Published online Nov 27, 2012. doi: 10.4254/wjh.v4.i11.319
Revised: September 6, 2012
Accepted: October 22, 2012
Published online: November 27, 2012
Pyogenic liver abscesses are rare but a life-threatening important condition. Dental procedures constitute only rare cases of pyogenic liver abscesses, with only a few cases in the literature. We report a patient with liver abscess following a dental procedure. A 74 years old diabetic male patient was admitted to our hospital with complaints of fatigue, 40 °C fever, rigors and right upper quadrant pain, 3-4 d after a dental procedure. Physical examination revealed fever and tenderness in the right upper quadrant. Laboratory examination revealed leucocytosis, elevated erythrocyte sedimentation rate and C-reactive protein and moderately elevated transaminases. An abscess was detected in radiological examination in the medial part of the left lobe of liver, neighboring the gall bladder. He was successfully treated with percutaneous abscess drainage and antibiotherapy.
- Citation: Gungor G, Biyik M, Polat H, Ciray H, Ozbek O, Demir A. Liver abscess after implantation of dental prosthesis. World J Hepatol 2012; 4(11): 319-321
- URL: https://www.wjgnet.com/1948-5182/full/v4/i11/319.htm
- DOI: https://dx.doi.org/10.4254/wjh.v4.i11.319
Pyogenic liver abscesses are uncommon but a life-threatening important condition. Pyogenic liver abscesses are caused by infections, usually originating from biliary or portal pathologies, gastrointestinal and subdiaphragmatic infections, traumas, sepsis (hematogenous route) or idiopathic.
Dental procedures and diseases are very rare etiologies of pyogenic liver abscesses and, to date, only a few cases of pyogenic liver abscess associated with dental procedures have been reported in the literature[1-5]. A case of hepatic abscess has also been reported that originated from the oral cavity other than dental diseases[6].
We report a patient with liver abscess following a dental procedure (implantation).
A 74 years old man underwent a dental prosthesis implantation approximately ten days ago. Three to four days after the dental prosthesis implantation, he suffered from malaise, abdominal pain in right upper quadrant and chills. He recoursed to the first medical center with 40 °C fever and increasing complaints. In the first hospital, liver abscess was suspected on ultrasonographic (USG) evaluation of the abdomen and with USG-guided drainage, this abscess material was sent for cultural analysis simultaneously with the blood culture. Intravenous empirical antibiotherapy with Ceftriaxone 1 gr bid + Metronidazole 500 mg bid was given in the first medical center. The patient recoursed to our center for treatment by his own desire.
He had a 3 years history of diabetes mellitus and used metformin irregularly. Physical examination revealed weakness, a blood pressure of 120/80 mmHg, a heart rate of 84 beats/min and a body temperature of 38.7 °C. His conjunctiva were pale. Tenderness was found in the right upper quadrant and other physical findings were unremarkable.
Laboratory examination revealed the following findings: hemoglobin was 11.2 g/dL; white blood cell count was 11.200 cells/mm³ with 87.6% neutrophils; serum transaminases and prothrombin time were mildly elevated; serum total bilirubin, alkaline phosphatase and gamma-glutamyl transferase levels were all within normal range; serum glucose was 256 mg/dL; glucosylated hemoglobin level was 6.8%; erythrocyte sedimentation rate was 69 mm/h; C-reactive protein level was 115 mg/L; serological tests for infection with hepatitis B, C, human immunodeficiency virus, brucella, salmonella and amebiasis were all negative; and urine analysis was unremarkable except for microalbuminuria. Chest radiograph was normal.
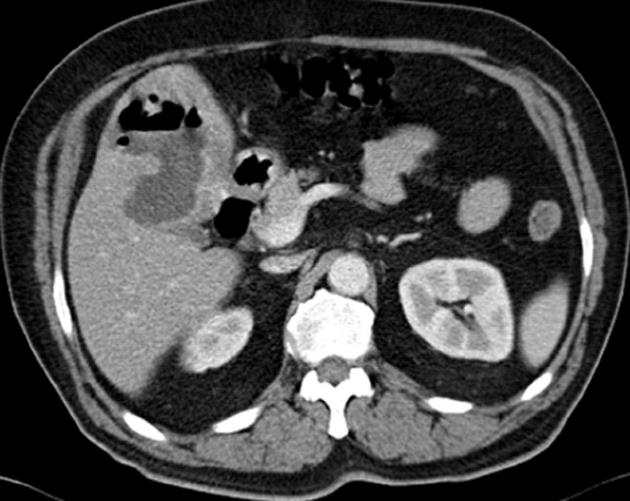
Ultrasonography of the abdomen revealed that the liver was steatotic and there was a single hypoechoic-heterogenous lesion 75 mm × 60 mm in diameter within the medial part of the left lobe, neighboring the gall bladder. Abdominal computed tomography (CT) scan showed no contrast involvement in the lesion after the contrast injection but showed involvement just around this lesion (Figure 1). Separately, the cortical cyst 10 mm in diameter was detected on the left kidney. Calibration of small and large intestinal segments and wall thickness were normal. Gall bladder and biliary tract were normal. No intraabdominal fluid collection was detected.
After diagnosis of liver abscess, the patient was treated with intravenous Imipenem 4 mg × 500 mg + Metronidazol 3 mg × 500 mg empirically. Hydration and subcutaneus insulin injections were applied. We learned that streptococcus subspecies was isolated from the abscess material culture and from the blood sample taken before empirical antibiotherapy. Urine culture was negative.
After that, USG-guided percutaneous drainage of the liver abscess was performed. A total of 400 mL foul-smelling purulent material was drained; 300 mL on the first day and 100 mL on the following day. His complaints improved after the drainage of the abscess and 14 d of antibiotherapy. The fever decreased, he was clinically well and the white blood cell count, CRP and sedimentation rate values returned to normal.
Control USG evaluation after 14 d showed almost complete resolution of the liver abscess. Levofloxacin 500 mg/d po for seven days was recommended after discharge from hospital.
In our case we demonstrated a liver abscess development with hematogenous route due to the dental procedure. Isolating the same microorganisms from both the blood sample and abscess material, the onset of his symptoms 3-4 d after the dental prosthesis implantation and no detection of another infection origin supported our opinion. This procedure might have caused bacteremia spreading to the liver via the hepatic artery.
Isolated pathogens from a liver abscess are usually gram negative bacterias like Escherichia Coli, Klebsiella, Pseudomonas; gram positive microorganisms like Streptococcus and Staphylococcus species; and anaerobic organisms like Fusibacterium, Bacteroides, Ameobas or fungal microorganisms[1].
Prognosis may be poor if diagnosis and treatment are delayed in pyogenic liver abscesses. CT scan and ultrasound-guided drainage of pyogenic liver abscesses are safe and effective methods of treatment[7]. Liver transplantation, diabetes and a history of malignancy are risk factors for pyogenic liver abscess[8].
Dental diseases or procedures are very rare etiological factors of liver abscesses. However, poor oral hygiene might also be an independent risk factor. Usually hematogenous spread of bacteria occurs after interventional treatment of dental disease.
Patients with increased risk of infection, such as immunosuppression, diabetes and malignancy, should be treated with prophylactic antibiotherapy during and after dental procedures. Appropriate oral care is clearly important, not only for dental, but also for systemic diseases.
Peer reviewer: Christian Philipp Selinger, MD, Salford Royal Hospital, 55 Miry Lane, Westhoughton, BL5 2HW Bolton, United Kingdom
S- Editor Song XX L- Editor Roemmele A E- Editor Li JY
| 1. | Tweedy CR, White WB. Multiple Fusobacterium nucleatum liver abscesses. Association with a persistent abnormality in humoral immune function. J Clin Gastroenterol. 1987;9:194-197. [PubMed] |
| 2. | Crippin JS, Wang KK. An unrecognized etiology for pyogenic hepatic abscesses in normal hosts: dental disease. Am J Gastroenterol. 1992;87:1740-1743. [PubMed] |
| 3. | Schiff E, Pick N, Oliven A, Odeh M. Multiple liver abscesses after dental treatment. J Clin Gastroenterol. 2003;36:369-371. [PubMed] |
| 4. | Kajiya T, Uemura T, Kajiya M, Kaname H, Hirano R, Uemura N, Tei C. Pyogenic liver abscess related to dental disease in an immunocompetent host. Intern Med. 2008;47:675-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 26] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 5. | Lei WY, Chang WH, Shih SC, Liu CJ, Shih CH. Pyogenic liver abscess with Prevotella species and Fusobacterium necrophorum as causative pathogens in an immunocompetent patient. J Formos Med Assoc. 2009;108:253-257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 6. | Wagner KW, Schön R, Schumacher M, Schmelzeisen R, Schulze D. Case report: brain and liver abscesses caused by oral infection with Streptococcus intermedius. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2006;102:e21-e23. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 36] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 7. | Zibari GB, Maguire S, Aultman DF, McMillan RW, McDonald JC. Pyogenic liver abscess. Surg Infect (Larchmt). 2000;1:15-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 34] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 8. | Kaplan GG, Gregson DB, Laupland KB. Population-based study of the epidemiology of and the risk factors for pyogenic liver abscess. Clin Gastroenterol Hepatol. 2004;2:1032-1038. [PubMed] |