Published online Oct 28, 2020. doi: 10.3748/wjg.v26.i40.6250
Peer-review started: May 14, 2020
First decision: May 21, 2020
Revised: September 4, 2020
Accepted: September 17, 2020
Article in press: September 17, 2020
Published online: October 28, 2020
Processing time: 166 Days and 8.7 Hours
Pancreatic adenoma can potentially transform into adenocarcinoma, so it is recommended to be resected surgically or endoscopically. Endoscopic papillectomy is one of the main treatments for papillary adenoma, and bleeding, perforation, and pancreatitis are the most frequent and critical adverse events that restrict its wider use. There is no standard procedure for endoscopic papillectomy yet. The procedure is relevant to postoperative adverse events.
To reduce the postoperative adverse event rates and improve patients’ postoperative condition, we developed a standard novel procedure for endoscopic papillectomy.
The novel endoscopic papillectomy had two main modifications based on the conventional method: The isolation of bile from pancreatic juice with a bile duct stent and wound surface protection with metal clips and fibrin glue. We performed a single-center retrospective comparison study on the novel and conventional methods to examine the feasibility of the novel method for reducing postoperative adverse events.
A total of 76 patients, of whom 23 underwent the novel procedure and 53 underwent the conventional procedure, were retrospectively evaluated in this study. The postoperative bleeding and pancreatitis rates of the novel method were significantly lower than those of the conventional method (0 vs 20.75%, P = 0.028, and 17.4% vs 41.5%, P = 0.042, respectively). After applying the novel method, the most critical adverse event, perforation, was entirely prevented, compared to a prevalence of 5.66% with the conventional method. Several postoperative symptoms, including fever, rapid pulse, and decrease in hemoglobin level, were significantly less frequent in the novel group (P = 0.042, 0.049, and 0.014, respectively). Overall, the total adverse event rate of the novel method was lower (0 vs 24.5%, P = 0.007) than that of the conventional method.
Patients who underwent the novel procedure had lower postoperative adverse event rates. This study demonstrates the potential efficacy and safety of the novel endoscopic papillectomy in reducing postoperative adverse events.
Core Tip: The novel method of endoscopic papillectomy mainly includes two modifications: Isolating bile from pancreatic juice with a bile duct stent and closing the wound surface with metal clips and fibrin glue. Our retrospective comparison showed that postoperative adverse events, including bleeding, pancreatitis, and overall adverse events, were less frequent in the novel endoscopic papillectomy group than in the conventional endoscopic papillectomy group. Postoperative perforation was entirely prevented by this novel method. This study demonstrates that novel endoscopic papillectomy is a potentially effective and safe method for reducing postoperative adverse events.
- Citation: Jiang L, Ling-Hu EQ, Chai NL, Li W, Cai FC, Li MY, Guo X, Meng JY, Wang XD, Tang P, Zhu J, Du H, Wang HB. Novel endoscopic papillectomy for reducing postoperative adverse events (with videos). World J Gastroenterol 2020; 26(40): 6250-6259
- URL: https://www.wjgnet.com/1007-9327/full/v26/i40/6250.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i40.6250
Tumors of the main duodenal papilla are rare, with a prevalence of only 0.04% to 0.12% in autopsy studies[1]. Although the main pathologic diagnosis, papillary adenoma (PA), is benign, it can potentially develop into adenocarcinoma through the adenoma-adenocarcinoma sequence[2]. Thus, the treatment of PA is essential and more critical than just follow-up. Pancreaticoduodenectomy and endoscopic papillectomy (EP) are the main treatments for PA, and EP is more recommended for benign papillary lesions because of its advantages over open surgery, including less trauma, satisfying treatment outcomes, fewer adverse events, and lower cost[3-6]. With all of these aforementioned advantages, EP seemed to be a promising treatment for PA; however, surgeons also found intraoperative and postoperative adverse events. These included bleeding, operation-related pancreatitis, perforation, cholangitis, and papillary stricture, and some events, especially bleeding and perforation, are critical and even fatal.
These adverse events could lead to extremely difficult results that require invasive surgical intervention or even result in death. Although this is not common, it does limit the wide application of EP. Thus, a better standard procedure of EP with fewer adverse events is urgently needed for the endoscopic treatment of papillary lesions.
To decrease the postoperative adverse event rate of EP and improve patients’ postoperative condition, we modified conventional endoscopic papillectomy and initiated a novel procedure for endoscopic papillectomy in 2016. To verify the safety and feasibility of this novel method, we reviewed the outcomes of patients undergoing novel or conventional EP.
We retrospectively studied patients who had PA and underwent novel or conventional EP at the endoscopy center of Chinese PLA General Hospital from January 2016 to September 2018. A total of 76 patients, of whom 23 underwent novel EP and 53 underwent conventional EP, were enrolled in this study. Sample size calculation with the adverse event rates of two groups (significance level of 5% and power of 80%) suggested a minimum sample size of 70. As a retrospective observational study, the study was not blindly randomized.
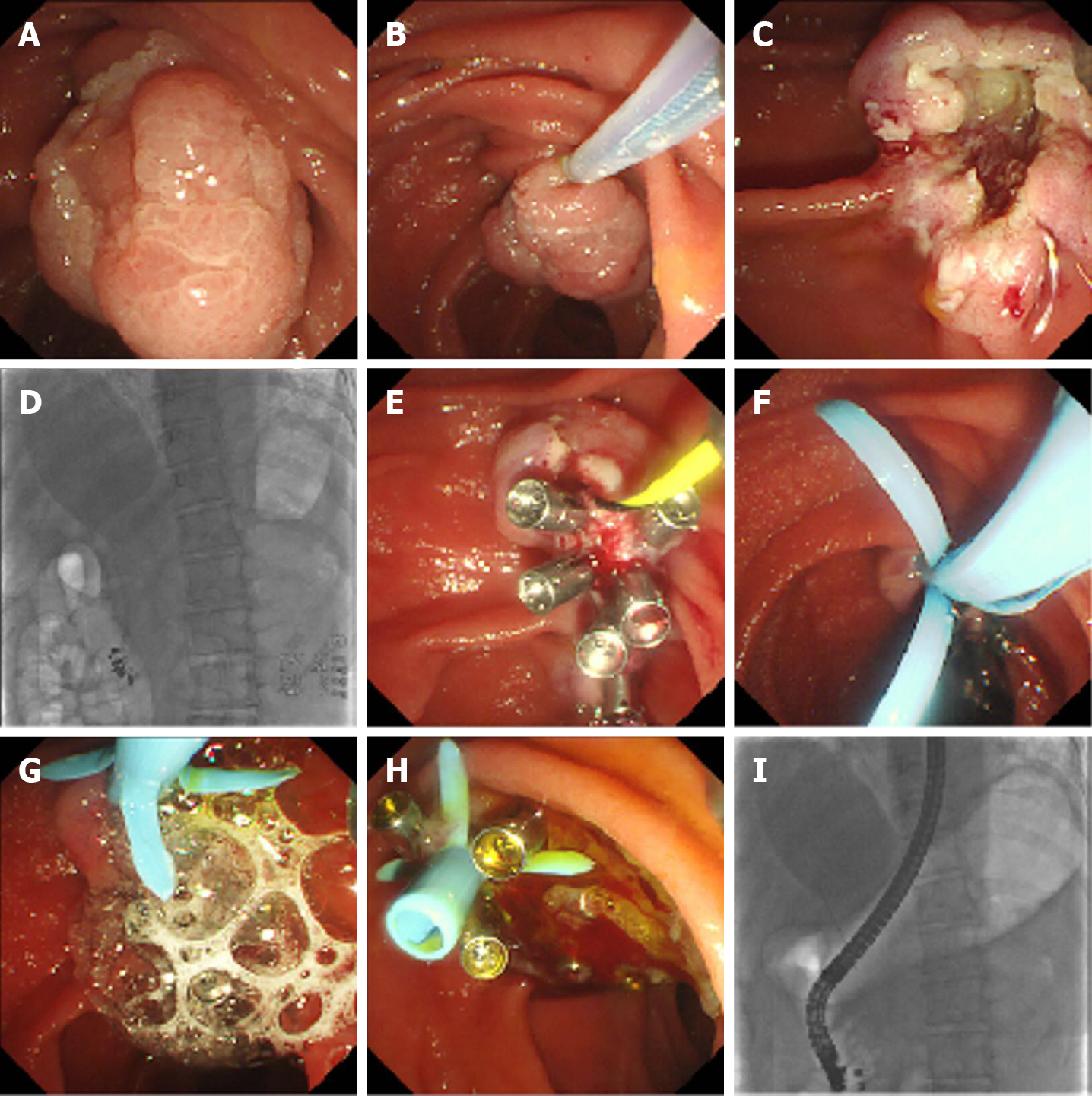
The standard procedure for novel EP was as follows (Figure 1, Video 1): (1) The patient was placed in a supine position under general anesthesia; (2) Duodenoscopy (TJF-240/TJF-260V; Olympus, Tokyo, Japan) was used to observe the papillary lesion; (3) A submucosal injection was performed to lift the lesion, or an injection was not performed; then, en bloc or blocking resection of the lesion was performed with an endoloop (SD-7P-1/SD-221L-25; Olympus, Tokyo, Japan). If the lesion was large, edge cutting could be performed before snaring; argon plasma coagulation was used for bleeding; (4) A guidewire was used to superselect the bile duct (BD), and the guidewire was retained in the BD; (5) Electric coagulation forceps and argon plasma coagulation were used for wound hemostasis, and then the wound was closed with metal clips; (6) A BD stent was implanted; (7) Fibrin glue (S201110020; Bioseal, Guangzhou, China) was sprayed on the closed wound; (8) Exhaust air in the stomach and stent placement were checked under X-ray; and (9) The sample was sent for pathology.
Patients were required to fast after the operation. At the same time, acid-inhibitory drugs, enzyme inhibitors (somatostatin), and total parenteral nutrition were given through intravenous infusion. Operation-related symptoms, including bleeding, abdominal pain, fever, and postoperative pancreatitis, were closely monitored. The BD stents were removed 3 to 4 wk after the operation.
The enrolled patients were classified into two groups: Novel group and conventional group. The baseline features of the patients, the postoperative indexes, and the occurrence rates of adverse events were compared by the t-test, Fisher’s test, chi-square test, or ANOVA, with P < 0.05 indicating statistical significance. All data were analyzed with SPSS statistics 19.0 (IBM, Armonk, NY, United States) and Prism 8.0 (GraphPad, San Diego, CA, United States).
No specific IRB approval was required for this study according to the Ethics Committee of PLA General Hospital.
A total of 76 patients who underwent EP from January 2016 to September 2018 were enrolled in the study, 23 of whom were randomly chosen by the surgeons for the novel EP, and the rest underwent the conventional EP. Table 1 shows the comparison of baseline features, including pre-, intra-, and postoperative characteristics, which are related to postoperative adverse events. No significant difference was found in the baseline characteristics between these two groups.
| N group (n = 23) | C group (n = 53) | P value | ||
| Age | 59.30 ± 9.55 | 58.06 ± 13.09 | 0.682 | |
| Sex | 9:14 | 27:26 | 0.343 | |
| Symptomatic | 13/23 | 32/53 | 0.753 | |
| Intra-operation | BD dilation | 2/23 | 10/53 | 0.327 |
| Resection size (cm2) | 3.668 ± 2.904 | 4.780 ± 6.536 | 0.582 | |
| Pathologic result | HGIN | 8 | 20 | |
| LGIN | 3 | 8 | ||
| Non-GIN | 10 | 16 | ||
| NT | 1 | 1 | 0.703 | |
| Postoperative hospital stay (d) | 10.35 ± 4.23 | 10.08 ± 3.60 | 0.783 | |
| Total cost (Yuan) | 45243.75 ± 11443.68 | 58158.77 ± 79290.51 | 0.473 | |
The blood test results and vital signs related to the postoperative condition are compared in Table 2. More patients in the conventional group than in the novel group had postoperative fever (17.4% vs 41.5%, P = 0.042), and the result was similar for postoperative rapid pulse (0 vs 15.1%, P = 0.049). No obvious decrease in hemoglobin (greater than 20 g/L) was observed in the novel group, while 22.6% of patients in the conventional group had a statistically significant (P = 0.014) decrease in hemoglobin. No significant differences were found in other indexes.
| N group | C group | P value | ||
| Fever | 17.4% (4/23) | 41.5% (22/53) | 0.0421 | |
| Breathing ratea (/m) | 18.09 ± 1.04 | 19.60 ± 7.43 | 0.335 | |
| Pulse (/m)a | Value | 79.70 ± 7.62 | 84.68 ± 18.84 | 0.226 |
| > 90/m | 0 (0/23) | 15.1% (8/53) | 0.0491 | |
| Abdominal pain scoreb | 2.09 ± 2.59 | 1.75 ± 1.93 | 0.585 | |
| Drop in hemoglobinc (g/L) | 0 (0/23) | 22.6% (12/53) | 0.0141 | |
| WBC (109/L) | Value | 8.84 ± 3.05 | 9.54 ± 3.39 | 0.395 |
| Abnormal | 13.0 (3/23) | 30.2% (16/53) | 0.113 | |
| C-RP (mg/L) | Value | 1.43 ± 2.66 | 2.93 ± 5.62 | 0.252 |
| Abnormal | 4.8% (1/21) | 9.5% (4/42) | 0.510 | |
| AMY (u/L) | 354.20 ± 399.12 | 324.35 ± 320.12 | 0.731 | |
| LPS (u/L) | 2052.68 ± 4220.83 | 1555.89 ± 2047.35 | 0.493 | |
| Hyperamylasemia | 39.1% (9/23) | 49.1% (26/53) | 0.425 | |
To further reveal the safety and effect of the novel procedure, we compared the rate of adverse events, as listed in Table 3. Postoperative bleeding was significantly more common in the conventional EP group than in the novel EP group (0 vs 20.75%, P = 0.028). The frequency of pancreatitis in the conventional group was also higher than that in the novel group (17.4% vs 41.5%, P = 0.042). Although no significant difference was observed in the prevalence of perforation, there was no single case of perforation in the novel EP group compared to three perforation cases in the conventional group. The incidences of cholangitis and multiple organ dysfunction were not significantly different between the groups, although the occurrence rates in the novel group were lower than those in the conventional group. The overall adverse event occurrence rate in the novel group was markedly lower than that in the conventional group (13.0% vs 41.5%, P = 0.018). All adverse events were cured or relieved after medical treatments or surgery with no mortality.
Unlike that of other gastrointestinal polyps, the resection of papillary lesions is far more complex and difficult for several reasons, including its complicated location in the duodenum, the extremely enriched vasculature, and the thin wall of the duodenum. Thus, for a long time, open or minimally invasive surgeries, such as pancreaticoduodenectomy, have been regarded as the only removal strategy for PAs[1]. The drawbacks of these invasive surgeries are obvious and include the high adverse event rate and mortality, long anesthesia duration, large operative trauma, high cost, etc[1,7].
Compared with conventional papillary surgery, EP has merits in various aspects that have popularized its rapid worldwide use. The merits are as follows: (1) For benign lesions, EP can be a curative therapy by resecting the lesion completely with less invasion; (2) For malignant lesions, EP can offer abundant tissues for pathological tests, and the identified pathological diagnosis could guide next-step treatments; and (3) For patients who are not eligible for long surgery, EP can reduce the surgical risk[8-10].
Although it was first performed in 1987[11] and has been used by surgeons worldwide, there are still no standard procedures or guidelines for EP. Several details, such as pancreatic duct (PD) stenting and submucosal injection, are still controversial[12-19].
Postoperative adverse events, which occur in approximately 29% of patients[9,20-34], have been a difficult issue for endoscopists and have been the main cause of death from endoscopic papillectomy in clinical practice since the wide application of EP. The mortality of EP is reported to be approximately 0.4%[35]. Bleeding and pancreatitis are the most common adverse events, with a reported prevalence of 4.5%-45% and 7%-20%, respectively[9,20,23,26-29,36-39]. Based on our experience, we designed a novel papillectomy procedure to decrease adverse events, especially bleeding and pancreatitis. After applying this novel procedure, we found that patients who underwent novel EP had better physical conditions, including less occurrence of fever, rapid pulse, and blood loss, after the surgery. For postoperative adverse events, the novel method showed clear superiority for reducing bleeding, EP-related pancreatitis, and the overall adverse event rate. We also found a decreasing trend of other adverse events, including perforation, cholangitis, and multiple organ dysfunction, in the novel group, even though the differences were not statistically significant. Perforation is one of the most critical and dangerous adverse events of EP that may lead to severe intra-abdominal infection and even death. Addressing perforation is rather difficult and usually requires the cooperation of hepatological surgery, which increases the surgical risk and cost and reverses the superiority of endoscopic treatment. Thus, the novel method could prevent perforation, making it an important contribution to improving the prognosis of EP.
After examining relevant studies, we found that all the adverse event rates of the conventional group in our study were consistent with the results of previous studies[9,12,13,20,23,26-29,37,38,40-42]. Accordingly, the adverse event rates of the novel method were significantly lower than those of previous studies[9,12,13,20,23,26-29,37,38,40-42]. Therefore, novel EP can improve postoperative physical conditions and decrease surgical adverse events, making it a safe and effective method.
The occurrence of post-EP adverse events is relevant to two main processes: The incomplete healing of surgical wounds and the invasion of pathogenic microorganisms. In the novel procedure, timely closure of the wound with metal clips could help with wound healing and could prevent perforation. It could also prevent bacteria from invading the wound surface or into ducts[43,44].
In conventional endoscopic papillectomy, the implantation of PD stents is usually recommended to release postoperative pancreatitis by promoting pancreatic drainage, while the efficacy of PD stents is controversial. Furthermore, the application of BD stents has not yet been clarified[8,12,27,40,45].
With conventional EP, in which a PD stent is implanted solely or with BD stents simultaneously, pancreatic juice and bile can drain out, cover the resection wound, and mix with each other. In the mixture of bile and pancreatic juice, bile can activate trypsinogen to trypsin, which has a strong digestive capacity. Active trypsin, especially within the pancreatic duct activated by reflux bile, can induce pancreatitis[46]. It can also produce new granulation tissue on the wound surface and inhibit the coagulation process, leading to postoperative bleeding[47-51]. Hence, we presume that isolating the contact between bile and pancreatic juice can prevent trypsinogen from being activated by bile and can reduce the contribution of pancreatic juice to postoperative adverse events. The novel therapy promotes bile drainage into the cavity of the duodenum directly without covering the wound surface, prevents pancreatic juice and bile from mixing, and does not activate trypsinogen. In addition, as a protein substance, we presume that inactive trypsinogen that covers the wound surface can act as a kind of “nutrition cover” and, together with fibrin glue, can protect and promote the healing process. Furthermore, this protective cover can also prevent pathogenic microorganisms from invading into the PD and can reduce the risk of pancreatitis[43]. Based on these results, the evidence for how novel EP prevents postoperative adverse events is by preventing wound bleeding and infection and promoting wound healing.
We sprayed fibrin glue on the closed wound to protect the wound surface and found that it was effective in clinical practice[52,53]. The fibrin glue kit consists of two main components, porcine fibrinogen with factor XIII (component 1) and porcine thrombin with calcium chloride (component 2). When these two components mix with each other, the mixture simulates the final stage of the clotting cascade. Thrombin proteolytically cleaves fibrinogen, which results in the formation of fibrin polymers. In the presence of calcium, thrombin catalyzes the conversion of factor XIII to activated factor XIII (factor Xllla). Factor Xllla crosslinks the fibrin polymers into a stable, insoluble fibrin clot. Mutignani et al[54] used a modified injection of fibrin glue on six patients with post-endoscopic retrograde cholangiopancreatography bleeding. They verified the hemostatic effect of fibrin glue and confirmed that fibrin glue was an efficient endoscopic hemostatic agent, as all six patients were successfully stanched. At the same time, two patients had fibrin glue-related adverse events because the glue concreted in the BD and led to obstruction. In our novel method, bile duct stenting can prevent glue from blocking the bile ducts.
Another commonly used endoscopic hemostasis method is hemostatic powder, such as Hemospray, a mineral blend powder that can rapidly solidify and form an adhesive layer on contact with weak bases, such as water and blood[55]. Several studies have shown that Hemospray was efficient in treating gastrointestinal tract bleeding endoscopically[56-60]. EP was recommended as a favorable Hemospray indication[58]. Although there are no comparative studies investigating the efficacy and safety of Hemospray and fibrin glue, both of these treatments are believed to be efficient in treating gastrointestinal tract bleeding. Hemospray is not commercially available in China now, but considering its efficacy, we still look forward to testing it in our novel EP procedure in the future.
In some cases, submucosal injection was performed before resection to lift the lesions and to help with electronic resection and snares, but whether it is necessary is still an open question[4]. Hyun et al[41] performed multicenter prospective research to investigate whether submucosal injection contributed to endoscopic papillary procedures. They found that submucosal injection did not improve the complete resection rate or reduce the adverse event rate compared to direct snare resection. Moreover, direct snare resection was faster than resection after submucosal injection. Therefore, direct snare resection is a simpler and more recommendable method. In our study, we did not find submucosal injection to be helpful in novel therapy; however, its effects need more clinical research and analysis for verification.
As a retrospective study, this study had some limitations: (1) It was not blindly randomized; and (2) The sample size was not large enough; although it reached the minimum calculated sample size, the samples were not divided evenly between the two groups. Therefore, a multicenter randomized controlled prospective study is required for further research.
Compared with conventional EP, the novel method can reduce postoperative adverse events and improve the postoperative condition of patients. The novel EP achieves its benefits by isolating the contact between bile and pancreatic juice with a bile stent and protecting the wound surface with metal clips and fibrin glue.
Endoscopic papillectomy (EP) is one of the main treatments for duodenal papillary lesions.
The postoperative adverse events of EP limit its wide application.
To reduce postoperative adverse events by a novel EP, and to verify its safety and feasibility.
The novel method of endoscopic papillectomy mainly includes two modifications: Isolating bile from pancreatic juice with a bile duct stent and closing the wound surface with metal clips and fibrin glue. We retrospectively compared the postoperative condition and adverse event rates of the novel EP and conventional EP to reveal the safety and feasibility of the novel EP.
Patients who underwent the novel EP had a better postoperative condition and lower adverse event rates compared with patients who underwent the conventional EP. Postoperative perforation was entirely prevented by the novel method.
This study demonstrates that novel endoscopic papillectomy is a potentially effective and safe method for reducing postoperative adverse events.
A randomized controlled prospective study is required to further verify the safety and feasibility of the novel EP. After the confirmation, it could be recommended for wider application.
We thank peer reviewers for the insightful and thorough suggestions on this manuscript. We thank Julia Jones for language editing and video contract recording.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: China
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Ribeiro IB S-Editor: Zhang L L-Editor: Wang TQ P-Editor: Liu JH
| 1. | Bohnacker S, Soehendra N, Maguchi H, Chung JB, Howell DA. Endoscopic resection of benign tumors of the papilla of vater. Endoscopy. 2006;38:521-525. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 53] [Cited by in RCA: 49] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 2. | Stolte M, Pscherer C. Adenoma-carcinoma sequence in the papilla of Vater. Scand J Gastroenterol. 1996;31:376-382. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 112] [Cited by in RCA: 97] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 3. | Yoon SM, Kim MH, Kim MJ, Jang SJ, Lee TY, Kwon S, Oh HC, Lee SS, Seo DW, Lee SK. Focal early stage cancer in ampullary adenoma: surgery or endoscopic papillectomy? Gastrointest Endosc. 2007;66:701-707. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 88] [Cited by in RCA: 73] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 4. | Standards of Practice Committee. Adler DG, Qureshi W, Davila R, Gan SI, Lichtenstein D, Rajan E, Shen B, Zuckerman MJ, Fanelli RD, Van Guilder T, Baron TH. The role of endoscopy in ampullary and duodenal adenomas. Gastrointest Endosc. 2006;64:849-854. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 132] [Cited by in RCA: 105] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 5. | Kim AL, Choi YI. Safety of duodenal ampullectomy for benign periampullary tumors. Ann Hepatobiliary Pancreat Surg. 2017;21:146-150. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 13] [Cited by in RCA: 13] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 6. | Klein A, Ahlenstiel G, Tate DJ, Burgess N, Richardson A, Pang T, Byth K, Bourke MJ. Endoscopic resection of large duodenal and papillary lateral spreading lesions is clinically and economically advantageous compared with surgery. Endoscopy. 2017;49:659-667. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 25] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 7. | ASGE Standards of Practice Committee. Chathadi KV, Khashab MA, Acosta RD, Chandrasekhara V, Eloubeidi MA, Faulx AL, Fonkalsrud L, Lightdale JR, Salztman JR, Shaukat A, Wang A, Cash BD, DeWitt JM. The role of endoscopy in ampullary and duodenal adenomas. Gastrointest Endosc. 2015;82:773-781. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 116] [Cited by in RCA: 134] [Article Influence: 13.4] [Reference Citation Analysis (0)] |
| 8. | Moon JH, Choi HJ, Lee YN. Current status of endoscopic papillectomy for ampullary tumors. Gut Liver. 2014;8:598-604. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 27] [Cited by in RCA: 23] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 9. | Napoleon B, Gincul R, Ponchon T, Berthiller J, Escourrou J, Canard JM, Boyer J, Barthet M, Ponsot P, Laugier R, Helbert T, Coumaros D, Scoazec JY, Mion F, Saurin JC; Sociéte Française d’Endoscopie Digestive (SFED, French Society of Digestive Endoscopy). Endoscopic papillectomy for early ampullary tumors: long-term results from a large multicenter prospective study. Endoscopy. 2014;46:127-134. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 69] [Cited by in RCA: 74] [Article Influence: 6.7] [Reference Citation Analysis (0)] |
| 10. | Ogawa T, Ito K, Fujita N, Noda Y, Kobayashi G, Horaguchi J, Koshita S, Kanno Y, Masu K, Ishii S. Endoscopic papillectomy as a method of total biopsy for possible early ampullary cancer. Dig Endosc. 2012;24:291. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15] [Cited by in RCA: 19] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 11. | Hernandez LV, Catalano MF. Endoscopic papillectomy. Curr Opin Gastroenterol. 2008;24:617-622. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.4] [Reference Citation Analysis (0)] |
| 12. | Kim SH, Moon JH, Choi HJ, Kim DC, Lee TH, Cheon YK, Cho YD, Park SH, Kim SJ. Usefulness of pancreatic duct wire-guided endoscopic papillectomy for ampullary adenoma for preventing post-procedure pancreatitis. Endoscopy. 2013;45:838-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 28] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 13. | Poincloux L, Scanzi J, Goutte M, Pereira B, Devaud H, Joubert J, Dapoigny M, Bommelaer G, Abergel A. Pancreatic intubation facilitated by methylene blue injection decreases the risk for postpapillectomy acute pancreatitis. Eur J Gastroenterol Hepatol. 2014;26:990-995. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 16] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 14. | Han J, Kim MH. Endoscopic papillectomy for adenomas of the major duodenal papilla (with video). Gastrointest Endosc. 2006;63:292-301. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 93] [Cited by in RCA: 83] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 15. | Lee SK, Kim MH, Seo DW, Lee SS, Park JS. Endoscopic sphincterotomy and pancreatic duct stent placement before endoscopic papillectomy: are they necessary and safe procedures? Gastrointest Endosc. 2002;55:302-304. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 13] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 16. | Moon JH, Cha SW, Cho YD, Ryu CB, Cheon YK, Kwon KW, Kim YS, Kim YS, Lee JS, Lee MS, Shim CS, Kim BS. Wire-guided endoscopic snare papillectomy for tumors of the major duodenal papilla. Gastrointest Endosc. 2005;61:461-466. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 37] [Article Influence: 1.9] [Reference Citation Analysis (0)] |
| 17. | Attaallah W, Gunal O, Mokhtare S, Ozmen T, Cingi A. Endoscopic snare excision of adenoma of the papilla of Vater without prophylactic pancreatic-duct stent. JOP. 2014;15:587-590. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 3] [Reference Citation Analysis (0)] |
| 18. | Napoléon B, Alvarez-Sanchez MV, Leclercq P, Mion F, Pialat J, Gincul R, Ribeiro D, Cambou M, Lefort C, Rodríguez-Girondo M, Scoazec JY. Systematic pancreatic stenting after endoscopic snare papillectomy may reduce the risk of postinterventional pancreatitis. Surg Endosc. 2013;27:3377-3387. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 24] [Article Influence: 2.0] [Reference Citation Analysis (0)] |
| 19. | Yamao T, Isomoto H, Kohno S, Mizuta Y, Yamakawa M, Nakao K, Irie J. Endoscopic snare papillectomy with biliary and pancreatic stent placement for tumors of the major duodenal papilla. Surg Endosc. 2010;24:119-124. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 58] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 20. | Katsinelos P, Paroutoglou G, Kountouras J, Beltsis A, Papaziogas B, Mimidis K, Zavos C, Dimiropoulos S. Safety and long-term follow-up of endoscopic snare excision of ampullary adenomas. Surg Endosc. 2006;20:608-613. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 56] [Cited by in RCA: 51] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 21. | Ghidirim G, Mişin I, Istrate V, Cazacu S. Endoscopic papillectomy into the treatment of neoplastic lesions of vater papilla. Curr Health Sci J. 2009;35:92-97. [PubMed] |
| 22. | Irani S, Arai A, Ayub K, Biehl T, Brandabur JJ, Dorer R, Gluck M, Jiranek G, Patterson D, Schembre D, Traverso LW, Kozarek RA. Papillectomy for ampullary neoplasm: results of a single referral center over a 10-year period. Gastrointest Endosc. 2009;70:923-932. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 145] [Cited by in RCA: 113] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 23. | Jung MK, Cho CM, Park SY, Jeon SW, Tak WY, Kweon YO, Kim SK, Choi YH. Endoscopic resection of ampullary neoplasms: a single-center experience. Surg Endosc. 2009;23:2568-2574. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 36] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 24. | Igarashi Y, Okano N, Ito K, Mimura T, Nakano S. Endoscopic snare excision of a major duodenal papillary tumor. Dig Surg. 2010;27:119-122. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 9] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 25. | Ahn DW, Ryu JK, Kim J, Yoon WJ, Lee SH, Kim YT, Yoon YB. Endoscopic papillectomy for benign ampullary neoplasms: how can treatment outcome be predicted? Gut Liver. 2013;7:239-245. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 16] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 26. | Will U, Müller AK, Fueldner F, Wanzar I, Meyer F. Endoscopic papillectomy: data of a prospective observational study. World J Gastroenterol. 2013;19:4316-4324. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 28] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 27. | De Palma GD, Luglio G, Maione F, Esposito D, Siciliano S, Gennarelli N, Cassese G, Persico M, Forestieri P. Endoscopic snare papillectomy: a single institutional experience of a standardized technique. A retrospective cohort study. Int J Surg. 2015;13:180-183. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 28. | Soma H, Miyata N, Hozawa S, Higuchi H, Yamagishi Y, Nakamura Y, Saeki K, Kameyama K, Masugi Y, Yahagi N, Kanai T. Ampullary Adenoma Treated by Endoscopic Double-Snare Retracting Papillectomy. Gut Liver. 2015;9:689-692. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 1.0] [Reference Citation Analysis (0)] |
| 29. | Tsuji S, Itoi T, Sofuni A, Mukai S, Tonozuka R, Moriyasu F. Tips and tricks in endoscopic papillectomy of ampullary tumors: single-center experience with large case series (with videos). J Hepatobiliary Pancreat Sci. 2015;22:E22-E27. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 30. | Attila T, Parlak E, Alper E, Dişibeyaz S, Çiçek B, Ödemiş B. Endoscopic papillectomy of benign ampullary lesions: Outcomes from a multicenter study. Turk J Gastroenterol. 2018;29:325-334. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 17] [Cited by in RCA: 18] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 31. | Kobayashi M, Ryozawa S, Iwano H, Araki R, Tanisaka Y, Fujita A, Kobatake T. The usefulness of wire-guided endoscopic snare papillectomy for tumors of the major duodenal papilla. PLoS One. 2019;14:e0211019. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 32. | Pérez-Cuadrado-Robles E, Piessevaux H, Moreels TG, Yeung R, Aouattah T, Komuta M, Dano H, Jouret-Mourin A, Deprez PH. Combined excision and ablation of ampullary tumors with biliary or pancreatic intraductal extension is effective even in malignant neoplasms. United European Gastroenterol J. 2019;7:369-376. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 29] [Cited by in RCA: 18] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 33. | Lee R, Huelsen A, Gupta S, Hourigan LF. Endoscopic ampullectomy for non-invasive ampullary lesions: a single-center 10-year retrospective cohort study. Surg Endosc. 2020;. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 19] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 34. | Taglieri E, Micelli-Neto O, Bonin EA, Goldman SM, Kemp R, Dos Santos JS, Ardengh JC. Analysis of risk factors associated with acute pancreatitis after endoscopic papillectomy. Sci Rep. 2020;10:4132. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 10] [Cited by in RCA: 12] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 35. | De Palma GD. Endoscopic papillectomy: indications, techniques, and results. World J Gastroenterol. 2014;20:1537-1543. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 63] [Cited by in RCA: 58] [Article Influence: 5.3] [Reference Citation Analysis (2)] |
| 36. | Kang SH, Kim KH, Kim TN, Jung MK, Cho CM, Cho KB, Han JM, Kim HG, Kim HS. Therapeutic outcomes of endoscopic papillectomy for ampullary neoplasms: retrospective analysis of a multicenter study. BMC Gastroenterol. 2017;17:69. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 55] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 37. | Ismail S, Marianne U, Heikki J, Jorma H, Leena K. Endoscopic papillectomy, single-centre experience. Surg Endosc. 2014;28:3234-3239. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 21] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 38. | Boix J, Lorenzo-Zúñiga V, Moreno de Vega V, Domènech E, Gassull MA. Endoscopic resection of ampullary tumors: 12-year review of 21 cases. Surg Endosc. 2009;23:45-49. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 52] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 39. | Nguyen N, Shah JN, Binmoeller KF. Outcomes of endoscopic papillectomy in elderly patients with ampullary adenoma or early carcinoma. Endoscopy. 2010;42:975-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 18] [Cited by in RCA: 12] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 40. | Ito K, Fujita N, Noda Y, Kobayashi G, Obana T, Horaguchi J, Koshita S, Kanno Y, Ogawa T, Kato Y, Yamashita Y. Impact of technical modification of endoscopic papillectomy for ampullary neoplasm on the occurrence of complications. Dig Endosc. 2012;24:30-35. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 38] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 41. | Hyun JJ, Lee TH, Park JS, Han JH, Jeong S, Park SM, Lee HS, Moon JH, Park SH. A prospective multicenter study of submucosal injection to improve endoscopic snare papillectomy for ampullary adenoma. Gastrointest Endosc. 2017;85:746-755. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 42. | Chang WI, Min YW, Yun HS, Lee KH, Lee JK, Lee KT, Rhee PL. Prophylactic pancreatic stent placement for endoscopic duodenal ampullectomy: a single-center retrospective study. Gut Liver. 2014;8:306-312. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 43. | Leal-Lopes C, Velloso FJ, Campopiano JC, Sogayar MC, Correa RG. Roles of Commensal Microbiota in Pancreas Homeostasis and Pancreatic Pathologies. J Diabetes Res. 2015;2015:284680. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 33] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 44. | Allison MC, Sandoe JA, Tighe R, Simpson IA, Hall RJ, Elliott TS; Endoscopy Committee of the British Society of Gastroenterology. Antibiotic prophylaxis in gastrointestinal endoscopy. Gut. 2009;58:869-880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 95] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 45. | Ito K, Fujita N, Noda Y. Endoscopic diagnosis and treatment of ampullary neoplasm (with video). Dig Endosc. 2011;23:113-117. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 32] [Cited by in RCA: 29] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 46. | van Geenen EJ, van der Peet DL, Bhagirath P, Mulder CJ, Bruno MJ. Etiology and diagnosis of acute biliary pancreatitis. Nat Rev Gastroenterol Hepatol. 2010;7:495-502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 71] [Article Influence: 4.7] [Reference Citation Analysis (0)] |
| 47. | Högström H, Haglund U, Zederfeldt B. Beneficial effect of proteinase inhibitors on early breaking strength of intestinal anastomoses. Acta Chir Scand. 1985;151:529-532. [PubMed] |
| 48. | Meddahi A, Benoit J, Ayoub N, Sézeur A, Barritault D. Heparin-like polymers derived from dextran enhance colonic anastomosis resistance to leakage. J Biomed Mater Res. 1996;31:293-297. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 2] [Reference Citation Analysis (0)] |
| 49. | Shogan BD, Belogortseva N, Luong PM, Zaborin A, Lax S, Bethel C, Ward M, Muldoon JP, Singer M, An G, Umanskiy K, Konda V, Shakhsheer B, Luo J, Klabbers R, Hancock LE, Gilbert J, Zaborina O, Alverdy JC. Collagen degradation and MMP9 activation by Enterococcus faecalis contribute to intestinal anastomotic leak. Sci Transl Med. 2015;7:286ra68. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 204] [Cited by in RCA: 286] [Article Influence: 31.8] [Reference Citation Analysis (0)] |
| 50. | Innerfield I, Schwarz A, Angrist A. Intravenous trypsin; its anticoagulant, fibrinolytic and thrombolytic effects. J Clin Invest. 1952;31:1049-1055. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 54] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 51. | Sarkar NK. Mechanism of clot lysis. Nature. 1960;185:624-625. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 9] [Article Influence: 0.3] [Reference Citation Analysis (0)] |
| 52. | Zhang WG, Linghu EQ, Li HK. Fibrin sealant for closure of mucosal penetration at the cardia during peroral endoscopic myotomy: A retrospective study at a single center. World J Gastroenterol. 2017;23:1637-1644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 17] [Cited by in RCA: 9] [Article Influence: 1.1] [Reference Citation Analysis (0)] |
| 53. | Li H, Linghu E, Wang X. Fibrin sealant for closure of mucosal penetration at the cardia during peroral endoscopic myotomy (POEM). Endoscopy. 2012;44 Suppl 2 UCTN:E215-E216. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 21] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 54. | Mutignani M, Seerden T, Tringali A, Feisal D, Perri V, Familiari P, Costamagna G. Endoscopic hemostasis with fibrin glue for refractory postsphincterotomy and postpapillectomy bleeding. Gastrointest Endosc. 2010;71:856-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 23] [Article Influence: 1.5] [Reference Citation Analysis (0)] |
| 55. | de Rezende DT, Brunaldi VO, Bernardo WM, Ribeiro IB, Mota RCL, Baracat FI, de Moura DTH, Baracat R, Matuguma SE, de Moura EGH. Use of hemostatic powder in treatment of upper gastrointestinal bleeding: a systematic review and meta-analysis. Endosc Int Open. 2019;7:E1704-E1713. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 21] [Cited by in RCA: 17] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 56. | Pittayanon R, Prueksapanich P, Rerknimitr R. The efficacy of Hemospray in patients with upper gastrointestinal bleeding from tumor. Endosc Int Open. 2016;4:E933-E936. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 19] [Cited by in RCA: 25] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 57. | Arena M, Masci E, Eusebi LH, Iabichino G, Mangiavillano B, Viaggi P, Morandi E, Fanti L, Granata A, Traina M, Testoni PA, Opocher E, Luigiano C. Hemospray for treatment of acute bleeding due to upper gastrointestinal tumours. Dig Liver Dis. 2017;49:514-517. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 42] [Cited by in RCA: 38] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 58. | Hagel AF, Albrecht H, Nägel A, Vitali F, Vetter M, Dauth C, Neurath MF, Raithel M. The Application of Hemospray in Gastrointestinal Bleeding during Emergency Endoscopy. Gastroenterol Res Pract. 2017;2017:3083481. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 26] [Cited by in RCA: 29] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 59. | Ofosu A, Ramai D, Latson W, Adler DG. Endoscopic management of bleeding gastrointestinal tumors. Ann Gastroenterol. 2019;32:346-351. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 6] [Cited by in RCA: 13] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 60. | Baracat FI, de Moura DTH, Brunaldi VO, Tranquillini CV, Baracat R, Sakai P, de Moura EGH. Randomized controlled trial of hemostatic powder versus endoscopic clipping for non-variceal upper gastrointestinal bleeding. Surg Endosc. 2020;34:317-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 38] [Cited by in RCA: 28] [Article Influence: 5.6] [Reference Citation Analysis (0)] |