Published online Dec 27, 2021. doi: 10.4254/wjh.v13.i12.1936
Peer-review started: February 25, 2021
First decision: May 13, 2021
Revised: May 26, 2021
Accepted: November 28, 2021
Article in press: November 28, 2021
Published online: December 27, 2021
Processing time: 304 Days and 6.4 Hours
The liver is commonly affected by metastatic disease. Therefore, it is essential to detect and characterize liver metastases, assuming that patient management and prognosis rely on it. The imaging techniques that allow non-invasive assessment of liver metastases include ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), positron emission tomography (PET)/CT, and PET/MRI. In this paper, we review the imaging findings of liver metastases, focusing on each imaging modality’s advantages and potential limitations. We also assess the importance of different imaging modalities for the management, follow-up, and therapy response of liver metastases. To date, both CT and MRI are the most appropriate imaging methods for initial lesion detection, follow-up, and assessment of treatment response. Multiparametric MRI is frequently used as a problem-solving technique for liver lesions and has evolved substantially over the past decade, including hardware and software developments and specific intravenous contrast agents. Several studies have shown that MRI performs better in small-sized metastases and moderate to severe liver steatosis cases. Although state-of-the-art MRI shows a greater sensitivity for detecting and characterizing liver metastases, CT remains the chosen method. We also present the controversial subject of the "economic implication" to use CT over MRI.
Core Tip: Several imaging methods are clinically available to evaluate and characterize liver metastases. Both computed tomography and magnetic resonance imaging (MRI) are currently the techniques that show the highest diagnostic performance and are also the most suitable for assessing therapy response and follow-up. Several studies have shown that MRI has a higher sensitivity for detecting and characterizing liver metastases; therefore, it may be the ideal imaging method for treatment planning before and after neoadjuvant chemotherapy. The traditional paradigm for ordering imaging studies emphasizes diagnostic accuracy, which is why we believe that MRI should be favored when available, the first-line imaging for detecting liver metastases, and pre- and post-treatment follow-up.
- Citation: Freitas PS, Janicas C, Veiga J, Matos AP, Herédia V, Ramalho M. Imaging evaluation of the liver in oncology patients: A comparison of techniques. World J Hepatol 2021; 13(12): 1936-1955
- URL: https://www.wjgnet.com/1948-5182/full/v13/i12/1936.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i12.1936
The liver is one of the most common organs involved with metastatic disease. Secondary lesions are about 18-40 × more common than primary liver tumors[1,2]. Liver metastases are most often secondary to colorectal carcinoma (CRC) (40%), stomach (20%), pancreas (20%), lung (10%), and breast cancer (10%)[3]. Other less frequent primary tumors include neuroendocrine tumors (NETs), gastrointestinal stromal tumors (GISTs), and renal cell carcinomas[3].
The spectrum of presentation is broad. Liver metastases frequently present as multifocal and separate lesions; however, they can also be solitary or less frequently manifest as confluent masses[4]. The solitary mass form of presentation is most often associated with colon cancer. Meanwhile, breast cancer metastases may infrequently diffusely involve the liver in a pseudocirrhosis pattern (mimicking cirrhosis), particularly following chemotherapy[3].
Solid liver metastases are typically supplied by arterial blood flow; hence they can be classified as hypovascular or hypervascular[1]. The main group of hypovascular metastases includes CRC, gastric, breast, and lung cancer[5]. On the other hand, hypervascular liver metastases are more commonly seen in renal cell carcinoma (especially clear-cell type), NETs, melanoma, thyroid carcinoma, and GISTs. Breast cancer liver metastases may appear hypovascular and hypervascular. Additionally, liver metastases may be cystic, arising from cystic primaries, such as ovarian carcinoma or mucinous cystadenocarcinoma of the GI and pancreas. These may also arise from GIST, leiomyosarcoma, malignant melanoma, carcinoid, and pheochromocytoma[1]. Calcification may be present in mucinous adenocarcinomas from the gastrointestinal tract or the ovary and in breast, lung, renal, and medullary thyroid carcinoma[6,7].
In the current perspective of oncologic liver surgery or local ablation, imaging shows a vital role in the detection, characterization, and determination of metastases' exact location, on a per-patient and per-lesion basis, even in patients with stage IV disease. Surgery and a variety of interventional radiologic techniques are also performed in selected patients with oligometastatic disease.
Stage IV CRC is defined as distant metastasis that either is confined to one organ or site (stage IVa) or affects more than one organ or site or the peritoneum (stage IVb). The past decade has seen a paradigm shift in stage IV or metastatic CRC (mCRC) management, leading to a significant increase in overall survival for these patients, from less than 6 mo to nearly 2 years[6]. Much of this success is credited to the increased utilization of hepatectomy in patients with oligometastatic liver disease, the development of newer chemotherapy regimens, and the identification of new molecular targets and their inhibitors. Imaging plays an essential role in the workup of patients with mCRC by helping enumerate the number and sites of metastases, determine resectability, assess response to systemic and liver-directed therapies, and detect drug toxicities and disease recurrences.
This paper aims to briefly review each imaging technique and subsequently evaluate them in assessing liver metastases, including detection, characterization, diagnosis, and treatment response evaluation.
Ultrasonography (US) is a safe, accessible, and inexpensive technique. Nevertheless, it has considerable limitations, including dependency on operator expertise, patient’s body habitus, cooperation, and bowel gas interposition[8]. The lower performance of this technique is also explained by limited spatial resolution, and for this reason, small (< 3-5 mm), isoechoic, and deep-seated metastases can be missed[1,8]. The conventional US's general sensitivity for detecting liver metastases is approximately 69% (sensitivity of 50%-76% in series with a true gold standard – intraoperative US or resection)[1,9]. This sensitivity is probably lower in patients with subdiaphragmatic lesions, chronic hepatic disease, and severe hepatic steatosis, which may be induced by chemotherapy. Moreover, the ambiguity in segmental localization leads to a lack of reproducibility compared to computed tomography (CT) and magnetic resonance imaging (MRI).
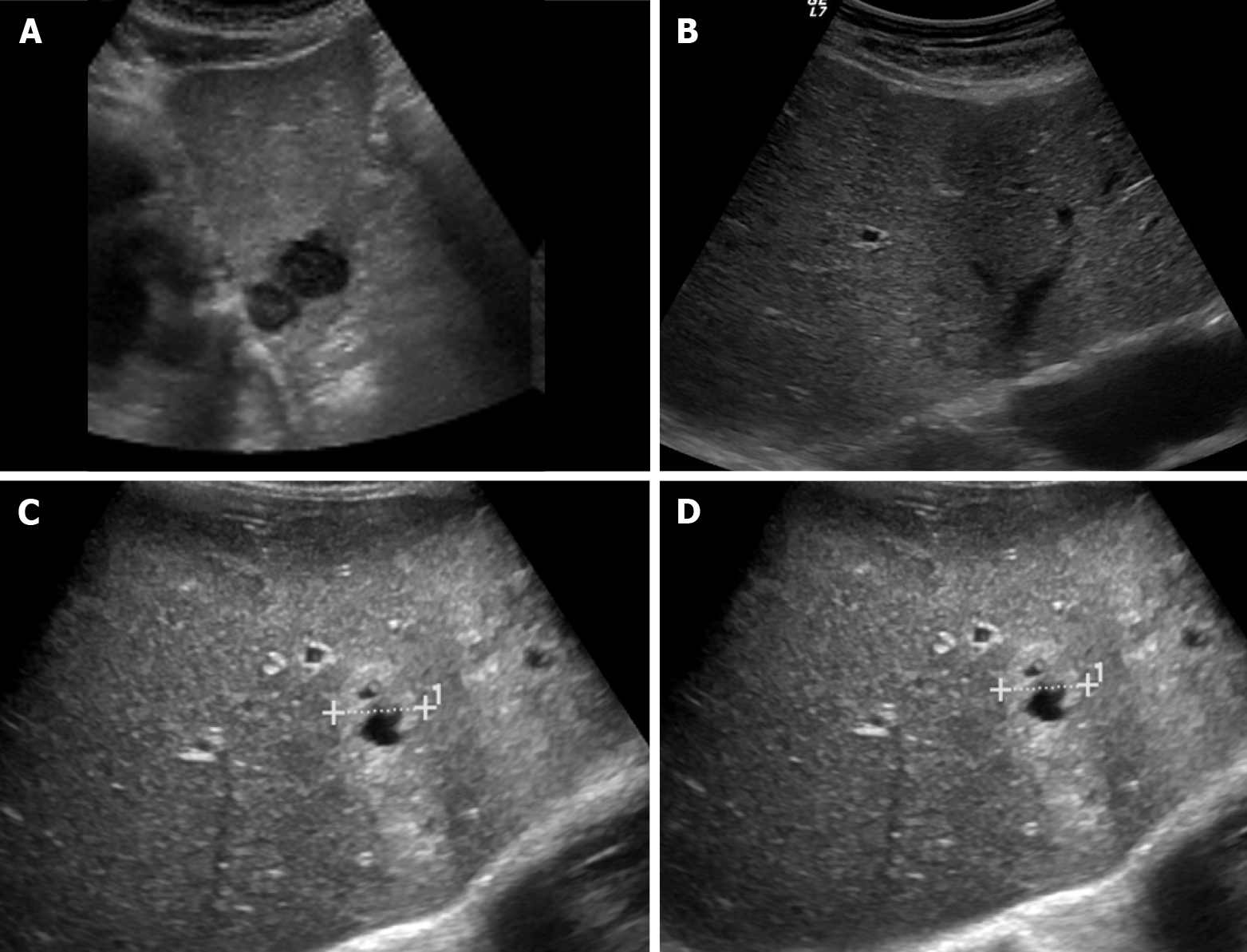
The appearance of metastases on ultrasound is diverse, but most appear rounded with sharp or smooth margins. They show variable echogenicity (hypo-, iso, or hyperechoic relative to the surrounding parenchyma), with the hypoechoic pattern being the most common (65%)[7]. Sometimes a hypoechoic halo is noted (40%), especially if the lesion is iso- or hyperechoic (Figure 1)[7]. Hepatic metastases of CRC are typically well-defined, solid, hypoechoic lesions and hypovascular on Doppler ultrasound, and occasionally present a peripheral halo ("target" or "bulls-eye" appearance)[8,9]. This broad spectrum of appearance makes the distinction between benign and malignant lesions difficult, reducing its specificity[8].
Contrast-enhanced ultrasound (CEUS) has improved the sensitivity for the detection of liver metastases. A study by Kong et al[10], including 240 patients with liver metastases, showed that diffuse homogeneous hyperenhancement followed by rapid washout was the most common pattern on CEUS (55.4% and 96.2%, respectively).
Regarding CEUS, reports differ, mainly because they depend more on operator expertise and other technical factors. Bernatik et al[11] found that CEUS detected 97% of the lesions diagnosed by CT[8,11]. Piscaglia et al[12] examined 109 patients with colorectal and gastric cancer. They showed that CEUS improves sensitivity in the detection of liver metastases to 95.4% when compared to conventional US (76.9%) and CT (90.8%)[12]. Cantisani et al[8,13] showed that CEUS improved US sensitivity from 67.4%-71.6% to 93.4%-95.8%. On the other hand, Vialle et al[14] reported that the CEUS sensitivity was inferior to CT in detecting hepatic metastases from colorectal cancer (CEUS 64.5% vs CT 80.4%). Moreover, since metastatic liver disease frequently shows multiple lesions, the per-lesion evaluation would need multiple doses of ultrasound contrast agent[7].
The accuracy for the detection of hepatic lesions may differ with the US mode. Two-dimensional (2D) CEUS shows limitations in evaluating liver metastases since it is more prone to sampling errors, such as imaging caption of a single section and plane-to-plane perfusion variation. On the other hand, three-dimensional (3D) CEUS imaging techniques can image the tumor as a whole, provide spatial information, and allow volumetric images. El Kaffas et al[15] showed that 3D dynamic CEUS is superior to 2D dynamic CEUS imaging by reducing the sampling errors from heterogeneous tumor perfusion. Other studies have shown no significant differences between the two modes concerning sensitivity[16]. Nevertheless, the perception of the feeding arteries is improved with the 3D CEUS, which might be helpful for the treatment of hypervascular liver metastases[16].
Cross-section imaging techniques, including CT, and positron emission tomography (PET)/CT, have advanced considerably, leading to early and accurate liver metastasis detection[17]. Multidetector CT is a reliable technique for detecting liver metastases and preoperative staging, allowing volumetric acquisition with high-quality multiplanar reformatted images, liver volume calculation, and 3D reconstructions preoperative tumor resection planning[3]. CT is fast and accessible, allows high-quality liver imaging and entire abdomen and chest coverage, and depicts extrahepatic disease[18]. CT shows a specificity of 77.3% and sensitivity up to 73.5% for the detection of liver metastases[19].
Liver metastases usually appear as hypo or iso-dense nodules on unenhanced CT. These nodules tend to be well-defined, but they can also be irregular, depending on size[6]. Necrosis and cystic transformation may be present, appearing as a central area of low attenuation. Besides, at times liver metastases may also show high attenuation due to hemorrhagic content[3].
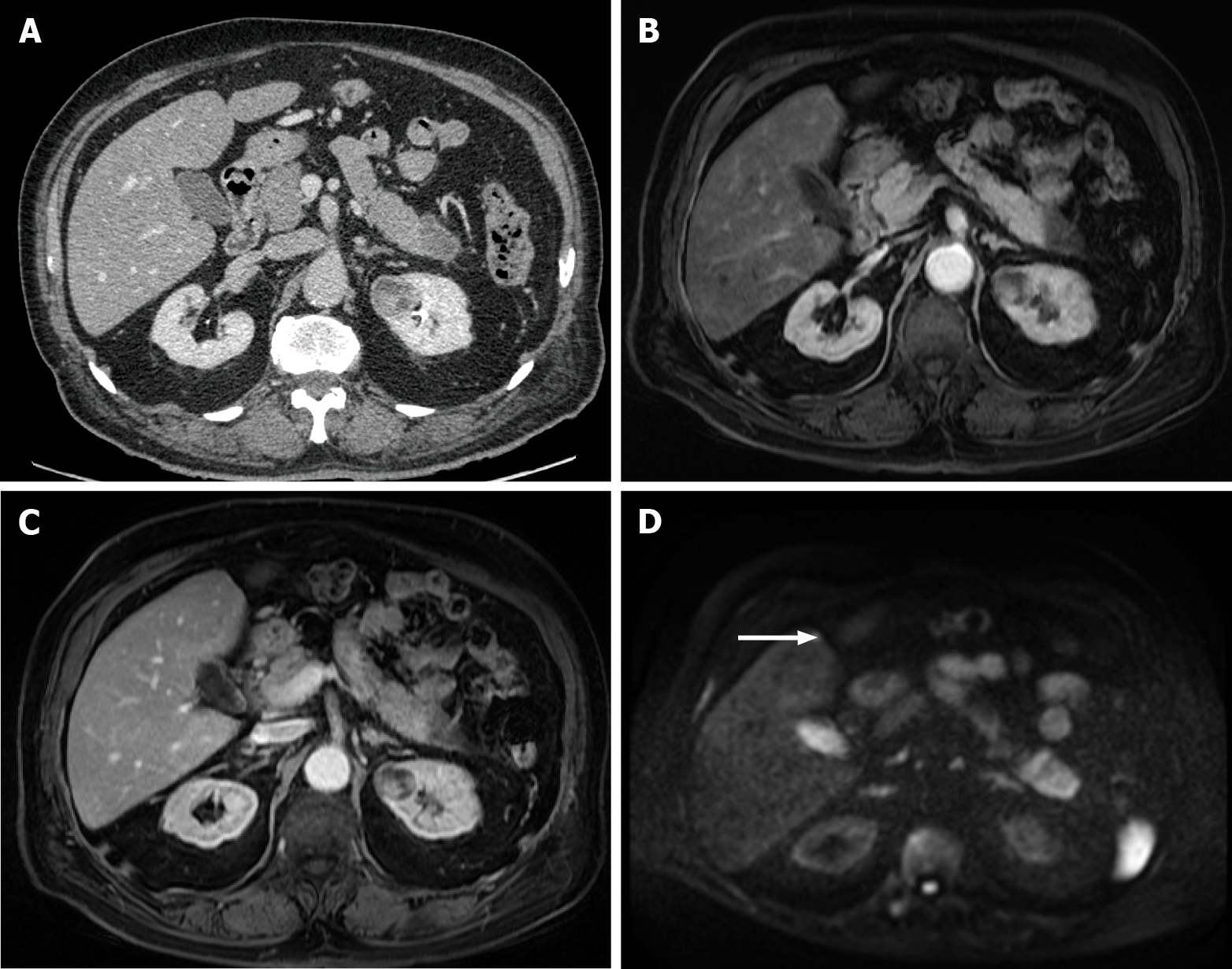
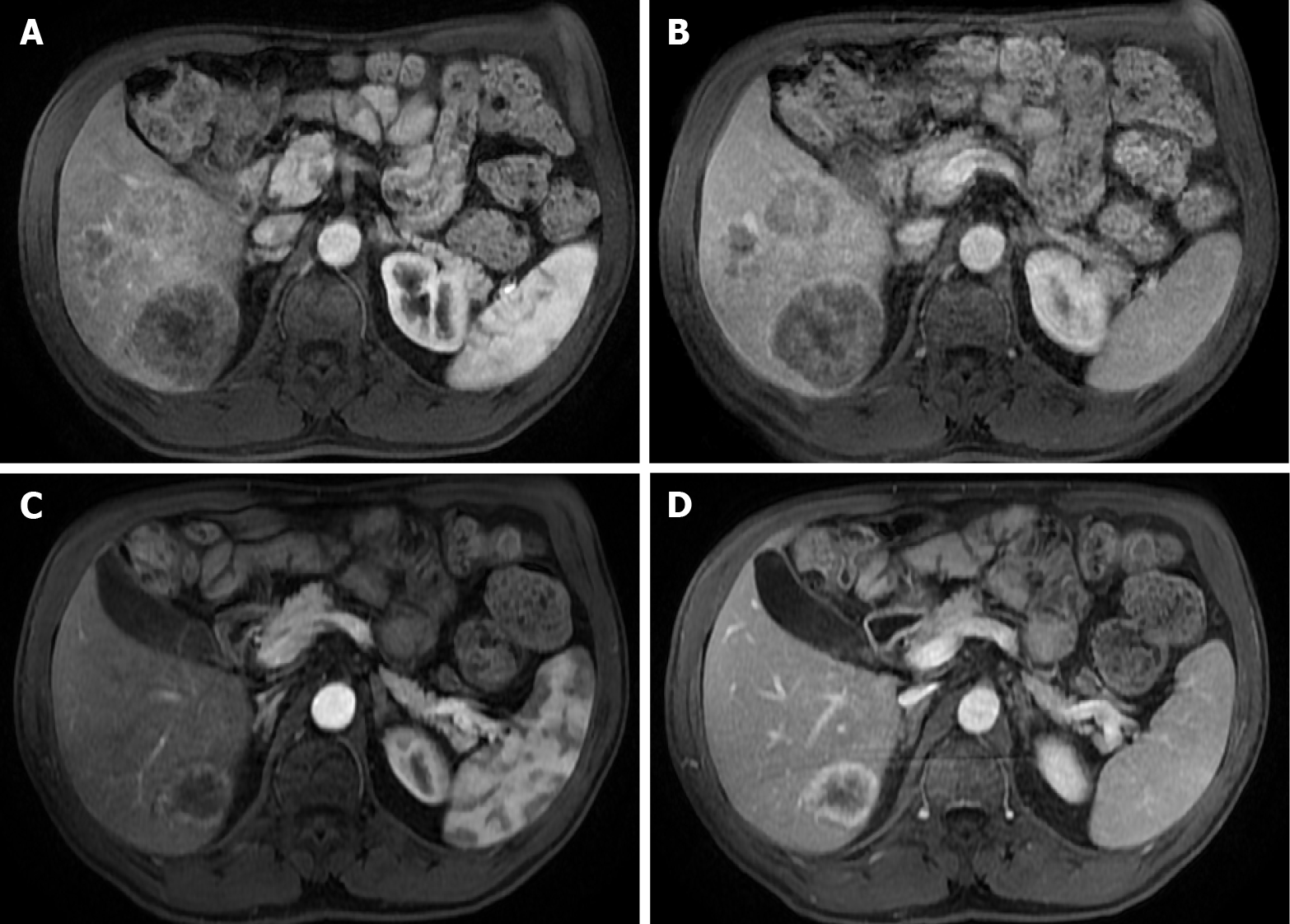
Dynamic imaging is crucial, and its concept, perception, and evaluation are similar between CT and MRI (Figure 2). Most liver metastases are hypovascular and are best detected during the portal venous phase (PVP), which begins approximately 60-80 s after the initial injection. In this phase, the liver parenchyma enhances through the dominant blood supply by the portal vein. Hypovascular metastases appear as hypodense/hypoattenuating lesions compared to the background liver parenchyma (Figure 3)[1]. They usually show a peripheral rim enhancement in the late arterial phase (LAP), which fades centrally in the venous phase (“target appearance”)[5,6]. On the other hand, hypervascular metastases enhance earlier in the LAP, which is demonstrated by contrast in the portal vein and absence in the hepatic veins. These lesions may fade and become isodense with the remaining liver parenchyma or show variable degrees of washout in the PVP and delayed acquisitions[5,6,20].
The PVP is considered the most critical phase, with a sensitivity of 91.5% for detecting hypovascular metastases[21]. However, the optimal number and choice of acquisition phases are still under debate, given the potential risks of higher radiation doses[1]. Honda et al[22] showed that adding a LAP improved liver metastases’ detectability, particularly in lesions smaller than 10 mm. However, other studies, such as that from Ferlay et al[23], concluded that for evaluating CRC liver metastases, the addition of the LAP and delayed phases did not improve the performance compared to the PVP alone.
For hypervascular metastases, non-contrast-enhanced CT (NE-CT) only adds a small incremental value to contrast-enhanced CT (CE-CT) for their detection and characterization based on existing evidence. It seems that it is not worth adding further radiation exposure and the increased number of images for interpretation associated with NE-CT acquisition[24]. Still, NE-CT may be helpful as calcifications are present in up to 11% of liver metastases at initial presentation[25,26].
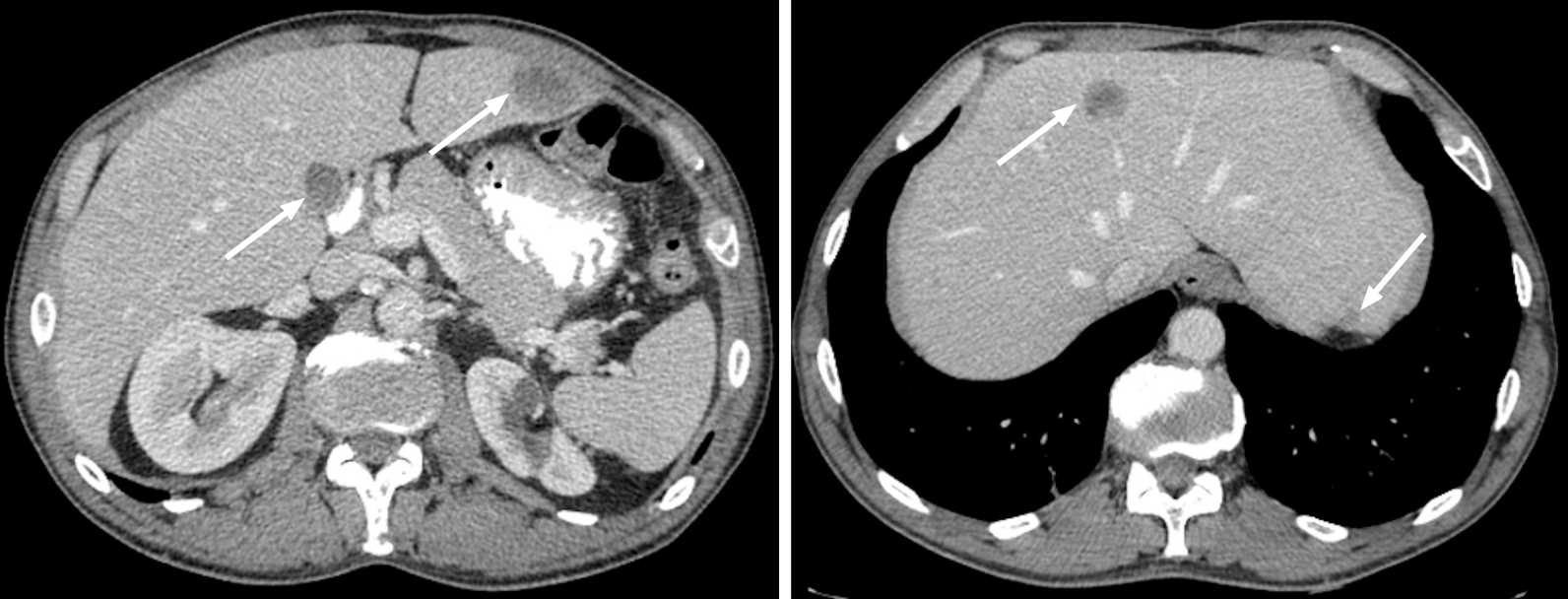
CT is the workhorse for abdominal imaging staging; however, liver metastases may be missed. The detection rate of lesions by CT declines as its diameter decreases, with a detection rate estimated at 72% for lesions measuring 10-20 mm and 16% for lesions smaller than 10 mm[19]. Benoist et al[27] showed that the rate of missed liver lesions after chemotherapy could be as high as 83%.
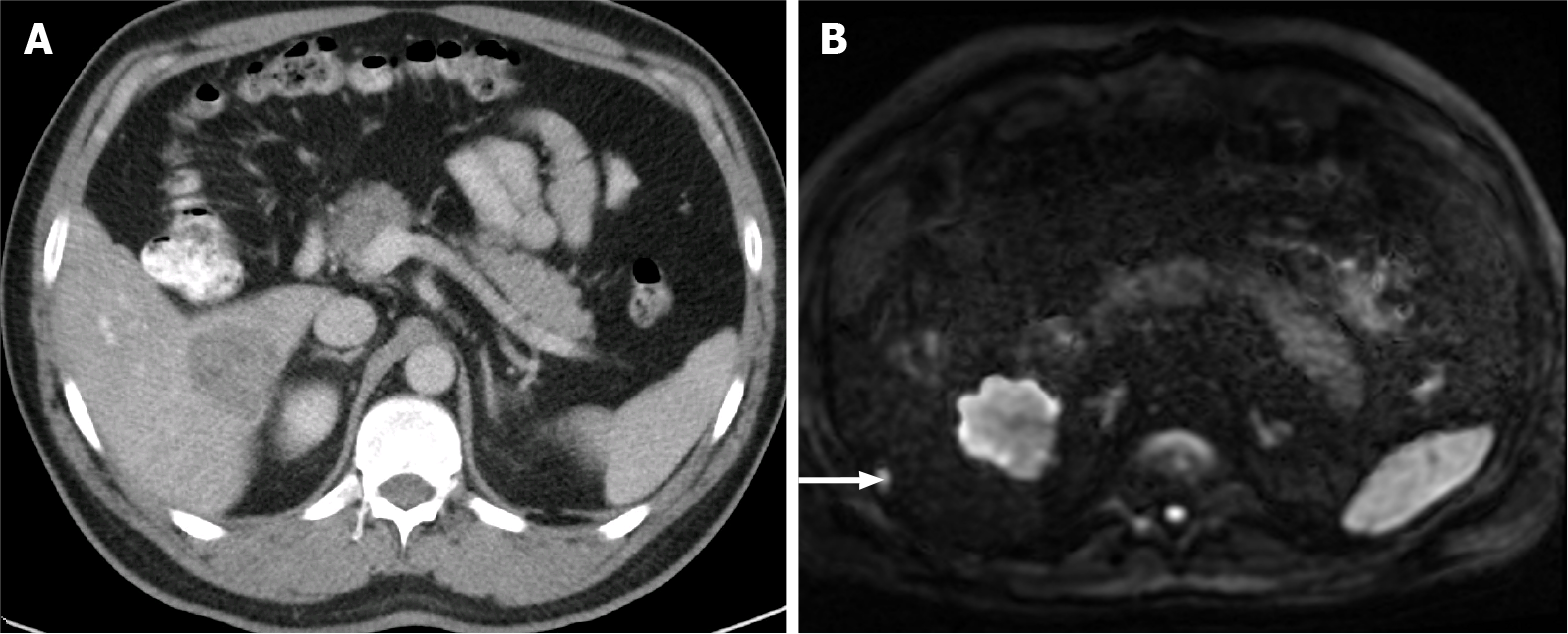
A recent study demonstrated that some liver metastases without sufficient contrast enhancement were more likely to be overlooked, as were subcapsular lesions, in case of liver steatosis or in cases of examination indication other than assessing malignant tumors[17].
It has been shown that imaging during the exact correct vascular phase of contrast and an adequate iodine concentration (300-400 mg/mL) is essential for improving the detectability of hypoattenuating metastases[28]. However, it is known that higher contrast concentration may harm renal impaired patients and may also lead to contrast-induced nephropathy. As most patients will frequently need repeated examinations and extended follow-up periods, radiation exposure should also be kept in consideration, representing one of the most critical limitations of CT.
Dual-energy CT (DE-CT) scanners are getting progressively more available. It involves the acquisition of two or more CT measurements with distinct energy spectra. Using the differential attenuation of tissues and materials at different X-ray energies, DE-CT allows the distinction of tissues and materials beyond what is possible with conventional CT[29].
A study comparing DE-CT-driven low-keV virtual monoenergetic imaging to standard linearly blended images concluded that low-keV images improved quantitative size measurements and diagnostic accuracy of CRC liver metastases[30]. Also, this new technique improves the CT accuracy in differentiating liver abscesses from liver metastases in the context of hypovascular metastases, a common clinical dilemma. This technique may increase hypervascular and hypovascular liver lesions' conspicuity, improving CT performance in detecting metastases, especially in cases of concomitant liver steatosis[31].
Multiparametric MRI is frequently used as a problem-solving technique in the evaluation of liver lesions. MRI has evolved substantially over the past decade, including hardware and software developments and specific intravenous contrast agents[3]. Technological improvements also potentially allow better quality imaging in non-cooperative patients, one of the main challenges in MRI. Therefore, when reviewing this imaging technique's performance, one should be aware of these recent advances in the field of MRI, preferring the recent literature.
MRI allows anatomic and morphologic evaluation, as well as functional imaging. The diagnostic sensitivity in detecting hepatic metastases is approximately 87% and has increased with the introduction of diffusion-weighted imaging (WI) in routine protocols and the development of hepatocyte-specific contrast agents, reaching a sensitivity of 95%[21,26]. This technique significantly improves the diagnostic efficacy and accuracy in the approach of liver metastases. Several studies reported the superiority in detecting liver lesions compared to CT, especially if they are small[32,33].
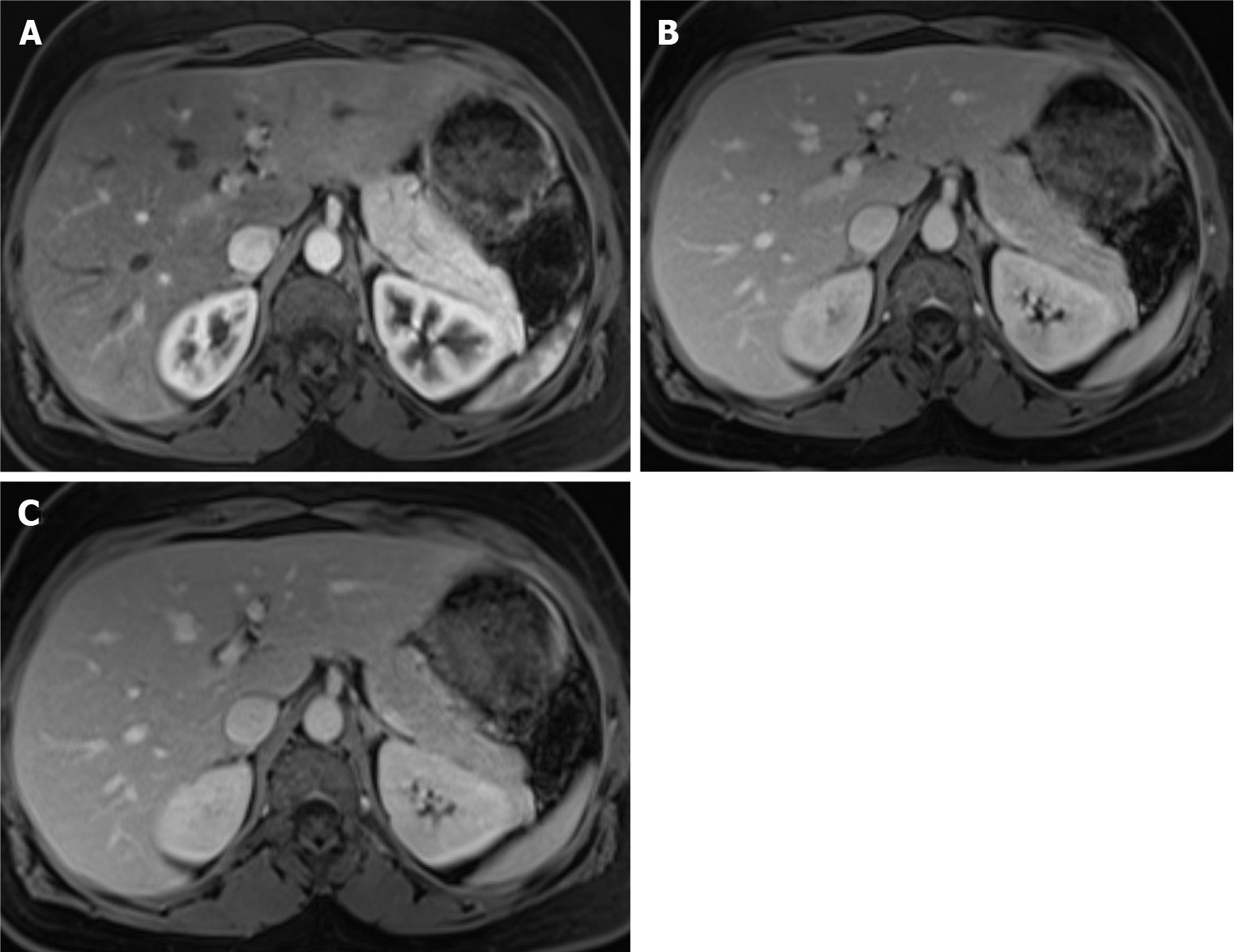
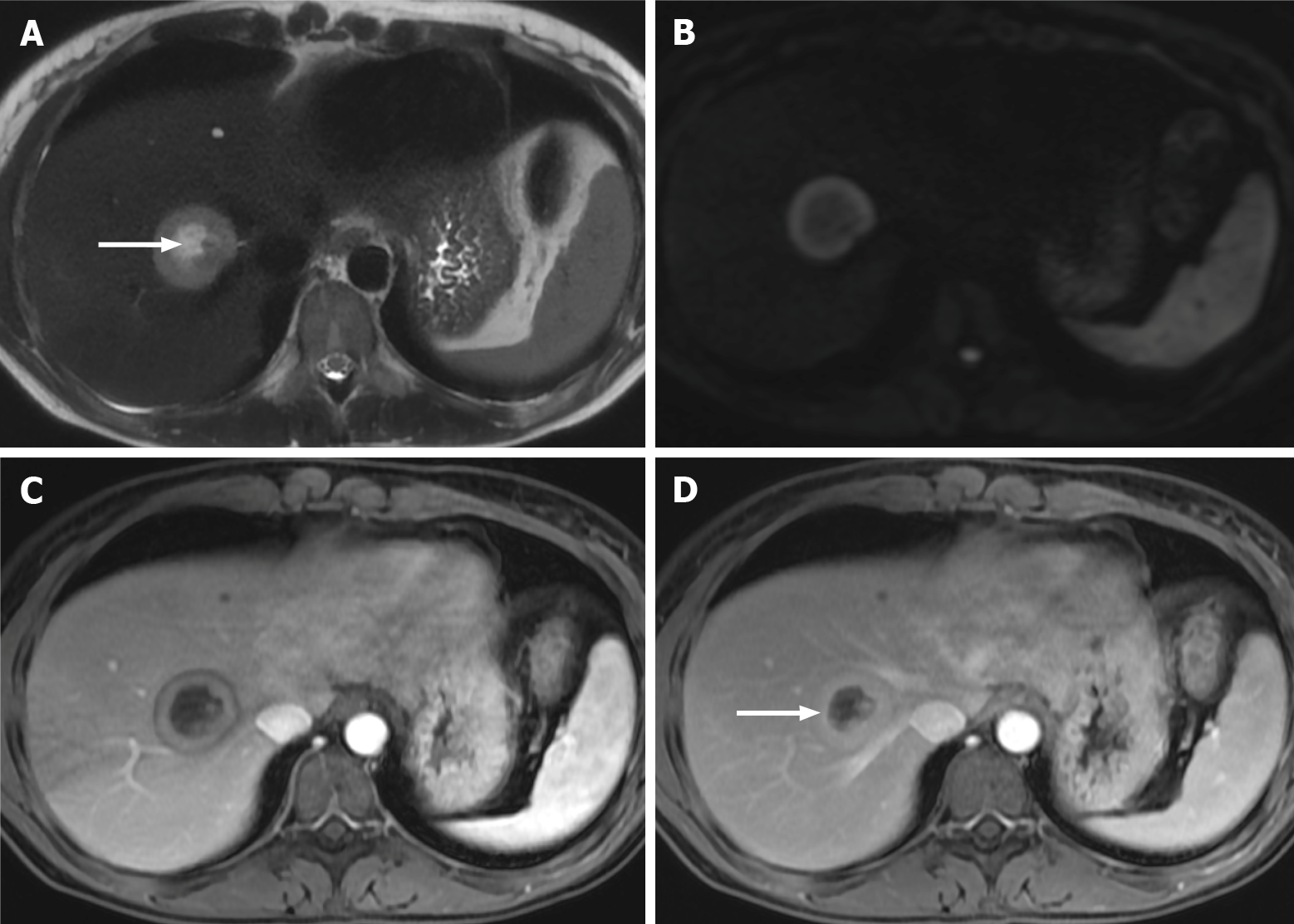
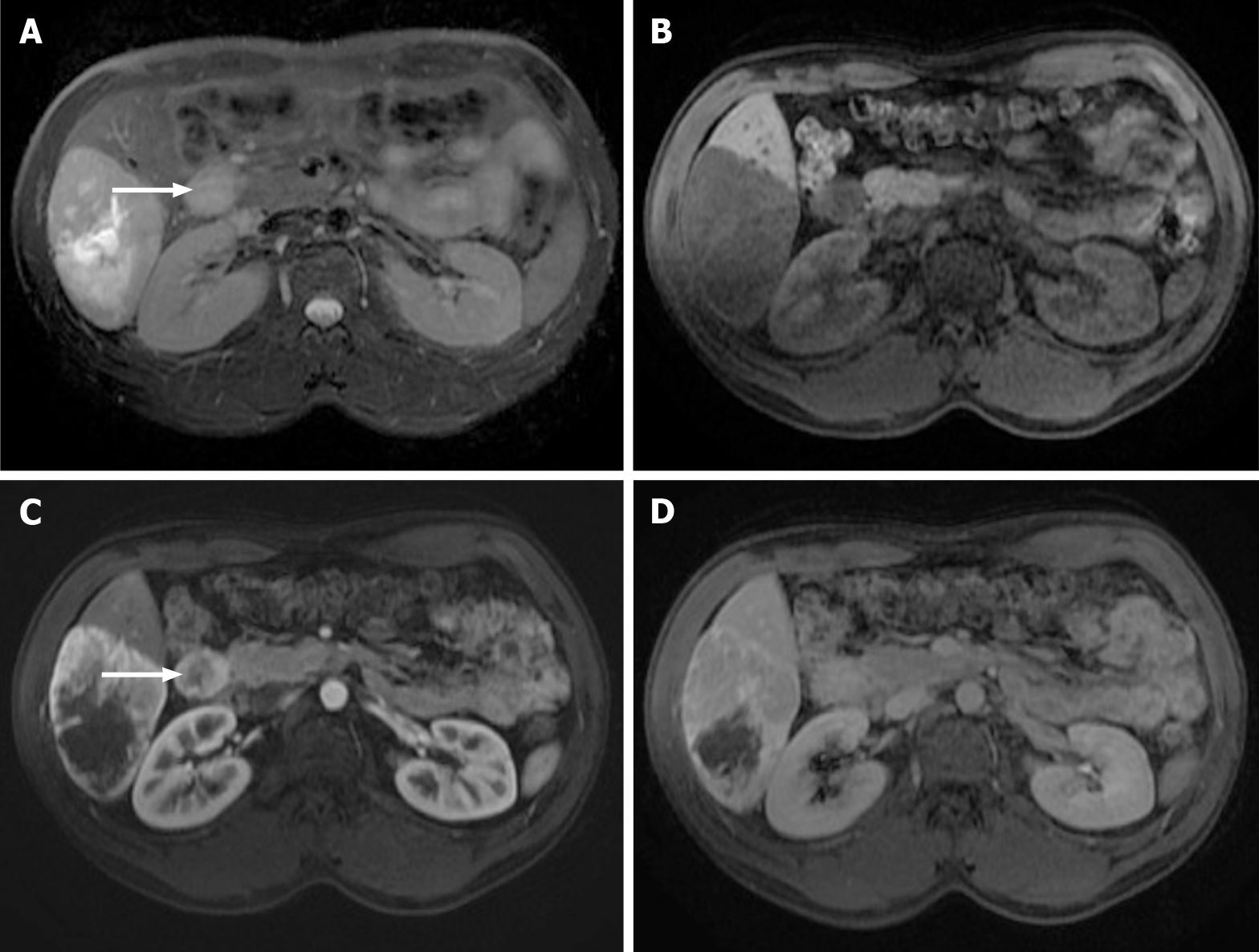
Contrary to CT, non-enhanced sequences in MRI are essential for the detection and characterization of liver metastases. Frequently, metastases are hypo- to isointense on T1-WI sequences and mildly hyperintense on T2-WI[1]. However, some liver metastases, such as those derived from NETs and sarcomas, may show moderately high signal on T2-WI. Moreover, cystic and necrotic metastases (such as from ovary tumors, NETs, melanoma, and sarcomas) may show moderately to markedly high T2 signal intensity[3]. Liver metastases may occasionally present intralesional hemorrhage, fat, or glycogen deposition and appear hyperintense on T1-WI. Also, melanoma and mucinous adenocarcinoma metastases may show high signal on T1-WI due to their high melanocytic and mucin content, respectively (Figure 4). Occasionally, they may appear as a target sign on T2-WI sequences, characterized by hyperintense central necrosis delimited by a lesser intense rim of viable tumor. On T1-WI, a hypointense rim surrounding a center of even lower signal intensity is known as the doughnut sign (Figure 5)[1,6].
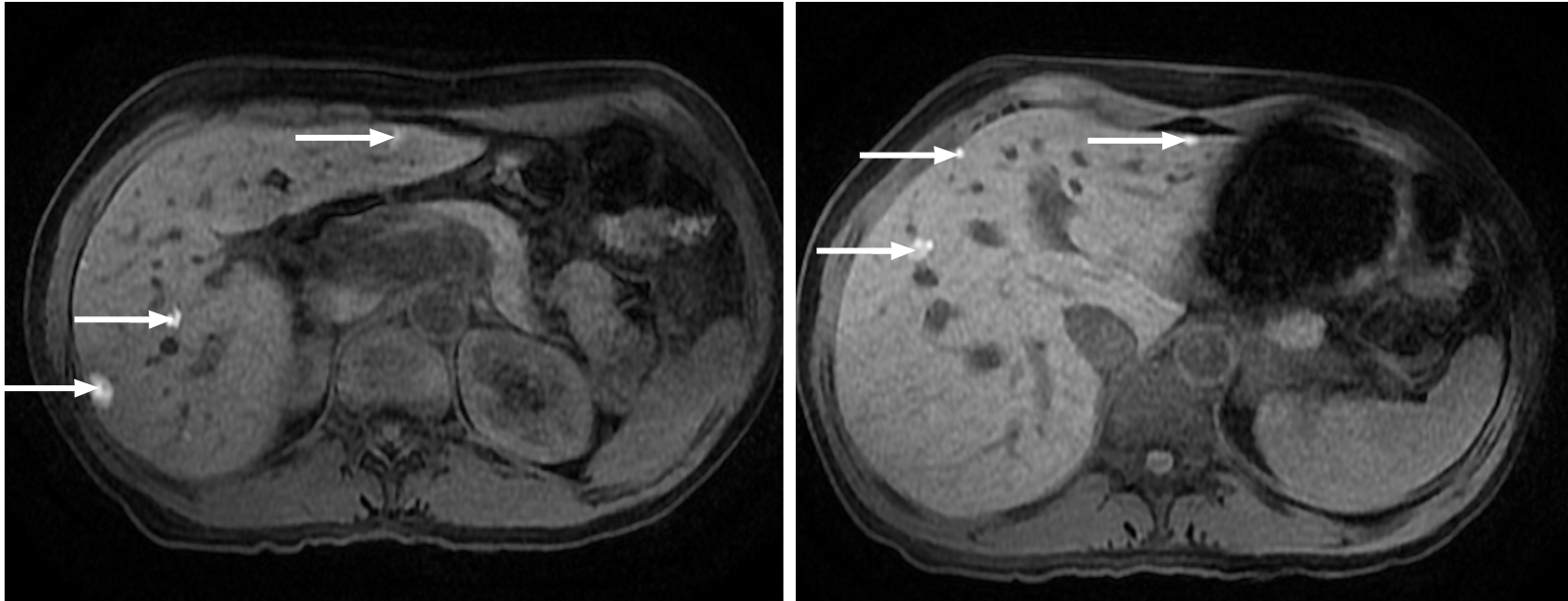
Diffusion-WI (DWI)-MRI allows the interrogation of lesions’ cellularity, taking advantage of water molecules’ movement. Tissues with high cellularity (tumor, fibrosis, abscess, and cytotoxic edema) show restricted diffusion[1]. Diffusion may be quantified by the apparent diffusion coefficient (ADC), and low ADC values correspond to restriction. ADC values are reported to vary between 0.94-2.87; however, there may be an overlap between the ADC values for primary malignant hepatocellular lesions, such as hepatocellular carcinoma and benign hepatocellular lesions[34]. In clinical practice, the evaluation of DWI relies on subjective appreciation. DWI may also pose disadvantages due to the inherent low spatial resolution, low signal-to-noise ratio, and predisposition to artifacts, especially for subcapsular/subdiaphragmatic lesions.
Kim et al[35] reported a higher sensitivity for DWI when compared to CT (79% vs 50%) in the detection of small liver metastases (< 1 cm) (Figure 6). Other studies concluded that DWI is more sensitive than unenhanced T2-WI (88%-91% vs 45%-62%), and the difference is even more obvious when only small metastases are considered (85% vs 35%)[36,37].
For the characterization of liver metastases, it is crucial to combine pre- and post-contrast sequences as mentioned above. After entering the liver via the portal vein and hepatic artery, the extracellular gadolinium-based contrast agent (GBCA) is distributed through the extracellular interstitial space[1]. The desired effect is tissue enhancement on T1-WI, which is achieved by shortening the T1 and T2 relaxation times of adjacent hydrogen protons. The suggested dose for liver imaging is 0.1 mmol/kg, administered through a bolus injection at 2-3 mL/s[38]. Compared to iodine-based contrast agents (used on CT), a greater sensitivity and greater perception of enhancement are observed with GBCAs. GBCAs are considered safe, primarily because they are not nephrotoxic at the recommended doses and show fewer acute reactions than iodinated contrast agents. Although some centers still refrain from using GBCAs in renal impaired patients, one should know that class II contrast agents are rarely associated with nephrogenic systemic fibrosis. A risk-benefit analysis for every individual is required[39,40].
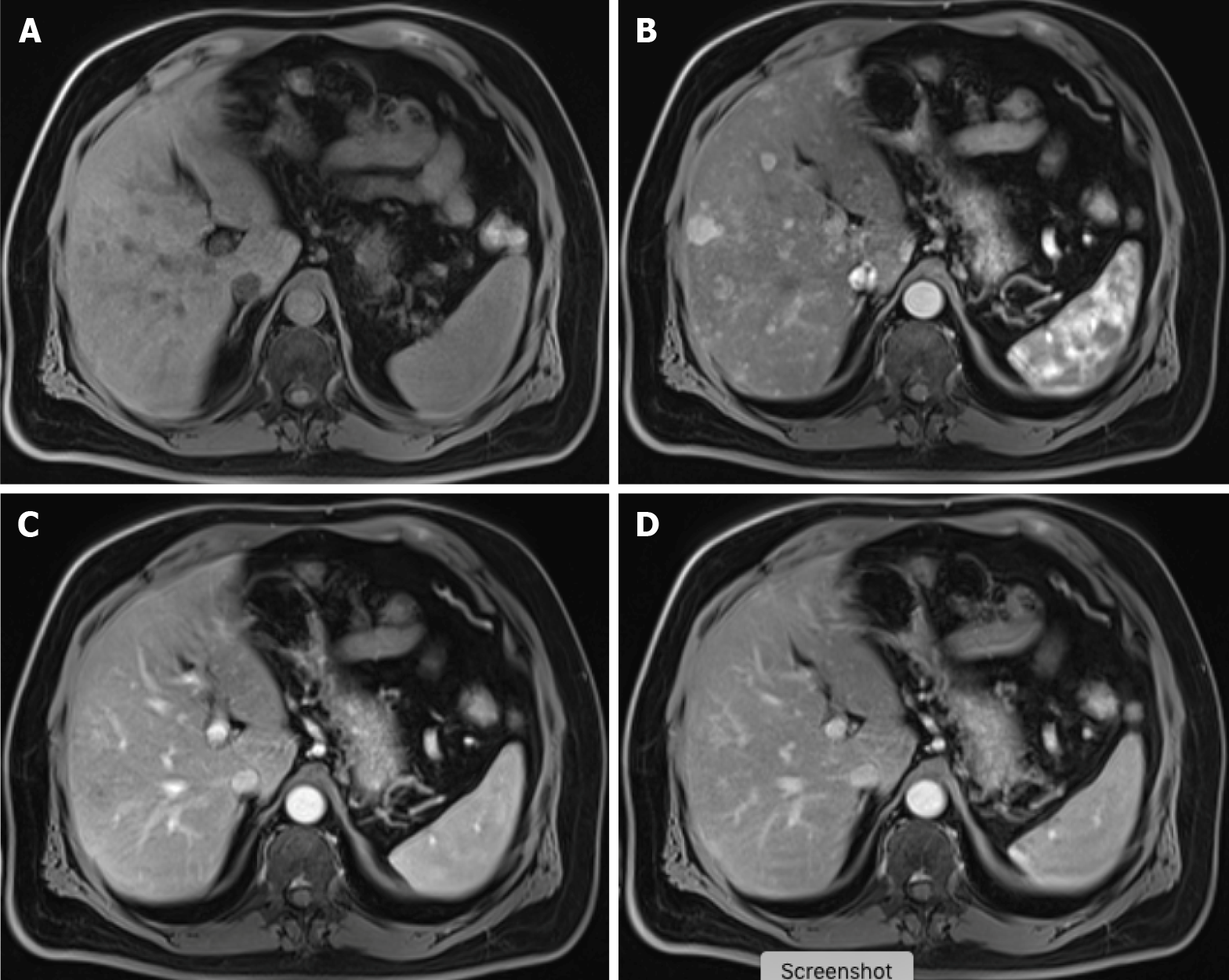
As observed with CT, the characteristics of liver metastases vary with the primary tumor. Hypervascular metastases show a hyperintense signal in the LAP, and hypovascular metastases appear hypointense in the PVP (Figure 7). Hypovascular metastases tend to show a thin peripheral rim type enhancement in the LAP and PVP, with progressive central enhancement in interstitial phases (Figure 8)[3]. In the LAP, hypervascular metastases may show homogeneous enhancement (if smaller than 2 cm) or heterogeneous enhancement (if larger than 2 cm), demonstrating variable degrees of washout or in delayed phases (Figure 9). Isovascular metastases may be seen in breast cancer and avascular metastases on cystic metastases, such as ovarian cancer, and may demonstrate septal or wall enhancement (Figure 10). Chemotherapy-treated metastases may appear isovascular or avascular.
After being distributed in the vascular and extra-vascular space during the LAP, PVP, and delayed phases, hepatocyte-specific contrast agents are incorporated by functioning hepatocytes. The available hepatocyte-specific MRI contrast agents are gadobenate dimeglumine (Gd-BOPTA; MultiHance), with a recommended dose of 0.1 mmol/kg, and gadoxetic acid (Gb-EOB-DTPA; Primovist/Eovist), with a recommended dose of 0.025 mmol/kg[38]. The hepatobiliary phase is acquired after 90-150 min for MultiHance and 15-20 min for Primovist. These temporal differences for the hepatocyte phases are related to the degree of biliary excretion, estimated at 3%-5% for MultiHance and 50% for Primovist[1]. The kidneys excrete the remaining.
The normal functioning hepatocytes uptake the hepatocyte-specific MRI contrast agents and excrete them into the biliary system due to cellular membrane transporters. The contrast agent is responsible for shortening the T1 relaxation, which results in higher signal intensity of the healthy liver parenchyma on T1-WI in the hepatobiliary phase[1]. In the later (hepatobiliary) phase, there is also a subsequent excretion into the biliary canaliculi, allowing imaging of the biliary pathways. Therefore, the hepatobiliary phase is easily recognized because the normal liver parenchyma and bile ducts appear enhanced[41]. Non-hepatocellular lesions, as well as lesions with impaired hepatocytes, appear hypointense. In short, as liver metastases lack functioning hepatocytes and biliary ducts, they appear hypointense in the hepatobiliary phase. Allergic reactions are infrequent and comparable with those of extracellular GBCAs.
In a recent meta-analysis, Zhang et al[42] showed that the sensitivity of gadobenate (MultiHance) for detecting liver metastases on a per-lesion basis for pre-contrast and combined dynamic, delayed hepatobiliary phase imaging was 77.8%, 88.1%, and 95.1%, respectively. These results are comparable to those reported for gadoxetate (Primovist/Eovist).
Resembling only the MRI's specificities, a meta-analysis published in 2016 showed that the sensitivity of DWI and gadoxetic acid-enhanced MRI (GA-MRI) was 87.1% and 90.6%, respectively. When both sequences were combined, the sensitivity for detecting liver metastases on a per-lesion basis was the highest (95.5%)[43].
Therefore, MRI plays a crucial role in evaluating liver metastases and is considered the ideal imaging method for detection and follow-up in many university hospitals.
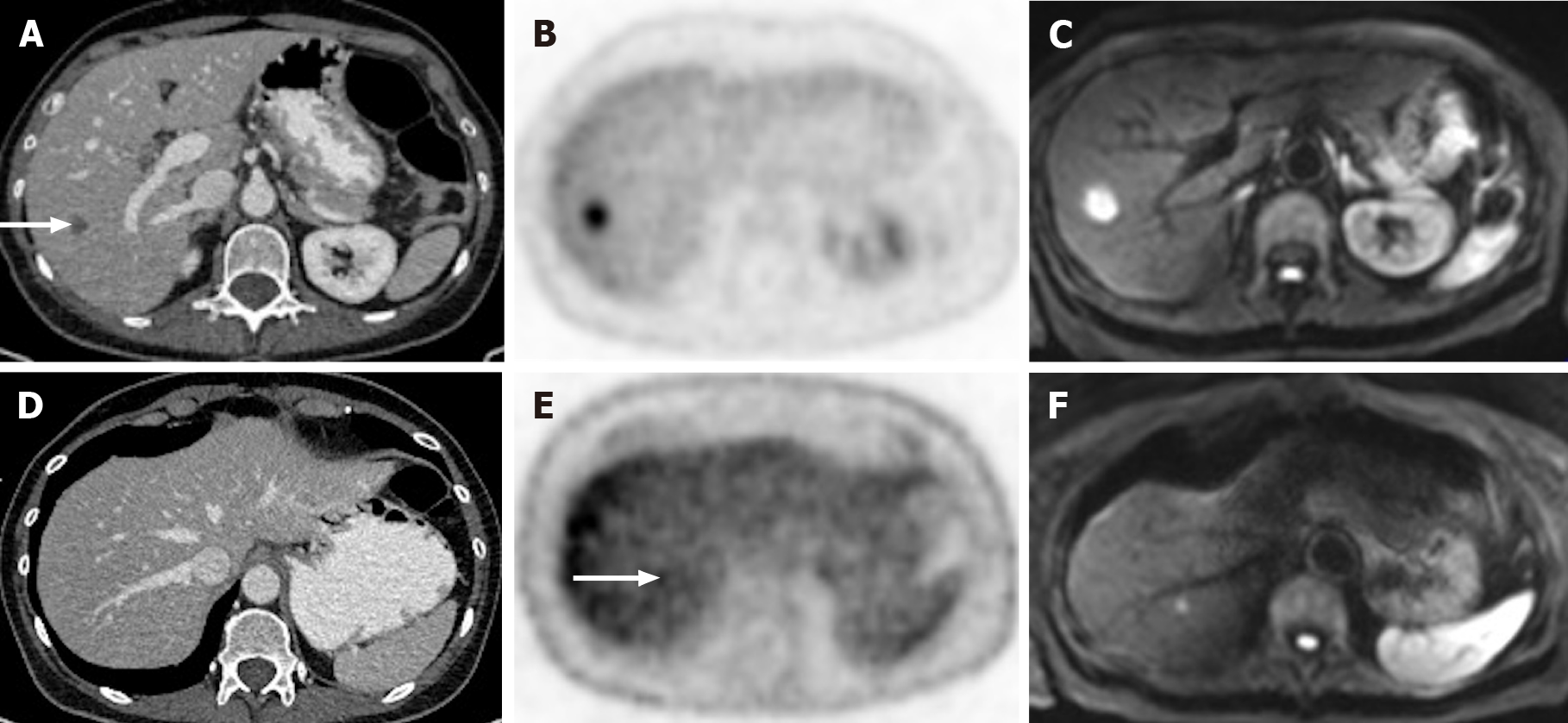
Liver metastases may have significant fluorine-18-labeled fluorodeoxyglucose (FDG) uptake. Previous investigations mentioned the impact of FDG-PET on the detection of such lesions (Figure 11). A meta-analysis published by Maffione et al[44] suggests that FDG-PET/CT is highly accurate in detecting liver metastases on a patient-based analysis, besides showing an added value in identifying extrahepatic disease. However, conventional PET proved to be less sensitive than MRI and CT in detecting CRLM, both on a patient-based (93% vs 100% vs 98%, respectively) and lesion-based analysis (66% vs 89% vs 79%, respectively). In addition to the detection of extrahepatic disease, PET/CT has the advantage of assessing treatment response (i.e., chemotherapy) of liver metastases, demonstrated by a decrease in FDG uptake[1]. However, false negatives may arise immediately after completing a chemotherapy cycle due to residual metabolic inhibition. For this reason, PET/CT is not recommended to be performed earlier than 4 wk after finishing chemotherapy, and a negative result must not be fully trusted[45].
PET/MRI is a more recent technique that combines the advantages of metabolic imaging (FDG-PET) with MRI sensitivity to assess liver metastases. PET/MRI is a helpful diagnostic technique in detecting small hepatic lesions and may improve the evaluation of treatment response after radiation and chemotherapy. Beiderwellen et al[46] demonstrated that PET/MRI has a higher diagnostic accuracy for detecting liver metastases than PET/CT or multidetector CT. However, according to Lake et al[47], there is no significant difference in the diagnostic performance between PET/MRI and Gd-EOB-DTPA MRI. Moreover, PET/MRI also shows an incremental value for detecting additional extrahepatic metastases[47].
It is crucial to detect hepatic metastases as accurately as possible in a per-patient and per-lesion manner to improve patient's clinical evolution, prognosis, and treatment planning. CT, MRI, and FDG-PET are historically the most accurate and precise imaging techniques for this purpose[45]. Below, we refer to various studies comparing these techniques, which will help choose the best option for evaluating liver metastases. Table 1 summarizes the pros and cons of cross sectional techniques.
| Imaging methods | Critical details |
| MRI | Pros: |
| Most accurate method, and superior to CT and PET-CT for the detection of liver metastases: | |
| Especially useful for smaller lesions (< 1 cm), characterization of hypervascular metastases, and in the setting of liver steatosis | |
| High grade of confidence in the distinction between malignant and benign lesions | |
| Anatomic and morphologic evaluation. | |
| Non-enhancing sequences play an important role | |
| Therapy response assessment | |
| Absence of ionizing radiation | |
| Less allergic reactions | |
| May be the most cost-effective option: | |
| Higher detection rate > more curative approach > avoids additional imaging examinations | |
| Cons: | |
| Lower availability | |
| Non-cooperative patients may result in suboptimal study | |
| Limited for pacemaker carriers | |
| Limited use if Glomerular filtration rate < 15 mL/min | |
| CT | Pros: |
| Low cost | |
| Higher availability | |
| Higher sensitivity compared to ultrasonography | |
| Whole-body evaluation | |
| Therapy response assessment | |
| Cons: | |
| Ionizing radiation | |
| Lower sensitivity for the detection of smaller metastases or in the setting of liver steatosis compared to MRI | |
| Low confidence in the distinction between malignant and benign lesions | |
| Not adequate for renal impaired patients | |
| PET-CT | Pros: |
| Accurate detection of extrahepatic disease | |
| Therapy response assessment | |
| Cons: | |
| False negatives after a chemotherapy cycle | |
| Lower sensitivity for small liver metastases | |
| Lower availability | |
| Highest ionizing radiation dose |
Several studies reported that CE-MRI is more sensitive and specific than CE-CT for detecting liver metastases, mainly due to high intrinsic soft-tissue contrast, technical versatility, sensitivity to blood flow, and contrast enhancement and biochemical information[6]. Vreugdenburg et al[32] confirmed in their systematic meta-analysis that in terms of per-lesion diagnostic accuracy, GA-MRI is superior to CE-CT (sensitivity 86.9%-100% vs 51.8%-84.6% and specificity 80.2%-98% vs 77.2%-98%). This difference is more evident in lesions smaller than 10 mm, in which GA-MRI is notably more sensitive but less specific. Based on the reported sensitivity, an equivocal result will happen more frequently with CE-CT, which leads to a modest impact on patient prognosis and management. In 2017, similar results were reported by Choi et al[48], who compared MRI, CT, and PET/CT for the detection of CRC liver metastases, showing a sensitivity of 93.1% vs 82.1% vs 74.1% and specificity of 87.3%, 73.5%, and 93.9%, respectively (Figure 12). MRI showed a better accuracy than CT in detecting CRC liver metastases and presented an incremental value when added to CT alone to detect additional metastases[48]. In this study, the authors reported that neoadjuvant chemotherapy decreases the sensitivity of both CT and MRI; however, it does not significantly affect the sensitivity of PET/CT[48].
The superiority of MRI is self-evident in small metastases. It is supported by various studies, including that by Schulz et al[49], where they reported that the detection of CRLM should rely on MRI. Overall sensitivity/specificity for MRI, CT, and PET was 90%/87%, 68%/94%, and 61%/99%, respectively; and the sensitivity/specificity for lesions smaller than 10 mm for MRI, CT, and PET was 74%/88%, 16%/96% and 9%/98%, respectively[49].
With the introduction of surgical removal of metastatic liver nodules, the overall survival rate has increased. Therefore, it is crucial to ensure the best imaging method to detect them, mainly the smaller ones, which can be easily missed. Ko et al[50] showed that the sensitivity of CT was 8%, 55%, 91%, and 100% for nodules of 1-5 mm, 6-10 mm, 11-15 mm, and > 20 mm, respectively. Consequently, it appears obvious that in metastases that are "too small to characterize," CT has a limited role, particularly for those smaller than 5 mm[50]. However, GA-MRI and CE-CT seem equivalent for detecting lesions larger than 10 mm[21,26].
Maegerlein et al[51] confirmed that MRI was significantly superior (sensitivity of 87.4%) compared to PET/CT (sensitivity of 68.2%).
For metastases in a fatty liver background, the sensitivity of MRI is approximately 85%-88% (vs 65%-68.3% for CE-CT)[18,52]. In these conditions, Kulemann et al[18] found that MRI detects 66% of lesions up to 10 mm, while CT detects only 11%. Therefore, they determined that MRI is superior to CT in detecting CRLM in liver steatosis, especially the smaller ones[18,52].
MRI also showed to be significantly better than CE-CT in the detection and characterization of hypervascular liver metastases. For instance, according to Seemann et al[53], MRI presented a sensitivity of 98.2%, and CT showed a sensitivity of only 37.1% for detecting carcinoid metastases.
Nowadays, debate continues over whether MRI should be a first-line imaging technique for suspected liver metastases. The current European Society for Medical Oncology (ESMO) guidelines for rectal cancer diagnosis and follow-up (2017) consider that MRI is the imaging method of choice for loco-regional staging. However, CT is preferred for distant metastases[54]. Still, these recommendations are relatively poor (level V), and curiously that manuscript does not make any reference to the use of hepatospecific contrast agents[55]. The American College of Radiology in 2017 also stated that "the available evidence supports that both MRI and CT detect liver lesions with high accuracy."
The updated NCCN guidelines (March 2019) for colon and rectal cancer suggest chest, abdominal, and pelvic CT for metastatic disease's initial workup[21,24,56]. However, if surgical resection of hepatic metastases is considered, contrast-enhanced MRI (extracellular or hepatospecific contrast agent) is preferred over CT to assess their number and distribution[56]. Also, PET-CT may be pondered in selected cases with surgical curable M1 disease[21,26].
Many clinicians use the “economic implication” to use CT instead of MRI[55]. Patients often are referred to CT rather than MRI because of the perceived impression that money is being saved in the healthcare system. Zech et al[57] compared the three imaging techniques (GA-MRI, CE-MRI, and CE-CT), considering the diagnostic workup and surgery costs for patients with CRLM. The countries analyzed included Austria, Germany, Italy, Sweden, Switzerland, and Thailand and all of them showed an overall lower cost with GA-MRI compared to the other techniques[57]. The reason is that no patient needed any additional imaging technique to achieve a decision concerning the treatment in the group that used GA-MRI as the initial imaging method. However, in the group of patients submitted to extracellular CE-MRI and CE-CT as an initial approach, approximately 18.1% and 39.7%, respectively, performed an additional examination. Furthermore, it was also noted that the costs of surgery were higher in the GA-MRI group since more liver metastases were detected and consequently needed surgery for a curative approach.
According to these data, we concur that GA-MRI shows a superior sensitivity in detecting hepatic metastases, which leads to a more curative approach, avoids additional imaging examinations, and can be the most cost-effective option. Sadly, these studies did not significantly affect the current clinical guidelines, especially the latest consensus of ESMO, where MRI is still considered a second-line method[45,54].
In addition, according to a recent study, laparoscopic liver ultrasound might improve liver staging for CRLM compared to liver-specific contrast-enhanced MRI (sensitivity of 93.1% vs 85.6%)[58].
Approximately 80% of CRLM are unresectable at initial presentation, and chemotherapy is the treatment of choice (Figure 13). Some studies have reported that some of these lesions might respond to chemotherapy and become resectable, showing better long-term results than “conversion chemotherapy”[59]. As above-mentioned, these patients submitted to neoadjuvant chemotherapy may then appear with liver steatosis, especially after irinotecan and 5-FU or with sinusoidal obstruction (oxaliplatin), which may limit CT liver evaluation[21,60].
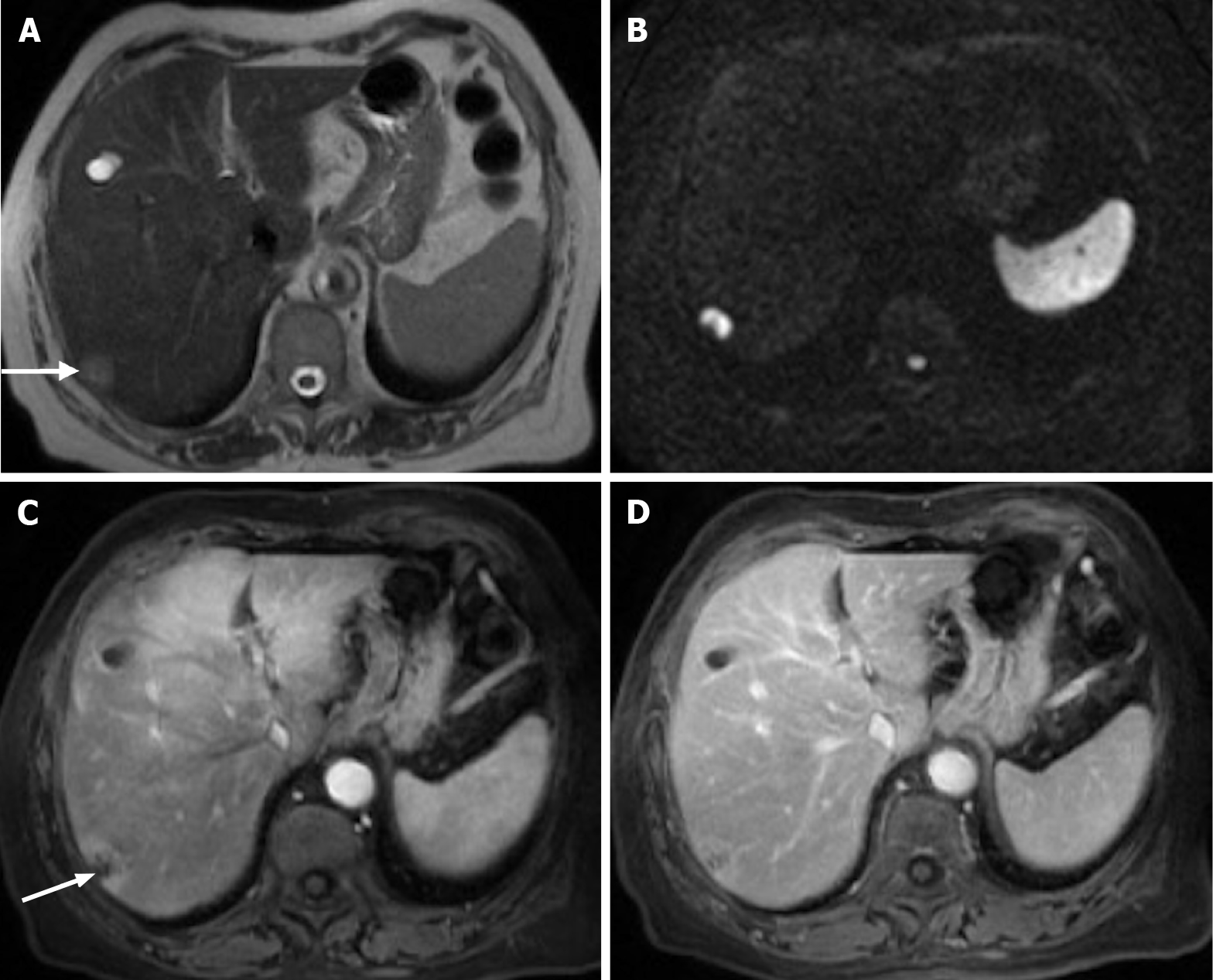
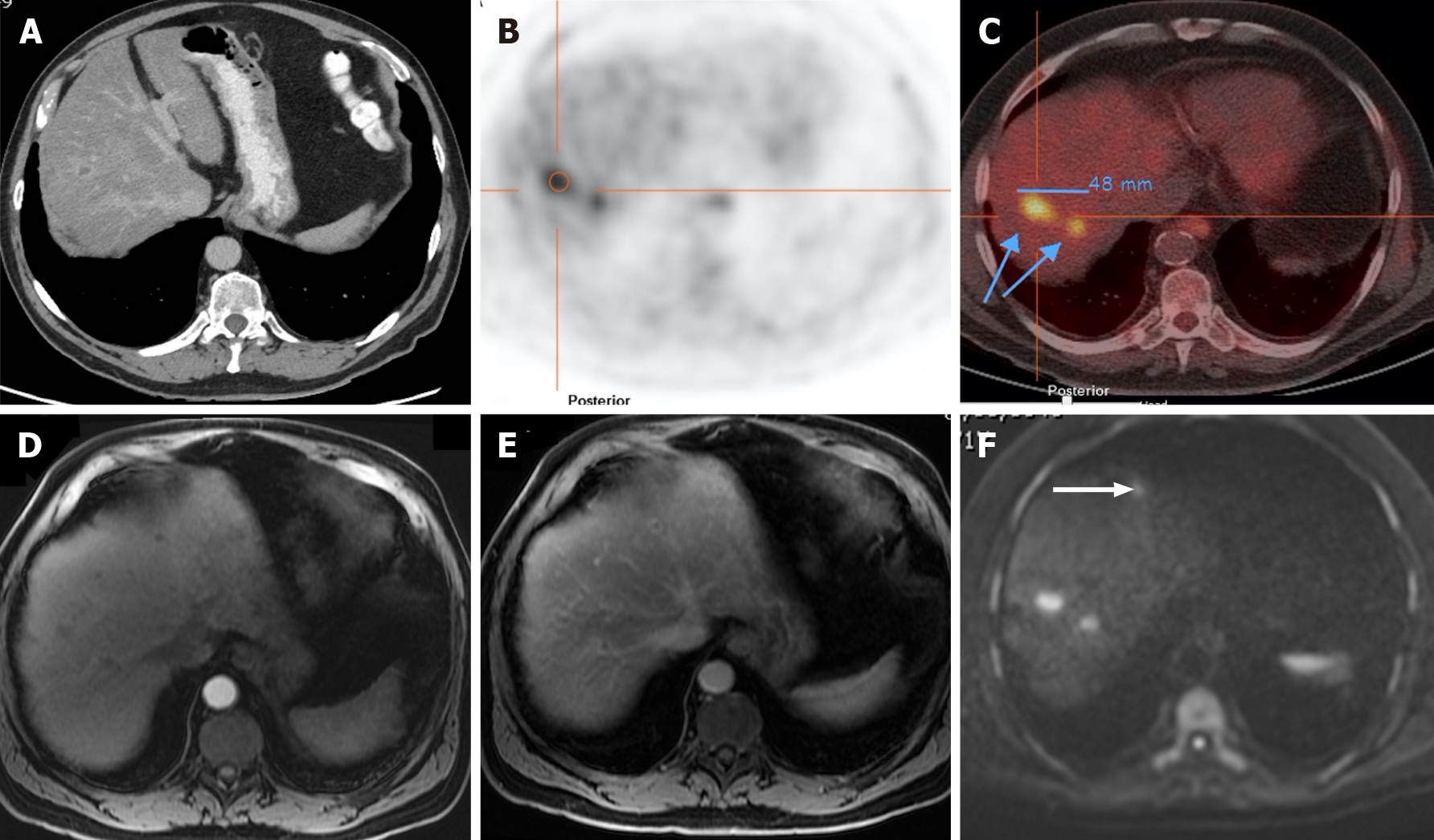
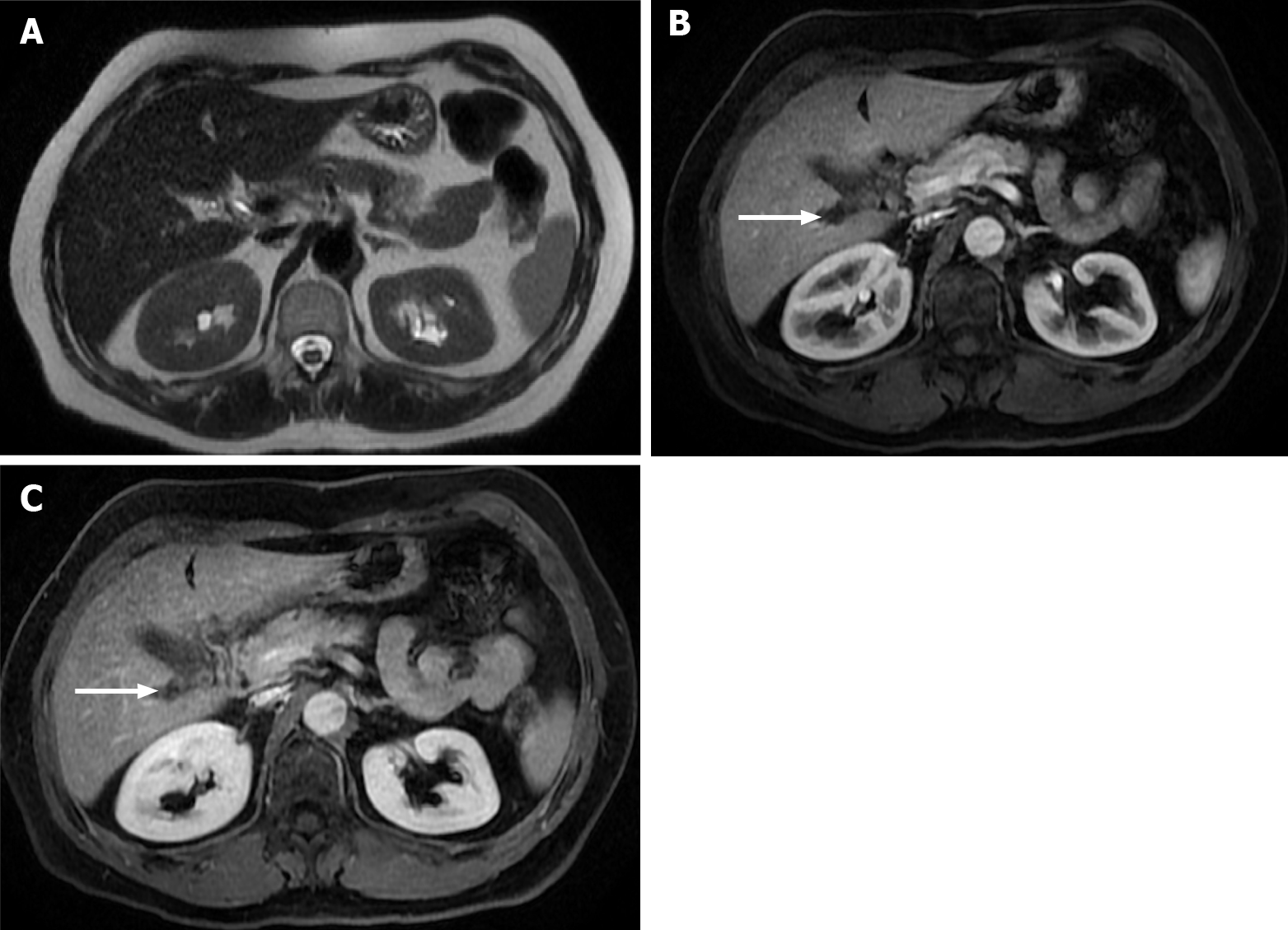
In follow-up studies of CRLM, CT may be used to evaluate response to systemic chemotherapy. In contrast, MRI (with hepatospecific contrast agent and DWI sequences) can be used to assess metastases after neoadjuvant chemotherapy, to assess resectability, and to estimate "disappearing” or “vanishing” metastases (DLM) (Figure 14)[21]. This term corresponds to complete radiologic response – treated metastases that are too small to be detected at follow-up imaging studies – ranging from 7%-24% in CRLM[21,61].
Barimani et al[61] showed that the combination of CE-CT, MRI, and intraoperative ultrasound (IOUS) showed promising results in detecting DLM in CRLM. Furthermore, it was suggested that when DLM remains undetectable by MRI and IOUS, it is a valid option to leave DLM in situ as an alternative approach to surgical resection.
According to Jhaveri et al[62], GA-MRI is superior to CE-CT for the detection of small CRLM (< 1 cm) in both categories of non-treated patients and those who underwent neoadjuvant chemotherapy.
In 2017, a study by Park et al[63] also concluded that MRI has a higher positive predictive value for the absence of tumors after chemotherapy than CT (78% vs 35.2%, respectively).
The RECIST criteria were developed to reach a standardized pattern of tumor response evaluation[64]. These criteria show limitations and appear inadequate for patients treated with immune checkpoint inhibitors due to the "pseudoprogression" phenomenon. Pseudoprogression may occur when molecular target agents diminish the tumor attenuation and enhancement to a lesser degree when compared to the surrounding liver, making the preexisting lesion now visible and mimicking disease progression. To assess this limitation, iRECIST criteria, based on RECIST-based measurements and immune-related response patterns, have been developed[55]. However, iRECIST criteria still need validation.
RECIST evaluation concerning CRLM often fails to identify clinically meaningful responses to bevacizumab-containing therapy. In this matter, Liu et al[65] created a developed-RECIST (D-RECIST) by combining CE-MRI and DWI-MRI. They showed that responders employing D-RECIST had a longer median disease-free survival than non-responders and that defined responses provided important prognostic information. It was concluded that D-RECIST might serve as a better response evaluation than RECIST in CRLM treated with bevacizumab-chemotherapy.
Some morphologic and dynamic features of liver metastases in MRI may predict the response before therapy[21,66]. For instance, a study showed that tumors with lower ADC values correlate with a better response to chemotherapy, while others report a poorer survival[67].
Besides chemotherapy, ablative therapies such as microwave ablation, transarterial chemoembolization, and radioembolization lead to a low-density lesion on CT and high T1 signal / low T2 signal on MRI due to coagulative necrosis[3]. These areas tend to shrink progressively with time. The existence of thick linear peripheral enhan
The liver is one of the most common organs involved with metastatic disease. Both CT and MRI are currently the techniques that show the highest diagnostic performance and are also the most suitable for assessing therapy response and follow-up. Studies have shown that MRI plays a crucial role and has a higher sensitivity in evaluating liver metastases. Therefore, it may be the ideal imaging method for treatment planning before and after neoadjuvant chemotherapy and is also considered the best technique for detection and follow-up in many university hospitals.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Radiology, nuclear medicine and medical imaging
Country/Territory of origin: Portugal
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Fakhradiyev I, Koustas E S-Editor: Liu M L-Editor: Wang TQ P-Editor: Liu M
| 1. | Lincke T, Zech CJ. Liver metastases: Detection and staging. Eur J Radiol. 2017;97:76-82. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 2. | Namasivayam S, Martin DR, Saini S. Imaging of liver metastases: MRI. Cancer Imaging. 2007;7:2-9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 144] [Cited by in RCA: 149] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 3. | Matos AP, Altun E, Ramalho M, Velloni F, AlObaidy M, Semelka RC. An overview of imaging techniques for liver metastases management. Expert Rev Gastroenterol Hepatol. 2015;9:1561-1576. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Silva AC, Evans JM, McCullough AE, Jatoi MA, Vargas HE, Hara AK. MR imaging of hypervascular liver masses: a review of current techniques. Radiographics. 2009;29:385-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 87] [Cited by in RCA: 61] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 5. | Gore RM, Thakrar KH, Wenzke DR, Newmark GM, Mehta UK, Berlin JW. That liver lesion on MDCT in the oncology patient: is it important? Cancer Imaging. 2012;12:373-384. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 6. | Sica GT, Ji H, Ros PR. CT and MR imaging of hepatic metastases. AJR Am J Roentgenol. 2000;174:691-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 118] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 7. | Albrecht T. Detection and Characterisation of Liver Metastases. In: Enhancing the Role of Ultrasound with Contrast Agents. 1st ed. Lencioni R editor. Milano: Springer, 2006: 53-67. [DOI] [Full Text] |
| 8. | Cantisani V, Grazhdani H, Fioravanti C, Rosignuolo M, Calliada F, Messineo D, Bernieri MG, Redler A, Catalano C, D'Ambrosio F. Liver metastases: Contrast-enhanced ultrasound compared with computed tomography and magnetic resonance. World J Gastroenterol. 2014;20:9998-10007. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 57] [Cited by in RCA: 57] [Article Influence: 5.2] [Reference Citation Analysis (1)] |
| 9. | Tirumani SH, Kim KW, Nishino M, Howard SA, Krajewski KM, Jagannathan JP, Cleary JM, Ramaiya NH, Shinagare AB. Update on the role of imaging in management of metastatic colorectal cancer. Radiographics. 2014;34:1908-1928. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 68] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 10. | Kong WT, Ji ZB, Wang WP, Cai H, Huang BJ, Ding H. Evaluation of Liver Metastases Using Contrast-Enhanced Ultrasound: Enhancement Patterns and Influencing Factors. Gut Liver. 2016;10:283-287. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 23] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 11. | Bernatik T, Strobel D, Hahn EG, Becker D. Detection of liver metastases: comparison of contrast-enhanced wide-band harmonic imaging with conventional ultrasonography. J Ultrasound Med. 2001;20:509-515. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 34] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 12. | Piscaglia F, Lencioni R, Sagrini E, Pina CD, Cioni D, Vidili G, Bolondi L. Characterization of focal liver lesions with contrast-enhanced ultrasound. Ultrasound Med Biol. 2010;36:531-550. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 79] [Cited by in RCA: 82] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 13. | Cantisani V, Ricci P, Erturk M, Pagliara E, Drudi F, Calliada F, Mortele K, D'Ambrosio U, Marigliano C, Catalano C, Marin D, Di Seri M, Longo F, Passariello R. Detection of hepatic metastases from colorectal cancer: prospective evaluation of gray scale US vs SonoVue® low mechanical index real time-enhanced US as compared with multidetector-CT or Gd-BOPTA-MRI. Ultraschall Med. 2010;31:500-505. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 41] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 14. | Vialle R, Boucebci S, Richer JP, Velasco S, Herpe G, Vesselle G, Ingrand P, Tasu JP. Preoperative detection of hepatic metastases from colorectal cancer: Prospective comparison of contrast-enhanced ultrasound and multidetector-row computed tomography (MDCT). Diagn Interv Imaging. 2016;97:851-855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 15. | El Kaffas A, Sigrist RMS, Fisher G, Bachawal S, Liau J, Wang H, Karanany A, Durot I, Rosenberg J, Hristov D, Willmann JK. Quantitative Three-Dimensional Dynamic Contrast-Enhanced Ultrasound Imaging: First-In-Human Pilot Study in Patients with Liver Metastases. Theranostics. 2017;7:3745-3758. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 39] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 16. | Lee JC, Yan K, Lee SK, Yang W, Chen MH. Focal Liver Lesions: Real-time 3-Dimensional Contrast-Enhanced Ultrasonography Compared With 2-Dimensional Contrast-Enhanced Ultrasonography and Magnetic Resonance Imaging. J Ultrasound Med. 2017;36:2015-2026. [PubMed] [DOI] [Full Text] |
| 17. | Nakai H, Arizono S, Isoda H, Togashi K. Imaging Characteristics of Liver Metastases Overlooked at Contrast-Enhanced CT. AJR Am J Roentgenol. 2019;212:782-787. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 27] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 18. | Kulemann V, Schima W, Tamandl D, Kaczirek K, Gruenberger T, Wrba F, Weber M, Ba-Ssalamah A. Preoperative detection of colorectal liver metastases in fatty liver: MDCT or MRI? Eur J Radiol. 2011;79:e1-e6. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 78] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 19. | Sahani DV, Bajwa MA, Andrabi Y, Bajpai S, Cusack JC. Current status of imaging and emerging techniques to evaluate liver metastases from colorectal carcinoma. Ann Surg. 2014;259:861-872. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 45] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 20. | Kamaya A, Maturen KE, Tye GA, Liu YI, Parti NN, Desser TS. Hypervascular liver lesions. Semin Ultrasound CT MR. 2009;30:387-407. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 33] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 21. | Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18694] [Cited by in RCA: 21371] [Article Influence: 2137.1] [Reference Citation Analysis (3)] |
| 22. | Honda Y, Higaki T, Higashihori H, Monzen Y, Tatsugami F, Date S, Awai K. Re-evaluation of detectability of liver metastases by contrast-enhanced CT: added value of hepatic arterial phase imaging. Jpn J Radiol. 2014;32:467-475. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 23. | Ferlay J, Colombet M, Soerjomataram I, Dyba T, Randi G, Bettio M, Gavin A, Visser O, Bray F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries and 25 major cancers in 2018. Eur J Cancer. 2018;103:356-387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1625] [Cited by in RCA: 1660] [Article Influence: 237.1] [Reference Citation Analysis (0)] |
| 24. | Sadigh G, Applegate KE, Baumgarten DA. Comparative accuracy of intravenous contrast-enhanced CT vs noncontrast CT plus intravenous contrast-enhanced CT in the detection and characterization of patients with hypervascular liver metastases: a critically appraised topic. Acad Radiol. 2014;21:113-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 25. | Hale HL, Husband JE, Gossios K, Norman AR, Cunningham D. CT of calcified liver metastases in colorectal carcinoma. Clin Radiol. 1998;53:735-741. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 25] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 26. | Paulatto L, Dioguardi Burgio M, Sartoris R, Beaufrère A, Cauchy F, Paradis V, Vilgrain V, Ronot M. Colorectal liver metastases: radiopathological correlation. Insights Imaging. 2020;11:99. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 24] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 27. | Benoist S, Brouquet A, Penna C, Julié C, El Hajjam M, Chagnon S, Mitry E, Rougier P, Nordlinger B. Complete response of colorectal liver metastases after chemotherapy: does it mean cure? J Clin Oncol. 2006;24:3939-3945. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 423] [Cited by in RCA: 385] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 28. | Jo BG, Song YG, Shim SG, Kim YW. Comparison of enhancement and image quality: different iodine concentrations for liver on 128-slice multidetector computed tomography in the same chronic liver disease patients. Korean J Intern Med. 2016;31:461-469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 29. | Rajiah P, Parakh A, Kay F, Baruah D, Kambadakone AR, Leng S. Update on Multienergy CT: Physics, Principles, and Applications. Radiographics. 2020;40:1284-1308. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 30. | Lenga L, Lange M, Arendt CT, Booz C, Yel I, Bodelle B, D'Angelo T, Hammerstingl RM, Huizinga NA, Vogl TJ, Martin SS, Albrecht MH. Measurement Reliability and Diagnostic Accuracy of Virtual Monoenergetic Dual-Energy CT in Patients with Colorectal Liver Metastases. Acad Radiol. 2020;27:e168-e175. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 21] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 31. | Lenga L, Lange M, Arendt CT, Yel I, Booz C, Durden J, Leithner D, Vogl TJ, Albrecht MH, Martin SS. Can Dual-energy CT-based Virtual Monoenergetic Imaging Improve the Assessment of Hypodense Liver Metastases in Patients With Hepatic Steatosis? Acad Radiol. 2021;28:769-777. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 32. | Vreugdenburg TD, Ma N, Duncan JK, Riitano D, Cameron AL, Maddern GJ. Comparative diagnostic accuracy of hepatocyte-specific gadoxetic acid (Gd-EOB-DTPA) enhanced MR imaging and contrast enhanced CT for the detection of liver metastases: a systematic review and meta-analysis. Int J Colorectal Dis. 2016;31:1739-1749. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 81] [Cited by in RCA: 75] [Article Influence: 8.3] [Reference Citation Analysis (1)] |
| 33. | Lee KH, Lee JM, Park JH, Kim JH, Park HS, Yu MH, Yoon JH, Han JK, Choi BI. MR imaging in patients with suspected liver metastases: value of liver-specific contrast agent gadoxetic acid. Korean J Radiol. 2013;14:894-904. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 34. | Kanematsu M, Kondo H, Goshima S, Kato H, Tsuge U, Hirose Y, Kim MJ, Moriyama N. Imaging liver metastases: review and update. Eur J Radiol. 2006;58:217-228. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 77] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 35. | Kim HJ, Lee SS, Byun JH, Kim JC, Yu CS, Park SH, Kim AY, Ha HK. Incremental value of liver MR imaging in patients with potentially curable colorectal hepatic metastasis detected at CT: a prospective comparison of diffusion-weighted imaging, gadoxetic acid-enhanced MR imaging, and a combination of both MR techniques. Radiology. 2015;274:712-722. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 90] [Cited by in RCA: 107] [Article Influence: 9.7] [Reference Citation Analysis (1)] |
| 36. | Bruegel M, Rummeny EJ. Hepatic metastases: use of diffusion-weighted echo-planar imaging. Abdom Imaging. 2010;35:454-461. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 30] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 37. | Ward J. New MR techniques for the detection of liver metastases. Cancer Imaging. 2006;6:33-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 56] [Cited by in RCA: 57] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 38. | Giuga M, De Gaetano AM, Guerra A, Infante A, Iezzi R, Spinelli I, Siciliano M, Grieco A, Rapaccini GL, Gasbarrini A, Pompili M, Bonomo L. An update on clinical applications of hepatospecific contrast media in magnetic resonance imaging of liver parenchyma. Eur Rev Med Pharmacol Sci. 2016;20:2515-2525. [PubMed] |
| 39. | Schieda N, Maralani PJ, Hurrell C, Tsampalieros AK, Hiremath S. Updated Clinical Practice Guideline on Use of Gadolinium-Based Contrast Agents in Kidney Disease Issued by the Canadian Association of Radiologists. Can Assoc Radiol J. 2019;70:226-232. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (0)] |
| 40. | Goldstein KM, Lunyera J, Mohottige D, Amrhein TJ, Alexopoulos AS, Campbell H, Cameron CB, Sagalla N, Crowley MJ, Dietch JR, Gordon AM, Kosinski AS, Cantrell S, Williams Jr JW, Gierisch JM. Risk of Nephrogenic Systemic Fibrosis after Exposure to Newer Gadolinium Agents. Washington (DC): Department of Veterans Affairs, 2019. [PubMed] |
| 41. | Hazhirkarzar B, Khoshpouri P, Shaghaghi M, Ghasabeh MA, Pawlik TM, Kamel IR. Current state of the art imaging approaches for colorectal liver metastasis. Hepatobiliary Surg Nutr. 2020;9:35-48. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 42. | Zhang L, Yu X, Huo L, Lu L, Pan X, Jia N, Fan X, Morana G, Grazioli L, Schneider G. Detection of liver metastases on gadobenate dimeglumine-enhanced MRI: systematic review, meta-analysis, and similarities with gadoxetate-enhanced MRI. Eur Radiol. 2019;29:5205-5216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 43. | Vilgrain V, Esvan M, Ronot M, Caumont-Prim A, Aubé C, Chatellier G. A meta-analysis of diffusion-weighted and gadoxetic acid-enhanced MR imaging for the detection of liver metastases. Eur Radiol. 2016;26:4595-4615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 106] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 44. | Maffione AM, Lopci E, Bluemel C, Giammarile F, Herrmann K, Rubello D. Diagnostic accuracy and impact on management of (18)F-FDG PET and PET/CT in colorectal liver metastasis: a meta-analysis and systematic review. Eur J Nucl Med Mol Imaging. 2015;42:152-163. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 96] [Cited by in RCA: 106] [Article Influence: 9.6] [Reference Citation Analysis (0)] |
| 45. | Glazer ES, Beaty K, Abdalla EK, Vauthey JN, Curley SA. Effectiveness of positron emission tomography for predicting chemotherapy response in colorectal cancer liver metastases. Arch Surg. 2010;145:340-5; discussion 345. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 46. | Beiderwellen K, Geraldo L, Ruhlmann V, Heusch P, Gomez B, Nensa F, Umutlu L, Lauenstein TC. Accuracy of [18F]FDG PET/MRI for the Detection of Liver Metastases. PLoS One. 2015;10:e0137285. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 50] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 47. | Lake ES, Wadhwani S, Subar D, Kauser A, Harris C, Chang D, Lapsia S. The influence of FDG PET-CT on the detection of extrahepatic disease in patients being considered for resection of colorectal liver metastasis. Ann R Coll Surg Engl. 2014;96:211-215. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 48. | Choi SH, Kim SY, Park SH, Kim KW, Lee JY, Lee SS, Lee MG. Diagnostic performance of CT, gadoxetate disodium-enhanced MRI, and PET/CT for the diagnosis of colorectal liver metastasis: Systematic review and meta-analysis. J Magn Reson Imaging. 2018;47:1237-1250. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (0)] |
| 49. | Schulz A, Viktil E, Godt JC, Johansen CK, Dormagen JB, Holtedahl JE, Labori KJ, Bach-Gansmo T, Kløw NE. Diagnostic performance of CT, MRI and PET/CT in patients with suspected colorectal liver metastases: the superiority of MRI. Acta Radiol. 2016;57:1040-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 50. | Ko Y, Kim J, Park JK, Kim H, Cho JY, Kang SB, Ahn S, Lee KJ, Lee KH. Limited detection of small (≤ 10 mm) colorectal liver metastasis at preoperative CT in patients undergoing liver resection. PLoS One. 2017;12:e0189797. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 51. | Maegerlein C, Fingerle AA, Souvatzoglou M, Rummeny EJ, Holzapfel K. Detection of liver metastases in patients with adenocarcinomas of the gastrointestinal tract: comparison of (18)F-FDG PET/CT and MR imaging. Abdom Imaging. 2015;40:1213-1222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 13] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 52. | Abdalla EK, Vauthey JN, Ellis LM, Ellis V, Pollock R, Broglio KR, Hess K, Curley SA. Recurrence and outcomes following hepatic resection, radiofrequency ablation, and combined resection/ablation for colorectal liver metastases. Ann Surg. 2004;239:818-825. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1364] [Cited by in RCA: 1291] [Article Influence: 61.5] [Reference Citation Analysis (0)] |
| 53. | Seemann MD, Meisetschlaeger G, Gaa J, Rummeny EJ. Assessment of the extent of metastases of gastrointestinal carcinoid tumors using whole-body PET, CT, MRI, PET/CT and PET/MRI. Eur J Med Res. 2006;11:58-65. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 54. | Glynne-Jones R, Wyrwicz L, Tiret E, Brown G, Rödel C, Cervantes A, Arnold D; ESMO Guidelines Committee. Rectal cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2017;28:iv22-iv40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1112] [Cited by in RCA: 1199] [Article Influence: 149.9] [Reference Citation Analysis (0)] |
| 55. | Renzulli M, Clemente A, Ierardi AM, Pettinari I, Tovoli F, Brocchi S, Peta G, Cappabianca S, Carrafiello G, Golfieri R. Imaging of Colorectal Liver Metastases: New Developments and Pending Issues. Cancers (Basel). 2020;12:151. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 38] [Article Influence: 7.6] [Reference Citation Analysis (0)] |
| 56. | Benson AB, Venook AP, Al-Hawary MM, Cederquist L, Chen YJ, Ciombor KK, Cohen S, Cooper HS, Deming D, Engstrom PF, Grem JL, Grothey A, Hochster HS, Hoffe S, Hunt S, Kamel A, Kirilcuk N, Krishnamurthi S, Messersmith WA, Meyerhardt J, Mulcahy MF, Murphy JD, Nurkin S, Saltz L, Sharma S, Shibata D, Skibber JM, Sofocleous CT, Stoffel EM, Stotsky-Himelfarb E, Willett CG, Wuthrick E, Gregory KM, Gurski L, Freedman-Cass DA. Rectal Cancer, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J Natl Compr Canc Netw. 2018;16:874-901. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 447] [Cited by in RCA: 683] [Article Influence: 113.8] [Reference Citation Analysis (0)] |
| 57. | Zech CJ, Ba-Ssalamah A, Berg T, Chandarana H, Chau GY, Grazioli L, Kim MJ, Lee JM, Merkle EM, Murakami T, Ricke J, B Sirlin C, Song B, Taouli B, Yoshimitsu K, Koh DM. Consensus report from the 8th International Forum for Liver Magnetic Resonance Imaging. Eur Radiol. 2020;30:370-382. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 57] [Article Influence: 11.4] [Reference Citation Analysis (0)] |
| 58. | Russolillo N, Borello A, Langella S, Casella M, Lo Tesoriere R, Ferrero A. Comparison of laparoscopic ultrasound and liver-specific magnetic resonance imaging for staging colorectal liver metastases. Surg Endosc. 2021;35:3547-3553. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 59. | Basso M, Dadduzio V, Ardito F, Lombardi P, Strippoli A, Vellone M, Orlandi A, Rossi S, Cerchiaro E, Cassano A, Giuliante F, Barone C. Conversion Chemotherapy for Technically Unresectable Colorectal Liver Metastases: A Retrospective, STROBE-Compliant, Single-Center Study Comparing Chemotherapy Alone and Combination Chemotherapy With Cetuximab or Bevacizumab. Medicine (Baltimore). 2016;95:e3722. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 60. | Rubbia-Brandt L, Audard V, Sartoretti P, Roth AD, Brezault C, Le Charpentier M, Dousset B, Morel P, Soubrane O, Chaussade S, Mentha G, Terris B. Severe hepatic sinusoidal obstruction associated with oxaliplatin-based chemotherapy in patients with metastatic colorectal cancer. Ann Oncol. 2004;15:460-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 752] [Cited by in RCA: 764] [Article Influence: 36.4] [Reference Citation Analysis (0)] |
| 61. | Barimani D, Kauppila JH, Sturesson C, Sparrelid E. Imaging in disappearing colorectal liver metastases and their accuracy: a systematic review. World J Surg Oncol. 2020;18:264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 23] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 62. | Jhaveri K, Cleary S, Audet P, Balaa F, Bhayana D, Burak K, Chang S, Dixon E, Haider M, Molinari M, Reinhold C, Sherman M. Consensus statements from a multidisciplinary expert panel on the utilization and application of a liver-specific MRI contrast agent (gadoxetic acid). AJR Am J Roentgenol. 2015;204:498-509. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 66] [Cited by in RCA: 70] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 63. | Park MJ, Hong N, Han K, Kim MJ, Lee YJ, Park YS, Rha SE, Park S, Lee WJ, Park SH, Lee CH, Nam CM, An C, Kim HJ, Kim H, Park MS. Use of Imaging to Predict Complete Response of Colorectal Liver Metastases after Chemotherapy: MR Imaging vs CT Imaging. Radiology. 2017;284:423-431. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 64. | Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M, Rubinstein L, Shankar L, Dodd L, Kaplan R, Lacombe D, Verweij J. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15860] [Cited by in RCA: 21630] [Article Influence: 1351.9] [Reference Citation Analysis (1)] |
| 65. | Liu LH, Zhou GF, Lv H, Wang ZC, Rao SX, Zeng MS. Identifying response in colorectal liver metastases treated with bevacizumab: development of RECIST by combining contrast-enhanced and diffusion-weighted MRI. Eur Radiol. 2021;31:5640-5649. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (1)] |
| 66. | Beckers RCJ, Lambregts DMJ, Lahaye MJ, Rao SX, Kleinen K, Grootscholten C, Beets GL, Beets-Tan RGH, Maas M. Advanced imaging to predict response to chemotherapy in colorectal liver metastases - a systematic review. HPB (Oxford). 2018;20:120-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 29] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 67. | Heijmen L, ter Voert EE, Oyen WJ, Punt CJ, van Spronsen DJ, Heerschap A, de Geus-Oei LF, van Laarhoven HW. Multimodality imaging to predict response to systemic treatment in patients with advanced colorectal cancer. PLoS One. 2015;10:e0120823. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |