Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.109801
Revised: June 7, 2025
Accepted: July 24, 2025
Published online: August 27, 2025
Processing time: 97 Days and 19.9 Hours
Artificial intelligence (AI) has become an indispensable tool in modern health care, offering transformative potential across clinical workflows and diagnostic innovations. This review explores the sation of AI technologies in synthesizing and analyzing multimodal data to enhance efficiency and accuracy in health care delivery. Specifically, deep learning models have demonstrated remarkable capabilities in identifying seven categories of hepatobiliary disorders using ocular imaging datasets, including slit-lamp, retinal fundus, and optical coherence tomography images. Leveraging ResNet-101 neural networks, researchers have developed screening models and independent diagnostic tools, showcasing how AI can redefine diagnostic practices and improve accessibility, particularly in resource-limited settings. By examining advancements in AI-driven health care solutions, this article sheds light on both the challenges and opportunities that lie ahead in integrating such technologies into routine clinical practice.
Core Tip: Artificial intelligence has taken the world by storm. It has various applications in the screening and diagnosis of many systemic diseases. In this review article, we will discuss AI-based ocular biomarkers in hepatobiliary disorders. As per studies, there are deep learning models to detect numerous categories of hepatobiliary disorders on two common types of ocular images: slit-lamp retinal fundus images and optical coherence tomography images.
- Citation: Parmar UPS, Morya AK, Gupta PC, Arora A, Verma N. Role of artificial intelligence-based ocular biomarkers in hepatobiliary diseases: A scoping review. World J Hepatol 2025; 17(8): 109801
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/109801.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.109801
The eye is increasingly recognized as a non-invasive window into systemic and hepatic health, owing to its accessibility and capacity to reflect underlying pathophysiological processes[1]. Ocular manifestations can provide early clues about underlying diseases elsewhere in the body, including the liver and biliary system. Hepatobiliary disorders are often silent until advanced stages. Non-invasive and easily accessible biomarkers are thus critical for early detection and monitoring. Hepatobiliary diseases, such as chronic hepatitis, cirrhosis, cholestatic disorders, and Wilson's disease, often lead to characteristic changes in ocular tissues[2]. Ocular biomarkers appeal due to the eye's transparent structures, which permit direct, non-invasive visualization of blood vessels, nerves, and pathological deposits. From yellow sclera in jaundice to Kayser-Fleischer (K-F) rings due to copper deposition in Wilson's disease, these ocular biomarkers have significant diagnostic and prognostic value[2]. Historically, clinicians have relied on direct observation and manual evaluation of these signs. Still, the advent of artificial intelligence (AI) and machine learning offers new opportunities to detect and quantify ocular biomarkers objectively and at scale. This review provides a comprehensive overview of AI-based ocular biomarkers in hepatobiliary diseases, emphasizing evidence from both human and animal studies, and outlines how AI is transforming diagnosis, management, and research in this evolving field.
We searched the keywords on scientific platforms like PubMed, Google Scholar, EMBASE, RCA, and Scopus. We included highly cited papers from the years 1997 to 2025 only. We excluded articles published in languages other than English.
Before the AI era, clinicians and researchers used traditional methods to identify and measure ocular biomarkers associated with liver and biliary diseases. While foundational, these conventional approaches often lack the sensitivity, objectivity, and scalability required for early detection and population-level screening. Early ocular signs may be subtle and missed on physical exam. Therefore, automation through AI can be a key step forward.
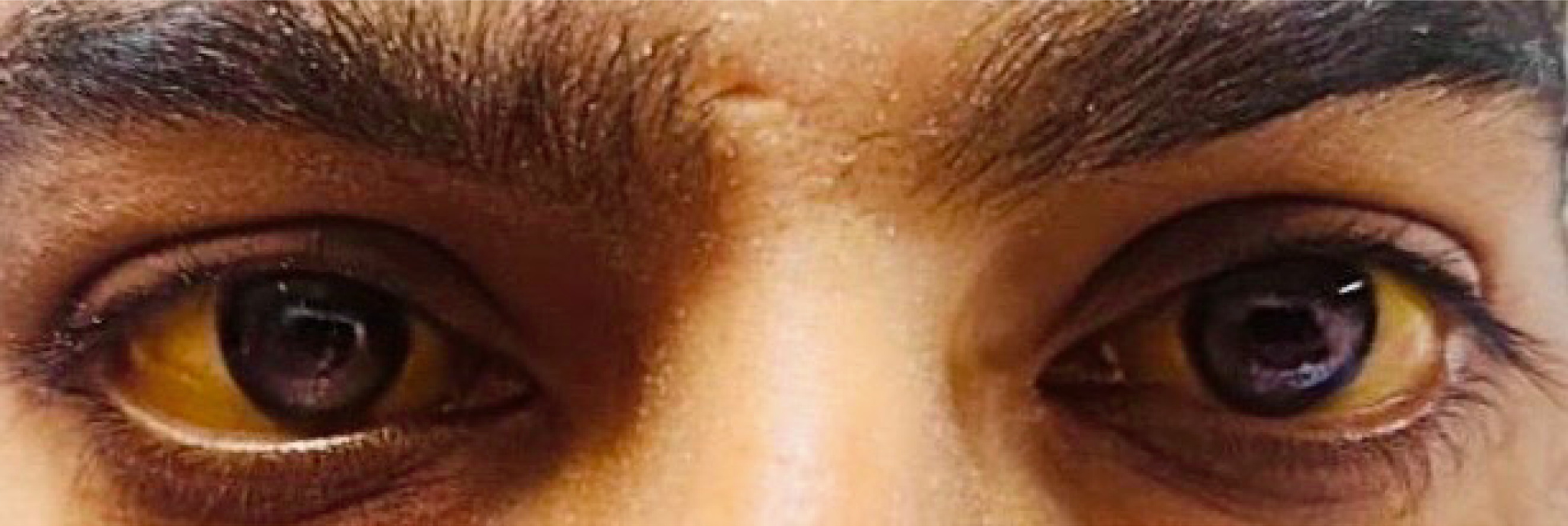
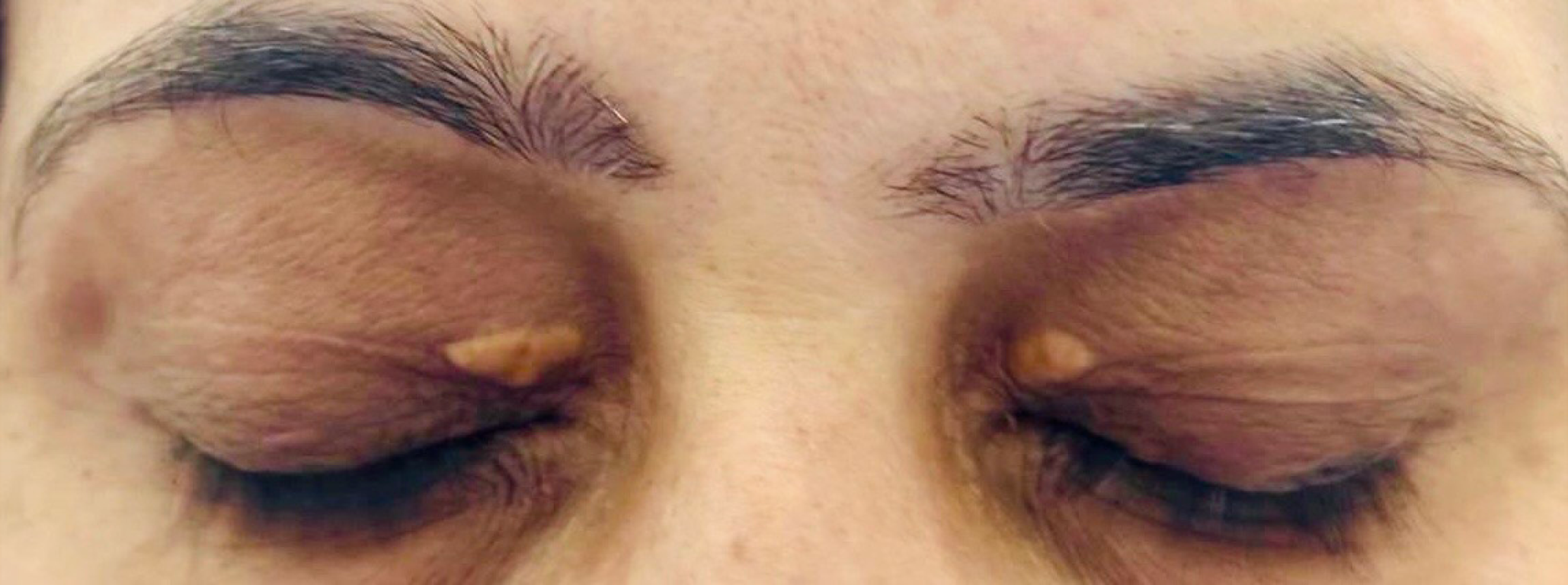
The most basic approach to ocular biomarkers has been the direct clinical examination of the eye. Physicians look for classic signs like scleral icterus, K-F rings in the cornea, corneal opacities, or retinal changes. For example, scleral icterus is observed by examining the exposed sclera under natural light, and a positive finding strongly suggests elevated bilirubin (> 2–3 mg/dL) and impending jaundice[3]. In Wilson's Disease, a careful slit-lamp exam can reveal the brownish K-F ring at the corneal periphery due to copper deposition[4]. Ophthalmologists may grade K-F rings by extent and density, but this is largely subjective and requires significant expertise. Likewise, vitamin A deficiency due to chronic liver disease is traditionally identified by signs like Bitot's spots, which are foamy keratin deposits on the conjunctiva, and night blindness, often confirmed by serum vitamin A levels[5]. Dry eye symptoms in hepatitis or cirrhosis patients are assessed via Schirmer's tests or tear break-up time, indicating tear film insufficiency, possibly from autoimmune damage as in the case of autoimmune hepatitis[6].
Beyond visible signs, traditional methods involve analyzing ocular fluids or tissues for biomarkers. Tear fluid, for instance, has been studied for liver disease markers because it can reflect systemic metabolic changes. Researchers have measured tear osmolarity and composition in patients with cirrhosis, finding that chronic liver disease can impair tear secretion and stability[7]. Con
There are several notable challenges with these conventional approaches. Many assessments, for instance, judging "mild" vs "moderate" scleral icterus, are subjective. Under poor lighting or in inexperienced hands, subtle signs can be overlooked. A slight yellow tinge might be missed, delaying diagnosis. Furthermore, human vision can only detect changes beyond a certain threshold. Bilirubin usually must exceed about 2 mg/dL for jaundice to become apparent[10]. More minor elevations, which might still indicate pathology, are often missed. Thus, early disease signs frequently go undetected by eye exam alone.
A thorough eye examination requires specialized equipment and expertise, including slit-lamp evaluation or fundus photography. Performing detailed ophthalmic exams on every patient at risk for liver disease isn't feasible, especially in primary care or screening settings. Also, obtaining ocular fluids like aqueous or vitreous humour for analysis is not done routinely due to the invasive nature. Even tear collection, while non-invasive, yields tiny samples and can be technically challenging to analyze for systemic biomarkers[11]. Lastly, many ocular findings in liver disease are not exclusive to hepatobiliary disorders. For example, dry eye and retinal vascular changes can result from numerous other conditions, including Sjögren's syndrome, diabetes, hypertension, normal ageing, among others. A given ocular sign might not definitively point to liver disease without corroborating systemic clues.
The application of AI, particularly Deep Learning, has begun to transform how ocular biomarkers are detected and quantified. By analyzing eye images, AI algorithms can uncover subtle features linked to systemic liver disease that human observers might miss. Several key advances illustrate the potential of these techniques.
In a landmark study, Xiao et al[12] trained a deep CNN on the ResNet architecture on eye photographs, including both external eye images and retinal fundus photos from over a thousand individuals. ResNet was selected for its depth and ability to handle image complexity. The model could accurately distinguish patients with conditions like cirrhosis and hepatocellular carcinoma from healthy controls, reporting area-under-the-curve (AUC) values around 0.90 for detecting cirrhosis from external eye photos. Predictions for milder conditions, such as non-alcoholic fatty liver disease (NAFLD) or chronic hepatitis, were less accurate (with AUCs in the 0.65–0.75 range). Nonetheless, this study proved that eye images contain diagnostic information about liver health. Another project by Song et al[13] used CNN models to detect K–F rings from corneal photographs automatically. Their system exceeded 95% sensitivity and specificity in identifying the presence of K–F rings, effectively matching expert ophthalmologists in recognizing this copper deposition. Such results demonstrate that AI can detect classic ocular signs of liver disease and do so with consistency and potential for quantification.
A study by Babenko et al[14] in 2023, using a deep learning model trained on frontal eye photos to predict various laboratory values, found that their model identified individuals with elevated liver enzymes, like high AST, or low serum albumin based on subtle color and pattern cues around the eye, with accuracy improvements of 5%–20% over non-image-based models. Their results suggest that chronic liver disease can cause changes like slight conjunctival pallor or scleral hue and can manifest in small but detectable ocular differences. Similarly, an earlier proof-of-concept system, BiliScreen, developed by Mariakakis et al[15] in 2017, used smartphone images of the sclera to estimate bilirubin levels. Their pilot study of 70 subjects successfully flagged cases of subclinical jaundice with about 90% sensitivity and 97% specificity by quantifying scleral colour changes. These examples highlight how AI can transform widely available devices into screening tools for liver dysfunction by measuring ocular biomarkers like scleral icterus.
The retinal fundus is another rich source of biomarkers. Although changes in the retina due to liver disease are subtler than those of other diseases like diabetic retinopathy, AI has found some associations. In Xiao et al's study, including retinal images modestly improved the detection of conditions like fatty liver and hepatitis, likely because chronic liver disease can affect retinal microvasculature, causing slight narrowing of retinal arterioles or other circulation changes related to the metabolic syndrome[12]. AI Retinal imaging plays a supporting role compared to external eye analysis for liver disease detection, but it remains an area of active research.
Region segmentation: Before classification, one may need to isolate the region of interest. In their study for detecting K-F rings, Song et al[13] used the You Only Look Once algorithm to detect the corneal region and then a U-Net model for segmenting the ring itself before grading its severity with the ResNet neural network. Likewise, segmenting the sclera in eye photos can also improve bilirubin detection by focusing on the white of the eye while avoiding confounders like lighting or conjunctival blood vessels[16].
Ensemble models: Combining multiple algorithms or inputs, like ensembling slit-lamp and fundus image models, can also improve robustness. Xiao et al[12] built separate models for external and retinal images and found that each excelled in different disease predictions. An ensemble that uses both might cover more ground. Another possible way to implement this is by fusing image analysis with non-imaging data like patient age or clinical parameters to improve predictive accuracy, moving toward a multimodal AI system[17].
Transfer learning: As curated medical image datasets are often limited in size, many projects leverage transfer learning, that is, using CNNs pre-trained on large general image databases and then fine-tuning them on ocular medical images[18]. This allows models to achieve good accuracy even when only a few hundred or thousands of training images are available by capitalizing on features the network has already learned from millions of generic images. This technique was used in the previously discussed study by Song et al[13], wherein they used six pre-trained K-F ring grading models.
Video and eye movement analysis: While static images dominate, AI is also applied to ocular function tests. Video-oculography (VOG) records eye movements, and AI can classify patterns. For instance, minimal hepatic encephalopathy (MHE), a subtle complication of cirrhosis, has been linked to impaired saccadic eye movements. Studies using machine learning on VOG data showed that MHE patients have longer saccade latencies and more errors in eye movement tasks compared to non-MHE cirrhotics[19,20]. Algorithms can quantify this functional biomarker to diagnose MHE non-invasively, which is traditionally challenging. AI could automate interpreting such complex eye movement data better than manual review of tracings.
Validation against clinicians: A key question is how AI compares to human experts. Early studies are encouraging. Xiao et al[12] reported that their AI performed on par with experienced ophthalmologists and hepatologists in identifying certain liver diseases from eye images. Furthermore, their model could pick up subtle conjunctival colour changes indicative of cirrhosis that a non-eye-specialist might overlook. In Wilson's disease, the tested models might help less experienced clinicians by reliably detecting a faint K–F ring, thereby preventing missed diagnoses of this treatable condition. These systems are intended to augment, not replace, physicians by acting as a consistent second observer that can quantify what it sees. AI can assess ocular biomarkers more objectively and reproducibly. In analogous fields like diabetic retinopathy screening, Food and Drug Administration-approved AI systems already achieve specialist-level sensitivity and specificity; similarly, we expect AI algorithms for liver-related ocular findings to reach expert-level performance as training data and techniques improve[12].
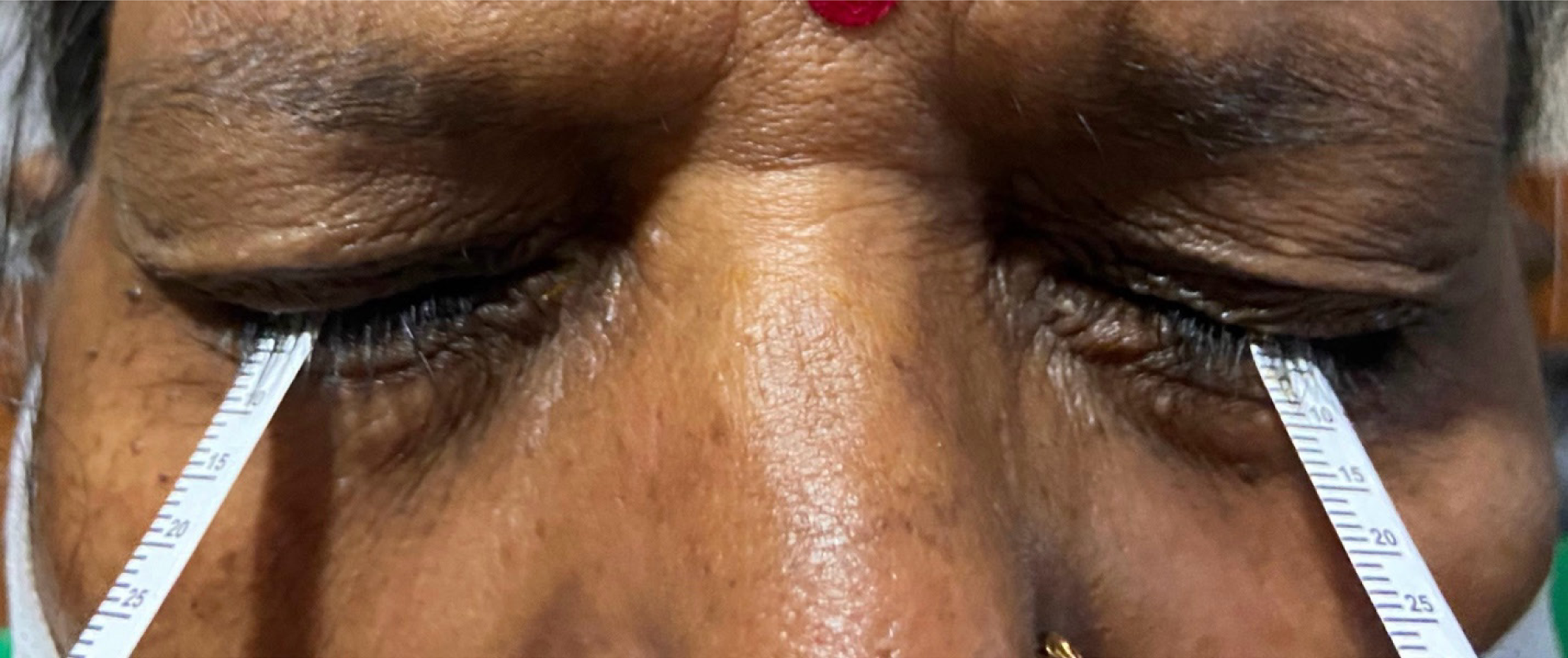
Scleral icterus: Clinically, scleral icterus often appears before skin jaundice, indicating serum bilirubin is typically above about 2 mg/dL[10]. In cirrhosis, regardless of cause, whether viral, alcoholic, or non-alcoholic steatohepatitis-related, the presence of scleral icterus signals advanced disease or an acute exacerbation[21]. AI can enhance the detection of this biomarker. Smartphone-based imaging has been used to quantify scleral yellowness as a continuous value. For example, Kazankov et al[22] showed that a 'scleral colour value' derived from eye photographs in patients with acute-on-chronic liver failure correlated almost linearly (ρ ~0.90) with serum bilirubin levels, and changes in this value tracked changes in bilirubin over time[22]. Similarly, the BiliScreen app mentioned earlier could detect even mild icterus invisible to the naked eye[15]. These tools demonstrate that digital ocular imaging can serve as an objective, non-invasive proxy for liver function in cirrhosis patients (Figure 1).
Hepatic retinopathy: Advanced liver disease, especially when accompanied by hepatic encephalopathy, can cause subtle retinal dysfunction. Electroretinography studies in patients with cirrhosis have shown reduced b-wave amplitudes and other perturbations in retinal signalling, likely due to neurotoxic factors like ammonia[23]. These disturbances improved after liver transplantation, indicating they were largely reversible[23]. Clinically, patients might not notice such slight vision changes, but the findings highlight that liver failure can affect the nervous system, including the retina. In the future, AI analysis of visual function tests or retinal scans might assist in detecting these subtle changes as a marker of hepatic encephalopathy.
Eye movement abnormalities in hepatic encephalopathy: MHE is tricky to diagnose using standard psychometric tests, but eye movement testing has emerged as a sensitive tool. Cirrhotic patients with MHE show longer latencies in initiating saccades and more frequent errors in antisaccade tasks[24]. A 2022 study quantified over a hundred oculomotor metrics and found significant differences in dozens of parameters in MHE patients compared to control cirrhosis patients without encephalopathy[20]. These patterns reflect cognitive slowing and impaired motor control of the eyes due to elevated ammonia and other toxins[24]. AI plays a promising role here: Machine learning algorithms can analyze complex eye-tracking data to classify which patients have encephalopathy. Early work suggests that a VOG test interpreted by AI could provide an objective, quick diagnostic for minimal encephalopathy, which is otherwise often missed[19]. In practical terms, an AI system might flag a cirrhosis patient as having early neurologic impairment based on an abnormal eye movement signature, prompting timely initiation of therapies to prevent progression to overt encephalopathy.
Chronic hepatitis viruses and autoimmune hepatitis can produce some ocular findings, but these are generally nonspecific or uncommon. Hepatitis C virus has been associated with dry eye symptoms[25], especially in those with coexisting autoimmune disorders and, in the interferon treatment era, a transient retinopathy, with cotton wool spots in the retina observed during therapy[26]. Hepatitis B virus has no distinctive ocular manifestation beyond what might occur from resultant cirrhosis. Autoimmune hepatitis sometimes overlaps with other autoimmune conditions that affect the eyes; for instance, a subset of patients develop severe dry eye or occasional uveitis[27]. These ocular issues are not specific to liver injury but reflect the broader autoimmune activity. No AI-driven ocular tools have been developed specifically for viral hepatitis or AIH, given the lack of unique eye signs. However, if needed, general AI systems for detecting dry eye or retinopathy could be used to monitor known complications like interferon-related retinopathy (Figure 2).
Primary biliary cholangitis (PBC) is characterized by chronic cholestasis and often hyperlipidemia, and a classic ass
Wilson's disease provides one of the most striking examples of an ocular biomarker for a systemic condition. The path
Such a tool could be used for screening; for example, any young patient with unexplained liver abnormalities or neuropsychiatric symptoms might have an eye photo taken in the clinic, and an AI algorithm could immediately indicate whether a K–F ring is likely present, expediting referral and treatment. Furthermore, AI could quantitatively monitor K–F rings over time. In Wilson's disease management, effective therapy with copper-chelating agents typically leads to the gradual fading of the ring. AI-driven analysis of serial corneal images could enable objective monitoring of K-F ring resolution as a digital surrogate for therapeutic efficacy in Wilson's disease. This is particularly useful since Wilson's disease requires long-term management and adherence; an unchanged or reappearing ring might signal non-compliance or a need for therapy adjustment. Overall, Wilson's disease is a case where an ocular biomarker is central to diagnosis and management, and AI can significantly enhance its detection and follow-up.
Chronic alcohol abuse can lead to nutritional deficiencies that manifest in the eyes. One classic example is vitamin A deficiency in patients with alcoholic cirrhosis and malnutrition. The liver stores vitamin A, and in advanced liver disease, those stores are depleted; combined with poor dietary intake, this can cause night blindness, conjunctival and corneal dryness, and Bitot's spots on the conjunctiva, which are hallmark signs of vitamin A deficiency[5]. Identifying these findings in an alcohol-use patient points to severe nutritional compromise and should prompt vitamin repletion. Thiamine (vitamin B1) deficiency in alcohol abuse can also cause ophthalmologic signs. Classically, Wernicke's encephalopathy presents with nystagmus and ophthalmoplegia, which is eye movement paralysis alongside confusion, but this is an acute neurological emergency rather than a chronic biomarker of liver disease. In routine alcoholic liver disease care, the primary ocular consideration is to be vigilant for vitamin deficiencies; an AI system could potentially assist by detecting subtle signs of malnutrition.
Early diagnosis and screening: AI-enhanced ocular biomarkers offer a transformative avenue for early, non-invasive detection of liver disease, particularly in settings with limited access to laboratory diagnostics. Because capturing an eye photograph is quick and painless, this approach could be used to screen large populations or at-risk groups. For example, a subtle yellowing of the sclera that is not apparent to the naked eye might be detected by an AI algorithm, prompting liver function tests and allowing a diagnosis of cholestatic disease or hepatitis before overt jaundice develops. Similarly, in a young patient with unexplained liver dysfunction or neuropsychiatric symptoms, an AI that automatically checks for a K–F ring could facilitate early identification of Wilson's disease, complementing traditional lab tests. Such screening could be particularly valuable in resource-limited areas: All that is needed is a smartphone or camera and the AI software, making it feasible to reach populations that lack regular blood test screening. These screening tools will need very high sensitivity to catch even mild cases while maintaining reasonable specificity to avoid too many false alarms. Combining ocular AI results with other risk factors, like a patient's known medical history or exposures, might improve the positive predictive value of identifying those needing follow-up[35].
Beyond initial detection, ocular biomarkers can help assess disease severity and prognosticate outcomes in patients with known liver disease. Some eye signs correlate with the severity of a condition. For instance, the intensity of scleral icterus tends to track bilirubin levels: An intensely yellow sclera signifies significant cholestasis or liver failure. AI quantification of scleral colouration provides a continuous metric that could be monitored over time. A study by Kazankov et al[22] confirmed this by noting that a digital scleral index correlated with clinical severity scores in acute-on-chronic liver failure[32]. Likewise, in Wilson's disease, the extent of the K-F ring might reflect the body's copper burden. Clinicians know that K–F rings fade with successful treatment; an AI that measures the ring's area or colour density could give an objective readout of treatment response or disease progression[13]. It is also conceivable that specific subtle retinal changes detected by AI, for example, signs of microvascular damage or neural impairment, could help stratify which patients with fatty liver disease are at higher risk of progressing to cirrhosis or developing complications, though such applications remain speculative. By adding quantitative ocular metrics to traditional lab and imaging data, clinicians could gain a more nuanced picture of a patient's prognosis[14]. For example, one might identify a patient with relatively 'mild' lab abnormalities but significant ocular signs as higher risk and therefore manage them more aggressively or surveil them more closely.
Many liver conditions require regular monitoring, and AI-based analysis of the eyes could become a convenient way to track disease status or detect complications between clinic visits[35]. Because patients can self-capture eye images (using a smartphone camera, for instance), ocular biomarkers lend themselves to remote monitoring. A patient with cirrhosis might take weekly pictures of their eyes; an AI algorithm can quantify their scleral icterus or other features and alert their doctor if there's a trend toward worsening. This kind of home monitoring could enable earlier intervention, such as adjusting medications if signs of hepatic decompensation appear, rather than waiting for the next appointment[35]. Similarly, in Wilson's disease, periodic AI analysis of corneal images could confirm that the K–F ring is diminishing with chelation therapy or signal if it reappears, indicating non-compliance or a need to intensify treatment. Even functional markers like eye movements could be tracked: A patient performing a quick eye movement test on a tablet each month might have the data analysed by AI for any slowing or errors that precede overt hepatic encephalopathy, prompting prophylactic treatment[19]. By integrating these tools into practice, healthcare providers could manage chronic liver disease in a more proactive, continuous way. This approach aligns with the trend toward telemedicine and precision medicine, providing individualised feedback and interventions based on an individual's real-time biomarker changes.
While AI-based ocular biomarkers hold great promise, several challenges must be addressed before they can be widely adopted.
Ocular images vary in quality based on camera type, lighting, and patient factors. A model trained on high-quality slit-lamp photos might falter on smartphone images taken under suboptimal lighting[36]. Likewise, differences in patient populations, such as skin pigmentation or scleral hue, can affect performance. Ensuring algorithms generalise well will require training on diverse datasets and validating models on external cohorts.
Implementing these tools in practice will require changes in clinical workflow. Eye images must be captured by a nurse, technician, or even by patients via an app and analysed without causing significant delays. The AI results must then be delivered to clinicians in a proper form, ideally integrated into the medical record. Training will be needed so clinicians understand the AI output and know how to act appropriately. Patient acceptance is also crucial, as some patients may be hesitant about an 'AI exam' or taking eye photographs for analysis, so clear communication about the tool's purpose and benefits is essential[37,38].
As with any medical AI, there are concerns around privacy, bias, and accountability. Eye photographs can potentially be identifying, so data handling must protect patient confidentiality[39]. If the training data are not representative, the AI could underperform in specific groups or propagate health disparities. It's essential to monitor and mitigate such biases to avoid unequal care. Regulatory approval will be needed before these tools can be used clinically; this means demon
It is essential to recognise that ocular biomarkers serve as surrogate indicators of liver function and require confirmatory testing for definitive diagnosis. An AI-detecting scleral icterus can strongly suggest liver dysfunction, but follow-up blood tests are still required to confirm and determine the cause. Similarly, confirmatory copper studies are needed if an AI flags a possible K–F ring. Thus, these tools will augment rather than replace existing diagnostics[38]. False negatives and positives can occur: A very early case of disease might not yet show any ocular signs, and conversely, a benign condition, for example, a naturally slightly yellow sclera or harmless pigmentation, might trigger an AI alert. Clinicians will need to interpret AI findings in context and have clear protocols for proceeding when an AI screening is positive.
Future work will focus on making AI models more robust across diverse settings. This will involve building larger, more varied training datasets through multi-centre collaborations, ensuring algorithms work well on different demographics and imaging conditions. Techniques like federated learning, training models on datasets from multiple hospitals without pooling sensitive data centrally[41], could help achieve large sample sizes while protecting privacy. Researchers are also exploring more explainable AI, where the algorithm highlights the features, for example, a region of sclera or iris, that influenced its prediction, to increase clinician trust and understanding[42].
Ocular biomarkers will likely be combined with other data for a comprehensive assessment. To improve diagnostic accuracy or risk prediction, an AI system might integrate an eye photograph with the patient's blood test results, genetic information, or other risk factors[43]. For instance, adding a fundus image analysis to standard clinical data could better stratify which NAFLD patients are at high risk of fibrosis or cardiovascular complications. Wearable or smartphone-based technology might also play a role. For example, smart devices could periodically scan a patient's sclera or monitor pupillary responses, feeding that data into an algorithm alongside other health metrics to detect decompensation earlier[44,45].
Rigorous prospective trials will be necessary to bring these AI tools into routine practice. We will likely see pilot programs where at-risk populations undergo AI eye screenings for liver disease, with outcomes compared to standard care. Similarly, trials in patients with established liver disease might test whether AI-guided monitoring, for example, home scleral imaging alerts, leads to earlier interventions or improved clinical outcomes. Successful trials will pave the way for regulatory approvals and clinical guidelines incorporating these tools.
As more data accumulate, AI-derived ocular biomarkers could help tailor treatments to individual patients. For instance, clinicians could escalate treatment or check adherence sooner if an AI algorithm tracking a Wilson's disease patient's K–F ring shows that the ring is not fading with standard therapy. Or an NAFLD patient whose ocular indicators, perhaps combined with other metrics, suggest high risk could be prioritized for aggressive interventions or enrollment in clinical trials for new therapies. In the long run, these tools could contribute to highly individualised care plans, a concept aligned with precision medicine[46].
Emerging technologies may reveal novel ocular signs of liver disease. High-resolution ocular imaging, like optical coherence tomography, could detect microscopic retinal or corneal changes due to liver disorders that are not visible on routine exams. Similarly, biochemical analysis of tears might uncover specific proteins or metabolites that correlate with liver fibrosis or cirrhosis. AI will be instrumental in sifting through these complex datasets to identify meaningful patterns. Additionally, dynamic eye tests, such as measuring pupillary reflex speed or tracking eye movements under specific tasks, might provide functional biomarkers for conditions like hepatic encephalopathy; machine learning could analyse those results to enhance early diagnosis of neurologic impairment.
Future AI systems may integrate multimodal inputs, including ocular images, biochemical parameters, and wearable sensor data, to deliver real-time, personalised risk stratification in hepatobiliary disease. As collaboration between hepatologists, ophthalmologists, and data scientists progresses, we expect eye-based assessments to become a regular part of the liver disease screening and management arsenal, pushing the boundaries of non-invasive diagnostics.
Validation across diverse populations: Current AI-based ocular biomarker systems require robust validation to ensure their reliability and effectiveness across various demographics and imaging conditions.
Integration into clinical practice: Achieving routine implementation demands regulatory approval, clinician training, and alignment with existing diagnostic methods.
Complementary nature of ocular findings: Ocular biomarkers should be used alongside traditional diagnostics such as blood tests and imaging, rather than as standalone tools.
Technical challenges: Developing AI models to work effectively in diverse settings and handle multimodal inputs remains a complex task.
Need for rigorous trials: Prospective clinical trials are essential to validate the effectiveness of AI-guided screenings and monitoring in improving patient outcomes.
Exploration of novel biomarkers: While promising, the discovery of new ocular biomarkers requires advanced imaging technologies and biochemical analyses, which may not be readily accessible.
These limitations highlight the importance of multidisciplinary research and collaboration to address current gaps, paving the way for integrating AI-based ocular biomarkers into precision hepatology (Table 1).
| Ref. | Aim | Population | Model details | Performance | Implications | Study type |
| Xiao et al[12] | Detect hepatobiliary diseases from eye images | 1000 individuals (multi-centre) | ResNet-101 on external and retinal images | Cirrhosis AUC: 0.90, HCC AUC: 0.93, NAFLD/Hepatitis AUC: 0.65–0.75 | Supports non-invasive AI-based liver disease screening using eye images | AI imaging study |
| Babenko et al[14] | Predict systemic biomarkers (AST, albumin) from eye photos | Diabetic patients | Custom deep learning system on external eye photos | Accuracy improvement of 5%–20% over non-image models | Demonstrates the potential of ocular imaging to reflect liver function | AI imaging study |
| Kazankov et al[32] | Correlate scleral colour with bilirubin levels | Cirrhosis patients with jaundice | Smartphone imaging with colour analysis (non-AI) | Correlation ρ: 0.90 with serum bilirubin | Enables remote, non-invasive bilirubin monitoring | Clinical validation (non-AI) |
| Mariakakis et al[15] | Detect jaundice via scleral colour using a smartphone app | 70 subjects (pilot study) | Smartphone app 'BiliScreen' with colour calibration | Sensitivity: 90%, Specificity: 97%, r = 0.89 with bilirubin | Feasible tool for at-home jaundice screening | Pilot study (AI with smartphone imaging) |
| Song et al[13] | Detect and grade Kayser-Fleischer rings in Wilson's disease | Database of 1850 corneal images | YOLO for detection, U-Net for segmentation, ResNet for grading | Accuracy > 95% (recall and specificity) | Facilitates early detection and monitoring of Wilson's disease | AI imaging study |
| Casanova-Ferrer et al[20] | Identify minimal hepatic encephalopathy via eye movements | Cirrhotic patients with and without minimal hepatic encephalopathy | Video-oculography with machine learning classification | 56/177 eye movement metrics were significantly different in minimal hepatic encephalopathy | Provides a functional ocular biomarker for minimal hepatic encephalopathy diagnosis | Clinical diagnostic study (AI with VOG) |
| Li et al[33] | Predict systemic diseases from retinal fundus images | General population | Deep learning models on retinal fundus images | Demonstrated potential in detecting diseases, including hepatobiliary conditions | Highlights the utility of retinal imaging in systemic disease screening | AI imaging study |
In summary, the application of AI to ocular biomarkers has revealed a unique opportunity to enhance the care of patients with hepatobiliary diseases. The eyes, long known to reflect systemic illness, can now be examined with machine precision to yield quantifiable indicators of liver health. Signs like scleral icterus, K–F rings, retinal microvascular changes, and even abnormal eye movements have demonstrated significant correlations with liver dysfunction. AI algorithms have achieved impressive accuracy in detecting these features in some cases, matching or exceeding human expert performance. These advances translate into concrete clinical benefits: The prospect of earlier diagnosis through accessible screening, for example, catching silent cirrhosis or Wilson's disease via an eye photograph; more refined assessments of disease severity by objectively tracking ocular signs of improvement or worsening; and more frequent, patient-friendly monitoring through telemedicine tools that watch for ocular evidence of complications.
Despite this progress, further work is needed to integrate AI-based ocular biomarkers into routine practice. Robust validation across diverse populations, regulatory approval, and clinician education will all be critical. It must also be remembered that ocular findings complement rather than replace traditional diagnostics—AI interpretations of eye images should be used alongside blood tests and imaging, not in isolation. Nevertheless, the momentum in this field is strong and growing. As multidisciplinary research continues to address current limitations, it is plausible that soon, an eye scan could become a standard component of liver disease evaluation. In essence, leveraging the eye-liver connection with modern AI represents a leap toward precision hepatology, using subtle, individualised ocular markers to guide timely interventions. This approach holds great promise for improving early detection and personalized management of hepatobiliary diseases, ultimately leading to better patient outcomes.
| 1. | Mortensen PW, Wong TY, Milea D, Lee AG. The Eye Is a Window to Systemic and Neuro-Ophthalmic Diseases. Asia Pac J Ophthalmol (Phila). 2022;11:91-93. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 2. | Patel R, Nair S, Choudhry H, Jaffry M, Dastjerdi M. Ocular manifestations of liver disease: an important diagnostic aid. Int Ophthalmol. 2024;44:177. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 3. | Pavlovic Markovic A, Stojkovic Lalosevic M, Mijac DD, Milovanovic T, Dragasevic S, Sokic Milutinovic A, Krstic MN. Jaundice as a Diagnostic and Therapeutic Problem: A General Practitioner's Approach. Dig Dis. 2022;40:362-369. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 4. | Chakraborty U, Chandra A. Kayser-Fleischer rings in Wilson's disease. Am J Med Sci. 2024;367:e47-e48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 5. | Das S, Chandra A. Bitot Spots: A Pathognomonic Sign of Vitamin A Deficiency. Am J Med. 2023;136:e195-e196. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 6. | Li S, Li A, Ruan F, Zhang W, Chen J, Huang C, Jie Y. Evaluation of the Clinical Characteristics of Dry Eye Secondary to Different Types of Liver Diseases. Ophthalmol Ther. 2023;12:2493-2503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 7. | Hagan S, Martin E, Enríquez-de-Salamanca A. Tear fluid biomarkers in ocular and systemic disease: potential use for predictive, preventive and personalised medicine. EPMA J. 2016;7:15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 162] [Cited by in RCA: 244] [Article Influence: 27.1] [Reference Citation Analysis (0)] |
| 8. | Singh R, Joseph A, Umapathy T, Tint NL, Dua HS. Impression cytology of the ocular surface. Br J Ophthalmol. 2005;89:1655-1659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 123] [Cited by in RCA: 134] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 9. | Lietman TM, Dhital SP, Dean D. Conjunctival impression cytology for vitamin A deficiency in the presence of infectious trachoma. Br J Ophthalmol. 1998;82:1139-1142. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 12] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 10. | Stillman AE. Jaundice. In: Clinical Methods: The History, Physical, and Laboratory Examinations. Boston: Butterworths, 1990. [PubMed] |
| 11. | Fotovat-Ahmadi N, Siddiqui O, Ong J, Thanitcul C, Reinhardt C, Cologna SM, Aakalu VK. The ocular surface tear film as a biomarker for systemic health. Ocul Surf. 2025;37:283-300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 12. | Xiao W, Huang X, Wang JH, Lin DR, Zhu Y, Chen C, Yang YH, Xiao J, Zhao LQ, Li JO, Cheung CY, Mise Y, Guo ZY, Du YF, Chen BB, Hu JX, Zhang K, Lin XS, Wen W, Liu YZ, Chen WR, Zhong YS, Lin HT. Screening and identifying hepatobiliary diseases through deep learning using ocular images: a prospective, multicentre study. Lancet Digit Health. 2021;3:e88-e97. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 61] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 13. | Song W, Xin L, Wang J. A grading method for Kayser Fleischer ring images based on ResNet. Heliyon. 2023;9:e16149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 14. | Babenko B, Traynis I, Chen C, Singh P, Uddin A, Cuadros J, Daskivich LP, Maa AY, Kim R, Kang EY, Matias Y, Corrado GS, Peng L, Webster DR, Semturs C, Krause J, Varadarajan AV, Hammel N, Liu Y. A deep learning model for novel systemic biomarkers in photographs of the external eye: a retrospective study. Lancet Digit Health. 2023;5:e257-e264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 15. | Mariakakis A, Banks MA, Phillipi L, Yu L, Taylor J, Patel SN. BiliScreen. Proc ACM Interact Mob Wearable Ubiquitous Technol. 2017;1:1-26. [DOI] [Full Text] |
| 16. | Miah OF, Hossain RM, Latif A, Sarkar U, Paul SK, Paul RS, Ahammod T, Islam MS, Dowel FA, Mahmud MA, Podder MK, Bhuiyan AS, Chowdhury UW. Pattern of Anaemia in Chronic Kidney Disease. Mymensingh Med J. 2019;28:1-7. [PubMed] [DOI] [Full Text] |
| 17. | Khalifa M, Albadawy M. AI in diagnostic imaging: Revolutionising accuracy and efficiency. Comput Methods Programs Biomed Update. 2024;5:100146. [DOI] [Full Text] |
| 18. | Salehi AW, Khan S, Gupta G, Alabduallah BI, Almjally A, Alsolai H, Siddiqui T, Mellit A. A Study of CNN and Transfer Learning in Medical Imaging: Advantages, Challenges, Future Scope. Sustainability. 2023;15:5930. [RCA] [DOI] [Full Text] [Cited by in Crossref: 280] [Cited by in RCA: 76] [Article Influence: 38.0] [Reference Citation Analysis (0)] |
| 19. | Calvo Córdoba A, García Cena CE, Montoliu C. Automatic Video-Oculography System for Detection of Minimal Hepatic Encephalopathy Using Machine Learning Tools. Sensors (Basel). 2023;23:8073. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 20. | Casanova-Ferrer F, García-Cena CE, Gallego JJ, Fiorillo A, Urios A, Calvo-Córdoba A, Ballester MP, Ríos MP, Durbán L, Hidalgo MR, García F, Felipo V, Montoliu C. Minimal hepatic encephalopathy is associated to alterations in eye movements. Sci Rep. 2022;12:16837. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 21. | Fortea J, Carrera IG, Puente A, Crespo J. Cirrosis hepática. Med - Programa de Formación Médica Continuada Acreditado. 2020;13:297-307. [DOI] [Full Text] |
| 22. | Kazankov K, Nixon-Hill M, Kumar R, Amin A, Alabsawy E, Chikhlia A, Leung TS, Mookerjee RP. A novel smartphone scleral-image based tool for assessing jaundice in decompensated cirrhosis patients. J Gastroenterol Hepatol. 2023;38:330-336. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 5] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 23. | Eckstein AK, Reichenbach A, Jacobi P, Weber P, Gregor M, Zrenner E. Hepatic retinopathia. Changes in retinal function. Vision Res. 1997;37:1699-1706. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 31] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 24. | Krismer F, Roos JC, Schranz M, Graziadei IW, Mechtcheriakov S, Vogel W, Carpenter RH, Zoller H. Saccadic latency in hepatic encephalopathy: a pilot study. Metab Brain Dis. 2010;25:285-295. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 25. | Jacobi C, Wenkel H, Jacobi A, Korn K, Cursiefen C, Kruse FE. Hepatitis C and ocular surface disease. Am J Ophthalmol. 2007;144:705-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 20] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 26. | Abd El-Badie Mohamed M, Abd-El Azeem Eed K. Retinopathy associated with interferon therapy in patients with hepatitis C virus. Clin Ophthalmol. 2012;6:1341-1345. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 27. | Alshahrani S, Aljumah AA, Alluhaidan A. Uveitis and autoimmune hepatitis, a real entity? A case report with review of the literature. Saudi J Ophthalmol. 2021;35:73-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 28. | Harris J, Cao S, Hile G, Eshaq M. Diffuse xanthomas in a patient with primary biliary cholangitis and lipoprotein X. JAAD Case Rep. 2021;7:30-32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 29. | Reshetnyak VI. Primary biliary cirrhosis: Clinical and laboratory criteria for its diagnosis. World J Gastroenterol. 2015;21:7683-7708. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 30. | Selmi C, Gershwin ME. Chronic Autoimmune Epithelitis in Sjögren's Syndrome and Primary Biliary Cholangitis: A Comprehensive Review. Rheumatol Ther. 2017;4:263-279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 31. | Langwińska-Wośko E, Litwin T, Dzieżyc K, Członkowska A. The sunflower cataract in Wilson's disease: pathognomonic sign or rare finding? Acta Neurol Belg. 2016;116:325-328. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 32. | Kazankov K, Novelli S, Chatterjee DA, Phillips A, Balaji A, Raja M, Foster G, Tripathi D, Boddu R, Kumar R, Jalan R, Mookerjee RP. Evaluation of CirrhoCare® - a digital health solution for home management of individuals with cirrhosis. J Hepatol. 2023;78:123-132. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 20] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 33. | Li Y, Zhang R, Dong L, Shi X, Zhou W, Wu H, Li H, Yu C, Wei W. Predicting systemic diseases in fundus images: systematic review of setting, reporting, bias, and models' clinical availability in deep learning studies. Eye (Lond). 2024;38:1246-1251. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 34. | Mantena S, Celi LA, Keshavjee S, Beratarrechea A. Improving community health-care screenings with smartphone-based AI technologies. Lancet Digit Health. 2021;3:e280-e282. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 35. | Li Z, Wang L, Wu X, Jiang J, Qiang W, Xie H, Zhou H, Wu S, Shao Y, Chen W. Artificial intelligence in ophthalmology: The path to the real-world clinic. Cell Rep Med. 2023;4:101095. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 59] [Reference Citation Analysis (0)] |
| 36. | Peng Q, Tseng RMWW, Tham YC, Cheng CY, Rim TH. Detection of Systemic Diseases From Ocular Images Using Artificial Intelligence: A Systematic Review. Asia Pac J Ophthalmol (Phila). 2022;11:126-139. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 37. | Madanian S, Nakarada-Kordic I, Reay S, Chetty T. Patients' perspectives on digital health tools. PEC Innov. 2023;2:100171. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 66] [Reference Citation Analysis (0)] |
| 38. | Al Kuwaiti A, Nazer K, Al-Reedy A, Al-Shehri S, Al-Muhanna A, Subbarayalu AV, Al Muhanna D, Al-Muhanna FA. A Review of the Role of Artificial Intelligence in Healthcare. J Pers Med. 2023;13:951. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 102] [Cited by in RCA: 176] [Article Influence: 88.0] [Reference Citation Analysis (0)] |
| 39. | Murdoch B. Privacy and artificial intelligence: challenges for protecting health information in a new era. BMC Med Ethics. 2021;22:122. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 250] [Article Influence: 62.5] [Reference Citation Analysis (0)] |
| 40. | Artificial Intelligence in Health Care: The Hope, the Hype, the Promise, the Peril. Washington (DC): National Academies Press (US), 2023 . [PubMed] |
| 41. | Yurdem B, Kuzlu M, Gullu MK, Catak FO, Tabassum M. Federated learning: Overview, strategies, applications, tools and future directions. Heliyon. 2024;10:e38137. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 42. | Rosenbacke R, Melhus Å, McKee M, Stuckler D. How Explainable Artificial Intelligence Can Increase or Decrease Clinicians' Trust in AI Applications in Health Care: Systematic Review. JMIR AI. 2024;3:e53207. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 43. | Rao D. The future of healthcare using multimodal AI: Technology that can read, see, hear and sense. Oral Oncol Rep. 2024;10:100340. [DOI] [Full Text] |
| 44. | Sabry F, Eltaras T, Labda W, Alzoubi K, Malluhi Q. Machine Learning for Healthcare Wearable Devices: The Big Picture. J Healthc Eng. 2022;2022:4653923. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 75] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 45. | Shajari S, Kuruvinashetti K, Komeili A, Sundararaj U. The Emergence of AI-Based Wearable Sensors for Digital Health Technology: A Review. Sensors (Basel). 2023;23:9498. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 86] [Reference Citation Analysis (0)] |
| 46. | Johnson KB, Wei WQ, Weeraratne D, Frisse ME, Misulis K, Rhee K, Zhao J, Snowdon JL. Precision Medicine, AI, and the Future of Personalized Health Care. Clin Transl Sci. 2021;14:86-93. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 531] [Article Influence: 106.2] [Reference Citation Analysis (0)] |