Published online Oct 27, 2020. doi: 10.4254/wjh.v12.i10.816
Peer-review started: May 25, 2020
First decision: June 12, 2020
Revised: June 26, 2020
Accepted: August 16, 2020
Article in press: August 16, 2020
Published online: October 27, 2020
Processing time: 151 Days and 21.6 Hours
Pyogenic liver abscess (PLA) is a rare disease with an estimated incidence that varies widely across the globe, being as high as 115.4/100000 habitants in Taiwan and as low as 1.1-1.2/100000 habitants in Europe and Canada. Even though there are multiple microorganisms capable of producing an abscess in the liver, including Entamoeba histolytica, fungi, and viruses, most abscesses are derived from bacterial infections. The epidemiology of PLA in Mexico is currently unknown.
To describe the clinical, demographic and microbiologic characteristics of PLA in Mexico.
This is a retrospective study carried out in two centers, and included patients seen between 2006 and 2018 with the diagnosis of pyogenic abscess. We collected demographic, clinical, and microbiological information, treatment, complications, and outcomes. A logistic regression analysis was used to determine the association between different variables and mortality rates.
A total of 345 patients were included in this study. 233 (67.5%) had confirmed PLA, 133 (30%) patients had no positive culture and negative serology and 9 (2.5%) had mixed abscesses. The mean age was 50 years (ranging from 16-97 years) and 63% were female. 65% of the patients had positive cultures for Extended Spectrum Beta-Lactamases (ESBL)-Escherichia coli and Klebsiella pneumoniae. Cefotaxime was administered in 60% of cases. The most common sources of infection were ascending cholangitis and cholecystitis in 34 (10%) and 31 (9%), respectively. The median length of hospital stay was 14 d. 165 patients underwent percutaneous catheter drainage. The inpatient mortality rate was 63%. Immunocompromised state [OR 3.9, 95%CI: 1.42-10.46], ESBL- Escherichia coli [OR 6.7, 95%CI: 2.7-16.2] and Klebsiella pneumoniae [OR 4-8, 95%CI: 1.6-14.4] predicted inpatient mortality by multivariate analysis.
The prevalence of PLA is increasing in Mexico and has a very high mortality rate. ESBL-Escherichia coli and Klebsiella pneumoniae are the most common microorganisms causing PLA and are independent predictors of inpatient mortality.
Core Tip: In this retrospective study we investigated the clinical, demographic, and microbiologic characteristics of pyogenic liver abscess (PLA) in Mexico. We found that the prevalence of PLA in Mexico is increasing and had a very high mortality rate (63%) in our study. Our data also indicated that the presence of ESBL-Escherichia coli and Klebsiella pneumoniae and an immunocompromised state were independent predictors of high-risk mortality with an adjusted OR of 6.7, 4.8 and 3.9, respectively.
- Citation: Pérez-Escobar J, Ramirez-Quesada W, Calle-Rodas DA, Chi-Cervera LA, Navarro-Alvarez N, Aquino-Matus J, Ramírez-Hinojosa JP, Moctezuma-Velázquez C, Torre A. Increased incidence of and microbiologic changes in pyogenic liver abscesses in the Mexican population. World J Hepatol 2020; 12(10): 816-828
- URL: https://www.wjgnet.com/1948-5182/full/v12/i10/816.htm
- DOI: https://dx.doi.org/10.4254/wjh.v12.i10.816
Although there are multiple microorganisms capable of producing an abscess in the liver, including Entamoeba histolytica, fungi, and viruses, most abscesses are derived from bacterial infections[1,2]. Pyogenic liver abscess (PLA) is a rare disease with an estimated incidence that varies widely across the globe, being as high as 115.4/100000 inhabitants in Taiwan and as low as 1.1-1.2/100000 inhabitants in Europe and Canada[1,2].
There has been a shift in the etiology of PLAs over the years. Previously, intraabdominal infections such as complicated appendicitis or diverticulitis with portal bacterial seeding have represented the most frequent causes[1], whereas currently, biliary tree diseases such as cholecystitis, choledocholithiasis, tumoral obstruction, strictures, congenital malformations, and hepatobiliary instrumentation are the most common etiologies[1-3]. With regard to bacterial isolates, before 1980, the most commonly reported agents were Escherichia coli, Enterobacteriaceae, anaerobes, and other members of the intestinal flora[1,2,4,5], but in recent years, some highly virulent strains of Klebsiella pneumoniae have been isolated in Taiwan and mainly in Singapore[6], where many series have been reported. There are also new cases in the United States and Europe[1].
In Mexico, the epidemiology of PLAs is unknown, with some small case series reporting up to 41 cases from all over the country with a predominance of Escherichia coli and a lower reported incidence[7]. The aim of this study was to describe the current clinical, demographic, and microbiologic characteristics of PLAs in two high-volume centers in Mexico.
This was a retrospective study that included patients who were seen between 2006 and 2018 at two referral centers in Mexico City: The Instituto Nacional de Ciencias Médicas y Nutrición Salvador Zubirán (INCMNSZ) and the Hospital General Dr. Manuel Gea González. Eligible patients were those with a diagnosis of liver abscess by imaging, and individuals with an unspecified type of abscess with negative amoeba serology, a mixed-type abscess defined by positive bacterial culture and positive amoeba serology and positive bacterial culture-confirmed PLAs were included. We excluded patients aged < 18 years and those with incomplete information; with confirmed amoebic abscesses, defined as those with positive serology and an absence of positive bacterial cultures; those in whom Entamoeba histolytica was isolated; and those in whom fungi were isolated. Patients with infected liver cysts and infected hydatid cysts were also excluded.
We collected demographic, clinical, and microbiological information from each patient's medical chart, including age, sex, symptoms, laboratory results, radiological characteristics, microbiological isolates, type of treatment, complications, length of hospital stay, and outcomes. Due to small numbers of specific cases, immunocompromise was defined as the presence of positive human immunodeficiency virus (HIV) status, underlying malignancy, or the presence of autoimmune disease. For the statistical analysis, data are presented as the means ± SD or frequencies and percentages, according to their distribution. Numeric and categorical variables were compared using the chi-squared test. To study the association between the different variables and mortality rates, we used a logistic regression model. Variables of interest plus variables with a P value less than 0.1 in univariate analysis were included in a multivariate regression analysis. Logistic regression using significant variables was applied with the Omnibus test and Hosmer-Lemeshow test to explain mortality (utilizing R2 Nagelkerke). Analyses were conducted with Stata version 12 (Stata Corp. LP, College Station, TX, United States).
All patients underwent abdominal ultrasound, computed tomography (CT) scanning or magnetic resonance imaging (MRI) studies. Patients with PLAs underwent at least one set of blood cultures before the administration of parenteral empiric broad-spectrum antibiotic agents in accordance with local antimicrobial resistance surveillance data.
The duration of parenteral antibiotic agents, the switch to oral formulations, and the total duration of therapy were decided by the individual physician and guided by clinical, biochemical, and radiologic responses.
The decision on percutaneous catheter drainage (PCD) depended on the clinical response to medical treatment and the size of the PLA. PCD was performed using CT guidance by interventional radiologists via the placement of a 10–12F pigtail catheter in situ. PCD was performed when any of the following criteria were met: (1) Size of the PLA > 4 cm, solitary or dominant; (2) Hemodynamic instability or the need for inotropic support upon admission; and (3) Failure of antibiotic therapy for PLA.
A giant PLA (GPLA) was defined as an abscess greater than or equal to 10 cm in diameter.
A multiloculated abscess was defined as an abscess with 2 or more septations within its cavity.
Nonresponse to interventional radiology-assisted drainage was defined as patients having persistent signs of sepsis after 5 d of intravenous (IV) antibiotics and PCD, thus requiring escalation of therapy.
Percutaneous drainage failure; patients with ongoing sepsis after antibiotics and PCD or patients with difficult-to-access sites such as the liver dome; abscesses not amenable to percutaneous drainage, such as multiloculated abscesses or those with thick purulent material that could not be aspirated; ruptured abscesses; and underlying causes requiring surgical removal, such as in the case of a foreign body.
Within the database, 404 patients were documented between both centers: 175 in the General Hospital and 229 in the INCMNSZ. Of these patients, 58 had amebic abscesses confirmed by Entamoeba histolytica serology, and only 1 patient developed a fungal abscess secondary to Candida albicans; those patients were excluded from the study, and a total of 345 patients were included.
We included 345 patients: 233 (67.5%) had a confirmed PLA, 133 (30%) were patients with no positive cultures and negative serology, and 9 (2.6%) had mixed abscesses. The mean age was 50 years (ranging from 16-97 years), and 217 patients (63%) were female. The initial presentation included fever, fatigue, right upper quadrant pain, and diarrhea in 293 (85%), 167 (48%), 259 (75%), and 52 (15%) patients, respectively. The mean time between the onset of symptoms and medical consultation was 22 d. The most frequent comorbidities were type 2 diabetes mellitus (T2DM), hypertension, underlying malignancy, and biliary tree lesions, which were found in 87 (25%), 32 (9.2%), 21 (6%), and 16 (4.6%) patients, respectively.
The most relevant laboratory findings were leukocytosis with an elevated neutrophil count, and among the liver biochemistry alterations, a mean total bilirubin level of 2.24 mg/dL and a mean alkaline phosphatase level of 256.37 U/L were observed; the rest of the clinical and analytical data are summarized in Table 1.
| BMI mean ± SD | 27 ± 5 |
| BMI classification | Patients, n (%) |
| Normal | 140 (41) |
| Overweight | 141 (41) |
| Obesity class 1 | 43 (12) |
| Obesity class 2 | 12 (3) |
| Obesity class 3 | 9 (3) |
| Sign/Symptom | |
| Fever | 293 (85) |
| Right upper quadrant pain | 259 (75) |
| Fatigue | 167 (48) |
| Diarrhea | 52 (15) |
| Laboratory findings | mean ± SD |
| Hemoglobin, g/dL | 11.7 ± 2.48 |
| Leucocyte count, × 103/µL | 15.52 ± 10.03 |
| Neutrophil percentage, % | 48.48 ± 31.89 |
| Lymphocyte percentage, % | 9.73 ± 11.46 |
| Platelet count, K/µL | 315 ± 180.4 |
| INR | 1.27 ± 0.51 |
| Glucose, mg/dL | 130 ± 68.12 |
| Creatinine, mg/dL | 1.14 ± 0.83 |
| Sodium, mmol/L | 132.3 ± 17.45 |
| Total bilirubin, mg/dL | 2.24 ± 2.91 |
| Albumin, g/dL | 2.63 ± 2.05 |
| AST, U/L | 59.3 ± 73.23 |
| ALT, U/L | 57.6 ± 82.09 |
| GGT, U/L | 133.5 ± 208.43 |
| AP, U/L | 256.37 ± 219.30 |
| Lesion size | n (%) |
| < 5 cm | 54 (16) |
| 5-10 cm | 138 (40) |
| > 10 cm | 28 (8) |
| < 500 mL | 77 (22) |
| ≥ 500 mL | 48 (14) |
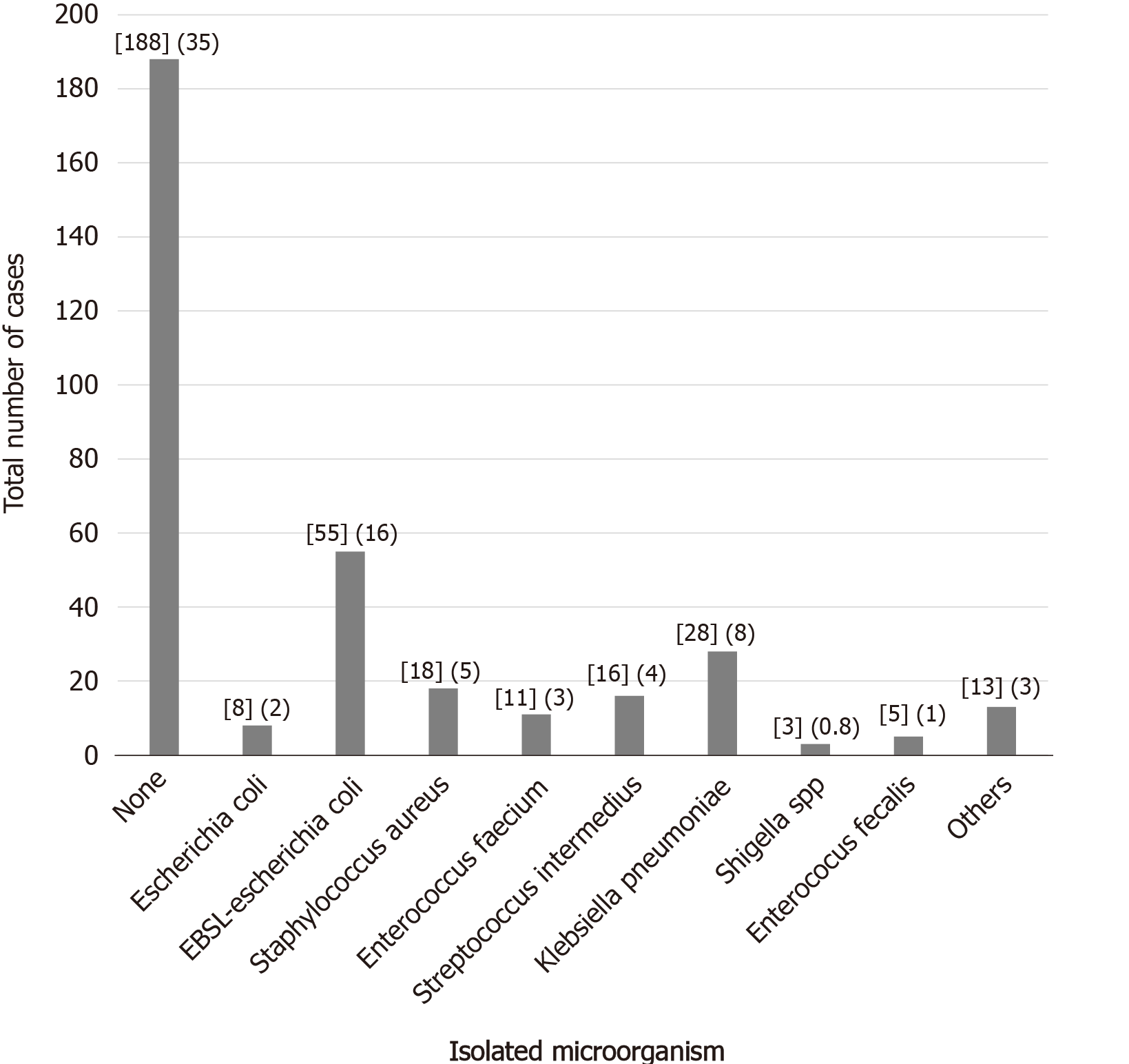
Abscess samples were obtained from direct-needle aspiration; 65% of the patients had positive cultures, and the most commonly isolated microorganisms were ESBL-Escherichia coli and Klebsiella pneumoniae, corresponding to 16% and 8%, respectively. In 59 cases, the culture isolated 2 different bacteria, and in 50 cases, more than two microorganisms were isolated. Figure 1 shows the frequency of the different bacteria cultured.
The antibiotic therapy administered varied considerably among the patients, with different approaches for each case; initial empiric treatment included cefotaxime in 60% cases, and in 90% of cases, it included a drug combination, most commonly with metronidazole. Interestingly, more than 90% of the cases required a combination with more than 1 drug; again, the previously mentioned third-generation cephalosporin was the most commonly used drug. Of those patients exposed to multiple regimens, at least 6 of them were exposed to six drug regimens, and 2 of them needed a seventh drug, of which vancomycin and cefuroxime were prescribed. In 82% of the cases, these regimens lasted for more than 14 d, with a median prescription time of 19 d (2-65 d).
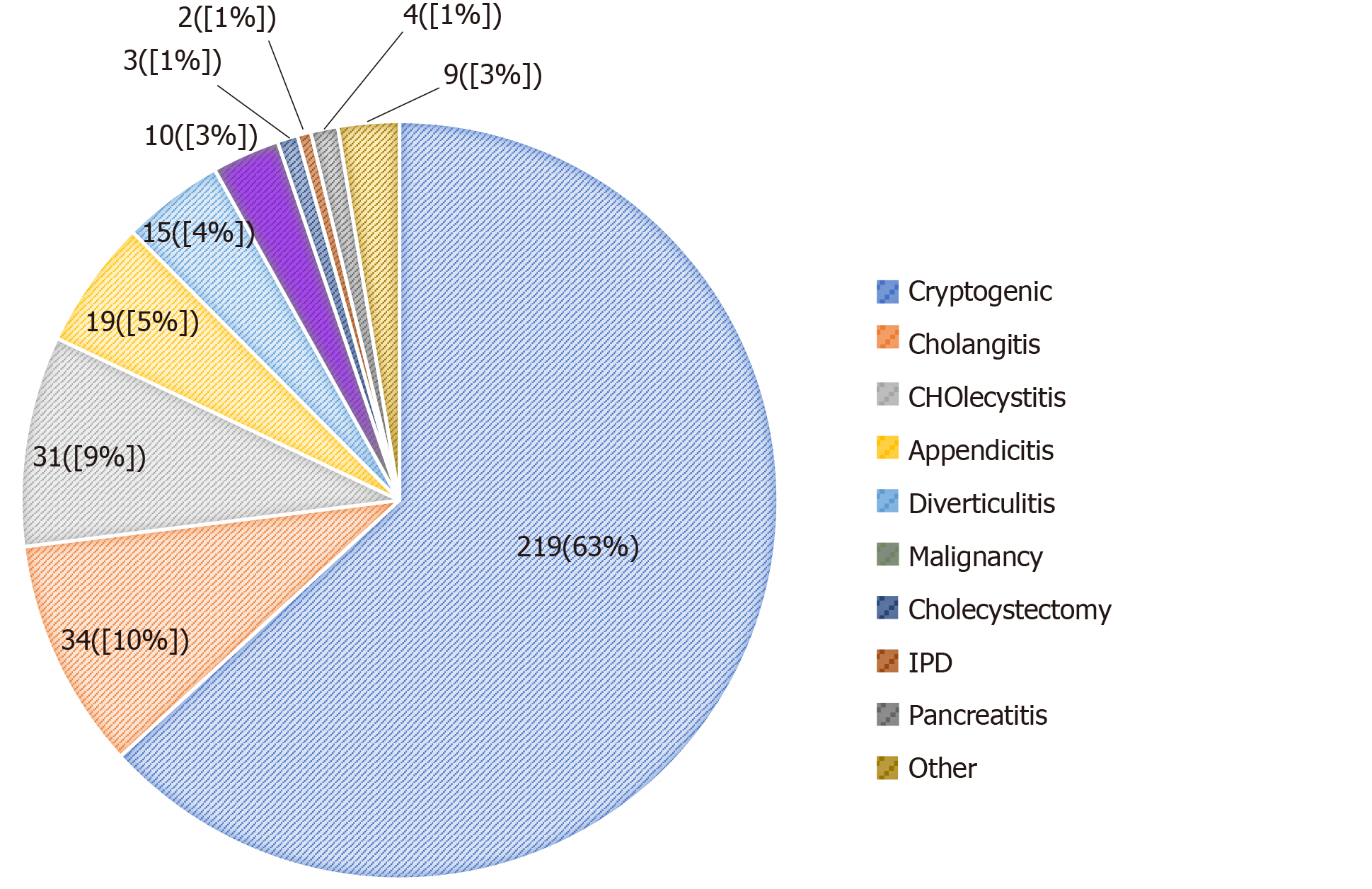
The most common causes were ascending cholangitis and cholecystitis in 34 (10%) and 31 (9%) patients, respectively, but in 40% of cases, no precipitating event was identified. Figure 2 shows the distribution of the different sources of infection with ascending cholangitis and cholecystitis, both being the most common precipitating events.
A total of 50.72% (n = 175) of the studied population had an abdominal ultrasound that initially suggested a diagnosis of PLA; for morphological diagnosis, a CT scan was performed in 320 patients, and MRI was carried out in just 14% of the cases for diagnosis confirmation.
The number of abscesses per case was quantified with the help of imaging techniques. In 51% of the cases (n = 178) only one documentable lesion was observed, 25% had at least 2 abscesses, and in 9% multiple abscesses in the liver parenchyma were identified.
86.7% of the lesions were located in the right lobe of the liver with segments VII, VI, and VIII being the most frequently affected in 32%, 28%, and 22% of the cases, respectively.
The median length of hospital stay was 14 d (1-53 d), 45 patients required intensive care unit admission with a median stay of 1 d (1-30 d), and most patients required regular inpatient beds for a median of 13 d (1-53 d).
In terms of source control, 165 patients underwent PCD, 64 underwent percutaneous needle aspiration (PNA), 48 underwent open drainage (OD), and 32 underwent laparoscopic drainage (LD). Twenty-four patients required surgical therapy after an incomplete response to interventional radiology-assisted drainage. Of the 345 patients, at least 66% (n = 229) required interventional radiology therapy. The two types of registered procedures were PNA and PCD. A total of 72% patients underwent PCD, and the remaining 28% of patients received PNA.
The other type of documented procedure was surgical OD or LD of the abscess. Only 22% of patients demanded a surgical intervention, of whom 48 (60%) corresponded to OD and the remaining 40% to LD. Fifty-five of those patients underwent direct surgical therapy, and only 24 who were previously exposed to interventional radiology procedures still required surgery.
A total of 120 (35%) patients had complications during their hospitalization; the most frequent complications were septic shock in 50 patients and sepsis in 18 (Third International Consensus Definitions for Sepsis and Septic Shock)[8]. Less frequent complications were biliary fistula, ruptured abscess, pneumonia, empyema, and myocardial infarction.
The inpatient mortality rate attributed to the abscess or its complications was 63%. In univariate analysis, variables associated with mortality were age, body mass index (BMI), T2DM, the presence of immunosuppression, ESBL-Escherichia coli, and septic shock, as shown in Table 2. In multivariate analysis, age and BMI were not significant, but the presence of immunocompromise, ESBL-Escherichia coli, and Klebsiella pneumoniae explained 15.8% of the mortality, with ESBL-Escherichia coli being the most critical component (adjusted OR 6.7), as shown in Table 3.
| High-risk variable | Univariate | ||||
| Dead | Alive | Total | OR (95%CI) | P value | |
| Age | |||||
| 52.7 ± 16.7 | 48.7 ± 16.9 | 0.02 | |||
| BMI | |||||
| 26.2 ± 4.4 | 27.4 ± 5.3 | 0.017 | |||
| T2DM2 | |||||
| Yes | 52 | 35 | 87 | 0.8 | 0.48 |
| No | 165 | 93 | 258 | ||
| Immunocompromise1 | |||||
| Yes | 30 | 5 | 35 | 3.9 | < 0.01 |
| No | 187 | 123 | 310 | ||
| E. coli | |||||
| Yes | 3 | 5 | 8 | 0.3 | 0.13 |
| No | 214 | 123 | 337 | ||
| ESBL2-Escherichia coli | |||||
| Yes | 49 | 6 | 55 | 5.9 | < 0.01 |
| No | 168 | 122 | 290 | ||
| Klebsiella pneumoniae | |||||
| Yes | 24 | 4 | 28 | 3.9 | < 0.01 |
| No | 193 | 124 | 317 | ||
| Septic shock | |||||
| Yes | 31 | 19 | 50 | 1 | < 0.89 |
| No | 186 | 109 | 295 | ||
| Total | 217 | 128 | 345 | ||
Repeat radiologic imaging (ultrasonography or CT) was performed for all patients at two- to three-week intervals to evaluate the response to treatment. Parenteral antibiotic agents were converted to oral formulations based on clinical progress and were guided by bacteriology results and down-trending inflammatory markers. Antibiotic agents were discontinued at clinical and near-radiologic resolution (absence or significant reduction in size of abscess on imaging).
In our study, the median age of the patients was 51 years, and the male to female ratio was 1:1.7, with a female predominance. Among the reported cases from the INCMNSZ by Téllez-Zenteno, the mean age was 52 ± 14 years, which was similar to the age in our series, but an inverse result in the sex ratio was observed, with a male predominance[7]. On the other hand, European series, such as the one published by Serraino et al[5], reported an average age of 65.4 years with a male predominance. According to the world literature, the peak incidence of PLA is found in the same age groups as those in our study but with a typical male predominance[1].
A total of 85% of our patients reported fever during their initial presentation, followed by 48% with right upper quadrant pain, both of which are the most commonly documented signs and symptoms in many of the world case series and in our previous series[5-10], but the clinical picture shows many nonspecific findings, such as jaundice, pleural effusion, anorexia, nausea and vomiting[1,2].
As previously mentioned, the etiology of PLA has suffered a shift from the expansion of intraabdominal infection to biliary tree-derived diseases. Nonetheless, 63% of the cases did not have any documentable cause of their PLA on their medical records after evaluating the pathological history, trauma, and possible postoperative complications; performing CT and cholangioresonance to evaluate the biliary tree as well as endoscopic retrograde cholangiopancreatography in the indicated cases; and searching for bacteremia from any source of sepsis and foreign bodies.
According to the series by Téllez-Zenteno, 53% of patients had an unidentified source of infection in contrast to most recent series, which report between 25% and 40% of cryptogenic PLA cases[5,7,11,12]. The reason for the high cryptogenic PLA count is not apparent but might be derived from previous exposure to antibiotics, delayed medical attention, and increased prevalence of risk factors such as T2DM regardless of this phenomenon seen worldwide[13,14]. Among the patients with an underlying cause, both cholangitis and cholecystitis accounted for 19% of the cases, and other series have reported similar findings with 20% up to 55% depending on the consulted literature[5,9,15,16]. According to the Mexican series, biliary tract diseases were present in 24% of the cases, which is analogous to the most recent findings[7].
A total of 58% of our patients reported at least one medical comorbidity, in which T2DM was present in 25% of the patients, akin to what is described in Mexican, European and United States series[5,7,15].
As already stated, Escherichia coli is the most important pathogen causing PLA in the world literature and even in the present and previous Mexican case series[1,2,6]. Even so, ESBL-Escherichia coli represented a majority of the isolated microorganisms in this series, where the increase compared to the series by Téllez-Zenteno might be explained by the widespread expansion of endoscopic procedures such as biliary stenting, thus promoting the risk of bacterial bile colonization[17]. Of outstanding importance is the absolute increase in Klebsiella pneumoniae cases compared to 20 years ago (n = 5 vs n = 28) due to emerging concern regarding a certain strain of this microorganism as it has been an increasing cause of PLA. There has been a steady increase in the number of reports on this particular pathogen in cryptogenic PLA not only in Asian but also in Western countries[18-20]; this variant has been associated with an abundant production of capsular material along with other heightened intracellular functions that result in a hypermucoviscous phenotype[1,2,18]. Cryptogenic PLAs associated with these Klebsiella pneumoniae strains are thought to be due to colonization of the GI tract with secondary invasion of the intestinal mucosa and portal venous blood, but the true pathogenesis is not completely understood [1,2,20]. Shelat et al[21] demonstrated that despite demographic and clinical differences, the overall outcomes of culture-negative pyogenic liver abscess (CNPLA) patients were equivalent to those of Klebsiella-positive PLA (KPPLA) patients: Hospital stay (15.7 d vs 16.8 d, P = 0.397) and overall 30-d mortality (14.0% vs 11.0%, P = 0.292) in the CNPLA and KPPLA groups, respectively. Hence, it is justified to treat these patients with empirical antibiotics targeting Klebsiella pneumoniae.
Intravenous antibiotic therapy was the therapeutic mainstay. According to the WSES guidelines on intra-abdominal infections, initial antibiotic therapy is typically empirical in nature because a patient with abdominal sepsis requires immediate treatment, and microbiological data (culture and susceptibility results) can require up to 48-72 h before they are available for a more detailed analysis. The selection of appropriate empiric antibiotic therapy is critical for preventing unnecessary morbidity and mortality[22].
In our study, the majority of patients were treated with third-generation cephalosporins ± metronidazole as the combination of choice, which is related to the local flora being represented by sensitive gram-negative enterobacteria and anaerobes. The prevalence of multidrug-resistant (MDR) organisms seems to be ever-rising secondarily due to endoscopic and surgical interventions predisposing patients to recurrent biliary infections and exposure to broad-spectrum antibiotics[23].
Infections caused by resistant gram-negative bacteria, such as Klebsiella pneumoniae, are rapidly emerging as a major source of multidrug-resistant infections worldwide and are the most common causative pathogen of gas-forming PLA (GFPLA). GFPLA represents a small proportion of PLA with a reported incidence of 7%-24%, and it has traditionally been associated with high mortality (25.7% to 37.1%). In one study, Chan et al[24] reported no significant differences between GFLPA and non-GFLPA in the need for percutaneous drainage [26/36 (72.2%) vs 47/72 (65.3%), respectively, P = 0.467] and in-hospital all-cause death [4/36 (11.1%) vs 7 (9.7%), P = 0.822].
The median treatment duration in this study was 19 d, with more than 80% of the patients having received at least 14 d of antibiotic therapy, which, according to the world literature, should be at least 2-6 wk[1,13,17].
Although no general consensus has been published, a small PLA (3-5 cm) is suitable for antibiotic therapy alone, while larger diameter abscesses might need drainage[25]. PCD can be performed when the PLA is > 4 cm, which is arbitrary and is based on the mathematical principles of sphere volumes. It becomes obvious that with increments in diameter from 4 to 5 cm, the volume of spheres doubles from 33 to 65 cc. The 4 cm size also allows for the satisfactory placement of pigtail drains and hence seems to be a logical cut-off value for PCD[6].
PNA or PCD was the interventional radiology method used to drain the PLA, with the latter being the most commonly used in our series due to its high efficacy. PNA is less expensive and more straightforward and does not require catheter care, and its potential complications and multiple abscess cavities can be aspirated but require multiple punctures; additionally, there is a high failure rate with viscous pus or greater cavities. On the other hand, PCD is a high-success rate therapy but might cause discomfort, pain, or cellulitis at the insertion point[26].
The other therapeutic approach to PLA is surgical drainage, either open or laparoscopic. PLA rupture, PNA/PCD failure, anatomic inaccessibility through percutaneous means, and multiloculated and coexisting pathology requiring surgery mandate a more invasive strategy[27]. In a retrospective study by Ahmed et al[28], 39 patients with GPLA were safely treated with IV antibiotics and PCD, and the authors concluded that lesion size was not a contraindication for PCD.
In our series, 22% of the patients underwent OD or LD; 70% of the patients underwent surgery as the primary therapy for the abscess, and the remaining 30% underwent salvage therapy when percutaneous methods failed. Some authors suggest that surgery should be the first-line treatment in patients with a high mortality risk, particularly in critically ill patients[29]. Tan et al[30] published a retrospective study comparing LD and percutaneous methods and failed to achieve clinical superiority of LD despite the theoretical benefits of laparoscopic surgery; the authors still recommend LD due to its lower tendency for developing complications due to sepsis and the possibility to wash the peritoneal cavity with ease[31]. Although in our study the ratio of OD vs LD was 3:2, some series have reported that both approaches are valid and safe in patients able to withstand the physiological stress of surgery and LD, offering advantages for the postoperative recovery of GI function and hospital stay[30].
Abdominal ultrasound is a safe and noninvasive method for evaluating the liver parenchyma and usually shows a hypoechoic mass (or masses), but its echogenicity is variable[1,32]. Therefore, another imaging method is usually needed; in our series, a CT scan was the primary confirmatory method, with a contrast-enhanced technique being used in 85% of the cases and with the remaining patients undergoing magnetic resonance for confirmation. Classic literature describes that PLAs are usually located in the right lobe, which is associated with dominant portal blood input to this anatomic region[1]; our patients' primary lesions were located in segments VII, VI, and VII, which correlates with frequently described findings.
Laboratory findings are usually nonspecific for PLAs but are on par with the inflammatory status of the patient; leukocytosis with an elevated PMN count, hypoalbuminemia, elevated transaminases and alkaline phosphatase, and hyperglycemia were among the most common findings in this study, which are in accordance with other case series[5,33,34].
Complications related to PLA are not uncommon, and rates can be as high as 72%, with bacteremia, empyema, septic shock, and metastatic infection being the most frequent[2]. Relating to our own series, the most frequent complications were septic shock and sepsis, both of which are frequently mentioned in the United States, European or Chinese series[11,12,15]. Septic shock has been associated with excess mortality in patients with PLAs, so identifying which factors relate to its development is a matter of interest. Cho et al[35] described that older age and malignancy were associated with 3.0- and 2.1-fold higher rates of septic shock, as well as elevated procalcitonin levels[36].
The mortality rates due to PLA have varied over the years; previous data have described that patients developing septic shock can reach a mortality rate of 19%[5], but more recent case series mention in-hospital mortality rates of up to 10%[5,7,13,28]. Interestingly, we demonstrated an extraordinarily high in-hospital mortality rate of 63% due to abscess-related complications; this could be due to many factors, including the advanced age of the studied population; the fact that both centers are tertiary care centers where patients from the whole country are referred, thus delaying hospitalization and appropriate antibiotic treatment; the presence of ESBL-Escherichia coli and Klebsiella pneumoniae related to PLA; and the high incidence of septic shock in the study population as well as hospital-acquired infections. Comorbidities also played an important role in the high mortality rate in the series because at least 60% of T2DM patients and 85% of patients with other causes of immunosuppression (HIV infection, underlying malignancy, and autoimmune diseases) impacted the increased mortality.
In our study, T2DM and the development of septic shock did not achieve statistical significance, but as expected, advanced age and higher BMI were high-risk characteristics. The presence of both ESBL-Escherichia coli and Klebsiella pneumoniae demonstrated adjusted ORs of 6.7 and 4.8, respectively, thus being the most critical high-risk characteristics related to mortality in this study, as well as the presence of compromised immune function, which in turn had a 4-fold greater risk of death. Through logistic regression, these 3 variables explained at least 16% of the mortality in our study, with other factors, such as the previously mentioned factors, playing an important role and could be a focal point for further studies.
We consider it important to highlight the measures aimed at reducing mortality, including clinical suspicion and early diagnosis, the availability of imaging studies such as CT, and deciding on a timely intervention for radiology and surgery in difficult to manage cases.
Even though demographic characteristics have remained almost constant in the past decade, an increased prevalence in both referral centers has been noted. More than half of the cases were cryptogenic PLA, which correlates with global epidemiology and a steady shift to a predominant Klebsiella pneumoniae-related abscess. Biliary tree disease was the most common source of infection with ESBL-Escherichia coli and Klebsiella pneumoniae being the most common microorganisms, both of which together with an immunocompromised state were independent predictors of high mortality risk.
In Mexico, the epidemiology of pyogenic liver abscess (PLA) is unknown, with some small case series. This study recruited patients from two high-level hospitals in Mexico.
PLA is a rare medical condition that has had a change in its etiology in recent years; however, more studies are needed to determine the epidemiology of PLA in Mexico.
The aim of this study was to describe the current clinical, demographic, and microbiologic characteristics of PLAs in two high-volume centers in Mexico. These data will allow us to understand the behavior of this disease in Mexico.
This is a retrospective analysis of patients with PLA from two high-level hospitals in Mexico. A chart review was performed to evaluate the clinical, demographic and microbiologic characteristics of PLA. A multivariate analysis was performed to identify independent risk factors associated with mortality.
The main isolated microorganisms were ESBL-Escherichia coli and Klebsiella pneumoniae. The inpatient mortality rate was 63%. In multivariate analysis, immunocompromised state, ESBL-Escherichia coli, and Klebsiella pneumoniae were independent predictors of high risk mortality
An increased prevalence in both referral centers has been noted. The mortality rate was significantly higher compared to previously reported rates worldwide, reaching 63%. There was a steady shift to a predominant Klebsiella pneumoniae-related abscess.
As a retrospective review, our study is limited. Prospective studies that monitor the mortality rate are required, which in this study was high compared to that reported in other series.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Mexico
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): D
Grade E (Poor): 0
P-Reviewer: Shelat VG, Sun WB S-Editor: Zhang H L-Editor: Webster JR P-Editor: Li JH
| 1. | Lübbert C, Wiegand J, Karlas T. Therapy of Liver Abscesses. Viszeralmedizin. 2014;30:334-341. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 41] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 2. | Longworth S, Han J. Pyogenic liver abscess. Clin Liver Dis (Hoboken). 2015;6:51-54. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 29] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 3. | Ko MC, Lin WH, Martini S, Chang YH, Chiu CT, Li CY. A cohort study of age and sex specific risk of pyogenic liver abscess incidence in patients with type 2 diabetes mellitus. Medicine (Baltimore). 2019;98:e15366. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 4. | Sharma A, Mukewar S, Mara KC, Dierkhising RA, Kamath PS, Cummins N. Epidemiologic Factors, Clinical Presentation, Causes, and Outcomes of Liver Abscess: A 35-Year Olmsted County Study. Mayo Clin Proc Innov Qual Outcomes. 2018;2:16-25. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 22] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 5. | Serraino C, Elia C, Bracco C, Rinaldi G, Pomero F, Silvestri A, Melchio R, Fenoglio LM. Characteristics and management of pyogenic liver abscess: A European experience. Medicine (Baltimore). 2018;97:e0628. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 77] [Cited by in RCA: 102] [Article Influence: 14.6] [Reference Citation Analysis (0)] |
| 6. | Shelat VG, Chia CL, Yeo CS, Qiao W, Woon W, Junnarkar SP. Pyogenic Liver Abscess: Does Escherichia Coli Cause more Adverse Outcomes than Klebsiella Pneumoniae? World J Surg. 2015;39:2535-2542. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 42] [Article Influence: 4.2] [Reference Citation Analysis (0)] |
| 7. | Téllez-Zenteno JF, Torre-Delgadillo A, Uscanga-Domínguez LF. Análisis descriptivo de 41 pacientes con absceso hepático piógeno [Descriptive analysis of 41 patients with a pyogenic hepatic abscess]. Rev Invest Clin. 2001;53:218-222. [PubMed] |
| 8. | Singer M, Deutschman CS, Seymour CW, Shankar-Hari M, Annane D, Bauer M, Bellomo R, Bernard GR, Chiche JD, Coopersmith CM, Hotchkiss RS, Levy MM, Marshall JC, Martin GS, Opal SM, Rubenfeld GD, van der Poll T, Vincent JL, Angus DC. The Third International Consensus Definitions for Sepsis and Septic Shock (Sepsis-3). JAMA. 2016;315:801-810. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15803] [Cited by in RCA: 17143] [Article Influence: 1904.8] [Reference Citation Analysis (2)] |
| 9. | Abbas MT, Khan FY, Muhsin SA, Al-Dehwe B, Abukamar M, Elzouki AN. Epidemiology, Clinical Features and Outcome of Liver Abscess: A single Reference Center Experience in Qatar. Oman Med J. 2014;29:260-263. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 17] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 10. | Rahimian J, Wilson T, Oram V, Holzman RS. Pyogenic liver abscess: recent trends in etiology and mortality. Clin Infect Dis. 2004;39:1654-1659. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 391] [Cited by in RCA: 393] [Article Influence: 18.7] [Reference Citation Analysis (0)] |
| 11. | Qian Y, Wong CC, Lai S, Chen H, He X, Sun L, Wu J, Zhou J, Yu J, Liu W, Zhou D, Si J. A retrospective study of pyogenic liver abscess focusing on Klebsiella pneumoniae as a primary pathogen in China from 1994 to 2015. Sci Rep. 2016;6:38587. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 12. | Peris J, Bellot P, Roig P, Reus S, Carrascosa S, González-Alcaide G, Palazón JM, Ramos JM. Clinical and epidemiological characteristics of pyogenic liver abscess in people 65 years or older versus people under 65: a retrospective study. BMC Geriatr. 2017;17:161. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 13. | Du ZQ, Zhang LN, Lu Q, Ren YF, Lv Y, Liu XM, Zhang XF. Clinical Charateristics and Outcome of Pyogenic Liver Abscess with Different Size: 15-Year Experience from a Single Center. Sci Rep. 2016;6:35890. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 14. | Chen J, Zhang M, Chen J, Ning Y, Cai X, Zhang L, Xu H, Guo J. Cryptogenic and non-cryptogenic liver abscess: A retrospective analysis of 178 cases revealed distinct characteristics. J Int Med Res. 2018;46:3824-3836. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 7] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 15. | Meddings L, Myers RP, Hubbard J, Shaheen AA, Laupland KB, Dixon E, Coffin C, Kaplan GG. A population-based study of pyogenic liver abscesses in the United States: incidence, mortality, and temporal trends. Am J Gastroenterol. 2010;105:117-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 187] [Cited by in RCA: 211] [Article Influence: 14.1] [Reference Citation Analysis (0)] |
| 16. | Chan KS, Chen CM, Cheng KC, Hou CC, Lin HJ, Yu WL. Pyogenic liver abscess: a retrospective analysis of 107 patients during a 3-year period. Jpn J Infect Dis. 2005;58:366-368. [PubMed] |
| 17. | Reuken PA, Torres D, Baier M, Löffler B, Lübbert C, Lippmann N, Stallmach A, Bruns T. Risk Factors for Multi-Drug Resistant Pathogens and Failure of Empiric First-Line Therapy in Acute Cholangitis. PLoS One. 2017;12:e0169900. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 1] [Reference Citation Analysis (0)] |
| 18. | Arena F, Henrici De Angelis L, D'Andrea MM, Cannatelli A, Fossati L, Di Pilato V, Giani T, Cavallo R, Rossolini GM. Infections caused by carbapenem-resistant Klebsiella pneumoniae with hypermucoviscous phenotype: A case report and literature review. Virulence. 2017;8:1900-1908. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 23] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 19. | Peermohamed S, Kogilwaimath S. Hypervirulent (hypermucoviscous) Klebsiella pneumoniae causing pyogenic liver abscess. CMAJ. 2018;190:E441. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 20. | Fazili T, Sharngoe C, Endy T, Kiska D, Javaid W, Polhemus M. Klebsiella pneumoniae Liver Abscess: An Emerging Disease. Am J Med Sci. 2016;351:297-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 58] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 21. | Shelat VG, Wang Q, Chia CL, Wang Z, Low JK, Woon WW. Patients with culture negative pyogenic liver abscess have the same outcomes compared to those with Klebsiella pneumoniae pyogenic liver abscess. Hepatobiliary Pancreat Dis Int. 2016;15:504-511. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 27] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 22. | Sartelli M, Chichom-Mefire A, Labricciosa FM, Hardcastle T, Abu-Zidan FM, Adesunkanmi AK, Ansaloni L, Bala M, Balogh ZJ, Beltrán MA, Ben-Ishay O, Biffl WL, Birindelli A, Cainzos MA, Catalini G, Ceresoli M, Che Jusoh A, Chiara O, Coccolini F, Coimbra R, Cortese F, Demetrashvili Z, Di Saverio S, Diaz JJ, Egiev VN, Ferrada P, Fraga GP, Ghnnam WM, Lee JG, Gomes CA, Hecker A, Herzog T, Kim JI, Inaba K, Isik A, Karamarkovic A, Kashuk J, Khokha V, Kirkpatrick AW, Kluger Y, Koike K, Kong VY, Leppaniemi A, Machain GM, Maier RV, Marwah S, McFarlane ME, Montori G, Moore EE, Negoi I, Olaoye I, Omari AH, Ordonez CA, Pereira BM, Pereira Júnior GA, Pupelis G, Reis T, Sakakhushev B, Sato N, Segovia Lohse HA, Shelat VG, Søreide K, Uhl W, Ulrych J, Van Goor H, Velmahos GC, Yuan KC, Wani I, Weber DG, Zachariah SK, Catena F. The management of intra-abdominal infections from a global perspective: 2017 WSES guidelines for management of intra-abdominal infections. World J Emerg Surg. 2017;12:29. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 282] [Cited by in RCA: 251] [Article Influence: 31.4] [Reference Citation Analysis (0)] |
| 23. | Shi S, Xia W, Guo H, Kong H, Zheng S. Unique characteristics of pyogenic liver abscesses of biliary origin. Surgery. 2016;159:1316-1324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 24. | Chan KS, Thng CB, Chan YH, Shelat VG. Outcomes of Gas-Forming Pyogenic Liver Abscess Are Comparable to Non-Gas-Forming Pyogenic Liver Abscess in the Era of Multi-Modal Care: A Propensity Score Matched Study. Surg Infect (Larchmt). 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Lardière-Deguelte S, Ragot E, Amroun K, Piardi T, Dokmak S, Bruno O, Appere F, Sibert A, Hoeffel C, Sommacale D, Kianmanesh R. Hepatic abscess: Diagnosis and management. J Visc Surg. 2015;152:231-243. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 108] [Cited by in RCA: 169] [Article Influence: 16.9] [Reference Citation Analysis (0)] |
| 26. | Singh S, Chaudhary P, Saxena N, Khandelwal S, Poddar DD, Biswal UC. Treatment of liver abscess: prospective randomized comparison of catheter drainage and needle aspiration. Ann Gastroenterol. 2013;26:332-339. [PubMed] |
| 27. | Eipel C, Glanemann M, Nuessler AK, Menger MD, Neuhaus P, Vollmar B. Ischemic preconditioning impairs liver regeneration in extended reduced-size livers. Ann Surg. 2005;241:477-484. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 126] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 28. | Ahmed S, Chia CL, Junnarkar SP, Woon W, Shelat VG. Percutaneous drainage for giant pyogenic liver abscess--is it safe and sufficient? Am J Surg. 2016;211:95-101. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 44] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 29. | Alkofer B, Dufay C, Parienti JJ, Lepennec V, Dargere S, Chiche L. Are pyogenic liver abscesses still a surgical concern? A Western experience. HPB Surg. 2012;2012:316013. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 35] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 30. | Tan L, Zhou HJ, Hartman M, Ganpathi IS, Madhavan K, Chang S. Laparoscopic drainage of cryptogenic liver abscess. Surg Endosc. 2013;27:3308-3314. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 12] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 31. | Tu JF, Huang XF, Hu RY, You HY, Zheng XF, Jiang FZ. Comparison of laparoscopic and open surgery for pyogenic liver abscess with biliary pathology. World J Gastroenterol. 2011;17:4339-4343. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 23] [Cited by in RCA: 17] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 32. | Bächler P, Baladron MJ, Menias C, Beddings I, Loch R, Zalaquett E, Vargas M, Connolly S, Bhalla S, Huete Á. Multimodality Imaging of Liver Infections: Differential Diagnosis and Potential Pitfalls. Radiographics. 2016;36:1001-1023. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 84] [Cited by in RCA: 73] [Article Influence: 8.1] [Reference Citation Analysis (0)] |
| 33. | Liu Y, Wang JY, Jiang W. An Increasing Prominent Disease of Klebsiella pneumoniae Liver Abscess: Etiology, Diagnosis, and Treatment. Gastroenterol Res Pract. 2013;2013:258514. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 61] [Cited by in RCA: 69] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 34. | Kong H, Yu F, Zhang W, Li X. Clinical and microbiological characteristics of pyogenic liver abscess in a tertiary hospital in East China. Medicine (Baltimore). 2017;96:e8050. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 31] [Cited by in RCA: 37] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 35. | Cho H, Lee ES, Lee YS, Kim YJ, Sohn CH, Ahn S, Seo DW, Lee JH, Kim WY, Lim KS. Predictors of septic shock in initially stable patients with pyogenic liver abscess. Scand J Gastroenterol. 2017;52:589-594. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 12] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 36. | Roediger R, Lisker-Melman M. Pyogenic and Amebic Infections of the Liver. Gastroenterol Clin North Am. 2020;49:361-377. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 52] [Article Influence: 10.4] [Reference Citation Analysis (1)] |