Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.108772
Revised: June 6, 2025
Accepted: July 22, 2025
Published online: August 27, 2025
Processing time: 126 Days and 22.7 Hours
Non-alcoholic fatty liver disease (NAFLD) is the fastest-growing global contributor to the disease burden associated with the consequences of chronic liver disease, including cirrhosis and liver cancer. It is projected that more than fifty percent of the adult population, including women, smokers, and individuals without metabolic syndrome, will have NAFLD by 2040. Various mechanisms linking the gut microbiome to NAFLD and the consequent fibrosis have been discerned, which suggest the dysbiosis-induced impairment of gut endothelial barrier function, leading to hepatic inflammation through the translocation of bacterial components. NAFLD is progressively associated with environmental variables, especially exposure to heavy metals that impair liver metabolism, produce oxidative stress, and exacerbate inflammation, hence accelerating its progression. These toxicants also modify the composition of gut microbiota, hence intensifying liver damage. Comprehending the processes by which heavy metals contribute to NAFLD is essential for formulating tailored therapies. This review examines strategies to alleviate liver toxicity caused by heavy metals, including chelation therapy, dietary modifications (antioxidants and hepatoprotective nutrients), gut microbiome modulation via probiotics and postbiotics like short-chain fatty acids to restore intestinal barrier function and use of essential minerals like selenium, with potent antioxidant characteristics. Employing these measures may offer an integrated approach for addressing NAFLD in individuals subjected to heavy metal poisoning.
Core Tip: A combination of factors, such as nutrition, oxidative stress, inflammation, gut dysbiosis, and heredity, causes a complicated metabolic disease, non-alcoholic fatty liver disease (NAFLD). Environmental pollutants are becoming more prominent factors in its development. In this review, we will be discussing the treatment regimens that incorporate genetic, microbiological, and metabolic characteristics that are essential. Along with lifestyle changes, new approaches like microbiota-targeted treatments and selenium supplementation show promise but require more research. To create accurate and efficient therapies for the prevention and management of NAFLD, future research should prioritize early screening, particularly in high-risk groups, and concentrate on the dynamics of the gut-liver axis.
- Citation: Sharma T, Kaushal N, Garla R. Toxins to treatments: Impact of environmental pollutants, gut microbiota, and natural compounds on non-alcoholic fatty liver disease progression. World J Hepatol 2025; 17(8): 108772
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/108772.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.108772
Non-alcoholic fatty liver disease (NAFLD) is associated with the accumulation of fats (mainly triglycerides) in the liver of non-alcohol-consuming subjects[1]. It was first described in the 1980s. Over three decades, a continuous increase has been observed in the crude global NAFLD prevalence (from 10.5% to 16.0%) among all ages[2]. South Asia has become the epicentre of the explosive increase in NAFLD prevalence rates, with India and China contributing the most. According to recent reports, the global incidence of NAFLD among adults is 30%, and among children is 7.4%[3]. NAFLD is also the leading cause of liver-related morbidity and mortality.
NAFLD diagnosis can only be performed in the absence of other chronic liver disorders like autoimmune diseases, alcohol consumption, etc.[1]. For the diagnosis, liver biopsy is the gold standard that is based on the presence of 5% hepatic steatosis[4]. Nowadays, new diagnostic criteria have been developed, which rely on non-invasive diagnostic techniques like ultrasound, computed tomography, and magnetic resonance imaging, along with metabolic parameters (changes in lipid composition, waist circumference, fasting sugar levels, aspartate aminotransferase (AST) and alanine aminotransferase (ALT) ratio, type 2 diabetes mellitus, body mass index (BMI), etc.[5]. Fatty liver index, NAFLD fat score, and NAFLD fibrosis score are examples of a few scoring systems developed for the diagnosis of NAFLD and liver content using these techniques[5,6]. However, all of these systems have some recognizable limitations.
Earlier, the pathogenesis of NAFLD was explained with the help of the ‘two-hit’ hypothesis[7]. Accumulation of lipids is considered to be the first hit that further increases the susceptibility of the liver to various factors that constitute the second hit, leading to inflammation, hepatic injury, and fibrosis[8]. Currently, this hypothesis is replaced by the ‘multiple hit’ hypothesis that considers multiple factors (such as lipotoxicity, diet, sedentary lifestyle, epigenetics, insulin resistance, innate immune activation, environmental pollution, etc.) responsible for the progression of NAFLD[9]. For example: Dietary variables, especially meals high in fat and sugar, changed microbial metabolites, including bile acids, endotoxins, and short-chain fatty acids (SCFAs), which worsen intestinal barrier dysfunction. Decreases in healthy SCFAs, such as acetate, butyrate, and propionate, weaken gut integrity and mucosal immunity, which makes it easier for bacterial components to circulate across the body's tissues. Along with genetic predisposition, epigenetic factors such as DNA methylation, histone modifications, and non-coding RNAs, which manage gene expression without changing the DNA sequence, play a significant role in the development of NAFLD.
Growing shreds of evidence supported that environmental pollution, especially heavy metal exposure, contributes to NAFLD risk[10]. The mechanisms of development of metal-induced hepatic steatosis were shown to involve oxidative stress, endoplasmic reticulum (ER) stress, pyroptosis, and induction of an inflammatory response[11,12]. Moreover, the epigenetic effects of the metals, as well as their effect on gut microbiota and gut wall integrity, have also been shown to mediate their role in NAFLD development[10]. The therapeutic approaches developed against metal exposure-induced NAFLD include strategies to increase the antioxidant capacity of cells or sequestration of metals with the help of chelators. Because of the side effects of regular use of chelators, the use of nutritional supplements (gut microbiota-based therapeutic approaches) is a better approach. In contrast to probiotics and prebiotics, which depend on the host's gut microbiota to function, postbiotics, like SCFAs, provide a more standardized approach. Three SCFAs, such as butyrate, propionate, and acetate, are excellent at reducing inflammation, improving insulin function, and improving mito
In this regard, the antioxidant properties of essential metals like zinc (Zn) and selenium (Se) are proven to be effective in providing protection against metal-induced hepatotoxicity[13]. Acting as a co-factor for more than 300 enzymes and proteins, Zn performs a crucial role in various physiological functions and cellular signaling pathways[14]. In various studies, a positive relationship between serum Zn concentrations and the probability of developing NAFLD has been documented[14-16]. Se, another essential metal, is the integral component of antioxidant selenoproteins like glutathione peroxidase (GPx) and thioredoxin reductases (TrxR), etc.[17]. It also plays a pivotal part in hormone activation, development, immune function, reproduction, and cancer prevention, etc.[17]. There are few studies suggesting link of Se with NAFLD[18,19]. The understanding of the role of Se in preventing the progression of NAFLD remains limited.
Despite the overwhelming literature available on NAFLD, we are still far from being able to predict several molecular and metabolic changes associated with it and tailored therapies for combating it. The current report will, therefore, delve into different perspectives from previous reviews on NAFLD. This review mainly focuses on the current state of the art used for alleviating liver toxicity caused by heavy metals, including dietary modifications (antioxidants and hepatoprotective nutrients), gut microbiome modulation via postbiotics like SCFAs and use of essential minerals like Se, with potent antioxidant characteristics.
In the three decades since its discovery, NAFLD is estimated to affect approximately one-third of the population worldwide[20]. The global prevalence of NAFLD has heightened from 25.3% in 1990-2006 to 38.0% in 2016-2019 in accordance with a systematic meta-analysis[21]. The highest rates of prevalence are observed in the Middle East (32%) and the lowest in Africa (14%). In Asia, the prevalence stands at 27%, followed by 24% in North America and 23% in Europe[21]. NAFLD has become the most rapidly increasing etiology of hepatocellular carcinoma in the United States, France, and the United Kingdom[22]. The peak prevalence of NAFLD is reported between the ages of 50 and 60 years in males, with a rate of 29.3%, whereas, 21.6%, for females. Although the prevalence is lower in women, it rises significantly after menopause[23]. Taking into consideration, the rising rates of obesity and diabetes among younger populations, several studies have begun to investigate the prevalence of NAFLD in these age groups. One such study reported that approximately 8% to 10% of children aged 2 to 19 years are affected by NAFLD, with a prevalence of around 26% among those classified as obese[24]. Another study, which analyzed autopsy data from children (aged 2 to 19 years) who died in New York city between 2005 and 2010, found a 4.5% prevalence of NAFLD, with 1.7% having non-alcoholic steatohepatitis (NASH)[25].
The prevalence of NAFLD among the Indian population varies from 9% to 53%[26]. The wide variation in prevalence can be attributed to the rural-urban disparity and geographical variation in the dietary patterns among Indian popu
NAFLD is intricately associated with insulin resistance, a metabolic disorder characterized by an inadequate cellular response to insulin that results in systemic metabolic dysfunction. Insulin resistance in the liver leads to excessive lipid buildup, a hallmark of hepatic steatosis[29]. Under conventional physiological circumstances, insulin inhibits lipolysis, which is the degradation of triglycerides into free fatty acids (FFAs) and glycerol in adipose tissue. When there is insulin resistance, the fat cells don't respond to insulin's ability to stop hormone-sensitive lipase, the primary enzyme that breaks down triglycerides[30]. Consequently, there is an unregulated discharge of FFAs into the bloodstream. The additional circulating FFAs are absorbed by the liver, where they facilitate hepatic lipid buildup and cause lipotoxicity[31]. Furthermore, the elevated FFA load intensifies insulin resistance in the liver by activating protein kinase C epsilon, so disrupting insulin receptor signaling and exacerbating metabolic dysfunction[32].
Alongside the elevated influx of FFAs from adipose tissue, the liver additionally produces its fatty acids via a process known as de novo lipogenesis (DNL)[33]. Insulin and glucose levels tightly control this process. They do this by turning on lipogenic transcription factors such as sterol regulatory element-binding protein-1c (SREBP-1c) and carbohydrate-responsive element-binding protein (ChREBP)[34]. In insulin-resistant conditions, hepatic insulin signaling is disrupted, resulting in the aberrant overexpression of SREBP-1C and ChREBP, which promote excessive lipogenesis. Fructose in food is a big reason for this imbalance.
The liver secretes triglycerides as very low-density lipoproteins (VLDL) to prevent lipid buildup. In NAFLD, this pathway becomes ineffective because of an imbalance between lipid production and exportation. A protein called apolipoprotein B (ApoB) is needed to make VLDL. ApoB is an important part of the structure of VLDL particles. In insulin-resistant conditions, the production and stability of ApoB are impaired, resulting in diminished VLDL secretion. This leads to an imbalance between triglyceride synthesis and elimination, resulting in hepatic steatosis[35].
The accumulation of excess lipids in hepatocytes triggers a cascade of metabolic disturbances that lead to cellular damage. Some of the main things that make the disease worse are mitochondrial dysfunction, reactive oxygen species (ROS) overproduction, ER stress, and hepatocyte apoptosis. These mechanisms collectively result in hepatocyte injury, inflammation, and fibrosis, accelerating the deterioration of liver function[36].
In a healthy liver, mitochondria effectively metabolize fatty acids via β-oxidation, producing ATP that sustains cellular processes[37]. In NAFLD, the increased input of FFAs surpasses the mitochondrial oxidative capability, resulting in inefficient fatty acid oxidation[38]. This mechanism leads to the accumulation of partially oxidized lipid intermediates, which compromise mitochondrial integrity and inhibit ATP production[39]. The overproduction of ROS is a significant effect of mitochondrial malfunction. Cellular antioxidant systems neutralize the trace levels of ROS produced by mitochondria as a result of oxidative phosphorylation under normal physiological conditions[40]. Therefore, in NAFLD, this excessive generation of ROS exceeds the liver's antioxidant defence capabilities.
Protein folding, lipid metabolism, and cellular homeostasis are all significantly impacted by the ER[41]. Hepatocytes in NAFLD struggle with an excessive lipid load, which impairs ER function and causes ER stress. The unfolded protein response, a cellular response to this stress, speeds up the breakdown of misfolded proteins, slows down total protein synthesis, and improves C/EBP homologous protein (CHOP), a key player in this process. CHOP levels up during long-term ER stress and triggers apoptosis by blocking pathways that help cells stay alive[42]. ER stress also makes hepatic lipid buildup and cell dysfunction worse by messing up the metabolism of lipids through misfolded proteins, which causes lipotoxicity[43]. The progress of hepatocyte apoptosis has a big effect on the progression of NAFLD because dying hepatocytes make damage-associated molecular patterns that turn on hepatic stellate cells (HSCs) and Kupffer cells[44]. The liver's setting exacerbates this activation, resulting in excessive extracellular matrix deposition and chronic inflammation. An increase in hepatocytes initiates a series of immune system reactions that intensify the environment's inflammation, attract immune cells from the bloodstream, and maintain the activity of resident macrophages[45].
The course of NAFLD is characterized by chronic inflammation, which progresses from basic hepatic steatosis to NASH and fibrosis. When hepatocytes get too much fat, it sets off a chain of immune reactions that make the inflammatory response stronger, bring in immune cells from the blood, and keep resident macrophages active. The dysregulated immune response exacerbates hepatocellular damage, promotes fibrogenesis, and ultimately contributes to the disease's progression[46].
The liver's resident macrophages, known as Kupffer cells, are essential in coordinating the inflammatory response in NAFLD. By eliminating apoptotic cells and microbial waste, Kupffer cells preserve immunological homeostasis under typical physiological circumstances. On the other hand, excessive lipid buildup in NAFLD causes lipotoxic stress, which activates Kupffer cells and causes them to release pro-inflammatory cytokines[47].
Tumor necrosis factor (TNF)-α is a big part of the inflammatory response. It causes insulin resistance, mitochondrial dysfunction, and the death of interleukin-6 (IL-6). IL-6 also makes the liver more inflamed by turning on the JAK/STAT pathway. This leads to more hepatocyte proliferation and fibrosis. By increasing oxidative stress and promoting neutrophil recruitment, interleukin-1β (IL-1β) intensifies inflammation even further[48,49]. Experimental evidence with bacterial endotoxins like lipopolysaccharide (LPS) demonstrates that the gut-liver axis also plays a part in activating Kupffer cells because it makes it easier for bacterial endotoxins like LPS to enter the portal circulation. By activating Kupffer cells via TLR4 signalling, LPS causes the generation of inflammatory mediators and the activation of NF-κB. This condition creates an atmosphere that supports fibrogenesis and prolongs hepatocellular damage[49,50].
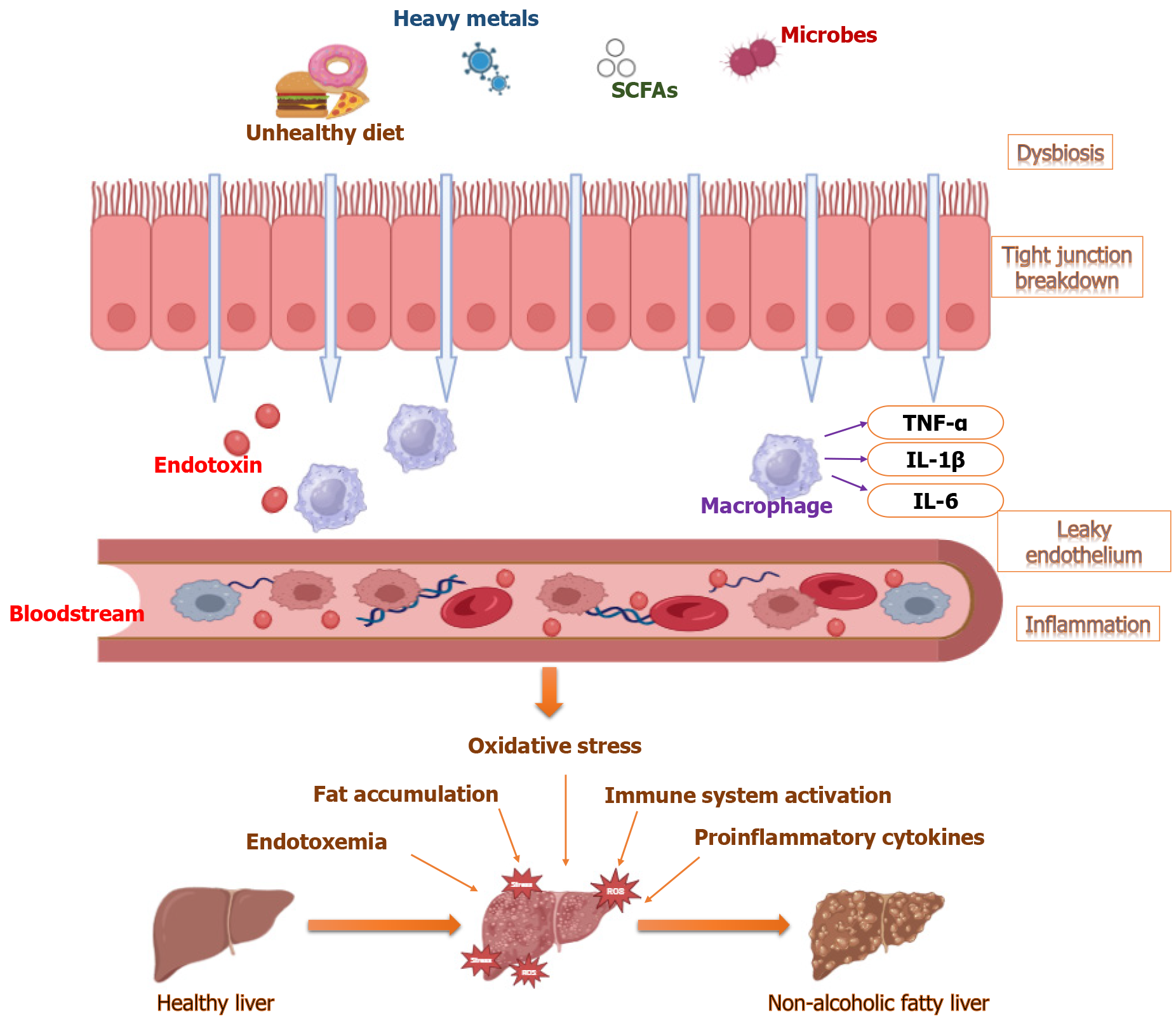
Energy balance, immunological responses, and host metabolism are all significantly influenced by the gut microbiome. Two signs of gut microbiota dysbiosis, i.e., a change in the variety of bacteria and an increase in species that cause inflammation, can alter the gut's microbial composition[51]. Figure 1 summarizes the role of gut-liver in the pathophysiology of NAFLD. Studies have shown that people with NAFLD have a different mix of microbes in their gut, with more Proteobacteria and Firmicutes and fewer Bacteroidetes. This causes metabolic problems that make hepatic steatosis and inflammation worse[5].
Impaired intestinal barrier integrity, which raises gut permeability, is a major effect of gut dysbiosis in NAFLD[52]. Tight junction proteins, like occludin, claudins, and zonula occludens-1, protect the lining of the intestine by controlling selective permeability and stopping the movement of products made by microbes into the bloodstream[53]. In NAFLD, gut dysbiosis causes tight junction dysfunction, leading to heightened intestinal permeability. Microbial components like LPS translocate to the liver because of increased intestinal permeability, where they activate hepatic cells. This leads to the development of hepatic inflammation, insulin resistance, and the transition from steatosis to NASH by inducing NF-κB signaling and the production of pro-inflammatory cytokines[52]. HSCs are also activated by the chronic inflammatory state, which promotes fibrosis and extracellular matrix deposition[54].
The aetiology of NAFLD is complicated and depends on many factors, including food patterns, gut microbiota com
One important factor influencing the development and susceptibility of NAFLD is genetic predisposition. Through modifications to inflammatory pathways, glucose control, and lipid metabolism, a number of gene variations play a role in the development of NAFLD. Triglyceride hydrolysis in hepatocytes is impeded by the PNPLA3 gene, especially the I148M variation, which causes lipid retention in the liver. This buildup may affect inflammation and fibrosis and result in steatosis[55].
Hepatic lipid processing is impacted by the TM6SF2 gene, particularly the E167K variation, which decreases the export of VLDL. This results in elevated hepatic fat content[56]. Changes in phospholipid remodelling in cell membranes caused by the MBOAT7 gene variation may impact inflammatory signalling and the buildup of liver fat[57].
Variants like P446 L that affect the GCKR gene increase hepatic glucose absorption and glucokinase activity. This alteration promotes DNL, which helps hepatocytes accumulate triglycerides[58]. However, in the context of hepatic steatosis, a variation in the HSD17B13 gene is linked to a lower risk of inflammation and fibrosis. By altering the activity of enzymes involved in lipid metabolism, this variation may have an impact on hepatic inflammation[59]. These genetic variables contribute to the onset and progression of NAFLD in a variety of populations, either alone or in combination with metabolic and environmental factors.
Along with genetic predisposition, epigenetic factors, which manage gene expression without changing the DNA sequence, play a big role in the development of NAFLD. Environmental and metabolic factors like food, gut microbiota, and insulin resistance affect epigenetic changes, such as DNA methylation, histone modifications, and non-coding RNAs[60]. One important epigenetic alteration that controls hepatic gene expression in NAFLD is DNA methylation. NAFLD patients have been found to have hypermethylated genes related to lipid metabolism (e.g., PPARα and CPT1A), insulin signalling (e.g., IRS2), and mitochondrial function (e.g., PGC-1α), which results in metabolic dysregulation and hepatocellular damage[61]. Additionally, promoter hypomethylation of pro-inflammatory cytokines like TNF-α and IL-6 makes liver inflammation worse and speeds up the disease's progression.
In NAFLD, histone changes like acetylation, methylation, and phosphorylation control gene transcription and chromatin accessibility. Histone methylation of fibrogenic genes (TGF-β and COL1A1) leads to more extracellular matrix buildup, which accelerates the fibrosis process. On the other hand, increased histone acetylation of SREBP-1c and FASN promotes the formation of new fat cells in the liver[62,63].
NAFLD pathogenesis is further influenced by non-coding RNAs (ncRNAs), especially microRNAs (miRNAs) and long ncRNAs. Important lipid metabolism regulators, miR-34a and miR-122, are turned up in people with NAFLD. This procedure makes fatty acid oxidation go down, and lipogenesis go up. Conversely, the downregulation of miR-192 and miR-223 encourages hepatic inflammation and fibrosis[64,65].
The pathophysiology of NAFLD is mostly driven by the disruption of the gut microbiota, which is essential for host metabolism, immunological response, and liver health. With a higher Firmicutes-to-Bacteroidetes ratio, an increase in Proteobacteria, and a decrease in beneficial butyrate-producing bacteria, such as Faecalibacterium prausnitzii and Roseburia spp., the gut microbial composition of individuals with NAFLD shifts towards pro-inflammatory and lipogenic taxa[66].
SCFAs, including acetate, propionate, and butyrate, are fermented byproducts of dietary fibres and resistant starches produced by gut bacteria. These metabolites are important modulators of inflammation, glucose homeostasis, and hepatic lipid metabolism[67]. However, in individuals with NAFLD, alterations in the gut microbiota can disrupt the balance of SCFA production, exacerbating fibrosis, insulin resistance, and hepatic steatosis[68].
As industrialization and urbanization accelerate, increased exposure to environmental pollutants—both through inhalation of particulate matter and through diet—has emerged as a potential risk factor for the development of NAFLD[69]. Persistent organic pollutants are a class of long-lasting, carbon-based chemicals commonly used in food production, particularly as pesticides[70]. These compounds tend to bioaccumulate in adipose tissue and may also be retained in the liver, where they can contribute to the development of metabolic disorders[71]. Evidence from multiple studies indicates that the pathogenic effects of PM2.5 on cardiovascular disease and diabetes are mediated primarily through inflammation, oxidative stress, and insulin resistance, all of which are core features in the progression of NAFLD[72,73]. Furthermore, increasing evidence suggests a potential link between exposure to certain toxic metals and both the initiation of NAFLD and its progression to NASH[74]. Exposure to cadmium, lead, mercury, and arsenic has been correlated with disruptions in hepatic biochemical markers, thereby contributing to an elevated risk of NAFLD[74]. Various epidemiological studies and case reports of severely intoxicated or occupationally exposed subjects have proven the connection between As and Lead exposure and hepatic steatosis. Recent experimental studies have provided insight into the prospective mechanisms underlying this link. Heavy metals promote the accumulation of hepatic lipids by dysregulating the lipid metabolism through up-regulation of lipogenic and down-regulation of lipolytic genes, which may be mediated by modulation of SREBP1, PPARα/γ, AMPK, and C/EBPs signalling[10,75]. Other contributing mechanisms involve oxidative stress linked to dysregulated SIRT-1 signaling, ER stress, ferroptosis, impaired autophagic processes, and heightened inflammation via NF-κB activation and NLRP3 inflammasome engagement[75].
Exposure to heavy metals triggers epigenetic modifications—such as altered DNA methylation patterns, dysregulated miRNA expression, and changes in histone structure[76,77]. In most of the animals, inorganic As gets methylated in liver. This process is accomplished by methyltransferases using S-adenosyl-methionine (SAM), as the methyl donor[78]. SAM is also required for most other cellular methylation reactions, including, DNA methylation. Therefore, it has been reported that chronic arsenic exposure in vivo induces hepatic DNA hypomethylation, and inappropriate expression of genes[76]. Contrarily, Hg exposure caused an increased DNA methylation at the promoter region of the GSTM1 in women and hypomethylation of the promoter region of the SEPP1[77]. Moreover, systemic (extrahepatic) effects of these metals may further exacerbate steatosis in the liver[76,77]. Corroborating the role of obesity as a leading risk factor for NAFLD, dysregulated adipogenesis, and adipose tissue dysfunction upon metal exposure may promote ectopic lipid accumulation[79].
Moreover, heavy metal-induced dysbiosis and compromised intestinal barrier function play a role in hepatic steatosis by facilitating increased LPS translocation into the bloodstream, thereby triggering inflammatory pathways[80]. Additionally, disruption of gut microbiota caused by metal exposure is associated with shifts in microbial metabolite production, which can significantly impact host lipid and glucose metabolism[10]. Table 1 presents a summary of key studies investigating the relationships among heavy metals, gut microbiota, and Se in the context of NAFLD.
| Metal | Study design | Model | Key findings |
| Cadmium exposure and NAFLD[189] | Animal study | HFD-fed mice + Cd | Cadmium worsened hepatic lipid accumulation and gut dysbiosis; increased Proteobacteria, decreased Lactobacillus |
| Pb exposure and gut-liver axis[190] | Animal study | Pb-exposed mice | Pb caused intestinal barrier dysfunction, elevated LPS, aggravated hepatic inflammation and steatosis |
| Selenium supplementation[191] | Animal study | HFD-fed mice | Selenium improved hepatic lipid metabolism, increased Akkermansia, improved antioxidant enzyme activity |
| Selenium and gut microbiota[192] | Animal study | HFD-fed mice | Selenium enriched SCFA-producing microbiota; reduction in hepatic TGs, decline in inflammatory cytokines |
| Arsenic exposure and microbiome[193] | Animal study | Mice + arsenic | Arsenic altered microbiota composition, increased gut permeability and promoted liver fibrosis |
| Cadmium, microbiota, and liver injury[189,194] | Animal study | Cd-exposed mice | Cd exposure induced Firmicutes/Bacteroidetes imbalance; elevation in inflammatory cytokines and liver damage |
| Selenium nanoparticles in NAFLD[195] | Animal study | HFD + SeNPs | SeNPs improved oxidative stress, modulated microbiota, and reduced NAFLD severity |
| Selenium status and NAFLD/NASH[166] | Meta-analysis | Human studies | Low selenium significantly associated with NAFLD and NASH risk; supplementation showed mixed results |
Reducing the toxic burden of metals and metalloids may represent a viable approach to lowering the risk of hepatotoxicity and the onset or exacerbation of NAFLD/NASH.
Approaches such as chelation with targeted agents[81], combination therapies[82], polyphenolic plant compounds[83], and supplementation with essential trace elements like Se[84] may effectively reduce metal toxicity by exerting antioxidant and anti-inflammatory actions and facilitating the elimination of metals.
Finally, considering the established role of gut dysbiosis in metal-associated NAFLD, the use of probiotics has demonstrated promise in mitigating heavy metal toxicity[80]. Nevertheless, the underlying mechanisms and clinical effectiveness of such interventions remain insufficiently understood. Therefore, further research is necessary to elucidate the complex relationship between heavy metal exposure and the development of NAFLD/NASH[10]. This includes a deeper investigation into the molecular pathways involved, which may ultimately lead to the identification of novel therapeutic targets for the treatment and prevention of metal-induced liver disease and its progression to more advanced pathological states.
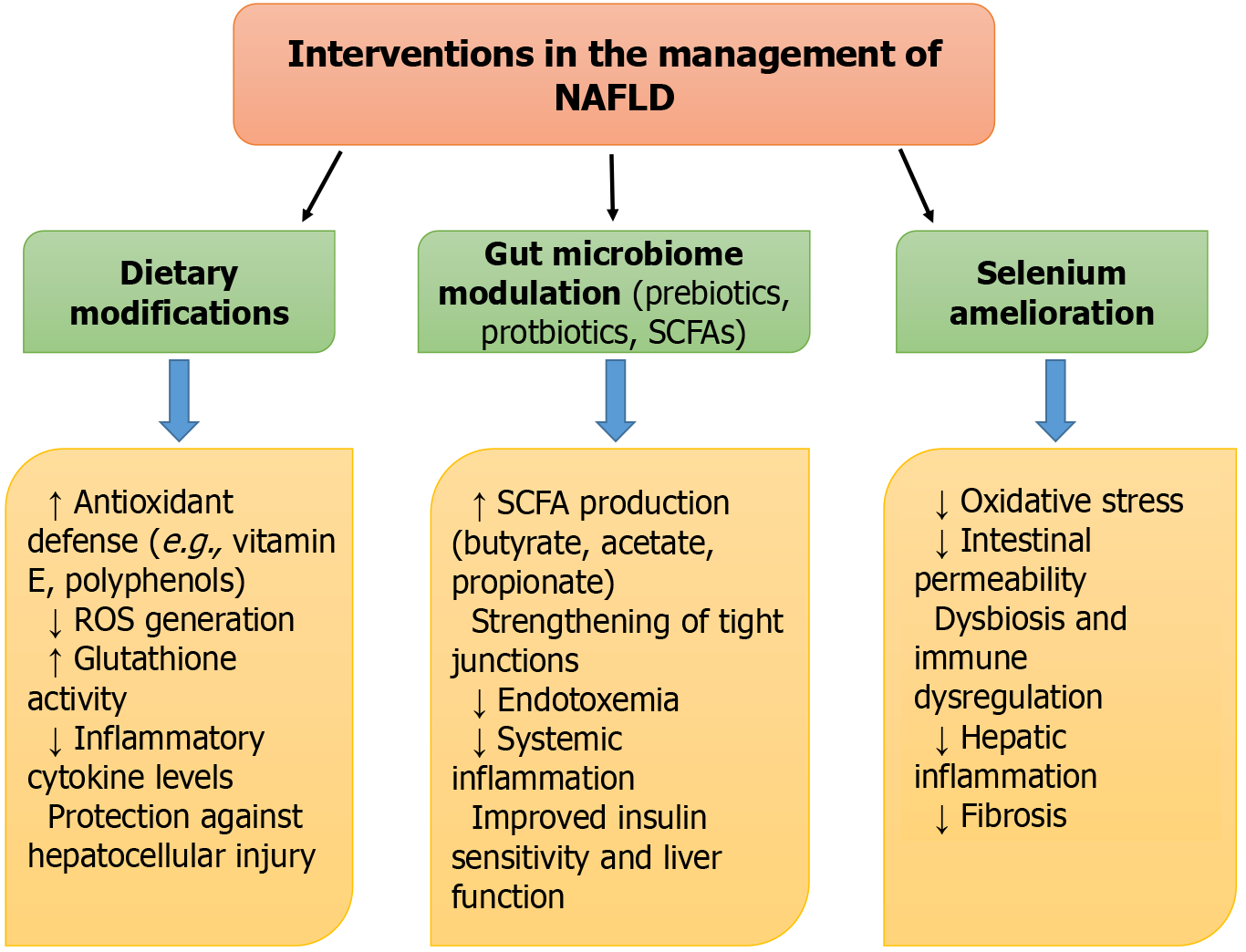
The management and treatment of NAFLD typically involves a combination of lifestyle modifications, pharmacologic interventions, gut microbiota and essential metals-based therapies compiled in Figure 2.
The primary management strategy for NAFLD is dietary treatments aimed at lowering inflammation, increasing insulin sensitivity, and lessening hepatic fat buildup. A crucial element is calorie restriction; even a 7%-10% drop in body weight shows notable improvements in hepatic steatosis, and histological indicators of disease development[85,86]. Particularly effective in lowering oxidative stress and hepatic lipid buildup is the mediterranean diet (MD), which is high in fiber, antioxidants, polyunsaturated fatty acids, and monounsaturated fatty acids. The MD is distinguished by a diet low in processed foods, refined carbohydrates, and saturated fats and high in nuts, fish, whole grains, and fresh vegetables[87]. This diet has anti-inflammatory and insulin-sensitizing effects because it contains a lot of omega-3 fatty acids, which inhibit hepatic lipogenesis and encourage fatty acid oxidation[88]. Low-carb and ketogenic diets have shown promise in treating NAFLD by lowering hepatic DNL and raising mitochondrial fatty acid oxidation[89]. These diets improve insulin signaling and liver lipid metabolism by reducing hepatic glucose input. However, long-term use of severely low-carb diets may change the composition of the gut microbiota, which calls for a balanced dietary strategy[90].
Given the detrimental effects of high fructose consumption on insulin resistance and hepatic steatosis, fructose restriction is another essential dietary approach. By turning on the ChREBP, a high-fructose diet raises the production of uric acid and DNL in the liver. This makes inflammation and oxidative stress worse. Systemic insulin resistance and intrahepatic triglyceride buildup have been shown to significantly decrease when dietary fructose intake is decreased[91].
Along with a healthy diet, exercise is a powerful non-pharmacological treatment for NAFLD, improving insulin sensitivity, systemic inflammation, and hepatic lipid metabolism. Even without noticeable weight reduction, exercise improves mitochondrial activity, speeds up the clearance of hepatic triglycerides, and increases liver glucose utilisation[92]. It has been demonstrated that resistance training and moderate-to-intense aerobic exercise can both successfully lower hepatic fat content and metabolic dysfunction. By activating AMPK and PGC-1α, aerobic exercise improves mitochondrial biogenesis in the liver. This process lowers hepatic lipogenesis and raises fatty acid oxidation[93]. In comparison to moderate-intensity continuous training, high-intensity interval training has shown itself to be a particularly successful method, resulting in higher decreases in hepatic fat content[94]. Resistance exercise reduces circulating FFAs that cause hepatic steatosis by promoting increased lipid oxidation, glucose absorption through skeletal muscle hypertrophy, and improved insulin signalling. Resistance exercise improves peripheral and hepatic insulin sensitivity by increasing GLUT4 translocation[95]. Frequent exercise also helps to modulate the gut microbiota, which increases the number of bacteria that produce butyrate and improves hepatic metabolic balance[96].
As evidence for the therapeutic benefits of bariatric surgery in NAFLD and NASH continues to grow, it has become a highly effective intervention for morbid obesity and metabolic diseases. Hepatic steatosis, insulin resistance, and systemic inflammation all significantly improve as a result of the substantial and long-lasting weight loss brought about by bariatric surgeries[97].
Of the many surgical procedures, sleeve gastrectomy and Roux-en-Y gastric bypass have shown the best results in reversing hepatic fibrosis, lowering hepatic lipid buildup, and improving metabolic dysfunction[97]. Bariatric surgery changes the metabolism by changing the composition of the gut microbiota, the way bile acids are broken down, and the release of gut hormones (GLP-1, PYY)[98]. Furthermore, the severe calorie restriction following surgery causes a rise in fatty acid β-oxidation and a decrease in hepatic DNL[99].
According to clinical research, weight loss surgery greatly reduces liver fibrosis and inflammatory scores in people with NASH. Twenty-five percent of the 293 patients who participated in a lifestyle weight loss intervention over 52 weeks achieved resolution of steatohepatitis, 47% exhibited a decrease in NASH activity score, and 19% showed regression of liver fibrosis. In a significant prospective study, 25% of the 293 patients who participated in a lifestyle weight loss intervention over 52 weeks achieved resolution of steatohepatitis, 47% exhibited a drop in NASH activity score, and 19% showed regression of liver fibrosis[100]. These surgeries are a suitable option for NAFLD patients who are severely overweight (BMI ≥ 35 kg/m²) and have metabolic syndrome because they have long-term metabolic benefits like better control of blood sugar, lower risk of heart disease, and better insulin sensitivity[101]. Post-operative monitoring is essential because of risks such as weight regain, post-surgical problems, and nutrient malabsorption[102].
Researchers have thoroughly studied pharmacological treatments that target hepatic and systemic insulin sensitivity, given the strong link between insulin resistance and NAFLD[29]. Metformin is a common biguanide used to treat T2D. It protects the liver by turning on AMPK, which lowers DNL, raises fatty acid oxidation, and stops the liver from making glucose[103,104]. Despite improving lipid metabolism and hepatic insulin signaling, metformin's effectiveness in slowing the progression of hepatic steatosis and fibrosis has been limited in clinical trials, which makes it less desirable as a stand-alone treatment for NAFLD[105,106]. By turning on PPAR-γ, thiazolidinediones, especially pioglitazone, help adipocytes change into different types of fat cells and move fat from the liver to peripheral adipose tissue. Pioglitazone raises the production of adiponectin, which helps the liver burn fat and stops inflammatory pathways. However, its extensive use is restricted because of worries about weight gain, fluid retention, and long-term safety[107,108].
Liraglutide and semaglutide are examples of GLP-1RAs drugs that have shown promise as treatments. These drugs can inhibit inflammation in the liver, lower the amount of fat stored in the liver, and enhance insulin sensitivity. These substances help people lose weight and increase their metabolism by delaying stomach emptying, decreasing hunger, and improving β-cell function[109,110].
SGLT2i, including empagliflozin and dapagliflozin, are another promising anti-inflammatory tactic. They increase the excretion of glucose in the urine, improve insulin sensitivity, and decrease the accumulation of lipids in the liver. According to studies, SGLT2 inhibitors reduce hepatic inflammation and oxidative stress, which makes them a promising therapy option for NAFLD[111,112].
Since the gut-liver axis is crucial to the pathophysiology of NAFLD, treatments that target the microbiota present an intriguing therapeutic option. Researchers have investigated the potential of prebiotics, probiotics, postbiotics, and fecal microbiota transplantation (FMT) to increase the integrity of the intestinal barrier, lower hepatic inflammation, and restore gut microbial balance.
The availability of substrate for microbial fermentation can be changed as a means of modifying gut-liver interactions in NAFLD. Prebiotics function as a fundamental element in this approach. The intestinal microbiota ferments non-digestible food substances called prebiotics. These substances encourage the development of microbial species that control immune responses, host metabolism, and barrier function[113]. Prebiotics affect the course of NAFLD through mechanisms that are dependent on the microbiota. Microorganisms such as Faecalibacterium prausnitzii, Bifidobacterium adolescentis, and Bifidobacterium longum use prebiotics like inulin, fructooligosaccharides, and galactooligosaccharides. They ferment to create SCFAs, particularly butyrate, and propionate, which control fatty acid oxidation, inflammatory signaling, and hepatic gluconeogenesis[114,115]. By maintaining the integrity of tight junctions, prebiotics decrease intestinal permeability and endotoxin transfer[116]. It has been demonstrated that prebiotic treatment lowers circulating aminotransferases, enhances insulin sensitivity, and lowers hepatic lipid levels in NAFLD models. Prebiotic effectiveness is influenced by the metabolic state of the host, substrate availability, and microbial makeup. To improve metabolism, they are used in conjunction with dietary and lifestyle changes[117].
In addition to prebiotic nutrition, direct administration of certain microbial strains provides an additional way to affect immune signaling and host metabolism in NAFLD. Probiotics are living microorganisms that affect host-microbe interactions and gut microbiota composition when taken. Probiotics are utilised to modify bile acid metabolism, lower inflammation, and restore microbial balance in NAFLD[118]. Bifidobacterium lactis, Lactobacillus rhamnosus, Lactobacillus plantarum, and Akkermansia muciniphila are among the strains frequently studied in NAFLD. These species improve the function of the epithelial barrier, suppress immunological activation, and lower endotoxin levels[118,119]. Additionally, Akkermansia muciniphila lowers metabolic endotoxemia and helps maintain the mucin layer[120]. Probiotics modulate NF-κB and MAPK signalling, which in turn reduces inflammatory cytokines like TNF-α, IL-1β, and IL-6[121]. Probiotics increase insulin sensitivity indicators and lessen hepatic steatosis in NAFLD through these pathways. Because of their restricted capacity for colonisation, their effects are strain-specific and may necessitate continual consumption[122].
While probiotics and prebiotics work by stimulating microbial growth and activity, postbiotics offer bioactive compounds that work without affecting the viability of microorganisms. Postbiotics are secreted metabolites or non-viable microbial derivatives that affect the immune system and metabolism of the host. Across various host microbiota compositions, they provide similar biological signals and function independently of microbial colonisation[123]. Postbiotics such butyrate, acetate, and propionate are produced by bacteria such as Eubacterium rectale, Roseburia intestinalis, and Faecalibacterium prausnitzii in NAFLD[124]. Through the activation of G-protein coupled receptors (such as GPR41 and GPR43) and the inhibition of histone deacetylases, these SCFAs alter hepatic metabolism and affect the expression of genes linked to inflammation and lipid oxidation[125]. Additionally, postbiotics consist of exopolysaccharides, microbial peptides, and cell wall constituents that interact with nucleotide-binding oligomerization domain proteins and TLRs. These compounds alter systemic immune responses, enhance epithelial barrier function, and decrease inflammatory signaling[126]. It has been demonstrated that direct butyrate administration enhances hepatic insulin signaling and decreases fat deposition in experimental models of NAFLD[127]. Postbiotics are suitable for targeted liver therapies because they offer a therapeutic approach that gets over the restrictions of microbial viability and variability.
Apart from specific microbial metabolites, FMT is an additional strategy that seeks to restore microbial diversity and functional capacity in the host by directly transferring an entire microbial community. FMT is a new treatment that seeks to change metabolic pathways that are linked to the progression of NAFLD and bring back microbial balance[128]. According to preliminary research, FMT from lean donors lowers hepatic fat accumulation, increases insulin sensitivity, and modifies inflammatory cytokine profiles. Restoring intestinal microbial equilibrium is a promising microbiota-targeted therapy that seeks to restore intestinal microbial balance and reduce hepatic inflammation[129]. It has gotten a lot of attention because gut microbiota dysbiosis is a key part of how NAFLD works. By sending microbiota from a healthy donor to a healthy recipient through feces, FMT strengthens the gut barrier, changes the way bile acid is used, and rearranges the microbe community[130].
Although preliminary research indicates possible enhancements in inflammatory markers, hepatic steatosis, and insulin sensitivity, a number of restrictions prevent its practical application. Small sample numbers, brief follow-up times, and uneven study endpoints constrain the available evidence[131]. Regarding the best FMT procedures, such as dosage, frequency, and manner of administration, there is also disagreement[132]. In terms of mechanism, it is unclear how long-term microbiota changes persist after FMT, and it is also unclear how precisely FMT affects liver pathology[133,134]. The majority of reported adverse events in terms of safety are minor gastrointestinal symptoms, including diarrhea and bloating[135]. Nonetheless, there have been reports of uncommon but dangerous infections, such as the spread of germs resistant to multiple drugs[136].
Despite the fact FMT has great potential, there are still several obstacles to its long-term safety and standardized treatment for NAFLD. To demonstrate its efficacy as a standard therapeutic option, further controlled clinical trials are needed[137]. Making next-generation probiotics and postbiotics, such as SCFAs, is another way to deal with metabolic dysfunction caused by gut microbiota in NAFLD[138].
Numerous randomized controlled trials have examined how probiotics affect NAFLD. One study showed, for example, that a multistrain probiotic program significantly improved liver histology, including lowering fibrosis, lobular inflammation, and hepatocyte ballooning. Furthermore, when compared to a placebo, there were significant drops in inflammatory markers and liver enzymes[139]. These results have been corroborated by systematic reviews and meta-analyses, which show that probiotics can improve lipid profiles, inflammatory markers, and liver enzymes in individuals with NAFLD[140]. The effects of a probiotic mixture comprising Lactobacillus and Bifidobacterium strains on NAFLD patients over a 12-week period were assessed in a recent double-blind, placebo-controlled experiment. When compared to the placebo group, this trial showed notable reductions in liver enzymes, metabolic parameters, and hepatic steatosis[141,142]. On the other hand, the effectiveness of a postbiotic formulation was assessed in a randomized, double-blind, placebo-controlled clinical trial in 50 Caucasian patients with metabolic syndrome and NAFLD. Zn gluconate (5 mg), vitamin D3 (500 IU), and calcium butyrate (500 mg) were given to participants every day for 12 weeks. When compared to the baseline and placebo, the study found that the fatty liver index and hepatic steatosis index significantly improved. Furthermore, decreases in GGT, triglycerides, and total cholesterol were noted, suggesting that this postbiotic combination may be helpful in the treatment of NAFLD[143]. The effectiveness of postbiotics and probiotics in NAFLD patients has not yet been directly compared in the same study in clinical trials, which highlights a significant research gap and the necessity of carefully monitored head-to-head comparison studies.
Building on the increasing interest in gut microbiota-based treatments for NAFLD, a number of researchers have started to investigate whether these interventions result in noticeable improvements in the liver's histopathology. As demonstrated by liver biopsies or non-invasive imaging correlated with histological markers, randomized controlled trials employing multistrain probiotics have documented notable decreases in hepatic steatosis, lobular inflammation, hepatocyte ballooning, and even mild fibrosis[144,145]. When compared to a placebo, individuals in one trial who received a multistrain probiotic showed improvements in the NAFLD activity score, including lobular inflammation and ballooning[145]. According to elastography, new clinical trials on postbiotics, like pasteurized Akkermansia muciniphila, have shown decreases in liver stiffness and lipid content, which indirectly implies improvements in histopathology[146,147]. Although there is minimal histological evaluation and the trials are still in their early phases, some have revealed enhanced insulin sensitivity and lower liver fat, which may indicate structural improvements later on[148]. Overall, the evidence suggests that microbiota-targeted treatments have a promising role in correcting structural liver damage in NAFLD, even though histopathological data are still scarce in comparison to biochemical outcomes.
It is crucial to think about whether suggested therapeutic measures are successful at all stages of NAFLD or if stage-specific therapies are needed. The effectiveness of microbiota-targeted treatments may vary depending on the stage of NAFLD. Studies in the past have shown that gut microbiota-based therapies have emerged as promising interventions across various stages of NAFLD, from simple steatosis to NASH and fibrosis[149]. Probiotics and synbiotics have demonstrated efficacy in reducing hepatic inflammation, liver stiffness, and steatosis[150]. A meta-analysis of 21 randomized controlled trials involving 1252 participants found that these therapies significantly lowered ALT levels and improved liver stiffness measurements, indicating reduced inflammation and fibrosis. Additionally, they increased the odds of hepatic steatosis improvement as assessed by ultrasound[5,140,150].
Prebiotics, such as inulin-type fructans (ITF), have shown potential in modulating gut microbiota composition, leading to increased levels of beneficial bacteria like Bifidobacterium and Faecalibacterium prausnitzii. In animal models, ITF supplementation improved liver histology and reduced portal propionate levels, suggesting benefits in early-stage NAFLD[151]. From a mechanistic viewpoint, dietary and microbiota-modulating techniques such as probiotics and prebiotics may be useful in restoring gut microbial balance and lowering hepatic fat content in cases of simple steatosis (early-stage fat accumulation without significant inflammation) by enhancing insulin sensitivity and lipid metabolism[11]. To improve liver histology and modulate pro-inflammatory pathways, more focused interventions such as postbiotics (e.g., pasteurized Akkermansia muciniphila) or synbiotics may be required as the disease advances to NASH, which is characterized by inflammation and hepatocyte ballooning[152,153]. During fibrotic stages, the therapeutic window becomes more limited due to the increased deposition of extracellular matrix[154]. Similarly, in high-fat diet-induced NASH mouse models, FMT increased the abundance of beneficial bacteria such as Christensenellaceae and Lactobacillus, alleviating endotoxemia, liver steatosis, and intra-hepatic inflammation[155]. Clinical trials have shown that FMT can improve insulin sensitivity and gut microbiota diversity in patients with metabolic syndrome, although its long-term efficacy and safety in NAFLD patients require further investigation[122,156,157].
However, concerns related to gut-derived endotoxins and microbial translocation, FMT must be used carefully, or specialised therapies that target immune regulation and gut barrier repair are required[158,159]. Therefore, the safety and effectiveness of microbiota-based therapies are anticipated to vary depending on the stage of NAFLD; non-invasive microbial modulation is more effective in earlier stages, whereas advanced stages necessitate customised, potentially supplementary, therapeutic approaches[114]. Overall, while gut microbiota-targeted therapies show promise in managing different stages of NAFLD, further well-designed clinical trials are necessary to establish standardized treatment protocols and long-term outcomes.
Chelation therapy is one of the principal approaches for mitigating metal toxicity and thus, metal-induced NAFLD. Chelators help mobilizing metals from tissues, allowing them to be excreted through the kidneys in urine or through the liver in bile[160]. In the context of heavy metal-induced NAFLD, chelation therapy may offer therapeutic benefits by limiting the interaction of toxic metals with critical biomolecules, thereby mitigating hepatotoxic effects. To date, numerous chelating agents have been reported in the literature for their efficacy against a variety of heavy metals. British anti-Lewisite (BAL), a dithioglycerol compound, was originally developed by the British during World War I to counteract the effects of Lewisite (β-chlorovinyldichloroarsine), a chemical warfare agent[161]. BAL effectively binds to arsenic, thereby preventing its interaction with critical cellular proteins and offering protective effects. In recent years, the development of lipophilic and less toxic chelators has emerged as a promising direction in the treatment of metal poisoning. Among the most widely used chelating agents are Sodium 2,3-dimercaptopropane-1-sulfonate, dimercaptosuccinic acid (DMSA), and its structural analogue, monoisoamyl-DMSA[161,162]. Studies have also been reported regarding the use of calcium disodium ethylenediaminetetraacetic acid chelation therapy in patients with cadmium induced chronic renal failure and those with ischemic heart disease The free iron deposition is linked with histological severity in NAFLD patients due to the production of ROS (Fenton reaction)[163]. Recent studies on animal models and various cell lines revealed that iron chelators successfully removed hepatic excess iron indicating the potential benefits of iron chelation therapy in the clinical management of NAFLD[164,165]. Although, the regular use of chelators has its repercussions. The adverse effects associated with chelating agents may vary from gastrointestinal disturbances, Zn depletion, leukopenia, transient agranulocytosis, and reversible musculoskeletal to arthralgic symptoms[18]. Hence, additional research is warranted to identify novel heavy metal chelators exhibiting enhanced chelating efficacy coupled with minimal toxicity. To overcome these challenges, natural compounds present a promising alternative due to their diverse bioactive properties, lower toxicity profiles, and potential to modulate multiple pathological pathways involved in disease progression.
Se is one of the essential metals, but unlike most metals, Se is incorporated into proteins as a key component of certain amino acids[166]. The Recommended Dietary Allowance is 55 mg per day of Se for both adult men and women[167]. The discovery of 25 selenoproteins encoded in the human genome has been reported so far. These mainly include GPx, TrxR, SPS2, and DIO etc. These proteins are largely associated with oxidoreductase functions and enhance the antioxidative capacity of the cells[168]. As NAFLD is strongly associated with the presence of oxidative stress, GPxs and TrxRs play an important role in the efficient removal of various ROS. SEPS1, another selenoprotein, located in the ER, has been shown to play a part in regulating inflammation by influencing the production of inflammatory cytokines[169]. Se plays a pivotal part in the hormone activation, development, immune function, reproduction and cancer prevention etc.[17]. In the mice model, Se supplementation is also associated with the increased mRNA expression of PPARα in hepatocytes, which can modulate the triglyceride accumulation[170]. Additionally, alterations in Se levels appear to play a significant role in the development of insulin resistance[171]. Insulin is essential for maintaining glucose homeostasis by regulating hepatic glucose production and promoting glucose uptake in muscle and adipose tissues. The treatment of sodium selenite was reported to increase GLP-1R, IRS-1, and preproinsulin mRNA both at transcriptional and translational levels in the liver of diabetic rats[172]. GLP-1 play major role in glycogen synthesis, henceforth reducing hepatic glucose production[172]. Based on glucose clamp studies, Iizuka et al[173] demonstrated that sodium selenate treatment enhanced insulin-stimulated glucose disposal in Goto-Kakizaki rats. In another study, the administration of high-dose sodium selenite was found to exacerbate hepatic insulin resistance in a murine model of T2D, partially via activation of the oxidative stress-mediated JNK signaling pathway[174].
Due to its involvement in numerous metabolic processes, Se has frequently been linked to NAFLD. However, the impact of Se on NAFLD remains a subject of ongoing debate. Shojaei Zarghani et al[175] reported a weak positive association between dietary Se intake and the risk of NAFLD in the subjects from the Prospective Epidemiological Research Studies in Iran Kavar cohort study. In another bioinformatics-based studies, lower Se levels have been reported in NAFLD liver than in a disease-free liver[176]. The study also offered evidence suggesting a connection between Se and iron metabolism, which is known to be disturbed in NAFLD. Contrarily, the studies had shown non-linear associations of serum Se with ALT activity and NAFLD prevalence[177]. In another study, higher plasma Se levels were linked to a greater prevalence of NAFLD in a Chinese population[178]. The findings of epidemiological studies exploring the link between Se and NAFLD are significantly affected by the criteria used to assess the diagnosis, prevalence, and risk factors of NAFLD in large populations. Currently, there is no universally accepted method for this assessment. Hence, more rigorous research is needed to establish a standardized approach and unbiased outcomes.
To sum up, the response to Se intake exhibits a U-shaped dose-dependent curve, where intake above the recommended level leads to hyperglycemia and hyperinsulinemia[179]. This dysregulation disrupts oxidative stress balance, impairs insulin signaling pathways, and alters both lipid and glucose metabolism[179]. The application of Se in the management of NAFLD requires meticulous evaluation of its narrow therapeutic window, balancing efficacy against potential toxicity. Precise determination of the optimal Se dosage is essential and continues to be an area of active investigation.
Apart from Se and SCFAs, a wide variety of natural substances have been thoroughly investigated for their potential as therapeutics in the treatment of NAFLD. Polyphenols, including resveratrol, curcumin, and epigallocatechin gallate (EGCG), have demonstrated significant hepatoprotective properties[180]. Resveratrol, which is present in berries and grapes, has anti-inflammatory and antioxidant properties that enhance insulin sensitivity and decrease the buildup of hepatic fat by modifying the SIRT1 and AMPK pathways[181]. According to reports, curcumin, the main ingredient in turmeric, reduces liver inflammation and fibrosis by inhibiting NF-κB signaling and downregulating pro-inflammatory cytokines (TNF-α, IL-6)[182]. By controlling lipid metabolism-related genes like SREBP-1c and PPAR-α, EGCG, the main catechin in green tea, lowers oxidative stress and fat buildup[183].
Omega-3 fatty acids have also drawn a lot of interest because of their capacity to improve lipid profiles and reduce hepatic triglyceride levels, especially eicosapentaenoic acid and docosahexaenoic acid[184]. In order to reduce hepatic steatosis, they activate PPAR-α and decrease the production of lipogenic genes[185]. Additionally, prebiotics and dietary fibers like fructooligosaccharides and inulin contribute to the improvement of the composition of the gut microbiota and the creation of advantageous microbial metabolites like SCFAs, which have an indirect positive impact on liver function[186].
Moreover, flavonoids with antioxidative and anti-fibrotic qualities, such quercetin, naringenin, and silymarin (found in milk thistle), promote liver regeneration and lower oxidative stress indicators in NAFLD[187]. Additionally, alkaloids such as berberine have shown promise in reducing lipogenesis and decreasing hepatic insulin resistance through AMPK pathway modulation[188]. These substances are appealing candidates for integrative and adjuvant NAFLD therapy because they collectively function through a variety of pathways that include lipid metabolism management, inflammation attenuation, gut-liver axis modulation, and oxidative stress reduction.
NAFLD is a complex metabolic disease characterized by chronic inflammation, lipotoxicity, oxidative stress, insulin resistance, and dysbiosis. The disease gets worse from simple hepatic steatosis to more severe stages like NASH, fibrosis, and cirrhosis through complex interactions between genes, epigenetic changes, dietary habits, and problems with the gut-liver axis. A growing volume of evidence has thrown light on the potential impact of environmental toxicants especially heavy metals on liver health, in particular, on NAFLD incidence and progression. However, the identification of specific biomarkers for metal-induced hepatotoxicity remains an ongoing area of research. Comprehending these mechanisms has been essential in creating focused treatment strategies. The most successful therapies for metabolic restoration in NAFLD are still lifestyle changes, such as calorie restriction, changes in macronutrient composition, and physical activity. These changes are the main emphasis of current management methods for NAFLD. Although their effectiveness and long-term safety need more research. Only those who are extremely obese benefit from bariatric surgery and other surgical procedures, which significantly improve metabolism. The ability of therapies based on gut microbiota to change or control the types of microbes that live in the gut and boost the production of SCFAs, host-dependent variability. "Does modulation of the gut microbiota offer a potential strategy to mitigate metal-induced NAFLD?’’, is still the hypothesis under investigation. Se may play a part in the development of NAFLD by regulating the anti-oxidative and inflammatory response. However, the use of Se supplementation in treating metabolic disorders warrants further investigation due to its narrow effective dose range. Despite these developments, the lack of a single treatment that works for everyone highlights the necessity for individualized treatment plans that incorporate dietary, microbial, and genetic therapies. To maximize therapeutic results, future research should concentrate on precise diagnostic techniques that make use of metabolic signatures, host-specific responses, and microbiome analysis. The development of next-generation therapies for the prevention and treatment of NAFLD will depend heavily on a deeper understanding of gut-liver interaction and its effects on systemic metabolism. During recent years, the awareness of NAFLD has markedly increased, leading to discoveries of pathogenetic mechanisms and innovative therapeutic opportunities. On the other hand, current treatments do not advance much beyond the recommendation of sustained body weight loss. The lack of efficacy or adverse effects of treatments are probably contributing to the proportional global increase in metabolic diseases and NAFLD, making them a progressive pandemic, which poses an important health and economic burden and significantly affects health-related quality of life. A proposal is to screen individuals with the highest genetic or acquired risk of disease progression, for example, the SIRD subgroup, initiate early prevention and treatment, and develop therapeutic concepts, targeting the earliest pathophysiological alterations, namely, adipocyte dysfunction and insulin resistance.
The authors gratefully acknowledge the support of Department of Science & Technology and University Grants Commission for their assistance in developing instrumental and laboratory facilities in the department.
| 1. | Galiero R, Caturano A, Vetrano E, Cesaro A, Rinaldi L, Salvatore T, Marfella R, Sardu C, Moscarella E, Gragnano F, Calabrò P, Sasso FC. Pathophysiological mechanisms and clinical evidence of relationship between Nonalcoholic fatty liver disease (NAFLD) and cardiovascular disease. Rev Cardiovasc Med. 2021;22:755-768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 60] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 2. | Paik JM, Henry L, Younossi Y, Ong J, Alqahtani S, Younossi ZM. The burden of nonalcoholic fatty liver disease (NAFLD) is rapidly growing in every region of the world from 1990 to 2019. Hepatol Commun. 2023;7:e0251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 85] [Article Influence: 42.5] [Reference Citation Analysis (0)] |
| 3. | Li J, Ha A, Rui F, Zou B, Yang H, Xue Q, Hu X, Xu Y, Henry L, Barakat M, Stave CD, Shi J, Wu C, Cheung R, Nguyen MH. Meta-analysis: global prevalence, trend and forecasting of non-alcoholic fatty liver disease in children and adolescents, 2000-2021. Aliment Pharmacol Ther. 2022;56:396-406. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 64] [Article Influence: 21.3] [Reference Citation Analysis (0)] |
| 4. | Rinaldi L, Pafundi PC, Galiero R, Caturano A, Morone MV, Silvestri C, Giordano M, Salvatore T, Sasso FC. Mechanisms of Non-Alcoholic Fatty Liver Disease in the Metabolic Syndrome. A Narrative Review. Antioxidants (Basel). 2021;10:270. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 109] [Cited by in RCA: 123] [Article Influence: 30.8] [Reference Citation Analysis (0)] |
| 5. | Sharpton SR, Ajmera V, Loomba R. Emerging Role of the Gut Microbiome in Nonalcoholic Fatty Liver Disease: From Composition to Function. Clin Gastroenterol Hepatol. 2019;17:296-306. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 125] [Article Influence: 20.8] [Reference Citation Analysis (0)] |
| 6. | Bedogni G, Bellentani S, Miglioli L, Masutti F, Passalacqua M, Castiglione A, Tiribelli C. The Fatty Liver Index: a simple and accurate predictor of hepatic steatosis in the general population. BMC Gastroenterol. 2006;6:33. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1238] [Cited by in RCA: 2038] [Article Influence: 107.3] [Reference Citation Analysis (0)] |
| 7. | Paschos P, Paletas K. Non alcoholic fatty liver disease two-hit process: multifactorial character of the second hit. Hippokratia. 2009;13:128. |
| 8. | Maurice J, Manousou P. Non-alcoholic fatty liver disease. Clin Med (Lond). 2018;18:245-250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 212] [Article Influence: 30.3] [Reference Citation Analysis (0)] |
| 9. | Zarghamravanbakhsh P, Frenkel M, Poretsky L. Metabolic causes and consequences of nonalcoholic fatty liver disease (NAFLD). Metabol Open. 2021;12:100149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 77] [Article Influence: 19.3] [Reference Citation Analysis (0)] |
| 10. | Tinkov AA, Aschner M, Santamaria A, Bogdanov AR, Tizabi Y, Virgolini MB, Zhou JC, Skalny AV. Dissecting the role of cadmium, lead, arsenic, and mercury in non-alcoholic fatty liver disease and non-alcoholic steatohepatitis. Environ Res. 2023;238:117134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 11. | Song YP, Lv JW, Zhao Y, Chen X, Zhang ZC, Fan YJ, Zhang C, Gao L, Huang Y, Wang H, Xu DX. DNA hydroxymethylation reprogramming of β-oxidation genes mediates early-life arsenic-evoked hepatic lipid accumulation in adult mice. J Hazard Mater. 2022;430:128511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 18] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 12. | Garla R, Sharma N, Shamli, Kaushal N, Garg ML. Effect of Zinc on Hepatic and Renal Tissues of Chronically Arsenic Exposed Rats: A Biochemical and Histopathological Study. Biol Trace Elem Res. 2021;199:4237-4250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 13. | Zwolak I. The Role of Selenium in Arsenic and Cadmium Toxicity: an Updated Review of Scientific Literature. Biol Trace Elem Res. 2020;193:44-63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 145] [Cited by in RCA: 166] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 14. | Ye M, He Y, Xia Y, Zhong Z, Kong X, Zhou Y, Xia W, Wang W, Fan H, Chen L, Wu X, Li Q. Association Between Serum Zinc and Non-Alcoholic Fatty Liver Disease and Advanced Liver Fibrosis: NHANES 2011-2016. Biol Trace Elem Res. 2025;203:1305-1316. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 15. | Li L, Huang L, Huang S, Luo X, Zhang H, Mo Z, Wu T, Yang X. Non-linear association of serum molybdenum and linear association of serum zinc with nonalcoholic fatty liver disease: Multiple-exposure and Mendelian randomization approach. Sci Total Environ. 2020;720:137655. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 16. | Asprouli E, Kalafati IP, Sakellari A, Karavoltsos S, Vlachogiannakos J, Revenas K, Kokkinos A, Dassenakis M, Dedoussis GV, Kalogeropoulos N. Evaluation of Plasma Trace Elements in Different Stages of Nonalcoholic Fatty Liver Disease. Biol Trace Elem Res. 2019;188:326-333. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 36] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 17. | Labunskyy VM, Hatfield DL, Gladyshev VN. Selenoproteins: molecular pathways and physiological roles. Physiol Rev. 2014;94:739-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 712] [Cited by in RCA: 923] [Article Influence: 83.9] [Reference Citation Analysis (0)] |
| 18. | Xu L, Lu Y, Wang N, Feng Y. The Role and Mechanisms of Selenium Supplementation on Fatty Liver-Associated Disorder. Antioxidants (Basel). 2022;11:922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 25] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 19. | Shidfar F, Faghihi A, Amiri HL, Mousavi SN. Regression of Nonalcoholic Fatty Liver Disease with Zinc and Selenium Co-supplementation after Disease Progression in Rats. Iran J Med Sci. 2018;43:26-31. [PubMed] |
| 20. | Wong VW, Ekstedt M, Wong GL, Hagström H. Changing epidemiology, global trends and implications for outcomes of NAFLD. J Hepatol. 2023;79:842-852. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 308] [Article Influence: 154.0] [Reference Citation Analysis (0)] |
| 21. | Younossi ZM, Golabi P, Paik JM, Henry A, Van Dongen C, Henry L. The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology. 2023;77:1335-1347. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 270] [Cited by in RCA: 1443] [Article Influence: 721.5] [Reference Citation Analysis (2)] |
| 22. | Huang DQ, El-Serag HB, Loomba R. Global epidemiology of NAFLD-related HCC: trends, predictions, risk factors and prevention. Nat Rev Gastroenterol Hepatol. 2021;18:223-238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 840] [Cited by in RCA: 1212] [Article Influence: 303.0] [Reference Citation Analysis (0)] |
| 23. | Golabi P, Paik JM, AlQahtani S, Younossi Y, Tuncer G, Younossi ZM. Burden of non-alcoholic fatty liver disease in Asia, the Middle East and North Africa: Data from Global Burden of Disease 2009-2019. J Hepatol. 2021;75:795-809. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 153] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 24. | Schwimmer JB, Behling C, Newbury R, Deutsch R, Nievergelt C, Schork NJ, Lavine JE. Histopathology of pediatric nonalcoholic fatty liver disease. Hepatology. 2005;42:641-649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 537] [Cited by in RCA: 532] [Article Influence: 26.6] [Reference Citation Analysis (1)] |
| 25. | Fernandes DM, Pantangi V, Azam M, Salomao M, Iuga AC, Lefkowitch JH, Gill J, Morotti R, Lavine JE, Mencin AA. Pediatric Nonalcoholic Fatty Liver Disease in New York City: An Autopsy Study. J Pediatr. 2018;200:174-180. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 36] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 26. | De A, Duseja A. Nonalcoholic Fatty Liver Disease: Indian Perspective. Clin Liver Dis (Hoboken). 2021;18:158-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (1)] |
| 27. | Mukherjee PS, Vishnubhatla S, Amarapurkar DN, Das K, Sood A, Chawla YK, Eapen CE, Boddu P, Thomas V, Varshney S, Hidangmayum DS, Bhaumik P, Thakur B, Acharya SK, Chowdhury A. Etiology and mode of presentation of chronic liver diseases in India: A multi centric study. PLoS One. 2017;12:e0187033. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 47] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 28. | Kuchay MS, Choudhary NS, Mishra SK, Bano T, Gagneja S, Mathew A, Singh MK, Kaur P, Gill HK, Wasir JS, Sud R, Mithal A. Prevalence of clinically relevant liver fibrosis due to nonalcoholic fatty liver disease in Indian individuals with type 2 diabetes. JGH Open. 2021;5:915-922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 29. | Palma R, Pronio A, Romeo M, Scognamiglio F, Ventriglia L, Ormando VM, Lamazza A, Pontone S, Federico A, Dallio M. The Role of Insulin Resistance in Fueling NAFLD Pathogenesis: From Molecular Mechanisms to Clinical Implications. J Clin Med. 2022;11:3649. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 45] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 30. | Richard AJ, White U, Elks CM, Stephens JM. Adipose Tissue: Physiology to Metabolic Dysfunction. 2020 Apr 4. In: Feingold KR, Ahmed SF, Anawalt B, Blackman MR, Boyce A, Chrousos G, Corpas E, de Herder WW, Dhatariya K, Dungan K, Hofland J, Kalra S, Kaltsas G, Kapoor N, Koch C, Kopp P, Korbonits M, Kovacs CS, Kuohung W, Laferrère B, Levy M, McGee EA, McLachlan R, Muzumdar R, Purnell J, Rey R, Sahay R, Shah AS, Singer F, Sperling MA, Stratakis CA, Trence DL, Wilson DP, editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000. [PubMed] |
| 31. | Mu C, Wang S, Wang Z, Tan J, Yin H, Wang Y, Dai Z, Ding D, Yang F. Mechanisms and therapeutic targets of mitochondria in the progression of metabolic dysfunction-associated steatotic liver disease. Ann Hepatol. 2025;30:101774. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 32. | Ragheb R, Shanab GM, Medhat AM, Seoudi DM, Adeli K, Fantus IG. Free fatty acid-induced muscle insulin resistance and glucose uptake dysfunction: evidence for PKC activation and oxidative stress-activated signaling pathways. Biochem Biophys Res Commun. 2009;389:211-216. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 84] [Cited by in RCA: 83] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 33. | Solinas G, Borén J, Dulloo AG. De novo lipogenesis in metabolic homeostasis: More friend than foe? Mol Metab. 2015;4:367-377. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 113] [Cited by in RCA: 138] [Article Influence: 13.8] [Reference Citation Analysis (0)] |
| 34. | Thiel G, Guethlein LA, Rössler OG. Insulin-Responsive Transcription Factors. Biomolecules. 2021;11:1886. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 35. | Huang JK, Lee HC. Emerging Evidence of Pathological Roles of Very-Low-Density Lipoprotein (VLDL). Int J Mol Sci. 2022;23:4300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 36. | Ma Y, Lee G, Heo SY, Roh YS. Oxidative Stress Is a Key Modulator in the Development of Nonalcoholic Fatty Liver Disease. Antioxidants (Basel). 2021;11:91. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 119] [Article Influence: 29.8] [Reference Citation Analysis (0)] |
| 37. | Ribas GS, Vargas CR. Evidence that Oxidative Disbalance and Mitochondrial Dysfunction are Involved in the Pathophysiology of Fatty Acid Oxidation Disorders. Cell Mol Neurobiol. 2022;42:521-532. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 38. | Engin A. Nonalcoholic Fatty Liver Disease and Staging of Hepatic Fibrosis. Adv Exp Med Biol. 2024;1460:539-574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 39. | Zong Y, Li H, Liao P, Chen L, Pan Y, Zheng Y, Zhang C, Liu D, Zheng M, Gao J. Mitochondrial dysfunction: mechanisms and advances in therapy. Signal Transduct Target Ther. 2024;9:124. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 348] [Cited by in RCA: 279] [Article Influence: 279.0] [Reference Citation Analysis (0)] |
| 40. | Kowalczyk P, Sulejczak D, Kleczkowska P, Bukowska-Ośko I, Kucia M, Popiel M, Wietrak E, Kramkowski K, Wrzosek K, Kaczyńska K. Mitochondrial Oxidative Stress-A Causative Factor and Therapeutic Target in Many Diseases. Int J Mol Sci. 2021;22:13384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 231] [Article Influence: 57.8] [Reference Citation Analysis (0)] |
| 41. | Xu J, Taubert S. Beyond Proteostasis: Lipid Metabolism as a New Player in ER Homeostasis. Metabolites. 2021;11:52. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 42. | Ajoolabady A, Kaplowitz N, Lebeaupin C, Kroemer G, Kaufman RJ, Malhi H, Ren J. Endoplasmic reticulum stress in liver diseases. Hepatology. 2023;77:619-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 188] [Article Influence: 94.0] [Reference Citation Analysis (0)] |
| 43. | Song MJ, Malhi H. The unfolded protein response and hepatic lipid metabolism in non alcoholic fatty liver disease. Pharmacol Ther. 2019;203:107401. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 105] [Cited by in RCA: 100] [Article Influence: 16.7] [Reference Citation Analysis (0)] |
| 44. | Park HJ, Choi J, Kim H, Yang DY, An TH, Lee EW, Han BS, Lee SC, Kim WK, Bae KH, Oh KJ. Cellular heterogeneity and plasticity during NAFLD progression. Front Mol Biosci. 2023;10:1221669. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 45. | Thibaut R, Gage MC, Pineda-Torra I, Chabrier G, Venteclef N, Alzaid F. Liver macrophages and inflammation in physiology and physiopathology of non-alcoholic fatty liver disease. FEBS J. 2022;289:3024-3057. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 56] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 46. | Wang M, Li L, Xu Y, Du J, Ling C. Roles of hepatic stellate cells in NAFLD: From the perspective of inflammation and fibrosis. Front Pharmacol. 2022;13:958428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 47. | Park SJ, Garcia Diaz J, Um E, Hahn YS. Major roles of kupffer cells and macrophages in NAFLD development. Front Endocrinol (Lausanne). 2023;14:1150118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 38] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 48. | Gordon S, Martinez FO. Alternative activation of macrophages: mechanism and functions. Immunity. 2010;32:593-604. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3290] [Cited by in RCA: 3122] [Article Influence: 208.1] [Reference Citation Analysis (0)] |
| 49. | Ziolkowska S, Binienda A, Jabłkowski M, Szemraj J, Czarny P. The Interplay between Insulin Resistance, Inflammation, Oxidative Stress, Base Excision Repair and Metabolic Syndrome in Nonalcoholic Fatty Liver Disease. Int J Mol Sci. 2021;22:11128. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 102] [Article Influence: 25.5] [Reference Citation Analysis (0)] |
| 50. | Rogero MM, Calder PC. Obesity, Inflammation, Toll-Like Receptor 4 and Fatty Acids. Nutrients. 2018;10:432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 324] [Cited by in RCA: 475] [Article Influence: 67.9] [Reference Citation Analysis (0)] |
| 51. | Safari Z, Gérard P. The links between the gut microbiome and non-alcoholic fatty liver disease (NAFLD). Cell Mol Life Sci. 2019;76:1541-1558. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 317] [Cited by in RCA: 340] [Article Influence: 56.7] [Reference Citation Analysis (0)] |
| 52. | Vachher M, Kaur K, Marothia M, Burman A, Deepanjana, Bansal S. Deciphering the role of dietary modifications and gut dysbiosis in Non-Alcoholic Fatty Liver Disease. Nutr Metab. 2025;40:200305. [DOI] [Full Text] |
| 53. | Kuo WT, Odenwald MA, Turner JR, Zuo L. Tight junction proteins occludin and ZO-1 as regulators of epithelial proliferation and survival. Ann N Y Acad Sci. 2022;1514:21-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 236] [Article Influence: 78.7] [Reference Citation Analysis (0)] |
| 54. | Kamm DR, McCommis KS. Hepatic stellate cells in physiology and pathology. J Physiol. 2022;600:1825-1837. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 152] [Article Influence: 50.7] [Reference Citation Analysis (0)] |
| 55. | Romeo S, Kozlitina J, Xing C, Pertsemlidis A, Cox D, Pennacchio LA, Boerwinkle E, Cohen JC, Hobbs HH. Genetic variation in PNPLA3 confers susceptibility to nonalcoholic fatty liver disease. Nat Genet. 2008;40:1461-1465. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2701] [Cited by in RCA: 2599] [Article Influence: 152.9] [Reference Citation Analysis (0)] |
| 56. | Kozlitina J, Smagris E, Stender S, Nordestgaard BG, Zhou HH, Tybjærg-Hansen A, Vogt TF, Hobbs HH, Cohen JC. Exome-wide association study identifies a TM6SF2 variant that confers susceptibility to nonalcoholic fatty liver disease. Nat Genet. 2014;46:352-356. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 724] [Cited by in RCA: 931] [Article Influence: 84.6] [Reference Citation Analysis (0)] |
| 57. | Mancina RM, Dongiovanni P, Petta S, Pingitore P, Meroni M, Rametta R, Borén J, Montalcini T, Pujia A, Wiklund O, Hindy G, Spagnuolo R, Motta BM, Pipitone RM, Craxì A, Fargion S, Nobili V, Käkelä P, Kärjä V, Männistö V, Pihlajamäki J, Reilly DF, Castro-Perez J, Kozlitina J, Valenti L, Romeo S. The MBOAT7-TMC4 Variant rs641738 Increases Risk of Nonalcoholic Fatty Liver Disease in Individuals of European Descent. Gastroenterology. 2016;150:1219-1230.e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 385] [Cited by in RCA: 515] [Article Influence: 57.2] [Reference Citation Analysis (0)] |
| 58. | Speliotes EK, Yerges-Armstrong LM, Wu J, Hernaez R, Kim LJ, Palmer CD, Gudnason V, Eiriksdottir G, Garcia ME, Launer LJ, Nalls MA, Clark JM, Mitchell BD, Shuldiner AR, Butler JL, Tomas M, Hoffmann U, Hwang SJ, Massaro JM, O'Donnell CJ, Sahani DV, Salomaa V, Schadt EE, Schwartz SM, Siscovick DS; NASH CRN; GIANT Consortium; MAGIC Investigators, Voight BF, Carr JJ, Feitosa MF, Harris TB, Fox CS, Smith AV, Kao WH, Hirschhorn JN, Borecki IB; GOLD Consortium. Genome-wide association analysis identifies variants associated with nonalcoholic fatty liver disease that have distinct effects on metabolic traits. PLoS Genet. 2011;7:e1001324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 775] [Cited by in RCA: 762] [Article Influence: 54.4] [Reference Citation Analysis (0)] |
| 59. | Abul-Husn NS, Cheng X, Li AH, Xin Y, Schurmann C, Stevis P, Liu Y, Kozlitina J, Stender S, Wood GC, Stepanchick AN, Still MD, McCarthy S, O'Dushlaine C, Packer JS, Balasubramanian S, Gosalia N, Esopi D, Kim SY, Mukherjee S, Lopez AE, Fuller ED, Penn J, Chu X, Luo JZ, Mirshahi UL, Carey DJ, Still CD, Feldman MD, Small A, Damrauer SM, Rader DJ, Zambrowicz B, Olson W, Murphy AJ, Borecki IB, Shuldiner AR, Reid JG, Overton JD, Yancopoulos GD, Hobbs HH, Cohen JC, Gottesman O, Teslovich TM, Baras A, Mirshahi T, Gromada J, Dewey FE. A Protein-Truncating HSD17B13 Variant and Protection from Chronic Liver Disease. N Engl J Med. 2018;378:1096-1106. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 445] [Cited by in RCA: 611] [Article Influence: 87.3] [Reference Citation Analysis (0)] |
| 60. | Choudhary NS, Duseja A. Genetic and epigenetic disease modifiers: non-alcoholic fatty liver disease (NAFLD) and alcoholic liver disease (ALD). Transl Gastroenterol Hepatol. 2021;6:2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 62] [Article Influence: 15.5] [Reference Citation Analysis (0)] |
| 61. | Rodríguez-Sanabria JS, Escutia-Gutiérrez R, Rosas-Campos R, Armendáriz-Borunda JS, Sandoval-Rodríguez A. An Update in Epigenetics in Metabolic-Associated Fatty Liver Disease. Front Med (Lausanne). 2021;8:770504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 22] [Reference Citation Analysis (0)] |
| 62. | Xu L, Fan YH, Zhang XJ, Bai L. Unraveling the relationship between histone methylation and nonalcoholic fatty liver disease. World J Hepatol. 2024;16:703-715. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 63. | Shi Y, Qi W. Histone Modifications in NAFLD: Mechanisms and Potential Therapy. Int J Mol Sci. 2023;24:14653. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 64. | Sulaiman SA, Muhsin NIA, Jamal R. Regulatory Non-coding RNAs Network in Non-alcoholic Fatty Liver Disease. Front Physiol. 2019;10:279. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 65. | Mukherjee AG, Wanjari UR, Gopalakrishnan AV, Katturajan R, Kannampuzha S, Murali R, Namachivayam A, Ganesan R, Renu K, Dey A, Vellingiri B, Prince SE. Exploring the Regulatory Role of ncRNA in NAFLD: A Particular Focus on PPARs. Cells. 2022;11:3959. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 66. | Mokhtari Z, Gibson DL, Hekmatdoost A. Nonalcoholic Fatty Liver Disease, the Gut Microbiome, and Diet. Adv Nutr. 2017;8:240-252. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 129] [Cited by in RCA: 121] [Article Influence: 15.1] [Reference Citation Analysis (0)] |
| 67. | Portincasa P, Bonfrate L, Vacca M, De Angelis M, Farella I, Lanza E, Khalil M, Wang DQ, Sperandio M, Di Ciaula A. Gut Microbiota and Short Chain Fatty Acids: Implications in Glucose Homeostasis. Int J Mol Sci. 2022;23:1105. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 515] [Cited by in RCA: 469] [Article Influence: 156.3] [Reference Citation Analysis (0)] |
| 68. | Li X, He M, Yi X, Lu X, Zhu M, Xue M, Tang Y, Zhu Y. A Systematic Review of Short-Chain Fatty Acids in Nonalcoholic Fatty Liver Disease: New Prospects for Short-Chain Fatty Acid Salts as Therapeutic Targets. 2023 Preprint. [DOI] [Full Text] |
| 69. | Guo W, Pan B, Sakkiah S, Yavas G, Ge W, Zou W, Tong W, Hong H. Persistent Organic Pollutants in Food: Contamination Sources, Health Effects and Detection Methods. Int J Environ Res Public Health. 2019;16:4361. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 198] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 70. | Reina-Pérez I, Artacho-Cordón F, Mustieles V, Castellano-Castillo D, Cardona F, Jiménez-Díaz I, López-Medina JA, Alcaide J, Ocaña-Wilhelmi L, Iribarne-Durán LM, Arrebola JP, Olea N, Tinahones FJ, Fernández MF. Cross-sectional associations of persistent organic pollutants measured in adipose tissue and metabolic syndrome in clinically diagnosed middle-aged adults. Environ Res. 2023;222:115350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 13] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 71. | Kumar J, Lind L, Salihovic S, van Bavel B, Ingelsson E, Lind PM. Persistent organic pollutants and liver dysfunction biomarkers in a population-based human sample of men and women. Environ Res. 2014;134:251-256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 42] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 72. | Chen J, Wu L, Yang G, Zhang C, Liu X, Sun X, Chen X, Wang N. The influence of PM(2.5) exposure on non-alcoholic fatty liver disease. Life Sci. 2021;270:119135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 41] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 73. | Xu MX, Ge CX, Qin YT, Gu TT, Lou DS, Li Q, Hu LF, Feng J, Huang P, Tan J. Prolonged PM2.5 exposure elevates risk of oxidative stress-driven nonalcoholic fatty liver disease by triggering increase of dyslipidemia. Free Radic Biol Med. 2019;130:542-556. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 107] [Cited by in RCA: 169] [Article Influence: 28.2] [Reference Citation Analysis (0)] |
| 74. | Renu K, Chakraborty R, Myakala H, Koti R, Famurewa AC, Madhyastha H, Vellingiri B, George A, Valsala Gopalakrishnan A. Molecular mechanism of heavy metals (Lead, Chromium, Arsenic, Mercury, Nickel and Cadmium) - induced hepatotoxicity - A review. Chemosphere. 2021;271:129735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 141] [Cited by in RCA: 292] [Article Influence: 73.0] [Reference Citation Analysis (0)] |
| 75. | Zhou Y, Addai FP, Zhang X, Liu Y, Wang Y, Lin F, Tuffour A, Gu J, Liu G, Shi H. Heavy metal-induced lipogenic gene aberration, lipid dysregulation and obesogenic effect: a review. Environ Chem Lett. 2022;20:1611-1643. [DOI] [Full Text] |
| 76. | Chen H, Li S, Liu J, Diwan BA, Barrett JC, Waalkes MP. Chronic inorganic arsenic exposure induces hepatic global and individual gene hypomethylation: implications for arsenic hepatocarcinogenesis. Carcinogenesis. 2004;25:1779-1786. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 200] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 77. | Khan F, Momtaz S, Abdollahi M. The relationship between mercury exposure and epigenetic alterations regarding human health, risk assessment and diagnostic strategies. J Trace Elem Med Biol. 2019;52:37-47. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 57] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 78. | Stýblo M, Drobná Z, Jaspers I, Lin S, Thomas DJ. The role of biomethylation in toxicity and carcinogenicity of arsenic: a research update. Environ Health Perspect. 2002;110 Suppl 5:767-771. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 180] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 79. | Tinkov AA, Aschner M, Ke T, Ferrer B, Zhou JC, Chang JS, Santamaría A, Chao JC, Aaseth J, Skalny AV. Adipotropic effects of heavy metals and their potential role in obesity. Fac Rev. 2021;10:32. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 38] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 80. | Duan H, Yu L, Tian F, Zhai Q, Fan L, Chen W. Gut microbiota: A target for heavy metal toxicity and a probiotic protective strategy. Sci Total Environ. 2020;742:140429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 134] [Article Influence: 26.8] [Reference Citation Analysis (0)] |
| 81. | Kim JJ, Kim YS, Kumar V. Heavy metal toxicity: An update of chelating therapeutic strategies. J Trace Elem Med Biol. 2019;54:226-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 225] [Cited by in RCA: 281] [Article Influence: 46.8] [Reference Citation Analysis (0)] |
| 82. | Aaseth J, Ajsuvakova OP, Skalny AV, Skalnaya MG, Tinkov AA. Chelator combination as therapeutic strategy in mercury and lead poisonings. Coord Chem Rev. 2018;358:1-12. [RCA] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 83. | Mishra DK, Awasthi H, Srivastava D, Fatima Z. Phytochemical: a treatment option for heavy metal induced neurotoxicity. J Complement Integr Med. 2022;19:513-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 84. | Rahman MM, Hossain KFB, Banik S, Sikder MT, Akter M, Bondad SEC, Rahaman MS, Hosokawa T, Saito T, Kurasaki M. Selenium and zinc protections against metal-(loids)-induced toxicity and disease manifestations: A review. Ecotoxicol Environ Saf. 2019;168:146-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 103] [Article Influence: 17.2] [Reference Citation Analysis (0)] |
| 85. | Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L, Torres-Gonzalez A, Gra-Oramas B, Gonzalez-Fabian L, Friedman SL, Diago M, Romero-Gomez M. Weight Loss Through Lifestyle Modification Significantly Reduces Features of Nonalcoholic Steatohepatitis. Gastroenterology. 2015;149:367-78.e5; quiz e14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1181] [Cited by in RCA: 1627] [Article Influence: 162.7] [Reference Citation Analysis (1)] |
| 86. | Romero-Gómez M, Zelber-Sagi S, Trenell M. Treatment of NAFLD with diet, physical activity and exercise. J Hepatol. 2017;67:829-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 844] [Cited by in RCA: 926] [Article Influence: 115.8] [Reference Citation Analysis (0)] |
| 87. | Schwingshackl L, Morze J, Hoffmann G. Mediterranean diet and health status: Active ingredients and pharmacological mechanisms. Br J Pharmacol. 2020;177:1241-1257. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 189] [Article Influence: 31.5] [Reference Citation Analysis (0)] |
| 88. | Sinha S, Haque M, Lugova H, Kumar S. The Effect of Omega-3 Fatty Acids on Insulin Resistance. Life (Basel). 2023;13:1322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 89. | Luukkonen PK, Dufour S, Lyu K, Zhang XM, Hakkarainen A, Lehtimäki TE, Cline GW, Petersen KF, Shulman GI, Yki-Järvinen H. Effect of a ketogenic diet on hepatic steatosis and hepatic mitochondrial metabolism in nonalcoholic fatty liver disease. Proc Natl Acad Sci U S A. 2020;117:7347-7354. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 139] [Cited by in RCA: 163] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 90. | Li L, Zhao X, Abdugheni R, Yu F, Zhao Y, Ma BF, Yang Z, Li R, Li Y, Maimaitiyiming Y, Maimaiti M. Gut microbiota changes associated with low-carbohydrate diet intervention for obesity. Open Life Sci. 2024;19:20220803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 91. | Softic S, Stanhope KL, Boucher J, Divanovic S, Lanaspa MA, Johnson RJ, Kahn CR. Fructose and hepatic insulin resistance. Crit Rev Clin Lab Sci. 2020;57:308-322. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 152] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 92. | Fromenty B, Roden M. Mitochondrial alterations in fatty liver diseases. J Hepatol. 2023;78:415-429. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 187] [Article Influence: 93.5] [Reference Citation Analysis (0)] |
| 93. | Sabag A, Barr L, Armour M, Armstrong A, Baker CJ, Twigg SM, Chang D, Hackett DA, Keating SE, George J, Johnson NA. The Effect of High-intensity Interval Training vs Moderate-intensity Continuous Training on Liver Fat: A Systematic Review and Meta-Analysis. J Clin Endocrinol Metab. 2022;107:862-881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 94. | McGee SL, Hargreaves M. Exercise performance and health: Role of GLUT4. Free Radic Biol Med. 2024;224:479-483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 95. | Cheng CK, Ye L, Wang Y, Wang YL, Xia Y, Wong SH, Chen S, Huang Y. Exercised gut microbiota improves vascular and metabolic abnormalities in sedentary diabetic mice through gut‒vascular connection. J Sport Health Sci. 2025;14:101026. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 96. | Sasaki A, Nitta H, Otsuka K, Umemura A, Baba S, Obuchi T, Wakabayashi G. Bariatric surgery and non-alcoholic Fatty liver disease: current and potential future treatments. Front Endocrinol (Lausanne). 2014;5:164. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 90] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 97. | Lim JK. The Role of Bariatric Surgery in the Management of Nonalcoholic Steatohepatitis. Gastroenterol Hepatol (N Y). 2016;12:397-399. [PubMed] |
| 98. | Softic S, Cohen DE, Kahn CR. Role of Dietary Fructose and Hepatic De Novo Lipogenesis in Fatty Liver Disease. Dig Dis Sci. 2016;61:1282-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 444] [Cited by in RCA: 485] [Article Influence: 53.9] [Reference Citation Analysis (0)] |
| 99. | Lim JK. The role of bariatric surgery in the management of nonalcoholic steatohepatitis. Gastroenterol Hepatol. 2016;12:397. [PubMed] |
| 100. | Ziogas IA, Zapsalis K, Giannis D, Tsoulfas G. Metabolic syndrome and liver disease in the era of bariatric surgery: What you need to know! World J Hepatol. 2020;12:709-721. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 101. | Kim TY, Kim S, Schafer AL. Medical Management of the Postoperative Bariatric Surgery Patient. 2020 Aug 24. In: Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc.; 2000. [PubMed] |
| 102. | Rena G, Hardie DG, Pearson ER. The mechanisms of action of metformin. Diabetologia. 2017;60:1577-1585. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1320] [Cited by in RCA: 1452] [Article Influence: 181.5] [Reference Citation Analysis (0)] |
| 103. | Goedeke L, Perry RJ, Shulman GI. Emerging Pharmacological Targets for the Treatment of Nonalcoholic Fatty Liver Disease, Insulin Resistance, and Type 2 Diabetes. Annu Rev Pharmacol Toxicol. 2019;59:65-87. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 61] [Article Influence: 12.2] [Reference Citation Analysis (0)] |
| 104. | Lavine JE, Schwimmer JB, Van Natta ML, Molleston JP, Murray KF, Rosenthal P, Abrams SH, Scheimann AO, Sanyal AJ, Chalasani N, Tonascia J, Ünalp A, Clark JM, Brunt EM, Kleiner DE, Hoofnagle JH, Robuck PR; Nonalcoholic Steatohepatitis Clinical Research Network. Effect of vitamin E or metformin for treatment of nonalcoholic fatty liver disease in children and adolescents: the TONIC randomized controlled trial. JAMA. 2011;305:1659-1668. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 864] [Cited by in RCA: 849] [Article Influence: 60.6] [Reference Citation Analysis (0)] |
| 105. | Jianfang F, Wanxia X, Xiling G, Jing X, Wenjuan Y, Jianrong L, Qingzhen H, Kaiyan M, Jingxuan L, Taixiong C, Qian X, Mengying L, Jie M, Qiuhe J. Effect and Safety of Pioglitazone-Metformin Tablets in the Treatment of Newly Diagnosed Type 2 Diabetes Patients with Nonalcoholic Fatty Liver Disease in Shaanxi Province: A Randomized, Double-Blinded, Double-Simulated Multicenter Study. J Diabetes Res. 2023;2023:2044090. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 106. | Basak S, Murmu A, Matore BW, Roy PP, Singh J. Thiazolidinedione an auspicious scaffold as PPAR-γ agonist: its possible mechanism to Manoeuvre against insulin resistant diabetes mellitus. Eur J Med Chem Rep. 2024;11:100160. [DOI] [Full Text] |
| 107. | Soccio RE, Chen ER, Lazar MA. Thiazolidinediones and the promise of insulin sensitization in type 2 diabetes. Cell Metab. 2014;20:573-591. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 335] [Cited by in RCA: 392] [Article Influence: 35.6] [Reference Citation Analysis (0)] |
| 108. | Zheng Z, Zong Y, Ma Y, Tian Y, Pang Y, Zhang C, Gao J. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal Transduct Target Ther. 2024;9:234. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 212] [Cited by in RCA: 169] [Article Influence: 169.0] [Reference Citation Analysis (0)] |
| 109. | Madsbad S, Holst JJ. The promise of glucagon-like peptide 1 receptor agonists (GLP-1RA) for the treatment of obesity: a look at phase 2 and 3 pipelines. Expert Opin Investig Drugs. 2025;34:197-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 110. | Androutsakos T, Nasiri-Ansari N, Bakasis AD, Kyrou I, Efstathopoulos E, Randeva HS, Kassi E. SGLT-2 Inhibitors in NAFLD: Expanding Their Role beyond Diabetes and Cardioprotection. Int J Mol Sci. 2022;23:3107. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 94] [Article Influence: 31.3] [Reference Citation Analysis (0)] |
| 111. | Jasleen B, Vishal GK, Sameera M, Fahad M, Brendan O, Deion S, Pemminati S. Sodium-Glucose Cotransporter 2 (SGLT2) Inhibitors: Benefits Versus Risk. Cureus. 2023;15:e33939. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 12] [Reference Citation Analysis (0)] |
| 112. | Davani-Davari D, Negahdaripour M, Karimzadeh I, Seifan M, Mohkam M, Masoumi SJ, Berenjian A, Ghasemi Y. Prebiotics: Definition, Types, Sources, Mechanisms, and Clinical Applications. Foods. 2019;8:92. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 604] [Cited by in RCA: 784] [Article Influence: 130.7] [Reference Citation Analysis (0)] |
| 113. | Gupta H, Min BH, Ganesan R, Gebru YA, Sharma SP, Park E, Won SM, Jeong JJ, Lee SB, Cha MG, Kwon GH, Jeong MK, Hyun JY, Eom JA, Park HJ, Yoon SJ, Choi MR, Kim DJ, Suk KT. Gut Microbiome in Non-Alcoholic Fatty Liver Disease: From Mechanisms to Therapeutic Role. Biomedicines. 2022;10:550. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 114. | Porras D, Nistal E, Martínez-Flórez S, González-Gallego J, García-Mediavilla MV, Sánchez-Campos S. Intestinal Microbiota Modulation in Obesity-Related Non-alcoholic Fatty Liver Disease. Front Physiol. 2018;9:1813. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 73] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 115. | Di Vincenzo F, Del Gaudio A, Petito V, Lopetuso LR, Scaldaferri F. Gut microbiota, intestinal permeability, and systemic inflammation: a narrative review. Intern Emerg Med. 2024;19:275-293. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 348] [Article Influence: 348.0] [Reference Citation Analysis (0)] |
| 116. | Mijangos-Trejo A, Nuño-Lambarri N, Barbero-Becerra V, Uribe-Esquivel M, Vidal-Cevallos P, Chávez-Tapia N. Prebiotics and Probiotics: Therapeutic Tools for Nonalcoholic Fatty Liver Disease. Int J Mol Sci. 2023;24:14918. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 23] [Reference Citation Analysis (0)] |
| 117. | Lu J, Shataer D, Yan H, Dong X, Zhang M, Qin Y, Cui J, Wang L. Probiotics and Non-Alcoholic Fatty Liver Disease: Unveiling the Mechanisms of Lactobacillus plantarum and Bifidobacterium bifidum in Modulating Lipid Metabolism, Inflammation, and Intestinal Barrier Integrity. Foods. 2024;13:2992. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 118. | Plaza-Diaz J, Ruiz-Ojeda FJ, Gil-Campos M, Gil A. Mechanisms of Action of Probiotics. Adv Nutr. 2019;10:S49-S66. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 901] [Cited by in RCA: 690] [Article Influence: 115.0] [Reference Citation Analysis (0)] |
| 119. | Zhao Y, Yang H, Wu P, Yang S, Xue W, Xu B, Zhang S, Tang B, Xu D. Akkermansia muciniphila: A promising probiotic against inflammation and metabolic disorders. Virulence. 2024;15:2375555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 120. | Vincenzi A, Goettert MI, Volken de Souza CF. An evaluation of the effects of probiotics on tumoral necrosis factor (TNF-α) signaling and gene expression. Cytokine Growth Factor Rev. 2021;57:27-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 121. | Chen J, Vitetta L. Gut Microbiota Metabolites in NAFLD Pathogenesis and Therapeutic Implications. Int J Mol Sci. 2020;21:5214. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 48] [Cited by in RCA: 208] [Article Influence: 41.6] [Reference Citation Analysis (0)] |
| 122. | Thorakkattu P, Khanashyam AC, Shah K, Babu KS, Mundanat AS, Deliephan A, Deokar GS, Santivarangkna C, Nirmal NP. Postbiotics: Current Trends in Food and Pharmaceutical Industry. Foods. 2022;11:3094. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 98] [Cited by in RCA: 128] [Article Influence: 42.7] [Reference Citation Analysis (0)] |
| 123. | Fusco W, Lorenzo MB, Cintoni M, Porcari S, Rinninella E, Kaitsas F, Lener E, Mele MC, Gasbarrini A, Collado MC, Cammarota G, Ianiro G. Short-Chain Fatty-Acid-Producing Bacteria: Key Components of the Human Gut Microbiota. Nutrients. 2023;15:2211. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 408] [Reference Citation Analysis (0)] |
| 124. | Lee DH, Kim MT, Han JH. GPR41 and GPR43: From development to metabolic regulation. Biomed Pharmacother. 2024;175:116735. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 21] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 125. | Jastrząb R, Graczyk D, Siedlecki P. Molecular and Cellular Mechanisms Influenced by Postbiotics. Int J Mol Sci. 2021;22:13475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 36] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 126. | Zhao ZH, Wang ZX, Zhou D, Han Y, Ma F, Hu Z, Xin FZ, Liu XL, Ren TY, Zhang F, Xue Y, Cui A, Liu Z, Bai J, Liu Y, Cai G, Su W, Dai X, Shen F, Pan Q, Li Y, Fan JG. Sodium Butyrate Supplementation Inhibits Hepatic Steatosis by Stimulating Liver Kinase B1 and Insulin-Induced Gene. Cell Mol Gastroenterol Hepatol. 2021;12:857-871. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 64] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 127. | Zheng L, Ji YY, Wen XL, Duan SL. Fecal microbiota transplantation in the metabolic diseases: Current status and perspectives. World J Gastroenterol. 2022;28:2546-2560. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 31] [Cited by in RCA: 34] [Article Influence: 11.3] [Reference Citation Analysis (4)] |
| 128. | Xue L, Luo W, Wu L, He X, Xia HH, Chen Y. Fecal Microbiota Transplantation for the Treatment of Nonalcoholic Fatty Liver Disease. Explor Res and Hypothesis Med. 2019;4:12-18. [RCA] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 129. | Khoruts A, Sadowsky MJ. Understanding the mechanisms of faecal microbiota transplantation. Nat Rev Gastroenterol Hepatol. 2016;13:508-516. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 377] [Cited by in RCA: 391] [Article Influence: 43.4] [Reference Citation Analysis (0)] |
| 130. | Qiu XX, Cheng SL, Liu YH, Li Y, Zhang R, Li NN, Li Z. Fecal microbiota transplantation for treatment of non-alcoholic fatty liver disease: Mechanism, clinical evidence, and prospect. World J Gastroenterol. 2024;30:833-842. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 8] [Reference Citation Analysis (1)] |
| 131. | Yu Y, Wang W, Zhang F. The Next Generation Fecal Microbiota Transplantation: To Transplant Bacteria or Virome. Adv Sci (Weinh). 2023;10:e2301097. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 30] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 132. | Bajaj JS, Fagan A, Gavis EA, Kassam Z, Sikaroodi M, Gillevet PM. Long-term Outcomes of Fecal Microbiota Transplantation in Patients With Cirrhosis. Gastroenterology. 2019;156:1921-1923.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 132] [Article Influence: 22.0] [Reference Citation Analysis (0)] |
| 133. | Stols-Gonçalves D, Mak AL, Madsen MS, van der Vossen EWJ, Bruinstroop E, Henneman P, Mol F, Scheithauer TPM, Smits L, Witjes J, Meijnikman AS, Verheij J, Nieuwdorp M, Holleboom AG, Levin E. Faecal Microbiota transplantation affects liver DNA methylation in Non-alcoholic fatty liver disease: a multi-omics approach. Gut Microbes. 2023;15:2223330. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 38] [Reference Citation Analysis (0)] |
| 134. | Dailey FE, Turse EP, Daglilar E, Tahan V. The dirty aspects of fecal microbiota transplantation: a review of its adverse effects and complications. Curr Opin Pharmacol. 2019;49:29-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 77] [Article Influence: 12.8] [Reference Citation Analysis (0)] |
| 135. | Karimi M, Shirsalimi N, Hashempour Z, Salehi Omran H, Sedighi E, Beigi F, Mortezazadeh M. Safety and efficacy of fecal microbiota transplantation (FMT) as a modern adjuvant therapy in various diseases and disorders: a comprehensive literature review. Front Immunol. 2024;15:1439176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 27] [Reference Citation Analysis (0)] |
| 136. | Jadhav PA, Thomas AB, Nanda RK, Chitlange SS. Correlation of non-alcoholic fatty liver disease and gut microflora: clinical reports and treatment options. Egypt Liver J. 2024;14:21. [DOI] [Full Text] |
| 137. | Zhu Y, Tan JK, Liu J, Goon JA. Roles of Traditional and Next-Generation Probiotics on Non-Alcoholic Fatty Liver Disease (NAFLD) and Non-Alcoholic Steatohepatitis (NASH): A Systematic Review and Network Meta-Analysis. Antioxidants (Basel). 2024;13:329. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 9] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 138. | Kobyliak N, Abenavoli L, Mykhalchyshyn G, Kononenko L, Boccuto L, Kyriienko D, Dynnyk O. A Multi-strain Probiotic Reduces the Fatty Liver Index, Cytokines and Aminotransferase levels in NAFLD Patients: Evidence from a Randomized Clinical Trial. J Gastrointestin Liver Dis. 2018;27:41-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 192] [Cited by in RCA: 177] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 139. | Pan Y, Yang Y, Wu J, Zhou H, Yang C. Efficacy of probiotics, prebiotics, and synbiotics on liver enzymes, lipid profiles, and inflammation in patients with non-alcoholic fatty liver disease: a systematic review and meta-analysis of randomized controlled trials. BMC Gastroenterol. 2024;24:283. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 16] [Reference Citation Analysis (0)] |
| 140. | Cao C, Shi M, Wang X, Yao Y, Zeng R. Effects of probiotics on non-alcoholic fatty liver disease: a review of human clinical trials. Front Nutr. 2023;10:1155306. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 13] [Reference Citation Analysis (0)] |
| 141. | Abd El Hamid AA, Mohamed AE, Mohamed MS, Amin GEE, Elessawy HAA, Allam MF. The effect of probiotic supplementation on non-alcoholic fatty liver disease (NAFLD) fibrosis score in patients attending a tertiary hospital clinic in Cairo, Egypt. BMC Gastroenterol. 2024;24:354. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (2)] |
| 142. | Fogacci F, Giovannini M, Di Micoli V, Grandi E, Borghi C, Cicero AFG. Effect of Supplementation of a Butyrate-Based Formula in Individuals with Liver Steatosis and Metabolic Syndrome: A Randomized Double-Blind Placebo-Controlled Clinical Trial. Nutrients. 2024;16:2454. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 12] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 143. | Kobyliak N, Conte C, Cammarota G, Haley AP, Styriak I, Gaspar L, Fusek J, Rodrigo L, Kruzliak P. Probiotics in prevention and treatment of obesity: a critical view. Nutr Metab (Lond). 2016;13:14. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 175] [Cited by in RCA: 208] [Article Influence: 23.1] [Reference Citation Analysis (0)] |
| 144. | Duseja A, Acharya SK, Mehta M, Chhabra S; Shalimar, Rana S, Das A, Dattagupta S, Dhiman RK, Chawla YK. High potency multistrain probiotic improves liver histology in non-alcoholic fatty liver disease (NAFLD): a randomised, double-blind, proof of concept study. BMJ Open Gastroenterol. 2019;6:e000315. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 96] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 145. | Depommier C, Everard A, Druart C, Plovier H, Van Hul M, Vieira-Silva S, Falony G, Raes J, Maiter D, Delzenne NM, de Barsy M, Loumaye A, Hermans MP, Thissen JP, de Vos WM, Cani PD. Supplementation with Akkermansia muciniphila in overweight and obese human volunteers: a proof-of-concept exploratory study. Nat Med. 2019;25:1096-1103. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 774] [Cited by in RCA: 1429] [Article Influence: 238.2] [Reference Citation Analysis (0)] |
| 146. | Lanthier N, Rodriguez J, Nachit M, Hiel S, Trefois P, Neyrinck AM, Cani PD, Bindels LB, Thissen JP, Delzenne NM. Microbiota analysis and transient elastography reveal new extra-hepatic components of liver steatosis and fibrosis in obese patients. Sci Rep. 2021;11:659. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 39] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 147. | Benedict M, Zhang X. Non-alcoholic fatty liver disease: An expanded review. World J Hepatol. 2017;9:715-732. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 547] [Cited by in RCA: 511] [Article Influence: 63.9] [Reference Citation Analysis (18)] |
| 148. | Swamikkannu DM, Dasarapu S, Siva RPV, Nallam J, Pabba S. The gut-liver nexus: exploring gut microbiota dysbiosis in non-alcoholic fatty liver disease and its therapeutic implications. Egypt Liver J. 2024;14:28. [DOI] [Full Text] |
| 149. | Mpountouridis A, Tsigalou C, Bezirtzoglou I, Bezirtzoglou E, Stavropoulou E. Gut microbiome in non-alcoholic fatty liver disease. Front Gastroenterol. 2025;3. [DOI] [Full Text] |
| 150. | Yaghmaei H, Bahanesteh A, Soltanipur M, Takaloo S, Rezaei M, Siadat SD. The Role of Gut Microbiota Modification in Nonalcoholic Fatty Liver Disease Treatment Strategies. Int J Hepatol. 2024;2024:4183880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 151. | Vallianou N, Christodoulatos GS, Karampela I, Tsilingiris D, Magkos F, Stratigou T, Kounatidis D, Dalamaga M. Understanding the Role of the Gut Microbiome and Microbial Metabolites in Non-Alcoholic Fatty Liver Disease: Current Evidence and Perspectives. Biomolecules. 2021;12:56. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 175] [Article Influence: 43.8] [Reference Citation Analysis (0)] |
| 152. | Fang H, Rodrigues E-Lacerda R, Barra NG, Kukje Zada D, Robin N, Mehra A, Schertzer JD. Postbiotic Impact on Host Metabolism and Immunity Provides Therapeutic Potential in Metabolic Disease. Endocr Rev. 2025;46:60-79. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 8] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 153. | Herrera J, Henke CA, Bitterman PB. Extracellular matrix as a driver of progressive fibrosis. J Clin Invest. 2018;128:45-53. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 250] [Cited by in RCA: 426] [Article Influence: 60.9] [Reference Citation Analysis (0)] |
| 154. | Maestri M, Santopaolo F, Pompili M, Gasbarrini A, Ponziani FR. Gut microbiota modulation in patients with non-alcoholic fatty liver disease: Effects of current treatments and future strategies. Front Nutr. 2023;10:1110536. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 44] [Reference Citation Analysis (0)] |
| 155. | Sharpton SR, Maraj B, Harding-Theobald E, Vittinghoff E, Terrault NA. Gut microbiome-targeted therapies in nonalcoholic fatty liver disease: a systematic review, meta-analysis, and meta-regression. Am J Clin Nutr. 2019;110:139-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 144] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 156. | Abenavoli L, Maurizi V, Rinninella E, Tack J, Di Berardino A, Santori P, Rasetti C, Procopio AC, Boccuto L, Scarpellini E. Fecal Microbiota Transplantation in NAFLD Treatment. Medicina (Kaunas). 2022;58:1559. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 28] [Reference Citation Analysis (0)] |
| 157. | Wang C, Li Q, Ren J. Microbiota-Immune Interaction in the Pathogenesis of Gut-Derived Infection. Front Immunol. 2019;10:1873. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 93] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 158. | Shen Y, Fan N, Ma SX, Cheng X, Yang X, Wang G. Gut Microbiota Dysbiosis: Pathogenesis, Diseases, Prevention, and Therapy. MedComm (2020). 2025;6:e70168. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 159. | Koyama H, Kamogashira T, Yamasoba T. Heavy Metal Exposure: Molecular Pathways, Clinical Implications, and Protective Strategies. Antioxidants (Basel). 2024;13:76. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 32] [Reference Citation Analysis (0)] |
| 160. | Gersl V, Hrdina R, Vávrová J, Holecková M, Palicka V, Voglová J, Mazurová Y, Bajgar J. Effects of repeated administration of dithiol chelating agent--sodium 2,3-dimercapto-1-propanesulphonate (DMPS)--on biochemical and haematological parameters in rabbits. Acta Medica (Hradec Kralove). 1997;40:3-8. [PubMed] |
| 161. | Flora SJ, Pachauri V. Chelation in metal intoxication. Int J Environ Res Public Health. 2010;7:2745-2788. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 648] [Cited by in RCA: 562] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 162. | Chen H. Iron metabolism in non-alcoholic fatty liver disease: A promising therapeutic target. Liver Res. 2022;6:203-213. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 163. | Rodrigues de Morais T, Gambero A. Iron chelators in obesity therapy - Old drugs from a new perspective? Eur J Pharmacol. 2019;861:172614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 164. | Nelson JE, Klintworth H, Kowdley KV. Iron metabolism in Nonalcoholic Fatty Liver Disease. Curr Gastroenterol Rep. 2012;14:8-16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 78] [Cited by in RCA: 92] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 165. | Orisakwe OE, Amadi CN, Frazzoli C. Management of Iron Overload in Resource Poor Nations: A Systematic Review of Phlebotomy and Natural Chelators. J Toxicol. 2020;2020:4084538. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 166. | Tóth RJ, Csapó J. The role of selenium in nutrition – A review. Acta Univ Sapientiae, Aliment. 2018;11:128-144. [RCA] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 167. | Papp LV, Lu J, Holmgren A, Khanna KK. From selenium to selenoproteins: synthesis, identity, and their role in human health. Antioxid Redox Signal. 2007;9:775-806. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 922] [Cited by in RCA: 894] [Article Influence: 49.7] [Reference Citation Analysis (0)] |
| 168. | Yu SS, Du JL. Selenoprotein S: a therapeutic target for diabetes and macroangiopathy? Cardiovasc Diabetol. 2017;16:101. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 34] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 169. | Speckmann B, Schulz S, Hiller F, Hesse D, Schumacher F, Kleuser B, Geisel J, Obeid R, Grune T, Kipp AP. Selenium increases hepatic DNA methylation and modulates one-carbon metabolism in the liver of mice. J Nutr Biochem. 2017;48:112-119. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 47] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 170. | Feng W, Cui X, Liu B, Liu C, Xiao Y, Lu W, Guo H, He M, Zhang X, Yuan J, Chen W, Wu T. Association of urinary metal profiles with altered glucose levels and diabetes risk: a population-based study in China. PLoS One. 2015;10:e0123742. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 71] [Cited by in RCA: 98] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 171. | Barakat GM, Moustafa ME, Bikhazi AB. Effects of selenium and exendin-4 on glucagon-like peptide-1 receptor, IRS-1, and Raf-1 in the liver of diabetic rats. Biochem Genet. 2012;50:922-935. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 172. | D'Alessio D, Vahl T, Prigeon R. Effects of glucagon-like peptide 1 on the hepatic glucose metabolism. Horm Metab Res. 2004;36:837-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 36] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 173. | Iizuka Y, Ueda Y, Yagi Y, Sakurai E. Significant improvement of insulin resistance of GK rats by treatment with sodium selenate. Biol Trace Elem Res. 2010;138:265-271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 174. | Zhou J, Xu G, Bai Z, Li K, Yan J, Li F, Ma S, Xu H, Huang K. Selenite exacerbates hepatic insulin resistance in mouse model of type 2 diabetes through oxidative stress-mediated JNK pathway. Toxicol Appl Pharmacol. 2015;289:409-418. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 175. | Shojaei Zarghani S, Rahimi Kashkooli N, Bagheri Z, Tabatabaei M, Fattahi MR, Safarpour AR. Dietary selenium intake in relation to non-alcoholic fatty liver disease assessed by fatty liver index and hepatic steatosis index; a cross-sectional study on the baseline data of prospective PERSIAN Kavar cohort study. BMC Endocr Disord. 2023;23:51. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 176. | Day K, Seale LA, Graham RM, Cardoso BR. Selenotranscriptome Network in Non-alcoholic Fatty Liver Disease. Front Nutr. 2021;8:744825. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 28] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 177. | Wang X, Seo YA, Park SK. Serum selenium and non-alcoholic fatty liver disease (NAFLD) in U.S. adults: National Health and Nutrition Examination Survey (NHANES) 2011-2016. Environ Res. 2021;197:111190. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 72] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 178. | Yang Z, Yan C, Liu G, Niu Y, Zhang W, Lu S, Li X, Zhang H, Ning G, Fan J, Qin L, Su Q. Plasma selenium levels and nonalcoholic fatty liver disease in Chinese adults: a cross-sectional analysis. Sci Rep. 2016;6:37288. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 55] [Cited by in RCA: 53] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 179. | Casanova P, Monleon D. Role of selenium in type 2 diabetes, insulin resistance and insulin secretion. World J Diabetes. 2023;14:147-158. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 9] [Cited by in RCA: 21] [Article Influence: 10.5] [Reference Citation Analysis (1)] |
| 180. | Cione E, La Torre C, Cannataro R, Caroleo MC, Plastina P, Gallelli L. Quercetin, Epigallocatechin Gallate, Curcumin, and Resveratrol: From Dietary Sources to Human MicroRNA Modulation. Molecules. 2019;25:63. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 154] [Cited by in RCA: 139] [Article Influence: 23.2] [Reference Citation Analysis (0)] |
| 181. | Izzo C, Annunziata M, Melara G, Sciorio R, Dallio M, Masarone M, Federico A, Persico M. The Role of Resveratrol in Liver Disease: A Comprehensive Review from In Vitro to Clinical Trials. Nutrients. 2021;13:933. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 75] [Article Influence: 18.8] [Reference Citation Analysis (0)] |
| 182. | Kunnumakkara AB, Hegde M, Parama D, Girisa S, Kumar A, Daimary UD, Garodia P, Yenisetti SC, Oommen OV, Aggarwal BB. Role of Turmeric and Curcumin in Prevention and Treatment of Chronic Diseases: Lessons Learned from Clinical Trials. ACS Pharmacol Transl Sci. 2023;6:447-518. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 69] [Article Influence: 34.5] [Reference Citation Analysis (0)] |
| 183. | Tang G, Xu Y, Zhang C, Wang N, Li H, Feng Y. Green Tea and Epigallocatechin Gallate (EGCG) for the Management of Nonalcoholic Fatty Liver Diseases (NAFLD): Insights into the Role of Oxidative Stress and Antioxidant Mechanism. Antioxidants (Basel). 2021;10:1076. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 78] [Cited by in RCA: 71] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 184. | Li X, Liu C, Zhang R, Li Y, Ye D, Wang H, He M, Sun Y. Biosynthetic deficiency of docosahexaenoic acid causes nonalcoholic fatty liver disease and ferroptosis-mediated hepatocyte injury. J Biol Chem. 2024;300:107405. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 185. | Todisco S, Santarsiero A, Convertini P, De Stefano G, Gilio M, Iacobazzi V, Infantino V. PPAR Alpha as a Metabolic Modulator of the Liver: Role in the Pathogenesis of Nonalcoholic Steatohepatitis (NASH). Biology (Basel). 2022;11:792. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 56] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 186. | Victoria Obayomi O, Folakemi Olaniran A, Olugbemiga Owa S. Unveiling the role of functional foods with emphasis on prebiotics and probiotics in human health: A review. J Funct Foods. 2024;119:106337. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 25] [Article Influence: 25.0] [Reference Citation Analysis (0)] |
| 187. | Liao Y, Lv F, Quan T, Wang C, Li J. Flavonoids in natural products for the therapy of liver diseases: progress and future opportunities. Front Pharmacol. 2024;15:1485065. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 188. | Och A, Och M, Nowak R, Podgórska D, Podgórski R. Berberine, a Herbal Metabolite in the Metabolic Syndrome: The Risk Factors, Course, and Consequences of the Disease. Molecules. 2022;27:1351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 46] [Article Influence: 15.3] [Reference Citation Analysis (0)] |
| 189. | Zhu Q, Chen B, Zhang F, Zhang B, Guo Y, Pang M, Huang L, Wang T. Toxic and essential metals: metabolic interactions with the gut microbiota and health implications. Front Nutr. 2024;11:1448388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 17] [Reference Citation Analysis (0)] |
| 190. | Liu ZH, Ai S, Xia Y, Wang HL. Intestinal toxicity of Pb: Structural and functional damages, effects on distal organs and preventive strategies. Sci Total Environ. 2024;931:172781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 191. | Rao Y, Kuang Z, Li C, Guo S, Xu Y, Zhao D, Hu Y, Song B, Jiang Z, Ge Z, Liu X, Li C, Chen S, Ye J, Huang Z, Lu Y. Gut Akkermansia muciniphila ameliorates metabolic dysfunction-associated fatty liver disease by regulating the metabolism of L-aspartate via gut-liver axis. Gut Microbes. 2021;13:1-19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 114] [Cited by in RCA: 191] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 192. | Rondanelli M, Borromeo S, Cavioni A, Gasparri C, Gattone I, Genovese E, Lazzarotti A, Minonne L, Moroni A, Patelli Z, Razza C, Sivieri C, Valentini EM, Barrile GC. Therapeutic Strategies to Modulate Gut Microbial Health: Approaches for Chronic Metabolic Disorder Management. Metabolites. 2025;15:127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 193. | Wu H, Wu R, Chen X, Geng H, Hu Y, Gao L, Fu J, Pi J, Xu Y. Developmental arsenic exposure induces dysbiosis of gut microbiota and disruption of plasma metabolites in mice. Toxicol Appl Pharmacol. 2022;450:116174. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 18] [Reference Citation Analysis (0)] |
| 194. | Li X, Brejnrod AD, Ernst M, Rykær M, Herschend J, Olsen NMC, Dorrestein PC, Rensing C, Sørensen SJ. Heavy metal exposure causes changes in the metabolic health-associated gut microbiome and metabolites. Environ Int. 2019;126:454-467. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 99] [Cited by in RCA: 135] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 195. | Luo Y, Peng S, Cheng J, Yang H, Lin L, Yang G, Jin Y, Wang Q, Wen Z. Chitosan-Stabilized Selenium Nanoparticles Alleviate High-Fat Diet-Induced Non-Alcoholic Fatty Liver Disease (NAFLD) by Modulating the Gut Barrier Function and Microbiota. J Funct Biomater. 2024;15:236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |