Published online Jun 27, 2021. doi: 10.4254/wjh.v13.i6.673
Peer-review started: January 8, 2021
First decision: March 29, 2021
Revised: April 12, 2021
Accepted: May 21, 2021
Article in press: May 21, 2021
Published online: June 27, 2021
Processing time: 148 Days and 4.6 Hours
In a previous paper, we reported a high prevalence of donor-specific antibody (DSA) in pediatric patients with chronic rejection and expressed the need for confirmation of these findings in a larger cohort.
To clarify the importance of DSAs on long-term graft survival in a larger cohort of pediatric patients.
We performed a retrospective analysis of 123 pediatric liver transplantation (LT) recipients who participated in yearly follow-ups including Luminex testing for DSA at our center. The cohort was split into two groups according to the DSA status (DSA-positive n = 54, DSA-negative n = 69). Groups were compared with regard to liver function, biopsy findings, graft survival, need for re-LT and immunosuppressive medication.
DSA-positive pediatric patients showed a higher prevalence of chronic rejection (P = 0.01), fibrosis (P < 0.001) and re-transplantation (P = 0.018) than DSA-negative patients. Class II DSAs particularly influenced graft survival. Alleles DQ2, DQ7, DQ8 and DQ9 might serve as indicators for the risk of chronic rejection and/or allograft fibrosis. Mean fluorescence intensity levels and DSA number did not impact graft survival. Previous episodes of chronic rejection might lead to DSA development.
DSA prevalence significantly affected long-term liver allograft performance and liver allograft survival in our cohort of pediatric LT. Screening for class II DSAs in combination with assessment of protocol liver biopsies for chronic antibody-mediated rejection improved early identification of patients at risk of graft loss.
Core Tip: This was a retrospective study to evaluate the impact of donor-specific antibodies (DSAs) on graft survival with pediatric liver transplantation. Graft fibrosis and graft loss was significantly higher in patients with DSAs. Screening for DSAs should be included in follow-ups to avoid delayed identification of patients at risk of graft loss (rejection), and may be even more relevant for patients with early histological signs of possible allograft dysfunction (fibrosis). Moreover, patients with DSAs may be poor candidates for reduction of initial immunosuppression or even weaning.
- Citation: Schotters FL, Beime J, Briem-Richter A, Binder T, Herden U, Grabhorn EF. Impact of donor-specific antibodies on long-term graft survival with pediatric liver transplantation. World J Hepatol 2021; 13(6): 673-685
- URL: https://www.wjgnet.com/1948-5182/full/v13/i6/673.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i6.673
The impact of anti-human leucocyte antigen (HLA) donor-specific antibodies (DSAs) on graft survival and graft function in kidney and heart transplantation is crucial[1-3]. However, their impact on liver transplantation (LT) is still controversial: Some research suggests that the overall impact on graft and patient outcome is limited[4], but other studies found that it may be an independent risk factor for patient death and graft loss[5-7].
For a long time, DSAs were suspected to have a minor influence on liver allografts[8,9], based on low vascular expression of HLA class II antigens, weak HLA class I expression on hepatocytes and a large endothelial surface diluting soluble antibodies and antigens[10]. However, HLA class II expression is present, especially on perivenular dendritic cells and endothelial cells of the septal venule, sinusoidal and central vein[11,12].
There is growing (but limited) data for pediatric LT suggesting that DSAs might have an impact on long-term graft survival by influencing the development of portal inflammation, portal fibrosis[13-15], perivenular or perisinusoidal fibrosis[15-18], or obliterative portal venopathy[19] and might lead to chronic antibody-mediated rejection (cAMR)[10,19,20].
According to Demetris et al[10,21-23] chronic rejection (CR) is characterized by a slow process based on alloreactivity. Histopathological findings of T-cell-mediated rejection (TCMR) include lesions or loss of small bile ducts, portal inflammation, venous endothelial inflammation, obliterative arteriopathy and low-grade necroinflammation. Since obliterative arteriopathy is rarely found in a percutaneous needle biopsy, ductopenia and signs of necroinflammation tend to be used for the diagnosis of chronic rejection in biopsy specimens[23]. According to O’Leary et al[24], ductopenia, biliary strictures and fibrosis are associated with DSAs in adult LT.
Initially, AMR was only suspected after ABO-incompatible transplantation, but it has since been reported in ABO-compatible LT as well[19,25,26]. It has also been suggested that TCMR and cAMR are linked, and that any form of TCMR might channel cAMR by increasing the presentation of intra- and extracellular donor antigens on dendritic cells, which would then stimulate the production of DSAs (second-hit hypothesis)[10,23,24,27,28].
This paper reports the results of a retrospective cross-sectional study of the influence of DSAs developed after LT on long-term graft survival in pediatric LT recipients.
From 1993 to 2015, 765 pediatric LTs were performed at our tertiary center. Testing of DSAs started in 2013, mostly with the ELISA technique. We performed a cross-sectional retrospective chart analysis of all patients coming for check-ups at our pediatric department between 2013 and 2017. All charts were checked for DSA measurement with the Luminex technique (single antigen bead assay) as the most sensitive test, as well as donor HLA typing and complete laboratory values. We included 123 patients in the present study after exclusions due to change of residency, follow-up in other transplant centers, missing donor typing and missing Luminex testing as well as deaths within the first year after LT.
DSA testing usually took place as part of the yearly follow-up routine at 1 to 19 years (mean 8.9 years) after first LT. There were 55 female (44.7%) and 68 male (55.3%) participants. The main diagnosis leading to LT was biliary atresia (n = 40, 32.5%), followed by metabolic disorders (n = 40, 32.5%), acute liver failure (n = 10, 8.1%), Alagille syndrome (n = 9, 7.3%) and others (n = 64, 52%). LT was performed either as full-graft (n = 22) or technically modified as a split graft (n = 74), reduced-size graft (n = 10) or living donor LT (n = 14). The majority of patients are still living with the first transplant (n = 87, 70.7%); 26 patients were retransplanted once (21.1%), 8 patients twice (6.5%) and two patients three times (1.6%). The main cause for re-transplantation was chronic rejection with chronic graft dysfunction; see baseline characteristics in Table 1 for more details.
| ‘DSA-positive’ group 1 (n = 54) | ‘DSA-negative’ group 2 (n = 69) | |
| Age | ||
| At LT (yr) | 3.3 (1 mo-17 yr) | 4.0 (1 mo-17.8 yr) |
| At follow-up (yr) | 13.8 (2-23) | 12.6 (1-24) |
| Gender | ||
| Female | n = 21 | n = 35 |
| Male | n = 33 | n = 35 |
| Main diagnosis | ||
| Biliary atresia | n = 20 | n = 20 |
| Alagille syndrome | n = 1 | n = 8 |
| Acute liver failure | n = 6 | n = 4 |
| Metabolic disorders1 | n = 14 | n = 26 |
| Others2 | n = 13 | n = 11 |
| Donor source | ||
| Ldlt | n = 9 | n = 11 |
| Ddlt | n = 45 | n = 58 |
| Full-graft | n = 8 | n = 12 |
| Split size | n = 30 | n = 43 |
| Reduced size | n = 7 | n = 3 |
| Cold ischemic time (min) | 543.3 (122-949) | 572.5 (145-943) |
| Relt | n = 22 | n = 14 |
| Graft loss due to | ||
| Cr | n = 23 | n = 7 |
| Alv | n = 7 | n = 3 |
| Thrombosis | n = 3 | n = 3 |
| Rond | n = 2 | n = 0 |
| Ssc | n = 2 | n = 0 |
| Time to DSA-testing | ||
| Years after current LT | 9.75 (1-19) | 7.98 (1-19) |
| Anti-HLA antibodies | n = 54 | n = 32 |
| Previous episodes | ||
| Of acute rejection | n = 12 | n = 7 |
| Of chronic rejection | n = 10 | n = 3 |
Yearly follow-ups included physical examination of the child, an ultrasound evaluation of the graft, extensive laboratory diagnostics (including detection of DSAs) and histopathological examination of liver biopsies if available.
Since we perform DSA detection with luminex testing as part of our routine clinical practice and because of the retrospective study design, our study was readily approved by the ethics committee.
In our center, we perform routine protocol liver biopsies every three to five years after LT. Liver biopsy is also indicated if there are laboratory signs of allograft dysfunction or if rejection is suspected. The histological features that we assessed are shown in Table 2. In case of fibrosis or rejection, grading and rating was performed using the Desmet score for fibrosis, the rejection activity index and Banff scores for chronic liver transplant rejection by experienced in-house pathologists.
| Group 1: ‘DSA-positive’ (n = 38) | Group 2: ‘DSA-negative’ (n = 34) | P value | |
| Fibrosis1 | n = 24 | n = 6 | |
| Low-grade | n = 23 | n = 6 | < 0.001 |
| High-grade | n = 1 | n = 0 | 0.5 |
| Cirrhosis | n = 3 | n = 0 | 0.5 |
| Steatosis | n = 6 | n = 6 | 0.8 |
| Portal inflammation | n = 28 | n = 18 | 0.005 |
| Perivenular/perisinusoidal inflammation | n = 11 | n = 2 | 0.011 |
| Ductular inflammation | n = 13 | n = 3 | 0.004 |
| Endothelitis | n = 6 | n = 2 | 0.1 |
| Biliary lesions/ductulopenia | n = 6 | n = 0 | 0.009 |
| Ductular cholestasis | n = 12 | n = 4 | 0.01 |
| Biliary tract strictures | n = 9 | n = 6 | 0.03 |
| Single cell necrosis | n = 5 | n = 0 | 0.02 |
| Chronic rejection2 | n = 7 | n = 0 | 0.009 |
| Possible camr3 | n = 9 | n = 0 | 0.002 |
We described the technique of HLA typing and HLA antibody testing in our previous paper[14]. In this study, we also used luminex single antigen class I and class II beads (One Lambda Inc., LABScreen®) for retrospective detection of anti-HLA-antibodies (A, -B, -Cw, -DR, -DQ, and -DP). Normalized mean fluorescence intensity (MFI) > 1500 was regarded as positive.
All data were statistically analyzed using IBM SPSS Statistics® software, Version 25. Due to their mainly Fisher’s exact non-Gaussian character, variables were analyzed with Pearson’s chi-square, Cramer and Phi, Mann-Whitney U and the Wilcoxon test. Freedom from events (graft loss and rejection) was estimated by the Kaplan-Meier method and was compared across groups with the log-rank test. Graft survival was computed from the date of LT to re-transplantation, or to biopsy-proven rejection. A P value < 0.05 was considered statistically significant. We performed binary correlation analysis and evaluation of odds ratios (95%CI, P < 0.05). Significant correlations (P < 0.02) were included to form predictive models for DSA development and chronic rejection with binary logistic regression and Cox regression analysis.
The population was divided into two groups according to DSA status (group 1 = DSA-positive, n = 54; group 2 = DSA-negative, n = 69). If DSAs against a certain HLA locus were found in more than four patients (n ≥ 5), they were analyzed separately to determine whether a single HLA locus was a common target for DSA formation or if it might be associated with histopathological changes, chronic rejection or retransplantation. To assess whether the number of DSAs influenced graft survival, we compared graft survival of patients with a single DSA (n = 26) with those who had multiple DSAs (n = 28). To determine whether high MFI levels influenced graft performance, we compared patients with very high MFI levels [MFI > 10000 (n = 24)] to patients with lower MFI levels [MFI < 10000 but > 1500 (n = 30)].
Not every patient with luminex testing received a liver biopsy, so we could not include every participant for histopathological analysis.
Initial immunosuppression (IS) within the first year and also maintenance therapy 1 year post-LT has already been described by our group[14]. In the present study population, patients mainly received immunosuppressive therapy with CNI (group 1: CSA n = 27; Tac n = 24; group 2: CSA n = 29; TAC n = 36) which was either monotherapy (group 1: 53.7%; group 2: 56.5%) or in combination with other medications. Detailed information is provided in Table 3. IS was modified if there were side effects or rejection episodes.
| Group 1 ‘DSA-positive’ | Group 2 ‘DSA-negative’ | |
| Monotherapy | n = 29 | n = 39 |
| CSA | n = 17 | n = 20 |
| Trough level | 109.5 µg/L (23-674 µg/L) median 73 µg/L | 58.0 µg/L (29-106 µg/L) |
| TAC | n = 10 | n = 17 |
| Trough level | 4.4µg/L (1.0-7.3 µg/L) | 5.5 µg/L (2.6-7.7 µg/L) |
| EVE | n = 1 | |
| Trough level | 10.3 µg/L | |
| SIR | n = 1 | |
| Trough level | 5.2 µg/L | |
| MMF | n = 1 | n = 1 |
| Combined therapy | n = 25 | n = 30 |
| CSA | ||
| + EVE | n = 3 | n = 4 |
| + MMF | n = 3 | n = 3 |
| + PRED | n = 2 | n = 1 |
| + EVE + PRED | n = 1 | |
| + MMF + PRED | n = 2 | |
| Trough level | 54.7µg/L (11-89 µg/L) | 72.7 µg/L (23-137 µg/L) |
| TAC | ||
| + EVE | n = 3 | n = 6 |
| + MMF | n = 5 | n = 7 |
| + PRED | n = 5 | n = 3 |
| + MMF + PRED | n = 1 | n = 3 |
| Trough level | 6.1 µg/L (3.0-9.9 µg/L) | 6.3 µg/L (2.1-15.1 µg/L) |
| EVE | ||
| + MMF | n = 1 | |
| + MMF + PRED | n = 1 | |
| Trough level | 3.7 µg/L (1.0-5.0 µg/L) | 4.3 µg/L (1.8-7.8 µg/L) |
| SIR | ||
| + MMF | n = 1 (trough levels, NA) | |
| Adherence rates | ||
| CSA | 81.5% | 72.4% |
| TAC | 87.5% | 88.9% |
| EVE | 75% | 84.9% |
| SIR | 100% | 100% |
There were 123 patients in the study. HLA antibodies were found in 74.1% of all patients (n = 106), and 43.9% (n = 54) presented with DSAs. The mean number of HLA antibodies per patient in group 1 was 10.9 (minimum of n = 1, maximum of n = 63, SD = 10.6) whereas group 2 had only 2.9 HLA antibodies per patient (minimum of n = 0, maximum of n = 33, SD = 6.2). The mean number of DSAs was 2.2 with a maximum of up to 6 DSAs per patient and graft (SD = 1.4).
All DSA-positive patients except one had DSAs of HLA class II (n = 53), while 14.8% (n = 8) had DSAs of both classes. Only one patient had DSAs exclusively in class I.
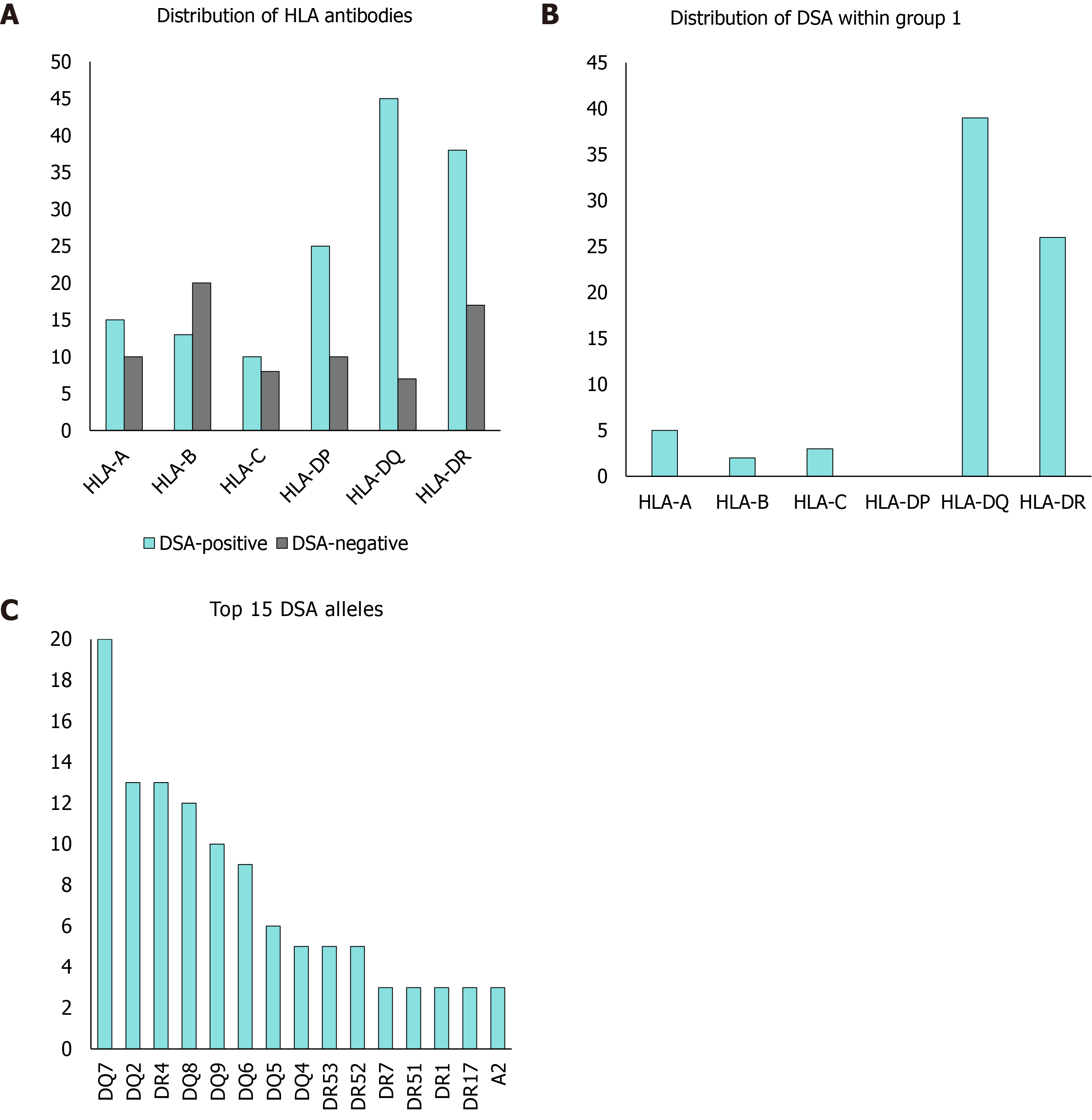
A detailed analysis of DSA HLA allele distribution showed that mainly HLA class II alleles, especially DR (n = 26 out of 54) and DQ (n = 39 out of 54) alleles were targeted. DP-HLA alleles could not be evaluated, because HLA donor typing was missing or incomplete for the majority of DP alleles. We could count these as HLA antibodies, but could not determine donor specificity. Nevertheless, the DSA-positive group showed a 46.3% prevalence of anti-HLA DP antibodies (n = 25 of 54), while the prevalence of these antibodies was only 14.5% (n = 10 of 69) in the DSA-negative group (Figure 1).
Liver biopsies (group 1 n = 38; group 2 n = 34) were mostly performed as protocol biopsies (67.3% in group 1 vs 85.7% in group 2), followed by suspected rejection (16.3% vs 10.7%). Recurrence of native disease was suspected in four children and confirmed in three (PFIC2 n = 1; AIH n = 1; PSC n = 1).
Comparing both groups, we found that fibrosis, portal inflammation, perivenular or perisinusoidal inflammation, ductular inflammation, biliary lesions/ductulopenia, ductular cholestasis, biliary tract strictures, single cell necrosis and chronic rejection were significantly more common in the DSA-positive group. Fibrosis was significantly correlated to class II HLA-DSAs (P < 0.001), especially to alleles DQ2 (P = 0.03), DQ7 (P < 0.001), DQ8 (P = 0.02) and DQ9 (P = 0.007). Low-grade fibrosis (F1 and F2) in particular was significantly higher in DSA-positive patients (P < 0.001) and was found in 17 routine protocol biopsies in group 1 (F1 and F2), whereas only 3 protocol biopsies showed signs of low-grade fibrosis (F1) in group 2.
We also found a higher incidence of high-grade fibrosis (F3), cirrhosis and endothelitis in group 1, although the difference was not significant. Steatosis, hepatocyte ballooning and other signs of toxic damage to the graft were either comparable or more likely to be found in group 2 (Table 2).
Correlation analysis showed a significant connection between biopsy-proven rejection and DSAs of HLA class II (P = 0.005), in particular against DQ2 (P = 0.02), DQ8 (P = 0.02) and DR52 (P = 0.03).
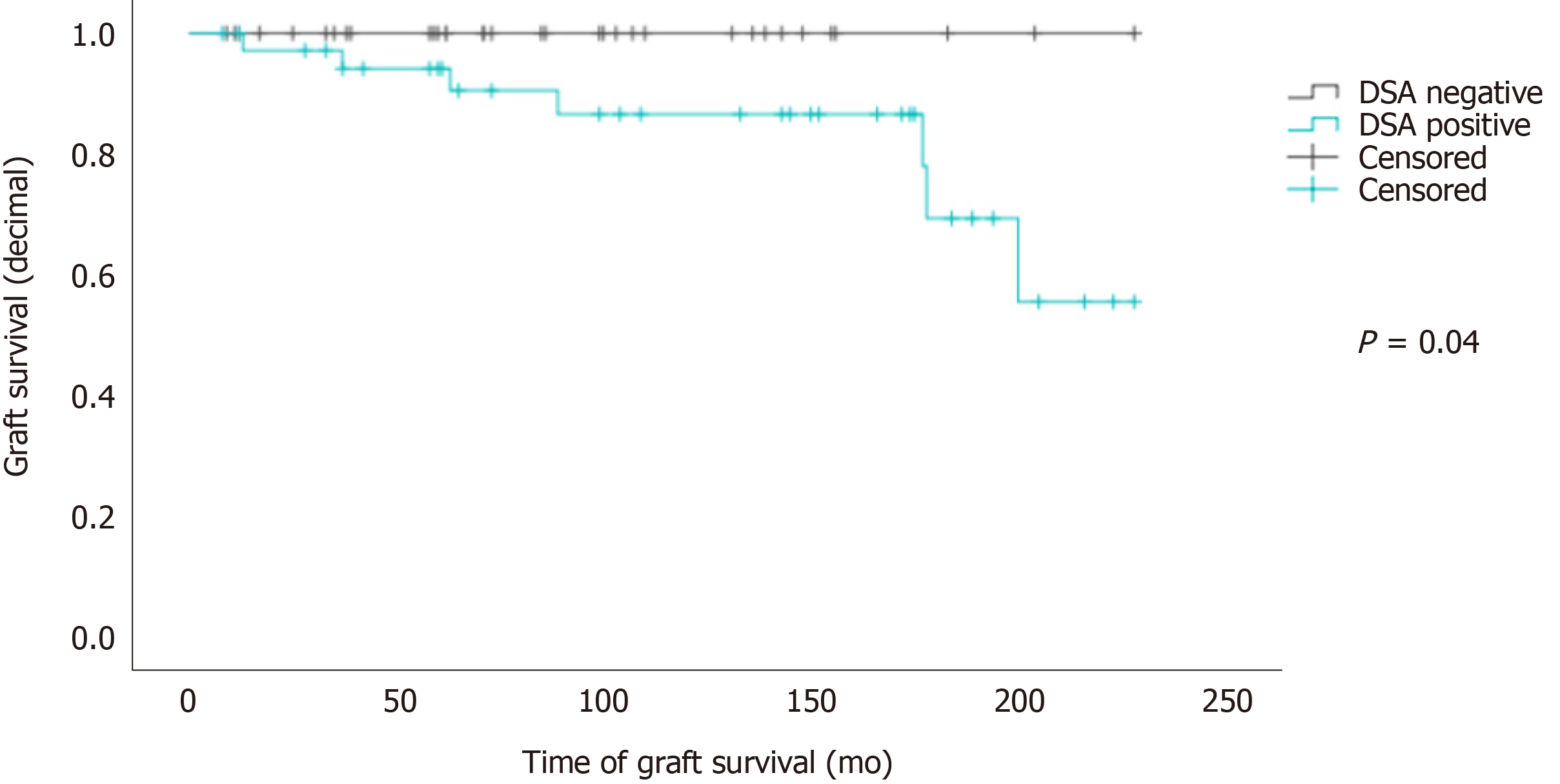
Overall graft survival according to Kaplan-Meier estimates was significantly lower for patients with DSAs (Mantel-Cox test: P = 0.04, Figure 2).
Clinical evaluation of liver enzymes, liver synthesis parameters and ultrasound criteria in CR-positive patients was not consistent with the histopathological status. Elevated levels of ALT and AST were only found in 10%-20% of CR-positive patients; γGT- and GLDH-elevation occurred in 50%-66%. None of these enzyme elevations reached statistical significance, nor did aberration of liver function parameters (albumin, bilirubin, international normalized ratio).
Histopathological indicators of possible cAMR were found in 9 patients of Group 1 (5 male, 4 female). Of these, three patients received monotherapy (CSA n = 2, MMF n = 1), while the other patients were treated with CNI in combination with EVE or MMF. Combined therapy was introduced if renal function was impaired or if there were previous signs of rejection. Trough levels were in the therapeutic range in all but two patients. Liver enzymes were normal except for elevated γGT in four patients. Alleles most targeted by DSAs belonged to HLA class II. DSAs against DQ alleles were particularly prevalent (DQ2 n = 2; DQ7 n = 4; DQ8 n = 4, DQ9 n = 3).
There was a significantly higher incidence of previous episodes of chronic rejection in our DSA-positive patients (P = 0.015). The rate of previous episodes of acute rejection was comparable in both groups.
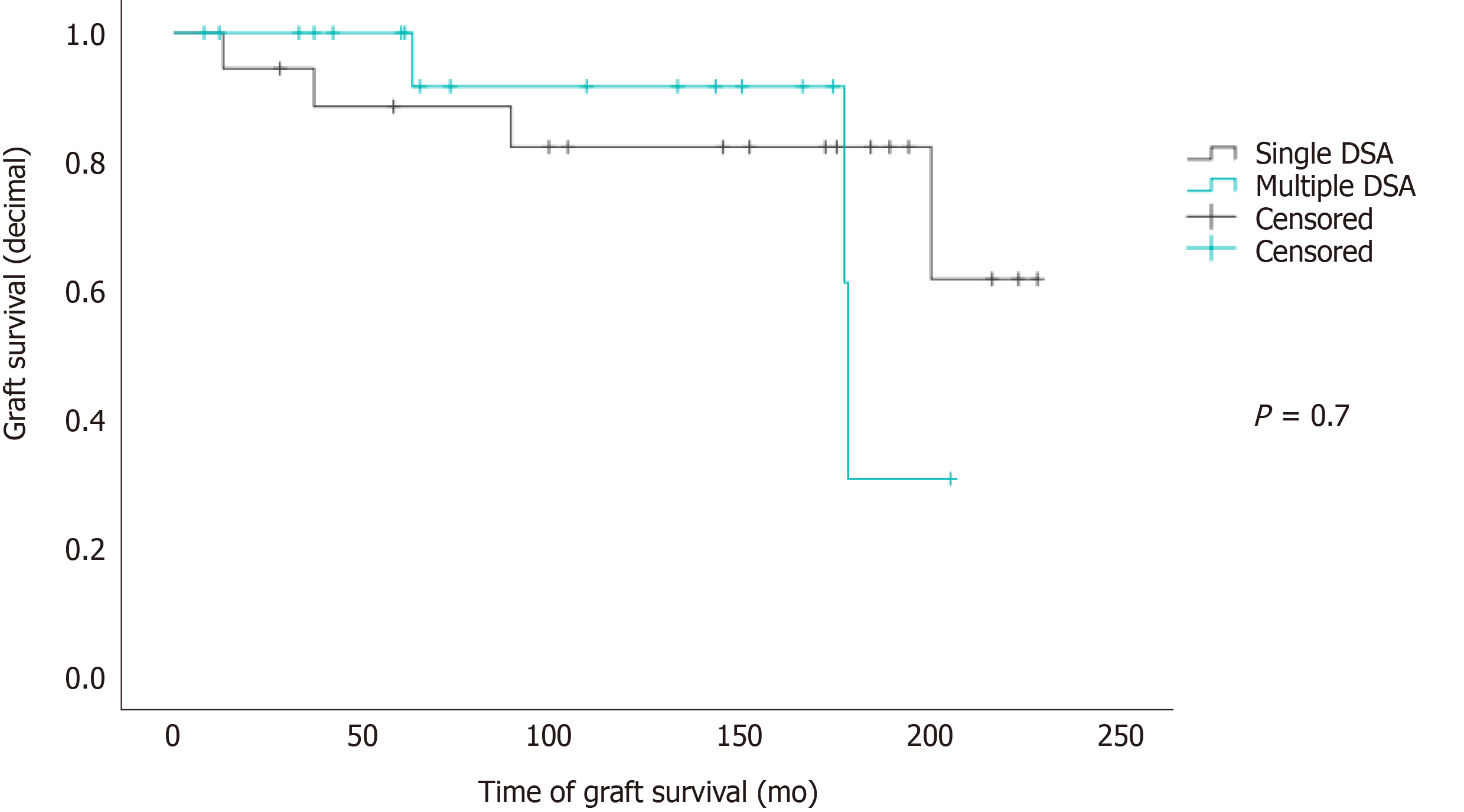
There was no correlation between DSA number and MFI levels with chronic rejection or re-LT. The Mantel-Cox test showed no significant influence of the number of DSAs on graft survival (P = 0.7; Figure 3).
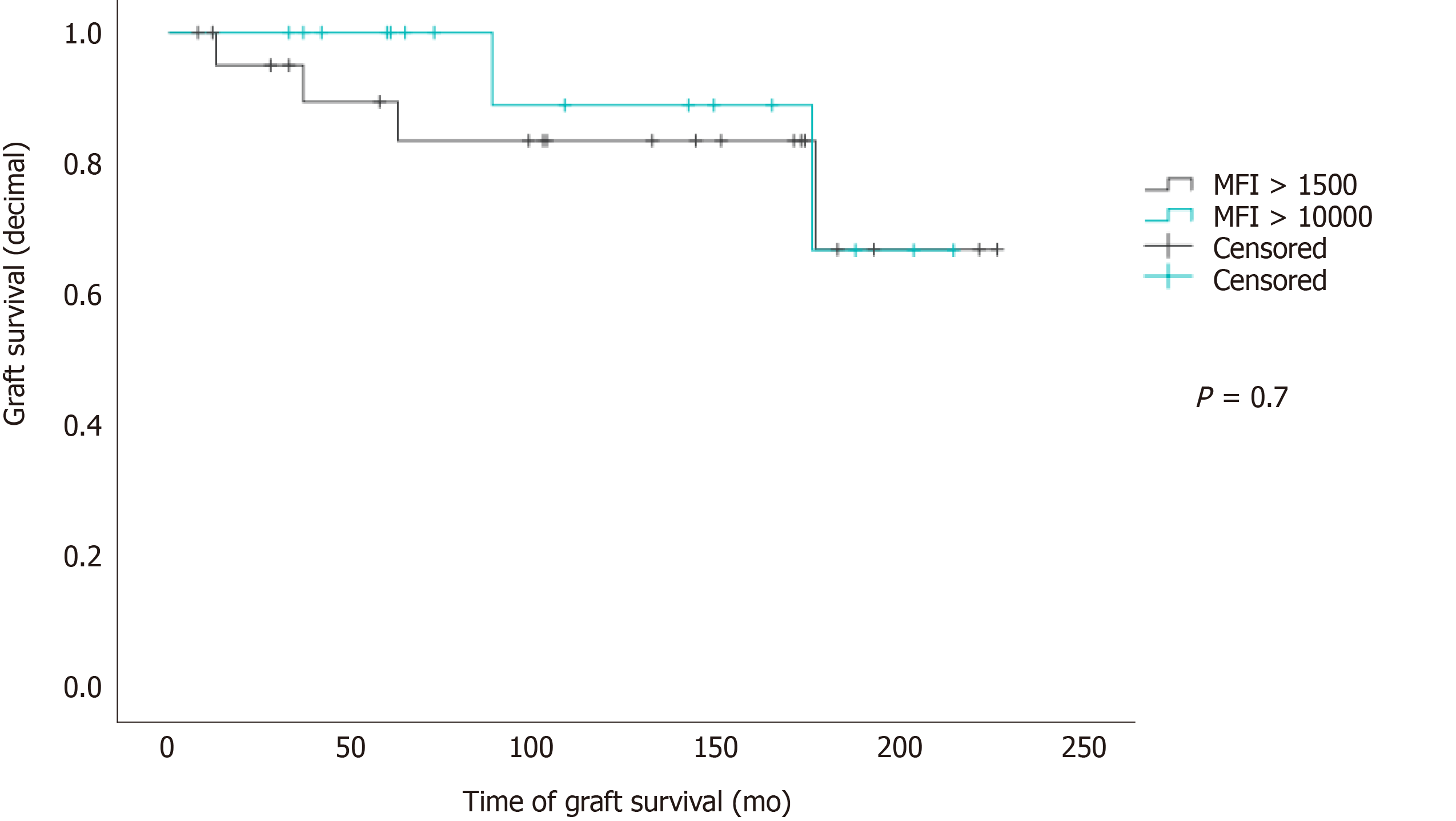
Single MFI levels ranged from 1668 to 31309 (cumulated MFI from 1678 to 120181; SD = 22333). Fibrosis, biopsy-proven chronic rejection, re-LT and numbers of re-LT were not significantly influenced by high MFI levels (MFI >10000 vs MFI > 1500 but < 10000). Graft survival was not significantly decreased in patients with high MFI levels (Mantel-Cox P = 0.7, Figure 4).
Comparing both groups, retransplantation was significantly more common in the DSA-positive group (n = 22 vs n = 14; P = 0.018). 79.9% of group 2 patients were able to maintain their first graft, but only 61.1% of group 1 maintained their initial grafts. The overall number of LTs was significantly higher in group 1 (1 to 4 LTs) than in Group 2 (1 to 3 LTs; P = 0.015). Also, the number of LTs was directly correlated with the presence of DSAs (P = 0.002). Retransplantation due to chronic rejection was significantly more common in group 1 (n = 9) than in group 2 (n = 2; P = 0.04).
The role of DSAs in the pathogenesis of chronic rejection in pediatric LT recipients has been subject to various studies but is still not fully understood. In a previous study, our group reported a higher prevalence of DSAs in patients with CR, although the statistical significance could not be determined due to the small cohort[14]. CR in DSA-positive patients was also described later by Wozniak et al[27] in a cross-sectional study of 50 pediatric patients.
The results of the present study show that histological indicators of CR have a significantly higher prevalence in DSA-positive patients, confirming O’Leary’s findings in a pediatric population and reaffirming the results of our previous study in a larger pediatric cohort. Furthermore, in all cases of biopsy-proven CR, patient sera were positive for DSAs. We also identified nine DSA-positive patients who possibly suffered from chronic antibody-mediated allograft rejection. Even biopsies of DSA-positive patients who received routine protocol biopsies and had no laboratory signs of impaired allograft function exhibited histological signs of fibrosis or rejection. This shows that correlating the aberration of laboratory parameters and CR or fibrosis is not a reliable clinical procedure. Ohlsson et al[29] recently confirmed the value of protocol biopsies in detecting silent immune-mediated allograft injuries, regularly associated with the presence of DSAs.
As this study had a cross-sectional design, we could not examine the development of DSAs over the full study period, especially after rejection episodes. Nevertheless, previous episodes of CR with the same graft (prior to the current follow-up, which did not lead to graft loss or re-LT) were significantly more common in our DSA-positive patients (P = 0.015).
Although there is growing evidence that DSAs impact graft survival, it is still unclear whether DSA number or quality matter. While Couchonnal et al[30] reported poorer long-term graft survival in patients with high MFI (> 10000), Wozniak et al[27] described a significantly higher impact of the overall presence of DQ-DSAs on graft survival, as opposed to the presence of any DSAs with high MFI levels (threshold > 13000).
We also used MFI levels of > 10000 to identify strong DSA effects, but we found no statistically significant impact of MFI levels on biopsy-proven CR, graft survival or need for re-transplantation. However, anti-HLA class II antibodies and especially anti-HLA-DQ2 and -DQ8-antibodies were significantly correlated with graft survival. We therefore support Demetris’ ‘second-hit’ hypothesis and regard it as probable that class II HLA-DSAs influence TCMR, cAMR and probably the need for reLT. This is supported by a significant influence of the presence of DSAs on graft survival in survival analysis. The use of anti-HLA-DQ2 and -DQ8-antibodies as screening markers needs to be assessed with further prospective studies.
Mild to moderate allograft fibrosis is a common finding in protocol biopsies obtained 5-10 years post-LT. So far, low IS trough levels or even weaning off are thought to promote the development of such fibrosis by enabling alloreactivity that leads to allograft inflammation[31] and fibrosis, with possible later development of cirrhosis and graft failure[17,20,32,33]. According to Briem-Richter et al[31], increasing IS dosage results in the resolution of histopathological signs of rejection and severity of fibrosis. In this present study, we found that portal, perivenular or perisinusoidal inflammation is very common in DSA-positive patients. Also, mild to moderate allograft fibrosis (grade 1-2) was significantly more common in DSA-positive patients. We therefore consider DSA presence as a symptom of such alloreactivity; this might help to identify children who are poor candidates for reducing IS levels.
Correlation between DSA and cirrhosis did not reach statistical significance, probably because of the small number of cirrhosis patients confirmed with histopathology (DSA-positive patients: n = 3 vs DSA-negative patients: n = 0).
The presence of HLA class II DSAs, especially anti-HLA-DQ antibodies, coincided significantly with allograft fibrosis. Furthermore, we were able to identify four specific HLA-DQ alleles that might serve as serological markers or have predictive value: DQ2, DQ7, DQ8 and DQ9.
As a liver biopsy was not performed for every study participant, one might argue that there is selection of patients with poorer graft performance, leading to a biased correlation of DSA presence with CR. However, the general group of poor allograft performance is still relatively small, thus reversing the suspected bias with an overall representative selection of the study population. Other limitations of this study were the incomplete HLA typing of DP alleles, errors in sampling, and missing HLA donor typing in general, which led to exclusion of participants. Also, C4d staining was not performed on liver biopsies that were taken prior to the updated Banff criteria in 2016, so that these biopsies could not be fully included in the evaluation. As these are parts of the general restrictions of retrospective studies, we plan to conduct a prospective clinical trial to assess the new issues that this study has raised.
Long-term allograft survival is even more valuable in pediatric LT than in adult LT, and with the decreased graft survival and increased prevalence of allograft dysfunction and retransplantation in DSA-positive patients, this important subject should not be underestimated.
Screening for DSA must be included in follow-ups to ensure identification of patients at risk of potential graft loss (rejection), and may be even more relevant for patients with early signs of allograft dysfunction (fibrosis). Moreover, patients with DSA might not be good candidates for reduction of IS or even weaning. According to our results, in the presence of DSA, selected patients should be considered for additional graft biopsies including assessment with Banff chronic cAMR criteria after C4d-staining, since routine laboratory parameters are not sufficiently accurate for monitoring the allograft status and cannot identify patients with silent immune-mediated allograft injuries. Whether the latter could be detected by ultrasound elastography could be the subject of a future clinical trial.
Since HLA class I DSAs are less common and have less impact on allograft fibrosis or rejection, screening could be limited to HLA class II DSA (-DQ, -DR -DP).
An impact of donor-specific antibodies (DSAs) on long-term liver allograft survival was found previously in a small cohort of pediatric patients, but the statistical significance was unclear.
The aim of this study was to clarify the importance of DSAs on long-term graft survival in a larger cohort of pediatric patients.
The objective of this study was to emphasize the importance of comprehensive follow-up examinations in clinical practice after pediatric liver transplantation (LT) and contribute to optimizing and standardizing LT follow-ups.
This was a cross-sectional retrospective cohort study that compared the outcomes of two patient groups after pediatric LT.
Our study showed that DSAs significantly impact liver allograft survival. The presence of human leucocyte antigen class II DSAs is associated with chronic rejection, chronic antibody-mediated rejection, graft fibrosis, graft failure, graft loss and re-LT.
Screening of DSAs and protocol liver biopsies including C4d immunostaining should be standard practice in follow-ups after pediatric LT.
Further prospective studies should be conducted to explore whether certain DQ-DSAs could be used as a serological marker for the risk of graft loss.
Manuscript source: Invited manuscript
Specialty type: Transplantation
Country/Territory of origin: Germany
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ferrarese A S-Editor: Zhang H L-Editor: A P-Editor: Li JH
| 1. | Hollander SA, Peng DM, Mills M, Berry GJ, Fedrigo M, McElhinney DB, Almond CS, Rosenthal DN. Pathological antibody-mediated rejection in pediatric heart transplant recipients: Immunologic risk factors, hemodynamic significance, and outcomes. Pediatr Transplant. 2018;22:e13197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 2. | Engen RM, Park GE, Schumacher CS, Gimferrer I, Warner P, Finn LS, Weiss NS, Smith JM. Donor-specific Antibody Surveillance and Graft Outcomes in Pediatric Kidney Transplant Recipients. Transplantation. 2018;102:2072-2079. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 3. | Barten MJ, Schulz U, Beiras-Fernandez A, Berchtold-Herz M, Boeken U, Garbade J, Hirt S, Richter M, Ruhpawar A, Sandhaus T, Schmitto JD, Schönrath F, Schramm R, Schweiger M, Wilhelm M, Zuckermann A. The clinical impact of donor-specific antibodies in heart transplantation. Transplant Rev (Orlando). 2018;32:207-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 53] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 4. | Vandevoorde K, Ducreux S, Bosch A, Guillaud O, Hervieu V, Chambon-Augoyard C, Poinsot D, André P, Scoazec JY, Robinson P, Boillot O, Dubois V, Dumortier J. Prevalence, Risk Factors, and Impact of Donor-Specific Alloantibodies After Adult Liver Transplantation. Liver Transpl. 2018;24:1091-1100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 5. | Kaneku H, O'Leary JG, Banuelos N, Jennings LW, Susskind BM, Klintmalm GB, Terasaki PI. De novo donor-specific HLA antibodies decrease patient and graft survival in liver transplant recipients. Am J Transplant. 2013;13:1541-1548. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 65] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 6. | San Segundo D, Alonso C, Ruiz P, Romon I, Arias-Loste MT, Cuadrado A, Puente A, Casafont F, López-Hoyos M, Crespo J, Fábrega E. De Novo Donor-Specific Anti-Human Leukocyte Antigen Antibody Detection in Long-Term Adult Liver Transplantation. Transplant Proc. 2016;48:2980-2982. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 7. | Musat AI, Agni RM, Wai PY, Pirsch JD, Lorentzen DF, Powell A, Leverson GE, Bellingham JM, Fernandez LA, Foley DP, Mezrich JD, D'Alessandro AM, Lucey MR. The significance of donor-specific HLA antibodies in rejection and ductopenia development in ABO compatible liver transplantation. Am J Transplant. 2011;11:500-510. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 159] [Cited by in RCA: 143] [Article Influence: 10.2] [Reference Citation Analysis (0)] |
| 8. | Donaldson PT, Thomson LJ, Heads A, Underhill JA, Vaughan RW, Rolando N, Williams R. IgG donor-specific crossmatches are not associated with graft rejection or poor graft survival after liver transplantation. An assessment by cytotoxicity and flow cytometry. Transplantation. 1995;60:1016-1023. [PubMed] |
| 9. | Lobo PI, Spencer C, Douglas MT, Stevenson WC, Pruett TL. The lack of long-term detrimental effects on liver allografts caused by donor-specific anti-HLA antibodies. Transplantation. 1993;55:1063-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 34] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 10. | Demetris AJ, Bellamy C, Hübscher SG, O'Leary J, Randhawa PS, Feng S, Neil D, Colvin RB, McCaughan G, Fung JJ, Del Bello A, Reinholt FP, Haga H, Adeyi O, Czaja AJ, Schiano T, Fiel MI, Smith ML, Sebagh M, Tanigawa RY, Yilmaz F, Alexander G, Baiocchi L, Balasubramanian M, Batal I, Bhan AK, Bucuvalas J, Cerski CTS, Charlotte F, de Vera ME, ElMonayeri M, Fontes P, Furth EE, Gouw ASH, Hafezi-Bakhtiari S, Hart J, Honsova E, Ismail W, Itoh T, Jhala NC, Khettry U, Klintmalm GB, Knechtle S, Koshiba T, Kozlowski T, Lassman CR, Lerut J, Levitsky J, Licini L, Liotta R, Mazariegos G, Minervini MI, Misdraji J, Mohanakumar T, Mölne J, Nasser I, Neuberger J, O'Neil M, Pappo O, Petrovic L, Ruiz P, Sağol Ö, Sanchez Fueyo A, Sasatomi E, Shaked A, Shiller M, Shimizu T, Sis B, Sonzogni A, Stevenson HL, Thung SN, Tisone G, Tsamandas AC, Wernerson A, Wu T, Zeevi A, Zen Y. 2016 Comprehensive Update of the Banff Working Group on Liver Allograft Pathology: Introduction of Antibody-Mediated Rejection. Am J Transplant. 2016;16:2816-2835. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 496] [Cited by in RCA: 431] [Article Influence: 47.9] [Reference Citation Analysis (0)] |
| 11. | Ballardini G, Bianchi FB, Mirakian R, Fallani M, Pisi E, Bottazzo GF. HLA-A,B,C, HLA-D/DR and HLA-D/DQ expression on unfixed liver biopsy sections from patients with chronic liver disease. Clin Exp Immunol. 1987;70:35-46. [PubMed] |
| 12. | Lautenschlager I, Taskinen E, Inkinen K, Lehto VP, Virtanen I, Häyry P. Distribution of the major histocompatibility complex antigens on different cellular components of human liver. Cell Immunol. 1984;85:191-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 44] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 13. | Ueno T, Zenitani M, Yamanaka H, Tanaka N, Uehara S, Tazuke Y, Bessho K, Okuyama H. Impact of Donor-Specific Antibodies on Graft Fibrosis After Pediatric Living Donor Liver Transplantation for Biliary Atresia. Transplant Proc. 2016;48:1095-1099. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 14. | Grabhorn E, Binder TM, Obrecht D, Brinkert F, Lehnhardt A, Herden U, Peine S, Nashan B, Ganschow R, Briem-Richter A. Long-term Clinical Relevance of De Novo Donor-Specific Antibodies After Pediatric Liver Transplantation. Transplantation. 2015;99:1876-1881. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 50] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 15. | Tokodai K, Miyagi S, Nakanishi C, Hara Y, Nakanishi W, Miyazawa K, Shimizu K, Murakami K, Sasano H, Goto M, Unno M, Kamei T. Association of post-transplant donor-specific HLA antibody with liver graft fibrosis during long-term follow-up after pediatric liver transplantation. Pediatr Transplant. 2018;22:e13169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 16. | Egawa H, Miyagawa-Hayashino A, Haga H, Teramukai S, Yoshizawa A, Ogawa K, Ogura Y, Okamoto S, Kaido T, Uemoto S. Non-inflammatory centrilobular sinusoidal fibrosis in pediatric liver transplant recipients under tacrolimus withdrawal. Hepatol Res. 2012;42:895-903. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 41] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 17. | Yamada H, Kondou H, Kimura T, Ikeda K, Tachibana M, Hasegawa Y, Kiyohara Y, Ueno T, Miyoshi Y, Mushiake S, Ozono K. Humoral immunity is involved in the development of pericentral fibrosis after pediatric live donor liver transplantation. Pediatr Transplant. 2012;16: 858-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 34] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 18. | Markiewicz-Kijewska M, Kaliciński P, Kluge P, Piątosa B, Jankowska I, Rękawek A, Kostecka E, Kurowski PN. Immunological factors and liver fibrosis in pediatric liver transplant recipients. Ann Transplant. 2015;20:279-284. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 19. | Guerra MR, Naini BV, Scapa JV, Reed EF, Busuttil RW, Cheng EY, Farmer DG, Vargas JH, Venick RS, McDiarmid SV, Wozniak LJ. Obliterative portal venopathy: A histopathologic finding associated with chronic antibody-mediated rejection in pediatric liver allografts. Pediatr Transplant. 2018;22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 13] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 20. | Dao M, Habès D, Taupin JL, Mussini C, Redon MJ, Suberbielle C, Jacquemin E, Gonzales E, Guettier C. Morphological characterization of chronic antibody-mediated rejection in ABO-identical or ABO-compatible pediatric liver graft recipients. Liver Transpl. 2018;24:897-907. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 17] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 21. | schema for grading liver allograft rejection: an international consensus document. Hepatology. 1997;25:658-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1016] [Cited by in RCA: 1002] [Article Influence: 35.8] [Reference Citation Analysis (1)] |
| 22. | Demetris A, Adams D, Bellamy C, Blakolmer K, Clouston A, Dhillon AP, Fung J, Gouw A, Gustafsson B, Haga H, Harrison D, Hart J, Hubscher S, Jaffe R, Khettry U, Lassman C, Lewin K, Martinez O, Nakazawa Y, Neil D, Pappo O, Parizhskaya M, Randhawa P, Rasoul-Rockenschaub S, Reinholt F, Reynes M, Robert M, Tsamandas A, Wanless I, Wiesner R, Wernerson A, Wrba F, Wyatt J, Yamabe H. Update of the International Banff Schema for Liver Allograft Rejection: working recommendations for the histopathologic staging and reporting of chronic rejection. An International Panel. Hepatology. 2000;31:792-799. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 360] [Article Influence: 14.4] [Reference Citation Analysis (1)] |
| 23. | Banff Working Group; Demetris AJ, Adeyi O, Bellamy CO, Clouston A, Charlotte F, Czaja A, Daskal I, El-Monayeri MS, Fontes P, Fung J, Gridelli B, Guido M, Haga H, Hart J, Honsova E, Hubscher S, Itoh T, Jhala N, Jungmann P, Khettry U, Lassman C, Ligato S, Lunz JG 3rd, Marcos A, Minervini MI, Mölne J, Nalesnik M, Nasser I, Neil D, Ochoa E, Pappo O, Randhawa P, Reinholt FP, Ruiz P, Sebagh M, Spada M, Sonzogni A, Tsamandas AC, Wernerson A, Wu T, Yilmaz F. Liver biopsy interpretation for causes of late liver allograft dysfunction. Hepatology. 2006;44:489-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 234] [Article Influence: 12.3] [Reference Citation Analysis (0)] |
| 24. | O'Leary JG, Demetris AJ, Friedman LS, Gebel HM, Halloran PF, Kirk AD, Knechtle SJ, McDiarmid SV, Shaked A, Terasaki PI, Tinckam KJ, Tomlanovich SJ, Wood KJ, Woodle ES, Zachary AA, Klintmalm GB. The role of donor-specific HLA alloantibodies in liver transplantation. Am J Transplant. 2014;14:779-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 162] [Article Influence: 14.7] [Reference Citation Analysis (0)] |
| 25. | Hübscher SG. Antibody-mediated rejection in the liver allograft. Curr Opin Organ Transplant. 2012;17:280-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 26. | Wozniak LJ, Naini BV, Hickey MJ, Bhattacharyya S, Reed EF, Busuttil RW, Farmer DG, Vargas JH, Venick RS, McDiarmid SV. Acute antibody-mediated rejection in ABO-compatible pediatric liver transplant recipients: case series and review of the literature. Pediatr Transplant. 2017;21. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 27. | Wozniak LJ, Hickey MJ, Venick RS, Vargas JH, Farmer DG, Busuttil RW, McDiarmid SV, Reed EF. Donor-specific HLA Antibodies Are Associated With Late Allograft Dysfunction After Pediatric Liver Transplantation. Transplantation. 2015;99:1416-1422. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 98] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 28. | Demetris AJ, Zeevi A, O'Leary JG. ABO-compatible liver allograft antibody-mediated rejection: an update. Curr Opin Organ Transplant. 2015;20:314-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 43] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 29. | Ohlsson S, Kathemann S, Pilic D, Prusinskas B, Baba HA, Theurer S, Dechene A, Paul A, Heinold A, Hoyer PF, Lainka E. Protocol liver biopsies in stable long-term pediatric liver transplant recipients: risk or benefit? Eur J Gastroenterol Hepatol. 2021;Publish ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 30. | Couchonnal E, Rivet C, Ducreux S, Dumortier J, Bosch A, Boillot O, Collardeau-Frachon S, Dubois R, Hervieu V, André P, Scoazec JY, Lachaux A, Dubois V, Guillaud O. Deleterious impact of C3d-binding donor-specific anti-HLA antibodies after pediatric liver transplantation. Transpl Immunol. 2017;45:8-14. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 31. | Briem-Richter A, Ganschow R, Sornsakrin M, Brinkert F, Schirmer J, Schaefer H, Grabhorn E. Liver allograft pathology in healthy pediatric liver transplant recipients. Pediatr Transplant. 2013;17:543-549. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 25] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 32. | Kivelä JM, Kosola S, Peräsaari J, Mäkisalo H, Jalanko H, Holmberg C, Pakarinen MP, Lauronen J. Donor-specific antibodies after pediatric liver transplantation: a cross-sectional study of 50 patients. Transpl Int. 2016;29:494-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 33. | Cheng EY. The Role of Humoral Alloreactivity in Liver Transplantation: Lessons Learned and New Perspectives. J Immunol Res. 2017;2017:3234906. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 14] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |