Published online Jun 7, 2019. doi: 10.3748/wjg.v25.i21.2603
Peer-review started: March 3, 2019
First decision: March 20, 2019
Revised: March 27, 2019
Accepted: April 19, 2019
Article in press: April 20, 2019
Published online: June 7, 2019
Processing time: 96 Days and 19.8 Hours
Given the complex pathogenesis of ulcerative colitis (UC), the conventional therapeutic methods are not fully curative. As a sort of systematic complementary and alternative medicine, traditional Chinese medicine (TCM) provides new options for the standard therapy. Nevertheless, there are still numerous problems with the promotion of TCM attributed to its complexity, and consequently, new research approaches are urgently needed. Thus, we explored the protective effects of Jian-Pi Qing-Chang (JPQC) decoction on UC based on systems pharmacology approach, which might fill the current innovation gap in drug discovery and clinical practice pertaining to TCM.
To investigate the protective mechanisms of JPQC decoction on UC based on systems pharmacology approach.
We performed systems pharmacology to predict the active ingredients, the matched targets, and the potential pharmacological mechanism of JPQC on UC. In vivo, we explored the effects of JPQC in a colitis model induced by dextran sulfate sodium. In vitro, we adopted the bone marrow-derived macrophages (BMDMs) as well as BMDMs co-cultured with Caco2 cells to verify the underlying mechanisms and effects of JPQC on UC under TNF-α stimulation.
Systems pharmacology revealed 170 targets for the 107 active ingredients of JPQC and 112 candidate targets of UC. Protein-protein interaction networks were established to identify the underlying therapeutic targets of JPQC on UC. Based on enrichment analyses, we proposed our hypothesis that JPQC might have a protective effect on UC via the NF-κB/HIF-1α signalling pathway. Subsequent experimental validation revealed that treatment with TNFα activated the NF-κB/HIF-1α signalling pathway in BMDMs, thereby damaging the epithelial barrier permeability in co-cultured Caco2 cells, while JPQC rescued this situation. The findings were also confirmed in a dextran sulfate sodium-induced colitis model.
JPQC could improve the mucosal inflammatory response and intestinal epithelial barrier function via the NF-κB/HIF-1α signalling pathway, which provides new perspectives on the pharmaceutical development and clinical practice of TCM.
Core tip: Given the complex pathogenesis of ulcerative colitis (UC), the conventional therapeutic methods are not fully curative. As a sort of systematic complementary and alternative medicine, traditional Chinese medicine (TCM) provides new options for the standard therapy. Nevertheless, there are still numerous problems with the promotion of TCM attributed to its complexity, and consequently, new research approaches are urgently needed. Thus, we explored the protective effects of Jian-Pi Qing-Chang decoction on UC based on systems pharmacology approach, which might fill the current innovation gap in drug discovery and clinical practice pertaining to TCM.
- Citation: Chen YL, Zheng YY, Dai YC, Zhang YL, Tang ZP. Systems pharmacology approach reveals protective mechanisms of Jian-Pi Qing-Chang decoction on ulcerative colitis. World J Gastroenterol 2019; 25(21): 2603-2622
- URL: https://www.wjgnet.com/1007-9327/full/v25/i21/2603.htm
- DOI: https://dx.doi.org/10.3748/wjg.v25.i21.2603
Ulcerative colitis (UC) is a common chronic disease of the gastrointestinal tract that often presents with diarrhoea, bloody purulent stools, fatigue, and unexplained weight loss[1]. As the vital pathogenic factor of UC, intestinal mucosal inflammation mediates impairment of epithelial barrier function, which is associated with a range of disease states, both intestinal and extra-intestinal disorders[2,3]. Given its persistent and recurrent nature, UC may also present patients with enormous psychological disturbance and financial burden, which poses a tremendous challenge for public health worldwide[4].
Mounting evidence highlights inflammation as the initiating factor in the development of UC, which can lead to a series of inflammatory cascades, thus causing profound changes in the intestinal microenvironment[5,6]. Such imbalanced intestinal epithelial homeostasis during mucosal inflammation can damage intercellular adhesions in a direct or indirect manner, which may result in increased barrier permeability, ultimately contributing to disordered systemic metabolism[6-8]. In addition, various studies in different fields, such as irritable bowel syndrome, obesity, and metabolic diseases[9-11], have set their sights on intestinal homeostasis; however, the underlying mechanism appears to be more complex than formerly thought.
Considering the routine therapeutic tactics for UC including aminosalicylic acid, steroids, and biological agents are not fully curative, treatment guidelines propose the recommendation of multimodality therapy, which is an emerging concept for most chronic inflammatory diseases[12,13]. As a sort of systematic complementary and alternative medicine, traditional Chinese medicine (TCM), which provides new options for the standard therapy, has attracted widespread attention and has gradually been accepted by an increasing number of people throughout the world[14]. Nevertheless, there are still numerous problems with the promotion of TCM attributed to its complexity, and consequently, new research approaches are urgently needed.
Systems pharmacology, an emerging systems-oriented methodology, which provides a promising avenue for holistically understanding mechanisms of drug actions by means of active compound screening, target matching, and network construction and analysis, may fill the current innovation gap in drug discovery and clinical practice of TCM[15,16]. In recent years, series of investigations of systems pharmacology on TCM have been delivered; however, studies on compound prescription are still rare[17]. Here, a preliminary exploration was conducted via systems pharmacology analysis of Jian-Pi Qing-Chang (JPQC) decoction, which has been demonstrated to be effective in previous clinical and experimental studies[18-20], and experimental validations were conducted subsequently.
Chemical compounds information was retrieved from the Traditional Chinese Medicine System Pharmacology (TCMSP) database for all the nine medicinal herbs. Compounds with oral bioavailability (OB) ≥ 20 % and drug-likeness (DL) ≥ 0.18 were obtained, and others with lower OB or DL values were manually selected owing to their extensive biological and pharmacological activities[21,22]. The Genetic Association Database (GAD), Therapeutic Target Database (TTD), and Online Mendelian Inheritance in Man database (OMIM) were screened to collect the known UC-related targets. We constituted protein-protein interaction (PPI) networks for candidate targets of JPQC and UC, respectively, and an intersection was performed to identify the putative targets of JPQC against UC. We applied the Cytoscape plugin, CytoNCA, and ClueGO, to calculate the degree centrality (DC) values as well as to perform the enrichment analyses, respectively[23,24].
For this study, we used age-matched (8 wk) male C57BL/6 mice purchased from SLAC Animal Laboratories. The mice were randomly divided into three groups (n = 10 in each group). Colitis was constructed by administration of 3.5 % dextran sulfate sodium (DSS), while the control group received water only. Subsequently, normal saline or JPQC (15 g/kg/d) was given intra-gastrically daily for the control/DSS groups and JPQC group, respectively. The disease activity index (DAI) scores used in this study to calculate the severity of colitis are described in Supplementary Table S5[25]. By the end of the trial, serum and colon samples were collected for further detection.
The animal experiment was conducted in accordance with the Laboratory Animal Regulations of the Institutional Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine, and was approved by the Institutional Animal Care and Use Committee of Shanghai University of Traditional Chinese Medicine.
For preparation of bone marrow-derived macrophages (BMDMs), the femur and tibia bones were isolated from 6-8 wk mice and bone marrow was flushed out into cold PBS + 2 % FBS with a 1 mL syringe. Next, the cells were filtrated through a 70 μm cell strainer, and three volumes of Red Blood Cell Lysis Buffer was added. After incubation on ice for 10 min, the cell pellet was resuspended and centrifuged at 500 g for 5 min twice. Finally, the isolated bone marrow cells were resuspended in IMDM containing 10 ng/mL M-CSF (2 × 105 cells/mL)[26]. Seeded cells in 6-well plates and changed fresh BMDM growth medium on day 3. To stimulate mature BMDMs, cells were incubated with IMDM containing TNF-α (20 ng/mL) alone or together with JPQC (100 μg/mL) or rapamycin (RAP) (2 μM) for 24 h, or pre-treated with pyrrolidinedithiocarbamate (PDTC; 50 μM) for 30 min.
For the co-culture assay, Caco2 cells were obtained from Shanghai Academy of Life Sciences. BMDMs were co-cultured in the upper chamber of a trans-well plate, with Caco2 cells placed in the lower chamber on day 5. Mature BMDMs were incubated with IMDM (the BMDM growth medium) containing TNF-α (20 ng/mL) alone or together with JPQC (100 μg/mL) or RAP (2 μM) for 24 h, or pre-treated with PDTC (50 μM) for 30 min on day 7.
We extracted total RNA according to protocols with TRIzol (Invitrogen, United States) and RT-PCR was conducted based on the Eppendorf PCR system. Actin was used to normalize the samples. Primer sequences are presented in Supplementary Table S6.
Primary antibodies against NF-κB, HIF-1α, p-mTOR (phospho S2448), TRAF2, p-IκB (Ser32), IκB, p-Akt (phospho S473), Akt, ZO-1, and Actin were obtained from CST, and antibodies against mTOR, p-PI3K (phospho Y607), and Claudin-1 were obtained from Abcam. The groupings were cut off from different blots under the same conditions and each protein band was extracted from a continuous part.
BMDMs and Caco2 cells were incubated with anti-HIF-1α (1:500), anti-NF-κB (1:500), anti-Tublin (1:1000), and anti-Claudin-1 (1:500). Secondary antibodies conjugated with 488/594 (1:1000) were used in this study. Hoechst (1:1000) was used to detect nuclei. Colon sections were incubated with anti-HIF-1α (1:100) and anti-Claudin-1 (1:100). Secondary antibodies conjugated with 488 (1:500) were used in this study. DAPI (1:500) was used to detect nuclei.
Colon samples were processed and stained with haematoxylin and eosin as well as immunohistochemical staining using anti-TNF-α as described previously[27].
Serum was detected through mouse-specific ELISA using anti-TNF-α and anti-IL-1β following the manufacturer’s protocols.
All data are expressed as the mean ± SD. Statistical differences between different groups were measured through one-way ANOVA, with P < 0.05 considered statistically significant. Data analyses were conducted using GraphPad Prism 7.0.
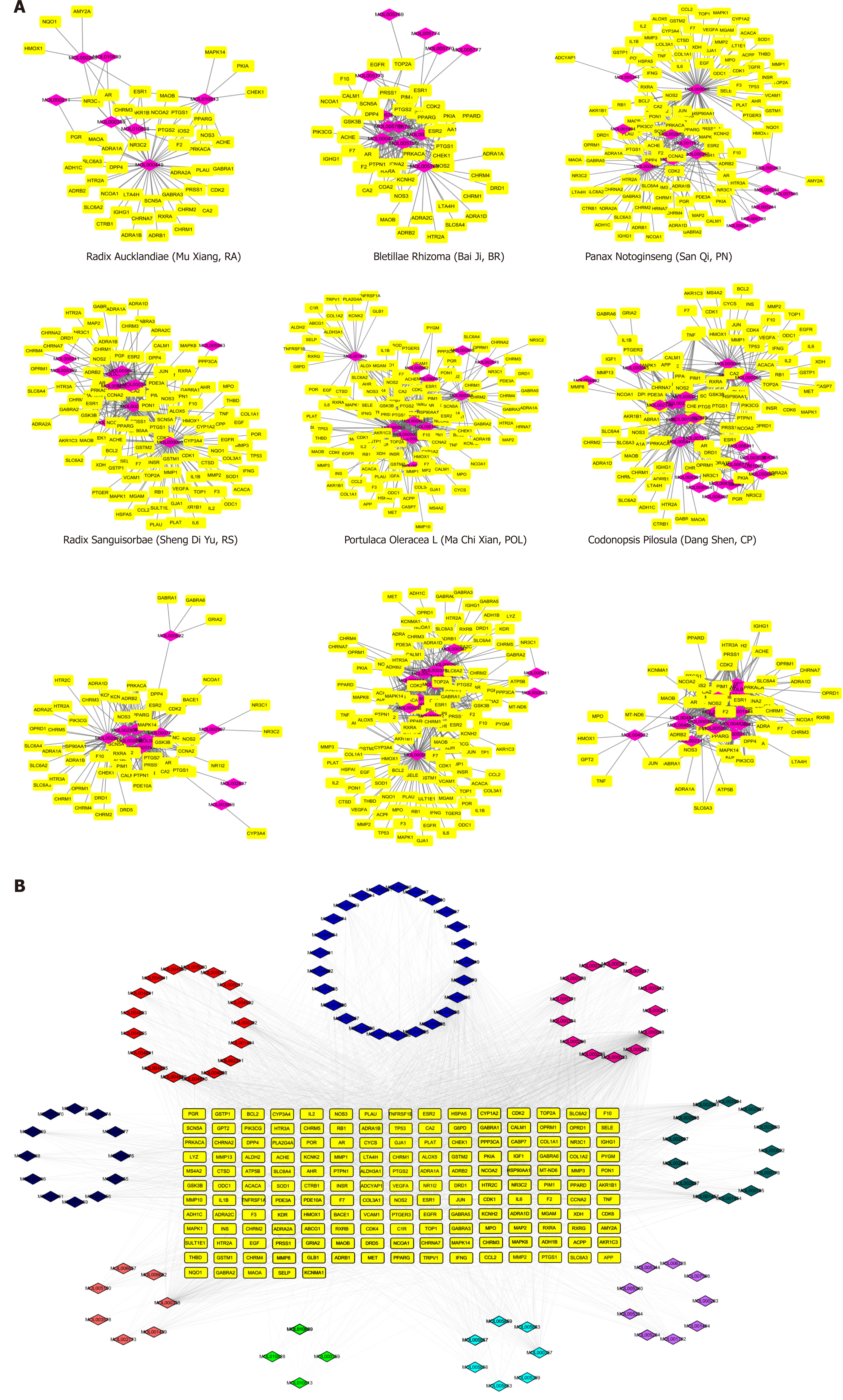
To holistically analyse the mechanisms of drug actions of JPQC, we retrieved the compounds information on the TCMSP database for all nine medicinal herbs, namely, Astragali mongholicus (AM), Codonopsis pilosula (CP), Portulaca oleracea L (POL), Radix sanguisorbae (RS), Panax notoginseng (PN), Bletillae rhizoma (BR), Radix aucklandiae (RA), Coptis chinensis (CC), and Radix glycyrrhizae (RG). As key indicators for selecting the drug-like compounds of bioactive molecules in traditional Chinese herbal drugs, OB and DL were used here to predict the active substances in JPQC[21,22]. A total of 101 potential compounds with OB ≥ 20 % and DL ≥ 0.18 were obtained from the herbal constituents of JPQC; moreover, six typical compounds with lower OB or DL values were manually selected owing to their extensive biological and pharmacological activities. Finally, 107 compounds were considered putative for further analyses, and the numbers of candidate compounds in AM, CP, POL, RS, PN, BR, RA, CC, and RG were 17, 27, 12, 15, 17, 14, 8, 17, and 17, respectively (Figure 1A and Supplementary Table S1). Among these, 37 compounds overlapped, indicating synergistic effects as a result of similar ingredients shared by the herbs.
Given that the integrated effects of TCM formulas rely on their complex compound-target interactions, we next predicted the therapeutic targets for all these 107 compounds[28]. In total, 170 potential targets were collected, and the numbers in AM, CP, POL, RS, PN, BR, RA, CC, and RG were 130, 101, 135, 114, 121, 47, 49, 62, and 61, respectively (Figure 1A and Supplementary Table S2 and Table S3), which also suggested synergistic effects by virtue of similar targets regulated by these different compounds. To further elucidate the complex interactions, we constructed a network consisting of 277 nodes and 2703 edges for all the 107 compounds and their corresponding targets (Figure 1B). The compound-target network revealed that most compounds exerted multi-targets effects, representing multifarious therapeutic effects.
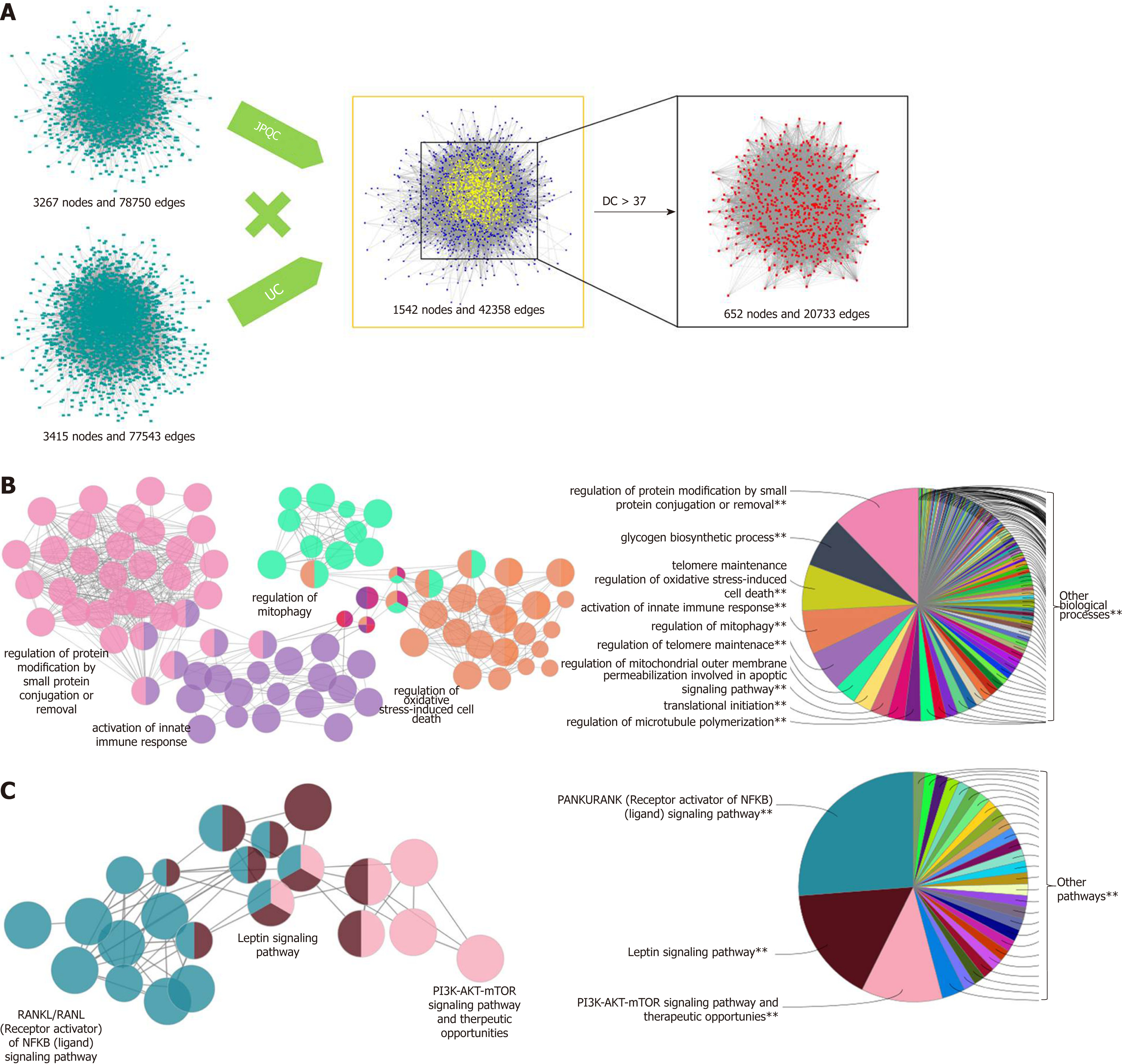
To explore the interpretation of the mechanisms of JPQC on UC, we screened the GAD, TTD, and OMIM to collect the known UC-related targets. One hundred and twelve candidate targets were harvested (Supplementary Table S4), and subsequently a PPI network for known UC-related targets (3415 nodes and 77543 edges) was constituted (Figure 2A). The PPI network was also constructed for the putative targets of JPQC (3267 nodes and 78750 edges); furthermore, an intersection of these two networks (1542 nodes and 42358 edges) was constructed to find the putative targets of JPQC on UC (Figure 2A). The DC value, an indicator representative of the correlation coefficient among targets, was calculated by CytoNCA in this article to filter the potential targets[23]. Finally, 652 putative targets with DC > 37 (the median value) were selected for the following analyses (Figure 2A).
In order to further comprehend the potential mechanisms of biological processes and signalling pathways of JPQC against UC, we performed ClueGO analyses for the targets using the Cytoscape plugin[24]. Specifically, the biological processes were largely related to regulation of protein modification by small protein conjugation or removal, glycogen biosynthetic process, telomere maintenance, regulation of oxidative stress-induced cell death, and activation of innate immune response (Figure 2B); whereas the signalling pathways mainly consisted of RANKL/RANK [Receptor activator of NF-κB (ligand)] signalling pathway, Leptin signalling pathway, and PI3K-AKT-mTOR signalling pathway (Figure 2C). Evidence shows that the NF-κB and PI3K-AKT-mTOR signalling pathways share the same downstream event of HIF-1α, while NF-κB and HIF-1α are both critically involved in the activation of inflammation as well as the deficiency of intestinal epithelial barrier function during the de-velopment of UC[29,30]. Herein, we proposed a hypothesis that JPQC inhibited the NF-κB/HIF-1α signalling pathway to protect against UC.
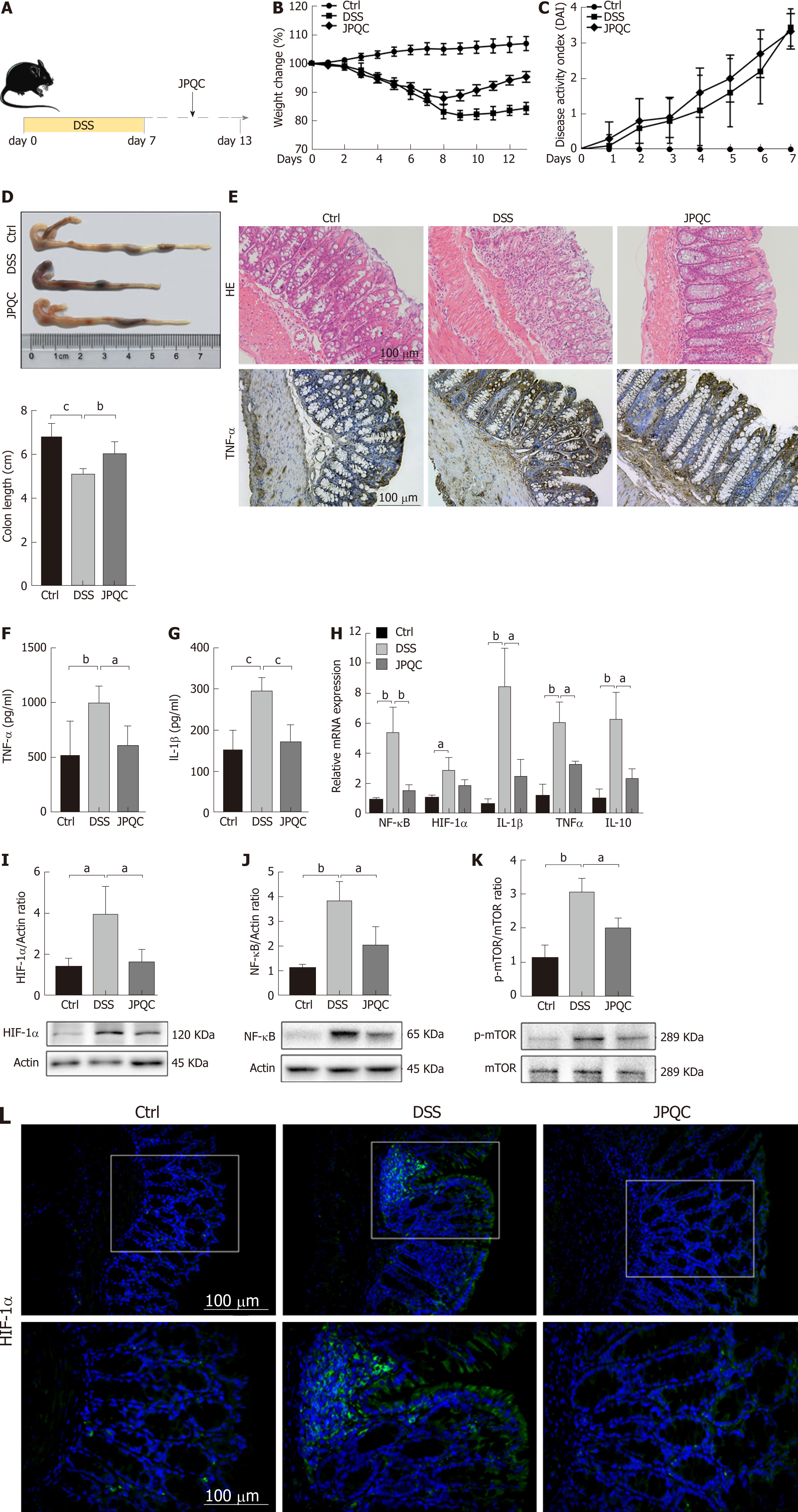
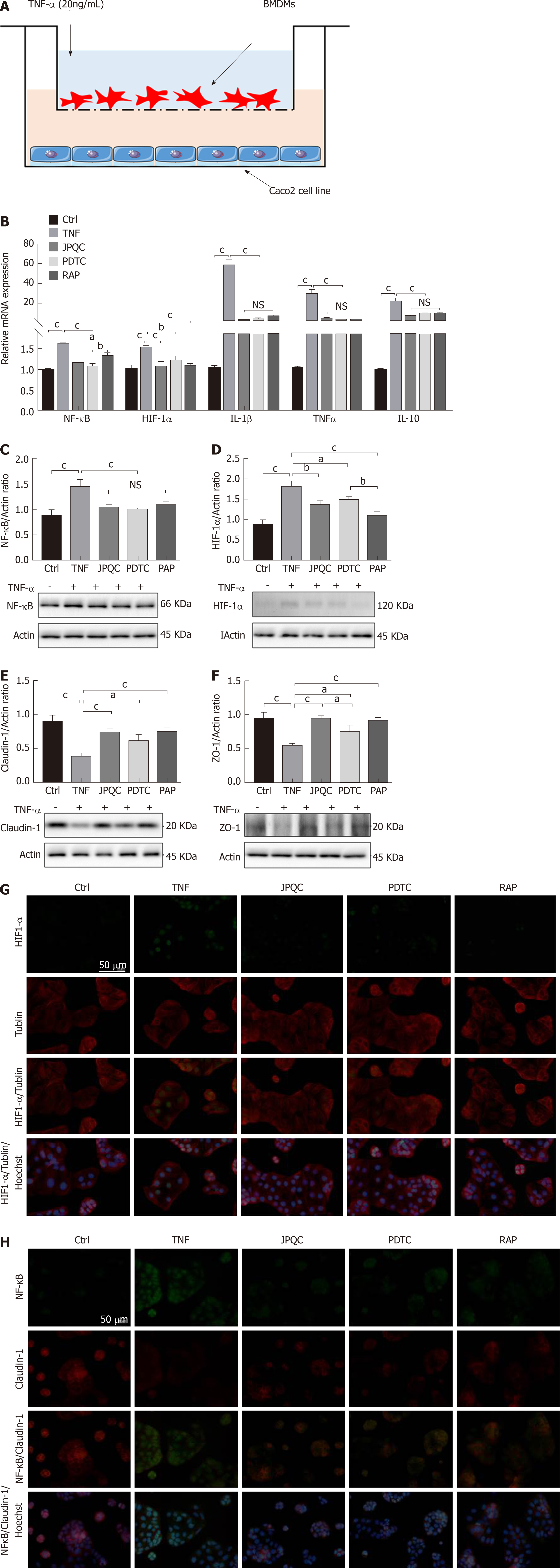
A murine model of colitis induced by DSS was used here to validate our previous presumption; the specific experiment flow is shown in Figure 3A. In general, DSS gave rise to considerable reductions in body weight and colon length as well as a dramatic increase in DAI scores, whereas JPQC provided protective therapeutic effects (Figure 3B-D). To evaluate the extent of the intestinal inflammatory response, HE staining was performed here and the result revealed leukocyte infiltration, crypt loss, and muscularis thickening in the colons under DSS stimulation (Figure 3E). Meanwhile, the expression levels of inflammatory factors, including TNF-α, IL-1β, and IL-10, were examined by ELISA and PCR analyses, and the outcomes also presented pro-inflammatory changes induced by DSS (Figure 3F-H). Fortunately, JPQC could distinctly improve all the DSS-induced inflammation, which was also confirmed by the immunohistochemical staining for TNF-α (Figure 3E).
Based on systems pharmacology analyses, we then examined the expression levels of HIF-1α, NF-κB, and mTOR. Western blot analyses revealed that the relative expression of HIF-1α/actin, NF-κB/actin, and p-mTOR/mTOR was all increased in the DSS group, while JPQC alleviated the DSS-induced elevations as expected (Figure 3I-K). PCR analyses of NF-κB and HIF-1α mRNA levels yielded similar results (Figure 3H), indicated that NF-κB, HIF-1α, and mTOR were all involved in the mecha-nisms by which JPQC ameliorated UC. As a common downstream factor, immunofluore-scence staining for HIF-1α was performed, and the result presented that JPQC could depress HIF-1α expression in DSS-induced colitis mice (Figure 3L).
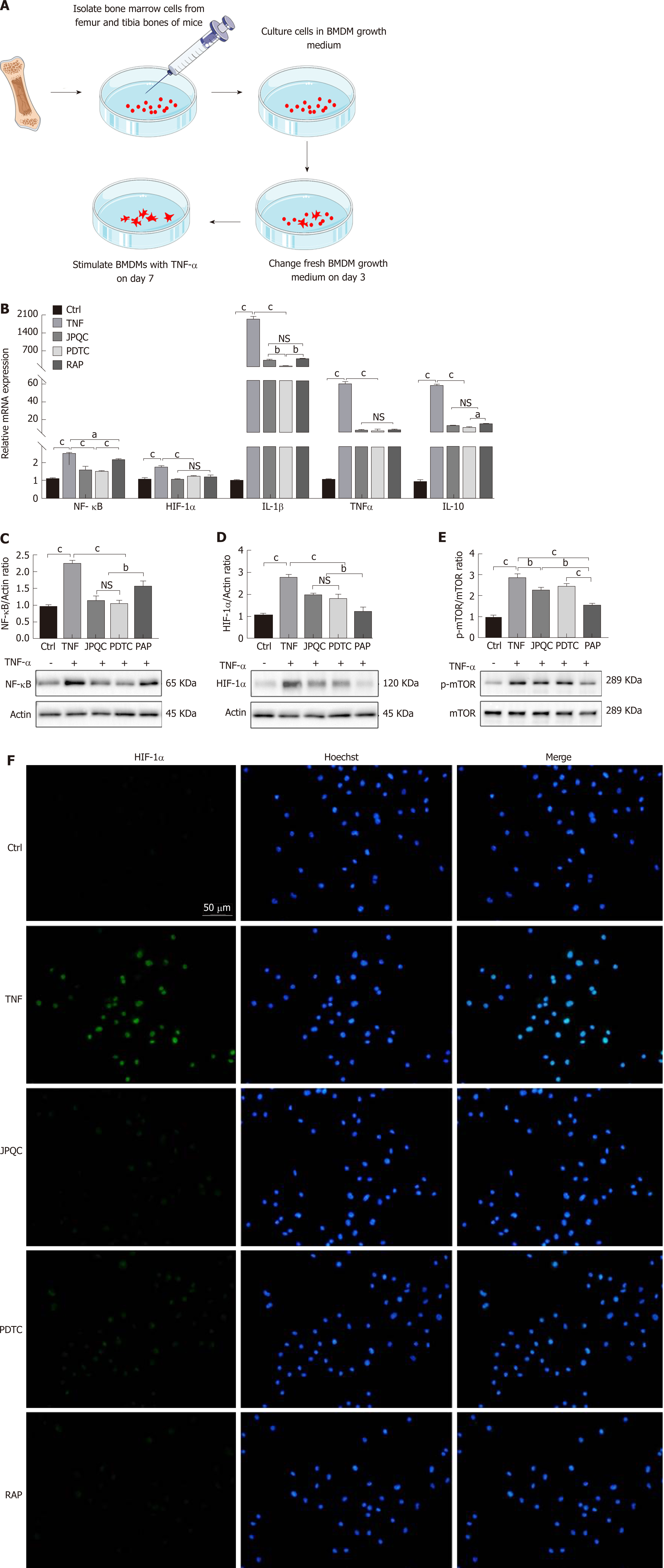
Next, BMDMs were extracted from wild-type C57BL/6 mice according to previously reported protocols[26]; the experimental method is illustrated in Figure 4A. Mature BMDMs were incubated with TNF-α (20 ng/mL) alone or together with JPQC (100 μg/mL), and the relative expression levels of NF-κB, HIF-1α, TNF-α, IL-1β, and IL-10 were measured by PCR analyses. The results showed that TNF-α stimulation raised the levels of all these indicators and JPQC mitigated the elevations as previously observed (Figure 4B). In addition, the ratios of HIF-1α/actin, NF-κB/actin, and p-mTOR/mTOR expression were also measured by Western blot analyses, and the results were corresponding to those in vivo (Figure 4C-E). These data indicated that JPQC restrained the HIF-1α signalling pathway in BMDMs, and the immu-nofluorescence staining for HIF-1α confirmed that finding (Figure 4F).
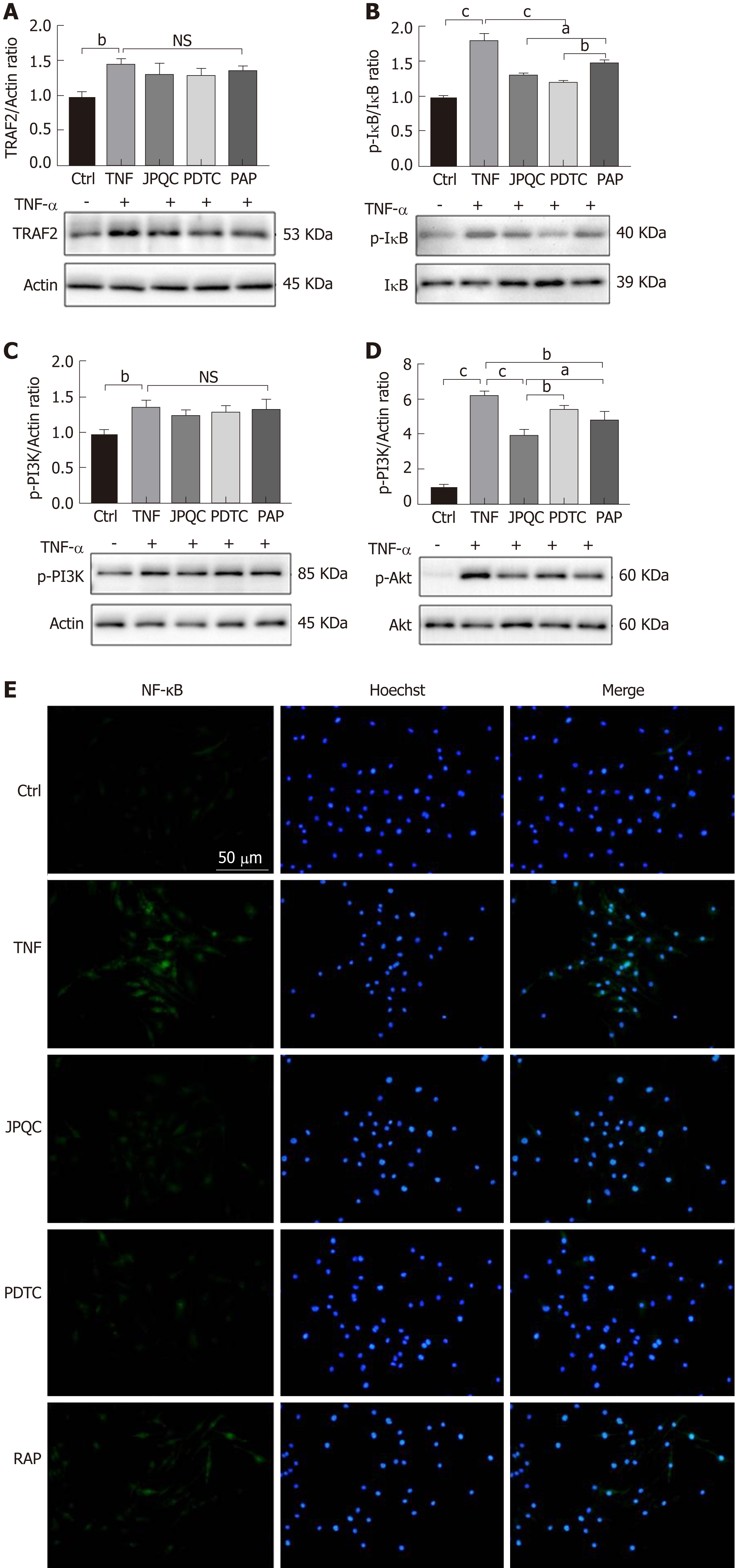
Here, PDTC and RAP, the inhibitors of NF-κB and mTOR, respectively, served as positive control agents, and both provided NF-κB and HIF-1α results similar to those of JPQC (Figure 4B-D and F); in contrast, PDTC had no influence on the phos-phorylation of mTOR (Figure 4E). As researches have demonstrated that the phos-phorylation of Akt can modulate the degradation of IκB, it appears that mTOR may play an important role in the process[31]. Thus, we further detected the upstream events of the NF-κB and mTOR signalling pathways mentioned previously. Interestingly, Western blot analyses presented similar results, showing that PDTC had no influence on Akt phosphorylation, whereas RAP significantly decreased IκB phosphorylation (Figure 5B and D). No changes in TRAF2 or p-PI3K expression were observed (Figure 5A and C). Immunofluorescence staining also revealed that JPQC and positive control agents offered therapeutic effects on NF-κB (Figure 5E), suggesting that JPQC could inhibit the NF-κB/HIF-1α signalling pathway in BMDMs.
Considering that epithelial barrier plays an important role in the pathogenesis of UC, Caco2 cells were co-cultured with BMDMs to evaluate the relevance between mucosal inflammation and intestinal barrier permeability in this study; the experimental procedure is shown in Figure 6A. We first examined the relevant inflammatory factors, and all the results of the relative mRNA expression levels of NF-κB, HIF-1α, TNF-α, IL-1β, and IL-10 as well as the ratios of NF-κB/actin and HIF-1α/actin expression displayed that JPQC could inhibit inflammation through the NF-κB/HIF-1α signalling pathway in Caco2 cells (Figure 6B-D). Next, we performed Western blot analyses for the tight junction protein. The results showed that the expression of Claudin-1 and ZO-1 was down-regulated in the TNF group and that JPQC improved this situation (Figure 6E and F), indicating that JPQC could regulate tight junctions in Caco2 cells. Double immunofluorescence staining for HIF-1α/Tublin and NF-κB/Claudin-1 confirmed the previous findings (Figure 6G and H). Taken together, JPQC could modulate tight junctions via the NF-κB/HIF-1α signalling pathway in Caco2 cells.
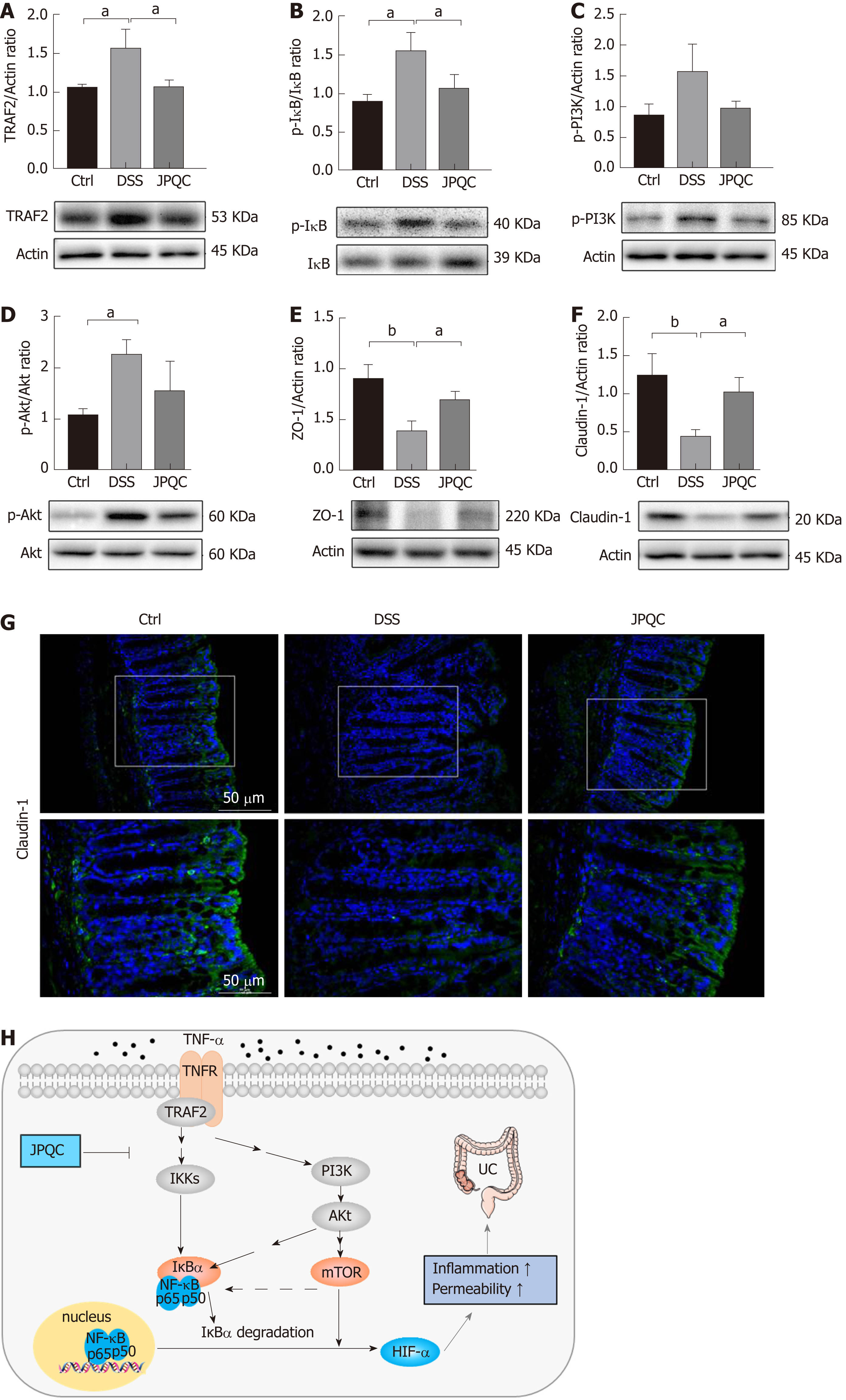
To verify the abovementioned findings, we examined the upstream events of the NF-κB/HIF-1α signalling pathway in a murine model. Western blot analyses showed that the ratios of TRAF2/actin and p-IκB/IκB expression were obviously reduced in the JPQC group compared with those in the DSS group, whereas the changes of p-PI3K/actin and p-Akt/Akt between the two groups were mild (Figure 7A-D). In the meantime, JPQC ameliorated the declined expression of Claudin-1 and ZO-1 induced by DSS (Figure 7E and F), suggesting that JPQC could improve intestinal epithelial barrier function in DSS-induced colitis mice, and the subsequent immunofluorescence staining for Claudin-1 further strengthened this finding (Figure 7G). Taken together, all these data suggested that JPQC could improve the mucosal inflammatory response and intestinal epithelial barrier function via the NF-κB/HIF-1α signalling pathway during the development of UC, and the specific pathway diagram is described in Figure H.
In the field of medical research, pharmaceutical development has consistently been an urgent challenge, especially for TCM attributed to the multi-target interactions of the complex ingredients of its herbal drugs[32]. One major dilemma is that the exact therapeutic tactics are hardly defined in conventional studies due to the mild curative effects for most individual targets. Fortunately, the synergistic effects exerted by multi-targets of many Chinese herbal compounds are prominent, which are responsible for the integral pharmacodynamics of TCM[33]. Thus, we tried to explore a promising approach to obtain a comprehensive and systematic cognization of drug action in the present study. By dint of systems pharmacology, we screened candidate compounds, matched putative targets, and constructed PPI networks to harvest the potential therapeutic targets of JPQC against UC, subsequently predicted the specific signalling pathway, and finally validated our hypothesis through experiments in vivo and in vitro.
First, we performed compound screening and target matching based on open-source initiatives and free web-based tools. Ultimately, 107 candidate compounds and 170 corresponding targets were obtained from JPQC, and then, PPI networks were constructed to identify the underlying therapeutic targets of JPQC on UC. ClueGO analyses revealed the RANKL/RANK signalling pathway and PI3K-AKT-mTOR signalling pathway for the potential therapeutic targets, both of which have been reported to be crucially involved in the inflammatory response. In our previous studies, JPQC has been proved protective for UC by inhibiting inflammation as well as by repairing intestinal epithelial barrier function, while HIF-1α, the common downstream event of the NF-κB and mTOR signalling pathways, was also demonstrated to be associated with those processes[19,20,29]. Hence, we proposed our hypothesis that JPQC might have a protective effect on UC via the NF-κB/HIF-1α signalling pathway.
HIF-1α, as a central link to regulate the expression of many transcription factors that impact inflammation, angiogenesis, and cancer, has been considered to mediate many diseases[34]. Evidence shows that the activation of the NF-κB and mTOR signalling pathways in inflammation can trigger the nuclear translocation and transcription of HIF-1α, leading to a series of inflammatory cascades[35,36]. Specifically, during the development of UC, the expression of HIF-1α can result in perpetual inflammation, causing deficiency of the intestinal epithelial barrier function[29,37].
First, we evaluated the curative effects of JPQC on DSS-induced colitis mice and confirmed that HIF-1α was involved in the process. Subsequently, considering the pivotal role of macrophages in intestinal inflammation, BMDMs were isolated from wild-type C57BL/6 mice to monitor the changes in the indicators of the NF-κB/HIF-1α signalling pathway in the inflammatory reactions induced by TNF-α and the pharmacological actions of JPQC. In addition, to better simulate the physiological environment in vivo, Caco2 cells were co-cultured with BMDMs to explore the protective effects of JPQC on intestinal epithelial mucosal barrier function. The results showed that treatment with TNFα activated the mTOR and NF-κB/HIF-1α signalling pathway in BMDMs, thereby damaging the epithelial barrier permeability in co-cultured Caco2 cells, while JPQC rescued this situation. Interestingly, we found that the indicators in the NF-κB signalling pathway were obviously restrained by RAP, whereas PDTC presented no similar influence on the indicators in the mTOR signalling pathway, suggesting that the phosphorylation of mTOR might regulate the NF-κB signalling pathway in some way, which has also been reported in latest investigations[38]. Furthermore, a murine model induced by DSS was used in this research to confirm our previous findings, and the results revealed that the mTOR and NF-κB/HIF-1α signalling pathways were highly involved in the therapeutic mechanism of JPQC against UC. Taken together, all the data in our present study demonstrated that JPQC could improve UC via the NF-κB/HIF-1α signalling pathway.
Limitations should be acknowledged that bioinformatics-based systems pharmacology is an analytical methodology based on the existing data network platform, where the bioinformatics is derived from massive existing studies. However, as an emerging research field, there are still many gaps in the current bioinformatics, which might bring about biases in our systematic pharmacological analyses. Beyond that, our research has proven the feasibility of systematic pharmacology in the pharmaceutical development of TCM. Hence, further interdisciplinary research and integrated analyses are necessary, and they may provide new perspectives on the development and clinical practice of TCM.
Ulcerative colitis (UC) is a kind of common chronic disease with complex etiology, and it is difficult to achieve a satisfactory curative effect by conventional drug treatments. Meanwhile, pharmaceutical development has always been an urgent dilemma in the field of medical research. Thus, promising and novel research methods are urgently needed to be explored for UC.
Mucosal inflammation and impairment of epithelial barrier function were thought to mediate the pathogenesis of UC. HIF-1α was also demonstrated to be associated with those processes. Thus, we explored the potential pharmacological mechanism of Jian-Pi Qing-Chang (JPQC) on UC, which might provide a novel complementary tactic to the standard treatment.
To explore the potential pharmacological mechanism of JPQC on UC based on systems pharmacology approach.
Systems pharmacology was used in this research to explore the potential pharmacological mechanism of JPQC on UC, and cell co-culture assay was applied to better simulate the physiological environment in vivo.
The study showed the protective effect of JPQC on UC through cell and animal experiments.
JPQC could improve the mucosal inflammatory response and intestinal epithelial barrier function via the NF-κB/HIF-1α signalling pathway.
The feasibility of systematic pharmacology was proved in this research, and further interdisciplinary research and integrated analyses are necessary in the pharmaceutical development of traditional Chinese medicine (TCM), which might provide new perspectives on the development and clinical practice of TCM.
Manuscript source: Unsolicited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: China
Peer-review report classification
Grade A (Excellent): A
Grade B (Very good): B, B
Grade C (Good): C, C
Grade D (Fair): 0
Grade E (Poor): 0
P-Reviewer: Can G, Matowicka-Karna J, M’Koma AE, Sulkowski S, Vradelis S S-Editor: Yan JP L-Editor: Wang TQ E-Editor: Ma YJ
| 1. | Ungaro R, Mehandru S, Allen PB, Peyrin-Biroulet L, Colombel JF. Ulcerative colitis. Lancet. 2017;389:1756-1770. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 2199] [Cited by in RCA: 2483] [Article Influence: 310.4] [Reference Citation Analysis (2)] |
| 2. | Brandl K, Schnabl B. Is intestinal inflammation linking dysbiosis to gut barrier dysfunction during liver disease? Expert Rev Gastroenterol Hepatol. 2015;9:1069-1076. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 44] [Cited by in RCA: 61] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 3. | Winer DA, Luck H, Tsai S, Winer S. The Intestinal Immune System in Obesity and Insulin Resistance. Cell Metab. 2016;23:413-426. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 297] [Cited by in RCA: 359] [Article Influence: 39.9] [Reference Citation Analysis (0)] |
| 4. | Sairenji T, Collins KL, Evans DV. An Update on Inflammatory Bowel Disease. Prim Care. 2017;44:673-692. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 217] [Cited by in RCA: 347] [Article Influence: 43.4] [Reference Citation Analysis (13)] |
| 5. | Sands BE. Biomarkers of Inflammation in Inflammatory Bowel Disease. Gastroenterology. 2015;149:1275-1285.e2. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 213] [Cited by in RCA: 288] [Article Influence: 28.8] [Reference Citation Analysis (0)] |
| 6. | Lechuga S, Ivanov AI. Disruption of the epithelial barrier during intestinal inflammation: Quest for new molecules and mechanisms. Biochim Biophys Acta Mol Cell Res. 2017;1864:1183-1194. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 187] [Article Influence: 23.4] [Reference Citation Analysis (0)] |
| 7. | Moschen AR, Adolph TE, Gerner RR, Wieser V, Tilg H. Lipocalin-2: A Master Mediator of Intestinal and Metabolic Inflammation. Trends Endocrinol Metab. 2017;28:388-397. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 175] [Cited by in RCA: 261] [Article Influence: 32.6] [Reference Citation Analysis (0)] |
| 8. | Bischoff SC, Barbara G, Buurman W, Ockhuizen T, Schulzke JD, Serino M, Tilg H, Watson A, Wells JM. Intestinal permeability--a new target for disease prevention and therapy. BMC Gastroenterol. 2014;14:189. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 893] [Cited by in RCA: 1218] [Article Influence: 110.7] [Reference Citation Analysis (0)] |
| 9. | Zhou Q, Costinean S, Croce CM, Brasier AR, Merwat S, Larson SA, Basra S, Verne GN. MicroRNA 29 targets nuclear factor-κB-repressing factor and Claudin 1 to increase intestinal permeability. Gastroenterology. 2015;148:158-169.e8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 106] [Cited by in RCA: 140] [Article Influence: 14.0] [Reference Citation Analysis (0)] |
| 10. | Thaiss CA, Levy M, Grosheva I, Zheng D, Soffer E, Blacher E, Braverman S, Tengeler AC, Barak O, Elazar M, Ben-Zeev R, Lehavi-Regev D, Katz MN, Pevsner-Fischer M, Gertler A, Halpern Z, Harmelin A, Aamar S, Serradas P, Grosfeld A, Shapiro H, Geiger B, Elinav E. Hyperglycemia drives intestinal barrier dysfunction and risk for enteric infection. Science. 2018;359:1376-1383. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 416] [Cited by in RCA: 620] [Article Influence: 88.6] [Reference Citation Analysis (0)] |
| 11. | Laurans L, Venteclef N, Haddad Y, Chajadine M, Alzaid F, Metghalchi S, Sovran B, Denis RGP, Dairou J, Cardellini M, Moreno-Navarrete JM, Straub M, Jegou S, McQuitty C, Viel T, Esposito B, Tavitian B, Callebert J, Luquet SH, Federici M, Fernandez-Real JM, Burcelin R, Launay JM, Tedgui A, Mallat Z, Sokol H, Taleb S. Genetic deficiency of indoleamine 2,3-dioxygenase promotes gut microbiota-mediated metabolic health. Nat Med. 2018;24:1113-1120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 174] [Cited by in RCA: 217] [Article Influence: 31.0] [Reference Citation Analysis (0)] |
| 12. | Eisenstein M. Ulcerative colitis: Towards remission. Nature. 2018;563:S33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 121] [Article Influence: 20.2] [Reference Citation Analysis (0)] |
| 13. | Barreiro-de Acosta M, Marín-Jiménez I, Panadero A, Guardiola J, Cañas M, Gobbo Montoya M, Modino Y, Alcaín G, Bosca-Watts MM, Calvet X, Casellas F, Chaparro M, Fernández Salazar L, Ferreiro-Iglesias R, Ginard D, Iborra M, Manceñido N, Mañosa M, Merino O, Rivero M, Roncero O, Sempere L, Vega P, Zabana Y, Mínguez M, Nos P, Gisbert JP. Recommendations of the Spanish Working Group on Crohn's Disease and Ulcerative Colitis (GETECCU) and the Association of Crohn's Disease and Ulcerative Colitis Patients (ACCU) in the management of psychological problems in Inflammatory Bowel Disease patients. Gastroenterol Hepatol. 2018;41:118-127. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 28] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 14. | Liang NC, Devereaux A. Integrative Medicine (Complementary and Alternative Medicine) for the Lungs. Am J Respir Crit Care Med. 2017;195:P21-P22. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 15. | Xie L, Draizen EJ, Bourne PE. Harnessing Big Data for Systems Pharmacology. Annu Rev Pharmacol Toxicol. 2017;57:245-262. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 35] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 16. | Fleisher B, Brown AN, Ait-Oudhia S. Application of pharmacometrics and quantitative systems pharmacology to cancer therapy: The example of luminal a breast cancer. Pharmacol Res. 2017;124:20-33. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 7] [Cited by in RCA: 10] [Article Influence: 1.3] [Reference Citation Analysis (0)] |
| 17. | Ru J, Li P, Wang J, Zhou W, Li B, Huang C, Li P, Guo Z, Tao W, Yang Y, Xu X, Li Y, Wang Y, Yang L. TCMSP: A database of systems pharmacology for drug discovery from herbal medicines. J Cheminform. 2014;6:13. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 1623] [Cited by in RCA: 3145] [Article Influence: 285.9] [Reference Citation Analysis (0)] |
| 18. | Dai YC, Zheng L, Zhang YL, Chen X, Chen DL, Tang ZP. Effects of Jianpi Qingchang decoction on the quality of life of patients with ulcerative colitis: A randomized controlled trial. Medicine (Baltimore). 2017;96:e6651. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 28] [Cited by in RCA: 30] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 19. | Zheng L, Zhang YL, Dai YC, Chen X, Chen DL, Dai YT, Tang ZP. Jianpi Qingchang decoction alleviates ulcerative colitis by inhibiting nuclear factor-κB activation. World J Gastroenterol. 2017;23:1180-1188. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 42] [Cited by in RCA: 44] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 20. | Dai YC, Zheng L, Zhang YL, Chen X, Chen DL, Wang LJ, Tang ZP. Jianpi Qingchang decoction regulates intestinal motility of dextran sulfate sodium-induced colitis through reducing autophagy of interstitial cells of Cajal. World J Gastroenterol. 2017;23:4724-4734. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 31] [Cited by in RCA: 31] [Article Influence: 3.9] [Reference Citation Analysis (0)] |
| 21. | Xu X, Zhang W, Huang C, Li Y, Yu H, Wang Y, Duan J, Ling Y. A novel chemometric method for the prediction of human oral bioavailability. Int J Mol Sci. 2012;13:6964-6982. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 564] [Cited by in RCA: 622] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 22. | Tao W, Xu X, Wang X, Li B, Wang Y, Li Y, Yang L. Network pharmacology-based prediction of the active ingredients and potential targets of Chinese herbal Radix Curcumae formula for application to cardiovascular disease. J Ethnopharmacol. 2013;145:1-10. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 368] [Cited by in RCA: 453] [Article Influence: 37.8] [Reference Citation Analysis (0)] |
| 23. | Tang Y, Li M, Wang J, Pan Y, Wu FX. CytoNCA: A cytoscape plugin for centrality analysis and evaluation of protein interaction networks. Biosystems. 2015;127:67-72. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 483] [Cited by in RCA: 779] [Article Influence: 70.8] [Reference Citation Analysis (0)] |
| 24. | Bindea G, Mlecnik B, Hackl H, Charoentong P, Tosolini M, Kirilovsky A, Fridman WH, Pagès F, Trajanoski Z, Galon J. ClueGO: A Cytoscape plug-in to decipher functionally grouped gene ontology and pathway annotation networks. Bioinformatics. 2009;25:1091-1093. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 3770] [Cited by in RCA: 4737] [Article Influence: 296.1] [Reference Citation Analysis (0)] |
| 25. | Chaudhary G, Mahajan UB, Goyal SN, Ojha S, Patil CR, Subramanya SB. Protective effect of Lagerstroemia speciose against dextran sulfate sodium induced ulcerative colitis in C57BL/6 mice. Am J Transl Res. 2017;9:1792-1800. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1] [Cited by in RCA: 1] [Article Influence: 0.1] [Reference Citation Analysis (0)] |
| 26. | Ying W, Cheruku PS, Bazer FW, Safe SH, Zhou B. Investigation of macrophage polarization using bone marrow derived macrophages. J Vis Exp. 2013;(76). [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 179] [Article Influence: 14.9] [Reference Citation Analysis (0)] |
| 27. | Chen YL, Zhang YL, Dai YC, Tang ZP. Systems pharmacology approach reveals the antiinflammatory effects of Ampelopsis grossedentata on dextran sodium sulfate-induced colitis. World J Gastroenterol. 2018;24:1398-1409. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in CrossRef: 20] [Cited by in RCA: 19] [Article Influence: 2.7] [Reference Citation Analysis (0)] |
| 28. | Li S, Zhang B. Traditional Chinese medicine network pharmacology: Theory, methodology and application. Chin J Nat Med. 2013;11:110-120. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 341] [Cited by in RCA: 627] [Article Influence: 57.0] [Reference Citation Analysis (0)] |
| 29. | Lei Q, Qiang F, Chao D, Di W, Guoqian Z, Bo Y, Lina Y. Amelioration of hypoxia and LPS-induced intestinal epithelial barrier dysfunction by emodin through the suppression of the NF-κB and HIF-1α signaling pathways. Int J Mol Med. 2014;34:1629-1639. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 30. | Xue X, Ramakrishnan S, Anderson E, Taylor M, Zimmermann EM, Spence JR, Huang S, Greenson JK, Shah YM. Endothelial PAS domain protein 1 activates the inflammatory response in the intestinal epithelium to promote colitis in mice. Gastroenterology. 2013;145:831-841. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 127] [Cited by in RCA: 145] [Article Influence: 12.1] [Reference Citation Analysis (0)] |
| 31. | Leonardi M, Perna E, Tronnolone S, Colecchia D, Chiariello M. Activated kinase screening identifies the IKBKE oncogene as a positive regulator of autophagy. Autophagy. 2018;1-15. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 18] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 32. | Fan W, Fan L, Peng C, Zhang Q, Wang L, Li L, Wang J, Zhang D, Peng W, Wu C. Traditional Uses, Botany, Phytochemistry, Pharmacology, Pharmacokinetics and Toxicology of Xanthium strumarium L.: A Review. Molecules. 2019;24:pii: E359. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 39] [Cited by in RCA: 66] [Article Influence: 11.0] [Reference Citation Analysis (0)] |
| 33. | Wang D, Calabrese EJ, Lian B, Lin Z, Calabrese V. Hormesis as a mechanistic approach to understanding herbal treatments in traditional Chinese medicine. Pharmacol Ther. 2018;184:42-50. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 59] [Article Influence: 7.4] [Reference Citation Analysis (0)] |
| 34. | Balamurugan K. HIF-1 at the crossroads of hypoxia, inflammation, and cancer. Int J Cancer. 2016;138:1058-1066. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 366] [Cited by in RCA: 436] [Article Influence: 48.4] [Reference Citation Analysis (1)] |
| 35. | Kim JY, Lee HJ, Lee SJ, Jung YH, Yoo DY, Hwang IK, Seong JK, Ryu JM, Han HJ. Palmitic Acid-BSA enhances Amyloid-β production through GPR40-mediated dual pathways in neuronal cells: Involvement of the Akt/mTOR/HIF-1α and Akt/NF-κB pathways. Sci Rep. 2017;7:4335. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 30] [Cited by in RCA: 57] [Article Influence: 7.1] [Reference Citation Analysis (0)] |
| 36. | Chiche J, Pommier S, Beneteau M, Mondragón L, Meynet O, Zunino B, Mouchotte A, Verhoeyen E, Guyot M, Pagès G, Mounier N, Imbert V, Colosetti P, Goncalvès D, Marchetti S, Brière J, Carles M, Thieblemont C, Ricci JE. GAPDH enhances the aggressiveness and the vascularization of non-Hodgkin's B lymphomas via NF-κB-dependent induction of HIF-1α. Leukemia. 2015;29:1163-1176. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 40] [Cited by in RCA: 53] [Article Influence: 4.8] [Reference Citation Analysis (0)] |
| 37. | Xie A, Robles RJ, Mukherjee S, Zhang H, Feldbrügge L, Csizmadia E, Wu Y, Enjyoji K, Moss AC, Otterbein LE, Quintana FJ, Robson SC, Longhi MS. HIF-1α-induced xenobiotic transporters promote Th17 responses in Crohn's disease. J Autoimmun. 2018;94:122-133. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 41] [Article Influence: 5.9] [Reference Citation Analysis (0)] |
| 38. | Laberge RM, Sun Y, Orjalo AV, Patil CK, Freund A, Zhou L, Curran SC, Davalos AR, Wilson-Edell KA, Liu S, Limbad C, Demaria M, Li P, Hubbard GB, Ikeno Y, Javors M, Desprez PY, Benz CC, Kapahi P, Nelson PS, Campisi J. MTOR regulates the pro-tumorigenic senescence-associated secretory phenotype by promoting IL1A translation. Nat Cell Biol. 2015;17:1049-1061. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 797] [Cited by in RCA: 838] [Article Influence: 83.8] [Reference Citation Analysis (0)] |