Revised: January 14, 2010
Accepted: January 21, 2010
Published online: January 27, 2010
Ruptured hepatocellular carcinoma is a rare, emergency occurrence in western countries with high mortality risk. A number of hypotheses have been formulated in order to explain the precise mechanism that leads to hepatocellular carcinoma (HCC) rupture: sub-capsular location, dimensions, portal hypertension, tumour necrosis, local increase of venous pressure due to the outflow reduction caused by neoplastic invasion, and the presence of a previous vascular injury which might predispose to HCC rupture. There is still a debate in the literature concerning the best approach in cases of HCC rupture. Surgery is the first option for treatment of acute abdominal bleeding. However the advent of endovascular treatments widens the range of possible therapies for acute bleeding control and subsequent ablation purposes. We report a case of hemoperitoneum from spontaneous rupture of undiagnosed HCC, that was treated successfully by emergency surgical resection followed by transarterial chemo-embolization for local recurrence.
- Citation: Rossetto A, Adani GL, Risaliti A, Baccarani U, Bresadola V, Lorenzin D, Terrosu G. Combined approach for spontaneous rupture of hepatocellular carcinoma. World J Hepatol 2010; 2(1): 49-51
- URL: https://www.wjgnet.com/1948-5182/full/v2/i1/49.htm
- DOI: https://dx.doi.org/10.4254/wjh.v2.i1.49
The first manifestation of unknown hepatocellular carcinoma (HCC) with hemoperitoneum caused by spontaneous rupture is a rare occurrence in western countries, with an incidence of less than 3%, but with a high mortality risk[1,2]. There is still a debate in the literature concerning the best approach in cases of HCC rupture.Surgery is the first option for treatment of acute abdominal bleeding. However the advent of endovascular treatments widens the range of possible therapies for acute bleeding control and subsequent ablation purposes. Short term survival benefit can be obtained with surgical treatment, associated or not with interventional radiology[3-5]. Long term survival seems to be unanimously correlated to the stage of the disease, its local spread after rupture and to the residual hepatic functionality[6,7]. We report a case of hemoperitoneum from spontaneous rupture of undiagnosed HCC successfully treated by emergency surgical resection followed by transarterial chemo-embolization (TACE) two months later for tumour recurrence. At a follow-up of one year the patient is still alive, with no signs of HCC recurrence.
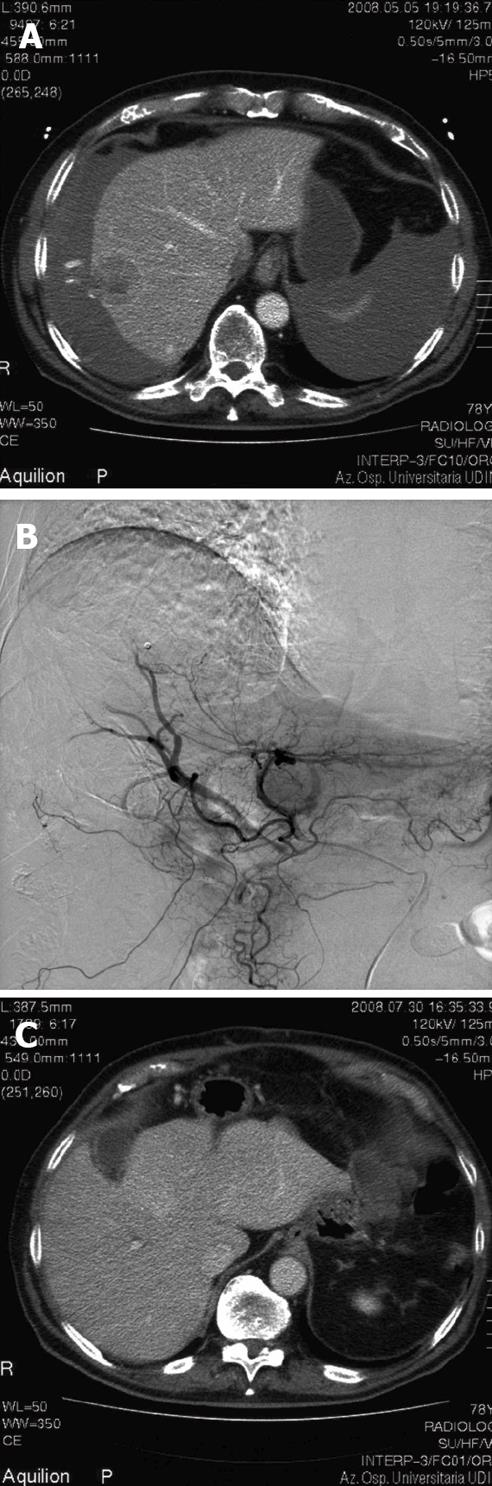
We report a 78-year-old male patient who came to the Emergency Room (ER) because of abdominal pain, hypotension (80/40 mmHg) associated with tachycardia, and hypothermia (35.7°C). Anamnesis was totally negative. Doppler ultrasound showed diffuse hemoperitoneum. Urgent abdomen Computed Tomography (CT) scan revealed an actively bleeding focal lesion of 3.5 centimeter diameter in the 6th hepatic segment, associated with perihepatic and perisplenic hemoperitoneum (Figure 1A). At emergency laparotomy, major hemoperitoneum of over 2000 mL was present. The source of bleeding was from a 3.5 cm, whitish, ligneous hepatic nodule in the 6th segment, which was lacerated in its central part. The liver was not cirrhotic. After cleaning of the abdomen, and liver mobilization, a 6th segment wedge resection was performed using Pringle manoeuvre (30 min of clamping). The post-operative course was uneventful, and at post-operative day 7 the patient was discharged from hospital in good general condition. Post-operative laboratory tests confirmed no evidence of hepatitis B or C positivity. Histological exam showed a lesion of 4.5 centimeter diameter partly covered with glissonian sheath compatible with HCC (grading 3, WHO 2001). Margins were free of HCC infiltration. Two months after the operation, local recurrence of HCC was evidenced by CT scan. Considering the general condition of the patient, and the high risk of a new hepatic resection, we performed TACE by selective catheterisation of the right hepatic artery, and injection of DC Bead 100-300 micron + 50 mg Doxorubicin (Figure 1B). The procedure was uneventful, and without evidence of liver failure. Two weeks later, an abdominal CT scan revealed complete devascularisation of the HCC (Figure 1C). At one year follow-up, no signs of tumour recurrence were evidenced: CT scan and Magnetic Resonance Imaging (MRI) were negative, and laboratory tests including alpha fetoprotein were in the normal range.
The occurrence of a ruptured HCC is an unusual find in western countries with an incidence of < 3% compared to Eastern countries (12%-14%), with a reported mortality rate up to 50%[1,2]. A number of hypotheses have been formulated in order to explain the precise mechanism that leads to HCC rupture: sub-capsular location, dimensions, portal hypertension, tumour necrosis, local increase of venous pressure due to the outflow reduction caused by neoplastic invasion[8], the presence of a previous vascular injury which might predispose to HCC rupture and to the rupture of smaller lesions in different locations[9]. With regard to diagnosis, according to the study of Yeh et al., abdominal pain was the only independent factor relating patients with ruptured HCC[3]. Preoperative diagnosis of HCC rupture is difficult in patients without a previous history of cirrhosis or HCC. Vergara et al[1], reported that an accurate pre-operative diagnosis of ruptured HCC was obtained in only 25% of cases, despite shock present in 33% to 90% of the patients. Doppler ultrasound and CT scan are useful to demonstrate the presence of hemoperitoneum and liver tumours. CT also has the advantage of demonstrating the patency of the portal vein but the site of active bleeding can seldom be demonstrated, conventional angiography demonstrates extravasations of contrast from the tumour only in 13%-35% of cases[10]. There is no single opinion about the best treatment for a ruptured HCC, even if patient’s general conditions are considered in all cases. If hepatic insufficiency, liver cirrhosis and hemoperitoneum are present, trans-arterial embolization (TAE) is an effective and well-tolerated treatment for the emergency management of hemoperitoneum[8]. In patients with normal liver function haemostasis followed by hepatic resection either in emergency or after staging has been suggested[2]. Literature reports such as[7] suggest that multidisciplinary management with TAE and postponed surgery in selected patients would improve the short term mortality. Liver resection has been advocated to achieve both haemostasis and to provide a definitive treatment in a single operation[1,4], even if liver resection should be reserved for patients with a small and easily accessible tumour in a non cirrhotic liver[4]. The studies on survival by Liu et al[5] and Yeh et al[3] supply contrasting data, therefore it is not clear whether there is a different survival expectation for the patients with ruptured HCC and the others[5,6]. The surgical treatment of intraperitoneal lesions caused by neoplastic dissemination during rupture can increase the long term survival.
In our case, the diagnosis of HCC was accidental in a patient with no history of hepatic disease. On his admission the patient had poor hemodynamic stability but normal liver function. After CT scan which confirmed hemoperitoneum secondary to hepatic rupture, we opted for emergency surgery, even because the lesion was small, and easily accessible.
A different approach could have been considered in cases of liver cirrhosis, with trans arterial embolization first, followed by surgical operation (laparoscopic/ laparotomic approach) or percutaneous drainage of the hemoperitoneum to reduce any possible development of liver decompensation.
In our patient, HCC recurred two months after treatment. Considering the age of the patient and the risk of a new hepatic resection, we completed surgery with TACE. One year follow-up was regular, hepatic function was normal, as was the αFP values, and radiological examinations showed no HCC recurrence.
Peer reviewer: Radha K Dhiman, Professor, Department of Hepatology,Postgraduate Institute of Medical Education and Research, Chandigarh 160012, India
| 1. | Vergara V, Muratore A, Bouzari H, Polastri R, Ferrero A, Galatola G, Capussotti L. Spontaneous rupture of hepatocelluar carcinoma: surgical resection and long-term survival. Eur J Surg Oncol. 2000;26:770-772. |
| 2. | Battula N, Madanur M, Priest O, Srinivasan P, O’Grady J, Heneghan MA, Bowles M, Muiesan P, Heaton N, Rela M. Spontaneous rupture of hepatocellular carcinoma: a Western experience. Am J Surg. 2009;197:164-167. |
| 3. | Yeh CN, Lee WC, Jeng LB, Chen MF, Yu MC. Spontaneous tumour rupture and prognosis in patients with hepatocellular carcinoma. Br J Surg. 2002;89:1125-1129. |
| 4. | Lai EC, Lau WY. Spontaneous rupture of hepatocellular carcinoma: a systematic review. Arch Surg. 2006;141:191-198. |
| 5. | Liu CL, Fan ST, Lo CM, Tso WK, Poon RT, Lam CM, Wong J. Management of spontaneous rupture of hepatocellular carcinoma: single-center experience. J Clin Oncol. 2001;19:3725-3732. |
| 6. | Tanaka A, Takeda R, Mukaihara S, Hayakawa K, Shibata T, Itoh K, Nishida N, Nakao K, Fukuda Y, Chiba T. Treatment of ruptured hepatocellular carcinoma. Int J Clin Oncol. 2001;6:291-295. |
| 7. | Buczkowski AK, Kim PT, Ho SG, Schaeffer DF, Lee SI, Owen DA, Weiss AH, Chung SW, Scudamore CH. Multidisciplinary management of ruptured hepatocellular carcinoma. J Gastrointest Surg. 2006;10:379-386. |
| 8. | Castells L, Moreiras M, Quiroga S, Alvarez-Castells A, Segarra A, Esteban R, Guardia J. Hemoperitoneum as a first manifestation of hepatocellular carcinoma in western patients with liver cirrhosis: effectiveness of emergency treatment with transcatheter arterial embolization. Dig Dis Sci. 2001;46:555-562. |