Published online Apr 14, 2020. doi: 10.3748/wjg.v26.i14.1601
Peer-review started: December 7, 2019
First decision: December 30, 2019
Revised: March 6, 2020
Accepted: March 27, 2020
Article in press: March 27, 2020
Published online: April 14, 2020
Processing time: 125 Days and 20.4 Hours
Some substances of plant origin have been reported to exert an effect in reducing intestinal neoplasm development, especially in animal models. Adenomatous polyposis coli multiple intestinal neoplasia - ApcMin/+ is the most studied murine model of genetic intestinal carcinogenesis.
To assess whether an enriched nutritional formulation (silymarin, boswellic acid and curcumin) with proven “in vitro” and “in vivo” anti-carcinogenetic properties may prevent inherited intestinal cancer in animal model.
Forty adenomatous polyposis coli multiple intestinal neoplasia - ApcMin/+ mice were used for the study of cancer prevention. They were divided into two groups: 20 assumed standard and 20 enriched diet. At the 110th d animals were sacrificed. In each group, four subgroups received intraperitoneal bromodeoxyuridine injection at different times (24, 48, 72 and 96 h before the sacrifice) in order to assess epithelial turnover. Moreover, we evaluated the following parameters: Intestinal polypoid lesion number and size on autoptic tissue, dysplasia and neoplasia areas by histological examination of the whole small intestine, inflammation by histology and cytokine mRNA expression by real-time polymerase chain reaction, bromodeoxyuridine and TUNEL immuno-fluorescence for epithelial turnover and apoptosis, respectively. Additionally, we performed western blotting analysis for the expression of estrogen alpha and beta receptors, cyclin D1 and cleaved caspase 3 in normal and polypoid tissues.
Compared to standard, enriched diet reduced the total number (203 vs 416) and the mean ± SD/animal (12.6 ± 5.0 vs 26.0 ± 8.8; P < 0.001) of polypoid lesions. In enriched diet group a reduction in polyp size was observed (P < 0.001). Histological inflammation and pro-inflammatory cytokine expression were similar in both groups. Areas of low-grade dysplasia (P < 0.001) and intestinal carcinoma (IC; P < 0.001) were significantly decreased in enriched diet group. IC was observed in 100% in standard and 85% in enriched formulation assuming animals. Enriched diet showed a faster epithelial migration and an increased apoptosis in normal mucosa and low-grade dysplasia areas (P < 0.001). At western blotting, estrogen receptor beta protein was well expressed in normal mucosa of enriched and standard groups, with a more marked trend associated to the first one. Estrogen receptor alpha was similarly expressed in normal and polypoid mucosa of standard and enriched diet group. Cleaved caspase 3 showed in normal mucosa a stronger signal in enriched than in standard diet. Cyclin D1 was more expressed in standard than enriched diet group of both normal and polypoid tissue.
Our results are suggestive of a chemo-preventive synergic effect of the components (silymarin, boswellic acid and curcumin) of an enriched formulation in inherited IC. This effect may be mediated by the reduction of epithelial proliferation, the increase of apoptosis and the acceleration of villous cell renewal due to dietary formulation intake.
Core tip: A dietetic formulation containing silymarin, boswellic acid and curcumin has shown “in vitro” and “in vivo” anti-carcinogenetic properties in animal model of inflammation-related intestinal carcinoma. Herein, we assessed whether it may prevent inherited intestinal cancer in animal model (adenomatous polyposis coli multiple intestinal neoplasia - ApcMin/+). Our results showed that the dietetic formulation reduced polypoid lesion number and size on autoptic tissue, histological dysplasia and neoplasia areas. This effect is related to increased epithelial renewal and apoptosis and decreased proliferation. Our data are suggestive of a chemo-preventive synergic effect of the components of the dietetic formulation in inherited intestinal carcinoma.
- Citation: Girardi B, Pricci M, Giorgio F, Piazzolla M, Iannone A, Losurdo G, Principi M, Barone M, Ierardi E, Di Leo A. Silymarin, boswellic acid and curcumin enriched dietetic formulation reduces the growth of inherited intestinal polyps in an animal model. World J Gastroenterol 2020; 26(14): 1601-1612
- URL: https://www.wjgnet.com/1007-9327/full/v26/i14/1601.htm
- DOI: https://dx.doi.org/10.3748/wjg.v26.i14.1601
Colorectal cancer (CRC) is the conclusive result of a progressive phenomenon that, in most cases, implies a succession of events (normal, pre-cancerous and neoplastic conditions)[1-3]. The progression to cancer may be due to genetic mutations leading to dysplasia and, then, to carcinoma, as in familial adenomatous polyposis and Lynch syndrome (inherited models of CRC). In humans, APC gene mutation is the genetic basis to carcinogenesis, making intestinal cells predisposed to cancer promotion and evolution with additional mutations by epigenetic changes, mostly affected by environmental stimuli[4,5].
Some substances of plant origin have been reported to exert an effect in reducing intestinal neoplasm development, especially in animal models. In detail, silymarin, a phytoestrogen compound derived from milk thistle (Silybum marianum) may decrease intestinal carcinogenesis through both anti-oxidant and estrogen receptor (ER)-beta agonist properties[6,7]. Previous studies have shown that silymarin is able to hamper intestinal carcinoma (IC) development in ApcMin/+ mice and familial adenomatous polyposis patients with ileal pouch-anal anastomosis[8,9]. Moreover, a case report suggests a similar effect even in Lynch syndrome[10]. Boswellia serrata is a plant with anti-inflammatory properties. Interestingly, boswellic acids, especially Acetyl-11-Keto-beta-Boswellic Acid (AKBA), a component of the gum resin of Boswellia serrata, has been recognized as a promising agent for the prevention of intestinal tumorigenesis in a mouse model of inherited carcinogenesis, i.e., APC multiple intestinal neoplasia (min) animals. The ApcMin/+ experimental model mimics familial adenomatous polyposis, even if the disease, differently from humans, is mainly confined to the small bowel and only minimally involves colonic district[11-13]. Curcuma is a phytochemical derived from turmeric (Curcuma longa), a plant similar to ginger. It has been demonstrated to exert anti-inflammatory and anti-neoplastic properties by interacting with several molecular targets, i.e., transcription factors, enzymes, cell cycle proteins, cytokines, receptors and adhesion molecules[14-16].
The background of this study was based on the possibility that a mixture of phytochemicals may result in health benefits more than what supplied by single components[17,18]. Therefore, in a previous experiment we tested “in vitro” the effect of the elements of a nutritional combination as well as the complete mixture on the proliferation of cultured colo-rectal neoplastic cells. Every molecule (silymarin, boswellic acids and curcumin) showed a relevant anti-proliferative action in comparison with control samples. In addition, the mixture of the three molecules significantly inhibited cellular growth more than single or double combination[19]. Moreover, in the same study, a nutritional formulation based on the combination of silymarin, boswellic acids and curcumin clearly demonstrated an anti-inflammatory and chemopreventive effect “in vivo” in an animal model of colorectal carcinoma arising from inflamed tissue[19].
On these bases, the present study had the primary aim of assessing whether the effect of the nutritional formulation (enriched dietary supplement) could exert an inhibitory activity on intestinal carcinogenesis in ApcMin/+ animal model. For this purpose, the dose of every substance was given “in vivo” under the maximal effective amount of single components. Furthermore, the doses were in agreement with their bioavailability based on daily intake amount able to achieve an appropriate plasma concentration by a complete intestinal absorption[20]. Additionally, some mechanistic features were investigated.
Forty ApcMin/+ animals were used for the experimental design. They were kept in controlled conditions of temperature, air and light (from 7 a.m. to 7 p.m.) and received food and water ad libitum. Animals did not receive any surgical or hormonal manipulation, but they were kept anatomically and physiologically intact. All animals received care in agreement with the “Guide for the Care and Use of Laboratory Animals”[21]. The study protocol was approved by the University of Bari Committee for Animal Experimentation (protocol number 6/12).
Dietary procedures were started at the 10th wk of age after a two-week period of settling. Forty wild type animals (20 receiving standard diet and 20 modified diet - enriched formulation, THD SpA, Correggio, Italy) were used for the preliminary assessment of the dietary supplement safety. Forty ApcMin/+ animals (20 receiving standard diet and 20 modified diet - enriched formulation) were used for the evaluation of dietary mixture chemopreventive effect. The feeding procedures, used in both phases of the study, were: (1) Standard diet (Harlan Teklad Rodent diet): 18.5% proteins, 3% oils and fats, 6% fibers, 7% crude ash and 65.5% of non-nitrogenous compounds - wheat, maize, toasted soybean meal, corn gluten feed, wheat straw, fish meal, Lucerne meal, mineral bicalcium phosphate, calcium carbonate, sodium chloride, whey powder, soybean oil, yeast and hazelnut skins, poly-vitamin complex (Mucedola Srl, Settimo Milanese, Italy); and (2) Modified Diet (enriched formulation): The standard diet was enriched by a formulation containing silymarin (4 g%), AKBA (3 g%), curcumin (2 g%), maltodextrins (69.553 g%) and excipients (soluble fibers 16.667 g%, citric acid 1 g%, silicon dioxide 1 g%, lignans 0.5 g%, sucralose 0.280 g%, orange flavor 2 g%) and was administered at the cumulative dose of 22.4 mg/100 g of body weight.
The enrichment dietetic formulation was established taking into account that: (1) Mice with a body weight of 20-40 g usually eat about 5 g of daily food amount (16.7 g/100 g of body weight according to Italian Association for Laboratory Animal Science, November 2012)[22]; (2) Daily intake was 0.892 mg of silymarin, 0.672 mg of AKBA and 0.448 mg of curcumin per 100 g of body weight for animal. In detail, these amounts were administered below the maximum order of magnitude of the effective concentrations of the single substances against cancer in animals[20]; and (3) Our supposition was that these substances, similarly to what observed “in vitro”, could exert beneficial effects beyond what provided by the single phytochemical.
ApcMin/+ mice were sacrificed 110 d after starting dietetic formulation assumption. All animals received intraperitoneal administration of bromodeoxyuridine (BrdU, 50 mg/kg) for epithelia turnover assessment. In both groups, mice were divided into four subgroups and sacrificed under metaphane anesthesia at 24th, 48th, 72th or 96th h after the injection, respectively. The small bowel from each animal was collected, washed and dissected along the longitudinal axis in order to evaluate both macroscopic and microscopic neoplastic lesions along the whole organ. After macroscopic evaluation, small and large bowel were fixed in 10% buffered formalin and embedded in paraffin. Samples from each animal were frozen at -80 °C for molecular biology and western blotting analyses. Macroscopic evaluation of polypoid lesions was based on their division into three groups according to the size: Small (< 3 mm), intermediate (3-7 mm) and large (> 7 mm).
Sections (4 micron thick) were sequentially stained with haematoxylin and eosin for histological examination. Inflammation score (range 0-5) was evaluated in normal tissue according to Yu et al[23]. For each animal, the number of low-grade dysplasia (LGD) and high-grade dysplasia (HGD) and IC areas were recorded along the whole small bowel and expressed as mean ± SD/animal. All tissues were examined by two expert observers in blind.
Pro-inflammatory cytokine [interferon gamma, interleukin 6 and tumor necrosis factor (TNF) alpha] gene expression was assessed by real time polymerase chain reaction to assess the amount of specific mRNA in frozen samples without polypoid lesions. Results were expressed as fold change compared to control. RNA was extracted from five sections of 10 microns, using QIAgen RNA mini kit (QIAGEN GmbH, Germany). Two-step reverse transcription polymerase chain reaction was performed using first-strand cDNA with a final concentration of 1 × TaqMan gene expression assay, i.e., TNF alpha and glyceraldehyde-3-phosphate-dehydrogenase (Applied Biosystems, Foster City, CA). The final reaction volume was 25 microliters and analyzed in triplicate (all experiments were repeated twice). A non-template control (RNAse-free water) was included on every plate. A further validation of our method was performed by enclosing in each assay fresh samples from at least three healthy mouse colonic mucosa. Specific thermal cycler conditions were employed by real time polymerase chain reaction System (Applied Biosystems, Foster City, CA, United States). A standard curve plus validation experiment were performed for each primer/probe set. A series of 6 serial dilutions (20 to 0.1 ng/microl) of tissue cDNA were used as a template.
BrdU expression in epithelial cells was investigated by a monoclonal mouse antibody cell signaling (Novus Biologicals, Milan, Italy). Sections were rinsed in PBS buffer with TWEEN 0.025% for 10 min and incubated in microwave oven (citric buffer pH 6.0, 10 min, 750 W) for antigen unmasking. Then, they were treated (2 h; room temperature) in 10% goat serum and 1% bovine serum albumin blocking solution. Successively, they were incubated with anti-BrdU antibody diluted 1:50 at 4 °C overnight. Alexa 488 fluorescent-conjugated goat anti-mouse (Invitrogen, Life Technologies, Monza, Italy) at a dilution of 1:200 represented the secondary antibody. All sections were observed with confocal microscopy at 630 × magnification. Ten well-oriented crypt/villous areas of histologically normal small bowel were selected for the analysis. Cell proliferation in the crypts and migration towards mucosal free surface (epithelial turnover) were evaluated, using the highest labeled cell along the villus as marker of the percentage of covered axis[24]. The analysis was performed in four subgroups in both enriched and standard diet in relation to the time of sacrifice after BrdU injection (i.e., 24, 48, 72 and 96 h).
Apoptosis was evaluated by TUNEL (Cell Death Detection kit, Roche, Mannheim, Germany). Briefly, sections were de-waxed and incubated in 0.1 mol citrate buffer (pH = 6.0) in microwave oven at 350 W for 10 min. Then, sections were incubated with TUNEL probe at 37 °C for one hour and counterstained with TOPRO-3 at a dilution of 1:3000 (Invitrogen Molecular Probes). Slides were analyzed with confocal microscopy at 630 × magnification. The count of marked cells was performed on 10 well-oriented crypt/villous configurations. Labeling index, i.e., percentage of positive cells, was used to quantify apoptosis. Cell count was executed in IC, HGD, LGD and normal tissue. Due to the large specimen size, all histological pictures were found in every section. Two expert observers analyzed the slides in blind.
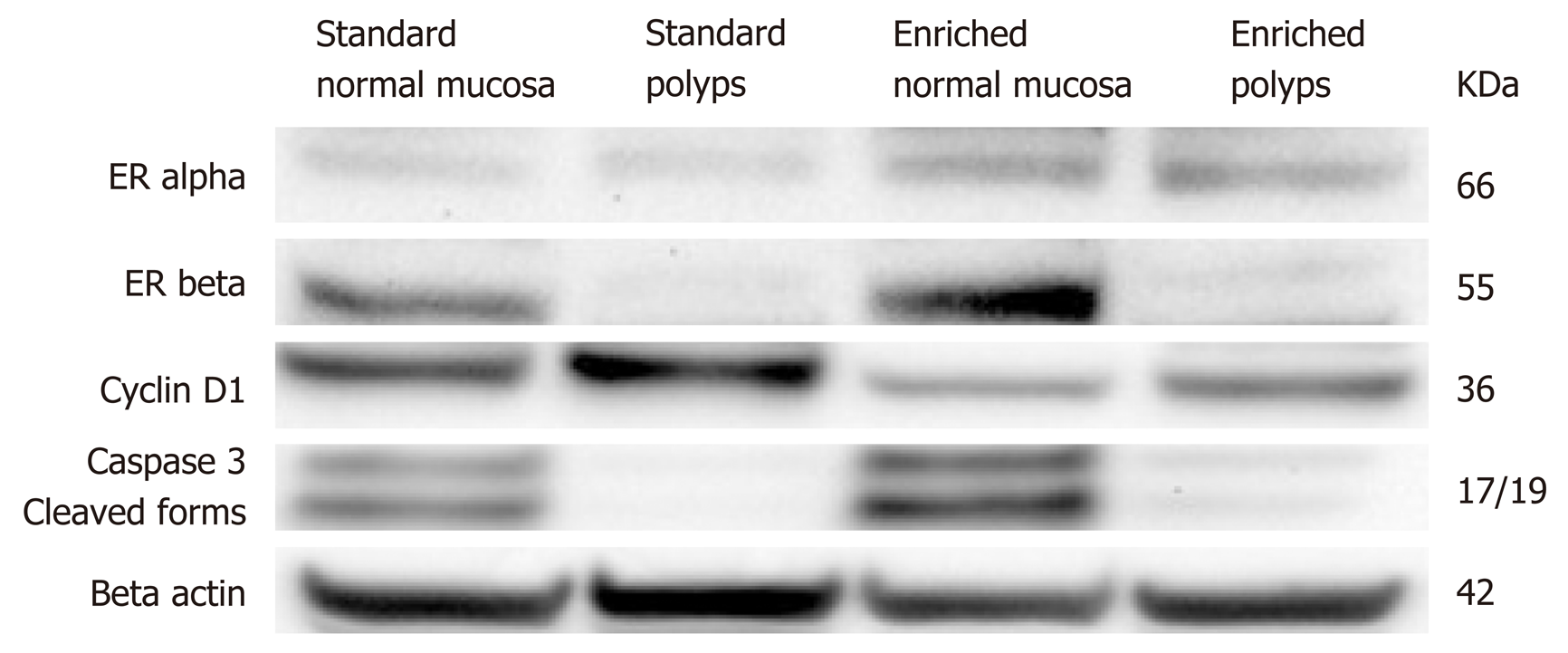
Frozen samples were homogenized in lysis buffer (25 mmol Tris-HCl pH 7.6, 150 mmol NaCl, 1% NP-40, 1% sodium deoxycholate, 0.1% sodium dodecyl sulphate, protease inhibitor cocktail; Roche Diagnostics, Germany). Lysates underwent centrifugation at 14000 rpm for 30 min a 4 °C. Protein levels were assayed with Bradford method (Bradford Reagent - Bio Rad, Milano, Italy). Aliquots containing 40 μg of total proteins were separated in 4%-12% precast polyacrylamide gels (Invitrogen, Thermo Fisher Scientific, Monza, Italy) and transferred to nitrocellulose membranes (Bio-Rad) for 1 h at 300 mA. Ponceau S stain (Sigma, Milano, Italy) was used to verify the even and complete transfer of proteins onto the membrane. The membranes were blocked with Tris-buffered saline (TBS) containing 5% dry milk powder and 0.1% Tween-20 (Sigma) for 2 h at room temperature. This step was followed by an overnight incubation at 4 °C with primary rabbit antibodies, diluted in TBS 0.1% Tween (T-TBS) with 5% dry milk powder, of ER alpha (Invitrogen cat. no. PA5-34577 - 1 µg/mL), ER beta (Invitrogen cat.no. PA1-310B – 1 µg/mL), Cyclin D1 (Invitrogen cat.no. MA5-14512 - 1:100), Caspase-3 cleaved forms (Invitrogen cat.no. 700182- 0.2 µg/mL) and Beta Actin, as housekeeping protein (Invitrogen cat.no. PA1-183 - 1:1000). After repeated washing with T-TBS, the membranes were incubated for 1 h at room temperature with a horseradish peroxidase conjugated secondary anti-rabbit antibody (Invitrogen cat.no. A27036 - 1:20000). The proteins were detected using an enhanced chemiluminescent substrate (ECL - Super Signal West Pico - Thermo Scientific) for the detection of horseradish peroxidase and the signal density of every protein obtained by the Molecular Imager ChemidocTM (Bio-Rad Laboratories).
The comparison of continuous variables in the two groups was performed by Student’s t test for unpaired data. One-way analysis of variance corrected by Bonferroni’s test was used to compare TUNEL expression in normal mucosa, LGD, HGD and IC. Categorical data were evaluated by Fisher’s exact test or chi squared test for trend. Values of P < 0.05 (two tails) were considered significant. The statistical software GraphPad Prism 5 (San Diego, California, United States) was used.
Enriched diet safety test had been performed in a previous experience[19] and did not show any adverse event.
In the enriched diet group, the total length of the small bowel was similar to that of standard diet group (26.4 cm ± 1.2 cm vs 25.7 cm ± 1.3 cm; P = 0.17).
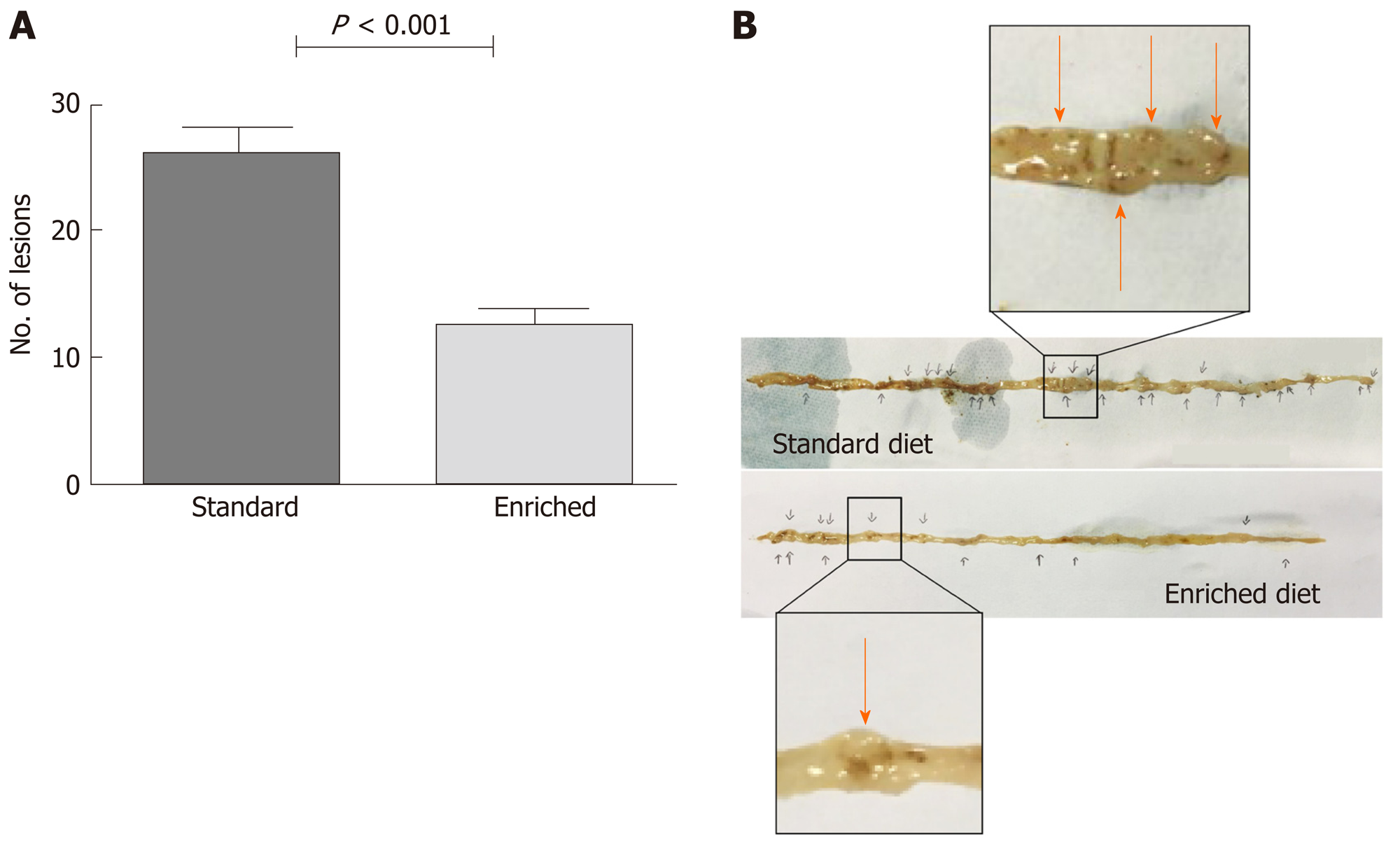
A very small amount of polypoid lesions was observed in the colon at sacrifice (0.6 ± 0.8 vs 0.8 ± 0.9 in the enriched and standard diet group, respectively; P = 0.54). The total number of solid lesions observed in the small bowel of sacrificed animals was 416 in the standard diet and 203 in the enriched diet group. In detail, the number of polypoid lesions per animal in the small bowel was reduced in the enriched compared to the standard diet group (12.6 ± 5.0 vs 26.0 ± 8.8; P < 0.001) (Figure 1).
The evaluation of polypoid lesion size showed that: (1) In the standard diet group, lesion size was > 7 mm (large) in 9/416 (2.2%), 4-7 mm (intermediate) in 205/416 (49.2%) and < 3 mm (small) in 202/416 (48.6%); (2) In the enriched diet group, the lesion size was > 7 mm (large) in 3/203 (1.4%), 4-7 mm (intermediate) in 57/203 (28.1%) and < 3 mm (small) in 143/203 (70.5%). The test for trend demonstrated that solid lesion size was significantly reduced (P < 0.001) in the enriched diet group.
There was no significant difference in the histological score of inflammation between the two groups (1.2 ± 0.4 in the standard and 1.5 ± 0.5 in the enriched diet group, P = 0.09) in normal tissue. Both scores indicate that inflammation was almost absent in ApcMin/+ mice in non-dysplastic/neoplastic areas.
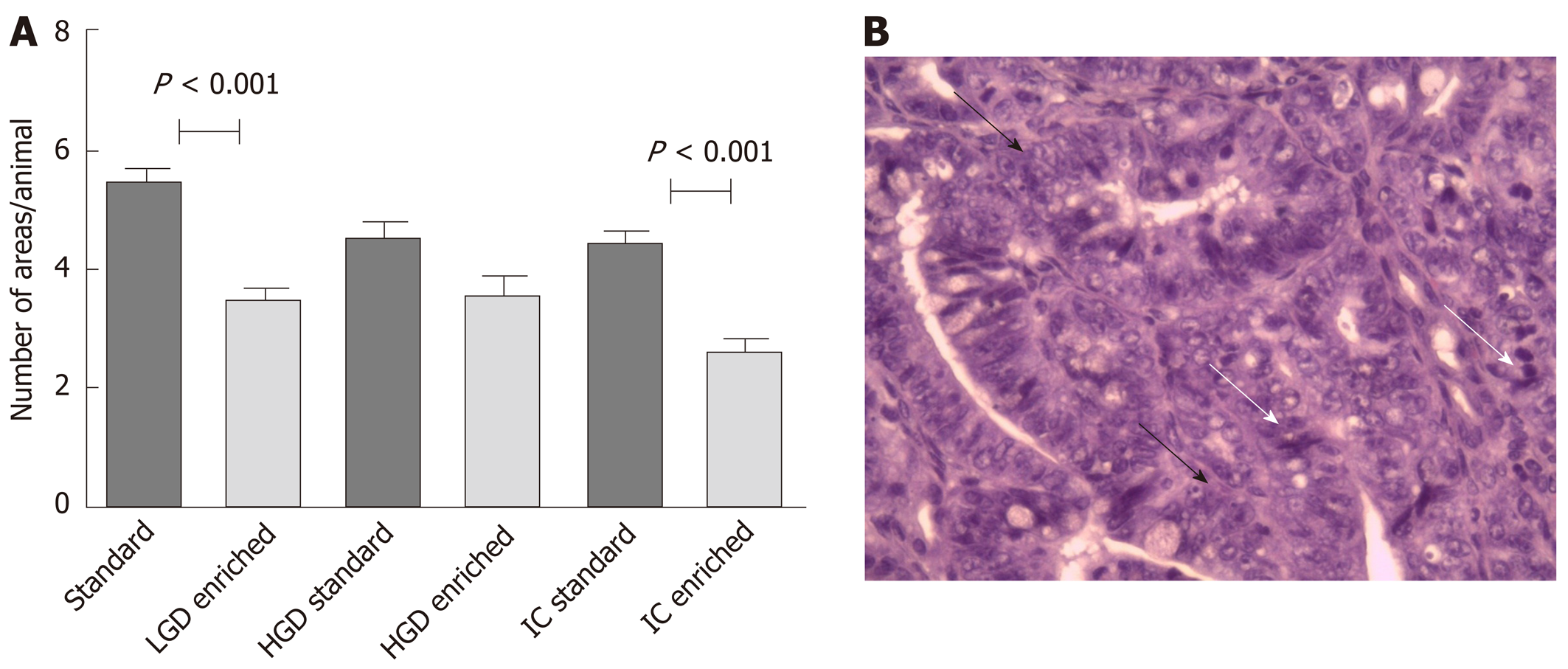
The mean number of LGD areas per mouse, recorded along the whole small bowel and expressed as mean ± SD/animal, was 5.4 ± 1.2 in the standard and 3.3 ± 1.2 in the enriched diet group (P < 0.001). The number of HGD areas was 4.5 ± 1.0 in the standard and 4.1 ± 1.3 in the enriched diet group (P = 0.10). The number of IC areas was 4.5 ± 0.7 in the standard and 2.6 ± 0.7 in the enriched diet group (P < 0.001). Results are summarized in Figure 2A, while in Figure 2B and an explanatory example of IC histological picture is shown.
IC was observed in 20/20 (100%) of the standard and 17/20 (85%) of the enriched diet group (P = 0.22).
Pro-inflammatory cytokine mRNA expression did not statistically differ in normal small bowel of the mice assuming the standard or the enriched diet. In detail, interferon-gamma showed a value of 0.5 ± 0.3 (standard) vs 0.5 ± 0.4 (enriched diet) fold-change; interleukin 6 showed a value of 0.4 ± 0.3 (standard) vs 0.4 ± 0.3 (enriched diet) fold-change; TNF alpha showed a value of 0.5 ± 0.4 (standard) vs 0.8 ± 0.4 (enriched diet) fold-change.
Epithelial turnover: In both standard and enriched diet groups, we observed in normal mucosa that positive cells were predominantly located in the crypts after 24 h, while a progression towards the villous district was observed after 48 h. In addition, we found positive cells only near the villous free surface at 72nd and 96th h with a decreasing number through the time progression.
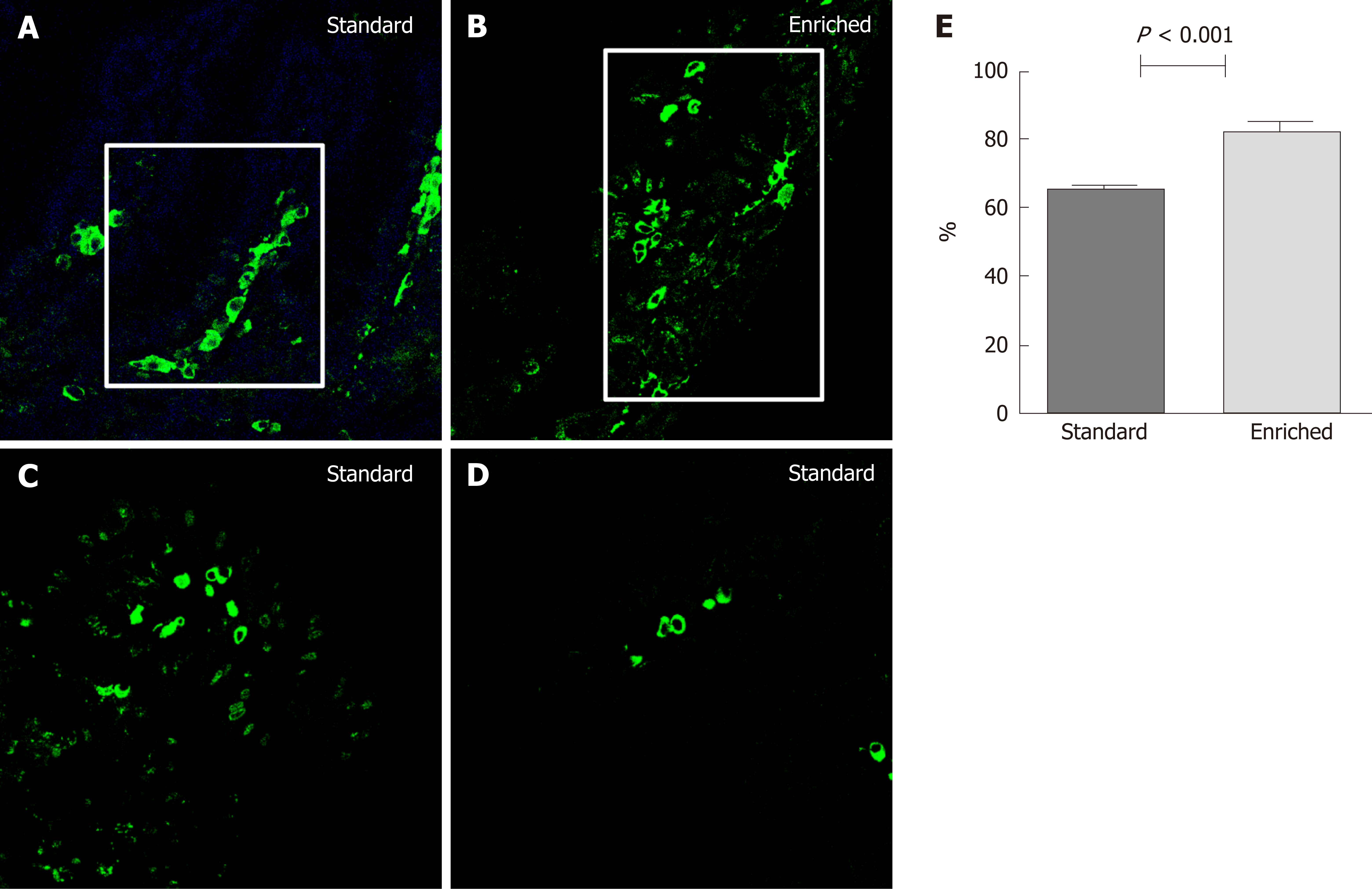
Figure 3 illustrates the percentage of villous axis covered by the highest labeled cell at the 48th h after injection in normal mucosa. This parameter was significantly higher in the enriched than in the standard diet group (82.2 ± 6.4 vs 65.3 ± 5.6 percent; P < 0.001). In LGD areas of the two groups, we found positive cells in both crypt and villous areas even at 24th h. Positive cells were found only at villous level after 48, 72 and 96 h with a decreasing LI. In HGD and IC the complete alteration of the crypt/villous structure did not allow evaluating the position of BrdU positive cells.
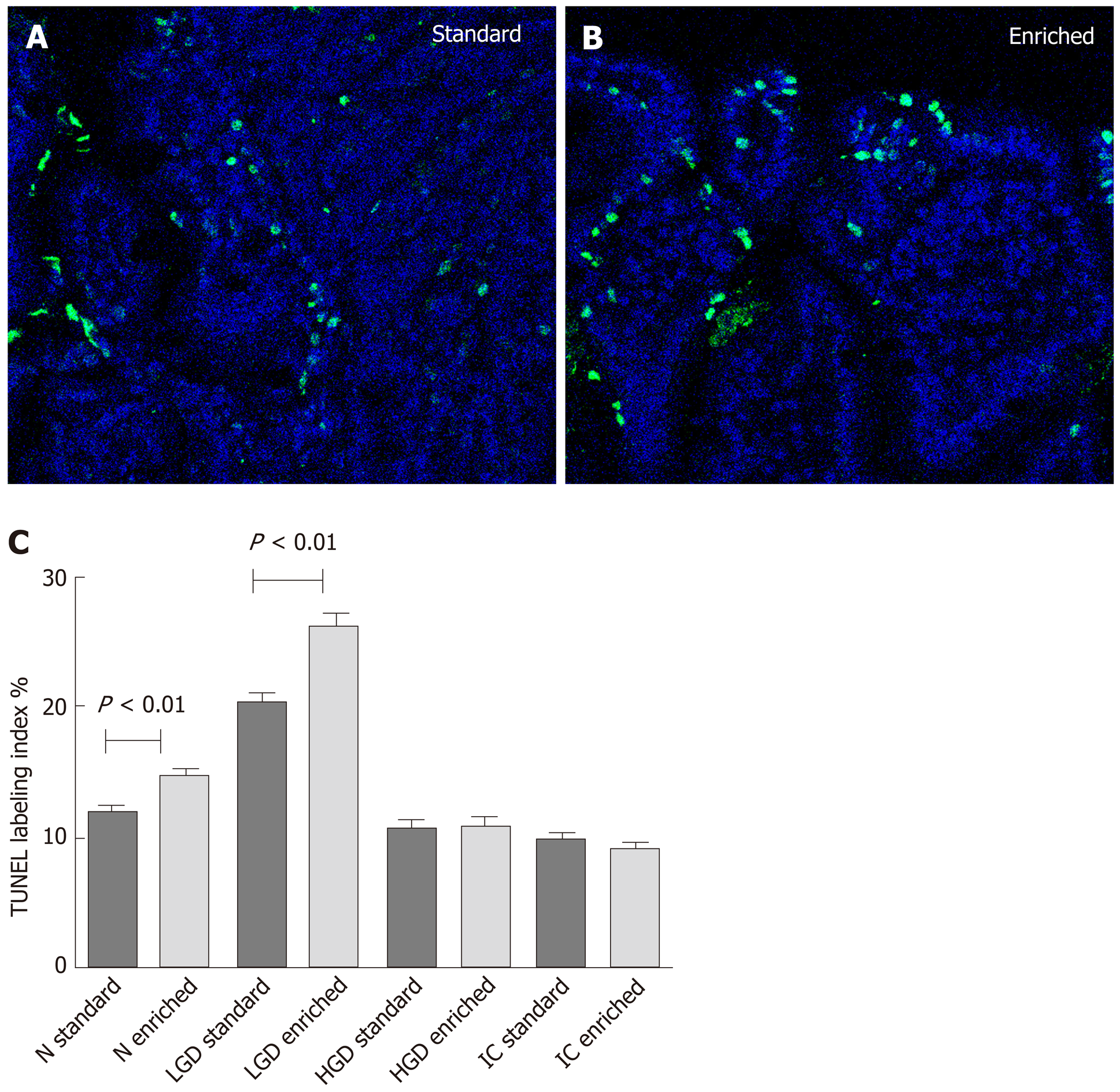
Epithelial apoptosis: Immuno-fluorescent TUNEL expression is shown in Figure 4. Statistical analysis displayed: N < LGD > HGD = IC both in the standard and enriched diet group (P < 0.001 for both groups). However, a significant higher level of epithelial apoptosis was observed in the enriched compared to the standard diet group in normal (P < 0.01) and LGD (P < 0.001) areas.
Western blotting findings are summarized in Figure 5. In detail we did not find any difference in ER alpha expression between normal and polypoid tissue, despite an increased protein level was observed in enriched compared to standard group. Conversely, ER beta protein was poorly expressed in polyps of both groups, while it was well expressed in normal mucosa of enriched and standard diet with a more marked signal in the first group. Cyclin D1 was more expressed showed lower levels in polyps of enriched compared to standard diet animals. Moreover, its expression was less evident in normal tissue of enriched compared to standard diet. The protein of cleaved form of caspase 3 showed a poor expression in polyps of both groups, while evident levels were found in normal mucosa of both groups with a stronger signal in enriched than in standard diet.
This study was planned to assess whether an enriched dietary supplement containing silymarin, boswellic acid and curcumin, could protect against polypoid lesion onset in a murine model of inherited intestinal cancer (ApcMin/+). The hypothesis of this study was that the combinations of phytochemicals under the maximal effective amount of single components may result in health benefits more than what supplied by single components. In order to support this hypothesis, in a previous experiment we tested “in vitro” the effect the elements of a nutritional combination as well as the complete mixture on the proliferation of cultured colo-rectal neoplastic cells. Every molecule (silymarin, boswellic acids and curcumin) showed a relevant anti-proliferative action in comparison with control samples. In addition, the mixture of the three molecules significantly inhibited cellular growth more than single or double combination[19]. Moreover, in the same study, the nutritional formulation based on the combination of silymarin, boswellic acids and curcumin clearly demonstrated an anti-inflammatory and chemopreventive effect in an animal model of colorectal carcinoma arising from inflamed tissue[19].
ApcMin/+ model mimics familial adenomatous polyposis even if, unlike humans, almost all of the polyps are located in the small bowel more than in the colon and duodenum[25]. However, the adenoma-carcinoma sequence is well represented[26].
The main result of this study concerns the effect of the dietetic formulation on carcinogenesis. Indeed, we found small bowel carcinomas in the 85% of the animals receiving enriched diet, while cancer was seen in the 100% of the standard diet group. However, enriched diet was associated with a significant decrease in the number and size of polypoid lesions. Finally, in mice assuming enriched diet a significant decrease of areas of dysplasia and carcinoma was detected along the whole small bowel when compared to animals fed with the standard diet.
Despite dietetic formulation has a well-demonstrated anti-inflammatory effect[19], we could not demonstrate it in ApcMin/+ animals for the almost absence of mucosal inflammation in normal mucosa. Inflammation evaluation was performed in non-dysplastic/neoplastic tissue, since our aim was to assess its role in carcinogenetic process. Therefore, we did not detect this parameter in pre-cancerous or cancerous lesions, where it is commonly found as a consequence of necrosis and not as a pro-carcinogenetic factor. As expected, we found a very low inflammatory histological score as well as a poor mucosal pro-inflammatory cytokine mRNA expression. Presumably, this finding suggests that anti-neoplastic effect of enriched dietetic formulation may not be mediated by dietetic formulation anti-inflammatory activity. Therefore, other mechanistic potential anti-neoplastic effects may be invoked.
A possible explanation for the anti-neoplastic effect of dietary treatment could be related to the well-demonstrated anti-oxidant and ER beta agonist effects of silymarin[7,27,28]. Indeed, Calabrese et al[29] demonstrated that a dietetic supplementation with silymarin induces a significant reduction of number and size of duodenal polyps in patients with familial adenomatous polyposis in the course of the follow up after ileal pouch-anal anastomosis. Moreover, Barone et al[8] showed that silymarin is able to lower IC development in ApcMin/+ mice and this process is associated with an increased crypt-villous epithelial cell migration and apoptosis. In this study, western blotting analysis demonstrated that silymarin is able to increase beta-ER/alpha ER ratio. Moreover, we have previously observed that beta ERs showed a progressive decline in the progressive steps of intestinal carcinogenesis, i.e., normal tissue, LGD, HGD and IC in subjects affected by familial adenomatous polyposis[30]. Interestingly, in the same study we demonstrated that beta-ERs and caspase 3, an early marker of apoptosis, were co-expressed in the same cell and this co-expression declined progressively from normal to neoplastic tissue. In the present study, we found a poor expression of ER beta in polypoid tissue and the presence of high grade dysplasia and carcinoma at this sites may explain this result, since ER beta is poorly expressed in high grade dysplasia and carcinoma areas[30]. However, in macroscopically normal mucosa, ER beta protein was well expressed in normal mucosa of enriched and standard diet with a more marked signal in the first group. Therefore, a stimulation of ER beta by silymarin could have occurred at this level and played a role in anti-neoplastic effect of dietary treatment.
Analogously, boswellic acid has shown interesting properties for the prevention of intestinal tumorigenesis in ApcMin/+ mice[11-13]. Its chemopreventive effect was attributed to a collection of activities including antiproliferation and apoptosis induction[12]. In detail, a decrease of proliferative (cyclin D1) and an increase of apoptotic markers (survivin and Bcl-xL) have been described as evidence that anticancer effects of boswellic acid[31].
Curcumin is able to modulate several molecular targets (transcription factors, enzymes, cell cycle proteins, cytokines, receptors and adhesion molecules) which are involved in carcinogenetic progression[14-16]. A recent meta-analysis by Tabrizi et al[32] suggests that taking curcumin-containing supplements may exert antioxidant properties. In detail, Shang et al[33] demonstrated that curcumin inhibited CRC cell proliferation and promoted apoptosis by down-regulating DJ-1 to regulate the activity of PTEN/PI3K/AKT pathway.
In the present study we found by western blotting that the dietetic formulation containg silymarin, boswellic acid and curcumin showed some mechanistic effects, which could explain their chemopreventive effect. Thoroughly, ER beta protein was well expressed in normal mucosa of enriched and standard diet, but this trend was more marked in animals assuming enriched diet. On the other hand, ER alpha was similarly expressed in normal and polypoid mucosa of standard and enriched diet group. The protein of cleaved form of caspase 3, moreover, showed in normal mucosa a stronger signal in enriched than in standard diet. Additionally, cyclin D1 was more expressed in standard than enriched diet group of polypoid tissue. In summary, we demonstrated that the dietetic formulation exerts a simultaneous anti-proliferative and pro-apoptotic effect. Moreover, the induction of ER beta increase may explain the silymarin main contribution to the process.
Another result of the present study is related to the effect of enriched diet on epithelial turnover. As expected, in both groups assuming control and supplemented diet, we found positive cells mainly located in the crypts of normal mucosa 24 h after BrdU injection. The step forward of positive cell migration to the villi was seen after 48 h. At this point, the highest labeled cell along the villus as marker of the percentage of covered axis was located much higher in the enriched than in the standard diet group. The mean timing of epithelial cell renewal in the small bowel has been assessed to be about 3 d[34-36]. This information clearly explains why the analysis at 48th h after BrdU injection was the most representative of cellular turnover state in our study. In detail, small bowel epitelial renewal process includes stem cell proliferation in the crypts and following migration towards the tip of the villi with a simultaneous differentiation and the final step of programmed death[37]. Therefore, our results suggest that epithelial turnover is faster in the enriched compared to the standard diet group. Consequently, enriched diet-related reduction of cellular half-life may decrease the time of exposure of proliferating cells to DNA mutations due to both intrinsic and extrinsic factors[38]. Finally, the enhancement of epithelial renewal induced by dietary supplementation agrees with the increase of apoptosis found in the same group of animals.
The results of the present study suggest an efficient synergic effect of combined diet based on substances of plant origin for the chemoprevention of intestinal genetic carcinogenesis. The synergic effect of our dietetic formulation is suggested even by the dosage used for the individual nutritional components, which turns out to be lower than that used for the single substances for “in vivo” experiments on the same animal model[13,39,40].
Some natural products derived from plants may have anti-carcinogenetic effect.
We looked for possible candidate molecules to arrest the development of intestinal cancer in an animal model.
To test a combination of phytochemicals in a mouse model of genetic intestinal carcinogenesis.
A combination of silymarin, boswellic acid and curcumin was given to forty adenomatous polyposis coli multiple intestinal neoplasia. Markers of proliferation/apoptosis were examined.
Compared to standard, enriched diet reduced the total number of polypoid lesions. In enriched diet group a reduction in polyp size was observed. Areas of low-grade dysplasia and intestinal carcinoma were significantly decreased in enriched diet group. Enriched diet showed a faster epithelial migration and an increased apoptosis in normal mucosa and low-grade dysplasia areas. Estrogen receptor beta protein was well expressed in normal mucosa of enriched and standard groups, with a more marked trend associated to the first one. Estrogen receptor alpha was similarly expressed in normal and polypoid mucosa of standard and enriched diet group. Cleaved caspase 3 showed in normal mucosa a stronger signal in enriched than in standard diet. Cyclin D1 was more expressed in standard than enriched diet group of both normal and polypoid tissue.
Our results are suggestive of a chemo-preventive synergic effect of silymarin, boswellic acid and curcumin in inherited intestinal cancer.
The dietetic formulation may be promising for patients with a genetic predisposition to develop intestinal polyps and carcinomas.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country/Territory of origin: Italy
Peer-review report’s scientific quality classification
Grade A (Excellent): 0
Grade B (Very good): B
Grade C (Good): C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Eren T, Patial V S-Editor: Dou Y L-Editor: A E-Editor: Zhang YL
| 1. | Brenner H, Kloor M, Pox CP. Colorectal cancer. Lancet. 2014;383:1490-1502. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1965] [Cited by in RCA: 2289] [Article Influence: 208.1] [Reference Citation Analysis (1)] |
| 2. | Leslie A, Carey FA, Pratt NR, Steele RJ. The colorectal adenoma-carcinoma sequence. Br J Surg. 2002;89:845-860. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 439] [Cited by in RCA: 462] [Article Influence: 20.1] [Reference Citation Analysis (0)] |
| 3. | Markowitz SD, Bertagnolli MM. Molecular origins of cancer: Molecular basis of colorectal cancer. N Engl J Med. 2009;361:2449-2460. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1274] [Cited by in RCA: 1392] [Article Influence: 87.0] [Reference Citation Analysis (2)] |
| 4. | Chen TH, Chang SW, Huang CC, Wang KL, Yeh KT, Liu CN, Lee H, Lin CC, Cheng YW. The prognostic significance of APC gene mutation and miR-21 expression in advanced-stage colorectal cancer. Colorectal Dis. 2013;15:1367-1374. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 46] [Cited by in RCA: 55] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 5. | Dickinson BT, Kisiel J, Ahlquist DA, Grady WM. Molecular markers for colorectal cancer screening. Gut. 2015;64:1485-1494. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 91] [Article Influence: 9.1] [Reference Citation Analysis (0)] |
| 6. | Di Leo A, Barone M, Maiorano E, Tanzi S, Piscitelli D, Marangi S, Lofano K, Ierardi E, Principi M, Francavilla A. ER-beta expression in large bowel adenomas: implications in colon carcinogenesis. Dig Liver Dis. 2008;40:260-266. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 40] [Article Influence: 2.4] [Reference Citation Analysis (0)] |
| 7. | Khorsandi L, Saki G, Bavarsad N, Mombeini M. Silymarin induces a multi-targeted cell death process in the human colon cancer cell line HT-29. Biomed Pharmacother. 2017;94:890-897. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 10] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 8. | Barone M, Tanzi S, Lofano K, Scavo MP, Pricci M, Demarinis L, Papagni S, Guido R, Maiorano E, Ingravallo G, Comelli MC, Francavilla A, Di Leo A. Dietary-induced ERbeta upregulation counteracts intestinal neoplasia development in intact male ApcMin/+ mice. Carcinogenesis. 2010;31:269-274. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 37] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 9. | Calabrese C, Rizzello F, Gionchetti P, Calafiore A, Pagano N, De Fazio L, Valerii MC, Cavazza E, Strillacci A, Comelli MC, Poggioli G, Campieri M, Spisni E. Can supplementation of phytoestrogens/insoluble fibers help the management of duodenal polyps in familial adenomatous polyposis? Carcinogenesis. 2016;37:600-606. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 6] [Cited by in RCA: 6] [Article Influence: 0.7] [Reference Citation Analysis (0)] |
| 10. | Bringiotti R, Ierardi E, De Tullio N, Fracella MR, Brindicci D, Marmo R, Albano F, Papagni S, Di Leo A, Principi M. Education and imaging. Gastroenterology: video capsule endoscopy disclosure of unprecedented therapeutic effect of Eviendep on small bowel polyposis in Lynch syndrome. J Gastroenterol Hepatol. 2015;30:801. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 5] [Cited by in RCA: 5] [Article Influence: 0.5] [Reference Citation Analysis (0)] |
| 11. | Pellegrini L, Milano E, Franceschi F, Belcaro G, Gizzi G, Feragalli B, Dugall M, Luzzi R, Togni S, Eggenhoffner R, Giacomelli L. Managing ulcerative colitis in remission phase: usefulness of Casperome®, an innovative lecithin-based delivery system of Boswellia serrata extract. Eur Rev Med Pharmacol Sci. 2016;20:2695-2700. [PubMed] |
| 12. | Liu HP, Gao ZH, Cui SX, Wang Y, Li BY, Lou HX, Qu XJ. Chemoprevention of intestinal adenomatous polyposis by acetyl-11-keto-beta-boswellic acid in APC(Min/+) mice. Int J Cancer. 2013;132:2667-2681. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 33] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 13. | Wang R, Wang Y, Gao Z, Qu X. The comparative study of acetyl-11-keto-beta-boswellic acid (AKBA) and aspirin in the prevention of intestinal adenomatous polyposis in APC(Min/+) mice. Drug Discov Ther. 2014;8:25-32. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 26] [Cited by in RCA: 28] [Article Influence: 2.5] [Reference Citation Analysis (0)] |
| 14. | Shishodia S, Sethi G, Aggarwal BB. Curcumin: getting back to the roots. Ann N Y Acad Sci. 2005;1056:206-217. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 448] [Cited by in RCA: 443] [Article Influence: 22.2] [Reference Citation Analysis (0)] |
| 15. | Imran M, Ullah A, Saeed F, Nadeem M, Arshad MU, Suleria HAR. Cucurmin, anticancer, & antitumor perspectives: A comprehensive review. Crit Rev Food Sci Nutr. 2018;58:1271-1293. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 63] [Article Influence: 7.9] [Reference Citation Analysis (0)] |
| 16. | Cruz-Correa M, Hylind LM, Marrero JH, Zahurak ML, Murray-Stewart T, Casero RA, Montgomery EA, Iacobuzio-Donahue C, Brosens LA, Offerhaus GJ, Umar A, Rodriguez LM, Giardiello FM. Efficacy and Safety of Curcumin in Treatment of Intestinal Adenomas in Patients With Familial Adenomatous Polyposis. Gastroenterology. 2018;155:668-673. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 75] [Cited by in RCA: 88] [Article Influence: 12.6] [Reference Citation Analysis (0)] |
| 17. | Pesakhov S, Khanin M, Studzinski GP, Danilenko M. Distinct combinatorial effects of the plant polyphenols curcumin, carnosic acid, and silibinin on proliferation and apoptosis in acute myeloid leukemia cells. Nutr Cancer. 2010;62:811-824. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 63] [Cited by in RCA: 64] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 18. | Cheung KL, Khor TO, Kong AN. Synergistic effect of combination of phenethyl isothiocyanate and sulforaphane or curcumin and sulforaphane in the inhibition of inflammation. Pharm Res. 2009;26:224-231. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 72] [Cited by in RCA: 75] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 19. | Girardi B, Principi M, Pricci M, Giorgio F, Iannone A, Losurdo G, Ierardi E, Di Leo A, Barone M. Chemoprevention of inflammation-related colorectal cancer by silymarin-, acetyl-11-keto-beta-boswellic acid-, curcumin- and maltodextrin-enriched dietetic formulation in animal model. Carcinogenesis. 2018;39:1274-1282. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 23] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 20. | Kidd PM. Bioavailability and activity of phytosome complexes from botanical polyphenols: the silymarin, curcumin, green tea, and grape seed extracts. Altern Med Rev. 2009;14:226-246. [PubMed] |
| 21. | National Research Council (US) Committee for the Update of the Guide for the Care and Use of Laboratory Animals. Guide for the Care and Use of Laboratory Animals. 8th edition. Washington (DC): National Academies Press (US); 2011. [PubMed] |
| 22. | Associazione Italiana per le Scienze degli Animali da Laboratorio. Dati Sulla Sperimentazione. [accessed 2020 Jan 3] Available from: https://www.aisal.org/normative-dati-risorse/dati-sulla-sperimentazione/. |
| 23. | Yu C, Zhang S, Song L, Wang Y, Hwaiz R, Luo L, Thorlacius H. Rac1 signaling regulates neutrophil-dependent tissue damage in experimental colitis. Eur J Pharmacol. 2014;741:90-96. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9] [Cited by in RCA: 9] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 24. | Javid SH, Moran AE, Carothers AM, Redston M, Bertagnolli MM. Modulation of tumor formation and intestinal cell migration by estrogens in the Apc(Min/+) mouse model of colorectal cancer. Carcinogenesis. 2005;26:587-595. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 51] [Cited by in RCA: 56] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 25. | Su LK, Kinzler KW, Vogelstein B, Preisinger AC, Moser AR, Luongo C, Gould KA, Dove WF. Multiple intestinal neoplasia caused by a mutation in the murine homolog of the APC gene. Science. 1992;256:668-670. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1121] [Cited by in RCA: 1131] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 26. | Nalbantoglu I, Blanc V, Davidson NO. Characterization of Colorectal Cancer Development in Apc (min/+) Mice. Methods Mol Biol. 2016;1422:309-327. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 27. | Seidlová-Wuttke D, Becker T, Christoffel V, Jarry H, Wuttke W. Silymarin is a selective estrogen receptor beta (ERbeta) agonist and has estrogenic effects in the metaphysis of the femur but no or antiestrogenic effects in the uterus of ovariectomized (ovx) rats. J Steroid Biochem Mol Biol. 2003;86:179-188. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 28. | Principi M, Barone M, Pricci M, De Tullio N, Losurdo G, Ierardi E, Di Leo A. Ulcerative colitis: from inflammation to cancer. Do estrogen receptors have a role? World J Gastroenterol. 2014;20:11496-11504. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 34] [Article Influence: 3.1] [Reference Citation Analysis (0)] |
| 29. | Calabrese C, Praticò C, Calafiore A, Coscia M, Gentilini L, Poggioli G, Gionchetti P, Campieri M, Rizzello F. Eviendep® reduces number and size of duodenal polyps in familial adenomatous polyposis patients with ileal pouch-anal anastomosis. World J Gastroenterol. 2013;19:5671-5677. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 15] [Cited by in RCA: 20] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 30. | Di Leo A, Nesi G, Principi M, Piscitelli D, Girardi B, Pricci M, Losurdo G, Iannone A, Ierardi E, Tonelli F. Epithelial turnover in duodenal familial adenomatous polyposis: A possible role for estrogen receptors? World J Gastroenterol. 2016;22:3202-3211. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 8] [Cited by in RCA: 7] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | Takahashi M, Sung B, Shen Y, Hur K, Link A, Boland CR, Aggarwal BB, Goel A. Boswellic acid exerts antitumor effects in colorectal cancer cells by modulating expression of the let-7 and miR-200 microRNA family. Carcinogenesis. 2012;33:2441-2449. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 98] [Cited by in RCA: 86] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 32. | Tabrizi R, Vakili S, Akbari M, Mirhosseini N, Lankarani KB, Rahimi M, Mobini M, Jafarnejad S, Vahedpoor Z, Asemi Z. The effects of curcumin-containing supplements on biomarkers of inflammation and oxidative stress: A systematic review and meta-analysis of randomized controlled trials. Phytother Res. 2019;33:253-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 109] [Cited by in RCA: 90] [Article Influence: 15.0] [Reference Citation Analysis (0)] |
| 33. | Shang H, Wang T, Shang F, Li M, Luo Y, Huang KM. Over-expression of DJ-1 attenuates effects of curcumin on colorectal cancer cell proliferation and apoptosis. Eur Rev Med Pharmacol Sci. 2019;23:3080-3087. [RCA] [PubMed] [DOI] [Full Text] [Cited by in RCA: 4] [Reference Citation Analysis (0)] |
| 34. | Bertalanffy FD, Nagy KP. Mitotic activity and renewal rate of the epithelial cells of human duodenum. Acta Anat (Basel). 1961;45:362-370. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 78] [Article Influence: 2.9] [Reference Citation Analysis (0)] |
| 35. | Lipkin M, Bell B, Sherlock P. CELL PROLIFERATION KINETICS IN THE GASTROINTESTINAL TRACT OF MAN. I. CELL RENEWAL IN COLON AND RECTUM. J Clin Invest. 1963;42:767-776. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 121] [Article Influence: 6.4] [Reference Citation Analysis (0)] |
| 36. | Gehart H, Clevers H. Tales from the crypt: new insights into intestinal stem cells. Nat Rev Gastroenterol Hepatol. 2019;16:19-34. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 393] [Cited by in RCA: 649] [Article Influence: 108.2] [Reference Citation Analysis (0)] |
| 37. | Principi M, Di Leo A, Pricci M, Scavo MP, Guido R, Tanzi S, Piscitelli D, Pisani A, Ierardi E, Comelli MC, Barone M. Phytoestrogens/insoluble fibers and colonic estrogen receptor β: randomized, double-blind, placebo-controlled study. World J Gastroenterol. 2013;19:4325-4333. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 29] [Cited by in RCA: 31] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 38. | Parris A, Williams MR. A human colonic crypt culture system to study regulation of stem cell-driven tissue renewal and physiological function. Methods Mol Biol. 2015;1212:141-161. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 8] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 39. | Rajamanickam S, Velmurugan B, Kaur M, Singh RP, Agarwal R. Chemoprevention of intestinal tumorigenesis in APCmin/+ mice by silibinin. Cancer Res. 2010;70:2368-2378. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 80] [Cited by in RCA: 76] [Article Influence: 5.1] [Reference Citation Analysis (0)] |
| 40. | Perkins S, Verschoyle RD, Hill K, Parveen I, Threadgill MD, Sharma RA, Williams ML, Steward WP, Gescher AJ. Chemopreventive efficacy and pharmacokinetics of curcumin in the min/+ mouse, a model of familial adenomatous polyposis. Cancer Epidemiol Biomarkers Prev. 2002;11:535-540. [PubMed] |