Published online May 27, 2021. doi: 10.4254/wjh.v13.i5.611
Peer-review started: January 2, 2021
First decision: January 18, 2021
Revised: February 4, 2021
Accepted: April 9, 2021
Article in press: April 9, 2021
Published online: May 27, 2021
Processing time: 137 Days and 22.6 Hours
Metastatic small bowel low-grade neuroendocrine tumors (NETs) have a good prognosis. Surgery is the only curative treatment; however, this may induce advanced liver disease, particularly in long-term survivor patients. Acquired hepatocerebral degeneration or Parkinsonism in cirrhosis is characterized by rapidly progressive extrapyramidal symptoms in patients with advanced liver disease.
A 70-year-old man presented to the emergency department with diminished consciousness and disorientation, and was diagnosed with hepatic encephalopathy. The patient was diagnosed in 1993 with a metastatic small bowel NET, for which he twice underwent hepatic surgery, with metastatic resection in 1993 and a right hepatectomy in 2002 to remove two hepatic metastases. In 2003, the patient started first-line chemotherapy and in 2004 started the first of three consecutive biological treatments, followed by radio-molecular therapy, achieving stable disease for 14 years. Disease progression was identified and he underwent an endoscopic retrograde cholangiopancreatography. However, in 2019 advanced liver disease was identified. We diagnosed the development of acquired hepatocerebral degeneration, an unusual long-term side effect after multiple hepatic procedures.
The importance of regular and ongoing surveillance in long-term NET survivors who undergo hepatic procedures should be integrated into the therapeutic management plan, as some of these negative outcomes could be prevented.
Core Tip: To the best of our knowledge, this is the first case report of acquired hepatocerebral degeneration in a metastatic small bowel neuroendocrine tumor long-term survivor, an uncommon irreversible extrapyramidal neurodegenerative condition encountered in patients with cirrhotic chronic liver disease, and resulting in widespread cerebral, basal ganglia, and cerebellar damage.
- Citation: Mirallas O, Saoudi N, Gómez-Puerto D, Riveiro-Barciela M, Merino X, Auger C, Landolfi S, Blanco L, Garcia-Burillo A, Molero X, Salcedo-Allende MT, Capdevila J. Acquired hepatocerebral degeneration in a metastatic neuroendocrine tumor long-term survivor — an update on neuroendocrine neoplasm’s treatment: A case report. World J Hepatol 2021; 13(5): 611-619
- URL: https://www.wjgnet.com/1948-5182/full/v13/i5/611.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i5.611
Neuroendocrine neoplasms (NENs) are a rare group of cancers accounting for about 0.05% of all newly diagnosed malignancies and 0.5% of all gastrointestinal and lung malignancies[1-3]. Nonetheless, the incidence rate increased 6.4-fold from 1973 to 2012[2,4]. NENs are a heterogeneous group of malignancies with a slightly higher female preponderance, and are most commonly found in the gastrointestinal tract and lungs[5].
The neuroendocrine system encompasses not only the endocrine glands but is also scattered throughout the exocrine parenchyma, the so-called diffuse endocrine system[6,7]. Histologically, NENs are clustered into two main groups. On one hand, neuroendocrine tumors (NETs) are typically well-differentiated tumors characterized by uniform nuclei with dense granules, histologically described as “salt and pepper.” By contrast, neuroendocrine carcinomas have a poorly defined phenotype with a high mitotic index, and up to 40% do not express neuroendocrine markers[6,7]. Diagnosis confirmation must always be accompanied by a biopsy of the primary tumor or metastases. The 2017 World Health Organization classification takes into account the grade of differentiation and the Ki-67 mitotic proliferation index, distinguishing four groups; G1, G2 and G3 NETs and neuroendocrine carcinomas. Ki-67 grading is an important prognostic factor, and is therefore a mandatory biomarker in pathological reporting[8-10].
Liver metastases represent another crucial prognostic factor. Surgery of metastases is the only treatment that offers a cure[11]. For unresectable lesions, there are multiple treatment options such as somatostatin analogues (SSA), interferon α, local liver therapies, chemotherapy, peptide-receptor radionuclide therapy, angiogenesis inhibitors, and mammalian rapamycin inhibitors. SSA have both anti-secretory and antiproliferative effects, improving progression-free survival in both the PROMID trial (octreotide LAR vs placebo) and the CLARINET (lanreotide vs placebo) trial[12,13]. The NETTER-1 trial reported prolongation of progression-free survival (PFS) after treatment with 177Lu-Dotatate compared to treatment with octreotide in patients with a well-differentiated midgut-NET[14]. Notably in the case of gastrointestinal NETs, the certainty of evidence is highest for the combination of SSA plus 177Lu-dotatate[15].
Nonetheless, the downside of these options is that many of these treatments can result in injury of healthy liver parenchyma and development of sinusoid liver fibrosis, and consequently induce portal hypertension with progression to advanced liver disease. The main complications of chronic liver disease are hepatocellular carcinoma and portal hypertension[16,17]. According to a study published in 2013 by Tryc et al[17], about 4% of cirrhotic patients develop progressive hypokinetic-rigid syndrome, which is not present in hepatic encephalopathy, recently referred to in the literature as “cirrhosis-related-Parkinsonism” or “acquired hepatocerebral degeneration (AHD).” The most commonly reported symptoms of patients with AHD are bradykinesia, cerebellar symptoms, tremor, and rigidity[16,18,19].
It has been hypothesized that AHD originates from increased manganese deposits in the basal ganglia, particularly in the globus pallidum, damaging the presynaptic dopamine transporters and post-synaptic dopamine receptors in cirrhotic patients[17,20,21]. Treatment with levodopa can be effective when D2 receptors are available[17,22]. The study by Rose et al[20] analyzing postmortem human brain tissue, demonstrated an increase in manganese deposits in several brain structures of cirrhotic patients. The two main causes of increased manganese deposits that the authors found to be statistically significant resulted both from portocaval-shunt and liver dysfunction[20]. This manuscript is the first case in the literature to report AHD in a metastatic gastrointestinal NET long-term survivor.
A 70-year-old male patient presented in January 2019 to the emergency department with diminished consciousness and disorientation, without any other relevant symptoms.
Neurological symptoms were first reported in May 2017 and asymmetric Parkinson’s disease diagnosed in June 2018 for which he received levodopa.
The patient had a medical history of high blood pressure, which was treated with diuretics. The patient denied use of potentially hepatotoxic drugs. He also had diabetes mellitus type 2 treated with metformin, without any other cardiovascular risk factors. He underwent a gastrectomy and Billroth II reconstruction in March 1993 for a gastric ulcer.
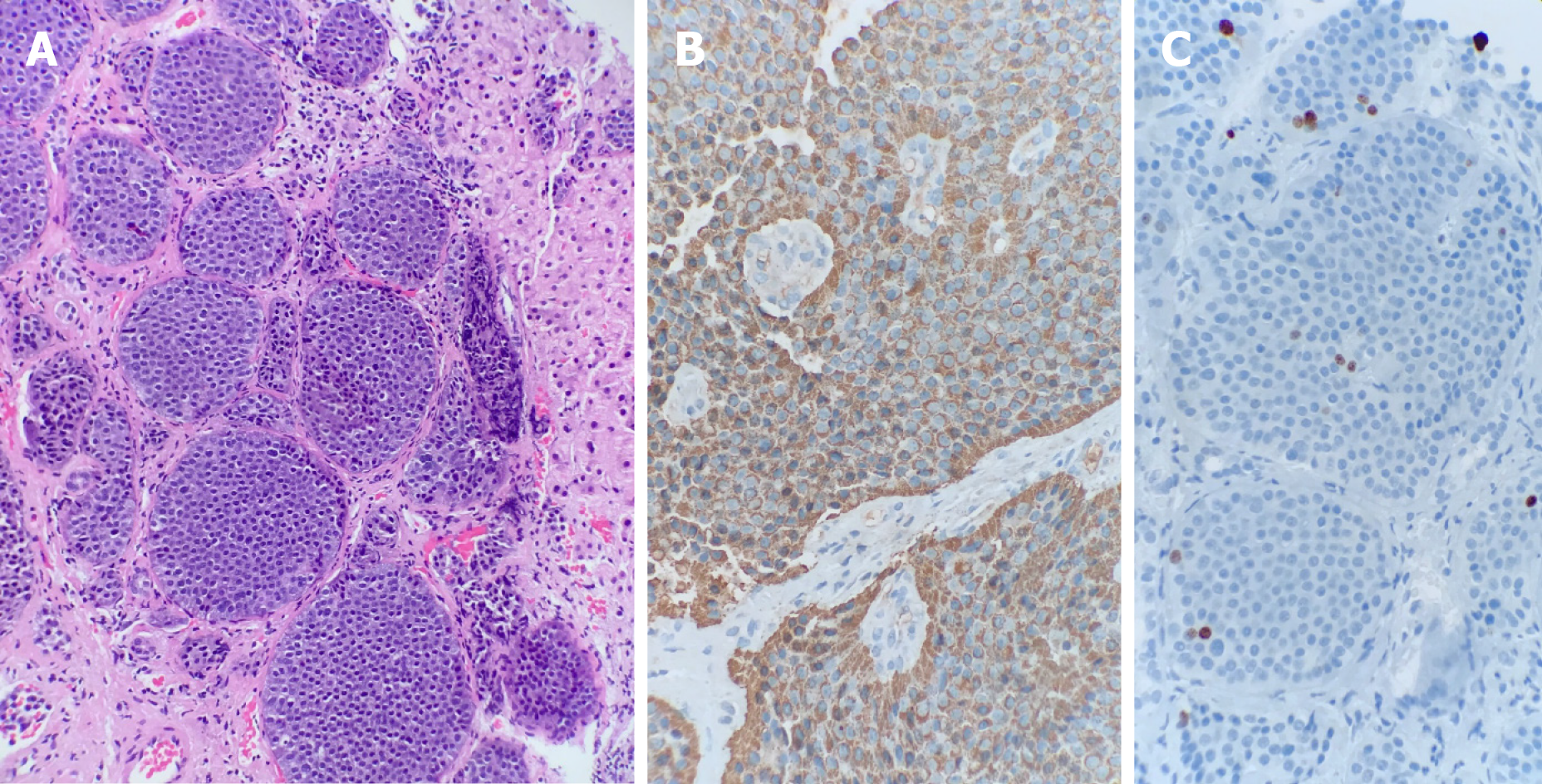
His oncological history started in 1993 when he was diagnosed with a metastatic midgut NET confirmed by a hepatic biopsy, in which the anatomic pathology reported a well-differentiated tumor with a Ki-67 expression of 1.26%, graded as a G1 tumor (Figure 1), and without hepatic enzyme alterations. He subsequently underwent two hepatic procedures: a single metastasis resection from the right hepatic lobe in July 1993 and a right hepatectomy was performed in 2002 to remove two hepatic metastases. There were no changes to laboratory data or computed tomography (CT) scans after both procedures.
In November 2003, the patient started first-line chemotherapy with streptozotocin and doxorubicin after a new hepatic lesion appeared. In 2004, the patient showed hepatic progression and began treatment with the biological agent octreotide. He achieved stable disease lasting until 2010, when a CT scan showed a new hepatic lesion in the surgical bed, three sub-centimeter hepatic lesions, and a new adenopathy in the hepatic hilum. The treatment was discontinued. Then the patient participated in the RAMSETE clinical trial evaluating the efficacy of everolimus at 10 mg daily in non-functioning extrapancreatic NETs, and was randomized to the active treatment arm. The lesions showed tumor shrinkage with a total reduction of 23%, corresponding to stable disease per RECIST v1.1. The only side effects were low platelet counts and grade 1 pneumonitis (CTCAE v5.0). After 5 years with stable disease, a CT scan showed progressive disease in July 2015, and he changed to a third biological treatment, lanreotide at 120 mg monthly, again achieving stable disease as the best response.
In June 2017, he presented to the emergency department with cholangitis due to extrinsic compression of the bile duct from hepatic lesions, and a choledochal stent was inserted by endoscopic retrograde cholangiopancreatography. The CT scan showed progressive disease in the liver. A somatostatin receptor scintigraphy revealed liver, hepatic hilum and peritoneal uptake, and he started peptide receptor radionuclide therapy (PRRT) with 177Lu-Dotatate for three sessions and 30 mg octreotide LAR monthly, achieving partial response as the best response. One year later in July 2018, the patient returned to the emergency room with a new episode of cholangitis. Hepatic magnetic resonance imaging (MRI) showed extensive progression in the surgical bed, invading the biliary stent and causing partial obstruction. A new endoscopic retrograde cholangiopancreatography was performed to unblock the bile duct and antibiotic treatment was administered. At discharge, the patient continued on octreotide LAR, and had an ongoing best response of stable disease at the time of presentation to the emergency department in January 2019.
It should be noted that in May 2017, the patient began to complain of memory loss and distal tremor, but it was not until June 2018 that he was diagnosed with asymmetric Parkinson’s disease by a neurologist. At this time, the CT scan showed heterogeneous liver with signs of portal hypertension. The patient began treatment with levodopa for Parkinson’s disease, without a significant clinical response.
There was no relevant family history.
On physical examination, he presented with flapping, jaundice, facial amimia, and cogwheel rigidity in both arms. There were no signs of Kayser-Fleischer rings. All other neurological examinations were normal.
Blood tests showed low platelet count, low albumin and evidence of cholestasis, with no other relevant alterations. Urine sediment and blood culture were negative, ruling out an infectious cause. The patient presented with Child-Pugh B and MELD 8 at admission. Ceruloplasmin was within normal ranges. There were no findings of hypovitaminosis, dyselectrolytemia, hypothyroidism, hepatitis virus serologies, or autoimmunity tests.
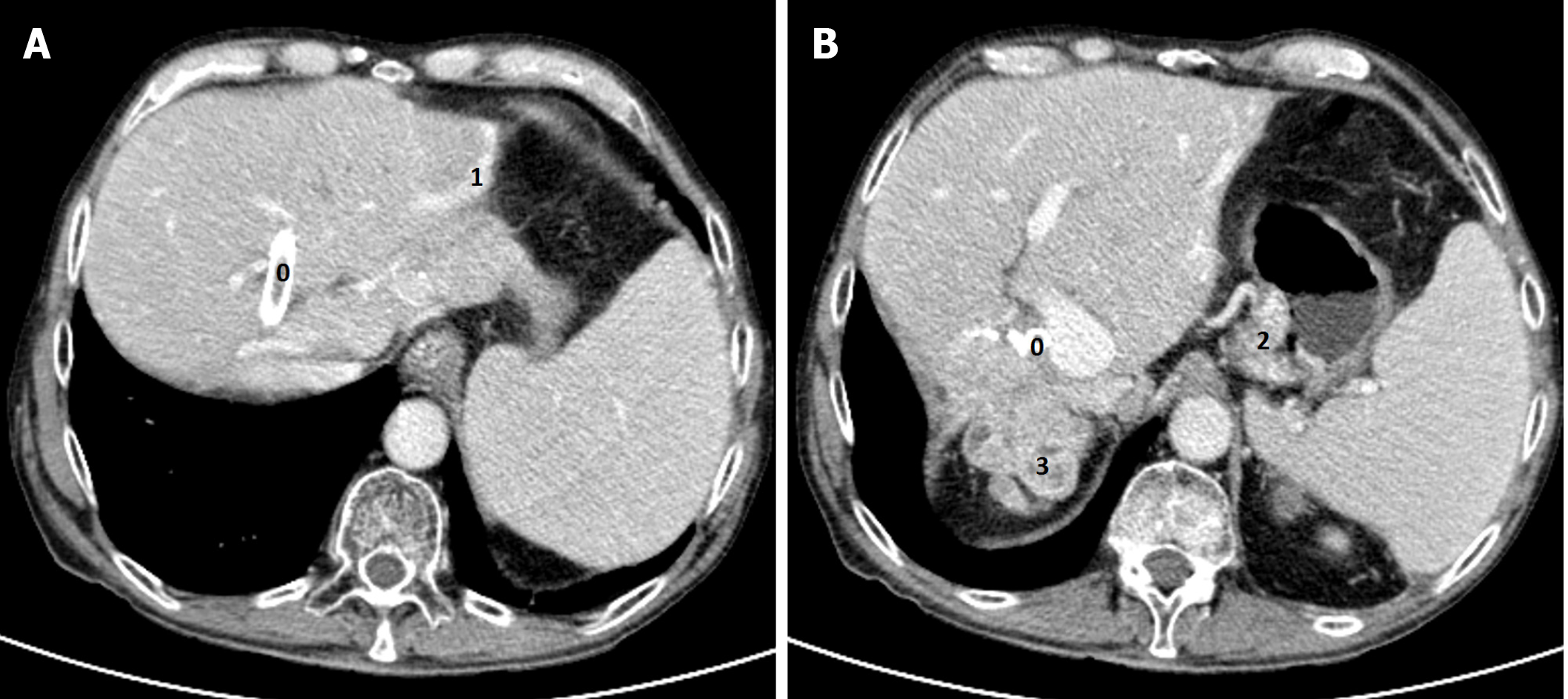
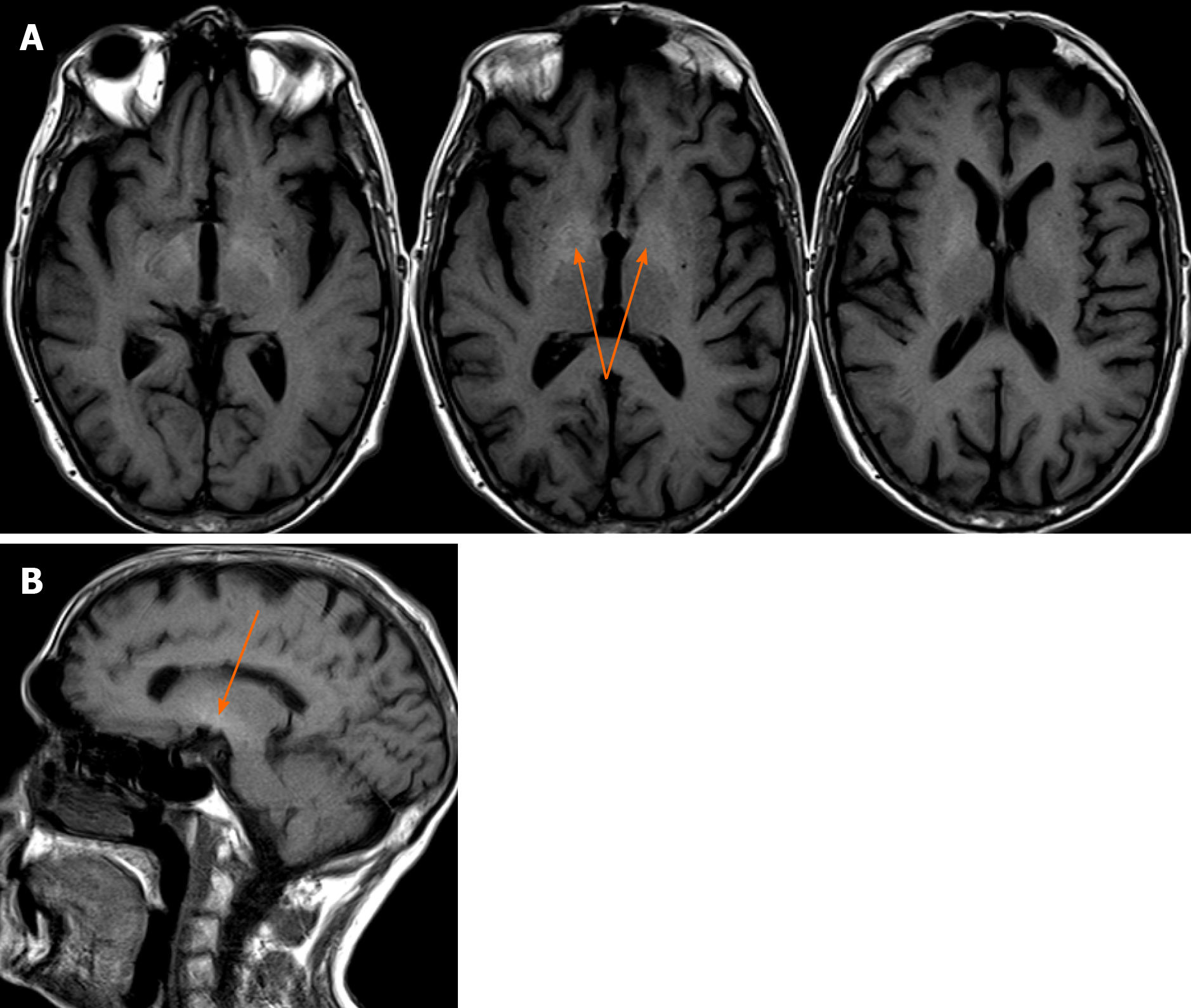
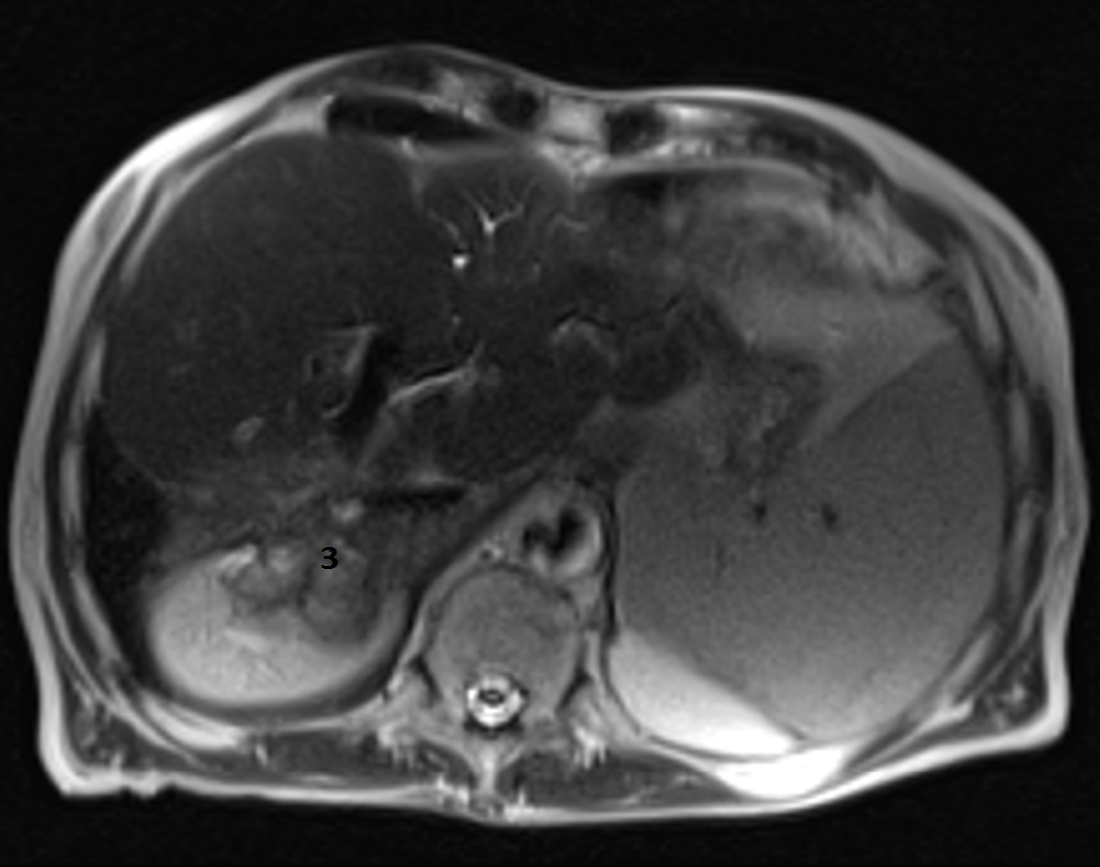
A head CT scan performed with intravenous contrast material revealed no evidence of intracranial hemorrhage, mass, or acute territorial infarct. However, an abdominal CT scan and brain MRI shed light on our case. The CT scan showed signs of portal hypertension, describing splenomegaly and splenic dilatation with collateral circulation, as well as an intrahepatic portosystemic shunt without biliary tract obstruction (Figure 2A and B). Surprisingly, brain MRI showed symmetric basal ganglia hyperintensity in a T1 alteration and asymmetric extension to cerebral peduncles, compatible with deposits of paramagnetic substances (Figure 3A and B) related to the intrahepatic shunt described in the previous CT scan. A video-electroencephalography was performed and showed neuronal dysfunction of metabolic-toxic origin. To complete our analysis, the patient underwent a gastroduodenoscopy, which showed grade 3 esophageal varices and the liver MRI revealed multiple irregular metastases (Figure 4).
The patient was diagnosed with hepatic encephalopathy and AHD secondary to advanced liver disease, most likely induced by a combination of previous hepatic resections, targeted therapies, and radionuclide treatment.
We started treatment with both oral and rectal laxatives, banding, and beta blockers at increasing dosage, improving the hepatic encephalopathy symptoms without developing side effects, thus restoring the patient to his basal state.
The patient was discharged after 2 wk of treatment with only a remaining rigidity of the superior left extremity, a sign of non-reversible neuronal damage due to AHD. We did not administer any further oncological treatment, and best supportive care was maintained. Levodopa treatment was stopped. The patient was alive 6 mo after discharge.
NENs are rare and heterogeneous tumors with the particularity of secreting hormones, adding a further layer of complexity to their clinical management. On the other hand, this also gave the treating physician the opportunity to target these tumor cells with multiple approaches. We must consider carefully the treatment modalities available, since our choices will impact our patients’ future. In this particular case, the patient developed an AHD after receiving multiple treatments for his metastatic midgut NET.
In particular, the patient underwent two hepatic surgeries and multiple hepatotoxic treatments, notably receiving four targeted therapies, three doses of PRRT, and lastly a somatostatin agonist. According to the NETTER-1 study, PRRT is superior to octreotide, but an important part of our treatment approach is to individualize the therapy according to the type of tumor, patient and treatments received previously. While PRRT treatment is a local radiotherapy that is highly selective for tumor tissue, it also affects the healthy surrounding hepatic parenchyma. Our patient had little healthy hepatic parenchyma left, and therefore had a greater susceptibility to local “ablative” therapies.
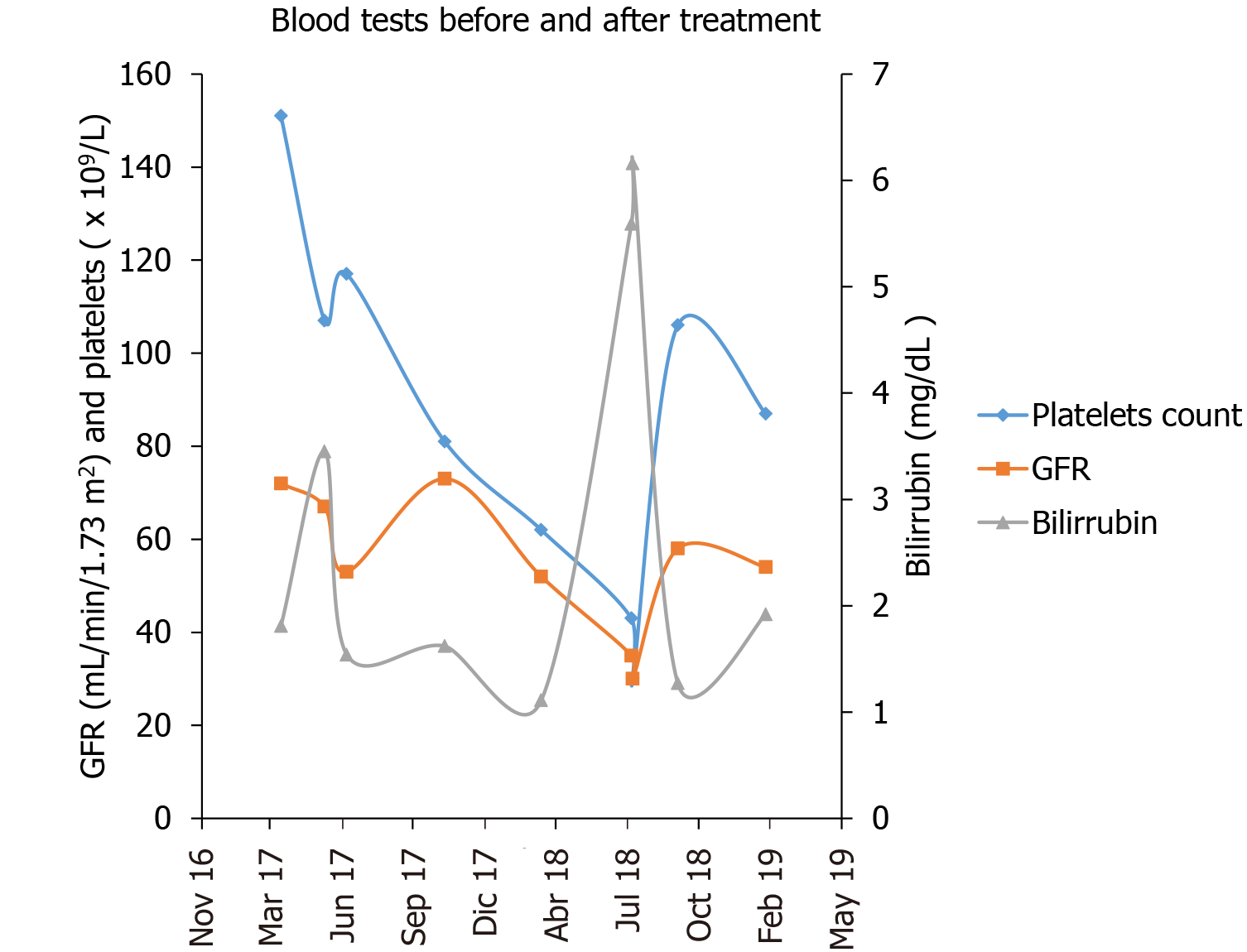
The patient experienced advanced liver disease after PRRT treatment, which likely acted as a trigger for AHD in a patient with unhealthy liver tissue, as we could identify in the CT scans and from consecutive liver laboratory tests before, during and after PRRT (Figure 5). The patient was not a candidate for closing the portosystemic shunt due to technical difficulties, and the patient’s severe portal hypertension (Child B cirrhosis) and a high bleeding risk, so his only options were preventive medical treatment. Thus, the AHD symptoms of our patient persisted, since the cause was not treated. On the other hand, the hepatic encephalopathy responded excellently, within days, to ammonia-lowering agents. The Parkinson’s disease was initially thought to be primary, but with the extensive paramagnetic deposits in the basal ganglia and the poor response to levodopa, it would be more reasonable to consider it a Parkinsonism secondary to the portosystemic shunts and hepatic cirrhosis. As stated previously, the presence of manganese in the basal ganglia of cirrhotic patients is diagnostic of AHD and can result in irreversible neuronal damage.
Herein, we report the first case described in the literature of AHD in a metastatic NET. Patients with NETs are typically long-time survivors, and we currently have multiple treatment modalities to choose from. Selection should be based on maximizing survival and reducing both the potential immediate and long-term side effects. The negative outcomes relating to hepatic injury in long-term NET survivors resemble those of patients with advanced liver disease. As such, regular monitoring and surveillance for potential complications in long-term cancer survivors should be recommended to rule out negative outcomes that may appear following treatment.
This work would not have been possible without the support of all personnel at Vall d’Hebron University Hospital who help take care of patients and excel at their everyday job. We are especially indebted to Dr. Capdevila and Dra. Riveiro, who both provided us with guidance and expertise. We would like to thank Carla Ainscow and Dr. Adam Lassiter Carrera for English revisions of the final manuscript. We would also like to thank the rest of the team who helped make this article possible. We would like to thank our family and friends for their continuous support. We must not conclude this section without thanking all of the patients and families who come to VHUH and make our everyday work gratifying, and on top of that, contribute to science by participating in research projects altruistically.
Manuscript source: Unsolicited manuscript
Specialty type: Oncology
Country/Territory of origin: Spain
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Jamali R, Kang KJ S-Editor: Zhang L L-Editor: Filipodia P-Editor: Wang LL
| 1. | Yao JC, Hassan M, Phan A, Dagohoy C, Leary C, Mares JE, Abdalla EK, Fleming JB, Vauthey JN, Rashid A, Evans DB. One hundred years after "carcinoid": epidemiology of and prognostic factors for neuroendocrine tumors in 35,825 cases in the United States. J Clin Oncol. 2008;26:3063-3072. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3022] [Cited by in RCA: 3215] [Article Influence: 189.1] [Reference Citation Analysis (0)] |
| 2. | Dasari A, Shen C, Halperin D, Zhao B, Zhou S, Xu Y, Shih T, Yao JC. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients With Neuroendocrine Tumors in the United States. JAMA Oncol. 2017;3:1335-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1510] [Cited by in RCA: 2399] [Article Influence: 299.9] [Reference Citation Analysis (4)] |
| 3. | Hallet J, Law CH, Cukier M, Saskin R, Liu N, Singh S. Exploring the rising incidence of neuroendocrine tumors: a population-based analysis of epidemiology, metastatic presentation, and outcomes. Cancer. 2015;121:589-597. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 446] [Cited by in RCA: 601] [Article Influence: 54.6] [Reference Citation Analysis (1)] |
| 4. | Byrne RM, Pommier RF. Small Bowel and Colorectal Carcinoids. Clin Colon Rectal Surg. 2018;31:301-308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 12] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 5. | Díez M, Teulé A, Salazar R. Gastroenteropancreatic neuroendocrine tumors: diagnosis and treatment. Ann Gastroenterol. 2013;26:29-36. [PubMed] |
| 6. | Oronsky B, Ma PC, Morgensztern D, Carter CA. Nothing But NET: A Review of Neuroendocrine Tumors and Carcinomas. Neoplasia. 2017;19:991-1002. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 405] [Cited by in RCA: 466] [Article Influence: 58.3] [Reference Citation Analysis (0)] |
| 7. | Rindi G, Wiedenmann B. Neuroendocrine neoplasms of the gut and pancreas: new insights. Nat Rev Endocrinol. 2011;8:54-64. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 133] [Cited by in RCA: 129] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 8. | Lloyd RV, Osamura RY, Klöppel G and Rosai J. WHO classification of tumours of endocrine organs. Vol. 10, 4th edition. IARC Publications, Lyon, France, 2017. |
| 9. | Inzani F, Petrone G, Rindi G. The New World Health Organization Classification for Pancreatic Neuroendocrine Neoplasia. Endocrinol Metab Clin North Am. 2018;47:463-470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 120] [Cited by in RCA: 147] [Article Influence: 21.0] [Reference Citation Analysis (0)] |
| 10. | Milione M, Maisonneuve P, Pellegrinelli A, Grillo F, Albarello L, Spaggiari P, Vanoli A, Tagliabue G, Pisa E, Messerini L, Centonze G, Inzani F, Scarpa A, Papotti M, Volante M, Sessa F, Fazio N, Pruneri G, Rindi G, Solcia E, La Rosa S, Capella C. Ki67 proliferative index of the neuroendocrine component drives MANEC prognosis. Endocr Relat Cancer. 2018;25:583-593. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 72] [Article Influence: 10.3] [Reference Citation Analysis (0)] |
| 11. | Pavel M, Baudin E, Couvelard A, Krenning E, Öberg K, Steinmüller T, Anlauf M, Wiedenmann B, Salazar R; Barcelona Consensus Conference participants. ENETS Consensus Guidelines for the management of patients with liver and other distant metastases from neuroendocrine neoplasms of foregut, midgut, hindgut, and unknown primary. Neuroendocrinology. 2012;95:157-176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 608] [Cited by in RCA: 589] [Article Influence: 45.3] [Reference Citation Analysis (0)] |
| 12. | Rinke A, Müller HH, Schade-Brittinger C, Klose KJ, Barth P, Wied M, Mayer C, Aminossadati B, Pape UF, Bläker M, Harder J, Arnold C, Gress T, Arnold R; PROMID Study Group. Placebo-controlled, double-blind, prospective, randomized study on the effect of octreotide LAR in the control of tumor growth in patients with metastatic neuroendocrine midgut tumors: a report from the PROMID Study Group. J Clin Oncol. 2009;27:4656-4663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1609] [Cited by in RCA: 1720] [Article Influence: 107.5] [Reference Citation Analysis (0)] |
| 13. | Caplin ME, Pavel M, Ćwikła JB, Phan AT, Raderer M, Sedláčková E, Cadiot G, Wolin EM, Capdevila J, Wall L, Rindi G, Langley A, Martinez S, Blumberg J, Ruszniewski P; CLARINET Investigators. Lanreotide in metastatic enteropancreatic neuroendocrine tumors. N Engl J Med. 2014;371:224-233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1142] [Cited by in RCA: 1265] [Article Influence: 115.0] [Reference Citation Analysis (0)] |
| 14. | Strosberg J, El-Haddad G, Wolin E, Hendifar A, Yao J, Chasen B, Mittra E, Kunz PL, Kulke MH, Jacene H, Bushnell D, O'Dorisio TM, Baum RP, Kulkarni HR, Caplin M, Lebtahi R, Hobday T, Delpassand E, Van Cutsem E, Benson A, Srirajaskanthan R, Pavel M, Mora J, Berlin J, Grande E, Reed N, Seregni E, Öberg K, Lopera Sierra M, Santoro P, Thevenet T, Erion JL, Ruszniewski P, Kwekkeboom D, Krenning E; NETTER-1 Trial Investigators. Phase 3 Trial of 177Lu-Dotatate for Midgut Neuroendocrine Tumors. N Engl J Med. 2017;376:125-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1702] [Cited by in RCA: 2171] [Article Influence: 271.4] [Reference Citation Analysis (0)] |
| 15. | Kaderli RM, Spanjol M, Kollár A, Bütikofer L, Gloy V, Dumont RA, Seiler CA, Christ ER, Radojewski P, Briel M, Walter MA. Therapeutic Options for Neuroendocrine Tumors: A Systematic Review and Network Meta-analysis. JAMA Oncol. 2019;5:480-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 60] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 16. | Weissenborn K. Hepatic Encephalopathy: Definition, Clinical Grading and Diagnostic Principles. Drugs. 2019;79:5-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 127] [Cited by in RCA: 166] [Article Influence: 27.7] [Reference Citation Analysis (0)] |
| 17. | Tryc AB, Goldbecker A, Berding G, Rümke S, Afshar K, Shahrezaei GH, Pflugrad H, Barg-Hock H, Strassburg CP, Hecker H, Weissenborn K. Cirrhosis-related Parkinsonism: prevalence, mechanisms and response to treatments. J Hepatol. 2013;58:698-705. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 50] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 18. | Pinarbasi B, Kaymakoglu S, Matur Z, Akyuz F, Demir K, Besisik F, Ozdil S, Boztas G, Cakaloglu Y, Mungan Z, Okten A. Are acquired hepatocerebral degeneration and hepatic myelopathy reversible? J Clin Gastroenterol. 2009;43:176-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 36] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 19. | Fernández-Rodriguez R, Contreras A, De Villoria JG, Grandas F. Acquired hepatocerebral degeneration: clinical characteristics and MRI findings. Eur J Neurol. 2010;17:1463-1470. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 58] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 20. | Rose C, Butterworth RF, Zayed J, Normandin L, Todd K, Michalak A, Spahr L, Huet PM, Pomier-Layrargues G. Manganese deposition in basal ganglia structures results from both portal-systemic shunting and liver dysfunction. Gastroenterology. 1999;117:640-644. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 210] [Cited by in RCA: 190] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 21. | Maffeo E, Montuschi A, Stura G, Giordana MT. Chronic acquired hepatocerebral degeneration, pallidal T1 MRI hyperintensity and manganese in a series of cirrhotic patients. Neurol Sci. 2014;35:523-530. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 34] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 22. | Butterworth RF. Parkinsonism in cirrhosis: pathogenesis and current therapeutic options. Metab Brain Dis. 2013;28:261-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 58] [Cited by in RCA: 57] [Article Influence: 4.8] [Reference Citation Analysis (0)] |