Published online Sep 28, 2018. doi: 10.3748/wjg.v24.i36.4152
Peer-review started: July 3, 2018
First decision: July 17, 2018
Revised: August 6, 2018
Accepted: August 24, 2018
Article in press: August 24, 2018
Published online: September 28, 2018
Processing time: 84 Days and 18.1 Hours
Sorafenib has been considered the standard of care for patients with advanced unresectable hepatocellular carcinoma (HCC) since 2007 and numerous studies have investigated the role of markers involved in the angiogenesis process at both the expression and genetic level and clinical aspect. What results have ten years of research produced? Several clinical and biological markers are associated with prognosis. The most interesting clinical parameters are adverse events, Barcelona Clinic Liver Cancer stage, and macroscopic vascular invasion, while several single nucleotide polymorphisms and plasma angiopoietin-2 levels represent the most promising biological biomarkers. A recent pooled analysis of two phase III randomized trials showed that the neutrophil-to-lymphocyte ratio, etiology and extra-hepatic spread are predictive factors of response to sorafenib, but did not identify any predictive biological markers. After 10 years of research into sorafenib there are still no validated prognostic or predictive factors of response to the drug in HCC. The aim of the present review was to summarize 10 years of research into sorafenib, looking in particular at the potential of associated clinical and biological markers to predict its efficacy in patients with advanced HCC.
Core tip: Sorafenib has been considered the standard of care for patients with advanced unresectable hepatocellular carcinoma, but after 10 years of research into sorafenib response or resistance, there are still no validated prognostic or predictive factors of response.
- Citation: Marisi G, Cucchetti A, Ulivi P, Canale M, Cabibbo G, Solaini L, Foschi FG, De Matteis S, Ercolani G, Valgiusti M, Frassineti GL, Scartozzi M, Casadei Gardini A. Ten years of sorafenib in hepatocellular carcinoma: Are there any predictive and/or prognostic markers? World J Gastroenterol 2018; 24(36): 4152-4163
- URL: https://www.wjgnet.com/1007-9327/full/v24/i36/4152.htm
- DOI: https://dx.doi.org/10.3748/wjg.v24.i36.4152
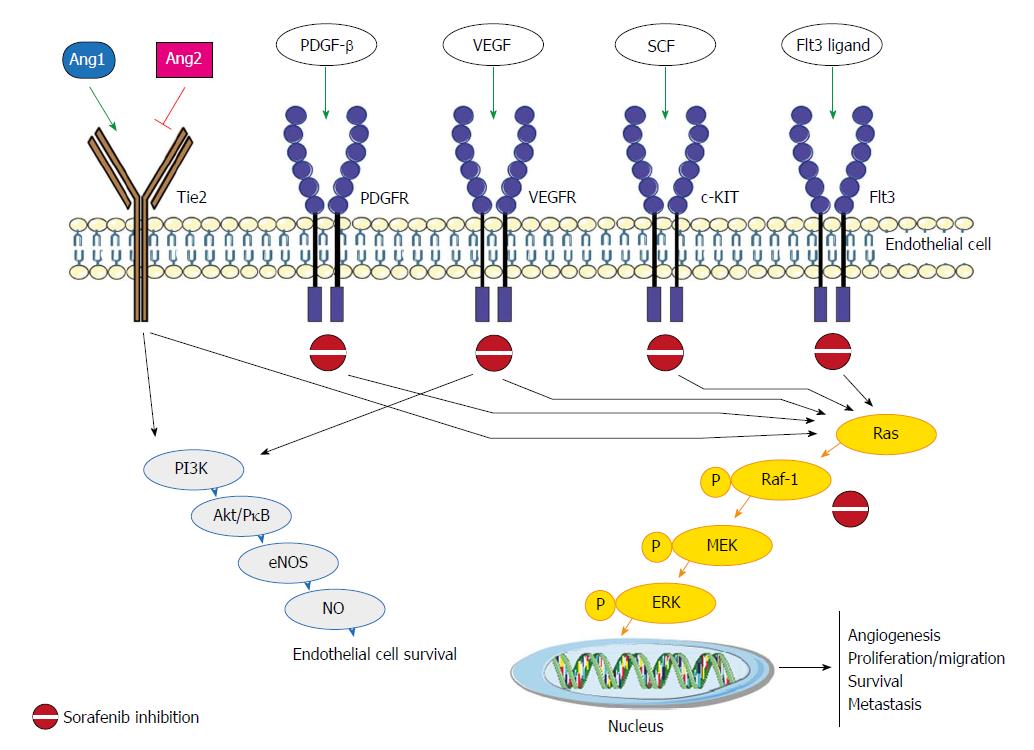
Sorafenib, an oral multikinase inhibitor, has been considered the standard of care for patients with advanced unresectable hepatocellular carcinoma (HCC) since 2007[1]. It works by inhibiting the activity of several tyrosine kinases involved in tumor angiogenesis and progression, including vascular endothelial growth factor receptor (VEGFR-2/3), platelet-derived growth factor receptor (PDGF-R), Flt3 and c-Kit, and also targets Raf kinases involved in the MAPK/ERK pathway[2] (Figure 1). The molecular mechanisms by which sorafenib exerts its activity have still not been fully elucidated, and both Raf/MEK/ERK-dependent and -independent mechanisms have been observed[3].
Sorafenib is expensive and associated with adverse events (AEs). Furthermore, a proportion of treated patients show no response to the drug. It would thus be useful to have predictive markers capable of identifying those who are more likely to benefit from therapy. The availability of more accurate predictive or prognostic factors would also help to spare potentially resistant patients from unnecessary toxicity.
Ten years have passed since sorafenib was first commercialized and about 2800 studies have been published on the kinase inhibitor. But how many associated prognostic and/or predictive markers have been identified? Numerous studies have focused on the role of markers involved in the angiogenesis process at both the expression and genetic levels. The largest biomarker study conducted to date is the SHARP trial[4], which included an adequate number of participants and a placebo-controlled group. Smaller single-arm studies exploring predictive or prognostic markers for sorafenib have also been conducted, but the results of these have yet to be validated.
The aim of the present review was to summarize 10 years of research into sorafenib, looking in particular at the potential of associated clinical and biological markers to predict its efficacy in patients with advanced HCC (Tables 1 and 2).
| Clinical markers | Predictive value | Prognostic value | Ref. |
| Alpha-fetoprotein | No | Yes | [6] |
| Adverse events | |||
| Hand-foot skin reaction | No | Yes | [13] |
| Hypertension | No | Uncertain | [16,19,20] |
| Diarrhea | No | Yes | [21] |
| Child-Pugh A vs B | No | Yes | [27-29] |
| Macroscopic vascular invasion | No | Yes | [6] |
| BCLC B vs C | No | Yes | [6,29,32] |
| Starting dose and dose reduction | No | Yes | [29,32] |
| Etiology HCV vs HBV | Yes | Yes | [6] |
| Chronic treatment with metformin | No | Yes | [35,36] |
| Neutrophil-to-lymphocyte ratio | Yes | Yes | [6,41,44] |
| Extra hepatic spread | Yes | Yes | [6] |
| Biological markers | Predictive value | Prognostic value | Ref. |
| Serum and plasma proteins | |||
| VEGF-A | No | Uncertain | [4,57] |
| Ang-2 | No | Yes | [4] |
| IGF-1 | No | No | [55] |
| Single nucleotide polymorphisms | |||
| VEGF-A rs2010963 | No | Yes | [65] |
| VEGF-C rs4604006 | No | Yes | [65] |
| eNOS (eNOS-786/eNOS VNTR) | No | Yes | [66] |
| Ang-2 rs55633437 | No | Yes | [67] |
| HIF-1 alpha rs12434438 | No | Yes | [68] |
| Amplifications | |||
| VEGF | No | Uncertain | [70] |
| FGF3/FGF4 | No | Uncertain | [71] |
| miRNAs | |||
| miR-425-3p | No | Yes | [74] |
| miR-224 | No | Yes | [75] |
| miR-181a-5p | No | Yes | [77] |
| miR-339-5p | No | Yes | [77] |
| miR-423-5p | No | Yes | [78] |
| miR-10b-3p | No | Yes | [79] |
| miR-221 | No | Uncertain | [76] |
| Tissue biomarker expression | |||
| Phospho-ERK | Uncertain | Uncertain | [81,82] |
| PDGFR-b | No | Yes | [84] |
| c-Met | No | No | [84] |
| VEGFR | No | No | [84] |
| p-c-Jun | No | Yes | [85] |
Alpha-fetoprotein (AFP) is secreted by about 50% of all HCCs and is the main serological marker used for the diagnosis of the tumor[5]. The SHARP trial[4] showed that high baseline AFP plasma levels (> 200 ng/mL) had a negative impact on overall survival (OS), a finding recently confirmed in a pooled analysis of the SHARP trial and the Asia Pacific trial by Bruix et al[6]. High baseline serum AFP levels (≥ 400 ng/mL) also appear to be associated with shorter time-to-progression (TTP). Notably, in an analysis of six prospective phase II trials evaluating systemic therapies for patients with advanced HCC, no association was observed between baseline AFP levels and prognosis[7].
Several studies[8-10] have highlighted a consistent correlation between an early decrease of > 20% in AFP levels following sorafenib and objective response and better outcome in advanced HCC patients. Shao et al[8] evaluated for the first time this aspect and they observed that patients with early AFP response had an improved progression-free survival (PFS) (7.5 mo vs 1.9 mo) and OS (15.3 mo vs 4.1 mo). This data was confirmed by Personeni et al[10] a few years later. They reported that early responders had a significantly better median OS and TTP than non-responders (13.8 mo vs 8.2 mo, P = 0.022 and 7.9 mo vs 2.4 mo, P = 0.004; respectively). Conversely, Nakazawa et al[11] did not observe such an association.
The main AEs of Sorafenib are hand-foot skin reaction (HFSR), hypertension and diarrhea. Several papers have highlighted a consistent correlation between AEs and survival in patients treated with Sorafenib.
Vincenzi et al[12] evaluated for the first time the correlation between HSFR and outcome. They showed, in a small series of patients treated with sorafenib, that patients with HSFR had a significantly higher disease control rate with respect to patients without HSFR. This data was confirmed in a prospective study of 147 patients by Reig et al[13]. They reported different OS when patients were subdivided according to the presence or not of skin toxicity during the first 60 d of treatment (18.2 mo vs 10.1 mo, respectively)[13] . A recently meta-analysis confirmed that HSFR was a good indicator of outcome for OS and TTP in HCC patients receiving sorafenib[14]
Hypertension (HTN) is frequently associated with the use of angiogenesis inhibitors[15]. Casadei Gardini et al[16] showed that early HTN (15 d after the start of treatment) rather than later onset HTN vs patients without HTN was associated with better PFS (6.0 mo vs 2.5 mo; P < 0.001) and OS (14.6 mo vs 3.9 mo; P = 0.003). This finding has been confirmed in some studies[17,18] but not in others[19,20].
Bettinger et al[21] reported for the first time that diarrhea was an independent positive prognostic factor (HR = 0.41; P = 0.001) in 112 patients with advanced HCC, a finding also confirmed by Koschny et al[22].
Finally, other authors showed that the number of AEs was associated with predict survival in patients treated with sorafenib. In particular, Di Costanzo et al[23] evaluated the potential of pretreatment clinical variables to predict survival. Three groups of patients were taken into account: patients without AEs (group 0), patients with one AE (group 1) and patients with two to three AEs (group 2). The study reported a strong correlation between this classification and disease progression at 3 mo (41.9%, 25.9% and 12.7% of patients in groups 0, 1 and 2, respectively; P = 0.014). These data were subsequently confirmed in the validation cohort[24]. A recent meta-analysis by Abdel-Rahman et al[25] revealed an association between specific side-effects (hypertension, HFSR and diarrhea) and patient outcome (HR = 0.38; 95%CI: 0.30-0.48; P < 0.00001).
Child-Pugh A vs Child-Pugh B: In the SHARP trial[4] and the Asia Pacific trial[26], more than 95% of patients were classified as having Child-Pugh A cirrhosis, thus preventing the investigation of the potential benefits of sorafenib in Child-Pugh B patients.
Hollebecque et al[27] reported for the first time the results from a prospective study on sorafenib efficacy in 120 advanced HCC patients, 20 of whom Child-Pugh B cirrhosis. OS was 11.1 mo, with a significantly longer median survival in Child-Pugh A patients than Child-Pugh B patients (13 mo vs 4.5 mo, P = 0.0008). A few years later, Pressiani et al[28] studied clinical outcome in a population of 300 consecutive patients; PFS in the Child-Pugh A group was 4.3 mo vs 2.1 mo in the Child-Pugh B arm (HR = 3.23; 95%CI: 2.38-4.39; P < 0.001), TTP was 4.2 mo vs 3.8 mo and OS was 10.0 mo vs 3.8 mo, respectively (P < 0.001).
The most important work on the use of sorafenib in Child-Pugh subgroups was the GIDEON study published in 2016[29]. This study observed that median OS was significantly longer in patients with Child-Pugh A (13.6 mo) than in those with Child Pugh B (5.2 mo) or Child-Pugh C (2.6 mo).
Macroscopic vascular invasion: It is widely acknowledged that the presence of macroscopic vascular invasion leads to a poorer prognosis[26,30]. The meta-analysis by Peng et al[31] confirmed its prognostic value and the pooled analysis by Bruix et al[6] affirmed the importance of macroscopic vascular invasion as a predictor of survival but not of response to treatment.
BCLC stage: In the SHARP trial, patients with Barcelona Clinic Liver Cancer (BCLC) B had a median OS of 14.5 mo compared to 9.7 mo for those with BCLC C[4]. Later, SOFIA[32] and GIDEON study[29] confirmed this data. In the SOFIA trial[32] the OS was 8.4 mo in BCLC C vs 20.6 mo in BCLC B patients (P < 0.0001), but the time to radiologic progression did not differ significantly between the 2 groups. In the GIDEON study[29], median OS according to BCLC by Child-Pugh cross-classification followed a similar trend, i.e. patients with Child-Pugh A and BCLC stage B showed longer OS than those with Child-Pugh B and BCLC B (19.5 mo vs 10.0 mo); and patients with Child-Pugh A and BCLC stage C had longer OS than those with Child-Pugh B and BCLC stage C (11.2 mo vs 3.8 mo).
Recently, Bruix et al[6]’s pooled analysis confirmed that BCLC C patients had a poorer prognosis than those with BCLC B HCC (HR = 1.59; P = 0.02).
Sorafenib starting dose and dose escalation/reduction: The two most important studies that evaluated sorafenib starting dose and dose escalation/reduction are SOFIA[32] and GIDEON trial[29].
In the SOFIA trial[32] sorafenib was down-dosed in 161 (54%) patients because of AEs (133 patients, 83%) and a reduction in liver function (28 patients, 17%). Median OS of the 77 patients receiving a half-dose of sorafenib for 70% of the treatment period was 21.6 mo (95%CI: 13.6-29.6) compared with 9.6 mo (95%CI: 6.9-12.3) for the remaining 219 patients who had a dose reduction for < 70% of the treatment period or who maintained the full dosage.
A sub-analysis of the GIDEON study[29] evaluated the starting dose of sorafenib with respect to clinical outcome and toxicity. Patients starting on 400 mg/d were slightly older, had baseline characteristics indicative of greater disease progression and had a higher incidence of AEs than those with a starting dose of 800 mg/d (96% vs 88%). Treatment duration (18.0 wk vs 13.0 wk) and median OS (12.1 mo vs 9.4 mo) were longer in patients receiving 800 mg/d.
Etiology: In the subgroup analysis of the SHARP study[4], the HR for OS was 0.76 in HBV-positive patients (95%CI: 0.38-1.50, P = not significant) and 0.50 (95%CI: 0.32-0.77) in HCV-positive patients. Results were similar for TTP (HR = 1.03 and 0.43 for HBV-positive and HCV-positive patients, respectively). Similar data were obtained for HBV-positive HCC patients in the phase III randomized Asia Pacific trial, i.e. the HR for OS was 0.74 (95%CI: 0.51-1.06, not significant) with respect to patients with the other etiology, for which the HR was 0.57 (95%CI: 0.29-1.33)[26]. Bruix et al[6]’s pooled analysis of the SHARP/Asia Pacific trial results showed that the absence of HCV was a potential prognostic factor for poorer OS (HR = 0.7, P = 0.02). The same authors revealed that HBV-positive patients did not show a significant difference in treatment response with respect to their HBV-negative counterparts (HR = 0.78; 95%CI: 0.57-1.06) and OS (HR = 1.128, P = 0.4538). We believe that the 2 etiologic groups respond differently to sorafenib and that further investigation is warranted in specific studies[33].
Metformin treatment: Type 2 diabetes is a significant risk factor for the development of malignancies, including HCC[34]. Casadei Gardini et al[35] published findings of reduced sorafenib efficacy in HCC patients treated chronically with or without metformin for type II diabetes mellitus (PFS 2.6 mo vs 5.0 mo, respectively; and OS 10.4 mo vs 15.1 mo, respectively). The same authors validated these data in a series of more than 250 cases[36], also highlighting a possible role of sirtuin-3 in resistance to sorafenib[37]. Di Costanzo et al[38] recently reported an increase in TTP and OS in diabetic with respect to non-diabetic HCC patients. However, no distinction was made between the different hypoglycemic therapies administered.
Systemic inflammatory responses have been shown to reflect the promotion of angiogenesis, DNA damage and tumor invasion through an upregulation of cytokines[39]. Previous research revealed that lymphocytes play a crucial role in tumor defense by inducing cytotoxic cell death and inhibiting tumor cell proliferation and migration[40]. Consequently, several inflammation and immune-based prognostic scores, such as lymphocyte count, neutrophil-lymphocyte ratio (NLR), and systemic immune-inflammation index (SII), have been developed to predict survival and recurrence in cancers, including HCC. Casadei Gardini et al[41] evaluated for the first time SII, NLR and platelet-lymphocyte ratio (PLR) in a small case series, observing that SII were independent prognostic factors for OS. Other studies showed that NLR was a significant independent risk factor for shorter survival[42,43]. NLR was also found to be an independent prognostic factor for both response and survival in Bruix et al[6]’s pooled analysis and Lue et al[44]’s retrospective study on Spanish patients.
The response to sorafenib does not correlate with a change in lesion dimension, but it is more correlate with intralesional vascularization. For this reason, the RECIST criteria[45,46] usually used for tumor response evaluation is inappropriate to evaluate the response to sorafenib in patients with advanced HCC. The modified RECIST (mRECIST) appear more indicate for evalutation the response. They include vascularization and tumor arterial enhancement changes of the target lesion on computed tomographic (CT). Several studies have demonstrated the superiority of the mRECIST criteria with respect to the RECIST criteria in assessing the response to treatment with sorafenib[47]. Various functional imaging tools were proposed to evaluate the antiangiogenic effects, but none of these has entered normal clinical practice[48-52]. Finally, a recently study showed that texture features on pretreatment contrast material-enhanced CT images can help predict OS and TTP in these patients[53] .
Although plasma biomarkers are the best candidates for evaluating sorafenib efficacy, only the SHARP trial produced results with borderline significance[4]. Baseline angiopoietin-2 (Ang-2) and vascular endothelial growth factor-A (VEGF-A) plasma levels independently predicted survival in both the entire patient population and the placebo cohort. Conversely, none of the tested biomarkers significantly predicted response to sorafenib[4]. Insulin-like growth factor (IGF)-1 levels have been found to decrease in patients with cirrhosis of the liver or HCC[54], and high pretreatment levels of IGF-1 predict better PFS and OS in advanced HCC patients receiving first-line antiangiogenic therapy[55].
The role of serum cytokines as biomarkers for the prediction of sorafenib responses is interesting, in particular Kim et al[56] developed a new prediction model for sorafenib response that combines relevant serum markers, tumor related factors, and cirrhosis-related factors in a scoring system.
VEGF-A: Llovet et al[4] showed that, although baseline plasma VEGF-A concentrations did not exhibit a predictive value, low plasma VEGF-A was associated with improved prognosis (HR = 1.48, 95%CI: 1.08-2.03, P = 0.015). However, other authors did not find any association between VEGF-A and prognosis in patients treated with sorafenib[57]. Tsuchiya et al[58]’s analysis of plasma VEGF concentrations during sorafenib treatment revealed that a decrease in the protein 8 wk after the start of therapy predicted better overall survival in advanced HCC patients (30.9 mo vs 14.4 mo; P = 0.038).
Ang-2: In the presence of VEGF, Ang-2 destabilizes blood vessels, promotes vascular sprouting, and is associated with an invasive and metastatic cancer phenotype[59]. Llovet et al[4] demonstrated that high baseline Ang-2 levels were correlated with more aggressive disease (HR = 1.58, 95%CI: 1.20-2.07, P = 0.001). Moreover, levels of the protein increased during treatment in the placebo group, suggestive of poor outcome related to disease progression in this cohort, whereas they remained constant during treatment with sorafenib, reflecting the generally more favorable outcome of this group. Overall increased Ang-2 expression levels were associated with poorer outcome in both groups, suggesting that this marker could be useful in monitoring treatment response. In agreement with Llovet’s study, Miyahara et al[57] reported that high baseline Ang-2 serum levels were associated with poor outcome in advanced HCC patients receiving sorafenib (HR = 2.51, 95%CI: 1.01-6.57, P = 0.048). Although these results indicate the potential prognostic value of Ang-2 in HCC, its role in predicting response to sorafenib remains to be verified.
IGF-1: Shao et al[55] found that high-pretreatment serum levels of IGF-1 were associated with a better DCR and improved PFS and OS in patients undergoing antiangiogenic therapy. Although the study did not have a control arm, the substantial significant difference in DCR between patients with high and low levels of IGF-1 (71% vs 39%) denotes the potential usefulness of IGF-1 as a predictive biomarker of response to antiangiogenic therapy.
Multiple-factor analyses: By using baseline serum basic fibroblast growth factor (bFGF) and hepatocyte growth factor (HGF) levels as covariates together etiology (B-viral), platelet count, BCLC stage and protein induced by vitamin K absence-II, Kim et al[56] reported that a total score of < 6 could be a relevant cutoff value for selecting patients who are most likely to benefit from sorafenib therapy. Moreover, Hayashi et al[60] found that serum interleukin (IL)-5, IL-8, CXCL9, PDGF-BB, TGF-α, and VEGF-A were elevated in the long survivors group among HCC patients who received sorafenib, potentially reflecting the activation of stromal signaling in the tumor microenvironment.
Molecular and genomic analyses from tumor and non-tumor tissue have proven useful in evaluating prognosis and could open up new avenues for tailoring treatment[61]. Genetic alterations, such as single nucleotide polymorphisms (SNPs) in genes encoding for proteins involved in the angiogenic process, have been studied as potential biomarkers for anti-angiogenic therapy. SNP evaluation would seem to be more advantageous than protein or gene expression analyses as it can be performed at any time during the course of the disease, is not substantially influenced by laboratory biases, and is relatively inexpensive. Some authors have focused on molecular profiling in formalin-fixed paraffin-embedded (FFPE) samples, comparing the mutation profiles of HCC biopsy samples and the response to sorafenib treatment[62]. Gene amplification, gene mutations and expression profiling of tumors have now become a research priority and are expected to lead to personalized treatment for HCC patients[63,64].
SNPs: Specific SNPs in VEGF and VEGFR genes have been found to be correlated with PFS and OS in HCC patients treated with sorafenib. In multivariate analysis, VEGF-A rs2010963 and VEGF-C rs4604006 were found to be independent factors influencing outcome in terms of PFS (HR = 0.25, 95%CI: 0.19-1.02, P = 0.0376 and HR = 0.22, 95%CI: 0.14-0.81, P = 0.004, respectively) and OS (HR = 0.28, 95%CI: 0.23-0.96, P = 0.02 and HR = 0.25, 95%CI: 0.17-0.99, P = 0.04, respectively)[65]. In the Italian multicenter, retrospective ePHAS [endothelial nitric oxide synthase (eNOS) polymorphisms in HCC and sorafenib] study, eNOS polymorphisms were analyzed in relation to PFS and OS. In univariate analysis, training cohort patients homozygous for eNOS haplotype (HT1:T-4b at eNOS-786/eNOS VNTR) showed a lower median PFS (2.6 mo vs 5.8 mo, HR = 5.43, 95%CI: 2.46-11.98, P < 0.0001) and OS (3.2 mo vs 14.6 mo, HR = 2.35, 95%CI: 1.12-4.91, P = 0.024) than those with other haplotypes. These results were confirmed in a validation set and multivariate analysis further substantiated this haplotype as the only independent prognostic factor[66]. More recently, evidence emerged that patients homozygous for ANGPT2 (Ang2 gene) rs55633437 GG genotype showed significantly longer PFS (P < 0.001) and OS (P < 0.001) than those with the other genotypes (GT+TT)[67].
In the ALICE-2 study, Faloppi et al[68] investigated the role of hypoxia-inducible factor 1-alpha (HIF-1α) SNPs, confirming the results of the ALICE-1 study[65]. In multivariate analysis, rs12434438 of HIF-1α, rs2010963 of VEGF-A and rs4604006 of VEGF-C were confirmed as independent factors and may help to identify patients who are more likely to respond to sorafenib[68]. The prospective INNOVATE study is ongoing to validate the role of VEGF, eNOS, Ang-2 and HIF-1α SNPs in relation to clinical outcome in advanced HCC patients treated with sorafenib (NCT02786342)[69].
Gene amplification, gene mutations and RNA expression: A relation between VEGF-A gene amplification and response to sorafenib was observed in a study performed on a mouse model of HCC[70]. The authors found that HCC patients with tumor VEGF-A amplification showed markedly better survival than those with non-amplified tumors, highlighting that VEGFA amplification is a potential biomarker of response to VEGF-A-blocking drugs in HCC[70]. Arao et al[71] observed that FGF3/FGF4 amplification and multiple lung metastases were frequently observed in responders to sorafenib, although the sample size was relatively small.
Sakai et al[62] used targeted DNA and RNA sequencing in FFPE specimens from fine-needle biopsy to identify candidate biomarkers of response to sorafenib in 46 HCC patients. A significant difference was observed in the number of oncogene mutations between progressing and non-progressing patients (P = 0.045), suggesting that tumor mutational burden may be predictive of sorafenib effectiveness. Tumor gene expression of NRG1, TGFa, and PECAM1 would also seem to be a marker of treatment response and PFS.
MicroRNAs (miRNAs) affect drug response directly or indirectly by regulating the expression of genes involved in drug transportation, metabolism, and downstream signaling pathways. The deregulation of various miRNAs has been reported in in vitro, in vivo and population studies[72,73], confirming its correlation with response to sorafenib. Some authors have evaluated the predictive role of miRNA expression in HCC tissue[74,75], while others have studied circulating miRNA levels prior to sorafenib treatment[76]. To date, the most interesting tissue miRNAs are miR-425-3p[74] and miR-224[75]. High levels of miR-425-3p have been associated with longer TTP and PFS (HR = 0.4, 95%CI: 0.2-0.7, P = 0.0008 and HR = 0.5, 95%CI: 0.3-0.9, P = 0.007, respectively), and elevated miR-224 expression have been correlated with increased PFS and OS (HR = 0.28, 95%CI: 0.09-0.92, P = 0.029 and HR = 0.0.24, 95%CI: 0.07-0.79, P = 0.012, respectively). Circulating miRNAs have also been studied in the serum of HCC patients to predict early response to sorafenib treatment, with miR-181a-5p and miR-339-5p associated with partial response and disease progression[77], miR-423-5p with stable disease or partial response[78] and miR-10b-3p with shorter survival[79].
Another potentially interesting circulating miRNA is miR-221, which was studied by Fornari et al[76] in both animal models and in a patient population. Patients with radiologic disease progression after 2-mo treatment had higher pretreatment miR-221 levels than responders (P = 0.007).
Larger confirmatory studies are needed before miRNAs can be considered valid biomarkers for clinical practice.
A number of studies have analyzed tissue biomarkers that may be very specific to the disease of interest[80]. In a phase II study of sorafenib in advanced HCC, Abou-Alfa et al[81] showed that patients whose tumors expressed higher baseline phospho-ERK levels had a longer TTP. However, other studies reported conflicting results[82,83]. With regard to the expression of angiogenic markers in tumor tissue, it has been observed that high platelet-derived growth factor receptor beta expression is correlated with poor OS but not with PFS in HCC patients receiving sorafenib. High expression of the proto-oncogene c-Met may predict the therapeutic effectiveness of sorafenib in HCC patients, but no differences in terms of outcome have been seen with respect to VEGFR-2 expression[84]. Hagiwara et al[85] studied another interesting tissue biomarker, phospho-c-Jun, reporting a significantly higher expression (P < 0.001) in non-responding compared to responding patients treated with sorafenib.
After 10 years of research into sorafenib, there are still no validated prognostic or predictive markers of response to sorafenib in hepatocellular carcinoma. Furthermore, the main results obtained to date come from 2 important randomized trials and from different subanalyses and pooled analyses rather than from normal clinical practice. The fact of there being only one drug for the treatment of these patients has certainly done nothing to stimulate research into identifying and validating predictors of response and prognosis. However, given the recent publication of a positive phase III trial[86] and the ongoing NCT01658878 immunotherapy study, the race is now on to see who will be the first to identify a prognostic and predictive factor for sorafenib and/or new drugs in this setting. In conclusion, the use of metabolomic profiling and whole genome analysis to examine the association between patient outcome and response to sorafenib could become alternative approaches to the search for new biomarkers in HCC.
The authors thank Gráinne Tierney and Cristiano Verna for editorial assistance.
Manuscript source: Invited manuscript
Specialty type: Gastroenterology and hepatology
Country of origin: Italy
Peer-review report classification
Grade A (Excellent): 0
Grade B (Very good): B, B, B
Grade C (Good): 0
Grade D (Fair): 0
Grade E (Poor): 0
P- Reviewer: Lin ZY, Sun XY, Yao DF S- Editor: Gong ZM L- Editor: A E- Editor: Huang Y
| 1. | Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378-390. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 9016] [Cited by in RCA: 10272] [Article Influence: 604.2] [Reference Citation Analysis (2)] |
| 2. | Cervello M, Bachvarov D, Lampiasi N, Cusimano A, Azzolina A, McCubrey JA, Montalto G. Molecular mechanisms of sorafenib action in liver cancer cells. Cell Cycle. 2012;11:2843-2855. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 102] [Cited by in RCA: 127] [Article Influence: 9.8] [Reference Citation Analysis (0)] |
| 3. | Tai WT, Cheng AL, Shiau CW, Huang HP, Huang JW, Chen PJ, Chen KF. Signal transducer and activator of transcription 3 is a major kinase-independent target of sorafenib in hepatocellular carcinoma. J Hepatol. 2011;55:1041-1048. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 125] [Cited by in RCA: 139] [Article Influence: 9.9] [Reference Citation Analysis (0)] |
| 4. | Llovet JM, Peña CE, Lathia CD, Shan M, Meinhardt G, Bruix J; SHARP Investigators Study Group. Plasma biomarkers as predictors of outcome in patients with advanced hepatocellular carcinoma. Clin Cancer Res. 2012;18:2290-2300. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 464] [Cited by in RCA: 455] [Article Influence: 35.0] [Reference Citation Analysis (2)] |
| 5. | Forner A, Llovet JM, Bruix J. Hepatocellular carcinoma. Lancet. 2012;379:1245-1255. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3249] [Cited by in RCA: 3597] [Article Influence: 276.7] [Reference Citation Analysis (4)] |
| 6. | Bruix J, Cheng AL, Meinhardt G, Nakajima K, De Sanctis Y, Llovet J. Prognostic factors and predictors of sorafenib benefit in patients with hepatocellular carcinoma: Analysis of two phase III studies. J Hepatol. 2017;67:999-1008. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 360] [Cited by in RCA: 453] [Article Influence: 56.6] [Reference Citation Analysis (0)] |
| 7. | Lin ZZ, Hsu C, Hu FC, Shao YY, Chang DY, Yang CH, Hong RL, Hsu CH, Cheng AL. Factors impacting prognosis prediction in BCLC stage C and Child-Pugh class A hepatocellular carcinoma patients in prospective clinical trials of systemic therapy. Oncologist. 2012;17:970-977. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 8] [Article Influence: 0.6] [Reference Citation Analysis (0)] |
| 8. | Shao YY, Lin ZZ, Hsu C, Shen YC, Hsu CH, Cheng AL. Early alpha-fetoprotein response predicts treatment efficacy of antiangiogenic systemic therapy in patients with advanced hepatocellular carcinoma. Cancer. 2010;116:4590-4596. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 119] [Cited by in RCA: 138] [Article Influence: 9.2] [Reference Citation Analysis (0)] |
| 9. | Yau T, Yao TJ, Chan P, Wong H, Pang R, Fan ST, Poon RT. The significance of early alpha-fetoprotein level changes in predicting clinical and survival benefits in advanced hepatocellular carcinoma patients receiving sorafenib. Oncologist. 2011;16:1270-1279. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 77] [Cited by in RCA: 85] [Article Influence: 6.1] [Reference Citation Analysis (0)] |
| 10. | Personeni N, Bozzarelli S, Pressiani T, Rimassa L, Tronconi MC, Sclafani F, Carnaghi C, Pedicini V, Giordano L, Santoro A. Usefulness of alpha-fetoprotein response in patients treated with sorafenib for advanced hepatocellular carcinoma. J Hepatol. 2012;57:101-107. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 172] [Cited by in RCA: 181] [Article Influence: 13.9] [Reference Citation Analysis (0)] |
| 11. | Nakazawa T, Hidaka H, Takada J, Okuwaki Y, Tanaka Y, Watanabe M, Shibuya A, Minamino T, Kokubu S, Koizumi W. Early increase in α-fetoprotein for predicting unfavorable clinical outcomes in patients with advanced hepatocellular carcinoma treated with sorafenib. Eur J Gastroenterol Hepatol. 2013;25:683-689. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 60] [Cited by in RCA: 66] [Article Influence: 5.5] [Reference Citation Analysis (0)] |
| 12. | Vincenzi B, Santini D, Russo A, Addeo R, Giuliani F, Montella L, Rizzo S, Venditti O, Frezza AM, Caraglia M. Early skin toxicity as a predictive factor for tumor control in hepatocellular carcinoma patients treated with sorafenib. Oncologist. 2010;15:85-92. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 138] [Cited by in RCA: 143] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 13. | Reig M, Torres F, Rodriguez-Lope C, Forner A, LLarch N, Rimola J, Darnell A, Ríos J, Ayuso C, Bruix J. Early dermatologic adverse events predict better outcome in HCC patients treated with sorafenib. J Hepatol. 2014;61:318-324. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 178] [Cited by in RCA: 198] [Article Influence: 18.0] [Reference Citation Analysis (0)] |
| 14. | Wang P, Tan G, Zhu M, Li W, Zhai B, Sun X. Hand-foot skin reaction is a beneficial indicator of sorafenib therapy for patients with hepatocellular carcinoma: a systemic review and meta-analysis. Expert Rev Gastroenterol Hepatol. 2018;12:1-8. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 46] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 15. | Scartozzi M, Galizia E, Chiorrini S, Giampieri R, Berardi R, Pierantoni C, Cascinu S. Arterial hypertension correlates with clinical outcome in colorectal cancer patients treated with first-line bevacizumab. Ann Oncol. 2009;20:227-230. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 264] [Cited by in RCA: 256] [Article Influence: 16.0] [Reference Citation Analysis (0)] |
| 16. | Casadei Gardini A, Scarpi E, Marisi G, Foschi FG, Donati G, Giampalma E, Faloppi L, Scartozzi M, Silvestris N, Bisulli M. Early onset of hypertension and serum electrolyte changes as potential predictive factors of activity in advanced HCC patients treated with sorafenib: results from a retrospective analysis of the HCC-AVR group. Oncotarget. 2016;7:15243-15251. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 24] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 17. | Zhong BY, Ni CF, Chen L, Zhu HD, Teng GJ. Early Sorafenib-related Biomarkers for Combination Treatment with Transarterial Chemoembolization and Sorafenib in Patients with Hepatocellular Carcinoma. Radiology. 2017;284:583-592. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 16] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 18. | Howell J, Pinato DJ, Ramaswami R, Bettinger D, Arizumi T, Ferrari C, Yen C, Gibbin A, Burlone ME, Guaschino G. On-target sorafenib toxicity predicts improved survival in hepatocellular carcinoma: a multi-centre, prospective study. Aliment Pharmacol Ther. 2017;45:1146-1155. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 41] [Cited by in RCA: 45] [Article Influence: 5.6] [Reference Citation Analysis (0)] |
| 19. | Shin SY, Lee YJ. Correlation of skin toxicity and hypertension with clinical benefit in advanced hepatocellular carcinoma patients treated with sorafenib. Int J Clin Pharmacol Ther. 2013;51:837-846. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 19] [Cited by in RCA: 19] [Article Influence: 1.7] [Reference Citation Analysis (0)] |
| 20. | Otsuka T, Eguchi Y, Kawazoe S, Yanagita K, Ario K, Kitahara K, Kawasoe H, Kato H, Mizuta T; Saga Liver Cancer Study Group. Skin toxicities and survival in advanced hepatocellular carcinoma patients treated with sorafenib. Hepatol Res. 2012;42:879-886. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 75] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 21. | Bettinger D, Schultheiss M, Knüppel E, Thimme R, Blum HE, Spangenberg HC. Diarrhea predicts a positive response to sorafenib in patients with advanced hepatocellular carcinoma. Hepatology. 2012;56:789-790. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 43] [Cited by in RCA: 46] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 22. | Koschny R, Gotthardt D, Koehler C, Jaeger D, Stremmel W, Ganten TM. Diarrhea is a positive outcome predictor for sorafenib treatment of advanced hepatocellular carcinoma. Oncology. 2013;84:6-13. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 53] [Article Influence: 4.1] [Reference Citation Analysis (0)] |
| 23. | Di Costanzo GG, de Stefano G, Tortora R, Farella N, Addario L, Lampasi F, Lanza AG, Cordone G, Imparato M, Caporaso N. Sorafenib off-target effects predict outcomes in patients treated for hepatocellular carcinoma. Future Oncol. 2015;11:943-951. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 22] [Cited by in RCA: 22] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 24. | Di Costanzo GG, Casadei Gardini A, Marisi G, Foschi FG, Scartozzi M, Granata R, Faloppi L, Cascinu S, Silvestris N, Brunetti O. Validation of a Simple Scoring System to Predict Sorafenib Effectiveness in Patients with Hepatocellular Carcinoma. Target Oncol. 2017;12:795-803. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 20] [Cited by in RCA: 21] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 25. | Abdel-Rahman O, Lamarca A. Development of sorafenib-related side effects in patients diagnosed with advanced hepatocellular carcinoma treated with sorafenib: a systematic-review and meta-analysis of the impact on survival. Expert Rev Gastroenterol Hepatol. 2017;11:75-83. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 52] [Article Influence: 6.5] [Reference Citation Analysis (0)] |
| 26. | Cheng AL, Guan Z, Chen Z, Tsao CJ, Qin S, Kim JS, Yang TS, Tak WY, Pan H, Yu S. Efficacy and safety of sorafenib in patients with advanced hepatocellular carcinoma according to baseline status: subset analyses of the phase III Sorafenib Asia-Pacific trial. Eur J Cancer. 2012;48:1452-1465. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 185] [Cited by in RCA: 225] [Article Influence: 17.3] [Reference Citation Analysis (0)] |
| 27. | Hollebecque A, Cattan S, Romano O, Sergent G, Mourad A, Louvet A, Dharancy S, Boleslawski E, Truant S, Pruvot FR. Safety and efficacy of sorafenib in hepatocellular carcinoma: the impact of the Child-Pugh score. Aliment Pharmacol Ther. 2011;34:1193-1201. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 86] [Cited by in RCA: 95] [Article Influence: 6.8] [Reference Citation Analysis (0)] |
| 28. | Pressiani T, Boni C, Rimassa L, Labianca R, Fagiuoli S, Salvagni S, Ferrari D, Cortesi E, Porta C, Mucciarini C. Sorafenib in patients with Child-Pugh class A and B advanced hepatocellular carcinoma: a prospective feasibility analysis. Ann Oncol. 2013;24:406-411. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 126] [Article Influence: 10.5] [Reference Citation Analysis (0)] |
| 29. | Marrero JA, Kudo M, Venook AP, Ye SL, Bronowicki JP, Chen XP, Dagher L, Furuse J, Geschwind JH, de Guevara LL. Observational registry of sorafenib use in clinical practice across Child-Pugh subgroups: The GIDEON study. J Hepatol. 2016;65:1140-1147. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 205] [Cited by in RCA: 297] [Article Influence: 33.0] [Reference Citation Analysis (0)] |
| 30. | Lee S, Kang JH, Kim DY, Ahn SH, Park JY, Kim BK, Kim SU, Han KH. Prognostic factors of sorafenib therapy in hepatocellular carcinoma patients with failure of transarterial chemoembolization. Hepatol Int. 2017;11:292-299. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 8] [Cited by in RCA: 6] [Article Influence: 0.8] [Reference Citation Analysis (0)] |
| 31. | Peng S, Zhao Y, Xu F, Jia C, Xu Y, Dai C. An updated meta-analysis of randomized controlled trials assessing the effect of sorafenib in advanced hepatocellular carcinoma. PLoS One. 2014;9:e112530. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 20] [Cited by in RCA: 24] [Article Influence: 2.2] [Reference Citation Analysis (0)] |
| 32. | Iavarone M, Cabibbo G, Piscaglia F, Zavaglia C, Grieco A, Villa E, Cammà C, Colombo M; SOFIA (SOraFenib Italian Assessment) study group. Field-practice study of sorafenib therapy for hepatocellular carcinoma: a prospective multicenter study in Italy. Hepatology. 2011;54:2055-2063. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 287] [Cited by in RCA: 299] [Article Influence: 21.4] [Reference Citation Analysis (0)] |
| 33. | Casadei Gardini A, Frassineti GL, Foschi FG, Ercolani G, Ulivi P. Sorafenib and Regorafenib in HBV- or HCV-positive hepatocellular carcinoma patients: Analysis of RESORCE and SHARP trials. Dig Liver Dis. 2017;49:943-944. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 17] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 34. | Donadon V, Balbi M, Mas MD, Casarin P, Zanette G. Metformin and reduced risk of hepatocellular carcinoma in diabetic patients with chronic liver disease. Liver Int. 2010;30:750-758. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 206] [Cited by in RCA: 223] [Article Influence: 14.9] [Reference Citation Analysis (1)] |
| 35. | Casadei Gardini A, Marisi G, Scarpi E, Scartozzi M, Faloppi L, Silvestris N, Masi G, Vivaldi C, Brunetti O, Tamberi S. Effects of metformin on clinical outcome in diabetic patients with advanced HCC receiving sorafenib. Expert Opin Pharmacother. 2015;16:2719-2725. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 48] [Cited by in RCA: 58] [Article Influence: 5.8] [Reference Citation Analysis (0)] |
| 36. | Casadei Gardini A, Faloppi L, De Matteis S, Foschi FG, Silvestris N, Tovoli F, Palmieri V, Marisi G, Brunetti O, Vespasiani-Gentilucci U. Metformin and insulin impact on clinical outcome in patients with advanced hepatocellular carcinoma receiving sorafenib: Validation study and biological rationale. Eur J Cancer. 2017;86:106-114. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 71] [Cited by in RCA: 72] [Article Influence: 9.0] [Reference Citation Analysis (0)] |
| 37. | De Matteis S, Granato AM, Napolitano R, Molinari C, Valgiusti M, Santini D, Foschi FG, Ercolani G, Vespasiani Gentilucci U, Faloppi L. Interplay Between SIRT-3, Metabolism and Its Tumor Suppressor Role in Hepatocellular Carcinoma. Dig Dis Sci. 2017;62:1872-1880. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 12] [Cited by in RCA: 14] [Article Influence: 1.8] [Reference Citation Analysis (0)] |
| 38. | Di Costanzo GG, Tortora R, Morisco F, Addario L, Guarino M, Cordone G, Falco L, Caporaso N. Impact of Diabetes on Outcomes of Sorafenib Therapy for Hepatocellular Carcinoma. Target Oncol. 2017;12:61-67. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 14] [Cited by in RCA: 18] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 39. | Ferrucci PF, Gandini S, Battaglia A, Alfieri S, Di Giacomo AM, Giannarelli D, Cappellini GC, De Galitiis F, Marchetti P, Amato G. Baseline neutrophil-to-lymphocyte ratio is associated with outcome of ipilimumab-treated metastatic melanoma patients. Br J Cancer. 2015;112:1904-1910. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 155] [Cited by in RCA: 197] [Article Influence: 19.7] [Reference Citation Analysis (0)] |
| 40. | Hu B, Yang XR, Xu Y, Sun YF, Sun C, Guo W, Zhang X, Wang WM, Qiu SJ, Zhou J. Systemic immune-inflammation index predicts prognosis of patients after curative resection for hepatocellular carcinoma. Clin Cancer Res. 2014;20:6212-6222. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 1414] [Cited by in RCA: 1366] [Article Influence: 124.2] [Reference Citation Analysis (2)] |
| 41. | Casadei Gardini A, Scarpi E, Faloppi L, Scartozzi M, Silvestris N, Santini D, de Stefano G, Marisi G, Negri FV, Foschi FG. Immune inflammation indicators and implication for immune modulation strategies in advanced hepatocellular carcinoma patients receiving sorafenib. Oncotarget. 2016;7:67142-67149. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 68] [Cited by in RCA: 82] [Article Influence: 11.7] [Reference Citation Analysis (0)] |
| 42. | Howell J, Pinato DJ, Ramaswami R, Arizumi T, Ferrari C, Gibbin A, Burlone ME, Guaschino G, Toniutto P, Black J. Integration of the cancer-related inflammatory response as a stratifying biomarker of survival in hepatocellular carcinoma treated with sorafenib. Oncotarget. 2017;8:36161-36170. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 32] [Article Influence: 4.6] [Reference Citation Analysis (0)] |
| 43. | da Fonseca LG, Barroso-Sousa R, Bento Ada S, Blanco BP, Valente GL, Pfiffer TE, Hoff PM, Sabbaga J. Pre-treatment neutrophil-to-lymphocyte ratio affects survival in patients with advanced hepatocellular carcinoma treated with sorafenib. Med Oncol. 2014;31:264. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 39] [Article Influence: 3.5] [Reference Citation Analysis (0)] |
| 44. | Lué A, Serrano MT, Bustamante FJ, Iñarrairaegui M, Arenas JI, Testillano M, Lorente S, Gil C, de la Torre M, Gomez A. Neutrophil-to-lymphocyte ratio predicts survival in European patients with hepatocellular carcinoma administered sorafenib. Oncotarget. 2017;8:103077-103086. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 29] [Cited by in RCA: 35] [Article Influence: 4.4] [Reference Citation Analysis (0)] |
| 45. | Therasse P, Arbuck SG, Eisenhauer EA, Wanders J, Kaplan RS, Rubinstein L, Verweij J, Van Glabbeke M, van Oosterom AT, Christian MC. New guidelines to evaluate the response to treatment in solid tumors. European Organization for Research and Treatment of Cancer, National Cancer Institute of the United States, National Cancer Institute of Canada. J Natl Cancer Inst. 2000;92:205-216. [PubMed] |
| 46. | Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, Ford R, Dancey J, Arbuck S, Gwyther S, Mooney M. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer. 2009;45:228-247. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 15860] [Cited by in RCA: 21632] [Article Influence: 1352.0] [Reference Citation Analysis (1)] |
| 47. | Ronot M, Bouattour M, Wassermann J, Bruno O, Dreyer C, Larroque B, Castera L, Vilgrain V, Belghiti J, Raymond E. Alternative Response Criteria (Choi, European association for the study of the liver, and modified Response Evaluation Criteria in Solid Tumors [RECIST]) Versus RECIST 1.1 in patients with advanced hepatocellular carcinoma treated with sorafenib. Oncologist. 2014;19:394-402. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 122] [Cited by in RCA: 118] [Article Influence: 10.7] [Reference Citation Analysis (1)] |
| 48. | Sugimoto K, Moriyasu F, Saito K, Rognin N, Kamiyama N, Furuichi Y, Imai Y. Hepatocellular carcinoma treated with sorafenib: early detection of treatment response and major adverse events by contrast-enhanced US. Liver Int. 2013;33:605-615. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 55] [Cited by in RCA: 52] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 49. | Zocco MA, Garcovich M, Lupascu A, Di Stasio E, Roccarina D, Annicchiarico BE, Riccardi L, Ainora ME, Ponziani F, Caracciolo G. Early prediction of response to sorafenib in patients with advanced hepatocellular carcinoma: the role of dynamic contrast enhanced ultrasound. J Hepatol. 2013;59:1014-1021. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 68] [Cited by in RCA: 68] [Article Influence: 5.7] [Reference Citation Analysis (0)] |
| 50. | Zhou C, Liu J, Li Y, Liu L, Zhang X, Ma CY, Hua SC, Yang M, Yuan Q. microRNA-1274a, a modulator of sorafenib induced a disintegrin and metalloproteinase 9 (ADAM9) down-regulation in hepatocellular carcinoma. FEBS Lett. 2011;585:1828-1834. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 47] [Cited by in RCA: 50] [Article Influence: 3.6] [Reference Citation Analysis (0)] |
| 51. | Hsu CY, Shen YC, Yu CW, Hsu C, Hu FC, Hsu CH, Chen BB, Wei SY, Cheng AL, Shih TT. Dynamic contrast-enhanced magnetic resonance imaging biomarkers predict survival and response in hepatocellular carcinoma patients treated with sorafenib and metronomic tegafur/uracil. J Hepatol. 2011;55:858-865. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 97] [Cited by in RCA: 97] [Article Influence: 6.9] [Reference Citation Analysis (0)] |
| 52. | Lee JH, Park JY, Kim DY, Ahn SH, Han KH, Seo HJ, Lee JD, Choi HJ. Prognostic value of 18F-FDG PET for hepatocellular carcinoma patients treated with sorafenib. Liver Int. 2011;31:1144-1149. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 52] [Cited by in RCA: 53] [Article Influence: 3.8] [Reference Citation Analysis (0)] |
| 53. | Mulé S, Thiefin G, Costentin C, Durot C, Rahmouni A, Luciani A, Hoeffel C. Advanced Hepatocellular Carcinoma: Pretreatment Contrast-enhanced CT Texture Parameters as Predictive Biomarkers of Survival in Patients Treated with Sorafenib. Radiology. 2018;288:445-455. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 81] [Article Influence: 11.6] [Reference Citation Analysis (0)] |
| 54. | Su WW, Lee KT, Yeh YT, Soon MS, Wang CL, Yu ML, Wang SN. Association of circulating insulin-like growth factor 1 with hepatocellular carcinoma: one cross-sectional correlation study. J Clin Lab Anal. 2010;24:195-200. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 35] [Cited by in RCA: 39] [Article Influence: 2.6] [Reference Citation Analysis (0)] |
| 55. | Shao YY, Huang CC, Lin SD, Hsu CH, Cheng AL. Serum insulin-like growth factor-1 levels predict outcomes of patients with advanced hepatocellular carcinoma receiving antiangiogenic therapy. Clin Cancer Res. 2012;18:3992-3997. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 33] [Cited by in RCA: 36] [Article Influence: 2.8] [Reference Citation Analysis (0)] |
| 56. | Kim HY, Lee DH, Lee JH, Cho YY, Cho EJ, Yu SJ, Kim YJ, Yoon JH. Novel biomarker-based model for the prediction of sorafenib response and overall survival in advanced hepatocellular carcinoma: a prospective cohort study. BMC Cancer. 2018;18:307. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 21] [Article Influence: 3.0] [Reference Citation Analysis (0)] |
| 57. | Miyahara K, Nouso K, Tomoda T, Kobayashi S, Hagihara H, Kuwaki K, Toshimori J, Onishi H, Ikeda F, Miyake Y. Predicting the treatment effect of sorafenib using serum angiogenesis markers in patients with hepatocellular carcinoma. J Gastroenterol Hepatol. 2011;26:1604-1611. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 65] [Cited by in RCA: 69] [Article Influence: 4.9] [Reference Citation Analysis (0)] |
| 58. | Tsuchiya K, Asahina Y, Matsuda S, Muraoka M, Nakata T, Suzuki Y, Tamaki N, Yasui Y, Suzuki S, Hosokawa T. Changes in plasma vascular endothelial growth factor at 8 weeks after sorafenib administration as predictors of survival for advanced hepatocellular carcinoma. Cancer. 2014;120:229-237. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 46] [Cited by in RCA: 54] [Article Influence: 4.5] [Reference Citation Analysis (0)] |
| 59. | Hu B, Cheng SY. Angiopoietin-2: development of inhibitors for cancer therapy. Curr Oncol Rep. 2009;11:111-116. [PubMed] |
| 60. | Hayashi T, Yamashita T, Terashima T, Suda T, Okada H, Asahina Y, Hayashi T, Hara Y, Nio K, Sunagozaka H. Serum cytokine profiles predict survival benefits in patients with advanced hepatocellular carcinoma treated with sorafenib: a retrospective cohort study. BMC Cancer. 2017;17:870. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 26] [Article Influence: 3.3] [Reference Citation Analysis (0)] |
| 61. | Villanueva A, Minguez B, Forner A, Reig M, Llovet JM. Hepatocellular carcinoma: novel molecular approaches for diagnosis, prognosis, and therapy. Annu Rev Med. 2010;61:317-328. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 170] [Cited by in RCA: 196] [Article Influence: 13.1] [Reference Citation Analysis (0)] |
| 62. | Sakai K, Takeda H, Nishijima N, Orito E, Joko K, Uchida Y, Izumi N, Nishio K, Osaki Y. Targeted DNA and RNA sequencing of fine-needle biopsy FFPE specimens in patients with unresectable hepatocellular carcinoma treated with sorafenib. Oncotarget. 2015;6:21636-21644. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 15] [Cited by in RCA: 14] [Article Influence: 1.6] [Reference Citation Analysis (0)] |
| 63. | Kan Z, Zheng H, Liu X, Li S, Barber TD, Gong Z, Gao H, Hao K, Willard MD, Xu J. Whole-genome sequencing identifies recurrent mutations in hepatocellular carcinoma. Genome Res. 2013;23:1422-1433. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 355] [Cited by in RCA: 412] [Article Influence: 34.3] [Reference Citation Analysis (0)] |
| 64. | Cleary SP, Jeck WR, Zhao X, Chen K, Selitsky SR, Savich GL, Tan TX, Wu MC, Getz G, Lawrence MS. Identification of driver genes in hepatocellular carcinoma by exome sequencing. Hepatology. 2013;58:1693-1702. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 216] [Cited by in RCA: 243] [Article Influence: 20.3] [Reference Citation Analysis (0)] |
| 65. | Scartozzi M, Faloppi L, Svegliati Baroni G, Loretelli C, Piscaglia F, Iavarone M, Toniutto P, Fava G, De Minicis S, Mandolesi A. VEGF and VEGFR genotyping in the prediction of clinical outcome for HCC patients receiving sorafenib: the ALICE-1 study. Int J Cancer. 2014;135:1247-1256. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 111] [Cited by in RCA: 105] [Article Influence: 9.5] [Reference Citation Analysis (0)] |
| 66. | Casadei Gardini A, Marisi G, Faloppi L, Scarpi E, Foschi FG, Iavarone M, Lauletta G, Corbelli J, Valgiusti M, Facchetti F. eNOS polymorphisms and clinical outcome in advanced HCC patients receiving sorafenib: final results of the ePHAS study. Oncotarget. 2016;7:27988-27999. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 22] [Cited by in RCA: 30] [Article Influence: 4.3] [Reference Citation Analysis (0)] |
| 67. | Marisi G, Scartozzi M, Faloppi L, Foschi GF, Lauletta G, Valgiusti M, Aprile G, Fornaro L, Iavarone M, Ercolani G. Ang-2 polymorphisms in relation to outcome in advanced HCC patients receiving sorafenib. J Clin Oncol. 2017;35 Suppl 15:e15666. [RCA] [DOI] [Full Text] [Cited by in Crossref: 3] [Cited by in RCA: 3] [Article Influence: 0.4] [Reference Citation Analysis (0)] |
| 68. | Faloppi L, Casadei Gardini A, Masi G, Silvestris N, Loretelli C, Ulivi P, Vivaldi C, Bianconi M, Giampieri R, Bittoni A. Angiogenesis polymorphisms profile in the prediction of clinical outcome of advanced HCC patients receiving sorafenib: Combined analysis of VEGF and HIF-1α. Final results of the ALICE-2 study. J Clin Oncol. 2016;34 Suppl 4:280. [RCA] [DOI] [Full Text] [Cited by in Crossref: 11] [Cited by in RCA: 11] [Article Influence: 1.2] [Reference Citation Analysis (0)] |
| 69. | Casadei Gardini A, Faloppi L, Aprile G, Brunetti O, Caparello C, Corbelli J, Chessa L, Bruno D, Ercolani G, Leonetti A. Multicenter Prospective Study of Angiogenesis Polymorphism Validation in HCC Patients Treated with Sorafenib. An INNOVATE Study Protocol. Tumori. 2017; : tj5000704 Epub ahead of print. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 13] [Cited by in RCA: 15] [Article Influence: 2.1] [Reference Citation Analysis (0)] |
| 70. | Horwitz E, Stein I, Andreozzi M, Nemeth J, Shoham A, Pappo O, Schweitzer N, Tornillo L, Kanarek N, Quagliata L. Human and mouse VEGFA-amplified hepatocellular carcinomas are highly sensitive to sorafenib treatment. Cancer Discov. 2014;4:730-743. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 151] [Cited by in RCA: 156] [Article Influence: 14.2] [Reference Citation Analysis (0)] |
| 71. | Arao T, Ueshima K, Matsumoto K, Nagai T, Kimura H, Hagiwara S, Sakurai T, Haji S, Kanazawa A, Hidaka H. FGF3/FGF4 amplification and multiple lung metastases in responders to sorafenib in hepatocellular carcinoma. Hepatology. 2013;57:1407-1415. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 115] [Cited by in RCA: 120] [Article Influence: 10.0] [Reference Citation Analysis (0)] |
| 72. | He C, Dong X, Zhai B, Jiang X, Dong D, Li B, Jiang H, Xu S, Sun X. MiR-21 mediates sorafenib resistance of hepatocellular carcinoma cells by inhibiting autophagy via the PTEN/Akt pathway. Oncotarget. 2015;6:28867-28881. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 171] [Cited by in RCA: 184] [Article Influence: 18.4] [Reference Citation Analysis (0)] |
| 73. | Xu Y, Huang J, Ma L, Shan J, Shen J, Yang Z, Liu L, Luo Y, Yao C, Qian C. MicroRNA-122 confers sorafenib resistance to hepatocellular carcinoma cells by targeting IGF-1R to regulate RAS/RAF/ERK signaling pathways. Cancer Lett. 2016;371:171-181. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 110] [Cited by in RCA: 137] [Article Influence: 13.7] [Reference Citation Analysis (0)] |
| 74. | Vaira V, Roncalli M, Carnaghi C, Faversani A, Maggioni M, Augello C, Rimassa L, Pressiani T, Spagnuolo G, Di Tommaso L. MicroRNA-425-3p predicts response to sorafenib therapy in patients with hepatocellular carcinoma. Liver Int. 2015;35:1077-1086. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 59] [Cited by in RCA: 63] [Article Influence: 6.3] [Reference Citation Analysis (0)] |
| 75. | Gyöngyösi B, Végh É, Járay B, Székely E, Fassan M, Bodoky G, Schaff Z, Kiss A. Pretreatment MicroRNA Level and Outcome in Sorafenib-treated Hepatocellular Carcinoma. J Histochem Cytochem. 2014;62:547-555. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 37] [Cited by in RCA: 41] [Article Influence: 3.7] [Reference Citation Analysis (0)] |
| 76. | Fornari F, Pollutri D, Patrizi C, La Bella T, Marinelli S, Casadei Gardini A, Marisi G, Baron Toaldo M, Baglioni M, Salvatore V. In Hepatocellular Carcinoma miR-221 Modulates Sorafenib Resistance through Inhibition of Caspase-3-Mediated Apoptosis. Clin Cancer Res. 2017;23:3953-3965. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 104] [Cited by in RCA: 130] [Article Influence: 16.3] [Reference Citation Analysis (0)] |
| 77. | Nishida N, Arizumi T, Hagiwara S, Ida H, Sakurai T, Kudo M. MicroRNAs for the Prediction of Early Response to Sorafenib Treatment in Human Hepatocellular Carcinoma. Liver Cancer. 2017;6:113-125. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 39] [Cited by in RCA: 49] [Article Influence: 6.1] [Reference Citation Analysis (1)] |
| 78. | Stiuso P, Potenza N, Lombardi A, Ferrandino I, Monaco A, Zappavigna S, Vanacore D, Mosca N, Castiello F, Porto S. MicroRNA-423-5p Promotes Autophagy in Cancer Cells and Is Increased in Serum From Hepatocarcinoma Patients Treated With Sorafenib. Mol Ther Nucleic Acids. 2015;4:e233. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 103] [Cited by in RCA: 112] [Article Influence: 11.2] [Reference Citation Analysis (0)] |
| 79. | Yoon EL, Yeon JE, Ko E, Lee HJ, Je JH, Yoo YJ, Kang SH, Suh SJ, Kim JH, Seo YS. An Explorative Analysis for the Role of Serum miR-10b-3p Levels in Predicting Response to Sorafenib in Patients with Advanced Hepatocellular Carcinoma. J Korean Med Sci. 2017;32:212-220. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 24] [Cited by in RCA: 32] [Article Influence: 4.0] [Reference Citation Analysis (0)] |
| 80. | Carvajal-Hausdorf DE, Schalper KA, Neumeister VM, Rimm DL. Quantitative measurement of cancer tissue biomarkers in the lab and in the clinic. Lab Invest. 2015;95:385-396. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 66] [Cited by in RCA: 66] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 81. | Abou-Alfa GK, Schwartz L, Ricci S, Amadori D, Santoro A, Figer A, De Greve J, Douillard JY, Lathia C, Schwartz B. Phase II study of sorafenib in patients with advanced hepatocellular carcinoma. J Clin Oncol. 2006;24:4293-4300. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 879] [Cited by in RCA: 908] [Article Influence: 47.8] [Reference Citation Analysis (0)] |
| 82. | Personeni N, Rimassa L, Pressiani T, Destro A, Ligorio C, Tronconi MC, Bozzarelli S, Carnaghi C, Di Tommaso L, Giordano L. Molecular determinants of outcome in sorafenib-treated patients with hepatocellular carcinoma. J Cancer Res Clin Oncol. 2013;139:1179-1187. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 33] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 83. | Chen D, Zhao P, Li SQ, Xiao WK, Yin XY, Peng BG, Liang LJ. Prognostic impact of pERK in advanced hepatocellular carcinoma patients treated with sorafenib. Eur J Surg Oncol. 2013;39:974-980. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 24] [Cited by in RCA: 28] [Article Influence: 2.3] [Reference Citation Analysis (0)] |
| 84. | Chu JS, Ge FJ, Zhang B, Wang Y, Silvestris N, Liu LJ, Zhao CH, Lin L, Brunetti AE, Fu YL. Expression and prognostic value of VEGFR-2, PDGFR-β, and c-Met in advanced hepatocellular carcinoma. J Exp Clin Cancer Res. 2013;32:16. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 69] [Cited by in RCA: 84] [Article Influence: 7.0] [Reference Citation Analysis (0)] |
| 85. | Hagiwara S, Kudo M, Nagai T, Inoue T, Ueshima K, Nishida N, Watanabe T, Sakurai T. Activation of JNK and high expression level of CD133 predict a poor response to sorafenib in hepatocellular carcinoma. Br J Cancer. 2012;106:1997-2003. [RCA] [PubMed] [DOI] [Full Text] [Full Text (PDF)] [Cited by in Crossref: 76] [Cited by in RCA: 86] [Article Influence: 6.6] [Reference Citation Analysis (0)] |
| 86. | Kudo M, Finn RS, Qin S, Han KH, Ikeda K, Piscaglia F, Baron A, Park JW, Han G, Jassem J. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163-1173. [RCA] [PubMed] [DOI] [Full Text] [Cited by in Crossref: 3128] [Cited by in RCA: 3832] [Article Influence: 547.4] [Reference Citation Analysis (1)] |