Published online Jun 21, 2022. doi: 10.3748/wjg.v28.i23.2582
Peer-review started: October 30, 2021
First decision: March 11, 2022
Revised: March 25, 2022
Accepted: May 7, 2022
Article in press: May 7, 2022
Published online: June 21, 2022
Processing time: 229 Days and 3.9 Hours
Infliximab trough level (ITL) severely affects therapeutic outcomes of Crohn’s disease (CD) patients under infliximab (IFX). Recently, frontier research has focused on identifying ITL based on different therapeutic targets. Although previous studies have elaborated clinical value of ITL monitoring on short-term outcomes in CD patients during therapy, studies contraposing the predictive value of ITL on long-term endoscopic outcomes in CD patients are still scarce domestically and overseas.
To explore the predictive value of ITL in combination with inflammatory biomarkers on long-term endoscopic outcomes in CD with clinical remission during IFX maintenance therapy.
CD patients with endoscopic remission under long-term IFX maintenance therapy in the First Affiliated Hospital of Zhejiang Chinese Medicine University from January 2012 to December 2020 were collected. ITL and inflammatory biomarkers were continuously monitored during the therapy. The Step I study was conducted from weeks 14 to 54 of IFX treatment. The Step II study was conducted from weeks 54 to 108 of IFX treatment. Endoscopic outcomes were defined as endoscopic activity (Crohn’s disease endoscopic index of severity score > 2 points or Rutgeerts score > i1) and endoscopic remission (Crohn’s disease endoscopic index of severity score ≤ 2 points or Rutgeerts ≤ i1). Endoscopic relapse free survival was defined as endoscopic remission at the beginning of the study stage and maintaining endoscopic remission during the study stage.
At week 14, low ITL [odds ratio (OR) = 0.666, 95% confidence interval (CI): 0.514-0.862, P < 0.01] and high fecal calprotectin (FCP) level (OR = 1.002, 95%CI: 1.001-1.004, P < 0.01) increased the risk of endoscopic activity at week 54. At week 54, low ITL (OR = 0.466, 95%CI: 0.247-0.877, P < 0.01) and high C-reactive protein (CRP) level (OR = 1.590, 95%CI: 1.007-2.510, P < 0.01) increased the risk of endoscopic activity at week 108. At week 14, ITL ≤ 5.60 μg/mL [area under the curve (AUC) = 0.83, 95%CI: 0.73-0.90, P < 0.001] and FCP > 238 μg/g (AUC = 0.82, 95%CI: 0.72-0.89, P < 0.001) moderately predicted endoscopic activity at week 54. ITL ≤ 5.60 μg/mL in combination with FCP > 238 μg/g indicated 82.0% possibility of endoscopic activity. At week 54, ITL ≤ 2.10 μg/mL (AUC = 0.85, 95%CI: 0.72-0.93, P < 0.001) and CRP > 3.00 mg/L (AUC = 0.73, 95%CI: 0.60-0.84, P = 0.012) moderately predicted moderate endoscopic activity at week 108. ITL ≤ 2.10 μg/mL in combination with CRP > 3.00 mg/L indicated 100.0% possibility of endoscopic activity. From weeks 14 to 54 of IFX treatment, patients with ITL > 5.60 μg/mL had higher rate of endoscopic relapse free survival than those with ITL ≤ 5.60 μg/mL (95.83% vs 46.67%). From weeks 54 to 108 of IFX treatment, patients with ITL > 2.10 μg/mL had higher rate of endoscopic survival free relapsed rate than those with ITL ≤ 2.10 μg/mL (92.68% vs 30.77%).
Combination of ITL, CRP, and FCP contribute to long-term endoscopic prognosis monitoring. During IFX maintenance treatment, low ITL, high CRP level, and high FCP level were independent risk factors of CD patients with clinical remission in adverse endoscopy outcomes within 1-year follow-up.
Core Tip: Previous investigations, contraposing Crohn’s disease patients under infliximab (IFX) maintenance therapy, have indicated that higher IFX trough levels (ITLs) were associated with sustained drug response and clinical remission in inflammatory bowel disease patients, while lower ITLs were linked to secondary unresponsiveness of IFX. Currently, endoscopic remission or mucosal healing has been considered the main goal of biological therapy. Our study manifested that Crohn’s disease patients with higher levels of IFX blood concentration and lower levels of inflammatory biomarkers tended to have a better long-term endoscopic prognosis. Combining ITL, fecal calprotectin and C-reactive protein monitoring was helpful for the timely adjustment of IFX treatment strategy.
- Citation: Cao WT, Huang R, Liu S, Fan YH, Xu MS, Xu Y, Ni H. Infliximab trough level combined with inflammatory biomarkers predict long-term endoscopic outcomes in Crohn’s disease under infliximab therapy. World J Gastroenterol 2022; 28(23): 2582-2596
- URL: https://www.wjgnet.com/1007-9327/full/v28/i23/2582.htm
- DOI: https://dx.doi.org/10.3748/wjg.v28.i23.2582
Crohn’s disease (CD) is a persistently progressive disease with nonspecific inflammation characterized by disease scope involving the whole digestive tract and disease depth involving the whole intestinal wall. The accumulation damage of intestinal walls contributes to the occurrence of stenosis, fistula and even abscess, reducing the life quality. Therefore, recent clinical studies have consistently concluded that therapeutic strategies and targets play key roles in controlling CD progression. Setting different therapeutic targets will have different disease outcomes. Clinical response, focusing only on the improvement of clinical symptoms, can improve the quality of daily life but not affect long-term treatment outcomes. CD patients who do not achieve deep remission may be aggravated persistently, while achieving deep remission could reduce long-term hospitalization and surgery rates. Deep remission is mainly defined in previous studies as endoscopic remission or mucosal healing. Biologics, as an important step in the therapeutic strategy of CD, can effectively control the disease progression if conducted early and completely.
In consideration of the wide use of infliximab (IFX), precisely predicting the long-term endoscopic outcomes is stressed by more and more inflammatory bowel disease (IBD) physicians. Although the IFX trough level (ITL) has been proven to be closely related to the outcome of CD, ITL alone may be biased in predicting the outcome of CD. Monitoring inflammation biomarkers is one of the important links of IFX therapy, including C-reaction protein (CRP), fecal calprotectin (FCP), etc. High inflammatory load affects the pharmacokinetics of IFX, inducing secondary nonresponse by decreasing blood drug concentration. Currently, it is believed that inflammatory biomarkers are good predictors of disease activity, but there is still a lack of reliable evidence for predicting disease remission. Therefore, this study intends to evaluate long-term endoscopic outcomes of CD patients receiving IFX treatment by combining the blood drug concentration and inflammatory biomarkers.
A single-center retrospective research was implemented at the First Affiliated Hospital of Zhejiang Chinese Medical University. CD patients under IFX therapy from January 2012 to December 2020 were collected. One hundred and eighty-one CD patients underwent IFX treatment. One hundred and fifty-one CD patients underwent endoscopy as well as serum concentration monitoring at week 14 after the third dose of IFX induction therapy. Inclusion criteria: (1) Endoscopic remission at week 14 [Crohn’s disease endoscopic index of severity (CDEIS) score ≤ 2 points or Rutgeerts ≤ i1]; (2) Clinical remission after IFX induction therapy without corticosteroids more than 6 mo; and (3) Therapeutic strategy during maintenance stage was designed as IFX 5 mg/kg every 8 wk combined with azathioprine (AZA) 50 mg every day. Therapeutic strategic was modulated if CD patients were confronted with clinical relapse or secondary loss of response (LOR), and data analysis focused on the treatment course when patients received IFX 5 mg/kg and AZA therapy regularly. Secondary LOR was defined as a recurrence of the disease during IFX maintenance therapy. Two criteria were met to determine LOR: The recurrence of symptoms of IBD in clinical remission after induction therapy and symptoms caused by the inflammatory activity of IBD itself. Clinical relapse means Crohn’s disease activity index > 150 points. Blood drug concentration monitoring and clinical, laboratory, endoscopic and imaging evaluation were implemented every 2 mo since the third dose of IFX induction therapy in all patients. The study was divided into two stages, step I study period defined as IFX maintenance therapy during week 14 to week 54 and step II study period defined as IFX maintenance therapy during week 54 to week 108.
General data included age, sex, course, disease location, disease behavior, medication history and history of intestinal surgery. Laboratory indicators include white blood cell count, blood platelet count, CRP, erythrocyte sedimentation rate, serum albumin, FCP, ITL and anti-IFX antibody. Evaluation indicators of disease severity included Crohn’s disease activity index score on clinical severity, CDEIS score on endoscopic severity in CD patients without intestinal surgery and Rutgeerts score on endoscopic severity in CD patients with intestinal surgery.
Endoscopic outcomes at week 54 and week 108 after IFX initial therapy were evaluated by specialist physicians on IBD under electronic colonoscopy. Endoscopic remission was defined as CDEIS score ≤ 2 or Rutgeerts score ≤ i1, and endoscopic activity was defined as CDEIS score > 2 or Rutgeerts score > i1. Survival outcomes during IFX maintenance therapy were concentrated on endoscopic relapse-free survival, defined as sustained endoscopic remission during step I study period or step II study period.
Descriptive statistical analysis was used to describe characteristics of CD patients. Number of cases (percentage) was used to describe categorial variable. mean ± SD was used to describe continuous variable. Nonparametric Mann-Whitney test was used to compare two groups in enumeration data or measurement data without normal distribution. Two-sample t test was used to compare two groups in measurement data with normal distribution. One-way analysis of variance was used to compare multi-group if data satisfied homogeneity of variance. Nonparametric Kruskal-Wallis test was used to compare multi-group if data not satisfied homogeneity of variance. SPSS 23.0 (Armonk, NY, United States) was used to analyze differences between groups. A P value < 0.05 was considered significant.
Receiver-operating characteristic (ROC) analysis was used to identify the best cut off level of ITL on predicting endoscopic remission as well as sensitivity, specificity, positive predictive value, negative predictive value, area under the curve and Youden Index. Univariate logistic regression analysis was used to identify the association between endoscopic activity and predictors. Log-rank test was used to identify the association between endoscopic relapse and predictors. GraphPad Prism9.0 (San Diego, CA, United States) was used to draw histograms and survival analysis curves and implement log-rank test. MedCalc19.0 was used to draw ROC curve and analyze the predictive value of indicators on endoscopic outcomes. A P value < 0.05 was considered significant.
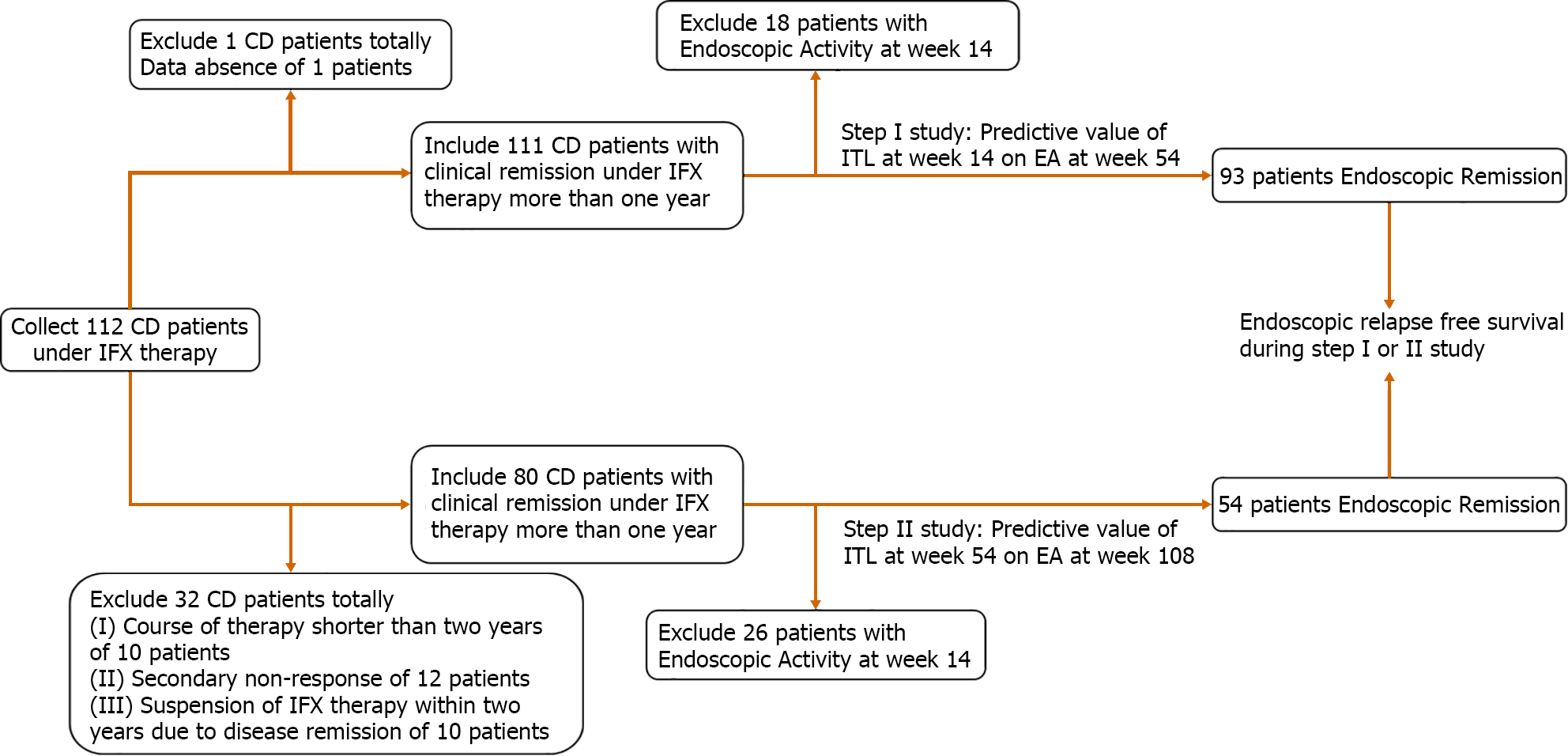
In total, the study cohort collected 112 CD patients achieving clinical remission after IFX induction therapy. In step I study, 19 CD patients were excluded due to data absence (n = 1, 5.26%) and endoscopic activity at week 14 (n = 18, 94.74%), while 93 CD patients with endoscopic remission at week 14 were included. In step II study, 58 CD patients were excluded due to course of therapy shorter than 2 years (n = 10, 17.24%), secondary non-response of IFX (n = 12, 20.69%), suspension of IFX therapy within 2 years for disease remission (n = 10, 17.24%) and endoscopic activity at week 54 (n = 26, 44.83%), while 54 CD patients with endoscopic remission at week 54 were included. These 12 patients did not satisfy indications of operation and received hormonotherapy as the primary choice to alleviate disease, for our center lacked other biological agents at that time. All CD patients under IFX maintenance therapy were combined with AZA (Figure 1). The dose of IFX was 5 mg/kg every 8 wk, and the dose of AZA was 50 mg every day. Characteristics of CD patients included in study are shown in Table 1.
| Variable | Week 14 (after initial IFX therapy) | Week 54 (after initial IFX therapy) n = 54 | ||||
| Total, n = 93 | ER at week 54, n = 67 | EA at week 54, n = 26 | Total, n = 54 | ER at week 108, n = 42 | EA at week 108, n = 12 | |
| Median age in yr, mean ± SD | 28.96 ± 9.37 | 29.03 ± 10.09 | 28.77 ± 7.39 | 27.57 ± 10.13 | 26.93 ± 9.87 | 29.83 ± 11.13 |
| Course in yr, median (IQR) | 3.0 (1.0, 6.0) | 2.0 (1.0, 6.0) | 3.5 (2.0, 8.3) | 2.0 (1.0, 5.0) | 2.0 (1.0, 5.0) | 2.0 (1.3, 8.5) |
| Male sex, n (%) | 55 (59.1) | 43 (64.2) | 12 (46.2) | 35 (64.8) | 28 (66.7) | 7 (58.3) |
| Disease location, n (%) | ||||||
| L1 (terminal ileum) | 18 (19.4) | 14 (20.9) | 4 (15.4) | 10 (18.5) | 9 (21.4) | 1 (8.3) |
| L2 (colon) | 11 (11.8) | 6 (9.0) | 5 (19.2) | 5 (9.3) | 4 (9.5) | 1 (8.3) |
| L3 (ileocolon) | 64 (68.8) | 47 (70.1) | 17 (65.4) | 39 (72.2) | 29 (69.0) | 10 (83.3) |
| L4 (upper digestive tract) | 22 (23.7) | 15 (22.4) | 7 (26.9) | 14 (25.9) | 11(26.2) | 3 (25.0) |
| Disease behavior, n (%) | ||||||
| B1 (no) | 19 (20.4) | 15 (22.4) | 4 (15.4) | 13 (24.1) | 9 (21.4) | 4 (33.3) |
| B2 (stenosis) | 11 (11.8) | 9 (13.4) | 2 (7.7) | 6 (11.1) | 5 (11.9) | 1 (8.3) |
| B3 (penetration) | 40 (43.0) | 27 (40.3) | 13 (50.0) | 23 (42.6) | 18 (42.9) | 5 (41.7) |
| B2 (stenosis) + B3 (penetration) | 23 (24.7) | 16 (23.9) | 7 (26.9) | 12 (22.2) | 10 (23.8) | 2 (16.7) |
| Perianal diseases, n (%) | 55 (59.1) | 38 (56.7) | 17 (65.4) | 32 (59.3) | 25 (59.5) | 7 (58.3) |
| Previous medical therapy, n (%) | 64 (68.8) | 46 (68.7) | 18 (69.2) | 34 (63.0) | 25 (59.5) | 9 (75.0) |
| Previous surgical therapy, n (%) | 15 (16.1) | 11 (16.4) | 4 (15.4) | 8 (14.8) | 8 (19.0) | 0 (0.0) |
| Laboratory indicators, mean ± SD | ||||||
| Fecal calprotectin, μg/g | 399.96 ± 562.47 | 178.62 ± 242.38 | 970.35 ± 734.49 | 353.17 ± 557.71 | 178.57 ± 276.56 | 964.25 ± 830.56 |
| IFX trough level, μg/ml | 6.12 ± 3.72 | 7.23 ± 3.48 | 3.25 ± 2.67 | 3.80 ± 2.25 | 4.37 ± 2.02 | 1.80 ± 1.90 |
| White blood count, × 109/L | 5.32 ± 1.87 | 5.16 ± 1.47 | 5.73 ± 2.65 | 5.27 ± 1.41 | 5.27 ± 1.43 | 5.28 ± 1.41 |
| Hematoglobin, g/L | 128.37 ± 20.42 | 130.96 ± 20.03 | 121.69 ± 20.27 | 136.59 ± 16.06 | 136.79 ± 16.94 | 135.92 ± 13.13 |
| Platelet, × 109/L | 210.66 ± 69.77 | 202.36 ± 58.34 | 232.04 ± 90.94 | 205.02 ± 46.19 | 204.62 ± 45.82 | 206.42 ± 49.54 |
| Erythrocyte sedimentation rate, mm/h | 9.88 ± 12.25 | 6.89 ± 7.96 | 17.46 ± 17.28 | 7.31 ± 10.35 | 6.33 ± 6.89 | 10.75 ± 17.97 |
| Albumin, g/L | 42.13 ± 4.17 | 42.65 ± 3.93 | 40.77 ± 4.54 | 44.51 ± 3.40 | 44.99 ± 3.38 | 42.85 ± 3.02 |
| C-reactive protein, mg/dl | 2.98 ± 5.43 | 1.82 ± 2.77 | 5.96 ± 8.69 | 1.99 ± 3.18 | 1.23 ± 1.79 | 4.66 ± 5.19 |
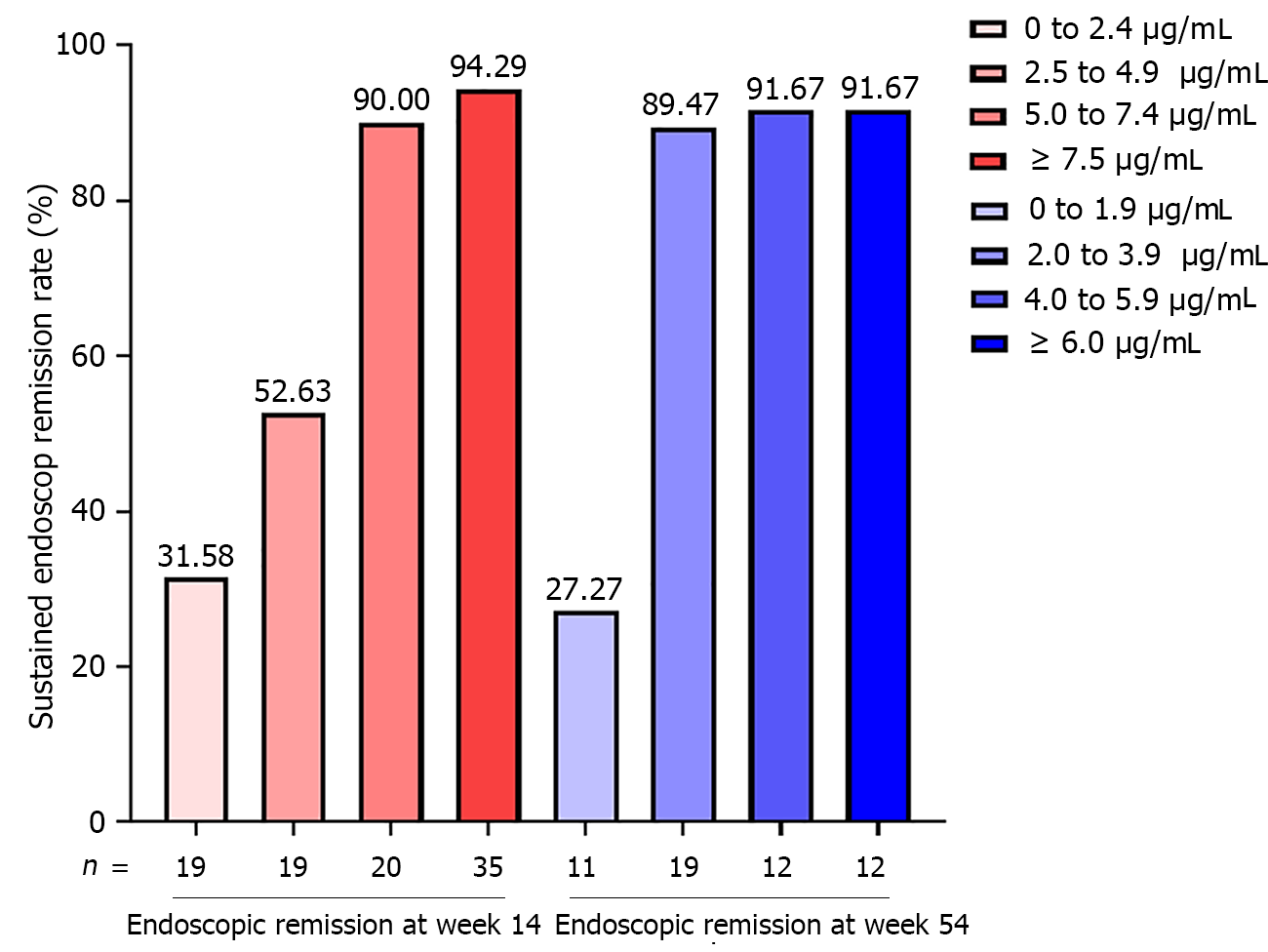
In step I study, 67/93 CD patients (72.04%) sustained endoscopic remission at week 54 among. Multivariable regression analysis revealed that only ITL (OR = 0.666, 95%CI: 0.514-0.862, P = 0.002) and FCP (OR = 1.002, 95%CI: 1.001-1.004, P = 0.002) were independent risk of endoscopic activity at week 54 (Table 2). Based on incremental gain analysis, an ITL range of 5.0-7.4 μg/mL was correlated with sustained endoscopic remission rate of more than 85% (Figure 2).
| Variable | Week 14 (after initial IFX therapy), n = 93 | Week 14 (after initial IFX therapy), n = 54 | ||||||
| Predict endoscopic relapse at week 54 | Predict endoscopic relapse at week 108 | |||||||
| Univariate analysis | Multivariable analysis | Univariate analysis | Multivariable analysis | |||||
| OR (95%CI) | P value | OR (95%CI) | P value | OR (95%CI) | P value | OR (95%CI) | P value | |
| Median age in year, median (IQR) | 0.997 (0.950-1.047) | 0.904 | — | — | 1.028 (0.967-1.093) | 0.381 | — | — |
| Course in year, median (IQR) | 1.054 (0.945-1.176) | 0.346 | — | — | 1.065 (0.906-1.253) | 0.444 | — | — |
| Male sex, n (%) | 0.478 (0.191-1.199) | 0.116 | — | — | 0.700 (0.188-2.607) | 0.595 | — | — |
| Disease location, n (%) | ||||||||
| L1 (terminal ileum) | 0.413 (0.114-1.495) | 0.178 | — | — | 1.158 0.117-11.454) | 0.900 | — | — |
| L2 (colon) | 1.453 (0.430-4.908) | 0.548 | — | — | 3.000 (0.341-26.427) | 0.322 | — | — |
| L3 (ileocolon) | — | — | — | — | — | — | — | — |
| L4 (upper digestive tract) | 1.277 (0.452-3.612) | 0.645 | — | — | 0.939 (0.215-4.113) | 0.934 | — | — |
| Disease behavior, n (%) | ||||||||
| B1 (no) | — | — | — | — | — | — | — | — |
| B2 (stenosis) | 0.889 (0.345-2.294) | 0.808 | — | — | 0.600 (0.141-2.561) | 0.490 | — | — |
| B3 (penetration) | 1.860 (0.658-5.264) | 0.242 | — | — | 0.700 (0.188-2.607) | 0.595 | — | — |
| B2 (stenosis) + B3 (penetration) | — | — | — | — | — | — | — | — |
| Perianal diseases, n (%) | 1.442 (0.562-3.696) | 0.446 | — | — | 0.952 (0.259-3.502) | 0.941 | — | — |
| Previous medical therapy, n (%) | 1.027 (0.386-2.736) | 0.957 | 2.040 (0.481-8.650) | 0.333 | ||||
| Previous surgical therapy, n (%) | 0.926 (0.266-3.218) | 0.903 | — | — | — | — | — | — |
| Laboratory indicators, median (IQR) | ||||||||
| Fecal calprotectin, μg/g | 1.003 (1.002-1.005) | 0.000 | 1.002 (1.001-1.004) | 0.002 | 1.002 (1.001-1.004) | 0.001 | NS | NS |
| IFX trough level, μg/mL | 0.650 (0.532-0.796) | 0.000 | 0.666 (0.514-0.862) | 0.002 | 0.470 (0.289-0.766) | 0.002 | 0.466 (0.247-0.877) | 0.018 |
| White blood count, x 109/L | 1.167 (0.921-1.478) | 0.201 | — | — | 1.004 (0.636-1.586) | 0.986 | — | — |
| Hematoglobin, g/L | 0.977 (0.954-1.000) | 0.053 | — | — | 0.997 (0.957-1.038) | 0.867 | — | — |
| Platelet, x 109/L | 1.006 (0.999-1.013) | 0.081 | — | — | 1.001 (0.987-1.015) | 0.904 | — | — |
| Erythrocyte sedimentation rate, mm/h | 1.073 (1.028-1.120) | 0.001 | NS | NS | 1.035 (0.978-1.096) | 0.239 | — | — |
| Albumin, g/L | 0.895 (0.800-1.002) | 0.054 | — | — | 0.821 (0.667-1.010) | 0.062 | — | — |
| C-reactive protein, mg/dL | 1.245 (1.080-1.435) | 0.003 | NS | NS | 1.389 (1.070-1.804) | 0.014 | 1.590 (1.007-2.510) | 0.047 |
In step II study, 42/54 CD patients (77.78%) sustained endoscopic remission at week 108. Multivariable regression analysis revealed that only ITL (OR = 0.466, 95%CI: 0.247-0.877, P = 0.018) and CRP (OR = 1.590, 95%CI: 1.007-2.510, P = 0.047) were independent risks of endoscopic activity at week 108 (Table 2). Based on incremental gain analysis, an ITL range of 2.0-3.9 μg/mL was correlated with sustained endoscopic remission rate of more than 85% (Figure 2).
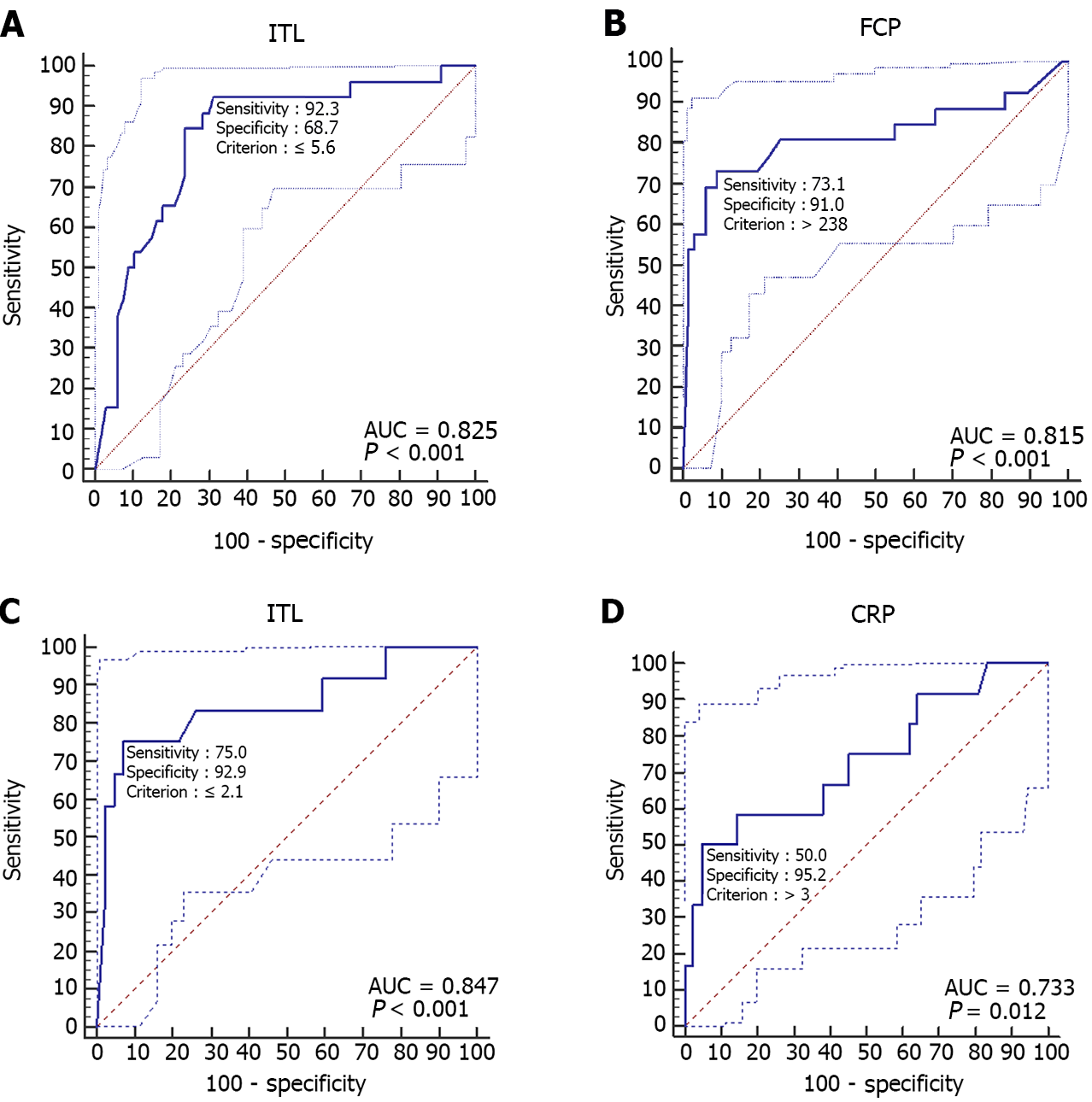
In step I study, the ROC analysis demonstrated that the best cut off level of ITL and FCP at week 14 on predicting endoscopic relapse at week 54 was 5.60 μg/ml (AUC = 0.83, 95%CI: 0.73-0.90, P < 0.001) and 238 μg/g (AUC = 0.82, 95%CI: 0.72-0.89, P < 0.001) (Table 3 and Figure 3). CD patients with ITL ≤ 5.60 μg/ml and FCP > 238 μg/g at week 14 had 82% probability of endoscopic relapse at week 54. However, CD patients with ITL > 5.60 μg/ml and FCP ≤ 238 μg/g at week 14 had 98% probability of sustained endoscopic remission at week 54.
| Youden index | Sensitivity | Specificity | Positive predictive value | Negative predictive value | Area under the ROC curve | P value | |
| Predictors at week 14 of Endoscopic relapse at week 54 | |||||||
| ITL ≤ 5.6 μg/mL | 0.61 (0.41-0.72) | 0.92 (0.75-0.99) | 0.69 (0.56-0.79) | 0.53 (0.44-0.62) | 0.96 (0.86-0.99) | 0.83 (0.73-0.90) | < 0.001 |
| FCP > 238 μg/g | 0.64 (0.38-0.78) | 0.73 (0.52-0.88) | 0.91 (0.82-0.97) | 0.76 (0.59-0.88) | 0.90 (0.82-0.94) | 0.82 (0.72-0.89) | < 0.001 |
| ITL ≤ 5.6 μg/mL and FCP > 238 μg/g | 0.63 (0.40-0.80) | 0.69 (0.48-0.86) | 0.94 (0.85-0.98) | 0.82 (0.63-0.92) | 0.89 (0.82-0.93) | 0.82 (0.72-0.89) | < 0.001 |
| ITL ≤ 5.6 μg/mL or FCP > 238 μg/g | 0.62 (0.47-0.74) | 0.96 (0.80-1.00) | 0.66 (0.53-0.77) | 0.52 (0.44-0.60) | 0.98 (0.87-1.00) | 0.81 (0.71-0.88) | < 0.001 |
| Predictors at week 54 of Endoscopic relapse at week 108 | |||||||
| ITL ≤ 2.1 μg/mL | 0.68 (0.40-0.87) | 0.75 (0.43-0.95) | 0.93 (0.81-0.99) | 0.75 (0.49-0.90) | 0.93 (0.83-0.97) | 0.85 (0.72-0.93) | < 0.001 |
| CRP > 3.0 mg/dL | 0.45 (0.20-0.68) | 0.50 (0.21-0.79) | 0.95 (0.84-0.99) | 0.75 (0.41-0.93) | 0.87 (0.79-0.92) | 0.73 (0.60-0.84) | 0.012 |
| ITL ≤ 2.1 μg/mL and CRP > 3.0 mg/dL | 0.33 (0.08-0.58) | 0.33 (0.10-0.65) | 1.00 (0.92-1.00) | 1.00 (1.00-1.00) | 0.84 (0.78-0.89) | 0.67 (0.53-0.79) | 0.019 |
| ITL ≤ 2.1 μg/mL or CRP > 3.0 mg/dL | 0.80 (0.50-0.93) | 0.92 (0.62-1.00) | 0.88 (0.74-0.96) | 0.69 (0.49-0.84) | 0.97 (0.85-1.00) | 0.90 (0.79-0.96) | <0.001 |
In step II study, the ROC analysis demonstrated that the best cut off level of ITL and CRP at week 54 on predicting endoscopic relapse at week 108 was 2.10 μg/mL (AUC = 0.85, 95%CI: 0.72-0.93, P < 0.001) and 3.00 mg/L (AUC = 0.73, 95%CI: 0.60-0.84, P = 0.012) (Table 3 and Figure 3). CD patients with ITL ≤ 2.10 μg/mL and CRP > 3.00 mg/L at week 54 had 100% probability of endoscopic relapse at week 108. However, CD patients with ITL > 2.10 μg/mL and CRP ≤ 3.00 mg/L at week 54 had 97% probability of sustained endoscopic remission at week 108.
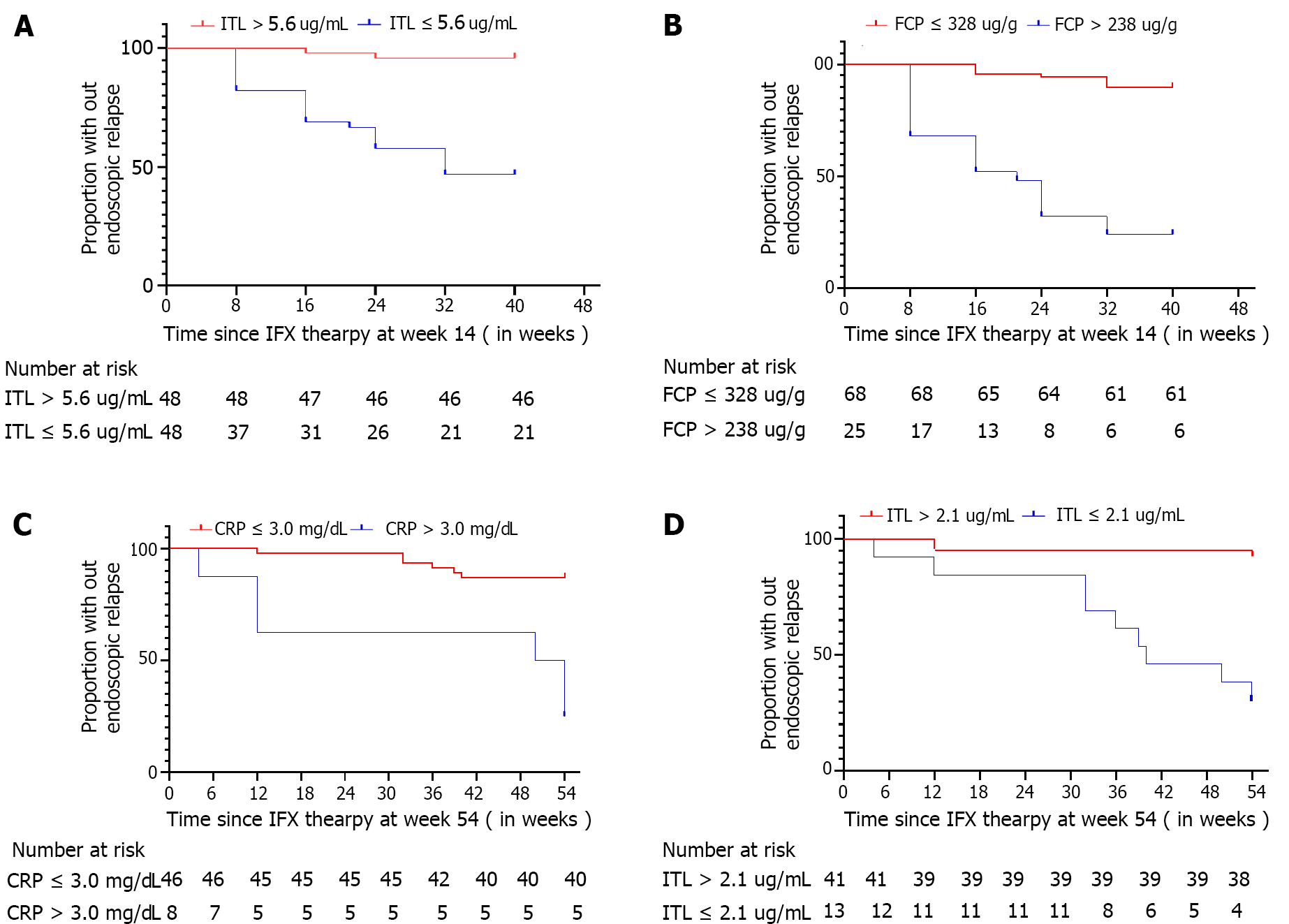
In step I study, 26/93 (27.96%) CD patients had experienced endoscopic relapse from week 14 to week 54 of IFX maintenance therapy. The estimated endoscopic relapse-free rate was 46/48 (95.83%) in CD patients with ITL > 5.6 μg/ml and 21/45 (46.67%) in CD patients with ITL ≤ 5.6 μg/ml. The median time to endoscopic relapse of CD patients with ITL ≤ 5.6 μg/ml was 32.00 wk shorter than those with ITL > 5.6 μg/ml [hazard ratio (HR) = 16.19, 95%CI: 7.44-35.22, P < 0.0001] (Figure 4A). The estimated endoscopic relapse-free rate was 6/25 (24.00%) in CD patients with FCP > 238 μg/g and 61/68 (89.71%) in CD patients with FCP ≤ 238 μg/g. The median time to endoscopic relapse of CD patients with FCP > 238 μg/g was 21.00 wk shorter than those with FCP ≤ 238 μg/g (HR = 11.25, 95%CI: 4.26-29.73, P < 0.0001) (Figure 4B).
In step II study, 12/54 (22.22%) CD patients had experienced endoscopic relapse from week 54 to week 108 of IFX maintenance therapy. The estimated endoscopic relapse-free rate was 38/41 (92.68%) in CD patients with ITL > 2.1 μg/mL and 4/13 (30.77%) in CD patients with ITL ≤ 2.1 μg/mL. The median time to endoscopic relapse of CD patients with ITL ≤ 2.1 μg/mL was 40.00 w, shorter than those with ITL > 2.1 μg/mL (HR = 13.14, 95%CI: 3.07-56.27, P < 0.0001) (Figure 4D). The estimated endoscopic relapse-free rate was 4/8 (50.00%) in CD patients with CRP > 3.00 mg/L and 40/46 (86.96%) in CD patients with CRP ≤ 3.00 mg/L. The median time to endoscopic relapse of CD patients with CRP > 3.00 mg/L was 50.00 wk shorter than those with CRP ≤ 3.00 mg/L (HR = 7.85, 95%CI: 1.31-46.85, P < 0.0001) (Figure 4C).
Several studies have confirmed that different ITLs brought about different outcomes of CD under IFX therapy (Table 4). Tang et al[1] discovered that CD patients achieving mucosal healing at week 14 of IFX therapy with ITL > 2.5 μg/mL at week 14 had 71% chance of mucosal healing at week 54, while patients with ITL < 2.5 μg/mL had only 33% chance. ITL ≥ 3 μg/mL at the beginning of IFX maintenance therapy was confirmed as a predictor of sustained response to IFX in CD patients[2]. Recently a prospective study in Japan verified that CD patients with ITL ≥ 3 μg/mL at week 14 after IFX initial therapy had much better long-term clinical outcomes than patients with ITL < 3 μg/mL, of which survival analysis indicated 100% probability of clinical remission at week 108 in the former and 33.3% probability in the latter[3]. A meta-analysis determined that ITL > 2.0 μg/mL of IBD patients under IFX maintenance therapy contributes to better prognosis such as clinical remission or mucosal healing[4]. Similarly, this study found that CD patients with ITL > 5.6 μg/mL at week 14 had a large chance of achieving sustained endoscopic remission during IFX maintenance therapy as well as CD patients with ITL > 2.1 μg/mL at week 54. Borren et al[5] implemented a multi-center retrospective study and concluded that low ITL in IBD patients during IFX maintenance therapy could not be a good predictor of clinical relapse in the next 2 years, suggesting that proactive therapeutic drug monitoring was not suitable in this group. However, this study discovered that CD patients with ITL ≤ 5.6 μg/mL at week 14 or ITL ≤ 2.1 μg/mL at week 54 were more likely to experience endoscopic relapse during the 1-year follow-up period.
| Study design | Optimal cut-off value | Predictive content | Yes/No, n | SE | SP | PPV | NPV | AUC |
| A retrospective observational single-center study in China | 4.85 μg/mL at week 14 | Mucosal healing (complete absence of any sign of ulceration) | 82/59 | 67% | 80% | 0.80 | ||
| 4.85 μg/mL at week 14 | Mucosal healing (CDEIS of < 3) | 84/57 | 68% | 83% | 0.79 | |||
| 2.85 μg/mL at week 30 | Mucosal healing (complete absence of any sign of ulceration) | 59/50 | 73% | 84% | 0.78 | |||
| 2.85 μg/mL at week 30 | Mucosal healing (CDEIS of < 3) | 62/47 | 68% | 81% | 0.73 | |||
| A retrospective observational single-center study in China | 2.50 μg/mL at week 14 | Mucosal healing (SES-CD/Rutgeerts of 0 or 1) at week 52 | 31/42 | 87% | 60% | 0.70 | ||
| 2.50 μg/mL at week 14 | Sustained remission (no treatment failure, no need for surgery or intensification of IFX nor new introduction during IFX therapy) at week 52 | 70/38 | 64% | 63% | 0.70 | |||
| A prospective multicenter study in Spanish | 3.40 μg/mL | (1) SES-CD<3 for CD patients; (2) Rutgeerts score < i2 for CD patients in the postoperative setting; and (3) Mayo endoscopic score < 2 for UC patients | 58/30 | 60% | 60% | 73% | 42% | 0.63 |
| A multicenter, randomized, double-blind, controlled trial in Europe | 23.10 mg/L at week 2 | Endoscopic remission (CDEIS < 3) at week 12 | 54/52 | 56% | 80% | 72% | 65% | 0.67 |
| 10.00 mg/L at week 6 | Endoscopic remission (CDEIS < 3) at week 12 | 54/52 | 37% | 89% | 76% | 59% | 0.64 | |
| 10.60 mg/L (dose escalation to 10 mg/kg) | The absence of ulcers at week 54 | 85/51 | 94% | 42% | 49% | 92% | 0.71 | |
| A retrospective multicenter study in United States | 9.70 μg/mL | Endoscopic remission (absence of any mucosal break (ulceration or erosion)/Rutgeerts score of ≤ i1) | 62/34 | 57% | 73% | 80% | 48% | 0.65 |
| 9.80 μg/mL | Histologic remissions (absence of active inflammation) | 43/44 | 63% | 66% | 64% | 64% | 0.62 | |
| 2.20 μg/mL | Biochemical remission (CRP ≤ 5 mg/dL) | 48/23 | 92% | 35% | 75% | 67% | 0.64 | |
| A retrospective observational single-center study in Japan | 4.00 μg/mL | Mucosal healing (modified Rutgeerts scoring system: 0 or 1) after 30 days | 20/58 | 71% | 70% | 0.63 | ||
| 0.60 μg/mL | CRP normalization (≤ 0.3 mg/dL) | 28/22 | 73% | 62% | 0.67 | |||
| 1.00 μg/mL | Serum albumin normalization (≥ 4.0 mg/dL) | 17/33 | 67% | 71% | 0.72 | |||
| 1.10 μg/mL | Fecal calprotectin (≥ 300 μg/g) | 13/25 | 72% | 56% | 0.63 | |||
| A retrospective cross-sectional multicenter study in South Korea | 4.20 μg/mL | Mucosal healing (SES-CD = 0) | 51/54 | 65% | 70% | 67% | 68% | 0.68 |
| 3.71 μg/mL | Partial mucosal healing (SES-CD < 3) | 63/42 | 70% | 71% | 79% | 61% | 0.73 | |
| 3.26 μg/mL | Clinical remission (PCDAI < 10) | 95/10 | 71% | 100% | 100% | 73% | 0.90 | |
| 2.52 μg/mL | Biochemical remission (CRP < 0.3 mg/dL) | 87/18 | 86% | 56% | 90% | 46% | 0.71 | |
| A prospective cohort multicenter study in Canada | 8.02 μg/mL | Histologic remission (an absence of active chronic inflammation) | 56/48 | 79% | 68% | 0.72 | ||
| 8.27 μg/mL | Sustained histologic remission (histologic remission documented at both the baseline and follow-up colonoscopies) | 36/16 | 88% | 72% | 0.77 | |||
| A retrospective cross-sectional study in United Kingdom | 7.10 μg/mL | Fistula healing (no spontaneous discharge or no discharge on palpation in the absence of seton drainage) | 18/11 | 78% | 100% | 0.93 | ||
| 7.10 μg/mL | Fistula closure (the absence of an external skin opening) | 13/16 | 64% | 100% | 0.97 |
According to previous studies, the challenge for IBD physicians is to frame the more suitable blood trough level of IFX to achieve better disease prognosis in clinical therapy. The elements associated with the blood trough level of IFX can be classified into three areas. Above all, the better the therapeutic goal desired by IBD physicians or patients, the higher the blood trough level of IFX is required. An observational study contraposing to CD patients with a history of intestinal surgery by Imaeda et al[6] verified that mucosal healing required higher ITLs as more than 4.0 μg/mL compared to those to achieve normalization of routine clinical markers. Papamichael et al[7] considered that ITL surpassing 9.7 μg/mL indicated 80% probability of endoscopic remission in CD patients under IFX maintenance therapy, while ITL surpassing 2.2 μg/mL was associated only with biochemical remission. Recently, a prospective study verified that ITL > 8.0 μg/mL was highly correlated with histological emission and sustained histological remission in IBD patients[8]. Perianal fistula, the most universal complications of CD patients, is another therapeutic goal. A retrospective cross-sectional study by Plevris et al[9] manifested that perianal fistula healing or closure is associated with higher ITLs as more than 7.1 μg/mL.
Secondly, each clinical study had different stages of IFX drug monitoring, especially during maintenance therapy. ITL continues to decrease as time passed during IFX maintenance therapy[10]. A cross-sectional study of IBD patients under IFX therapy with a fixed dose more than 6 mo found that IBD patients with ITL ≥ 3.4 μg/mL had a 73% chance of endoscopic mucosal healing[11]. Kang et al[12] showed that ITL ≥ 5 μg/mL during IFX maintenance therapy could identify mucosal healing in pediatric CD patients with 80% specificity. Feng et al[13] innovatively integrated ITL levels in different time stages to identify endoscopic mucosal healing in CD patients, indicating that patients with ITL > 4.85 μg/mL at week 14 and ITL > 2.85 μg/mL at week 30 had an 80% chance of achieving endoscopic mucosal healing. Based on incremental gain analysis in our study, sustained endoscopic remission rate at week 54 reached only 54.63% at an ITL range of 2.5 to 4.9 μg/mL at week 14 while corresponding numbers at week 108 was 89.47% at an ITL range of 2.0 to 3.9 μg/mL at week 54. Therefore, the study held the view that CD patients with endoscopic remission need higher ITL at the beginning of IFX maintenance therapy (≥ 5.6 μg/mL at week 14) than after IFX maintenance therapy over a half year (≥ 2.1 μg/mL at week 54). In addition, CD patients achieving endoscopic remission after IFX induction therapy and sustained endoscopic remission more than a half year may not need high ITL to maintain endoscopic remission.
The third element is therapeutic optimization of IFX in IBD. Adverse IFX response as high ATI level or low ITL may occur in a few CD patients during IFX maintenance therapy. Several clinical studies held the view that severe inflammatory activity of CD patients could change pharmacokinetics of anti-tumor necrosis factor α biology[14-16]. Therapeutic optimization as increasing fixed dose from 5 mg/kg to 10 mg/kg or shortening injection interval form every 8 wk to 4-6 wk contributes to increase ITL and decrease ATI level. A study from Greece demonstrated that, for the initial measurement after therapeutic adjustment, ITL increased from 1.47 μg/mL to 8.50 μg/mL in patients with therapeutic optimization while ITL decreased from 5.65 μg/mL to 3.8μg/ml in patients without therapeutic optimization[10]. A multi-center randomized clinical trial conducted by Dreesen et al[17] showed that CD patients under IFX maintenance therapy as 5 mg/kg had high probability of no mucosal ulcer under endoscopy at week 54 with ITL more than 7.3mg/L, and CD patients under intensified dose IFX therapy as 10 mg/kg had 94% probability of no mucosal ulcer under endoscopy with ITL rising to more than 10.6 mg/L. Therefore, intensified therapy may contribute to mucosal healing in CD patients with ulceration if IFX injection dose is less than 10 mg/kg and ITL is less than 10.6 μg/mL. However, a few CD patients will accept combination therapy of IFX and immunosuppressant to boost the efficacy, especially AZA and mercaptopurine. AZA is a precursor of mercaptopurine, and two components ultimately produce thioguanine to exert clinical effect during metabolism. A study verified that thioguanine concentration more than 125 pmol/8 × 108 red blood cells could enhance ITL to 8.3 μg/mL or more and decrease positive rate of ATI[18,19]. Hence, this study mainly included CD patients with sustained clinical remission more than 6 mo under fixed therapeutic strategy of IFX 5 mg/kg every 8 wk combined with AZA 50 mg every day. Retrospective records of CD patients included would suspend if therapy strategy changed, such as intensive therapy of IFX, conversion therapy of other biologics and combination therapy of surgery or other medications. The study design eliminated the influence of therapeutic adjustment on ITL.
The greatest strength of inflammatory biomarkers compared with blood trough level is that they are unaffected by time during different monitoring stages of biological therapy in IBD patients. This study showed that ITL > 5.6 μg/mL combined with FCP ≤ 238 μg/g at week 14 moderately predicted sustained endoscopic remission during the 1-year follow-up period on CD patients with positive predictive value more than 95% as well as ITL > 2.1 μg/mL combined with CRP ≤ 3.00 mg/L at week 54, superior to use ITL as the only predictor. FCP and CRP are considered as the most universal and typical biomarkers of inflammatory evaluation in IBD, also verified to be the independent risk factors of adverse endoscopic outcomes. The study confirmed that combining blood trough level with inflammatory biomarkers contributed to improving the accuracy of the prediction on endoscopic outcomes. A post hoc analysis from the CALM study manifested that CD patients with FCP < 250 μg/g mostly achieved CDEIS < 4 without deep ulceration, regardless of whether CRP < 5 mg/L. However, among patients with CRP < 5 mg/L but FCP ≥ 250 μg/g, only 16.7% achieved CDEIS < 4 without deep ulceration[20]. The result indicated that the correlation between FCP normalization and endoscopic mucosal healing in CD patients was stronger than that of CRP normalization. A previous study verified that FCP is suitable for distinguishing mild endoscopic activity from endoscopic remission, while it is difficult to distinguish partial endoscopic remission from complete endoscopic remission[21]. Similar to blood trough level, the optimal cut off value of FCP for distinguishing endoscopic activity from endoscopic remission ranges from 71 μg/g to 250 μg/g with moderate diagnostic performance[22-27]. The study identified that FCP > 276 μg/g predicted endoscopic activity at week 54 of CD patients with clinical remission at week 14 moderately with 84.6% sensitivity and 92.1% specificity. Unlike FCP, the sensitivity of CRP to mild intestinal inflammation was low and the level of CRP increased much more dramatically in CD patients with moderate to severe inflammation. Therefore, the previous study preferred to utilize CRP to distinguish moderate to severe endoscopic activity from mild to moderate endoscopic activity rather than distinguish mild endoscopic activity from endoscopic remission. A Spanish study showed that FCP > 155 μg/g in combination with CRP > 6.7 mg/L could identify endoscopic activity with 82% specificity[27].
However, the study has shortcomings in some areas. Firstly, the retrospective single-center study with small sample, inferior to prospective multi-center with greater sample, comprised some confounding factors. More real-world studies and randomized controlled trials on guidance significance of ITL to therapeutic outcomes in IBD need to be conducted. Secondly, the study primarily concentrated on mucosal inflammation located in large intestine, ignoring small intestine due to the high cost and the incomplete scoring system of small intestinal evaluation accompanied by the poor compliance of patients and the laborious operation of endoscopists. Correlation between ITL and various small intestinal examinations including endoscopy or imageology may be the focus of the future study. Last but not least, definition of deep remission on CD has been tightened. Considering transmural inflammation of CD, endoscopy is confined to mucosal inflammation and macroscopical evaluation while imageology can accurately evaluate complete volume of intestinal wall and histopathological examination, which contributes to microscopical examination. Notwithstanding endoscopic remission considered as the main targets and histological remission considered as a novel target, the new concept of ‘disease clearance’, which includes clinical, endoscopic and microscopic remission, has drawn more and more attention from IBD physicians and may bring about a new upsurge of studies on IFX monitoring and new therapeutic targets[28].
In conclusion, during IFX maintenance treatment, low ITL, high CRP level and high FCP level were independent risk factors of long-term adverse endoscopy outcomes in CD patients with clinical remission. Combination of ITL, CRP and FCP contribute to long-term endoscopic prognosis monitoring. The best cut off values of ITL for predicting endoscopic activity within 1-year follow up were 5.60 μg/mL at week 14 and 2.10 μg/mL at week 54. In addition, ITL ≤ 5.60 μg/mL in combination with FCP > 238 μg/g at week 14 as well as ITL ≤ 2.10 μg/mL in combination with CRP > 3.00 mg/L at week 54 increased the precision of prediction on endoscopic outcomes at week 54 and week 108, respectively. Therapeutic optimization is still recommended in CD patients achieving endoscopic remission, provided that low ITLs or high levels of inflammatory biomarkers, such as CRP or FCP, arise to prevent endoscopic recurrence as soon as possible.
Existing studies have confirmed that infliximab (IFX) blood concentration is closely related to remission and recurrence of Crohn's disease (CD) patients under IFX therapy. In addition, monitoring inflammatory biomarkers regularly is another important tool for prognosis assessment of CD patients. Current studies have confirmed that C-reactive protein (CRP) and fecal calprotectin (FCP) are good predictors of disease activity, but there is still a lack of reliable evidence for predicting disease remission. Therefore, in the early stage of IFX treatment, combination of IFX blood concentration and inflammatory biomarkers may contribute to predict the change of CD outcomes.
The best therapeutic goal of CD was initially defined as clinical remission, and then the definition was converted to endoscopic remission with precise therapy. Nowadays, some clinicians even pursue whole-wall healing with individualized therapy. However, long-term clinical prognosis rather than long-term endoscopic prognosis is still a research priority of clinical studies contrapose to CD patients under IFX therapy. Therefore, prediction on long-term endoscopic prognosis of CD patients under IFX therapy has been based solely on models because of a lack of available data.
To explore the predictive value of blood drug concentration on long-term endoscopic outcomes of IFX therapy for CD and establish a comprehensive outcome prediction model combining IFX blood drug concentration, CRP and FCP, so as to provide a basis for clinical decision making.
A single-center retrospective research has been implemented in the First Affiliated Hospital of Zhejiang Chinese Medical University. CD patients under IFX therapy from January 2012 to December 2020 were collected. One hundred and eighty-one CD patients underwent IFX treatment. One hundred and fifty-three CD patients underwent endoscopy as well as serum concentration monitoring at week 14 after the third dose of IFX induction therapy. Inclusion criteria: (1) Endoscopic remission at week 14 [Crohn’s disease endoscopic index of severity (CDEIS) score ≤ 2 points or Rutgeerts ≤ i1]; (2) Clinical remission after IFX induction therapy without corticosteroids more than 6 mo; and (3) Therapeutic strategy during maintenance stage was designed as IFX 5 mg/kg every 8 wk combined with azathioprine 50 mg every day. The study was divided into two stages, the Step I study was conducted from week 14 to 54 of IFX treatment, and the Step II study was conducted from week 54 to 108 of IFX treatment. Endoscopic outcomes were defined as endoscopic activity (CDEIS score > 2 points or Rutgeerts score > i1) and endoscopic remission (CDEIS score ≤ 2 points or Rutgeerts ≤ i1). Endoscopic relapse free survival was defined as endoscopic remission at the beginning of the study stage and maintaining endoscopic remission during the study stage.
In step I study, 67/93 CD patients (72.04%) sustained endoscopic remission at week 54. Multivariable regression analysis demonstrated that only ITL [odds ratio (OR) = 0.666, 95% confidence interval (CI): 0.514-0.862, P = 0.002] and FCP (OR = 1.002, 95%CI: 1.001-1.004, P = 0.002) were independent risk of endoscopic activity at week 54. The receiver-operating characteristic analysis demonstrated that the best cut off level of ITL and FCP at week 14 on predicting endoscopic relapse at week 54 was 5.60 μg/mL [area under the curve (AUC) = 0.83, 95%CI: 0.73-0.90, P < 0.001] and 238 μg/g (AUC = 0.82, 95%CI: 0.72-0.89, P < 0.001). The median time to endoscopic relapse of CD patients with ITL ≤ 5.6 μg/ml was 32.00 wk shorter than those with ITL > 5.6 μg/mL [hazard ratio (HR) = 16.19, 95%CI: 7.44-35.22, P < 0.0001]. The median time to endoscopic relapse of CD patients with FCP > 238 μg/g was 21.00 wk shorter than those with FCP ≤ 238 μg/g (HR = 11.25, 95%CI: 4.26-29.73, P < 0.0001). (II) In step II study, 42/54 CD patients (77.78%) sustained endoscopic remission at week 108. Multivariable regression analysis found that only ITL (OR = 0.466, 95%CI: 0.247-0.877, P = 0.018) and CRP (OR = 1.590, 95%CI: 1.007-2.510, P = 0.047) were independent risks of endoscopic activity at week 108. The receiver-operating characteristic analysis demonstrated that the best cut off level of ITL and CRP at week 54 on predicting endoscopic relapse at week 108 was 2.10 μg/ml (AUC = 0.85, 95%CI: 0.72-0.93, P < 0.001) and 3.00 mg/L (AUC = 0.73, 95%CI: 0.60-0.84, P = 0.012). The median time to endoscopic relapse of CD patients with ITL ≤ 2.1 μg/mL was 40.00 w shorter than those with ITL > 2.1 μg/mL (HR = 13.14, 95%CI: 3.07-56.27, P < 0.0001). The median time to endoscopic relapse of CD patients with CRP > 3.00 mg/L was 50.00 wk shorter than those with CRP ≤ 3.00 mg/L (HR = 7.85, 95%CI: 1.31-46.85, P< 0.0001).
The best cut off values of ITL for predicting endoscopic activity within 1-year follow up was 5.60 μg/mL at week 14 and 2.10 μg/mL at week 54. In addition, ITL ≤ 5.60 μg/mL in combination with FCP > 238 μg/g at week 14 as well as ITL ≤ 2.10 μg/mL in combination with CRP > 3.00 mg/L at week 54 increased the precision of prediction on endoscopic outcomes at week 54 and week 108, respectively.
In view of the fact that conduction of intensive monitoring for biological management plays a vital role in precise treatment for CD patients, much larger and more stringent prospective studies are warranted to provide the best predictive models as acknowledged globally in allusion to long-term endoscopic outcomes of CD patients under IFX therapy.
The authors would like to thank Dr. Bin Lv, Full Professor and Chief Physician at the First Affiliated Hospital of Zhejiang Chinese Medical University, Department of Gastroenterology, for his help in revising this paper, and Shan Liu, at the First Affiliated Hospital of Zhejiang Chinese Medical University, Clinical Evaluation Center, for help with statistical analysis.
Provenance and peer review: Unsolicited article; Externally peer reviewed.
Peer-review model: Single blind
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): 0
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Leite A, Brazil; Taxonera C, Spain S-Editor: Ma YJ L-Editor: Filipodia P-Editor: Cai YX
| 1. | Tang J, Yang Q, Huang Z, Guo H, Chao K, Hu P, Gao X. Early serum infliximab trough level and mucosal healing could be predictors for one-year outcome after initiating therapy in Crohn's disease. Scand J Gastroenterol. 2020;55:1035-1040. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2] [Cited by in RCA: 3] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 2. | Bortlik M, Duricova D, Malickova K, Machkova N, Bouzkova E, Hrdlicka L, Komarek A, Lukas M. Infliximab trough levels may predict sustained response to infliximab in patients with Crohn's disease. J Crohns Colitis. 2013;7:736-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 180] [Cited by in RCA: 202] [Article Influence: 16.8] [Reference Citation Analysis (0)] |
| 3. | Ishida N, Miyazu T, Sugiyama T, Tamura S, Kagami T, Tani S, Yamade M, Iwaizumi M, Hamaya Y, Osawa S, Furuta T, Sugimoto K. The effect of early trough level of infliximab on subsequent disease course in patients with Crohn disease: A prospective cohort study. Medicine (Baltimore). 2020;99:e21226. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 5] [Cited by in RCA: 4] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 4. | Moore C, Corbett G, Moss AC. Systematic Review and Meta-Analysis: Serum Infliximab Levels During Maintenance Therapy and Outcomes in Inflammatory Bowel Disease. J Crohns Colitis. 2016;10:619-625. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 99] [Cited by in RCA: 94] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 5. | Borren NZ, Paulides E, Frinack JL, Olson RN, Willrich MAV, van der Woude CJ, Ananthakrishnan AN. Infliximab Trough Levels Are Not Predictive of Relapse in Patients with IBD in Endoscopic Remission: A Multicenter Cohort Study. Dig Dis Sci. 2021;66:3548-3554. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 8] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 6. | Imaeda H, Bamba S, Takahashi K, Fujimoto T, Ban H, Tsujikawa T, Sasaki M, Fujiyama Y, Andoh A. Relationship between serum infliximab trough levels and endoscopic activities in patients with Crohn's disease under scheduled maintenance treatment. J Gastroenterol. 2014;49:674-682. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 82] [Cited by in RCA: 87] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 7. | Papamichael K, Rakowsky S, Rivera C, Cheifetz AS, Osterman MT. Association Between Serum Infliximab Trough Concentrations During Maintenance Therapy and Biochemical, Endoscopic, and Histologic Remission in Crohn's Disease. Inflamm Bowel Dis. 2018;24:2266-2271. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 70] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 8. | Wilson A, Choi B, Sey M, Ponich T, Beaton M, Kim RB. High infliximab trough concentrations are associated with sustained histologic remission in inflammatory bowel disease: a prospective cohort study. BMC Gastroenterol. 2021;21:77. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 11] [Cited by in RCA: 10] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Plevris N, Jenkinson PW, Arnott ID, Jones GR, Lees CW. Higher anti-tumor necrosis factor levels are associated with perianal fistula healing and fistula closure in Crohn's disease. Eur J Gastroenterol Hepatol. 2020;32:32-37. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 52] [Article Influence: 10.4] [Reference Citation Analysis (0)] |
| 10. | Orfanoudaki E, Gazouli M, Foteinogiannopoulou K, Theodoraki E, Legaki E, Romanos I, Mouzas I, Koutroubakis IE. Infliximab trough levels are decreasing over time in patients with inflammatory bowel disease on maintenance treatment with infliximab. Eur J Gastroenterol Hepatol. 2019;31:187-191. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 10] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 11. | Chaparro M, Barreiro-de Acosta M, Echarri A, Almendros R, Barrio J, Llao J, Gomollón F, Vera M, Cabriada JL, Guardiola J, Guerra I, Beltrán B, Roncero O, Busquets D, Taxonera C, Calvet X, Ferreiro-Iglesias R, Ollero Pena V, Bernardo D, Donday MG, Garre A, Godino A, Díaz A, Gisbert JP. Correlation Between Anti-TNF Serum Levels and Endoscopic Inflammation in Inflammatory Bowel Disease Patients. Dig Dis Sci. 2019;64:846-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 27] [Cited by in RCA: 24] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 12. | Kang B, Choi SY, Choi YO, Lee SY, Baek SY, Sohn I, Choe BH, Lee HJ, Choe YH. Infliximab Trough Levels Are Associated With Mucosal Healing During Maintenance Treatment With Infliximab in Paediatric Crohn's Disease. J Crohns Colitis. 2019;13:189-197. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 42] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 13. | Feng T, Chen B, Ungar B, Qiu Y, Zhang S, He J, Lin S, He Y, Zeng Z, Ben-Horin S, Chen M, Mao R. Association of Infliximab Levels With Mucosal Healing Is Time-Dependent in Crohn's Disease: Higher Drug Exposure Is Required Postinduction Than During Maintenance Treatment. Inflamm Bowel Dis. 2019;25:1813-1821. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 14. | Beltrán B, Iborra M, Sáez-González E, Marqués-Miñana MR, Moret I, Cerrillo E, Tortosa L, Bastida G, Hinojosa J, Poveda-Andrés JL, Nos P. Fecal Calprotectin Pretreatment and Induction Infliximab Levels for Prediction of Primary Nonresponse to Infliximab Therapy in Crohn's Disease. Dig Dis. 2019;37:108-115. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 23] [Cited by in RCA: 31] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 15. | Hemperly A, Vande Casteele N. Clinical Pharmacokinetics and Pharmacodynamics of Infliximab in the Treatment of Inflammatory Bowel Disease. Clin Pharmacokinet. 2018;57:929-942. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 62] [Cited by in RCA: 114] [Article Influence: 19.0] [Reference Citation Analysis (0)] |
| 16. | Dreesen E, Berends S, Laharie D, D'Haens G, Vermeire S, Gils A, Mathôt R. Modelling of the relationship between infliximab exposure, faecal calprotectin and endoscopic remission in patients with Crohn's disease. Br J Clin Pharmacol. 2021;87:106-118. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 17. | Dreesen E, Baert F, Laharie D, Bossuyt P, Bouhnik Y, Buisson A, Lambrecht G, Louis E, Oldenburg B, Pariente B, Pierik M, van der Woude CJ, D'Haens G, Vermeire S, Gils A. Monitoring a Combination of Calprotectin and Infliximab Identifies Patients With Mucosal Healing of Crohn's Disease. Clin Gastroenterol Hepatol. 2020;18:637-646.e11. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 57] [Cited by in RCA: 70] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 18. | Yarur AJ, Kubiliun MJ, Czul F, Sussman DA, Quintero MA, Jain A, Drake KA, Hauenstein SI, Lockton S, Deshpande AR, Barkin JS, Singh S, Abreu MT. Concentrations of 6-thioguanine nucleotide correlate with trough levels of infliximab in patients with inflammatory bowel disease on combination therapy. Clin Gastroenterol Hepatol. 2015;13:1118-24.e3. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 125] [Article Influence: 12.5] [Reference Citation Analysis (0)] |
| 19. | Vermeire S, Noman M, Van Assche G, Baert F, D'Haens G, Rutgeerts P. Effectiveness of concomitant immunosuppressive therapy in suppressing the formation of antibodies to infliximab in Crohn's disease. Gut. 2007;56:1226-1231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 450] [Cited by in RCA: 469] [Article Influence: 26.1] [Reference Citation Analysis (0)] |
| 20. | Reinisch W, Panaccione R, Bossuyt P, Baert F, Armuzzi A, Hébuterne X, Travis S, Danese S, Sandborn WJ, Schreiber S, Berg S, Zhou Q, Kligys K, Neimark E, Suleiman AA, D'Haens G, Colombel JF. Association of Biomarker Cutoffs and Endoscopic Outcomes in Crohn's Disease: A Post Hoc Analysis From the CALM Study. Inflamm Bowel Dis. 2020;26:1562-1571. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 34] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 21. | Kawashima K, Ishihara S, Yuki T, Fukuba N, Sonoyama H, Kazumori H, Yamashita N, Tada Y, Kusunoki R, Oka A, Oshima N, Mishima Y, Moriyama I, Kinoshita Y. Fecal Calprotectin More Accurately Predicts Endoscopic Remission of Crohn's Disease than Serological Biomarkers Evaluated Using Balloon-assisted Enteroscopy. Inflamm Bowel Dis. 2017;23:2027-2034. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 22. | D'Haens G, Ferrante M, Vermeire S, Baert F, Noman M, Moortgat L, Geens P, Iwens D, Aerden I, Van Assche G, Van Olmen G, Rutgeerts P. Fecal calprotectin is a surrogate marker for endoscopic lesions in inflammatory bowel disease. Inflamm Bowel Dis. 2012;18:2218-2224. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 553] [Cited by in RCA: 624] [Article Influence: 48.0] [Reference Citation Analysis (0)] |
| 23. | Kostas A, Siakavellas SI, Kosmidis C, Takou A, Nikou J, Maropoulos G, Vlachogiannakos J, Papatheodoridis GV, Papaconstantinou I, Bamias G. Fecal calprotectin measurement is a marker of short-term clinical outcome and presence of mucosal healing in patients with inflammatory bowel disease. World J Gastroenterol. 2017;23:7387-7396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 61] [Cited by in RCA: 60] [Article Influence: 7.5] [Reference Citation Analysis (0)] |
| 24. | Vázquez Morón JM, Pallarés Manrique H, Machancoses FH, Ramos Lora M, Ruiz Frutos C. Accurate cut-offs for predicting endoscopic activity and mucosal healing in Crohn's disease with fecal calprotectin. Rev Esp Enferm Dig. 2017;109:130-136. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 16] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 25. | Inokuchi T, Kato J, Hiraoka S, Takashima S, Nakarai A, Takei D, Sugihara Y, Takahara M, Kawano S, Harada K, Okada H. Fecal Immunochemical Test Versus Fecal Calprotectin for Prediction of Mucosal Healing in Crohn's Disease. Inflamm Bowel Dis. 2016;22:1078-1085. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 50] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 26. | D'Arcangelo G, Oliva S, Dilillo A, Viola F, Civitelli F, Isoldi S, Cucchiara S, Aloi M. Predictors of Long-term Clinical and Endoscopic Remission in Children With Crohn Disease Treated With Infliximab. J Pediatr Gastroenterol Nutr. 2019;68:841-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 15] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 27. | Penna FGC, Rosa RM, Pereira FH, Cunha PFS, Sousa SCS, Ferrari TCA, Cara C, Ferrari MLA. Combined evaluation of fecal calprotectin and C-reactive protein as a therapeutic target in the management of patients with Crohn's disease. Gastroenterol Hepatol. 2021;44:87-95. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 28. | Dal Buono A, Roda G, Argollo M, Zacharopoulou E, Peyrin-Biroulet L, Danese S. Treat to target or 'treat to clear' in inflammatory bowel diseases: one step further? Expert Rev Gastroenterol Hepatol. 2020;14:807-817. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 18] [Article Influence: 3.6] [Reference Citation Analysis (0)] |