10%), hepatic hilar (25%) or extrahepatic (65%) bile ducts[1]. Among gastrointestinal (GI) cancers, CCA is the most difficult to detect and diagnose with a 5 year survival of less than 5%[2]. Recently, endoscopic ultrasound (EUS) has emerged as an important modality in the diagnosis of CCA[3]. EUS guided fine needle aspirate (FNA) has a specificity of 100%, and a sensitivity of 43%-86% depending upon the location of the cholangiocarcinoma[4]. The negative predictive value for EUS-FNA for cholangiocarcinoma is reported at 29%[5]. The additional benefit of EUS-FNA is to sample regional lymph nodes to stage the disease particularly in the context of liver transplant evaluation[4].
Herein a unique case of autoimmune hepatitis that presents with jaundice is described. Here we report that in a cirrhotic patient, EUS was extremely helpful in making the diagnosis of CCA through identification of suspected lesions. EUS and subsequent endoscopic retrograde cholangiopancreatography (ERCP) spared the patient an exploratory laprotomy which has inherent risks related to general anesthesia, intubation, abdominal insufflation and biopsy of masses. EUS is especially helpful in noting differences in echogenicity among normal tissue, lymph nodes and neoplasms. This distinction makes EUS a selective tool for subsequent biopsy. In this way, the pre-test probability is higher compared to gross visualization during an exploratory laprotomy.
A 64-year-old Caucasian female was referred to University Medical Center (UMC) following a diagnosis of autoimmune hepatitis and cirrhosis that was made at an outside hospital. She had used an over-the-counter herbal remedy “CholestOff” for six months. Prior to using CholesOff, her liver function tests (LFTs) were normal, as documented by her primary care physician (PCP). During her six months of CholestOff therapy, LFTs were evaluated showing a total bilirubin of 1.2 g/dL (0.2-1.0), alkaline phosphatase of 272 IU/L (38-126), aspartate aminotransferase (AST) of 310 IU/L (7-40), and alanine aminotransferase (ALT) of 223 IU/L (7-40). CholestOff therapy was ceased and over a period of two months her LFTs showed a downward trend with an alkaline phosphatase of 167 IU/L, an AST of 207 IU/L, and ALT of 145 IU/L. Four months later, her LFTs were repeated again and showed an alkaline phosphatase of 272 IU/L, AST of 496 IU/L, and ALT of 420 IU/L and bilirubin of 0.8 mg/dL.
Two weeks later, the patient presented to an outside hospital with painless jaundice. Her laboratory results showed an alkaline phosphatase of 357 IU/L, AST of 1464 IU/L, ALT of 1090 IU/L, total bilirubin of 4.6 mg/dL, international normalized ratio (INR) greater than 9.4 and albumin of 2.9 g/dL (3.5-5.5). She tested positive for anti-nuclear antibody (ANA) and antimitochondrial antibody (AMA) at 11.4 (0.0-0.9) and 0.2 (< 0.1) respectively. The antismooth muscle antibody was negative. Her gamma globulins were elevated at 2.4 g/dL (0.6-1.6) while her acute hepatitis panel was negative. Further investigation lead to a transjugular liver biopsy. The results indicated portal inflammation with mixed infiltrate comprising of predominantly lymphocytes, readily identifiable plasma cells and cirrhosis. Bile duct injury and bile duct proliferation were also noted.
A Computed tomography (CT) scan was performed and showed mild intrahepatic and extrahepatic ductal dilation with a nodular contour of the liver. Multiple prominent aortocaval, retrocrural and portacaval nodes as well as hypodensity of the distal common bile duct were also noted. An ERCP was performed and showed a subtle, two centimeter common bile duct (CBD) stricture. Cytology sampling was followed by biliary sphincterotomy and placement of a 7 cm/10 Fr. biliary stent. The cytological analysis did not identify malignant cells. The patient was placed on a combination of prednisone and azathioprine therapy with resolution of her jaundice.
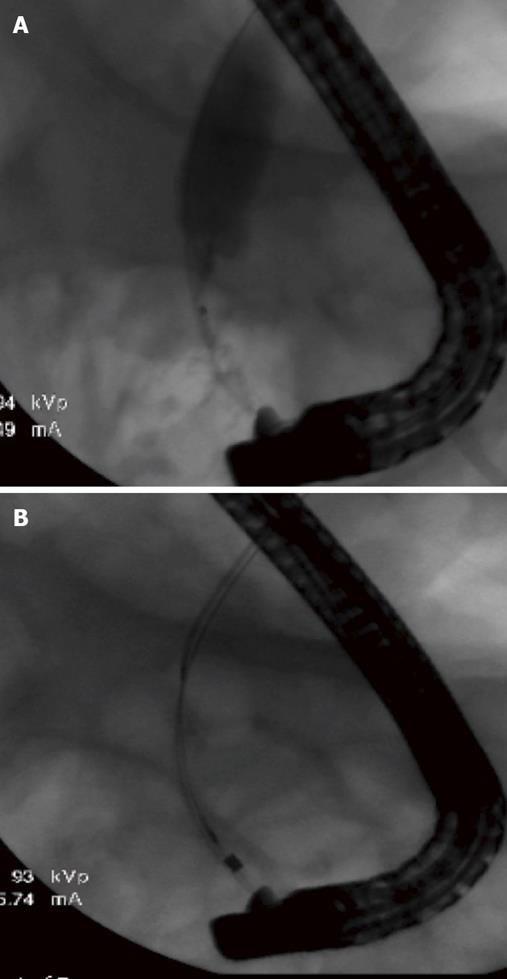
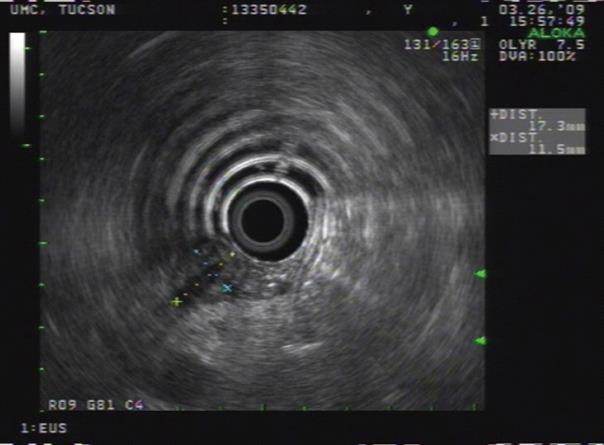
Three months later the patient underwent further evaluation at UMC with an EUS examination. At that time a 15 mm hypoechoic mass in the distal CBD (Figure 1A) was identified. Multiple biopsies followed by biliary brushings of the distal CBD mass were accomplished during an ERCP (Figure 1B). The CBD biopsy demonstrated benign acute inflammation and the brushings of the CBD mass were positive for CCA on cytological analysis. While the stricture seen at ERCP was subtle, the CBD mass was very apparent on EUS (Figure 2) and led to a rigorous work up.
The case highlights that EUS is a safe and valuable tool in establishing the diagnosis and staging of CCA in patients with cirrhosis. EUS-FNA has been successfully used for the staging of CCA before consideration of liver transplantation[4-8]. The technique has been extensively used to biopsy the bile duct, gallbladder[6,7], hepatic hilum[8], regional lymph nodes[9] pancreatic lesions[10] and hepatic lesions[11] as well as for aspiration of malignant ascites[12]. CT guided FNA has been utilized to biopsy peritoneal and omental masses[13].
This case also delineates the difficulties encountered while managing patients with cholangiocarcinoma and cirrhosis[14,15]. Moreover, the association of autoimmune hepatitis with cholangiocarcinoma is interesting. Only one other case of autoimmune hepatitis has been described in association with CCA and the authors of the report suggested that autoimmune hepatitis is a potential risk factor for the development of CCA[16]. In that particular case, the patient had a diagnosis of autoimmune hepatitis for 30 years and was treated with azathioprine and prednisone and was found to have a small hepatic lesion. That patient underwent a liver transplantation with the presumed diagnosis of hepatocellular carcinoma and developed recurrence of cholangiocarcinoma in distant lymph nodes within a few months of liver transplantation and expired[16].
Our patient underwent EUS that showed a 15-mm hypoechoic CBD mass. EUS guided FNA was not done on this particular patient because of the risk of potential tumor seeding. Instead, the patient underwent a second ERCP with brushing and cytological analysis which documented the correct diagnosis of cholangiocarcinoma, sparing her exploratory laporotomy, general anesthesia, insufflation of the abdomen and tissue biopsy.
This case describes a rare association of CCA with autoimmune hepatitis. The patient developed hepatitis while taking an herbal medication CholestOff which consists of plant sterols/stanols, tribasic calcium phosphate, croscarmellose sodium, calcium carbonate, hydroxypropyl methyl-cellulose, silicon dioxide, magnesium stearate, taranium dioxide, polyethyelene glycol, triethyl citrate, polysorbate 80 and sodium citrate. Only one other case of autoimmune hepatitis has been described in association with CCA and it has been suggested that autoimmune hepatitis is a potential risk factor for the development of CCA[16]. It is possible that the herbal medicine may have caused bile duct toxicity, autoimmune hepatitis and resulted in transformation to malignant cells. However, it appears more plausible that the herbal remedy simply resulted in drug-induced hepatitis with histological findings that mimic autoimmune hepatitis[17]. The case also reinforces the suspicion that the association of autoimmune hepatitis with CCA may be more than mere coincidence.
Peer reviewers: Vasileios Panteris, MD, FEBG, Department of Gastroenterology and Hepatology, Erasmus MC University Medical Center, Rotterdam, The Netherlands; David J Desilets, MD, PhD, Chief, Division of Gastroenterology, Springfield Bldg, Rm S2606, Department of Medicine; Assistant Professor of Clinical Medicine, Tufts University School of Medicine, Springfield Campus, Baystate Medical Center, Springfield, MA 01199, United States; Mohammad Al-Haddad, MD, Assistant Professor of Clinical Medicine, Director, Endoscopic Ultrasound Fellowship Program, Indiana University School of Medicine, 550 N. University Blvd, Suite 4100, Indianapolis, IN 46202, United States
S- Editor Zhang HN L- Editor Roemmele A E- Editor Liu N
| 1. | Lim JH. Cholangiocarcinoma: morphologic classification according to growth pattern and imaging findings. AJR. 2003;181:819-827. |
| 2. | Mosconi S, Beretta GD, Labianca R, Zampino MG, Gatta G, Heinemann V. Cholangiocarcinoma. Crit Rev Oncol Hematol. 2009;69:259-270. |
| 3. | Nguyen K, Sing JT Jr. Review of endoscopic techniques in the diagnosis and management of cholangiocarcinoma. World J Gastroenterol. 2008;14:2995-2999. |
| 4. | Harewood GC. Endoscopic tissue diagnosis of cholangiocarcinoma. Curr Opin Gastroenterol. 2008;24:627-630. |
| 5. | Rauws EA, Kloek JJ, Gouma DJ, Van Gulik TM. Staging of cholangiocarcinoma: the role of endoscopy. HPB (Oxford). 2008;10:110-112. |
| 6. | Meara RS, Jhala D, Eloubeidi MA, Eltoum I, Chhieng DC, Crowe DR, Varadarajulu S, Jhala N. Endoscopic ultrasound-guided FNA biopsy of bile duct and gallbladder: analysis of 53 cases. Cytopathology. 2006;17:42-49. |
| 7. | Eloubeidi MA, Chen VK, Jhala NC, Eltoum IE, Jhala D, Chhieng DC, Syed SA, Vickers SM, Mel Wilcox C. Endoscopic ultrasound-guided fine needle aspiration biopsy of suspected cholangiocarcinoma. Clin Gastroenterol Hepatol. 2004;2:209-213. |
| 8. | Fritscher-Ravens A, Broering DC, Sriram PV, Topalidis T, Jaeckle S, Thonke F, Soehendra N. EUS-guided fine-needle aspiration cytodiagnosis of hilar cholangiocarcinoma: a case series. Gastrointest Endosc. 2000;52:534-540. |
| 9. | Gleeson FC, Rajan E, Levy MJ, Clain JE, Topazian MD, Harewood GC, Papachristou GI, Takahashi N, Rosen CB, Gores GJ. EUS-guided FNA of regional lymph nodes in patients with unresectable hilar cholangiocarcinoma. Gastrointest Endosc. 2008;67:438-443. |
| 10. | Hahn M, Faigel DO. Frequency of mediastinal lymph node metastases in patients undergoing EUS evaluation of pancreaticobiliary masses. Gastrointest Endosc. 2001;54:331-335. |
| 11. | Singh P, Erickson RA, Mukhopadhyay P, Gopal S, Kiss A, Khan A, Ulf Westblom T. EUS for detection of the hepatocellular carcinoma: results of a prospective study. Gastrointest Endosc. 2007;66:265-273. |
| 12. | Kaushik N, Khalid A, Brody D, McGrath K. EUS-guided paracentesis for the diagnosis of malignant ascites. Gastrointest Endosc. 2006;64:908-913. |
| 13. | Souza FF, Mortelé KJ, Cibas ES, Erturk SM, Silverman SG. Predictive value of percutaneous imaging-guided biopsy of peritoneal and omental masses: results in 111 patients. AJR. 2009;192:131-136. |
| 14. | Maggs J, Cullen S. Management of autoimmune liver disease. Minerva Gastroenterol Dietol. 2009;55:173-206. |
| 15. | Saich R, Chapman R. Primary sclerosing cholangitis, autoimmune hepatitis and overlap syndromes in inflammatory bowel disease. World J Gastroenterol. 2008;14:331-337. |
| 16. | Elfaki DH, Gossard AA, Lindor KD. Cholangiocarcinoma: expanding the spectrum of risk factors. J Gastrointest Cancer. 2008;39:114-117. |
| 17. | Krawitt EL. Clinical features and management of autoimmune hepatitis. World J Gastroenterol. 2008;14:3301-3305. |