Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.108606
Revised: May 11, 2025
Accepted: July 14, 2025
Published online: August 27, 2025
Processing time: 131 Days and 14.7 Hours
The liver is a central metabolic organ that regulates numerous physiological processes, including glucose and lipid metabolism, detoxification, and the synthesis of essential proteins and bile. Bile acids (BAs), synthesized from cholesterol in hepatocytes, not only facilitate the emulsification and absorption of dietary fats but also act as potent signaling molecules through receptors such as the farnesoid X receptor (FXR) and Takeda G-protein-coupled receptor 5. Metabolic dysfunction-associated steatotic liver disease (MASLD) is a leading cause of chronic liver disease globally, closely linked with obesity, insulin resis
Core Tip: Bile acids (BAs) play a central role in lipid metabolism, influencing lipid digestion, absorption, storage and oxidation. Through their actions, as signaling molecules, they regulate a wide range of metabolic processes, including lipid and glucose metabolism, fatty acid oxidation, energy expenditure, and composition of the gut microbiota. The dysregulation of bile acid metabolism is linked to various metabolic diseases, highlighting the importance of BAs in maintaining metabolic homeostasis. Understanding these mechanisms is of great importance and may be crucial for the development of new therapies for these diseases.
- Citation: Zerem E, Kunosic S, Kurtcehajic A, Zerem D, Zerem O. Bile acids in metabolic dysfunction-associated steatotic liver disease. World J Hepatol 2025; 17(8): 108606
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/108606.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.108606
Normal liver function ensures a balance between energy production from lipids and the regulation of lipid levels in the bloodstream. Liver dysfunction can result in lipid metabolism disorders and cause serious health consequences. Fatty liver disease, known as metabolic dysfunction-associated steatotic liver disease (MASLD), is the leading cause of chronic liver disease globally[1-5]. MASLD is usually asymptomatic, but the disease sometimes progresses to a more severe clinical form, called metabolic dysfunction-associated steatohepatitis (MASH), with histological manifestations of inflammation and fibrosis. Further progression of the disease leads to liver cirrhosis, with the risk of developing hepatocellular carcinoma and/or liver failure, as well as all the other consequences that this stage of the disease causes. Despite great interest and numerous publications concerning research into the pathogenesis of this disease, the mechanism by which MASLD progresses to MASH and subsequent cirrhosis has not been fully elucidated[6-9].
Bile acids (BAs) play a very important role in lipid metabolism, influencing the digestion, absorption, storage, and oxidation of lipids. Traditionally, BAs have been considered agents for emulsifying fats and facilitating their intestinal absorption, which contributes to the establishment of cholesterol homoeostasis and nutrient absorption[10-12]. However, more recent insights have highlighted their additional role as signaling molecules that activate receptors such as the farnesoid X receptor (FXR) and Takeda G-protein-coupled receptor 5 (TGR5). These receptors mediate a variety of metabolic processes, positioning BAs as important regulators beyond their classical digestive function[13-15].
All these discoveries have contributed to the fact that BAs and their receptors have become a central focus in understanding the pathophysiology and treatment of fatty liver disease. This minireview aims to clarify the role of BAs in hepatic lipid regulation and explore current and potential therapeutic strategies targeting MASLD and MASH.
BAs are steroid derivatives primarily produced in the liver through the enzymatic conversion of cholesterol, representing the final step in its catabolic pathway. Their synthesis is tightly controlled by a feedback loop that maintains metabolic homeostasis. A central component of this regulatory system is the nuclear receptor FXR, which detects BA concentrations in the liver and intestines and modulates their synthesis accordingly. Elevated BA levels activate FXR, leading to the downregulation of crucial biosynthetic enzymes, most notably cholesterol 7α-hydroxylase [cytochrome P450 family 7 subfamily A member 1 (CYP7A1)], thereby suppressing further production. Conversely, when BA levels decline, this inhibition is lifted, allowing synthesis to resume[16-18].
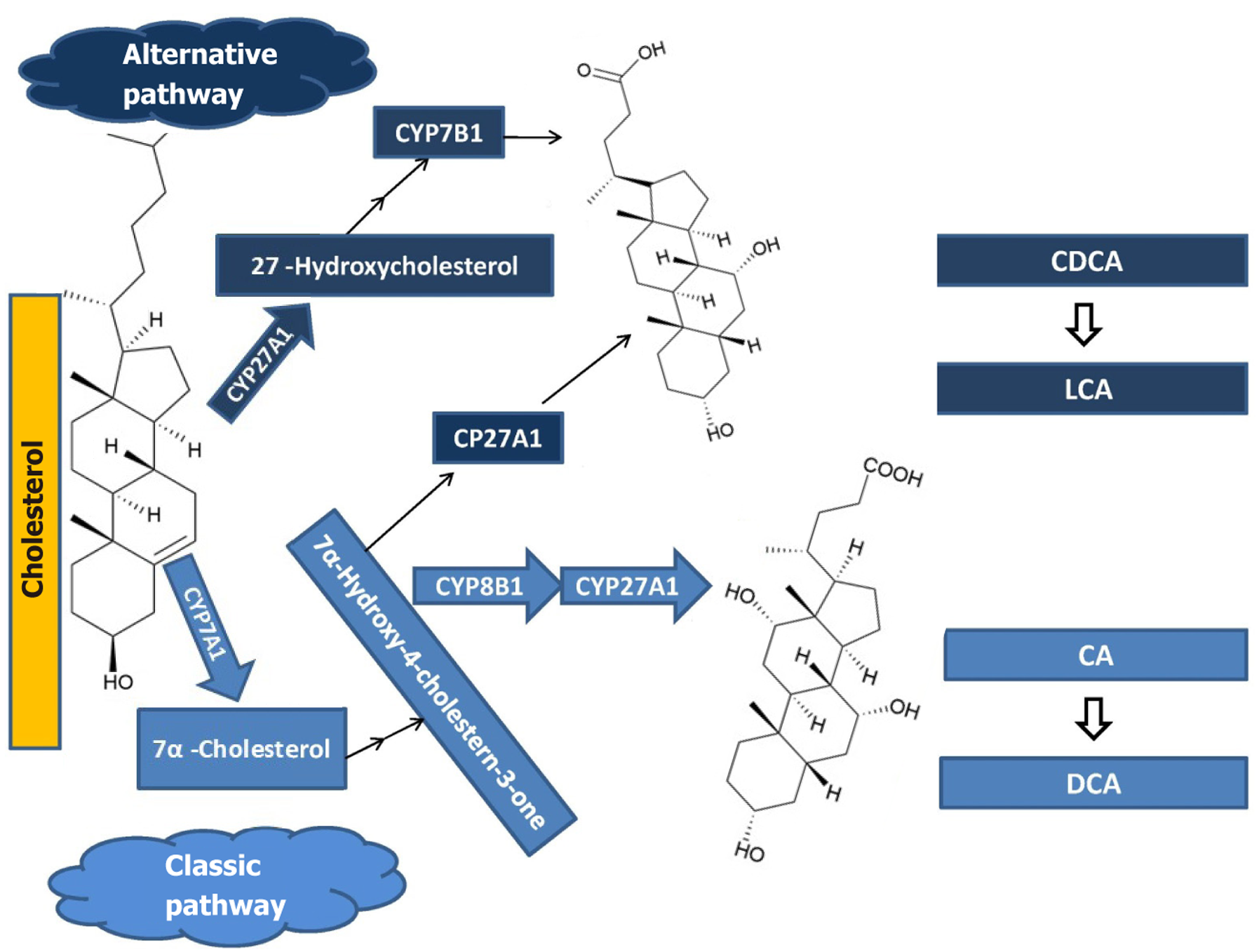
The synthesis of BAs occurs via two distinct pathways: The classical (or neutral) pathway and the alternative (or acidic) pathway (Figure 1).
The classical (neutral) pathway is predominant for the synthesis of BAs in the liver. The process begins with the hydroxylation of cholesterol, catalyzed by the enzyme CYP7A1, which introduces a hydroxyl group at the 7α-position of cholesterol, converting it to 7α-hydroxycholesterol which regulates the rate of BA synthesis. After that, BAs are further modified through hydroxylation and oxidation reactions, resulting in the formation of primary BAs–cholic acid (CA) and chenodeoxycholic acid (CDCA). These are then conjugated with the amino acids glycine or taurine, forming bile salts, which are easily excreted into the small intestine via bile because they are more soluble in water. In the small intestine, primary BAs undergo bacterial modification and are converted into secondary BAs such as deoxycholic acid (DCA) and lithocholic acid (LCA)[16-18].
The alternative (acidic) pathway is carried out by the hydroxylation of cholesterol at positions other than the 7α-position. The key enzyme in this pathway is CYP27A1, which catalyzes the conversion of cholesterol to 27-hydroxycholesterol, which is then, through a series of enzymatic processes, converted to secondary BAs[16-18].
BA synthesis is a very important biological process that ensures proper digestion and absorption of fats and regulates cholesterol metabolism. Furthermore, BAs also act as signaling molecules, influencing various metabolic pathways, including glucose and lipid metabolism, and contributing to the regulation of the composition of the intestinal microbiota[10-13,19].
BAs have a broad influence on metabolic regulation through interaction with various receptors. By acting as signaling molecules, they regulate glucose and lipid metabolism through interaction with nuclear and G-protein-coupled receptors. These receptors are present in various tissues, including the liver, intestine, adipose tissue, and pancreas, where they modulate gene expression and metabolic functions[12-15,19]. Among the key receptors involved in BA-mediated signaling are the FXR and TGR5.
FXR is a nuclear receptor that plays a key role in the regulation of bile acid homeostasis, glucose and lipid metabolism. FXR is activated upon binding to BAs, particularly the primary BAs CA and CDCA and their derivatives, and initiates the expression of genes involved in BA synthesis and transport. FXR inhibits CYP7A1, reducing BA synthesis, and modulates the expression of genes involved in lipid metabolism, including those regulating triglyceride synthesis and lipoprotein metabolism. Its activation enhances the expression of genes that increase fatty acid oxidation and decrease lipid storage in the liver, thereby improving lipid homeostasis. It also regulates the transcription of several genes involved in glucose metabolism. The activation of FXR in the liver suppresses hepatic glucose production by lowering the expression of genes involved in gluconeogenesis, such as phosphoenolpyruvate carboxykinase and glucose-6-phosphatase. In addition, its activation[12-15] increases insulin sensitivity by modulating the expression of insulin signaling genes[19-23].
TGR5 is activated predominantly by secondary BAs such as LCA and ursodeoxycholic acid (UDCA) and is expressed in various tissues, including the gallbladder, intestine, liver and brown adipose tissue. This leads to the activation of signaling pathways that promote the release of glucagon-like peptide 1 (GLP-1), an intestinal hormone that increases insulin secretion and improves insulin sensitivity. It also stimulates the production of BAs in the liver and stimulates energy consumption by stimulating the thermogenic activity of brown fat tissue, which enhances thermogenesis and positively affects glucose metabolism[19,23-25].
Through these receptor-mediated mechanisms, BAs function as crucial modulators of systemic energy metabolism, linking nutrient availability with coordinated hormonal and metabolic responses.
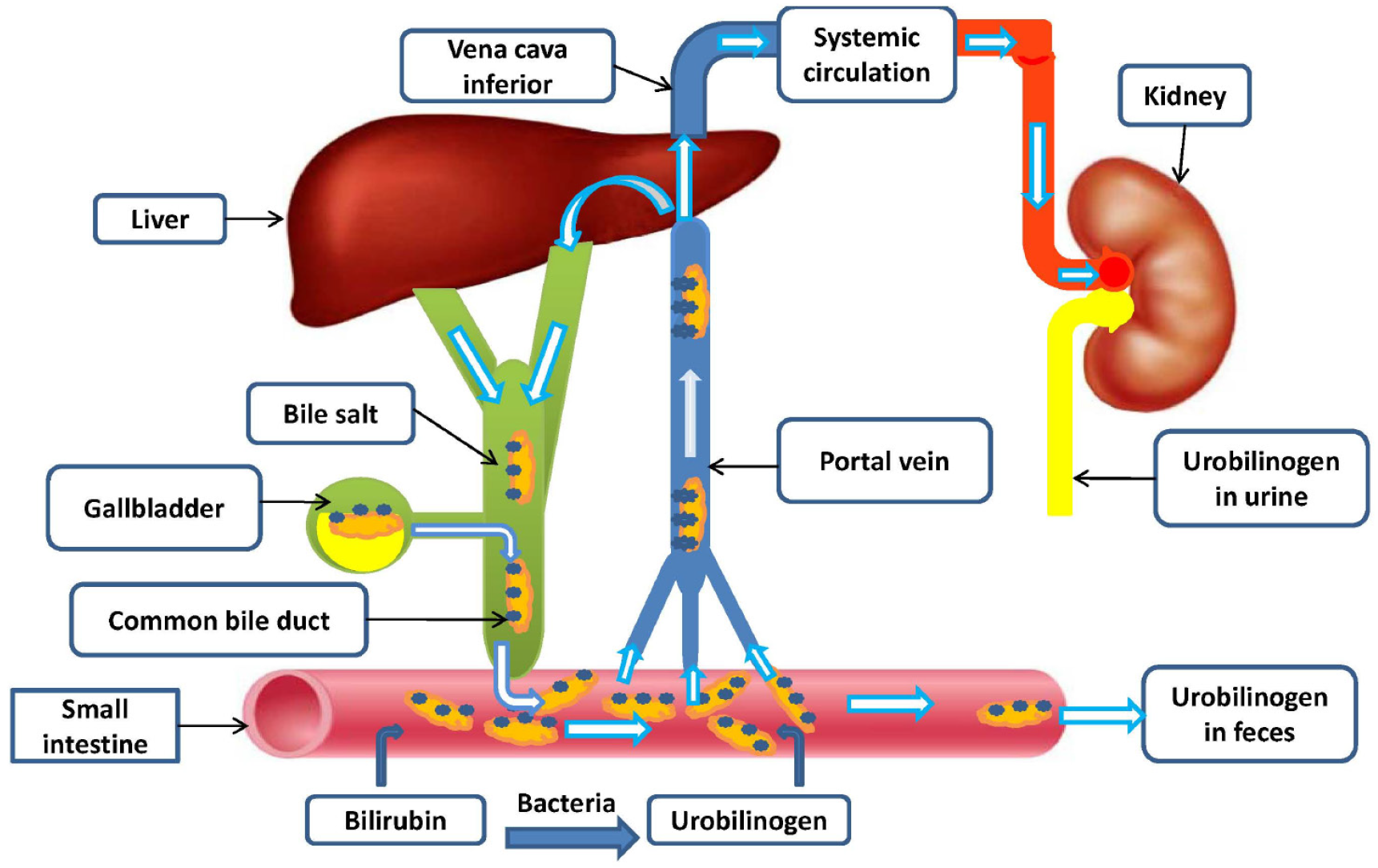
The enterohepatic circulation of BAs is a precisely regulated process that maintains cholesterol homeostasis and normal liver and gastrointestinal tract function. This recycling mechanism enables the continuous movement of BAs between the liver, gallbladder, and intestines, facilitating their reuse and minimizing fecal loss, thereby reducing the need for de novo synthesis (Figure 2).
Approximately 95% of the excreted BAs are reabsorbed in the terminal ileum by active transport mediated by the apical sodium-dependent bile salt cotransporter. Through the portal circulation, reabsorbed BAs are transported back to the liver, where they are taken up by hepatocytes using specific transporters, such as sodium taurocholate cotransporter polypeptide and organic anion transport polypeptides. Hepatocytes then resecrete these BAs into the bile ducts, completing the enterohepatic cycle[26,27]. Enterohepatic circulation, wherein BAs are reabsorbed in the ileum and returned to the liver, is essential for maintaining their homeostasis. A smaller fraction of BAs escape reabsorption in the ileum and reach the colon, where they are transformed by gut microbiota into secondary BAs, including DCA and LCA, which can also be reabsorbed and returned to the liver (Figure 2).
The proper regulation of enterohepatic circulation depends primarily on feedback information obtained through nuclear receptors, such as FXR, which inhibits the expression of CYP7A1, reducing the synthesis of new BAs when their concentration is high. In addition, fibroblast growth factor 19, which is secreted in the intestine, acts as an inhibitor of bile acid synthesis in the liver[26-29].
Enterohepatic circulation dysfunction can be associated with various pathological conditions, such as ileum damage (Crohn’s disease, terminal ileum resection) or the use of drugs such as BA sequestrants (cholestyramine), as well as chronic liver diseases (cirrhosis, cholestatic liver disease), in which disorders in BA metabolism can cause disease progression[26-28]. The complex regulation of BA synthesis and enterohepatic circulation ensures a balance between the synthesis, excretion, and absorption of BAs. Imbalances in this system may lead to a range of metabolic and digestive disorders, highlighting the importance of maintaining functional enterohepatic dynamics[26-29].
Lipid metabolism begins with both mechanical and enzymatic processing in the stomach and continues primarily in the small intestine. In the stomach, fat droplets are physically fragmented into smaller units through mechanical mixing, which increases their surface area and facilitates enzymatic access. Concurrently, a partial enzymatic hydrolysis of triglycerides also occurs in the stomach under the action of gastric lipase. The partially digested lipid fragments then pass into the small intestine, where most of their digestion takes place. In the small intestine, BAs emulsify lipid droplets, further breaking them into even smaller ones (micelles), which additionally increases their surface area and allows pancreatic lipase to efficiently break down triglycerides into free fatty acids and monoglycerides. These digestion products remain within micelles, enabling their passage through the intestinal lumen to the enterocytes. Inside enterocytes, monoglycerides and fatty acids are re-esterified into triglycerides, which are then packaged into chylomicrons. These lipoprotein particles enter the lymphatic system and are subsequently released into the bloodstream for systemic distribution. Once absorbed, lipids are processed in the liver through various metabolic pathways that regulate their distribution, storage, and conversion into energy, ensuring homeostasis between energy supply and lipid concentration in circulation[30-32].
BAs are indispensable in this process not only because of their emulsifying properties which facilitate the digestion and absorption of fats, but also because of their role as signaling molecules. Their amphipathic nature allows them to effectively solubilize dietary fats and fat-soluble vitamins (A, D, E, K). Beyond this classical function, BAs regulate metabolic processes by activating receptors such as FXR and TGR5, influencing lipid and glucose homeostasis as well as the composition of gut microbiota[33-36].
These regulatory mechanisms involve: (1) Modulation of lipid synthesis (lipogenesis): BAs influence transcriptional regulators and enzymes involved in lipogenesis. FXR activation downregulates genes responsible for triglyceride synthesis while simultaneously suppressing sterol regulatory element-binding proteins, which are central to lipid biosynthesis[33,34]; (2) Stimulation of fatty acid oxidation: FXR promotes the expression of peroxisome proliferator-activated receptor alpha, a key regulator of mitochondrial and peroxisomal fatty acid oxidation. TGR5 activation promotes energy expenditure through thermogenesis, particularly by enhancing the conversion of white adipose tissue to a more metabolically active brown-like phenotype[35,36]; and (3) Regulation of cholesterol balance: BAs contribute to cholesterol turnover by modulating gene networks involved in its synthesis, uptake, and elimination. FXR activation leads to the suppression of CYP7A1, a key enzyme in BA biosynthesis, thus modulating cholesterol catabolism. It also promotes pathways that enhance ketogenesis and cholesterol excretion into bile, reinforcing systemic lipid balance[37].
Impairments in BA metabolism or receptor signaling have been linked to a range of metabolic conditions. The dysregulation of BA synthesis or receptor function can lead to an altered lipid metabolism, contributing to the accumulation of ectopic fat in tissues such as the liver and muscle, and increasing the risk of metabolic diseases. In MASLD, bile acid homeostasis is often disrupted, resulting in altered lipid accumulation and hepatic inflammation. Impaired BA signaling via FXR or TGR5 can exacerbate liver lipid deposition and insulin resistance, assisting in the progression of MASLD[38,39].
BAs play an important role in the regulation of glucose metabolism, acting as signaling molecules that modulate metabolic processes through interaction with nuclear receptors. These receptors are present in various tissues, including the liver, intestine, adipose tissue and pancreas, where they modulate gene expression and metabolic functions. The effects of BAs on insulin sensitivity, glucose production and energy expenditure bring them into focus as potential drugs for the treatment of metabolic diseases such as type 2 diabetes mellitus and MASLD[40-42]. Their metabolic effects are exerted through several key mechanisms.
BAs can improve insulin sensitivity through their effects on FXR and TGR5. FXR activation in the liver, reduces insulin resistance by suppressing inflammatory markers and modulating glucose production. TGR5 indirectly stimulates the secretion of GLP-1 which not only enhances insulin release but also elevates insulin sensitivity in peripheral tissues such as muscle and adipose tissue. Reduced TGR5 activity lessens energy expenditure and promotes fat accumulation. Conversely, enhanced TGR5 activation boosts energy expenditure through thermogenesis which contributes to fat reduction and improved glucose homeostasis[41-43].
Intestinal microbes contribute to the conversion of primary into secondary BAs. Altered intestinal microbiota can lead to changes in the composition of BAs and affect glucose homeostasis. Therefore, maintaining a healthy gut microbiota represents a promising strategy for regulating BA metabolism and improving insulin sensitivity[44-46].
FXR in the liver downregulates the expression of key gluconeogenic enzymes, such as phosphoenolpyruvate carboxykinase and glucose-6-phosphatase, thereby reducing glucose production from non-carbohydrate sources, especially during fasting or metabolic stress. BAs may also indirectly modulate glucose production through their effects on hepatic lipid metabolism, preventing hepatic lipid accumulation and lowering insulin resistance[47].
The use of BA sequestrants, FXR agonists and TGR5 activators is being explored as a means to improve insulin sensitivity and regulate glucose production. BA sequestrants, such as colesevelam, are drugs that bind to BAs in the intestine, preventing their reabsorption and promoting their excretion. This leads to an increase in BA synthesis from cholesterol, which may enhance FXR and TGR5 signaling[48].
The interaction between BAs and the gut microbiota plays a crucial role in maintaining metabolic balance and preserving normal liver function. While BAs were traditionally viewed as emulsifying agents that aid in the digestion and absorption of dietary fats, recent research has unequivocally confirmed their role as hormone-like signaling molecules, which influence glucose, lipid and energy metabolism through the activation of nuclear and membrane-bound receptors in the liver and intestines[49-51].
The intestinal microbiota significantly modulates the composition and biological activity of BAs. Through processes such as deconjugation, dihydroxylation, and epimerization, intestinal bacteria convert primary BAs (e.g., CA and CDCA) into secondary forms (such as DCA and LCA)[52]. These microbiota-mediated transformations alter the size and composition of the BA pool and directly impact FXR and TGR5 signaling. Conversely, BAs influence the structure of the gut microbiota through their intrinsic antimicrobial properties, establishing a bidirectional relationship between the host metabolism and gut microbial community[52].
An increasing body of evidence highlights the central role of this interaction in the development of MASLD. BAs and the microbiota engage in a complex, reciprocal communication that affects hepatic lipid metabolism, disease progression, and inflammatory processes. Disruptions in the intestinal microbial community, known as dysbiosis, are characterized by reduced microbial diversity and alterations in microbial composition, which in turn impair BA metabolism and the signaling pathways involved in liver homeostasis[52-54].
In MASLD patients, there is often a decline in bacteria capable of de-conjugating and 7α-dehydroxylating BAs, limiting the production of beneficial secondary BAs. Simultaneously, pro-inflammatory microbial taxa increase, promoting the generation of hepatotoxic BAs. This microbial shift leads to a dominance of primary BAs and a reduction in anti-inflammatory secondary forms[55,56].
Altered BA composition due to intestinal dysbiosis may disrupt FXR signaling. Reduced FXR activity in the intestine leads to increased hepatic lipogenesis and decreased insulin sensitivity, which are key features in the progression of MASLD. Additionally, the accumulation of toxic BAs, such as DCA and LCA, induces hepatocyte damage through oxidative stress, mitochondrial dysfunction, and the activation of pro-inflammatory and pro-fibrotic pathways[57,58].
The interaction between BAs and intestinal microbiota is now recognized as a crucial determinant of MASLD pathogenesis. Disturbances in this correlation contribute to the formation of liver steatosis, inflammation and disease progression.
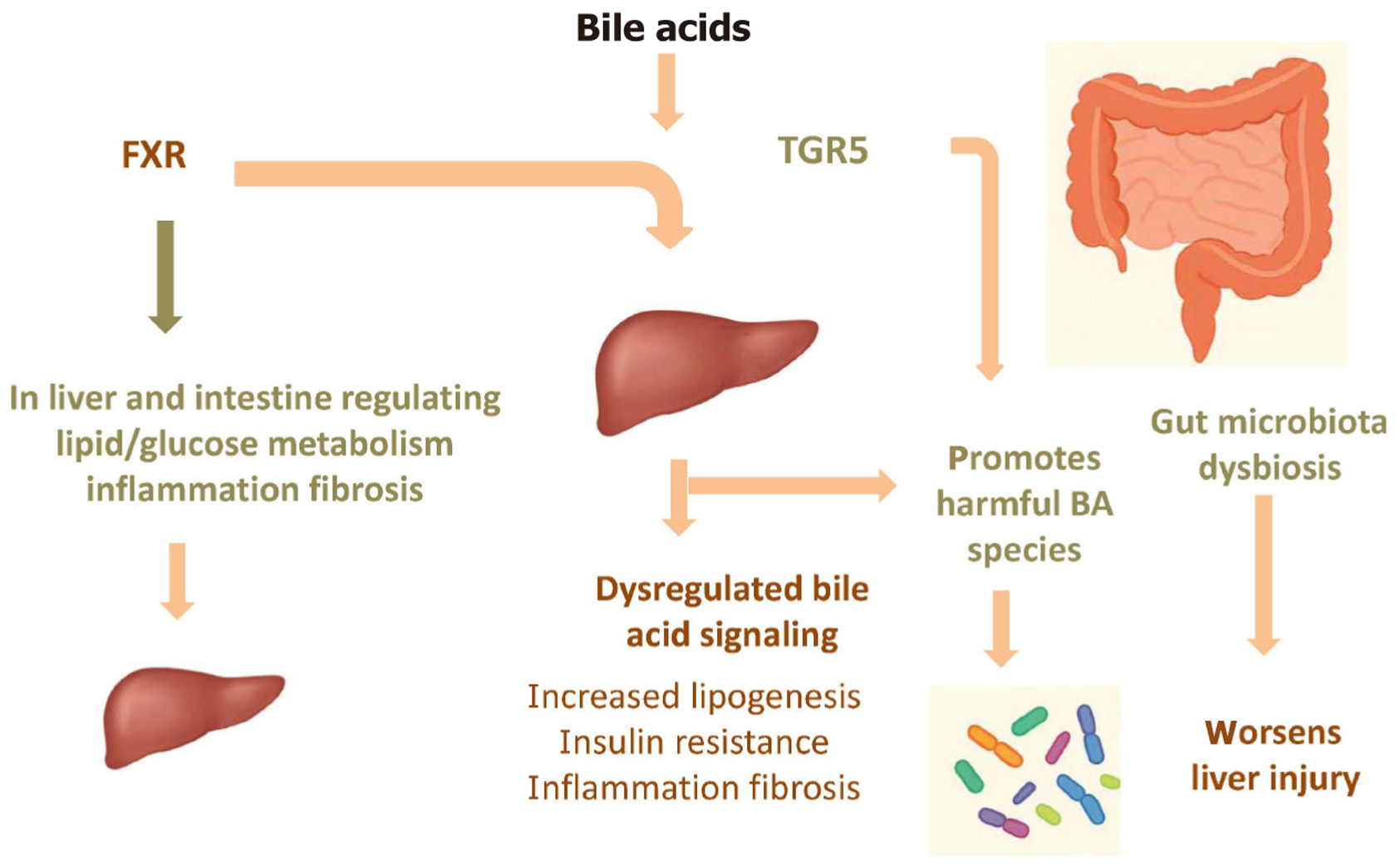
MASLD is not merely a condition of fat accumulation in the liver but a complex biological disorder arising from the intricate interplay of BAs, metabolic pathways, and gut microbiota, as well as inflammation and fibrogenesis. These elements form a closely interconnected network that exacerbates liver tissue injury. The progression of the disease is driven by dynamic and reciprocal interactions among these factors, as illustrated in Figure 3.
Recent studies highlight the role of BAs as powerful signaling molecules that modulate a number of metabolic processes. These effects are primarily mediated through specific receptors, notably FXR and TGR5, which are crucial in maintaining lipid and glucose homeostasis. In MASLD, disturbances in BA synthesis and receptor signaling contribute to insulin resistance, liver steatosis and chronic inflammation[55-58].
MASLD reflects a state of metabolic dysfunction marked by abnormal lipid accumulation in hepatocytes due to enhanced de novo lipogenesis, reduced fatty acid oxidation and disordered lipoprotein metabolism. Hyperglycemia and insulin resistance further promote hepatic lipid buildup by upregulating lipogenic transcription factors (Figure 3). Impaired bile acid signaling worsens these metabolic disturbances, fueling a self-sustaining cycle of lipid overload and hepatic damage[57-59].
The gut–liver axis plays a central role in the pathogenesis of MASLD (Figure 3). Alterations in the composition and function of the gut microbiota disrupt BA metabolism and increase intestinal permeability. This so-called “leaky gut” permits the translocation of microbial-derived products, such as lipopolysaccharide, into the portal circulation, triggering hepatic inflammation through the activation of innate immune pathways. In addition, microbial metabolites can affect BA composition, thereby modulating key signaling pathways mediated by FXR and TGR5, which contributes to impaired metabolic regulation and promotes liver fibrogenesis[14,54,58,59].
Continuous metabolic stress coupled with gut-derived microbial signals activates innate immune responses in the liver. Kupffer cells, hepatocytes, and hepatic stellate cells (HSCs) react by releasing proinflammatory cytokines, including tumor necrosis factor-alpha and interleukin-6, which sustain chronic hepatic inflammation. Dysregulated BA pathways further amplify these inflammatory responses, aggravating hepatocyte damage and setting the stage for fibrotic remodeling[14,54,59,60].
In response to ongoing inflammation and continuous liver tissue damage, HSCs undergo activation and transdifferentiate into myofibroblast-like cells, becoming the primary source of extracellular matrix proteins. These proteins drive the development of liver fibrosis, which is a key histological and clinical indicator of disease severity in MASLD (Figure 3). The activation of HSCs is regulated by a complex synergistic interaction of additional factors, including BAs, proinflammatory cytokines, and microbial-derived toxins, such as lipopolysaccharide[14,55,59,60].
A better understanding of these multifactorial interactions offers promising therapeutic opportunities. Future treatment strategies will likely require multi-targeted approaches aimed at simultaneously modulating inflammation, BA signaling and intestinal microbiota to effectively halt or potentially reverse the fibrotic progression in MASLD.
MASLD encompasses a broad spectrum of liver damage, ranging from simple steatosis to steatohepatitis and advanced fibrosis. The dysregulation of BA synthesis and signaling plays a key role in the pathogenesis of the disease and significantly contributes to its progression. Alterations in the size and composition of the BA pool, along with distur
Among the potential pharmacologic agents, UDCA, a hydrophilic BA, has demonstrated multiple hepatoprotective effects. Its mechanisms of action include the inhibition of oxidative and endoplasmic reticulum stress as well as the stabilization of mitochondrial membranes and modulation of nuclear receptors involved in inflammatory and apoptotic signaling pathways[66-68]. UDCA[14,17,59] exerts these effects through interactions with receptors such as the FXR and pregnane X receptor, which are crucial in BA homeostasis, lipid metabolism and inflammation control[69-73]. Preclinical studies[29] in animal models of MASLD have shown that high-dose UDCA can decrease serum alanine aminotransferase levels, reduce liver steatosis, improve insulin sensitivity, and suppress inflammatory signaling pathways[72-76].
However, clinical studies have not consistently demonstrated that UDCA has significant antifibrotic effects. Moreover, high-dose therapy has been associated with gastrointestinal side effects such as diarrhea, nausea and bloating. Rarely, paradoxical hepatotoxicity or worsening cholestasis has been reported, particularly in patients with pre-existing liver dysfunction[67].
Clinical evidence supports the use of UDCA in the early stages of MASLD, as improvements in liver biochemistry and histological features have been observed, particularly reductions in inflammation and hepatocyte ballooning. The inability of UDCA to directly inhibit the activation of HSCs, which represents a crucial mechanism in the progression of fibrosis, limits its therapeutic use in advanced disease stages. Nonetheless, given its favorable safety profile and good tolerability, UDCA remains a useful therapeutic option in the early stage of MASLD, as early treatment of inflammation and apoptosis may help delay or prevent progression to advanced liver fibrosis, especially when used in combination with lifestyle modifications or agents with antifibrotic properties[65,77].
Furthermore, several studies indicate that UDCA positively influences gut microbiota composition and BA metabolism, thereby indirectly improving liver function by modulating the gut–liver axis. Specific gut bacteria can convert primary BAs into secondary forms, helping to restore BA homeostasis and alleviate the metabolic dysfunction characteristic of MASLD[46,52,78-80].
Combination therapy involving UDCA and bioactive compounds such as quercetin has demonstrated synergistic effects in improving liver function and lessening systemic inflammation, especially in patients with comorbidities such as atrial fibrillation[81]. Similarly, the combination of UDCA with alpha-lipoic acid has been shown to promote the normalization of liver enzymes and reduce steatosis and fibrosis in MASLD patients[82]. Emerging evidence suggests that UDCA may be most effective as part of a multimodal treatment strategy targeting oxidative stress, inflammation and metabolic dysfunction[81-85]. In addition to UDCA, other BAs under investigation or in clinical use for MASLD therapy include obeticholic acid (OCA) and tauroursodeoxycholic acid (TUDCA).
OCA is a semi-synthetic derivative of CDCA and a potent FXR agonist, with significantly greater nanomolar potency compared to natural BAs[86,87]. It has been approved at a daily dose of 25 mg for patients with MASH based on evidence from clinical trials demonstrating improvements in fibrosis and biochemical markers of liver injury[86,88,89]. However, despite its positive impact on disease progression, OCA has not shown a significant benefit on hard clinical outcomes. Additionally, adverse effects such as dose-dependent pruritus and elevations in LDL cholesterol levels have raised concerns regarding its risk-benefit profile. These safety concerns ultimately led to the Food and Drug Administration’s denial of accelerated approval and limited its broader therapeutic application[1,89,90].
TUDCA, a taurine-conjugated form of UDCA, has shown promising results in preclinical models of MASLD. In murine studies, TUDCA administration was associated with reduced intestinal inflammation, improved gut barrier integrity, and the correction of gut microbiota dysbiosis. Nevertheless, its translation into clinical practice remains uncertain and further human studies are needed to confirm its therapeutic potential[91,92].
Experimental BA derivatives, including norUDCA, cilofexor, tropifexor, and INT-767, represent a new class of therapeutics that act as dual FXR/TGR5 agonists or structurally modified BAs designed to enhance efficacy and reduce toxicity. Most of these agents are currently in early or mid-stage clinical trials, and long-term safety and efficacy data are still lacking. Their effectiveness may depend on receptor expression patterns and individual metabolic phenotypes, complicating patient selection[93-96].
BAs are key regulators of hepatic lipid metabolism, glucose homeostasis, and inflammatory responses, primarily through the activation of nuclear and membrane-bound receptors such as FXR and TGR5. In MASLD, alterations in BA synthesis, composition, and receptor-mediated signaling play a crucial role in disease pathogenesis, contributing to hepatic steatosis, insulin resistance, and progression to steatohepatitis and fibrosis. A comprehensive understanding of the complex crosstalk between BAs and lipid metabolism is essential for the development of effective therapeutic strategies for metabolic disorders. Emerging evidence highlights the pleiotropic hepatoprotective effects of BAs, which include the modulation of lipid and glucose metabolism, reduction of oxidative and endoplasmic reticulum stress, and regulation of immune and inflammatory pathways. The therapeutic potential of BAs in MASLD is largely attributed to their dual action on hepatic and intestinal compartments via the gut–liver axis. UDCA has demonstrated a favorable safety profile and therapeutic benefit in early disease stages. However, its efficacy in reversing fibrosis or halting disease progression in advanced MASLD remains limited. Future research should focus on optimizing BA-based therapies, identifying reliable biomarkers for disease staging and treatment response, and evaluating long-term clinical outcomes across diverse and heterogeneous patient populations.
| 1. | European Association for the Study of the Liver (EASL); European Association for the Study of Diabetes (EASD); European Association for the Study of Obesity (EASO). EASL-EASD-EASO Clinical Practice Guidelines on the management of metabolic dysfunction-associated steatotic liver disease (MASLD). J Hepatol. 2024;81:492-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 449] [Article Influence: 449.0] [Reference Citation Analysis (1)] |
| 2. | Kim JH, Lee Y, Nam CM, Kwon YJ, Lee JW. Impact of cardiometabolic risk factors for metabolic dysfunction-associated steatotic liver disease on mortality. Nutr Metab Cardiovasc Dis. 2025;35:103965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 3. | Castera L, Alazawi W, Bugianesi E, Caussy C, Federici M, Romero-Gómez M, Schattenberg JM, Basuroy R, Prasad P, Estulin D, Lazarus JV. A European Survey to Identify Challenges in the Management of Metabolic Dysfunction-Associated Steatotic Liver Disease. Liver Int. 2025;45:e16224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 4. | Zisis M, Chondrogianni ME, Androutsakos T, Rantos I, Oikonomou E, Chatzigeorgiou A, Kassi E. Linking Cardiovascular Disease and Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD): The Role of Cardiometabolic Drugs in MASLD Treatment. Biomolecules. 2025;15:324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 4] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 5. | Kowada A. Risk-stratified hepatocellular carcinoma screening according to the degree of obesity and progression to cirrhosis for diabetic patients with metabolic dysfunction-associated steatotic liver disease (MASLD) in Japan: a cost-effectiveness study. BMJ Open. 2024;14:e080549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 6. | An G, Song J, Ying W, Lim W. Overview of the hazardous impacts of metabolism-disrupting chemicals on the progression of fatty liver diseases. Mol Cell Toxicol. 2025;21:387-397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 7. | Gadi Z, Kwanten WJ, Vonghia L, Francque SM. MASH to cirrhosis: bridging the gaps in MASLD management. Acta Clin Belg. 2024;79:441-450. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 8. | Alferink LJM, Radjabzadeh D, Erler NS, Vojinovic D, Medina-Gomez C, Uitterlinden AG, de Knegt RJ, Amin N, Ikram MA, Janssen HLA, Kiefte-de Jong JC, Metselaar HJ, van Duijn CM, Kraaij R, Darwish Murad S. Microbiomics, Metabolomics, Predicted Metagenomics, and Hepatic Steatosis in a Population-Based Study of 1,355 Adults. Hepatology. 2021;73:968-982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 54] [Article Influence: 13.5] [Reference Citation Analysis (0)] |
| 9. | Singh S, Allen AM, Wang Z, Prokop LJ, Murad MH, Loomba R. Fibrosis progression in nonalcoholic fatty liver vs nonalcoholic steatohepatitis: a systematic review and meta-analysis of paired-biopsy studies. Clin Gastroenterol Hepatol. 2015;13:643-54.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 932] [Cited by in RCA: 1238] [Article Influence: 123.8] [Reference Citation Analysis (0)] |
| 10. | Cheng Z, Chen Y, Schnabl B, Chu H, Yang L. Bile acid and nonalcoholic steatohepatitis: Molecular insights and therapeutic targets. J Adv Res. 2024;59:173-187. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 17] [Article Influence: 17.0] [Reference Citation Analysis (0)] |
| 11. | Chow MD, Lee YH, Guo GL. The role of bile acids in nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Mol Aspects Med. 2017;56:34-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 76] [Cited by in RCA: 113] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 12. | Radun R, Trauner M. Role of FXR in Bile Acid and Metabolic Homeostasis in NASH: Pathogenetic Concepts and Therapeutic Opportunities. Semin Liver Dis. 2021;41:461-475. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 13. | Hernandez GV, Smith VA, Melnyk M, Burd MA, Sprayberry KA, Edwards MS, Peterson DG, Bennet DC, Fanter RK, Columbus DA, Steibel JP, Glanz H, Immoos C, Rice MS, Santiago-Rodriguez TM, Blank J, VanderKelen JJ, Kitts CL, Piccolo BD, La Frano MR, Burrin DG, Maj M, Manjarin R. Dysregulated FXR-FGF19 signaling and choline metabolism are associated with gut dysbiosis and hyperplasia in a novel pig model of pediatric NASH. Am J Physiol Gastrointest Liver Physiol. 2020;318:G582-G609. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 30] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 14. | Arab JP, Karpen SJ, Dawson PA, Arrese M, Trauner M. Bile acids and nonalcoholic fatty liver disease: Molecular insights and therapeutic perspectives. Hepatology. 2017;65:350-362. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 349] [Cited by in RCA: 458] [Article Influence: 57.3] [Reference Citation Analysis (1)] |
| 15. | Duparc T, Plovier H, Marrachelli VG, Van Hul M, Essaghir A, Ståhlman M, Matamoros S, Geurts L, Pardo-Tendero MM, Druart C, Delzenne NM, Demoulin JB, van der Merwe SW, van Pelt J, Bäckhed F, Monleon D, Everard A, Cani PD. Hepatocyte MyD88 affects bile acids, gut microbiota and metabolome contributing to regulate glucose and lipid metabolism. Gut. 2017;66:620-632. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 96] [Cited by in RCA: 132] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 16. | Šarenac TM, Mikov M. Bile Acid Synthesis: From Nature to the Chemical Modification and Synthesis and Their Applications as Drugs and Nutrients. Front Pharmacol. 2018;9:939. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 101] [Article Influence: 14.4] [Reference Citation Analysis (0)] |
| 17. | Kwong E, Li Y, Hylemon PB, Zhou H. Bile acids and sphingosine-1-phosphate receptor 2 in hepatic lipid metabolism. Acta Pharm Sin B. 2015;5:151-157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 107] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 18. | Guan B, Tong J, Hao H, Yang Z, Chen K, Xu H, Wang A. Bile acid coordinates microbiota homeostasis and systemic immunometabolism in cardiometabolic diseases. Acta Pharm Sin B. 2022;12:2129-2149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 108] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 19. | Thibaut MM, Bindels LB. Crosstalk between bile acid-activated receptors and microbiome in entero-hepatic inflammation. Trends Mol Med. 2022;28:223-236. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 101] [Article Influence: 33.7] [Reference Citation Analysis (0)] |
| 20. | Ali AH, Carey EJ, Lindor KD. Recent advances in the development of farnesoid X receptor agonists. Ann Transl Med. 2015;3:5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 103] [Reference Citation Analysis (0)] |
| 21. | Ding L, Yang L, Wang Z, Huang W. Bile acid nuclear receptor FXR and digestive system diseases. Acta Pharm Sin B. 2015;5:135-144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 180] [Cited by in RCA: 317] [Article Influence: 31.7] [Reference Citation Analysis (0)] |
| 22. | Mudaliar S, Henry RR, Sanyal AJ, Morrow L, Marschall HU, Kipnes M, Adorini L, Sciacca CI, Clopton P, Castelloe E, Dillon P, Pruzanski M, Shapiro D. Efficacy and safety of the farnesoid X receptor agonist obeticholic acid in patients with type 2 diabetes and nonalcoholic fatty liver disease. Gastroenterology. 2013;145:574-82.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 656] [Cited by in RCA: 738] [Article Influence: 61.5] [Reference Citation Analysis (0)] |
| 23. | Rachubik P, Grochowalska K, Audzeyenka I, Rogacka D, Piwkowska A. Role of bile acid-dependent Takeda G-coupled protein receptor 5 (TGR5) in regulating AMPK expression in human podocytes. Biochem Biophys Res Commun. 2025;759:151671. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 24. | Park JY, Ha ES, Lee J, Brun PJ, Kim Y, Chung SS, Hwang D, Lee SA, Park KS. The brown fat-specific overexpression of RBP4 improves thermoregulation and systemic metabolism by activating the canonical adrenergic signaling pathway. Exp Mol Med. 2025;57:554-566. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 25. | Zhou H, Hylemon PB. Bile acids are nutrient signaling hormones. Steroids. 2014;86:62-68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 191] [Cited by in RCA: 210] [Article Influence: 19.1] [Reference Citation Analysis (0)] |
| 26. | Zhu Z, Xu Y, Xia Y, Jia X, Chen Y, Liu Y, Zhang L, Chai H, Sun L. Review on chronic metabolic diseases surrounding bile acids and gut microbiota: What we have explored so far. Life Sci. 2024;336:122304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 27. | Dawson PA, Lan T, Rao A. Bile acid transporters. J Lipid Res. 2009;50:2340-2357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 523] [Article Influence: 32.7] [Reference Citation Analysis (0)] |
| 28. | Ferrebee CB, Dawson PA. Metabolic effects of intestinal absorption and enterohepatic cycling of bile acids. Acta Pharm Sin B. 2015;5:129-134. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 92] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 29. | Wang Y, Xu H, Zhou X, Chen W, Zhou H. Dysregulated bile acid homeostasis: unveiling its role in metabolic diseases. Med Rev (2021). 2024;4:262-283. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 30. | Koziolek M, Carrière F, Porter CJH. Lipids in the Stomach - Implications for the Evaluation of Food Effects on Oral Drug Absorption. Pharm Res. 2018;35:55. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 48] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 31. | Lindquist S, Hernell O. Lipid digestion and absorption in early life: an update. Curr Opin Clin Nutr Metab Care. 2010;13:314-320. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 184] [Cited by in RCA: 182] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 32. | Bakala N'Goma JC, Amara S, Dridi K, Jannin V, Carrière F. Understanding the lipid-digestion processes in the GI tract before designing lipid-based drug-delivery systems. Ther Deliv. 2012;3:105-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 122] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 33. | Yang W, Liu R, Xia C, Chen Y, Dong Z, Huang B, Li R, Li M, Xu C. Effects of different fatty acids on BRL3A rat liver cell damage. J Cell Physiol. 2020;235:6246-6256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 14] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 34. | Liu N, Zhao J, Wang J, Teng H, Fu Y, Yuan H. Farnesoid X receptor ligand CDCA suppresses human prostate cancer cells growth by inhibiting lipid metabolism via targeting sterol response element binding protein 1. Am J Transl Res. 2016;8:5118-5124. [PubMed] |
| 35. | Zhou S, You H, Qiu S, Yu D, Bai Y, He J, Cao H, Che Q, Guo J, Su Z. A new perspective on NAFLD: Focusing on the crosstalk between peroxisome proliferator-activated receptor alpha (PPARα) and farnesoid X receptor (FXR). Biomed Pharmacother. 2022;154:113577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 35] [Reference Citation Analysis (0)] |
| 36. | Houten SM, Violante S, Ventura FV, Wanders RJ. The Biochemistry and Physiology of Mitochondrial Fatty Acid β-Oxidation and Its Genetic Disorders. Annu Rev Physiol. 2016;78:23-44. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 301] [Cited by in RCA: 594] [Article Influence: 59.4] [Reference Citation Analysis (0)] |
| 37. | Fiorucci S, Distrutti E. Bile Acid-Activated Receptors, Intestinal Microbiota, and the Treatment of Metabolic Disorders. Trends Mol Med. 2015;21:702-714. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 273] [Cited by in RCA: 369] [Article Influence: 36.9] [Reference Citation Analysis (0)] |
| 38. | Sun J, Dahboul F, Pujos-Guillot E, Petera M, Chu-Van E, Colsch B, Weil D, Di Martino V, Demidem A, Abergel A. PLD2 is a marker for MASLD-HCC with early-stage fibrosis: revealed by lipidomic and gene expression analysis. Metabolomics. 2025;21:39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 39. | Dahboul F, Sun J, Buchard B, Abeywickrama-Samarakoon N, Pujos-Guillot E, Durand S, Petera M, Centeno D, Guerrieri F, Cocca M, Levrero M, Rossary A, Weil D, Di Martino V, Demidem A, Abergel A. Simultaneous Activation of Beta-Oxidation and De Novo Lipogenesis in MASLD-HCC: A New Paradigm. Liver Int. 2025;45:e70006. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 40. | González-Regueiro JA, Moreno-Castañeda L, Uribe M, Chávez-Tapia NC. The Role of Bile Acids in Glucose Metabolism and Their Relation with Diabetes. Ann Hepatol. 2017;16:16-21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 196] [Article Influence: 24.5] [Reference Citation Analysis (0)] |
| 41. | Rajani C, Jia W. Bile acids and their effects on diabetes. Front Med. 2018;12:608-623. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 42] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 42. | Shapiro H, Kolodziejczyk AA, Halstuch D, Elinav E. Bile acids in glucose metabolism in health and disease. J Exp Med. 2018;215:383-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 184] [Cited by in RCA: 329] [Article Influence: 47.0] [Reference Citation Analysis (0)] |
| 43. | Chiang JY, Pathak P, Liu H, Donepudi A, Ferrell J, Boehme S. Intestinal Farnesoid X Receptor and Takeda G Protein Couple Receptor 5 Signaling in Metabolic Regulation. Dig Dis. 2017;35:241-245. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 53] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 44. | Ionescu RF, Enache RM, Cretoiu SM, Gaspar BS. Gut Microbiome Changes in Gestational Diabetes. Int J Mol Sci. 2022;23:12839. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 32] [Article Influence: 10.7] [Reference Citation Analysis (0)] |
| 45. | Wang X, Liu H, Li Y, Huang S, Zhang L, Cao C, Baker PN, Tong C, Zheng P, Qi H. Altered gut bacterial and metabolic signatures and their interaction in gestational diabetes mellitus. Gut Microbes. 2020;12:1-13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 89] [Article Influence: 17.8] [Reference Citation Analysis (0)] |
| 46. | Li J, Wang M, Ma S, Jin Z, Yin H, Yang S. Association of gastrointestinal microbiome and obesity with gestational diabetes mellitus-an updated globally based review of the high-quality literatures. Nutr Diabetes. 2024;14:31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 5] [Reference Citation Analysis (0)] |
| 47. | Panzitt K, Wagner M. FXR in liver physiology: Multiple faces to regulate liver metabolism. Biochim Biophys Acta Mol Basis Dis. 2021;1867:166133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 96] [Article Influence: 24.0] [Reference Citation Analysis (0)] |
| 48. | Staels B, Handelsman Y, Fonseca V. Bile acid sequestrants for lipid and glucose control. Curr Diab Rep. 2010;10:70-77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 63] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 49. | Li T, Chiang JY. Bile acid signaling in metabolic disease and drug therapy. Pharmacol Rev. 2014;66:948-983. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 728] [Cited by in RCA: 696] [Article Influence: 63.3] [Reference Citation Analysis (0)] |
| 50. | Li T, Chiang JYL. Bile Acid Signaling in Metabolic and Inflammatory Diseases and Drug Development. Pharmacol Rev. 2024;76:1221-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 11] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 51. | Ridlon JM, Harris SC, Bhowmik S, Kang DJ, Hylemon PB. Consequences of bile salt biotransformations by intestinal bacteria. Gut Microbes. 2016;7:22-39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 532] [Cited by in RCA: 742] [Article Influence: 82.4] [Reference Citation Analysis (0)] |
| 52. | Wahlström A, Sayin SI, Marschall HU, Bäckhed F. Intestinal Crosstalk between Bile Acids and Microbiota and Its Impact on Host Metabolism. Cell Metab. 2016;24:41-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1317] [Cited by in RCA: 1852] [Article Influence: 205.8] [Reference Citation Analysis (0)] |
| 53. | Albillos A, de Gottardi A, Rescigno M. The gut-liver axis in liver disease: Pathophysiological basis for therapy. J Hepatol. 2020;72:558-577. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 542] [Cited by in RCA: 1265] [Article Influence: 253.0] [Reference Citation Analysis (1)] |
| 54. | Tripathi A, Debelius J, Brenner DA, Karin M, Loomba R, Schnabl B, Knight R. The gut-liver axis and the intersection with the microbiome. Nat Rev Gastroenterol Hepatol. 2018;15:397-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 872] [Cited by in RCA: 973] [Article Influence: 139.0] [Reference Citation Analysis (0)] |
| 55. | Jiao N, Baker SS, Nugent CA, Tsompana M, Cai L, Wang Y, Buck MJ, Genco RJ, Baker RD, Zhu R, Zhu L. Gut microbiome may contribute to insulin resistance and systemic inflammation in obese rodents: a meta-analysis. Physiol Genomics. 2018;50:244-254. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 118] [Cited by in RCA: 208] [Article Influence: 29.7] [Reference Citation Analysis (0)] |
| 56. | Wang B, Jiang X, Cao M, Ge J, Bao Q, Tang L, Chen Y, Li L. Altered Fecal Microbiota Correlates with Liver Biochemistry in Nonobese Patients with Non-alcoholic Fatty Liver Disease. Sci Rep. 2016;6:32002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 167] [Cited by in RCA: 274] [Article Influence: 30.4] [Reference Citation Analysis (0)] |
| 57. | Sayin SI, Wahlström A, Felin J, Jäntti S, Marschall HU, Bamberg K, Angelin B, Hyötyläinen T, Orešič M, Bäckhed F. Gut microbiota regulates bile acid metabolism by reducing the levels of tauro-beta-muricholic acid, a naturally occurring FXR antagonist. Cell Metab. 2013;17:225-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1367] [Cited by in RCA: 1683] [Article Influence: 140.3] [Reference Citation Analysis (0)] |
| 58. | Jia W, Xie G, Jia W. Bile acid-microbiota crosstalk in gastrointestinal inflammation and carcinogenesis. Nat Rev Gastroenterol Hepatol. 2018;15:111-128. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1355] [Cited by in RCA: 1261] [Article Influence: 180.1] [Reference Citation Analysis (0)] |
| 59. | Fiorucci S, Biagioli M, Zampella A, Distrutti E. Bile Acids Activated Receptors Regulate Innate Immunity. Front Immunol. 2018;9:1853. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 202] [Cited by in RCA: 372] [Article Influence: 53.1] [Reference Citation Analysis (0)] |
| 60. | Seki E, Schwabe RF. Hepatic inflammation and fibrosis: functional links and key pathways. Hepatology. 2015;61:1066-1079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 548] [Cited by in RCA: 742] [Article Influence: 74.2] [Reference Citation Analysis (0)] |
| 61. | Ferslew BC, Xie G, Johnston CK, Su M, Stewart PW, Jia W, Brouwer KL, Barritt AS 4th. Altered Bile Acid Metabolome in Patients with Nonalcoholic Steatohepatitis. Dig Dis Sci. 2015;60:3318-3328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 284] [Cited by in RCA: 273] [Article Influence: 27.3] [Reference Citation Analysis (0)] |
| 62. | Simental-Mendía M, Sánchez-García A, Simental-Mendía LE. Effect of ursodeoxycholic acid on liver markers: A systematic review and meta-analysis of randomized placebo-controlled clinical trials. Br J Clin Pharmacol. 2020;86:1476-1488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 16] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 63. | Hofmann AF, Hagey LR. Bile acids: chemistry, pathochemistry, biology, pathobiology, and therapeutics. Cell Mol Life Sci. 2008;65:2461-2483. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 651] [Cited by in RCA: 609] [Article Influence: 35.8] [Reference Citation Analysis (1)] |
| 64. | Lin X, Mai M, He T, Huang H, Zhang P, Xia E, Guo H. Efficiency of ursodeoxycholic acid for the treatment of nonalcoholic steatohepatitis: A systematic review and meta-analysis. Expert Rev Gastroenterol Hepatol. 2022;16:537-545. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 17] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 65. | Ratziu V, de Ledinghen V, Oberti F, Mathurin P, Wartelle-Bladou C, Renou C, Sogni P, Maynard M, Larrey D, Serfaty L, Bonnefont-Rousselot D, Bastard JP, Rivière M, Spénard J; FRESGUN. A randomized controlled trial of high-dose ursodesoxycholic acid for nonalcoholic steatohepatitis. J Hepatol. 2011;54:1011-1019. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 208] [Cited by in RCA: 237] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 66. | Amaral JD, Viana RJ, Ramalho RM, Steer CJ, Rodrigues CM. Bile acids: regulation of apoptosis by ursodeoxycholic acid. J Lipid Res. 2009;50:1721-1734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 264] [Article Influence: 16.5] [Reference Citation Analysis (0)] |
| 67. | Beuers U, Trauner M, Jansen P, Poupon R. New paradigms in the treatment of hepatic cholestasis: from UDCA to FXR, PXR and beyond. J Hepatol. 2015;62:S25-S37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 332] [Cited by in RCA: 382] [Article Influence: 38.2] [Reference Citation Analysis (0)] |
| 68. | Rodrigues CM, Steer CJ. The therapeutic effects of ursodeoxycholic acid as an anti-apoptotic agent. Expert Opin Investig Drugs. 2001;10:1243-1253. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 69. | Mao Q, Lin B, Zhang W, Zhang Y, Zhang Y, Cao Q, Xu M. Understanding the role of ursodeoxycholic acid and gut microbiome in non-alcoholic fatty liver disease: current evidence and perspectives. Front Pharmacol. 2024;15:1371574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 8] [Reference Citation Analysis (0)] |
| 70. | Chávez-Talavera O, Tailleux A, Lefebvre P, Staels B. Bile Acid Control of Metabolism and Inflammation in Obesity, Type 2 Diabetes, Dyslipidemia, and Nonalcoholic Fatty Liver Disease. Gastroenterology. 2017;152:1679-1694.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 458] [Cited by in RCA: 679] [Article Influence: 84.9] [Reference Citation Analysis (1)] |
| 71. | Lee JM, Gang GT, Kim DK, Kim YD, Koo SH, Lee CH, Choi HS. Ursodeoxycholic acid inhibits liver X receptor α-mediated hepatic lipogenesis via induction of the nuclear corepressor SMILE. J Biol Chem. 2014;289:1079-1091. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 40] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 72. | Lin YN, Wang CCN, Chang HY, Chu FY, Hsu YA, Cheng WK, Ma WC, Chen CJ, Wan L, Lim YP. Ursolic Acid, a Novel Liver X Receptor α (LXRα) Antagonist Inhibiting Ligand-Induced Nonalcoholic Fatty Liver and Drug-Induced Lipogenesis. J Agric Food Chem. 2018;66:11647-11662. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 73. | Fujita K, Iguchi Y, Une M, Watanabe S. Ursodeoxycholic Acid Suppresses Lipogenesis in Mouse Liver: Possible Role of the Decrease in β-Muricholic Acid, a Farnesoid X Receptor Antagonist. Lipids. 2017;52:335-344. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 74. | Li T, Chiang JYL. Bile acid-based therapies for non-alcoholic steatohepatitis and alcoholic liver disease. Hepatobiliary Surg Nutr. 2020;9:152-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 66] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 75. | Mueller M, Castro RE, Thorell A, Marschall HU, Auer N, Herac M, Rodrigues CMP, Trauner M. Ursodeoxycholic acid: Effects on hepatic unfolded protein response, apoptosis and oxidative stress in morbidly obese patients. Liver Int. 2018;38:523-531. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 76. | Oh AR, Bae JS, Lee J, Shin E, Oh BC, Park SC, Cha JY. Ursodeoxycholic acid decreases age-related adiposity and inflammation in mice. BMB Rep. 2016;49:105-110. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 24] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 77. | Lindor KD, Kowdley KV, Heathcote EJ, Harrison ME, Jorgensen R, Angulo P, Lymp JF, Burgart L, Colin P. Ursodeoxycholic acid for treatment of nonalcoholic steatohepatitis: results of a randomized trial. Hepatology. 2004;39:770-778. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 516] [Cited by in RCA: 493] [Article Influence: 23.5] [Reference Citation Analysis (0)] |
| 78. | Fuchs CD, Simbrunner B, Baumgartner M, Campbell C, Reiberger T, Trauner M. Bile acid metabolism and signalling in liver disease. J Hepatol. 2025;82:134-153. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 20] [Reference Citation Analysis (0)] |
| 79. | Li H, Wang Q, Chen P, Zhou C, Zhang X, Chen L. Ursodeoxycholic Acid Treatment Restores Gut Microbiota and Alleviates Liver Inflammation in Non-Alcoholic Steatohepatitic Mouse Model. Front Pharmacol. 2021;12:788558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 53] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 80. | Zazueta A, Valenzuela-Pérez L, Ortiz-López N, Pinto-León A, Torres V, Guiñez D, Aliaga N, Merino P, Sandoval A, Covarrubias N, Pérez de Arce E, Cattaneo M, Urzúa A, Roblero JP, Poniachik J, Gotteland M, Magne F, Beltrán CJ. Alteration of Gut Microbiota Composition in the Progression of Liver Damage in Patients with Metabolic Dysfunction-Associated Steatotic Liver Disease (MASLD). Int J Mol Sci. 2024;25:4387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 15] [Reference Citation Analysis (0)] |
| 81. | Teslenko O, Fedorov S, Bielinskyi M, Herashchenko A, Nyshchuk-oliinyk N. Synergistic effects of ursodeoxycholic acid, and quercetin on liver function and systemic inflammation in non-alcoholic fatty liver disease and atrial fibrillation patients. Gastroenterology. 2024;58:102-108. [DOI] [Full Text] |
| 82. | Gianturco V, Troisi G, Bellomo A, Bernardini S, D'Ottavio E, Formosa V, Iacono CL, Verrusio W, Marigliano B, Marigliano V. Impact of combined therapy with alpha-lipoic and ursodeoxycolic acid on nonalcoholic fatty liver disease: double-blind, randomized clinical trial of efficacy and safety. Hepatol Int. 2013;7:570-576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 22] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 83. | Prikhodko VA, Bezborodkina NN, Okovityi SV. Pharmacotherapy for Non-Alcoholic Fatty Liver Disease: Emerging Targets and Drug Candidates. Biomedicines. 2022;10:274. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 84. | Zhang Y, Lu J, Dai W, Wang F, Shen M, Yang J, Zhu R, Zhang H, Chen K, Cheng P, He L, Wang C, Xu L, Zhou Y, Guo C. Combination therapy of ursodeoxycholic Acid and corticosteroids for primary biliary cirrhosis with features of autoimmune hepatitis: a meta-analysis. Gastroenterol Res Pract. 2013;2013:490731. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 17] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 85. | Jiang L, Yi R, Chen H, Wu S. Quercetin alleviates metabolic-associated fatty liver disease by tuning hepatic lipid metabolism, oxidative stress and inflammation. Anim Biotechnol. 2025;36:2442351. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 6] [Reference Citation Analysis (0)] |
| 86. | Pellicciari R, Fiorucci S, Camaioni E, Clerici C, Costantino G, Maloney PR, Morelli A, Parks DJ, Willson TM. 6alpha-ethyl-chenodeoxycholic acid (6-ECDCA), a potent and selective FXR agonist endowed with anticholestatic activity. J Med Chem. 2002;45:3569-3572. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 565] [Cited by in RCA: 612] [Article Influence: 26.6] [Reference Citation Analysis (0)] |
| 87. | Fiorucci S, Baldoni M, Ricci P, Zampella A, Distrutti E, Biagioli M. Bile acid-activated receptors and the regulation of macrophages function in metabolic disorders. Curr Opin Pharmacol. 2020;53:45-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 42] [Article Influence: 8.4] [Reference Citation Analysis (0)] |
| 88. | Neuschwander-Tetri BA, Loomba R, Sanyal AJ, Lavine JE, Van Natta ML, Abdelmalek MF, Chalasani N, Dasarathy S, Diehl AM, Hameed B, Kowdley KV, McCullough A, Terrault N, Clark JM, Tonascia J, Brunt EM, Kleiner DE, Doo E; NASH Clinical Research Network. Farnesoid X nuclear receptor ligand obeticholic acid for non-cirrhotic, non-alcoholic steatohepatitis (FLINT): a multicentre, randomised, placebo-controlled trial. Lancet. 2015;385:956-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1726] [Cited by in RCA: 1799] [Article Influence: 179.9] [Reference Citation Analysis (3)] |
| 89. | Sanyal AJ, Ratziu V, Loomba R, Anstee QM, Kowdley KV, Rinella ME, Sheikh MY, Trotter JF, Knapple W, Lawitz EJ, Abdelmalek MF, Newsome PN, Boursier J, Mathurin P, Dufour JF, Berrey MM, Shiff SJ, Sawhney S, Capozza T, Leyva R, Harrison SA, Younossi ZM. Results from a new efficacy and safety analysis of the REGENERATE trial of obeticholic acid for treatment of pre-cirrhotic fibrosis due to non-alcoholic steatohepatitis. J Hepatol. 2023;79:1110-1120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 65] [Article Influence: 32.5] [Reference Citation Analysis (0)] |
| 90. | Rinella ME, Dufour JF, Anstee QM, Goodman Z, Younossi Z, Harrison SA, Loomba R, Sanyal AJ, Bonacci M, Trylesinski A, Natha M, Shringarpure R, Granston T, Venugopal A, Ratziu V. Non-invasive evaluation of response to obeticholic acid in patients with NASH: Results from the REGENERATE study. J Hepatol. 2022;76:536-548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 108] [Article Influence: 36.0] [Reference Citation Analysis (0)] |
| 91. | Wang W, Zhao J, Gui W, Sun D, Dai H, Xiao L, Chu H, Du F, Zhu Q, Schnabl B, Huang K, Yang L, Hou X. Tauroursodeoxycholic acid inhibits intestinal inflammation and barrier disruption in mice with non-alcoholic fatty liver disease. Br J Pharmacol. 2018;175:469-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 152] [Cited by in RCA: 150] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 92. | Lu Q, Jiang Z, Wang Q, Hu H, Zhao G. The effect of Tauroursodeoxycholic acid (TUDCA) and gut microbiota on murine gallbladder stone formation. Ann Hepatol. 2021;23:100289. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 30] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 93. | Patel K, Harrison SA, Elkhashab M, Trotter JF, Herring R, Rojter SE, Kayali Z, Wong VW, Greenbloom S, Jayakumar S, Shiffman ML, Freilich B, Lawitz EJ, Gane EJ, Harting E, Xu J, Billin AN, Chung C, Djedjos CS, Subramanian GM, Myers RP, Middleton MS, Rinella M, Noureddin M. Cilofexor, a Nonsteroidal FXR Agonist, in Patients With Noncirrhotic NASH: A Phase 2 Randomized Controlled Trial. Hepatology. 2020;72:58-71. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 263] [Article Influence: 52.6] [Reference Citation Analysis (0)] |
| 94. | Alkhouri N, Herring R, Kabler H, Kayali Z, Hassanein T, Kohli A, Huss RS, Zhu Y, Billin AN, Damgaard LH, Buchholtz K, Kjær MS, Balendran C, Myers RP, Loomba R, Noureddin M. Safety and efficacy of combination therapy with semaglutide, cilofexor and firsocostat in patients with non-alcoholic steatohepatitis: A randomised, open-label phase II trial. J Hepatol. 2022;77:607-618. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 147] [Article Influence: 49.0] [Reference Citation Analysis (0)] |
| 95. | Traussnigg S, Schattenberg JM, Demir M, Wiegand J, Geier A, Teuber G, Hofmann WP, Kremer AE, Spreda F, Kluwe J, Petersen J, Boettler T, Rainer F, Halilbasic E, Greinwald R, Pröls M, Manns MP, Fickert P, Trauner M; Austrian/German NAFLD-norUDCA study group. Norursodeoxycholic acid versus placebo in the treatment of non-alcoholic fatty liver disease: a double-blind, randomised, placebo-controlled, phase 2 dose-finding trial. Lancet Gastroenterol Hepatol. 2019;4:781-793. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 65] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 96. | Carlsson B, Lindén D, Brolén G, Liljeblad M, Bjursell M, Romeo S, Loomba R. Review article: the emerging role of genetics in precision medicine for patients with non-alcoholic steatohepatitis. Aliment Pharmacol Ther. 2020;51:1305-1320. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 59] [Cited by in RCA: 130] [Article Influence: 26.0] [Reference Citation Analysis (0)] |