Published online Apr 18, 2015. doi: 10.4254/wjh.v7.i5.761
Peer-review started: August 30, 2014
First decision: November 19, 2014
Revised: November 26, 2014
Accepted: December 16, 2014
Article in press: December 17, 2014
Published online: April 18, 2015
Processing time: 233 Days and 19.9 Hours
Impaired renal function is associated with a high risk of chronicity of hepatitis B virus (HBV) infection. Patients on hemodialysis (HD) or peritoneal dialysis are at an increased risk of viral transmission due to frequent necessity of blood product transfer as well as use of contaminated dialysate or dialysis materials. Additionally, health professionals may cause viral spread via contaminated hands and carelessness against hygiene rules. The frequency of chronic HBV infection may be as high as 80% in patients on renal replacement therapies. This is because HBV vaccination is essential to eliminate chronic HBV infection. However, response rates of HD patients to HBV vaccination vary between 10%-50%. Dialysis adequacy and early vaccination before the onset of dialysis therapy seem to be major determinants of high seroconversion rates. Older age, male gender, duration of dialysis therapy and nutritional status are other well-known factors associated with seroconversion rate. There are controversial reports regarding the role of the presence of diabetes mellitus, HCV positivity, erythropoietin resistance, hyperparathyroidism, and vitamin D inadequacy. The role of genetic alteration in the functions or production of cytokines still needs to be elucidated.
Core tip: Due to immunesuppresive effect of uremia and dialyser membranes, chronicity of hepatitis B virus (HBV) infection is frequently observed. Rates of seroconversion induced by HBV vaccine is diminished in chronic kidney disease patients when compared to the general population, which gradually decrease as renal functions deteriorates. Efficient dialysis is a major determinant of response to HBV vaccination. In contrast to three doses of 20 μg HBV vaccine for the general population, patients on hemodialysis or peritoneal dialysis usually require four doses of 40 μg HBV vaccine.
- Citation: Sit D, Esen B, Atay AE, Kayabaşı H. Is hemodialysis a reason for unresponsiveness to hepatitis B vaccine? Hepatitis B virus and dialysis therapy. World J Hepatol 2015; 7(5): 761-768
- URL: https://www.wjgnet.com/1948-5182/full/v7/i5/761.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i5.761
Hepatitis B virus (HBV) infection is an important public health problem affecting approximately 500 million people worldwide[1-3]. According to 2010 data, 360 million people have chronic HBV infection that leads to more than 1 million deaths/year due to acute hepatitis, cirrhosis or hepatocellular carcinoma[4,5].
Patients with chronic kidney disease (CKD) exhibit an impaired immune response against host agents including HBV due to bone marrow suppression caused by uremia and loss of CD4 T cells by use of bio-incompatible dialysate and membranes[6,7]. Patients on hemodialysis (HD) or peritoneal dialysis (PD) have an increased risk of HBV related complications. On the other hand, the rates of seroconversion induced by HBV vaccination in patients with CKD is significantly lower than those in the general population[8,9].
Chronic HBV infection is associated with high morbidity and mortality by leading to carrier state or chronic infection[10-14]. Pediatric population, especially newborns, as well as individuals at an advanced age are at an increased risk of chronicity of HBV infection[15]. Clinical course of chronic HBV infection may vary from asymptomatic carrier state to cirrhosis or even hepatocellular carcinoma[16].
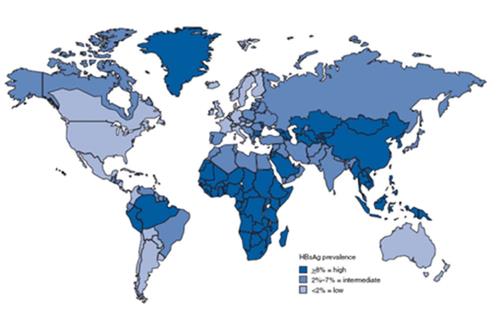
Recently, the rates of hepatitis B surface antigen (HBsAg) positivity is 0.1% in Western countries[17]. However, it is significantly higher in some areas like southeastern Asia and Middle East. The majority of southeast Asia and Middle East countries have an intermediate or high endemicity of HBV infection[18]. Based on the data in 2009, the rate of HBsAg positivity was 4.4% in the Turkish population (ranging from 2.5% to 9.1%)[19]. Figure 1 shows the geographic distribution of chronic HBV infection.
The chronicity rate of HBV infection is 5%-10% in the general population, whereas it may be as high as 60%-80% in patients receiving renal replacement therapy (RRT)[20]. Nucleoside analogues and interferon (IFN) are choices of treatment; however, a sustained viral response is achieved in only 30%-40% of patients on dialysis[21]. Owing to the fact that the chronicity rate of HBV infection is high and success rate of antiviral therapy is low in dialysis population, preventive measures against HBV infection is of vital importance.
Since the first recommendation of HBV vaccination by the Center for Disease Control and Prevention, the United States in 1982, administration of recombinant HBV vaccine which is composed of HBsAg is routinely used[22].
Former vaccines were derived from human plasma; however, as a consequence of innovations in vaccine technology, vaccines produced by recombinant DNA technology were introduced[23]. Recombinant HBV vaccine composed of HBsAg is associated with high seroconversion rates[24]. Recombinant HBV vaccine contains 20 μg HBsAg solution and 0.5 mg aluminium salt[25]. A number of adjuvants including levamisole, zinc, interferon, interleukin-2 (IL-2) and thymopoietin were added to increase the effectiveness[26-32].
Neutralizing antibodies against HBsAg indicate prior infection with HBV or triggered immune response against HBsAg in HBV vaccination[33-37]. Exposure to HBV is defined as appearance of HBsAg with or without antibody to hepatitis B e antigen (HBeAg) and hepatitis B core antigen (HBcAg)[38]. A group of patients may be in the window period which is associated with sole appearance of IgM class antibody against HBcAg[39]. Seroconversion of HBV is defined as appearance of antibodies to HBsAg (antiHBs) in the absence of HBsAg, HBeAg, HBcAg and undetectable HBV DNA[40]. Table 1 summarizes the interpretation of serologic results.
| HBsAg | Total anti HBc | IgM anti HBc | AntiHBs | Interpretation |
| - | - | - | - | Noninfected |
| + | - | - | - | Acute infection (early phase) |
| + | + | + | - | Acute infection |
| - | + | + | - | Recovering acute infection |
| - | + | - | + | İmmunized patient, past infection |
| + | + | - | - | Chronic infection |
| - | + | - | - | Chronic infection with low level viremia or false positive |
| - | - | - | + | Immunized |
HBV vaccination should be started before the initiation of RRT[41]. Currently, intramuscular administration HBV vaccine at 0, 1, 2 and 6 mo at a dose of 40 μg is recommended. Instead of gluteal region which contains muscle and fat, deltoid muscle is a preferable area to increase response rates[42].
There are variable response rates to HBV vaccination among HD patients. Inadequate seroconversion rates in the general population and patients on RRT are 5%-10% and 40%-50%, respectively[43]. According to another report, 20% of vaccinated patients on HD still does not achieve antibody formation against HBsAg[44].
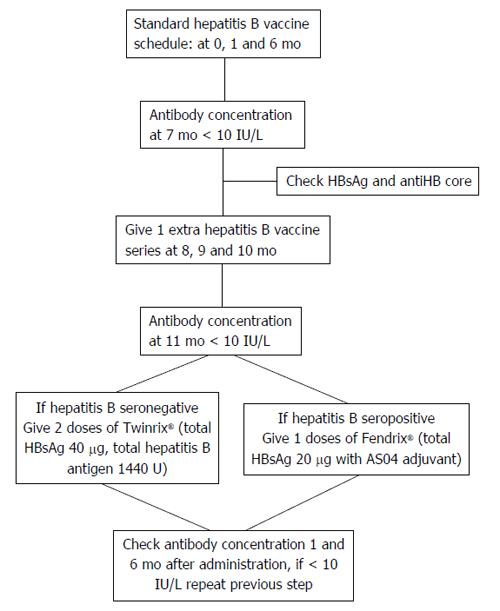
Lack of consensus exists regarding determining optimal vaccination schedule for patients with CKD at predialysis stage. For patients on RRT, the recommended vaccination schedule contains twice the dose of the general population (40 μg) in 4 cycles at intervals of 0, 1, 2 and 6 mo administered by intramuscular route at one site[45]. Additional three cycles of HBV vaccine should be administered to patients who do not respond to primary schedule[46,47] (Figure 2).
Because patients on RRT have blunted immune response, they exhibited an unsatisfactory response to HBV vaccination when compared to healthy individuals[48]. Dacko et al[49] concluded that efficient hemodialysis, age, nutritional status and systemic inflammation are determinants of an adequate response to HBV vaccination[49]. Similarly, Hashemi et al[50] stated that duration of dialysis, hemoglobin, and parathyroid hormone level and accompanying HCV infection do not affect immune response to HBV vaccination[50].
Seroconversion and adequate response are defined as anti-HBs > 10 IU/mL and > 100 IU/mL, respectively. Buti et al[51] stated that seroconversion was achieved in 76.7% of HD patients whereas adequate response was observed only in 53.5% at the third month of vaccination[51]. In a report from Saudia Arabia, adequate response rates reached 89.5% in HD patients[52]. Similarly, some reports determined satisfactory seroconversion rates among HD patients. Jadoul et al[53] showed that the seroconversion rate among HD patients was 89.65%[53]. A suboptimal response to HBV vaccine in HD patients is probably related to immunologic factors and poor nutritional status. Patients on RRT have impaired humoral and cellular immune response leading to underproduction of antibody.
Seroconversion rates may vary in different stages of CKD. Agarwal et al[47] performed a study to determine response rates to HBV vaccine in mild (creatinine 1.5 mg/dL to 3.0 mg/dL), moderate (creatinine 3.0 mg/dL to 6.0 mg/dL) and severe (creatinine > 6.0 mg/dL) CKD[47]. They pointed that seroconversion rates by three doses of 20 μg HBV vaccine in mild, moderate and severe CKD were 87.5%, 66.6% and 35.7%, respectively, which were significantly lower than seroconversion rates achieved by four doses of 40 μg (100%, 77% and 36.4%, respectively).
There are some reports with regard to the role of administration route on the rate of serconversion in HD patients. In a meta-analysis including 14 studies and 718 adult patients on HD, Fabrizi et al[54] concluded that seroconversion rate associated with intramuscular administration of HBV vaccine is significantly lower than that with intradermal administration [odds ratio (OR) = 0.454, 95%CI: 0.30-0.67, P = 0.001)[54].
There are controversial reports regarding success rate of HBV vaccination in patients at predialysis stage and patients on dialysis therapy. Taheri et al[55] indicated that response rate to HBV vaccination in predialysis patients is similar to that in dialysis patients. In contrast, Seaworth et al[56] observed that patients at predialysis stage have a more favorable outcome than patients at dialysis stage, suggesting that vaccination should be given as early as possible.
In conclusion, several factors including advanced age, DR3, DR7 and DQ2 positivity and the absence of A2 alleles may influence a response to hepatitis B vaccine in HD patients. Natural HBV infection achieves higher seroconversion rates than HBV vaccination; however, current HBV vaccination schedule provides remarkable seroconversion rates.
HBV vaccination stimulates specific antibody production by the activation of B cells, which is mediated by CD8+ cytotoxic T cells and CD4+ helper T cells[57]. As previously known, uremia is associated with an impaired immune response via several ways including cellular and humoral immune mechanisms. Patients on dialysis have lymphocytopenia, shortened life duration of lymphocytes and/or dysfunctional lymphocytes. Adequate CD4+ lymphocyte count is essential to provide antibody production subsequent to vaccination[58,59].
Sengar et al[60] showed that an impaired immune response to HBV transmission is linked to a group of human leukocyte antigens (HLAs). Alper et al[61] determined an association between an inadequate response to HBV vaccine and HLA-DR3 and HLA-B8 in the Caucasian population. Some HLA groups were identified as predictors of low response to HBV vaccine. Pol et al[62] and Höhler et al[63] showed that low responders to HBV vaccine have enhanced expression of DRB 1 × 3, DRB 1 × 7 and DRB 1 × 14[62,63].
Walker et al[64] pointed out that nonresponders to HBV vaccine exhibit excess of HLA-DR7 and absence of HLA-DR1. In accordance with this study, patients with HLA-DR1, -DR5, -DR2, -DQ5 and-DP4 usually well respond to HBV vaccine and usually seroconvert[63].
Albumin level as a nutritional marker has been shown to directly affect antibody response to HBV vaccination. Brown et al[65] showed that patients with hypoalbuminemia are unable to produce adequate titers of antiHBs. Creatinine level is an indicator of protein intake and nutrition in the general population; however, due to lower excretion rate in patients with CKD, it is not a suitable marker for the assessment of nutritional status.
Age is another factor that may affect antibody response to vaccination[66]. Owing to the fact that bone marrow depression by aging, humoral and cellular responses are impaired in elderly patients. Patients at an advanced age have lymphocytopenia, monocytopenia and neutropenia as well as functional deterioration of these cells. Lymphocytes mediate humoral response against viral antigens in different steps. Only 15% of responders were older than 60 years; however, 55% of nonresponders were above 60 years of age[47]. Decline of antiHBs level is quicker in older patients, suggesting defective function of T lymphocytes and inadequate production of interleukins. In a study from Egypt, rate of seroconversion caused by HBV vaccination may be as high as 89% while it was only 51% in patients above 60 years of age[67]. Seroconversion rates significantly decline in older patients. The mean age of responders was 40.6 years while that of nonresponders was 59.6 years in the same study.
Also, male patients on dialysis have a significantly diminished antibody response to HBV vaccine when compared to female patients. Male gender is associated with an impaired response to vaccine. Seroconversion rates in female and male dialysis patients were 85.6% and 68.3%, respectively, and only 29% of patients with seroconversion were male[21].
Body weight, diabetes mellitus, hyperparathyroidism, erythropoietin resistance, vitamin D deficiency, use of low bio-incompatible dialysis material, iron overload, high number of blood product transfer, vitamin deficiency and hepatitis C positivity are well-known factors that are associated with poor response to vaccination[68-71]. On the other hand, Roozbeh et al[72] stated that age, gender, body mass index and serum albumin level do not significantly affect seroconversion rates.
Dialysis adequacy is probably a globally validated determinant of seroconversion rates. Seroconversion rates significantly correlate with renal function. Ghadiani et al[73] reported that seroconversion rates in patients with GFR < 15 mL/min, 15 to 60 mL/min and > 90 mL/min are 44%, 90% and 96%, respectively.
Controversy exists about the role of diabetes mellitus in response to HBV vaccine. Al Saran et al[52] concluded that the presence of diabetes mellitus has no significant effect on seroconversion rates. However, Chin et al[74] stated that dialysis patients with diabetes mellitus have a poor response to HBV vaccine.
Afsar et al[69] carried out a study in dialysis patients to evaluate the relation of erythropoietin resistance and response to HBV vaccine, and observed that erythropoietin resistance inversely influences the response to HBV vaccine.
A vast majority of reports determined that HCV positivity is related with a poor response to HBV vaccination[75]. However, some recent reports failed to demonstrate a negative impact of HCV positivity on response to HBV vaccination[76]. Table 2 summarizes the factors involved in the pathogenesis of unresponsiveness to HBV vaccination.
| General population | Patients with chronic kidney disease |
| Obesity | Dialysis |
| Smoking | Inflammation |
| Administration route of vaccine | |
| Diabetes mellitus | |
| Hyperparathyroidism | |
| Lymphomas | |
| Co-existing HCV | |
| Newborns and advanced age | Advanced age |
| Inflammation | Vitamin D deficiency |
| Celiac disease | Male gender |
| Hypoalbuminemia | |
| Erythropoietin resistance | |
| IL-18 and IFN-y gene polymorphisms |
Patients on dialysis therapy have functionally and/or numerically defective regulatory T cells, leading to immunodeficiency and dysintegration between antigen presenting cells and CD4 T cells[77]. Accordingly, patients on HD had deteriorated neutrophil and macrophage functions resulting from inhibited chemotaxis and opsonization, both of which play a reactive role against host antigens. Selective T cell depletion is a frequently observed immunologic defect in dialysis patients, which causes diminished production of IL-1, IL-2, IL-6 and tumor necrosis factor-α[78]. In addition, interferon-gamma is produced by T cells and induces endocellular lysis of microorganisms and antigens.
Immunodeficiency is less frequently detected in patients receiving PD. They generally have depressed bactericidal activities of macrophages like opsonization, phagocytosis and lymphocytopenia, which reflects diminished peritoneal host defense[79]. Dialysis membranes and use of reaginic dialysis material are associated with excessive but non-effective immune response[80].
Regulation of immune response and interaction of mediators involved in immune response are complex processes and some unknown factors may influence their functions[81]. Roy et al[82] stated that decreased levels of cytokines that mediate the function of T helper cells may be associated with a low response to HBV vaccine. Deficiency of Th-1 like cells and defective or inadequate production of some cytokines by Th-1 cells are associated with immunosupression and a low response to viral agents[83]. IL-1, IL-2, IL-6, IL-12 and IFN-gamma are major cytokines involved in response to viral agents. Genetic polymorphisms and polymorphic variant of specific cytokines are associated with unresponsiveness to HBV vaccine[84].
The recommended antibody titer to HBsAg should be > 100 IU/mL[85]. An important proportion of dialysis patients who achieve an adequate response (> 100 IU/mL) require a booster dose in every 5 years to maintain antiHBs titer[86]. Patients who failed to produce an adequate antibody response should undergo booster vaccination at 1 year and at 5 years of primary vaccination schedule[73].
The antibody titer < 10 IU/mL is defined as hyporesponse and > 10 IU/mL is accepted as positive seroconversion[87]. However, anti-HBs titer below 100 IU/mL is evidence of a low response.
Positive seroconversion (antiHBs > 10 IU/mL) does not always warrant protection against HBV infection in dialysis patients. Lombardi et al[88] suggested that antiHBs titer of at least 50 IU/mL should be a target level in HD patients.
Because the exact reason of lower serconversion rates to HBV vaccine is not known, the best strategy to overcome the unresponsiveness is to administer additional HBV vaccine. Wismans et al[89] showed that seroconversion rates after one and three additional 20 μg dose of HBV vaccine were 38% and 75%, respectively. Similarly, another study demonstrated a 61% seroconversion rate after additional vaccination[90].
On the other hand, a group of dialysis patients who well respond to HBV vaccination and produce neutralizing antibodies against HBsAg do not maintain the antibody level with time. Although a decline in antiHBs titer by time is globally known in the general population as well as dialysis patients, it is significantly frequent and quicker in patients on RRT.
At the first year of vaccination, antiHBs > 10 IU/mL is induced in 82.5% of the general population by three doses of 20 μg, however, it was only 53% in dialysis patients by four doses of 40 μg[91]. At the third year of vaccination, the vast majority of HD patients have undetectable antiHbs level. American Association for the Study of Liver Diseases recommends annual screening of antiHBs titers and booster vaccination as antiHBs titer is around 10 IU/mL[40].
Innovations in recombinant DNA vaccine technology may be hopeful to increase seroconversion rates and sustained response. IL-12-based vaccination therapies may restore HBV-specific CD4(+) T cell responses and augment seroconversion[92]. In agreement with Zeng et al[92], Lau et al[93] showed that combination of HBV vaccine with interferon-gamma or IL-12 may enhance therapeutic efficacy[93]. Accordingly, Somi et al[94] mentioned that IFN-adjuvanted HBV vaccination may be beneficial for hyporesponsive patients. In addition, nano-adjuvants seem to be frequently used to overcome unresponsiveness[95].
Despite increased awareness against HBV and improvement in hygiene preservations, patients receiving RRT are still at an increased risk of HBV transmission. Additionally, due to immunosuppresive effect of uremia and dialyser membranes, chronicity of HBV infection is frequently observed. Rates of seroconversion induced by HBV vaccine is diminished in CKD patients when compared to the general population, which gradually decrease as renal functions deteriorate. Efficient dialysis is a major determinant of response to HBV vaccination. That is why early vaccination against HBV as soon as possible is essential to overcome unresponsiveness to HBV vaccine. In contrast to three doses of 20 μg HBV vaccine for the general population, patients on HD or PD usually require four doses of 40 μg HBV vaccine. Patients with CKD should be screened annually to detect decline of antiHBs titer and administered additional doses of HBV vaccine.
P- Reviewer: Farzin R, McQuillan GM, Sonzogni A S- Editor: Ji FF L- Editor: Wang TQ E- Editor: Liu SQ
| 1. | Shepard CW, Simard EP, Finelli L, Fiore AE, Bell BP. Hepatitis B virus infection: epidemiology and vaccination. Epidemiol Rev. 2006;28:112-125. [PubMed] |
| 2. | Ozaras R, Inanc Balkan I, Yemisen M, Tabak F. Epidemiology of HBV subgenotypes D. Clin Res Hepatol Gastroenterol. 2015;39:28-37. [PubMed] |
| 3. | Margolis HS, Alter MJ, Hadler SC. Hepatitis B: evolving epidemiology and implications for control. Semin Liver Dis. 1991;11:84-92. [PubMed] |
| 4. | Kaić B, Vilibić-Cavlek T, Filipović SK, Nemeth-Blazić T, Pem-Novosel I, Vucina VV, Simunović A, Zajec M, Radić I, Pavlić J. [Epidemiology of viral hepatitis]. Acta Med Croatica. 2013;67:273-279. [PubMed] |
| 5. | Güthle M, Dollinger MM. [Epidemiology and risk factors of hepatocellular carcinoma]. Radiologe. 2014;54:654-659. [PubMed] |
| 6. | Noori S, Gol-Mohamadi A, Sarbazi MR, Safaee A, Farsar AR. Epidemiological features of hepatitis B and C infection in a high risk population: results of screening programs. Gastroenterol Hepatol Bed Bench. 2013;6:136-140. [PubMed] |
| 7. | Akbaş H, Yalcin K, Isi H, Tekes S, Atay AE, Akkus Z, Budak T. Role of p53 codon 72 polymorphism in chromosomal aberrations and mitotic index in patients with chronic hepatitis B. Braz J Med Biol Res. 2012;45:1011-1016. [PubMed] |
| 8. | Liu CJ, Huang YJ, Chen HL, Lin HT, Lin HL, Kuo ML, Huang JL, Chen DS, Chen PJ, Wu HL. Characterization of interleukin 8 in woodchucks with chronic hepatitis B and hepatocellular carcinoma. Genes Immun. 2009;10:27-36. [PubMed] |
| 9. | Thanachartwet V, Phumratanaprapin W, Desakorn V, Sahassananda D, Wattanagoon Y, Chaiprasert A, Aimpun P, Supaporn T. Viral hepatitis infections among dialysis patients: Thailand registry report. Nephrology (Carlton). 2007;12:399-405. [PubMed] |
| 10. | Do TN, Nam S. Knowledge, Awareness and Medical Practice of Asian Americans/Pacific Islanders on Chronic Hepatitis B Infection: Review of Current Psychosocial Evidence. Pogon Sahoe Yongu. 2011;31:341-364. [PubMed] |
| 11. | World Health Organization. Hepatitis B, fact sheet no. 204. 2012; Available from: http://www.who.int/mediacentre/factsheets/fs204/en/. |
| 12. | Liaw YF, Brunetto MR, Hadziyannis S. The natural history of chronic HBV infection and geographical differences. Antivir Ther. 2010;15 Suppl 3:25-33. [PubMed] |
| 13. | Gust ID. Epidemiology of hepatitis B infection in the Western Pacific and South East Asia. Gut. 1996;38 Suppl 2:S18-S23. [PubMed] |
| 14. | Croagh CM, Lubel JS. Natural history of chronic hepatitis B: phases in a complex relationship. World J Gastroenterol. 2014;20:10395-10404. [PubMed] |
| 15. | Gentile I, Borgia G. Vertical transmission of hepatitis B virus: challenges and solutions. Int J Womens Health. 2014;6:605-611. [PubMed] |
| 16. | Luca AS, Dorobăţ C, Ursu RG, Luca MC, Vâţă A, Iancu LS. Epidemiological and laboratory features of chronic hepatitis B cases in the interval 2010-2013. Rev Med Chir Soc Med Nat Iasi. 2014;118:479-484. [PubMed] |
| 17. | Beckett GA, Ramirez G, Vanderhoff A, Nichols K, Chute SM, Wyles DL, Schoenbachler BT, Bedell DT, Cabral R, Ward JW. Early identification and linkage to care of persons with chronic hepatitis B virus infection--three U.S. sites, 2012-2014. MMWR Morb Mortal Wkly Rep. 2014;63:399-401. [PubMed] |
| 18. | Ibrahim S, el-Din S, Bazzal I. Antibody level after hepatitis-B vaccination in hemodialysis patients: impact of dialysis adequacy, chronic inflammation, local endemicity and nutritional status. J Natl Med Assoc. 2006;98:1953-1957. [PubMed] |
| 19. | Sayan M, Cavdar C, Dogan C. Naturally occurring polymerase and surface gene variants of hepatitis B virus in Turkish hemodialysis patients with chronic hepatitis B. Jpn J Infect Dis. 2012;65:495-501. [PubMed] |
| 21. | Grzegorzewska AE. Hepatitis B vaccination in chronic kidney disease patients: a call for novel vaccines. Expert Rev Vaccines. 2014;13:1317-1326. [PubMed] |
| 22. | Centers for Disease Control (CDC). Recommendation of the Immunization Practices Advisory Committee (ACIP). Inactivated hepatitis B virus vaccine. MMWR Morb Mortal Wkly Rep. 1982;31:317-322, 327-328. [PubMed] |
| 23. | Bruguera M, Cremades M, Mayor A, Sánchez Tapias JM, Rodés J. Immunogenicity of a recombinant hepatitis B vaccine in haemodialysis patients. Postgrad Med J. 1987;63 Suppl 2:155-158. [PubMed] |
| 24. | Stevens CE, Taylor PE, Tong MJ, Toy PT, Vyas GN, Nair PV, Weissman JY, Krugman S. Yeast-recombinant hepatitis B vaccine. Efficacy with hepatitis B immune globulin in prevention of perinatal hepatitis B virus transmission. JAMA. 1987;257:2612-2616. [PubMed] |
| 25. | Norrby E. Prospects for new viral vaccines. Microbiol Sci. 1987;4:202-205. [PubMed] |
| 26. | Zhang W, Du X, Zhao G, Jin H, Kang Y, Xiao C, Liu M, Wang B. Levamisole is a potential facilitator for the activation of Th1 responses of the subunit HBV vaccination. Vaccine. 2009;27:4938-4946. [PubMed] |
| 27. | Afsharian M, Vaziri S, Janbakhsh AR, Sayad B, Mansouri F, Nourbakhsh J, Qadiri K, Najafi F, Shirvanii M. The effect of zinc sulfate on immunologic response to recombinant hepatitis B vaccine in elderly: Zinc sulfate and immunologic response to recombinant hepatitis B vaccine. Hepat Mon. 2011;11:32-35. [PubMed] |
| 28. | Tan W, Meng Y, Li H, Chen Y, Han S, Zeng J, Huang A, Li B, Zhang Y, Guo Y. A bispecific antibody against two different epitopes on hepatitis B surface antigen has potent hepatitis B virus neutralizing activity. MAbs. 2013;5:946-955. [PubMed] |
| 29. | Jiang YF, Ma ZH, Zhao PW, Pan Y, Liu YY, Feng JY, Niu JQ. Effect of thymosin-α(1) on T-helper 1 cell and T-helper 2 cell cytokine synthesis in patients with hepatitis B virus e antigen-positive chronic hepatitis B. J Int Med Res. 2010;38:2053-2062. [PubMed] |
| 30. | Saini V, Jain V, Sudheesh MS, Dixit S, Gaur RL, Sahoo MK, Joseph SK, Verma SK, Jaganathan KS, Murthy PK. Humoral and cell-mediated immune-responses after administration of a single-shot recombinant hepatitis B surface antigen vaccine formulated with cationic poly(l-lactide) microspheres. J Drug Target. 2010;18:212-222. [PubMed] |
| 31. | Dikici B, Bosnak M, Haspolat K. The effect of thymosine 1 alpha and Lamivudine combination therapy in chronic hepatitis B patients. Indian Pediatr. 2002;39:701-702. [PubMed] |
| 32. | Baj Z, Pokoca L, Tchórzewski H, Dworniak D, Tkacz B. The effect of long-term thymosine factor x/TFx/treatment on T lymphocyte subpopulations and peripheral blood mononuclear cell cytotoxicity in chronic active hepatitis B. J Investig Allergol Clin Immunol. 1991;1:239-246. [PubMed] |
| 33. | Simmonds P, Midgley S. Recombination in the genesis and evolution of hepatitis B virus genotypes. J Virol. 2005;79:15467-15476. [PubMed] |
| 34. | Sumi H, Yokosuka O, Seki N, Arai M, Imazeki F, Kurihara T, Kanda T, Fukai K, Kato M, Saisho H. Influence of hepatitis B virus genotypes on the progression of chronic type B liver disease. Hepatology. 2003;37:19-26. [PubMed] |
| 35. | Hadziyannis SJ, Papatheodoridis GV. Hepatitis B e antigen-negative chronic hepatitis B: natural history and treatment. Semin Liver Dis. 2006;26:130-141. [PubMed] |
| 36. | de Franchis R, Meucci G, Vecchi M, Tatarella M, Colombo M, Del Ninno E, Rumi MG, Donato MF, Ronchi G. The natural history of asymptomatic hepatitis B surface antigen carriers. Ann Intern Med. 1993;118:191-194. [PubMed] |
| 37. | Zarski JP, Marcellin P, Leroy V, Trepo C, Samuel D, Ganne-Carrie N, Barange K, Canva V, Doffoel M, Cales P. Characteristics of patients with chronic hepatitis B in France: predominant frequency of HBe antigen negative cases. J Hepatol. 2006;45:355-360. [PubMed] |
| 38. | Chu CJ, Keeffe EB, Han SH, Perrillo RP, Min AD, Soldevila-Pico C, Carey W, Brown RS, Luketic VA, Terrault N. Prevalence of HBV precore/core promoter variants in the United States. Hepatology. 2003;38:619-628. [PubMed] |
| 39. | Gaeta GB, Stornaiuolo G, Precone DF, Lobello S, Chiaramonte M, Stroffolini T, Colucci G, Rizzetto M. Epidemiological and clinical burden of chronic hepatitis B virus/hepatitis C virus infection. A multicenter Italian study. J Hepatol. 2003;39:1036-1041. [PubMed] |
| 40. | Lok AS, McMahon BJ. Chronic hepatitis B. Hepatology. 2007;45:507-539. [PubMed] |
| 41. | Magnani G, Calzetti C, Campari M, Lehndorff H, Pizzaferri P, Rossi E. [Immune response to hepatitis B vaccine and duration of protection in a dialysis unit]. Acta Biomed Ateneo Parmense. 1987;58:41-47. [PubMed] |
| 42. | Janus N, Vacher LV, Karie S, Ledneva E, Deray G. Vaccination and chronic kidney disease. Nephrol Dial Transplant. 2008;23:800-807. [PubMed] |
| 43. | Grzegorzewska AE, Wobszal PM, Sowińska A, Mostowska A, Jagodziński PP. Association of the interleukin-12 polymorphic variants with the development of antibodies to surface antigen of hepatitis B virus in hemodialysis patients in response to vaccination or infection. Mol Biol Rep. 2013;40:6899-6911. [PubMed] |
| 44. | Lin SY, Liu JH, Wang SM, Wang IK, Tsai CA, Liu YL, Lin HH, Chang CC, Huang CC. Association of response to hepatitis B vaccination and survival in dialysis patients. BMC Nephrol. 2012;13:97. [PubMed] |
| 45. | Ramezani A, Eslami far A, Ahmadi F, Maziar S, Razeghi E, Kalantar E. Is any factor influence on hepatitis B vaccination response in hemodialysis patients. Internet J Nephrol. 2009;3:1540-1547. |
| 46. | Stachowski J, Pollok M, Barth C, Maciejewski J, Baldamus CA. Non-responsiveness to hepatitis B vaccination in haemodialysis patients: association with impaired TCR/CD3 antigen receptor expression regulating co-stimulatory processes in antigen presentation and recognition. Nephrol Dial Transplant. 1994;9:144-152. [PubMed] |
| 47. | Agarwal SK, Irshad M, Dash SC. Comparison of two schedules of hepatitis B vaccination in patients with mild, moderate and severe renal failure. J Assoc Physicians India. 1999;47:183-185. [PubMed] |
| 48. | Fernández E, Betriu MA, Gómez R, Montoliu J. Response to the hepatitis B virus vaccine in haemodialysis patients: influence of malnutrition and its importance as a risk factor for morbidity and mortality. Nephrol Dial Transplant. 1996;11:1559-1563. [PubMed] |
| 49. | Dacko C, Holley JL. The influence of nutritional status, dialysis adequacy, and residual renal function on the response to hepatitis B vaccination in peritoneal dialysis patients. Adv Perit Dial. 1996;12:315-317. [PubMed] |
| 50. | Hashemi B, Mahdavi-Mazdeh M, Abbasi M, Hosseini-Moghaddam SM, Zinat NH, Ahmadi F. Efficacy of HBV vaccination in various stages of chronic kidney disease: is earlier better? Hepat Mon. 2011;11:816-820. [PubMed] |
| 51. | Buti M, Viladomiu L, Jardi R, Olmos A, Rodriguez JA, Bartolome J, Esteban R, Guardia J. Long-term immunogenicity and efficacy of hepatitis B vaccine in hemodialysis patients. Am J Nephrol. 1992;12:144-147. [PubMed] |
| 52. | Al Saran K, Sabry A, Al Halawany Z, Ismail M. Factors affecting response to hepatitis B vaccine among hemodialysis patients in a large Saudi Hemodialysis Center. Saudi J Kidney Dis Transpl. 2014;25:185-191. [PubMed] |
| 53. | Jadoul M, Goubau P. Is anti-hepatitis B virus (HBV) immunization successful in elderly hemodialysis (HD) patients? Clin Nephrol. 2002;58:301-304. [PubMed] |
| 54. | Fabrizi F, Dixit V, Magnini M, Elli A, Martin P. Meta-analysis: intradermal vs. intramuscular vaccination against hepatitis B virus in patients with chronic kidney disease. Aliment Pharmacol Ther. 2006;24:497-506. [PubMed] |
| 55. | Taheri S, Shahidi S, Moghtaderi J, Seirafian S, Emami A, Eftekhari S. Response rate to Hepatitis B vaccination in patients with chronic renal failure and end-stage- renal- disease: Influence of diabetes mellitus. J Res Med Sci. 2005;10:384-390. |
| 56. | Seaworth B, Drucker J, Starling J, Drucker R, Stevens C, Hamilton J. Hepatitis B vaccines in patients with chronic renal failure before dialysis. J Infect Dis. 1988;157:332-337. [PubMed] |
| 57. | Rahman F, Dahmen A, Herzog-Hauff S, Böcher WO, Galle PR, Löhr HF. Cellular and humoral immune responses induced by intradermal or intramuscular vaccination with the major hepatitis B surface antigen. Hepatology. 2000;31:521-527. [PubMed] |
| 58. | Chatenoud L, Herbelin A, Beaurain G, Descamps-Latscha B. Immune deficiency of the uremic patient. Adv Nephrol Necker Hosp. 1990;19:259-274. [PubMed] |
| 59. | Descamps-Latscha B, Chatenoud L. T cells and B cells in chronic renal failure. Semin Nephrol. 1996;16:183-191. [PubMed] |
| 60. | Sengar DP, McLeish WA, Sutherland M, Couture RA, Rashid A. Hepatitis B antigen (HBAg) infection in a hemodialysis unit. I. HL-A8 and immune response to HBAg. Can Med Assoc J. 1975;112:968, 971. [PubMed] |
| 61. | Alper CA, Kruskall MS, Marcus-Bagley D, Craven DE, Katz AJ, Brink SJ, Dienstag JL, Awdeh Z, Yunis EJ. Genetic prediction of nonresponse to hepatitis B vaccine. N Engl J Med. 1989;321:708-712. [PubMed] |
| 62. | Pol S, Legendre C, Mattlinger B, Berthelot P, Kreis H. Genetic basis of nonresponse to hepatitis B vaccine in hemodialyzed patients. J Hepatol. 1990;11:385-387. [PubMed] |
| 63. | Höhler T, Meyer CU, Notghi A, Stradmann-Bellinghausen B, Schneider PM, Starke R, Zepp F, Sänger R, Clemens R, Meyer zum Büschenfelde KH. The influence of major histocompatibility complex class II genes and T-cell Vbeta repertoire on response to immunization with HBsAg. Hum Immunol. 1998;59:212-218. [PubMed] |
| 64. | Walker M, Szmuness W, Stevens C, Rubinstein P. Genetics of anti-HBs responsiveness. I. HLA-DR7 and non-responsiveness to hepatitis vaccination. Transfusion. 1981;21:601. |
| 65. | Brown CM, Donlon S, O’Kelly P, Casey AM, Collier C, Conlon PJ, Walshe JJ. A prospective study of hepatitis B vaccination - a comparison of responders versus nonresponders. Ren Fail. 2011;33:276-279. [PubMed] |
| 66. | Tang S, Lai KN. Chronic viral hepatitis in hemodialysis patients. Hemodial Int. 2005;9:169-179. [PubMed] |
| 67. | Shatat HZ, Kotkat AM, Farghaly AG. Immune response to hepatitis B vaccine in haemodialysis patients. J Egypt Public Health Assoc. 2000;75:257-275. [PubMed] |
| 68. | Zitt E, Sprenger-Mähr H, Knoll F, Neyer U, Lhotta K. Vitamin D deficiency is associated with poor response to active hepatitis B immunisation in patients with chronic kidney disease. Vaccine. 2012;30:931-935. [PubMed] |
| 69. | Afsar B. The relationship between erythropoietin resistance and antibody response to hepatitis B vaccine in hemodialysis patients. Nephrourol Mon. 2013;5:806-812. [PubMed] |
| 70. | Navarro JF, Teruel JL, Mateos ML, Marcen R, Ortuno J. Antibody level after hepatitis B vaccination in hemodialysis patients: influence of hepatitis C virus infection. Am J Nephrol. 1996;16:95-97. [PubMed] |
| 71. | Alavian SM, Tabatabaei SV. The effect of diabetes mellitus on immunological response to hepatitis B virus vaccine in individuals with chronic kidney disease: A meta-analysis of current literature. Vaccine. 2010;28:3773-3777. [PubMed] |
| 72. | Roozbeh J, Moini M, Lankarani KB, Sagheb MM, Shahpoori S, Bastani B. Low dose intradermal versus high dose intramuscular hepatitis B vaccination in patients on chronic hemodialysis. ASAIO J. 2005;51:242-245. [PubMed] |
| 73. | Ghadiani MH, Besharati S, Mousavinasab N, Jalalzadeh M. Response rates to HB vaccine in CKD stages 3-4 and hemodialysis patients. J Res Med Sci. 2012;17:527-533. [PubMed] |
| 74. | Chin AI. Hepatitis B virus vaccine response in hemodialysis: baseline patient characteristics. Hemodial Int. 2003;7:296-303. [PubMed] |
| 75. | Elzouki AN, Elgamay SM, Zorgani A, Elahmer O. Hepatitis B and C status among health care workers in the five main hospitals in eastern Libya. J Infect Public Health. 2014;7:534-541. [PubMed] |
| 76. | Kovacić V, Sain M, Vukman V. [Does efficient haemodialysis improve the response to hepatitis B virus vaccination?]. Lijec Vjesn. 2004;126:133-137. [PubMed] |
| 77. | Afzali B, Edozie FC, Fazekasova H, Scottà C, Mitchell PJ, Canavan JB, Kordasti SY, Chana PS, Ellis R, Lord GM. Comparison of regulatory T cells in hemodialysis patients and healthy controls: implications for cell therapy in transplantation. Clin J Am Soc Nephrol. 2013;8:1396-1405. [PubMed] |
| 78. | Hosking CS, Atkins RC, Scott DF, Holdsworth SR, Fitzgerald MG, Shelton MJ. Immune and phagocytic function in patients on maintenance dialysis and post-transplantation. Clin Nephrol. 1976;6:501-505. [PubMed] |
| 79. | Kazancioglu R. Peritoneal defense mechanisms--the effects of new peritoneal dialysis solutions. Perit Dial Int. 2009;29 Suppl 2:S198-S201. [PubMed] |
| 80. | Lamperi S, Carozzi S. Immunological defenses in CAPD. Blood Purif. 1989;7:126-143. [PubMed] |
| 81. | Lamperi S, Carozzi S. Interferon-gamma (IFN-gamma) as in vitro enhancing factor of peritoneal macrophage defective bactericidal activity during continuous ambulatory peritoneal dialysis (CAPD). Am J Kidney Dis. 1988;11:225-230. [PubMed] |
| 82. | Roy MJ, Wu MS, Barr LJ, Fuller JT, Tussey LG, Speller S, Culp J, Burkholder JK, Swain WF, Dixon RM. Induction of antigen-specific CD8+ T cells, T helper cells, and protective levels of antibody in humans by particle-mediated administration of a hepatitis B virus DNA vaccine. Vaccine. 2000;19:764-778. [PubMed] |
| 83. | Duan Z, Chen X, Liang Z, Zeng Y, Zhu F, Long L, McCrae MA, Zhuang H, Shen T, Lu F. Genetic polymorphisms of CXCR5 and CXCL13 are associated with non-responsiveness to the hepatitis B vaccine. Vaccine. 2014;32:5316-5322. [PubMed] |
| 84. | Grzegorzewska AE, Pajzderski D, Sowińska A, Jagodziński PP. Polymporphism of monocyte chemoattractant protein 1 (MCP1 -2518 A/G) and responsiveness to hepatitis B vaccination in hemodialysis patients. Pol Arch Med Wewn. 2014;124:10-18. [PubMed] |
| 85. | McNulty CA, Bowen JK, Williams AJ. Hepatitis B vaccination in predialysis chronic renal failure patients a comparison of two vaccination schedules. Vaccine. 2005;23:4142-4147. [PubMed] |
| 86. | Teo CG, Locarnini SA. Potential threat of drug-resistant and vaccine-escape HBV mutants to public health. Antivir Ther. 2010;15:445-449. [PubMed] |
| 87. | Kara IH, Yilmaz ME, Suner A, Kadiroglu AK, Isikoglu B. The evaluation of immune responses that occur after HBV infection and HBV vaccination in hemodialysis patients. Vaccine. 2004;22:3963-3967. [PubMed] |
| 88. | Lombardi M, Pizzarelli F, Righi M, Cerrai T, Dattolo P, Nigrelli S, Michelassi S, Sisca S, Alecci A, Di Geronimo P. Hepatitis B vaccination in dialysis patients and nutritional status. Nephron. 1992;61:266-268. [PubMed] |
| 89. | Wismans PJ, van Hattum J, de Gast GC, Bouter KP, Diepersloot RJ, Maikoe T, Mudde GC. A prospective study of in vitro anti-HBs producing B cells (spot-ELISA) following primary and supplementary vaccination with a recombinant hepatitis B vaccine in insulin dependent diabetic patients and matched controls. J Med Virol. 1991;35:216-222. [PubMed] |
| 90. | Wismans P, van Hattum J, Stelling T, Poel J, de Gast GC. Effect of supplementary vaccination in healthy non-responders to hepatitis B vaccination. Hepatogastroenterology. 1988;35:78-79. [PubMed] |
| 91. | Shakhgil’dian IV, Khukhlovich PA, Savin EA, Kuzin SN, Anan’ev VA, Sergeeva NA, Khasanova VA, Shostka GD, Vu Z, Vasil’ev AN. [Risk of infection with hepatitis B and C viruses of medical workers, patients in the hemodialysis ward, and vaccine prophylaxis of hepatitis B infection in these populations]. Vopr Virusol. 1994;39:226-229. [PubMed] |
| 92. | Zeng Z, Kong X, Li F, Wei H, Sun R, Tian Z. IL-12-based vaccination therapy reverses liver-induced systemic tolerance in a mouse model of hepatitis B virus carrier. J Immunol. 2013;191:4184-4193. [PubMed] |
| 93. | Lau GK. Use of immunomodulatory therapy (other than interferon) for the treatment of chronic hepatitis B virus infection. J Gastroenterol Hepatol. 2000;15 Suppl:E46-E52. [PubMed] |
| 94. | Somi MH, Hajipour B. Improving hepatitis B vaccine efficacy in end-stage renal diseases patients and role of adjuvants. ISRN Gastroenterol. 2012;2012:960413. [PubMed] |
| 95. | Fakharzadeh S, Kalanaky S, Hafizi M, Goya MM, Masoumi Z, Namaki S, Shakeri N, Abbasi M, Mahdavi M, Nazaran MH. The new nano-complex, Hep-c, improves the immunogenicity of the hepatitis B vaccine. Vaccine. 2013;31:2591-2597. [PubMed] |
| 96. | Weinbaum S. National Center for HIV/AIDS, Viral Hepatitis, STD, and TB Prevention. 2006;. |
| 97. | Schillie SF, Spradling PR, Murphy TV. Immune response of hepatitis B vaccine among persons with diabetes: a systematic review of the literature. Diabetes Care. 2012;35:2690-2697. [PubMed] |