Published online Jul 27, 2020. doi: 10.4254/wjh.v12.i7.378
Peer-review started: January 11, 2020
First decision: April 18, 2020
Revised: May 25, 2020
Accepted: June 10, 2020
Article in press: June 10, 2020
Published online: July 27, 2020
Processing time: 193 Days and 6.4 Hours
Non-alcoholic fatty liver disease (NAFLD) is a systemic disease with bidirectional relationships with cardiovascular disease (CVD). Non-alcoholic steatohepatitis (NASH) is a more severe subtype of NAFLD. Patients with NASH exhibit more intra and extrahepatic inflammation, procoagulant imbalances and proatherogenic lipid profiles. Whether NASH increases the risk of ischemic heart disease is currently unclear.
To investigate the relationship between acute myocardial infarction (MI) and NASH in a large cohort of subjects in the United States.
We reviewed data from a large commercial database (Explorys IBM) that aggregates electronic health records from 26 large nationwide healthcare systems. Using systemized nomenclature of clinical medical terms (SNOMED CT), we identified adult with the diagnosis of NASH from 1999-2019. We included patients with the diagnosis of acute MI from 2018-2019. Comorbidities known to be associated with NASH and MI such as obesity, diabetes mellitus, hyperlipidemia, smoking, male gender, and hypertension were collected. Univariable and multivariable analyses were performed to investigate whether NASH is independently associated with the risk of MI.
Out of 55099280 patients, 43170 were diagnosed with NASH (0.08%) and 107000 (0.194%) had a MI within 2018-2019. After adjusting for traditional risk factors, NASH conferred greater odds of MI odds ratio (OR) 1.5 [95% confidence interval (CI): 1.40-1.62]. Hyperlipidemia had the strongest association with MI OR 8.39 (95%CI: 8.21-8.58) followed by hypertension OR 3.11 (95%CI: 3.05-3.17) and smoking OR 2.83 (95%CI: 2.79-2.87). NASH had a similar association with MI as the following traditional risk factors like age above 65 years OR 1.47 (95%CI: 1.45-1.49), male gender OR 1.53 (95%CI: 1.51-1.55) diabetes mellitus OR 1.89 (95%CI: 1.86-1.91).
MI appears to be a prevalent disease in NASH. Patients with NASH may need early identification and aggressive cardiovascular risk modification.
Core tip: Non-alcoholic steatohepatitis (NASH) is a severe subtype of nonalcoholic fatty liver disease. The progression of non-alcoholic fatty liver disease is mirrored by activation of hepatic and systemic inflammatory cascades that maybe implicated in the pathogenesis of cardiovascular disease. Using a large electronic medical record database, we performed a national-based population study to investigate the association between NASH and myocardial infarction (MI). In this large cohort study, NASH was associated with increased risk of MI independent of traditional risk factors. Close follow up and aggressive risk modification maybe indicated to prevent major cardiovascular events in patients with NASH.
- Citation: Ghoneim S, Dhorepatil A, Shah AR, Ram G, Ahmad S, Kim C, Asaad I. Non-alcoholic steatohepatitis and the risk of myocardial infarction: A population-based national study. World J Hepatol 2020; 12(7): 378-388
- URL: https://www.wjgnet.com/1948-5182/full/v12/i7/378.htm
- DOI: https://dx.doi.org/10.4254/wjh.v12.i7.378
Non-alcoholic fatty liver disease (NAFLD) is the hepatic manifestation of metabolic syndrome (MetS) defined by the presence of central obesity, insulin resistance and dyslipidemia[1-3]. The severity of NAFLD ranges from simple steatosis to non-alcoholic steatohepatitis (NASH), liver fibrosis progressing to cirrhosis and hepatocellular carcinoma[1,2]. Non-alcoholic liver disease is a world-wide chronic disease and the most common liver disorder in North America[2-5]. The prevalence of NAFLD in the United States is reported to be 10%-46% with approximately 7%-30% meeting the criteria of NASH[6].
NAFLD is not only associated with increased liver-related morbidity and mortality, it is also associated with increased risk of mortality due to cardiovascular disease (CVD)[7-9]. The distillation of NAFLD as an independent risk factor for CVD is impeded by overlap with established risk factors for CVD that are also risk factors for NAFLD. Even in the absence of cardiovascular mortality, CVD is highly prevalent in patients with NAFLD[7-9]. There is convincing data supporting the association of NAFLD with subclinical atherosclerosis, carotid arthrosclerosis and venous thromboembolic events[10-13]. The severity of coronary artery disease is also higher in patients with NAFLD diagnosed by coronary angiography[14]. Several mechanisms have been proposed by which NAFLD may give rise to CVD. Endothelial damage is considered the first step in atherosclerosis. Intrahepatic and systemic endothelial damage have been noted in NAFLD and are more pronounced in NASH[15]. Second, inflammation fuels CVD and promotes endothelial damage, by altering vascular tone and enhancing platelet dysfunction[16]. Circulating proinflammatory cytokines are increased in NAFLD and are highly expressed in NASH[17]. Moreover, the liver plays a key role in whole body lipid metabolism. Serum lipid profiles mirror disease severity and are significantly higher in NASH[17]. Furthermore disturbances in haemostasis are seen in NAFLD and correlate with disease severity[18]. Nonetheless, the role of NAFLD as an independent cardiovascular risk is still debatable with research yielding contradictory results. In one meta-analysis that included 34043 subjects, NALFD was associated with increased overall mortality deriving from liver-related and CVD odds ratio (OR) 1.57 [95% confidence interval (CI): 1.18-2.10)[19]. In a prospective study, Baratta et al[20] reported NAFLD to have more than a 2-fold risk increase and those with fibrosis had a fourfold increase in cardiovascular events defined as fatal or nonfatal ischemic stroke or myocardial infarction (MI).
Other studies have failed to confirm this association[7,8]. This data however should be interpreted with caution because of methodological issues. To illustrate, the diagnosis of NAFLD relied on ultrasound imaging or biochemical testing both of which correlate poorly with histological findings. Furthermore, the degree of disease severity was not stratified in any of these studies. As we highlighted above, the progression of NAFLD is mirrored by increased activation of bidirectional pathways that maybe involved in ischemic CVD. Whether NASH is associated with an increased risk of MI in the United States population remains unclear. Given the lack of consensus on this important issue, and the ambiguity of data around this question, the aim of this study is to investigate the association between NASH and MI in United States population.
This is a retrospective analysis of a large electronic health record (EHR)-based commercial database called Explorys (IBM). Explorys platform assimilates patient information from 26 major healthcare systems spread over 50 states in the United States. It stores over 60 million unique patient records[20]. Patient information is then de-identified, standardized and stored in a cloud database[20,21]. The Explorys platform uses SNOMED CT for medical diagnoses and procedures. For diagnoses, International Classification of Disease, Ninth Revision, Clinical Modification (ICD-9-CM) codes are mapped into the SNOMED-CT hierarchy[20]. Cohorts can further be refined demographically and comorbid diseases can be extracted[20,21]. The use of Explorys has been validated in multiple fields including Cardiology and Gastroenterology[20]. This platform is Health Insurance Portability and Accountability Act and Health Information Technology for Economic and Clinical Health Act compliant[20]. The Case Western Reserve University/Metrohealth Medical Center Institutional Review Board deemed studies using Explorys, as the dataset of record, as exempt from approval because all patient information is de-identified. Explorys protects patient confidentiality by approximating each population count to 10. All counts between 0 and 10 are treated equally. For the purpose of this study, counts between 0 and 10 were approximated to be five[21].
Subjects with active electronic health records since 1999 were identified using the search tool in Explorys. Using the SNOMED-CT diagnosis “Non-alcoholic steatohepatitis”, we identified patients who were diagnosed with NASH between 1999-2019. Patients with the diagnosis of “fatty liver disease”, “alcoholic hepatitis” and “alcoholic fatty liver disease” were excluded. Patients with MI were identified as those with the following SNOMED-CT diagnosis “Acute myocardial infarction”. To establish a temporal relationship with NASH, only those with the diagnosis of acute MI within the last year of the study, 2018-2019, were included. Controls were identified as those patients without the diagnosis of NASH. For our analysis we identified cohorts of patients with NASH, with and without MI. It was not possible to perform propensity-score matching because this database only provides population-level data and not individual cases.
The following characteristics were collected: demographic factors such as age, sex, and comorbidities, known to be associated with NASH and MI (hyperlipidemia, hypertension, obesity, diabetes mellitus, smoking age and gender), by searching the database for their respective SNOMED-CT terms.
To assess the association between MI with risk factors we divided the whole cohort of patients into MI and Non-MI patients. The prevalence of MI was calculated by dividing the number of patients with MI in each risk group (NASH, hypertension, diabetes mellitus, obesity, hyperlipidemia, age, gender and smoking). Frequency and percentages were used for statistical description of categorical variables and compared using the Pearson χ2 test. ORs are presented with 95%CIs. Univariate binary logistic model was constructed using MI as dependent variable and other variables as independent variables. To adjust for possible confounding, a multivariable model adjusting for all covariates mentioned in univariate variables were added to test the main effect. Analyses were performed using R version 3.6.1 (The R Foundation, Vienna, Austria). Independence among covariate risk factors was assessed using the variance inflating factor (VIF) with cut-off of significant collinearity set at VIF > 1.5. “Goodness-of-fit” was assessed for all regression models using the Hosmer-Lemeshow test, with P > 0.05 indicating good fit. Two-sided P values were used and were considered statistically significant if P < 0.05.
Out of 55099280 patients, 43170 patients were diagnosed with NASH (0.08%) and 107000 (0.194%) had acute MI within 2018-2019. Baseline characteristics of subjects included in this study are displayed in Table 1.
| Total cohort | Proportion (%) | NASH cohort | Proportion (%) | |
| Total cohort | 55099280 | 43170 | ||
| Male gender | 24569555 | 45 | 17380 | 40 |
| Age above 65 yr | 15333620 | 28 | 17020 | 39 |
| Essential hypertension | 11043970 | 20 | 32040 | 74 |
| Hyperlipidemia | 9539460 | 17 | 31030 | 72 |
| Smoking | 4993910 | 9 | 9920 | 23 |
| Diabetes mellitus | 4785840 | 9 | 24640 | 57 |
| Obesity | 4226640 | 8 | 25570 | 59 |
The prevalence of MI in subjects with NASH was 10.24% and 0.18% in the non-NASH group. In overall unadjusted analysis, the unadjusted OR for MI in patients with NASH was 10.66 (95%CI: 9.58-10.94). Hyperlipidemia was strongly associated with increased risk of MI OR 40.13 (95%CI: 39.47-40.79), followed by hypertension OR 27.43 (95%CI: 27.00-7.86) and the male gender OR 17.61 (95%CI: 17.41-17.81). Diabetes, obesity, smoking and age above 65 years had slightly lower association with MI than NASH OR 12.57 (95%CI: 12.44–12.71), 8.19 (95%CI: 8.09-8.28), 6.71 (95%CI: 6.64-6.79) and 4.97 (95%CI: 4.92-5.03), respectively (Table 2).
| Odds ratio | 95%CIb | |
| Hyperlipidemia | 40.13 | 39.47-40.79 |
| Essential hypertension | 27.43 | 27.00-7.86 |
| Male gender | 17.61 | 17.41-17.81 |
| Diabetes mellitus | 12.57 | 12.44–12.71 |
| NASH | 10.66 | 9.58-10.94 |
| Obesity | 8.19 | 8.09-8.28 |
| Smoking | 6.71 | 6.64-6.79 |
| Age above 65 yr | 4.97 | 4.92-5.03 |
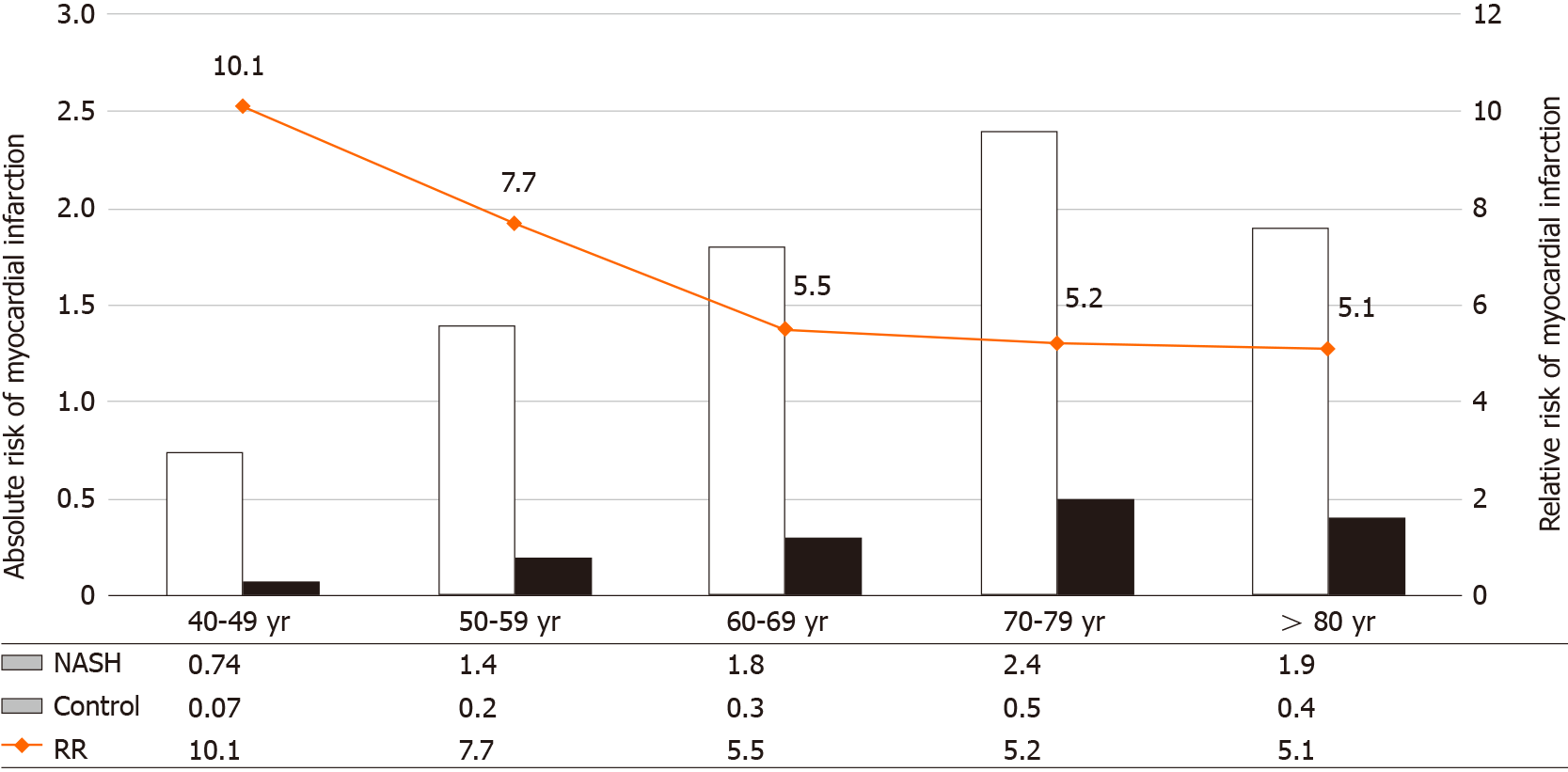
Compared to the older NASH population, the younger NASH population had a higher relative risk of MI. The relative risk of MI decreased with older age [40-49 (10.1, P < 0.0001), 50-59 (7.7, P < 0.0001), 60-69 (5.5, P < 0.0001), 70-79 (5.2, P < 0.0001) and age above 80 years (5.1, P < 0.0001)] (Figure 1).
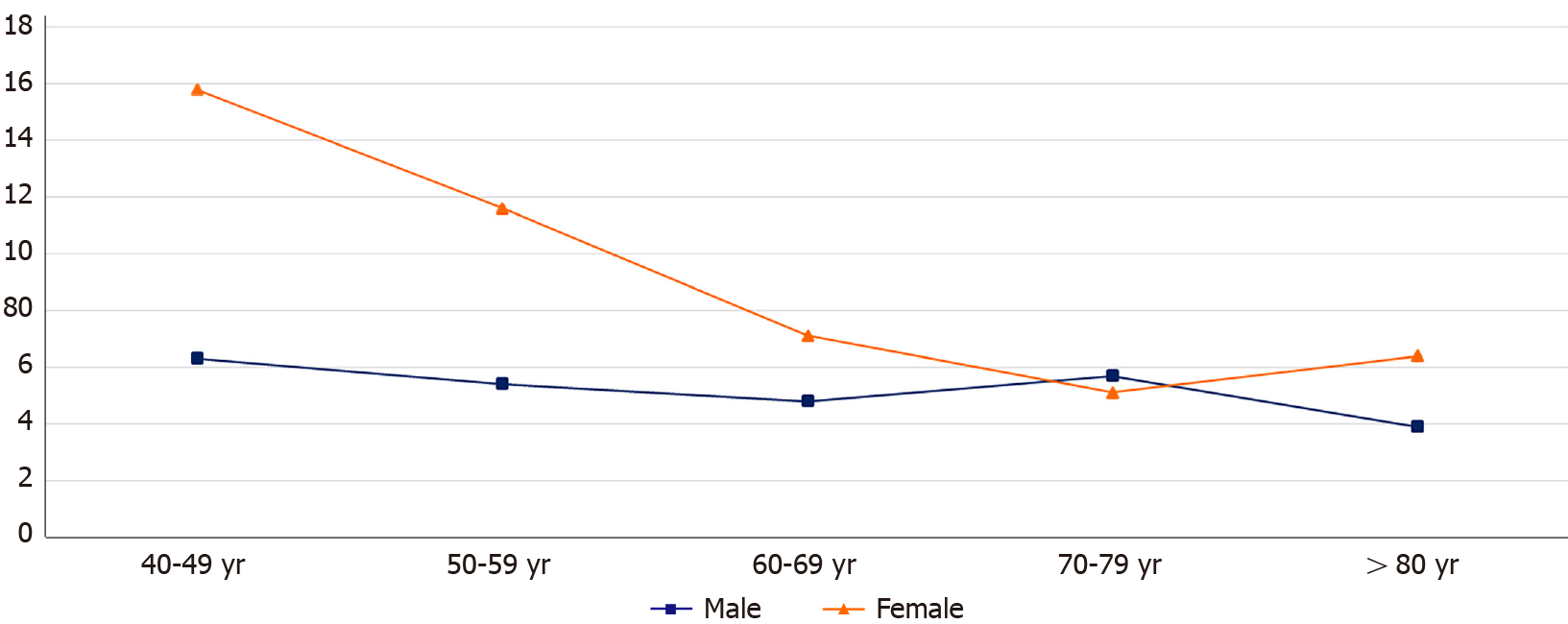
Gender differences in NASH contributed to the disparities observed in the relative risk of MI among each age group. Within the 40-49 years group, females had a higher risk of MI compared to their male counterparts (15.8 vs 6.3, P < 0.0001).
Compared to the younger NASH females, the older group had a lower relative risk of MI; while males with NASH maintained a relatively more stable relative risk of MI throughout their lifetime [age 50-59 (11.6 vs 5.4, P < 0.0001); age 60-69 (7.1 vs 4.8, P < 0.0001); age 70-79 (5.1 vs 5.7, P < 0.0001)] (Figure 2).
The absolute risk of MI was also higher in subjects with NASH than non-NASH and increased with age [40-49 (0.74 vs 0.07), 50-59 (1.4 vs 0.2), 60-69 (1.8 vs 0.3), age 70-79 (2.4 vs 0.5), and above 80 years (1.9 vs 0.4)] (Figure 1).
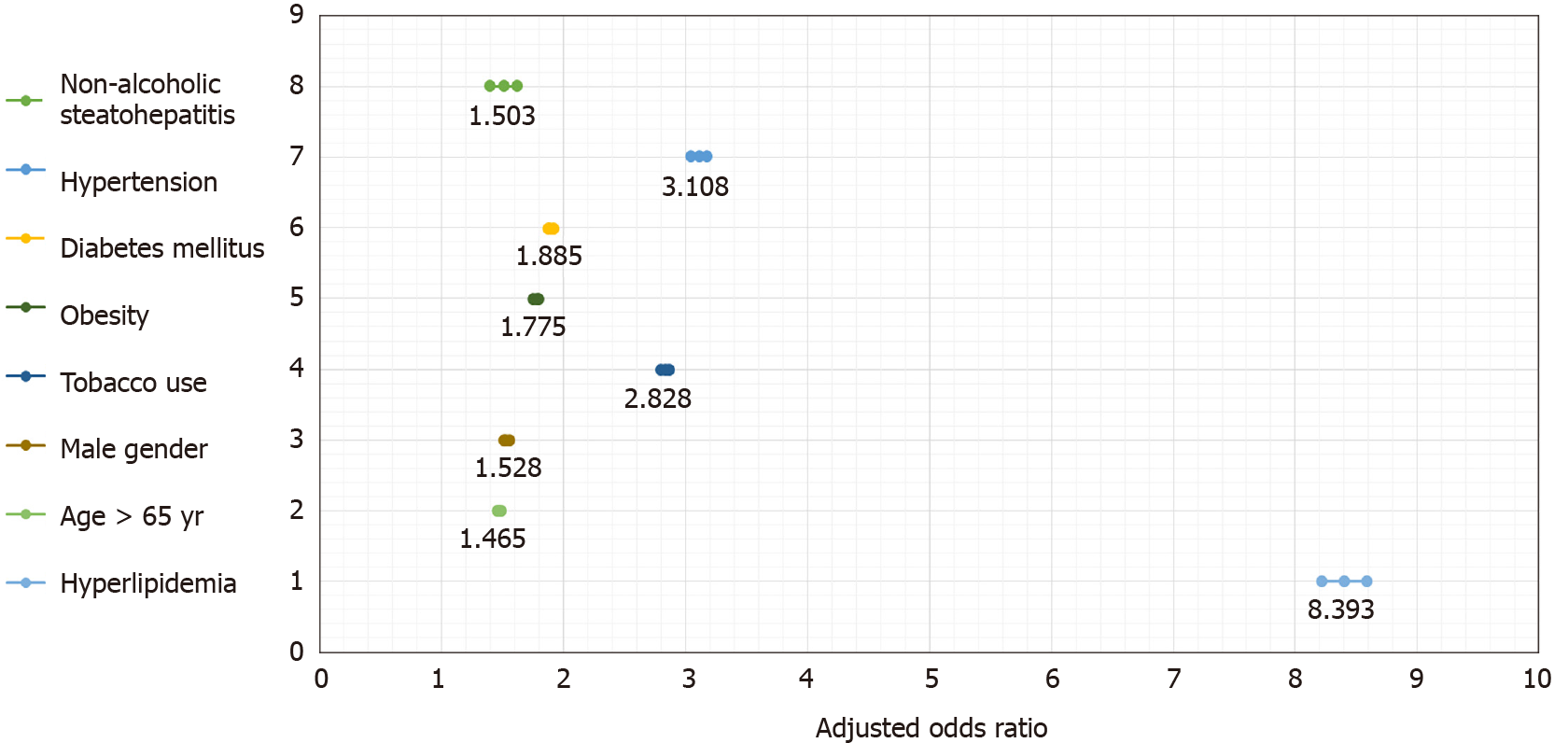
After adjusting for age, gender, smoking, hyperlipidemia, hypertension, diabetes and smoking, the diagnosis of NASH had a significant association with MI OR 1.5 (95%CI: 1.40-1.62) (Table 3).
| Odds ratio | 95%CIb | |
| Hyperlipidemia | 8.39 | 8.21-8.58 |
| Hypertension | 3.11 | 3.05-3.17 |
| Smoking | 2.83 | 2.79-2.87 |
| Diabetes mellitus | 1.89 | 1.86-1.91 |
| Obesity | 1.78 | 1.75-1.80 |
| Male | 1.53 | 1.51-1.55 |
| NASH | 1.5 | 1.40-1.62 |
| Age above 65 yr | 1.47 | 1.45-1.49 |
Hyperlipidemia had the strongest association with MI OR 8.39 (95%CI: 8.21-8.58) followed by hypertension OR 3.11 [95%CI: 3.05-3.17), and smoking OR 2.83 [95%CI: 2.79-2.87) (Figure 3). NASH had comparable association with MI when compared to traditional risk factors such as age above 65 OR 1.47 (95%CI: 1.45-1.49), male gender OR 1.53 (95%CI: 1.51-1.55) and diabetes mellitus OR 1.89 (95%CI: 1.86-1.91) (Table 3).
There is increasing evidence suggesting the relationship between NAFLD and CVD extends beyond risk factors common to both chronic conditions[22,23]. A number of population studies have also confirmed this association and have suggested that individuals with NAFLD should be screened for CVD. Other studies have shown that CVD risk increases with the severity of NAFLD[24,25]. Our study used a large population-based database to evaluate the relationship between NASH and MI in the United States. We included over 55 million patients in our study, of which 43170 were diagnosed with NASH and 107000 had an acute MI within 2018-2019.
The main findings of our study are: (1) NASH is associated with MI independent of traditional risk factors; (2) Compared to the older NASH population, younger patients were more likely to have a higher relative risk of MI; and (3) MI is a prevalent outcome among patients with NASH.
Although an association between ischemic heart disease and NAFLD has been suggested, the strength of this relationship remains controversial. In this study, NASH was independently associated with MI OR 1.5 (95%CI: 1.40-1.62) after adjusting for classically known risk factors for the latter. Our study is in line with other studies that have shown an association between NASH or NAFLD and ischemic heart disease[19,20].
At the pathophysiological level, this observation could be explained by several mechanisms. First, NASH is considered a more severe manifestation of NAFLD with augmented hepatic steatosis and aberrant intrahepatic inflammation[26]. Oxidative stress is thought to be the driving force of NASH propagating intra-hepatic proinflammatory cascades[13,27-30]. Free fatty acids themselves contribute to inflammatory mechanisms when they undergo esterification[31]. The liver is an important immune tissue containing the largest number of macrophages and other immune cells[32]. Hence, diseased liver will drain an increasing number of cytokines into systemic circulation and promote extrahepatic inflammation. Systemic cytokines and inflammatory markers are also associated with secondary cardiovascular effects[29,30]. Chronic inflammatory diseases such as rheumatoid arthritis and systemic lupus erythematosus have been associated with higher risks of atherosclerotic diseases and arterial thromboembolic events including MI[33,34]. A procoagulant imbalance exists and is worse in NASH[18]. Since NASH is a severe inflammatory subtype of NAFLD we hypothesize that NASH is an independent risk factor MI. Previous cross-sectional studies have grouped NASH and NAFLD as a single disease entity. We believe by doing so, the authors might have created a null effect and subsequently contributed to the heterogeneity in outcomes reported. Hence, further methodologically stringent studies with long-term follow up are needed to gain mechanistic insight into the pathology of NASH-MI axis.
NAFLD is part of a systemic disease with complex multidirectional relationships between MetS and CVD. The liver plays a crucial role in lipid and glucose metabolism. Even though dyslipidemia is an independent risk factor for CVD, abnormal lipid profiles are seen in NAFLD and are more deranged with increased histological inflammation[17]. A stepwise increase in lipid ratios correlates with histological severity of liver injury and is associated with increased risk of CVD and an atherogenic lipid profile[17]. Insulin resistance and alterations in lipid metabolism might also precede the development of hyperlipidemia and diabetes mellitus both of which are associated with increased risk of ischemic heart disease[11]. In our study, NASH was significantly associated with MI when these variables were partially controlled. Even though we observed an independent association between NASH and MI, to completely control these variables would blunt the contribution NASH might have on ischemic heart disease[19,20].
The prevalence of NASH is reported to be the greatest among subjects between the ages of 40-49 years[35]. It is conceivable to say that the presence of NASH in the younger population might increase their relative risk of MI compared to those without NASH (Figure 1). In our study the relative risk of MI was highest in the younger NASH population, suggesting increased inflammation and more aggressive disease in the younger group. Although age correlates with the duration of NAFLD, advanced disease may not be attributed to age alone. Furthermore, with aging, the non-NASH group might have accumulated traditional factors that may have reduced the relative contribution of inflammation on arthrosclerosis. Hence the contribution of NASH towards the total burden of cardiovascular risk maybe reduced with advancing age. This being said, we were not able to control for the duration or the severity of disease as this was an observational study. Nonetheless, we observed a step wise increase in the absolute risk of MI in both cohorts with increasing age (Figure 1).
There is increasing evidence that menopause is associated with the progression of NAFLD[36]. Although more data is needed, this is likely due the loss of estrogen’s hepato-protective role post menopause. In the current study, premenopausal females with NASH had a higher relative risk of MI compared to post-menopausal females (Figure 2). Our findings maybe partially explained by the lower CVD risk profile at baseline of the younger NASH females compared to their older counterparts. This supports the growing body of evidence suggesting a pathological association between inflammation and ischemic heart disease at least in the younger patient group[13,26-30].
Across almost all age groups, the risk of MI was higher in females compared to their male counterparts. The relative risk of MI in females with NASH decreased with increasing age, while males maintained a relatively more stable relative risk of MI throughout their lifetime (Figure 2). Current data suggests young women with MI are more likely than their male counterparts to have comorbidities[37]. Additionally, although the risk of MI on average is higher in men than women, the presence of comorbidities has been shown to confer an excess greater risk of MI in women than men and this risk doesn’t attenuate with age[37]. However given that data on time trends in risk factors was not available in Explorys, we do not know whether there were sex-related differences in risk factor levels and risk factor control before MI and our findings indicate further studies are needed to investigate the phenomenon reported in the current study.
An important strength of our study is that we used a large national database and reported data on over 55 million active adult subjects. We also provided comprehensive epidemiological information on the risk of MI in patients with NASH based on classical risk factors for both entities. Our results are therefore consistent with established data suggesting that NASH might be significantly associated with acute CVD events such as MI.
There are several limitations to this study that should be addressed. Observational studies preclude us from addressing causality. Due to the design of Explorys, we could not establish temporal relationships between the duration of NASH, severity of the disease and impact of interventions on the risk of cardiovascular events in our cohort. Second, the prevalence and natural disease progression of NAFLD is difficult to estimate as accurate diagnosis requires tissue analysis. The prevalence of NASH is estimated to be between 1.5%-6.45% based on a few biopsy series[38]. As this is a retrospective study reliant on diagnosed SNOMED-CT codes, it is impossible to verify the accuracy of diagnoses and it is prone to coding errors. The reported prevalence of NASH in our study was 0.08%. We excluded those with the diagnosis of fatty liver disease in an attempt to increase accuracy in our reporting; as the latter may be caused by drugs, viruses or alcohol. But even though the NASH population was underrepresented in the current study, MI was significantly associated with this disease entity.
An important limitation of our study is the validity of diagnosis of acute MI. We were unable to differentiate between MI secondary to coronary artery disease and MI resulting from demand-related events (Type II MI). Validation was also not possible as patient information in this platform is de-identified. Further, direct temporal relationships between NASH and MI cannot be defined due to the inherent design of Explorys. We acknowledged this limitation and included only those with a diagnosis of acute MI within the last year of the study to circumvent this issue. However, using a large national population-based sample allows for the ability to generalize our results to the United States population and offset these limitations.
In our large United States-based cohort study, we found acute MI to be a prevalent diagnosis among patients with NASH. Although our findings need to be further confirmed by prospective studies, we propose that patients with NAFLD at risk of NASH, should be identified early and screened for associated cardiovascular risk factors. Current AASLD-AGA guidelines recommend modification of CVD risk factors in patients with NAFLD[39]. Whereas, the European association for the study of liver view CVD as an extra-hepatic manifestation of NAFLD[40]. As we have mentioned above, severe inflammation, as seen in patients with NASH, is associated with accelerated atherosclerosis. A major problem that hampers screening for CVD in this subgroup is the unawareness of health care providers of the relevance of this disease, and as a consequence, NASH is under diagnosed in the general population. Even in our study, out of 55 million patients, only 43170 (0.08%) of patients had an ICD-coded diagnosis of NASH, despite a reported prevalence of NAFLD as high as 24%[35].
In conclusion, there is a significant relationship between NASH and acute MI. The use of already existing non-invasive scoring systems to stratify disease severity in NAFLD can overcome the need for liver biopsy and allow for early detection and aggressive risk factor modification.
Non-alcoholic fatty liver disease (NAFLD) is the most common chronic liver disease in the United States. The severity of NAFLD ranges from simple steatosis to non-alcoholic steatohepatitis which can progress to fibrosis, cirrhosis and hepatocellular carcinoma. Recent evidence suggests that the diagnosis of NAFLD may be associated with an increased risk of cardiovascular disease (CVD) independent of traditional risk factors. We believe that patients with non-alcoholic steatohepatitis (NASH) are at a higher risk of serious cardiovascular events such as myocardial infarction (MI).
There is an overlap between the risk factors that give rise to NAFLD and CVD. NASH remains underdiagnosed in the general population as tissue analysis is needed for accurate diagnosis. It is a more severe subtype of NAFLD associated with hepatic and systemic inflammation. Inflammation is implicated in the pathogenesis of atherosclerosis. Whether NASH is associated with serious cardiovascular events such as MI has major economic and public health implications.
The aim of this study was to assess the prevalence of acute MI among patients with NASH and to investigate the contribution of age and gender on the relative risk of MI in a large cohort of subjects in the United States.
This was a large retrospective study that included over 50 million patients from over 50 states in the United States. Patients diagnosed with NASH between 1999-2019 and those with acute MI within 2018-2019 were identified. Traditional risk factors associated with both diseases were also collected. Univariable and multivariable analyses were performed to assess the association between NASH and MI.
After adjusting for traditional risk factors, there was an independent associated between NASH and MI (1.5, 95%CI: 1.40-1.62, P < 0.0001). The relative risk of MI in patients with NASH appeared to be the highest in the younger patient population (< 49 years old, RR 10.1, P < 0.0001) suggesting inflammation might be the driving force for this observation. Women with NASH had a higher relative risk of MI compared to their male counterparts (15.8 vs 11.6, P < 0.0001, respectively). Overall, the absolute risk of MI was higher in the older population.
Acute MI is a prevalent diagnosis among patients with NASH. According to our dataset, NASH continues to be underdiagnosed in the United States population. Systemic inflammatory cascades are exaggerated in NASH and might be implicated in the pathogenesis of arthrosclerosis. Identification of NAFLD patients at high risk of NASH might allow for primary prevention and aggressive cardiovascular risk modification.
Performing large scale prospective studies with long-term follow-up are needed to gain mechanistic insight into the pathology of NASH-MI axis.
We gratefully acknowledge Dr. Joseph Sudano for ensuring the data analysis is appropriate for this research.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: United States
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Kim SE, Morozov S, Rezaee-Zavareh MS, Sirin G, Ta T S-Editor: Dou Y L-Editor: A E-Editor: Li X
| 1. | Sayiner M, Koenig A, Henry L, Younossi ZM. Epidemiology of Nonalcoholic Fatty Liver Disease and Nonalcoholic Steatohepatitis in the United States and the Rest of the World. Clin Liver Dis. 2016;20:205-214. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 323] [Cited by in RCA: 400] [Article Influence: 44.4] [Reference Citation Analysis (3)] |
| 2. | Kanwar P, Kowdley KV. The Metabolic Syndrome and Its Influence on Nonalcoholic Steatohepatitis. Clin Liver Dis. 2016;20:225-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 78] [Article Influence: 8.7] [Reference Citation Analysis (0)] |
| 3. | Williams CD, Stengel J, Asike MI, Torres DM, Shaw J, Contreras M, Landt CL, Harrison SA. Prevalence of nonalcoholic fatty liver disease and nonalcoholic steatohepatitis among a largely middle-aged population utilizing ultrasound and liver biopsy: a prospective study. Gastroenterology. 2011;140:124-131. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1522] [Cited by in RCA: 1619] [Article Influence: 115.6] [Reference Citation Analysis (1)] |
| 4. | Vernon G, Baranova A, Younossi ZM. Systematic review: the epidemiology and natural history of non-alcoholic fatty liver disease and non-alcoholic steatohepatitis in adults. Aliment Pharmacol Ther. 2011;34:274-285. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2405] [Cited by in RCA: 2292] [Article Influence: 163.7] [Reference Citation Analysis (0)] |
| 5. | Lazo M, Hernaez R, Eberhardt MS, Bonekamp S, Kamel I, Guallar E, Koteish A, Brancati FL, Clark JM. Prevalence of nonalcoholic fatty liver disease in the United States: the Third National Health and Nutrition Examination Survey, 1988-1994. Am J Epidemiol. 2013;178:38-45. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 633] [Cited by in RCA: 625] [Article Influence: 52.1] [Reference Citation Analysis (1)] |
| 6. | Dixon JB, Bhathal PS, O'Brien PE. Nonalcoholic fatty liver disease: predictors of nonalcoholic steatohepatitis and liver fibrosis in the severely obese. Gastroenterology. 2001;121:91-100. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 941] [Cited by in RCA: 901] [Article Influence: 37.5] [Reference Citation Analysis (0)] |
| 7. | Ekstedt M, Hagström H, Nasr P, Fredrikson M, Stål P, Kechagias S, Hultcrantz R. Fibrosis stage is the strongest predictor for disease-specific mortality in NAFLD after up to 33 years of follow-up. Hepatology. 2015;61:1547-1554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1353] [Cited by in RCA: 1703] [Article Influence: 170.3] [Reference Citation Analysis (1)] |
| 8. | Söderberg C, Stål P, Askling J, Glaumann H, Lindberg G, Marmur J, Hultcrantz R. Decreased survival of subjects with elevated liver function tests during a 28-year follow-up. Hepatology. 2010;51:595-602. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 529] [Cited by in RCA: 561] [Article Influence: 37.4] [Reference Citation Analysis (0)] |
| 9. | Angulo P, Kleiner DE, Dam-Larsen S, Adams LA, Bjornsson ES, Charatcharoenwitthaya P, Mills PR, Keach JC, Lafferty HD, Stahler A, Haflidadottir S, Bendtsen F. Liver Fibrosis, but No Other Histologic Features, Is Associated With Long-term Outcomes of Patients With Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015;149:389-97.e10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2304] [Cited by in RCA: 2229] [Article Influence: 222.9] [Reference Citation Analysis (0)] |
| 10. | Salvi P, Ruffini R, Agnoletti D, Magnani E, Pagliarani G, Comandini G, Praticò A, Borghi C, Benetos A, Pazzi P. Increased arterial stiffness in nonalcoholic fatty liver disease: the Cardio-GOOSE study. J Hypertens. 2010;28:1699-1707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 98] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 11. | Mellinger JL, Pencina KM, Massaro JM, Hoffmann U, Seshadri S, Fox CS, O'Donnell CJ, Speliotes EK. Hepatic steatosis and cardiovascular disease outcomes: An analysis of the Framingham Heart Study. J Hepatol. 2015;63:470-476. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 131] [Cited by in RCA: 171] [Article Influence: 17.1] [Reference Citation Analysis (0)] |
| 12. | Lee YJ, Shim JY, Moon BS, Shin YH, Jung DH, Lee JH, Lee HR. The relationship between arterial stiffness and nonalcoholic fatty liver disease. Dig Dis Sci. 2012;57:196-203. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 49] [Cited by in RCA: 61] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 13. | Sookoian S, Pirola CJ. Non-alcoholic fatty liver disease is strongly associated with carotid atherosclerosis: a systematic review. J Hepatol. 2008;49:600-607. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 277] [Cited by in RCA: 306] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 14. | Ong JP, Pitts A, Younossi ZM. Increased overall mortality and liver-related mortality in non-alcoholic fatty liver disease. J Hepatol. 2008;49:608-612. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 529] [Cited by in RCA: 504] [Article Influence: 29.6] [Reference Citation Analysis (0)] |
| 15. | Villanova N, Moscatiello S, Ramilli S, Bugianesi E, Magalotti D, Vanni E, Zoli M, Marchesini G. Endothelial dysfunction and cardiovascular risk profile in nonalcoholic fatty liver disease. Hepatology. 2005;42:473-480. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 443] [Cited by in RCA: 473] [Article Influence: 23.7] [Reference Citation Analysis (0)] |
| 16. | Sookoian S, Gianotti TF, Rosselli MS, Burgueño AL, Castaño GO, Pirola CJ. Liver transcriptional profile of atherosclerosis-related genes in human nonalcoholic fatty liver disease. Atherosclerosis. 2011;218:378-385. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 79] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 17. | Alkhouri N, Tamimi TA, Yerian L, Lopez R, Zein NN, Feldstein AE. The inflamed liver and atherosclerosis: a link between histologic severity of nonalcoholic fatty liver disease and increased cardiovascular risk. Dig Dis Sci. 2010;55:2644-2650. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 80] [Cited by in RCA: 85] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 18. | Verrijken A, Francque S, Mertens I, Prawitt J, Caron S, Hubens G, Van Marck E, Staels B, Michielsen P, Van Gaal L. Prothrombotic factors in histologically proven nonalcoholic fatty liver disease and nonalcoholic steatohepatitis. Hepatology. 2014;59:121-129. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 145] [Article Influence: 13.2] [Reference Citation Analysis (0)] |
| 19. | Targher G, Byrne CD, Lonardo A, Zoppini G, Barbui C. Non-alcoholic fatty liver disease and risk of incident cardiovascular disease: A meta-analysis. J Hepatol. 2016;65:589-600. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1049] [Cited by in RCA: 1003] [Article Influence: 111.4] [Reference Citation Analysis (0)] |
| 20. | Baratta F, Pastori D, Angelico F, Balla A, Paganini AM, Cocomello N, Ferro D, Violi F, Sanyal AJ, Del Ben M. Nonalcoholic Fatty Liver Disease and Fibrosis Associated With Increased Risk of Cardiovascular Events in a Prospective Study. Clin Gastroenterol Hepatol. 2019;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 171] [Cited by in RCA: 156] [Article Influence: 31.2] [Reference Citation Analysis (0)] |
| 21. | IBM Explorys-Accelerating Insights from Real-World Evidence. 2019. Available from: http://www-01.ibm.com/common/ssi/cgi-bin/ssialias?htmlfid=HL912350U-SEN. |
| 22. | Kaelber DC, Foster W, Gilder J, Love TE, Jain AK. Patient characteristics associated with venous thromboembolic events: a cohort study using pooled electronic health record data. J Am Med Inform Assoc. 2012;19:965-972. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 107] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 23. | Satapathy SK, Sanyal AJ. Epidemiology and Natural History of Nonalcoholic Fatty Liver Disease. Semin Liver Dis. 2015;35:221-235. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 266] [Cited by in RCA: 247] [Article Influence: 24.7] [Reference Citation Analysis (0)] |
| 24. | Perseghin G. The role of non-alcoholic fatty liver disease in cardiovascular disease. Dig Dis. 2010;28:210-213. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 43] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 25. | Sung KC, Ryan MC, Wilson AM. The severity of nonalcoholic fatty liver disease is associated with increased cardiovascular risk in a large cohort of non-obese Asian subjects. Atherosclerosis. 2009;203:581-586. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 93] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 26. | Targher G, Day CP, Bonora E. Risk of cardiovascular disease in patients with nonalcoholic fatty liver disease. N Engl J Med. 2010;363:1341-1350. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1326] [Cited by in RCA: 1485] [Article Influence: 99.0] [Reference Citation Analysis (0)] |
| 27. | Chalasani N, Deeg MA, Crabb DW. Systemic levels of lipid peroxidation and its metabolic and dietary correlates in patients with nonalcoholic steatohepatitis. Am J Gastroenterol. 2004;99:1497-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 252] [Cited by in RCA: 260] [Article Influence: 12.4] [Reference Citation Analysis (0)] |
| 28. | Stoner L, Lucero AA, Palmer BR, Jones LM, Young JM, Faulkner J. Inflammatory biomarkers for predicting cardiovascular disease. Clin Biochem. 2013;46:1353-1371. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 100] [Cited by in RCA: 134] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 29. | Wieckowska A, Papouchado BG, Li Z, Lopez R, Zein NN, Feldstein AE. Increased hepatic and circulating interleukin-6 levels in human nonalcoholic steatohepatitis. Am J Gastroenterol. 2008;103:1372-1379. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 395] [Cited by in RCA: 442] [Article Influence: 26.0] [Reference Citation Analysis (0)] |
| 30. | Hamirani YS, Katz R, Nasir K, Zeb I, Blaha MJ, Blumenthal RS, Kronmal RN, Budoff MJ. Association between inflammatory markers and liver fat: The Multi-Ethnic Study of Atherosclerosis. J Clin Exp Cardiolog. 2014;5. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 38] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 31. | Neuschwander-Tetri BA. Hepatic lipotoxicity and the pathogenesis of nonalcoholic steatohepatitis: the central role of nontriglyceride fatty acid metabolites. Hepatology. 2010;52:774-788. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 726] [Cited by in RCA: 840] [Article Influence: 56.0] [Reference Citation Analysis (0)] |
| 32. | Racanelli V, Rehermann B. The liver as an immunological organ. Hepatology. 2006;43:S54-S62. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 870] [Cited by in RCA: 967] [Article Influence: 50.9] [Reference Citation Analysis (0)] |
| 33. | Solomon DH, Reed GW, Kremer JM, Curtis JR, Farkouh ME, Harrold LR, Hochberg MC, Tsao P, Greenberg JD. Disease activity in rheumatoid arthritis and the risk of cardiovascular events. Arthritis Rheumatol. 2015;67:1449-1455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 239] [Article Influence: 23.9] [Reference Citation Analysis (0)] |
| 34. | Manzi S, Meilahn EN, Rairie JE, Conte CG, Medsger TA, Jansen-McWilliams L, D'Agostino RB, Kuller LH. Age-specific incidence rates of myocardial infarction and angina in women with systemic lupus erythematosus: comparison with the Framingham Study. Am J Epidemiol. 1997;145:408-415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1280] [Cited by in RCA: 1271] [Article Influence: 45.4] [Reference Citation Analysis (0)] |
| 35. | Sanyal AJ; American Gastroenterological Association. AGA technical review on nonalcoholic fatty liver disease. Gastroenterology. 2002;123:1705-1725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 759] [Cited by in RCA: 781] [Article Influence: 34.0] [Reference Citation Analysis (0)] |
| 36. | Venetsanaki V, Polyzos SA. Menopause and Non-Alcoholic Fatty Liver Disease: A Review Focusing on Therapeutic Perspectives. Curr Vasc Pharmacol. 2019;17:546-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 31] [Cited by in RCA: 59] [Article Influence: 11.8] [Reference Citation Analysis (0)] |
| 37. | Arora S, Stouffer GA, Kucharska-Newton AM, Qamar A, Vaduganathan M, Pandey A, Porterfield D, Blankstein R, Rosamond WD, Bhatt DL, Caughey MC. Twenty Year Trends and Sex Differences in Young Adults Hospitalized With Acute Myocardial Infarction. Circulation. 2019;139:1047-1056. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 438] [Cited by in RCA: 488] [Article Influence: 81.3] [Reference Citation Analysis (0)] |
| 38. | Younossi ZM. The epidemiology of nonalcoholic steatohepatitis. Clin Liver Dis (Hoboken). 2018;11:92-94. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 66] [Article Influence: 9.4] [Reference Citation Analysis (0)] |
| 39. | Chalasani N, Younossi Z, Lavine JE, Charlton M, Cusi K, Rinella M, Harrison SA, Brunt EM, Sanyal AJ. The diagnosis and management of nonalcoholic fatty liver disease: Practice guidance from the American Association for the Study of Liver Diseases. Hepatology. 2018;67:328-357. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3544] [Cited by in RCA: 4946] [Article Influence: 706.6] [Reference Citation Analysis (9)] |
| 40. | Ratziu V, Bellentani S, Cortez-Pinto H, Day C, Marchesini G. A position statement on NAFLD/NASH based on the EASL 2009 special conference. J Hepatol. 2010;53:372-384. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 723] [Cited by in RCA: 789] [Article Influence: 52.6] [Reference Citation Analysis (1)] |