Published online Oct 16, 2021. doi: 10.4253/wjge.v13.i10.529
Peer-review started: January 28, 2021
First decision: March 31, 2021
Revised: April 30, 2021
Accepted: August 2, 2021
Article in press: August 2, 2021
Published online: October 16, 2021
Processing time: 258 Days and 11.8 Hours
Duodenal duplications are rare congenital anomalies of the gastrointestinal tract. As the periampullary variant is much rarer, literature is scant and only few authors have reported their experience in diagnosis and treatment, particularly with operative endoscopy.
To report our experience with the endoscopic treatment in a series of children with periampullary duodenal duplication cysts, focusing on the importance of obtaining an accurate preoperative anatomic assessment of the malformations. The pediatric periampullary duodenal duplication cyst literature is reviewed. We conducted a systematic review according to the PRISMA guidelines. The PubMed database was searched for original studies on “duodenal duplication”, “periampullary duplication” or “endoscopic management” published since 1990, involving patients younger than 18 years of age. Eligible study designs were case report, case series and reviews. We analyzed the data and reported the results in table and text. Fifteen eligible articles met the inclusion criteria with 16 patients, and analysis was extended to our additional 4 cases. Median age at diagnosis was 13.5 years. Endoscopic treatment was performed in 10 (50%) patients, with only 2 registered complications.
Periampullary duodenal duplication cysts in pediatric patients are very rare. Our experience suggests that an accurate preoperative assessment is critical. In the presence of sludge or stones inside the duplication, endoscopic retrograde cholangio-pancreatography is mandatory to demonstrate a communication with the biliary tree. Endoscopic treatment resulted in a safe, minimally invasive and effective treatment. In periampullary duodenal duplication cyst endoscopically treated children, long-term follow-up is still necessary considering the potential malignant transformation at the duplication site.
Core Tip: Periampullary duodenal duplications are extremely uncommon in children. The authors report a series of 4 patients and provide a detailed literature review.
- Citation: Bulotta AL, Stern MV, Moneghini D, Parolini F, Bondioni MP, Missale G, Boroni G, Alberti D. Endoscopic treatment of periampullary duodenal duplication cysts in children: Four case reports and review of the literature. World J Gastrointest Endosc 2021; 13(10): 529-542
- URL: https://www.wjgnet.com/1948-5190/full/v13/i10/529.htm
- DOI: https://dx.doi.org/10.4253/wjge.v13.i10.529
Duodenal duplications (DD) are rare congenital anomalies of the gastrointestinal tract, which usually arise during the first decade of life[1-3]. Due to variability of location and size, DD do not display pathognomonic clinical presentation, but they can manifest with a variety of complications including pancreatitis, bleeding, perforation and duodenal obstruction[1]. Unfortunately, little is reported about the anatomical details of DD, which can be divided into two groups: periampullary and non-periampullary duplication cyst. Periampullary duodenal duplication cysts (PADDC) are defined as cysts located near the major papilla and the biliary-pancreatic ampulla, sometimes with a small aberrant pancreatic duct drained into the cyst[4]. As the periampullary variant is much rarer, literature is scant and only few authors have reported their experience in diagnosis and treatment. Moreover, the recent introduction of operative endoscopy for DD treatment in adults has also been extended to the pediatric population with promising results[5-10].
The aim of this paper is to report our experience with the endoscopic treatment (ET) in a series of children with PADDC, focusing on the importance of obtaining an accurate preoperative anatomic assessment of the malformations. The pediatric PADDC literature is reviewed.
All consecutive children with PADDC managed at our tertiary-level institution from 2015 to 2020 were retrospectively reviewed. A written consent was obtained from all patients. All data were retrospectively collected and recorded according to the Declaration of Helsinki.
Case 1, 2 and 4: Abdominal pain.
Case 3: Abdominal pain and vomiting.
Case 1: A 14-year-old boy was admitted with a 1-year history of recurrent pancreatitis. The abdominal computed tomography (CT) scan, previously performed at another center, showed a cyst within the duodenal lumen.
Case 2: A 16-year-old girl was admitted to our emergency room with abdominal pain.
Case 3: A Chinese 11-year-old girl was admitted for 1-year history of epigastric pain with vomiting and weight loss.
Case 4: An 11-year-old girl was admitted to our unit with abdominal pain and vomiting.
Case 1: His previous history was unremarkable.
Case 2: In the past 2 years she had suffered from recurrent abdominal pain due to pancreatitis.
Case 3: The girl was previously examined in her country, and a CT scan showed a cyst in the second part of the duodenum.
Case 4: Unremarkable.
Unremarkable.
Case 1: On inspection, the abdomen was distended with tenderness in epigastrium upon superficial and deep palpation.
Case 2: Physical examination at admission showed a mild distended abdomen and diffuse tenderness upon superficial and deep palpation.
Case 3: Physical examination showed mild diffuse abdominal tenderness upon superficial and deep palpation.
Case 4: Physical examination showed severe tenderness upon superficial and deep palpation of the upper abdomen.
Case 1: Laboratory values revealed an increased serum levels of lipase (1077 UI/L; normal value (n.v.) 70-280 UI/L), amylase 514 UI/L (n.v. 15-53 UI/L) and C-reactive protein 168 mg/dL (n.v. < 5 mg/L), while gamma glutamyl transferase 69 U/L (n.v. 6-42 UI/L), count of blood cells, white cell count, total and conjugated bilirubin, alkaline phosphatase level, aspartate aminotransferase and alanine aminotransferase were normal.
Case 2: Blood samples revealed increased serum levels of lipase (2365 UI/L; n.v. 70-280 UI/L); the full panel of liver tests including cholestasis indexes were normal. US showed the presence of an anechoic cystic lesion within the pancreatic head. Intrahepatic and extrahepatic biliary ducts were normal.
Case 3: Laboratory values revealed increased serum levels of lipase (43440 UI/L; n.v. 70-280 UI/L). The full panel of liver tests was normal.
Case 4: Biochemical investigation revealed hyperlipasemia (5497 UI/L; n.v. 70-280 UI/L) and increased levels of aspartate aminotransferase (5.3 x n.v.), alanine aminotransferase (9.2 x n.v.) and gamma-glutamyl transferase (169 UI/L, n.v. 6-42).
Case 1: The radiological workup first included an abdominal ultrasound (US) that showed a heterogeneous hyperechogenicity of the whole pancreas and an intraluminal duodenal cyst (5.8 cm x 4.5 cm x 4.0 cm in size) near the pancreas head. An 8.5 mm dilatation of the main common bile duct (CBD) was also detected. Intrahepatic biliary ducts and gallbladder were normal.
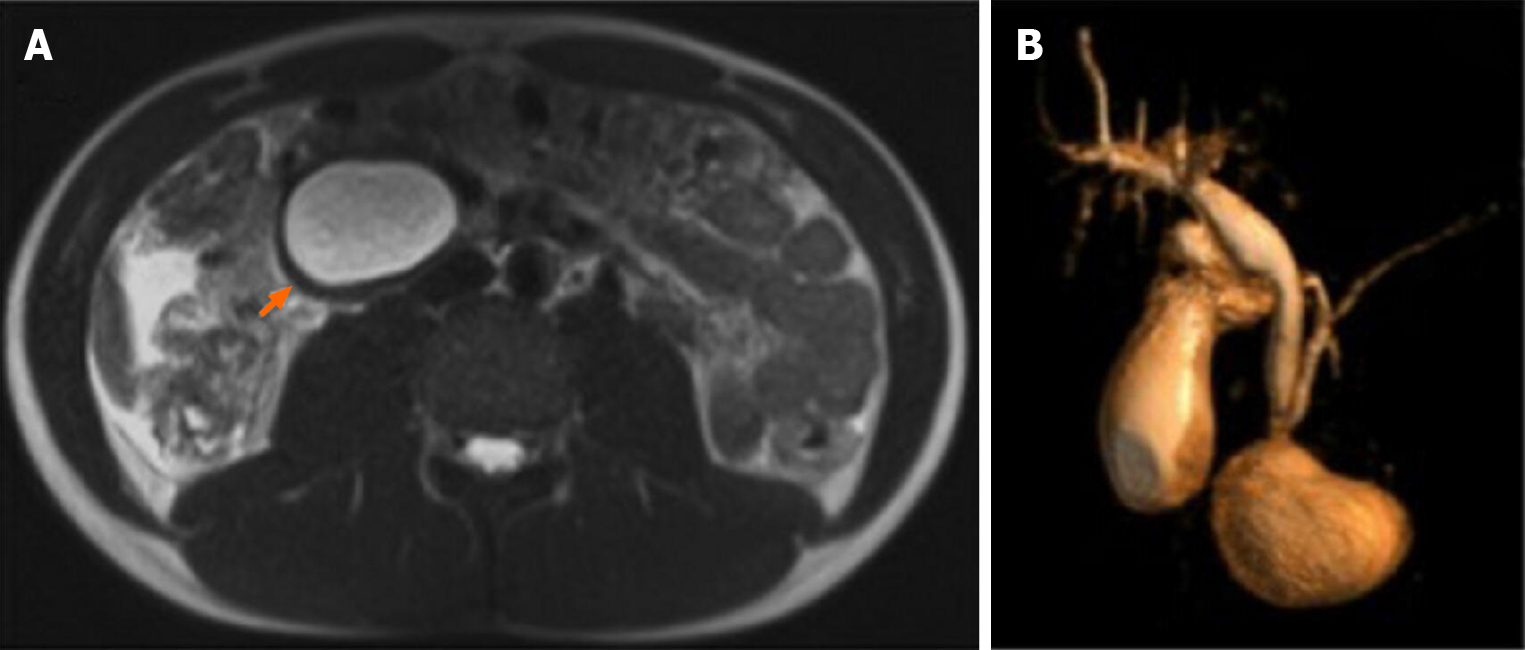
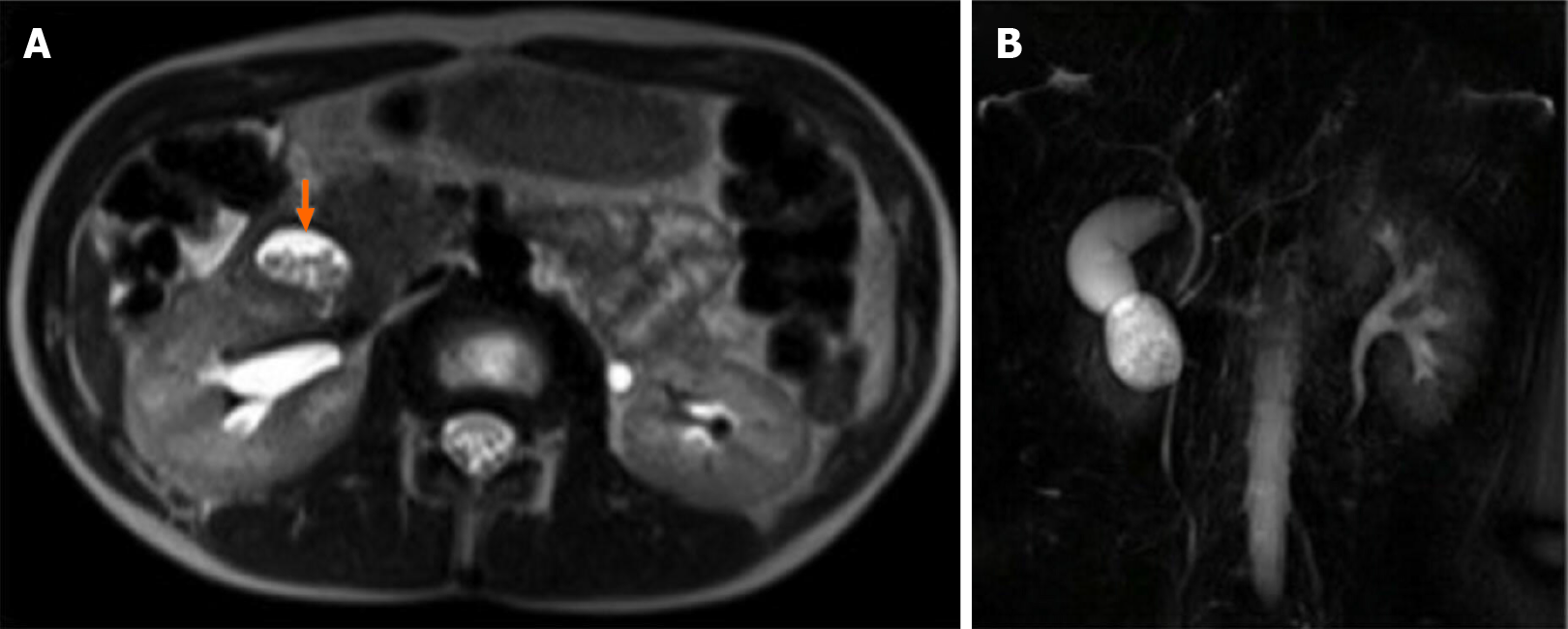
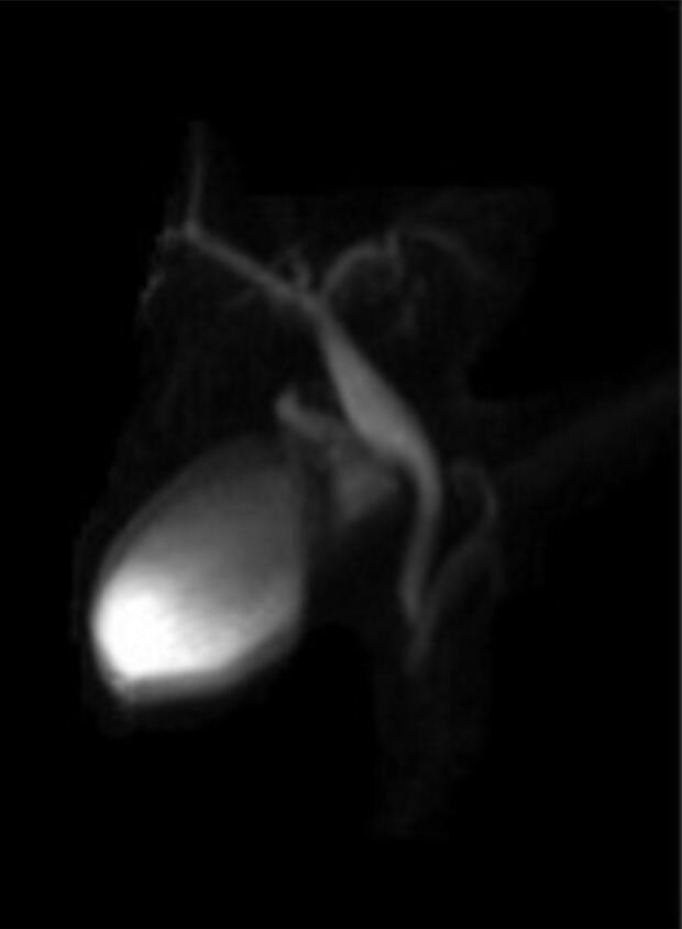
A magnetic resonance imaging (MRI) on HASTE T2-w sequence showed a homogeneously hyperintense cyst below the pancreatic head, located within a partially occluded duodenum (Figure 1A). On cholangiographic reconstruction the intrahepatic bile ducts were normal, the cystic duct appeared dilated with a tortuous course and the common hepatic duct presented saccular dilation. CBD had a caliber at the upper limits of the normal range with a regular course; the Wirsung duct was normal (Figure 1B).
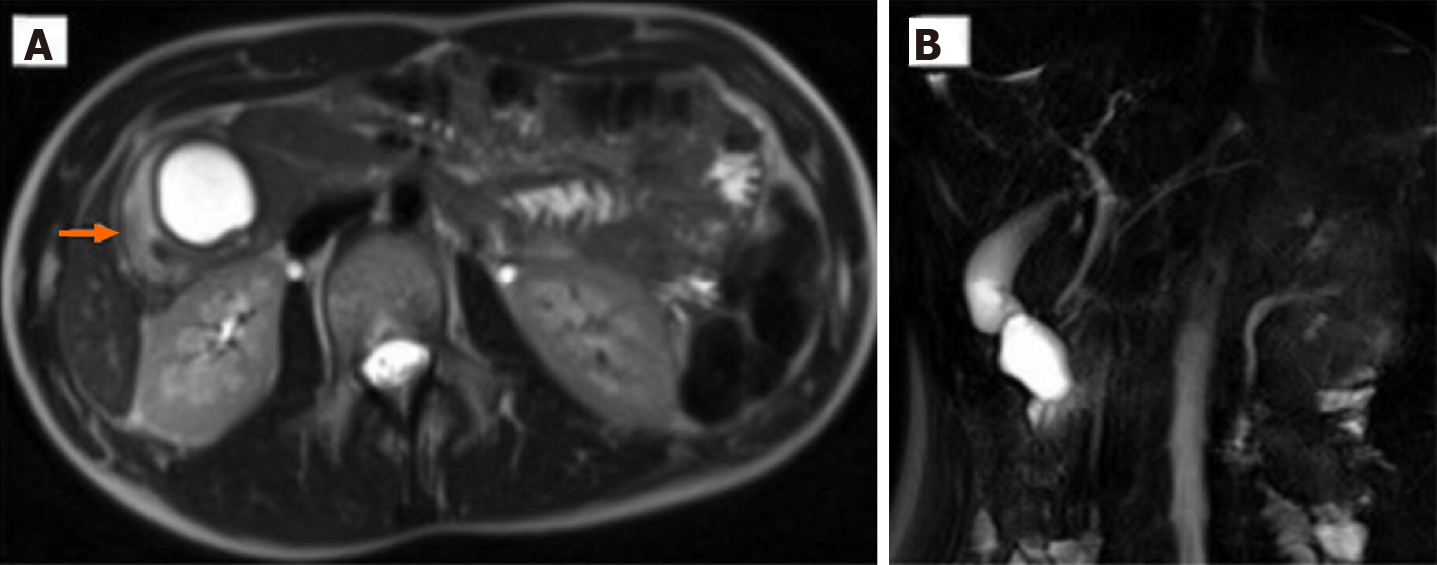
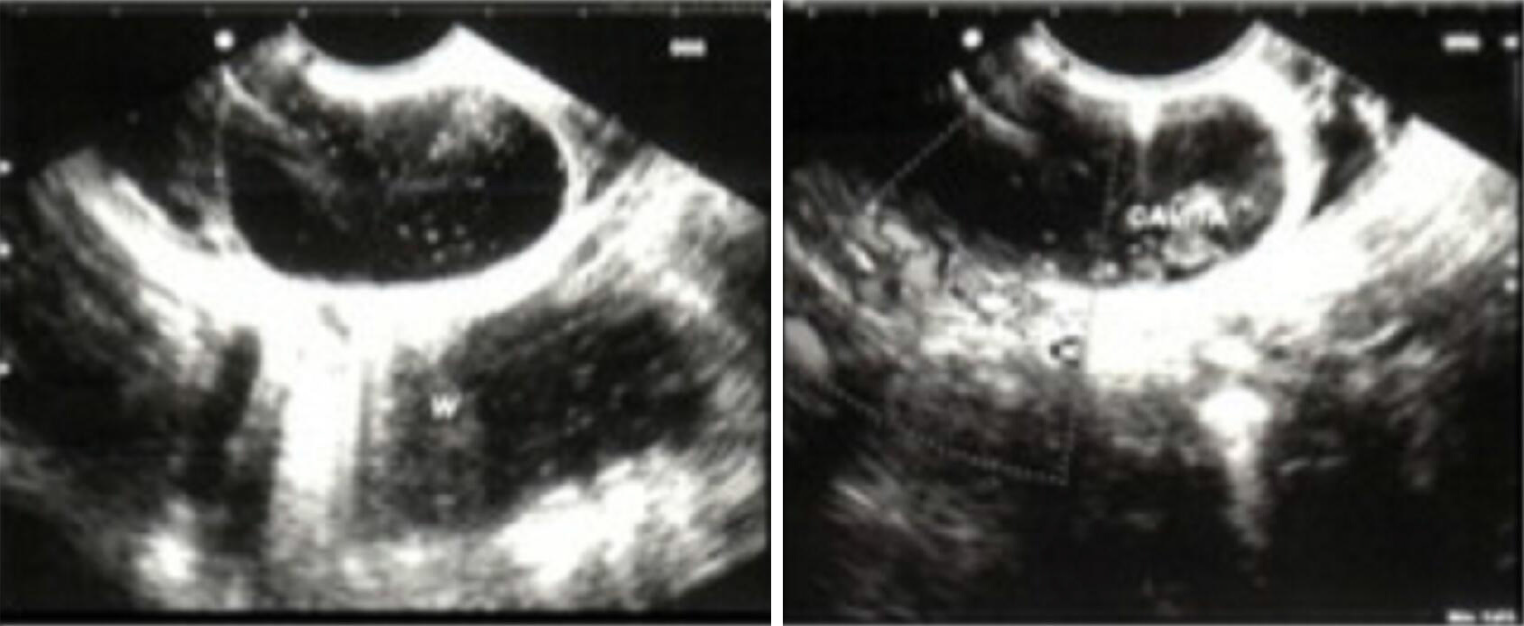
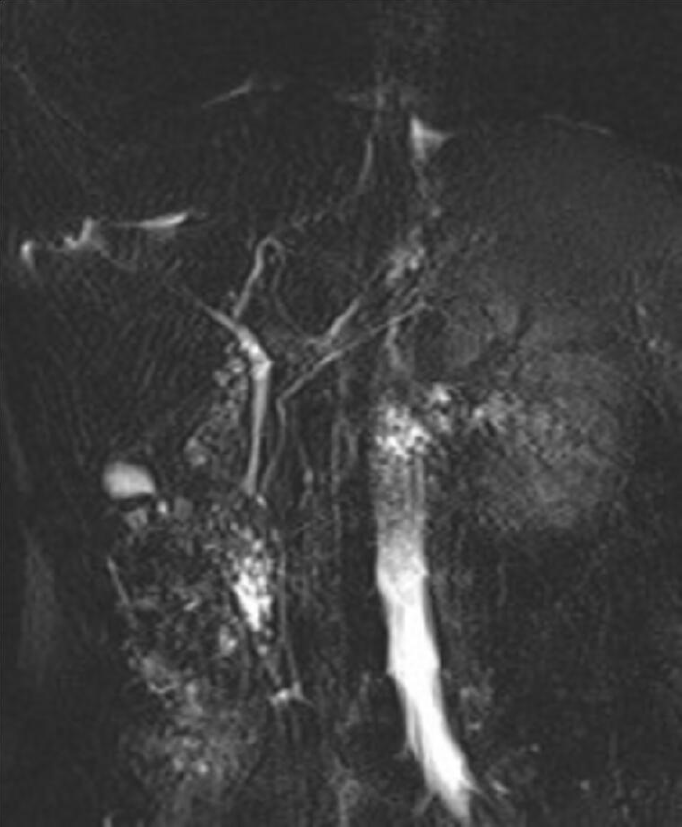
Case 2: An MRI on HASTE T2 w sequence revealed (Figure 2) a round homogeneous hyperintense lesion on the pancreas uncinate process, determining a major compression of the second portion of the duodenum. At cholangiographic reconstruction, the intra- and extrahepatic biliary tree along with the pancreatic ductal system were normal (Figure 2B).
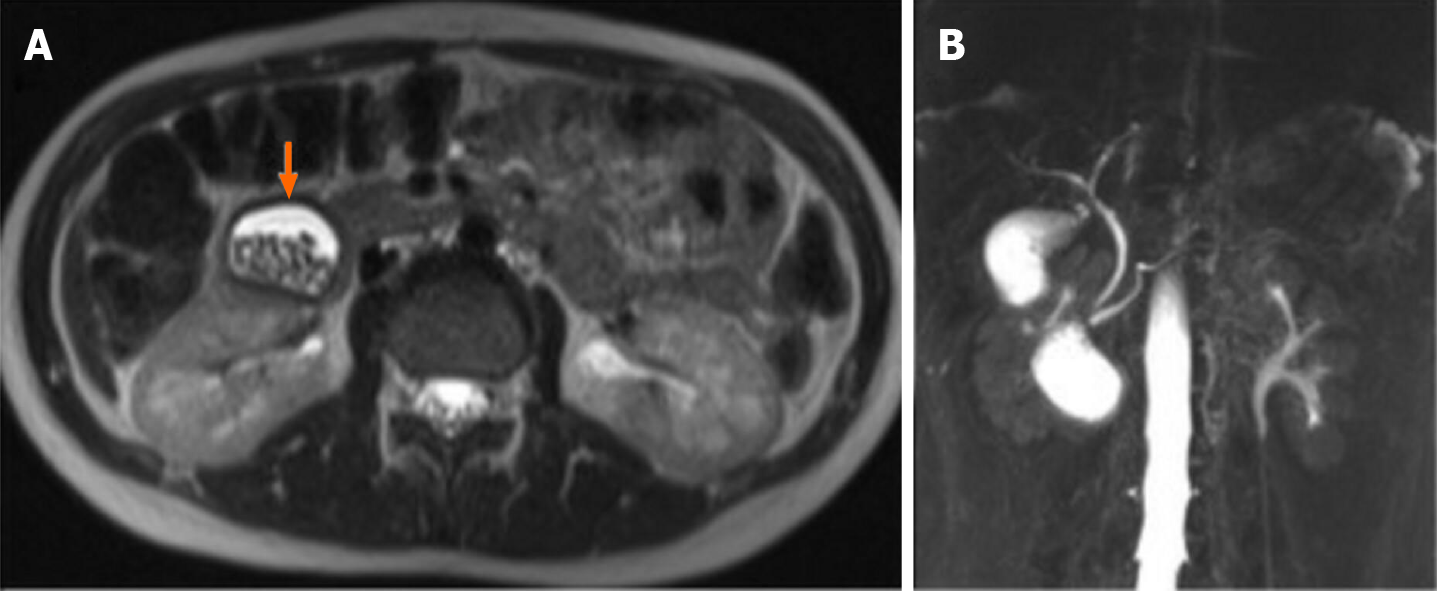
Case 3: An MRI on HASTE T2 w sequence showed an oval heterogeneous hyper
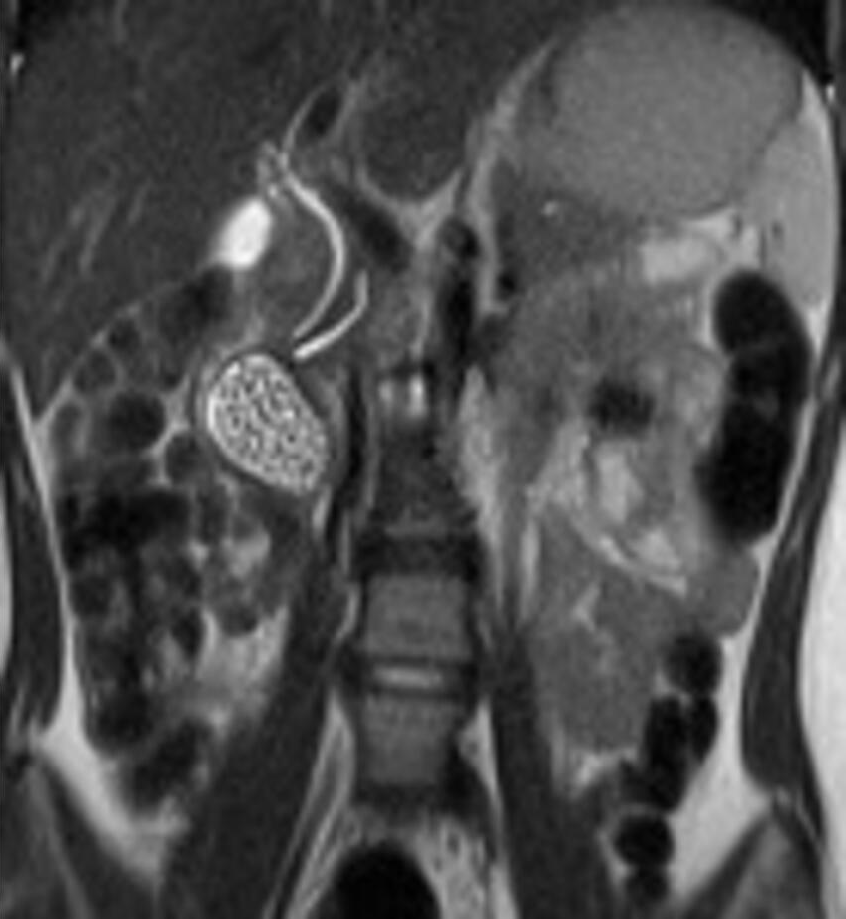
Case 4: US examination found a cyst (2.5 cm × 2.5 cm × 1.6 cm) sharing bowel wall stratification with the second part of the duodenum and full of hyperechogenic debris. An MRI on HASTE T2 sequence detected an oval mass, located below the gallbladder and laterally to the CBD and pancreatic duct (Figure 5), adjacent to the pancreatic head. The cyst was filled with fluid and multiple stones. Cholangiographic reconstruction indicated a normal gallbladder and intra- and extrahepatic bile ducts.
Endoscopic ultrasound (EUS) showed a bulging in the second duodenal portion, covered with normal mucosa, next to the Vater’s papilla and filled with biliary sludge (Figure 6). The lesion preserved a five-layer wall consisting with the typical echoendoscopic feature for the gastrointestinal wall consistent with a PADDC, and ET was proposed to parents.
A EUS showed an anechoic cystic lesion within the second duodenal portion, characterized by normal echographic bowel wall stratification and containing multiple hyperechoic stones; the cyst was not in communication with the CBD, and thereby PADDC was diagnosed.
A EUS revealed an anechoic cystic lesion characterized by a normal echographic bowel wall stratification and containing biliary sludge.
Duodenoscopy revealed an intraduodenal cyst, next to the papilla of Vater and not in communication with the duodenal lumen.
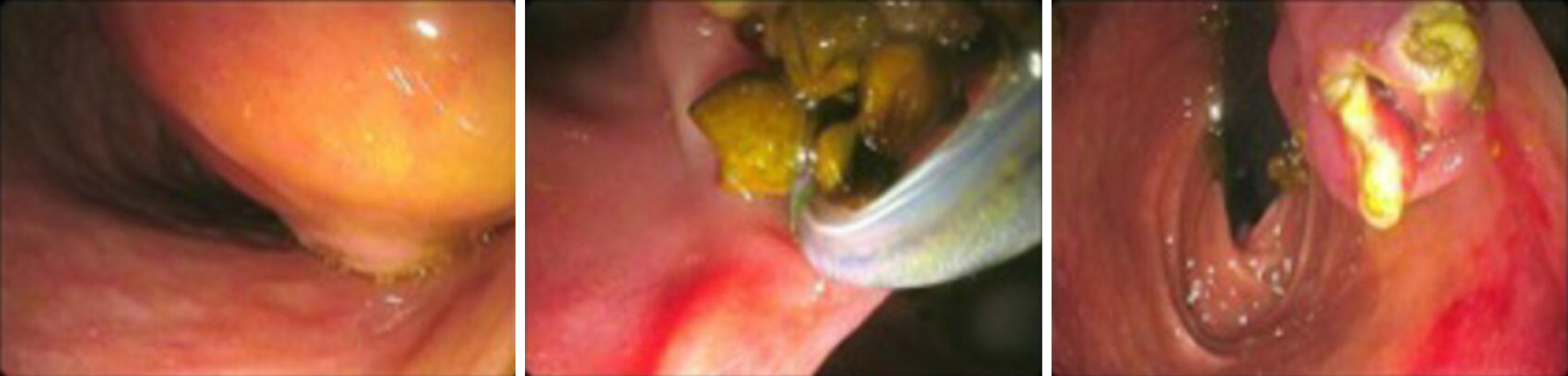
Upon endoscopic retrograde cholangio-pancreatography (ERCP), elective cannulation of the CBD showed a direct communication with the cyst and multiple stones in its lumen. A sphincterotome incision of the wall cyst, laterally to the papilla, was performed, and the stones were removed.
Upon ERCP, a small orifice on the lateral surface of the cyst was cannulated; a contrast injection failed to demonstrate any communication with the CBD. Intracystic stones were confirmed. The DD wall was incised with sphincterotome,, and stones were removed.
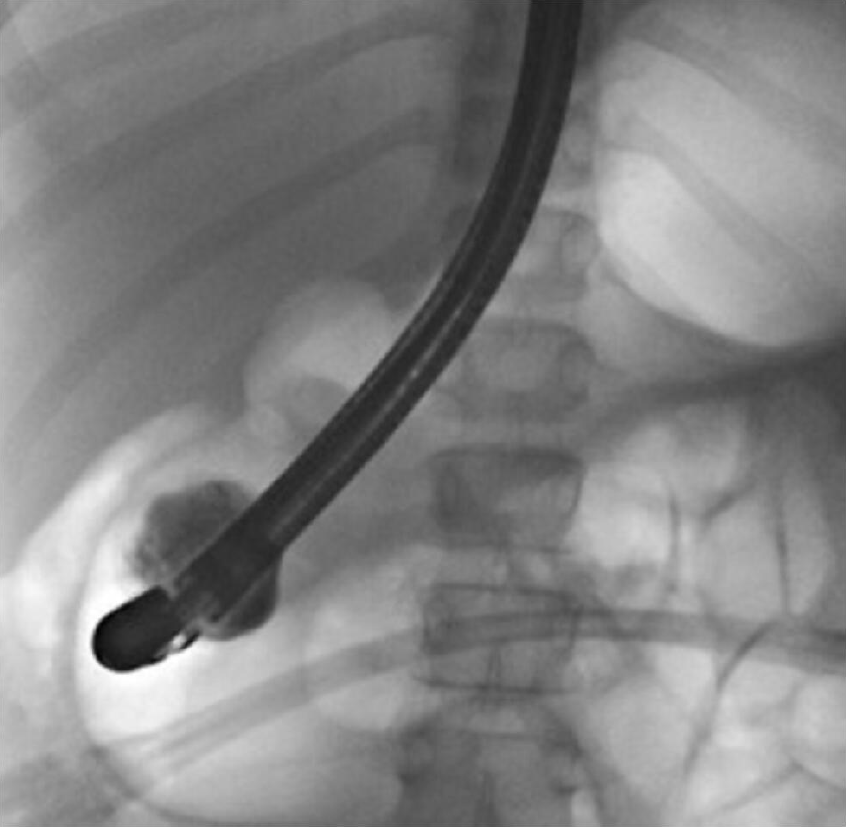
ERCP showed a regular main pancreatic duct; after distal papillotomy, contrast was injected, and it filled the PADDC (Figure 7). Marsupialization of the cyst with sphincterotome was then performed.
ERCP showed a normal pancreatic duct, dilation of CBD (20 mm diameter) without a detectable communication with the cyst. Cyst marsupialization was performed with subsequent extraction of biliary microstones (Figure 8).
The patient had an uneventful postoperative course and was discharged home 8 d later with a quick resolution of the abdominal pain and normalization of serum pancreatic enzymes. Ursodeoxycholic acid therapy and a hypolipic diet were continued until the next follow-up. At the 3 mo follow-up, magnetic resonance cholangiopancreatography (MRCP) control after ET, PADDC was no longer detected (Figure 9). At the 10-year follow-up the patient is doing well, without any therapy or further episodes of pancreatitis.
The patient had an uneventful recovery and was discharged home 2 d after the procedure with low fat meals. The 9 mo follow-up MRCP did not show any residual duplication (Figure 10), and at 8 years follow-up no further pancreatitis episodes were reported.
The postoperative course was complicated by severe melena on day 3, which required packed red cell transfusion. Esophagogastroduodenoscopy detected bleeding at the cyst section site. Endoscopic metallic clip placement was effective for bleeding control. The patient showed a progressive normalization of the serum lipase, and she was discharged home with ursodeoxycholic acid therapy and a low-fat diet. MRCP, done 2 mo later, did not show any duodenal cyst or intra- or extrahepatic bile and pancreatic duct dilatation. At the 4-year follow-up, she was well, and no further episodes of abdominal pain were reported.
The patient had an uneventful recovery and was discharged home 10 d after the procedure, with an ursodeoxycholic acid therapy and low-fat meals for 3 mo.
At the 2-year follow-up, she was totally asymptomatic, abdominal US was normal, and she eats a free diet.
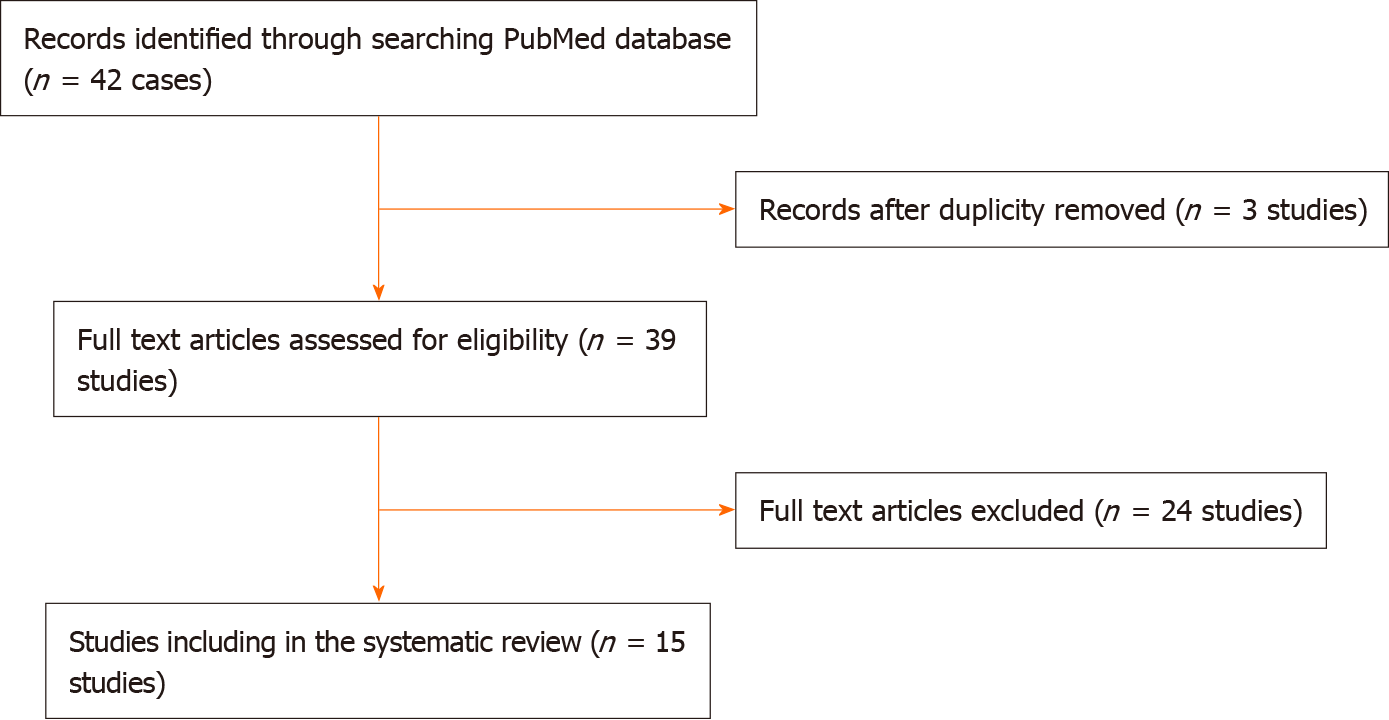
This literature review was performed according to preferred reporting items for systematic reviews and meta-analyses (PRISMA) guidelines[11] (Figure 11). The PubMed database was searched for original studies on “duodenal duplication,” “periampullary duplication” or “endoscopic management” published since 1990, involving patients younger than 18 years of age. Eligible study designs were case reports, case series and reviews. We omitted reports in which abstracts indicated an adult population (> 18 years) and improper reporting of the diagnosis and treatment methods. We then evaluated the full text of the selected articles and consider PADDC only where that diagnosis was confirmed by authors.
According to Tröbs et al[4], PADDC were defined as cysts located near the major papilla and the biliary-pancreatic ampulla that can have a small aberrant pancreatic duct drained into the cyst. We excluded all patients with a diagnosis of biliary/gallbladder disease (including acute acalculous cholecystitis) or with a diagnosis of duodenal duplication not located near the major papilla.
The date of the last search was December 2020. For each study, data were extracted for two primary outcomes (diagnostic assessment and type of treatment) and several secondary outcomes (including sex and age at presentation, clinical presentation, pathological examination and outcome). Analysis was extended to our additional 4 cases.
The initial PubMed search yielded 42 potentially relevant studies. Eventually, 16 eligible articles met the inclusion criteria, involving a total of 17 children with PADDC[1,3,4,6-9,12-20] (Table 1 and Figure 11). All selected studies were case reports (class of evidence Ⅲ and rating scale of evidence E) and clearly reported the two primary outcomes.
| Ref. | Year | Age | Sex | Clinical | Laboratory data | US | MR/CT | EUS | ERCP | Description | Treatment and complications |
| Mattioli et al[13] | 1999 | 11 yr | F | Abdominal pain | NA | Yes | Yes (CT) | No | Yes | Periampullary duplication | Surgical resection |
| Zamir et al[16] | 1999 | 17 yr | M | Abdominal pain, duodeno-jejunal intussusception | AST/ALT, 50/140; ALP 250, GGT 400 | Yes | Yes (CT) | No | No | Periampullary duplication | Surgical cyst marsupialization |
| Niehues et al[18] | 2005 | 16 yr | M | Abdominal pain, jaundice | Lipase 3343 | Yes | Yes (CT and MRCP) | No | Yes | Periampullary duplication | Surgical resection and cholecystectomy |
| Guarise et al[2] | 2006 | 18 yr | M | Abdominal pain, pancreatitis | NA | Yes | Yes (CT and MRCP) | Yes | Yes | Periampullary duplication | Surgical resection |
| Chryssostalis et al[8] | 2007 | 17 yr | - | Abdominal pain Recurrent pancreatitis | NA | Yes | Yes (CT) | No | Yes | Periampullary duplication | Endoscopic excision of the cyst |
| Ozel et al[14] | 2008 | 8 yr | F | Abdominal pain, pancreatitis | Amylase 1287 | Yes | Yes (CT) | No | No | Periampullary duplications | Surgical resection |
| Chen et al[3] | 2009 | 8 yr | F | Abdominal pain, pancreatitis | Amylase 155; lipase 109 | Yes | Yes (CT and MRCP) | No | Yes | Periampullary duplication | Surgical cyst marsupialization |
| Tröbs et al[4] | 2009 | 8 yr | M | Abdominal pain, pancreatitis, hepatitis | Lipase 3000 | Yes | Yes (CT and MRCP) | No | No | Periampullary duplication | Surgicalcyst marsupialization |
| Tekin et al[7] | 2009 | 18 yr | F | Abdominal pain, pancreatitis | NA | Yes | Yes (CT) | No | Yes | Periampullary duplication | Endoscopic sphincterotomy and stent implantation |
| Criblez et al[17] | 2011 | 17 yr | M | Abdominal pain | Lipase 5400 | Yes | Yes (CT) | No | Yes | Periampullary duplication | Endoscopic cyst marsupialization and sphincterotomy |
| Romeo et al[9] | 2011 | - | - | Recurrent pancreatitis | NA | Yes | Yes (CT and MRCP) | Yes | Yes | Periampullary duplication | Surgical resection of common wall |
| - | - | Recurrent pancreatitis | NA | Yes | Yes (CT and MRCP) | Yes | No | Periampullary duplication | Endoscopic cyst wall resection | ||
| Meier et al[6] | 2012 | 9 yr | M | Abdominal pain | Amylase 270 U/ml; Lipase 824 U/ml | Yes | Yes (CT and MRCP) | No | Yes | Periampullary duplication | Endoscopic opening of cyst wall |
| Koffie et al[12] | 2012 | 13 yr | M | Abdominal pain, hepatitis and pancreatitis | Lipase 1363; Amylase 401, direct bilirubin 9.1 | Yes | Yes (CT and MRCP) | No | No | Periampullary duplication | Surgical resection |
| Taghavi et al[15] | 2017 | 17 yr | M | Recurrent pancreatitis | NA | Yes | Yes (MRCP) | No | No | Periampullary duplication | Surgical resection, sphincteroplasty of terminal pancreatic duct and stent positioning. |
| Salazar et al[19] | 2018 | 3 yr | M | Abdominal pain, pancreatitis | NA | Yes | Yes (MRCP) | Yes | No | Periampullary duplication | Endoscopic cyst marsupialization |
| This case | 2019 | 14 yr | M | Recurrent pancreatitis and abdominal pain | Lipase 1077, Amylase 514 GGT 69 | Yes | Yes (CT in another center, MRCP) | Yes | Yes | Periampullary duplication | Endoscopic distal papillotomy and cyst incision |
| 16 yr | F | Recurrent pancreatitis and abdominal pain | Lipase 2365 | Yes | Yes (CT in another center, MRCP) | Yes | Yes | Periampullary duplication | Endoscopic cyst incision | ||
| 11 yr | F | Recurrent pancreatitis, abdominal pain and weight loss | Lipase 43440 | Yes | Yes (CT in another center, MRCP) | Yes | Yes | Periampullary duplication | Endoscopic cyst incision (bleeding treated with metallic clips placement and blood transfusion) | ||
| 11 yr | F | Pancreatitis | Lipase 5497, AST/ALT 315/532; GGT 169 | Yes | Yes (MRCP) | Yes | Yes | Periampullary duplication | Sphincterotomy |
The patients’ median age at diagnosis was 14 years (range: 3-18 years), and PADDC was reported in 10 males and 8 females. For 3 patients, data were not available. Clinical presentation was unspecific, with abdominal pain reported in all cases. Recurrent pancreatitis was the most common complication and was observed in 14 cases (70%), followed by cholestasis, jaundice and intussusception.
All patients underwent abdominal ultrasound, followed by abdominal CT scan in 18 cases (90%), ERCP in 13 (65%), MRCP in 7 (35%) and EUS in 8 (4%); 1 patient was only examined with ERCP (5%) (Table 1).
Endoscopic treatment was performed in 10 patients (50%), with two reported complications, namely bleeding at the duplication incision site, which were treated with packed red cell transfusion and endoscopic clipping of the bleeding site in one case and with local injection of epinephrine in the other case (Table 1) [9]. The median follow-up was 22.5 mo (range: 4-108 mo); all endoscopically treated patients are doing well with disappearance of the duplication on imaging. No case of malignancy was reported.
Duodenal duplications are uncommon congenital anomalies of the gastrointestinal tract, which usually present during the first decade of life[4,5]. They represent 5%-7% of all gastrointestinal duplications and result from disturbances in the embryonic development, probably due to duodenal epithelial pinching during the outgrowth of the dorsal pancreatic bud or secondary to an epithelial sequestration[4]. The majority of them are cystic, adherent and located on the mesenteric side of the second or third portion of the duodenum, with an epithelial mucosal lining and a smooth muscle layer[10,21]. A communication with the duodenal lumen has been reported in up to 25% of cases[1], and some authors have also described the possibility of a pancreato-biliary involvement in 30% of patients, although this cannot always be the only explanation of pancreatitis[5,6].
Three different mechanisms have been reported as responsible for pancreatitis: (1) External papilla obstruction by duplication enlargement; (2) Presence of an aberrant pancreatic duct within the duplication, which can become obstructed by mucus and debris; and (3) Migration of biliary sludge and/or microstones from the cyst into the bilio-pancreatic duct[3,4]. Migration of biliary sludge and/or microstones from the cyst to the bilio-pancreatic duct is possible only due to a communication between the duplication and the bilio-pancreatic duct with stone formation due to the bile stasis within the duplication seeing as its peristalsis is intermittent[2]. For this reason, the presence of stones or biliary sludge inside a duodenal mass do not ruled out the possibility of a DD.
DD can be divided into two subgroups: periampullary (PADDC) and non-periampullary duplication cyst. According to Tröbs et al[4] periampullary duodenal duplication is defined as a duplication cyst located near the major papilla and the biliary-pancreatic ampulla, sometimes with a small aberrant pancreatic duct drained into the cyst[4].
Our experience suggests the possibility of communication between PADDC and the CBD and pancreatic duct, which explains both the possibility of observing sludge or calculi in the cyst and the pancreatitis. Unfortunately, detailed descriptions of the relationships between duplication and major papilla and/or pancreatic ampulla are lacking, and our review found that only 17 out of 49 pediatric patients reported a detailed description of the DD that can be classified as periampullary type (Table 1).
PADDC cases have been reported in childhood with a median age of diagnosis of 14 years (range: 3-18 years); this was consistent also in our series (Table 1).
The first radiological tool for diagnosis was US, which is highly suggestive for a DD when peristalsis and pathognomonic “double wall sign,” consisting of an outer hypoechoic muscular layer, an internal echogenic mucosal layer and corpuscular fluid inside the lesion, are found[22]. However, this finding should be confirmed with a more exhaustive radiological work-up by abdominal CT scan or preferably by MRCP[23], which provides more information about the location, size, enhancement and multilayered duplication cyst wall as well as anatomical details of the biliary and pancreatic ductal system. Furthermore, ionizing radiation should be limited as much as possible in childhood.
Moreover, we suggest performing an EUS in children with a cystic lesion next to the papilla. In our experience, EUS offered two major advantages: (1) Endoscopic vision allowed a better definition of the intraluminal duodenal lesion and an accurate localization of the papilla; and (2) US vision highlighted the presence of an anechoic structure surrounded by a five layer wall, consisting with the typical echo-endoscopic feature for the gastrointestinal wall, distinguishing DD from the other cystic and neoplastic duodenal or pancreatic masses, including cystic dystrophy of the duodenal wall, pseudocysts, cystic lymphangiomas, mesenteric cysts and choledochocele[4,24].
In particular, the performance of EUS to identify the presence of normal echographic bowel wall stratification at the DD allowed us to make differential diagnosis with choledochocele, where that hallmark is absent, but which represents the most frequent and challenging differential diagnosis. Furthermore, although many authors consider biopsy as the gold standard for the differential diagnosis between DD and choledochocele, duodenal type mucosa has been reported in choledochocele[25-27]. Sarris and Tsang reported 15 cases of choledochocele with duodenal mucosa at pathological examination[27,28].
Eventually, EUS can well indicate the relationships between the duplication and biliary-pancreatic duct. Therefore, when a PADDC is suspected, we suggest con
Despite having carried out the EUS, before proceeding with the endoscopic duplication unroofing, ERCP would have to be mandatory in order to obtain a detailed anatomic view of the bilio-pancreatic system and to detect a possible communication between the duplication and the biliary and/or pancreatic duct, particularly in patients with stones or sludge inside the cyst.
Endoscopic treatment of children with PADDC was first described in 2007[8], and a later meta-analysis of the pediatric population confirmed the safety, feasibility and effectiveness of this approach in this population[10]. Our review revealed that 10/20 patients with PADDC (50%) underwent ET[6-9,17,19].
Two postoperative complications occurred (bleeding) and were both endoscopically treated; this point stresses the importance of ensuring a careful coagulation of the severed edges of the duplication. When planning an ET we thereby advise that a thorough preoperative radiological imaging encompassing EUS be mandatory, and our experience suggests that the real incidence of PADDC is underestimated because of incomplete preoperative imaging.
The anatomic location of the PADD and the possible communication with the biliary and/or pancreatic ductal system makes an open surgical approach highly demanding and not necessarily safer than ET. Furthermore, surgery has several disadvantages over ET, including worse postoperative pain, higher risk of postoperative complications, visible scars and longer hospitalization time.
Endoscopic cyst marsupialization was highly effective in relieving symptoms and cyst disappearance even at long-term follow-up.
Undoubtedly endoscopic management of PADDC requires a skilled multidisciplinary team, and the still limited use of the endoscopic strategy in a pediatric setting is probably explained, other than the rarity of PADDC, by the unavailability of a trained ERCP endoscopic team.
We suggest considering ET as a first line approach after a complete EUS study and reserving a surgical approach only when it is impossible to understand the relationship between PADDC and the pancreato-biliary tree.
ET provides marsupialization or incision of PADDC, therefore it is rare, but possible, to leave ectopic gastric or pancreatic tissue with potential risk of malignant degeneration.
Eventually, although DD (PADDC included) are generally benign lesions and only a few cases of malignant transformation have been reported in literature[5,29,30], a long-term follow up is mandatory in endoscopically treated patients, even in asymptomatic ones.
PADDC in pediatric patients are very rare. Our experience suggests that an accurate preoperative assessment with EUS is essential to differentiating the duplication from other duodenal lesions. In the presence of sludge or stones inside the duplication, ERCP is mandatory to demonstrate a communication with the biliary tree. ET is a safe, minimally invasive and effective treatment in children with PADDC. Long-term follow-up of this population throughout adulthood is mandatory and necessary considering that malignant degeneration of duodenal duplication has been described[5,29,30].
Manuscript source: Invited manuscript
Specialty type: Surgery
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Khayyat YM, Yang WY S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Liu JH
| 1. | Rockx MA, McAlister VC. Endoscopic fenestration of a duodenal duplication cyst to resolve recurrent pancreatitis. JOP. 2007;8:795-798. [PubMed] |
| 2. | Guarise A, Faccioli N, Ferrari M, Romano L, Parisi A, Falconi M. Duodenal duplication cyst causing severe pancreatitis: imaging findings and pathological correlation. World J Gastroenterol. 2006;12:1630-1633. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 34] [Cited by in RCA: 31] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 3. | Chen JJ, Lee HC, Yeung CY, Chan WT, Jiang CB, Sheu JC. Meta-analysis: the clinical features of the duodenal duplication cyst. J Pediatr Surg. 2010;45:1598-1606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 77] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 4. | Tröbs RB, Hemminghaus M, Cernaianu G, Liermann D. Stone-containing periampullary duodenal duplication cyst with aberrant pancreatic duct. J Pediatr Surg. 2009;44:e33-e35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 5. | Gjeorgjievski M, Manickam P, Ghaith G, Cappell MS. Safety and Efficacy of Endoscopic Therapy for Nonmalignant Duodenal Duplication Cysts: Case Report and Comprehensive Review of 28 Cases Reported in the Literature. Medicine (Baltimore). 2016;95:e3799. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 6. | Meier AH, Mellinger JD. Endoscopic management of a duodenal duplication cyst. J Pediatr Surg. 2012;47:e33-e35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 7. | Tekin F, Ozutemiz O, Ersoz G, Tekesin O. A new endoscopic treatment method for a symptomatic duodenal duplication cyst. Endoscopy. 2009;41 Suppl 2:E32-E33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 8. | Chryssostalis A, Ribiere O, Prat F. Endoscopic management of a duodenal duplication cyst filled with stones and revealed by recurrent pancreatitis. Clin Gastroenterol Hepatol. 2007;5:e31-e32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 9. | Romeo E, Torroni F, Foschia F, De Angelis P, Caldaro T, Santi MR, di Abriola GF, Caccamo R, Monti L, Dall'Oglio L. Surgery or endoscopy to treat duodenal duplications in children. J Pediatr Surg. 2011;46:874-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Wada S, Higashizawa T, Tamada K, Tomiyama T, Ohashi A, Satoh Y, Sugano K, Nagai H. Endoscopic partial resection of a duodenal duplication cyst. Endoscopy. 2001;33:808-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 26] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 11. | Moher D, Liberati A, Tetzlaff J, Altman DG; PRISMA Group. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. Int J Surg. 2010;8:336-341. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9207] [Cited by in RCA: 8044] [Article Influence: 536.3] [Reference Citation Analysis (2)] |
| 12. | Koffie RM, Lee S, Perez-Atayde A, Mooney DP. Periampullary duodenal duplication cyst masquerading as a choledochocele. Pediatr Surg Int. 2012;28:1035-1039. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Mattioli G, Buffa P, Pesce F, Barabino A, Ganduglia P, Fratino G, Granata C, Torre M, Magnano G, Gambini C, Ivani G, Jasonni V. Pancreatitis caused by duodenal duplication. J Pediatr Surg. 1999;34:645-648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 17] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 14. | Ozel A, Uysal E, Tufaner O, Erturk SM, Yalcin M, Basak M. Duodenal duplication cyst: a rare cause of acute pancreatitis in children. J Clin Ultrasound. 2008;36:584-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 15. | Taghavi K, Wilms H, Bann S, Stringer MD. Duodenal duplication cyst causing recurrent pancreatitis. J Paediatr Child Health. 2017;53:814-816. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 5] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 16. | Zamir G, Gross E, Shmushkevich A, Bar-Ziv J, Durst AL, Jurim O. Duodenal duplication cyst manifested by duodeno-jejunal intussusception and hyperbilirubinemia. J Pediatr Surg. 1999;34:1297-1299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 17. | Criblez D, Mitschele T, Scheiwiller A. A rare cause of acute pancreatitis in an adolescent. Juxtapapillary duodenal duplication cyst as a rare cause of acute pancreatitis in an adolescent. Gastroenterology. 2011;140:783, 1110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 18. | Niehues R, Dietl KH, Bettendorf O, Domschke W, Pohle T. Duodenal duplication cyst mimicking pancreatic cyst in a patient with pancreatitis. Gastrointest Endosc. 2005;62:190-192. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 19. | Salazar E, Sin EI, Low Y, Khor CJL. Insulated-tip knife: an alternative method of marsupializing a symptomatic duodenal duplication cyst in a 3-year-old child. VideoGIE. 2018;3:356-357. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 20. | Alatas FS, Masumoto K, Matsuura T, Pudjiadi AH, Taguchi T. Modified Puestow Procedure for Chronic Pancreatitis in a Child Due to Annular Pancreas and Duodenal Duplication: A Case Report. Pediatr Gastroenterol Hepatol Nutr. 2020;23:304-309. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 21. | Merrot T, Anastasescu R, Pankevych T, Tercier S, Garcia S, Alessandrini P, Guys JM. Duodenal duplications. Clinical characteristics, embryological hypotheses, histological findings, treatment. Eur J Pediatr Surg. 2006;16:18-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 46] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 22. | Cheng G, Soboleski D, Daneman A, Poenaru D, Hurlbut D. Sonographic pitfalls in the diagnosis of enteric duplication cysts. AJR Am J Roentgenol. 2005;184:521-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 86] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 23. | Wong AM, Wong HF, Cheung YC, Wan YL, Ng KK, Kong MS. Duodenal duplication cyst: MRI features and the role of MR cholangiopancreatography in diagnosis. Pediatr Radiol. 2002;32:124-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 24. | Ko SY, Ko SH, Ha S, Kim MS, Shin HM, Baeg MK. A case of a duodenal duplication cyst presenting as melena. World J Gastroenterol. 2013;19:6490-6493. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 18] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 25. | Ghazi A, Slone E. Endoscopic management of choledochocele. A case report and review of the English literature. Surg Endosc. 1987;1:151-154. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 26. | Schimpl G, Sauer H, Goriupp U, Becker H. Choledochocele: importance of histological evaluation. J Pediatr Surg. 1993;28:1562-1565. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 37] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 27. | Sarris GE, Tsang D. Choledochocele: case report, literature review, and a proposed classification. Surgery. 1989;105:408-414. [PubMed] |
| 28. | Zhu L, Lv Z, Liu J, Xu W. Choledochocele: A Case Report and Discussion of Diagnosis Criteria. European J Pediatr Surg Rep. 2015;3:85-89. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 29. | Seeliger B, Piardi T, Marzano E, Mutter D, Marescaux J, Pessaux P. Duodenal duplication cyst: a potentially malignant disease. Ann Surg Oncol. 2012;19:3753-3754. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Rai BK, Zaman S, Mirza B, Hanif G, Sheikh A. Duodenal Duplication Cyst having Ectopic Gastric and Pancreatic Tissues. APSP J Case Rep. 2012;3:15. [PubMed] |