Published online Dec 27, 2021. doi: 10.4254/wjh.v13.i12.1828
Peer-review started: March 1, 2021
First decision: July 6, 2021
Revised: July 16, 2021
Accepted: November 13, 2021
Article in press: November 13, 2021
Published online: December 27, 2021
Processing time: 300 Days and 7 Hours
Hepatobiliary disorders are among the most common extraintestinal manifestations in inflammatory bowel diseases (IBD), both in Crohn’s disease and ulcerative colitis (UC), and therefore represent a diagnostic challenge. Immune-mediated conditions include primary sclerosing cholangitis (PSC) as the main form, variant forms of PSC (namely small-duct PSC, PSC-autoimmune hepatitis overlap syndrome and IgG4-related sclerosing cholangitis) and granulomatous hepatitis. PSC is by far the most common, presenting in up to 8% of IBD patients, more frequently in UC. Several genetic foci have been identified, but environmental factors are preponderant on disease pathogenesis. The course of the two diseases is typically independent. PSC diagnosis is based mostly on typical radiological findings and exclusion of secondary cholangiopathies. Risk of cholangiocarcinoma is significantly increased in PSC, as well as the risk of colorectal cancer in patients with PSC and IBD-related colitis. No disease-modifying drugs are approved to date. Thus, PSC management is directed against symptoms and complications and includes medical therapies for pruritus, endoscopic treatment of biliary stenosis and liver transplant for end-stage liver disease. Other non-immune-mediated hepatobiliary disorders are gallstone disease, whose incidence is higher in IBD and reported in up to one third of IBD patients, non-alcoholic fatty liver disease, pyogenic liver abscess and portal vein thrombosis. Drug-induced liver injury (DILI) is an important issue in IBD, since most IBD therapies may cause liver toxicity; however, the incidence of serious adverse events is low. Thiopurines and methotrexate are the most associated with DILI, while the risk related to anti-tumor necrosis factor-α and anti-integrins is low. Data on hepatotoxicity of newer drugs approved for IBD, like anti-interleukin 12/23 and tofacitinib, are still scarce, but the evidence from other rheumatic diseases is reassuring. Hepatitis B reactivation during immunosuppressive therapy is a major concern in IBD, and adequate screening and vaccination is warranted. On the other hand, hepatitis C reactivation does not seem to be a real risk, and hepatitis C antiviral treatment does not influence IBD natural history. The approach to an IBD patient with abnormal liver function tests is complex due to the wide range of differential diagnosis, but it is of paramount importance to make a quick and accurate diagnosis, as it may influence the therapeutic management.
Core Tip: Hepatobiliary disorders are commonly associated with inflammatory bowel diseases (IBD) and represent a management challenge. They include (1) Immune-mediated diseases that can coexist with IBD, mainly primary sclerosing cholangitis; (2) Other non-immune-mediated disorders like gallstone disease; (3) Liver injury induced by drugs used in IBD; and (4) Risks related to concomitant viral hepatitis B and C. All these conditions are summarized in this review, according to the latest literature evidence and the current clinical practice guidelines.
- Citation: Mazza S, Soro S, Verga MC, Elvo B, Ferretti F, Cereatti F, Drago A, Grassia R. Liver-side of inflammatory bowel diseases: Hepatobiliary and drug-induced disorders. World J Hepatol 2021; 13(12): 1828-1849
- URL: https://www.wjgnet.com/1948-5182/full/v13/i12/1828.htm
- DOI: https://dx.doi.org/10.4254/wjh.v13.i12.1828
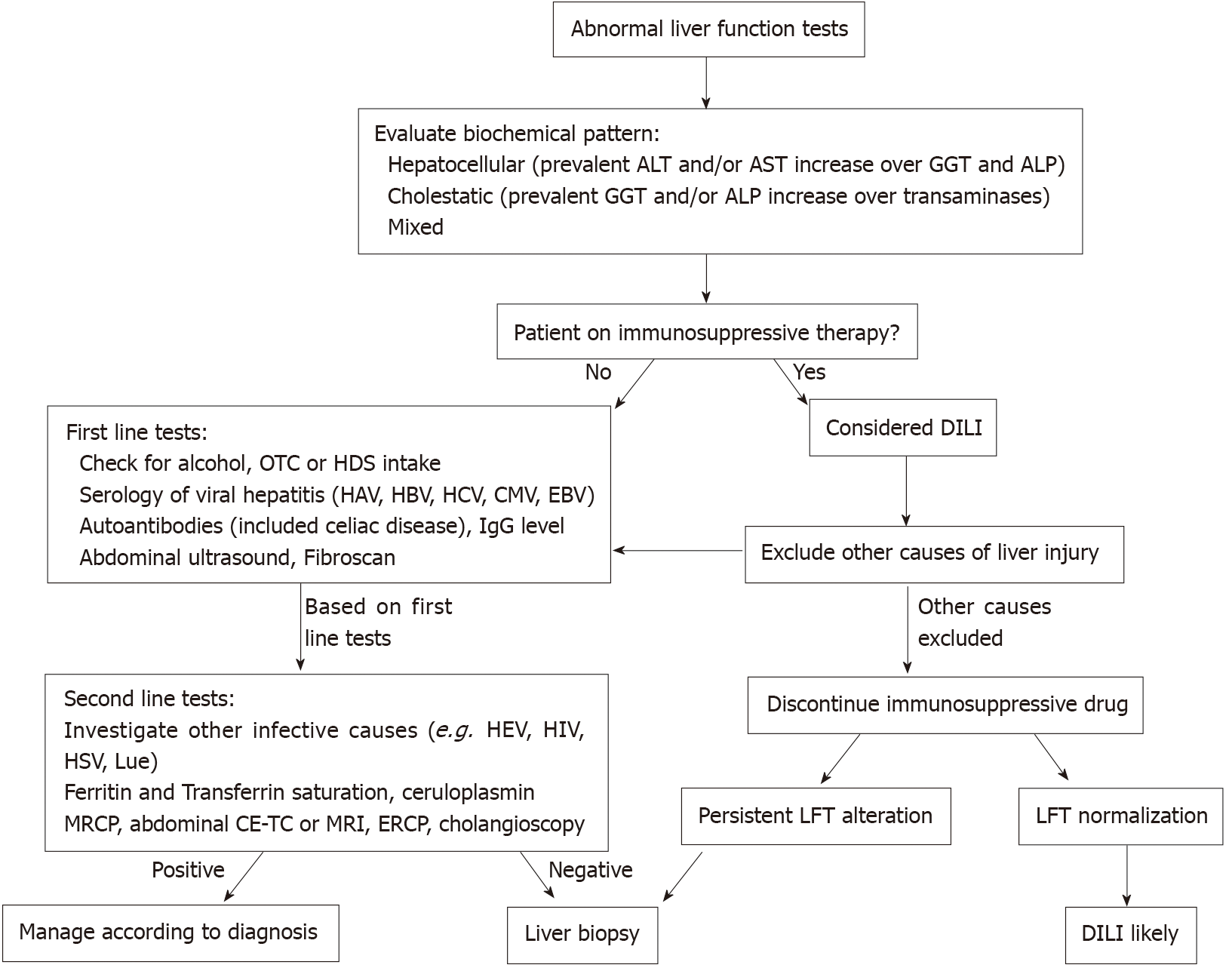
Hepatobiliary disorders are common extraintestinal manifestations of inflammatory bowel diseases (IBD) and may occur in both Crohn’s disease (CD) and ulcerative colitis (UC). The range of IBD-associated hepatobiliary disorders is wide and can underlie different pathogenetic mechanisms. They include diseases with immune-mediated pathogenesis, which typically have a course independent of intestinal activity, the most common being primary sclerosing cholangitis (PSC); variant form of PSC, like small-duct PSC, must also be considered. Other non-immune-mediated conditions include gallstone disease, whose incidence is increased in IBD patients, non-alcoholic fatty liver disease (NAFLD), pyogenic liver abscess and portal vein thrombosis. Drug-induced liver diseases is another important chapter, since several drugs used in IBD, mainly thiopurines, methotrexate and anti-tumor necrosis factor-α (anti-TNF) may induce liver toxicity. Concomitant viral hepatitis B and C in IBD is also a relevant issue, particularly hepatitis B reactivation under immunosuppressive therapy; however, the recent introduction of potent antiviral drugs for both the infections and the spread of the anti-hepatitis B virus vaccine (HBV) contributed to significantly lower the risk. The diagnosis of such hepatobiliary conditions is of great importance, since they may influence the management and therapeutic approach to IBD, contraindicate the use of some therapies, or prevent the evolution towards the end stage of liver disease. The main hepatobiliary disorders, which are discussed in this review, are summarized in Table 1. A proposed practical approach to abnormal liver function tests (LFT) in a patient with IBD is presented in Figure 1.
| Hepatobiliary manifestation | Main features |
| Immune-mediated | |
| PSC | The most frequent (50%-80% of PSC patients have IBD, and 2%-8% of IBD patients have PSC) |
| No medical treatment approved. Therapies directed towards PSC complications | |
| Increased risk of cholangiocarcinoma and colorectal cancer (surveillance needed) | |
| Small duct PSC | Histological evidence of PSC, but normal cholangiogram |
| More benign disease course than classic PSC (cholangiocarcinoma risk not increased) | |
| PSC-AIH overlap syndrome | Coexistence of biochemical and histological features of AIH and PSC-associated biliary tract alterations |
| Better response to steroids and immunosuppressants than PSC | |
| IgG4-related sclerosing cholangitis | Part of the IgG4-related systemic disease |
| Characterized by histological evidence of IgG4+ plasma cells infiltrate | |
| Good response to steroids | |
| Granulomatous hepatitis | Rare, generally in Crohn’s disease |
| Autoimmune or drug-induced pathogenesis | |
| Good response to steroids | |
| Non-immune-mediated | |
| Gallstone disease | Incidence increased in IBD, more in Crohn’s disease |
| Bile salts malabsorption underlying the pathogenesis | |
| NAFLD | Not strictly associated with IBD; similar risk factors in the general population |
| Higher NAFLD prevalence in patients with severe IBD activity | |
| Pyogenic liver abscess | Rare, mainly in Crohn’s disease |
| Penetrating disease, steroid treatment and malnutrition are risk factors | |
| Portal vein thrombosis | Increased risk in IBD, especially during severe disease flare and after surgery. Prophylactic treatment indicated in these settings |
| DILI | |
| Aminosalicylates | Low risk of DILI |
| LFT monitoring not necessary | |
| Thiopurines | DILI quite frequent (prevalence of about 3%); both dose-independent and dose-dependent toxicities are possible |
| Regular LFT monitoring indicated | |
| Methotrexate | DILI quite frequent, with a prevalent dose-dependent mechanism |
| Regular LFT monitoring indicated | |
| Folic acid supplementation indicated during treatment | |
| Anti-tumour necrosis factor-α | Low risk of DILI, mainly with infliximab |
| LFT monitoring not necessary | |
| Anti-integrins | Low risk of DILI |
| LFT monitoring not necessary | |
| Anti-interleukin 12/23 | Low risk of DILI |
| LFT monitoring not necessary | |
| Tofacitinib | Data in IBD still scarce |
| Alanine aminotransferase elevation quite frequent in rheumatoid arthritis, but generally mild | |
| Hepatitis B reactivation | A relevant concern |
| Antiviral therapy indicated in HBsAg positive patients | |
| LFT monitoring indicated in HBsAg negative/anti-HBc positive patients | |
| Vaccination indicated in naïve patients | |
| Hepatitis C reactivation | Not a relevant concern |
PSC is the most common hepatobiliary manifestation associated with IBD. It is a rare, idiopathic, chronic cholestatic syndrome characterized by chronic inflammation, fibrosis and finally destruction of intra- and/or extra-hepatic bile ducts. PSC is a progressive disease, leading to liver biliary cirrhosis and portal hypertension.
Epidemiology: According to a recent systematic review, the incidence and prevalence rates of PSC range from 0 to 1.3 per 100.000 inhabitants/year and from 0 to 16.2 per 100.000 inhabitants, respectively. There is a 2:1 male predominance and a peak of incidence between 30 to 40 years old[1]. PSC is commonly associated with IBD, with about 50%-80% of patients with PSC having concomitant IBD, more frequently UC[2], and about 2%-8% of patients with IBD having PSC[3]. PSC diagnosis usually precedes that of IBD, although PSC may be diagnosed many years after proctocolectomy for colitis[4].
Etiology: The exact etiology of PSC is unknown. A multifactorial pathogenesis has been proposed, in which genetic, immunological, and environmental factors contribute to the development of the disease. The increased risk of PSC in first-degree relatives suggests a genetic predisposition. Multiple human leukocyte antigen (HLA) haplotypes related to PSC susceptibility have been reported: HLA-B8, HLA-DRB1*0301 (DR3), HLA-DRB3*0101 (DRw52a) and HLA-DRB1*0401 (DR4)[5]. Interestingly, three UC susceptibility loci, harboring the genes REL, IL2, and CARD9, have been linked to PSC, supporting the association UC-PSC as a separate disease entity. However, genetic factors are implicated in a minority of PSC cases, clearly emphasizing the predominant role of environmental risk factors in the overall disease liability[6,7]; colonic toxins, gut microbiota, portal bacteria and viral infections[6], are some of the main environmental determinants, which are discussed below. Based on the association between certain HLA haplotypes, the acute and chronic inflammatory infiltrate at histology, and given the association with several other autoimmune conditions, PSC has been classically considered an autoimmune disease[8]. Several autoantibodies may be present, including antinuclear antibodies in 24%-53%, smooth muscle antibodies in 13%-20%, and anti-perinuclear cytoplasmic antibodies (pANCA) in 65%-88% of patients[9]. However, none of these autoantibodies are reliable for diagnosis and there is no significant response of the disease to immunosuppressants. Chronic portal bacteremia is another important mechanism postulated: the bacterial translocation from the gut into the portal system can lead to biliary inflammation and recurrent cholangitis, probably through activation of the innate immune response in susceptible individuals[10]. Growing evidence suggests a relevant role of the gut microbiome in the pathogenesis of PSC, independently of IBD. Patients with PSC are characterized by a fecal overrepresentation of Escherichia, Lactobacillus, Fusobacterium, Enterococcus and Ruminococcus, and decreased populations of Clostridium cluster II, Prevotella and Bacteroides, compared to healthy individuals and patients with IBD alone[11-13]. Gut dysbiosis has been linked to an increase Gut dysbiosis has been linked to an increase in gut permeability and bacterial translocation that enter the enterohepatic circulation[14]. Other etiologic mechanisms such as ischemia and chronic viral infections have been postulated, but more evidence is needed.
Clinical presentation and diagnosis: Since most patients with PSC are asymptomatic at diagnosis, the disease is frequently suspected after routine liver biochemical tests. When the disease is symptomatic, the most common symptoms are pruritus, fatigue, right upper abdominal pain, and weight loss. Acute cholangitis is the first clinical manifestation of PSC in about 15% of cases[15]. Biochemical tests typically show a cholestatic pattern: An increased alkaline phosphatase (ALP) is the most frequent alteration, usually together with a raise of gamma-glutamyl transpeptidase. Notably, although an elevated ALP is a sensitive diagnostic marker, a normal level does not exclude PSC[6]. A high level of serum bilirubin is observed in an advanced stage of disease and is a marker of poor prognosis. Aminotransferases are often normal or mildly raised. As mentioned above, multiple autoantibodies, most frequently pANCA, have been associated with PSC, but they are not specific nor related to disease activity and prognosis[16]. Diagnosis is confirmed if the typical morphological alterations of biliary ducts are identified and causes of secondary sclerosing cholangitis are excluded. Magnetic resonance cholangiopancreatography (MRCP) should be the technique of choice for the investigation of suspected PSC, with a sensitivity and specificity for diagnosis of 0.86 and 0.94, respectively[17]. MRCP demonstrates diffuse, multifocal strictures and dilations of the intra- and extra-hepatic bile ducts. In about 40% of cases, the gallbladder and cystic duct are also involved[18]. Endoscopic retrograde cholangiopancreatography (ERCP) should be reserved for patients with biliary strictures requiring tissue acquisition (e.g. cytological brushing) or when therapeutic intervention is indicated (e.g. jaundice or acute cholangitis)[6]. In recent years, peroral cholangioscopy has emerged as a useful endoscopic tool in PSC management. It can provide a direct intraductal visualization, which allows guided biliary biopsies and can be helpful in distinguishing between benign and malignant strictures. A recent meta-analysis found a sensitivity and specificity of cholangioscopy-directed biopsies for all indications (i.e., not limited to PSC) of 71.9% and 99.1%, respectively[19,20]; however, data on patients with PSC are still limited. Moreover, cholangioscopy has been recently used in the treatment of biliary stones in patients with PSC, with promising results[19]. Liver biopsy is not required to establish a diagnosis of a “classic” form of PSC. However, it is essential in presence of abnormal liver tests and normal cholangiogram to investigate small duct PSC, or in PSC patients with disproportionately elevated serum aminotransferase values to exclude PSC–autoimmune hepatitis (AIH) overlap syndrome. The most specific histological finding of PSC is periductal fibrosis with an “onion skin” pattern. In clinical practice, however, histological assessment is often non-specific, demonstrating general features of cholestasis that are similar to those find in primary biliary cirrhosis. Liver biopsy can also play a role in staging the disease and in defining the prognosis[6].
Complications and prognosis: PSC is a progressive disease that leads to severe complications involving liver, biliary tree and intestine. Fibrotic obliteration of intra-hepatic bile ducts finally evolves into liver cirrhosis, hepatic failure and portal hypertension. Disease progression towards end-stage liver disease is unavoidable in most patients, and liver transplantation (LT) is considered the only curative treatment option[21]. In the literature, the median time from diagnosis to death or LT range from 7 to 22 years, with higher survival rates observed in overall PSC populations respected to cohorts of patients from liver transplant centers, which suffer from referral bias[22,23]. In IBD patients, performing colectomy before PSC diagnosis was associated with lower risk of LT and death in a large cohort study in Sweden[14]. Portal hypertension is a frequent complication of PSC, and the presence of esophageal varices at diagnosis or history of variceal hemorrhage are considered predictors of worse prognosis[24]. PSC patients are at increased risk of cholangiocarcinoma (CCA), gallbladder carcinoma, hepatocellular carcinoma (HCC), and colorectal carcinoma (CCR). The estimated annual incidence of CCA in patients with PSC range from 0.5% to 1.5%[25,26], with 20%-30% of CCA found synchronously at PSC diagnosis, and 50% of CCA occurring within 1 year[25]. According to a large international, multicentre, PSC cohort study (7121 patients from 37 countries), 10.9% of PSC patients developed a hepatopancreatobiliary malignancy, which was CCA in about 80% of cases[27]. Importantly, concomitant UC was a risk factor for future development of hepatopancreatobiliary malignancies[27]. Gallbladder cancer and HCC are less frequent complication of PSC, with a lifetime incidence of 3%-14% and 0.3%-2.8%, respectively[28]. An increased risk of CCR has been clearly demonstrated in patients with PSC-IBD, compared to patients with IBD or PSC alone. According to a recent meta-analysis of observational studies, patients with IBD and PSC were at increased risk of colorectal cancer compared with patients with IBD alone, with an odds ratio of 3.41 (95%CI: 2.13-5.48). Interestingly, stratification by IBD type revealed that PSC was a risk factor for colorectal cancer in patients with UC, but not in CD patients[29]. In addition, unlike in patients with UC alone, CCR risk in PSC-UC seems to manifest soon after the combined diagnosis, with a peak of incidence within the first 2 years of diagnosis[30]; thus, cancer surveillance is strongly recommended in PSC-UC, even in patients with ileal pouch-anal anastomosis (IPAA) after colectomy[31]. Finally, IBD patients with IPAA and concomitant PSC are at increased risk of pouchitis, with an almost double incidence at 10 years as compared to patients without PSC[32].
Treatment: Treatment of PSC associated with IBD does not differ from PSC without IBD. To date, no medical treatments have been demonstrated to modify the course of “classic” PSC. In particular, ursodeoxycholic acid (UDCA) has shown to improve LFT in several studies, but two meta-analyses and a large multicentre study failed to show benefit from UDCA towards important clinical outcomes (e.g. complications and death) in patients with PSC[33,34]. Despite previous studies suggested a role of UDCA in prevention of cancer (CCR or CCA) in PSC, more recent meta-analyses and a randomized control trial did not confirm this effect[35,36]. UDCA is not currently recommended by PSC guidelines for either the treatment or cancer prevention[6,37]. Despite the presumed immune-mediated pathogenesis of the disease, corticosteroids and immunosuppressants are not recommended as well[6]. Thus, treatments goals in PSC are directed to the control of symptoms and management of complications, such as varices, liver decompensation, cholangitis, jaundice, pruritus, and malignancies. Endoscopic interventions, mainly ERCP, are a mainstay of PSC management, and specific guidelines have been published from collaboration of European Society of Gastrointestinal Endoscopy and European Association for the Study of the Liver (EASL)[38]. Main indications of ERCP in PSC are acute cholangitis, treatment of dominant strictures and suspicion of CCA. LT is a potential resolutive therapy in PSC patients with end-stage liver disease. Other disease-specific indications are intractable pruritus, recurrent cholangitis, and limited cases of very early stage of CCA[3]. A single-center experience from the Mayo Clinic reported survival rates after LT for PSC-related end-stage liver disease of 86% at 5 years and 70% at 10 years[39]. Recurrence of PSC after LT is a concern, occurring in 12%-37% of cases and causing significant impact on long term graft and recipient survival[40].
Small duct PSC: A minority of patients with cholestatic biochemistry and typical liver histology with concentric ‘onion skin’ fibrosis around the bile ducts, but with entirely normal cholangiogram, was first described by Wee and Ludwig[41] in 1985; they coined the term “small duct PSC”. In a large multicentre study, 81% of patients with small-duct PSC had IBD, predominantly UC (78%) compared to CD (21%). In this study, none of the patients developed CCA or other intestinal malignancies during a median follow-up of 13 years, but 28% of them shown evidence of progression to large duct PSC at repeated cholangiography[42]. In a large bicentric study from United Kingdom and Norway, only 12% of small duct PSC patients either required LT or died, compared to 47% of patients with “classic” PSC[43].
Overlap between PSC and AIH: PSC/AIH overlap syndrome is a rare disorder characterized by concomitant occurrence of the biochemical and histological features of AIH and the cholangiography abnormalities found in PSC. In a cohort of 211 PSC patients from United States, according to the International AIH group scoring system, AIH was diagnosed as “definite” in 1.4% and “probable” in 6% of patients[44]. An Italian cohort of PSC/AIH patients showed a lower mean age at presentation and higher alanine aminotransferase (ALT) and aspartate aminotransferase (AST) values compared to “classic” PSC[45]. There is also a strong association between PSC/AIH and IBD; according to a recent systematic review, IBD was present in 44% of PSC/AIH patients, that was UC in 68% of cases[46]. Patients with an established diagnosis of AIH who also have IBD should be evaluated for concomitant PSC. Patients with PSC/AIH seem to benefit from treatment with immunosuppressive medications and have a better prognosis compared to patients with PSC alone[45].
IgG4-related sclerosing cholangitis: IgG4-related sclerosing cholangitis (IgG4-SC) is the biliary manifestation of the multi-organ inflammatory IgG4-related disease. Diagnosis requires histological evidence of IgG4+ plasma cells infiltrate (> 10 per high-power field), imaging of biliary tract involvement (which may be indistinguishable from the “classic” PSC), elevated serum IgG4 levels (> 135 mg/dL), evidence of other organ involvement and response to steroid treatment[47]. Autoimmune pancreatitis is the most frequent organ involvement associated with IgG4-SC, being present in > 90% of cases[48]. An increase in serum IgG4 is reported in 9%-22% of patients with PSC overall[48], making it difficult to distinguish a PSC with high serum IgG4 levels from a “true” IgG4-SC. EASL Cholestatic Liver Disease Guidelines recommends measurement of serum IgG4 in all patients with large-duct PSC at diagnosis[37]. While association with IBD is prevalent in PSC, this is rarely seen in IgG4-SC; high serum IgG4 levels have been observed in about 5% of IBD patients[49]. Unlike in PSC, response to steroid treatment in IgG4-SC is excellent. However, relapse after steroid withdrawal is common[50]; in these cases, second-line treatments include immuno
AIH is an immune-mediated chronic liver disease characterized by hepatocellular inflammation, necrosis and progression to cirrhosis. The clinical presentation varies from persistent mild elevation of AST and ALT to fulminant forms of acute hepatitis. Mean age at presentation shows a bimodal pattern with one peak during childhood/teenage years and another between the 4th and 6th decade of life. The diagnosis of AIH must be suspected in presence of autoantibodies (mainly antinuclear, smooth muscle, soluble liver antigen/liver pancreas and liver/kidney microsomal type 1 antibodies), IgG elevation, consistent liver histology and exclusion of other forms of hepatitis[52]. Despite most of the data about AIH/IBD coexistence comes from studies focusing on PSC and AIH/PSC overlap syndrome, a higher prevalence of AIH has been found in patients with IBD, compared to subjects without IBD. In the cross-sectional study by Halling et al[53], AIH was more frequent in males and females with IBD compared with matched controls without IBD, with an odds ratio of 7.8 and 17.9, respectively[53]. Another study by Perdigoto et al[54] found a 16% prevalence of UC in patients with AIH, 42% of whom had also PSC features at cholangiography[54]. In this study, patients with colitis failed treatment for AIH more commonly and progressed to cirrhosis more frequently; similar results emerged from the study by Perdigoto et al[54].
Granulomatous hepatitis is a rare complication of IBD, with only a few cases of IBD-associated granulomatous hepatitis reported in literature[55-57]. It occurs more frequently in CD and can underlie an autoimmune pathogenesis or be induced by mesalamine or sulfasalazine therapy[58]. Clinical manifestations include fever, hepatomegaly and increase in cholestatic enzymes, although patients can be completely asymptomatic[59]. Response to corticosteroid therapy is generally good; methotrexate may be considered as second-line therapy in patients relapsing after steroids[60]. Prognosis is usually benign[61].
Several studies and a meta-analysis showed a prevalence of cholelithiasis in CD ranging from 8% to 34%, with a 2- to 5-fold increased risk compared to the general population[62-68]. Three studies also evaluated UC patients, reporting a prevalence of gallstone disease of 4%-10%; only one of these found a significantly higher risk compared to a population without UC[62], while the other two studies, including the aforementioned meta-analysis, did not demonstrate this increased risk[64-66]. Most studies relied on abdominal ultrasound to diagnose the lithiasis. A recent case-cohort study on a large cohort of IBD patients reported an incidence of cholelithiasis of 5.21/1000 persons/year, compared to a 3.49/1000 persons/year incidence of a matched non-IBD cohort (P < 0.001); the significance was also maintained by differentiating CD and UC[69]. Another case-control study reported an incidence of gallstone disease in CD and UC of 14.35/1000 persons/year and 7.48/1000 persons/year, respectively, that were significantly higher than those of the matched control populations[70]. In all studies assessing both CD and UC, prevalence of gallstone disease was higher in CD compared to UC. Among the risk factors, ileal disease location, previous ileal resection and long-standing disease were the most frequently associated with gallstone disease in IBD[62-64,67,68,70]. The pathogenesis of cholelithiasis in IBD patients is usually attributed to bile salts malabsorption at the terminal ileum; this leads to a decrease in the total bile acid pool, leading to supersaturated bile in gallbladder, which predispose to stone formation[71,72]. Lapidus and Einarsson[71] reported that patients with ileal resection due to CD are characterized by lower cholesterol saturation, but increased bilirubin concentration in fasting duodenal bile, compared to healthy controls; therefore, these patients seem not predisposed to the formation of cholesterol stones, but rather at risk of developing pigment stones[71].
NAFLD refers to a clinical and pathological syndrome that includes a spectrum of histological findings ranging from benign steatosis to non-alcoholic steatohepatitis. Non-alcoholic steatohepatitis is defined by histological evidence of hepatic steatosis associated with inflammation, and can progress to hepatic fibrosis and cirrhosis. A recent meta-analysis reported a worldwide prevalence of NAFLD of 25% in the general population[73], a prevalence that seems to be worryingly increasing over time[74]. In the literature, the prevalence of NAFLD in patients with IBD is variable. Two recent meta-analyses reported a pooled prevalence of NAFLD in IBD of 27.5%[75] and 32%[76]; older age, obesity, type 2 diabetes, longer IBD duration and previous surgery were the main risk factors associated with the development of NAFLD[77]. A further meta-analysis specifically addressing the role of IBD treatment on the risk of NAFLD found no significant association between medications of all types (i.e., steroids, biological agents, immunomodulators, methotrexate) and the risk of developing NAFLD[78]. Several studies also reported a higher prevalence of NAFLD among IBD patients with severe disease activity at the time of liver evaluation, compared to mild-moderate IBD cases[77,79,80].
Pyogenic liver abscesses are rarely seen in IBD, with only a few cases reported in literature, mainly in CD. A nationwide case-cohort study from Taiwan reported an incidence of pyogenic liver abscess in IBD patients of 6.7 cases/10000 persons/year, which was significantly higher compared to controls without IBD[81]. Clinical manifestations include fever, chills, anorexia, weight loss and abdominal pain with right upper quadrant tenderness, which can mimic an IBD flare and lead to a diagnostic delay. Moreover, hepatic abscesses have been reported as the initial presentation of CD in several cases[82,83]. Risk factors predisposing to liver abscesses in IBD include abdominal surgery, fistulizing disease, intra-abdominal abscess, malnutrition, and corticosteroid treatment[84]. Dissemination from intra-abdominal abscesses and portal bacteremia secondary to impaired intestinal permeability are the most involved pathogenic mechanism[84].
IBD patients are at increased risk of venous thromboembolism (VTE)[85]. In two studies on large cohorts of IBD patients with a follow-up time over 10 years, thromboembolic complications were reported in about 1% of patients, with an incidence of VTE of 2.6/1000 persons/year[85,86]. Porto-mesenteric venous system is a frequent site of thrombosis in IBD and is a potentially catastrophic complication, which may lead to bowel ischemia or infarction and to acute or chronic portal hypertension; the mortality rate range between 3%-25%[86,87]. Incidence is higher during disease flares and after surgical procedures[88-90], and prophylactic treatment with low-molecular-weight heparin in severely active disease is indicated by guidelines to reduce the risk of thromboembolism[91]. However, about 30%-50% of thrombosis occurs in remission phases of the disease[92-94], indicating that factors other than inflammatory status can be involved in the pathogenesis of the thrombotic event. Immobilization, extensive colonic disease, central catheters, corticosteroids, and smoking are other known prothrombotic risk factors[90,95]. A hematologic prothrombotic condition can be found in up to 40% of portal vein thrombotic events in IBD, hyperhomocysteinemia being the most frequently found[95]. Thrombocytosis is frequently seen during IBD flares and may result from systemic inflammatory activity and/or iron-deficiency anemia[96]; however, no data on a possible association between thrombocytosis and VTE in IBD is available to date, since large clinical studies addressing this association are still lacking[97]. Moreover, IBD are associated with significant changes in circulating levels of various coagulation factors, as result of an imbalance between procoagulant and anti-coagulant pathways. Specifically, higher levels of prothrombin fragment 1 and 2, fibrinogen, factors V and VIII, thrombin-antithrombin complex, plasmin-α2-antiplasmin complex, and an impairment of the protein C pathway have been described in IBD[97-99]. Specific mutations in clotting factors, e.g. Factor V Leiden, are rare, but important to be identified as they may indicate long-term anticoagulant treatment[100]. European Crohn’s and Colitis Organization (ECCO) guidelines recommend appropriate screening for prothrombotic condition after IBD diagnosis and anticoagulant treatment in accordance with international guidelines[95].
The therapeutic armamentarium for the treatment of IBD is gradually expanding. This certainly offers greater potential for therapeutic benefit, but the risk of hepatotoxicity is a concern. Although the overall risk of serious adverse events is low, cases of drug-induced liver injury (DILI) have been reported for most drugs used in IBD, and some therapies carry a significant risk of liver toxicity. DILI induced by IBD drugs can be allergic/idiosyncratic (dose-independent) or related to hepatotoxins (typically dose-dependent). In addition, some drugs can cause hepatotoxicity with more than one pathogenic mechanism. According to EASL guidelines, the exclusion of other causes of hepatotoxicity is necessary for the diagnosis of DILI, and recovery after drug discontinuation is an important criterion for the causality assessment[101] (Figure 1). The following paragraphs will describe the association between the main drugs used in IBD and the risk and type of DILI.
Sulfasalazine was the first aminosalicylate approved for the induction and maintenance of remission in mild-to-moderate UC. Within the bowel, sulfasalazine is cleaved into sulfapyridine and 5-aminosalicylic acid, most called mesalamine. Sulfapyridine, a sulfa-containing antibacterial agent, is then absorbed from the colon into the bloodstream, transported to the liver, and acetylated; acetylation was reported to be genetically programmed, with slow acetylators having higher levels of free sulfasalazine and more drug-induced adverse events[102]. Mesalamine is minimally absorbed and largely excreted in the stools and is primarily responsible for the anti-inflammatory effect on the colon. The introduction of the various mesalamine formulations has almost completely supplanted the use of sulfasalazine in UC, while the utility of aminosalicylates in CD remains unclear[91,103]. Both sulfapyridine and mesalamine are rarely associated with liver injury. According to the United Kingdom’s Committee on the Safety of Medicines, from 1991 to 1998 the incidence of hepatitis in patients treated with mesalamine was 3.1 cases per million, compared to 6 cases per million in patients treated with sulfasalazine[104]. A French pharmacovigilance study on mesalamine microgranules (Pentasa®) reported 0.79 cases of LFT elevations per million treatment days over a 2-year period[105]. The toxic effect almost always occurs within the first 2 mo of treatment, and LFT normalize in most cases after drug discontinuation[105]. For sulfasalazine, sporadic cases of granulomatous hepatitis or fulminant hepatitis have been reported[106-108]. Due to this low risk of hepatotoxicity, a close monitoring of liver chemistries is not necessary in patients treated with aminosalicylates.
Azathioprine (AZA) and its metabolite 6-mercaptopurine (6-MP) are two thiopurine analogues widely used for the treatment of IBD. Main indication of AZA and 6-MP is the maintenance of remission in steroid-dependent CD and UC[91,103]. Purine analogues act as DNA synthesis inhibitors by antagonizing endogenous purines, and lead to both cytotoxic and immunosuppressive effects[109]. Overall, adverse events due to thiopurines are frequent and occur in 15%-40% of patients, leading to dose reduction or drug withdrawal[110]. Thiopurine-related adverse events are classified into dose-independent (or allergic/idiosyncratic) and dose-dependent. The former are thought to be immune-mediated and include rash, fever, arthralgia, and pancreatitis; the latter include myelotoxicity as the main manifestation. Thiopurine-induced hepatotoxicity can be both dose-dependent and independent, based on the pathogenetic mechanism involved[111,112]. Dose-independent liver toxicity usually occurs within 3 mo of therapy and includes hypersensitivity and idiosyncratic reactions[111]; type of hepatotoxicity can be described as acute hepatocellular hepatitis, with prevalent increase of aminotransferase levels, acute cholestatic hepatitis, with prevalent increase of serum ALP, or mixed[113,114]. Other less frequent findings include peliosis hepatis, hepatic sinusoidal dilatation, veno-occlusive disease, perisinusoidal and portal fibrosis, and nodular regenerative hyperplasia[113]. Thiopurine-related DILI has been related to thiopurine metabolites. After absorption, AZA is metabolized in the liver to 6-MP, which undergo a complex metabolization by three enzymes; one of them is the thiopurine S-methyltransferase (TPMT), that lead to 6-methylmercaptopurine (6-MMP) formation. 6-MMP is a non-effective metabolite which is important in hepatotoxicity development[109]. Approximately 15%–20% of IBD patients treated with thiopurines demonstrate hypermethylation (or shunting), a phenomenon due to a high TPMT activity that leads to preferential methylation of 6-MP to 6-MMP over bioactivation to thioguanine nucleotides (TGNs); the usual definition of hypermethylation is a ratio of 6-MMP to TGNs of > 11. Subtherapeutic TGNs level results in a poor response to therapy, while a high 6-MMP level (> 5700 pmol/8 × 108 erythrocytes) has been correlated with a 3-fold increased risk of liver toxicity[115]. Allopurinol is a xanthine oxidase inhibitor that prevents the breakdown of thiopurines into thiouric acid (TUA), thus increasing the bioavailability of 6-MP. Several studies have demonstrated that the combination of low dose thiopurine, i.e. 25%–50% of the standard dose, with 100 mg of allopurinol corrects hypermethylation in patients who have experienced thiopurines-induced hepatotoxicity or who have had a poor response to thiopurines treatment[116,117]. However, Shaye et al[118] showed that about 90% of patients with 6-MMP > 5700 pmol/8 × 108 erythrocytes have no hepatotoxicity and almost 40% of subjects with hepatotoxicity had 6-MMP levels below this cut-off[118]. Moreover, a recent case-control study and a meta-analysis failed to demonstrate any correlations between TPMT gene polymorphisms and hepatic adverse events in IBD patients[119,120]. The reported frequency of thiopurine-related hepatotoxicity varies widely among studies, ranging from to 3% to 17%[108,115,121,122]; a systematic review by Gisbert et al[113] reported a mean prevalence of thiopurine-induced liver injury of 3%, with a mean annual rate of 1.4%[113]. In a prospective cohort study, abnormal liver function (defined by ALT or ALP levels > 50% the upper normal limit) occurred in 13% of patients, while hepatotoxicity (defined by ALT or ALP levels greater than twice the upper normal limit) developed in 10%[111]. CD, liver steatosis and concomitant steroid therapy are reported risk factors for liver injury during thiopurine therapy[108,111,123]. It has been shown that most cases of thiopurine-induced liver injury completely resolved after dose reduction, while the need to discontinue therapy only occurred in about 3%-4% of cases[111,118,124]. Switching from AZA to 6-MP in the case of AZA-induced DILI is a possible strategy, which is effective in resolving the liver toxicity in 71%-87% of cases[114,125]. Despite an optimal frequency has not yet been established, regular monitoring of blood tests should be performed for the entire duration of thiopurine treatment, more frequently in the first 3 mo of therapy[113,126,127]. British Society of Gastroenterology (BSG) guidelines on IBD recommend the monitoring of full blood count and LFT at 2, 4, 8 and 12 wk of thiopurine therapy, and every 12 wk thereafter[127].
Methotrexate (MTX) is a folic acid analogue with inhibitory activity against many enzymes in the metabolic pathway of folic acid. MTX inhibits production of thymidylate, purines, and methionine and leads to accumulation of adenosine, which has a potent anti-inflammatory activity. These actions inhibit cellular proliferation and tissue migration, and decrease production of inflammatory mediators[128]. MTX is currently indicated for the maintenance of remission in steroid-dependent CD[129], while its role in UC is still controversial[130]. The hepatotoxic potential of MTX is well known. A meta-analysis of clinical trials on IBD patients treated with MTX reported a pooled incidence rate of abnormal hepatic aminotransferase levels, which the author defined as up to a 2-fold increase over the upper limit of the normal, of 1.4 per 100 person-months. The rate of hepatotoxicity, defined as aminotransferase levels greater than a 2-fold over the upper normal limit, was 0.9 per 100 person-months. The rate of withdrawal of MTX due to these abnormalities was 0.8 per 100 person-months[112,131]. Alcohol intake is a main risk factor for MTX-induced hepatotoxicity and should be strictly avoided. Other potential risk factors are obesity, diabetes mellitus and chronic viral hepatitis[112,131,132]. Folic acid supplementation has been correlated with reduction of methotrexate-induced hepatic adverse events and is therefore recommended[133]. Regular liver chemistry tests are recommended for the monitoring of hepatotoxicity, every 2 wk for the first 2 mo and then every 2-3 mo[134]; the drug should be stopped if transaminases exceed twice the upper normal limit[127]. Although liver biopsy was previously indicated after an MTX-treatment cumulative dose ≥ 1.5 g, this practice is no longer recommended by current rheumatologic guidelines[134], this is based on recent evidence that show a low incidence of liver injury in patients receiving a chronic low dose of MTX[112]. In a retrospective study on 87 IBD patients with a mean MTX cumulative dose of 1813 mg, 76% of patients maintained normal liver chemistry tests throughout MTX therapy; a liver biopsy was performed in 11 patients after a cumulative dose ≥ 1.5 g and found no case of moderate or severe fibrosis[112]. Another study evaluating 20 liver biopsies after a cumulative methotrexate dose of ≥ 1.5 g (mean dose 2.6 g) found mild histological abnormalities in 95% of patients; abnormal liver chemistry tests were present in 30% of patients and did not correlate with histological toxicity[135]. However, liver biopsy should be performed in cases of persistent alteration of transaminases, especially in case of no reduction after lowering the drug dose. Transient elastography is a promising tool for the monitoring of liver fibrosis in MTX-treated patients and can be useful in selecting patients for liver biopsy[136].
Anti-TNF-α: Since its introduction in the 1990s, anti-TNF-α antibody therapy has revolutionized the treatment of IBD. Anti-TNFs, which include infliximab, adalimumab, golimumab and certolizumab pegol, are approved for the treatment of moderate-to-severe CD and UC and demonstrated high efficacy in the induction and maintenance of both clinical and endoscopic remission[91,103]. Several types of anti-TNF-related adverse events have been reported, mostly of infectious, auto-immune and tumoral types. DILI caused by anti-TNF is uncommon, mostly mild and related to infliximab. However, cases of liver failure requiring transplantation has rarely been reported[137-139]. Shelton et al[140] evaluated the incidence of liver enzyme elevation in a large cohort of IBD patients treated with anti-TNF: Only 102 out of 1753 patients (6%) developed ALT elevation, and in about half of cases this could clearly be linked to an alternative etiology. Infliximab was the involved anti-TNF in 96% cases. Compared to a control population of anti-TNF-treated patients without liver enzyme elevation, no difference in concomitant immunomodulator therapy, body max index, age and gender was found. The majority of patients with ALT elevation continued anti-TNF, most of them normalizing the liver enzyme during the follow-up. In 10 patients switching to a second anti-TNF was performed, without recurrence of liver injury[140]. Ghabril et al[141] identified 34 cases of DILI related to anti-TNF used for a variety of auto-immune conditions from a review of the United States DILI Network database and PubMed research. The drug presumed to have caused DILI was infliximab in 76% of cases. The liver injury was scored as mild-to-moderate in 93% of cases. Fifteen of the 17 patients undergoing liver biopsy showed clear features of autoimmunity. All patients improved after discontinuation of the anti-TNF[141]. The mechanism underlying liver toxicity remains to be elucidated. Infliximab-related hepatitis seems to be sustained by an immune-mediated mechanism, mimicking the characteristics of AIH type I, although a direct liver damage cannot be ruled out[112]. Currently, Food and Drug Administration classifies infliximab as a Most-DILI-concern drug, adalimumab as a Less-DILI-concern drug, and golimumab and certolizumab as Ambiguous-DILI-concern drug[142]. The current consensus recommends the use of infliximab in selected cases of patients with significant liver disease, and that treatment should be discontinued or avoided in patients with transaminases above three times the upper limit of normal[143].
Anti-integrins: Natalizumab is a monoclonal antibody that antagonizes both the integrins α4β1 and α4β7, which are necessary for the homing of lymphocytes to brain and gut, respectively. Natalizumab is therefore approved for the treatment of multiple sclerosis, and has been tested with good results in CD; however, the risk of JC virus-associated progressive multifocal leukoencephalopathy (PML) has limited its use in IBD[144-146]. Vedolizumab is a humanized monoclonal antibody which, unlike natalizumab, specifically inhibits α4β7 integrin, thus eliminating the risk of PML[147]. Vedolizumab is approved for the treatment of both moderate to severely active CD and UC since 2014[91,103]. To date, only sporadic cases of liver injury during vedoli
Anti-interleukin 12/23: Ustekinumab is a human monoclonal antibody directed against the p40 subunit, which is a component of both interleukin (IL)-12 and IL-23, allowing this drug to simultaneously inhibit both these cytokines. Ustekinumab has been recently approved as a second line therapy for moderate-to-severe CD and UC since 2016 and 2019, respectively[153,154]. Although current data are limited, liver injury related to ustekinumab seems to be very uncommon. In the phase III trial on CD, a similar rate of adverse events compared to placebo was reported, with no mention of hepatotoxicity[153]. According to the Clinical and Research Information on DILI database, mild-to-moderate serum aminotransferase elevation was reported in 0.5% to 1.4% of patients during ustekinumab therapy. However, this event was no more frequent than placebo and resolved without discontinuing the drug[155]. Risankizumab is a monoclonal antibody directed against p19 subunit of IL-23 and therefore selectively inhibit this cytokine. Phase II and III trials in IBD are ongoing and safety data are still limited[156].
Tofacitinib is an oral Janus kinase inhibitor and is the first drug of this class approved for the treatment of IBD, specifically UC since May 2018[157], while others are currently being tested in phase II and III trials[158]. Tofacitinib is indicated for the treatment of adult patients with moderately to severely active UC, who have had an inadequate response or who are intolerant to anti-TNF[127,158]. Data about hepatotoxic effects of tofacitinib mainly derive from rheumatoid arthritis, where a slight ALT elevation was reported in about 30% of patients, but elevation above 3 times the upper normal limit occurred in 1%-2% of patients[159-161]. Data regarding tofacitinib-induced liver toxicity in IBD are still limited. However, no increased incidence of liver injury has been reported either in the pivotal trial or in subsequent real-life studies[162-165].
In literature, the reported prevalence of hepatitis B surface antigen (HBsAg) and anti-HBc positivity in IBD patients ranges from 0.6%-5.7% and from 1.6%-41.6%, respectively, depending on the geographic area considered[166]. Despite previous studies reported a higher prevalence of HBV positivity in IBD patients compared to the general population, more recent studies indicated an equal or lower prevalence which tends to decrease over time, suggesting that preventive measures like vaccination, use of disposable materials and implementation of transfusion safety programs are effective[166,167]. The risk of viral reactivation is a major concern in HBV patients treated with immunosuppressants. This event is closely related both to the stage of the infection and the type of immunosuppressive drug used. HBV reactivation, defined as the increase in HBV viremia of more than 1 Log10 IU/mL, is characterized by a broad spectrum of clinical manifestations, that range from viremia without clinically relevant manifestations to fulminant life-threatening hepatitis[168]. For this reason, both ECCO and BSG guidelines recommend hepatitis B screening immediately after diagnosis of IBD, checking for HBsAg, anti-HBs, and anti-HBc[127,169]. If screening was not performed at the time of diagnosis, it should be performed before immunosuppressive therapy initiation[127,169]. HBsAg-positive/anti-HBc–positive patients carry the higher risk of reactivation, and should receive potent anti-viral agents (nucleoside/nucleotide analogues with high barrier to resistance) such as tenofovir and entecavir. Prophylactic treatment should be started 3-4 wk before immunosuppressive therapy and continued until at least 12 mo after the end of treatment[169]. HBsAg-negative/anti-HBc–positive patients are considered to have occult infection and viral reactivation is rare in this group with types of immunosuppressants used in IBD; in this case, HBV viremia (HBV-DNA) should be checked every 2-3 mo during the treatment and antiviral treatment started when HBV-DNA is detected[169]. Hepatitis B vaccination in all seronegative patients at IBD diagnosis is recommended by ECCO guidelines[169], while BSG guidelines indicate vaccination in high-risk groups[127]. Anti-HBs level should be measured after vaccination to confirm response; however, a reduction in vaccination during immunosuppressive therapy (mainly immunomodulators and anti-TNF) has emerged from several studies[170] and a recent meta-analysis[171]. Indications for the management of the IBD patient undergoing immunosuppressive therapy according to HBV status are summarized in Table 2.
| Hepatitis B status | Indications |
| HBsAg positive/anti-HBc positive (chronic hepatitis B) | Antiviral treatment (start 3-4 wk before and continue at least 12 mo after the immunosuppressive treatment) |
| HBsAg negative/anti-HBc positive (occult hepatitis B) | Liver function tests monitoring every 2-3 mo |
| HBsAg negative/anti-HBc negative/anti-HBs negative (naïve for hepatitis B) | Vaccination (indicated at diagnosis) |
| HBsAg negative/anti-HBc negative/anti-HBs positive | Check previous hepatitis B vaccination. Dose hepatitis B virus-DNA if uncertainty |
Hepatitis C prevalence in IBD is similar to the general population[168]. The risk of HCV reactivation under immunosuppressive therapies used in IBD is low[172,173]. Small case series reported successful treatment of hepatitis C with direct-acting antiviral (DAA) in patients on anti-TNF therapy[170] and no drug-drug interaction between DAA and anti-TNF has emerged[174]; thus, concomitant treatment with DAA and anti-TNF seems to be safe, although more studies specifically addressing this setting are needed.
Hepatobiliary disorders are frequently seen in IBD, and PSC represents the most common of them. A broad spectrum of pathogenic mechanisms may underlie the disorders, ranging from autoimmune conditions, metabolic diseases, infections up to drug toxicity, and two or more diseases can co-exist in the same patient. Moreover, liver disease severity can range from mild, which only requires monitoring over time, to liver failure, that may require LT. A step-by-step approach to the IBD patient with abnormal LFTs is extremely important to make the correct diagnosis, prevent complications, and identify those cases that warrant early and aggressive treatment. Finally, the diagnostic complexity often requires a multidisciplinary management involving gastroenterologist and hepatologist.
Provenance and peer review: Invited article; Externally peer reviewed.
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Kitamura K, Knudsen T S-Editor: Fan JR L-Editor: A P-Editor: Fan JR
| 1. | Moon AM, Singal AG, Tapper EB. Contemporary Epidemiology of Chronic Liver Disease and Cirrhosis. Clin Gastroenterol Hepatol. 2020;18:2650-2666. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 805] [Cited by in RCA: 719] [Article Influence: 143.8] [Reference Citation Analysis (0)] |
| 2. | de Vries AB, Janse M, Blokzijl H, Weersma RK. Distinctive inflammatory bowel disease phenotype in primary sclerosing cholangitis. World J Gastroenterol. 2015;21:1956-1971. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 120] [Cited by in RCA: 146] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 3. | Chapman R, Fevery J, Kalloo A, Nagorney DM, Boberg KM, Shneider B, Gores GJ; American Association for the Study of Liver Diseases. Diagnosis and management of primary sclerosing cholangitis. Hepatology. 2010;51:660-678. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 888] [Cited by in RCA: 835] [Article Influence: 55.7] [Reference Citation Analysis (0)] |
| 4. | Schaeffer DF, Win LL, Hafezi-Bakhtiari S, Cino M, Hirschfield GM, El-Zimaity H. The phenotypic expression of inflammatory bowel disease in patients with primary sclerosing cholangitis differs in the distribution of colitis. Dig Dis Sci. 2013;58:2608-2614. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 27] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 5. | Karlsen TH, Schrumpf E, Boberg KM. Genetic epidemiology of primary sclerosing cholangitis. World J Gastroenterol. 2007;13:5421-5431. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 60] [Cited by in RCA: 57] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 6. | Chapman MH, Thorburn D, Hirschfield GM, Webster GGJ, Rushbrook SM, Alexander G, Collier J, Dyson JK, Jones DE, Patanwala I, Thain C, Walmsley M, Pereira SP. British Society of Gastroenterology and UK-PSC guidelines for the diagnosis and management of primary sclerosing cholangitis. Gut. 2019;68:1356-1378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 116] [Cited by in RCA: 183] [Article Influence: 30.5] [Reference Citation Analysis (1)] |
| 7. | Jiang X, Karlsen TH. Genetics of primary sclerosing cholangitis and pathophysiological implications. Nat Rev Gastroenterol Hepatol. 2017;14:279-295. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 70] [Cited by in RCA: 103] [Article Influence: 12.9] [Reference Citation Analysis (1)] |
| 8. | Farh KK, Marson A, Zhu J, Kleinewietfeld M, Housley WJ, Beik S, Shoresh N, Whitton H, Ryan RJ, Shishkin AA, Hatan M, Carrasco-Alfonso MJ, Mayer D, Luckey CJ, Patsopoulos NA, De Jager PL, Kuchroo VK, Epstein CB, Daly MJ, Hafler DA, Bernstein BE. Genetic and epigenetic fine mapping of causal autoimmune disease variants. Nature. 2015;518:337-343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1618] [Cited by in RCA: 1376] [Article Influence: 137.6] [Reference Citation Analysis (0)] |
| 9. | Terjung B, Spengler U. Role of auto-antibodies for the diagnosis of chronic cholestatic liver diseases. Clin Rev Allergy Immunol. 2005;28:115-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 23] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 10. | Tabibian JH, Varghese C, LaRusso NF, O'Hara SP. The enteric microbiome in hepatobiliary health and disease. Liver Int. 2016;36:480-487. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 11. | Bajer L, Kverka M, Kostovcik M, Macinga P, Dvorak J, Stehlikova Z, Brezina J, Wohl P, Spicak J, Drastich P. Distinct gut microbiota profiles in patients with primary sclerosing cholangitis and ulcerative colitis. World J Gastroenterol. 2017;23:4548-4558. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 215] [Cited by in RCA: 262] [Article Influence: 32.8] [Reference Citation Analysis (4)] |
| 12. | Milosevic I, Russo E, Vujovic A, Barac A, Stevanovic O, Gitto S, Amedei A. Microbiota and viral hepatitis: State of the art of a complex matter. World J Gastroenterol. 2021;27:5488-5501. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 12] [Cited by in RCA: 11] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 13. | Little R, Wine E, Kamath BM, Griffiths AM, Ricciuto A. Gut microbiome in primary sclerosing cholangitis: A review. World J Gastroenterol. 2020;26:2768-2780. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 82] [Article Influence: 16.4] [Reference Citation Analysis (3)] |
| 14. | Mertz A, Nguyen NA, Katsanos KH, Kwok RM. Primary sclerosing cholangitis and inflammatory bowel disease comorbidity: an update of the evidence. Ann Gastroenterol. 2019;32:124-133. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 43] [Article Influence: 7.2] [Reference Citation Analysis (0)] |
| 15. | Prokopič M, Beuers U. Management of primary sclerosing cholangitis and its complications: an algorithmic approach. Hepatol Int. 2021;15:6-20. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 29] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 16. | Hov JR, Boberg KM, Karlsen TH. Autoantibodies in primary sclerosing cholangitis. World J Gastroenterol. 2008;14:3781-3791. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 109] [Cited by in RCA: 117] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 17. | Dave M, Elmunzer BJ, Dwamena BA, Higgins PD. Primary sclerosing cholangitis: meta-analysis of diagnostic performance of MR cholangiopancreatography. Radiology. 2010;256:387-396. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 158] [Cited by in RCA: 156] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 18. | Vázquez-Elizondo G, Muciño-Bermejo J, Méndez-Sánchez N. Gallbladder disease in patients with primary sclerosing cholangitis. Ann Hepatol. 2008;7:182-183. [PubMed] |
| 19. | Fung BM, Fejleh MP, Tejaswi S, Tabibian JH. Cholangioscopy and its Role in Primary Sclerosing Cholangitis. Eur Med J Hepatol. 2020;8:42-53. [PubMed] |
| 20. | Badshah MB, Vanar V, Kandula M, Kalva N, Badshah MB, Revenur V, Bechtold ML, Forcione DG, Donthireddy K, Puli SR. Peroral cholangioscopy with cholangioscopy-directed biopsies in the diagnosis of biliary malignancies: a systemic review and meta-analysis. Eur J Gastroenterol Hepatol. 2019;31:935-940. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 21. | Aadland E, Schrumpf E, Fausa O, Elgjo K, Heilo A, Aakhus T, Gjone E. Primary sclerosing cholangitis: a long-term follow-up study. Scand J Gastroenterol. 1987;22:655-664. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 214] [Cited by in RCA: 181] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 22. | Ponsioen CY, Vrouenraets SM, Prawirodirdjo W, Rajaram R, Rauws EA, Mulder CJ, Reitsma JB, Heisterkamp SH, Tytgat GN. Natural history of primary sclerosing cholangitis and prognostic value of cholangiography in a Dutch population. Gut. 2002;51:562-566. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 194] [Cited by in RCA: 198] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 23. | Barner-Rasmussen N, Pukkala E, Jussila A, Färkkilä M. Epidemiology, risk of malignancy and patient survival in primary sclerosing cholangitis: a population-based study in Finland. Scand J Gastroenterol. 2020;55:74-81. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 50] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 24. | Goode EC, Clark AB, Mells GF, Srivastava B, Spiess K, Gelson WTH, Trivedi PJ, Lynch KD, Castren E, Vesterhus MN, Karlsen TH, Ji SG, Anderson CA, Thorburn D, Hudson M, Heneghan MA, Aldersley MA, Bathgate A, Sandford RN, Alexander GJ, Chapman RW, Walmsley M; UK-PSC Consortium, Hirschfield GM, Rushbrook SM. Factors Associated With Outcomes of Patients With Primary Sclerosing Cholangitis and Development and Validation of a Risk Scoring System. Hepatology. 2019;69:2120-2135. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 37] [Cited by in RCA: 67] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 25. | Fevery J, Verslype C, Lai G, Aerts R, Van Steenbergen W. Incidence, diagnosis, and therapy of cholangiocarcinoma in patients with primary sclerosing cholangitis. Dig Dis Sci. 2007;52:3123-3135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 97] [Article Influence: 5.4] [Reference Citation Analysis (0)] |
| 26. | Lleo A, de Boer YS, Liberal R, Colombo M. The risk of liver cancer in autoimmune liver diseases. Ther Adv Med Oncol. 2019;11:1758835919861914. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 27. | Weismüller TJ, Trivedi PJ, Bergquist A, Imam M, Lenzen H, Ponsioen CY, Holm K, Gotthardt D, Färkkilä MA, Marschall HU, Thorburn D, Weersma RK, Fevery J, Mueller T, Chazouillères O, Schulze K, Lazaridis KN, Almer S, Pereira SP, Levy C, Mason A, Naess S, Bowlus CL, Floreani A, Halilbasic E, Yimam KK, Milkiewicz P, Beuers U, Huynh DK, Pares A, Manser CN, Dalekos GN, Eksteen B, Invernizzi P, Berg CP, Kirchner GI, Sarrazin C, Zimmer V, Fabris L, Braun F, Marzioni M, Juran BD, Said K, Rupp C, Jokelainen K, Benito de Valle M, Saffioti F, Cheung A, Trauner M, Schramm C, Chapman RW, Karlsen TH, Schrumpf E, Strassburg CP, Manns MP, Lindor KD, Hirschfield GM, Hansen BE, Boberg KM; International PSC Study Group. Patient Age, Sex, and Inflammatory Bowel Disease Phenotype Associate With Course of Primary Sclerosing Cholangitis. Gastroenterology. 2017;152:1975-1984.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 345] [Cited by in RCA: 364] [Article Influence: 45.5] [Reference Citation Analysis (1)] |
| 28. | Tabibian JH, Ali AH, Lindor KD. Primary Sclerosing Cholangitis, Part 2: Cancer Risk, Prevention, and Surveillance. Gastroenterol Hepatol (N Y). 2018;14:427-432. [PubMed] |
| 29. | Clarke WT, Feuerstein JD. Colorectal cancer surveillance in inflammatory bowel disease: Practice guidelines and recent developments. World J Gastroenterol. 2019;25:4148-4157. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 91] [Cited by in RCA: 120] [Article Influence: 20.0] [Reference Citation Analysis (0)] |
| 30. | Navaneethan U, Kochhar G, Venkatesh PG, Lewis B, Lashner BA, Remzi FH, Shen B, Kiran RP. Duration and severity of primary sclerosing cholangitis is not associated with risk of neoplastic changes in the colon in patients with ulcerative colitis. Gastrointest Endosc. 2012;75:1045-1054.e1. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 26] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 31. | Rabbenou W, Ullman TA. Risk of Colon Cancer and Recommended Surveillance Strategies in Patients with Ulcerative Colitis. Gastroenterol Clin North Am. 2020;49:791-807. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 32. | Gionchetti P, Calabrese C, Laureti S, Poggioli G, Rizzello F. Pouchitis: Clinical Features, Diagnosis, and Treatment. Int J Gen Med. 2021;14:3871-3879. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 14] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 33. | Shi J, Li Z, Zeng X, Lin Y, Xie WF. Ursodeoxycholic acid in primary sclerosing cholangitis: meta-analysis of randomized controlled trials. Hepatol Res. 2009;39:865-873. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 50] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 34. | Triantos CK, Koukias NM, Nikolopoulou VN, Burroughs AK. Meta-analysis: ursodeoxycholic acid for primary sclerosing cholangitis. Aliment Pharmacol Ther. 2011;34:901-910. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 83] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 35. | Hansen JD, Kumar S, Lo WK, Poulsen DM, Halai UA, Tater KC. Ursodiol and colorectal cancer or dysplasia risk in primary sclerosing cholangitis and inflammatory bowel disease: a meta-analysis. Dig Dis Sci. 2013;58:3079-3087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 37] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 36. | Palmela C, Peerani F, Castaneda D, Torres J, Itzkowitz SH. Inflammatory Bowel Disease and Primary Sclerosing Cholangitis: A Review of the Phenotype and Associated Specific Features. Gut Liver. 2018;12:17-29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 116] [Article Influence: 16.6] [Reference Citation Analysis (0)] |
| 37. | European Association for the Study of the Liver. EASL Clinical Practice Guidelines: management of cholestatic liver diseases. J Hepatol. 2009;51:237-267. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1382] [Cited by in RCA: 1200] [Article Influence: 75.0] [Reference Citation Analysis (1)] |
| 38. | Aabakken L, Karlsen TH, Albert J, Arvanitakis M, Chazouilleres O, Dumonceau JM, Färkkilä M, Fickert P, Hirschfield GM, Laghi A, Marzioni M, Fernandez M, Pereira SP, Pohl J, Poley JW, Ponsioen CY, Schramm C, Swahn F, Tringali A, Hassan C. Role of endoscopy in primary sclerosing cholangitis: European Society of Gastrointestinal Endoscopy (ESGE) and European Association for the Study of the Liver (EASL) Clinical Guideline. Endoscopy. 2017;49:588-608. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 141] [Article Influence: 17.6] [Reference Citation Analysis (0)] |
| 39. | Gidwaney NG, Pawa S, Das KM. Pathogenesis and clinical spectrum of primary sclerosing cholangitis. World J Gastroenterol. 2017;23:2459-2469. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 21] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 40. | Hildebrand T, Pannicke N, Dechene A, Gotthardt DN, Kirchner G, Reiter FP, Sterneck M, Herzer K, Lenzen H, Rupp C, Barg-Hock H, de Leuw P, Teufel A, Zimmer V, Lammert F, Sarrazin C, Spengler U, Rust C, Manns MP, Strassburg CP, Schramm C, Weismüller TJ; German PSC Study Group. Biliary strictures and recurrence after liver transplantation for primary sclerosing cholangitis: A retrospective multicenter analysis. Liver Transpl. 2016;22:42-52. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 89] [Cited by in RCA: 100] [Article Influence: 11.1] [Reference Citation Analysis (0)] |
| 41. | Wee A, Ludwig J. Pericholangitis in chronic ulcerative colitis: primary sclerosing cholangitis of the small bile ducts? Ann Intern Med. 1985;102:581-587. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 146] [Cited by in RCA: 104] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 42. | Björnsson E, Olsson R, Bergquist A, Lindgren S, Braden B, Chapman RW, Boberg KM, Angulo P. The natural history of small-duct primary sclerosing cholangitis. Gastroenterology. 2008;134:975-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 237] [Cited by in RCA: 183] [Article Influence: 10.8] [Reference Citation Analysis (0)] |
| 43. | Ricciuto A, Kamath BM, Griffiths AM. The IBD and PSC Phenotypes of PSC-IBD. Curr Gastroenterol Rep. 2018;20:16. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 58] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 44. | Rust C, Beuers U. Overlap syndromes among autoimmune liver diseases. World J Gastroenterol. 2008;14:3368-3373. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 89] [Cited by in RCA: 111] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 45. | Floreani A, Rizzotto ER, Ferrara F, Carderi I, Caroli D, Blasone L, Baldo V. Clinical course and outcome of autoimmune hepatitis/primary sclerosing cholangitis overlap syndrome. Am J Gastroenterol. 2005;100:1516-1522. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 166] [Cited by in RCA: 153] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 46. | Ballotin VR, Bigarella LG, Riva F, Onzi G, Balbinot RA, Balbinot SS, Soldera J. Primary sclerosing cholangitis and autoimmune hepatitis overlap syndrome associated with inflammatory bowel disease: A case report and systematic review. World J Clin Cases. 2020;8:4075-4093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 17] [Cited by in RCA: 17] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 47. | Ohara H, Okazaki K, Tsubouchi H, Inui K, Kawa S, Kamisawa T, Tazuma S, Uchida K, Hirano K, Yoshida H, Nishino T, Ko SB, Mizuno N, Hamano H, Kanno A, Notohara K, Hasebe O, Nakazawa T, Nakanuma Y, Takikawa H; Research Committee of IgG4-related Diseases; Research Committee of Intractable Diseases of Liver and Biliary Tract; Ministry of Health, Labor and Welfare, Japan; Japan Biliary Association. Clinical diagnostic criteria of IgG4-related sclerosing cholangitis 2012. J Hepatobiliary Pancreat Sci. 2012;19:536-542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 267] [Cited by in RCA: 251] [Article Influence: 20.9] [Reference Citation Analysis (0)] |
| 48. | Manganis CD, Chapman RW, Culver EL. Review of primary sclerosing cholangitis with increased IgG4 Levels. World J Gastroenterol. 2020;26:3126-3144. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (0)] |
| 49. | Song DJ, Shen J, Chen MH, Liu ZJ, Cao Q, Hu PJ, Gao X, Qian JM, Wu KC, Lai LJ, Ran ZH. Association of Serum Immunoglobulins Levels With Specific Disease Phenotypes of Crohn's Disease: A Multicenter Analysis in China. Front Med (Lausanne). 2021;8:621337. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 50. | Löhr JM, Beuers U, Vujasinovic M, Alvaro D, Frøkjær JB, Buttgereit F, Capurso G, Culver EL, de-Madaria E, Della-Torre E, Detlefsen S, Dominguez-Muñoz E, Czubkowski P, Ewald N, Frulloni L, Gubergrits N, Duman DG, Hackert T, Iglesias-Garcia J, Kartalis N, Laghi A, Lammert F, Lindgren F, Okhlobystin A, Oracz G, Parniczky A, Mucelli RMP, Rebours V, Rosendahl J, Schleinitz N, Schneider A, van Bommel EF, Verbeke CS, Vullierme MP, Witt H; UEG guideline working group. European Guideline on IgG4-related digestive disease - UEG and SGF evidence-based recommendations. United European Gastroenterol J. 2020;8:637-666. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 163] [Cited by in RCA: 143] [Article Influence: 28.6] [Reference Citation Analysis (0)] |
| 51. | Kamisawa T, Nakazawa T, Tazuma S, Zen Y, Tanaka A, Ohara H, Muraki T, Inui K, Inoue D, Nishino T, Naitoh I, Itoi T, Notohara K, Kanno A, Kubota K, Hirano K, Isayama H, Shimizu K, Tsuyuguchi T, Shimosegawa T, Kawa S, Chiba T, Okazaki K, Takikawa H, Kimura W, Unno M, Yoshida M. Clinical practice guidelines for IgG4-related sclerosing cholangitis. J Hepatobiliary Pancreat Sci. 2019;26:9-42. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 98] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 52. | Tanaka A. Autoimmune Hepatitis: 2019 Update. Gut Liver. 2020;14:430-438. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 34] [Cited by in RCA: 63] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 53. | Halling ML, Kjeldsen J, Knudsen T, Nielsen J, Hansen LK. Patients with inflammatory bowel disease have increased risk of autoimmune and inflammatory diseases. World J Gastroenterol. 2017;23:6137-6146. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 124] [Cited by in RCA: 163] [Article Influence: 20.4] [Reference Citation Analysis (3)] |
| 54. | Perdigoto R, Carpenter HA, Czaja AJ. Frequency and significance of chronic ulcerative colitis in severe corticosteroid-treated autoimmune hepatitis. J Hepatol. 1992;14:325-331. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 117] [Cited by in RCA: 98] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 55. | Khandelwal A, Gorsi U, Marginean EC, Papadatos D, George U. Isolated granulomatous hepatitis-A histopathological surprise mimicking cholangiocarcinoma in ulcerative colitis. Ann Hepatol. 2013;12:332-335. [PubMed] |
| 56. | Hilzenrat N, Lamoureux E, Sherker A, Cohen A. Cholestasis in Crohn's disease: a diagnostic challenge. Can J Gastroenterol. 1997;11:35-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 57. | Patedakis Litvinov BI, Pathak AP. Granulomatous hepatitis in a patient with Crohn's disease and cholestasis. BMJ Case Rep. 2017;2017. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 58. | Kim JM, Cheon JH. Pathogenesis and clinical perspectives of extraintestinal manifestations in inflammatory bowel diseases. Intest Res. 2020;18:249-264. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 43] [Article Influence: 8.6] [Reference Citation Analysis (0)] |
| 59. | Rojas-Feria M, Castro M, Suárez E, Ampuero J, Romero-Gómez M. Hepatobiliary manifestations in inflammatory bowel disease: the gut, the drugs and the liver. World J Gastroenterol. 2013;19:7327-7340. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 84] [Cited by in RCA: 92] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 60. | Knox TA, Kaplan MM, Gelfand JA, Wolff SM. Methotrexate treatment of idiopathic granulomatous hepatitis. Ann Intern Med. 1995;122:592-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 24] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 61. | Restellini S, Chazouillères O, Frossard JL. Hepatic manifestations of inflammatory bowel diseases. Liver Int. 2017;37:475-489. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 62. | Lorusso D, Leo S, Mossa A, Misciagna G, Guerra V. Cholelithiasis in inflammatory bowel disease. A case-control study. Dis Colon Rectum. 1990;33:791-794. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 41] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 63. | Fraquelli M, Losco A, Visentin S, Cesana BM, Pometta R, Colli A, Conte D. Gallstone disease and related risk factors in patients with Crohn disease: analysis of 330 consecutive cases. Arch Intern Med. 2001;161:2201-2204. [PubMed] |
| 64. | Bargiggia S, Maconi G, Elli M, Molteni P, Ardizzone S, Parente F, Todaro I, Greco S, Manzionna G, Bianchi Porro G. Sonographic prevalence of liver steatosis and biliary tract stones in patients with inflammatory bowel disease: study of 511 subjects at a single center. J Clin Gastroenterol. 2003;36:417-420. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 103] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 65. | Kratzer W, Haenle MM, Mason RA, von Tirpitz C, Kaechele V. Prevalence of cholelithiasis in patients with chronic inflammatory bowel disease. World J Gastroenterol. 2005;11:6170-6175. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 24] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 66. | Zhang FM, Xu CF, Shan GD, Chen HT, Xu GQ. Is gallstone disease associated with inflammatory bowel diseases? J Dig Dis. 2015;16:634-641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 30] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 67. | Sturdik I, Krajcovicova A, Jalali Y, Adamcova M, Tkacik M, Sekac J, Koller T, Huorka M, Payer J, Hlavaty T. Pathophysiology and risk factors for cholelithiasis in patients with Crohn's disease. Physiol Res. 2019;68:S173-S182. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 68. | Fagagnini S, Heinrich H, Rossel JB, Biedermann L, Frei P, Zeitz J, Spalinger M, Battegay E, Zimmerli L, Vavricka SR, Rogler G, Scharl M, Misselwitz B. Risk factors for gallstones and kidney stones in a cohort of patients with inflammatory bowel diseases. PLoS One. 2017;12:e0185193. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 48] [Article Influence: 6.0] [Reference Citation Analysis (0)] |
| 69. | Chen CH, Lin CL, Kao CH. Association between Inflammatory Bowel Disease and Cholelithiasis: A Nationwide Population-Based Cohort Study. Int J Environ Res Public Health. 2018;15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 70. | Parente F, Pastore L, Bargiggia S, Cucino C, Greco S, Molteni M, Ardizzone S, Porro GB, Sampietro GM, Giorgi R, Moretti R, Gallus S. Incidence and risk factors for gallstones in patients with inflammatory bowel disease: a large case-control study. Hepatology. 2007;45:1267-1274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 92] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 71. | Lapidus A, Einarsson C. Bile composition in patients with ileal resection due to Crohn's disease. Inflamm Bowel Dis. 1998;4:89-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 16] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 72. | Heaton KW. Disturbances of bile acid metabolism in intestinal disease. Clin Gastroenterol. 1977;6:69-89. [PubMed] |
| 73. | Dietrich CG, Rau M, Geier A. Screening for nonalcoholic fatty liver disease-when, who and how? World J Gastroenterol. 2021;27:5803-5821. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 10] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (2)] |
| 74. | Ge X, Zheng L, Wang M, Du Y, Jiang J. Prevalence trends in non-alcoholic fatty liver disease at the global, regional and national levels, 1990-2017: a population-based observational study. BMJ Open. 2020;10:e036663. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 73] [Cited by in RCA: 129] [Article Influence: 25.8] [Reference Citation Analysis (0)] |
| 75. | Voss J, Schneider CV, Kleinjans M, Bruns T, Trautwein C, Strnad P. Hepatobiliary phenotype of individuals with chronic intestinal disorders. Sci Rep. 2021;11:19954. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 12] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 76. | Yen HH, Su PY, Huang SP, Wu L, Hsu TC, Zeng YH, Chen YY. Evaluation of non-alcoholic fatty liver disease in patients with inflammatory bowel disease using controlled attenuation parameter technology: A Taiwanese retrospective cohort study. PLoS One. 2021;16:e0252286. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 26] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 77. | Likhitsup A, Dundulis J, Ansari S, Patibandla S, Hutton C, Kennedy K, Helzberg JH, Chhabra R. High prevalence of non-alcoholic fatty liver disease in patients with inflammatory bowel disease receiving anti-tumor necrosis factor therapy. Ann Gastroenterol. 2019;32:463-468. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 78. | Lapumnuaypol K, Kanjanahattakij N, Pisarcik D, Thongprayoon C, Wijarnpreecha K, Cheungpasitporn W. Effects of inflammatory bowel disease treatment on the risk of nonalcoholic fatty liver disease: a meta-analysis. Eur J Gastroenterol Hepatol. 2018;30:854-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 79. | Hoffmann P, Jung V, Behnisch R, Gauss A. Prevalence and risk factors of nonalcoholic fatty liver disease in patients with inflammatory bowel diseases: A cross-sectional and longitudinal analysis. World J Gastroenterol. 2020;26:7367-7381. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 14] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (1)] |
| 80. | Sartini A, Gitto S, Bianchini M, Verga MC, Di Girolamo M, Bertani A, Del Buono M, Schepis F, Lei B, De Maria N, Villa E. Non-alcoholic fatty liver disease phenotypes in patients with inflammatory bowel disease. Cell Death Dis. 2018;9:87. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 58] [Cited by in RCA: 70] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 81. | Lin JN, Lin CL, Lin MC, Lai CH, Lin HH, Kao CH. Pyogenic liver abscess in patients with inflammatory bowel disease: a nationwide cohort study. Liver Int. 2016;36:136-144. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 28] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 82. | McGreal S, Sayers R, Wurm P, West K. Crohn's disease presenting with pyogenic liver abscess: a case report. Case Rep Gastrointest Med. 2012;2012:762480. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 83. | Navot-Mintzer D, Koren A, Shahbari A, Nussinson E, Sakran W. Liver abscesses as the presenting manifestation of Crohn's disease in an adolescent. Inflamm Bowel Dis. 2006;12:666-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 6] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 84. | Margalit M, Elinav H, Ilan Y, Shalit M. Liver abscess in inflammatory bowel disease: report of two cases and review of the literature. J Gastroenterol Hepatol. 2004;19:1338-1342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 52] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 85. | Grainge MJ, West J, Card TR. Venous thromboembolism during active disease and remission in inflammatory bowel disease: a cohort study. Lancet. 2010;375:657-663. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 452] [Cited by in RCA: 556] [Article Influence: 37.1] [Reference Citation Analysis (0)] |
| 86. | Talbot RW, Heppell J, Dozois RR, Beart RW Jr. Vascular complications of inflammatory bowel disease. Mayo Clin Proc. 1986;61:140-145. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 408] [Cited by in RCA: 413] [Article Influence: 10.6] [Reference Citation Analysis (0)] |
| 87. | Gupta S, Hidalgo J, Singh B, Iyer A, Yang Y, Short A, Singh S, Bhatt H, Gupta S. Usage of Direct Acting Oral Anticoagulants in Cirrhotic and Non-Cirrhotic Portal Vein Thrombosis: A Systematic Review. Cureus. 2021;13:e16922. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 4] [Cited by in RCA: 3] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 88. | Remzi FH, Fazio VW, Oncel M, Baker ME, Church JM, Ooi BS, Connor JT, Preen M, Einstein D. Portal vein thrombi after restorative proctocolectomy. Surgery. 2002;132:655-61; discussion 661. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 73] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 89. | Jackson LM, O'Gorman PJ, O'Connell J, Cronin CC, Cotter KP, Shanahan F. Thrombosis in inflammatory bowel disease: clinical setting, procoagulant profile and factor V Leiden. QJM. 1997;90:183-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 137] [Cited by in RCA: 143] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 90. | Sleutjes JAM, van Lennep JER, van der Woude CJ, de Vries AC. Thromboembolic and atherosclerotic cardiovascular events in inflammatory bowel disease: epidemiology, pathogenesis and clinical management. Therap Adv Gastroenterol. 2021;14:17562848211032126. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 6] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 91. | Harbord M, Eliakim R, Bettenworth D, Karmiris K, Katsanos K, Kopylov U, Kucharzik T, Molnár T, Raine T, Sebastian S, de Sousa HT, Dignass A, Carbonnel F; European Crohn’s and Colitis Organisation [ECCO]. Third European Evidence-based Consensus on Diagnosis and Management of Ulcerative Colitis. Part 2: Current Management. J Crohns Colitis. 2017;11:769-784. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 938] [Cited by in RCA: 875] [Article Influence: 109.4] [Reference Citation Analysis (0)] |
| 92. | Zezos P, Kouklakis G, Saibil F. Inflammatory bowel disease and thromboembolism. World J Gastroenterol. 2014;20:13863-13878. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 126] [Cited by in RCA: 109] [Article Influence: 9.9] [Reference Citation Analysis (2)] |
| 93. | Jackson CS, Fryer J, Danese S, Vanagunas A, Polensky S, Buchman AL. Mesenteric vascular thromboembolism in inflammatory bowel disease: a single center experience. J Gastrointest Surg. 2011;15:97-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 19] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 94. | Sinagra E, Aragona E, Romano C, Maisano S, Orlando A, Virdone R, Tesè L, Modesto I, Criscuoli V, Cottone M. The role of portal vein thrombosis in the clinical course of inflammatory bowel diseases: report on three cases and review of the literature. Gastroenterol Res Pract. 2012;2012:916428. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 95. | Olivera PA, Zuily S, Kotze PG, Regnault V, Al Awadhi S, Bossuyt P, Gearry RB, Ghosh S, Kobayashi T, Lacolley P, Louis E, Magro F, Ng SC, Papa A, Raine T, Teixeira FV, Rubin DT, Danese S, Peyrin-Biroulet L. International consensus on the prevention of venous and arterial thrombotic events in patients with inflammatory bowel disease. Nat Rev Gastroenterol Hepatol. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 90] [Article Influence: 22.5] [Reference Citation Analysis (0)] |
| 96. | Voudoukis E, Karmiris K, Koutroubakis IE. Multipotent role of platelets in inflammatory bowel diseases: a clinical approach. World J Gastroenterol. 2014;20:3180-3190. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 63] [Cited by in RCA: 64] [Article Influence: 5.8] [Reference Citation Analysis (1)] |
| 97. | Koutroubakis IE. The relationship between coagulation state and inflammatory bowel disease: current understanding and clinical implications. Expert Rev Clin Immunol. 2015;11:479-488. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 11] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 98. | Chamouard P, Grunebaum L, Wiesel ML, Frey PL, Wittersheim C, Sapin R, Baumann R, Cazenave JP. Prothrombin fragment 1 + 2 and thrombin-antithrombin III complex as markers of activation of blood coagulation in inflammatory bowel diseases. Eur J Gastroenterol Hepatol. 1995;7:1183-1188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 99. | Dolapcioglu C, Soylu A, Kendir T, Ince AT, Dolapcioglu H, Purisa S, Bolukbas C, Sokmen HM, Dalay R, Ovunc O. Coagulation parameters in inflammatory bowel disease. Int J Clin Exp Med. 2014;7:1442-1448. [PubMed] |
| 100. | Magro F, Soares JB, Fernandes D. Venous thrombosis and prothrombotic factors in inflammatory bowel disease. World J Gastroenterol. 2014;20:4857-4872. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 67] [Cited by in RCA: 76] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 101. | European Association for the Study of the Liver. Clinical Practice Guideline Panel: Chair:; Panel members; EASL Governing Board representative: EASL Clinical Practice Guidelines: Drug-induced liver injury. J Hepatol. 2019;70:1222-1261. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 715] [Cited by in RCA: 666] [Article Influence: 111.0] [Reference Citation Analysis (0)] |
| 102. | Azad Khan AK, Nurazzaman M, Truelove SC. The effect of the acetylator phenotype on the metabolism of sulphasalazine in man. J Med Genet. 1983;20:30-36. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 29] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 103. | Torres J, Bonovas S, Doherty G, Kucharzik T, Gisbert JP, Raine T, Adamina M, Armuzzi A, Bachmann O, Bager P, Biancone L, Bokemeyer B, Bossuyt P, Burisch J, Collins P, El-Hussuna A, Ellul P, Frei-Lanter C, Furfaro F, Gingert C, Gionchetti P, Gomollon F, González-Lorenzo M, Gordon H, Hlavaty T, Juillerat P, Katsanos K, Kopylov U, Krustins E, Lytras T, Maaser C, Magro F, Marshall JK, Myrelid P, Pellino G, Rosa I, Sabino J, Savarino E, Spinelli A, Stassen L, Uzzan M, Vavricka S, Verstockt B, Warusavitarne J, Zmora O, Fiorino G. ECCO Guidelines on Therapeutics in Crohn's Disease: Medical Treatment. J Crohns Colitis. 2020;14:4-22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 991] [Cited by in RCA: 903] [Article Influence: 180.6] [Reference Citation Analysis (2)] |
| 104. | Ransford RA, Langman MJ. Sulphasalazine and mesalazine: serious adverse reactions re-evaluated on the basis of suspected adverse reaction reports to the Committee on Safety of Medicines. Gut. 2002;51:536-539. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 222] [Cited by in RCA: 219] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 105. | Ham M, Moss AC. Mesalamine in the treatment and maintenance of remission of ulcerative colitis. Expert Rev Clin Pharmacol. 2012;5:113-123. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 114] [Article Influence: 8.8] [Reference Citation Analysis (0)] |
| 106. | Namias A, Bhalotra R, Donowitz M. Reversible sulfasalazine-induced granulomatous hepatitis. J Clin Gastroenterol. 1981;3:193-198. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 107. | Marinos G, Riley J, Painter DM, McCaughan GW. Sulfasalazine-induced fulminant hepatic failure. J Clin Gastroenterol. 1992;14:132-135. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 25] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 108. | Hirten R, Sultan K, Thomas A, Bernstein DE. Hepatic manifestations of non-steroidal inflammatory bowel disease therapy. World J Hepatol. 2015;7:2716-2728. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 109. | Cuffari C, Théorêt Y, Latour S, Seidman G. 6-Mercaptopurine metabolism in Crohn's disease: correlation with efficacy and toxicity. Gut. 1996;39:401-406. [RCA] [DOI] [Full Text] [Cited by in Crossref: 260] [Cited by in RCA: 247] [Article Influence: 8.5] [Reference Citation Analysis (0)] |
| 110. | Axelrad JE, Roy A, Lawlor G, Korelitz B, Lichtiger S. Thiopurines and inflammatory bowel disease: Current evidence and a historical perspective. World J Gastroenterol. 2016;22:10103-10117. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 36] [Cited by in RCA: 40] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 111. | Bastida G, Nos P, Aguas M, Beltrán B, Rubín A, Dasí F, Ponce J. Incidence, risk factors and clinical course of thiopurine-induced liver injury in patients with inflammatory bowel disease. Aliment Pharmacol Ther. 2005;22:775-782. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 85] [Cited by in RCA: 84] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 112. | Tran-Minh ML, Sousa P, Maillet M, Allez M, Gornet JM. Hepatic complications induced by immunosuppressants and biologics in inflammatory bowel disease. World J Hepatol. 2017;9:613-626. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 22] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 113. | Gisbert JP, González-Lama Y, Maté J. Thiopurine-induced liver injury in patients with inflammatory bowel disease: a systematic review. Am J Gastroenterol. 2007;102:1518-1527. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 134] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 114. | Gisbert JP, Chaparro M, Gomollón F. Common misconceptions about 5-aminosalicylates and thiopurines in inflammatory bowel disease. World J Gastroenterol. 2011;17:3467-3478. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 25] [Cited by in RCA: 21] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 115. | Sousa P, Estevinho MM, Dias CC, Ministro P, Kopylov U, Danese S, Peyrin-Biroulet L, Magro F. Thiopurines' Metabolites and Drug Toxicity: A Meta-Analysis. J Clin Med. 2020;9. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 116. | Thomsen SB, Allin KH, Burisch J, Jensen CB, Hansen S, Gluud LL, Theede K, Kiszka-Kanowitz M, Nielsen AM, Jess T. Outcome of concomitant treatment with thiopurines and allopurinol in patients with inflammatory bowel disease: A nationwide Danish cohort study. United European Gastroenterol J. 2020;8:68-76. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 16] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 117. | Warner B, Johnston E, Arenas-Hernandez M, Marinaki A, Irving P, Sanderson J. A practical guide to thiopurine prescribing and monitoring in IBD. Frontline Gastroenterol. 2018;9:10-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 38] [Cited by in RCA: 54] [Article Influence: 7.7] [Reference Citation Analysis (0)] |
| 118. | Shaye OA, Yadegari M, Abreu MT, Poordad F, Simon K, Martin P, Papadakis KA, Ippoliti A, Vasiliauskas E, Tran TT. Hepatotoxicity of 6-mercaptopurine (6-MP) and Azathioprine (AZA) in adult IBD patients. Am J Gastroenterol. 2007;102:2488-2494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 71] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 119. | Ribaldone DG, Adriani A, Caviglia GP, Nicolò A, Agnesod D, Simiele M, Riganò D, Pellicano R, Canaparo R, Perri GD, D'Avolio A, Luzza F, Saracco GM, Astegiano M. Correlation between Thiopurine S-Methyltransferase Genotype and Adverse Events in Inflammatory Bowel Disease Patients. Medicina (Kaunas). 2019;55. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 12] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 120. | Liu YP, Wu HY, Yang X, Xu HQ, Li YC, Shi DC, Huang JF, Huang Q, Fu WL. Association between thiopurine S-methyltransferase polymorphisms and thiopurine-induced adverse drug reactions in patients with inflammatory bowel disease: a meta-analysis. PLoS One. 2015;10:e0121745. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 121. | Moran GW, Dubeau MF, Kaplan GG, Yang H, Eksteen B, Ghosh S, Panaccione R. Clinical predictors of thiopurine-related adverse events in Crohn's disease. World J Gastroenterol. 2015;21:7795-7804. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 26] [Cited by in RCA: 24] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 122. | McLean LP, Cross RK. Adverse events in IBD: to stop or continue immune suppressant and biologic treatment. Expert Rev Gastroenterol Hepatol. 2014;8:223-240. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 80] [Article Influence: 7.3] [Reference Citation Analysis (0)] |
| 123. | Philips CA, Ahamed R, Rajesh S, George T, Mohanan M, Augustine P. Comprehensive review of hepatotoxicity associated with traditional Indian Ayurvedic herbs. World J Hepatol. 2020;12:574-595. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 42] [Cited by in RCA: 31] [Article Influence: 6.2] [Reference Citation Analysis (3)] |
| 124. | Aithal GP. Hepatotoxicity related to antirheumatic drugs. Nat Rev Rheumatol. 2011;7:139-150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 110] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 125. | Renna S, Cottone M, Orlando A. Optimization of the treatment with immunosuppressants and biologics in inflammatory bowel disease. World J Gastroenterol. 2014;20:9675-9690. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 66] [Cited by in RCA: 69] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 126. | Meijer B, Mulder CJ, van Bodegraven AA, de Boer NK. How I treat my inflammatory bowel disease-patients with thiopurines? World J Gastrointest Pharmacol Ther. 2016;7:524-530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (1)] |
| 127. | Lamb CA, Kennedy NA, Raine T, Hendy PA, Smith PJ, Limdi JK, Hayee B, Lomer MCE, Parkes GC, Selinger C, Barrett KJ, Davies RJ, Bennett C, Gittens S, Dunlop MG, Faiz O, Fraser A, Garrick V, Johnston PD, Parkes M, Sanderson J, Terry H; IBD guidelines eDelphi consensus group, Gaya DR, Iqbal TH, Taylor SA, Smith M, Brookes M, Hansen R, Hawthorne AB. British Society of Gastroenterology consensus guidelines on the management of inflammatory bowel disease in adults. Gut. 2019;68:s1-s106. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1402] [Cited by in RCA: 1564] [Article Influence: 260.7] [Reference Citation Analysis (0)] |
| 128. | Egan LJ, Sandborn WJ. Methotrexate for inflammatory bowel disease: pharmacology and preliminary results. Mayo Clin Proc. 1996;71:69-80. [PubMed] |
| 129. | Patel V, Wang Y, MacDonald JK, McDonald JW, Chande N. Methotrexate for maintenance of remission in Crohn's disease. Cochrane Database Syst Rev. 2014;CD006884. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 51] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 130. | Wang Y, MacDonald JK, Vandermeer B, Griffiths AM, El-Matary W. Methotrexate for maintenance of remission in ulcerative colitis. Cochrane Database Syst Rev. 2015;CD007560. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 27] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 131. | Coskun M, Steenholdt C, de Boer NK, Nielsen OH. Pharmacology and Optimization of Thiopurines and Methotrexate in Inflammatory Bowel Disease. Clin Pharmacokinet. 2016;55:257-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 132. | Almeyda J, Baker H, Levene GM, Barnardo D, Landells JW. Methotrexate, alcohol, and liver damage. Br Med J. 1971;2:167. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 133. | Prey S, Paul C. Effect of folic or folinic acid supplementation on methotrexate-associated safety and efficacy in inflammatory disease: a systematic review. Br J Dermatol. 2009;160:622-628. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 92] [Cited by in RCA: 93] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 134. | Visser K, Katchamart W, Loza E, Martinez-Lopez JA, Salliot C, Trudeau J, Bombardier C, Carmona L, van der Heijde D, Bijlsma JW, Boumpas DT, Canhao H, Edwards CJ, Hamuryudan V, Kvien TK, Leeb BF, Martín-Mola EM, Mielants H, Müller-Ladner U, Murphy G, Østergaard M, Pereira IA, Ramos-Remus C, Valentini G, Zochling J, Dougados M. Multinational evidence-based recommendations for the use of methotrexate in rheumatic disorders with a focus on rheumatoid arthritis: integrating systematic literature research and expert opinion of a broad international panel of rheumatologists in the 3E Initiative. Ann Rheum Dis. 2009;68:1086-1093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 372] [Cited by in RCA: 324] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 135. | Seow CH, de Silva S, Kaplan GG, Devlin SM, Ghosh S, Panaccione R. Managing the risks of IBD therapy. Curr Gastroenterol Rep. 2009;11:509-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 136. | Barbero-Villares A, Mendoza J, Trapero-Marugan M, Gonzalez-Alvaro I, Daudén E, Gisbert JP, Moreno-Otero R. Evaluation of liver fibrosis by transient elastography in methotrexate treated patients. Med Clin (Barc). 2011;137:637-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 23] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 137. | Tobon GJ, Cañas C, Jaller JJ, Restrepo JC, Anaya JM. Serious liver disease induced by infliximab. Clin Rheumatol. 2007;26:578-581. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 114] [Cited by in RCA: 112] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 138. | Lopetuso LR, Mocci G, Marzo M, D'Aversa F, Rapaccini GL, Guidi L, Armuzzi A, Gasbarrini A, Papa A. Harmful Effects and Potential Benefits of Anti-Tumor Necrosis Factor (TNF)-α on the Liver. Int J Mol Sci. 2018;19. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 54] [Cited by in RCA: 61] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 139. | Hagel S, Bruns T, Theis B, Herrmann A, Stallmach A. Subacute liver failure induced by adalimumab. Int J Clin Pharmacol Ther. 2011;49:38-40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 140. | Shelton E, Chaudrey K, Sauk J, Khalili H, Masia R, Nguyen DD, Yajnik V, Ananthakrishnan AN. New onset idiosyncratic liver enzyme elevations with biological therapy in inflammatory bowel disease. Aliment Pharmacol Ther. 2015;41:972-979. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 36] [Cited by in RCA: 45] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 141. | Ghabril M, Bonkovsky HL, Kum C, Davern T, Hayashi PH, Kleiner DE, Serrano J, Rochon J, Fontana RJ, Bonacini M; US Drug-Induced Liver Injury Network. Liver injury from tumor necrosis factor-α antagonists: analysis of thirty-four cases. Clin Gastroenterol Hepatol. 2013;11:558-564.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 173] [Cited by in RCA: 155] [Article Influence: 12.9] [Reference Citation Analysis (0)] |
| 142. | FDA. U.S. Food and Drug Administration. [cited 17 January 2021]. Available from: https://www.fda.gov/science-research/Liver-toxicity-knowledge-base-ltkb/drug-induced-liver-injury-rank-dilirank-dataset. |
| 143. | Shah P, Sundaram V, Björnsson E. Biologic and Checkpoint Inhibitor-Induced Liver Injury: A Systematic Literature Review. Hepatol Commun. 2020;4:172-184. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 25] [Cited by in RCA: 45] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 144. | Biswas S, Bryant RV, Travis S. Interfering with leukocyte trafficking in Crohn's disease. Best Pract Res Clin Gastroenterol. 2019;38-39:101617. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 14] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 145. | Sandborn WJ, Colombel JF, Enns R, Feagan BG, Hanauer SB, Lawrance IC, Panaccione R, Sanders M, Schreiber S, Targan S, van Deventer S, Goldblum R, Despain D, Hogge GS, Rutgeerts P; International Efficacy of Natalizumab as Active Crohn's Therapy (ENACT-1) Trial Group; Evaluation of Natalizumab as Continuous Therapy (ENACT-2) Trial Group. Natalizumab induction and maintenance therapy for Crohn's disease. N Engl J Med. 2005;353:1912-1925. [RCA] [DOI] [Full Text] [Cited by in Crossref: 698] [Cited by in RCA: 664] [Article Influence: 33.2] [Reference Citation Analysis (0)] |
| 146. | Targan SR, Feagan BG, Fedorak RN, Lashner BA, Panaccione R, Present DH, Spehlmann ME, Rutgeerts PJ, Tulassay Z, Volfova M, Wolf DC, Hernandez C, Bornstein J, Sandborn WJ; International Efficacy of Natalizumab in Crohn's Disease Response and Remission (ENCORE) Trial Group. Natalizumab for the treatment of active Crohn's disease: results of the ENCORE Trial. Gastroenterology. 2007;132:1672-1683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 508] [Cited by in RCA: 456] [Article Influence: 25.3] [Reference Citation Analysis (0)] |
| 147. | Wyatt NJ, Speight RA, Stewart CJ, Kirby JA, Lamb CA. Targeting Leukocyte Trafficking in Inflammatory Bowel Disease. BioDrugs. 2021;35:473-503. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 148. | Lenti MV, Levison S, Eliadou E, Willert R, Kemp K, Carter A, Stansfield C, Assadsangabi A, Singh S, Crooks B, Tattersall S, Fairhurst F, Kenneth C, Subramanian S, Probert C, Storey D, Gregg B, Smith P, Liu E, Limdi JK, Johnston A, Hamlin PJ, Selinger CP. A real-world, long-term experience on effectiveness and safety of vedolizumab in adult patients with inflammatory bowel disease: The Cross Pennine study. Dig Liver Dis. 2018;50:1299-1304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 45] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 149. | Clinton JW, Kiparizoska S, Aggarwal S, Woo S, Davis W, Lewis JH. Drug-Induced Liver Injury: Highlights and Controversies in the Recent Literature. Drug Saf. 2021;44:1125-1149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 46] [Article Influence: 11.5] [Reference Citation Analysis (0)] |
| 150. | LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012–. . [PubMed] |
| 151. | Sandborn WJ, Feagan BG, Rutgeerts P, Hanauer S, Colombel JF, Sands BE, Lukas M, Fedorak RN, Lee S, Bressler B, Fox I, Rosario M, Sankoh S, Xu J, Stephens K, Milch C, Parikh A; GEMINI 2 Study Group. Vedolizumab as induction and maintenance therapy for Crohn's disease. N Engl J Med. 2013;369:711-721. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1416] [Cited by in RCA: 1567] [Article Influence: 130.6] [Reference Citation Analysis (1)] |
| 152. | Feagan BG, Rutgeerts P, Sands BE, Hanauer S, Colombel JF, Sandborn WJ, Van Assche G, Axler J, Kim HJ, Danese S, Fox I, Milch C, Sankoh S, Wyant T, Xu J, Parikh A; GEMINI 1 Study Group. Vedolizumab as induction and maintenance therapy for ulcerative colitis. N Engl J Med. 2013;369:699-710. [RCA] [DOI] [Full Text] [Cited by in Crossref: 1576] [Cited by in RCA: 1865] [Article Influence: 155.4] [Reference Citation Analysis (1)] |
| 153. | Sandborn WJ, Gasink C, Gao LL, Blank MA, Johanns J, Guzzo C, Sands BE, Hanauer SB, Targan S, Rutgeerts P, Ghosh S, de Villiers WJ, Panaccione R, Greenberg G, Schreiber S, Lichtiger S, Feagan BG; CERTIFI Study Group. Ustekinumab induction and maintenance therapy in refractory Crohn's disease. N Engl J Med. 2012;367:1519-1528. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 810] [Cited by in RCA: 872] [Article Influence: 67.1] [Reference Citation Analysis (0)] |
| 154. | Sands BE, Sandborn WJ, Panaccione R, O'Brien CD, Zhang H, Johanns J, Adedokun OJ, Li K, Peyrin-Biroulet L, Van Assche G, Danese S, Targan S, Abreu MT, Hisamatsu T, Szapary P, Marano C; UNIFI Study Group. Ustekinumab as Induction and Maintenance Therapy for Ulcerative Colitis. N Engl J Med. 2019;381:1201-1214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 498] [Cited by in RCA: 827] [Article Influence: 137.8] [Reference Citation Analysis (1)] |
| 155. | LiverTox: Clinical and Research Information on Drug-Induced Liver Injury [Internet]. Bethesda (MD): National Institute of Diabetes and Digestive and Kidney Diseases; 2012–. Ustekinumab. . [PubMed] |
| 156. | Chiu HY, Chen CH, Wu MS, Cheng YP, Tsai TF. The safety profile of ustekinumab in the treatment of patients with psoriasis and concurrent hepatitis B or C. Br J Dermatol. 2013;169:1295-1303. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 114] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 157. | FDA. U.S. Food and Drug Administration. FDA approves new treatment for moderately to severely active ulcerative colitis. 2018 May 1. [cited 06 February 2021]. Available from: https://www.fda.gov/news-events/press-announcements/fda-approves-new-treatment-moderately-severely-active-ulcerative-colitis. |
| 158. | Harris C, Cummings JRF. JAK1 inhibition and inflammatory bowel disease. Rheumatology (Oxford). 2021;60:ii45-ii51. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 32] [Cited by in RCA: 48] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 159. | van Vollenhoven RF, Fleischmann R, Cohen S, Lee EB, García Meijide JA, Wagner S, Forejtova S, Zwillich SH, Gruben D, Koncz T, Wallenstein GV, Krishnaswami S, Bradley JD, Wilkinson B; ORAL Standard Investigators. Tofacitinib or adalimumab vs placebo in rheumatoid arthritis. N Engl J Med. 2012;367:508-519. [RCA] [DOI] [Full Text] [Cited by in Crossref: 673] [Cited by in RCA: 733] [Article Influence: 56.4] [Reference Citation Analysis (0)] |
| 160. | Fleischmann R, Cutolo M, Genovese MC, Lee EB, Kanik KS, Sadis S, Connell CA, Gruben D, Krishnaswami S, Wallenstein G, Wilkinson BE, Zwillich SH. Phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) or adalimumab monotherapy vs placebo in patients with active rheumatoid arthritis with an inadequate response to disease-modifying antirheumatic drugs. Arthritis Rheum. 2012;64:617-629. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 315] [Article Influence: 24.2] [Reference Citation Analysis (0)] |
| 161. | Burmester GR, Blanco R, Charles-Schoeman C, Wollenhaupt J, Zerbini C, Benda B, Gruben D, Wallenstein G, Krishnaswami S, Zwillich SH, Koncz T, Soma K, Bradley J, Mebus C; ORAL Step investigators. Tofacitinib (CP-690,550) in combination with methotrexate in patients with active rheumatoid arthritis with an inadequate response to tumour necrosis factor inhibitors: a randomised phase 3 trial. Lancet. 2013;381:451-460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 509] [Cited by in RCA: 553] [Article Influence: 46.1] [Reference Citation Analysis (0)] |
| 162. | Sandborn WJ, Su C, Sands BE, D'Haens GR, Vermeire S, Schreiber S, Danese S, Feagan BG, Reinisch W, Niezychowski W, Friedman G, Lawendy N, Yu D, Woodworth D, Mukherjee A, Zhang H, Healey P, Panés J; OCTAVE Induction 1, OCTAVE Induction 2, and OCTAVE Sustain Investigators. Tofacitinib as Induction and Maintenance Therapy for Ulcerative Colitis. N Engl J Med. 2017;376:1723-1736. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 896] [Cited by in RCA: 1186] [Article Influence: 148.3] [Reference Citation Analysis (0)] |
| 163. | Lair-Mehiri L, Stefanescu C, Vaysse T, Laharie D, Roblin X, Rosa I, Treton X, Abitbol V, Amiot A, Bouguen G, Dib N, Fumery M, Pariente B, Carbonnel F, Peyrin-Biroulet L, Simon M, Viennot S, Bouhnik Y. Real-world evidence of tofacitinib effectiveness and safety in patients with refractory ulcerative colitis. Dig Liver Dis. 2020;52:268-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 34] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 164. | D'Amico F, Parigi TL, Fiorino G, Peyrin-Biroulet L, Danese S. Tofacitinib in the treatment of ulcerative colitis: efficacy and safety from clinical trials to real-world experience. Therap Adv Gastroenterol. 2019;12:1756284819848631. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 49] [Cited by in RCA: 50] [Article Influence: 8.3] [Reference Citation Analysis (0)] |
| 165. | Deepak P, Alayo QA, Khatiwada A, Lin B, Fenster M, Dimopoulos C, Bader G, Weisshof R, Jacobs M, Gutierrez A, Ciorba MA, Christophi GP, Patel A, Hirten RP, Colombel JF, Rubin DT, Ha C, Beniwal-Patel P, Ungaro RC, Syal G, Pekow J, Cohen BL, Yarur A. Safety of Tofacitinib in a Real-World Cohort of Patients With Ulcerative Colitis. Clin Gastroenterol Hepatol. 2021;19:1592-1601.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 79] [Article Influence: 19.8] [Reference Citation Analysis (0)] |
| 166. | Coban S, Kekilli M, Köklü S. Approach and management of patients with chronic hepatitis B and C during the course of inflammatory bowel disease. Inflamm Bowel Dis. 2014;20:2142-2150. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 167. | Chevaux JB, Bigard MA, Bensenane M, Oussalah A, Jarlot S, Belle A, Nani A, Bronowicki JP, Peyrin-Biroulet L. Inflammatory bowel disease and hepatitis B and C. Gastroenterol Clin Biol. 2009;33:1082-1093. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 17] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 168. | Sansone S, Guarino M, Castiglione F, Rispo A, Auriemma F, Loperto I, Rea M, Caporaso N, Morisco F. Hepatitis B and C virus reactivation in immunosuppressed patients with inflammatory bowel disease. World J Gastroenterol. 2014;20:3516-3524. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 169. | Rahier JF, Magro F, Abreu C, Armuzzi A, Ben-Horin S, Chowers Y, Cottone M, de Ridder L, Doherty G, Ehehalt R, Esteve M, Katsanos K, Lees CW, Macmahon E, Moreels T, Reinisch W, Tilg H, Tremblay L, Veereman-Wauters G, Viget N, Yazdanpanah Y, Eliakim R, Colombel JF; European Crohn's and Colitis Organisation (ECCO). Second European evidence-based consensus on the prevention, diagnosis and management of opportunistic infections in inflammatory bowel disease. J Crohns Colitis. 2014;8:443-468. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 694] [Cited by in RCA: 746] [Article Influence: 67.8] [Reference Citation Analysis (0)] |
| 170. | Borman ZA, Côté-Daigneault J, Colombel JF. The risk for opportunistic infections in inflammatory bowel disease with biologics: an update. Expert Rev Gastroenterol Hepatol. 2018;12:1101-1108. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 27] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 171. | Jiang HY, Wang SY, Deng M, Li YC, Ling ZX, Shao L, Ruan B. Immune response to hepatitis B vaccination among people with inflammatory bowel diseases: A systematic review and meta-analysis. Vaccine. 2017;35:2633-2641. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 172. | Craviotto V, Furfaro F, Loy L, Zilli A, Peyrin-Biroulet L, Fiorino G, Danese S, Allocca M. Viral infections in inflammatory bowel disease: Tips and tricks for correct management. World J Gastroenterol. 2021;27:4276-4297. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 11] [Cited by in RCA: 13] [Article Influence: 3.3] [Reference Citation Analysis (4)] |
| 173. | Morisco F, Castiglione F, Rispo A, Stroffolini T, Sansone S, Vitale R, Guarino M, Biancone L, Caruso A, D'Inca R, Marmo R, Orlando A, Riegler G, Donnarumma L, Camera S, Zorzi F, Renna S, Bove V, Tontini G, Vecchi M, Caporaso N. Effect of immunosuppressive therapy on patients with inflammatory bowel diseases and hepatitis B or C virus infection. J Viral Hepat. 2013;20:200-208. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 26] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 174. | Imperatore N, Castiglione F, Rispo A, Sessa A, Caporaso N, Morisco F. Timing Strategies of Direct-Acting Antivirals and Biologics Administration in HCV-Infected Subjects with Inflammatory Bowel Diseases. Front Pharmacol. 2017;8:867. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 7] [Article Influence: 0.9] [Reference Citation Analysis (0)] |