Published online Aug 27, 2025. doi: 10.4254/wjh.v17.i8.107041
Revised: May 8, 2025
Accepted: July 9, 2025
Published online: August 27, 2025
Processing time: 161 Days and 3.6 Hours
Pediatric liver masses encompass a diverse spectrum of benign and malignant lesions, with distinct patterns based on patient age. Optimal imaging is critical for timely diagnosis, management, and prognosis. This pictorial minireview cate
Core Tip: Pediatric liver masses are diverse, with distinct patterns based on age. This minireview categorizes benign and malignant hepatic lesions by age group, emphasizing imaging characteristics for accurate diagnosis and management. Key findings include the prominence of hepatoblastoma in infants, hepatocellular carcinoma in older children, and various benign lesions such as focal nodular hyperplasia and hepatocellular adenoma. Advanced imaging techniques, including ultrasonography, computed tomography, and magnetic resonance imaging, play a crucial role in identifying and staging these masses, helping clinicians tailor effective management strategies in pediatric hepatology, particularly in lower-middle income countries.
- Citation: Shahid M, Hilal K, Khan M, Ejaz ZH, Altaf S, Islam S, Khandwala K. Imaging insights into pediatric liver masses: A comprehensive minireview for hepatology practice. World J Hepatol 2025; 17(8): 107041
- URL: https://www.wjgnet.com/1948-5182/full/v17/i8/107041.htm
- DOI: https://dx.doi.org/10.4254/wjh.v17.i8.107041
Primary hepatic tumors are rare in children, accounting for only 5%-6% of all intra-abdominal masses and 0.5%-2% of all pediatric neoplasms. The incidence of primary hepatic lesions varies with patient age and is approximately 0.4-1.9 per million children each year[1]. This minireview aims to review both benign and malignant liver lesions in the pediatric population on the basis of age groups.
Ultrasonography is typically the first imaging modality for the initial assessment of hepatic lesions. For further characterization, computed tomography (CT) and magnetic resonance imaging (MRI) are commonly performed. In most cases, CT and MRI provide conclusive assessments without the need for additional imaging. Only in select cases are nuclear medicine studies required for further evaluation.
Despite significant advances in imaging technologies globally—including diffusion-weighted imaging (DWI) and perfusion imaging, quantitative MRI, and artificial intelligence-assisted diagnostic tools—the application of these modalities remains limited in low-income and middle-income countries (LMICs) owing to resource constraints and a lack of widespread access. As such, this minireview focuses primarily on conventional imaging techniques, namely ultrasound (US), CT, and MRI, which remain the main methods for evaluating pediatric liver lesions in LMIC.
Hepatic lesions in children can be categorized in various ways. In this pictorial minireview, we classify pediatric hepatic lesions into three groups: (1) Those frequently observed from birth to six years of age; (2) Those typically observed in children older than six years; and (3) Those that can occur across all age groups.
Hepatoblastoma is the most common hepatic malignancy in children, predominantly presenting before the age of five years, accounting for 91% of primary hepatic malignancies in this age group. The incidence of hepatoblastoma is approximately 10.5 cases per million in children less than one year of age and 5.2 cases per million in children between one and four years of age[2].
Hepatoblastoma is a malignant hepatic tumor of both epithelial and mesenchymal origin. Low birth weight and prematurity are recognized risk factors for its development[3]. Other predisposing conditions include Beckwith-Wiedemann syndrome and familial adenomatous polyposis. When diagnosed prenatally, delivery via cesarean section is recommended, as spontaneous vaginal delivery may be complicated by tumor rupture, resulting in perinatal hemorrhage and cardiovascular compromise.
Children with hepatoblastoma typically present with a large, painless abdominal mass or abdominal distension, often accompanied by nonspecific symptoms such as fatigue, anorexia, weight loss, and pain. Alpha-fetoprotein (AFP) levels are usually markedly elevated in children with this malignancy.
Hepatoblastomas can occur sporadically, be familial, or be associated with syndromic conditions such as Beckwith-Wiedemann syndrome. Many affected fetuses are found to have macroglossia in utero, with some developing hydrops fetalis and polyhydramnios. Hepatomegaly and nephromegaly may also be observed in these patients. Maternal AFP levels may be elevated in pregnancies involving fetuses with hepatoblastoma. When diagnosed prenatally, hepatoblastoma is typically associated with a poor prognosis.
Hepatoblastomas are typically large, solitary masses, often measuring more than 10 cm in size. However, in approximately 20% of cases, these lesions may be multifocal. Owing to their large size, they frequently cause compression and displacement of adjacent structures and are more commonly found in the right lobe of the liver than in the left lobe.
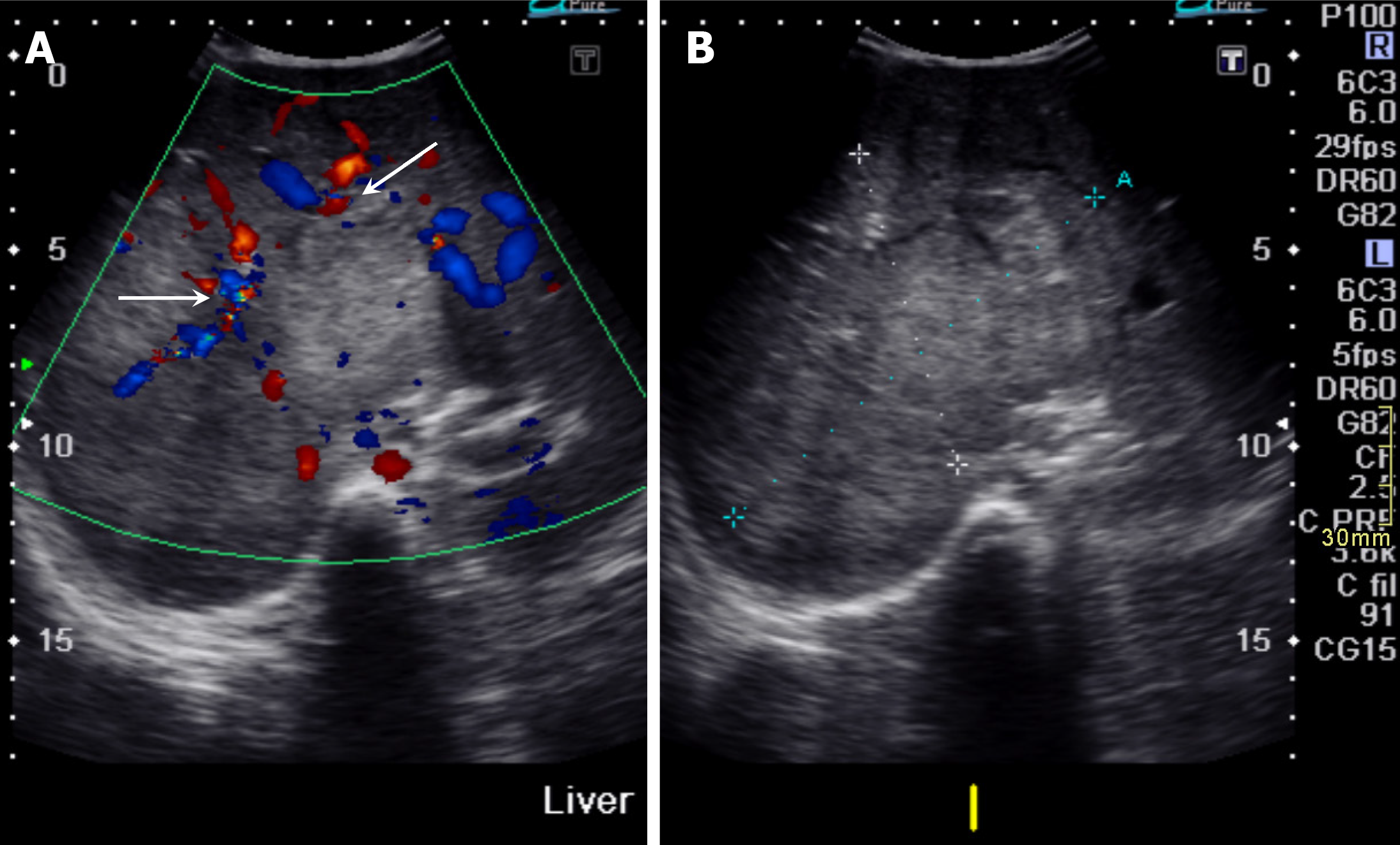
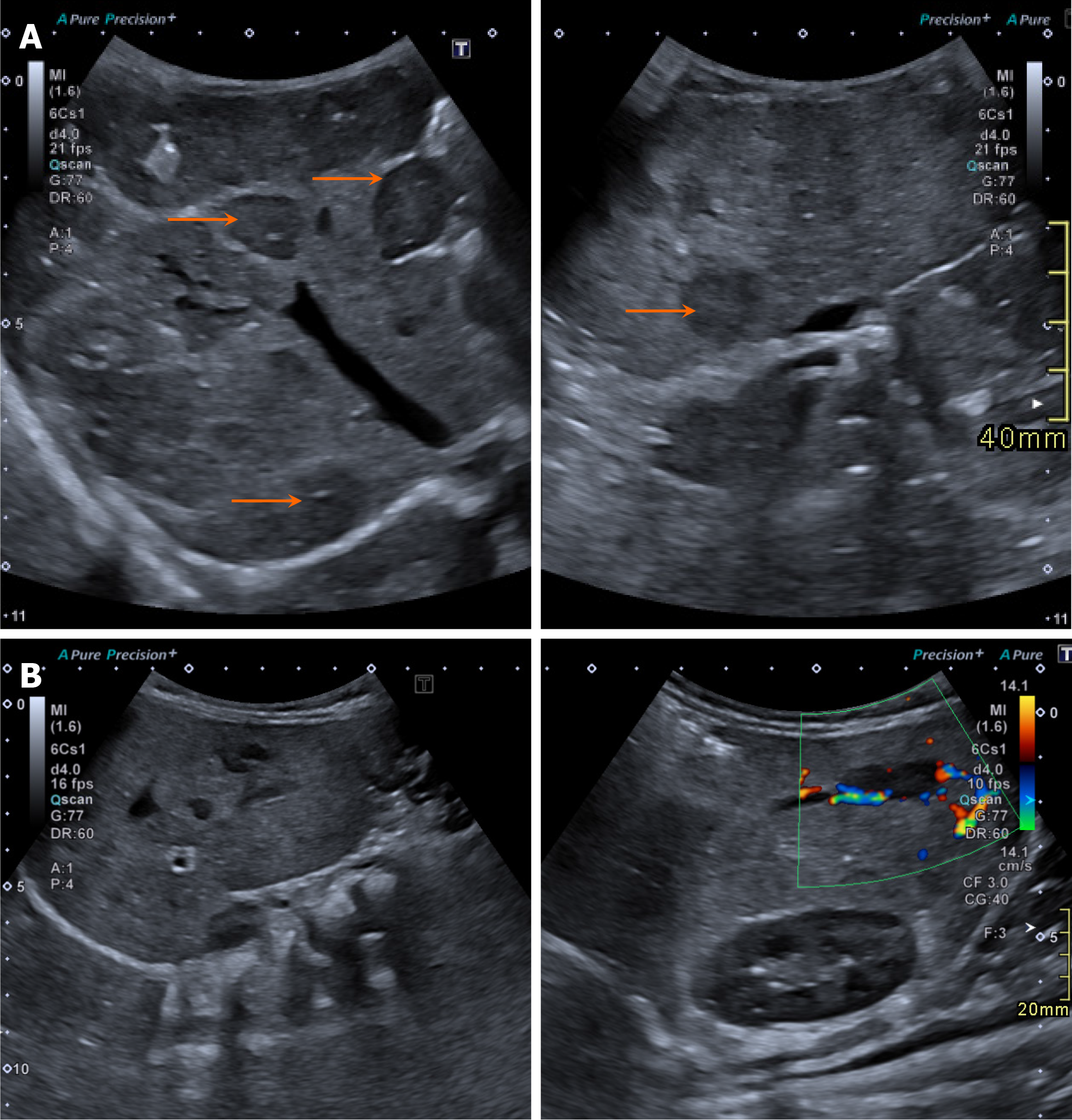
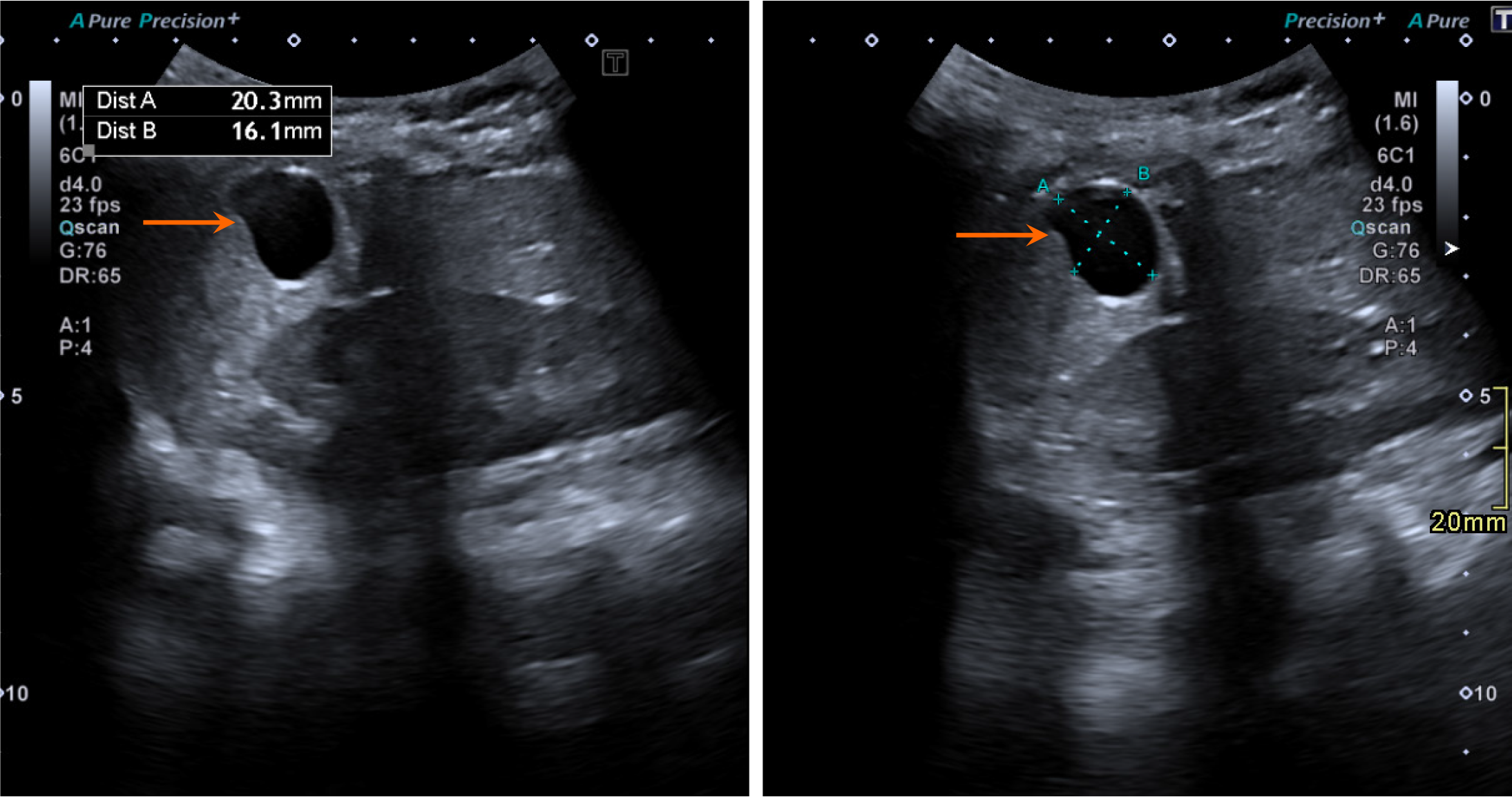
On ultrasonography, hepatoblastomas appear as solid masses with variable echogenicity and mild to moderate internal vascularity on color Doppler imaging (Figure 1). Cystic changes or a more heterogeneous appearance may be observed in cases of internal necrosis or hemorrhage.
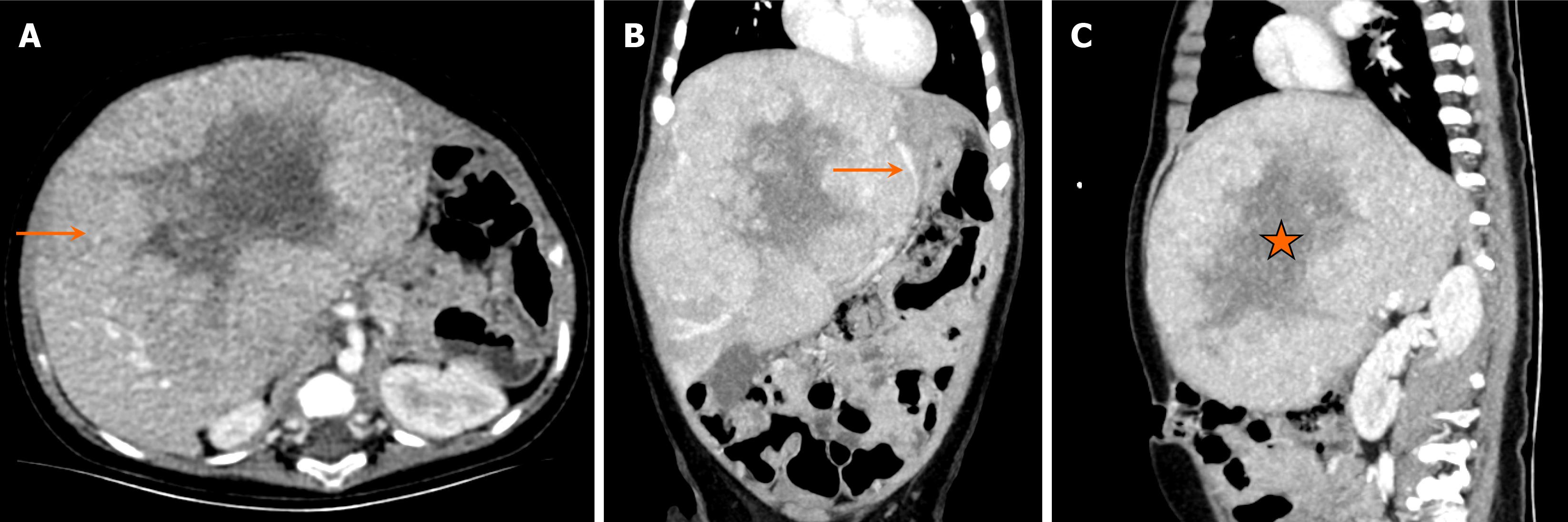
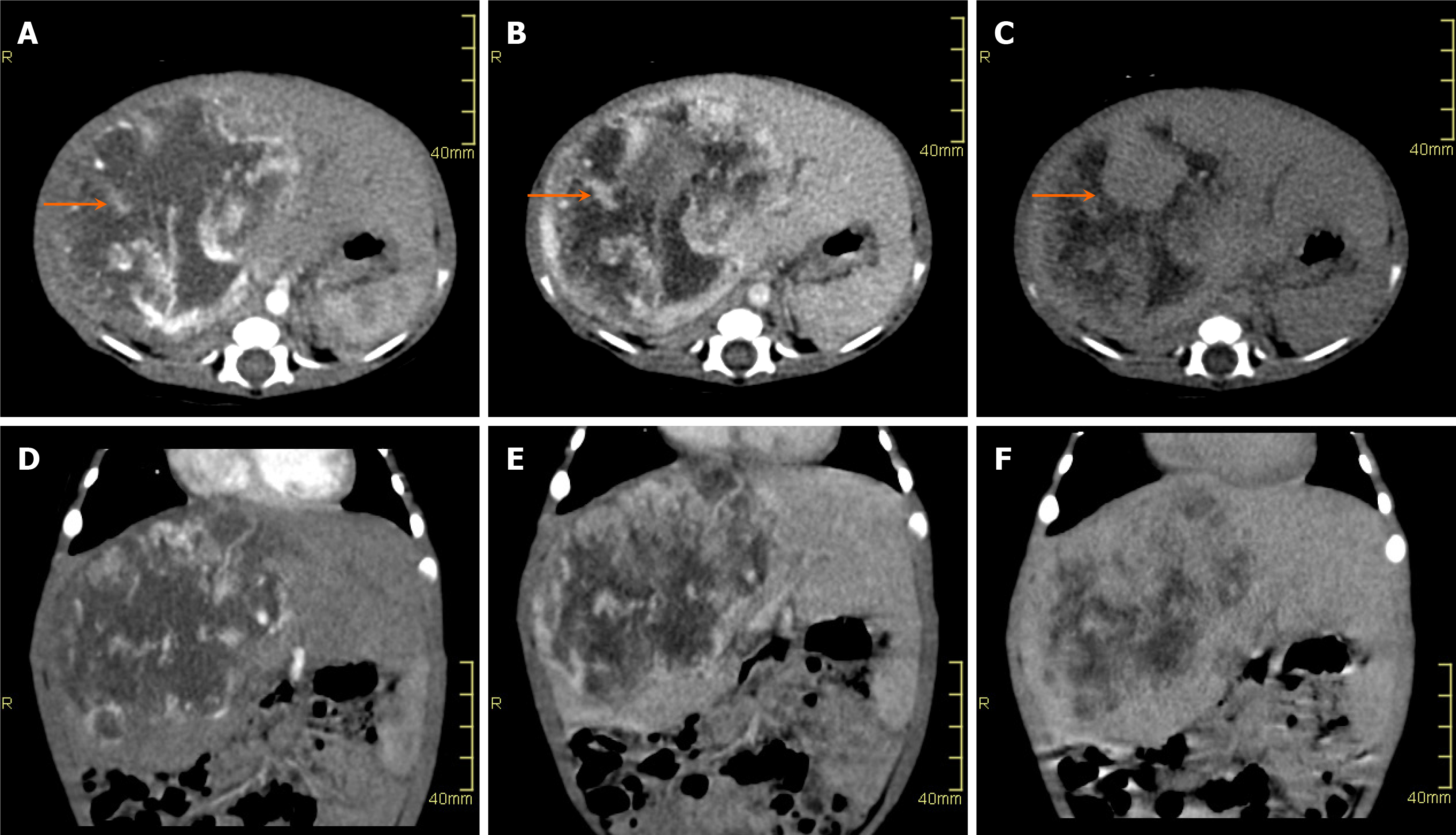
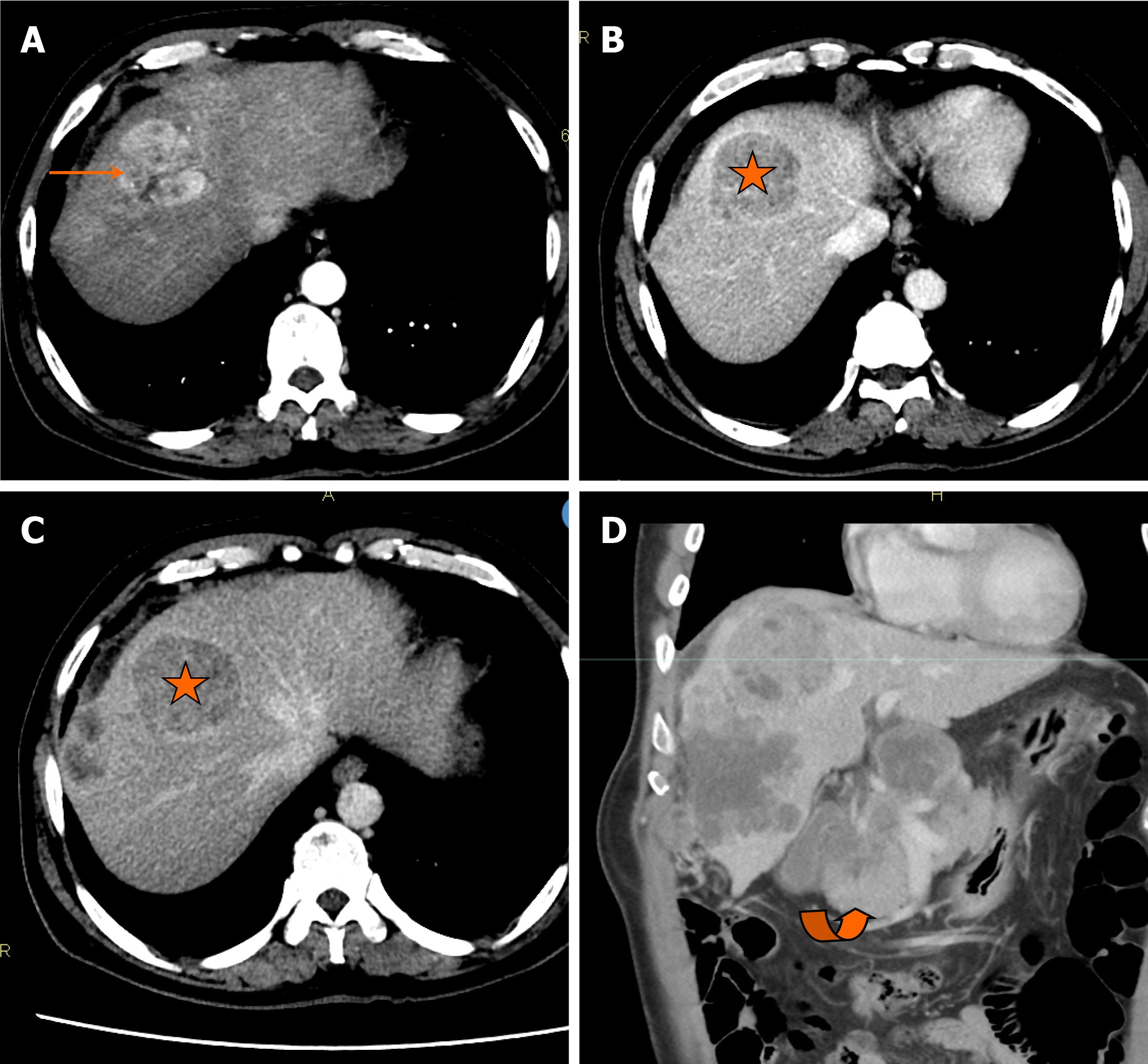
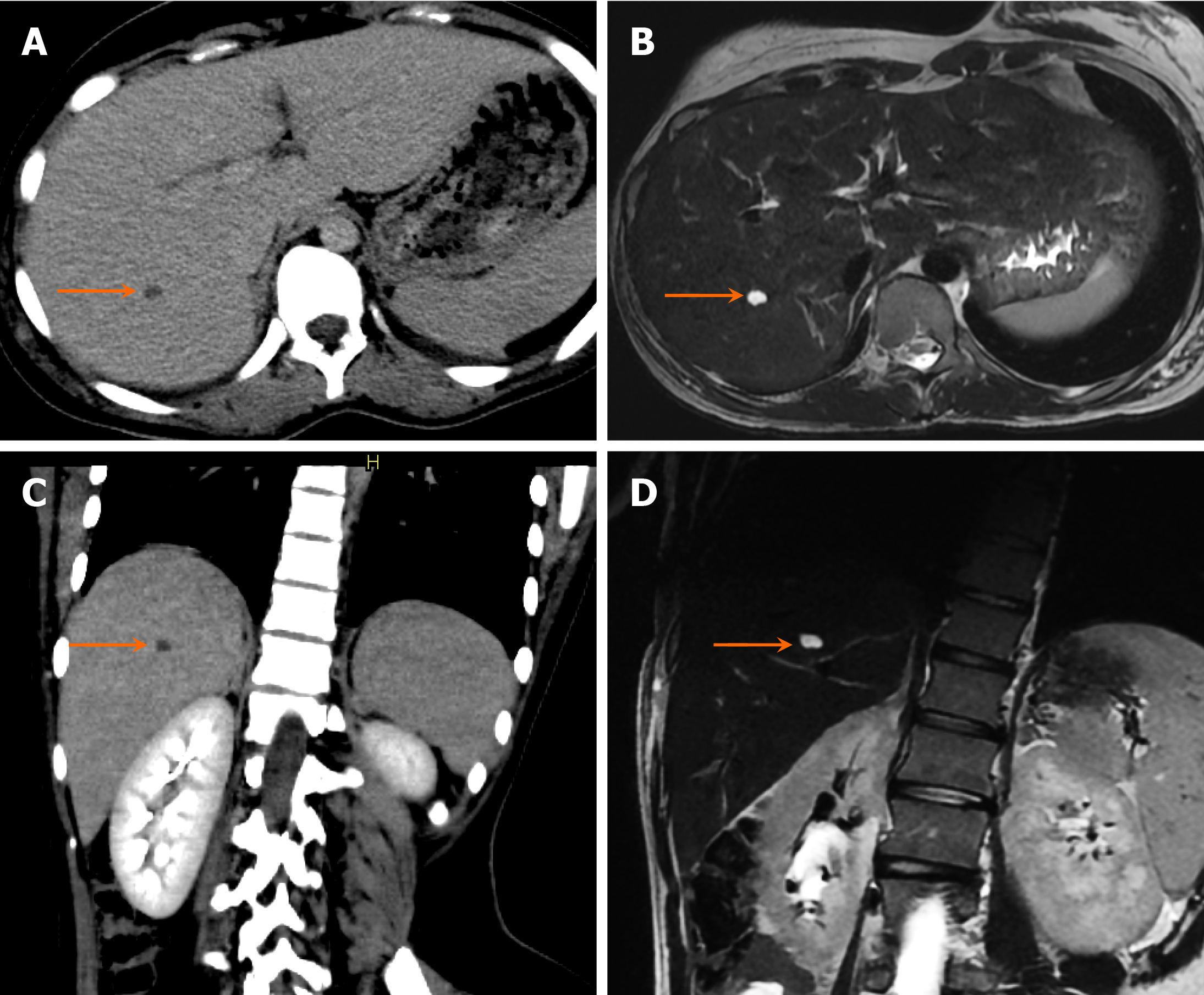
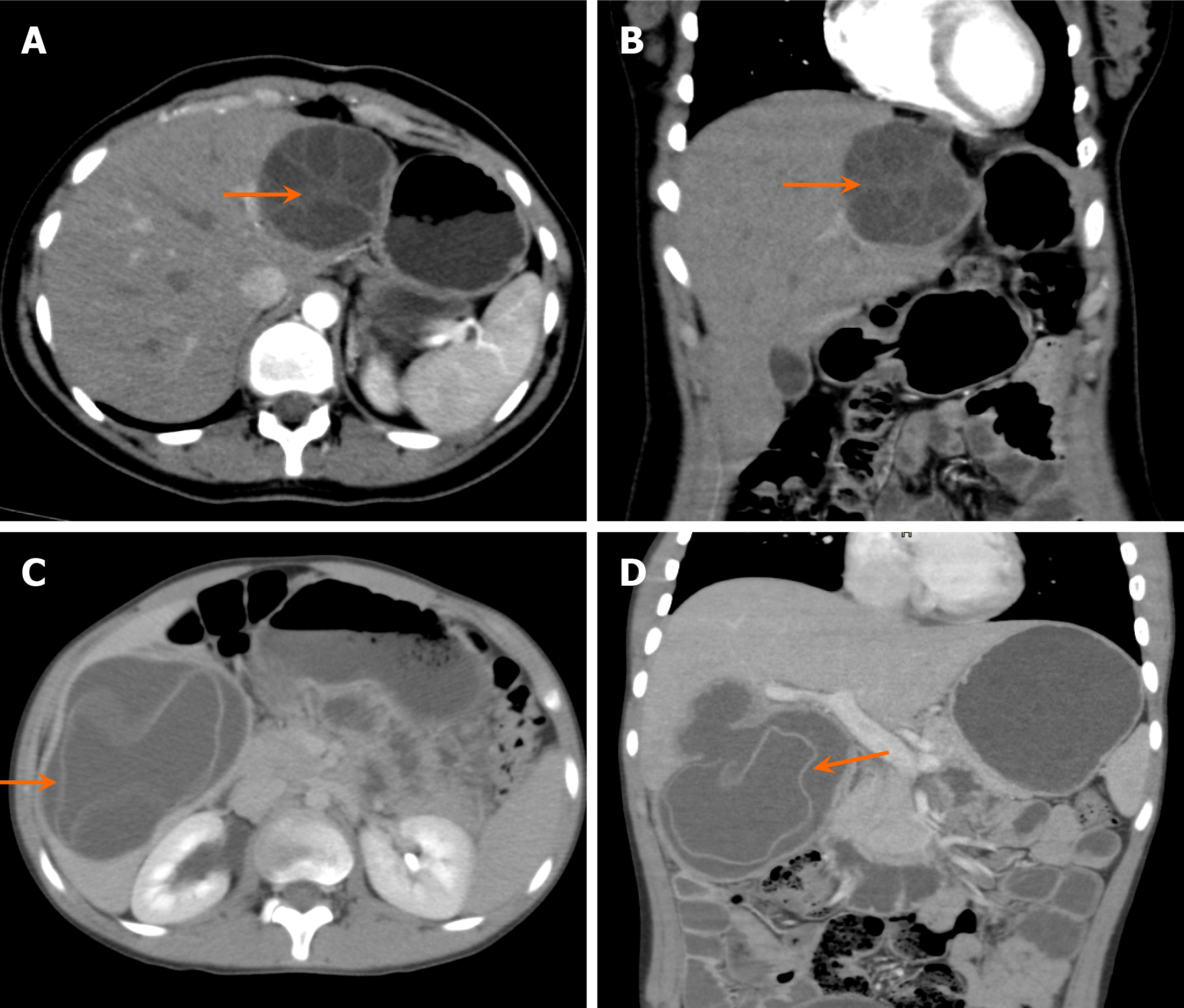
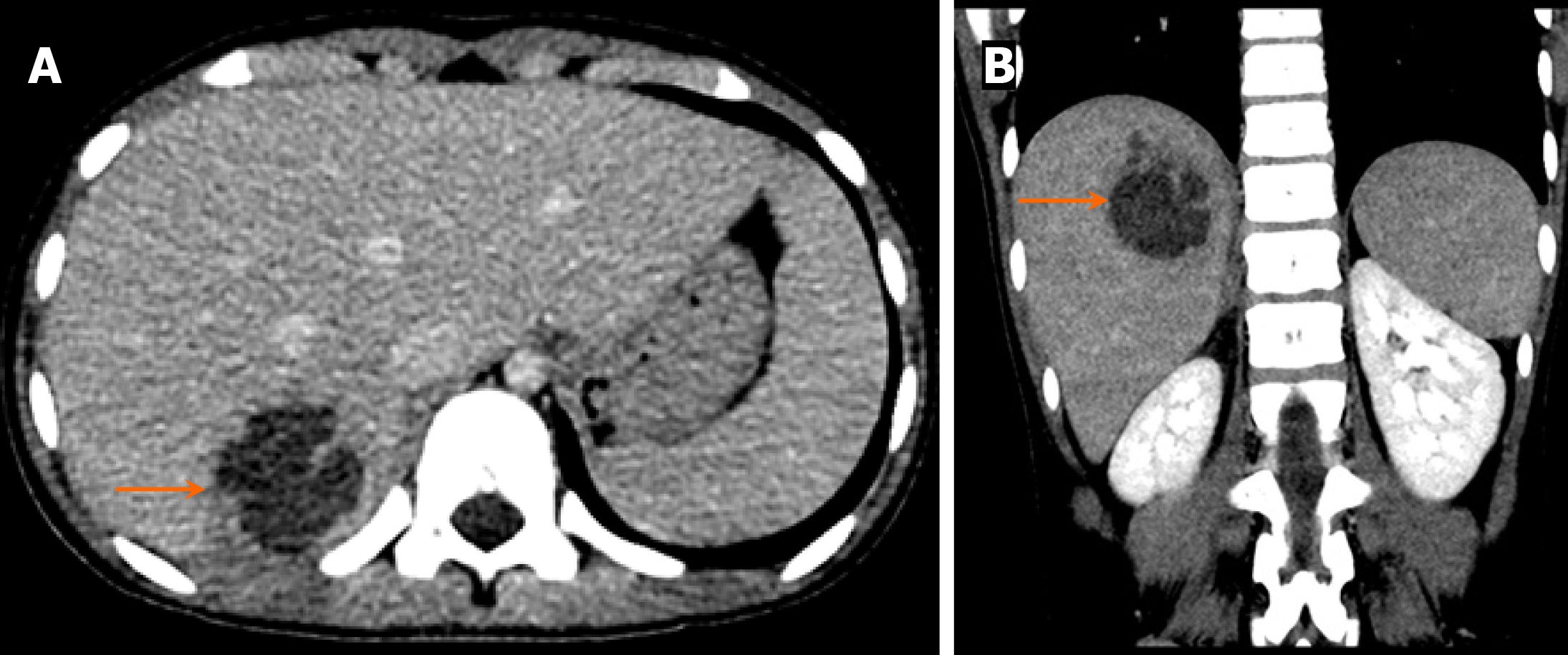
On CT, hepatoblastomas typically present as well-circumscribed heterogeneous lesions with internal areas of necrosis or hemorrhage (Figure 2). They usually demonstrate lower enhancement than normal liver parenchyma in all phases. Calcifications are observed in approximately 40%-50% of the cases.
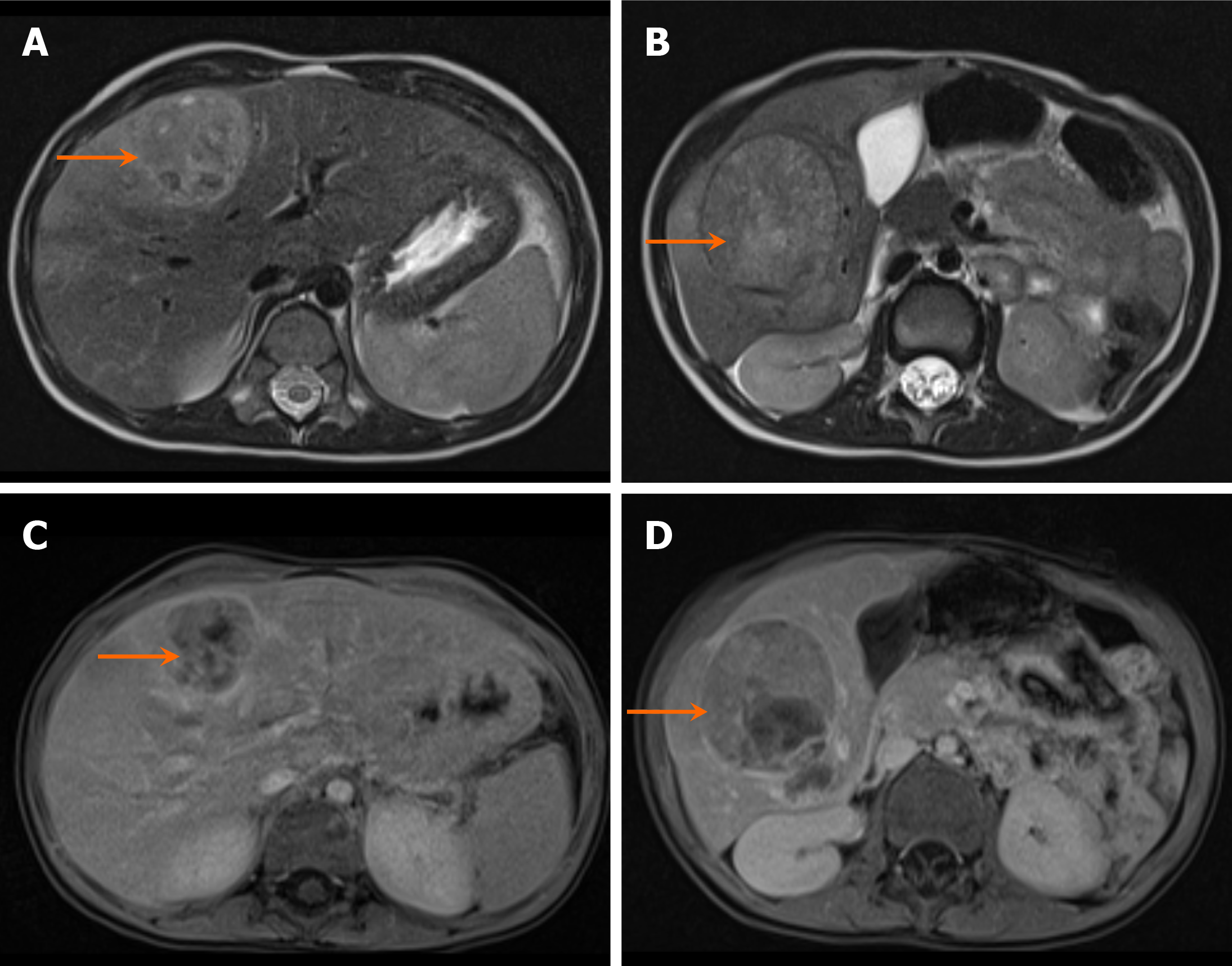
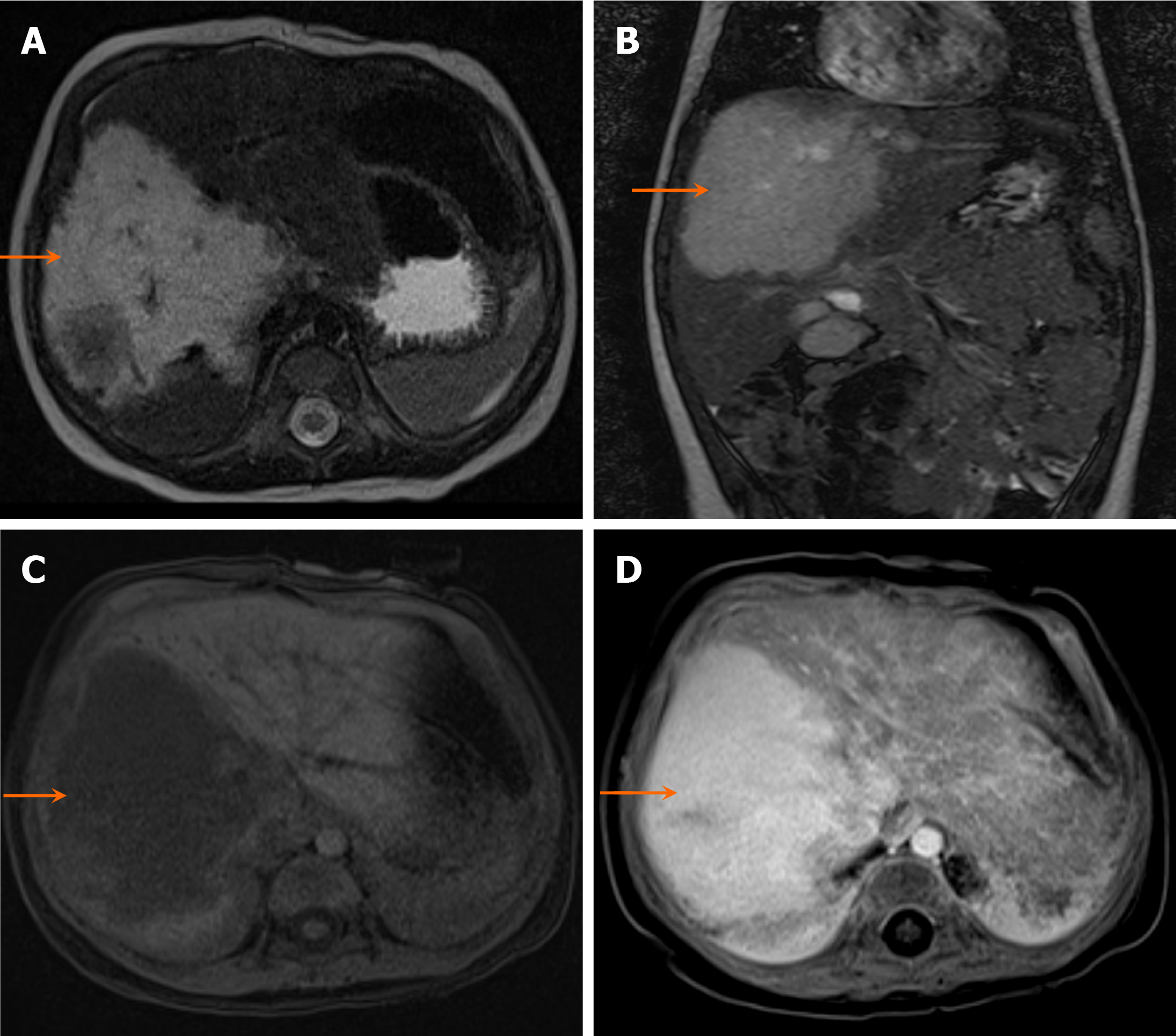
On MRI, hepatoblastomas generally appear hypointense on T1-weighted images, often with internal hyperintense foci secondary to hemorrhage, and hyperintense on T2-weighted images (Figure 3). Contrast-enhanced MRI with hepatobiliary phase imaging is generally recommended for the evaluation of pediatric hepatic tumors[4].
Staging of hepatoblastoma is performed via the pretreatment extent of disease (PRETEXT) system. This system is based on the involvement of four liver sections, rather than segments. The four sections include the left medial (segments IVa and IVb), left lateral (segments II and III), right anterior (segments V and VIII), and right posterior (segments VI and VII) sections (Table 1). The caudate lobe (C-positive disease) is considered separately, although a tumor confined to the caudate lobe is classified as PRETEXT II. Additional risk factors include vascular involvement of the hepatic veins and inferior vena cava (V-positive disease), portal vein thrombosis (P-positive disease), extrahepatic diaphragmatic or peritoneal spread (E-positive disease), multifocal disease (F-positive disease) or tumor rupture (R-positive disease). The most common sites of metastasis are the lymph nodes (N-positive disease) and lungs (M-positive disease)[5].
| Stage | Definition |
| PRETEXT I | Three contiguous hepatic sections free of tumor. Tumor can only be present in left lateral or right posterior sections |
| PRETEXT II | Involvement of either one or two sections, with no tumor seen in two contiguous sections. Involvement limited to the caudate lobe is also PRETEXT II disease |
| PRETEXT III | Involves portions of both right and left lobes of the liver with involvement of two or three sections. Only one contiguous section remains tumor free |
| PRETEXT IV | Mostly multifocal disease with involvement of all four sections |
Hepatic hemangioma, previously known as infantile hemangioendothelioma, is the most common benign vascular tumor of infancy, and typically develops within the first few weeks to months of life[6]. Hepatic hemangiomas are broadly categorized into congenital and infantile types.
Children with hepatic hemangiomas usually present with a palpable abdominal mass and high-output cardiac failure. Congenital hemangiomas, also referred to as focal hemangiomas, are typically discovered in the perinatal period and do not grow after birth. These lesions may be associated with anemia and consumptive coagulopathy. Congenital hema
Infantile hemangiomas, on the other hand, develop during the first few weeks of life and are categorized into focal, multifocal and diffuse subtypes[9]. Multifocal hemangiomas can present with congestive heart failure or hypothyroidism, whereas diffuse hemangiomas may cause massive hepatomegaly and hepatic failure[3]. Some authors suggest that multifocal hemangiomas may progress to the diffuse subtype, representing a spectrum of the same disease. Diffuse hepatic hemangiomas can lead to abdominal compartment syndrome, potentially resulting in multisystem organ failure[10].
Children with cutaneous hemangiomas are at increased risk of developing multifocal and diffuse hepatic hema
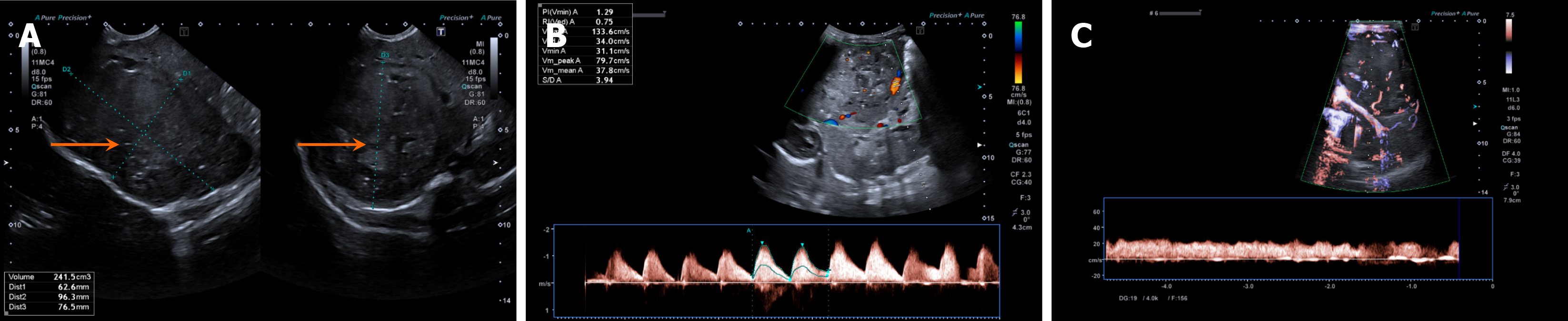
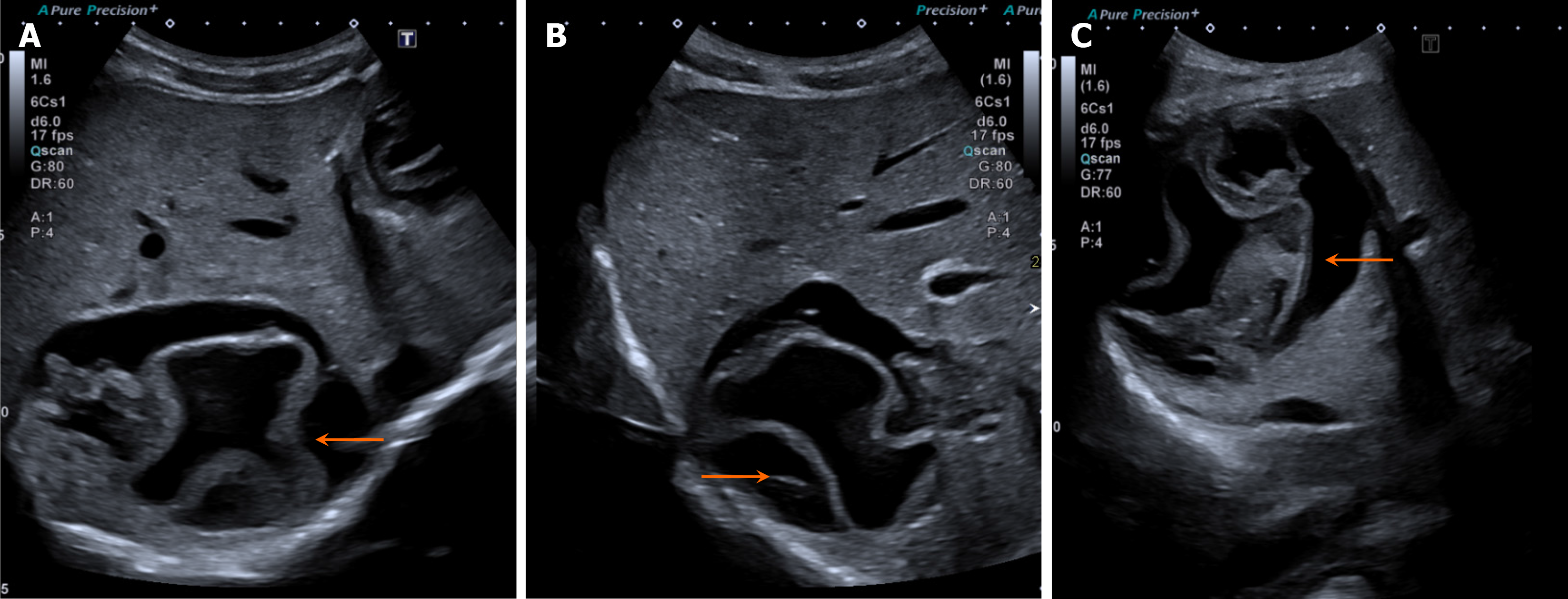
Congenital hepatic hemangiomas typically appear as solitary heterogeneous masses on US, with increased peripheral vascularity on color Doppler. Arteriovenous shunts may also be visualized. Infantile hemangiomas often present as hypoechoic solid masses with internal anechoic areas representing blood vessels. Color Doppler examination commonly reveals increased vascularity within the lesion, whereas pulsed Doppler may reveal a low-resistance arterial waveform during the proliferative phase (Figure 4)[12]. Hepatic artery velocities are usually elevated during proliferation and decrease during involution.
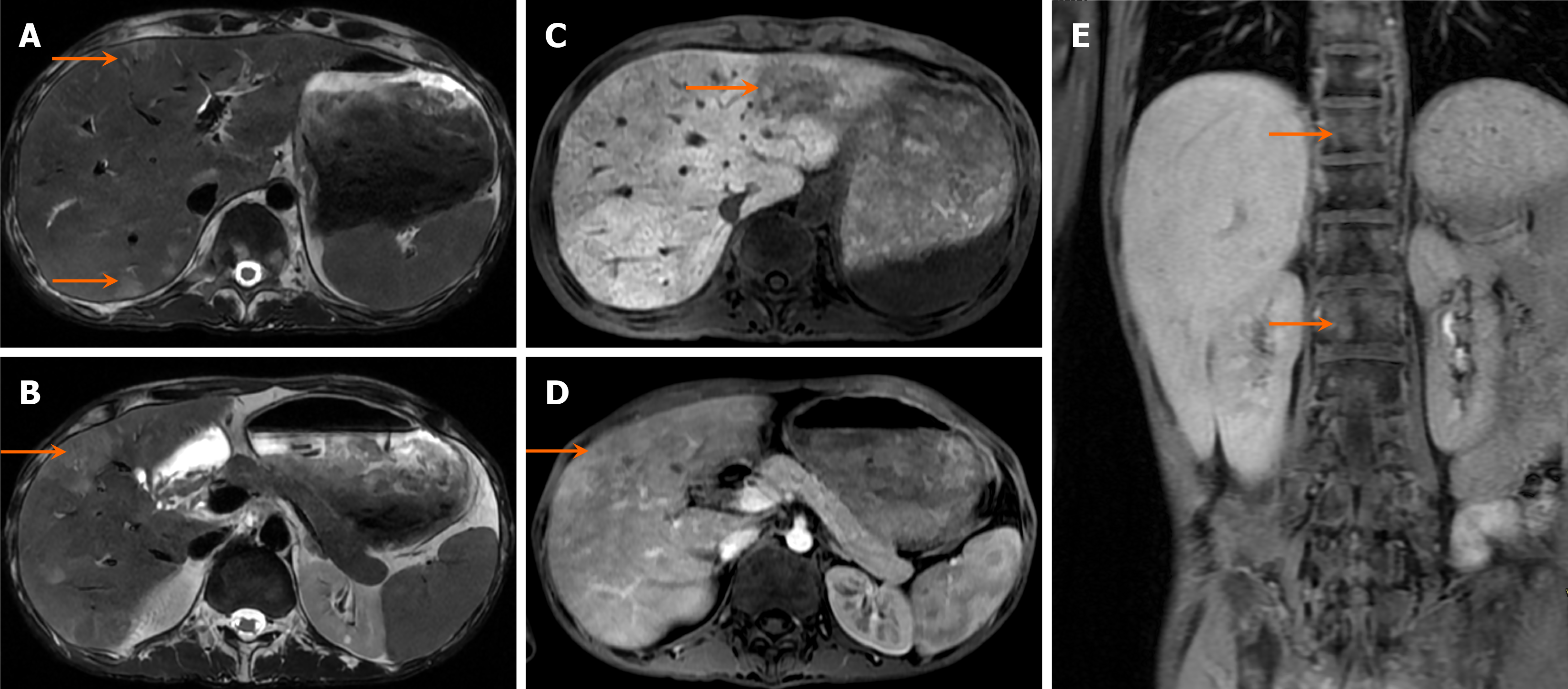
On CT, congenital hemangiomas typically appear as hypodense masses with nodular peripheral enhancement and central hypodense areas, demonstrating progressive fill-in on delayed imaging (Figure 5). Infantile hemangiomas may also appear hypodense, showing peripheral enhancement during the early arterial phase, with central enhancement in the portal venous and delayed phases. A possible differentiating feature between congenital and infantile hemangiomas may be calcifications, which are often present in congenital hemangiomas but almost never present in infantile hema
On MRI, both congenital and infantile hemangiomas are generally hypointense on T1-weighted images and hyperintense on T2-weighted images. Congenital hemangiomas may show central areas of hyperintensity on T1-weighted images, indicating hemorrhage. Compared with the surrounding hepatic parenchyma, infantile hemangiomas typically appear hyperintense. The enhancement pattern may, however, be variable with some lesions demonstrating avid homogeneous enhancement (Figure 6).
MR angiography and venography may reveal enlargement of the abdominal aorta, celiac axis, hepatic arteries, hepatic veins, and inferior vena cava. However, owing to the increased vascular supply to the lesions, a reduction in the caliber of the abdominal aorta distal to the celiac artery origin—known as mid-aortic syndrome—is often observed[13].
Oral propranolol has been shown to be effective in the management of infantile hemangiomas (Figure 7). A randomized controlled trial conducted in 2015 reported regression of the lesion in 88% of patients receiving propranolol, although side effects such as bradycardia and hypoglycemia were reported[14].
Mesenchymal hamartomas are benign cystic hepatic tumors, that are often considered developmental anomalies rather than true neoplasms. They are typically detected in children under two years of age and are twice as common in males[9].
Patients with mesenchymal hamartomas usually present with an enlarging abdominal mass. In neonates, the presentation may be acute due to the rapid growth of the lesion. Large mesenchymal hamartomas can cause massive abdominal distension, potentially leading to respiratory distress or apnea. AFP levels are typically normal in children with these cystic lesions.
On US, mesenchymal hamartomas commonly appear as large, multiseptated cystic masses with both cystic and solid components. Internal vascularity may be observed on color Doppler imaging (Figure 8). On CT, they typically present as low attenuation, multiseptated lesions with enhancing septations, creating a "Swiss cheese" appearance.
On MRI, mesenchymal hamartomas appear as hyperintense cystic structures on T2-weighted images with hypointense intervening stroma. T1 signal intensity varies depending on the amount of stromal elements and proteinaceous content within the lesion[15]. Multifocality is rare in mesenchymal hamartomas. Table 2 provides a summary of hepatic masses in neonates and children up to six years of age.
| Lesion | Age | AFP | Calcifications | Imaging appearance | Quantitative metrics |
| Hepatoblastoma | < 5 years (peak: 6 months-3 years) | Markedly elevated | Chunky calcifications commonly seen in 50% of cases | US: Heterogeneous, solid mass, may have necrosis and calcifications. CT: Enhancing soft tissue with coarse calcifications. MRI: Isoechoic/hypoechoic on T1, hyperintense on T2, with heterogeneous enhancement | Size: > 5 cm at diagnosis. AFP: |
| Hepatic hemangioma | Neonates and infants ( | Normal or mildly elevated (< 100 ng/mL). Limited diagnostic value | Rare. May be present in the congenital forms of hemangioma but absent in the infantile type | US: Hypoechoic or mixed echotexture, high vascularity with Doppler. CT/MRI: Vascular lesion with peripheral enhancement and centripetal fill-in | Size: Ranges from small (< 3 cm) to giant (> 5 cm). May cause high-output cardiac failure in giant hemangiomas |
| Mesenchymal hamartoma | < 2 years (typically | Normal | Rare | US and CT: Multiseptated cystic lesion interspersed with solid component. Minimal enhancement | Size: Often large |
Hepatocellular carcinoma (HCC) is a neoplasm that typically occurs in adolescents and adults. It is less common in children; however, when present, it tends to be more extensive and is associated with a poor prognosis. HCC generally presents in children older than 12 years of age but is more common than hepatoblastoma in children over six years of age. Risk factors for the development of HCC in children include chronic liver disease secondary to viral hepatitis, progressive familial intrahepatic cholestasis, galactosemia, glycogen storage disease type 1, and hereditary tyrosinemia[9].
Patients with HCC typically present with an abdominal mass and pain, which may radiate to the shoulder or back, along with jaundice and weight loss. On physical examination, the liver is often hard and nodular upon palpation. AFP levels are invariably elevated in children with HCC.
Fibrolamellar HCC is a distinct subtype of HCC, that is more common in younger patients without underlying liver disease[3]. This variant is associated with paraneoplastic syndromes and does not typically present with elevated AFP levels.
On grayscale US, HCC typically exhibits mixed echogenicity, with hypoechoic areas representing solid tumor components and hyperechoic areas indicating fatty changes. If encapsulated, the lesion may display a thin, surrounding hypoechoic band. Color Doppler imaging generally reveals increased vascularity.
On CT, HCC demonstrates early arterial enhancement followed by washout on venous and delayed phase images (Figure 9). The tumor may also show areas of necrosis, fat components, and internal calcifications. On MRI, HCC is usually hyperintense on T2-weighted images, whereas T1-weighted images demonstrate variable signal intensity depending on the fat or blood content within the lesion. Dynamic postcontrast MRI shows arterial enhancement with washout on venous and delayed phases, a characteristic feature of HCC[16].
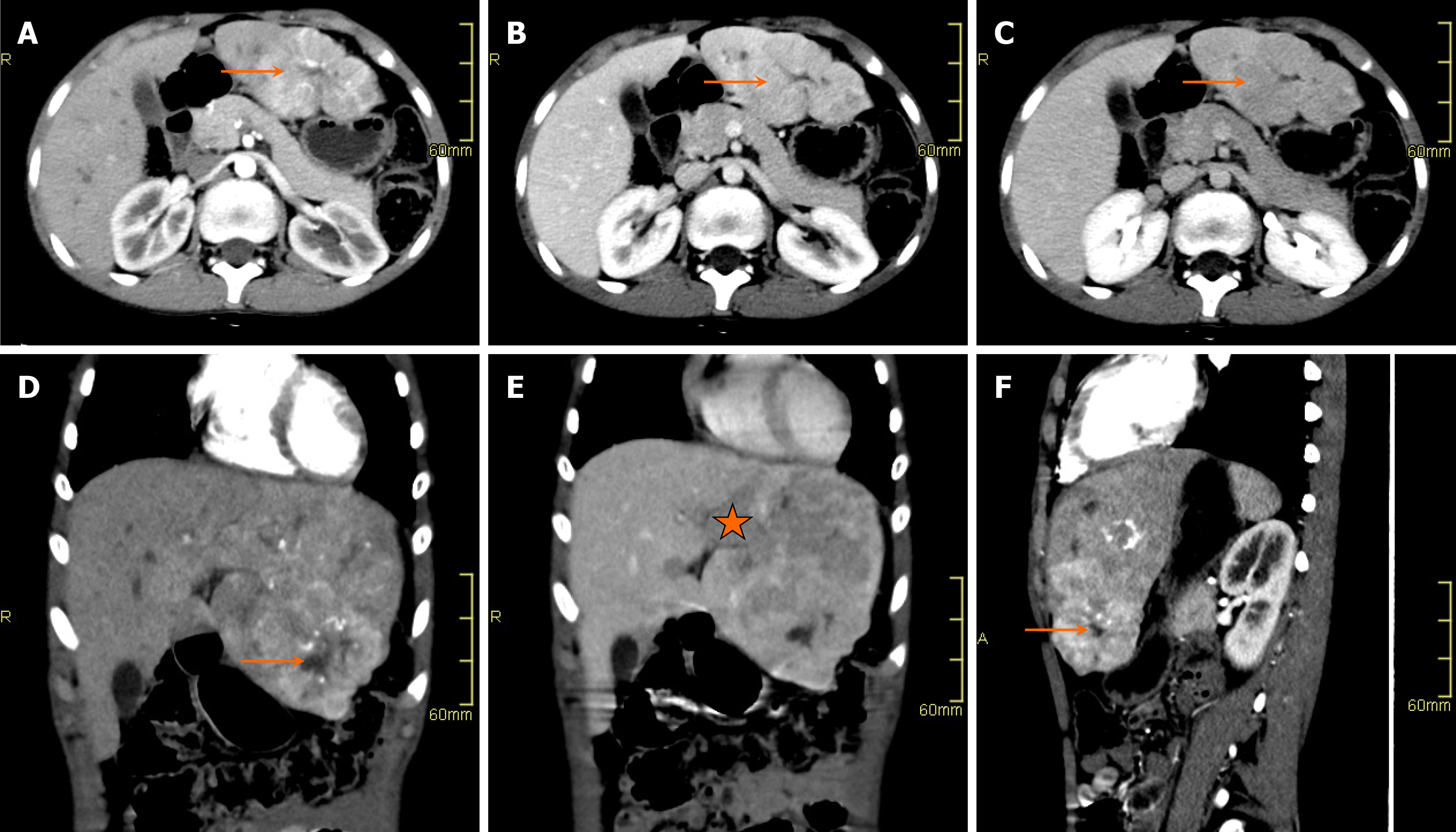
Fibrolamellar HCC typically appears as a well-defined lobulated lesion with a central hyperechoic scar on grayscale US. On CT, it presents as a mass with a central hypodense scar on unenhanced images. Calcifications and necrosis may be observed in approximately 70% of cases. On the arterial phase, fibrolamellar HCC may demonstrate hyperattenuation but can appear heterogeneous, with iso-attenuation to hypoattenuation on portal venous and delayed phases (Figure 10). The central scar often enhances on delayed-phase imaging. Biliary or vascular invasion, as well as nodal and lung metastases, may also be observed.
On MRI, fibrolamellar HCC typically shows hypointense signals on T1-weighted images and hyperintense signals on T2-weighted images, with the central scar appearing hypointense on both sequences. Arterial and portal venous phases usually reveal heterogeneous enhancement of the tumor, whereas the scar shows no enhancement on the arterial phase, partial enhancement on the portal venous phase, and complete enhancement on delayed phase imaging[17].
Focal nodular hyperplasia (FNH) is a benign hyperplastic parenchymal nodule with abnormal vasculature. It is more common in adults, with a female predilection, but can also be observed in children with chronic liver diseases such as portal vein agenesis/hypoplasia and Budd-Chiari syndrome[18,19].
FNH is usually asymptomatic and is often discovered incidentally. Abdominal pain and distension are common presenting complaints in symptomatic patients. Necrosis, hemorrhage, and rupture are rare[3]. Children with extra
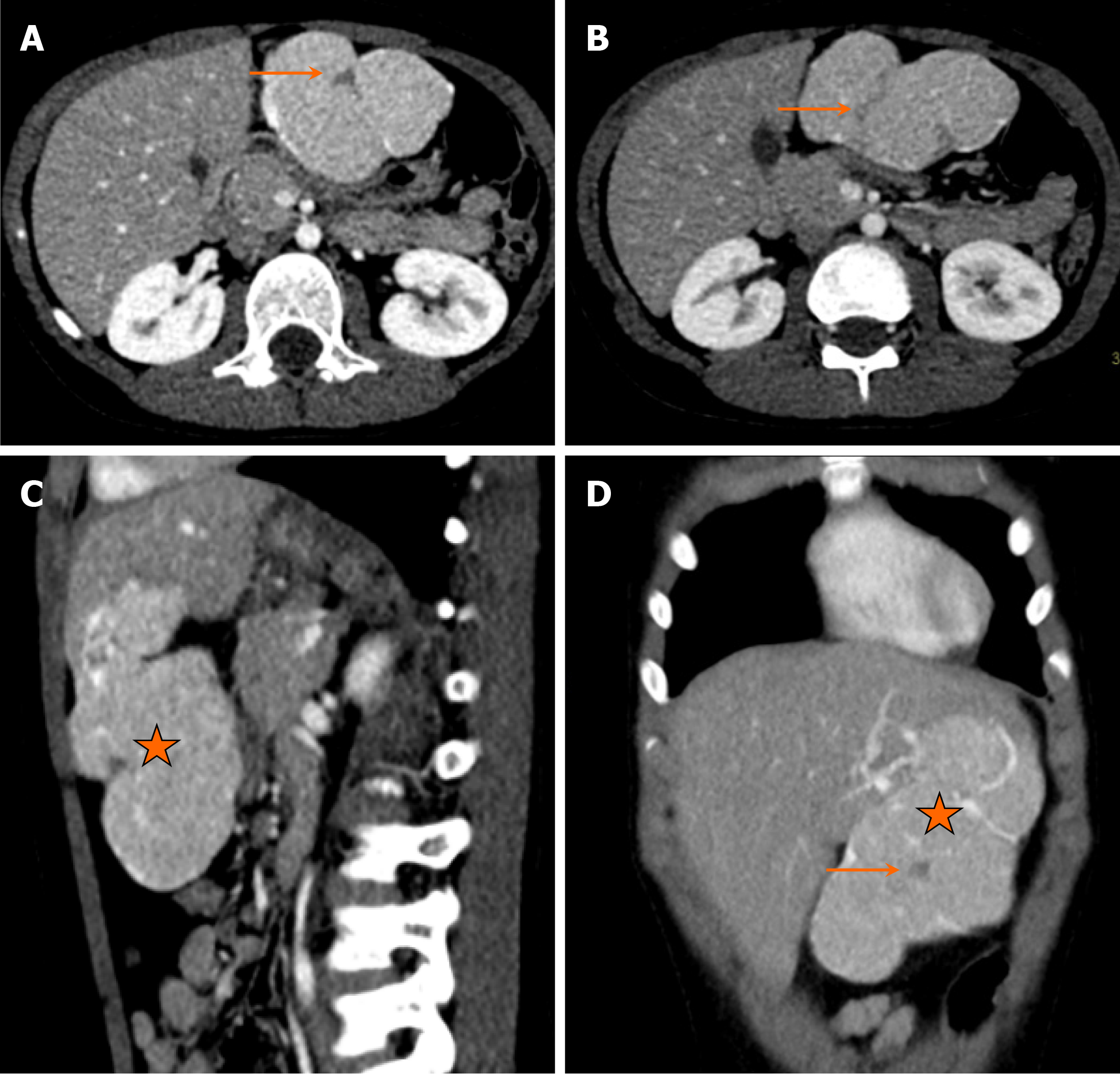
On US, FNH typically appears as a well-circumscribed, solid, isoechoic hepatic mass with a central scar. The "spoke-wheel" pattern of vascularity can be demonstrated on color Doppler imaging.
On CT, FNH commonly shows arterial phase enhancement with a hypodense central scar. The lesion becomes isodense to the liver parenchyma on portal venous and delayed phase imaging, with the central scar potentially showing enh
On MRI, the lesion and central scar are usually hypointense on T1-weighted images and hyperintense on T2-weighted images. Postcontrast images typically demonstrate arterial phase hyperenhancement, isointensity on venous phase, and greater enhancement of the central scar on delayed phase imaging[20]. On hepatobiliary phase imaging, the lesion may show mild enhancement, with relatively lower enhancement of the scar.
Nuclear scintigraphy via sulphur colloid and hepatobiliary iminodiacetic acid (HIDA) scans can assist in diagnosing FNH. Sulphur colloids are absorbed by the reticuloendothelial system, and normal Kupffer cell function in FNH results in normal or increased radiotracer uptake, distinguishing it from lesions with impaired hepatocellular function[21]. On HIDA scans, increased blood flow, rapid tracer uptake, and delayed clearance from the lesion are characteristic findings of FNH.
Hepatic adenomas (HAs) are benign tumors derived from hepatocytes. Risk factors for HA formation in children include anabolic steroid use, diabetes mellitus, and glycogen storage disease[21,22]. HAs are more common in females.
Patients typically present with right upper quadrant pain, which may be due to hemorrhage. liver function tests (LFTs) are often elevated in such cases. However, in many instances, the lesions are discovered incidentally.
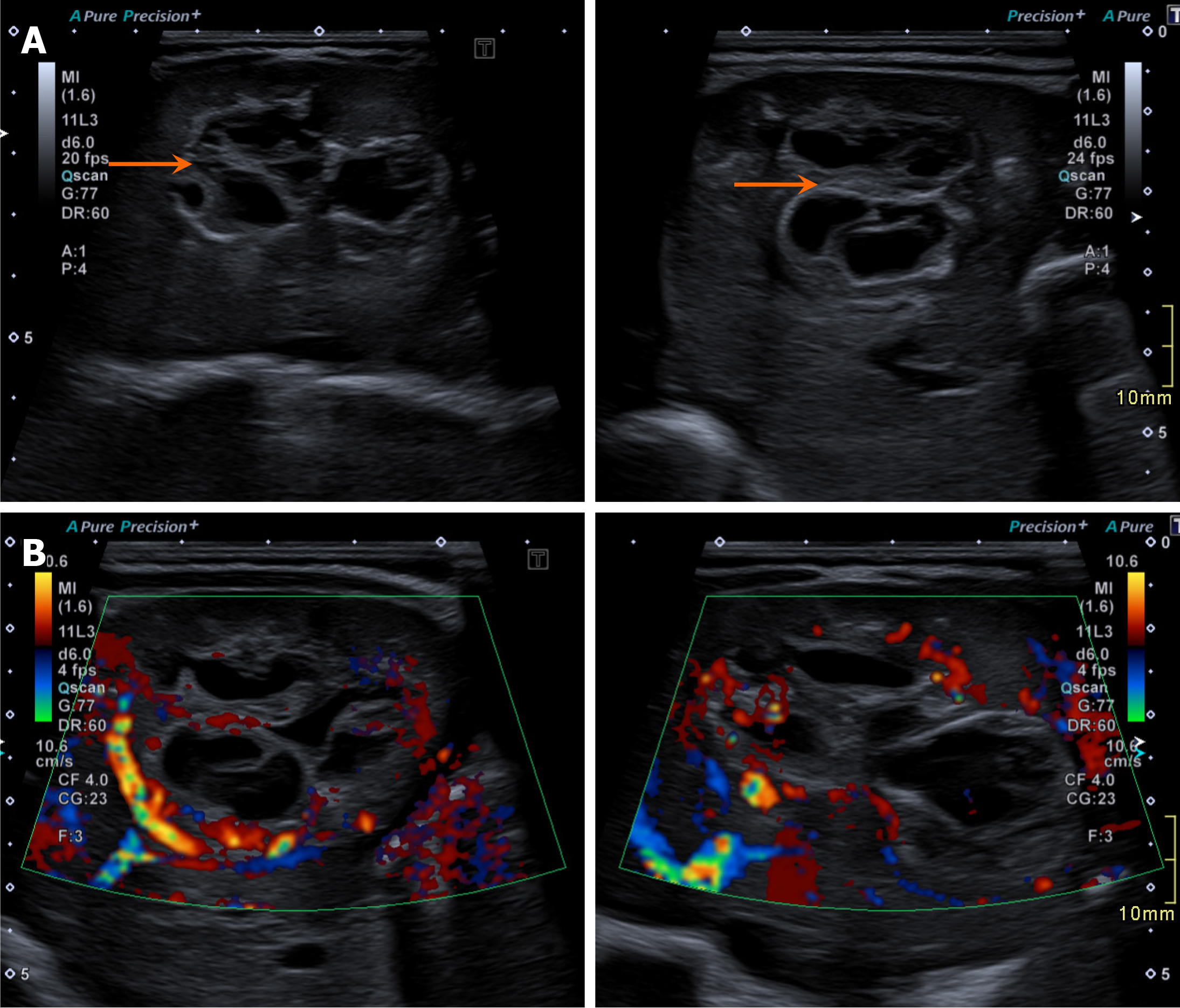
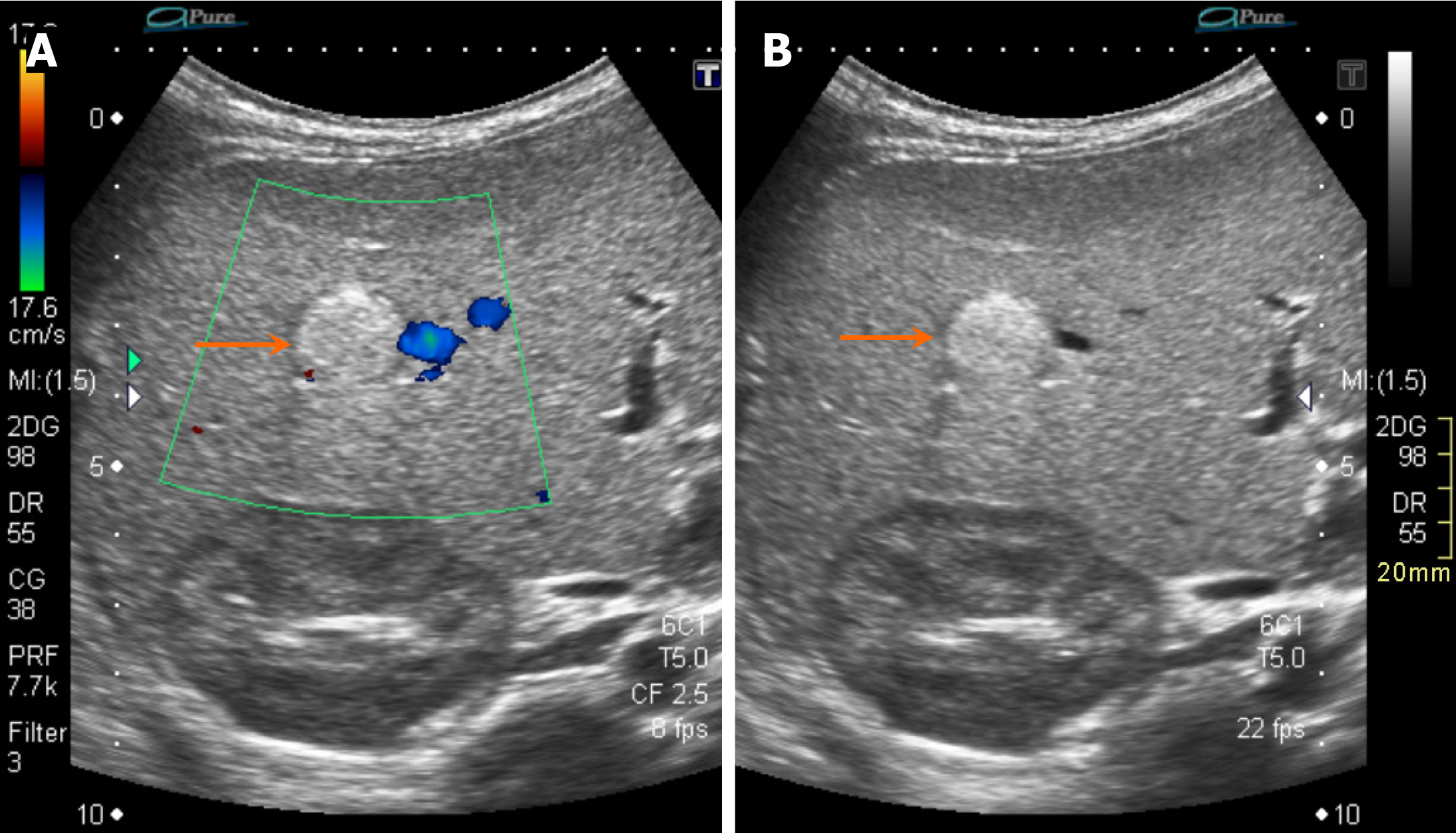
On grayscale US, HA usually appears as a well-defined, lobulated lesion with mixed echogenicity. Intraperitoneal or intratumoral hemorrhage is a potential acute complication. On color Doppler, HA may show increased vascularity with large peripheral arteries and veins, although some lesions may lack internal vascularity (Figure 12). The hepatic artery can often be identified as the arterial supply, and intratumoral veins may occasionally be observed, unlike in FNH.
On CT, HAs are typically hypodense on unenhanced imaging, with heterogeneous arterial-phase enhancement and hypoenhancement on the delayed phase.
On MRI, HAs demonstrate isoechoic to hyperintense signals on T1-weighted images and hyperintense signals on T2-weighted images. Signal intensity varies in cases of internal hemorrhage. The "Atoll sign", characterized by a rim of mild T2 hyperintensity, may be observed in the inflammatory subtype[23]. Postcontrast images typically show arterial-phase hyperenhancement with variable signal intensity on the portal venous phase. Loss of signal on out-of-phase images due to increased lipid content can also be noted.
On fluorodeoxyglucose positron emission tomography (FDG-PET) imaging, HAs generally appear intensely hypermetabolic, mimicking metastases or other aggressive lesions. Table 3 shows a summary of hepatic masses in children aged six years and older.
| Age | Gender | Alpha-fetoprotein | Imaging appearance | Quantitative metrics | |
| Hepatocellular carcinoma | > 6 years of age (bimodal peak: 10-14 years) | No gender predilection | Elevated in 50%-70% (variable; can be | US: Heterogeneous mass, may show necrosis. CT/MRI: Arterial phase enhancing lesion with washout on portal/delayed phases | Size: > 5 cm at presentation. Occasional calcifications (approximately 20%). Risk factors: Chronic liver disease, metabolic disorders (e.g., tyrosinemia) |
| Focal nodular hyperplasia | > 6 years (mean age of 12 years) | More common in females | Normal | US: Isoechoic to hypoechoic; spoke wheel pattern of vascularity. CT and MRI: Enhancing lesion with central scar. T2 hyperintense scar, arterial enhancement with delayed central scar fill-in | Size: < 5 cm. Central scar: Present in approximately 80%. Calcification is rare |
| Hepatic adenoma | > 6 years (mean age of 12 years) | More common in females | Normal | US: Solid, hyperechoic or isoechoic. CT: Arterial enhancement, isoechoic/hypodense in venous phase. MRI: Variable T1/T2, early enhancement, no central scar. May show fat or hemorrhage | Size: Variable; > 5 cm increases risk of hemorrhage. Risk of rupture/malignant transformation in β-catenin-mutated subtype. Risk factors: Oral contraceptive pills or steroid use |
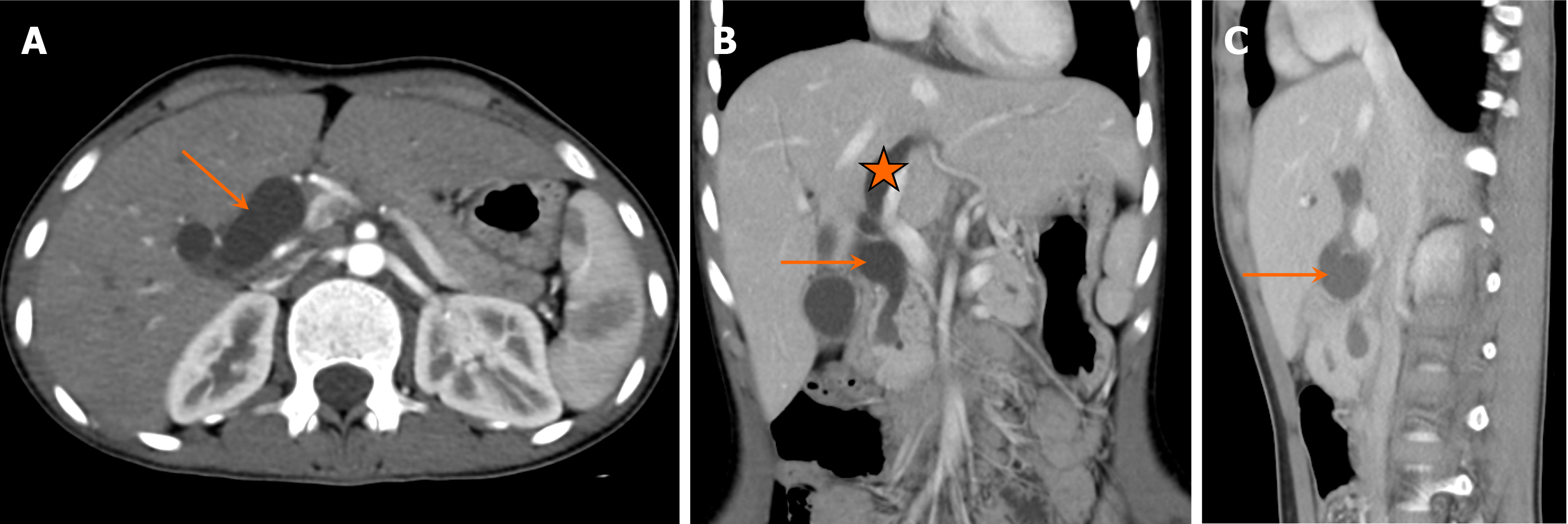
A simple hepatic cyst is a benign, fluid-filled structure that typically arises from the biliary ducts without communication with the biliary system. These cysts are lined by a single layer of cuboidal bile duct epithelium surrounded by fibrous tissue and may be solitary or multiple. If more than 10 cysts are present, conditions such as adult polycystic liver disease, adult polycystic kidney disease, and biliary hamartomas should be considered[24].
Hepatic cysts are usually discovered incidentally. However, if complications arise, they may cause abdominal pain and/or nausea. Large cysts can exert a mass effect on surrounding structures, leading to symptoms. Abdominal pain often results from capsular stretching[25]. In cases where the mass effect significantly obstructs the biliary channels, the patient may present with jaundice. Simple hepatic cysts can occur in any age group but are more common in females. Multiple cysts can be observed in cases of fibropolycystic liver disease or phakomatoses such as von Hippel-Lindau disease[26].
On grayscale US, a simple hepatic cyst appears as a well-defined, anechoic structure with thin walls. Septations may be present but are typically thin and sometimes imperceptible. Calcifications or mural nodules are usually absent. Characteristic posterior acoustic enhancement is observed (Figure 13). Simple hepatic cysts generally occupy a single liver segment without crossing into another segment. If the cyst is hemorrhagic or infected, internal debris may be observed. On color Doppler imaging, no internal vascularity is demonstrated.
On unenhanced CT, a hepatic cyst appears as a well-defined, hypodense lesion with thin, smooth walls, without evidence of intramural nodules or calcifications. These lesions typically have a CT density between -10 and +10 Hounsfield units. No or barely perceptible septations may be observed (Figure 14). If the cyst is complicated by hemorrhage or infection, layering debris will be visible along its dependent wall. On postcontrast imaging, simple hepatic cysts show no enhancement.
On MRI, simple hepatic cysts appear hypointense on T1-weighted images and uniformly hyperintense on T2-weighted images (Figure 14). These cysts do not communicate with the biliary system, which can be best evaluated using magnetic resonance cholangiopancreatography (MRCP). In the case of a complicated cyst, variable signal intensity may be observed on both T1-weighted sequences and T2-weighted sequences[27].
A choledochal cyst forms due to a malformation involving the intrahepatic and extrahepatic bile ducts. Dilatation of the biliary tree may be focal or diffuse. Patients with choledochal cysts have an increased risk of developing cholangiocarcinoma, and some studies suggest that this risk persists even after cyst excision[28].
Neonates with choledochal cysts often present with prolonged cholestasis. Infants may exhibit jaundice, acholic stools, hepatomegaly, or a palpable abdominal mass. Two-thirds of cases are diagnosed before the age of 10 years. There is a higher incidence of choledochal cysts in females. The Todani classification is used to categorize choledochal cysts into six groups (Table 4)[29].
| Todani classification | Subtypes | |
| Type I | Fusiform dilatation of the extrahepatic biliary ducts | Ia: Dilatation of entire extrahepatic bile duct. Ib: Focal segmental dilatation of extraheptic bile duct. Ic: Dilatation of the common bile duct |
| Type II | Saccular outpouching from the intra and extrahepatic bile duct, also known as bile duct diverticulum | |
| Type III | Protrusion of a focally dilated intramural segment of the common bile duct into the duodenum, also known as choledochocele | |
| Type IV | Communicating intra and extrahepatic duct cysts | IVa: Dilatation of the entire extrahepatic bile duct along with dilatation of the intrahepatic bile ducts. IVb: Cystic dilatation involving only the extrahepatic bile duct |
| Type V | Cystic dilatation of intrahepatic bile ducts, also known as Caroli disease | |
| Type VI | Dilatation of only the cystic duct, a rare entity |
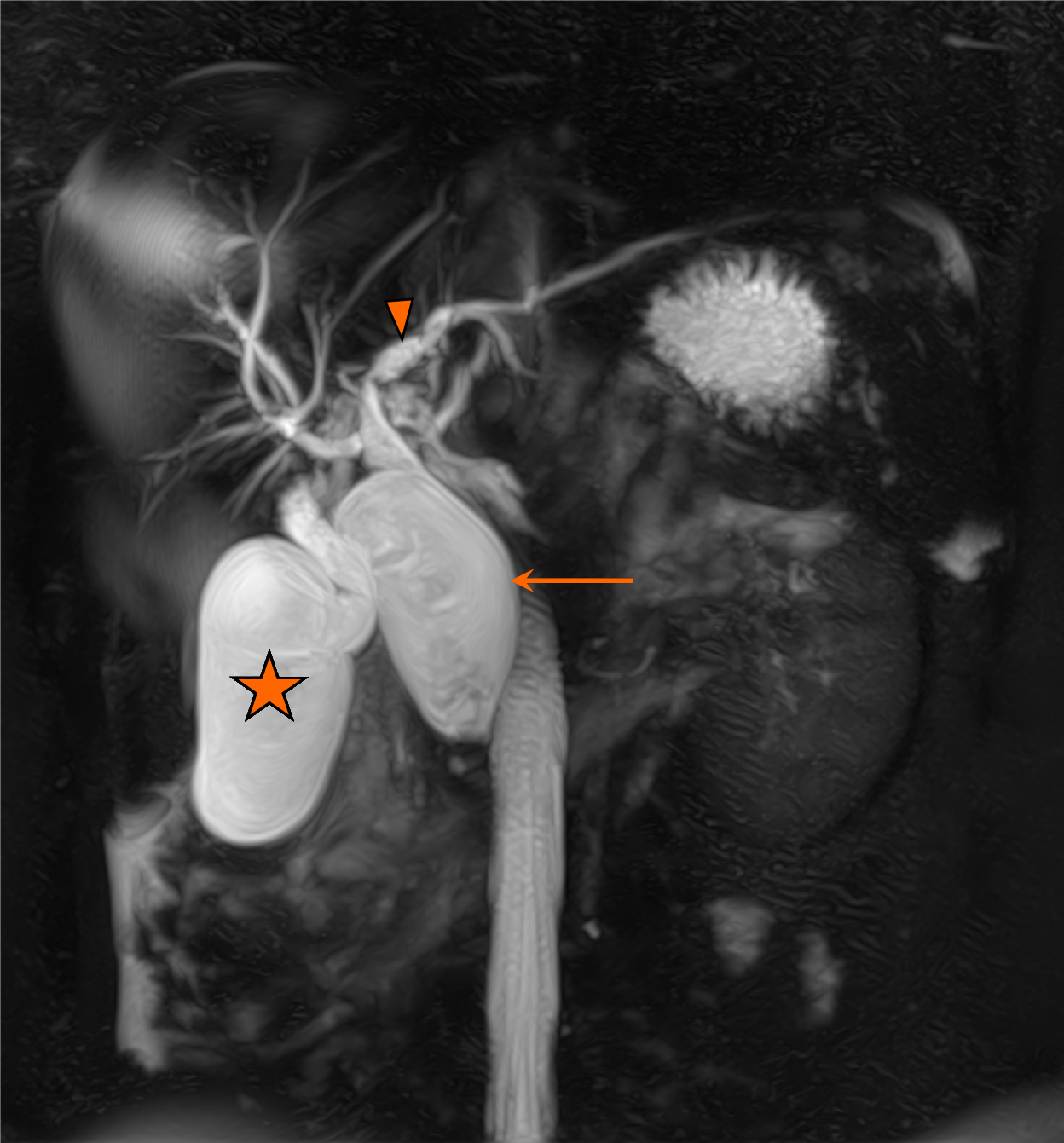
On US, a round or tubular cystic structure is observed in the right upper quadrant, separate from the gallbladder. Intrahepatic biliary dilatation may or may not be present. Dilatation of the common bile duct greater than 10 mm, with a fusiform or saccular shape, is suggestive of a choledochal cyst. On color Doppler imaging, no internal vascularity is observed within these dilated cystic structures.
On CT, choledochal cysts appear as continuous or multifocal intrahepatic and/or extrahepatic ductal dilatations (Figure 15). MRCP is the preferred imaging modality for assessing the anatomy of dilated biliary ducts. T2-weighted MRCP images typically show hyperintense cystic structures in communication with the biliary tree, which is distinct from the gallbladder (Figure 16). Delayed T1 postcontrast images can further confirm the biliary connection of these cysts. MRCP is usually performed for detailed preoperative assessment.
In cases of chronic biliary obstruction, cirrhosis and portal hypertension may develop[30]. Most choledochal cysts are associated with an anomalous pancreaticobiliary junction, resulting in an abnormally long common channel.
Hydatid disease is caused by Echinococcus granulosus, a parasitic infection that can affect any part of the body. Hydatid cysts are commonly observed in LMICs. Humans become infected secondarily by ingesting food or water contaminated with Echinococcus eggs. The liver is the most commonly affected site[31].
Hydatid cysts are typically acquired in childhood but may remain undiagnosed until the patient is 30-40 years of age. There is no gender predilection. Initially, hydatid cysts are often asymptomatic but can become symptomatic as they increase in size, become infected, or rupture. Patients commonly present with fever, pain, jaundice, and hepatomegaly.
In cases where a cyst ruptures into the peritoneal cavity, patients may develop a life-threatening anaphylactic reaction[31]. Signs of portal hypertension may also be observed. Laboratory investigations typically reveal eosinophilia, elevated serologic titers, and increased levels of alkaline phosphatase and gamma-glutamyl transpeptidase.
On grayscale US, the appearance of a hydatid cyst varies depending on its stage of evolution. It may present as a predominantly anechoic structure with internal debris or as a multiseptated cyst containing surrounding daughter cysts and intervening echogenic material, which are typical manifestations of hydatid disease. A characteristic feature is the presence of a floating, undulating membrane within the cyst, known as the "water lily sign" (Figure 17). Dense calcifications may also be observed within the cyst[31].
On CT, hydatid cysts appear as unilocular or multilocular cystic lesions with multiple surrounding daughter cysts of lower density. Detached endocysts may float in the cyst fluid, producing the characteristic water lily sign (Figure 18). Calcifications of the cyst wall may be present, and if they are completely circumferential, active infection can be ruled out. Enhancement of the cyst wall and septa may also be noted. Intrahepatic biliary dilatation may occur due to biliary compression or cyst rupture.
On MRI, hydatid cysts typically appear hyperintense on T2-weighted images with a hypointense rim. The matrix demonstrates intermediate signal intensity on T1-weighted images. Floating membranes appear as low to intermediate signal intensity on T2-weighted images. On T1 postcontrast images, enhancement of the cyst walls and septations may be observed.
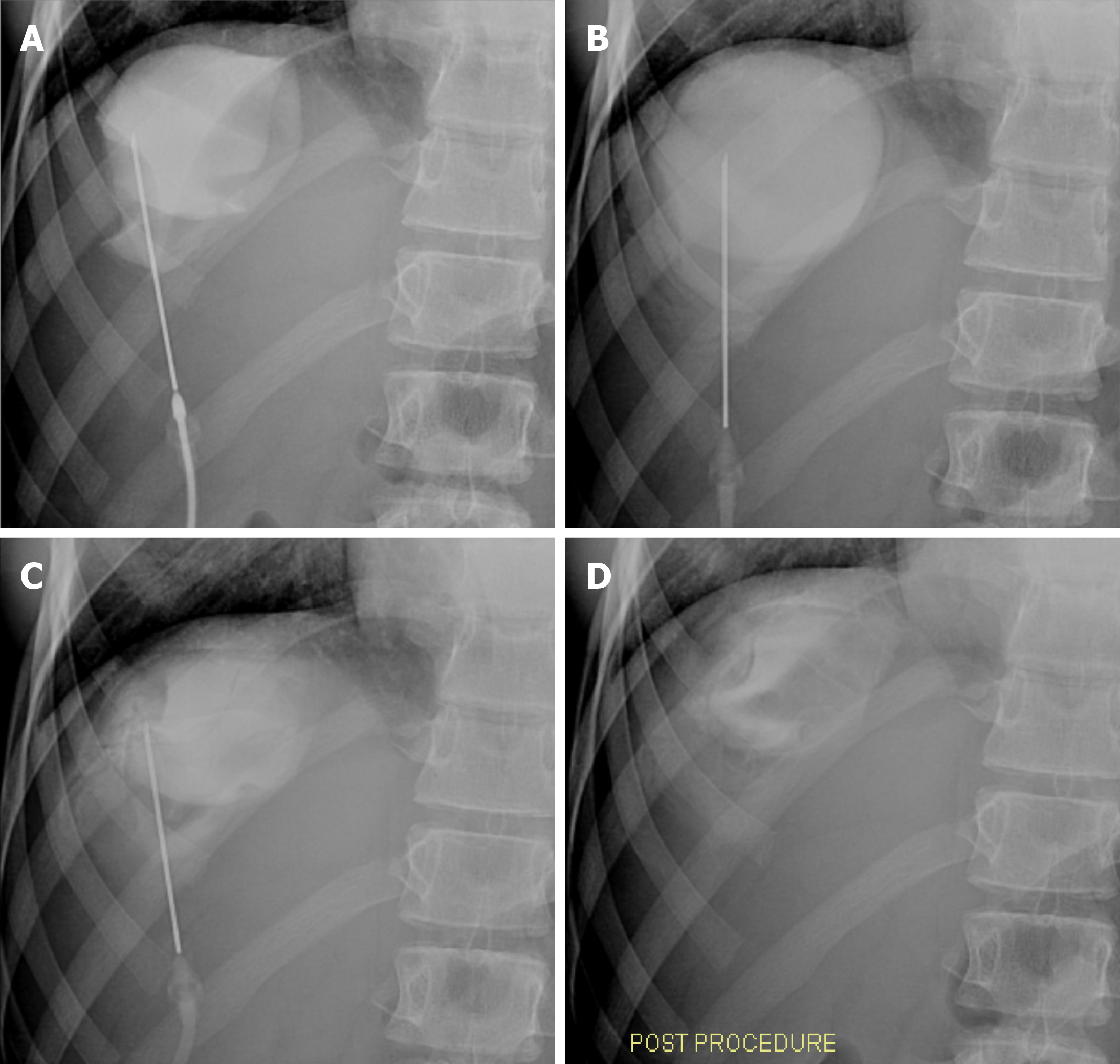
Percutaneous puncture, aspiration, injection, and reaspiration is a commonly performed procedure for the management of hydatid cysts (Figure 19). Under US guidance, a sheathed needle is used to puncture the cyst, followed by aspiration of the cyst fluid. A scolicidal agent is then injected into the cyst, and reaspiration is performed after a 20-minute interval[32].
Hepatic abscesses are localized collections of necrotic tissue, which can be related to fungal, bacterial, or parasitic infections. There are five major routes through which hepatic abscesses develop: (1) The portal vein; (2) Biliary tract; (3) Hepatic artery; (4) Direct extension; and (5) Trauma[33].
Portal vein-associated abscesses most commonly involve the right lobe of the liver and are usually solitary. Biliary tract-associated abscesses typically involve both lobes, particularly near the biliary ducts, and often present as multiple small abscesses. Parasitic abscesses are more prevalent in developing countries.
Clinically, patients with hepatic abscesses present with fever, right upper quadrant pain, rigors, and malaise. Some patients may also experience nausea, vomiting, and tender hepatomegaly. If the abscess is subphrenic, associated pulmonary complications such as atelectasis and pleural effusion may occur. Laboratory investigations commonly reveal leukocytosis and elevated serum alkaline phosphatase levels. The diagnosis is established through fine needle aspiration.
Among hepatic abscesses, pyogenic abscesses are the most common type. In children, the most common causative organism for pyogenic abscesses is Staphylococcus aureus[34]. Amoebic abscesses are caused by Entamoeba histolytica, whereas fungal abscesses most commonly result from Candida albicans.
On grayscale US, hepatic abscesses generally appear as poorly demarcated areas in the liver with variable echogenicity. Fluid levels, debris, and gas may be observed within the abscess, along with internal septations. The abscess walls may be thick or thin. Early lesions tend to be more irregular, with increased internal echogenicity, and may evolve into well-demarcated anechoic lesions. The adjacent hepatic parenchyma appears hypoechoic due to surrounding edema. On color Doppler imaging, vascularity may be observed in the wall of the abscess, with hypervascular hepatic parenchyma surrounding the lesion due to hyperemia.
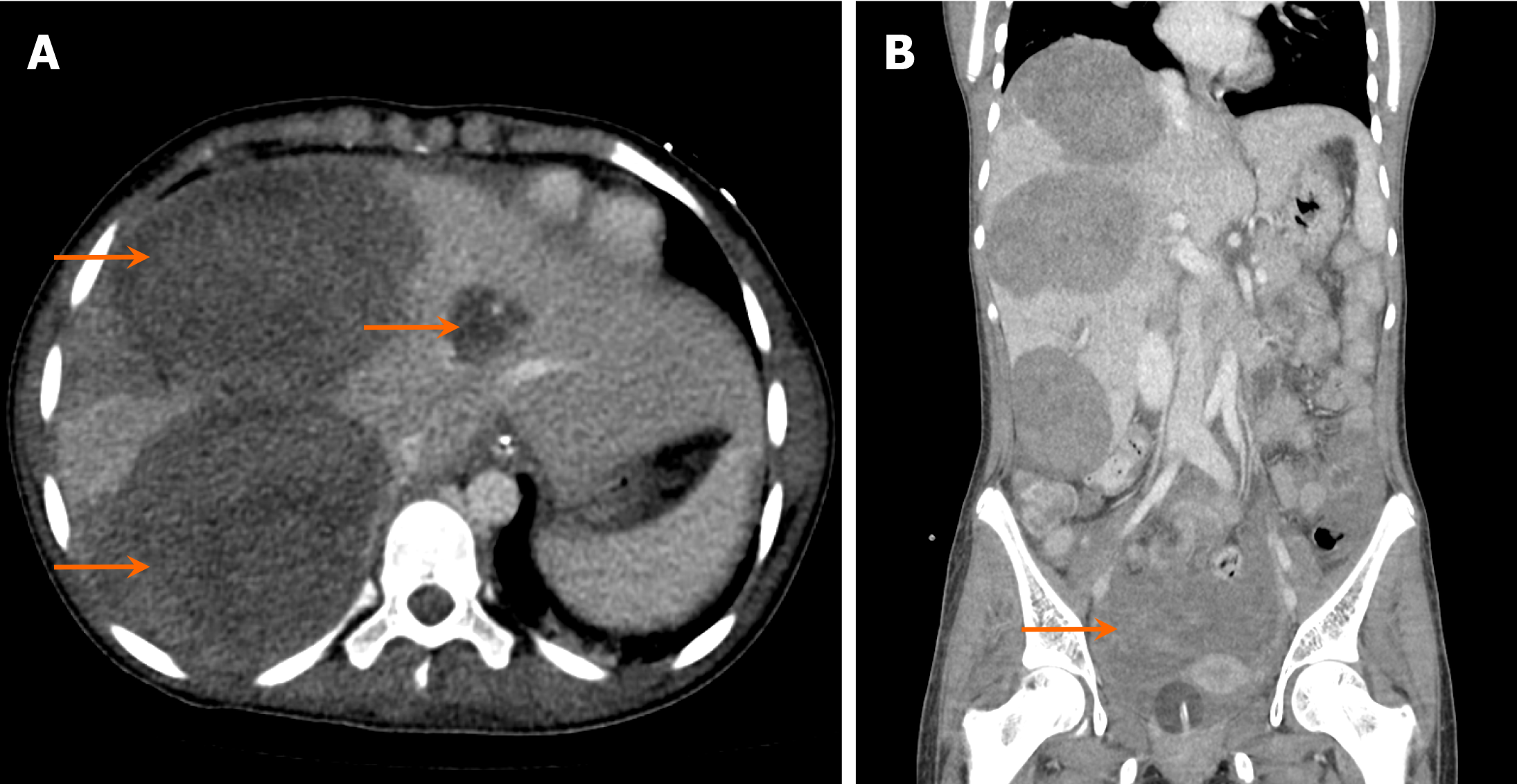
On unenhanced CT, a simple abscess appears as a well-defined hypodense lesion. Multiple small abscesses may coalesce to form a single large septated cavity, known as the “cluster sign” (Figure 20). A complex pyogenic abscess presents as a target lesion, with a central hypodensity and a hyperdense capsule. On contrast-enhanced CT, a “double target sign” may be observed, characterized by central hypodensity, a surrounding hyperdense rim, and an outer hypodense zone (Figure 21). Capsular and septal enhancement may also be noted. In cases of subphrenic abscess, right lower lobe atelectasis and pleural effusion may be present[34].
On MRI, hepatic abscesses typically appear as T2 hyperintense and T1 hypointense lesions, with surrounding hyperintense hepatic parenchyma on T2-weighted images, indicating edema. On postcontrast T1 images, capsular enhancement may be observed, and smaller abscesses (< 1 cm) may demonstrate homogeneous enhancement. The presence of gas appears as a signal void on gradient or in-phase images. Restricted diffusion is observed on DWI[35].
Hepatobiliary or sulfur colloid scans often reveal rounded cold areas, whereas gallium scans reveal a mixed lesion with a cold center and a hot rim corresponding to the abscess. On white blood cell (WBC) scans, hot lesions are observed due to the accumulation of WBCs, which is highly suggestive of pyogenic abscesses.
Hepatic tuberculosis (TB) is observed in up to 70% of patients with miliary TB; however, isolated hepatic involvement is rare, with an incidence of less than 1%. Hepatic spread of the bacteria occurs either through hematogenous dissemination from other organs or via the portal venous route from ingested bacilli in the bowel, with direct extension from adjacent organs such as the lung or pleura being rare. Granuloma formation typically occurs in the periportal region (Zone 1 of Rappaport) because of its optimal oxygen supply. Tuberculomas can merge, forming large areas of caseation with central necrosis[36]. Recognized forms of hepatic TB are categorized as miliary, local and biliary. The radiological appearance of TB can mimic hepatic malignancies and metastasis, potentially leading to unnecessary hepatic resection.
In immunocompetent children, isolated hepatic TB presents with nonspecific symptoms such as fever, weight loss, abdominal pain, and occasionally jaundice. The most common presentation is nonspecific hepatomegaly.
On grayscale US, hepatic TB may present with multiple liver nodules that are generally hypoechoic but can rarely be hyperechoic. Focal hepatic lesions larger than 2 mm are classified as macronodular or pseudotumoral TB[37,38].
On CT, the imaging appearance can vary from a hypodense lesion to a heterogeneous density mass with a necrotic center and “bull’s eye” calcification depending on the stage of the disease[39]. Like pyogenic abscesses, tubercular abscesses may occasionally demonstrate a “honeycomb” pattern, characterized by multiple enhancing septations interspersed with areas of necrosis. In some instances, the lesions can undergo extensive necrosis, resembling cystic structures with absent or minimal peripheral enhancement[40].
On MRI, hepatic TB can present with multiple T2 hyperintense nodules in patients with miliary TB and granulomas, with variable peripheral enhancement. Caseating abscesses have central T2 hyperintensity with a peripheral hypointense rim (capsule) showing rim enhancement on postcontrast images and diffusion restriction on DWI, depending on the degree of internal liquefaction. Calcified granulomas are usually T1 and T2 hypointense. Pseudotumoral TB usually presents with large T1 hypointense and T2 hyperintense masses with peripheral enhancement, but findings are often nonspecific and difficult to differentiate from those of other lesions (Figure 22)[40].
Lymphomatous deposits in the liver are commonly observed in patients with lymphoma, and most frequently occur in the periportal region because of the high lymphatic content in these areas. In children, these lesions are most often secondary to non-Hodgkin lymphoma or acquired immunodeficiency syndrome-associated lymphoma, but the liver can also rarely be a site for primary lymphoma[41,42].
Patients with lymphomatous liver lesions typically present with hepatomegaly, jaundice, right upper quadrant pain, fever, weight loss, and night sweats. Laboratory investigations commonly reveal deranged LFTs with elevated lactate dehydrogenase levels.
On grayscale US, multiple well-defined hypoechoic or anechoic lesions are typically observed. In some cases, conglomerate masses with internal septations may mimic abscesses. Associated splenomegaly with splenic deposits is frequently observed. Periportal, para-aortic, or mesenteric lymphadenopathy is common in patients with lymphoma. In cases of more extensive involvement, bowel wall thickening and ascites may also be present.
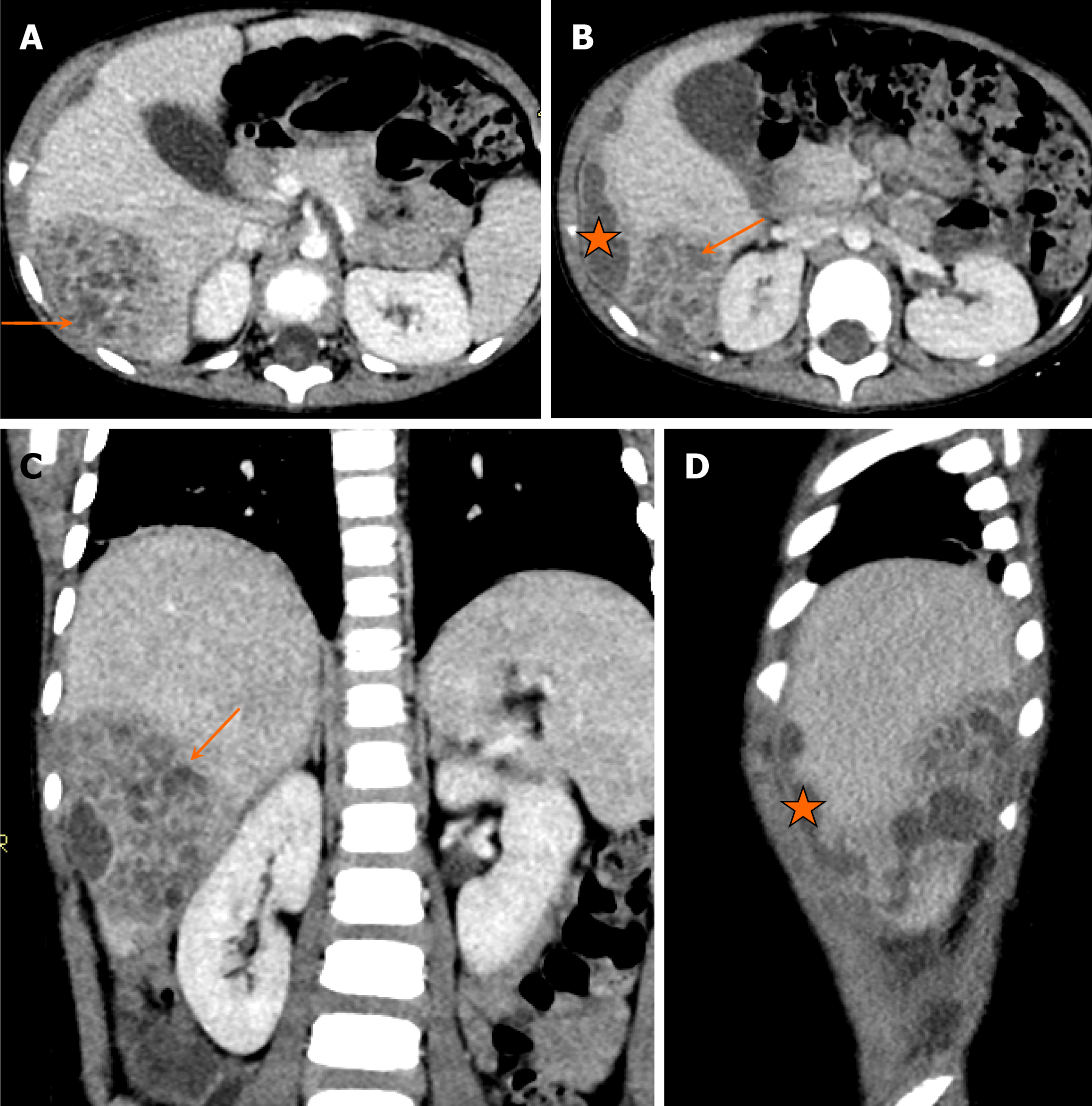
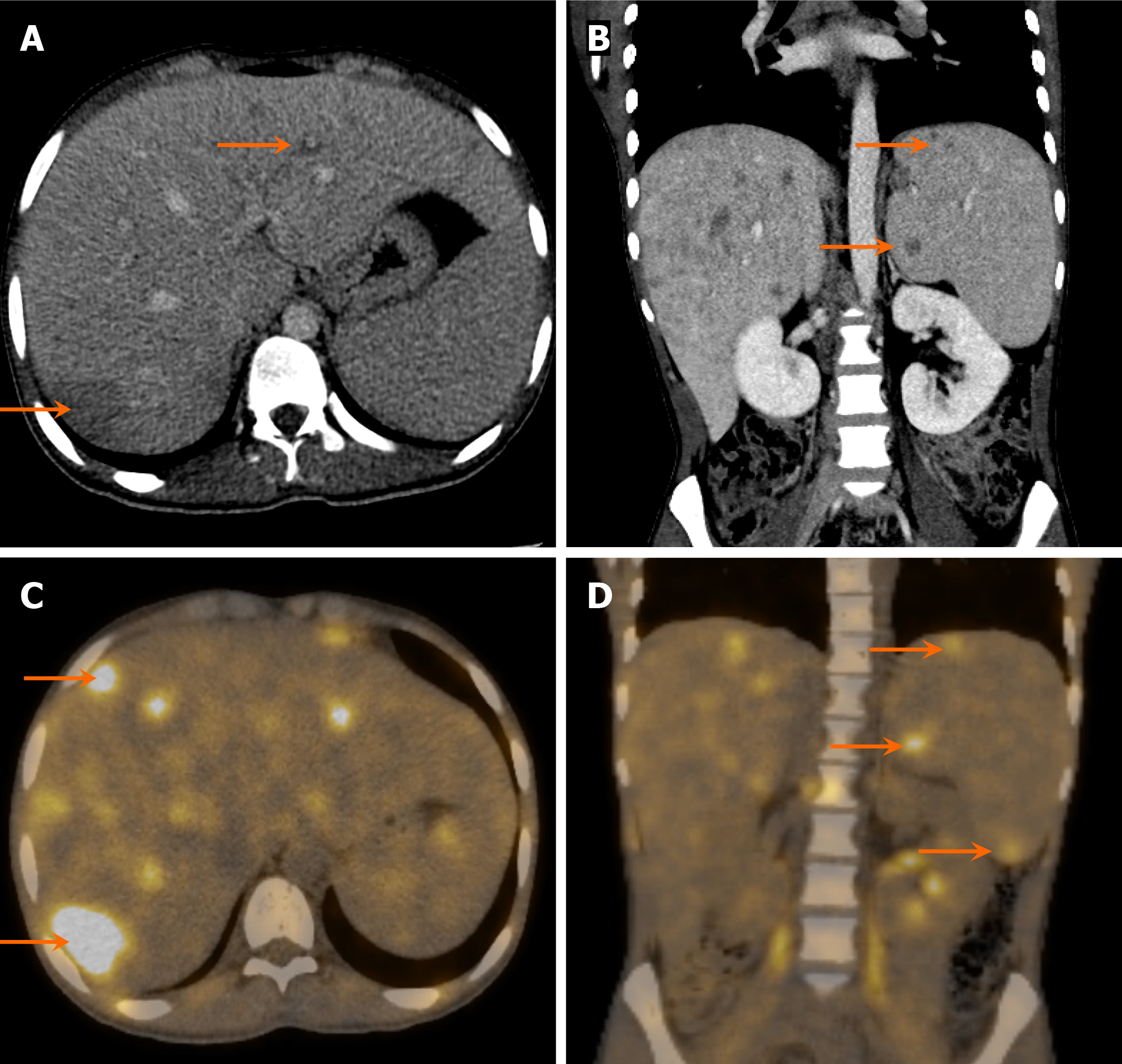
On CT, solid homogeneous lesions with poor contrast enhancement and thin rim enhancement are characteristic (Figure 23). On MRI, the lesions typically appear hypointense on T1-weighted images and hyperintense on T2-weighted images, with variable (mostly poor) enhancement on T1 postcontrast images. FDG-PET/CT is particularly useful in detecting fluorodeoxyglucose-avid lesions at sites of lymphomatous involvement that may be occult on conventional imaging (Figure 24)[43].
Liver metastases occur in various malignancies and are more common than primary hepatic tumors. In children, they are frequently associated with neu
Patients typically present with a palpable right upper quadrant mass, hepatomegaly, fever, and weight loss. Neuroblastoma hepatic metastasis is often referred to as Pepper syndrome. Laboratory investigations generally reveal deranged LFTs.
Metastasis can have a variety of appearances on imaging. On grayscale US, metastatic liver lesions often appear as rounded masses exerting mass effect on the surrounding hepatic parenchyma and vessels. These lesions are predominantly hypoechoic, but they can also present as cystic, echogenic, calcified, or infiltrative. Internal vascularity may be observed on color Doppler imaging.
On contrast-enhanced CT, metastases typically appear as multifocal hypodense or hyperdense masses of varying sizes (Figure 23)[44]. Peripheral enhancement on postcontrast images creates a characteristic “target” appearance, with central filling on the venous phase and subsequent washout on the delayed phase.
On MRI, metastatic lesions usually appear hypointense on T1-weighted images and hyperintense on T2-weighted images. Postcontrast imaging often demonstrates lesional enhancement, with smaller lesions exhibiting homogeneous enhancement, whereas larger lesions display a more heterogeneous pattern[45]. In some cases, perilesional enhancement may be more prominent than intralesional enhancement. Diffusion restriction is commonly noted within lesions on DWI and apparent diffusion coefficient sequences, depending on the cellularity of the primary neoplasm. Table 5 shows a summary of the hepatic masses encountered in all age groups of children.
| Lesion | Clinical features | Relevant laboratory investigations | Imaging appearances |
| Simple hepatic cyst | Asymptomatic; incidental finding; large cysts may cause abdominal pain or mass effect | Normal LFTs; no infection markers | US: Anechoic lesion with posterior acoustic enhancement. CT/MRI: Nonenhancing, fluid-filled lesion with thin wall |
| Choledochal cyst | Jaundice, abdominal pain, palpable mass, vomiting | LFTs abnormal in cholestasis | Magnetic resonance cholangiopancreatography/CT: Fusiform or saccular bile duct dilation showing communication with the biliary tree |
| Hydatid cyst | Asymptomatic in early stages; abdominal pain; rupture may cause anaphylaxis | Eosinophilia; positive Echinococcus serology | CT/MRI: Multiloculated cyst with calcifications, detached membrane (water lily sign) |
| Hepatic abscess | Fever, pain, jaundice, hepatomegaly; may mimic malignancy | Elevated white blood cell, CRP, ESR; positive blood cultures; abnormal LFTs | US: Hypoechoic/complex lesion, internal septations. CT: Peripheral enhancement, cluster sign, double target sign |
| Tuberculous liver lesions | Fever, weight loss, hepatosplenomegaly; often part of disseminated TB | Positive TB test, elevated CRP/ESR, possible acid fast bacilli in biopsy | CT/MRI: Calcified granulomas, miliary nodules, caseating necrosis, abscess formation |
| Lymphoma (hepatic involvement) | Fever, weight loss, hepatomegaly | Elevated lactate dehydrogenase; abnormal peripheral blood counts; possible lymphoma markers | US: Hypoechoic, ill-defined masses. CT/MRI: Multiple low-attenuation lesions, mild enhancement |
| Hepatic metastasis | Hepatomegaly, pain, systemic symptoms related to primary malignancy | Tumor markers (alpha-fetoprotein, catecholamines, etc.), abnormal LFTs | CT/MRI: Multiple lesions, variable enhancement, necrotic centers, target appearance |
This pictorial minireview highlights the clinical and radiological manifestations of common hepatic lesions in the pediatric population, many of which present with overlapping features. By emphasizing their distinct radiological characteristics, we aim to simplify the diagnostic process and support timely, accurate diagnosis, ultimately facilitating appropriate and effective management. However, clinical challenges persist, particularly in the accurate differentiation of rare lesions, which may lead to misdiagnosis or delayed treatment. Future research should focus on validating imaging-based diagnostic algorithms, integrating advanced techniques such as multiparametric MRI, and exploring the role of minimally invasive tools such as liquid biopsy. Addressing these gaps could significantly increase diagnostic accuracy and inform management strategies, especially in resource-limited settings, thereby advancing pediatric clinical hepatology.
| 1. | Chiorean L, Cui XW, Tannapfel A, Franke D, Stenzel M, Kosiak W, Schreiber-Dietrich D, Jüngert J, Chang JM, Dietrich CF. Benign liver tumors in pediatric patients - Review with emphasis on imaging features. World J Gastroenterol. 2015;21:8541-8561. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 91] [Cited by in RCA: 76] [Article Influence: 7.6] [Reference Citation Analysis (2)] |
| 2. | El Asmar A, El Rassi Z. Hepatoblastoma in childhood, long term survival achieved: 2 case reports and literature review. Int J Surg Case Rep. 2016;21:55-58. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 3. | Lucas B, Ravishankar S, Pateva I. Pediatric Primary Hepatic Tumors: Diagnostic Considerations. Diagnostics (Basel). 2021;11:333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 31] [Article Influence: 7.8] [Reference Citation Analysis (0)] |
| 4. | Yang T, Whitlock RS, Vasudevan SA. Surgical Management of Hepatoblastoma and Recent Advances. Cancers (Basel). 2019;11:1944. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 5. | Towbin AJ, Meyers RL, Woodley H, Miyazaki O, Weldon CB, Morland B, Hiyama E, Czauderna P, Roebuck DJ, Tiao GM. 2017 PRETEXT: radiologic staging system for primary hepatic malignancies of childhood revised for the Paediatric Hepatic International Tumour Trial (PHITT). Pediatr Radiol. 2018;48:536-554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 170] [Article Influence: 24.3] [Reference Citation Analysis (0)] |
| 6. | Chavhan GB, Schooler GR, Tang ER, Squires JH, Rees MA, Nguyen HN, Morin CE, Kolbe AB, Khanna G, Infante JC, Alazraki AL, Towbin AJ. Optimizing Imaging of Pediatric Liver Lesions: Guidelines from the Pediatric LI-RADS Working Group. Radiographics. 2023;43:e220043. [RCA] [PubMed] [DOI] [Full Text] [Reference Citation Analysis (0)] |
| 7. | Brás S, Mendes-Bastos P, Amaro C. Rapidly involuting congenital hemangioma. An Bras Dermatol. 2017;92:861-863. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 9] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 8. | Cordier F, Hoorens A, Van Dorpe J, Creytens D. Pediatric vascular tumors of the liver: Review from the pathologist's point of view. World J Hepatol. 2021;13:1316-1327. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 1.8] [Reference Citation Analysis (1)] |
| 9. | Adeyiga AO, Lee EY, Eisenberg RL. Focal hepatic masses in pediatric patients. AJR Am J Roentgenol. 2012;199:W422-W440. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 10. | Al Tasseh F, El-Khansa M, Abd O, Abdel Khalek A, El-Rifai N. Diffuse hepatic hemangioma with single cutaneous hemangioma: an alerting occurrence. Clin Case Rep. 2017;5:887-890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 11. | D'souza D, Di Muzio B, Yap J. Kasabach-Merritt syndrome. Radiopaedia.org. 2008;. [DOI] [Full Text] |
| 12. | Esposito F, D'Auria D, Ferrara D, Esposito P, Gaglione G, Zeccolini M, Tomà P. Hepatic hemangiomas in childhood: the spectrum of radiologic findings. A pictorial essay. J Ultrasound. 2023;26:261-276. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 2] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 13. | Radswiki T, Silverstone L, Campos A. Infantile hepatic haemangioma. Radiopaedia.org. 2010;. [DOI] [Full Text] |
| 14. | Léauté-Labrèze C, Hoeger P, Mazereeuw-Hautier J, Guibaud L, Baselga E, Posiunas G, Phillips RJ, Caceres H, Lopez Gutierrez JC, Ballona R, Friedlander SF, Powell J, Perek D, Metz B, Barbarot S, Maruani A, Szalai ZZ, Krol A, Boccara O, Foelster-Holst R, Febrer Bosch MI, Su J, Buckova H, Torrelo A, Cambazard F, Grantzow R, Wargon O, Wyrzykowski D, Roessler J, Bernabeu-Wittel J, Valencia AM, Przewratil P, Glick S, Pope E, Birchall N, Benjamin L, Mancini AJ, Vabres P, Souteyrand P, Frieden IJ, Berul CI, Mehta CR, Prey S, Boralevi F, Morgan CC, Heritier S, Delarue A, Voisard JJ. A randomized, controlled trial of oral propranolol in infantile hemangioma. N Engl J Med. 2015;372:735-746. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 488] [Cited by in RCA: 500] [Article Influence: 50.0] [Reference Citation Analysis (0)] |
| 15. | Kim SH, Kim WS, Cheon JE, Yoon HK, Kang GH, Kim IO, Yeon KM. Radiological spectrum of hepatic mesenchymal hamartoma in children. Korean J Radiol. 2007;8:498-505. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 32] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 16. | Rees MA, Schooler GR, Chavhan GB, Towbin AJ, Alazraki AL, Squires JH, Fraum TJ, Zhang C, Khanna G. Imaging Features of Hepatocellular Carcinoma in Children With and Without Underlying Risk Factors. AJR Am J Roentgenol. 2022;219:647-654. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 17. | Ganeshan D, Szklaruk J, Kundra V, Kaseb A, Rashid A, Elsayes KM. Imaging features of fibrolamellar hepatocellular carcinoma. AJR Am J Roentgenol. 2014;202:544-552. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 67] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 18. | Liu QY, Zhang WD, Lai DM, Ou-Yang Y, Gao M, Lin XF. Hepatic focal nodular hyperplasia in children: imaging features on multi-slice computed tomography. World J Gastroenterol. 2012;18:7048-7055. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 12] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 19. | Sintusek P, Phewplung T, Sanpavat A, Poovorawan Y. Liver tumors in children with chronic liver diseases. World J Gastrointest Oncol. 2021;13:1680-1695. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 6] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 20. | Cha DI, Yoo SY, Kim JH, Jeon TY, Eo H. Clinical and imaging features of focal nodular hyperplasia in children. AJR Am J Roentgenol. 2014;202:960-965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 21. | Welch TJ, Sheedy PF 2nd, Johnson CM, Stephens DH, Charboneau JW, Brown ML, May GR, Adson MA, McGill DB. Focal nodular hyperplasia and hepatic adenoma: comparison of angiography, CT, US, and scintigraphy. Radiology. 1985;156:593-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 159] [Cited by in RCA: 118] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 22. | Espinoza AF, Vasudevan SA, Masand PM, Lòpez-Terrada DH, Patel KR. Pediatric Hepatocellular Adenomas: What Is Known and What Is New? Cancers (Basel). 2023;15:4790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 23. | Grazioli L, Olivetti L, Mazza G, Bondioni MP. MR Imaging of Hepatocellular Adenomas and Differential Diagnosis Dilemma. Int J Hepatol. 2013;2013:374170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 46] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 24. | Tsuruya K, Nishizaki Y, Tatemichi M, Mishima Y, Shimma Y, Arase Y, Hirose S, Shiraishi K, Kagawa T. The prevalence and natural history of hepatic cysts examined by ultrasound: a health checkup population retrospective cohort study. Sci Rep. 2022;12:12797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 7] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 25. | Rogers TN, Woodley H, Ramsden W, Wyatt JI, Stringer MD. Solitary liver cysts in children: not always so simple. J Pediatr Surg. 2007;42:333-339. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 33] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 26. | Riedesel EL, Richer EJ, Taylor SD, Tao T, Gagnon MH, Braithwaite KA, Alazraki AL, Khanna G. Pediatric Hepatic Cystic Lesions: Differential Diagnosis and Multimodality Imaging Approach. Radiographics. 2022;42:1514-1531. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 27. | Chenin M, Paisant A, Lebigot J, Bazeries P, Debbi K, Ronot M, Laurent V, Aubé C. Cystic liver lesions: a pictorial review. Insights Imaging. 2022;13:116. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in RCA: 9] [Reference Citation Analysis (0)] |
| 28. | Madadi-Sanjani O, Wirth TC, Kuebler JF, Petersen C, Ure BM. Choledochal Cyst and Malignancy: A Plea for Lifelong Follow-Up. Eur J Pediatr Surg. 2019;29:143-149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 29. | Radswiki T, Bell D, Alhusseiny K. Todani classification of bile duct cysts. Radiopaedia.org. 2010;. [DOI] [Full Text] |
| 30. | Khandelwal C, Anand U, Kumar B, Priyadarshi RN. Diagnosis and management of choledochal cysts. Indian J Surg. 2012;74:29-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 31. | Mehta P, Prakash M, Khandelwal N. Radiological manifestations of hydatid disease and its complications. Trop Parasitol. 2016;6:103-112. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 62] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 32. | Paksoy Y, Odev K, Sahin M, Arslan A, Koç O. Percutaneous treatment of liver hydatid cysts: comparison of direct injection of albendazole and hypertonic saline solution. AJR Am J Roentgenol. 2005;185:727-734. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 33. | Malekzadeh S, Widmer L, Salahshour F, Egger B, Ronot M, Thoeny HC. Typical imaging finding of hepatic infections: a pictorial essay. Abdom Radiol (NY). 2021;46:544-561. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 18] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 34. | Salahi R, Dehghani SM, Salahi H, Bahador A, Abbasy HR, Salahi F. Liver abscess in children: a 10-year single centre experience. Saudi J Gastroenterol. 2011;17:199-202. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 35. | Hegde SV, Dillman JR, Lopez MJ, Strouse PJ. Imaging of multifocal liver lesions in children and adolescents. Cancer Imaging. 2013;12:516-529. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 8] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 36. | Reynolds M. Pediatric liver tumors. Cancer Treat Res. 2001;109:299-312. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 5] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 37. | Peters NJ, Samujh R, Gunasekaran V, Sodhi KS, Duseja R. Pediatric PseudoTumoral Hepatic Tuberculosis. A Great Mimicker!! J Indian Assoc Pediatr Surg. 2022;27:368-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 38. | Wu Z, Wang WL, Zhu Y, Cheng JW, Dong J, Li MX, Yu L, Lv Y, Wang B. Diagnosis and treatment of hepatic tuberculosis: report of five cases and review of literature. Int J Clin Exp Med. 2013;6:845-850. [PubMed] |
| 39. | Dursun I, Yikilmaz A, Poyrazoglu H, Soyuer I, Gunduz Z. Radiological findings of isolated hepatic tuberculosis in a child with tubulointerstitial nephritis-uveitis syndrome. Pediatr Radiol. 2009;39:302-305. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 7] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 40. | Kakkar C, Polnaya AM, Koteshwara P, Smiti S, Rajagopal KV, Arora A. Hepatic tuberculosis: a multimodality imaging review. Insights Imaging. 2015;6:647-658. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 45] [Cited by in RCA: 51] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 41. | Marie E, Navallas M, Katz DS, Farajirad E, Punnett A, Davda S, Shammas A, Oudjhane K, Vali R. Non-Hodgkin Lymphoma Imaging Spectrum in Children, Adolescents, and Young Adults. Radiographics. 2022;42:1214-1238. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 7] [Reference Citation Analysis (0)] |
| 42. | Maher MM, McDermott SR, Fenlon HM, Conroy D, O'Keane JC, Carney DN, Stack JP. Imaging of primary non-Hodgkin's lymphoma of the liver. Clin Radiol. 2001;56:295-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 83] [Cited by in RCA: 76] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 43. | Alves AMA, Torres US, Velloni FG, Ribeiro BJ, Tiferes DA, D'Ippolito G. The many faces of primary and secondary hepatic lymphoma: imaging manifestations and diagnostic approach. Radiol Bras. 2019;52:325-330. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 44. | Fernandez-Pineda I, Sandoval JA, Davidoff AM. Hepatic metastatic disease in pediatric and adolescent solid tumors. World J Hepatol. 2015;7:1807-1817. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 45. | Das CJ, Dhingra S, Gupta AK, Iyer V, Agarwala S. Imaging of paediatric liver tumours with pathological correlation. Clin Radiol. 2009;64:1015-1025. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 26] [Article Influence: 1.6] [Reference Citation Analysis (0)] |