Published online Jun 18, 2015. doi: 10.4254/wjh.v7.i11.1553
Peer-review started: September 23, 2014
First decision: November 27, 2014
Revised: March 16, 2015
Accepted: April 10, 2015
Article in press: April 14, 2015
Published online: June 18, 2015
Processing time: 265 Days and 19.9 Hours
Patients with hepatocellular carcinoma (HCC) accompanying portal vein tumor thrombosis (PVTT) have relatively few therapeutic options and an extremely poor prognosis. These patients are classified into barcelona clinic liver cancer stage C and sorafenib is suggested as the standard therapy of care. However, overall survival (OS) gain from sorafenib is unsatisfactory and better treatment modalities are urgently required. Therefore, we critically appraised recent data for the various treatment strategies for patients with HCC accompanying PVTT. In suitable patients, even surgical resection can be considered a potentially curative strategy. Transarterial chemoembolization (TACE) can be performed effectively and safely in a carefully chosen population of patients with reserved liver function and sufficient collateral blood flow nearby the blocked portal vein. A recent meta-analysis demonstrated that TACE achieved a substantial improvement of OS in HCC patients accompanying PVTT compared with best supportive care. In addition, transarterial radioembolization (TARE) using yttrium-90 microspheres achieves quality-of-life advantages and is as effective as TACE. A large proportion of HCC patients accompanying PVTT are considered to be proper for TARE. Moreover, TACE or TARE achieved comparable outcomes to sorafenib in recent studies and it was also reported that the combination of radiotherapy with TACE achieved a survival gain compared to sorafenib in HCC patients accompanying PVTT. Surgical resection-based multimodal treatments, transarterial approaches including TACE and TARE, and TACE-based appropriate combination strategies may improve OS of HCC patients accompanying PVTT.
Core tip: Given the modest survival gain and the limitation of sorafenib, such as resistance and tolerability, there are still clinical unmet needs in the management of patients with hepatocellular carcinoma (HCC) accompanying portal vein tumor thrombosis (PVTT). Surgical resection-based multimodal treatments including liver transplantation and transarterial chemoembolization-based appropriate combination strategies for resectable HCC accompanying PVTT may improve overall survival in these patients.
- Citation: Yu SJ, Kim YJ. Effective treatment strategies other than sorafenib for the patients with advanced hepatocellular carcinoma invading portal vein. World J Hepatol 2015; 7(11): 1553-1561
- URL: https://www.wjgnet.com/1948-5182/full/v7/i11/1553.htm
- DOI: https://dx.doi.org/10.4254/wjh.v7.i11.1553
Globally, hepatocellular carcinoma (HCC) is one of the main reasons of malignancy related death[1,2]. Most HCCs are detected in an advanced stage in spite of surveillance programs for high risk populations, and the prognosis for these patients is poor. Consequently, a minority of patients is eligible for liver resection.
Portal vein tumor thrombosis (PVTT) arises in about 10%-40% of patients at diagnosis[3-5]; lower rates are reported when HCC is diagnosed early usually as a consequence of screening[3] and is apparent in up to 44% of patients with HCC at the end of life[6]. PVTT has a profound adverse effect on prognosis, with the median survival time of patients with unresectable HCC accompanying PVTT being significantly reduced (2-4 mo) compared to those not accompanying PVTT (10-24 mo)[4,5,7]. The range and position of PVTT further affect the prognosis. PVTT is related with poor prognosis probably because of the intensified risk of tumor spread, raised portal pressure inducing variceal bleeding and reduced portal flow causing jaundice, ascites, hepatic encephalopathy and hepatic failure[4,8].
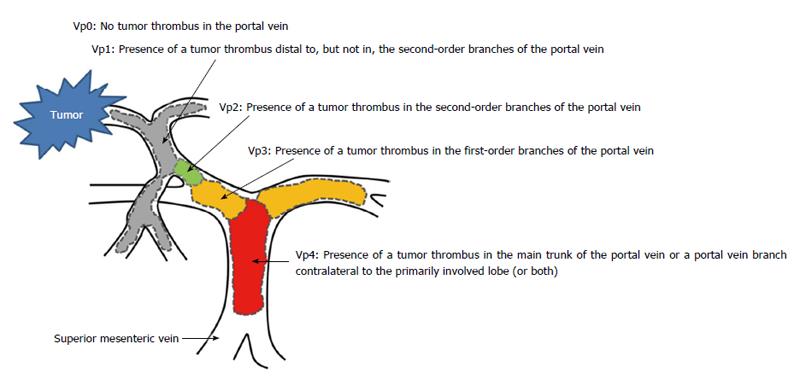
The Liver Cancer Study Group of Japan suggested a macroscopic classification for PVTT: categorized into five grades, Vp0-Vp4 (Figure 1). Each one is defined as follows: no PVTT, Vp0; existence of PVTT not in, but distal to, the 2nd-order branches of the portal vein, Vp1; existence of PVTT in the 2nd-order branches of the portal vein, Vp2; existence of PVTT in the 1st-order branches of the portal vein, Vp3; and existence of PVTT in the main trunk of the portal vein or a portal vein branch contralateral to the mainly involved lobe (or both), Vp4[9]. This classification is helpful, because it is established by surgical outcomes and by the clinical, imaging, and pathological findings.
The presence of PVTT also limits the treatment options, with HCC treatment guidelines often considering PVTT a contraindication for transplantation, curative resection and transarterial chemoembolization (TACE)[10-12]. Current guidelines recommend sorafenib for the patients with HCC with PVTT. Sorafenib is an oral multiple tyrosine kinases inhibitor that suppresses angiogenesis and tumor-cell proliferation and augments the rate of apoptosis[13]. In the Sorafenib HCC Assessment Randomized Protocol (SHARP) study[14] and multicenter study in Asian-Pacific region[15], sorafenib was proved to be efficacious and safe to patients with advanced HCC. Nevertheless, subgroup analyses for macroscopic vascular invasion (MVI) in these two pivotal studies showed only a marginal survival benefit for sorafenib over placebo[16,17]. Therefore, there are still clinical unmet needs in the treatment of patients with HCC accompanying PVTT.
This article review recent data for the various treatment strategies for the patients with HCC accompanying PVTT.
HCC is relatively resistant to traditional chemotherapy and liver dysfunction complicates the use of chemotherapeutic agents that undergo hepatic metabolism[8,11]. Sorafenib, a multiple tyrosine kinases inhibitor that blocks tumor angiogenesis and tumor cell proliferation, was the 1st systemic agent proven to significantly increase survival in advanced-stage HCC in randomized controlled trials[14,15]. Sub-analyses of SHARP trial[17] identified 231 patients staged barcelona clinic liver cancer (BCLC) C due to MVI and demonstrated that the sorafenib group (n = 108) achieved a longer median overall survival (OS) (8.1 mo vs 4.9 mo) and time to progression (TTP) (4.1 mo vs 2.7 mo) than the control group (n = 123) received placebo. In the sub-group analyses of the Asia-Pacific trial[16], patients with MVI and/or extrahepatic spread who received sorafenib (n = 118) showed a better clinical outcome than in placebo group (n = 61): median OS (5.6 mo vs 4.1 mo), TTP (2.7 mo vs 1.3 mo) and disease control rate (30.5% vs 11.5%), respectively. Although the authors argued that the survival benefit with sorafenib was evident regardless of the presence of PVTT in those two pivotal studies, subgroup analyses for MVI showed only a marginal survival benefit of sorafenib over placebo.
Two key trials and a meta-analysis indicated that TACE can improve survival (median 19-20 mo compared to 16 mo for untreated patients in clinical trials) in intermediate-stage HCC[18-20]. However, PVTT is generally considered a contraindication for TACE because of concerns that interruption to hepatic arterial blood supply could result in an enormous segment of hepatic necrosis in patients whose blood supply is already compromised[8,12]. Nevertheless, there is evidence that selected patients with PVTT can tolerate a modified delivery of TACE provided they have good liver function and collateral blood flow around the obstructed portal vein[4,21]. Recent two studies reported improvements in survival compared to conservative care in HCC patients accompanying PVTT[22,23]. Luo et al[22] a performed a prospective nonrandomized study and reported significantly better survival with TACE (n = 84) compared to conservative treatment (n = 80) either in non-cirrhotic or Child A cirrhotic HCC patients accompanying PVTT. The median OS, the 1-, and 2-year survival rates were 7.1 mo, 30.9%, and 9.2% for the TACE arm and 4.1 mo, 3.8%, and 0% for the conservative arm, respectively (P < 0.001)[22]. In the TACE group, the 40 patients with Vp1 or Vp2 survived longer than the 44 patients with Vp3 or Vp4 (median OS 10.2 mo vs 5.3 mo)[22]. In the second study, Chung et al[23] reported that TACE (n = 83) significantly improved survival compared to supportive care (n = 42; median OS 5.6 mo vs 2.2 mo, respectively; P < 0.001) in HCC patients with Vp4. Regardless of treatment (TACE or supportive care), patients with Child class B had worse outcomes (median OS 2.8 mo vs 1.9 mo) than those with Child class A (median OS 7.4 mo vs 2.6 mo)[23]. In addition, a recent meta-analysis evaluating 8 controlled trials (total 1601 HCC patients) demonstrated that TACE significantly improved the 6-mo (HR = 0.41; 95%CI: 0.32-0.53; P = 0.000) and 1-year (HR = 0.44; 95%CI: 0.34-0.57; P = 0.000) OS of HCC patients accompanying PVTT compared with best supportive treatment[24]. Moreover, another recent study comparing TACE and sorafenib in BCLC stage C HCC patients showed that TACE attained a comparable clinical outcome to sorafenib: the median OS was 9.2 mo (95%CI: 6.1-12.3 mo) for TACE group and 7.4 mo (95%CI: 5.6-9.2 mo) for sorafenib group (P = 0.377)[25]. The proportion of patients who had high-grade adverse events (grade ≥ 3) was significantly lower in the sorafenib arm (17%) than in the TACE arm (38%) (P = 0.024).
TACE using DC Bead, drug-eluting microsphere (Biocompatibles UK Ltd, Farnham, United Kingdom), is a relatively novel modality related with favorable systemic doxorubicin exposure/toxicity and liver-specific toxicity compared to conventional TACE[26]. A recent study involving BCLC B HCC patients showed that DC Bead TACE resulted in a significantly better clinical outcome compared to conventional TACE[27]. However, Sellers et al[26] reported poor OS in HCC patients accompanying PVTT underwent DC Bead TACE. Further studies are warranted to evaluate the efficacy of DC Bead TACE and sorafenib in HCC patients accompanying PVTT.
Transarterial radioembolization (TARE) is a form of catheter-directed, selective internal radiation therapy which delivers 25-32.5 μm sized microspheres loaded with high-energy radioisotope of yttrium-90 (90Y), pure β-ray, into tumor tissue[28]. Tumoricidal radiation doses are delivered with minimal toxicity to functional liver parenchyma and minimal alteration in vascularity with TARE[29,30]. However, there is only microembolization (minimal to moderate embolization)[8,31]. Studies report improved median OS (7-41.6 mo) in BCLC B to C HCC patients following TARE and objective response rates (20%-77%)[32]. Although previous studies reported comparable efficacy for TARE and TACE in terms of tumor response and OS, patients receiving TARE tended to experience fewer complications and fewer days in hospital (typically 0-1.7 d with TARE compared to 1.8-6 d with TACE)[33-36], which are important quality-of-life considerations in patients with unresectable HCC.
Moreover, there is increasing evidence that TARE can be delivered safely and effectively in suitable HCC patients with PVTT, with several studies reporting median OS rates of approximately 10 mo following the procedure in these patients[34,37-42]. Again the extent of PVTT affected survival outcome. Salem et al[36] reported that the median OS for patients with Child class A (without extrahepatic spread) ranged from a median 16.6 mo for patients with branch involvement to 7.4 mo for those with Vp4. Median OS in patients accompanying PVTT and Child class B was only 5.6 mo. The risk of death due to underlying liver disease rather than tumor progression becomes a factor in Child class B patients as evidenced by a median OS of only 7.7 mo in the total Child class B cohort despite a TTP of 8.4 mo[43]. Overall, the tolerability of TARE in patients with PVTT appeared to be comparable to that in those without PVTT[37,38,41,42]. When safety issues were specifically investigated, liver decompensation was not observed in the 2-mo period following TARE among HCC patients with PVTT[39], and clinical and laboratory adverse events in the 90-d period after TARE were not more frequent in BCLC C HCC patients than in BCLC A to B HCC patients[38]. Recently, Gramenzi et al[44] performed a cohort study directly comparing TARE and sorafenib in patients with intermediate-locally advanced HCC. Median OS of the two groups were comparable even after matching for independent prognostic factors including PVTT: sorafenib group (median OS: 13.1 mo; 95%CI: 1.2-25.9) and TARE group (median OS: 11.2 mo; 95%CI: 6.7-15.7).
The most studies regarding hepatic arterial infusion (HAI) used a combined regimen of cisplatin and 5-fluorouracil. The best results were reported by Ando et al[45]. The 5-year OS rate was 11.0% and the median OS was 10.2 mo in that study involving 48 patients treated with Vp2 to Vp4 by HAI with cisplatin plus 5-fluorouracil.
In a small sample sized retrospective study (n = 13), radiofrequency ablation could ablaze both single intrahepatic medium-sized (3.7-5 cm) HCCs and the accompanying Vp4 with high efficacy and safety. The 3-year cumulative survival rate was 77%. There were no major adverse events. Mild ascites and elevated transaminase levels were observed in only three patients[46].
In a retrospective study, Lu et al[47] evaluated the application of percutaneous laser ablation as a treatment for PVTT in 108 patients and demonstrated that 3 years survival rate was 22.38%.
Most patients with HCC with Vp4 are considered technically unsuitable for curative resection, and the presence of PVTT is usually considered a contraindication for liver transplantation due to higher tumor recurrence rates[8]. Surgical resection in HCC patients accompanying PVTT is rare in Occidental area where the BCLC staging system which regards PVTT as a contraindication for surgery is endorsed[8]. However, throughout Oriental area, operation is considered a potentially curative treatment in suitable patients with PVTT as reflected in the consensus recommendations of Asia-Pacific Association for the Study of the Liver[11], although only about 10% of patients undergoing surgery have PVTT[48,49]. Surgical resection in these patients may improve portal venous pressure, liver function, quality of life and survival[8]. The range and position of PVTT significantly affect the potential clinical results following resection[8]. Previous studies have shown that HCC patients accompanying Vp2-Vp3 have better clinical outcomes after resection compared to those with Vp4 or beyond (Table 1)[48-50]. Surgical resection provided survival gains for patients with resectable HCC accompanying PVTT compared with TACE: the 1-, 3-, and 5-year OS rates were 42.0%, 14.1%, and 11.1% for the surgical group and 37.8%, 7.3%, and 0.5% for the TACE group, respectively (P < 0.001)[51]. A sub-group analysis by the PVTT type identified increased survival in the surgical group compared with the TACE group in patients accompanying type I PVTT (Vp1-Vp2) or type II PVTT (Vp3) (P < 0.001, P = 0.002, respectively)[51]. However, there were no significant differences in OS between the resection group and the TACE group for patients accompanying type III PVTT (Vp4) and type IV PVTT (tumor thrombi involving the superior mesenteric vein) (P = 0.541, P = 0.371, respectively)[51]. In this study, after resection, there was only one postoperative in-hospital mortality caused by postoperative hepatic failure (0.5%), and the major complication rate was 4.0% (8 of 201). If PVTT is not stick to the portal vein wall, total thrombectomy is possible. However, when the PVTT is adhered to the wall of the portal vein, there is a high chance of intramural invasion of HCC cells into the vessel wall on pathological examination after resection[52]. Therefore, in case of Vp4, the prognosis is extremely poor if the involved wall of portal vein is not resected. Although PVTT is generally considered a contraindication to liver transplantation, some centers have reported their positive results for transplant in the setting of gross vascular invasion. Xu et al[53] performed a study involving 24 patients undergoing liver transplantation for HCC accompanying PVTT (10 at main trunk, 10 at right branch, and 4 at left branch) and demonstrated a 6-mo, 1-year, and 2-year OS of 66.7%, 29.5%, and 23.6%, respectively.
Advances in technology, including three-dimensional conformal radiotherapy, proton beam radiotherapy and stereotactic body radiosurgery, have allowed selective delivery of increased radiation doses to tumors with minimal doses to normal tissue[54]. A number of mostly retrospective studies have examined the use of these new technologies in selected patients accompanying PVTT: median OS (6.7-11 mo), and 1-, 2-, and 5-year survival rates (30%-40%, 20%-30%, and 5.1%-24%, respectively)[55-61]. In a recent retrospective study assessing radiotherapy and surgical resection in 371 resectable HCC patients accompanying PVTT enrolled from two tertiary referral centers, the median OS was 12.3 mo for radiotherapy (n = 185) and 10.0 mo for resection (n = 186). The 1-, 2-, and 3-year OS were 51.6%, 28.4%, and 19.9% for radiotherapy group and 40.1%, 17.0%, and 13.6% for surgical group, respectively (P = 0.029)[62]. More recently, Nakazawa et al[63] did a retrospective study comparing the survival benefits of sorafenib vs radiotherapy in unresectable HCC patients accompanying PVTT (Vp3 or Vp4). Median OS did not differ significantly between the sorafenib and the radiotherapy group (4.3 mo vs 5.9 mo, respectively; P = 0.115)[63]. However, after propensity score matching (n = 28 per group), better median OS was noted in the radiotherapy than in the sorafenib group (10.9 mo vs 4.8 mo, respectively; P = 0.025)[63]. In the sorafenib group, 90% (25 of 28) patients permanently discontinued sorafenib owing to disease progression (n = 10) or adverse events (n = 15). However, there was no high-grade (grade ≥ 3) gastrointestinal or hepatic toxicity in the radiotherapy group. Future large scale prospective studies are warranted to approve the results of these retrospective studies.
Zhu et al[64] conducted a retrospective study comparing the efficacy and safety of TACE plus sorafenib in 91 HCC patients accompanying PVTT (46 TACE-sorafenib vs 45 TACE alone). TACE plus sorafenib showed significant survival benefits over TACE alone in patients with Vp3 (median OS, 13 mo vs 6 mo; P = 0.002) or Vp1-2 (median OS, 15 mo vs 10 mo; P = 0.003). However, the control arm of this study was TACE alone instead of sorafenib alone. A randomized, controlled phase III trial of sorafenib with or without conventional TACE in patients with advanced HCC is recruiting participants (NCT01829035). The result of this study is awaited to answer whether TACE, as a powerful complimentary armament for sorafenib, could be allowed for HCC patients accompanying PVTT.
The recent advances with a co-treatment modality of TACE combined with radiotherapy have demonstrated superior results over TACE alone[65]. In addition, the survival benefit has been reported in patients accompanying PVTT who have been treated with TACE plus radiotherapy[66-68]. Recently, Cho et al[69] conducted a retrospective study comparing TACE combined with radiotherapy (n = 67) with sorafenib (n = 49) in 116 patients accompanying PVTT and demonstrated that OS in the TACE plus radiotherapy group was significantly prolonged over the sorafenib group (14.1 mo vs 3.3 mo, P < 0.001). Even in the matched cohort by propensity score, the TACE combined with radiotherapy group demonstrated extended OS over the sorafenib group (6.7 mo vs 3.1 mo, P < 0.001)[69].
There have been several studies of surgical resection-based multimodality treatment including surgical resection after TACE; surgical resection followed by TACE, HAI, and portal vein infusion chemotherapy; 90Y plus doxorubicin or preoperative intravenous chemotherapy with doxorubicin, cisplatin and 5-fluorouracil plus subcutaneous interferon-α (PIAF); postoperative percutaneous isolated hepatic perfusion; surgical resection followed by interferon with 5-fluorouracil; and surgical resection after radiotherapy. The median OS after surgical resection-based multidisciplinary treatments ranged from 13.0 to 22.1 mo, implying that multimodality therapy contributed to prolonged long-term survival[70-77]. In a controlled trial by Peng et al[77], 126 HCC patients accompanying PVTT (Vp3-4) were randomized into TACE after surgical resection (TACE group) or surgical resection alone (control group). The median OS was better in the TACE group (13 mo) than in the control group (9 mo). The estimated survival rates for 1-, 3-, and 5 years were significantly improved in the TACE group (50.9%, 33.8%, and 21.5%; respectively) than in the control group (33.3%, 17.0%, and 8.5%, respectively; P = 0.0094). The available evidence shows that surgical resection-based multimodality treatments are effective and should be estimated in further trials.
For HCC patients accompanying PVTT with Child class B, portal hypertension, or Eastern Cooperative Oncology Group (ECOG) 2, sorafenib would be best option as recommended in BCLC guideline. For HCC patients accompanying PVTT with Child class C, portal hypertension, or ECOG > 2, we have to treat these patients with best supportive care. For HCC patients accompanying PVTT with Child class A, no portal hypertension, and ECOG 0-1, we could treat these patients with individualized treatment plans, as follows: (1) Single HCC (≤ 2cm) with PVTT: In this setting, we could consider surgical resection as best options other than sorafenib. Alternatively, TACE and external beam radiotherapy (EBRT) could be other good options; (2) Single HCC (> 2 cm) with PVTT: For single HCC larger than 2 cm with PVTT, we still consider surgical resection as best option for patients with resectable tumor, reserved hepatic function and sufficient post-operative remnant hepatic volume. If tumor size is 10 cm or less, TACE and EBRT could be alternative options. For single huge HCC larger than 10 cm with PVTT, sorafenib would be 1st line option; (3) Multiple (maximal tumor size ≤ 2 cm) with PVTT: If maximal tumor size is 2 cm or less, we could adopt TACE as best option for multiple HCC. Sorafenib would be another best option for these patients; and (4) Multiple (maximal tumor size > 2 cm) with PVTT: In this setting, sorafenib would be 1st line option. However, we could still consider TACE as alternative option if maximal tumor size is 10 cm or less and tumor extent ≤ 50% of liver volume.
Although direct appraisals of the clinical outcomes of treatment are inappropriate by the differences in the patients’ baseline characteristics (Table 2), in HCC patients accompanying PVTT, evidence from retrospective and prospective studies suggests that multidisciplinary approaches including TACE and/or radiotherapy, TARE, and surgical resection-based multimodal treatments in selected patients may provide better outcomes than sorafenib. For resectable single nodular HCC patients with PVTT, we could treat these patients with surgical resection as 1st line treatment if they have Child class A, no portal hypertension, and ECOG 0-1. TACE, EBRT, and sorafenib would be alternative treatment options for these patients. For multi-nodular HCC patients accompanying PVTT, we could treat these patients with TACE or sorafenib if they have Child class A, no portal hypertension, and ECOG 0-1. TACE would be 1st line if maximal tumor size is 2 cm or less and sorafenib would be 1st line if maximal tumor size is greater than 2 cm. For HCC patients accompanying PVTT with Child class B, portal hypertension, or ECOG 2, sorafenib would be best option. However, for HCC patients accompanying PVTT with Child class C, portal hypertension, or ECOG > 2, we should treat these patients with best supportive care as recommended in BCLC guideline. Given the modest survival gain of sorafenib, surgical resection-based multimodal treatments for resectable HCC accompanying PVTT and TACE-based appropriate combined therapies for unresectable HCC accompanying PVTT may enhance the clinical outcomes of HCC patients with PVTT.
| Indication | Advantages | Disadvantages | |
| Sorafenib | BCLC stage C | Showing survival benefit | Modest efficacy compared to placebo control |
| in infiltrative type HCC | Hand-foot skin reaction | ||
| TACE | Nodular type HCC up to Vp4 | Wide indication | Post TACE syndrome |
| Child A liver function | Potential risk of liver failure | ||
| TARE | Tumor extension ≤ 50% of liver volume | Down-staging allowing | Requiring additional lung shunt study |
| Unilobar | liver transplantation | due to the risk of lung injury | |
| Nodular type | |||
| Up to Vp4 | |||
| RFA | Single medium-sized HCCs (3-5 cm) | Less invasive | If the intraparenchymal tumor was not completely ablated by RFA, complete effects on the thrombus probably would not be produced |
| Surgery | Up to Vp4 | Less expensive technic | Invasive and expensive technic |
| Single medium-sized HCCs ( ≤ 7 cm) | Better outcomes than other patients | Potential risk of liver failure | |
| Up to Vp4 | with HCC who are BCLC stage C | ||
| No HV/IVC invasion | with Child A liver function | ||
| External beam | AFP ≤ 30 ng/mL | Combined to multimodal strategies | Potential risk of radiation induced liver disease |
| radiotherapy | Tumor extension ≤ 60% of liver volume | Potential risk of GI tract toxicities |
P- Reviewer: Huang CYF, Zhu PQ S- Editor: Ji FF L- Editor: A E- Editor: Liu SQ
| 1. | Beasley RP. Hepatitis B virus. The major etiology of hepatocellular carcinoma. Cancer. 1988;61:1942-1956. [PubMed] |
| 2. | Bosch FX, Ribes J, Cléries R, Díaz M. Epidemiology of hepatocellular carcinoma. Clin Liver Dis. 2005;9:191-211, v. [PubMed] |
| 3. | Cheung TK, Lai CL, Wong BC, Fung J, Yuen MF. Clinical features, biochemical parameters, and virological profiles of patients with hepatocellular carcinoma in Hong Kong. Aliment Pharmacol Ther. 2006;24:573-583. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 74] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 4. | Minagawa M, Makuuchi M. Treatment of hepatocellular carcinoma accompanied by portal vein tumor thrombus. World J Gastroenterol. 2006;12:7561-7567. [PubMed] |
| 5. | Llovet JM, Bustamante J, Castells A, Vilana R, Ayuso Mdel C, Sala M, Brú C, Rodés J, Bruix J. Natural history of untreated nonsurgical hepatocellular carcinoma: rationale for the design and evaluation of therapeutic trials. Hepatology. 1999;29:62-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 839] [Cited by in RCA: 907] [Article Influence: 34.9] [Reference Citation Analysis (1)] |
| 6. | Pirisi M, Avellini C, Fabris C, Scott C, Bardus P, Soardo G, Beltrami CA, Bartoli E. Portal vein thrombosis in hepatocellular carcinoma: age and sex distribution in an autopsy study. J Cancer Res Clin Oncol. 1998;124:397-400. [PubMed] |
| 7. | Schöniger-Hekele M, Müller C, Kutilek M, Oesterreicher C, Ferenci P, Gangl A. Hepatocellular carcinoma in Central Europe: prognostic features and survival. Gut. 2001;48:103-109. [PubMed] |
| 8. | Lau WY, Sangro B, Chen PJ, Cheng SQ, Chow P, Lee RC, Leung T, Han KH, Poon RT. Treatment for hepatocellular carcinoma with portal vein tumor thrombosis: the emerging role for radioembolization using yttrium-90. Oncology. 2013;84:311-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 126] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 9. | Katagiri S, Yamamoto M. Multidisciplinary treatments for hepatocellular carcinoma with major portal vein tumor thrombus. Surg Today. 2014;44:219-226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | Bruix J, Sherman M. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020-1022. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5972] [Cited by in RCA: 6573] [Article Influence: 469.5] [Reference Citation Analysis (1)] |
| 11. | Omata M, Lesmana LA, Tateishi R, Chen PJ, Lin SM, Yoshida H, Kudo M, Lee JM, Choi BI, Poon RT. Asian Pacific Association for the Study of the Liver consensus recommendations on hepatocellular carcinoma. Hepatol Int. 2010;4:439-474. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 797] [Cited by in RCA: 841] [Article Influence: 56.1] [Reference Citation Analysis (0)] |
| 12. | Jelic S, Sotiropoulos GC. Hepatocellular carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2010;21 Suppl 5:v59-v64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 130] [Cited by in RCA: 165] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 13. | Wilhelm SM, Carter C, Tang L, Wilkie D, McNabola A, Rong H, Chen C, Zhang X, Vincent P, McHugh M. BAY 43-9006 exhibits broad spectrum oral antitumor activity and targets the RAF/MEK/ERK pathway and receptor tyrosine kinases involved in tumor progression and angiogenesis. Cancer Res. 2004;64:7099-7109. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2966] [Cited by in RCA: 3147] [Article Influence: 149.9] [Reference Citation Analysis (0)] |
| 14. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10266] [Article Influence: 603.9] [Reference Citation Analysis (2)] |
| 15. | Cheng AL, Kang YK, Chen Z, Tsao CJ, Qin S, Kim JS, Luo R, Feng J, Ye S, Yang TS. Efficacy and safety of sorafenib in patients in the Asia-Pacific region with advanced hepatocellular carcinoma: a phase III randomised, double-blind, placebo-controlled trial. Lancet Oncol. 2009;10:25-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3854] [Cited by in RCA: 4649] [Article Influence: 273.5] [Reference Citation Analysis (0)] |
| 16. | Cheng AL, Guan Z, Chen Z, Tsao CJ, Qin S, Kim JS, Yang TS, Tak WY, Pan H, Yu S. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma according to baseline status: subset analyses of the phase III Sorafenib Asia-Pacific trial. Eur J Cancer. 2012;48:1452-1465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 225] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 17. | Bruix J, Raoul JL, Sherman M, Mazzaferro V, Bolondi L, Craxi A, Galle PR, Santoro A, Beaugrand M, Sangiovanni A. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma: subanalyses of a phase III trial. J Hepatol. 2012;57:821-829. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 664] [Cited by in RCA: 653] [Article Influence: 50.2] [Reference Citation Analysis (0)] |
| 18. | Llovet JM, Real MI, Montaña X, Planas R, Coll S, Aponte J, Ayuso C, Sala M, Muchart J, Solà R. Arterial embolisation or chemoembolisation versus symptomatic treatment in patients with unresectable hepatocellular carcinoma: a randomised controlled trial. Lancet. 2002;359:1734-1739. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2502] [Cited by in RCA: 2611] [Article Influence: 113.5] [Reference Citation Analysis (0)] |
| 19. | Lo CM, Ngan H, Tso WK, Liu CL, Lam CM, Poon RT, Fan ST, Wong J. Randomized controlled trial of transarterial lipiodol chemoembolization for unresectable hepatocellular carcinoma. Hepatology. 2002;35:1164-1171. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1904] [Cited by in RCA: 1987] [Article Influence: 86.4] [Reference Citation Analysis (0)] |
| 20. | Llovet JM, Bruix J. Systematic review of randomized trials for unresectable hepatocellular carcinoma: Chemoembolization improves survival. Hepatology. 2003;37:429-442. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2207] [Cited by in RCA: 2270] [Article Influence: 103.2] [Reference Citation Analysis (0)] |
| 21. | Lee HS, Kim JS, Choi IJ, Chung JW, Park JH, Kim CY. The safety and efficacy of transcatheter arterial chemoembolization in the treatment of patients with hepatocellular carcinoma and main portal vein obstruction. A prospective controlled study. Cancer. 1997;79:2087-2094. [PubMed] |
| 22. | Luo J, Guo RP, Lai EC, Zhang YJ, Lau WY, Chen MS, Shi M. Transarterial chemoembolization for unresectable hepatocellular carcinoma with portal vein tumor thrombosis: a prospective comparative study. Ann Surg Oncol. 2011;18:413-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 227] [Cited by in RCA: 273] [Article Influence: 18.2] [Reference Citation Analysis (0)] |
| 23. | Chung GE, Lee JH, Kim HY, Hwang SY, Kim JS, Chung JW, Yoon JH, Lee HS, Kim YJ. Transarterial chemoembolization can be safely performed in patients with hepatocellular carcinoma invading the main portal vein and may improve the overall survival. Radiology. 2011;258:627-634. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 189] [Cited by in RCA: 250] [Article Influence: 17.9] [Reference Citation Analysis (0)] |
| 24. | Xue TC, Xie XY, Zhang L, Yin X, Zhang BH, Ren ZG. Transarterial chemoembolization for hepatocellular carcinoma with portal vein tumor thrombus: a meta-analysis. BMC Gastroenterol. 2013;13:60. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 95] [Cited by in RCA: 133] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 25. | Pinter M, Hucke F, Graziadei I, Vogel W, Maieron A, Königsberg R, Stauber R, Grünberger B, Müller C, Kölblinger C. Advanced-stage hepatocellular carcinoma: transarterial chemoembolization versus sorafenib. Radiology. 2012;263:590-599. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 140] [Cited by in RCA: 165] [Article Influence: 12.7] [Reference Citation Analysis (0)] |
| 26. | Sellers MT, Huggins S, Kegley K, Pollinger HS, Shrestha R, Johnson MW, Stein LL, Panjala C, Tan M, Arepally A. Multivariate analysis of prognostic factors for survival following doxorubicin-eluting bead transarterial chemoembolization for hepatocellular carcinoma. J Vasc Interv Radiol. 2013;24:647-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 15] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 27. | Song MJ, Chun HJ, Song do S, Kim HY, Yoo SH, Park CH, Bae SH, Choi JY, Chang UI, Yang JM. Comparative study between doxorubicin-eluting beads and conventional transarterial chemoembolization for treatment of hepatocellular carcinoma. J Hepatol. 2012;57:1244-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 193] [Cited by in RCA: 196] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 28. | Sangro B, Iñarrairaegui M, Bilbao JI. Radioembolization for hepatocellular carcinoma. J Hepatol. 2012;56:464-473. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 202] [Cited by in RCA: 231] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 29. | Liapi E, Geschwind JF. Intra-arterial therapies for hepatocellular carcinoma: where do we stand? Ann Surg Oncol. 2010;17:1234-1246. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 61] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 30. | Kennedy A, Nag S, Salem R, Murthy R, McEwan AJ, Nutting C, Benson A, Espat J, Bilbao JI, Sharma RA. Recommendations for radioembolization of hepatic malignancies using yttrium-90 microsphere brachytherapy: a consensus panel report from the radioembolization brachytherapy oncology consortium. Int J Radiat Oncol Biol Phys. 2007;68:13-23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 506] [Cited by in RCA: 512] [Article Influence: 28.4] [Reference Citation Analysis (0)] |
| 31. | Sato K, Lewandowski RJ, Bui JT, Omary R, Hunter RD, Kulik L, Mulcahy M, Liu D, Chrisman H, Resnick S. Treatment of unresectable primary and metastatic liver cancer with yttrium-90 microspheres (TheraSphere): assessment of hepatic arterial embolization. Cardiovasc Intervent Radiol. 2006;29:522-529. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 183] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 32. | Sangro B, Salem R, Kennedy A, Coldwell D, Wasan H. Radioembolization for hepatocellular carcinoma: a review of the evidence and treatment recommendations. Am J Clin Oncol. 2011;34:422-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 33. | Lewandowski RJ, Kulik LM, Riaz A, Senthilnathan S, Mulcahy MF, Ryu RK, Ibrahim SM, Sato KT, Baker T, Miller FH. A comparative analysis of transarterial downstaging for hepatocellular carcinoma: chemoembolization versus radioembolization. Am J Transplant. 2009;9:1920-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 437] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 34. | Kooby DA, Egnatashvili V, Srinivasan S, Chamsuddin A, Delman KA, Kauh J, Staley CA, Kim HS. Comparison of yttrium-90 radioembolization and transcatheter arterial chemoembolization for the treatment of unresectable hepatocellular carcinoma. J Vasc Interv Radiol. 2010;21:224-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 135] [Cited by in RCA: 149] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 35. | Carr BI, Kondragunta V, Buch SC, Branch RA. Therapeutic equivalence in survival for hepatic arterial chemoembolization and yttrium 90 microsphere treatments in unresectable hepatocellular carcinoma: a two-cohort study. Cancer. 2010;116:1305-1314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 148] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 36. | Salem R, Lewandowski RJ, Kulik L, Wang E, Riaz A, Ryu RK, Sato KT, Gupta R, Nikolaidis P, Miller FH. Radioembolization results in longer time-to-progression and reduced toxicity compared with chemoembolization in patients with hepatocellular carcinoma. Gastroenterology. 2011;140:497-507.e2. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 498] [Cited by in RCA: 504] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 37. | Hilgard P, Hamami M, Fouly AE, Scherag A, Müller S, Ertle J, Heusner T, Cicinnati VR, Paul A, Bockisch A. Radioembolization with yttrium-90 glass microspheres in hepatocellular carcinoma: European experience on safety and long-term survival. Hepatology. 2010;52:1741-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 316] [Cited by in RCA: 353] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 38. | Sangro B, Carpanese L, Cianni R, Golfieri R, Gasparini D, Ezziddin S, Paprottka PM, Fiore F, Van Buskirk M, Bilbao JI. Survival after yttrium-90 resin microsphere radioembolization of hepatocellular carcinoma across Barcelona clinic liver cancer stages: a European evaluation. Hepatology. 2011;54:868-878. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 464] [Cited by in RCA: 505] [Article Influence: 36.1] [Reference Citation Analysis (0)] |
| 39. | Iñarrairaegui M, Thurston KG, Bilbao JI, D’Avola D, Rodriguez M, Arbizu J, Martinez-Cuesta A, Sangro B. Radioembolization with use of yttrium-90 resin microspheres in patients with hepatocellular carcinoma and portal vein thrombosis. J Vasc Interv Radiol. 2010;21:1205-1212. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 110] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 40. | Tsai AL, Burke CT, Kennedy AS, Moore DT, Mauro MA, Dixon RD, Stavas JM, Bernard SA, Khandani AH, O’Neil BH. Use of yttrium-90 microspheres in patients with advanced hepatocellular carcinoma and portal vein thrombosis. J Vasc Interv Radiol. 2010;21:1377-1384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 42] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 41. | Woodall CE, Scoggins CR, Ellis SF, Tatum CM, Hahl MJ, Ravindra KV, McMasters KM, Martin RC. Is selective internal radioembolization safe and effective for patients with inoperable hepatocellular carcinoma and venous thrombosis? J Am Coll Surg. 2009;208:375-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 55] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 42. | Kulik LM, Carr BI, Mulcahy MF, Lewandowski RJ, Atassi B, Ryu RK, Sato KT, Benson A, Nemcek AA, Gates VL. Safety and efficacy of 90Y radiotherapy for hepatocellular carcinoma with and without portal vein thrombosis. Hepatology. 2008;47:71-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 457] [Article Influence: 26.9] [Reference Citation Analysis (0)] |
| 43. | Salem R, Lewandowski RJ, Mulcahy MF, Riaz A, Ryu RK, Ibrahim S, Atassi B, Baker T, Gates V, Miller FH. Radioembolization for hepatocellular carcinoma using Yttrium-90 microspheres: a comprehensive report of long-term outcomes. Gastroenterology. 2010;138:52-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 738] [Cited by in RCA: 781] [Article Influence: 52.1] [Reference Citation Analysis (0)] |
| 44. | Gramenzi A, Golfieri R, Mosconi C, Cappelli A, Granito A, Cucchetti A, Marinelli S, Pettinato C, Erroi V, Fiumana S. Yttrium-90 radioembolization vs sorafenib for intermediate-locally advanced hepatocellular carcinoma: a cohort study with propensity score analysis. Liver Int. 2015;35:1036-1047. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 89] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 45. | Ando E, Tanaka M, Yamashita F, Kuromatsu R, Yutani S, Fukumori K, Sumie S, Yano Y, Okuda K, Sata M. Hepatic arterial infusion chemotherapy for advanced hepatocellular carcinoma with portal vein tumor thrombosis: analysis of 48 cases. Cancer. 2002;95:588-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 241] [Cited by in RCA: 259] [Article Influence: 11.3] [Reference Citation Analysis (0)] |
| 46. | Giorgio A, Di Sarno A, de Stefano G, Farella N, Scognamiglio U, de Stefano M, Giorgio V. Hepatocellular carcinoma with cirrhosis: are patients with neoplastic main portal vein invasion eligible for percutaneous radiofrequency ablation of both the nodule and the portal venous tumor thrombus? AJR Am J Roentgenol. 2009;193:948-954. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 16] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 47. | Lu ZH, Shen F, Yan ZL, Li J, Yang JH, Zong M, Shi LH, Wu MC. Treatment of portal vein tumor thrombus of hepatocellular carcinoma with percutaneous laser ablation. J Cancer Res Clin Oncol. 2009;135:783-789. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 15] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 48. | Chen XP, Qiu FZ, Wu ZD, Zhang ZW, Huang ZY, Chen YF, Zhang BX, He SQ, Zhang WG. Effects of location and extension of portal vein tumor thrombus on long-term outcomes of surgical treatment for hepatocellular carcinoma. Ann Surg Oncol. 2006;13:940-946. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 110] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 49. | Shi J, Lai EC, Li N, Guo WX, Xue J, Lau WY, Wu MC, Cheng SQ. Surgical treatment of hepatocellular carcinoma with portal vein tumor thrombus. Ann Surg Oncol. 2010;17:2073-2080. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 222] [Article Influence: 14.8] [Reference Citation Analysis (0)] |
| 50. | Lin DX, Zhang QY, Li X, Ye QW, Lin F, Li LL. An aggressive approach leads to improved survival in hepatocellular carcinoma patients with portal vein tumor thrombus. J Cancer Res Clin Oncol. 2011;137:139-149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 45] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 51. | Peng ZW, Guo RP, Zhang YJ, Lin XJ, Chen MS, Lau WY. Hepatic resection versus transcatheter arterial chemoembolization for the treatment of hepatocellular carcinoma with portal vein tumor thrombus. Cancer. 2012;118:4725-4736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 131] [Article Influence: 10.1] [Reference Citation Analysis (0)] |
| 52. | Wu CC, Hsieh SR, Chen JT, Ho WL, Lin MC, Yeh DC, Liu TJ, P’eng FK. An appraisal of liver and portal vein resection for hepatocellular carcinoma with tumor thrombi extending to portal bifurcation. Arch Surg. 2000;135:1273-1279. [PubMed] |
| 53. | Xu X, Zheng SS, Liang TB, Wang WL, Jin J, Shen Y, Wu J, Yu J. Orthotopic liver transplantation for patients with hepatocellular carcinoma complicated by portal vein tumor thrombi. Hepatobiliary Pancreat Dis Int. 2004;3:341-344. [PubMed] |
| 54. | Hawkins MA, Dawson LA. Radiation therapy for hepatocellular carcinoma: from palliation to cure. Cancer. 2006;106:1653-1663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 183] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 55. | Lin CS, Jen YM, Chiu SY, Hwang JM, Chao HL, Lin HY, Shum WY. Treatment of portal vein tumor thrombosis of hepatoma patients with either stereotactic radiotherapy or three-dimensional conformal radiotherapy. Jpn J Clin Oncol. 2006;36:212-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 65] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 56. | Shirai S, Sato M, Suwa K, Kishi K, Shimono C, Kawai N, Tanihata H, Minamiguchi H, Nakai M. Single photon emission computed tomography-based three-dimensional conformal radiotherapy for hepatocellular carcinoma with portal vein tumor thrombus. Int J Radiat Oncol Biol Phys. 2009;73:824-831. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 17] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 57. | Hata M, Tokuuye K, Sugahara S, Kagei K, Igaki H, Hashimoto T, Ohara K, Matsuzaki Y, Tanaka N, Akine Y. Proton beam therapy for hepatocellular carcinoma with portal vein tumor thrombus. Cancer. 2005;104:794-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 85] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 58. | Nakagawa K, Yamashita H, Shiraishi K, Nakamura N, Tago M, Igaki H, Hosoi Y, Shiina S, Omata M, Makuuchi M. Radiation therapy for portal venous invasion by hepatocellular carcinoma. World J Gastroenterol. 2005;11:7237-7241. [PubMed] |
| 59. | Zeng ZC, Fan J, Tang ZY, Zhou J, Qin LX, Wang JH, Sun HC, Wang BL, Zhang JY, Jiang GL. A comparison of treatment combinations with and without radiotherapy for hepatocellular carcinoma with portal vein and/or inferior vena cava tumor thrombus. Int J Radiat Oncol Biol Phys. 2005;61:432-443. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 144] [Cited by in RCA: 142] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 60. | Kim DY, Park W, Lim DH, Lee JH, Yoo BC, Paik SW, Kho KC, Kim TH, Ahn YC, Huh SJ. Three-dimensional conformal radiotherapy for portal vein thrombosis of hepatocellular carcinoma. Cancer. 2005;103:2419-2426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 152] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 61. | Zhang FJ, Li CX, Jiao DC, Zhang NH, Wu PH, Duan GF, Wu YX. CT guided 125iodine seed implantation for portal vein tumor thrombus in primary hepatocellular carcinoma. Chin Med J (Engl). 2008;121:2410-2414. [PubMed] |
| 62. | Tang QH, Li AJ, Yang GM, Lai EC, Zhou WP, Jiang ZH, Lau WY, Wu MC. Surgical resection versus conformal radiotherapy combined with TACE for resectable hepatocellular carcinoma with portal vein tumor thrombus: a comparative study. World J Surg. 2013;37:1362-1370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 63. | Nakazawa T, Hidaka H, Shibuya A, Okuwaki Y, Tanaka Y, Takada J, Minamino T, Watanabe M, Kokubu S, Koizumi W. Overall survival in response to sorafenib versus radiotherapy in unresectable hepatocellular carcinoma with major portal vein tumor thrombosis: propensity score analysis. BMC Gastroenterol. 2014;14:84. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 98] [Article Influence: 8.9] [Reference Citation Analysis (0)] |
| 64. | Zhu K, Chen J, Lai L, Meng X, Zhou B, Huang W, Cai M, Shan H. Hepatocellular carcinoma with portal vein tumor thrombus: treatment with transarterial chemoembolization combined with sorafenib--a retrospective controlled study. Radiology. 2014;272:284-293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 136] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 65. | Meng MB, Cui YL, Lu Y, She B, Chen Y, Guan YS, Zhang RM. Transcatheter arterial chemoembolization in combination with radiotherapy for unresectable hepatocellular carcinoma: a systematic review and meta-analysis. Radiother Oncol. 2009;92:184-194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 103] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 66. | Yoon SM, Lim YS, Won HJ, Kim JH, Kim KM, Lee HC, Chung YH, Lee YS, Lee SG, Park JH. Radiotherapy plus transarterial chemoembolization for hepatocellular carcinoma invading the portal vein: long-term patient outcomes. Int J Radiat Oncol Biol Phys. 2012;82:2004-2011. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 156] [Cited by in RCA: 180] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 67. | Shim SJ, Seong J, Han KH, Chon CY, Suh CO, Lee JT. Local radiotherapy as a complement to incomplete transcatheter arterial chemoembolization in locally advanced hepatocellular carcinoma. Liver Int. 2005;25:1189-1196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 96] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 68. | Kim KM, Kim JH, Park IS, Ko GY, Yoon HK, Sung KB, Lim YS, Lee HC, Chung YH, Lee YS. Reappraisal of repeated transarterial chemoembolization in the treatment of hepatocellular carcinoma with portal vein invasion. J Gastroenterol Hepatol. 2009;24:806-814. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 117] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 69. | Cho JY, Paik YH, Park HC, Yu JI, Sohn W, Gwak GY, Choi MS, Lee JH, Koh KC, Paik SW. The feasibility of combined transcatheter arterial chemoembolization and radiotherapy for advanced hepatocellular carcinoma. Liver Int. 2014;34:795-801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 47] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 70. | Minagawa M, Makuuchi M, Takayama T, Ohtomo K. Selection criteria for hepatectomy in patients with hepatocellular carcinoma and portal vein tumor thrombus. Ann Surg. 2001;233:379-384. [PubMed] |
| 71. | Fan J, Wu ZQ, Tang ZY, Zhou J, Qiu SJ, Ma ZC, Zhou XD, Ye SL. Multimodality treatment in hepatocellular carcinoma patients with tumor thrombi in portal vein. World J Gastroenterol. 2001;7:28-32. [PubMed] |
| 72. | Fukuda S, Okuda K, Imamura M, Imamura I, Eriguchi N, Aoyagi S. Surgical resection combined with chemotherapy for advanced hepatocellular carcinoma with tumor thrombus: report of 19 cases. Surgery. 2002;131:300-310. [PubMed] |
| 73. | Lau WY, Ho SK, Yu SC, Lai EC, Liew CT, Leung TW. Salvage surgery following downstaging of unresectable hepatocellular carcinoma. Ann Surg. 2004;240:299-305. [PubMed] |
| 74. | Ku Y, Iwasaki T, Tominaga M, Fukumoto T, Takahashi T, Kido M, Ogata S, Takahashi M, Kuroda Y, Matsumoto S. Reductive surgery plus percutaneous isolated hepatic perfusion for multiple advanced hepatocellular carcinoma. Ann Surg. 2004;239:53-60. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 40] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 75. | Nagano H, Miyamoto A, Wada H, Ota H, Marubashi S, Takeda Y, Dono K, Umeshita K, Sakon M, Monden M. Interferon-alpha and 5-fluorouracil combination therapy after palliative hepatic resection in patients with advanced hepatocellular carcinoma, portal venous tumor thrombus in the major trunk, and multiple nodules. Cancer. 2007;110:2493-2501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 69] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 76. | Kamiyama T, Nakanishi K, Yokoo H, Tahara M, Nakagawa T, Kamachi H, Taguchi H, Shirato H, Matsushita M, Todo S. Efficacy of preoperative radiotherapy to portal vein tumor thrombus in the main trunk or first branch in patients with hepatocellular carcinoma. Int J Clin Oncol. 2007;12:363-368. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 37] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 77. | Peng BG, He Q, Li JP, Zhou F. Adjuvant transcatheter arterial chemoembolization improves efficacy of hepatectomy for patients with hepatocellular carcinoma and portal vein tumor thrombus. Am J Surg. 2009;198:313-318. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 130] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 78. | Chen JS, Wang Q, Chen XL, Huang XH, Liang LJ, Lei J, Huang JQ, Li DM, Cheng ZX. Clinicopathologic characteristics and surgical outcomes of hepatocellular carcinoma with portal vein tumor thrombosis. J Surg Res. 2012;175:243-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 79. | Matono R, Yoshiya S, Motomura T, Toshima T, Kayashima H, Masuda T, Yoshizumi T, Taketomi A, Shirabe K, Maehara Y. Factors linked to longterm survival of patients with hepatocellular carcinoma accompanied by tumour thrombus in the major portal vein after surgical resection. HPB (Oxford). 2012;14:247-253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 20] [Article Influence: 1.5] [Reference Citation Analysis (0)] |