Published online May 8, 2015. doi: 10.4254/wjh.v7.i7.980
Peer-review started: August 29, 2014
First decision: October 14, 2014
Revised: December 22, 2014
Accepted: February 4, 2015
Article in press: February 9, 2015
Published online: May 8, 2015
Processing time: 258 Days and 3.8 Hours
Hepatocellular carcinoma (HCC) is a common cancer worldwide with a poor prognosis. Few strategies have been proven efficient in HCC treatment, particularly for those patients not indicated for curative resection or transplantation. Immunotherapy has been developed for decades for cancer control and is attaining more attention as a result of encouraging outcomes of new strategies such as chimeric antigen receptor T cells and immune checkpoint blockade. Right at the front of the new era of immunotherapy, we review the immunotherapy in HCC treatment, from basic research to clinical trials, covering anything from immunomodulators, tumor vaccines and adoptive immunotherapy. The mechanisms, efficacy and safety as well as the approach particulars are unveiled to assist readers to gain a concise but extensive understanding of immunotherapy of HCC.
Core tip: This paper supplies a comprehensive review of immunotherapy for hepatocellular carcinoma from basic experiments to clinical trials. The development of interferon, chemokines, tumor vaccines, adoptive immunotherapy, including natural killer, natural killer T and T cells armed with chimeric antigen receptor, as well as regulatory T cell is summarized.
- Citation: Hong YP, Li ZD, Prasoon P, Zhang Q. Immunotherapy for hepatocellular carcinoma: From basic research to clinical use. World J Hepatol 2015; 7(7): 980-992
- URL: https://www.wjgnet.com/1948-5182/full/v7/i7/980.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i7.980
Hepatocellular carcinoma (HCC) accounts for 95% of primary liver cancer[1], the second most common cause of cancer-associated death worldwide and estimated to be responsible for around 746000 deaths in 2012[2]. Only liver resection and liver transplantation are considered curative, with poor efficiency of other modalities such as radiofrequency ablation (RFA) and transarterial chemoembolization (TACE). However, very few patients are indicated for liver resection and donors of liver are usually exceptional. Postoperative recurrence frequently occurs, resulting in a dismal prognosis in most affected individuals[3].
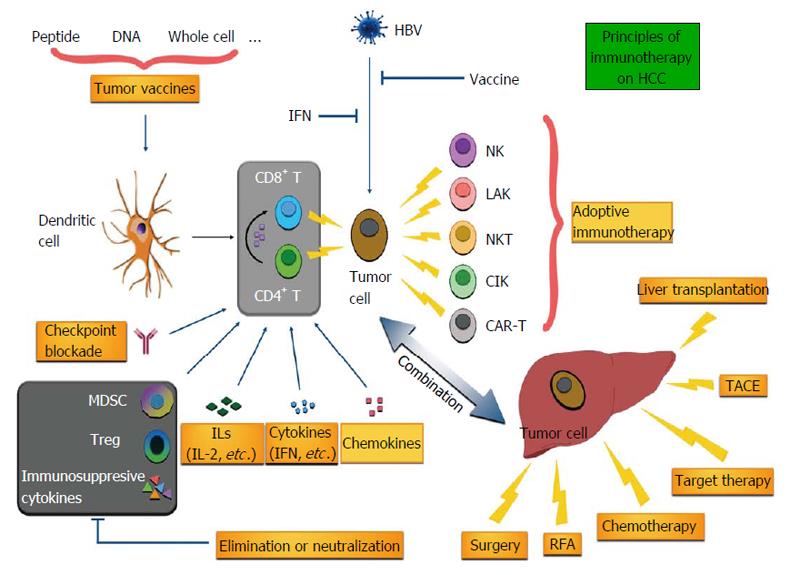
Immunotherapy has been explored in HCC for decades[4] and carries high expectations due to the recent progress in other malignancies such as melanoma. Different from other organs, liver shows its distinguished characteristics, such as an “immune organ”, and patients with HCC present with unique anti- or pro-tumor responses during the development and progression of HCC[5]. Immunotherapy can be categorized into several types according to their distinct strategies. For instance, immunomodulators and tumor vaccines are used to enhance the immune response to HCC in an indirect way; adoptive immunotherapy introduces a great amount of effective immune cells to directly remove tumor cells. In this review, we summarize the critical immune characteristics of liver and cover the immunotherapy of HCC in animal models as well as clinical trials (Figure 1).
The inherent immune tolerance of liver hinders immune surveillance and therefore makes the carcinogenesis of HCC possible. Liver confronts abundant xenogenous antigens within blood from the gut via the portal vein. Specific mechanisms with regards to immune tolerance are activated to inhibit unneeded immune responses. Unfortunately, these mechanisms, such as recruitment of regulatory T cells (Tregs) and myeloid-derived suppressor cells, as well as overexpression of inhibitory ligands such as programmed death ligand 1 (PD-L1), contribute to weaken anti-tumor immune responses[6].
As an “immune privileged” organ, multiple pathways exist within liver to maintain its function. Not only can hepatocytes cause T cell anergy in certain conditions, but many other nonparenchymal cells, including stellate cells, hepatic dendritic cells and liver sinusoidal endothelial cells, also induce tolerance or apoptosis of effective or naive T cells. In particular, HCC lacks major histocompatibility complex (MHC) class II, the activator of CD4+ T helper (Th) cells[6].
Clinically, various cytokines dysregulate and contribute to HCC progression[7]. Increased immunosuppressed cells in patients are in parallel with a poorer prognosis. Th17 and its secretory product interleukin 17 (IL-17) promote angiogenesis of HCC and recruit neutrophils to enhance angiogenesis[8,9]. The effector function of CD8+ T cells is prone to be impaired by increased Tregs, which predicts a poor prognosis of HCC patients[10]. In addition, the functional impairment of other cells like natural killer (NK) cells also contributes to tumor progression[11].
Various cytokines are involved in immune responses. Certain cytokines can directly inhibit tumor cell growth or enhance the capacity of relevant immune cells to delay tumor development.
Interferon is well known for immunomodulation, anti-proliferation and anti-angiogenesis. They were found to be decreased in serum of patients with HCC. All three types of interferon (IFN-α, IFN-β, IFN-γ) have been proved to be effective in inhibiting HCC by inducing tumor cell apoptosis or autophagy[12-14]. However, the efficiencies among different types of interferon are under debate. Although some investigators have addressed that IFN-β showed better anti-HCC effects[15], the use of IFN-α in HCC treatment have been more frequently reported. However, IFN-α alone did not show satisfactory survival benefit in patients with unresectable HCC, confirmed by randomized controlled trials (RCTs)[16,17]. IFN-α-2b also failed to decrease the risk of postoperative recurrence[18]. In contrast, interferon showed some benefits when combined with other modalities such as chemotherapy, curative resection and TACE[19-22]. Two meta-analyses revealed that adjuvant interferon therapy after curative therapy for HCC could improve both overall survival and recurrence-free survival[23,24]. Combination of IFN-α with sorafenib was also reported to be efficient in a mouse model[25] but this has not been tested in humans. In addition, interferon treatment may gain further benefits for HCC patients with hepatitis B or C virus infection from removing the viruses[26,27]. Currently, two registered clinical trials regarding IFN-α are still recruiting participants. One multicenter RCT is planning to test IFN-α as an adjuvant therapy in HCC with low miR-26 expression (NCT011681446) and the other phase II trial is trying to combine IFN-α with fluorouracil to treat HCC patients who underwent liver resection (NCT01834963). Generally, IFN-α showed demonstrated equivocal effects and should only be applied to selected patients as supportive or adjuvant therapy within the assumption of the current evidence.
ILs have been applied to enhance anti-tumor responses of the immune system. However, few studies concerning these ILs have been performed in humans. Small scale clinical studies evaluated the efficacies of IL-2 or IL-12 alone or combined together in HCC treatment but the results were inconclusive[28,29]. Other sorts of ILs were also studied. For instance, IL-37 was found to selectively recruit NK cells to conduct anti-tumor activity in HCC patients[30]. IL-24 was reported to show in vivo anti-tumor activity in the presence of apoptin[31]. However, clinical trials are lacking to determine the therapeutic effects of ILs in human.
Chemokines regulate activities and behavior of cells, including hepatocytes, immune cells and the tumor microenvironment. By mediating pro- and anti-inflammatory responses, chemokines can regulate leukocyte recruitment, angiogenesis and tumor progression[32]. Several chemokine-associated signaling, including CXCR4/CXCL12, CCR6/CCL20 axes, were evident in promoting HCC[33,34]. Blockade of these signalings by relevant receptors seems logical to control HCC.
Chemokines can regulate the function of immune cells by interacting with the receptors on the membrane of these cells. Tumor infiltrating anti-tumor cells, including T cells, NK cells and natural killer T (NKT) cells, showed enhanced expression of certain chemokine receptors[35]. Consistently, overexpression of certain chemokine genes, such as CXCL10, CCL5 and CCL2, in HCC tissue correlated with Th1, cytolytic T lymphocyte (CTL) and NK cells and predicted a better prognosis[36]. Regulated release of chemokines or genetic modification of chemokine receptors in immune cells may enhance anti-tumor immune response. Unfortunately, only preclinical data can be found in this field.
Due to the great achievements in melanoma, immune checkpoint blockade therapy sheds light on other solid tumors, including HCC. Co-inhibitory signals diminish the intensity of anti-tumor response even although HCC specific antigen has presented with MHC receptors. To overcome these, immune checkpoints should be a promising approach to restore anti-tumor function of immune cells. Many immune checkpoints have been identified in the lab. In addition to PD-1 and CTLA-4 which have been intensively studied, there are also other potential checkpoints, like TIM-3, OX40, VISTA, LAG-3 and BTLA[37].
Anti-CTLA-4 antibody blocks the binding of CTLA-4 and CD80/86, which is a defunct antigen-presenting cell (APC) and results in suppressed anti-tumor immune responses mediated by T cells[38,39]. Basically, the efficacy of CTLA-4 blockade correlates with the immunogenicity of the tumor. A phase I trial of tremelimumab (anti-CTLA-4 monoclonal antibody) in HCC patients was reported in 2013[40]. The study enrolled 21 patients with advanced HCC not amenable to percutaneous ablation or TACE and showed that tremelimumab was well tolerated. Partial response was found in 17.6% of the patients and 45% of the cases had stable disease for more than 6 mo. Another phase I clinical trial of tremelimumab combined with RFA or TACE is now ongoing (NCT01853618; Table 1).
| Interventions | Design | Start year | Main inclusion criteria | Primary outcomes | Registered No. | Status |
| CP-675,206: anti-CTLA-4 antibody | Phase II | 2008 | Unresectable disease not amenable to loco regional treatment, HCV chronic infection | Tumor response | NCT01008358 | Completed |
| CT-011 (Pidilizumab): anti-PD-1 antibody | Phase I/II | 2009 | HCC not eligible for surgery, TACE, or other systematic treatments | Safety and tolerability | NCT00966251 | Terminated because of slow accrual |
| Nivolumab: anti-PD-1 antibody | Phase I | 2012 | Advanced HCC, failed in previous one line therapy | Adverse events | NCT01658878 | Recruiting |
| Tremelimumab: anti-CTLA-4 antibody, combined with TACE or RFA | Phase I | 2013 | Not amenable to curative resection, transplantation or ablation | Safety and feasibility | NCT01853618 | Recruiting |
Anti-PD-1 and anti-PD-L1 antibodies interfere with the binding of PD-1 and PD-L1/2 which inhibits T cell proliferation and cytokine release[41]. Although CTLA-4 and PD-1 were found predominantly expressed in T cells with anti-tumor function[42,43] and showed similar effects when used alone, different mechanisms and indicated patients regarding the two pathways were suggested by clinical observations[44]. Unfortunately, the phase I/II trial (NCT00966251) of a new PD-1 blockade CT-011 was terminated because of slow accrual. Another phase I trial of nivolumab (anti-PD-1 monoclonal antibody) is ongoing. This trial plans to recruit three cohorts of patients stratified by viral infection hepatitis C virus, hepatitis B virus (HBV) and no viral infection.
With the satisfactory effects in animal models, more and more trials are being conducted to investigate the role of immune checkpoint blockade therapy in HCC treatment (Table 1). Immune checkpoint blockade therapy is considered to be a strategy with a bright future and is cast into the limelight by oncologists worldwide.
Although prophylactic vaccines for HCC such as HBV vaccine contributed to the decrease of HCC patients[45], a therapeutic vaccine for HCC is still awaited. Numerous approaches have been investigated, seeking to trigger the host immune system to remove cancer cells. The most important constraint for progression of a tumor vaccine is the lack of tumor specific antigens or tumor associated antigens (TAA). With all the evolving understanding of tumor heterogeneity, it appears to be unprecedentedly challenging to exterminate cancer cells by a tumor vaccine alone. On the other hand, it could possibly be an effective approach to exhibit positive aspects in certain patients and play important roles in regimens.
Alpha-fetoprotein (AFP) and glypican-3 (GPC3) are two frequently used TAAs in HCC vaccines. GPC3 can be overexpressed in more than 80% of HCC and AFP can be positive in 60%-80% of HCC. Other HCC biomarkers that might be candidates for a vaccine involving antigens include squamous cell carcinoma antigen, heat shock protein 70, NY-ESO-1b, etc[46,47].
AFP is rare in healthy adults but can be highly expressed in HCC, making it an ideal target for anti-HCC immunotherapy. Actually, AFP is currently the most well studied target antigen for HCC immunotherapy. The earliest relevant clinical trial was more than a decade ago, testing AFP-specific T cell response to an AFP-derived peptide in six patients[48]. GPC3 (144-152) (FVGEFFTDV) and GPC3 (298-306) (EYILSLEEL) peptides were proven to induce specific CD8+ CTLs in HCC patients with HLA-A2 and HLA-A24 restriction, respectively[49]. Encouraged by this result, a phase II trial of a GPC3-based vaccine as adjuvant therapy for patients after surgery or RFA was registered and is now ongoing (UMIN-CTR: 000002614).
Proper design of epitopes with cross-recognition of wild-type antigens can enhance immune responses. To overcome the limitation of weak immune responses induced by native TAAs, Hong et al[50] created a highly immunogenic AFP via computer-guided methodical epitope optimization. This genetic modified AFP vaccine showed amazing anti-tumor effects in xenograft and diethylnitrosamine-induced mouse model of HCC by means of activating CD8+ T cells[50]. Polypeptide or fusion peptide was another method to amplify anti-tumor immune responses. A combination of full-length HBV core protein, HBV-X protein (HBx)52-60, HBx140-148, AFP158-166 and melanoma antigen gene-A271-279 is an example for a HCC vaccine[51]. Innovative creation of a fusion peptide containing different epitopes that involve multiple steps of the immune response was also proved to inhibit HCC in animals[52].
For a peptide-based tumor vaccine, the choice of peptide is critical for clinical response. Not all proteins that contribute to tumor progression are suitable for vaccine development. For instance, although expression and activity of telomerase was found up-regulated in most HCCs[53,54], telomerase peptide did not lead to any complete or partial responses in a phase II study on advanced HCC[55]. Additionally, the origin of the peptide affects induction efficiency of CLTs and consequent anti-tumor effects. Peptides originated from endogenously presented antigen are thought to be sparse on tumor cells and inefficient in inducing CTLs[56]. Intratumoral peptide injection was thus developed to enhance tumor cell antigenicity[56]; however, this needs further investigation.
A DNA-based vaccine assumes that DNA directly injected into the body undergoes transcription and translation in host cells and that the expressed peptide induces immune responses. Theoretically, all peptide vaccines can be transformed into DNA vaccines. AFP and GPC3 DNA vaccines were both developed and tested in the lab and showed tumor growth inhibition and survival improvement in mouse models[57-59]. In a recent small scale clinical observation, two HCC (stage II) patients after locoregional therapy underwent AFP DNA vaccine and adenovirus boost immunization. This approach was confirmed to be safe and well tolerated; however, both patients experienced HCC recurrence after a mere nine and eighteen months, respectively[60]. To our best information, no clinical trial regarding DNA vaccines on HCC has been reported or is currently ongoing.
APCs play a key role in anti-tumor function of immune responses. Dendritic cells (DCs) are the most potent APC and are closely related to HCC. Numerous studies have proved that DCs from peripheral blood and lymph nodes of HCC patients were decreased, with an immature phenotype and an impaired function[61-63]. A study revealed that the more DCs that were detected in HCC nodules, the better the prognosis would be. Infiltration of DCs in HCC nodules was strongly associated with the prognosis of HCC patients after surgical resection[64]. The composition of DCs in the hepatic lymph nodes of HCC patients was aberrant which may be one of the causes of the inadequate T cell response against HCC in these patients[65]. In addition, some tumor-derived factors, such as vascular endothelial growth factor, granulocyte macrophage colony-stimulating factor, IL-6 and IL-10, influence the differentiation, number and phenotype of DCs[66].
Given the importance in cancer development, DCs are increasingly applied to vaccination in various cancers, including HCC. A DC-based vaccine was reported to not only induce tumor antigen-specific CTLs[67], but also to activate NK cells and inhibit Tregs in HCC patients[51,68]. Logically, DCs pulsed with tumor tissue of an individual patient should be used. However, the tumor tissue is not always available. Therefore, peptides or cell line lysate was commonly used to substitute tumor tissue by many investigators. At least in HCC, this replacement strategy proved to be feasible and safe[69]. DCs infused with cancer cells, transfected with total RNA of cancer cells or transfected with designed plasmids were all able to mature and prime Th1 cells and CTLs[69-71].
Based upon these characteristics and success in preclinical studies, many clinical trials were carried out to evaluate the efficacy of DC-based immunotherapy to treat HCC patients. Two phase I studies showed immunization by tumor lysate pulsed DCs was feasible for end stage HCC patients[72,73]. Another clinical trial of a DC vaccine pulsed with autologous tumor lysate addressed that 12.9% of advanced HCC patients had partial response (PR) and 54.8% had SD[74]. Notably, a monthly boost vaccination resulted in a significantly better 1 year survival[74]. In another RCT on advanced HCC, DCs pulsed with HepG2 cell lysate resulted in 13.3% patients with PR and 60% with SD after 6 mo of treatment[75]. However, the proportion of clinical response with this therapy is relatively low. As an illustration, one phase II study using DCs pulsed with tumor lysate in HCC revealed only one out of 39 patients exhibited PR[76]. Furthermore, a phase I/II study using a multiple TAA-pulsed DC vaccine showed clinical response in only one out of five patients with advanced HCC[77]. Some studies also evaluated the efficacy of DC immunotherapy combined with local radiation[78] or TACE[79,80] but the results showed that DC infusion could not prevent HCC recurrence. Therefore, further studies are needed to increase the efficacy of this therapeutic approach. A new phase I trial on DC vaccine for HCC was registered last year and is now recruiting participants (NCT 01974661; Table 2).
| Interventions | Design | Start year | Main inclusion criteria | Primary outcomes | Registered No. | Status |
| AFP + GM-CSF plasmid prime and AFP adenoviral vector boost | Phase I/II | 2008 | Locoregionally treated HCC | Dose, toxicity, and immunological response rate | NCT00669136 | Terminated because of poor accrual |
| DC loaded with autologous tumor | Phase II | 2008 | Metastatic HCC, available of tumor tissue | 2-mo response rate | NCT00610389 | Unknown |
| DC loaded with specific peptides of AFP | Phase I/II | 2009 | Patients with previous treatment, AFP ≥ 40 ng/mL, HLA A 0201 group | Adverse events | NCT01128803 | Terminated |
| DEC-205-NY-ESO-1 fusion protein vaccine | Phase I | 2012 | After resection and TACE for HCC | Adverse events | NCT01522820 | Recruiting |
| COMBIG-DC: allogeneic DC cancer vaccine | Phase I | 2013 | Not eligible for curative treatment or TACE, BCLC stage B and C | Adverse events | NCT01974661 | Recruiting |
| In-situ therapeutic cancer vaccine | Phase I | 2013 | Refractory HCC, not eligible for or failed any treatment, AFP > 30 | Safety | NCT01923233 | Recruiting |
| V5 therapeutic vaccine | Phase III | 2014 | Advanced HCC | Changes in plasma AFP | NCT02232490 | Not yet recruiting |
Adoptive immunotherapy is now promising in the scenario of potential approaches for the treatment of solid tumors which are refractory to conventional therapies. An increasing amount of the literature discusses the efficacy of adoptive immunotherapy to control tumors. Meanwhile, many clinical trials have demonstrated that adoptive immunotherapy showed potentially promising anti-tumor effects on various cancers, including HCC.
First reported in the early 1980s, lymphokine-activated killer (LAK) cells are cytotoxic effector lymphocytes whose cytolytic activities are not restricted by MHC and are capable of killing tumor cells as well as NK-resistant tumor cell lines. There are actually constrained studies concerning the effectiveness of LAK cells for HCC treatment. A report stated that dealing with LAK cells cultivated by IL-2 reduced the recurrence rate in postoperative HCC patients[81]. Another study found that LAK cell-based immunotherapy was not an ideal adjuvant strategy after hepatic resection[82]. Results from a clinical trial indicated that tumor-specific CTL therapy is more effective than LAK cell therapy in advanced HCC patients[83]. The enthusiasm of study on LAK for HCC treatment has significantly declined over the last decade.
Cytokine-induced killer (CIK) cells exhibit potent, non-MHC restricted cytolytic activities against susceptible tumor cells of both autologous and allogeneic origins[84]. CIK cells are characterized by expression of both T cell biomarker CD3 and NK cell biomarker CD56[85] and can be generated from the human peripheral blood mononuclear cells (PBMC) induced by IFN-γ, anti-CD3 antibody and IL-2[84,86,87].
Compared with other immune cells, CIK cells possess some advantages. Firstly, CIK cells have a higher proliferation rate and can be obtained from cancer patients by in vitro culture[88]. Secondly, CIK cells have strong cytolytic activities and cover a broad spectrum of targeted tumors, including those that are insusceptible to LAK cells or NK cells[89]. Finally, CIK cells show minimal toxicity and do not cause graft-vs-host disease[84,90]. These merits make CIK cells a preferential adoptive immunotherapy for selected cancer patients[91].
Recently, escalating proof from clinical trials demonstrated that CIK cells adoptive transfer demonstrated a substantial anti-tumor effect in patients with solid tumors and hematological malignancies[92-94]. Some reports showed that CIK adjuvant immunotherapy significantly improved the outcomes of HCC patients[95-101]. In these studies, CIK cell transfusion reduced the relapse rate of HCC in patients after TACE and RFA therapy and prolonged the disease-free survival and OS for HCC patients after radical resection or TACE. A meta-analysis including 13 RCTs evaluated the efficacy of CIK immunotherapy in the treatment of HCC and revealed a significant superiority in prolonging the OS and progressive-free survival of patients[102]. Recently, another meta-analysis assessed the efficacy of CIK therapy after TACE or TACE plus RFA and showed that CIK therapy combined with TACE plus RFA treatment was associated with a higher 1 year recurrent free survival rate and 1, 2 year OS rates[103]. However, due to the limited number of patients in this field, the efficacy of CIK immunotherapy for HCC is still not convincing.
CIK cells are an encouraging tool within cancer adoptive immunotherapy but more basic research and clinical trials with high quality are urgently desired. To date, we have witnessed at least four ongoing registered clinical trials regarding CIK therapy for HCC (Table 3).
| Interventions | Design | Start year | Main inclusion criteria | Primary outcomes | Registered No. | Status |
| Immuncell-LC: activated T lymphocyte | Phase III | 2008 | Stage I and II, complete resection within 12 wk | Efficacy and safety | NCT00699816 | Completed |
| CIK | Phase III | 2008 | After radical resection of HCC, no prior anti-cancer therapy | Time to recurrence | NCT01749865 | Recruiting |
| CIK | Phase III | 2008 | After radical resection, no prior anti-cancer treatment | Time to recurrence | NCT00769106 | Recruiting |
| Ex vivo expanded autologous immune killer cell, combined with TACE | Phase II/III | 2009 | Never receive TACE treatment, BCLC stage B and C | 2-yr reduction of tumor cells | NCT01024530 | Unknown |
| NK cells, combined with liver transplantation | Phase I | 2010 | After liver transplantation for HCC | Side effect | NCT01147380 | Ongoing, but not recruiting |
| Young TIL | Phase II | 2010 | Metastatic HCC with at least one lesion resectable | Tumor regression rate | NCT01174121 | Recruiting |
| Autologous tumor infiltrating lymphocytes, combined with IL-2 | Phase I | 2011 | Metastatic HCC | Safety and tolerability | NCT01462903 | Unknown |
| CIK, combined with Licartin | Phase IV | 2012 | Postoperative patients | 1-yr PFS | NCT01758679 | Recruiting |
| Dendritic and cytokine-induced killer cells | Phase II | 2013 | After complete resection or TACE | PFS | NCT01821482 | Not yet recruiting |
| CTL induced by DC loaded with multiple antigens | Phase I | 2013 | Complete tumor resection within 8 wk | 2-yr PFS and adverse events | NCT02026362 | Recruiting |
| DC incubated with irradiated autologous tumor stem cells + GM-CSF | Phase I | 2013 | Candidates for HCC resection | Vital signs, physical examinations and adverse events | NCT01828762 | Completed |
| Cord blood-derived CIK | Phase I | 2013 | After radical resection | Adverse events | NCT01914263 | Not yet recruiting |
| Autologous NKT cells | Phase I | 2013 | Advanced HCC, refractory to standard treatments | Adverse events | NCT01801852 | Recruiting |
| Immuncell-LC, combined with Nexavar | Phase II | 2013 | Stage III and IV, receiving or ready for Nexavar treatment | 2-yr PFS | NCT01897610 | Recruiting |
| MG4101: ex vivo expanded allogeneic NK cell | Phase II | 2013 | Stage III, after curative resection | 1-yr DFS | NCT02008929 | Recruiting |
NK cells belong to the innate immune system, predominantly reside in the liver and play a critical role in the host defense against tumorigenesis[104,105]. Carcinogenesis is under close surveillance of NK cells and other members of immune system. In addition to the capability of killing tumor cells directly, NK cells are able to release immunomodulatory cytokines which can activate leukocytes of both the innate and adaptive immune system[105]. Unfortunately, cytotoxicity of NK cells was significantly inhibited in patients with advanced HCC[104]. In line with this, NK cells derived from HCC patients displayed a reduced cytotoxicity against HCC cell lines after stimulation with IL-2 in vivo[106]. The authors suggested that functional defects of NK cells might be responsible for the failure of anti-tumor immune responses. The NK cells from HCC patients were also impaired in their IFN-γ production and cytotoxicity and this functional impairment was found to be associated with increased Tregs[107]. Meanwhile, myeloid-derived suppressor cells inhibited NK cell cytotoxicity and cytokine secretion[108]. This evidence suggested that HCC patients could benefit from reactivation of NK cells. In a mouse model, administration of IL-12 and IL-18 increased NK cells in the liver and resulted in reduction of intrahepatic tumor nodules[109]. A similar outcome was obtained in additional research which established that activation of NK cells increased survival in a xenograft mouse model[110]. Thus, the approach of enhancing the function of NK cells could possibly be accomplished in human HCC treatment. Although much solid evidence showed the role of NK cells in an anti-tumor reaction, there are, however, insufficient clinical studies to corroborate the efficacy of NK cell immunotherapy in HCC. Recently, a study demonstrated that RFA could activate the peripheral blood circulating NK cells in HCC patients[111]. Two ongoing clinical trials are trying to assess NK cell therapy combined with liver resection (NCT02008929) or liver transplantation for HCC (NCT01147380; Table 3). In the future, it would be of great interest to investigate the efficacy of NK cells combined with other strategies to improve immunotherapy in HCC.
NK T (NKT) cells are a heterogeneous group of T cells with a range of characteristics different from conventional T cells. Human NKT cells are found in small numbers in healthy liver (0.5%) and blood (0.02%)[112,113]; however, they are critical players in the regulation of anti-tumor immunity[114-116]. NKT cells are best known for their immunosuppressive functions; however, they can interact with many other immune cells, such as DCs, macrophages as well as NK cells, and the outcome of NKT cell stimulation depends on these interactions and the cytokine milieu[115]. NKT cells can manifest anti-tumor effects mediated by their reactivation with exogenous cytokines or ligands but recently the natural role of NKT cells in anti-tumor immunity was reported[112,115]. A phase I trial using autologous NKT cells to treat advanced HCC is now ongoing (NCT01801852; Table 3). Further investigation is needed to elucidate the role of NKT cells in human HCC.
One of the most important aims of T cell engineering is to generate tumor-targeted T cells through the genetic transfer of antigen specific receptors. T cell engineering consists of either physiological, MHC-restricted T cell receptors (TCRs) or non-MHC-restricted chimeric antigen receptors. The conception of the chimeric antigen receptor (CAR) originally generated from the growing understanding of the barriers to effective immune therapy of various types of cancers. T cells armed with CARs (CAR-T cells) are able to recognize the cell surface antigens directly and are not blunted by tumor variations possessing lower surface expression of major MHC antigens which are considered a common mechanism of tumor immune escape[117,118]. The three basic elements of CAR are an extracellular antigen binding domain, a transmembrane domain and a cytoplasmic signaling domain.
To date, three generations of CARs have been developed. The first generation CAR only contains a T cell signaling domain that transmits the activation signal. It owns a feature of a single signaling domain most commonly derived from the CD3ζ component of the TCR/CD3 complex. The full activation of T cells needs multiple signals and it is obvious that the signaling from these first-generation CARs only provided the so-called “signal one” that could drive T cell effector functions; however, due to the absence of further signals, the T cells are unable to fully engage their effector machinery[119]. Considering this modular nature of the CAR, later designs aimed to add additional signaling domains which would increase the potency of the CARs and the consequent effector function of T cells. The second generation CARs incorporate a single co-stimulatory molecule endodomain, such as the endodomain of CD28 or 4-1BB[120]. The third generation CARs incorporate at least two co-stimulatory molecule endodomains, such as the endodomains of CD28 and 4-1BB[121]. Ligation of CD28 on CAR-T cells through the expression of B7 co-stimulatory ligands on target cells or co-expression of the CD28 molecule together with the scFv (specific monoclonal antibody) and CD3ζ domain of the CAR was shown to promote the proliferation of CAR-modified T cells and anti-tumor activity[122-124].
Plenty of studies have evaluated the efficacy of CAR-T cell treatment in hematological malignancies such as lymphoid leukemia[125,126] and acute myeloid leukemia[127,128]. Nonetheless, the clinical efficacy of CAR-T cells continues to be marginal in solid tumors compared to leukemia. So far, as we have acknowledged, there are no reported clinical trials evaluating the efficacy of CAR-T in HCC. Only one research stated that genetically modified T cells could be used to reconstitute virus-specific T cell immunity in chronic HBV patients and target tumors in HBV-related HCC. They found TCR re-directed HBV-specific T cells produced from PBMC of hepatitis B related HCC patients were capable of recognizing HCC tumor cells[129].
Besides the failed clinical trials of CAR-T cells in renal cell cancer and ovarian cancer[130-132], the safety and tolerance of CAR-T cells likewise need further assessment since the trial ceased and the demise of patients was often documented[124,133,134]. In a clinical trial of renal cell cancer, a maximum of 10 infusions of a total 0.2 to 2.1 × 109 CAR-T cells administered to patients manifested in liver enzyme disturbances and the treatment was finally stopped[133]. A patient receiving CAR-T cells based on a Her2/neu-specific CARs died soon after the treatment[124]. These data from renal carcinoma seemed to indicate a dim future for CAR-T applications in solid tumors. To achieve a better clinical response, CAR-T cells need to overcome two major barriers, which are insufficient T-cell migration into the lesions and highly immunosuppressive microenvironments within the tumors[118,135,136].
Considered collectively, the original studies highlighted two important lessons. To begin with, clinical anti-tumor responses of T cells seem to be proportional to T cell persistence. Consequently, we ought to enhance the T cell persistence and then try to efficiently traffic adequate quantities of CAR-T cells from the peripheral blood towards tumor tissues. Once there, these CAR-T cells must functionally respond against tumor cells within such an immunosuppressive context. Secondly, understanding and defining the specific target is crucial for the safety of CAR-T treatment. Increasing evidence demonstrates that a careful choice of target antigen, including an understanding of accessibility and expression level, must be under consideration for future CAR-T clinical trials.
Treg mediated immunosuppression is the essential mechanism accountable for tumor immune evasion and could be the primary hurdle of tumor immunotherapy[137]. Tregs, characterized by CD4+CD25+FoxP3+, play a critical role in immune homeostasis and suppress function of immune cells, such as CD8+ T cells, CD4+ T cells and NKT cells. Many reports have demonstrated that the amount of Tregs in solid tumor decreased and was inversely related to the prognosis of HCC patients[10,138-141].
Targeting Tregs has been of great interest to change the immune suppression milieu and enhance tumor specific immune responses. Depletion of Tregs using anti-CD25 monoclonal antibody has been shown to have substantial anti-tumor effects in murine tumor models[142]. Nonspecific approach of CD25+ T cell depletion by injection of PC61 antibody was also tested on an orthotropic HCC model and led to a significant protection against tumor development[143]. No such strategy has been performed on humans yet.
An array of translational research and pilot clinical trials have revealed that immunotherapy is safe and tolerated by patients with cancers. The efficacies are also offered in some types of immunotherapy in selected patients. In HCC, more studies, including basic and clinical research, are urgently required to improve the outcomes of immunotherapy with best cost performance. Currently, immune checkpoint blockade and CAR-T strategies are specifically expected. In addition, it is become obvious that incorporating standard anti-tumor therapies with immunotherapy is the most likely effective alternative. Ultimately, more substantial randomized, controlled trials are required to authenticate the efficacy of immunotherapy for HCC patients.
P- Reviewer: Lakatos PL, Petrovic D S- Editor: Song XX L- Editor: Roemmele A E- Editor: Liu SQ
| 1. | Lau WY. Primary liver tumors. Semin Surg Oncol. 2000;19:135-144. [PubMed] |
| 2. | Cancer IAfRo. GLOBOCAN 2012: estimated cancer incidence, motality and prevalence worldwide in 2012. [Accessed in 26 August, 2014]. Available from: http: //globocan.iarc.fr/Pages/fact_sheets_cancer.aspx. |
| 3. | Bruix J, Sherman M; American Association for the Study of Liver Diseases. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020-1022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5972] [Cited by in RCA: 6573] [Article Influence: 469.5] [Reference Citation Analysis (1)] |
| 4. | Greten TF, Manns MP, Korangy F. Immunotherapy of HCC. Rev Recent Clin Trials. 2008;3:31-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Flecken T, Spangenberg HC, Thimme R. Immunobiology of hepatocellular carcinoma. Langenbecks Arch Surg. 2012;397:673-680. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 6. | Miamen AG, Dong H, Roberts LR. Immunotherapeutic approaches to hepatocellular carcinoma treatment. Liver Cancer. 2012;1:226-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 7. | Budhu A, Wang XW. The role of cytokines in hepatocellular carcinoma. J Leukoc Biol. 2006;80:1197-1213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 200] [Cited by in RCA: 239] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 8. | Kuang DM, Zhao Q, Wu Y, Peng C, Wang J, Xu Z, Yin XY, Zheng L. Peritumoral neutrophils link inflammatory response to disease progression by fostering angiogenesis in hepatocellular carcinoma. J Hepatol. 2011;54:948-955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 346] [Cited by in RCA: 391] [Article Influence: 27.9] [Reference Citation Analysis (0)] |
| 9. | Zhang JP, Yan J, Xu J, Pang XH, Chen MS, Li L, Wu C, Li SP, Zheng L. Increased intratumoral IL-17-producing cells correlate with poor survival in hepatocellular carcinoma patients. J Hepatol. 2009;50:980-989. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 378] [Cited by in RCA: 418] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 10. | Fu J, Xu D, Liu Z, Shi M, Zhao P, Fu B, Zhang Z, Yang H, Zhang H, Zhou C. Increased regulatory T cells correlate with CD8 T-cell impairment and poor survival in hepatocellular carcinoma patients. Gastroenterology. 2007;132:2328-2339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 598] [Cited by in RCA: 693] [Article Influence: 38.5] [Reference Citation Analysis (0)] |
| 11. | Li T, Yang Y, Hua X, Wang G, Liu W, Jia C, Tai Y, Zhang Q, Chen G. Hepatocellular carcinoma-associated fibroblasts trigger NK cell dysfunction via PGE2 and IDO. Cancer Lett. 2012;318:154-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 261] [Cited by in RCA: 295] [Article Influence: 22.7] [Reference Citation Analysis (0)] |
| 12. | Herzer K, Hofmann TG, Teufel A, Schimanski CC, Moehler M, Kanzler S, Schulze-Bergkamen H, Galle PR. IFN-alpha-induced apoptosis in hepatocellular carcinoma involves promyelocytic leukemia protein and TRAIL independently of p53. Cancer Res. 2009;69:855-862. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 54] [Cited by in RCA: 69] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 13. | Li P, Du Q, Cao Z, Guo Z, Evankovich J, Yan W, Chang Y, Shao L, Stolz DB, Tsung A. Interferon-γ induces autophagy with growth inhibition and cell death in human hepatocellular carcinoma (HCC) cells through interferon-regulatory factor-1 (IRF-1). Cancer Lett. 2012;314:213-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 131] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 14. | Obora A, Shiratori Y, Okuno M, Adachi S, Takano Y, Matsushima-Nishiwaki R, Yasuda I, Yamada Y, Akita K, Sano T. Synergistic induction of apoptosis by acyclic retinoid and interferon-beta in human hepatocellular carcinoma cells. Hepatology. 2002;36:1115-1124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 15. | Damdinsuren B, Nagano H, Wada H, Kondo M, Ota H, Nakamura M, Noda T, Natsag J, Yamamoto H, Doki Y. Stronger growth-inhibitory effect of interferon (IFN)-beta compared to IFN-alpha is mediated by IFN signaling pathway in hepatocellular carcinoma cells. Int J Oncol. 2007;30:201-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 131] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 16. | Lai CL, Lau JY, Wu PC, Ngan H, Chung HT, Mitchell SJ, Corbett TJ, Chow AW, Lin HJ. Recombinant interferon-alpha in inoperable hepatocellular carcinoma: a randomized controlled trial. Hepatology. 1993;17:389-394. [PubMed] |
| 17. | Llovet JM, Sala M, Castells L, Suarez Y, Vilana R, Bianchi L, Ayuso C, Vargas V, Rodés J, Bruix J. Randomized controlled trial of interferon treatment for advanced hepatocellular carcinoma. Hepatology. 2000;31:54-58. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 188] [Cited by in RCA: 182] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 18. | Chen LT, Chen MF, Li LA, Lee PH, Jeng LB, Lin DY, Wu CC, Mok KT, Chen CL, Lee WC. Long-term results of a randomized, observation-controlled, phase III trial of adjuvant interferon Alfa-2b in hepatocellular carcinoma after curative resection. Ann Surg. 2012;255:8-17. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 19. | Yeo W, Mok TS, Zee B, Leung TW, Lai PB, Lau WY, Koh J, Mo FK, Yu SC, Chan AT. A randomized phase III study of doxorubicin versus cisplatin/interferon alpha-2b/doxorubicin/fluorouracil (PIAF) combination chemotherapy for unresectable hepatocellular carcinoma. J Natl Cancer Inst. 2005;97:1532-1538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 467] [Cited by in RCA: 455] [Article Influence: 22.8] [Reference Citation Analysis (0)] |
| 20. | Kasai K, Ushio A, Kasai Y, Sawara K, Miyamoto Y, Oikawa K, Kuroda H, Takikawa Y, Suzuki K. Therapeutic efficacy of combination therapy with intra-arterial 5-fluorouracil and systemic pegylated interferon α-2b for advanced hepatocellular carcinoma with portal venous invasion. Cancer. 2012;118:3302-3310. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 21. | Lee D, Chung YH, Kim JA, Park WH, Jin YJ, Shim JH, Ryu SH, Jang MK, Yu E, Lee YJ. Safety and efficacy of adjuvant pegylated interferon therapy for metastatic tumor antigen 1-positive hepatocellular carcinoma. Cancer. 2013;119:2239-2246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 22. | Li M, Lu C, Cheng J, Zhang J, Cao C, Xu J, Xu J, Pan H, Zhong B, Tucker S. Combination therapy with transarterial chemoembolization and interferon-alpha compared with transarterial chemoembolization alone for hepatitis B virus related unresectable hepatocellular carcinoma. J Gastroenterol Hepatol. 2009;24:1437-1444. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 23. | Shen YC, Hsu C, Chen LT, Cheng CC, Hu FC, Cheng AL. Adjuvant interferon therapy after curative therapy for hepatocellular carcinoma (HCC): a meta-regression approach. J Hepatol. 2010;52:889-894. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 115] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 24. | Wang J, He XD, Yao N, Liang WJ, Zhang YC. A meta-analysis of adjuvant therapy after potentially curative treatment for hepatocellular carcinoma. Can J Gastroenterol. 2013;27:351-363. [PubMed] |
| 25. | Wang L, Jia D, Duan F, Sun Z, Liu X, Zhou L, Sun L, Ren S, Ruan Y, Gu J. Combined anti-tumor effects of IFN-α and sorafenib on hepatocellular carcinoma in vitro and in vivo. Biochem Biophys Res Commun. 2012;422:687-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Perrillo R. Benefits and risks of interferon therapy for hepatitis B. Hepatology. 2009;49:S103-S111. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 189] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 27. | Xu G, Yang F, Ding CL, Wang J, Zhao P, Wang W, Ren H. MiR-221 accentuates IFN׳s anti-HCV effect by downregulating SOCS1 and SOCS3. Virology. 2014;462-463:343-350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 28. | Lygidakis NJ, Kosmidis P, Ziras N, Parissis J, Kyparidou E. Combined transarterial targeting locoregional immunotherapy-chemotherapy for patients with unresectable hepatocellular carcinoma: a new alternative for an old problem. J Interferon Cytokine Res. 1995;15:467-472. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Sangro B, Mazzolini G, Ruiz J, Herraiz M, Quiroga J, Herrero I, Benito A, Larrache J, Pueyo J, Subtil JC. Phase I trial of intratumoral injection of an adenovirus encoding interleukin-12 for advanced digestive tumors. J Clin Oncol. 2004;22:1389-1397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 230] [Cited by in RCA: 233] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 30. | Zhao JJ, Pan QZ, Pan K, Weng DS, Wang QJ, Li JJ, Lv L, Wang DD, Zheng HX, Jiang SS. Interleukin-37 mediates the antitumor activity in hepatocellular carcinoma: role for CD57+ NK cells. Sci Rep. 2014;4:5177. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 89] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 31. | Yuan L, Zhao H, Zhang L, Liu X. The efficacy of combination therapy using adeno-associated virus-mediated co-expression of apoptin and interleukin-24 on hepatocellular carcinoma. Tumour Biol. 2013;34:3027-3034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 32. | Marra F, Tacke F. Roles for chemokines in liver disease. Gastroenterology. 2014;147:577-594.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 507] [Cited by in RCA: 628] [Article Influence: 57.1] [Reference Citation Analysis (0)] |
| 33. | Ghanem I, Riveiro ME, Paradis V, Faivre S, de Parga PM, Raymond E. Insights on the CXCL12-CXCR4 axis in hepatocellular carcinoma carcinogenesis. Am J Transl Res. 2014;6:340-352. [PubMed] |
| 34. | Du D, Liu Y, Qian H, Zhang B, Tang X, Zhang T, Liu W. The effects of the CCR6/CCL20 biological axis on the invasion and metastasis of hepatocellular carcinoma. Int J Mol Sci. 2014;15:6441-6452. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 35. | Liu Y, Poon RT, Hughes J, Feng X, Yu WC, Fan ST. Chemokine receptors support infiltration of lymphocyte subpopulations in human hepatocellular carcinoma. Clin Immunol. 2005;114:174-182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 29] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 36. | Chew V, Chen J, Lee D, Loh E, Lee J, Lim KH, Weber A, Slankamenac K, Poon RT, Yang H. Chemokine-driven lymphocyte infiltration: an early intratumoural event determining long-term survival in resectable hepatocellular carcinoma. Gut. 2012;61:427-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 229] [Cited by in RCA: 278] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 37. | Hato T, Goyal L, Greten TF, Duda DG, Zhu AX. Immune checkpoint blockade in hepatocellular carcinoma: current progress and future directions. Hepatology. 2014;60:1776-1782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 188] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 38. | Grohmann U, Orabona C, Fallarino F, Vacca C, Calcinaro F, Falorni A, Candeloro P, Belladonna ML, Bianchi R, Fioretti MC. CTLA-4-Ig regulates tryptophan catabolism in vivo. Nat Immunol. 2002;3:1097-1101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 889] [Cited by in RCA: 882] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 39. | Schneider H, Downey J, Smith A, Zinselmeyer BH, Rush C, Brewer JM, Wei B, Hogg N, Garside P, Rudd CE. Reversal of the TCR stop signal by CTLA-4. Science. 2006;313:1972-1975. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 457] [Cited by in RCA: 481] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 40. | Sangro B, Gomez-Martin C, de la Mata M, Iñarrairaegui M, Garralda E, Barrera P, Riezu-Boj JI, Larrea E, Alfaro C, Sarobe P. A clinical trial of CTLA-4 blockade with tremelimumab in patients with hepatocellular carcinoma and chronic hepatitis C. J Hepatol. 2013;59:81-88. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 634] [Cited by in RCA: 751] [Article Influence: 62.6] [Reference Citation Analysis (0)] |
| 41. | Okazaki T, Maeda A, Nishimura H, Kurosaki T, Honjo T. PD-1 immunoreceptor inhibits B cell receptor-mediated signaling by recruiting src homology 2-domain-containing tyrosine phosphatase 2 to phosphotyrosine. Proc Natl Acad Sci USA. 2001;98:13866-13871. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 590] [Cited by in RCA: 655] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 42. | Manzotti CN, Liu MK, Burke F, Dussably L, Zheng Y, Sansom DM. Integration of CD28 and CTLA-4 function results in differential responses of T cells to CD80 and CD86. Eur J Immunol. 2006;36:1413-1422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 43. | Francisco LM, Salinas VH, Brown KE, Vanguri VK, Freeman GJ, Kuchroo VK, Sharpe AH. PD-L1 regulates the development, maintenance, and function of induced regulatory T cells. J Exp Med. 2009;206:3015-3029. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1346] [Cited by in RCA: 1623] [Article Influence: 101.4] [Reference Citation Analysis (0)] |
| 44. | Hamid O, Robert C, Daud A, Hodi FS, Hwu WJ, Kefford R, Wolchok JD, Hersey P, Joseph RW, Weber JS. Safety and tumor responses with lambrolizumab (anti-PD-1) in melanoma. N Engl J Med. 2013;369:134-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2712] [Cited by in RCA: 2725] [Article Influence: 227.1] [Reference Citation Analysis (0)] |
| 45. | Chang MH. Cancer prevention by vaccination against hepatitis B. Recent Results Cancer Res. 2009;181:85-94. [PubMed] |
| 46. | Zhao YJ, Ju Q, Li GC. Tumor markers for hepatocellular carcinoma. Mol Clin Oncol. 2013;1:593-598. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 157] [Cited by in RCA: 174] [Article Influence: 14.5] [Reference Citation Analysis (0)] |
| 47. | Shang XY, Chen HS, Zhang HG, Pang XW, Qiao H, Peng JR, Qin LL, Fei R, Mei MH, Leng XS. The spontaneous CD8+ T-cell response to HLA-A2-restricted NY-ESO-1b peptide in hepatocellular carcinoma patients. Clin Cancer Res. 2004;10:6946-6955. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 57] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 48. | Butterfield LH, Ribas A, Meng WS, Dissette VB, Amarnani S, Vu HT, Seja E, Todd K, Glaspy JA, McBride WH. T-cell responses to HLA-A*0201 immunodominant peptides derived from alpha-fetoprotein in patients with hepatocellular cancer. Clin Cancer Res. 2003;9:5902-5908. [PubMed] |
| 49. | Komori H, Nakatsura T, Senju S, Yoshitake Y, Motomura Y, Ikuta Y, Fukuma D, Yokomine K, Harao M, Beppu T. Identification of HLA-A2- or HLA-A24-restricted CTL epitopes possibly useful for glypican-3-specific immunotherapy of hepatocellular carcinoma. Clin Cancer Res. 2006;12:2689-2697. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 134] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 50. | Hong Y, Peng Y, Guo ZS, Guevara-Patino J, Pang J, Butterfield LH, Mivechi NF, Munn DH, Bartlett DL, He Y. Epitope-optimized alpha-fetoprotein genetic vaccines prevent carcinogen-induced murine autochthonous hepatocellular carcinoma. Hepatology. 2014;59:1448-1458. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 51. | Chen Y, Yang D, Li S, Gao Y, Jiang R, Deng L, Frankel FR, Sun B. Development of a Listeria monocytogenes-based vaccine against hepatocellular carcinoma. Oncogene. 2012;31:2140-2152. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 38] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 52. | Zhang Y, Xu J, Zhao R, Liu J, Wu J. Inhibition effects on liver tumors of BALB/c mice bearing H22 cells by immunization with a recombinant immunogen of GnRH linked to heat shock protein 65. Vaccine. 2007;25:6911-6921. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 53. | Saini N, Srinivasan R, Chawla Y, Sharma S, Chakraborti A, Rajwanshi A. Telomerase activity, telomere length and human telomerase reverse transcriptase expression in hepatocellular carcinoma is independent of hepatitis virus status. Liver Int. 2009;29:1162-1170. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 45] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 54. | Shimada M, Hasegawa H, Gion T, Utsunomiya T, Shirabe K, Takenaka K, Otsuka T, Maehara Y, Sugimachi K. The role of telomerase activity in hepatocellular carcinoma. Am J Gastroenterol. 2000;95:748-752. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 23] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 55. | Greten TF, Forner A, Korangy F, N’Kontchou G, Barget N, Ayuso C, Ormandy LA, Manns MP, Beaugrand M, Bruix J. A phase II open label trial evaluating safety and efficacy of a telomerase peptide vaccination in patients with advanced hepatocellular carcinoma. BMC Cancer. 2010;10:209. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 138] [Cited by in RCA: 163] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 56. | Nobuoka D, Yoshikawa T, Sawada Y, Fujiwara T, Nakatsura T. Peptide vaccines for hepatocellular carcinoma. Hum Vaccin Immunother. 2013;9:210-212. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 57. | Lan YH, Li YG, Liang ZW, Chen M, Peng ML, Tang L, Hu HD, Ren H. A DNA vaccine against chimeric AFP enhanced by HSP70 suppresses growth of hepatocellular carcinoma. Cancer Immunol Immunother. 2007;56:1009-1016. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 26] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 58. | Li SQ, Lin J, Qi CY, Fu SJ, Xiao WK, Peng BG, Liang LJ. GPC3 DNA vaccine elicits potent cellular antitumor immunity against HCC in mice. Hepatogastroenterology. 2014;61:278-284. [PubMed] |
| 59. | Morozov AV, Morozov VA, Astakhova TM, Timofeev AV, Karpov VL. [DNA vaccine encoding alpha-fetoprotein with fused ornithine decarboxylase degradation signal significantly suppresses hepatocellular carcinoma growth in mice]. Mol Biol (Mosk). 2012;46:434-451. [PubMed] |
| 60. | Butterfield LH, Economou JS, Gamblin TC, Geller DA. Alpha fetoprotein DNA prime and adenovirus boost immunization of two hepatocellular cancer patients. J Transl Med. 2014;12:86. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 61. | Kakumu S, Ito S, Ishikawa T, Mita Y, Tagaya T, Fukuzawa Y, Yoshioka K. Decreased function of peripheral blood dendritic cells in patients with hepatocellular carcinoma with hepatitis B and C virus infection. J Gastroenterol Hepatol. 2000;15:431-436. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 83] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 62. | Ormandy LA, Farber A, Cantz T, Petrykowska S, Wedemeyer H, Horning M, Lehner F, Manns MP, Korangy F, Greten TF. Direct ex vivo analysis of dendritic cells in patients with hepatocellular carcinoma. World J Gastroenterol. 2006;12:3275-3282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 73] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 63. | Ninomiya T, Akbar SM, Masumoto T, Horiike N, Onji M. Dendritic cells with immature phenotype and defective function in the peripheral blood from patients with hepatocellular carcinoma. J Hepatol. 1999;31:323-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 104] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 64. | Cai XY, Gao Q, Qiu SJ, Ye SL, Wu ZQ, Fan J, Tang ZY. Dendritic cell infiltration and prognosis of human hepatocellular carcinoma. J Cancer Res Clin Oncol. 2006;132:293-301. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 81] [Cited by in RCA: 72] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 65. | Tang TJ, Vukosavljevic D, Janssen HL, Binda RS, Mancham S, Tilanus HW, Ijzermans JN, Drexhage H, Kwekkeboom J. Aberrant composition of the dendritic cell population in hepatic lymph nodes of patients with hepatocellular carcinoma. Hum Pathol. 2006;37:332-338. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 16] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 66. | Gabrilovich D. Mechanisms and functional significance of tumour-induced dendritic-cell defects. Nat Rev Immunol. 2004;4:941-952. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 754] [Cited by in RCA: 749] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 67. | Sun JC, Pan K, Chen MS, Wang QJ, Wang H, Ma HQ, Li YQ, Liang XT, Li JJ, Zhao JJ. Dendritic cells-mediated CTLs targeting hepatocellular carcinoma stem cells. Cancer Biol Ther. 2010;10:368-375. [PubMed] |
| 68. | Bray SM, Vujanovic L, Butterfield LH. Dendritic cell-based vaccines positively impact natural killer and regulatory T cells in hepatocellular carcinoma patients. Clin Dev Immunol. 2011;2011:249281. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 69. | Cao DY, Yang JY, Yue SQ, Tao KS, Song ZS, Wang DS, Yang YL, Dou KF. Comparative analysis of DC fused with allogeneic hepatocellular carcinoma cell line HepG2 and autologous tumor cells as potential cancer vaccines against hepatocellular carcinoma. Cell Immunol. 2009;259:13-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 70. | Zhang HM, Zhang LW, Liu WC, Cheng J, Si XM, Ren J. Comparative analysis of DC fused with tumor cells or transfected with tumor total RNA as potential cancer vaccines against hepatocellular carcinoma. Cytotherapy. 2006;8:580-588. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 71. | Homma S, Komita H, Sagawa Y, Ohno T, Toda G. Antitumour activity mediated by CD4+ cytotoxic T lymphocytes against MHC class II-negative mouse hepatocellular carcinoma induced by dendritic cell vaccine and interleukin-12. Immunology. 2005;115:451-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 72. | Iwashita Y, Tahara K, Goto S, Sasaki A, Kai S, Seike M, Chen CL, Kawano K, Kitano S. A phase I study of autologous dendritic cell-based immunotherapy for patients with unresectable primary liver cancer. Cancer Immunol Immunother. 2003;52:155-161. [PubMed] |
| 73. | Ladhams A, Schmidt C, Sing G, Butterworth L, Fielding G, Tesar P, Strong R, Leggett B, Powell L, Maddern G. Treatment of non-resectable hepatocellular carcinoma with autologous tumor-pulsed dendritic cells. J Gastroenterol Hepatol. 2002;17:889-896. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 74. | Lee WC, Wang HC, Hung CF, Huang PF, Lia CR, Chen MF. Vaccination of advanced hepatocellular carcinoma patients with tumor lysate-pulsed dendritic cells: a clinical trial. J Immunother. 2005;28:496-504. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 141] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 75. | El Ansary M, Mogawer S, Elhamid SA, Alwakil S, Aboelkasem F, Sabaawy HE, Abdelhalim O. Immunotherapy by autologous dendritic cell vaccine in patients with advanced HCC. J Cancer Res Clin Oncol. 2013;139:39-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 78] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 76. | Palmer DH, Midgley RS, Mirza N, Torr EE, Ahmed F, Steele JC, Steven NM, Kerr DJ, Young LS, Adams DH. A phase II study of adoptive immunotherapy using dendritic cells pulsed with tumor lysate in patients with hepatocellular carcinoma. Hepatology. 2009;49:124-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 215] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 77. | Tada F, Abe M, Hirooka M, Ikeda Y, Hiasa Y, Lee Y, Jung NC, Lee WB, Lee HS, Bae YS. Phase I/II study of immunotherapy using tumor antigen-pulsed dendritic cells in patients with hepatocellular carcinoma. Int J Oncol. 2012;41:1601-1609. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 92] [Cited by in RCA: 95] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 78. | Chi KH, Liu SJ, Li CP, Kuo HP, Wang YS, Chao Y, Hsieh SL. Combination of conformal radiotherapy and intratumoral injection of adoptive dendritic cell immunotherapy in refractory hepatoma. J Immunother. 2005;28:129-135. [PubMed] |
| 79. | Nakamoto Y, Mizukoshi E, Tsuji H, Sakai Y, Kitahara M, Arai K, Yamashita T, Yokoyama K, Mukaida N, Matsushima K. Combined therapy of transcatheter hepatic arterial embolization with intratumoral dendritic cell infusion for hepatocellular carcinoma: clinical safety. Clin Exp Immunol. 2007;147:296-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 42] [Article Influence: 2.3] [Reference Citation Analysis (1)] |
| 80. | Mizukoshi E, Nakamoto Y, Arai K, Yamashita T, Mukaida N, Matsushima K, Matsui O, Kaneko S. Enhancement of tumor-specific T-cell responses by transcatheter arterial embolization with dendritic cell infusion for hepatocellular carcinoma. Int J Cancer. 2010;126:2164-2174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 22] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 81. | Une Y, Kawata A, Uchino J. [Adopted immunochemotherapy using IL-2 and spleen LAK cell--randomized study]. Nihon Geka Gakkai Zasshi. 1991;92:1330-1333. [PubMed] |
| 82. | Kawata A, Une Y, Hosokawa M, Wakizaka Y, Namieno T, Uchino J, Kobayashi H. Adjuvant chemoimmunotherapy for hepatocellular carcinoma patients. Adriamycin, interleukin-2, and lymphokine-activated killer cells versus adriamycin alone. Am J Clin Oncol. 1995;18:257-262. [PubMed] |
| 83. | Haruta I, Yamauchi K, Aruga A, Komatsu T, Takasaki K, Hayashi N, Hanyu F. Analytical study of the clinical response to two distinct adoptive immunotherapies for advanced hepatocellular carcinoma: comparison between LAK cell and CTL therapy. J Immunother Emphasis Tumor Immunol. 1996;19:218-223. [PubMed] |
| 84. | Introna M, Golay J, Rambaldi A. Cytokine Induced Killer (CIK) cells for the treatment of haematological neoplasms. Immunol Lett. 2013;155:27-30. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 85. | Linn YC, Hui KM. Cytokine-induced killer cells: NK-like T cells with cytotolytic specificity against leukemia. Leuk Lymphoma. 2003;44:1457-1462. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 42] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 86. | Schmidt-Wolf IG, Negrin RS, Kiem HP, Blume KG, Weissman IL. Use of a SCID mouse/human lymphoma model to evaluate cytokine-induced killer cells with potent antitumor cell activity. J Exp Med. 1991;174:139-149. [PubMed] |
| 87. | Zoll B, Lefterova P, Csipai M, Finke S, Trojaneck B, Ebert O, Micka B, Roigk K, Fehlinger M, Schmidt-Wolf GD. Generation of cytokine-induced killer cells using exogenous interleukin-2, -7 or -12. Cancer Immunol Immunother. 1998;47:221-226. [PubMed] |
| 88. | Alvarnas JC, Linn YC, Hope EG, Negrin RS. Expansion of cytotoxic CD3+ CD56+ cells from peripheral blood progenitor cells of patients undergoing autologous hematopoietic cell transplantation. Biol Blood Marrow Transplant. 2001;7:216-222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 69] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 89. | Jiang J, Wu C, Lu B. Cytokine-induced killer cells promote antitumor immunity. J Transl Med. 2013;11:83. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 103] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 90. | Nishimura R, Baker J, Beilhack A, Zeiser R, Olson JA, Sega EI, Karimi M, Negrin RS. In vivo trafficking and survival of cytokine-induced killer cells resulting in minimal GVHD with retention of antitumor activity. Blood. 2008;112:2563-2574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 162] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 91. | Mesiano G, Todorovic M, Gammaitoni L, Leuci V, Giraudo Diego L, Carnevale-Schianca F, Fagioli F, Piacibello W, Aglietta M, Sangiolo D. Cytokine-induced killer (CIK) cells as feasible and effective adoptive immunotherapy for the treatment of solid tumors. Expert Opin Biol Ther. 2012;12:673-684. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 112] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 92. | Sangiolo D. Cytokine induced killer cells as promising immunotherapy for solid tumors. J Cancer. 2011;2:363-368. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 102] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 93. | Sangiolo D, Mesiano G, Carnevale-Schianca F, Piacibello W, Aglietta M, Cignetti A. Cytokine induced killer cells as adoptive immunotherapy strategy to augment graft versus tumor after hematopoietic cell transplantation. Expert Opin Biol Ther. 2009;9:831-840. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 43] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 94. | Ma Y, Zhang Z, Tang L, Xu YC, Xie ZM, Gu XF, Wang HX. Cytokine-induced killer cells in the treatment of patients with solid carcinomas: a systematic review and pooled analysis. Cytotherapy. 2012;14:483-493. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 56] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 95. | Weng DS, Zhou J, Zhou QM, Zhao M, Wang QJ, Huang LX, Li YQ, Chen SP, Wu PH, Xia JC. Minimally invasive treatment combined with cytokine-induced killer cells therapy lower the short-term recurrence rates of hepatocellular carcinomas. J Immunother. 2008;31:63-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 120] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 96. | Hui D, Qiang L, Jian W, Ti Z, Da-Lu K. A randomized, controlled trial of postoperative adjuvant cytokine-induced killer cells immunotherapy after radical resection of hepatocellular carcinoma. Digestive and Liver Disease. 2009;41:36-41. [RCA] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 97. | Hao MZ, Lin HL, Chen Q, Ye YB, Chen QZ, Chen MS. Efficacy of transcatheter arterial chemoembolization combined with cytokine-induced killer cell therapy on hepatocellular carcinoma: a comparative study. Chin J Cancer. 2010;29:172-177. [PubMed] |
| 98. | Zhou P, Liang P, Dong B, Yu X, Han Z, Xu Y. Phase I clinical study of combination therapy with microwave ablation and cellular immunotherapy in hepatocellular carcinoma. Cancer Biol Ther. 2011;11:450-456. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 48] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 99. | Huang ZM, Li W, Li S, Gao F, Zhou QM, Wu FM, He N, Pan CC, Xia JC, Wu PH. Cytokine-induced killer cells in combination with transcatheter arterial chemoembolization and radiofrequency ablation for hepatocellular carcinoma patients. J Immunother. 2013;36:287-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 58] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 100. | Pan K, Li YQ, Wang W, Xu L, Zhang YJ, Zheng HX, Zhao JJ, Qiu HJ, Weng DS, Li JJ. The efficacy of cytokine-induced killer cell infusion as an adjuvant therapy for postoperative hepatocellular carcinoma patients. Ann Surg Oncol. 2013;20:4305-4311. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 101. | Wang FS, Liu MX, Zhang B, Shi M, Lei ZY, Sun WB, Du QY, Chen JM. Antitumor activities of human autologous cytokine-induced killer (CIK) cells against hepatocellular carcinoma cells in vitro and in vivo. World J Gastroenterol. 2002;8:464-468. [PubMed] |
| 102. | Ma Y, Xu YC, Tang L, Zhang Z, Wang J, Wang HX. Cytokine-induced killer (CIK) cell therapy for patients with hepatocellular carcinoma: efficacy and safety. Exp Hematol Oncol. 2012;1:11. [PubMed] |
| 103. | Li X, Dai D, Song X, Liu J, Zhu L, Xu W. A meta-analysis of cytokine-induced killer cells therapy in combination with minimally invasive treatment for hepatocellular carcinoma. Clin Res Hepatol Gastroenterol. 2014;38:583-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 31] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 104. | Jinushi M, Takehara T, Tatsumi T, Hiramatsu N, Sakamori R, Yamaguchi S, Hayashi N. Impairment of natural killer cell and dendritic cell functions by the soluble form of MHC class I-related chain A in advanced human hepatocellular carcinomas. J Hepatol. 2005;43:1013-1020. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 164] [Cited by in RCA: 178] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 105. | Smyth MJ, Hayakawa Y, Takeda K, Yagita H. New aspects of natural-killer-cell surveillance and therapy of cancer. Nat Rev Cancer. 2002;2:850-861. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 536] [Cited by in RCA: 553] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 106. | Ishiyama K, Ohdan H, Ohira M, Mitsuta H, Arihiro K, Asahara T. Difference in cytotoxicity against hepatocellular carcinoma between liver and periphery natural killer cells in humans. Hepatology. 2006;43:362-372. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 126] [Cited by in RCA: 147] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 107. | Cai L, Zhang Z, Zhou L, Wang H, Fu J, Zhang S, Shi M, Zhang H, Yang Y, Wu H. Functional impairment in circulating and intrahepatic NK cells and relative mechanism in hepatocellular carcinoma patients. Clin Immunol. 2008;129:428-437. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 195] [Cited by in RCA: 229] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 108. | Hoechst B, Voigtlaender T, Ormandy L, Gamrekelashvili J, Zhao F, Wedemeyer H, Lehner F, Manns MP, Greten TF, Korangy F. Myeloid derived suppressor cells inhibit natural killer cells in patients with hepatocellular carcinoma via the NKp30 receptor. Hepatology. 2009;50:799-807. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 533] [Cited by in RCA: 527] [Article Influence: 32.9] [Reference Citation Analysis (0)] |
| 109. | Subleski JJ, Hall VL, Back TC, Ortaldo JR, Wiltrout RH. Enhanced antitumor response by divergent modulation of natural killer and natural killer T cells in the liver. Cancer Res. 2006;66:11005-11012. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 71] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 110. | Tsuchiyama T, Nakamoto Y, Sakai Y, Marukawa Y, Kitahara M, Mukaida N, Kaneko S. Prolonged, NK cell-mediated antitumor effects of suicide gene therapy combined with monocyte chemoattractant protein-1 against hepatocellular carcinoma. J Immunol. 2007;178:574-583. [PubMed] |
| 111. | Zerbini A, Pilli M, Laccabue D, Pelosi G, Molinari A, Negri E, Cerioni S, Fagnoni F, Soliani P, Ferrari C. Radiofrequency thermal ablation for hepatocellular carcinoma stimulates autologous NK-cell response. Gastroenterology. 2010;138:1931-1942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 135] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 112. | Bendelac A, Savage PB, Teyton L. The biology of NKT cells. Annu Rev Immunol. 2007;25:297-336. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1664] [Cited by in RCA: 1771] [Article Influence: 98.4] [Reference Citation Analysis (0)] |
| 113. | Kenna T, Golden-Mason L, Porcelli SA, Koezuka Y, Hegarty JE, O’Farrelly C, Doherty DG. NKT cells from normal and tumor-bearing human livers are phenotypically and functionally distinct from murine NKT cells. J Immunol. 2003;171:1775-1779. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 146] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 114. | Berzofsky JA, Terabe M. NKT cells in tumor immunity: opposing subsets define a new immunoregulatory axis. J Immunol. 2008;180:3627-3635. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 90] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 115. | Smyth MJ, Crowe NY, Hayakawa Y, Takeda K, Yagita H, Godfrey DI. NKT cells - conductors of tumor immunity? Curr Opin Immunol. 2002;14:165-171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 224] [Cited by in RCA: 204] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 116. | Godfrey DI, MacDonald HR, Kronenberg M, Smyth MJ, Van Kaer L. NKT cells: what’s in a name? Nat Rev Immunol. 2004;4:231-237. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 882] [Cited by in RCA: 954] [Article Influence: 45.4] [Reference Citation Analysis (0)] |
| 117. | Sadelain M, Brentjens R, Rivière I. The promise and potential pitfalls of chimeric antigen receptors. Curr Opin Immunol. 2009;21:215-223. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 328] [Cited by in RCA: 374] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 118. | Gilham DE, Debets R, Pule M, Hawkins RE, Abken H. CAR-T cells and solid tumors: tuning T cells to challenge an inveterate foe. Trends Mol Med. 2012;18:377-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 144] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 119. | Curran KJ, Pegram HJ, Brentjens RJ. Chimeric antigen receptors for T cell immunotherapy: current understanding and future directions. J Gene Med. 2012;14:405-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 184] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 120. | Lipowska-Bhalla G, Gilham DE, Hawkins RE, Rothwell DG. Targeted immunotherapy of cancer with CAR T cells: achievements and challenges. Cancer Immunol Immunother. 2012;61:953-962. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 79] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 121. | Dotti G, Savoldo B, Brenner M. Fifteen years of gene therapy based on chimeric antigen receptors: “are we nearly there yet?”. Hum Gene Ther. 2009;20:1229-1239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 70] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 122. | Savoldo B, Ramos CA, Liu E, Mims MP, Keating MJ, Carrum G, Kamble RT, Bollard CM, Gee AP, Mei Z. CD28 costimulation improves expansion and persistence of chimeric antigen receptor-modified T cells in lymphoma patients. J Clin Invest. 2011;121:1822-1826. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 837] [Cited by in RCA: 821] [Article Influence: 58.6] [Reference Citation Analysis (0)] |
| 123. | Morgan RA, Yang JC, Kitano M, Dudley ME, Laurencot CM, Rosenberg SA. Case report of a serious adverse event following the administration of T cells transduced with a chimeric antigen receptor recognizing ERBB2. Mol Ther. 2010;18:843-851. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1626] [Cited by in RCA: 1972] [Article Influence: 131.5] [Reference Citation Analysis (0)] |
| 124. | Kowolik CM, Topp MS, Gonzalez S, Pfeiffer T, Olivares S, Gonzalez N, Smith DD, Forman SJ, Jensen MC, Cooper LJ. CD28 costimulation provided through a CD19-specific chimeric antigen receptor enhances in vivo persistence and antitumor efficacy of adoptively transferred T cells. Cancer Res. 2006;66:10995-11004. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 357] [Cited by in RCA: 400] [Article Influence: 21.1] [Reference Citation Analysis (0)] |
| 125. | Porter DL, Levine BL, Kalos M, Bagg A, June CH. Chimeric antigen receptor-modified T cells in chronic lymphoid leukemia. N Engl J Med. 2011;365:725-733. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2975] [Cited by in RCA: 2799] [Article Influence: 199.9] [Reference Citation Analysis (0)] |
| 126. | Grupp SA, Kalos M, Barrett D, Aplenc R, Porter DL, Rheingold SR, Teachey DT, Chew A, Hauck B, Wright JF. Chimeric antigen receptor-modified T cells for acute lymphoid leukemia. N Engl J Med. 2013;368:1509-1518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2465] [Cited by in RCA: 2760] [Article Influence: 230.0] [Reference Citation Analysis (0)] |
| 127. | Pizzitola I, Anjos-Afonso F, Rouault-Pierre K, Lassailly F, Tettamanti S, Spinelli O, Biondi A, Biagi E, Bonnet D. Chimeric antigen receptors against CD33/CD123 antigens efficiently target primary acute myeloid leukemia cells in vivo. Leukemia. 2014;28:1596-1605. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 229] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 128. | June C, Rosenberg SA, Sadelain M, Weber JS. T-cell therapy at the threshold. Nat Biotechnol. 2012;30:611-614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 48] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 129. | Gehring AJ, Xue SA, Ho ZZ, Teoh D, Ruedl C, Chia A, Koh S, Lim SG, Maini MK, Stauss H. Engineering virus-specific T cells that target HBV infected hepatocytes and hepatocellular carcinoma cell lines. J Hepatol. 2011;55:103-110. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 177] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 130. | Kershaw MH, Westwood JA, Parker LL, Wang G, Eshhar Z, Mavroukakis SA, White DE, Wunderlich JR, Canevari S, Rogers-Freezer L. A phase I study on adoptive immunotherapy using gene-modified T cells for ovarian cancer. Clin Cancer Res. 2006;12:6106-6115. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1006] [Cited by in RCA: 968] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 131. | Lamers CH, Sleijfer S, Vulto AG, Kruit WH, Kliffen M, Debets R, Gratama JW, Stoter G, Oosterwijk E. Treatment of metastatic renal cell carcinoma with autologous T-lymphocytes genetically retargeted against carbonic anhydrase IX: first clinical experience. J Clin Oncol. 2006;24:e20-e22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 640] [Cited by in RCA: 712] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 132. | Lamers CH, Langeveld SC, Groot-van Ruijven CM, Debets R, Sleijfer S, Gratama JW. Gene-modified T cells for adoptive immunotherapy of renal cell cancer maintain transgene-specific immune functions in vivo. Cancer Immunol Immunother. 2007;56:1875-1883. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 78] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 133. | Lamers CH, Sleijfer S, van Steenbergen S, van Elzakker P, van Krimpen B, Groot C, Vulto A, den Bakker M, Oosterwijk E, Debets R. Treatment of metastatic renal cell carcinoma with CAIX CAR-engineered T cells: clinical evaluation and management of on-target toxicity. Mol Ther. 2013;21:904-912. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 460] [Cited by in RCA: 551] [Article Influence: 45.9] [Reference Citation Analysis (0)] |
| 134. | Brentjens R, Yeh R, Bernal Y, Riviere I, Sadelain M. Treatment of chronic lymphocytic leukemia with genetically targeted autologous T cells: case report of an unforeseen adverse event in a phase I clinical trial. Mol Ther. 2010;18:666-668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 291] [Cited by in RCA: 312] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 135. | Disis ML. Immune regulation of cancer. J Clin Oncol. 2010;28:4531-4538. |
| 136. | Whiteside TL. Immune responses to malignancies. J Allergy Clin Immun. 2010;125:S272-S283. [RCA] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 134] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 137. | Sakaguchi S, Powrie F. Emerging challenges in regulatory T cell function and biology. Science. 2007;317:627-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 185] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 138. | Zhou J, Ding T, Pan W, Zhu LY, Li L, Zheng L. Increased intratumoral regulatory T cells are related to intratumoral macrophages and poor prognosis in hepatocellular carcinoma patients. Int J Cancer. 2009;125:1640-1648. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 193] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 139. | Kobayashi N, Hiraoka N, Yamagami W, Ojima H, Kanai Y, Kosuge T, Nakajima A, Hirohashi S. FOXP3+ regulatory T cells affect the development and progression of hepatocarcinogenesis. Clin Cancer Res. 2007;13:902-911. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 315] [Cited by in RCA: 323] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 140. | Gao Q, Qiu SJ, Fan J, Zhou J, Wang XY, Xiao YS, Xu Y, Li YW, Tang ZY. Intratumoral balance of regulatory and cytotoxic T cells is associated with prognosis of hepatocellular carcinoma after resection. J Clin Oncol. 2007;25:2586-2593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 787] [Cited by in RCA: 892] [Article Influence: 49.6] [Reference Citation Analysis (0)] |
| 141. | Huang Y, Wang FM, Wang T, Wang YJ, Zhu ZY, Gao YT, Du Z. Tumor-infiltrating FoxP3+ Tregs and CD8+ T cells affect the prognosis of hepatocellular carcinoma patients. Digestion. 2012;86:329-337. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 78] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 142. | Shimizu J, Yamazaki S, Sakaguchi S. Induction of tumor immunity by removing CD25+CD4+ T cells: a common basis between tumor immunity and autoimmunity. J Immunol. 1999;163:5211-5218. [PubMed] |
| 143. | Cany J, Tran L, Gauttier V, Judor JP, Vassaux G, Ferry N, Conchon S. Immunotherapy of hepatocellular carcinoma: is there a place for regulatory T-lymphocyte depletion? Immunotherapy. 2011;3:32-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 1.4] [Reference Citation Analysis (0)] |