Published online Jun 27, 2011. doi: 10.4254/wjh.v3.i6.142
Revised: May 9, 2011
Accepted: May 16, 2011
Published online: June 27, 2011
With the arrival of the new millennium, gastroenterologists have been faced with the problem of metabolic liver diseases associated with obesity. The active role of the liver in metabolism and inflammation make it a key organ in the war against the rapidly-spreading world-wide epidemic of obesity. Many lives and much money could be saved if the work of hepatologists led to the development of effective diagnostic and therapeutic strategies against this growing leader of cirrhosis.
- Citation: Balaban YH. A key problem and challenge for hepatology: Obesity-related metabolic liver diseases. World J Hepatol 2011; 3(6): 142-146
- URL: https://www.wjgnet.com/1948-5182/full/v3/i6/142.htm
- DOI: https://dx.doi.org/10.4254/wjh.v3.i6.142
The term “metabolic liver disease” classically implies rare disorders of metabolism causing liver diseases; such as hereditary hemachromatosis, Wilson’s disease or alpha-1 anti-trypsin deficiency. However, knowledge of lipid and glucose metabolism has increased considerably since obesity and its associated complications have been recognized as a world-wide epidemic. This necessitates a redefinitionof the concept of metabolic liver disease, which now covers common liver diseases accompanying metabolic syndrome[1,2]. The main component of metabolic syndrome is obesity (particularly visceral); others are glucose intolerance (impaired glucose tolerance test, impaired fasting glucose, and type 2 diabetes mellitus), hypertension, dyslipidaemia (hipertriglyceridemia, low HDL, increased LDL), and atherosclerotic cardiovascular disease. Since each of these components is characterized by insulin resistance, metabolic syndrome is also referred to as “insulin resistance syndrome”[3].
Fatty liver disease occurs if fat content exceeds 5% of the total weight of liver. It can develop either secondary to various factors (HCV, Wilson disease, drugs, alcohol, hipotiroidism, etc) or as a primary metabolic disorder, similar to diabetes and hypertension. Non-alcoholic fatty liver disease (NAFLD) associates the primary fatty liver diseases with a wide spectrum of histopathologic changes, ranging from simple steatosis to steatohepatitis. Non-alcoholic steatohepatitis (NASH) represents the most severe part of the NAFLD spectrum, in which hepatic steatosis is associated with hepatic inflammation. Since NAFLD is strongly associated with obesity and insulin resistance, it is classically identified as the hepatic component of metabolic syndrome. Recently Brunt EM et al, have strongly argued for a change in the nomenclature to “metabolic fatty liver disease” or “metabolic steatohepatitis”, in order to drop the negative definition of ‘‘non-alcoholic” and to recognize the likely causal role of insulin resistance in NAFLD[1].
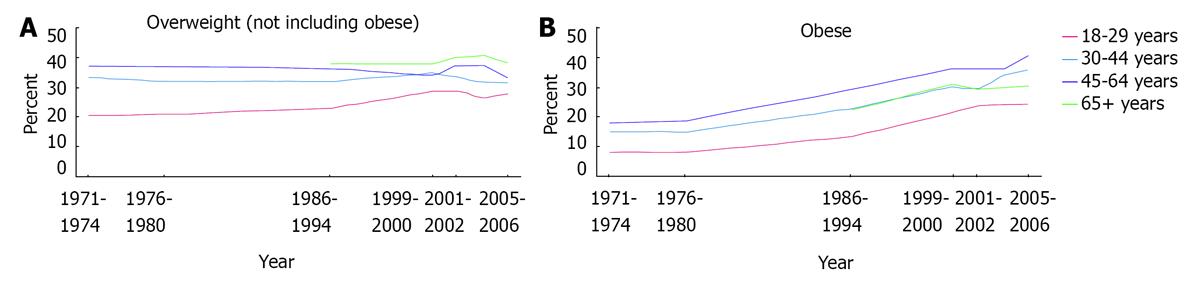
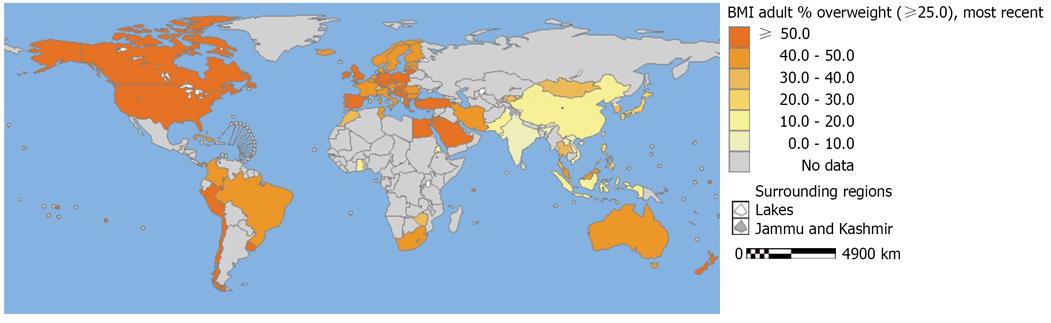
Obesity seems to be the major factor that moves the world away from the target of increased general health in the world population by 2020. Since weight-gain has historically been associated with wealth and prosperity in many cultures, in the past, obesity has been neglected as a public health problem. Moreover, during the last century, global eating habits have shifted from fiber-rich foods to an obesity prone Western diet[4]. Figure 1 presents a critical example of the steady and significant increase rates of obesity in the United States since 1970s[4]. Obesity has affected about one third of US adults aged 20 older in 2010. Similarly, the age-adjusted prevalence of clinically diagnosed diabetes has increased to 59 cases per 1000 population in 2008, from the 40 cases per 1000 in 1997. Because of these accelerating rates, one of the aims of the high-priority areas for motivating societal action in connection with health in the US, entitled “10 Leading Health Indicators and Focus Areas for Healthy People 2010”, is to decrease the incidence of diabetes to 25 cases per 1000 in 2020[5]. Estimates for the UK show that the rate of obesity in adults will rise from 23% in 2007 to 50% in 2050[7]. Although the epidemiological data is lacking for most parts of the world (Figure 2), available data warn that obesity is a global epidemic “disease” threatening the future health of the world[8].
The cost of obesity increases as more people in a population become obese. The total direct costs attributable to overweight and obesity have been calculated to reach $6.0 billion in Canada, a 15% increase from 2004 to 2006. The attributed ratios for overweight and obesity are approximately 35% and 65% of this $6.0 billion. The cost of obesity in 2006 corresponded to 4.1% of the total health expenditure in Canada[9].
The commonest chronic liver disease in the Western world and also in the Asia-Pacific Region in the new millennium is going to be NAFLD[10,11]. The sizes of world populations infected with HBV and HCV are respectively 350 and 180 million, whereas the current number of obese adults in the world is approximately 312 million. Furthermore, at least 2.5 billion people, which is one third of the estimated world population, are predicted to be obese in 2020[12]. The frequencies of NAFLD and NASH can be as high as 80% and 30% among the obese, so the expected number of patients with NAFLD and NASH in the world will be 2 billion and 800 million, respectively. Advanced fibrosis develops in 10%-30% of NAFLD patients, so 200-600 million people will face the risks of cirrhosis and hepatocellular carcinoma, both of which are well established complications of NAFLD. These numbers clearly identify metabolic liver disease as the leading cause of cirrhosis and its complications in the new millennium. Although the definition of NAFLD requires the exclusion of other etiologies of chronic liver disease, the high prevalence of clinical and histopathologic features of NAFLD causes problems in the diagnosis and management of liver disease. The metabolic effects underlying NAFLD are shown to exacerbate ongoing liver damage, particular in chronic hepatitis C, alcoholic liver disease and hemochromatosis[13,14,15]. From all these, it can be concluded that the hepatologists of 2020 will mainly be struggling with metabolic liver diseases and their complications.
NAFLD results from a series of liver insults, commonly referred to as the “multi-hit” hypothesis. Bearing in mind that this is a hypothesis that needs to be proven and is thus prone to changes, the main components this hypothesis are insulin resistance, lipotoxicity and adiposopathy. The underlying metabolic, genetic and/or environmental factors determine the degree of steatosis, inflammation and fibrosis in the liver, and, consequently, the severity of NAFLD in an individual. The NAFD is the underwater portion of the iceberg of obesity, due to high regeneration capacity of liver.
Obesity results from the metabolic need to deposit unused calories coming into the body. Excessive food intake and lack of physical exercise cause overload of the body with fat, which is accompanied by a specific etio-pathologic condition, insulin resistance. The adipose tissue in the body is redistributed from the subcutaneous area to the intra-abdominal/omental side. An ectopic fat accumulation occurs in muscle, liver, and heart, as well as “fat infiltration” in other tissues. The end results are cellular dysfunction, apoptosis and consequent pathologic stress on organs, such as heart failure, β-cell dysfunction in the pancreas, peripheral insulin resistance in both the liver and muscle tissue[16,17,18].
“Lipotoxicity” is the term that describes the deleterious effect of tissue fat accumulation on glucose metabolism. The insulin resistance accompanying obesity results in increased lipolysis in adipocytes, leading to an increase in plasma non-esterified fatty acids (NEFA). The high levels of plasma NEFA shift the substrate preference of mitochondrial oxidation from glucose to NEFA, and this diminishes the insulin secretor response of islet β-cells to glucose, leading to relative insulin insufficiency. On the other hand, the enhanced influx of NEFA into muscle and liver decreases fat oxidation at these sites. The increased intracellular fat causes peripheral insulin resistance through over-expression of lipoprotein lipase in the liver and muscle tissue. The over-expressed lipoprotein lipase increases hepatic fat content, induces severe hepatic insulin resistance and impairs hepatic insulin signaling. As a result, steatosis of the liver is established by means of this vicious cycle between lipotoxicity and insulin resistance[3,19] .
Adipokines are the cyokines secreted by adipocytes and mononuclear cells infiltrating adipose tissue. Adipocytokines travel to distant sites such as, muscular, liver, and arterial tissue; and affect metabolism and vascular functions negatively. These distant effects of adipokines are another form of lipotoxicity, called adiposopathy[3]. Visceral adipose tissue is particularly prone to inflammation; and it can contribute to hepatic fibro-inflammation by releasing inflammatory adipokines direct into the portal vein. Additionally, a systemic state of chronic inflammation is created by macrophages of visceral adipose tissue in obese and type 2 diabetic patients.
TLR4 on adipocytes and macrophages initiates insulin resistance and inflammatory response by functioning as a sensor for elevated NEFA concentrations. The detrimental complications of obesity-associated insulin resistance develop when the balance between adipokines of pro-inflammatory M1 macrophages (TNF-α, IL-6, and IL-1 β) and those of the anti-inflammatory M2 macrophages (IL-10, adiponectin and the IL-1 receptor antagonist), shift towards M1 macrophages. Pro-inflammatory adipokines exert insulin-resistance, in contrast to the insulin-sensitizing effect of anti-inflammatory ones. Similar to adipose tissue, activation of Kupffer cells promotes a pro-inflammatory Th1 immune response in a liver affected by NAFLD[18,20,21].
The peroxisome proliferator-activated receptors (PPARs) and liver X receptors (LXRs) are subgroups of the nuclear receptor superfamilies that regulate pro-inflammatory cytokine production by macrophages. Differentiation into M1 phenotype macrophages is inhibited by PPARγ signaling. The activation of PPARγ or PPARδvia IL-4, promotes polarization of macrophages toward M2 phenotype and thus avoids metabolic inflammation associated with insulin resistance. Another potential basis for the initiation of inflammation in obesity is endoplasmic reticulum (ER) stress. ER stress induces inflammatory signaling and in so doing, directly contributes to insulin resistance at insulin target cells. Therefore, dysregulation of macrophage-mediated inflammation by PPARs, LXRs and ER stress underlies the development of insulin resistance and inflammation in obesity[5,18,19].
NAFLD is a neglected component of metabolic syndrome due to the high regenerative capacity of the liver. However, the liver is the pathogenetic as well as the targeted organ of obesity-induced insulin resistance and lipotoxicity, which form two components of the vicious cycle ending in metabolic syndrome. Furthermore, NAFLD has been shown to precede other components of metabolic syndrome, such as diabetes, or atherosclerotic heart disease, by years[22]. Significant, but clinically silent liver disease can be detected during initial diagnosis of diabetes and is called “diabetic hepatopathy”[23,24]. All these facts indicate that diagnosis and successful treatment of NAFLD can have an impact not only on the liver but on the whole body.
The obesity epidemic poses enormous medical challenges for the near future. The patients have to be protected from deleterious psychological and social problems, as well as its systemic complications, which include atherosclerosis, diabetes and metabolic liver diseases. Treatment should mainly be based on prevention of obesity, since significant and sustained weight loss is very hard to achieve in real-life. As one of the systemic complications of obesity, treatment of NAFLD should be primarily directed at improving insulin resistance, and complemented by the treatment of other accompanying components of metabolic syndrome, such as diabetes, dyslipidemia or hypertension.
When the obesity has been brought under control, treatment options are dietary changes with exercise, medication, endoscopic or surgical bariatric approaches. The goal of the treatment is the loss of 8%-10% of body weight, by reducing total body fat content, which can bring about significant improvements in insulin sensitivity, adiponectin level, and liver histopathology, including steatosis, ballooning, inflammation and NAFLD score (NAS). Current medication such as ortistat or sibutramine, are most appropriate in patients who are highly motivated and have mild obesity. Because of the difficulties in achieving a “sustained” weight loss only by medical treatment, the weight loss by bariatric surgery, together with dietary changes and exercise is preferred to medication-assisted weight loss in patients with moderate to severe obesity [11].
The role of insulin-sensitizing drugs is another hot topic in the prevention and/or treatment of metabolic complications associated with obesity. The major insulin sensitizers are metformin and thiazolidinediones (TZDs). While metformin treatment may or may not be improving histopathology in NAFLD, it has utility due to its ability to improve insulin resistance and the cardio-metabolic risk factors often accompanying NAFLD[15]. TZDs are PPARγ agonists, such as rosiglitazone and pioglitazone. TZDs have unique effects, such as decreasing plasma NEFA by inhibition of lipolysis, reduction of muscle long-chain fatty acyl CoA levels, and redistribution of fat within the body from the visceral to the subcutaneous side. Even though they cause significant weight gain, TZDs can ameliorate lipotoxicity and adiposopathy overall[3]. As a treatment option in NAFLD, TZDs improve steatosis, transaminase levels, adiponectin, and insulin sensitivity, but have no significant effect on fibrosis or NAS. A recent study has shown that pioglitazone has more limited benefit than vitamin E for the treatment of NASH in adults without diabetes. Long-term treatment with TZDs may be necessary in NAFLD patients, but concerns about cardiotoxicity, congestive heart failure, edema, osteoporosis, weight gain and also hepatotoxicity exist[11].
There is also a long list of agents with some shown efficacy in treatment of NAFLD, such as ursodeoxycholic acid (UDCA), omega-3 fatty acids, pentoxifylline, betaine, S-adenosylmethionine (SAM), N-acetyl-cysteine (NAC), probiotics, and angiotensin-receptor blockers. Fibrates activate PPAR-α, leading to increased HDL and decreased triglycerides, LDL, and VLDL. Similarly, statins also improve liver enzymes, but not insulin resistance or histopathology.
NAFLD has become the most common cause of liver disease in the world of the new millennium. It affects approximately 20%-30% of adults and as high a percentage as 70%-90% of patients with obesity or diabetes. The liver is not only a central organ of metabolism, but also has active role in immunity and inflammation. The liver should therefore be the pathogenic as well as targeted organ of insulin resistance and inflammation associated with obesity. One of the corner stones of the current approach to obesity should be to recognize and to treat NAFLD as an early indicator complication of metabolic syndrome. Today, there is no single treatment modality or pharmacological agent that has established efficacy in the treatment of NAFLD. Since policies on awareness of metabolic liver diseases could potentially save lives and millions of dollars, metabolic liver diseases have become a hot topic of hepatologists.
Peer reviewers: Joel Faintuch, MD, PhD, Department of Gastroenterology, Hospital das Clinicas, ICHC, 9th Floor, Room 9077, Sao Paulo 05403-900, Brazil; Amedeo Lonardo, MD, Internal Medicine, Endocrinology, Metabolism and Geriatrics, University of Modena and Reggio, Emilia 41100 Modena, Italy
S- Editor Zhang HN L- Editor Herholdt A E- Editor Zhang L
| 1. | Brunt EM. What’s in a NAme? Hepatology. 2009;50:663-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 22] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 2. | Loria P, Lonardo A, Carulli N. Should nonalcoholic fatty liver disease be renamed? Dig Dis. 2005;23:72-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 63] [Cited by in RCA: 62] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 3. | DeFronzo RA. Insulin resistance, lipotoxicity, type 2 diabetes and atherosclerosis: the missing links. The Claude Bernard Lecture 2009. Diabetologia. 2010;53:1270-1287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 621] [Cited by in RCA: 616] [Article Influence: 41.1] [Reference Citation Analysis (0)] |
| 4. | Obesity: preventing and managing the global epidemic. Report of a WHO Consultation (WHO Technical Report Series 894). Geneva: World Health Organization; 2000; 252. |
| 5. | Available from: http: //www.cdc.gov/nchs/data/hus/hus08.pdf. |
| 6. | Koh HK. A 2020 vision for healthy people. N Engl J Med. 2010;362:1653-1656. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 139] [Article Influence: 9.3] [Reference Citation Analysis (0)] |
| 7. | Zeyda M, Stulnig TM. Obesity, inflammation, and insulin resistance--a mini-review. Gerontology. 2009;55:379-386. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 268] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 8. | Available from: http: //apps.who.int/bmi/index.jsp. |
| 9. | Anis AH, Zhang W, Bansback N, Guh DP, Amarsi Z, Birmingham CL. Obesity and overweight in Canada: an updated cost-of-illness study. Obes Rev. 2010;11:31-40. [PubMed] |
| 10. | Haslam DW, James WP. Obesity. Lancet. 2005;366:1197-1209. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3099] [Cited by in RCA: 3210] [Article Influence: 160.5] [Reference Citation Analysis (0)] |
| 11. | Lam B, Younossi ZM. Treatment options for nonalcoholic fatty liver disease. Therap Adv Gastroenterol. 2010;3:121-137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 81] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 12. | Available from: http: //www.un.org/esa/population/publications/longrange2/WorldPop2300final.pdf. |
| 13. | Jacobson IM, Davis GL, El-Serag H, Negro F, Trépo C. Prevalence and challenges of liver diseases in patients with chronic hepatitis C virus infection. Clin Gastroenterol Hepatol. 2010;8:924-33; quiz e117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 87] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 14. | Williams R. Global challenges in liver disease. Hepatology. 2006;44:521-526. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 526] [Cited by in RCA: 546] [Article Influence: 28.7] [Reference Citation Analysis (0)] |
| 15. | Balsano C, Alisi A, Nobili V. Liver fibrosis and therapeutic strategies: the goal for improving metabolism. Curr Drug Targets. 2009;10:505-512. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 21] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 16. | Hill MJ, Metcalfe D, McTernan PG. Obesity and diabetes: lipids, ‘nowhere to run to’. Clin Sci (Lond). 2009;116:113-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 46] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 17. | Capeau J. Insulin resistance and steatosis in humans. Diabetes Metab. 2008;34:649-657. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 77] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 18. | Balistreri CR, Caruso C, Candore G. The role of adipose tissue and adipokines in obesity-related inflammatory diseases. Mediators Inflamm. 2010;2010:802078. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 314] [Cited by in RCA: 336] [Article Influence: 22.4] [Reference Citation Analysis (0)] |
| 19. | Fuentes L, Roszer T, Ricote M. Inflammatory mediators and insulin resistance in obesity: role of nuclear receptor signaling in macrophages. Mediators Inflamm. 2010;2010:219583. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 65] [Cited by in RCA: 80] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 20. | Baffy G. Kupffer cells in non-alcoholic fatty liver disease: the emerging view. J Hepatol. 2009;51:212-223. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 371] [Cited by in RCA: 374] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 21. | Tordjman J, Guerre-Millo M, Clément K. Adipose tissue inflammation and liver pathology in human obesity. Diabetes Metab. 2008;34:658-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 44] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 22. | Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med. 2010;363:1341-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1326] [Cited by in RCA: 1485] [Article Influence: 99.0] [Reference Citation Analysis (0)] |
| 23. | Porepa L, Ray JG, Sanchez-Romeu P, Booth GL. Newly diagnosed diabetes mellitus as a risk factor for serious liver disease. CMAJ. 2010;182:E526-E531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 87] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 24. | Sanyal AJ, Chalasani N, Kowdley KV, McCullough A, Diehl AM, Bass NM, Neuschwander-Tetri BA, Lavine JE, Tonascia J, Unalp A. Pioglitazone, vitamin E, or placebo for nonalcoholic steatohepatitis. N Engl J Med. 2010;362:1675-1685. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2642] [Cited by in RCA: 2469] [Article Influence: 164.6] [Reference Citation Analysis (2)] |