Published online Feb 21, 2022. doi: 10.3748/wjg.v28.i7.693
Peer-review started: October 26, 2021
First decision: December 27, 2021
Revised: January 4, 2022
Accepted: January 22, 2022
Article in press: January 22, 2022
Published online: February 21, 2022
Processing time: 114 Days and 0.4 Hours
Early gastric cancer (EGC) is an invasive carcinoma involving only the stomach mucosa or submucosa, independently of lymph node status. EGC represents over 50% of cases in Japan and in South Korea, whereas it accounts only for approximately 20% of all newly diagnosed gastric cancers in Western countries. The main classification systems of EGC are the Vienna histopathologic classification and the Paris endoscopic classification of polypoid and non-polypoid lesions. A careful endoscopic assessment is fundamental to establish the best treatment of EGC. Generally, EGCs are curable if the lesion is completely removed by endoscopic resection or surgery. Some types of EGC can be resected endoscopically; for others the most appropriate treatment is surgical resection and D2 lymphadenectomy, especially in Western countries. The favorable oncological prognosis, the extended lymphadenectomy and the reconstruction of the intestinal continuity that excludes the duodenum make the prophylactic cholecystectomy mandatory to avoid the onset of biliary complications.
Core Tip: Early gastric cancer (EGC) is an invasive stomach cancer confined to the mucosal or submucosal lining and represents approximately 20% of gastric cancers in Western countries. A correct classification allows the most appropriate treatment. Some types of EGC are adequately treated by endoscopic mucosal resection, whilst others need gastrectomy. In Western countries, due to a higher incidence of the diffuse histotype and the less widespread advanced endoscopic procedures, surgical resection and D2 lymphadenectomy are regarded as the “gold standard” treatment.
- Citation: Chiarello MM, Fico V, Pepe G, Tropeano G, Adams NJ, Altieri G, Brisinda G. Early gastric cancer: A challenge in Western countries. World J Gastroenterol 2022; 28(7): 693-703
- URL: https://www.wjgnet.com/1007-9327/full/v28/i7/693.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i7.693
In its initial stages, gastric cancer tends to spread in the lamina propria, infiltrating the muscularis mucosae and the submucosal layer of the gastric wall[1-4]. The Japanese Society for Gastroenterological Endoscopy’s definition of early gastric cancer (EGC) is an invasive gastric cancer that invades no more deeply than the submucosa, irrespective of lymph node metastases (LNM). Thanks to rigorous screening programs in the Asian countries, up to 50% of patients treated for a gastric cancer have EGC[5,6]. Such a high incidence is not found in European countries, where advanced gastric cancer is prevalent.
The difference in incidence between Asian and Western countries may be due to a wide use of magnifying upper endoscopy with high-resolution images and chromoendoscopy in Japan and South Korea.
A correct classification and an accurate diagnosis are fundamental to plan the most effective treatment[7-10]. Some forms of EGC can be treated by endoscopic resection; in other forms surgical treatment is mandatory[10-14].
EGC carries an excellent prognosis if the lesion is completely removed by endoscopic resection or surgery[15-17]. Most of the Japanese studies have reported 5-year and 10-year survival rates of more than 90% for the patients with EGC. In the Western studies, 5-year survival rates are variable, ranging from 68% to 92%. EGCs recur in at least 1.9% of cases after resection with time intervals ranging from 4 mo to more than 10 years. Important risk factors for the recurrence are the presence of submucosal invasion, LNMs and undifferentiated histology.
The most common classification is the TNM, which relates information about the primitive lesion (T), node involvement (N) and distant metastases (M) thus providing information about the diffusion of the disease.
The macroscopic classification of EGC, as defined by the Japanese Gastric Cancer Association (JGCA), identifies different subtypes: type 0-I (protruding-polypoid tumors); type 0-IIa (superficial elevated tumors); type 0-IIb (tumors without elevation or depression); type 0-IIc (slightly depressed tumors); and type 0-III (excavated tumors), referring to the morphological features of the lesion on the mucosal surface[18].
The Vienna classification (Table 1) is a histologic classification. While the TNM defines EGC as a cancer invading no more deeply than the submucosa, irrespective of LNMs, the Vienna classification is the most exhaustive[19,20]. In particular, the intramucosal EGC is classified in 3 categories, while the submucosal EGC in 2, according to the depth of infiltration. This distinction is not present in the TNM system and is fundamental for the treatment planning. In fact, it allows the option for an endoscopic therapy [(endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD)] or surgery if mandatory.
| Category | Definition | Treatment |
| 1 | Negative for dysplasia/neoplasia | No treatment |
| 2 | Indefinite for dysplasia/neoplasia | Follow-up, recheck |
| 3 | Non-invasive, low-grade dysplasia | Follow-up |
| 4 | Non-invasive, high-grade dysplasia | Endoscopic resection, surgery |
| 4.1 | High-grade adenoma/dysplasia | |
| 4.2 | Non-invasive carcinoma (CIS) | |
| 4.3 | Suspicious of invasive carcinoma | |
| 5 | Invasive neoplasia | Surgery (recently ESD) |
| 5.1 | Intramucosal carcinoma | |
| M1 | Mucosa only | |
| M2 | Mucosa with preservation of muscularis mucosae | |
| M3 | Not further than muscularis mucosae | |
| 5.2 | Submucosal carcinoma or beyond | |
| sm1 | Invasion of muscularis mucosae < 0.5 µm | |
| sm2 | Invasion of muscularis mucosae > 0.5 µm |
The Paris classification of polypoid and non-polypoid lesions including flat lesions is useful to evaluate possible endoscopic treatments. This classification was created for the superficial colon neoplasms, undoubtedly the most frequent in the Western World and with some small changes was adopted to the superficial gastric neoplasms. One of the changes is the thickness of the wall layer, as in the submucosa, are thinner in the stomach.
A correct endoscopic diagnosis is fundamental in EGC[21]. The Japanese Society for Gastroenterological Endoscopy suggests that the length of the endoscopy, the accurate stomach wall distension and the further removal of mucous from the stomach lining are proportional to diagnostic accuracy and lesion detection rate[22]. As far as the length of the procedure is concerned, the most appropriate protocol is the Systematic Screening Stomach, which requires careful vision of the gastric lumen and acquisition of 22 pictures (12 in anterograde vision and 10 in retro vision) with a global length of the exam not inferior to 12 min[22]. Sedation is advised, while trans-nasal endoscopy is not.
Regarding diagnostic accuracy, the use of traditional or virtual cromo-endoscopy is advised to look for the “minimal changes” of the Japanese authors to evaluate the size and morphology of the lesion and the glandular and vascular pattern. Lesions should be described according to the Paris classification (Type 0I protruding, Type 0-IIa elevated, Type 0-IIb flat, Type 0-IIc depressed, Type 0-III excavated)[23].
Endoscopic resection must be taken into account when the risk of LNMs is low or when the location and size of the tumor allows a safe “en-bloc” complete resection[24,25]. Selected EGCs can be treated with EMR or ESD with good results in Western patients[26-28]. Absolute indications for endoscopic resection, according to the Japanese gastric cancer treatment guidelines, are: cT1a differentiated-type adenocarcinoma without ulcerative findings or cT1a differentiated-type adenocarcinoma with ulcerative findings and a diameter 3 cm. Endoscopic resection for cT1a undifferentiated carcinoma without ulcerative findings and with a diameter 2 cm is considered an expanded indication. Therefore, endoscopic resection should be considered for elderly patients with a high operative risk (relative indication)[18,29].
According to JGCA guidelines (Table 2), en-bloc endoscopic resection for differentiated pT1a cancer without ulcerative findings (UL0), negative horizontal margin (HM0), negative vertical margin (VM0) and no lymphovascular infiltration (Ly0, V0) or en-bloc endoscopic resection for pT1a cancer with ulcerative findings, tumor size 3 cm, HM0, VM0, Ly0 and V0 are classified as endoscopic curability A. However, the resection is classified as endoscopic curability B for undifferentiated pT1a cancer resected en-bloc, tumor size 2 cm, without ulcerative findings, HM0, VM0, Ly0 and V0 or for en-bloc resected pT1b differentiated cancer, with a depth of infiltration < 500 µm, tumor size 3 cm, HM0, VM0, Ly0 and V0. When the resection fulfills criteria to be classified as endoscopic curability A or endoscopic curability B, but the cancer was not resected en-bloc or has positive horizontal margin, it is classified as endoscopic curability C-1. All other resections are classified as endoscopic curability C-2.
| Mucosal cancer | Submucosal cancer | |||||
| No ulcer | Ulcer present | SM1 | SM2 | |||
| Size | ≤ 20 | > 20 | ≤ 30 | > 30 | ≤ 30 | Any size |
| Differentiated | Y | G | G | R | G | R |
| Undifferentiated | Gray | R | R | R | R | R |
Both endoscopic curability A and endoscopic curability B are considered to have low risk of LNMs and do not require additional surgery, so the patient can go to follow-up. Endoscopic curability C-1 has a low risk of LNMs but should be aware of residual or local recurrence, so the patient could be treated with another ESD, surgical resection, close observation or endoscopic coagulation. After an endoscopic resection classified as endoscopic curability C-2, gastrectomy with lymphadenectomy should be considered the standard-of-care[18].
The results of endoscopic treatment (EMR and ESD) have been compared to traditional surgical treatment in patients with EGC in several meta-analyses[30]. Overall survival after EMR or ESD is not significantly different when compared to surgical resection (hazard ratio: 0.995, 95% confidence interval: 0.836-1.185; P = 0.9). As for recurrence free survival, it has been proved that the recurrence risk after EMR is significantly higher compared to surgical resection (hazard ratio: 3,946, 95% confidence interval: 1.233-12.632, P = 0.02)[30]. However, it has been noted that in adequately selected patients endoscopic resection offers results that are similar to those of traditional surgery[31]. In patients who underwent EMR/ESD, strict endoscopic surveillance is mandatory, given the higher risk of endoluminal recurrence.
Complications of EMR or ESD for EGC include pain, bleeding and perforation[32]. Pain after resection is typically mild. Bleeding occurs in up to 8% of patients undergoing endoscopic treatment. Immediate bleeding appears more common with EMR/ESD of tumors located in the upper third of the stomach. Perforation occurs in 4% of the patients during ESD[15].
An innovative procedure called laparoscopic endoscopic cooperative surgery (LECS) that combines the strongest points of interventional endoscopy and laparoscopic surgery for the removal of gastric wall tumors was developed by Hiki et al[33]. In the original procedure of LECS, the stomach wall is opened for resection of the tumor, and the lumen is exposed to intraperitoneal space. LECS involves precutting around the tumor with an endoscope and artificial perforation of the gastric wall. Next, excision of the tumor with laparoscopy and repair of the gastric wall with a stapler are performed[34,35].
The indications for LECS are currently limited to lesions that are normally managed by ESD but are technically challenging to resect via ESD because of the presence of ulcers, or if the tumor size is more than 30 mm in diameter[36].
When a LECS procedure and sentinel node biopsy were combined, an extremely minimally invasive procedure that is adequate for radical oncological resection of EGC is achieved. Sentinel node navigation surgery is an ideal surgical option for preservation of most parts of the stomach and consequent maintenance of normal gastric function to improve quality of life in patients with EGC. Although many previous studies and clinical trials have demonstrated the safety and feasibility of the sentinel node concept in gastric cancer, the clinical application of sentinel node navigation surgery is debatable. Several issues regarding technical standardization and oncological safety need to be resolved. Recently several studies to resolve these problems are being actively performed, and sentinel node navigation surgery might be an important surgical option in the treatment of gastric cancer in the future.
Other techniques (Table 3) have been developed to avoid spread of the tumor to the peritoneum[37-39]. When performed by expert teams they show a lot of promise and achieve solid oncologic results[40,41]. Use and standardization of these minimally invasive surgical procedures contributes to reduction of unnecessary gastrectomy for gastric submucosal tumors[42]. Most of the clinical experiences are Japanese, and the small number of treated cases does not allow a comparison with the longest used endoscopic and surgical techniques. More research and clinical trials about LECS for EGC are expected. Furthermore, surgeons need to select one of these minimally invasive procedures according to the characteristics of the tumor, the localization of the tumor in the stomach, the personal surgical experience and the technological characteristics of the health institution.
| Procedure | Characteristics |
| Laparoscopic endoscopic cooperative surgery | Endoscopic dissection of the mucosal or submucosal layers with laparoscopic seromuscular resection. |
| Endoscope-assisted laparoscopic wedge resection | The procedure is performed to remove tumors with a laparoscope after localization by an intraoperative endoscope. EAWR is difficult to implement in sites where strictures may occur, such as pylorus and the gastroesophageal junction. |
| Laparoscopy-assisted endoscopic resection | The concept of LAER is contrary to that of EAWR. The procedure is an ESD procedure assisted by laparoscopy. |
| Endoscope-assisted laparoscopic transgastric resection | The procedure involves opening of the gastric wall under the direct view of an endoscope, tagging the tumor with a laparoscopic suture and performing wedge resection with a laparoscopic stapler. |
| Laparoscopic intragastric surgery | Procedure can be used in laparoscopic surgery performed within the stomach. The incision in the wall of the stomach is minimized and laparoscopic trocars are inserted into the gastric lumen. |
| Single-incision intragastric resection | This is a single-port laparoscopic surgery. |
| Endoscopic submucosal dissection with laparoscopic lymph node dissection | This procedure is the same as LAER with laparoscopic perigastric lymph node dissection. The advantage is that the stomach can be preserved. However, the main procedure is ESD, which requires a skilled endoscopist. |
| Single-incision endoscopic submucosa dissection with laparoscopic lymph node dissection | The procedure is similar to SI-IGR, where sentinel node navigation surgery with unilateral perigastric laparoscopic lymph node dissection is performed with a single-port. Then ESD is performed through a single-port. |
| Laparoscopy-assisted endoscopic full-thickness resection | If the tumor invades deeper than the muscle layer of the wall of the stomach, full-thickness resection with an endoscope is performed and a laparoscope is used for repair. |
| Non-exposed wall-inversion surgery | The procedure was developed so that EFTR could be performed without spillage. The disadvantages are that the procedure time is long, as it involves ESD and endoscopic closure, and it is difficult to apply to the pyloric area and gastroesophageal junction. |
| Clean no-exposure technique | Similar to NEWS, this procedure has also been developed to avoid cancer cell spillage. Clean-NET can be applied to EGCs in most locations, except for pyloric area and gastroesophageal junction. |
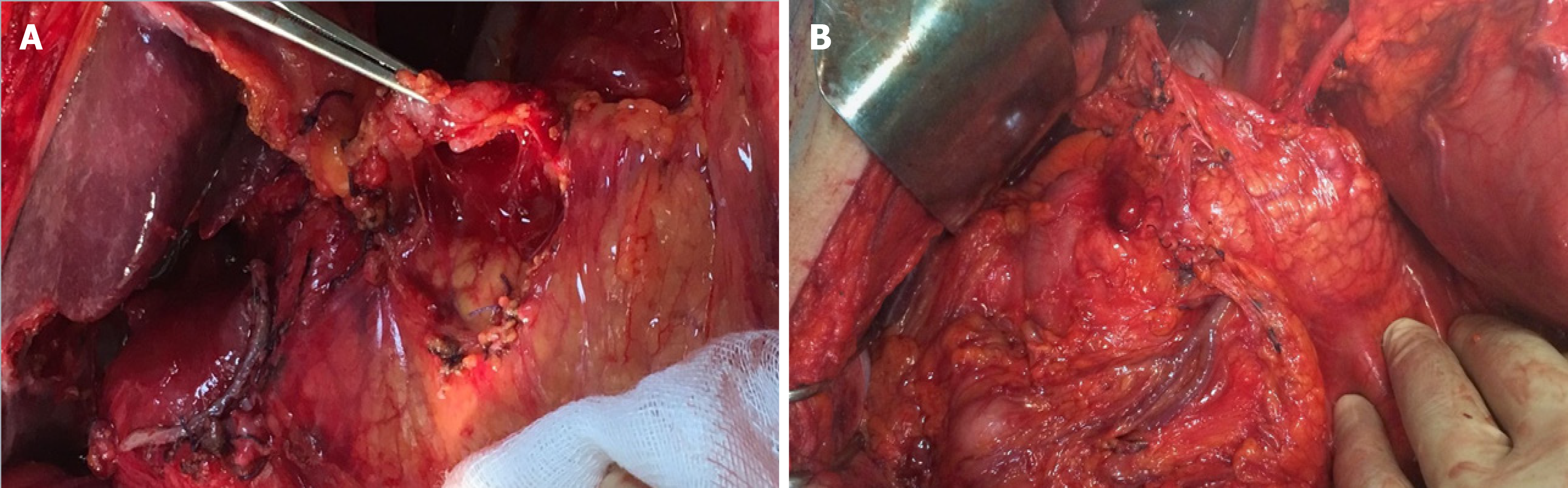
In the case of EGC without the above features, total or partial gastrectomy with a free margin of at least 2 cm is the treatment of choice. Adequate lymphadenectomy offers a high chance of curing patients with EGC[43-45]. The lymph nodes of the stomach are classified into stations numbered as in Table 4. The regional stations are the lymph node stations 1-12 and LN station 14v (Figure 1A). The remnant stations are considered as distant stations, and metastases to these nodes are classified as M1. The JGCA defined the extent of systematic lymphadenectomy according to the type (total or distal) of gastrectomy indicated (Table 5).
| Nr. | Definition |
| 1 | Right paracardial LNs, including those along the first branch of the ascending limb of the left gastric artery |
| 2 | Left paracardial LNs including those along the esophagocardiac branch of the left subphrenic artery |
| 3 | 3a: Lesser curvature LNs along the branches of the left gastric artery; 3b: Lesser curvature LNs along the 2nd branch and distal part of the right gastric artery |
| 4 | (1) 4sa: Left greater curvature LNs along the short gastric arteries (perigastric area); (2) 4sb: Left greater curvature LNs along the left gastroepiploic artery (perigastric area); and (3) 4d: Right greater curvature LNs along the 2nd branch and distal part of the right gastroepiploic artery |
| 5 | Suprapyloric LNs along the 1st branch and proximal part of the right gastric artery |
| 6 | Infrapyloric LNs along the first branch and proximal part of the right gastroepiploic artery down to the confluence of the right gastroepiploic vein and the anterior superior pancreatoduodenal vein |
| 7 | LNs along the trunk of left gastric artery between its root and the origin of its ascending branch |
| 8 | 8a: Anterosuperior LNs along the common hepatic artery; 8p: Posterior LNs along the common hepatic artery |
| 9 | Coeliac artery |
| 10 | Splenic hilar LNs including those adjacent to the splenic artery distal to the pancreatic tail, and those on the roots of the short gastric arteries and those along the left gastroepiploic artery proximal to its 1st gastric branch |
| 11 | (1) 11p: Proximal splenic artery LNs from its origin to halfway between its origin and the pancreatic tail end; and (2) 11d: Distal splenic artery LNs from halfway between its origin and the pancreatic tail end to the end of the pancreatic tail |
| 12 | (1) 12a: Hepatoduodenal ligament LNs along the proper hepatic artery, in the caudal half between the confluence of the right and left hepatic ducts and the upper border of the pancreas; (2) 12b: Hepatoduodenal ligament LNs along the bile duct, in the caudal half between the confluence of the right and left hepatic ducts and the upper border of the pancreas; and (3) 12p: Hepatoduodenal ligament LNs along the portal vein in the caudal half between the confluence of the right and left hepatic ducts and the upper border of the pancreas |
| 13 | LNs on the posterior surface of the pancreatic head cranial to the duodenal papilla |
| 14 | LNs along the superior mesenteric vein |
| 15 | LNs along the middle colic vessels |
| 16 | (1) 16a1: Paraaortic LNs in the diaphragmatic aortic hiatus; (2) 16a2: Paraaortic LNs between the upper margin of the origin of the celiac artery and the lower border of the left renal vein; (3) 16b1: Paraaortic LNs between the lower border of the left renal vein and the upper border of the origin of the inferior mesenteric artery; and (4) 16b2: Paraaortic LNs between the upper border of the origin of the inferior mesenteric artery and the aortic bifurcation |
| 17 | LNs on the anterior surface of the pancreatic head beneath the pancreatic sheath |
| 18 | LNs along the inferior border of the pancreatic body |
| 19 | Infradiaphragmatic LNs predominantly along the subphrenic artery |
| 20 | Paraesophageal LNs in the diaphragmatic esophageal hiatus |
| Lymphadenectomy | Gastrectomy | |
| Total | Distal | |
| D1 | Lymph node stations from N. 1 to 7 | Lymph node stations N. 1, 3, 4sb, 4d, 5, 6 and 7 |
| D1+ | D1 stations plus stations N. 8a, 9 and 11p | D1 stations plus stations N. 8a and 9 |
| D2 | D1 stations plus stations N. 8a, 9, 10, 11p, 11d and 12a | D1 stations plus stations N. 8a, 9, 11p and 12a. |
Surgical treatment is also mandatory when there is a lack of training in complex endoscopic procedures. In hospitals with a low volume of advanced endoscopic procedures, gastrectomy remains the gold standard for treatment of EGC.
In EGC lymphatic invasion is early and is linked to the distribution of the lymphatic vessels in the mucosa and submucosa, which form a rich plexus located just above, inside and below the muscularis mucosae. Infiltration of the mucosa (EGC T1a) is sufficient to cause LNMs. Submucosal infiltration (EGC T1b) entails a higher incidence of LNMs. In addition, the risk is higher in cases where the submucosal invasion has destroyed the muscularis mucosae (5.3% of intramucosal cancer and in 19.6%-64.3% of submucosal neoplasms)[46,47].
As already known, tumor grading and vascular-lymphatic invasion were found to be significant risk factors. A study on 517 patients with EGC found that LNMs are present in 22% of cases (114 patients), directly related to an ulcerated lesion of more than 2 cm diameter. Furthermore, the risk of LNMs is higher in males (P = 0.03), elderly patients (P = 0.01), in the presence of depressed tumor (P = 0.01) and with submucosal invasion (P = 0.03). Further risk factors include tumor localization in the body (P = 0.04) or at the angulus (P = 0.02). In these areas, the submucosa is thinner, and the lymphatic vessels more widespread in the lamina propria of the mucosa.
Similar results have been documented in a recent paper, where age, gender, tumor size, type of differentiation, Lauren classification and lympho-vascular and perineural invasion showed a significant correlation with the rate of LNMs in EGC by univariate and multivariate analyses in 1033 patients who underwent radical gastrectomy with lymphadenectomy. Patients with T1b gastric cancer had an older age, a higher proportion of proximal lesions, larger tumor size, more frequent vascular lymphatic invasion, perineural invasion and more LNMs than patients with T1a gastric cancer[48-51].
In a series of 5265 Japanese patients that underwent gastrectomy and lymphadenectomy, 3016 lesions were intramucosal EGCs, and 2249 were infiltrating the submucosa. LNMs were present in 65 (2.2%) of the patients with intramucosal EGC and in 402 (17.9%) of those with penetrating EGC. The authors documented that in the intramucosal cancer, the risk of LNMs is higher in depressed or ulcerated lesions, in lesions bigger than 30 mm, in those with an undifferentiated type or in those with vascular and lymphatic invasion. In submucosal EGCs a higher incidence of LNMs was documented in submucosal invasion ≥ 500 µm (23.7%) compared to submucosal invasion < 500 µm (8.8%, P < 0.0001), when there was vascular lymphatic invasion (36.6% vs 9.8% if no infiltration, P < 0.0001), in case of elevated EGC (24.8%) compared to depressed type (17.1%, P = 0.0003) and in tumors larger than 30 mm (P <0.0001). Moreover, extra-gastric LNMs (level II) were found in 10% of the cases, in the absence of involvement of the peri-gastric lymph nodes.
To optimize lymphadenectomy, the sentinel node search has been proposed. Said procedure is now standardized in breast cancer surgery but still experimental in the case of EGC[52,53]. Two endoscopically administered tracers are used sequentially: the 99Tc radioisotope (at least 3 h before the intervention) and the blue isosulphan dye (during the intervention). Since skip LNMs are frequent (about 20% of cases in EGCs), a negative sentinel node does not exclude the possibility that subsequent lymph node stations are involved in the disease. The usefulness of the sentinel lymph node would therefore not consist in excluding LNMs, as initially proposed, but in identifying the lymphatic drainage basin that must be removed. Thus, the sentinel lymph node would therefore become an aid to identify the relationship between gastric anatomy and gastric lymphatic drainage.
LNM strongly affects the prognosis[14,54]. This is a topic of not negligible importance because EGC is believed to be a curable albeit malignant disease. In T1a EGCs, the 10-year survival rates are about 100% in patients undergoing gastric resection and extended (D2) lymphadenectomy[55]. Five-year and ten-year survival in these patients was 2%–3% higher than in those with limited (D1) lymphadenectomy. In the case of submucosal EGC, the 10-year survival rate was about 100% in patients undergoing gastric resection and extended lymphadenectomy. Five-year and ten-year survival was 10% higher in these patients than in those with limited lymphadenectomy. Looking at the postoperative lymph node positive (pN+) patient group, 10-year survival was 87% if extended lymphadenectomy was performed, and 55% if lymphadenectomy was limited.
In patients with a radical resection, when a D2 lymphadenectomy (Figure 1B) was performed and the duodenum was excluded in the intestinal reconstruction, cholecy
The use of 300 mg or 600 mg of ursodeoxycholic acid compared with placebo resulted in a significantly decreased proportion of patients developing gallstones within 12 mo after surgery for gastric cancer (5.3% in the 300 mg group, 4.3% in the 600 mg group and 16.7% in the placebo group)[56]. Gallstone formation and its complications may be prevented using ursodeoxycholic acid rather than prophylactic cholecystectomy. However, the results raise some questions. As Pitt and Nakeeb[57] maintained, the study was conducted in patients undergoing gastrectomy for stomach cancer but follow-up did not last more than a year. Furthermore, more than 80% of patients have EGCs with a presumptively satisfactory oncological prognosis, given that 84% of cancers occurred in the distal stomach and 57% of lymphadenectomies were D2[56]. In this setting, we believe that prophylactic cholecystectomy is necessary for patients with a good cancer prognosis, as suggested by Pitt and Nakeeb[57].
Studies on the subject conclude that prophylactic cholecystectomy does not have a significant impact on the natural course of the disease[58]. However, it leads to a reduction in the number of biliary complications (which may affect up to 15% of the operated patients) and does not induce an increase in mortality and morbidity rates. In one study, a mortality rate of 1.8% was reported in the case of cholecystectomy performed during an intervention other than that of the gastric cancer treatment. Prophylactic cholecystectomy seems to be unnecessary only in cases where the continuity of the digestive tract involves the use of the duodenum[59]. In a study on 969 patients, after gastrectomy, gallbladder stones developed in 6.1% of patients (59 out of 969) with an incidence of cholecystitis /cholangitis of 1.2% of cases (12/969). It has been found that the method used to restore intestinal continuity, with preservation of the duodenal transit or excluding the duodenum, is an independent risk factor for both the development of cholelithiasis (P = 0.018) and cholecystitis and cholangitis (P = 0.006). It has been also confirmed that in patients who develop cholelithiasis, the incidence of cholecystitis and cholangitis is particularly high when the duodenal transit was excluded (31.3%) compared to those with duodenal transit maintained (7.4%).
In agreement with the JGCA treatment guidelines, D1 lymphadenectomy can be sufficient to treat EGCs not suitable for endoscopic treatment when the regional nodes are clinically free from disease. It is important to point out, however, that a relevant percentage of EGCs in Western countries are of the diffuse type and therefore associated with a high risk of LNMs, especially when the submucosa is invaded. Moreover, in Western countries, endoscopic resections are not very common, and preoperative diagnosis of lymph node status has some limitations, despite advancements in radiological techniques. Considering these limitations, D2 lymphadenectomy in EGCs not susceptible to radical endoscopic treatment is the surgical treatment of choice. In this area, based on a good prognosis and intestinal reconstruction that excludes the duodenum from gastrointestinal transit, we believe that prophylactic cholecystectomy is necessary.
Provenance and peer review: Invited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Surgery
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): A
Grade B (Very good): 0
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Yoshikawa T, Zhang H S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Ma YJ
| 1. | Yang Q, Feng S, Liu H, Zhang X, Cao J, Zhu Y, Zheng H, Song H. Clinicopathological features and lymph node metastasis risk in early gastric cancer with WHO criteria in China: 304 cases analysis. Ann Diagn Pathol. 2021;50:151652. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 2. | Ahn JY, Kim YI, Shin WG, Yang HJ, Nam SY, Min BH, Jang JY, Lim JH, Kim J-, Lee WS, Lee BE, Joo MK, Park JM, Lee HL, Gweon TG, Park MI, Choi J, Tae CH, Kim YW, Park B, Choi IIJ. Comparison between endoscopic submucosal resection and surgery for the curative resection of undifferentiated-type early gastric cancer within expanded indications: a nationwide multi-center study. Gastric Cancer. 2021;24:731-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 21] [Article Influence: 5.3] [Reference Citation Analysis (0)] |
| 3. | Abe S, Takizawa K, Oda I, Mizusawa J, Kadota T, Ono H, Hasuike N, Yano T, Yamamoto Y, Horiuchi Y, Nagata S, Yoshikawa T, Terashima M, Muto M. Incidence and treatment outcomes of metachronous gastric cancer occurring after curative endoscopic submucosal dissection of undifferentiated-type early gastric cancer: Japan Clinical Oncology Group study-post hoc analysis of JCOG1009/1010. Gastric Cancer. 2021;24:1123-1130. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 4. | Cai F, Dong Y, Wang P, Zhang L, Yang Y, Liu Y, Wang X, Zhang R, Liang H, Sun Y, Deng J. Risk assessment of lymph node metastasis in early gastric cancer: Establishment and validation of a 7-point scoring model. Surgery. 2021;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (0)] |
| 5. | Hamashima C. Cancer screening guidelines and policy making: 15 years of experience in cancer screening guideline development in Japan. Jpn J Clin Oncol. 2018;48:278-286. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 76] [Article Influence: 10.9] [Reference Citation Analysis (1)] |
| 6. | Hamashima C; Systematic Review Group and Guideline Development Group for Gastric Cancer Screening Guidelines. Update version of the Japanese Guidelines for Gastric Cancer Screening. Jpn J Clin Oncol. 2018;48:673-683. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 281] [Cited by in RCA: 268] [Article Influence: 38.3] [Reference Citation Analysis (0)] |
| 7. | Chang JW, Jung DH, Park JC, Shin SK, Lee SK, Lee YC. Long-Term Outcomes and Prognostic Factors of Endoscopic Submucosal Dissection for Early Gastric Cancer in Patients Aged ≥75 Years. Cancers (Basel). 2020;12. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 16] [Cited by in RCA: 10] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 8. | Mejía R, Sáez J, Norero E, Briones P, Ceroni M, Martínez C, Díaz A, Calvo A, Sharp A. Long-term Results of Endoscopic Submucosal Dissection (ESD) for the Treatment of Early Gastric Cancer (EGC) in a High-volume Latin American Center. Surg Laparosc Endosc Percutan Tech. 2020;31:165-169. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 2] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 9. | Shin EJ, Gong CS, Kim BS, Kim SO, Ko CS, Kim HS. Early Surgical and Long-term Oncological Outcomes of Totally Laparoscopic Near-total Gastrectomy in >150 Cases. Surg Laparosc Endosc Percutan Tech. 2020;30:529-533. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 10. | Abe N, Takeuchi H, Ohki A, Yanagida O, Masaki T, Mori T, Sugiyama M. Long-term outcomes of combination of endoscopic submucosal dissection and laparoscopic lymph node dissection without gastrectomy for early gastric cancer patients who have a potential risk of lymph node metastasis. Gastrointest Endosc. 2011;74:792-797. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 45] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 11. | Ahmad R, Setia N, Schmidt BH, Hong TS, Wo JY, Kwak EL, Rattner DW, Lauwers GY, Mullen JT. Predictors of Lymph Node Metastasis in Western Early Gastric Cancer. J Gastrointest Surg. 2016;20:531-538. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 30] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 12. | Wang S, Zhang Z, Liu M, Li S, Jiang C. Endoscopic Resection Compared with Gastrectomy to Treat Early Gastric Cancer: A Systematic Review and Meta-Analysis. PLoS One. 2015;10:e0144774. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 25] [Article Influence: 2.5] [Reference Citation Analysis (1)] |
| 13. | Kim HW, Lee YJ, Kim JH, Park JJ, Youn YH, Park H, Kim JW, Choi SH, Noh SH. The role of tumor size in surgical decision making after endoscopic resection for early gastric cancer. Surg Endosc. 2016;30:2799-2803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 6] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 14. | Brisinda G, Crocco A, Tomaiuolo P, Santullo F, Mazzari A, Vanella S. Extended or limited lymph node dissection? Ann Surg. 2012;256:e30-e31. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 15. | Gotoda T. Endoscopic resection of early gastric cancer. Gastric Cancer. 2007;10:1-11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 479] [Cited by in RCA: 495] [Article Influence: 27.5] [Reference Citation Analysis (1)] |
| 16. | Bollschweiler E, Berlth F, Baltin C, Mönig S, Hölscher AH. Treatment of early gastric cancer in the Western World. World J Gastroenterol. 2014;20:5672-5678. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 50] [Cited by in RCA: 59] [Article Influence: 5.4] [Reference Citation Analysis (1)] |
| 17. | Espinel J, Pinedo E, Ojeda V, Del Rio MG. Treatment modalities for early gastric cancer. World J Gastrointest Endosc. 2015;7:1062-1069. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 19] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 18. | Japanese Gastric Cancer Association. Japanese gastric cancer treatment guidelines 2018 (5th edition). Gastric Cancer. 2021;24:1-21. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 735] [Cited by in RCA: 1338] [Article Influence: 334.5] [Reference Citation Analysis (2)] |
| 19. | Hu W, Ai XB, Zhu YM, Han TM, Shen B, Pan WS. Combination of Paris and Vienna classifications may optimize follow-up of gastric epithelial neoplasia patients. Med Sci Monit. 2015;21:992-1001. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 20. | Kuan J, Ratcliffe E, Hayes S, McGrath S, Ang Y. Accuracy of the revised Vienna Classification for predicting postendoscopic resection outcomes for gastric and oesophageal neoplasms: a retrospective cohort study of patients from a UK tertiary referral centre. J Clin Pathol. 2020;73:493-501. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.2] [Reference Citation Analysis (0)] |
| 21. | Arribas Anta J, Dinis-Ribeiro M. Early gastric cancer and Artificial Intelligence: Is it time for population screening? Best Pract Res Clin Gastroenterol. 2021;52-53:101710. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 22. | Yao K, Uedo N, Kamada T, Hirasawa T, Nagahama T, Yoshinaga S, Oka M, Inoue K, Mabe K, Yao T, Yoshida M, Miyashiro I, Fujimoto K, Tajiri H. Guidelines for endoscopic diagnosis of early gastric cancer. Dig Endosc. 2020;32:663-698. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 64] [Cited by in RCA: 132] [Article Influence: 26.4] [Reference Citation Analysis (0)] |
| 23. | Miyaoka M, Yao K, Tanabe H, Kanemitsu T, Otsu K, Imamura K, Ono Y, Ishikawa S, Yasaka T, Ueki T, Ota A, Haraoka S, Iwashita A. Diagnosis of early gastric cancer using image enhanced endoscopy: a systematic approach. Transl Gastroenterol Hepatol. 2020;5:50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 11] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 24. | Ahmed Y, Othman M. EMR/ESD: Techniques, Complications, and Evidence. Curr Gastroenterol Rep. 2020;22:39. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 67] [Cited by in RCA: 60] [Article Influence: 12.0] [Reference Citation Analysis (0)] |
| 25. | Ishikawa S, Togashi A, Inoue M, Honda S, Nozawa F, Toyama E, Miyanari N, Tabira Y, Baba H. Indications for EMR/ESD in cases of early gastric cancer: relationship between histological type, depth of wall invasion, and lymph node metastasis. Gastric Cancer. 2007;10:35-38. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 101] [Cited by in RCA: 110] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 26. | Min YW, Min BH, Lee JH, Kim JJ. Endoscopic treatment for early gastric cancer. World J Gastroenterol. 2014;20:4566-4573. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 39] [Cited by in RCA: 40] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 27. | Bausys R, Bausys A, Maneikis K, Belogorceva V, Stratilatovas E, Strupas K. Safety of expanded criteria for endoscopic resection of early gastric cancer in a Western cohort. BMC Surg. 2018;18:79. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 6] [Article Influence: 0.9] [Reference Citation Analysis (0)] |
| 28. | Kume K. Endoscopic therapy for early gastric cancer: standard techniques and recent advances in ESD. World J Gastroenterol. 2014;20:6425-6432. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 18] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 29. | Fan T, Sun Q, Cao S, Fan X, Huang Q, Zhang S, Lv Y, Zhang X, Ling T, Wang L, Zou X, Xu G. Clinical outcomes of early gastric cardiac cancer treated with endoscopic submucosal dissection in patients with different indications. BMC Gastroenterol. 2021;21:119. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 30. | Kato M, Yahagi N. Advanced Endoscopic Treatment of Gastric and Duodenal Neoplasms: Beyond Standard EMR and ESD. Am J Gastroenterol. 2018;113:1423-1426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 10] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 31. | Wang Z, Zhang X, Hu J, Zeng W, Zhou Z. Clinicopathological features and outcomes in patients undergoing radical resection for early gastric cancer with signet ring cell histology. J Visc Surg. 2015;152:357-361. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 20] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 32. | Yang K, Lu L, Liu H, Wang X, Gao Y, Yang L, Li Y, Su M, Jin M, Khan S. A comprehensive update on early gastric cancer: defining terms, etiology, and alarming risk factors. Expert Rev Gastroenterol Hepatol. 2021;15:255-273. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 42] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 33. | Hiki N, Nunobe S. Laparoscopic endoscopic cooperative surgery (LECS) for the gastrointestinal tract: Updated indications. Ann Gastroenterol Surg. 2019;3:239-246. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 36] [Cited by in RCA: 48] [Article Influence: 8.0] [Reference Citation Analysis (1)] |
| 34. | Abe N, Takeuchi H, Ooki A, Nagao G, Masaki T, Mori T, Sugiyama M. Recent developments in gastric endoscopic submucosal dissection: towards the era of endoscopic resection of layers deeper than the submucosa. Dig Endosc. 2013;25 Suppl 1:64-70. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 35. | Aisu Y, Yasukawa D, Kimura Y, Hori T. Laparoscopic and endoscopic cooperative surgery for gastric tumors: Perspective for actual practice and oncological benefits. World J Gastrointest Oncol. 2018;10:381-397. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 16] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 36. | Nunobe S, Hiki N, Gotoda T, Murao T, Haruma K, Matsumoto H, Hirai T, Tanimura S, Sano T, Yamaguchi T. Successful application of laparoscopic and endoscopic cooperative surgery (LECS) for a lateral-spreading mucosal gastric cancer. Gastric Cancer. 2012;15:338-342. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 106] [Article Influence: 8.2] [Reference Citation Analysis (0)] |
| 37. | Matsuda T, Nunobe S, Ohashi M, Hiki N. Laparoscopic endoscopic cooperative surgery (LECS) for the upper gastrointestinal tract. Transl Gastroenterol Hepatol. 2017;2:40. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 38. | Gotoda T, Kusano C, Moriyasu F. Future perspective of gastric cancer endotherapy. Ann Transl Med. 2014;2:25. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 11] [Reference Citation Analysis (0)] |
| 39. | Min JS, Seo KW, Jeong SH. Choice of LECS Procedure for Benign and Malignant Gastric Tumors. J Gastric Cancer. 2021;21:111-121. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 40. | Saito H, Nishimura A, Sakimura Y, Tawara H, Hayashi K, Kato K, Tsuji T, Yamamoto D, Kitamura H, Kadoya S, Bando H. Closed laparoscopic and endoscopic cooperative surgery for early gastric cancer with difficulty in endoscopic submucosal dissection: a report of three cases. Surg Case Rep. 2020;6:235. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 2] [Cited by in RCA: 6] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 41. | Li H, Chen L, Huo Z, Xi H, Cui J, Zhao X. Defining a Subgroup Treatable for Laparoscopic and Endoscopic Cooperative Surgery in Undifferentiated Early Gastric Cancer: the Role of Lymph Node Metastasis. J Gastrointest Surg. 2015;19:1763-1768. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 42. | Ntourakis D, Mavrogenis G. Cooperative laparoscopic endoscopic and hybrid laparoscopic surgery for upper gastrointestinal tumors: Current status. World J Gastroenterol. 2015;21:12482-12497. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 57] [Cited by in RCA: 52] [Article Influence: 5.2] [Reference Citation Analysis (0)] |
| 43. | Oh SE, An JY, Choi MG, Sohn TS, Bae JM, Kim S, Lee JH. Long term oncological outcome of patients with grossly early gastric cancer-mimicking advanced gastric cancer. Eur J Surg Oncol. 2020;46:1262-1268. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 44. | Kano K, Yamada T, Yamamoto K, Komori K, Watanabe H, Takahashi K, Maezawa Y, Fujikawa H, Numata M, Aoyama T, Tamagawa H, Cho H, Yukawa N, Yoshikawa T, Rino Y, Masuda M, Ogata T, Oshima T. Evaluation of Lymph Node Staging Systems as Independent Prognosticators in Remnant Gastric Cancer Patients with an Insufficient Number of Harvested Lymph Nodes. Ann Surg Oncol. 2021;28:2866-2876. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 15] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 45. | Milhomem LM, Milhomem-Cardoso DM, da Mota OM, Mota ED, Kagan A, Filho JBS. Risk of lymph node metastasis in early gastric cancer and indications for endoscopic resection: is it worth applying the east rules to the west? Surg Endosc. 2021;35:4380-4388. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 4] [Cited by in RCA: 22] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 46. | Abe I, Kinoshita T, Kaito A, Sunagawa H, Watanabe M, Sugita S, Tonouchi A, Sato R. Five-year Survival Associated with Stage I Gastric Cancer after Resection of Early Recurrence at Nodal Station No. 14v: a Case Report. J Gastric Cancer. 2017;17:186-191. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3] [Cited by in RCA: 4] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 47. | Aizawa M, Honda M, Hiki N, Kinoshita T, Yabusaki H, Nunobe S, Shibasaki H, Matsuki A, Watanabe M, Abe T. Oncological outcomes of function-preserving gastrectomy for early gastric cancer: a multicenter propensity score matched cohort analysis comparing pylorus-preserving gastrectomy versus conventional distal gastrectomy. Gastric Cancer. 2017;20:709-717. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (1)] |
| 48. | Asakawa Y, Ohtaka M, Maekawa S, Fukasawa M, Nakayama Y, Yamaguchi T, Inoue T, Uetake T, Sakamoto M, Sato T, Kawaguchi Y, Fujii H, Mochizuki K, Hada M, Oyama T, Yasumura T, Omata K, Nishiyama A, Naito K, Hata H, Haba Y, Miyata K, Saitoh H, Yamadera Y, Miura K, Kawaoi A, Abe T, Tsunoda H, Honda Y, Kurosaki M, Enomoto N. Stratifying the risk of lymph node metastasis in undifferentiated-type early gastric cancer. World J Gastroenterol. 2015;21:2683-2692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 21] [Article Influence: 2.1] [Reference Citation Analysis (1)] |
| 49. | Roviello F, Rossi S, Marrelli D, Pedrazzani C, Corso G, Vindigni C, Morgagni P, Saragoni L, de Manzoni G, Tomezzoli A. Number of lymph node metastases and its prognostic significance in early gastric cancer: a multicenter Italian study. J Surg Oncol. 2006;94:275-80; discussion 274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 113] [Cited by in RCA: 129] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 50. | Zhao X, Cai A, Xi H, Chen L, Peng Z, Li P, Liu N, Cui J, Li H. Predictive Factors for Lymph Node Metastasis in Undifferentiated Early Gastric Cancer: a Systematic Review and Meta-analysis. J Gastrointest Surg. 2017;21:700-711. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 27] [Article Influence: 3.4] [Reference Citation Analysis (0)] |
| 51. | Kim DG, An JY, Kim H, Shin SJ, Choi S, Seo WJ, Roh CK, Cho M, Son T, Kim HI, Cheong JH, Hyung WJ, Noh SH, Choi YY. Clinical Implications of Microsatellite Instability in Early Gastric Cancer. J Gastric Cancer. 2019;19:427-437. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 8] [Cited by in RCA: 19] [Article Influence: 3.2] [Reference Citation Analysis (0)] |
| 52. | Kinami S, Kosaka T. Laparoscopic sentinel node navigation surgery for early gastric cancer. Transl Gastroenterol Hepatol. 2017;2:42. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 8] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 53. | Hiramatsu Y, Takeuchi H, Goto O, Kikuchi H, Kitagawa Y. Minimally Invasive Function-Preserving Gastrectomy with Sentinel Node Biopsy for Early Gastric Cancer. Digestion. 2019;99:14-20. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 54. | Brisinda G, Chiarello MM, Crocco A, Adams NJ, Fransvea P, Vanella S. Postoperative mortality and morbidity after D2 lymphadenectomy for gastric cancer: A retrospective cohort study. World J Gastroenterol. 2022;28:381-398. [RCA] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 15] [Article Influence: 5.0] [Reference Citation Analysis (4)] |
| 55. | An L, Gaowa S, Cheng H, Hou M. Long-Term Outcomes Comparison of Endoscopic Resection With Gastrectomy for Treatment of Early Gastric Cancer: A Systematic Review and Meta-Analysis. Front Oncol. 2019;9:725. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 8] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 56. | Lee SH, Jang DK, Yoo MW, Hwang SH, Ryu SY, Kwon OK, Hur H, Man Yoon H, Eom BW, Ahn HS, Son T, Song KY, Lee HH, Choi MG, An JY, Lee SI, Lee KH, Ahn S, Park YS, Park DJ; Efficacy and Safety of DWJ1319 in the Prevention of Gallstone Formation after Gastrectomy in Patient with Gastric Cancer: A Multicenter, Randomized, Double-blind, Placebo-controlled Study (PEGASUS-D) Group. Efficacy and Safety of Ursodeoxycholic Acid for the Prevention of Gallstone Formation After Gastrectomy in Patients With Gastric Cancer: The PEGASUS-D Randomized Clinical Trial. JAMA Surg. 2020;155:703-711. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 40] [Cited by in RCA: 35] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 57. | Pitt HA, Nakeeb A. Prevention of Gallstone Formation After Gastrectomy. JAMA Surg. 2020;155:712. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 58. | Bencini L, Marchet A, Alfieri S, Rosa F, Verlato G, Marrelli D, Roviello F, Pacelli F, Cristadoro L, Taddei A, Farsi M; Italian Research Group for Gastric Cancer (GIRCG). The Cholegas trial: long-term results of prophylactic cholecystectomy during gastrectomy for cancer-a randomized-controlled trial. Gastric Cancer. 2019;22:632-639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 25] [Cited by in RCA: 22] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 59. | Kimura J, Kunisaki C, Takagawa R, Makino H, Ueda M, Ota M, Oba M, Kosaka T, Akiyama H, Endo I. Is Routine Prophylactic Cholecystectomy Necessary During Gastrectomy for Gastric Cancer? World J Surg. 2017;41:1047-1053. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 15] [Article Influence: 1.7] [Reference Citation Analysis (0)] |