Published online Oct 16, 2022. doi: 10.4253/wjge.v14.i10.616
Peer-review started: June 6, 2022
First decision: August 1, 2022
Revised: August 5, 2022
Accepted: October 4, 2022
Article in press: October 4, 2022
Published online: October 16, 2022
Processing time: 127 Days and 8.6 Hours
Nutrition is one of the fundamental needs of both patient and non-patient po
To describe cases and technique of laparoscopic Janeway gastrostomy (LJG) and perform a systematic review of the data.
We successfully performed two LJG procedures, after which we conducted a literature review of all documented cases of LJG from 1991 to 2022. We surveyed these cases to show the efficacy of LJG and provide comparisons to other existing procedures with primary outcomes of operative time, complications, duration of gastrostomy use, and application settings. The data were then extracted and assessed on the basis of the Reference Citation Analysis (https://www.referencecitationanalysis.com/).
We presented two cases of LJG, detailing the simplicity and benefits of this tec
The LJG is a simple, reproducible procedure with a favorable complication profile. By its technical ease and benefits relating to the gastric tube formed, we propose this procedure as a viable, fa
Core Tip: This systematic review identifies that the laparoscopic Janeway gastrostomy may be advantageous as a first line option for enteral access in specific patient populations, when compared to percutaneous endoscopic gastrostomy, or other surgical gastrostomy options, by virtue of the gastric tube created and its resistance to dislodgment and ensuing complications. Patients with high risk for tube dislodgment, including those with neurocognitive disorders, seizures, dementia, or patients requiring permanent enteral feeding access, may benefit the most from this intervention as a first-line option.
- Citation: Murray-Ramcharan M, Fonseca Mora MC, Gattorno F, Andrade J. Laparoscopic Janeway gastrostomy as preferred enteral access in specific patient populations: A systematic review and case series. World J Gastrointest Endosc 2022; 14(10): 616-627
- URL: https://www.wjgnet.com/1948-5190/full/v14/i10/616.htm
- DOI: https://dx.doi.org/10.4253/wjge.v14.i10.616
Nutrition is one of the fundamental needs of the hospitalized patient, with feeding access providing many unique challenges within different patient subgroups. From stable patients to those requiring intensive care unit treatment, all have specific metabolic demands and requirements necessary for progression towards optimization. Within a hospital setting, there have been extensive studies ex
For long-term feeding accesses, the percutaneous endoscopic gastrostomy (PEG) or percutaneous radiographic endoscopy (PRG)[6,7] remain the first line and preferred procedure. First described in the literature in 1980, the PEG has become widely popularized due to simplicity of performance, ability to perform as a bedside procedure, cost-effectiveness, and low complications profile by non-surgical approach[8]. What historically was the only viable option for feeding access, now the second line in the event of failure or infeasibility of PEG, exists the surgical gastrostomies (and jejunostomies). The Stamm gastrostomy, introduced in 1894[9]; is achieved via an incision made in the anterior stomach wall with a purse-string suture securing a tube brought out through the anterior abdominal wall. Performed open or laparoscopically, this technique is simple to perform with low morbidity and revision rates[10]. The Witzel gastrostomy, initially described in 1891, is performed with a tube or catheter (exiting the anterior abdominal wall) introduced into a gastrostomy on the anterior stomach, with parallel folds fashioned into a tunnel around the tube. This procedure had limited response as a gastrostomy, and multiple variations have led it to be performed instead as a jejunostomy creation technique. As a result, this is a rarely performed gastrostomy procedure with minimal literature documenting its utility as such[9]. The Janeway gastrostomy, the focus of this paper, was introduced into practice in 1913, with the unique creation of a gastric tube from the anterior stomach wall exteriorized as a stoma boasts the advantage of permanence and resilience in the setting of tube dislodgement in comparison with other techniques[9]. Initially used for feeding in cases of advanced head and neck tumors[11], following several modifications, this technique is commonly performed laparoscopically for a variety of indications. This literature review explores the versatility of the laparoscopic Janeway gastrostomy (LJG) for patients requiring long-term or permanent enteral feeding access with the aid of two presented cases.
We retrieved the records of the patients who underwent LJG creation on (n = 2) in Woodhull Hospital Center of New York Health and Hospitals (Brooklyn, New York) from 2021 to 2022. Two patients were identified and their respective clinical courses relevant to their procedure were documented, making note of technical details, ensuing postoperative courses and complications.
A comprehensive search of the literature was conducted through MEDLINE/PubMed, Cochrane Central Register of Controlled Trials, and Cochrane Database of Systematic Reviews to identify relevant articles. Before initiation of the search authors determined titles, keywords, and text words of im
Relevant studies were identified and selected by individual reviewers separately based on title and abstract content. Supporting evidence included randomized and non-randomized controlled trials, systematic reviews, prospective and retrospective studies, case series, reviews, and letters to editors. Analysis and evaluation of Spanish articles were performed independently by native Spanish-speaking physicians.
The articles included in this selection were English or Spanish articles published between 1984 and 2022. We included patients of all ages and articles of all types. Exclusion criteria consisted only of articles written in other languages such as French or German, to prevent inaccurate translation. This search was performed and reviewed for inclusion in the review by authors MMR and MCF independently on 22nd February 2022.
The methodological quality of the studies was assessed using the 2010 American Association of Clinical Endocrinologists Protocol for Production of Clinical Practices Guidelines: Evidence Rating (Table 1). Data quality and recommendations for clinical application were categorized based on the evidence level.
| Numerical descriptor (evidence level) | Semantic descriptor (reference methodology) |
| 1 | Meta-analysis of randomized controlled trials |
| 1 | Randomized controlled trial |
| 2 | Meta-analysis of nonrandomized prospective or case-controlled trials |
| 2 | Nonrandomized controlled trial |
| 2 | Prospective cohort study |
| 2 | Retrospective case-control study/Retrospective cohort study |
| 3 | Cross-sectional study |
| 3 | Surveillance study (registries, surveys, epidemiologic study) |
| 3 | Consecutive case series |
| 3 | Single case reports |
| 4 | No evidence (theory, opinion, consensus, or review) |
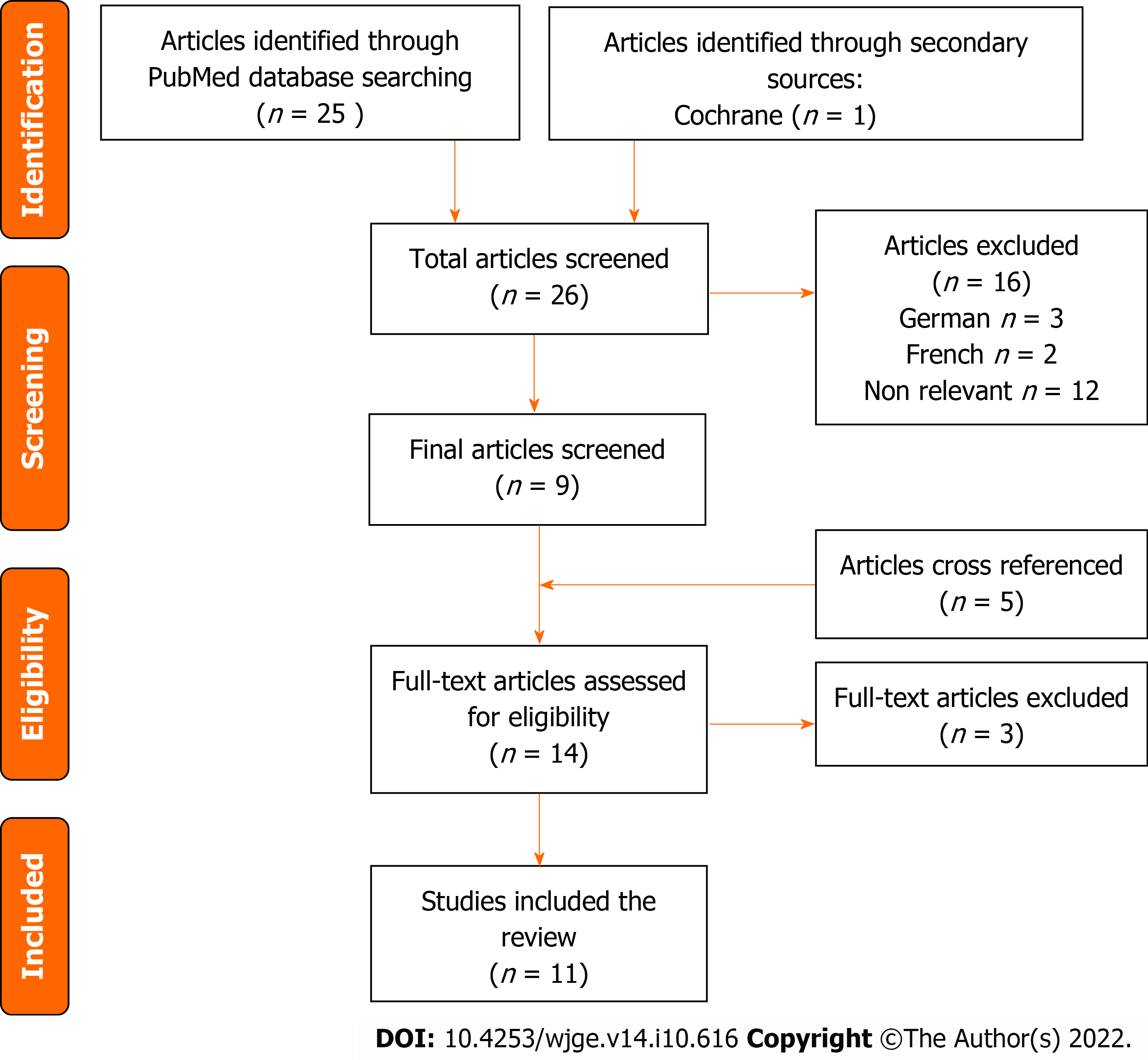
An initial assessment of articles’ abstracts and titles was performed with a total preliminary outcome of 26 articles. After this initial screening, the 26 articles were evaluated in more detail with proper screening against inclusion and exclusion criteria. 15 articles were excluded; of those three had content in German and two in the French language, the remaining twelve referred to content that was not pertinent to the outcomes being evaluated in this review, by either discussing animal trials or JG for additional procedures (trans-gastric endoscopic retrograde cholangiopancreatography in complicated anatomy) rather than enteral access. An addition of five references was found and of those, three were included after cross-referencing articles. After a thorough selection of articles using the PRISMA criteria (Figure 1) a total of 11 articles resulted in the following breakdown: Five case series, one case report, two short communications articles with associated case reports, one technical innovation article with associated case series, one comprehensive review article, and one original article.
From the analyzed studies on LJG (Table 2), of the total 56 patients with LJG 43 patients had doc
| Ref. | Evidence rating | Case | Outcomes | Complications |
| Haggie et al[18], 1992 | 3 | n = 1 pt; Age= 65 yr (M); Esophageal occlusion of pharyngeal SCC s/p CTX and RTX | ORT: N/M; MUTs: 3 wk (death 2/2 primary disease) | Leakage of gastric contents easily managed; D: 1; R: 1; TC: 2 |
| Serrano et al[13], 1994 | 3 | n = 7 pt; Age = 48-83 yr; Esophageal cancer stage IV: 85% (n = 6); Traumatic peri-esophageal hematoma: 14.2% (n = 1) | ORT: 30-40 min. Average 35 min. MUTs: N/M | TC: 0; D: 0; R: 0; Mortality: 0 |
| Ritz et al[12], 1998 | 3 | n = 15 pt; Age average: 61 yr; Esophageal or paraesophageal tumors | ORT: 20-55 min. MUTs: 3.5 mo (death) | Stoma necrosis to Witzel gastrostoma: 6.6% (n = 1); Self-limiting skin irritation: 20% (n = 3); D: 0; R: 0; TC: 2 |
| Molloy M et al[17], 1997 | 3 | n = 2 pt (M); Age= 63 yr and 77 yr; Organic neurologic disorders + pulled out PEG (placed 48 h prior); Perforation along greater curvature (minimal contamination) | ORT: N/M. MUTs: N/M | C: N/M; D: N/M; R: N/M |
| Raakow et al[14], 2001 | 2 | n = 21 pt (19 M; 2 F); Age = 53-78 yr; Extensive tumors of: Hypopharynx 57.1% (n = 12) Esophagus 42.8% (n = 9); Prior UGI surgery 19% (n = 4) to (2 OCh, 1 PCJ, 1 repair DP) | ORT: 24-50 min. Average 38 mins. MUT: 3.4 mo 2/2 death due to primary | C: Self-limiting skin irritation (method dependent): 9.6% (n = 2); D: N/M; R: N/M; Mortality from advanced cancer; MUTs: 26 d to 6.5 mo (average 3.4 mo) |
| Tous Romero et al[19], 2012 | 2 | n = 57 pt; Age = 51 yr; 10 LJG, 47 OJG; Esophageal cancer: 38.6% (n = 22); Head & neck: 26.3% (n = 15); Neuro deficit 26.3% (n = 15) | ORT: N/M. MUTs: N/M | TC: 5 (some patients had multiple complications); D: N/M; R: N/M; Gastric content leakage: 30% (n = 3); Abd wall irritation: 30% (n = 3); No C: 50% (n = 5); Exudate: 10% ( n = 1); Exudate with + culture: 20% ( n = 2); Granuloma: 10%( n = 1); Balloon rupture: 10% (n = 1); Loss of peristomal content: 0 |
| Ref. | Evidence rating | Case | Outcomes | Complications |
| McGovern et al[21], 1984 | 3 | n = 14 children (> 7lb); Severe cerebral palsy without pharyngeal musculature coordination and risk of aspiration | ORT: N/M, MUTs: N/M | C: GT stenosis treated with dilation: 7.14% (n = 1); Stomal granulations treated with cautery: 7.14% (n = 1); Mortality: 0; D: N/M; R: N/M |
| Laughlin et al[20], 1989 | 3 | n = 5 pt. Advanced esophageal cancer; Age/gender: N/M | ORT/MUTs: N/M | C: Stomal tip necrosis with stomal stenosis: 20% (n = 1); Mortality: 0; D: N/M; R: N/M |
| Vassilopoulos et al[11], 1998 | 3 | n = 24 pt (21M; 3F); Age average: 67.19 yr; Advanced head/neck cancer; Advanced UGI malignancy: 1.2% (n = 5); Prior UGI surgery: 0.48% (n = 2) | ORT: < 40 min; MUTs: N/M | C: Midline wound SSI treated with antibiotics: 16.6% (n = 4); Mortality: 0; D: N/M; R: N/M |
| Koivusalo et al[15], 2006 | 33 | n = 4 pt; Age = 0-6 yr; Recurrent gastrostomy prolapses and peristomal infection undergoing modified OJG revision; 3: OSG to 2 closure + PEG; 1: Initial PEG; Prior abdominal surgeries (OGT/PEG) | MUTs: 9 mo | C: 0;D: N/M; R: N/M content |
| Abdel-Lah et al[16], 2006 | 3 | Total procedure 287: JT: 46% (n = 167); SG: 18% (n = 40); OJG: 4% (n = 8); SNY double lumen: 32% (n = 72); Head & neck cancer; Total permanent gastrostomies n = 27: Balloon catheter/Fontan (LE < 37 d): n = 19; OJG (LE > 6 mo): n = 8 | MUTs; JG = 164 d | Morbidity 12.5% (n = 5): D (Migration)/peristomal abrasion- no fixation to parietal peritoneum; Mortality (open jejunostomy) 4.2% (n = 12); Esophageal 3% ( n = 9); Esophagojejunal: 1.2% (n = 3); R: N/M |
| Tous Romero et al[19], 2012 | 2 | n = 57 pt; Age average: 57, 51 yr 10 LJG, 47 OJG; Esophageal cancer: 38.6% (n = 22); Head & neck: 26.3% n = 15); Neuro deficit: 26.3% (n = 15) | ORT/MUTs: N/M | Gastric content leakage: 89.4% (n = 42); Abd wall irritation: 83% (n = 39); No C: 2.1% (n = 1); Exudate: 23.4% (n = 11); Granuloma: 4.3% (n = 4); Balloon rupture: 21.3% (n = 10); Loss of peristomal content: 17% (n = 8) |
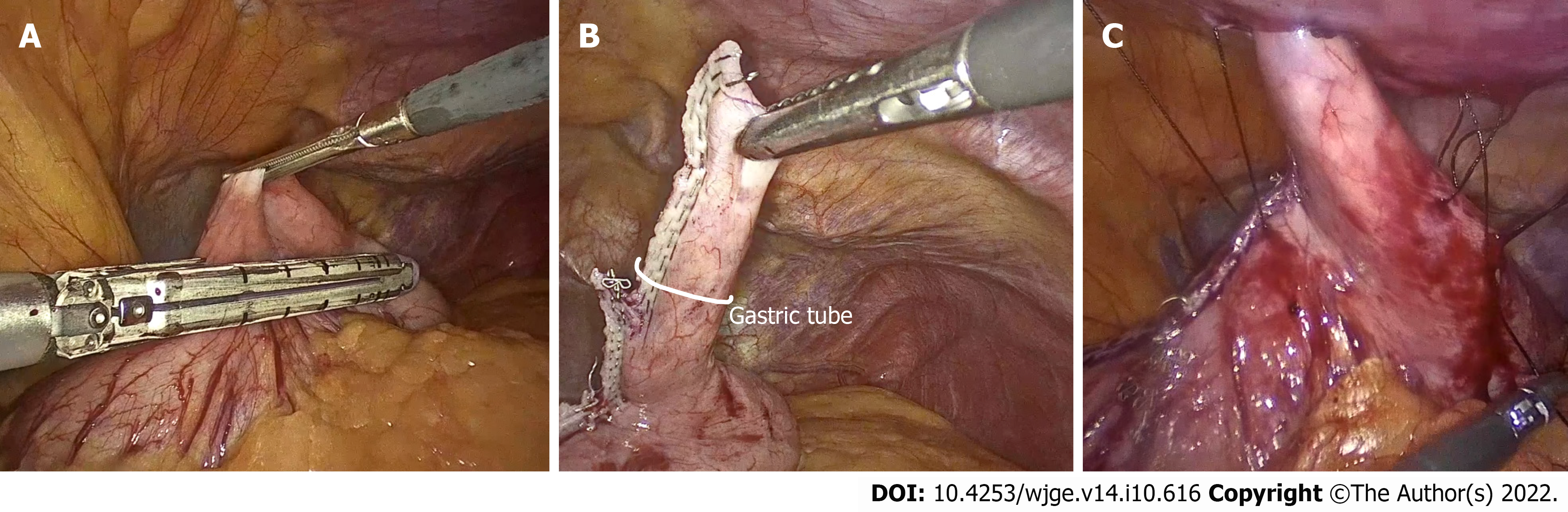
There exist several modifications of the original JG, with further modifications introduced with the inception of laparoscopy into commonplace surgical practice[14]. We describe the laparoscopic tec
Patient A: This is a 77-year-old woman with a past medical history of dementia, hypertension, and depression who was being managed in the hospital for altered mental status and mental decline following infection with coronavirus disease 2019 (COVID-19) a few months prior (Table 4). During the hospital stay, the patient experienced a further decline from baseline, with worsening dementia and refusal of oral intake and malnutrition. The primary team requested enteral feeding access, and with the agreement of the patient’s healthcare proxy, we advocated for LJG tube placement. We suggested this procedure due to the patient’s dementia, need for permanent/long-term feeding, and a high risk of the patient pulling out tubes. The procedure was performed by the technique described above, and the patient was followed postoperatively. There were no noted complications, and the gastrostomy tube was used for feeding immediately postoperatively without any complications noted and was discharged safely the following day. The gastrostomy tube remained intact with no complications until the patient passed away as a result of complications of primary disease while in hospice care 3 mo later.
| Case | Selection of LJG vs others | Indications | Outcomes | Complications |
| Patient A: 77 yr female | Instead of PEG; Patient is high risk of pulling out tubes | Worsening dementia and AMS. Need for long term/permanent feeding | ORT: 87 min. MUTs: 3 mo | D: 0; R: 0; TC: 0 |
| Patient B: 58 yr male; s/p tracheostomy and recent PEG tube placement | Instead of PEG. C: Dislodgement of PEG and septic shock | Cerebral palsy, seizure disorder self-removed PEG. Prior PEG removal + replacement | ORT: 76 min. MUTs: 3 mo | D: 0; R: 0; TC: 0 |
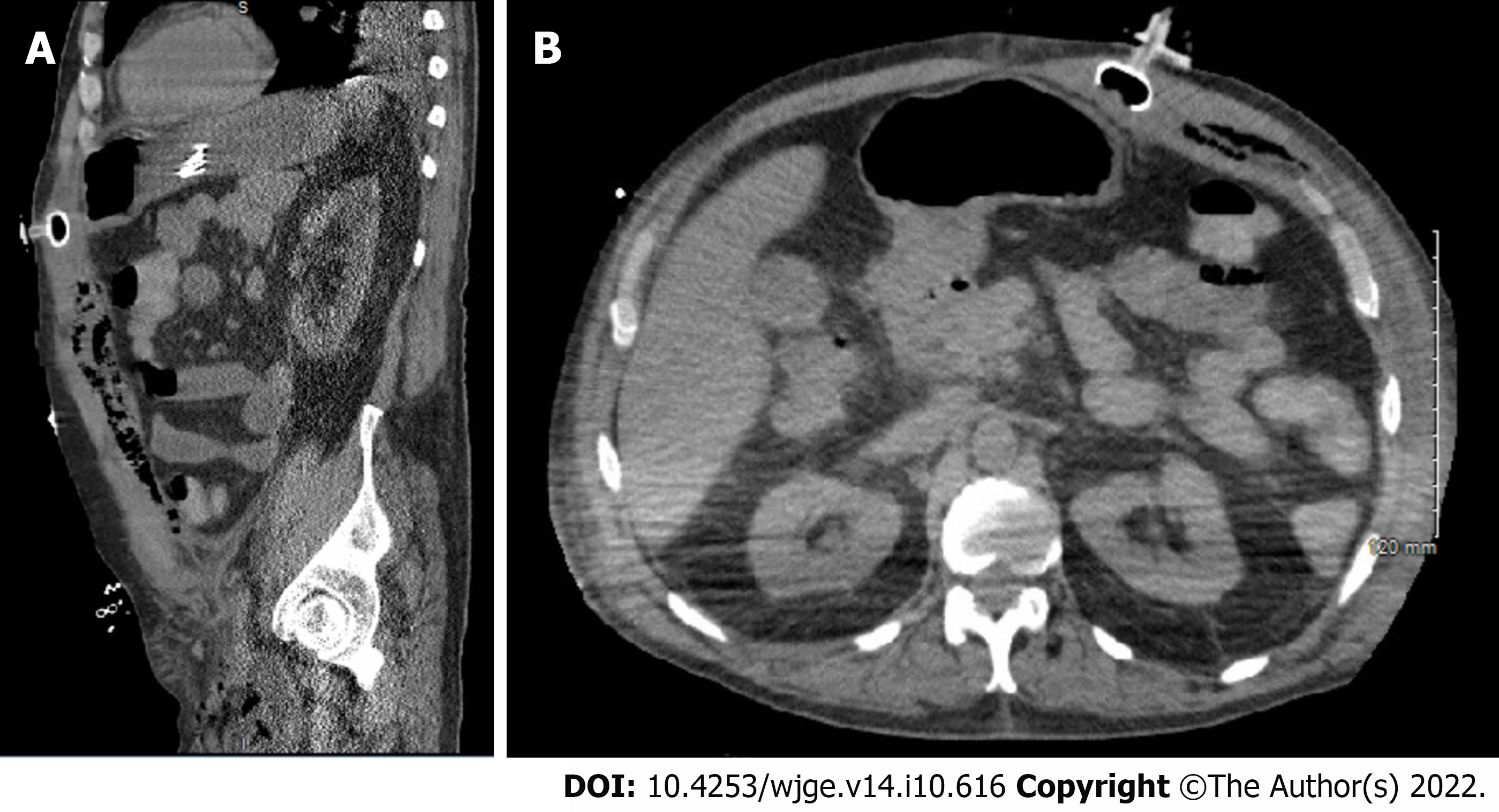
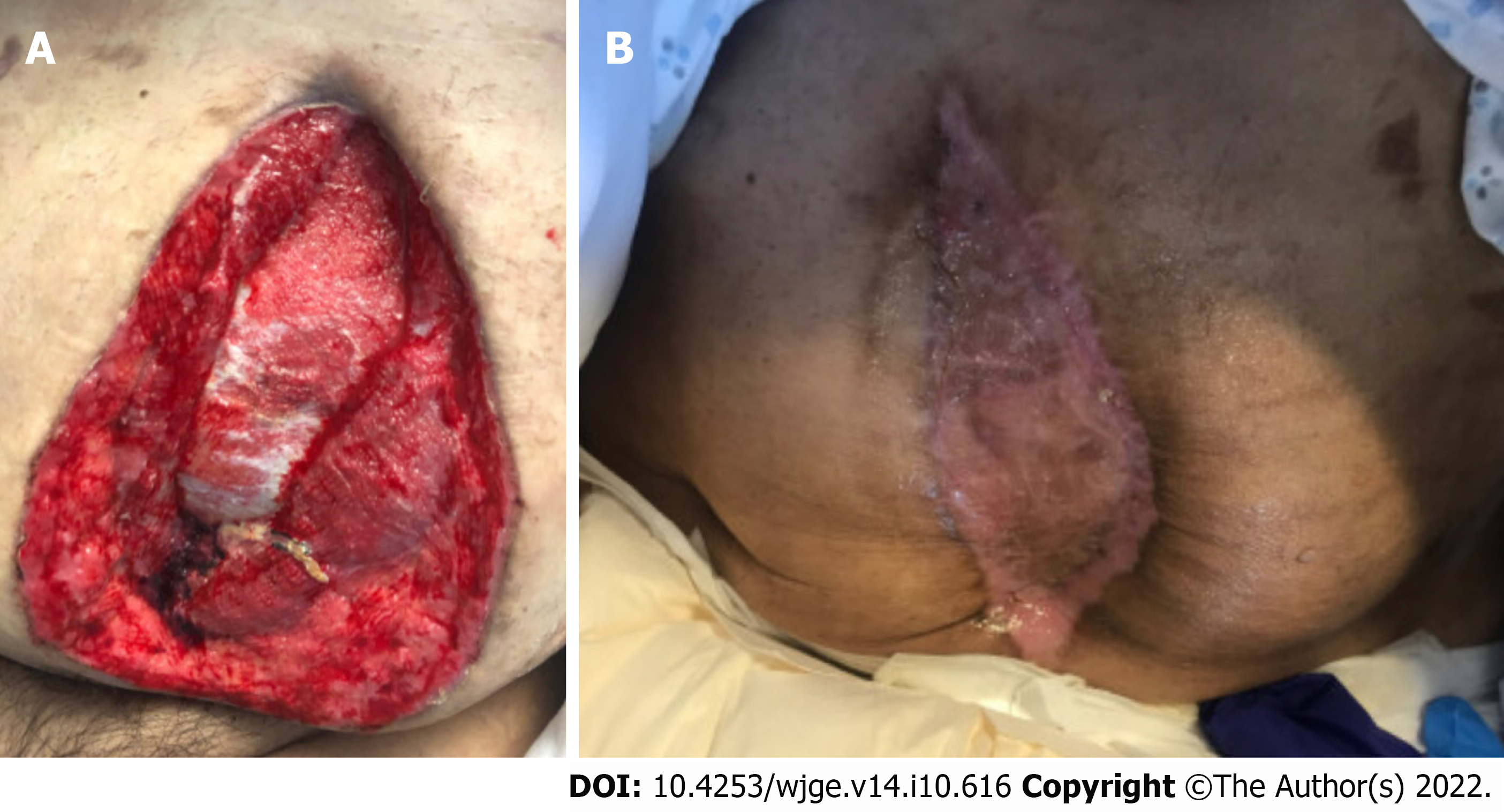
Patient B: This is a 58-year-old man who resides in a nursing home, with a past medical history of cerebral palsy, seizure disorder, diabetes, hypertension, and a past surgical history of tracheostomy and recent PEG tube placement after distant COVID-19 pneumonia (Table 4). After the PEG was placed, the patient was discharged back to his nursing home once his pneumonia resolved, during which time he removed his PEG tube in instances of agitation multiple times, each with subsequent replacement. Several months after initial placement, the patient was brought to the emergency department in septic shock with a tender and distended abdomen. Due to his neurologic conditions, he was unable to provide any history, and he underwent a computed tomography scan which revealed that the balloon of his gastrostomy feeding tube was embedded in the anterior abdominal wall, and there was significant subcutaneous air and fluid along the rectus sheath adjacent to the gastrostomy tube along with a fragment of the apparatus within the stomach. (Figure 3A and B). He underwent an emergent surgery where tube feeds and purulent fluid were found within the soft tissue above the fascia and the abdominal cavity. He underwent debridement and washout of this fluid, fascia closed and the wound was left to heal by secondary intention. After he recovered from septic shock in the intensive care unit, a skin graft was performed due to poor healing from this procedure (Figure 4A and B). Due to his hostile anatomy after these procedures, his high risk of removal or dislodgement of the tube, and the continued need for permanent feeding access due to his cerebral palsy, we elected to perform LJG. The procedure was by the technique described above, and the patient was followed postoperatively. There were no noted complications, and the gastrostomy tube was used for feeding immediately postoperatively. The gastrostomy tube was removed by the patient twice within the first 3 wk postoperatively (postoperative days 11 and 18), and two more times within the first 2 mo post-procedure (postoperative days 48 and 61) with subsequent replacement without issue. The patient was discharged approximately 2 mo after the procedure after the management of his primary disease, during which time no further complications were noted. A month later, the patient passed away as a result of complications of primary disease while in hospice care.
When comparing the standard of care (PEG) to LJG, we can see advantages concerning the fistula tract. In a PEG, there is rapid obliteration of the fistula if the tube becomes dislodged, which allows for only a small window in which replacement of the tube may be possible. In these settings, repeat instrumentation or another procedure for enteral access may be required[17], in addition to possible complications of the gastric leak[18]. The LJG does not share this complication, due to the mucous layer surrounding the gastrostomy tube, as well as the maturation of the gastric tube to the skin. A feeding tube can be safely replaced without concern, or in certain circumstances may be removed and replaced freely and intermittently when feeding is needed. Additionally, this type of gastrostomy is performed via an objectively easy and reproducible procedure with few steps. We draw reference to the described cases above, both performed almost entirely by surgical residents and in an identical fashion. Even in the case of patient A, with prior intra-abdominal surgery as well as abdominal wall surgery, the procedure was performed with no significant adjustments. Several modifications to the original technique exist; in our cases we utilized trans-fascial anchoring sutures to the base of the gastric tube. This serves to relieve any tension on the gastric tube, increasing the surface area of anterior abdominal wall adherence. Another modification is the use of a port site as the site of the gastrostomy, limiting additional incisions. In earlier techniques of LJG, the gastric tube was created with the base of the gastric tube near the lesser curvature, in contrast to the modification used in the presented case where the base was at the greater curvature (Figure 3). This simple but strategic modification described in our cases allows for preservation of the blood supply of the gastric tube by the gastro-epiploic vessels, as well as allows for more desirable positioning of the gastrostomy lateral to the midline with an exit through the rectus muscles. The fixation of the exteriorized gastrostomy to the skin, akin to the maturation of an ostomy, is not performed in surgical gastrostomies. This creates a definitive track that leads to the permanence and longevity of the LJG. The gastrostomy creation not only spares the need for a constant indwelling catheter but also provides continence as it exits through the rectus abdominis[12], with a sphincteric mechanism via the rectus muscles preventing reflux or incontinence[14]. This configuration may be advantageous in the population of patients with disorders such as seizures or cerebral palsy. Compared to PEG which lacks an anti-reflux mechanism, the sphincter created during the LJG may be more preventative against complications of convulsive patterns including reflux, leakage from the stoma, and stomal prolapse[15].
This systematic review was performed with a focus on technical ease and reproducibility of procedure, resistance to complications such as tube dislodgement, and evaluating the use of the LJG as a permanent or long-term feeding access option as it compares to the alternatives. In terms of operative times, most of the studies published share a very similar range and mean duration; with an average time of 35.3 min for all the 43 patients with their times documented. We propose three main reasons for the difference between these studies and the 2 case reports of our own (with an average operating time of 81 min). One is likely due to the procedures in our studies being performed almost entirely by residents, with a large focus on education and laparoscopic skill development. The other proposed reason is that in “Patient B”, the procedure was initially delayed by a transient intolerance to pneumoperitoneum, after which, following optimization by anesthesia, we were able to proceed. This delay was factoring into the total operative time which is a series of only 2 patients may lead to a greatly extended average operating time. The third proposed reason for time discrepancies relates to the technique used; in our two described cases, we employed the use of intracorporeal anchoring sutures to affix the base of the gastric tube to the anterior abdominal wall - an optional modification to the LJG to provide additional support, not performed in other reports. With regards to use as a long-term option for feeding access, there exists an objective theoretical advantage for LJG. By the creation of a gastric tube and maturation to the skin, a technique unique to the JG/LJG, there cannot be spontaneous closure of the fistula, making this ideal for long-term, palliative, or permanent enteral access. This systematic review looked at the documented MUTs of LJG (Table 2) to establish its role in longevity. This proved difficult, since the LJG by these benefits, was used quite extensively in populations consisting of terminal patients, or patients residing in nursing homes with expectedly poor follow-up.
We acknowledge that the goals of this paper are to demonstrate characteristics of the laparoscopic Janeway specifically, but we believe that with regards to MUT post-procedure, we may be able to utilize data from the subset of OJG analyzed (Table 3), as the result of these procedures is the same regarding gastrostomy use. The average MUT between the LJG and OJG groups is approximately 4 mo, however these results obtained do not reflect the true permanence of this procedure. In the above studies we had no documented cases of reversal of the gastrostomy, and due to the essential nature of the indications for this procedure, we can extrapolate that the LJG likely lasted the intended length of time: the rest of the respective patients’ lives. Of the 56 patients who underwent LJG in the analyzed articles, we note 13 total complications and 0 mortalities related to the procedure; reported mortalities were related to the medical condition itself as seen in our case series. We attempted to stratify these into major and minor complications. The only identified major complication occurred in 1 patient in this series, in the case of Ritz et al[12], which documents a case of stomal necrosis, attributed by the authors to the creation of a gastric tube that was too small. This case necessitated surgical revision and conversion to a Witzel gastrostomy, with the remainder of the post-operative course unremarkable. With regards to the minor complications, we note 8 total cases of skin irritation[12,14,19] all of which were self-limiting. Tous Romero et al[19] documented one case in which a stoma granuloma formed, and this did not affect the functioning of the gastrostomy nor the quality of patient life, demonstrating the preferable complication profile for the LJG.
A significant complication of most gastrostomy procedures is tube dislodgement. This highlights possibly the most desirable feature of the LJG, that tube dislodgement at any time post-operatively does not cause any complication and poses no significant risks to the patient. This benefit is not only theoretical; we see it in clinical practice. In Raakow et al[14], the authors had the gastrostomy tubes removed from the gastrostomy intermittently, beginning on postoperative days 10-14 without any complications related to removal or reinsertion. We saw this in our case of “Patient B” in the presented clinical case, where the patient himself removed the gastrostomy tube on postoperative days 11, 18, 48, and 61, with no concerning sequelae following bedside replacement. There may have been a need for reoperation, especially with the first two removals, had the procedure been any other gastrostomy than an LJG. Comparing the complication profile of the LJG to that of a PEG, Ritz et al[12] demonstrate that PEG has a complication rate up to 30% (minor) and 9% (major) with a 1%-2% mortality. This is further corroborated by Rahnemai-Azar et al[6] in a comprehensive literature review, which identifies 8 minor and 6 major complications associated with PEG. The dislodgement of the PEG tube is seen to occur in approximately 12.8% of patients, with management strategies including replacement or new PEG or surgical gastrostomy creation. Other major complications of PEG described that may be mitigated by the use of LJG include buried bumper syndrome, not using the classic PEG tube, and hollow viscus inadvertent injury, as direct visualization is possible[6].
Comparing LJG to other surgical gastrostomies, data from the existing literature advocates a more benign complication profile as compared to the other surgical alternatives. Ritz et al[12] compared complications of open Witzel, Stamm, Kader, and Janeway gastrostomy. The OJG had a complication rate of 0%-25%, with a mortality rate of 0%-11%, favorable to that of the other open surgical alternatives with a collective complication rate of 13%-42% and a mortality rate of 10%-23%. These rates in OJG were then compared to those of LJG, with LJG having a 0%-6% complication rate and 0% mortality[20,21]. For completion, laparoscopic Kader gastrostomy was also compared to the rates for LJG, with complication rates of 6%-9% and mortality rate of 0%-5%, illustrating the preferable results of the LJG. Raakow et al[14] further supplemented these results by noting that when the Janeway technique is applied, the risks of developing postoperative leakage are notably decreased (approximate 0%-1%) when compared to approximately 9% as seen in the other surgical gastrostomies[14]. Abdel-Lah et al[16] in a more recent study, compared the LJG directly to the OJG. However, no statistical differences were noted given the variety of the population and the lack of specific primary outcomes. This highlights the need for more studies to investigate these differences.
We identify several limitations in this literature review. Firstly, the majority of the studies analyzed had a relatively low sample size, with a total of 158 patients analyzed (56 patients with LJG and 102 with OJG). Another limitation is that there are no randomized controlled trials available in the literature that compares LJG to other gastrostomy creation techniques. This is the gold standard for inferring causation from correlation, and without this type of study we acknowledge less strength of the presented literature. In addition, there is limited research on the use of LJG, as evidenced by the small number of articles retrieved with broad search terms. Another limitation of this review is that many of the indications for LJG described in the literature are for palliative purposes with a large cohort of patients having advanced-stage cancers. This confounds the investigated MUT of the gastrostomy tube, which may have been longer had the patients not had poor prognoses. This limits the ability of this study for long-term analysis. Lastly, we noted that scarce recent data has been published on JGs, as evidenced by including articles published over 20 - 40 years ago. A proposed reason for this chronology is that surgical gastrostomies have been seldom performed in recent years due to the popularity of the PEG and indicates strong potential for future studies where recent data is lacking.
The LJG is a viable technique for the creation of permanent or long-term enteral access, by its simple, reproducible technique and desirable complication profile, especially with for tube removal or dislodgement. As seen in many of the cases reviewed, this can be performed by advanced laparoscopists, surgical residents, and general surgeons without formal laparoscopic fellowship training. We acknowledge the data supporting PEG as a first-line feeding option, and advocate that the LJG should be strongly considered as a first-line option in specific patient populations, those who require permanent enteral access who may be at risk of tube dislodgement or removal due to agitation or neurologic disease. Another role for LJG as a first-line option may be in the setting where PEG is infeasible, for example, in cases of advanced head and neck cancer, severe abdominal wall scarring, and inability to get transillumination, as seen in the cases reviewed. LJG also has a beneficial potential role as a second-line option should a PEG be unable to be performed or unsuccessful, for any sign of long-term feeding access. This literature review, besides describing the many advantages of this procedure, has made us aware of the need for further study and randomized controlled trials of this promising technique.
LJG, when initially described, was used as one of the first-line enteral access options, and has since been replaced by the advent and popularity of PEG. The significance of this study is that it demonstrates that the laparoscopic modification may be an acceptable first-line procedure for specific indications due to its longevity and ease of completion.
The main topics of this paper are that LJG may have more clinical relevance than previously considered. The problems this paper addresses is the complication rate including those caused due to dislodgement and tube removal with the PEG procedure. This procedure ameliorates these complications and may have a role in first-line access for specific indications.
The main objectives of this project was to describe cases of LJG as well as perform a systematic review of the available data as it relates to LJG for enteral access. We realized from this review, that LJG may serve as a viable alternative to PEG as a first-line option for enteral access in specific populations. The significance of this realization can result in lower morbidity and mortality as it relates to the complications of PEG dislodgements in specific patient populations.
A systematic review was performed of all available data of LJG relating to use for enteral access. This data was analyzed by the reviewers to realize the objectives. To our knowledge, no large systematic reviews of LJG have been recently performed for this purpose.
Our findings describe relatively low rate of complications from LJG, largely as a result of the permanent gastrostomy tube formed in the procedure. We also note significant technical ease in completion of the procedure.
This study proposes that LJG may be a viable alternative to PEG as a first-line procedure in specific patient populations.
This study describes the laparoscopic modification of Janeway gastrostomy and notes the technical ease and reproducibility.
The direction for future research in this topic may include prospective studies and randomized controlled trials to determine true comparative data between LJG and PEG and other gastrostomy alternatives, and also to provide objective data to guide optimal patient selection.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Surgery
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Anandan H, India; Shalaby MN, Egypt S-Editor: Xing YX L-Editor: A P-Editor: Xing YX
| 1. | Shi J, Wei L, Huang R, Liao L. Effect of combined parenteral and enteral nutrition versus enteral nutrition alone for critically ill patients: A systematic review and meta-analysis. Medicine (Baltimore). 2018;97:e11874. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Al-Dorzi HM, Arabi YM. Nutrition support for critically ill patients. JPEN J Parenter Enteral Nutr. 2021;45:47-59. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 17] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 3. | Pancorbo-Hidalgo PL, García-Fernandez FP, Ramírez-Pérez C. Complications associated with enteral nutrition by nasogastric tube in an internal medicine unit. J Clin Nurs. 2001;10:482-490. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 106] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 4. | Strong RM, Condon SC, Solinger MR, Namihas BN, Ito-Wong LA, Leuty JE. Equal aspiration rates from postpylorus and intragastric-placed small-bore nasoenteric feeding tubes: a randomized, prospective study. JPEN J Parenter Enteral Nutr. 1992;16:59-63. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 165] [Cited by in RCA: 143] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 5. | Pars H, Çavuşoğlu H. A Literature Review of Percutaneous Endoscopic Gastrostomy: Dealing With Complications. Gastroenterol Nurs. 2019;42:351-359. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 6. | Rahnemai-Azar AA, Rahnemaiazar AA, Naghshizadian R, Kurtz A, Farkas DT. Percutaneous endoscopic gastrostomy: indications, technique, complications and management. World J Gastroenterol. 2014;20:7739-7751. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 416] [Cited by in RCA: 340] [Article Influence: 30.9] [Reference Citation Analysis (8)] |
| 7. | Strijbos D, Keszthelyi D, Gilissen LPL, Lacko M, Hoeijmakers JGJ, van der Leij C, de Ridder RJJ, de Haan MW, Masclee AAM. Percutaneous endoscopic versus radiologic gastrostomy for enteral feeding: a retrospective analysis on outcomes and complications. Endosc Int Open. 2019;7:E1487-E1495. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 8. | Gauderer MW, Ponsky JL, Izant RJ Jr. Gastrostomy without laparotomy: a percutaneous endoscopic technique. J Pediatr Surg. 1980;15:872-875. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1408] [Cited by in RCA: 1299] [Article Influence: 28.9] [Reference Citation Analysis (1)] |
| 9. | Minard G. The history of surgically placed feeding tubes. Nutr Clin Pract. 2006;21:626-633. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 10. | Mizrahi I, Garg M, Divino CM, Nguyen S. Comparison of laparoscopic versus open approach to gastrostomy tubes. JSLS. 2014;18:28-33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Vassilopoulos PP, Filopoulos E, Kelessis N, Gontikakis M, Plataniotis G. Competent gastrostomy for patients with head and neck cancer. Support Care Cancer. 1998;6:479-481. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 12. | Ritz JP, Germer CT, Buhr HJ. Laparoscopic gastrostomy according to Janeway. Surg Endosc. 1998;12(6):894-897 [PMID: 9602016 DOI: 10. 1007/s004649900739. |
| 13. | Serrano L, García I, Dominguez J, Sánchez JM, Charlo T. [Laparoscopic gastrostomy]. Rev Esp Enferm Dig. 1994;86:577-580. [PubMed] |
| 14. | Raakow R, Hintze R, Schmidt S, Adler A, Neuhaus P. The laparoscopic Janeway gastrostomy. An alternative technique when percutaneous endoscopic gastrostomy is impractical. Endoscopy. 2001;33:610-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 8] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 15. | Koivusalo A, Pakarinen MP, Pyörälä S, Salminen P, Rintala RJ. Revision of prolapsed feeding gastrostomy with a modified Janeway 'gastric tube'. Pediatr Surg Int. 2006;22:202-204. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 16. | Abdel-Lah Mohamed A, Abdel-Lah Fernández O, Sánchez Fernández J, Pina Arroyo J, Gómez Alonso A. [Surgical access routes in enteral nutrition]. Cir Esp. 2006;79:331-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 17. | Molloy M, Ose KJ, Bower RH. Laparoscopic Janeway gastrostomy: an alternative to celiotomy for the management of a dislodged percutaneous gastrostomy. J Am Coll Surg. 1997;185:187-189. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 11] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 18. | Haggie JA. Laparoscopic tube gastrostomy. Ann R Coll Surg Engl. 1992;74:258-259. [PubMed] |
| 19. | Tous Romero MC, Alarcón del Agua I, Parejo Campos J, Oliva Rodríguez R, Serrano Aguayo P, Hisnard Cadet Dussort JM, Pereira Cunill JL, Morales-Conde S, García-Luna PP. [Comparison of two types of surgical gastrostomies, open and laparoscopic in home enteral nutrition]. Nutr Hosp. 2012;27:1304-1308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 21. | McGovern B. Janeway gastrostomy in children with cerebral palsy. J Pediatr Surg. 1984;19:800-802. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 9] [Article Influence: 0.2] [Reference Citation Analysis (0)] |